Comparison of the Antibacterial Effect of Silver Nanoparticles and a Multifunctional Antimicrobial Peptide on Titanium Surface
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of AgNPs
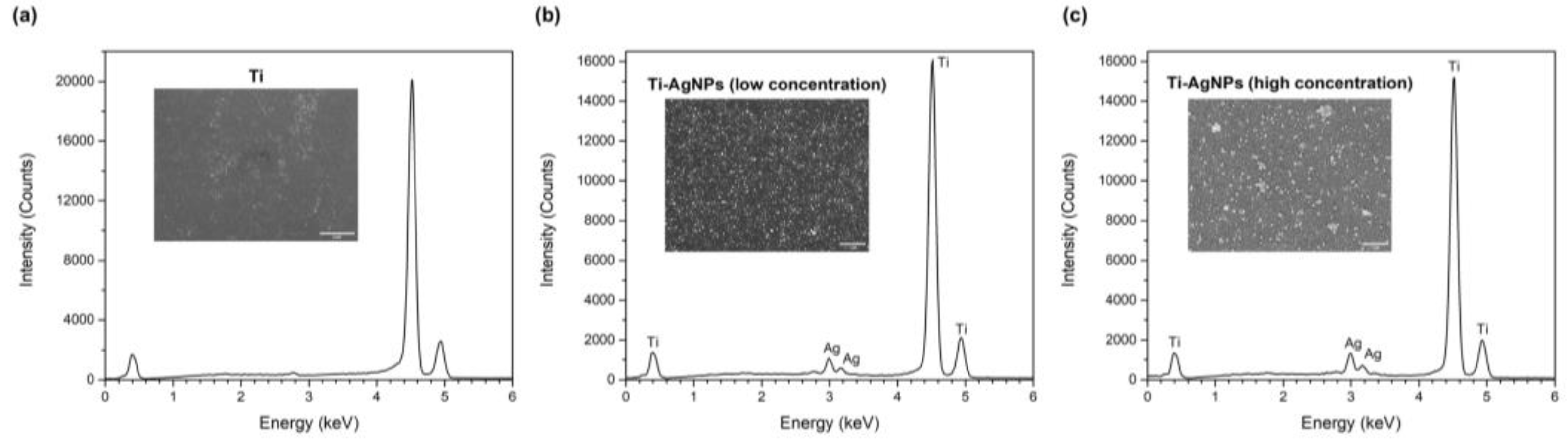
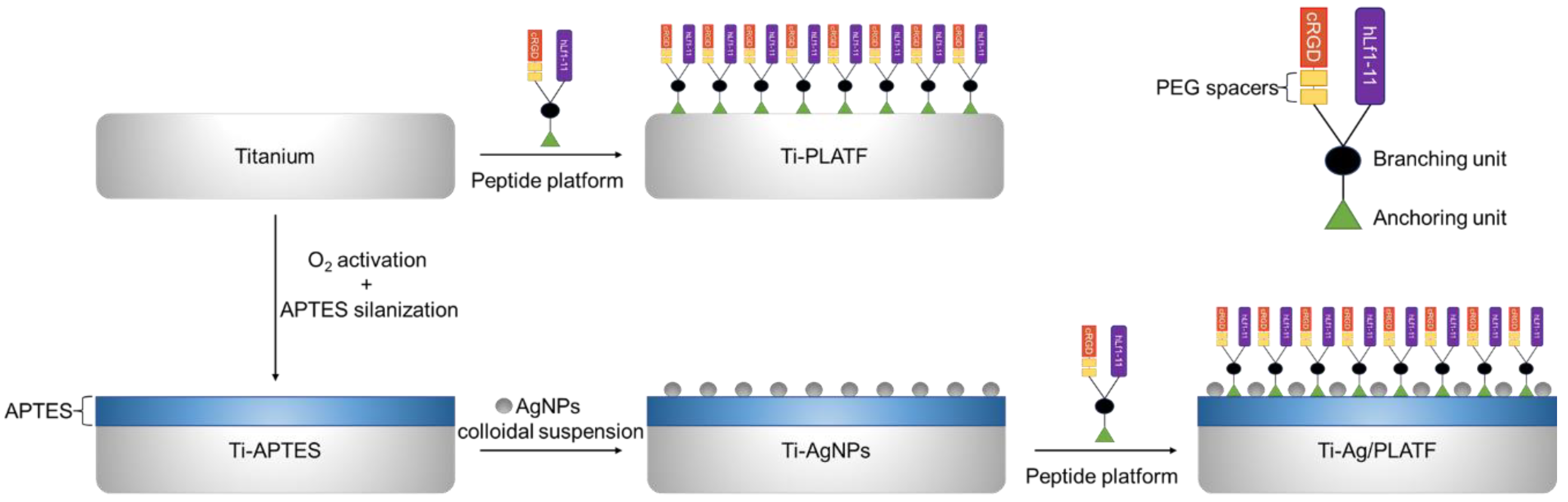
2.2. Surface Characterization of Functionalized Ti
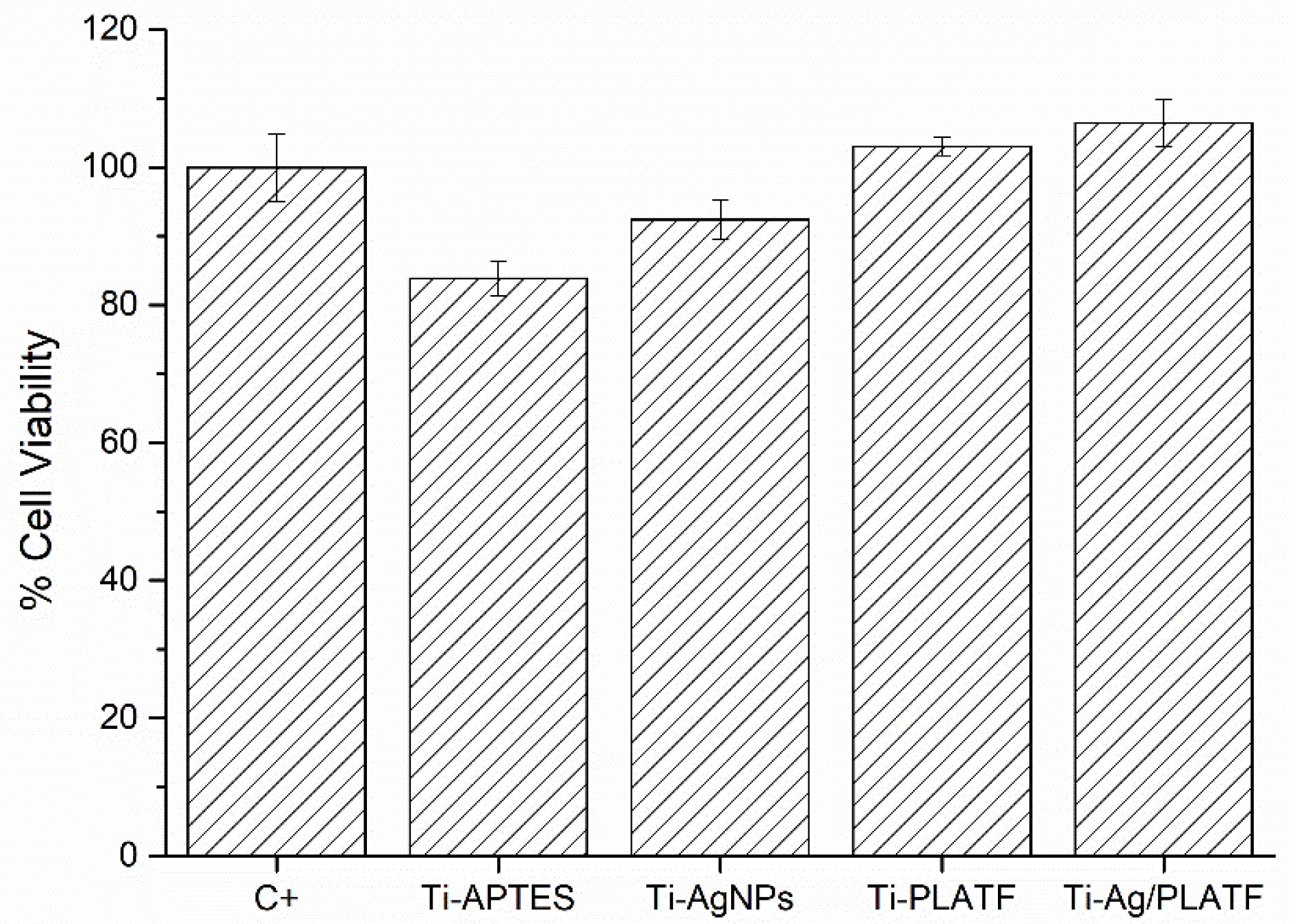
2.3. Biocompatibility Evaluation
2.4. Antibacterial Properties
3. Materials and Methods
3.1. Substrate Preparation
3.2. Synthesis of Silver Nanoparticles
3.3. AgNPs Characterization
3.4. Substrate Functionalization with AgNPs
3.5. Peptide Synthesis
3.6. Ti Functionalization with Peptide Platform
3.7. Physicochemical Surface Characterization
3.8. Evaluation of Antibacterial Properties
3.9. Biocompatibility Evaluation
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- OECD; European Union. Health at a Glance: Europe 2020; OECD Publishing: Paris, France, 2020; ISBN 9789264365643. [Google Scholar]
- Kapadia, B.H.; Berg, R.A.; Daley, J.A.; Fritz, J.; Bhave, A.; Mont, M.A. Periprosthetic Joint Infection. Lancet 2016, 387, 386–394. [Google Scholar] [CrossRef]
- Lum, Z.C.; Natsuhara, K.M.; Shelton, T.J.; Giordani, M.; Pereira, G.C.; Meehan, J.P. Mortality during Total Knee Periprosthetic Joint Infection. J. Arthroplast. 2018, 33, 3783–3788. [Google Scholar] [CrossRef]
- Mas-Moruno, C.; Su, B.; Dalby, M.J. Multifunctional Coatings and Nanotopographies: Toward Cell Instructive and Antibacterial Implants. Adv. Healthc. Mater. 2019, 8, 1801103. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, A.; Song, J. Anti-Periprosthetic Infection Strategies: From Implant Surface Topographical Engineering to Smart Drug-Releasing Coatings. ACS Appl. Mater. Interfaces 2021, 13, 20921–20937. [Google Scholar] [CrossRef] [PubMed]
- Buxadera-Palomero, J.; Calvo, C.; Torrent-Camarero, S.; Gil, F.J.; Mas-Moruno, C.; Canal, C.; Rodríguez, D. Biofunctional Polyethylene Glycol Coatings on Titanium: An In Vitro-Based Comparison of Functionalization Methods. Colloids Surf. B. Biointerfaces 2017, 152, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Sadowska, J.M.; Genoud, K.J.; Kelly, D.J.; O’Brien, F.J. Bone Biomaterials for Overcoming Antimicrobial Resistance: Advances in Non-Antibiotic Antimicrobial Approaches for Regeneration of Infected Osseous Tissue. Mater. Today 2021, 46, 136–154. [Google Scholar] [CrossRef]
- Iglesias-Fernandez, M.; Buxadera-Palomero, J.; Sadowska, J.-M.; Espanol, M.; Ginebra, M.-P. Implementation of Bactericidal Topographies on Biomimetic Calcium Phosphates and the Potential Effect of Its Reactivity. Biomater. Adv. 2022, 136, 212797. [Google Scholar] [CrossRef]
- Xuan, J.; Feng, W.; Wang, J.; Wang, R.; Zhang, B.; Bo, L.; Chen, Z.-S.; Yang, H.; Sun, L. Antimicrobial Peptides for Combating Drug-Resistant Bacterial Infections. Drug Resist. Updat. 2023, 68, 100954. [Google Scholar] [CrossRef]
- Tang, S.-S.; Prodhan, Z.H.; Biswas, S.K.; Le, C.-F.; Sekaran, S.D. Antimicrobial Peptides from Different Plant Sources: Isolation, Characterisation, and Purification. Phytochemistry 2018, 154, 94–105. [Google Scholar] [CrossRef]
- Singh, J.; Joshi, S.; Mumtaz, S.; Maurya, N.; Ghosh, I.; Khanna, S.; Natarajan, V.T.; Mukhopadhyay, K. Enhanced Cationic Charge Is a Key Factor in Promoting Staphylocidal Activity of α-Melanocyte Stimulating Hormone via Selective Lipid Affinity. Sci. Rep. 2016, 6, 31492. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.D.T.; Sothiselvam, S.; Lu, T.K.; de la Fuente-Nunez, C. Peptide Design Principles for Antimicrobial Applications. J. Mol. Biol. 2019, 431, 3547–3567. [Google Scholar] [CrossRef]
- Godoy-Gallardo, M.; Mas-Moruno, C.; Fernández-Calderón, M.C.; Pérez-Giraldo, C.; Manero, J.M.; Albericio, F.; Gil, F.J.; Rodríguez, D. Covalent Immobilization of HLf1-11 Peptide on a Titanium Surface Reduces Bacterial Adhesion and Biofilm Formation. Acta Biomater. 2014, 10, 3522–3534. [Google Scholar] [CrossRef] [PubMed]
- Godoy-Gallardo, M.; Rodríguez-Hernández, A.G.; Delgado, L.M.; Manero, J.M.; Javier Gil, F.; Rodríguez, D. Silver Deposition on Titanium Surface by Electrochemical Anodizing Process Reduces Bacterial Adhesion of Streptococcus Sanguinis and Lactobacillus Salivarius. Clin. Oral Implant. Res. 2015, 26, 1170–1179. [Google Scholar] [CrossRef] [PubMed]
- Kurtuldu, F.; Mutlu, N.; Boccaccini, A.R.; Galusek, D. Gallium Containing Bioactive Materials: A Review of Anticancer, Antibacterial, and Osteogenic Properties. Bioact. Mater. 2022, 17, 125–146. [Google Scholar] [CrossRef]
- Ye, Z.; Sang, T.; Li, K.; Fischer, N.G.; Mutreja, I.; Echeverría, C.; Kumar, D.; Tang, Z.; Aparicio, C. Hybrid Nanocoatings of Self-Assembled Organic-Inorganic Amphiphiles for Prevention of Implant Infections. Acta Biomater. 2022, 140, 338–349. [Google Scholar] [CrossRef]
- Yin, I.X.; Zhang, J.; Zhao, I.S.; Mei, M.L.; Li, Q.; Chu, C.H. The Antibacterial Mechanism of Silver Nanoparticles and Its Application in Dentistry. Int. J. Nanomed. 2020, 15, 2555–2562. [Google Scholar] [CrossRef] [PubMed]
- Mahlapuu, M.; Björn, C.; Ekblom, J. Antimicrobial Peptides as Therapeutic Agents: Opportunities and Challenges. Crit. Rev. Biotechnol. 2020, 40, 978–992. [Google Scholar] [CrossRef]
- Andersson, D.I.; Hughes, D.; Kubicek-Sutherland, J.Z. Mechanisms and Consequences of Bacterial Resistance to Antimicrobial Peptides. Drug Resist. Updat. Rev. Comment. Antimicrob. Anticancer Chemother. 2016, 26, 43–57. [Google Scholar] [CrossRef]
- Kubicek-Sutherland, J.Z.; Lofton, H.; Vestergaard, M.; Hjort, K.; Ingmer, H.; Andersson, D.I. Antimicrobial Peptide Exposure Selects for Staphylococcus Aureus Resistance to Human Defence Peptides. J. Antimicrob. Chemother. 2017, 72, 115–127. [Google Scholar] [CrossRef]
- Niño-Martínez, N.; Salas Orozco, M.F.; Martínez-Castañón, G.-A.; Torres Méndez, F.; Ruiz, F. Molecular Mechanisms of Bacterial Resistance to Metal and Metal Oxide Nanoparticles. Int. J. Mol. Sci. 2019, 20, 2808. [Google Scholar] [CrossRef]
- Oliver-Cervelló, L.; Martin-Gómez, H.; Mas-Moruno, C. New Trends in the Development of Multifunctional Peptides to Functionalize Biomaterials. J. Pept. Sci. 2022, 28, e3335. [Google Scholar] [CrossRef]
- Hoyos-Nogués, M.; Velasco, F.; Ginebra, M.-P.; Manero, J.M.; Gil, F.J.; Mas-Moruno, C. Regenerating Bone via Multifunctional Coatings: The Blending of Cell Integration and Bacterial Inhibition Properties on the Surface of Biomaterials. ACS Appl. Mater. Interfaces 2017, 9, 21618–21630. [Google Scholar] [CrossRef] [PubMed]
- Martin-Gómez, H.; Oliver-Cervelló, L.; Buxadera-Palomero, J.; Ginebra, M.P.; Mas-Moruno, C. Chemically Diverse Multifunctional Peptide Platforms with Antimicrobial and Cell Adhesive Properties. ChemBioChem 2021, 22, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Pal, I.; Brahmkhatri, V.P.; Bera, S.; Bhattacharyya, D.; Quirishi, Y.; Bhunia, A.; Atreya, H.S. Enhanced Stability and Activity of an Antimicrobial Peptide in Conjugation with Silver Nanoparticle. J. Colloid Interface Sci. 2016, 483, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Huo, L.; Zhang, K.; Ling, J.; Peng, Z.; Huang, X.; Liu, H.; Gu, L. Antimicrobial and DNA-Binding Activities of the Peptide Fragments of Human Lactoferrin and Histatin 5 against Streptococcus Mutans. Arch. Oral Biol. 2011, 56, 869–876. [Google Scholar] [CrossRef]
- De Leersnyder, I.; De Gelder, L.; Van Driessche, I.; Vermeir, P. Revealing the Importance of Aging, Environment, Size and Stabilization Mechanisms on the Stability of Metal Nanoparticles: A Case Study for Silver Nanoparticles in a Minimally Defined and Complex Undefined Bacterial Growth Medium. Nanomater 2019, 9, 1684. [Google Scholar] [CrossRef]
- Sun, J.; Ma, D.; Zhang, H.; Liu, X.; Han, X.; Bao, X.; Weinberg, G.; Pfänder, N.; Su, D. Toward Monodispersed Silver Nanoparticles with Unusual Thermal Stability. J. Am. Chem. Soc. 2006, 128, 15756–15764. [Google Scholar] [CrossRef]
- Dheyab, M.A.; Aziz, A.A.; Jameel, M.S.; Noqta, O.A.; Khaniabadi, P.M.; Mehrdel, B. Simple Rapid Stabilization Method through Citric Acid Modification for Magnetite Nanoparticles. Sci. Rep. 2020, 10, 10793. [Google Scholar] [CrossRef]
- Ranoszek-Soliwoda, K.; Tomaszewska, E.; Socha, E.; Krzyczmonik, P.; Ignaczak, A.; Orlowski, P.; Krzyzowska, M.; Celichowski, G.; Grobelny, J. The Role of Tannic Acid and Sodium Citrate in the Synthesis of Silver Nanoparticles. J. Nanopart. Res. 2017, 19, 273. [Google Scholar] [CrossRef]
- La Spina, R.; Mehn, D.; Fumagalli, F.; Rossi, F.; Gilliland, D.; Holland, M.; Reniero, F. Synthesis of Citrate-Stabilized Silver Nanoparticles Modified by Thermal and Ph Preconditioned Tannic Acid. Nanomaterials 2020, 10, 2031. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Ji, X.; Jing, J.; Liu, H.; Wu, H.; Yang, W. Size Control over Spherical Silver Nanoparticles by Ascorbic Acid Reduction. Colloids Surf. A Physicochem. Eng. Asp. 2010, 372, 172–176. [Google Scholar] [CrossRef]
- Huang, K.-W.; Hsieh, C.-W.; Kan, H.-C.; Hsieh, M.-L.; Hsieh, S.; Chau, L.-K.; Cheng, T.-E.; Lin, W.-T. Improved Performance of Aminopropylsilatrane over Aminopropyltriethoxysilane as a Linker for Nanoparticle-Based Plasmon Resonance Sensors. Sens. Actuators B Chem. 2012, 163, 207–215. [Google Scholar] [CrossRef]
- Dharanivasan, G.; Rajamuthuramalingam, T.; Michael Immanuel Jesse, D.; Rajendiran, N.; Kathiravan, K. Gold Nanoparticles Assisted Characterization of Amine Functionalized Polystyrene Multiwell Plate and Glass Slide Surfaces. Appl. Nanosci. 2015, 5, 39–50. [Google Scholar] [CrossRef]
- PJM Brouwer, C. Structure-Activity Relationship Study of Synthetic Variants Derived from the Highly Potent Human Antimicrobial Peptide HLF(1-11). Cohesive J. Microbiol. Infect. Dis. 2018, 1, 1–21. [Google Scholar] [CrossRef]
- Gebru, H.; Cui, S.; Li, Z.; Wang, X.; Pan, X.; Liu, J.; Guo, K. Facile PH-Dependent Synthesis and Characterization of Catechol Stabilized Silver Nanoparticles for Catalytic Reduction of 4-Nitrophenol. Catal. Lett. 2017, 147, 2134–2143. [Google Scholar] [CrossRef]
- Lin, Y.; Chen, C.; Wang, C.; Pu, F.; Ren, J.; Qu, X. Silver Nanoprobe for Sensitive and Selective Colorimetric Detection of Dopaminevia Robust Ag–Catechol Interaction. Chem. Commun. 2011, 47, 1181–1183. [Google Scholar] [CrossRef]
- Godoy-Gallardo, M.; Guillem-Marti, J.; Sevilla, P.; Manero, J.M.; Gil, F.J.; Rodriguez, D. Anhydride-Functional Silane Immobilized onto Titanium Surfaces Induces Osteoblast Cell Differentiation and Reduces Bacterial Adhesion and Biofilm Formation. Mater. Sci. Eng. C 2016, 59, 524–532. [Google Scholar] [CrossRef]
- Noronha, V.T.; Paula, A.J.; Durán, G.; Galembeck, A.; Cogo-Müller, K.; Franz-Montan, M.; Durán, N. Silver Nanoparticles in Dentistry. Dent. Mater. 2017, 33, 1110–1126. [Google Scholar] [CrossRef]
- Hoyos-Nogués, M.; Falgueras-Batlle, E.; Ginebra, M.-P.; Manero, J.; Gil, J.; Mas-Moruno, C. A Dual Molecular Biointerface Combining RGD and KRSR Sequences Improves Osteoblastic Functions by Synergizing Integrin and Cell-Membrane Proteoglycan Binding. Int. J. Mol. Sci. 2019, 20, 1429. [Google Scholar] [CrossRef] [PubMed]
- Hoyos-Nogués, M.; Buxadera-Palomero, J.; Ginebra, M.-P.; Manero, J.M.; Gil, F.J.; Mas-Moruno, C. All-in-One Trifunctional Strategy: A Cell Adhesive, Bacteriostatic and Bactericidal Coating for Titanium Implants. Colloids Surfaces B Biointerfaces 2018, 169, 30–40. [Google Scholar] [CrossRef]
- Harris, M.R.; Coote, P.J. Combination of Caspofungin or Anidulafungin with Antimicrobial Peptides Results in Potent Synergistic Killing of Candida Albicans and Candida Glabrata in Vitro. Int. J. Antimicrob. Agents 2010, 35, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Lupetti, A.; Paulusma-Annema, A.; Welling, M.M.; Dogterom-Ballering, H.; Brouwer, C.P.J.M.; Senesi, S.; Van Dissel, J.T.; Nibbering, P.H. Synergistic Activity of the N-Terminal Peptide of Human Lactoferrin and Fluconazole against Candida Species. Antimicrob. Agents Chemother. 2003, 47, 262–267. [Google Scholar] [CrossRef]
- Łoboda, D.; Kozłowski, H.; Rowińska-Żyrek, M. Antimicrobial Peptide–Metal Ion Interactions—A Potential Way of Activity Enhancement. New J. Chem. 2018, 42, 7560–7568. [Google Scholar] [CrossRef]
- Mohanty, S.; Jena, P.; Mehta, R.; Pati, R.; Banerjee, B.; Patil, S.; Sonawane, A. Cationic Antimicrobial Peptides and Biogenic Silver Nanoparticles Kill Mycobacteria without Eliciting DNA Damage and Cytotoxicity in Mouse Macrophages. Antimicrob. Agents Chemother. 2013, 57, 3688–3698. [Google Scholar] [CrossRef]
- Wu, G.; Ding, J.; Li, H.; Li, L.; Zhao, R.; Shen, Z.; Fan, X.; Xi, T. Effects of Cations and PH on Antimicrobial Activity of Thanatin and S-Thanatin against Escherichia Coli ATCC25922 and B. Subtilis ATCC 21332. Curr. Microbiol. 2008, 57, 552–557. [Google Scholar] [CrossRef]
- Zharkova, M.S.; Golubeva, O.Y.; Orlov, D.S.; Vladimirova, E.V.; Dmitriev, A.V.; Tossi, A.; Shamova, O.V. Silver Nanoparticles Functionalized With Antimicrobial Polypeptides: Benefits and Possible Pitfalls of a Novel Anti-Infective Tool. Front. Microbiol. 2021, 12, 750556. [Google Scholar] [CrossRef]
- Li, W.; Li, Y.; Sun, P.; Zhang, N.; Zhao, Y.; Qin, S.; Zhao, Y. Antimicrobial Peptide-Modified Silver Nanoparticles for Enhancing the Antibacterial Efficacy. RSC Adv. 2020, 10, 38746–38754. [Google Scholar] [CrossRef] [PubMed]
- Wigginton, N.S.; de Titta, A.; Piccapietra, F.; Dobias, J.; Nesatyy, V.J.; Suter, M.J.F.; Bernier-Latmani, R. Binding of Silver Nanoparticles to Bacterial Proteins Depends on Surface Modifications and Inhibits Enzymatic Activity. Environ. Sci. Technol. 2010, 44, 2163–2168. [Google Scholar] [CrossRef]
- Dadosh, T. Synthesis of Uniform Silver Nanoparticles with a Controllable Size. Mater. Lett. 2009, 63, 2236–2238. [Google Scholar] [CrossRef]
- Mehta, B.K.; Chhajlani, M.; Shrivastava, B.D. Green Synthesis of Silver Nanoparticles and Their Characterization by XRD. J. Phys. Conf. Ser. 2017, 836, 12050. [Google Scholar] [CrossRef]
- Martin-Gómez, H.; Oliver-Cervelló, L.; Sánchez-Campillo, I.; Marchán, V.; Ginebra, M.-P.; Mas-Moruno, C. A Versatile Click Chemistry-Based Approach for Functionalizing Biomaterials of Diverse Nature with Bioactive Peptides. Chem. Commun. 2021, 57, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Oliver-Cervelló, L.; Martin-Gómez, H.; Mandakhbayar, N.; Jo, Y.W.; Cavalcanti-Adam, E.A.; Kim, H.W.; Ginebra, M.P.; Lee, J.H.; Mas-Moruno, C. Mimicking Bone Extracellular Matrix: From BMP-2-Derived Sequences to Osteogenic-Multifunctional Coatings. Adv. Healthc. Mater. 2022, 11, e2201339. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Ni, Y.; Kong, X.; Wang, Y.; Zhang, L. A Mussel-like Inspired Modification of BaTiO3 Nanopartciles Using Catechol/Polyamine Co-Deposition and Silane Grafting for High-Performance Dielectric Elastomer Composites. Compos. Part B Eng. 2019, 172, 621–627. [Google Scholar] [CrossRef]
- Tran, N.T.; Flanagan, D.P.; Orlicki, J.A.; Lenhart, J.L.; Proctor, K.L.; Knorr, D.B.J. Polydopamine and Polydopamine–Silane Hybrid Surface Treatments in Structural Adhesive Applications. Langmuir 2018, 34, 1274–1286. [Google Scholar] [CrossRef] [PubMed]




| DLS | XRD | TEM | |
|---|---|---|---|
| AgNPs mean diameter [nm] | 64.0 ± 1.0 | 47.5 ± 5.6 | 32.1 ± 9.4 |
| Condition | C (1s) | N (1s) | O (1s) | Si (2p) | Ag (3d) | Ti (2p) |
|---|---|---|---|---|---|---|
| Ti | 28.4 ± 6.5 | 0.5 ± 0.1 | 53.1 ± 4.6 | N.D. | N.D. | 17.8 ± 1.9 |
| Ti-APTES | 36.4 ± 0.9 | 4.6 ± 0.1 | 42.7 ± 0.4 | 5.9 ± 0.1 | N.D. | 10.3 ± 0.4 |
| Ti-AgNPs | 39.2 ± 0.8 | 3.1 ± 0.6 | 26.9 ± 0.6 | 2.9 ± 0.1 | 22.1 ± 1.1 | 5.9 ± 0.4 |
| Ti-PLATF | 40.4 ± 0.4 | 8.1 ± 0.1 | 38.4 ± 0.2 | 0.7 ± 0.1 | N.D. | 12.3 ± 0.1 |
| Ti-Ag/PLATF | 46.4 ± 0.3 | 7.9 ± 0.1 | 25.0 ± 0.2 | 1.8 ± 0.2 | 13.4 ± 0.1 | 5.4 ± 0.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno, D.; Buxadera-Palomero, J.; Ginebra, M.-P.; Manero, J.-M.; Martin-Gómez, H.; Mas-Moruno, C.; Rodríguez, D. Comparison of the Antibacterial Effect of Silver Nanoparticles and a Multifunctional Antimicrobial Peptide on Titanium Surface. Int. J. Mol. Sci. 2023, 24, 9739. https://doi.org/10.3390/ijms24119739
Moreno D, Buxadera-Palomero J, Ginebra M-P, Manero J-M, Martin-Gómez H, Mas-Moruno C, Rodríguez D. Comparison of the Antibacterial Effect of Silver Nanoparticles and a Multifunctional Antimicrobial Peptide on Titanium Surface. International Journal of Molecular Sciences. 2023; 24(11):9739. https://doi.org/10.3390/ijms24119739
Chicago/Turabian StyleMoreno, Daniel, Judit Buxadera-Palomero, Maria-Pau Ginebra, José-María Manero, Helena Martin-Gómez, Carlos Mas-Moruno, and Daniel Rodríguez. 2023. "Comparison of the Antibacterial Effect of Silver Nanoparticles and a Multifunctional Antimicrobial Peptide on Titanium Surface" International Journal of Molecular Sciences 24, no. 11: 9739. https://doi.org/10.3390/ijms24119739
APA StyleMoreno, D., Buxadera-Palomero, J., Ginebra, M.-P., Manero, J.-M., Martin-Gómez, H., Mas-Moruno, C., & Rodríguez, D. (2023). Comparison of the Antibacterial Effect of Silver Nanoparticles and a Multifunctional Antimicrobial Peptide on Titanium Surface. International Journal of Molecular Sciences, 24(11), 9739. https://doi.org/10.3390/ijms24119739









