Tetracycline Removal through the Synergy of Catalysis and Photocatalysis by Novel NaYF4:Yb,Tm@TiO2-Acetylacetone Hybrid Core-Shell Structures
Abstract
1. Introduction
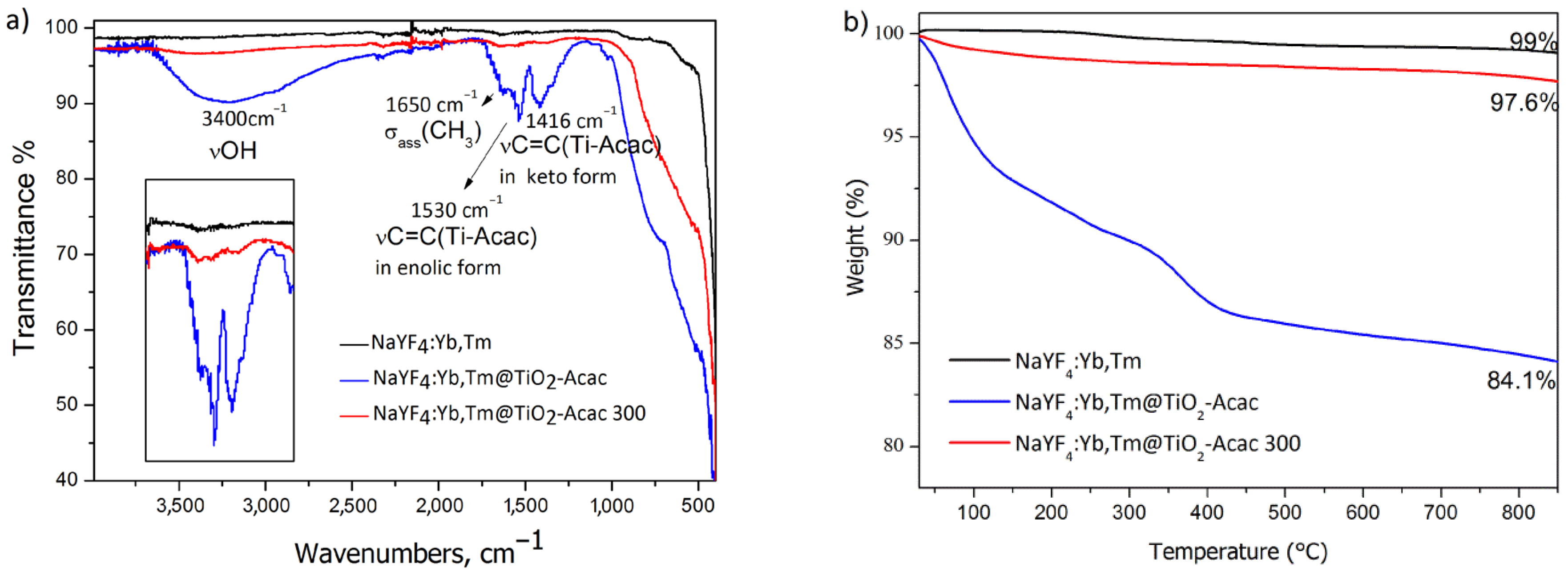
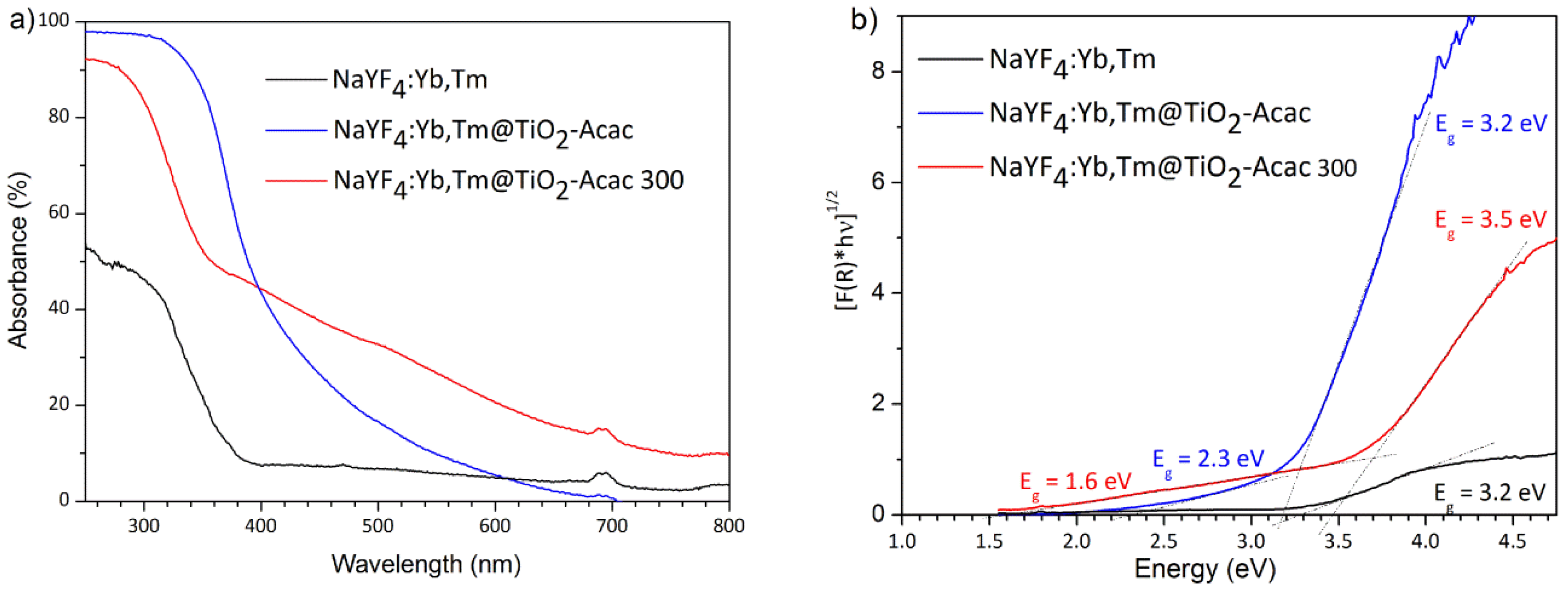
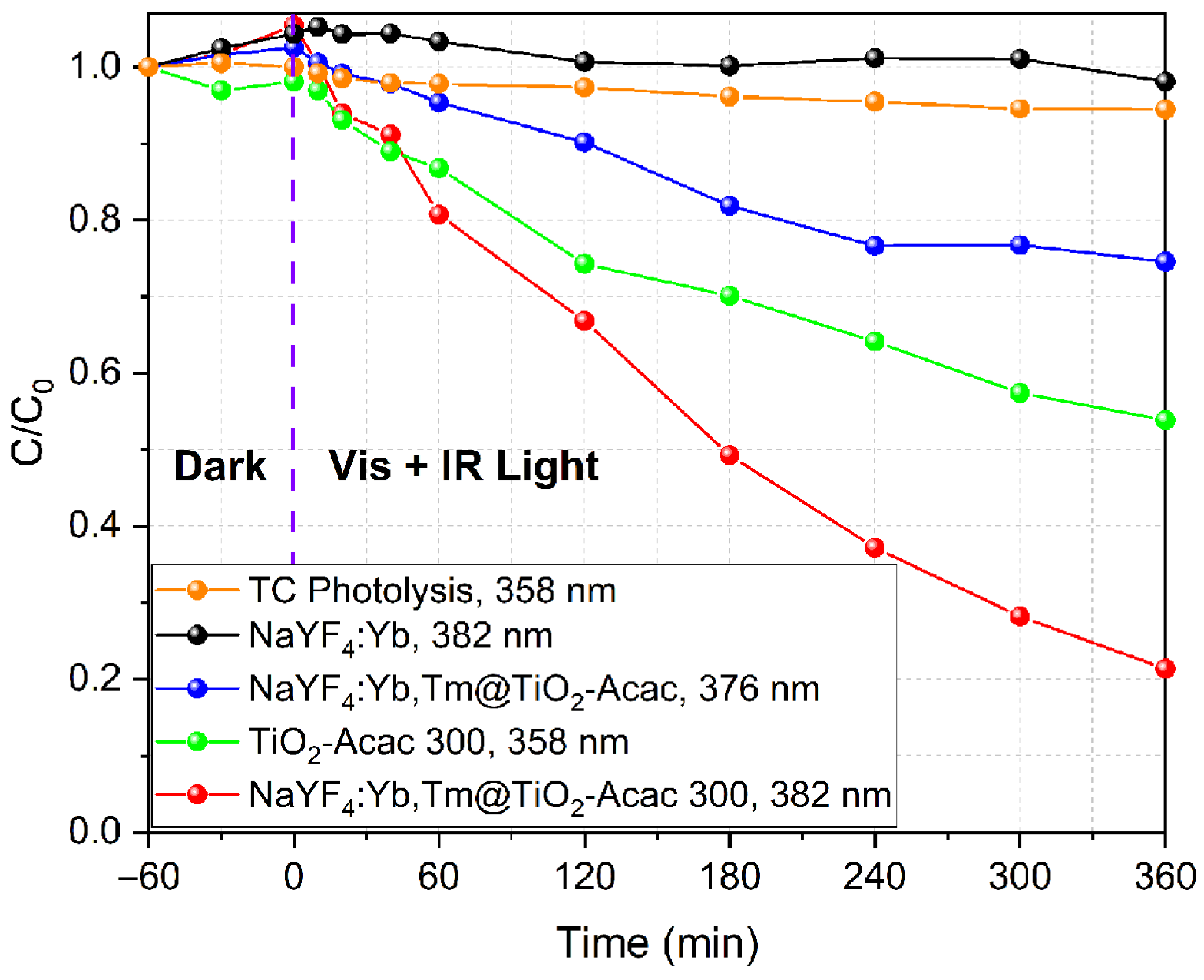
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Synthesis of β-NaYF4:Yb,Tm Particles (Core)
4.3. Formation of TiO2-Acac Shell onto β-NaYF4:Yb,Tm Particles
4.4. Characterization
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piątkowska, A.; Janus, M.; Szymański, K.; Mozia, S. C-,N- and S-Doped TiO2 Photocatalysts: A Review. Catalysts 2021, 11, 144. [Google Scholar] [CrossRef]
- Zhao, H.; Pan, F.; Li, Y. A review on the effects of TiO2 surface point defects on CO2 photoreduction with H2O. J. Materomics 2017, 3, 17–32. [Google Scholar] [CrossRef]
- Wei, L.; Yu, C.; Zhang, Q.; Liu, H.; Wang, Y. TiO2-based heterojunction photocatalysts for photocatalytic reduction of CO2 into solar fuels. J. Mater. Chem. A 2018, 6, 22411–22436. [Google Scholar] [CrossRef]
- Luciani, G.; Imparato, C.; Vitiello, G. Photosensitive Hybrid Nanostructured Materials: The Big Challenges for Sunlight Capture. Catalysts 2020, 10, 103. [Google Scholar] [CrossRef]
- Lv, Y.; Yue, L.; Li, Q.; Shao, B.; Zhao, S.; Wang, H.; Wu, S.; Wang, Z. Recyclable (Fe3O4-NaYF4:Yb,Tm)@TiO2 nanocomposites with near-infrared enhanced photocatalytic activity. Dalton Trans. 2018, 47, 1666–1673. [Google Scholar] [CrossRef]
- Liu, Z.; Hao, J.; Wang, Y.; Sun, Q.; Zhang, D.; Gan, Y. Decorating Ag3PO4 nanodots on mesoporous silica-functionalized NaYF4:Yb,Tm@NaLuF4 for efficient sunlight-driven photocatalysis: Synergy of broad spectrum absorption and pollutant adsorption-enrichment. Inorg. Chem. Front. 2019, 6, 3529–3538. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, W.; Chen, X.; Wang, J.; Zhu, Y. Photocatalytic activity enhancement of core-shell structure g-C3N4@TiO2 via controlled ultrathin g-C3N4 layer. Appl. Catal. B Environ. 2018, 220, 337–347. [Google Scholar] [CrossRef]
- Su, Y.; Liu, L.; Wen, S. Broadband NaYF4:Yb,Tm@NaYF4:Yb,Nd@TiO2 Nanoparticles Anchored on SiO2/Carbon Electrospun Fibers for Photocatalytic Degradation of Organic Pollutants. ACS Appl. Nano Mater. 2021, 4, 12576–12587. [Google Scholar] [CrossRef]
- Mazierski, P.; Roy, J.K.; Mikolajczyk, A.; Wyrzykowska, E.; Grzyb, T.; Caicedo, P.N.A.; Wei, Z.; Kowalska, E.; Zaleska-Medynska, A.; Nadolna, J. Systematic and detailed examination of NaYF4-Er-Yb-TiO2 photocatalytic activity under Vis–NIR irradiation: Experimental and theoretical analyses. Appl. Surf. Sci. 2021, 536, 147805. [Google Scholar] [CrossRef]
- Qin, W.; Zhang, D.; Zhao, D.; Wang, L.; Zheng, K. Near-infrared photocatalysis based on YF3:Yb3+,Tm3+/TiO2 core/shell nanoparticles. Chem. Commun. 2010, 46, 2304–2306. [Google Scholar] [CrossRef]
- Wu, S.; Lv, J.; Wang, F.; Duan, N.; Li, Q.; Wang, Z. Photocatalytic degradation of microcystin-LR with a nanostructured photocatalyst based on upconversion nanoparticles@TiO2 composite under simulated solar lights. Sci. Rep. 2017, 7, 14435. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Di, W.; Zhai, X.; Yang, R.; Qin, W. NIR-Responsive Photocatalytic Activity and Mechanism of NaYF4:Yb,Tm@TiO2 Core–Shell Nanoparticles. ACS Catal. 2013, 3, 405–412. [Google Scholar] [CrossRef]
- Pan, Y.; Feng, P.; Yin, M.; Fang, H.; Pan, C. Facile Synthesis of NaYF4:Yb,Tm@TiO2 Core-Shell Structured Composite with Enhanced Photocatalytic Activity for Degradation of RhB Dye. ChemistrySelect 2019, 4, 11346–11353. [Google Scholar] [CrossRef]
- Ullah, S.; Hazra, C.; Ferreira-Neto, E.P.; Silva, T.C.; Rodrigues-Filho, U.P.; Ribeiro, S.J.L. Microwave-assisted synthesis of NaYF4:Yb3+/Tm3+ upconversion particles with tailored morphology and phase for the design of UV/NIR-active NaYF4:Yb3+/Tm3+@TiO2 core@shell photocatalysts. CrystEngComm 2017, 19, 3465–3475. [Google Scholar] [CrossRef]
- Zhang, Y.; Hong, Z. Synthesis of lanthanide-doped NaYF4@TiO2 core–shell composites with highly crystalline and tunable TiO2 shells under mild conditions and their upconversion-based photocatalysis. Nanoscale 2013, 5, 8930–8933. [Google Scholar] [CrossRef]
- Ma, Y.; Li, S. NaYF4:Yb,Tm@TiO2 core@shell structures for optimal photocatalytic degradation of ciprofloxacin in the aquatic environment. RSC Adv. 2019, 9, 33519–33524. [Google Scholar] [CrossRef]
- Almeida, L.A.; Habran, M.; dos Santos Carvalho, R.; Maia da Costa, M.E.H.; Cremona, M.; Silva, B.C.; Krambrock, K.; GinoblePandoli, O.; Morgado, E., Jr.; Marinkovic, B.A. The Influence of Calcination Temperature on Photocatalytic Activity of TiO2-Acetylacetone Charge Transfer Complex towards Degradation of NOx under Visible Light. Catalysts 2020, 10, 1463. [Google Scholar] [CrossRef]
- Almeida, L.A.; Dosen, A.; Viol, J.; Marinkovic, B.A. TiO2-Acetylacetone as an Efficient Source of Superoxide Radicals under Reduced Power Visible Light: Photocatalytic Degradation of Chlorophenol and Tetracycline. Catalysts 2022, 12, 116. [Google Scholar] [CrossRef]
- Gil-Londoño, J.; Krambrock, K.; de Oliveira, R.; Cremona, M.; Maia da Costa, M.E.H.; Marinkovic, B.A. Extrinsic Point Defects in TiO2–Acetylacetone Charge-Transfer Complex and Their Effects on Optical and Photochemical Properties. Inorg. Chem. 2023, 62, 2273–2288. [Google Scholar] [CrossRef]
- Gil-Londoño, J.; Cremona, M.; Krambrock, K.; Maia da Costa, M.E.H.; Almeida, L.A.; Marinkovic, B.A. Functionalization of anatase nanoparticles with Glutaric acid and their photochemical and photocatalytic properties under visible light. Ceram. Int. 2023, 49, 17123–17134. [Google Scholar] [CrossRef]
- Li, Q.; Jiang, L.; Li, Y.; Wang, X.; Zhao, L.; Huang, P.; Chen, D.; Wang, J. Enhancement of Visible-Light Photocatalytic Degradation of Tetracycline by Co-Doped TiO2 Templated by Waste Tobacco Stem Silk. Molecules 2023, 28, 386. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ding, H.; Ji, H.; Dai, W.; Guo, J.; Du, G. Photocatalytic Degradation of Tetracycline Hydrochloride via a CdS-TiO2 Heterostructure Composite under Visible Light Irradiation. Nanomaterials 2018, 8, 415. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Mandal, M.K.; Pandey, S.; Kumar, R.; Kumar Dubey, K. Visible-Light-Driven Photocatalytic Degradation of Tetracycline Using Heterostructured Cu2O–TiO2 Nanotubes, Kinetics, and Toxicity Evaluation of Degraded Products on Cell Lines. ACS Omega. 2022, 7, 33572–33586. [Google Scholar] [CrossRef] [PubMed]
- Hunge, Y.M.; Yadav, A.A.; Kang, S.W.; Kim, H. Photocatalytic degradation of tetracycline antibiotics using hydrothermally synthesized two-dimensional molybdenum disulfide/titanium dioxide composites. J. Colloid. Interface Sci. 2022, 606, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Wang, P.; Ao, Y.; Wang, C.; Hou, J.; Qian, J. Visible light activated photocatalytic degradation of tetracycline by a magnetically separable composite photocatalyst: Graphene oxide/magnetite/cerium-doped titania. J. Colloid. Interface Sci. 2016, 467, 129–139. [Google Scholar] [CrossRef]
- Zheng, X.; Fu, W.; Kang, F.; Peng, H.; Wen, J. Enhanced photo-Fenton degradation of tetracycline using TiO2-coated α-Fe2O3 core–shell heterojunction. J. Ind. Eng. Chem. 2018, 68, 14–23. [Google Scholar] [CrossRef]
- Habran, M.; Pontón, P.I.; Mancic, L.; Pandoli, O.; Krambrock, K.; Maia da Costa, M.E.H.; Letichevsky, S.; Costa, A.M.L.M.; Morgado, E., Jr.; Marinkovic, B.A. Visible light sensitive mesoporous nanohybrids of lepidocrocite-like ferrititanate coupled to a charge transfer complex: Synthesis, characterization and photocatalytic degradation of NO. J. Photochem. Photobiol. A 2018, 365, 133–144. [Google Scholar] [CrossRef]
- Sannino, F.; Pernice, P.; Imparato, C.; Aronne, A.; D’Errico, G.; Minieri, L.; Perfettic, M.; Pirozzi, D. Hybrid TiO2–acetylacetonate amorphous gel-derived material with stably adsorbed superoxide radical active in oxidative degradation of organic pollutants. RSC Adv. 2015, 5, 93831–93839. [Google Scholar] [CrossRef]
- Dinic, I.Z.; Mancic, L.T.; Rabanal, M.E.; Yamamoto, K.; Ohara, S.; Tamura, S.; Koji, T.; Costa, A.M.L.M.; Marinkovic, B.A.; Milosevic, O.B. Compositional and structural dependence of up-converting rare earth fluorides obtained through EDTA assisted hydro/solvothermal synthesis. Adv. Powder. Technol. 2017, 28, 73–82. [Google Scholar] [CrossRef]
- Ding, M.; Chen, D.; Yin, S.; Ji, Z.; Zhong, J.; Ni, Y.; Lu, C.; Xu, Z. Simultaneous morphology manipulation and upconversion luminescence enhancement of β-NaYF4:Yb3+/Er3+ microcrystals by simply tuning the KF dosage. Sci. Rep. 2015, 5, 12745. [Google Scholar] [CrossRef]
- Lojpur, V.; Nikolić, M.; Mančić, L.; Milošević, O.; Dramićanin, M.D. Up-conversion luminescence in Ho3+ and Tm3+ co-doped Y2O3:Yb3+ fine powders obtained through aerosol decomposition. Opt. Mater. 2012, 35, 38–44. [Google Scholar] [CrossRef]
- Kolobkova, E.; Grabtchikov, A.; Khodasevich, I. Upconversion luminescence and energy transfer in the fluorophosphate glasses with low phosphate content doped with Tm3+ and Yb3+ ions. J. Non-Cryst. Solids X 2021, 11–12, 100065. [Google Scholar] [CrossRef]
- Wessels, J.M.; Ford, W.E.; Szymczak, W.; Schneider, S. The Complexation of Tetracycline and Anhydrotetracycline with Mg2+ and Ca2+: A Spectroscopic Study. J. Phys. Chem. B 1998, 102, 9323–9331. [Google Scholar] [CrossRef]
- Yu, X.; He, J.; Zhang, Y.; Hu, J.; Chen, F.; Wang, Y.; He, G.; Liu, J.; He, Q. Effective photodegradation of tetracycline by narrow-energy band gap photocatalysts La2-xSrxNiMnO6 (x = 0, 0.05, 0.10, and 0.125). J. Alloys Compd. 2019, 806, 451–463. [Google Scholar] [CrossRef]
- Jin, C.; Li, W.; Chen, Y.; Li, R.; Huo, J.; He, Q.; Wang, Y. Efficient Photocatalytic Degradation and Adsorption of Tetracycline over Type-II Heterojunctions Consisting of ZnO Nanorods and K-Doped Exfoliated g-C3N4 Nanosheets. Ind. Eng. Chem. Res. 2020, 59, 2860–2873. [Google Scholar] [CrossRef]
- Brahmi, C.; Benltifa, M.; Ghali, M.; Dumur, F.; Simonnet-Jegat, C.; Valerie, S.; Morlet-Savary, F.; Bousselmi, L.; Lalevee, J. Performance improvement of the photocatalytic process for the degradation of pharmaceutical compounds using new POM/polymer photocatalysts. J. Environ. Chem. Eng. 2021, 9, 106015. [Google Scholar] [CrossRef]
- Zhong, S.; Li, C.; Shen, M.; Lv, C.; Zhang, S. Synthesis of modified bismuth tungstate and the photocatalytic properties on tetracycline degradation and pathways. J. Mater Res. Technol. 2019, 8, 1849–1858. [Google Scholar] [CrossRef]
- Li, Z.; Xu, K.; Liang, B.; Li, Y. Determination of trace tetracyclines in surface water by aluminum hydroxide coprecipitation coupled with high-performance liquid chromatography. Anal. Methods 2013, 5, 3516–3522. [Google Scholar] [CrossRef]
- Wang, H.; Yao, H.; Sun, P.; Li, D.; Huang, C.-H. Transformation of Tetracycline Antibiotics and Fe(II) and Fe(III) Species Induced by Their Complexation. Environ. Sci. Technol. 2016, 50, 145–153. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mančić, L.; Almeida, L.A.; Machado, T.M.; Gil-Londoño, J.; Dinić, I.; Tomić, M.; Marković, S.; Jardim, P.; Marinkovic, B.A. Tetracycline Removal through the Synergy of Catalysis and Photocatalysis by Novel NaYF4:Yb,Tm@TiO2-Acetylacetone Hybrid Core-Shell Structures. Int. J. Mol. Sci. 2023, 24, 9441. https://doi.org/10.3390/ijms24119441
Mančić L, Almeida LA, Machado TM, Gil-Londoño J, Dinić I, Tomić M, Marković S, Jardim P, Marinkovic BA. Tetracycline Removal through the Synergy of Catalysis and Photocatalysis by Novel NaYF4:Yb,Tm@TiO2-Acetylacetone Hybrid Core-Shell Structures. International Journal of Molecular Sciences. 2023; 24(11):9441. https://doi.org/10.3390/ijms24119441
Chicago/Turabian StyleMančić, Lidija, Lucas A. Almeida, Tamires M. Machado, Jessica Gil-Londoño, Ivana Dinić, Miloš Tomić, Smilja Marković, Paula Jardim, and Bojan A. Marinkovic. 2023. "Tetracycline Removal through the Synergy of Catalysis and Photocatalysis by Novel NaYF4:Yb,Tm@TiO2-Acetylacetone Hybrid Core-Shell Structures" International Journal of Molecular Sciences 24, no. 11: 9441. https://doi.org/10.3390/ijms24119441
APA StyleMančić, L., Almeida, L. A., Machado, T. M., Gil-Londoño, J., Dinić, I., Tomić, M., Marković, S., Jardim, P., & Marinkovic, B. A. (2023). Tetracycline Removal through the Synergy of Catalysis and Photocatalysis by Novel NaYF4:Yb,Tm@TiO2-Acetylacetone Hybrid Core-Shell Structures. International Journal of Molecular Sciences, 24(11), 9441. https://doi.org/10.3390/ijms24119441







