COVID-19 Therapeutic Potential of Natural Products
Abstract
1. Introduction
2. SARS-CoV-2 and Its Life Cycle
3. Methodology
4. Pre-Infection Inhibitors, Their Proposed Mechanisms, and Their Potency against SARS-CoV-2
5. Post-Infection Inhibitors, Their Proposed Mechanisms, and Their Potency against SARS-CoV-2
6. Multi-Stage Inhibitors, Their Proposed Mechanisms, and Their Potency against SARS-CoV-2
7. Natural Products with Immunomodulatory Effects
8. Application Strategies
8.1. Combinational Therapy of Natural Compounds
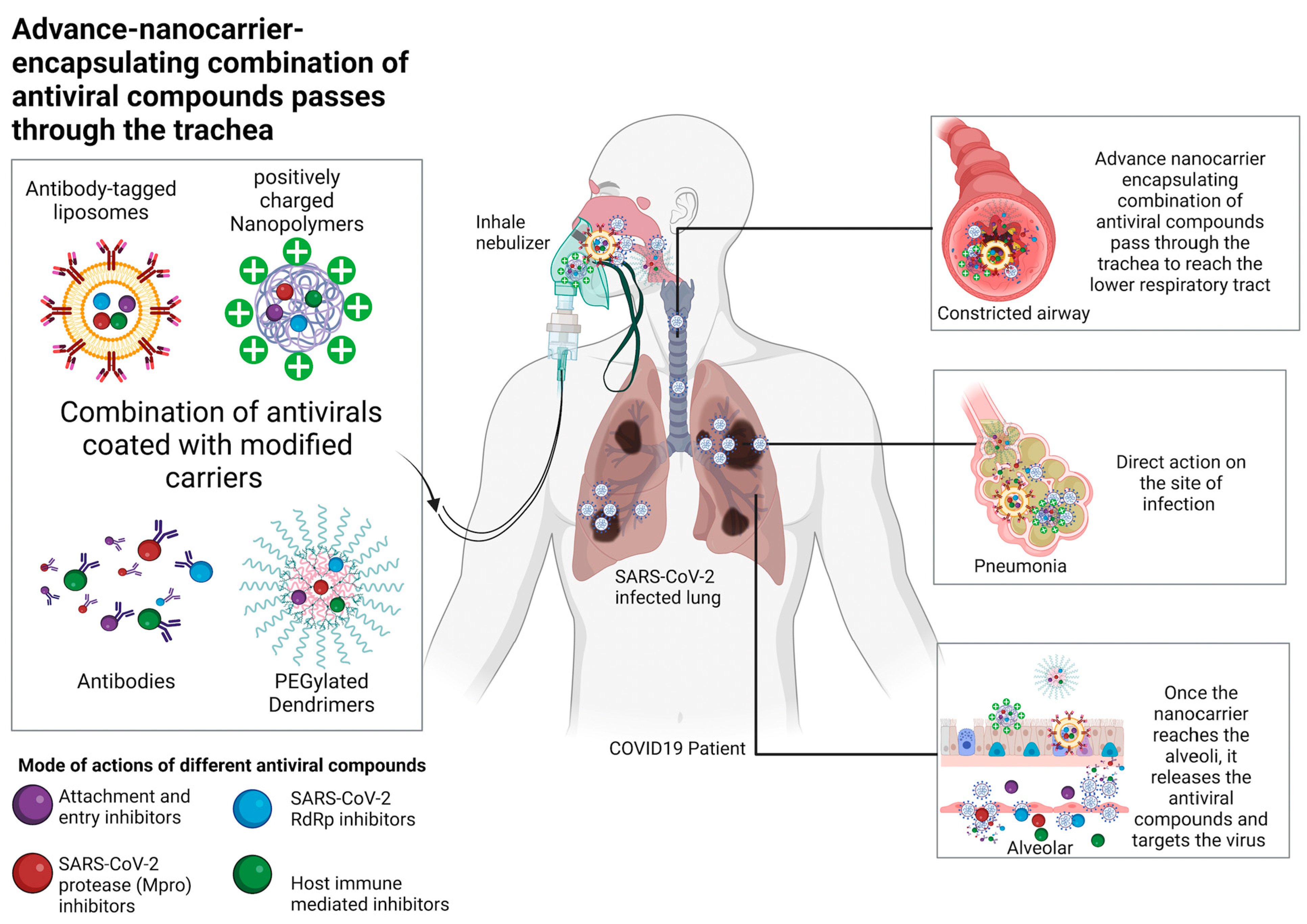
8.2. Potential Delivery Methods of Natural Compounds
8.3. Potential Route of Administration
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability statement
Acknowledgments
Conflicts of Interest
References
- Worldometer Coronavirus. Available online: https://www.worldometers.info/coronavirus/ (accessed on 17 September 2021).
- Sharma, O.; Sultan, A.A.; Ding, H.; Triggle, C.R. A Review of the Progress and Challenges of Developing a Vaccine for COVID-19. Front. Immunol. 2020, 11, 585354. [Google Scholar] [CrossRef] [PubMed]
- Smadja, D.M.; Yue, Q.-Y.; Chocron, R.; Sanchez, O.; Lillo-Le Louet, A. Vaccination against COVID-19: Insight from Arterial and Venous Thrombosis Occurrence Using Data from VigiBase. Eur. Respir. J. 2021, 58, 2100956. [Google Scholar] [CrossRef] [PubMed]
- Albert, E.; Aurigemma, G.; Saucedo, J.; Gerson, D.S. Myocarditis Following COVID-19 Vaccination. Radiol. Case Rep. 2021, 16, 2142–2145. [Google Scholar] [CrossRef] [PubMed]
- McLean, K.; Johnson, T.J. Myopericarditis in a Previously Healthy Adolescent Male Following COVID-19 Vaccination: A Case Report. Acad. Emerg. Med. 2021, 28, 918–921. [Google Scholar] [CrossRef] [PubMed]
- Ison, M.G.; Wolfe, C.; Boucher, H.W. Emergency Use Authorization of Remdesivir: The Need for a Transparent Distribution Process. JAMA 2020, 323, 2365–2366. [Google Scholar] [CrossRef]
- Chen, P.; Nirula, A.; Heller, B.; Gottlieb, R.L.; Boscia, J.; Morris, J.; Huhn, G.; Cardona, J.; Mocherla, B.; Stosor, V.; et al. SARS-CoV-2 Neutralizing Antibody LY-CoV555 in Outpatients with COVID-19. N. Engl. J. Med. 2021, 384, 229–237. [Google Scholar] [CrossRef]
- COVID-19 Treatment Guidelines Hospitalized Adults: Therapeutic Management. Available online: https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/hospitalized-adults--therapeutic-management/ (accessed on 20 October 2021).
- Usher, A.D. The Global COVID-19 Treatment Divide. Lancet 2022, 399, 779–782. [Google Scholar] [CrossRef]
- Josephson, A.; Kilic, T.; Michler, J.D. Socioeconomic Impacts of COVID-19 in Low-Income Countries. Nat. Hum. Behav. 2021, 5, 557–565. [Google Scholar] [CrossRef]
- Sun, Y.Y.; Li, M.; Lenzen, M.; Malik, A.; Pomponi, F. Tourism, Job Vulnerability and Income Inequality during the COVID-19 Pandemic: A Global Perspective. Ann. Tour. 2022, 3, 100046. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Bianchi, M.; Benvenuto, D.; Giovanetti, M.; Angeletti, S.; Ciccozzi, M.; Pascarella, S. SARS-CoV-2 Envelope and Membrane Proteins: Structural Differences Linked to Virus Characteristics? BioMed Res. Int. 2020, 2020, 4389089. [Google Scholar] [CrossRef] [PubMed]
- Mariano, G.; Farthing, R.J.; Lale-Farjat, S.L.M.; Bergeron, J.R.C. Structural Characterization of SARS-CoV-2: Where We Are, and Where We Need to Be. Front. Mol. Biosci. 2020, 7, 344. [Google Scholar] [CrossRef]
- Huang, Y.; Yang, C.; Xu, X.; Xu, W.; Liu, S. Structural and Functional Properties of SARS-CoV-2 Spike Protein: Potential Antivirus Drug Development for COVID-19. Acta Pharmacol. Sin. 2020, 41, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A Pneumonia Outbreak Associated with a New Coronavirus of Probable Bat Origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef]
- Mousavizadeh, L.; Ghasemi, S. Genotype and Phenotype of COVID-19: Their Roles in Pathogenesis. J. Microbiol. Immunol. Infect. 2021, 54, 159–163. [Google Scholar] [CrossRef]
- Kim, D.; Lee, J.Y.; Yang, J.S.; Kim, J.W.; Kim, V.N.; Chang, H. The Architecture of SARS-CoV-2 Transcriptome. Cell 2020, 181, 914–921. [Google Scholar] [CrossRef]
- Brant, A.C.; Tian, W.; Majerciak, V.; Yang, W.; Zheng, Z.-M. SARS-CoV-2: From Its Discovery to Genome Structure, Transcription, and Replication. Cell Biosci. 2021, 11, 136. [Google Scholar] [CrossRef] [PubMed]
- Moustaqil, M.; Ollivier, E.; Chiu, H.-P.; van Tol, S.; Rudolffi-Soto, P.; Stevens, C.; Bhumkar, A.; Hunter, D.J.B.; Freiberg, A.N.; Jacques, D.; et al. SARS-CoV-2 Proteases PLpro and 3CLpro Cleave IRF3 and Critical Modulators of Inflammatory Pathways (NLRP12 and TAB1): Implications for Disease Presentation across Species. Emerg. Microbes Infect. 2021, 10, 178–195. [Google Scholar] [CrossRef]
- Yan, S.; Wu, G. Potential 3-Chymotrypsin-like Cysteine Protease Cleavage Sites in the Coronavirus Polyproteins Pp1a and Pp1ab and Their Possible Relevance to COVID-19 Vaccine and Drug Development. FASEB J. 2021, 35, 21573. [Google Scholar] [CrossRef]
- Michel, C.J.; Mayer, C.; Poch, O.; Thompson, J.D. Characterization of Accessory Genes in Coronavirus Genomes. Virol. J. 2020, 17, 131. [Google Scholar] [CrossRef]
- Gordon, D.E.; Jang, G.M.; Bouhaddou, M.; Xu, J.; Obernier, K.; White, K.M.; O’Meara, M.J.; Rezelj, V.; Guo, J.Z.; Swaney, D.L.; et al. A SARS-CoV-2 Protein Interaction Map Reveals Targets for Drug Repurposing. Nature 2020, 583, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Long, X.; Zhang, B.; Zhang, W.; Chen, X.; Zhang, Z. ACE2 Expression in Pancreas May Cause Pancreatic Damage After SARS-CoV-2 Infection. Clin. Gastroenterol. Hepatol. 2020, 18, 2128–2130. [Google Scholar] [CrossRef] [PubMed]
- Trypsteen, W.; Van Cleemput, J.; van Snippenberg, W.; Gerlo, S.; Vandekerckhove, L. On the Whereabouts of SARS-CoV-2 in the Human Body: A Systematic Review. PLoS Pathog. 2020, 16, e1009037. [Google Scholar] [CrossRef] [PubMed]
- da Silva, S.R.; Ju, E.; Meng, W.; Mondolfi, A.E.P.; Dacic, S.; Green, A.; Bryce, C.; Grimes, Z.; Fowkes, M.; Sordillo, E.M.; et al. Broad SARS-CoV-2 Cell Tropism and Immunopathology in Lung Tissues from Fatal COVID-19. J. Infect. Dis. 2021, 223, 1842–1854. [Google Scholar] [CrossRef]
- Liu, J.; Li, Y.; Liu, Q.; Yao, Q.; Wang, X.; Zhang, H.; Chen, R.; Ren, L.; Min, J.; Deng, F.; et al. SARS-CoV-2 Cell Tropism and Multiorgan Infection. Cell Discov. 2021, 7, 17. [Google Scholar] [CrossRef]
- Datta, P.K.; Liu, F.; Fischer, T.; Rappaport, J.; Qin, X. SARS-CoV-2 Pandemic and Research Gaps: Understanding SARS-CoV-2 Interaction with the ACE2 Receptor and Implications for Therapy. Theranostics 2020, 10, 7448–7464. [Google Scholar] [CrossRef]
- Hamming, I.; Timens, W.; Bulthuis, M.; Lely, A.; Navis, G.; Goor, H. van Tissue Distribution of ACE2 Protein, the Functional Receptor for SARS Coronavirus. A First Step in Understanding SARS Pathogenesis. J. Pathol. 2004, 203, 631. [Google Scholar] [CrossRef] [PubMed]
- To, K.; Tong, J.H.; Chan, P.K.; Au, F.W.; Chim, S.S.; Chan, K.A.; Cheung, J.L.; Liu, E.Y.; Tse, G.M.; Lo, A.W.; et al. Tissue and Cellular Tropism of the Coronavirus Associated with Severe Acute Respiratory Syndrome: An In-situ Hybridization Study of Fatal Cases. J. Pathol. 2004, 202, 157. [Google Scholar] [CrossRef]
- Shang, J.; Ye, G.; Shi, K.; Wan, Y.; Luo, C.; Aihara, H.; Geng, Q.; Auerbach, A.; Li, F. Structural Basis of Receptor Recognition by SARS-CoV-2. Nature 2020, 581, 221–224. [Google Scholar] [CrossRef]
- Shang, J.; Wan, Y.; Luo, C.; Ye, G.; Geng, Q.; Auerbach, A.; Li, F. Cell Entry Mechanisms of SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2020, 117, 11727–11734. [Google Scholar] [CrossRef]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 Spike Receptor-Binding Domain Bound to the ACE2 Receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Örd, M.; Faustova, I.; Loog, M. The Sequence at Spike S1/S2 Site Enables Cleavage by Furin and Phospho-Regulation in SARS-CoV2 but Not in SARS-CoV1 or MERS-CoV. Sci. Rep. 2020, 10, 16944. [Google Scholar] [CrossRef] [PubMed]
- Bestle, D.; Heindl, M.R.; Limburg, H.; Van, T.V.L.; Pilgram, O.; Moulton, H.; Stein, D.A.; Hardes, K.; Eickmann, M.; Dolnik, O.; et al. TMPRSS2 and Furin Are Both Essential for Proteolytic Activation of SARS-CoV-2 in Human Airway Cells. Life Sci. Alliance 2020, 3, e202000786. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Jaimes, J.A.; Bidon, M.K.; Straus, M.R.; Daniel, S.; Whittaker, G.R. Proteolytic Activation of SARS-CoV-2 Spike at the S1/S2 Boundary: Potential Role of Proteases beyond Furin. ACS Infect. Dis. 2021, 7, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Romano, M.; Ruggiero, A.; Squeglia, F.; Maga, G.; Berisio, R. A Structural View of SARS-CoV-2 RNA Replication Machinery: RNA Synthesis, Proofreading and Final Capping. Cells 2020, 9, 1267. [Google Scholar] [CrossRef]
- Hillen, H.S.; Kokic, G.; Farnung, L.; Dienemann, C.; Tegunov, D.; Cramer, P. Structure of Replicating SARS-CoV-2 Polymerase. Nature 2020, 584, 154–156. [Google Scholar] [CrossRef]
- Yan, L.; Zhang, Y.; Ge, J.; Zheng, L.; Gao, Y.; Wang, T.; Jia, Z.; Wang, H.; Huang, Y.; Li, M.; et al. Architecture of a SARS-CoV-2 Mini Replication and Transcription Complex. Nat. Commun. 2020, 11, 5874. [Google Scholar] [CrossRef]
- Decroly, E. Crystal Structure and Functional Analysis of the SARS-Coronavirus RNA Cap 2’-O-Methyltransferase Nsp10/Nsp16 Complex. PLoS Pathog. 2011, 7, e1002059. [Google Scholar] [CrossRef]
- Chen, Y. Biochemical and Structural Insights into the Mechanisms of SARS Coronavirus RNA Ribose 2’-O-Methylation by Nsp16/Nsp10 Protein Complex. PLoS Pathog. 2011, 7, e1002294. [Google Scholar] [CrossRef]
- Ma, Y.; Wu, L.; Shaw, N.; Gao, Y.; Wang, J.; Sun, Y.; Lou, Z.; Yan, L.; Zhang, R.; Rao, Z. Structural Basis and Functional Analysis of the SARS Coronavirus Nsp14–Nsp10 Complex. Proc. Natl. Acad. Sci. USA 2015, 112, 9436–9441. [Google Scholar] [CrossRef]
- Knoops, K.; Kikkert, M.; Worm, S.H.E.; Zevenhoven-Dobbe, J.C.; van der Meer, Y.; Koster, A.J.; Mommaas, A.M.; Snijder, E.J. SARS-Coronavirus Replication Is Supported by a Reticulovesicular Network of Modified Endoplasmic Reticulum. PLoS Biol. 2008, 6, e226. [Google Scholar] [CrossRef]
- Kanjanahaluethai, A.; Chen, Z.; Jukneliene, D.; Baker, S.C. Membrane Topology of Murine Coronavirus Replicase Nonstructural Protein 3. Virology 2007, 361, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Oostra, M.; Lintelo, E.G.; Deijs, M.; Verheije, M.H.; Rottier, P.J.M.; Haan, C.A.M. de Localization and Membrane Topology of Coronavirus Nonstructural Protein 4: Involvement of the Early Secretory Pathway in Replication. J. Virol. 2007, 81, 12323. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus Infections and Immune Responses. J. Med. Virol. 2020, 92, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Florindo, H.F.; Kleiner, R.; Vaskovich-Koubi, D.; Acúrcio, R.C.; Carreira, B.; Yeini, E.; Tiram, G.; Liubomirski, Y.; Satchi-Fainaro, R. Immune-Mediated Approaches against COVID-19. Nat. Nanotechnol. 2020, 15, 630–645. [Google Scholar] [CrossRef]
- Mendonça, L.; Howe, A.; Gilchrist, J.B.; Sheng, Y.; Sun, D.; Knight, M.L.; Zanetti-Domingues, L.C.; Bateman, B.; Krebs, A.-S.; Chen, L.; et al. Correlative Multi-Scale Cryo-Imaging Unveils SARS-CoV-2 Assembly and Egress. Nat. Commun. 2021, 12, 4629. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.; Cao, Y.; Liu, W.; Li, J. The SARS-CoV-2 Nucleocapsid Protein and Its Role in Viral Structure, Biological Functions, and a Potential Target for Drug or Vaccine Mitigation. Viruses 2021, 13, 1115. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Gao, Y.; Liu, X.; Nie, Z.; Sun, H.; Lin, K.; Peng, H.; Wang, S. Identification and Functional Analysis of the SARS-CoV-2 Nucleocapsid Protein. BMC Microbiol. 2021, 21, 58. [Google Scholar] [CrossRef]
- He, R.; Leeson, A.; Ballantine, M.; Andonov, A.; Baker, L.; Dobie, F.; Li, Y.; Bastien, N.; Feldmann, H.; Strocher, U.; et al. Characterization of Protein–Protein Interactions between the Nucleocapsid Protein and Membrane Protein of the SARS Coronavirus. Virus Res. 2004, 105, 121–125. [Google Scholar] [CrossRef]
- Lu, S.; Ye, Q.; Singh, D.; Cao, Y.; Diedrich, J.K.; Yates, J.R.; Villa, E.; Cleveland, D.W.; Corbett, K.D. The SARS-CoV-2 Nucleocapsid Phosphoprotein Forms Mutually Exclusive Condensates with RNA and the Membrane-Associated M Protein. Nat. Commun. 2021, 12, 502. [Google Scholar] [CrossRef]
- Su, H.X.; Yao, S.; Zhao, W.F.; Li, M.J.; Liu, J.; Shang, W.J.; Xie, H.; Ke, C.Q.; Hu, H.C.; Gao, M.N.; et al. Anti-SARS-CoV-2 Activities in Vitro of Shuanghuanglian Preparations and Bioactive Ingredients. Acta Pharmacol. Sin. 2020, 41, 1167–1177. [Google Scholar] [CrossRef] [PubMed]
- Zandi, K.; Musall, K.; Oo, A.; Cao, D.; Liang, B.; Hassandarvish, P.; Lan, S.; Slack, R.L.; Kirby, K.A.; Bassit, L.; et al. Baicalein and Baicalin Inhibit SARS-CoV-2 Rna-Dependent-Rna Polymerase. Microorganisms 2021, 9, 893. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Pan, W.; Li, R.; Liu, B.; Li, C.; Xie, Y.; Wang, Z.; Zhao, J.; Jiang, H.; Huang, J.; et al. Liu Shen Capsule Shows Antiviral and Anti-Inflammatory Abilities against Novel Coronavirus SARS-CoV-2 via Suppression of NF-ΚB Signaling Pathway. Pharmacol. Res. 2020, 158, 104850. [Google Scholar] [CrossRef] [PubMed]
- Runfeng, L.; Yunlong, H.; Jicheng, H.; Weiqi, P.; Qinhai, M.; Yongxia, S.; Chufang, L.; Jin, Z.; Zhenhua, J.; Haiming, J.; et al. Lianhuaqingwen Exerts Anti-Viral and Anti-Inflammatory Activity against Novel Coronavirus (SARS-CoV-2). Pharmacol. Res. 2020, 156, 104761. [Google Scholar] [CrossRef]
- Kageyama, Y.; Aida, K.; Kawauchi, K.; Morimoto, M.; Ebisui, T.; Akiyama, T.; Nakamura, T. Jinhua Qinggan Granule, a Chinese Herbal Medicine against COVID-19, Induces Rapid Changes in the Plasma Levels of IL-6 and IFN-γ. World Acad. Sci. J. 2021, 4, 1–8. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Z.; Zhang, S.; Wang, W.; Jiang, X. Clinical Observation of Xuebijing in the Treatment of COVID-19. Chin. J. Hosp. Pharm. 2020, 40, 964–967. [Google Scholar] [CrossRef]
- Tsai, K.-C.; Huang, Y.-C.; Liaw, C.-C.; Tsai, C.-I.; Chiou, C.-T.; Lin, C.-J.; Wei, W.-C.; Lin, S.J.-S.; Tseng, Y.-H.; Yeh, K.-M.; et al. A Traditional Chinese Medicine Formula NRICM101 to Target COVID-19 through Multiple Pathways: A Bedside-to-Bench Study. Biomed. Pharmacother. 2021, 133, 111037. [Google Scholar] [CrossRef]
- Jan, J.-T.; Cheng, T.-J.R.; Juang, Y.-P.; Ma, H.-H.; Wu, Y.-T.; Yang, W.-B.; Cheng, C.-W.; Chen, X.; Chou, T.-H.; Shie, J.-J.; et al. Identification of Existing Pharmaceuticals and Herbal Medicines as Inhibitors of SARS-CoV-2 Infection. Proc. Natl. Acad. Sci. USA 2021, 118, e2021579118. [Google Scholar] [CrossRef]
- Du, R.; Cooper, L.; Chen, Z.; Lee, H.; Rong, L.; Cui, Q. Discovery of Chebulagic Acid and Punicalagin as Novel Allosteric Inhibitors of SARS-CoV-2 3CLpro. Antivir. Res. 2021, 190, 105075. [Google Scholar] [CrossRef]
- Nair, M.S.; Huang, Y.; Fidock, D.A.; Polyak, S.J.; Wagoner, J.; Towler, M.J.; Weathers, P.J. Artemisia Annua L. Extracts Inhibit the in Vitro Replication of SARS-CoV-2 and Two of Its Variants. J. Ethnopharmacol. 2021, 274, 114016. [Google Scholar] [CrossRef]
- Roshdy, W.H.; Rashed, H.A.; Kandeil, A.; Mostafa, A.; Moatasim, Y.; Kutkat, O.; Shama, N.M.A.; Gomaa, M.R.; El-Sayed, I.H.; El Guindy, N.M.; et al. EGYVIR: An Immunomodulatory Herbal Extract with Potent Antiviral Activity against SARS-CoV-2. PLoS ONE 2020, 15, e0241739. [Google Scholar] [CrossRef] [PubMed]
- Plante, K.S.; Dwivedi, V.; Plante, J.A.; Fernandez, D.; Mirchandani, D.; Bopp, N.; Aguilar, P.V.; Park, J.G.; Tamayo, P.P.; Delgado, J.; et al. Antiviral Activity of Oleandrin and a Defined Extract of Nerium Oleander against SARS-CoV-2. Biomed. Pharmacother. 2021, 138, 111457. [Google Scholar] [CrossRef] [PubMed]
- Zannella, C.; Giugliano, R.; Chianese, A.; Buonocore, C.; Vitale, G.A.; Sanna, G.; Sarno, F.; Manzin, A.; Nebbioso, A.; Termolino, P.; et al. Antiviral Activity of Vitis Vinifera Leaf Extract against SARS-CoV-2 and HSV-1. Viruses 2021, 13, 1263. [Google Scholar] [CrossRef] [PubMed]
- Enmozhi, S.K.; Raja, K.; Sebastine, I.; Joseph, J. Andrographolide as a Potential Inhibitor of SARS-CoV-2 Main Protease: An in Silico Approach. J. Biomol. Struct. Dyn. 2020, 39, 3092–3098. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.-H.; Huang, Y.-L.; Chen, C.-C.; Pi, W.-C.; Hsu, Y.-L.; Lo, L.-C.; Chen, W.-Y.; Fu, S.-L.; Lin, C.-H. Andrographolide and Its Fluorescent Derivative Inhibit the Main Proteases of 2019-NCoV and SARS-CoV through Covalent Linkage. Biochem. Biophys. Res. Commun. 2020, 533, 467–473. [Google Scholar] [CrossRef]
- Sangiamsuntorn, K.; Suksatu, A.; Pewkliang, Y.; Thongsri, P.; Kanjanasirirat, P.; Manopwisedjaroen, S.; Charoensutthivarakul, S.; Wongtrakoongate, P.; Pitiporn, S.; Chaopreecha, J.; et al. Anti-SARS-CoV-2 Activity of Andrographis Paniculata Extract and Its Major Component Andrographolide in Human Lung Epithelial Cells and Cytotoxicity Evaluation in Major Organ Cell Representatives. J. Nat. Prod. 2021, 84, 1261–1270. [Google Scholar] [CrossRef]
- Takeda, Y.; Jamsransuren, D.; Matsuda, S.; Crea, R.; Ogawa, H. The SARS-CoV-2-Inactivating Activity of Hydroxytyrosol-Rich Aqueous Olive Pulp Extract (HIDROX®) and Its Use as a Virucidal Cream for Topical Application. Viruses 2021, 13, 232. [Google Scholar] [CrossRef]
- Lee, Y.-G.; Kang, K.W.; Hong, W.; Kim, Y.H.; Oh, J.T.; Park, D.W.; Ko, M.; Bai, Y.-F.; Seo, Y.-J.; Lee, S.-M.; et al. Potent Antiviral Activity of Agrimonia Pilosa, Galla Rhois, and Their Components against SARS-CoV-2. Bioorg. Med. Chem. 2021, 45, 116329. [Google Scholar] [CrossRef]
- Henss, L.; Auste, A.; Schürmann, C.; Schmidt, C.; von Rhein, C.; Mühlebach, M.D.; Schnierle, B.S. The Green Tea Catechin Epigallocatechin Gallate Inhibits SARS-CoV-2 Infection. J. Gen. Virol. 2021, 102, 001574. [Google Scholar] [CrossRef]
- Khayrani, A.C.; Irdiani, R.; Aditama, R.; Pratami, D.K.; Lischer, K.; Ansari, M.J.; Chinnathambi, A.; Alharbi, S.A.; Almoallim, H.S.; Sahlan, M. Evaluating the Potency of Sulawesi Propolis Compounds as ACE-2 Inhibitors through Molecular Docking for COVID-19 Drug Discovery Preliminary Study. J. King Saud. Univ. Sci. 2021, 33, 101297. [Google Scholar] [CrossRef]
- Dewi, L.K.; Sahlan, M.; Pratami, D.K.; Agus, A.; Agussalim; Sabir, A. Identifying Propolis Compounds Potential to Be COVID-19 Therapies by Targeting SARS-CoV-2 Main Protease. Int. J. Appl. Pharm. 2021, 13, 103–110. [Google Scholar] [CrossRef]
- Signer, J.; Jonsdottir, H.R.; Albrich, W.C.; Strasser, M.; Züst, R.; Ryter, S.; Ackermann-Gäumann, R.; Lenz, N.; Siegrist, D.; Suter, A.; et al. In Vitro Virucidal Activity of Echinaforce®, an Echinacea Purpurea Preparation, against Coronaviruses, Including Common Cold Coronavirus 229E and SARS-CoV-2. Virol. J. 2020, 17, 136. [Google Scholar] [CrossRef] [PubMed]
- Ao, Z.; Chan, M.; Ouyang, M.J.; Olukitibi, T.A.; Mahmoudi, M.; Kobasa, D.; Yao, X. Identification and Evaluation of the Inhibitory Effect of Prunella Vulgaris Extract on SARS-Coronavirus 2 Virus Entry. PLoS ONE 2021, 16, e0251649. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Ta, W.; Tang, W.; Hua, R.; Wang, J.; Wang, C.; Lu, W. Potential Antiviral Activity of Isorhamnetin against SARS-CoV-2 Spike Pseudotyped Virus in Vitro. Drug Dev. Res. 2021, 82, 1124–1130. [Google Scholar] [CrossRef] [PubMed]
- Owis, A.I.; El-Hawary, M.S.; El Amir, D.; Aly, O.M.; Abdelmohsen, U.R.; Kamel, M.S. Molecular Docking Reveals the Potential of Salvadora Persica Flavonoids to Inhibit COVID-19 Virus Main Protease. RSC Adv. 2020, 10, 19570–19575. [Google Scholar] [CrossRef]
- Pandey, P.; Rane, J.S.; Chatterjee, A.; Kumar, A.; Khan, R.; Prakash, A.; Ray, S. Targeting SARS-CoV-2 Spike Protein of COVID-19 with Naturally Occurring Phytochemicals: An in Silico Study for Drug Development. J. Biomol. Struct. Dyn. 2020, 39, 6306–6316. [Google Scholar] [CrossRef]
- Zhang, Z.R.; Zhang, Y.N.; Li, X.D.; Zhang, H.Q.; Xiao, S.Q.; Deng, F.; Yuan, Z.M.; Ye, H.Q.; Zhang, B. A Cell-Based Large-Scale Screening of Natural Compounds for Inhibitors of SARS-CoV-2. Signal Transduct. Target. Ther. 2020, 5, 218. [Google Scholar] [CrossRef]
- Abdallah, H.M.; El-Halawany, A.M.; Sirwi, A.; El-Araby, A.M.; Mohamed, G.A.; Ibrahim, S.R.M.; Koshak, A.E.; Asfour, H.Z.; Awan, Z.A.; Elfaky, M.A. Repurposing of Some Natural Product Isolates as SARS-CoV-2 Main Protease Inhibitors via In Vitro Cell Free and Cell-Based Antiviral Assessments and Molecular Modeling Approaches. Pharmaceuticals 2021, 14, 213. [Google Scholar] [CrossRef]
- Ellen, B.M.; Kumar, N.D.; Bouma, E.M.; Troost, B.; van de Pol, D.P.I.; van der Ende-Metselaar, H.H.; Apperloo, L.; van Gosliga, D.; van den Berge, M.; Nawijn, M.C.; et al. Resveratrol and Pterostilbene Potently Inhibit SARS-CoV-2 Replication In Vitro. Viruses 2021, 13, 1335. [Google Scholar] [CrossRef]
- Kwon, P.S.; Oh, H.; Kwon, S.-J.; Jin, W.; Zhang, F.; Fraser, K.; Hong, J.J.; Linhardt, R.J.; Dordick, J.S. Sulfated Polysaccharides Effectively Inhibit SARS-CoV-2 in Vitro. Cell Discov. 2020, 6, 50. [Google Scholar] [CrossRef]
- Zhang, S.; Pei, R.; Li, M.; Sun, H.; Su, M.; Ding, Y.; Chen, X.; Du, Z.; Jin, C.; Huang, C.; et al. Structural Characterization of Cocktail-like Targeting Polysaccharides from Ecklonia Kurome Okam and Their Anti-SARS-CoV-2 Activities Invitro. bioRxiv 2021. [Google Scholar] [CrossRef]
- Song, S.; Peng, H.; Wang, Q.; Liu, Z.; Dong, X.; Wen, C.; Ai, C.; Zhang, Y.; Wang, Z.; Zhu, B. Inhibitory Activities of Marine Sulfated Polysaccharides against SARS-CoV-2. Food Funct. 2020, 11, 7415–7420. [Google Scholar] [CrossRef] [PubMed]
- Orfali, R.; Rateb, M.E.; Hassan, H.M.; Alonazi, M.; Gomaa, M.R.; Mahrous, N.; GabAllah, M.; Kandeil, A.; Perveen, S.; Abdelmohsen, U.R.; et al. Sinapic Acid Suppresses SARS-CoV-2 Replication by Targeting Its Envelope Protein. Antibiotics 2021, 10, 420. [Google Scholar] [CrossRef] [PubMed]
- Svenningsen, E.B.; Thyrsted, J.; Blay-Cadanet, J.; Liu, H.; Lin, S.; Moyano-Villameriel, J.; Olagnier, D.; Idorn, M.; Paludan, S.R.; Holm, C.K.; et al. Ionophore Antibiotic X-206 Is a Potent Inhibitor of SARS-CoV-2 Infection in Vitro. Antivir. Res. 2021, 185, 104988. [Google Scholar] [CrossRef]
- Braga, L.; Ali, H.; Secco, I.; Chiavacci, E.; Neves, G.; Goldhill, D.; Penn, R.; Jimenez-Guardeño, J.M.; Ortega-Prieto, A.M.; Bussani, R.; et al. Drugs That Inhibit TMEM16 Proteins Block SARS-CoV-2 Spike-Induced Syncytia. Nature 2021, 594, 88–93. [Google Scholar] [CrossRef]
- Zhang, Y.-N.; Zhang, Q.-Y.; Li, X.-D.; Xiong, J.; Xiao, S.-Q.; Wang, Z.; Zhang, Z.-R.; Deng, C.-L.; Yang, X.-L.; Wei, H.-P.; et al. Gemcitabine, Lycorine and Oxysophoridine Inhibit Novel Coronavirus (SARS-CoV-2) in Cell Culture. Emerg. Microbes Infect. 2020, 9, 1170–1173. [Google Scholar] [CrossRef]
- Marsh, M.; Helenius, A. Virus Entry: Open Sesame. Cell 2006, 124, 729–740. [Google Scholar] [CrossRef]
- Martín, C.S.S.; Liu, C.Y.; Kielian, M. Dealing with Low PH: Entry and Exit of Alphaviruses and Flaviviruses. Trends Microbiol. 2009, 17, 514–521. [Google Scholar] [CrossRef]
- Ohgitani, E.; Shin-Ya, M.; Ichitani, M.; Kobayashi, M.; Takihara, T.; Kawamoto, M.; Kinugasa, H.; Mazda, O. Significant Inactivation of SARS-CoV-2 In Vitro by a Green Tea Catechin, a Catechin-Derivative, and Black Tea Galloylated Theaflavins. Molecules 2021, 26, 3572. [Google Scholar] [CrossRef]
- Rauš, K.; Pleschka, S.; Klein, P.; Schoop, R.; Fisher, P. Effect of an Echinacea-Based Hot Drink Versus Oseltamivir in Influenza Treatment: A Randomized, Double-Blind, Double-Dummy, Multicenter, Noninferiority Clinical Trial. Curr. Ther. Res. Clin. Exp. 2015, 77, 66–72. [Google Scholar] [CrossRef]
- Schapowal, A. Efficacy and Safety of Echinaforce® in Respiratory Tract Infections. Wien. Med. Wochenschr. 2012, 163, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Jawad, M.; Schoop, R.; Suter, A.; Klein, P.; Eccles, R. Safety and Efficacy Profile of Echinacea Purpurea to Prevent Common Cold Episodes: A Randomized, Double-Blind, Placebo-Controlled Trial. Evid.-Based Complement. Altern. Med. 2012, 2012, 841315. [Google Scholar] [CrossRef] [PubMed]
- Nicolussi, S.; Ardjomand-Woelkart, K.; Stange, R.; Gancitano, G.; Klein, P.; Ogal, M. Echinacea as a Potential Force against Coronavirus Infections? A Mini-Review of Randomized Controlled Trials in Adults and Children. Microorganisms 2022, 10, 211. [Google Scholar] [CrossRef]
- Bansal, P.; Goyal, A.; IV, A.C.; Lahan, S.; Dhaliwal, H.S.; Bhyan, P.; Bhattad, P.B.; Aslam, F.; Ranka, S.; Dalia, T.; et al. Hydroxychloroquine: A Comprehensive Review and Its Controversial Role in Coronavirus Disease 2019. Ann. Med. 2020, 53, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Misra, D.P.; Gasparyan, A.Y.; Zimba, O. Benefits and Adverse Effects of Hydroxychloroquine, Methotrexate and Colchicine: Searching for Repurposable Drug Candidates. Rheumatol. Int. 2020, 40, 1741–1751. [Google Scholar] [CrossRef] [PubMed]
- Gevers, S.; Kwa, M.S.G.; Wijnans, E.; Nieuwkoop, C. van Safety Considerations for Chloroquine and Hydroxychloroquine in the Treatment of COVID-19. Clin. Microbiol. Infect. 2020, 26, 1276–1277. [Google Scholar] [CrossRef] [PubMed]
- Karolyi, M.; Omid, S.; Pawelka, E.; Jilma, B.; Stimpfl, T.; Schoergenhofer, C.; Laferl, H.; Seitz, T.; Traugott, M.; Wenisch, C.; et al. High Dose Lopinavir/Ritonavir Does Not Lead to Sufficient Plasma Levels to Inhibit SARS-CoV-2 in Hospitalized Patients With COVID-19. Front. Pharmacol. 2021, 12, 704767. [Google Scholar] [CrossRef]
- Owa, A.B.; Owa, O.T. Lopinavir/Ritonavir Use in COVID-19 Infection: Is It Completely Non-Beneficial? J. Microbiol. Immunol. Infect. 2020, 53, 674–675. [Google Scholar] [CrossRef]
- Vecchio, G.; Zapico, V.; Catanzariti, A.; Carboni Bisso, I.; Las Heras, M. Adverse Effects of Lopinavir/Ritonavir in Critically Ill Patients with COVID-19. Medicina 2020, 80, 439–441. [Google Scholar]
- Cao, B.; Wang, Y.; Wen, D.; Liu, W.; Wang, J.; Fan, G.; Ruan, L.; Song, B.; Cai, Y.; Wei, M.; et al. A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe COVID-19. N. Engl. J. Med. 2020, 382, 1787–1799. [Google Scholar] [CrossRef]
- Gagliardini, R.; Cozzi-Lepri, A.; Mariano, A.; Taglietti, F.; Vergori, A.; Abdeddaim, A.; Di Gennaro, F.; Mazzotta, V.; Amendola, A.; D’Offizi, G.; et al. No Efficacy of the Combination of Lopinavir/Ritonavir Plus Hydroxychloroquine Versus Standard of Care in Patients Hospitalized With COVID-19: A Non-Randomized Comparison. Front. Pharmacol. 2021, 12, 621676. [Google Scholar] [CrossRef] [PubMed]
- Reis, G.; Silva, E.A.D.S.M.; Silva, D.C.M.; Thabane, L.; Singh, G.; Park, J.J.H.; Forrest, J.I.; Harari, O.; dos Santos, C.V.Q.; de Almeida, A.P.F.G.; et al. Effect of Early Treatment With Hydroxychloroquine or Lopinavir and Ritonavir on Risk of Hospitalization Among Patients With COVID-19: The TOGETHER Randomized Clinical Trial. JAMA Netw. Open 2021, 4, e216468. [Google Scholar] [CrossRef] [PubMed]
- Axfors, C.; Schmitt, A.M.; Janiaud, P.; van’t Hooft, J.; Abd-Elsalam, S.; Abdo, E.F.; Abella, B.S.; Akram, J.; Amaravadi, R.K.; Angus, D.C.; et al. Mortality Outcomes with Hydroxychloroquine and Chloroquine in COVID-19 from an International Collaborative Meta-Analysis of Randomized Trials. Nat. Commun. 2021, 12, 3001. [Google Scholar] [CrossRef] [PubMed]
- Funnell, S.G.P.; Dowling, W.E.; Muñoz-Fontela, C.; Gsell, P.-S.; Ingber, D.E.; Hamilton, G.A.; Delang, L.; Rocha-Pereira, J.; Kaptein, S.; Dallmeier, K.H.; et al. Emerging Preclinical Evidence Does Not Support Broad Use of Hydroxychloroquine in COVID-19 Patients. Nat. Commun. 2020, 11, 4253. [Google Scholar] [CrossRef] [PubMed]
- Saghir, S.A.; AlGabri, N.A.; Alagawany, M.M.; Attia, Y.A.; Alyileili, S.R.; Elnesr, S.S.; Shafi, M.E.; Al-shargi, O.Y.; Al-balagi, N.; Alwajeeh, A.S.; et al. Chloroquine and Hydroxychloroquine for the Prevention and Treatment of COVID-19: A Fiction, Hope or Hype? An Updated Review. Ther. Clin. Risk Manag. 2021, 17, 371–387. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, Q.; Zhou, W.; Gao, S.; Lin, H.; Ye, S.; Xu, Y.; Cai, J. Chinese Medicine Injection Shuanghuanglian for Treatment of Acute Upper Respiratory Tract Infection: A Systematic Review of Randomized Controlled Trials. Evid. Based Complement. Altern. Med. 2013, 2013, 987326. [Google Scholar] [CrossRef]
- Gu, S.; Yin, N.; Pei, J.; Lai, L. Understanding Traditional Chinese Medicine Anti-Inflammatory Herbal Formulae by Simulating Their Regulatory Functions in the Human Arachidonic Acid Metabolic Network. Mol. Biosyst. 2013, 9, 1931–1938. [Google Scholar] [CrossRef]
- Tian, S.; He, G.; Song, J.; Wang, S.; Xin, W.; Zhang, D.; Du, G. Pharmacokinetic Study of Baicalein after Oral Administration in Monkeys. Fitoterapia 2012, 83, 532–540. [Google Scholar] [CrossRef]
- Salehi, B.; Fokou, P.V.T.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The Therapeutic Potential of Naringenin: A Review of Clinical Trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef]
- Varughese, J.K.; Libin, K.L.J.; Sindhu, K.S.; Rosily, A.V.; Abi, T.G. Investigation of the Inhibitory Activity of Some Dietary Bioactive Flavonoids against SARS-CoV-2 Using Molecular Dynamics Simulations and MM-PBSA Calculations. J. Biomol. Struct. Dyn. 2022, 40, 6755–6770. [Google Scholar] [CrossRef]
- Pasquereau, S.; Nehme, Z.; Ahmad, S.H.; Daouad, F.; Van Assche, J.; Wallet, C.; Schwartz, C.; Rohr, O.; Morot-Bizot, S.; Herbein, G. Resveratrol Inhibits HCoV-229E and SARS-CoV-2 Coronavirus Replication In Vitro. Viruses 2021, 13, 354. [Google Scholar] [CrossRef]
- Yang, M.; Wei, J.; Huang, T.; Lei, L.; Shen, C.; Lai, J.; Yang, M.; Liu, L.; Yang, Y.; Liu, G.; et al. Resveratrol Inhibits the Replication of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in Cultured Vero Cells. Phytother. Res. 2021, 35, 1127–1129. [Google Scholar] [CrossRef]
- McCreary, M.R.; Schnell, P.M.; Rhoda, D.A. Randomized Double-Blind Placebo-Controlled Proof-of-Concept Trial of Resveratrol for Outpatient Treatment of Mild Coronavirus Disease (COVID-19). Res. Sq. 2021, 12, 10978. [Google Scholar] [CrossRef]
- Meng, T.; Xiao, D.; Muhammed, A.; Deng, J.; Chen, L.; He, J. Anti-Inflammatory Action and Mechanisms of Resveratrol. Molecules 2021, 26, 229. [Google Scholar] [CrossRef] [PubMed]
- She, G.-M.; Xu, C.; Liu, B.; Shi, R.-B. Polyphenolic Acids from Mint (the Aerial of Mentha Haplocalyx Briq.) with DPPH Radical Scavenging Activity. J. Food Sci. 2010, 75, C359–C362. [Google Scholar] [CrossRef] [PubMed]
- Tiryaki Sönmez, G.; Çolak, M.; Sönmez, S.; Schoenfeld, B. Effects of Oral Supplementation of Mint Extract on Muscle Pain and Blood Lactate. Biomed. Hum. Kinet. 2010, 2, 66–69. [Google Scholar] [CrossRef]
- Pengfei, L.; Tiansheng, D.; Xianglin, H.; Jianguo, W. Antioxidant Properties of Isolated Isorhamnetin from the Sea Buckthorn Marc. Plant Foods Hum. Nutr. 2009, 64, 141–145. [Google Scholar] [CrossRef]
- Olas, B.; Skalski, B.; Ulanowska, K. The Anticancer Activity of Sea Buckthorn [Elaeagnus rhamnoides (L.) A. Nelson]. Front. Pharmacol. 2018, 9, 232. [Google Scholar] [CrossRef]
- Dayem, A.A.; Choi, H.Y.; Kim, Y.B.; Cho, S.-G. Antiviral Effect of Methylated Flavonol Isorhamnetin against Influenza. PLoS ONE 2015, 10, e0121610. [Google Scholar] [CrossRef]
- Matboli, M.; Saad, M.; Hasanin, A.H.; Saleh, L.; Baher, W.; Bekhet, M.M.; Eissa, S. New Insight into the Role of Isorhamnetin as a Regulator of Insulin Signaling Pathway in Type 2 Diabetes Mellitus Rat Model: Molecular and Computational Approach. Biomed. Pharmacother. 2021, 135, 111176. [Google Scholar] [CrossRef]
- Liu, C.-M.; Westley, J.W.; Hermann, T.E.; Prosser, B.L.T.; Palleroni, N.; Evans, R.H.; Miller, P.A. Novel Polyether Antibiotics X-14873A, G and H Produced by a Streptomyces: Taxonomy of the Producing Culture, Fermentation, Biological and Ionophorous Properties of the Antibiotics. J. Antibiot. 1986, 39, 1712–1718. [Google Scholar] [CrossRef]
- Liu, C.M.; Westley, J.W.; Chu, J.; Hermann, T.E.; Liu, M.; Palleroni, N. Three Novel Polyether Antibiotics X-14889a, c, and d from a Streptomycete Taxonomy of the Producing Ogranism, Fermentation Production and Biological Properties of the Antibiotics. J. Antibiot. 1993, 46, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Rausch, K.; Hackett, B.; Weinbren, N.; Reeder, S.; Sadovsky, Y.; Hunter, C.; Schultz, D.C.; Coyne, C.; Cherry, S. Screening Bioactives Reveals Nanchangmycin as a Broad Spectrum Antiviral Active against Zika Virus. Cell Rep. 2017, 18, 804–815. [Google Scholar] [CrossRef]
- Nakamura, M.; Kunimoto, S.; Takahashi, Y.; Naganawa, H.; Sakaue, M.; Inoue, S.; Ohno, T.; Takeuchi, T. Inhibitory Effects of Polyethers on Human Immunodeficiency Virus Replication. Antimicrob. Agents Chemother. 1992, 36, 492–494. [Google Scholar] [CrossRef] [PubMed]
- Velthuis, A.J.W.; van den Worm, S.H.E.; Sims, A.C.; Baric, R.S.; Snijder, E.J.; Hemert, M.J. van Zn2+ Inhibits Coronavirus and Arterivirus RNA Polymerase Activity In Vitro and Zinc Ionophores Block the Replication of These Viruses in Cell Culture. PLoS Pathog. 2010, 6, e1001176. [Google Scholar] [CrossRef] [PubMed]
- Cairns, D.M.; Dulko, D.; Griffiths, J.K.; Golan, Y.; Cohen, T.; Trinquart, L.; Price, L.L.; Beaulac, K.R.; Selker, H.P. Efficacy of Niclosamide vs Placebo in SARS-CoV-2 Respiratory Viral Clearance, Viral Shedding, and Duration of Symptoms Among Patients With Mild to Moderate COVID-19: A Phase 2 Randomized Clinical Trial. JAMA Netw. Open 2022, 5, e2144942. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, N.; Rasul, A.; Hussain, G.; Anwar, H.; Shah, M.A.; Sarfraz, I.; Riaz, A.; Batool, R.; Shahbaz, M.; Hussain, A.; et al. Oleandrin: A Bioactive Phytochemical and Potential Cancer Killer via Multiple Cellular Signaling Pathways. Food Chem. Toxicol. 2020, 143, 111570. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Saleem, M.; Aziz, M.H.; Mukhtar, H. Inhibition of 12-O-Tetradecanoylphorbol-13-Acetate-Induced Tumor Promotion Markers in CD-1 Mouse Skin by Oleandrin. Toxicol. Appl. Pharmacol. 2004, 195, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Zhang, Y.; Zhao, W.; Zhou, X.; Wang, C.; Deng, F. The Cardiac Glycoside Oleandrin Induces Apoptosis in Human Colon Cancer Cells via the Mitochondrial Pathway. Cancer Chemother. Pharmacol. 2017, 80, 91–100. [Google Scholar] [CrossRef]
- Yang, C.-W.; Lee, Y.-Z.; Hsu, H.-Y.; Jan, J.-T.; Lin, Y.-L.; Chang, S.-Y.; Peng, T.-T.; Yang, R.-B.; Liang, J.-J.; Liao, C.-C.; et al. Inhibition of SARS-CoV-2 by Highly Potent Broad-Spectrum Anti-Coronaviral Tylophorine-Based Derivatives. Front. Pharmacol. 2020, 11, 2056. [Google Scholar] [CrossRef]
- Diamond, M.S.; Kanneganti, T.D. Innate Immunity: The First Line of Defense against SARS-CoV-2. Nat. Immunol. 2022, 23, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Primorac, D.; Vrdoljak, K.; Brlek, P.; Pavelić, E.; Molnar, V.; Matišić, V.; Erceg Ivkošić, I.; Parčina, M. Adaptive Immune Responses and Immunity to SARS-CoV-2. Front. Immunol. 2022, 13, 2035. [Google Scholar] [CrossRef] [PubMed]
- Sette, A.; Crotty, S. Adaptive Immunity to SARS-CoV-2 and COVID-19. Cell 2021, 184, 861–880. [Google Scholar] [CrossRef] [PubMed]
- Sheikhi, K.; Shirzadfar, H.; Sheikhi, M. A Review on Novel Coronavirus (COVID-19): Symptoms, Transmission and Diagnosis Tests. Res. Infect. Dis. Trop. Med. 2020, 2, 1–8. [Google Scholar] [CrossRef]
- An, P.; Song, P.; Lian, K.; Wang, Y. CT Manifestations of Novel Coronavirus Pneumonia: A Case Report. Balkan Med. J. 2020, 37, 163–165. [Google Scholar] [CrossRef]
- Dupuis, C.; de Montmollin, E.; Buetti, N.; Goldgran-Toledano, D.; Reignier, J.; Schwebel, C.; Domitile, J.; Neuville, M.; Ursino, M.; Siami, S.; et al. Impact of Early Corticosteroids on 60-Day Mortality in Critically Ill Patients with COVID-19: A Multicenter Cohort Study of the OUTCOMEREA Network. PLoS ONE 2021, 16, e0255644. [Google Scholar] [CrossRef]
- van Paassen, J.; Vos, J.S.; Hoekstra, E.M.; Neumann, K.M.I.; Boot, P.C.; Arbous, S.M. Corticosteroid Use in COVID-19 Patients: A Systematic Review and Meta-Analysis on Clinical Outcomes. Crit. Care 2020, 24, 696. [Google Scholar] [CrossRef] [PubMed]
- Group, T.R.C. Dexamethasone in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2020, 384, 693–704. [Google Scholar] [CrossRef]
- Tang, X.; Feng, Y.-M.; Ni, J.-X.; Zhang, J.-Y.; Liu, L.-M.; Hu, K.; Wu, X.-Z.; Zhang, J.-X.; Chen, J.-W.; Zhang, J.-C.; et al. Early Use of Corticosteroid May Prolong SARS-CoV-2 Shedding in Non-Intensive Care Unit Patients with COVID-19 Pneumonia: A Multicenter, Single-Blind, Randomized Control Trial. Respiration 2021, 100, 116–126. [Google Scholar] [CrossRef]
- Berton, A.M.; Prencipe, N.; Giordano, R.; Ghigo, E.; Grottoli, S. Systemic Steroids in Patients with COVID-19: Pros and Contras, an Endocrinological Point of View. J. Endocrinol. Investig. 2021, 44, 873–875. [Google Scholar] [CrossRef]
- Wang, J.; Yang, W.; Chen, P.; Guo, J.; Liu, R.; Wen, P.; Li, K.; Lu, Y.; Ma, T.; Li, X.; et al. The Proportion and Effect of Corticosteroid Therapy in Patients with COVID-19 Infection: A Systematic Review and Meta-Analysis. PLoS ONE 2021, 16, e0249481. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Guan, W.J.; Bi, Y.; Zhang, W.; Li, L.; Zhang, B.; Liu, Q.; Song, Y.; Li, X.; Duan, Z.; et al. Efficacy and Safety of Lianhuaqingwen Capsules, a Repurposed Chinese Herb, in Patients with Coronavirus Disease 2019: A Multicenter, Prospective, Randomized Controlled Trial. Phytomedicine 2021, 85, 153242. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.X.; Li, M.Y.; Chen, X.L.; Ma, M.Y.; Hu, J.H. Clinical Efficacy of Lianhua Qingwen Integrated with Western Medicine on COVID-19 by Meta-Analysis. Chin. Tradit. Herb. Drugs 2020, 14, 3763–3769. [Google Scholar] [CrossRef]
- Yan, H.; Zou, C. Mechanism and Material Basis of LianhuaQingwen Capsule for Improving Clinical Cure Rate of COVID-19: A Study Based on Network Pharmacology and Molecular Docking Technology. Nan Fang Yi Ke Da Xue Xue Bao J. South. Med. Univ. 2021, 41, 20–30. [Google Scholar] [CrossRef]
- Ling, X.Y.; Tao, J.L.; Sun, X.; Yuan, B. Exploring Material Basis and Mechanism of Lianhua Qingwen Prescription against Coronavirus Based on Network Pharmacology. Chin. Tradit. Herb. Drugs 2020, 51, 1723–1730. [Google Scholar] [CrossRef]
- Zhang, Y.; Yao, Y.; Yang, Y.; Wu, H. Investigation of Anti-SARS, MERS, and COVID-19 Effect of Jinhua Qinggan Granules Based on a Network Pharmacology and Molecular Docking Approach. Nat. Prod. Commun. 2021, 16, 1934578X211020619. [Google Scholar] [CrossRef]
- Xing, Y.; Hua, Y.R.; Shang, J.; Ge, W.H.; Liao, J. Traditional Chinese Medicine Network Pharmacology Study on Exploring the Mechanism of Xuebijing Injection in the Treatment of Coronavirus Disease 2019. Chin. J. Nat. Med. 2020, 18, 941–951. [Google Scholar] [CrossRef]
- Ren, Y.; Yin, Z.; Dai, J.; Yang, Z.; Ye, B.; Ma, Y.; Zhang, T.; Shi, Y. Evidence-Based Complementary and Alternative Medicine Exploring Active Components and Mechanism of Jinhua Qinggan Granules in Treatment of COVID-19 Based on Virus-Host Interaction. Nat. Prod. Commun. 2020, 15, 1934578X20947213. [Google Scholar] [CrossRef]
- Nader-Macías, M.E.F.; De Gregorio, P.R.; Silva, J.A. Probiotic Lactobacilli in Formulas and Hygiene Products for the Health of the Urogenital Tract. Pharmacol. Res. Perspect. 2021, 9, e00787. [Google Scholar] [CrossRef]
- Nava, G.M.; Bielke, L.R.; Callaway, T.R.; Castañeda, M.P. Probiotic Alternatives to Reduce Gastrointestinal Infections: The Poultry Experience. Anim. Health Res. Rev. 2005, 6, 105–118. [Google Scholar] [CrossRef]
- Michail, S. The Role of Probiotics in Allergic Diseases. Allergy Asthma Clin. Immunol. 2009, 5, 5. [Google Scholar] [CrossRef]
- Makino, S.; Ikegami, S.; Kano, H.; Sashihara, T.; Sugano, H.; Horiuchi, H.; Saito, T.; Oda, M. Immunomodulatory Effects of Polysaccharides Produced by Lactobacillus Delbrueckii Ssp. Bulgaricus OLL1073R-1. J. Dairy Sci. 2006, 89, 2873–2881. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Kang, S.S.; Woo, S.J.; Park, O.J.; Ahn, K.B.; Song, K.D.; Lee, H.K.; Yun, C.H.; Han, S.H. Lipoteichoic Acid of Probiotic Lactobacillus Plantarum Attenuates Poly I:C-Induced IL-8 Production in Porcine Intestinal Epithelial Cells. Front. Microbiol. 2017, 8, 1827. [Google Scholar] [CrossRef]
- Zielińska, D.; Sionek, B.; Kołożyn-Krajewska, D. Safety of Probiotics. In Diet, Microbiome and Health; Academic Press: Cambridge, MA, USA, 2018; pp. 131–161. [Google Scholar] [CrossRef]
- Chen, M.F.; Weng, K.F.; Huang, S.Y.; Liu, Y.C.; Tseng, S.N.; Ojcius, D.M.; Shih, S.R. Pretreatment with a Heat-Killed Probiotic Modulates Monocyte Chemoattractant Protein-1 and Reduces the Pathogenicity of Influenza and Enterovirus 71 Infections. Mucosal Immunol. 2017, 10, 215–227. [Google Scholar] [CrossRef]
- Hart, A.L.; Lammers, K.; Brigidi, P.; Vitali, B.; Rizzello, F.; Gionchetti, P.; Campieri, M.; Kamm, M.A.; Knight, S.C.; Stagg, A.J. Modulation of Human Dendritic Cell Phenotype and Function by Probiotic Bacteria. Gut 2004, 53, 1602–1609. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, R.; Yamamoto, N.; Yamada, S.; Fujii, T.; Yamamoto, N.; Kanauchi, O. Induction of Anti-Viral Genes Mediated by Humoral Factors upon Stimulation with Lactococcus Lactis Strain Plasma Results in Repression of Dengue Virus Replication in Vitro. Antivir. Res. 2018, 160, 101–108. [Google Scholar] [CrossRef]
- Suzuki, H.; Tsuji, R.; Sugamata, M.; Yamamoto, N.; Yamamoto, N.; Kanauchi, O. Administration of Plasmacytoid Dendritic Cell-Stimulative Lactic Acid Bacteria Is Effective against Dengue Virus Infection in Mice. Int. J. Mol. Med. 2019, 43, 426–434. [Google Scholar] [CrossRef]
- Zhang, L.; Han, H.; Li, X.; Chen, C.; Xie, X.; Su, G.; Ye, S.; Wang, C.; He, Q.; Wang, F.; et al. Probiotics Use Is Associated with Improved Clinical Outcomes among Hospitalized Patients with COVID-19. Therap. Adv. Gastroenterol. 2021, 14, 17562848211035670. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, Y.; Nishizaki, Y.; Aida, K.; Yayama, K.; Ebisui, T.; Akiyama, T.; Nakamura, T. Lactobacillus Plantarum Induces Innate Cytokine Responses That Potentially Provide a Protective Benefit against COVID-19: A Single-Arm, Double-Blind, Prospective Trial Combined with an in Vitro Cytokine Response Assay. Exp. Ther. Med. 2022, 23, 20. [Google Scholar] [CrossRef]
- Gutiérrez-Castrellón, P.; Gandara-Martí, T.; Abreu, A.T.; Nieto-Rufino, C.D.; López-Orduña, E.; Jiménez-Escobar, I.; Jiménez-Gutiérrez, C.; López-Velazquez, G.; Espadaler-Mazo, J. Efficacy and Safety of Novel Probiotic Formulation in Adult Covid19 Outpatients: A Randomized, Placebo-Controlled Clinical Trial. medRxiv 2021. [Google Scholar] [CrossRef]
- Chaiyasut, B.; Sundaram, S.; Kesika, P.; Fujii, K.; Kubo, Y.; Noguchi, T.; Tobita, K. Effects of Bacillus Subtilis Natto Strains on Antiviral Responses in Resiquimod-Stimulated Human M1-Phenotype Macrophages. Foods 2023, 12, 313. [Google Scholar] [CrossRef]
- Rathi, A.; Jadhav, S.B.; Shah, N. A Randomized Controlled Trial of the Efficacy of Systemic Enzymes and Probiotics in the Resolution of Post-COVID Fatigue. Medicines 2021, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- What to Start: Initial Combination Regimens for the Antiretroviral-Naive Patient|NIH. Available online: https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/what-start-initial-combination-regimens (accessed on 16 March 2023).
- Jockusch, S.; Tao, C.; Li, X.; Chien, M.; Kumar, S.; Morozova, I.; Kalachikov, S.; Russo, J.J.; Ju, J. Sofosbuvir Terminated RNA Is More Resistant to SARS-CoV-2 Proofreader than RNA Terminated by Remdesivir. Sci. Rep. 2020, 10, 16577. [Google Scholar] [CrossRef] [PubMed]
- Jácome, R.; Campillo-Balderas, J.A.; Ponce de León, S.; Becerra, A.; Lazcano, A. Sofosbuvir as a Potential Alternative to Treat the SARS-CoV-2 Epidemic. Sci. Rep. 2020, 10, 9294. [Google Scholar] [CrossRef] [PubMed]
- Sacramento, C.Q.; Fintelman-Rodrigues, N.; Temerozo, J.R.; Silva, A. de P.D. Da; Dias, S. da S.G.; Silva, C. dos S. da; Ferreira, A.C.; Mattos, M.; Pão, C.R.R.; Freitas, C.S. de; et al. In Vitro Antiviral Activity of the Anti-HCV Drugs Daclatasvir and Sofosbuvir against SARS-CoV-2, the Aetiological Agent of COVID-19. J. Antimicrob. Chemother. 2021, 76, 1874–1885. [Google Scholar] [CrossRef]
- Houston, D.M.J.; Bugert, J.J.; Denyer, S.P.; Heard, C.M. Potentiated Virucidal Activity of Pomegranate Rind Extract (PRE) and Punicalagin against Herpes Simplex Virus (HSV) When Co-Administered with Zinc (II) Ions, and Antiviral Activity of PRE against HSV and Aciclovir-Resistant HSV. PLoS ONE 2017, 12, e0179291. [Google Scholar] [CrossRef]
- Haidari, M.; Ali, M.; Ward Casscells, S.; Madjid, M. Pomegranate (Punica Granatum) Purified Polyphenol Extract Inhibits Influenza Virus and Has a Synergistic Effect with Oseltamivir. Phytomedicine 2009, 16, 1127–1136. [Google Scholar] [CrossRef]
- Saadh, M.; Almaaytah, A.; Alaraj, M.; Dababneh, M.; Sa’adeh, I.; Aldalaen, S.; Kharshid, A.; Alboghdadly, A.; Hailat, M.; Khaleel, A.; et al. Punicalagin and Zinc (II) Ions Inhibit the Activity of SARS-CoV-2 3CL-Protease in Vitro. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 3908–3913. [Google Scholar] [CrossRef]
- Colunga Biancatelli, R.M.L.; Berrill, M.; Catravas, J.D.; Marik, P.E. Quercetin and Vitamin C: An Experimental, Synergistic Therapy for the Prevention and Treatment of SARS-CoV-2 Related Disease (COVID-19). Front. Immunol. 2020, 11, 1451. [Google Scholar] [CrossRef]
- Wakaskar, R.R. General Overview of Lipid–Polymer Hybrid Nanoparticles, Dendrimers, Micelles, Liposomes, Spongosomes and Cubosomes. J. Drug Target. 2018, 26, 311–318. [Google Scholar] [CrossRef]
- Günther, S.C.; Maier, J.D.; Vetter, J.; Podvalnyy, N.; Khanzhin, N.; Hennet, T.; Stertz, S. Antiviral Potential of 3′-Sialyllactose- and 6′-Sialyllactose-Conjugated Dendritic Polymers against Human and Avian Influenza Viruses. Sci. Rep. 2020, 10, 768. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-K. Liposomes for Enhanced Bioavailability of Water-Insoluble Drugs: In Vivo Evidence and Recent Approaches. Pharmaceutics 2020, 12, 264. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Wu, W.; Liu, Q.; Chen, S. Long-Circulating Nanoliposomes (LCNs) Sustained Delivery of Baicalein (BAI) with Desired Oral Bioavailability in Vivo. Drug Deliv. 2013, 20, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Refaat, H.; Mady, F.M.; Sarhan, H.A.; Rateb, H.S.; Alaaeldin, E. Optimization and Evaluation of Propolis Liposomes as a Promising Therapeutic Approach for COVID-19. Int. J. Pharm. 2021, 592, 120028. [Google Scholar] [CrossRef]
- Fu, M.; Han, X.; Chen, B.; Guo, L.; Zhong, L.; Hu, P.; Pan, Y.; Qiu, M.; Cao, P.; Chen, J. Cancer Treatment: From Traditional Chinese Herbal Medicine to the Liposome Delivery System. Acta Mater. Med. 2022, 1, 486–506. [Google Scholar] [CrossRef]
- Que, H.; Hong, W.; Lan, T.; Zeng, H.; Chen, L.; Wan, D.; Bi, Z.; Ren, W.; Luo, M.; Yang, J.; et al. Tripterin Liposome Relieves Severe Acute Respiratory Syndrome as a Potent COVID-19 Treatment. Signal Transduct. Target. Ther. 2022, 7, 399. [Google Scholar] [CrossRef]
- Boroumand, H.; Badie, F.; Mazaheri, S.; Seyedi, Z.S.; Nahand, J.S.; Nejati, M.; Baghi, H.B.; Abbasi-Kolli, M.; Badehnoosh, B.; Ghandali, M.; et al. Chitosan-Based Nanoparticles Against Viral Infections. Front. Cell. Infect. Microbiol. 2021, 11, 175. [Google Scholar] [CrossRef]
- Ju, J.; Kim, J.; Choi, Y.; Jin, S.; Kim, S.; Son, D.; Shin, M. Punicalagin-Loaded Alginate/Chitosan-Gallol Hydrogels for Efficient Wound Repair and Hemostasis. Polymers 2022, 14, 3248. [Google Scholar] [CrossRef]
- Wang, D.; Mehrabi Nasab, E.; Athari, S.S. Study Effect of Baicalein Encapsulated/Loaded Chitosan-Nanoparticle on Allergic Asthma Pathology in Mouse Model. Saudi J. Biol. Sci. 2021, 28, 4311–4317. [Google Scholar] [CrossRef]
- Saadh, M.J.; Aldalaen, S.M. Inhibitory Effects of Epigallocatechin Gallate (EGCG) Combined with Zinc Sulfate and Silver Nanoparticles on Avian Influenza A Virus Subtype H5N1. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 2630–2636. [Google Scholar] [CrossRef]
- Ciolkowski, M.; Petersen, J.F.; Ficker, M.; Janaszewska, A.; Christensen, J.B.; Klajnert, B.; Bryszewska, M. Surface Modification of PAMAM Dendrimer Improves Its Biocompatibility. Nanomedicine 2012, 8, 815–817. [Google Scholar] [CrossRef] [PubMed]
- Elbayoumi, T.A.; Torchilin, V.P. Liposomes for Targeted Delivery of Antithrombotic Drugs. Expert Opin. Drug Deliv. 2008, 5, 1185–1198. [Google Scholar] [CrossRef] [PubMed]
- Gabizon, A.; Shmeeda, H.; Barenholz, Y. Pharmacokinetics of Pegylated Liposomal Doxorubicin: Review of Animal and Human Studies. Clin. Pharm. 2003, 42, 419–436. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.D.; Kumar, P.V.; Selvam, T.; Rao, K.S. Prolonged Drug Delivery System of PEGylated PAMAM Dendrimers with a Anti-HIV Drug. Res. Pharm. 2015, 3, 8–17. [Google Scholar]
- Hama, S.; Sakai, M.; Itakura, S.; Majima, E.; Kogure, K. Rapid Modification of Antibodies on the Surface of Liposomes Composed of High-Affinity Protein A-Conjugated Phospholipid for Selective Drug Delivery. Biochem. Biophys. Rep. 2021, 27, 101067. [Google Scholar] [CrossRef]
- Low-Income, Uninsured Face Hurdles to Obtain Covid Antivirals. Available online: https://www.nbcnews.com/health/health-news/antiviral-hurdles-low-income-people-furthers-divisions-covid-rcna14565 (accessed on 26 April 2023).
- Eedara, B.B.; Alabsi, W.; Encinas-Basurto, D.; Polt, R.; Ledford, J.G.; Mansour, H.M. Inhalation Delivery for the Treatment and Prevention of COVID-19 Infection. Pharmaceutics 2021, 13, 1077. [Google Scholar] [CrossRef]
- Sungnak, W.; Huang, N.; Bécavin, C.; Berg, M.; Queen, R.; Litvinukova, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Sampaziotis, F.; et al. SARS-CoV-2 Entry Factors Are Highly Expressed in Nasal Epithelial Cells Together with Innate Immune Genes. Nat. Med. 2020, 26, 681–687. [Google Scholar] [CrossRef]
- Ahn, J.H.; Kim, J.; Hong, S.P.; Choi, S.Y.; Yang, M.J.; Ju, Y.S.; Kim, Y.T.; Kim, H.M.; Rahman, M.T.; Chung, M.K.; et al. Nasal Ciliated Cells Are Primary Targets for SARS-CoV-2 Replication in the Early Stage of COVID-19. J. Clin. Investig. 2021, 131, e148517. [Google Scholar] [CrossRef]
- Drusano, G.L. Infection Site Concentrations: Their Therapeutic Importance and the Macrolide and Macrolide-like Class of Antibiotics. Pharmacotherapy 2005, 25, 150–158. [Google Scholar] [CrossRef]
- Fazekas, T.; Eickhoff, P.; Pruckner, N.; Vollnhofer, G.; Fischmeister, G.; Diakos, C.; Rauch, M.; Verdianz, M.; Zoubek, A.; Gadner, H.; et al. Lessons Learned from a Double-Blind Randomised Placebo-Controlled Study with a Iota-Carrageenan Nasal Spray as Medical Device in Children with Acute Symptoms of Common Cold. BMC Complement. Altern. Med. 2012, 12, 147. [Google Scholar] [CrossRef]
- Fais, F.; Juskeviciene, R.; Francardo, V.; Mateos, S.; Constant, S.; Borelli, M.; Hohenfeld, I.P.; Meyer, T. Drug-Free Nasal Spray as a Barrier against SARS-CoV-2 Infection: Safety and Efficacy in Human Nasal Airway Epithelia. bioRxiv 2021. [Google Scholar] [CrossRef]
- Pelletier, J.S.; Tessema, B.; Frank, S.; Westover, J.B.; Brown, S.M.; Capriotti, J.A. Efficacy of Povidone-Iodine Nasal and Oral Antiseptic Preparations Against Severe Acute Respiratory Syndrome-Coronavirus 2 (SARS-CoV-2). Ear Nose Throat J. 2021, 100, 192S–196S. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Yin, Q.; Pei, R.; Zhang, Q.; Qu, Y.; Pan, Y.; Sun, L.; Gao, D.; Liang, C.; Yang, J.; et al. Nasal Delivery of Broadly Neutralizing Antibodies Protects Mice from Lethal Challenge with SARS-CoV-2 Delta and Omicron Variants. Virol. Sin. 2022, 37, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Go, C.C.; Pandav, K.; Sanchez-Gonzalez, M.A.; Ferrer, G. Potential Role of Xylitol Plus Grapefruit Seed Extract Nasal Spray Solution in COVID-19: Case Series. Cureus 2020, 12, e11315. [Google Scholar] [CrossRef]
- Meister, T.L.; Todt, D.; Brüggemann, Y.; Steinmann, J.; Banava, S.; Brill, F.H.H.; Pfaender, S.; Steinmann, E. Virucidal Activity of Nasal Sprays against Severe Acute Respiratory Syndrome Coronavirus-2. J. Hosp. Infect. 2022, 120, 9. [Google Scholar] [CrossRef]
- Yip, K.M.; Lee, K.M.; Ng, T.B.; Xu, S.; Yung, K.K.L.; Qu, S.; Cheung, A.K.L.; Sze, S.C.W. An Anti-Inflammatory and Anti-Fibrotic Proprietary Chinese Medicine Nasal Spray Designated as Allergic Rhinitis Nose Drops (ARND) with Potential to Prevent SARS-CoV-2 Coronavirus Infection by Targeting RBD (Delta)- Angiotensin Converting Enzyme 2 (ACE2) Binding. Chin. Med. 2022, 17, 88. [Google Scholar] [CrossRef]
- Ng, K.K.; Ng, M.K.; Zhyvotovska, A.; Singh, S.; Shevde, K. Acute Respiratory Failure Secondary to COVID-19 Viral Pneumonia Managed With Hydroxychloroquine/Azithromycin Treatment. Cureus 2020, 12, e8268. [Google Scholar] [CrossRef]
- Buehler, P.K.; Zinkernagel, A.S.; Hofmaenner, D.A.; Wendel Garcia, P.D.; Acevedo, C.T.; Gómez-Mejia, A.; Mairpady Shambat, S.; Andreoni, F.; Maibach, M.A.; Bartussek, J.; et al. Bacterial Pulmonary Superinfections Are Associated with Longer Duration of Ventilation in Critically Ill COVID-19 Patients. Cell Rep. Med. 2021, 2, 100229. [Google Scholar] [CrossRef]
- Labiris, N.R.; Dolovich, M.B. Pulmonary Drug Delivery. Part II: The Role of Inhalant Delivery Devices and Drug Formulations in Therapeutic Effectiveness of Aerosolized Medications. Br. J. Clin. Pharmacol. 2003, 56, 600. [Google Scholar] [CrossRef]
- Olivares-Morales, A.; Hatley, O.J.; Turner, D.; Galetin, A.; Aarons, L.; Rostami-Hodjegan, A. The Use of ROC Analysis for the Qualitative Prediction of Human Oral Bioavailability from Animal Data. Pharm. Res. 2014, 31, 720–730. [Google Scholar] [CrossRef]
- Price, G.; Patel, D.A. Drug Bioavailability; StatPearls Publishing: Tampa, FL, USA, 2020. [Google Scholar]
- Strong, P.; Ito, K.; Murray, J.; Rapeport, G. Current Approaches to the Discovery of Novel Inhaled Medicines. Drug Discov. Today 2018, 23, 1705–1717. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, S.; Nicolau, D.V.; Langford, B.; Mahdi, M.; Jeffers, H.; Mwasuku, C.; Krassowska, K.; Fox, R.; Binnian, I.; Glover, V.; et al. Inhaled Budesonide in the Treatment of Early COVID-19 (STOIC): A Phase 2, Open-Label, Randomised Controlled Trial. Lancet Respir. Med. 2021, 9, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Rathod, R.; Mohindra, R.; Vijayakumar, A.; Soni, R.K.; Kaur, R.; Kumar, A.; Hegde, N.; Anand, A.; Sharma, S.; Suri, V.; et al. Essential Oil Nebulization in Mild COVID-19(EONCO): Early Phase Exploratory Clinical Trial. J. Ayurveda Integr. Med. 2022, 13, 100626. [Google Scholar] [CrossRef] [PubMed]
- Amirav, I.; Newhouse, M.T. Transmission of Coronavirus by Nebulizer: A Serious, Underappreciated Risk. CMAJ 2020, 192, E346. [Google Scholar] [CrossRef]
- Cazzola, M.; Ora, J.; Bianco, A.; Rogliani, P.; Matera, M.G. Guidance on Nebulization during the Current COVID-19 Pandemic. Respir. Med. 2021, 176, 106236. [Google Scholar] [CrossRef] [PubMed]
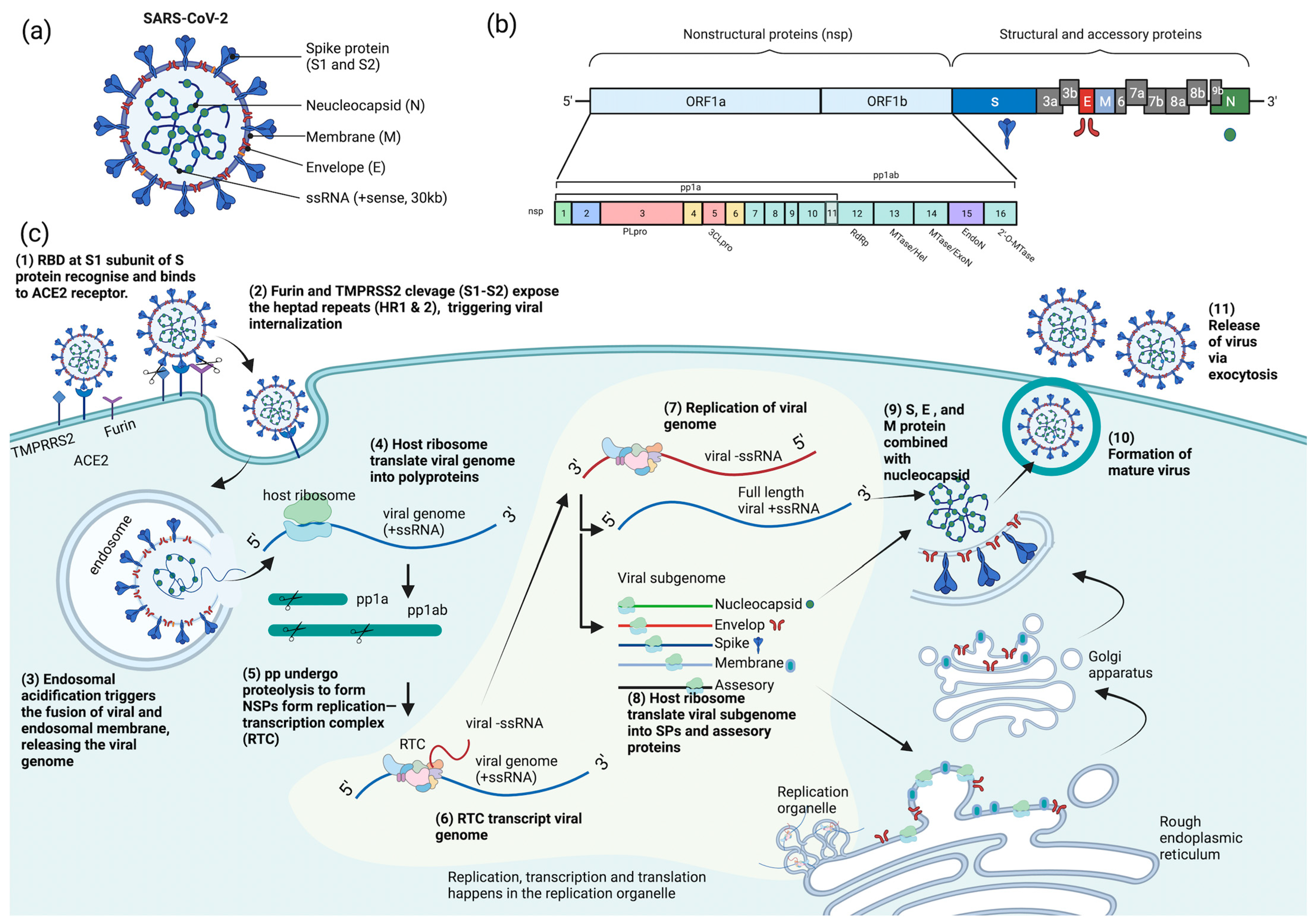

| Natural Compounds | Compound Structure | Genotype/Strain | IC50 | CC50 | Stages of Inhibition | Suggested Mechanism | RCTs | References |
|---|---|---|---|---|---|---|---|---|
| Traditional Chinese Medicine (TCM) | ||||||||
| Shuanghuanglian preparations | NA | 2019-nCoV | 0.064–0.090 µL/mL A,1 0.010 mg/mL A,2 3.65–4.44 µL/mL A,3 0.14 mg/mL A,4 0.93–1.20 μL/mL B | >12.5 μL/mL | Polypeptide processing and replication | Inhibited catalytic activity 3CLpro Inhibited polymerization of RdRp. | NA | [53,54] |
| Baicalein | 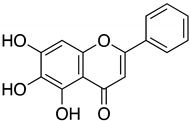 | 0.94 μM A 2.94 μM B | >200 μM | NA | ||||
| Baicalin | 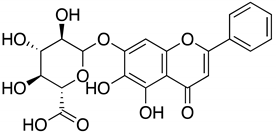 | 6.41 µM A 27.87 µM B | >200 μM | NA | ||||
| Liu Shen | NA | MT123290.1 | 0.6024 μg/mL C | 4.930 μg/mL | Post-infection | Targeted both the virus and host factor. Down-regulate NF-κB/MAPK signaling pathway and reduce the production of proinflammatory cytokines. | NA | [55] |
| Lianhuaqingwen (LHQW) | NA | Genebank accession no. MT123290.1 | 411.2 μg/mL C | 1157 μg/mL | Immune modulation Virucidal | Reduced production of proinflammatory cytokines such as TNF-α, IL-6, CCL-2/MCP-1, and CXCL-10/IP-10. Altered the morphology of extracellular virus. | NCT05366231 (Completed, Phase 4) | [56] |
| Jinhua Qinggan (JHQG) | NA | NA | NA | NA | Immune modulation | Reduced production of IL-6 and increased the production of IFN-γ | NCT04723524 (Completed, Phase 2) NCT05507489 (recruiting, unknown phase) | [57] |
| Xuebijing (XBJ) | NA | NA | NA | NA | Immune modulation | NA | NA | [58] |
| NRICM101 | NA | TCDC#4 from Taiwan CDC | 0.22 mg/mL A 0.41 mg/mL F 0.28 mg/mL G 0.42–1.18 mg/mL H | 1.77 mg/mL | Pre- and post-infection Immune modulation | Blocked the binding of S protein to ACE2 Inhibited 3CL protease Reduced production of IL-6 and increased the production of TNF-α | NCT04664049 (Unknown status, unknown phase) | [59] |
| Mentha haplocalyx extract | NA | hCoV-19/Taiwan/4/2020 | NA | NA | Prophylactic effect | NA | NA | [60] |
| Natural extracts and their active ingredients | ||||||||
| Punicalagin (PUG) from Pomegranate peel extract (PPE) | 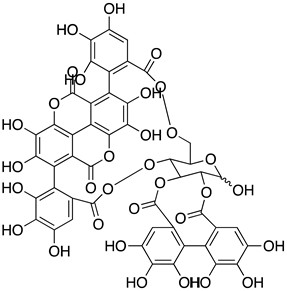 | Isolate USA-WA1/2020 | 7.20 µM D 4.62 µM A | 100 µM | Polypeptide processing | Acted as an allosteric Inhibitor and inhibited catalytic activity 3CLpro. | NA | [61] |
| Artemisia annua L. extracts | NA | USA/WA12020 | 0.01–0.14 μg/mL C | >500 µg/mL | Replication | NA | NA | [62] |
| EGYVIR | NA | hCoV-19/Egypt/NRC-03/2020 | 0.57 μg/mL D | NA | Virucidal effect | Inactivated extracellular SARS-CoV-2. Modulated the immune system by inhibiting nuclear translocation of p50 and down-regulating Ikβα, TNF-α, and IL-6, thus preventing cytokine storm. | NA | [63] |
| Netrium oleander extract and oleandrin | 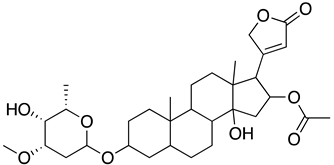 | USA-WA1/2020 strain | 7.07–11.98 ng/ml D | >1 µg/mL | Prophylactic effect | NA | NA | [64] |
| Vitis Vinifera leaf Extract | NA | clinical isolate from Lazzaro Spallanzani Hospital | 5–10 μg/mL D | >500 μg/mL | Attachment and entry | NA | NA | [65] |
| Andrographolide from Andrographis paniculata extract | 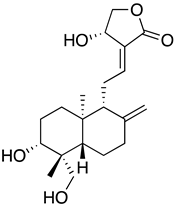 | SARS-CoV2/01/human/Jan2020/Thailand | 0.034 μM D 5–15.05 μM A | 13.19–81.52 μM | Polypeptide processing | Covalently linked to the active site of Mpro. | NCT05019326 (Recruiting, unknown phase) | [66,67,68] |
| Hydroxytyrosol-Rich Olive Pulp Extract (HIDROX) | NA | JPN/TY/WK-521 strain | NA | NA | Virucidal effect | Interacted, changed the structure and aggregated the S protein. | NA | [69] |
| APRG64, mixture of Agrimonia pilosa (AP) and Galla rhois (RG) | NA | NCCP43326 | NA | NA | Entry | Active ingredients such as ursolic acid, quercetin, and 1,2,3,4,6-penta-O-galloyl-β-d-glucose interacted with RBD of S protein. | NA | [70] |
| epigallocatechin gallate (EGCG) from green tea extract | 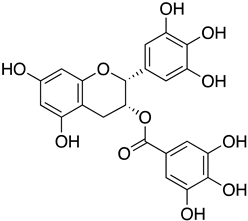 | MUC-IMB-1 | 2.47 μg/mL E | >20 μg/mL | Entry | Bound to RBD of S protein | NCT04446065 (Unknow status, Phase 2 and 3) | [71] |
| Propolis extract | NA | NA | NA | NA | Entry Replication | NA | NCT04680819 (Active unknown phase) NCT04480593 (Completed Phase 2 and 3) NCT04800224 (Completed Phase 2 and 3) NCT04916821 (Active, unknown phase) | [72,73] |
| Echinaforce | NA | BetaCoV/France/IDF0372/2020 | NA | >100 µg/ml | Virucidal effect | NA | NCT04999098 (Recruiting, Phase 4) | [74] |
| Prunella vilgaris (NhPV) extract | NA | hCoV-19/Canada/ON-VIDO-01/2020, GISAID accession# EPI_ISL_425177 | 30 μg/mL E | >200 μg/mL | Entry | Bound to ACE2 receptor and prevent viral entry | NA | [75] |
| Pure natural compounds isolated from natural origin | ||||||||
| Isorhamnetin | 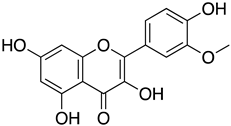 | SARS-CoV-2 pseudovirus | NA | NA | Entry and polypeptide processing | Interacted with ACE2, S protein and inhibited the catalytic activity of Mpro. | NA | [76,77,78] |
| Bufalin | 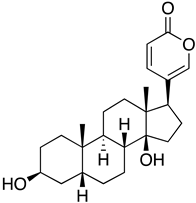 | WIV-04 | 0.018 μM C | >2000 μM | Replication | Targeted the ion exchange function of Na+/K+-ATPase | NA | [79] |
| Naringenin | 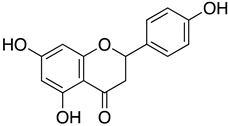 | hCoV-19/Egypt/NRC-03/2020 (Accession Number on GSAID: EPI_ISL_430820) | 28.35 µg/mL C 92 nM A | 178.75 µg/mL | Polypeptide processing | Inhibited catalytic activity Mpro. | NA | [80] |
| Resveratrol | 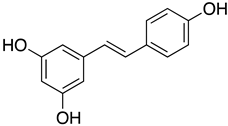 | NL/2020 (EVAg-010V-03903) | 66 µM D | NA | Post-entry | NA | NCT04666753 (Completed, unknown phase) NCT04799743 (Recruiting, unknown phase) | [81] |
| Pterostilbene | 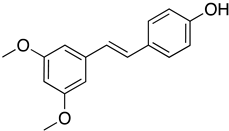 | 19 µM D | NA | |||||
| Sulfated polysaccharide (RPI-27) | NA | NA | 83 nM D | >500 µg/mL | Entry | Interacted with RBD on S protein. | NCT04777981 (Not yet recruiting, unknown phase) | [82] |
| Crude polysaccharide 375 | NA | WIV04 | 0.48 µM A 27 nM B | 136 mg/Kg on mice | Polypeptide processing | Inhibited catalytic activity Mpro. | [83] | |
| Sea cucumber sulfated polysaccharide | NA | NA | 9.10 μg/mL D | NA | Entry | Interacted with S protein. | [84] | |
| Sinapic acid | 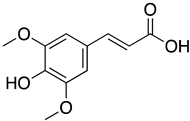 | NA | 2.69 µg/mL C | 189.3 µg/mL | Entry Assembly and release | Interacted with E protein | NA | [85] |
| X-206 | NA | NA | 14 nM C | 8.2 µM | Pre- and post-infection stage | NA | NA | [86] |
| Niclosamide | 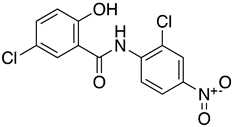 | England/IC19/2020 (IC19) | 0.34 µM D | NA | Replication | Blocked intracellular calcium release and prevented syncytia formation | NCT04399356 (Completed, phase 2) NCT05087381 (Completed, phase 4) | [87] |
| lycorine | 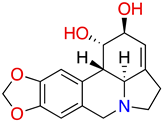 | WIV04 | 0.31 µM B | >40 μM | Post-infection | NA | NA | [88] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Low, Z.; Lani, R.; Tiong, V.; Poh, C.; AbuBakar, S.; Hassandarvish, P. COVID-19 Therapeutic Potential of Natural Products. Int. J. Mol. Sci. 2023, 24, 9589. https://doi.org/10.3390/ijms24119589
Low Z, Lani R, Tiong V, Poh C, AbuBakar S, Hassandarvish P. COVID-19 Therapeutic Potential of Natural Products. International Journal of Molecular Sciences. 2023; 24(11):9589. https://doi.org/10.3390/ijms24119589
Chicago/Turabian StyleLow, Zhaoxuan, Rafidah Lani, Vunjia Tiong, Chitlaa Poh, Sazaly AbuBakar, and Pouya Hassandarvish. 2023. "COVID-19 Therapeutic Potential of Natural Products" International Journal of Molecular Sciences 24, no. 11: 9589. https://doi.org/10.3390/ijms24119589
APA StyleLow, Z., Lani, R., Tiong, V., Poh, C., AbuBakar, S., & Hassandarvish, P. (2023). COVID-19 Therapeutic Potential of Natural Products. International Journal of Molecular Sciences, 24(11), 9589. https://doi.org/10.3390/ijms24119589







