A Current State of Proteomics in Adult and Pediatric Inflammatory Bowel Diseases: A Systematic Search and Review
Abstract
1. Introduction
1.1. The Value of Proteomics
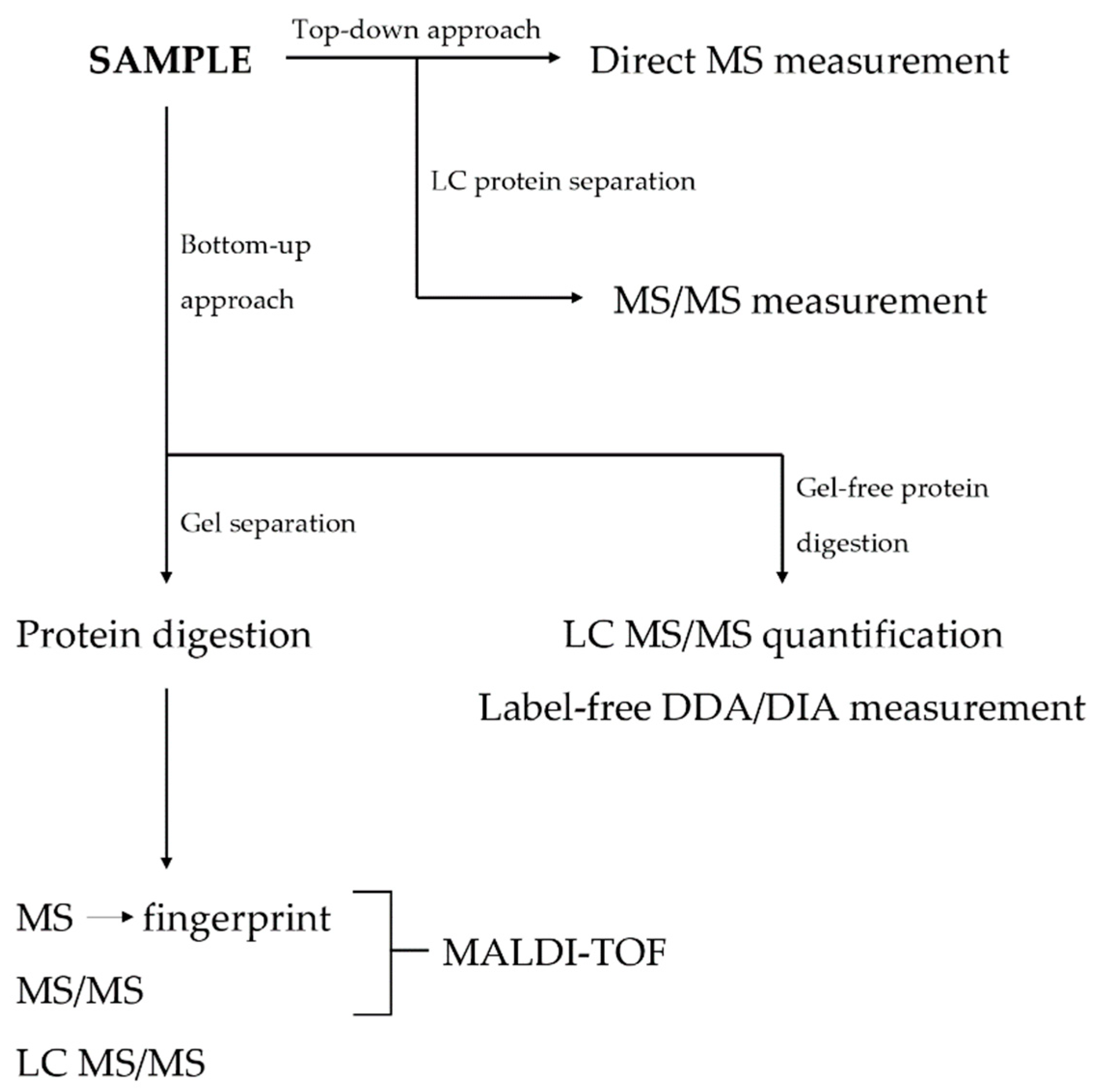
1.2. Techniques in Proteomics
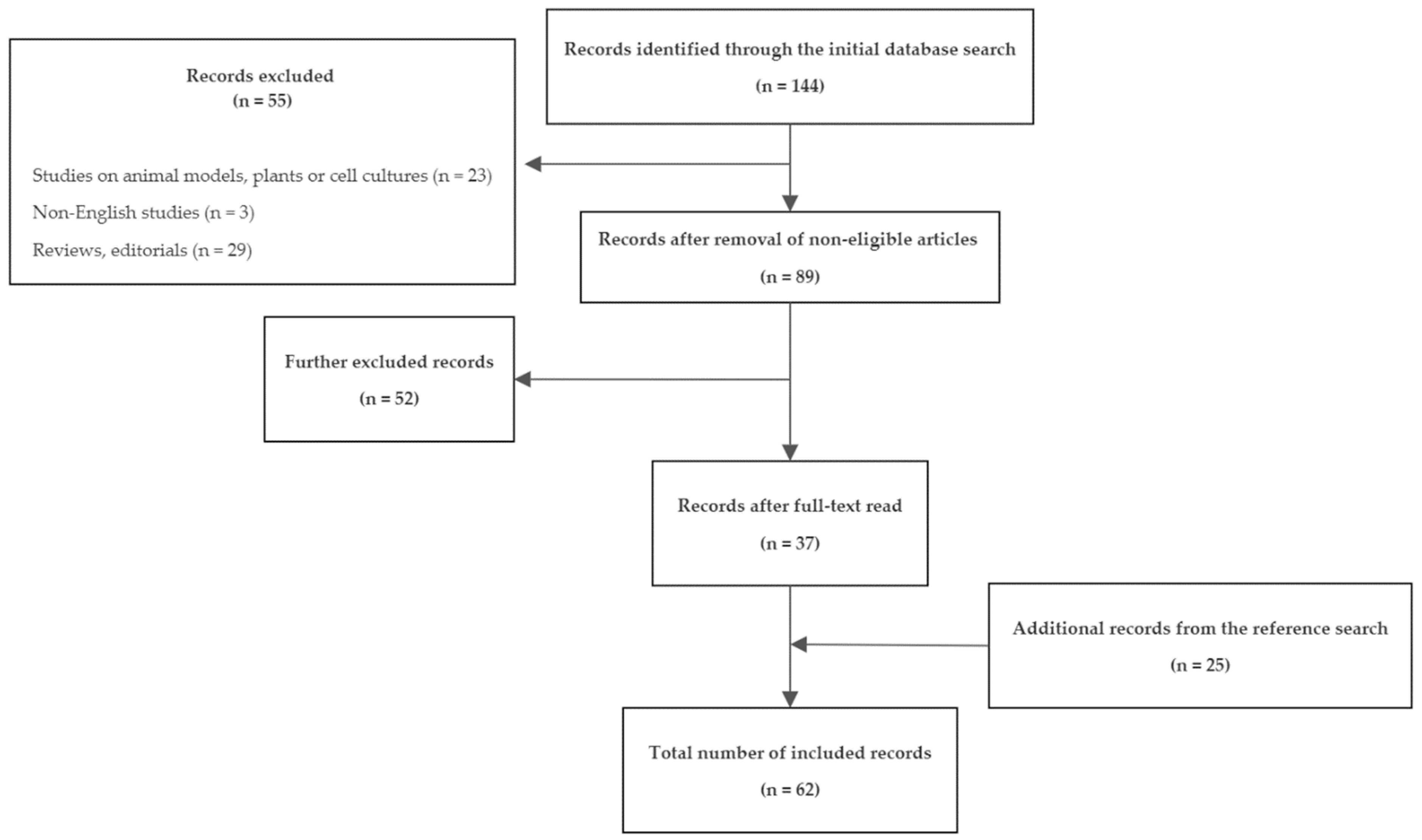
2. Methods
3. Results and Discussion
3.1. Proteomics for Distinguishing IBD Patients from Other Intestinal Diseases or Healthy Controls
3.2. Proteomics for Differentiating CD from UC
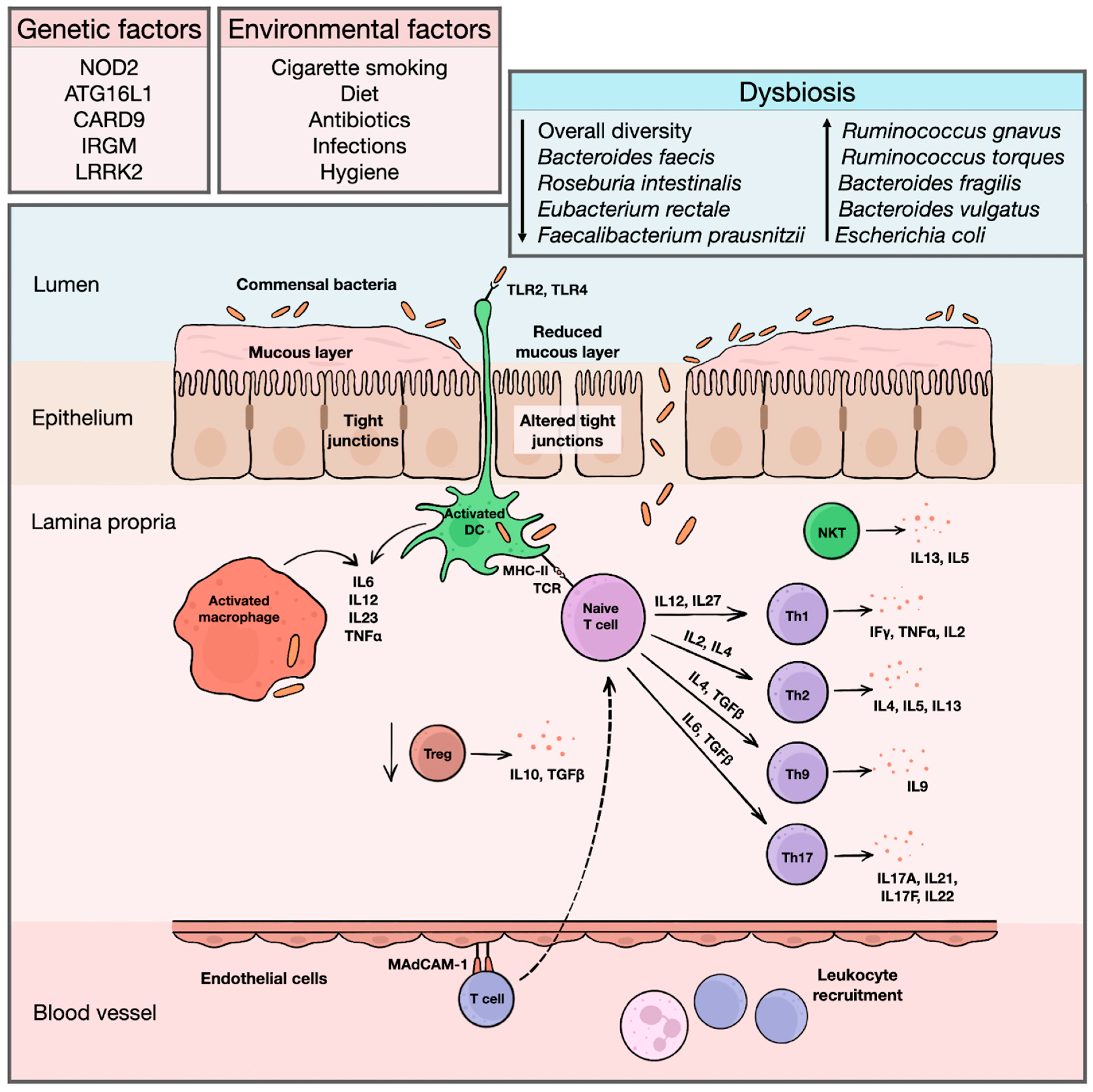
3.3. Role of Proteomics in the Understanding IBD Pathogenesis
3.4. Proteomics and Analysis of the Disease Behavior
3.5. Proteomics for Prediction of Treatment Response
3.6. Proteomics for the Selection of Biomarkers of Neoplastic Transformation
3.7. Proteomics in Pediatric IBD
3.8. Proteomics in PSC-IBD
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Magro, F.; Langner, C.; Driessen, A.; Ensari, A.; Geboes, K.; Mantzaris, G.J.; Villanacci, V.; Becheanu, G.; Borralho Nunes, P.; Cathomas, G.; et al. European consensus on the histopathology of inflammatory bowel disease. J. Crohns Colitis 2013, 7, 827–851. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.; Koletzko, S.; Turner, D.; Escher, J.C.; Cucchiara, S.; de Ridder, L.; Kolho, K.L.; Veres, G.; Russell, R.K.; Paerregaard, A.; et al. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012, 142, 46–54.e42, quiz e30. [Google Scholar] [CrossRef] [PubMed]
- Benchimol, E.I.; Fortinsky, K.J.; Gozdyra, P.; Van den Heuvel, M.; Van Limbergen, J.; Griffiths, A.M. Epidemiology of pediatric inflammatory bowel disease: A systematic review of international trends. Inflamm. Bowel Dis. 2011, 17, 423–439. [Google Scholar] [CrossRef] [PubMed]
- Burisch, J.; Pedersen, N.; Cukovic-Cavka, S.; Brinar, M.; Kaimakliotis, I.; Duricova, D.; Shonova, O.; Vind, I.; Avnstrom, S.; Thorsgaard, N.; et al. East-West gradient in the incidence of inflammatory bowel disease in Europe: The ECCO-EpiCom inception cohort. Gut 2014, 63, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Conrad, M.A.; Rosh, J.R. Pediatric Inflammatory Bowel Disease. Pediatr. Clin. North Am. 2017, 64, 577–591. [Google Scholar] [CrossRef]
- Prenzel, F.; Uhlig, H.H. Frequency of indeterminate colitis in children and adults with IBD—A metaanalysis. J. Crohns Colitis 2009, 3, 277–281. [Google Scholar] [CrossRef]
- Baumgart, D.C.; Carding, S.R. Inflammatory bowel disease: Cause and immunobiology. Lancet 2007, 369, 1627–1640. [Google Scholar] [CrossRef]
- Rifai, N.; Gillette, M.A.; Carr, S.A. Protein biomarker discovery and validation: The long and uncertain path to clinical utility. Nat. Biotechnol. 2006, 24, 971–983. [Google Scholar] [CrossRef]
- Tyers, M.; Mann, M. From genomics to proteomics. Nature 2003, 422, 193–197. [Google Scholar] [CrossRef]
- Assadsangabi, A.; Evans, C.A.; Corfe, B.M.; Lobo, A. Application of Proteomics to Inflammatory Bowel Disease Research: Current Status and Future Perspectives. Gastroenterol. Res. Pract. 2019, 2019, 1426954. [Google Scholar] [CrossRef] [PubMed]
- Baldan-Martin, M.; Chaparro, M.; Gisbert, J.P. Tissue Proteomic Approaches to Understand the Pathogenesis of Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2021, 27, 1184–1200. [Google Scholar] [CrossRef] [PubMed]
- Bennike, T.; Birkelund, S.; Stensballe, A.; Andersen, V. Biomarkers in inflammatory bowel diseases: Current status and proteomics identification strategies. World J. Gastroenterol. 2014, 20, 3231–3244. [Google Scholar] [CrossRef] [PubMed]
- Lawrance, I.C.; Klopcic, B.; Wasinger, V.C. Proteomics: An overview. Inflamm. Bowel Dis. 2005, 11, 927–936. [Google Scholar] [CrossRef]
- Liu, Y.; Beyer, A.; Aebersold, R. On the Dependency of Cellular Protein Levels on mRNA Abundance. Cell 2016, 165, 535–550. [Google Scholar] [CrossRef]
- Vaiopoulou, A.; Gazouli, M.; Theodoropoulos, G.; Zografos, G. Current advantages in the application of proteomics in inflammatory bowel disease. Dig. Dis. Sci. 2012, 57, 2755–2764. [Google Scholar] [CrossRef]
- Gisbert, J.P.; Chaparro, M. Clinical Usefulness of Proteomics in Inflammatory Bowel Disease: A Comprehensive Review. J. Crohns Colitis 2019, 13, 374–384. [Google Scholar] [CrossRef]
- Chan, P.P.; Wasinger, V.C.; Leong, R.W. Current application of proteomics in biomarker discovery for inflammatory bowel disease. World J. Gastrointest. Pathophysiol. 2016, 7, 27–37. [Google Scholar] [CrossRef]
- Aebersold, R.; Mann, M. Mass spectrometry-based proteomics. Nature 2003, 422, 198–207. [Google Scholar] [CrossRef]
- Zhang, H.; Ge, Y. Comprehensive analysis of protein modifications by top-down mass spectrometry. Circ. Cardiovasc. Genet. 2011, 4, 711. [Google Scholar] [CrossRef]
- Naryzhny, S. Inventory of proteoforms as a current challenge of proteomics: Some technical aspects. J. Proteom. 2019, 191, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Durbin, K.R.; Fornelli, L.; Fellers, R.T.; Doubleday, P.F.; Narita, M.; Kelleher, N.L. Quantitation and Identification of Thousands of Human Proteoforms below 30 kDa. J. Proteome Res. 2016, 15, 976–982. [Google Scholar] [CrossRef] [PubMed]
- Baggerman, G.; Vierstraete, E.; De Loof, A.; Schoofs, L. Gel-based versus gel-free proteomics: A review. Comb. Chem. High Throughput Screen. 2005, 8, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Westbrook, J.A.; Noirel, J.; Brown, J.E.; Wright, P.C.; Evans, C.A. Quantitation with chemical tagging reagents in biomarker studies. Proteom. Clin. Appl. 2015, 9, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Canas, B.; Pineiro, C.; Calvo, E.; Lopez-Ferrer, D.; Gallardo, J.M. Trends in sample preparation for classical and second generation proteomics. J. Chromatogr. A 2007, 1153, 235–258. [Google Scholar] [CrossRef]
- Alex, P.; Gucek, M.; Li, X. Applications of proteomics in the study of inflammatory bowel diseases: Current status and future directions with available technologies. Inflamm. Bowel Dis. 2009, 15, 616–629. [Google Scholar] [CrossRef]
- Aslam, B.; Basit, M.; Nisar, M.A.; Khurshid, M.; Rasool, M.H. Proteomics: Technologies and Their Applications. J. Chromatogr. Sci. 2017, 55, 182–196. [Google Scholar] [CrossRef]
- Coskun, O. Separation techniques: Chromatography. North Clin. Istanb. 2016, 3, 156–160. [Google Scholar]
- Ludwig, C.; Gillet, L.; Rosenberger, G.; Amon, S.; Collins, B.C.; Aebersold, R. Data-independent acquisition-based SWATH-MS for quantitative proteomics: A tutorial. Mol. Syst. Biol. 2018, 14, e8126. [Google Scholar] [CrossRef]
- Bateman, N.W.; Goulding, S.P.; Shulman, N.J.; Gadok, A.K.; Szumlinski, K.K.; MacCoss, M.J.; Wu, C.C. Maximizing peptide identification events in proteomic workflows using data-dependent acquisition (DDA). Mol. Cell. Proteom. 2014, 13, 329–338. [Google Scholar] [CrossRef]
- Ross, P.L.; Huang, Y.N.; Marchese, J.N.; Williamson, B.; Parker, K.; Hattan, S.; Khainovski, N.; Pillai, S.; Dey, S.; Daniels, S.; et al. Multiplexed protein quantitation in Saccharomyces cerevisiae using amine-reactive isobaric tagging reagents. Mol. Cell. Proteom. 2004, 3, 1154–1169. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.; Schafer, J.; Kuhn, K.; Kienle, S.; Schwarz, J.; Schmidt, G.; Neumann, T.; Johnstone, R.; Mohammed, A.K.; Hamon, C. Tandem mass tags: A novel quantification strategy for comparative analysis of complex protein mixtures by MS/MS. Anal. Chem. 2003, 75, 1895–1904. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.E.; Mann, M. Stable isotope labeling by amino acids in cell culture for quantitative proteomics. Methods Mol. Biol. 2007, 359, 37–52. [Google Scholar] [PubMed]
- Ong, S.E.; Blagoev, B.; Kratchmarova, I.; Kristensen, D.B.; Steen, H.; Pandey, A.; Mann, M. Stable isotope labeling by amino acids in cell culture, SILAC, as a simple and accurate approach to expression proteomics. Mol. Cell. Proteom. 2002, 1, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Schoeters, F.; Van Dijck, P. Protein-Protein Interactions in Candida albicans. Front. Microbiol. 2019, 10, 1792. [Google Scholar] [CrossRef]
- Han, X.; Aslanian, A.; Yates, J.R., 3rd. Mass spectrometry for proteomics. Curr. Opin. Chem. Biol. 2008, 12, 483–490. [Google Scholar] [CrossRef]
- Olshina, M.A.; Sharon, M. Mass Spectrometry: A Technique of Many Faces. Q. Rev. Biophys. 2016, 49, e18. [Google Scholar] [CrossRef]
- Nadler, W.M.; Waidelich, D.; Kerner, A.; Hanke, S.; Berg, R.; Trumpp, A.; Rosli, C. MALDI versus ESI: The Impact of the Ion Source on Peptide Identification. J. Proteome Res. 2017, 16, 1207–1215. [Google Scholar] [CrossRef]
- Selcuk, A.A. A Guide for Systematic Reviews: PRISMA. Turk. Arch. Otorhinolaryngol. 2019, 57, 57–58. [Google Scholar] [CrossRef]
- Meuwis, M.A.; Fillet, M.; Geurts, P.; de Seny, D.; Lutteri, L.; Chapelle, J.P.; Bours, V.; Wehenkel, L.; Belaiche, J.; Malaise, M.; et al. Biomarker discovery for inflammatory bowel disease, using proteomic serum profiling. Biochem. Pharmacol. 2007, 73, 1422–1433. [Google Scholar] [CrossRef]
- Meuwis, M.A.; Fillet, M.; Lutteri, L.; Maree, R.; Geurts, P.; de Seny, D.; Malaise, M.; Chapelle, J.P.; Wehenkel, L.; Belaiche, J.; et al. Proteomics for prediction and characterization of response to infliximab in Crohn’s disease: A pilot study. Clin. Biochem. 2008, 41, 960–967. [Google Scholar] [CrossRef] [PubMed]
- Shkoda, A.; Werner, T.; Daniel, H.; Gunckel, M.; Rogler, G.; Haller, D. Differential protein expression profile in the intestinal epithelium from patients with inflammatory bowel disease. J. Proteome Res. 2007, 6, 1114–1125. [Google Scholar] [CrossRef] [PubMed]
- Brentnall, T.A.; Pan, S.; Bronner, M.P.; Crispin, D.A.; Mirzaei, H.; Cooke, K.; Tamura, Y.; Nikolskaya, T.; Jebailey, L.; Goodlett, D.R.; et al. Proteins That Underlie Neoplastic Progression of Ulcerative Colitis. Proteom. Clin. Appl. 2009, 3, 1326. [Google Scholar] [CrossRef]
- Kanmura, S.; Uto, H.; Numata, M.; Hashimoto, S.; Moriuchi, A.; Fujita, H.; Oketani, M.; Ido, A.; Kodama, M.; Ohi, H.; et al. Human neutrophil peptides 1–3 are useful biomarkers in patients with active ulcerative colitis. Inflamm. Bowel Dis. 2009, 15, 909–917. [Google Scholar] [CrossRef] [PubMed]
- M’Koma, A.E.; Seeley, E.H.; Washington, M.K.; Schwartz, D.A.; Muldoon, R.L.; Herline, A.J.; Wise, P.E.; Caprioli, R.M. Proteomic profiling of mucosal and submucosal colonic tissues yields protein signatures that differentiate the inflammatory colitides. Inflamm. Bowel Dis. 2011, 17, 875–883. [Google Scholar] [CrossRef]
- May, D.; Pan, S.; Crispin, D.A.; Lai, K.; Bronner, M.P.; Hogan, J.; Hockenbery, D.M.; McIntosh, M.; Brentnall, T.A.; Chen, R. Investigating neoplastic progression of ulcerative colitis with label-free comparative proteomics. J. Proteome Res. 2011, 10, 200–209. [Google Scholar] [CrossRef]
- Zhao, X.; Kang, B.; Lu, C.; Liu, S.; Wang, H.; Yang, X.; Chen, Y.; Jiang, B.; Zhang, J.; Lu, Y.; et al. Evaluation of p38 MAPK pathway as a molecular signature in ulcerative colitis. J. Proteome Res. 2011, 10, 2216–2225. [Google Scholar] [CrossRef]
- Poulsen, N.A.; Andersen, V.; Moller, J.C.; Moller, H.S.; Jessen, F.; Purup, S.; Larsen, L.B. Comparative analysis of inflamed and non-inflamed colon biopsies reveals strong proteomic inflammation profile in patients with ulcerative colitis. BMC Gastroenterol. 2012, 12, 76. [Google Scholar] [CrossRef]
- Seeley, E.H.; Washington, M.K.; Caprioli, R.M.; M’Koma, A.E. Proteomic patterns of colonic mucosal tissues delineate Crohn’s colitis and ulcerative colitis. Proteom. Clin. Appl. 2013, 7, 541–549. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, H.; Gu, G.; Wang, G.; Wu, W.; Zhang, C.; Ren, J. Immunoproteomic to identify antigens in the intestinal mucosa of Crohn’s disease patients. PLoS ONE 2013, 8, e81662. [Google Scholar] [CrossRef]
- Han, N.Y.; Choi, W.; Park, J.M.; Kim, E.H.; Lee, H.; Hahm, K.B. Label-free quantification for discovering novel biomarkers in the diagnosis and assessment of disease activity in inflammatory bowel disease. J. Dig. Dis. 2013, 14, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Gazouli, M.; Anagnostopoulos, A.K.; Papadopoulou, A.; Vaiopoulou, A.; Papamichael, K.; Mantzaris, G.; Theodoropoulos, G.E.; Anagnou, N.P.; Tsangaris, G.T. Serum protein profile of Crohn’s disease treated with infliximab. J. Crohns Colitis 2013, 7, e461–e470. [Google Scholar] [CrossRef] [PubMed]
- Vaiopoulou, A.; Gazouli, M.; Papadopoulou, A.; Anagnostopoulos, A.K.; Karamanolis, G.; Theodoropoulos, G.E.; M’Koma, A.; Tsangaris, G.T. Serum protein profiling of adults and children with Crohn disease. J. Pediatr. Gastroenterol. Nutr. 2015, 60, 42–47. [Google Scholar] [CrossRef]
- Bennike, T.B.; Carlsen, T.G.; Ellingsen, T.; Bonderup, O.K.; Glerup, H.; Bogsted, M.; Christiansen, G.; Birkelund, S.; Stensballe, A.; Andersen, V. Neutrophil Extracellular Traps in Ulcerative Colitis: A Proteome Analysis of Intestinal Biopsies. Inflamm. Bowel Dis. 2015, 21, 2052–2067. [Google Scholar] [CrossRef] [PubMed]
- Townsend, P.; Zhang, Q.; Shapiro, J.; Webb-Robertson, B.J.; Bramer, L.; Schepmoes, A.A.; Weitz, K.K.; Mallette, M.; Moniz, H.; Bright, R.; et al. Serum Proteome Profiles in Stricturing Crohn’s Disease: A Pilot Study. Inflamm. Bowel Dis. 2015, 21, 1935–1941. [Google Scholar] [CrossRef] [PubMed]
- Corfe, B.M.; Majumdar, D.; Assadsangabi, A.; Marsh, A.M.; Cross, S.S.; Connolly, J.B.; Evans, C.A.; Lobo, A.J. Inflammation decreases keratin level in ulcerative colitis; inadequate restoration associates with increased risk of colitis-associated cancer. BMJ Open Gastroenterol. 2015, 2, e000024. [Google Scholar] [CrossRef]
- Starr, A.E.; Deeke, S.A.; Ning, Z.; Chiang, C.K.; Zhang, X.; Mottawea, W.; Singleton, R.; Benchimol, E.I.; Wen, M.; Mack, D.R.; et al. Proteomic analysis of ascending colon biopsies from a paediatric inflammatory bowel disease inception cohort identifies protein biomarkers that differentiate Crohn’s disease from UC. Gut 2017, 66, 1573–1583. [Google Scholar] [CrossRef]
- Stidham, R.W.; Wu, J.; Shi, J.; Lubman, D.M.; Higgins, P.D. Serum Glycoproteome Profiles for Distinguishing Intestinal Fibrosis from Inflammation in Crohn’s Disease. PLoS ONE 2017, 12, e0170506. [Google Scholar] [CrossRef]
- Moriggi, M.; Pastorelli, L.; Torretta, E.; Tontini, G.E.; Capitanio, D.; Bogetto, S.F.; Vecchi, M.; Gelfi, C. Contribution of Extracellular Matrix and Signal Mechanotransduction to Epithelial Cell Damage in Inflammatory Bowel Disease Patients: A Proteomic Study. Proteomics 2017, 17, 1700164. [Google Scholar] [CrossRef]
- Ning, L.; Shan, G.; Sun, Z.; Zhang, F.; Xu, C.; Lou, X.; Li, S.; Du, H.; Chen, H.; Xu, G. Quantitative Proteomic Analysis Reveals the Deregulation of Nicotinamide Adenine Dinucleotide Metabolism and CD38 in Inflammatory Bowel Disease. Biomed. Res. Int. 2019, 2019, 3950628. [Google Scholar] [CrossRef]
- Erdmann, P.; Bruckmueller, H.; Martin, P.; Busch, D.; Haenisch, S.; Muller, J.; Wiechowska-Kozlowska, A.; Partecke, L.I.; Heidecke, C.D.; Cascorbi, I.; et al. Dysregulation of Mucosal Membrane Transporters and Drug-Metabolizing Enzymes in Ulcerative Colitis. J. Pharm. Sci. 2019, 108, 1035–1046. [Google Scholar] [CrossRef]
- Schniers, A.; Goll, R.; Pasing, Y.; Sorbye, S.W.; Florholmen, J.; Hansen, T. Ulcerative colitis: Functional analysis of the in-depth proteome. Clin. Proteom. 2019, 16, 4. [Google Scholar] [CrossRef] [PubMed]
- Pierre, N.; Salee, C.; Massot, C.; Bletard, N.; Mazzucchelli, G.; Smargiasso, N.; Morsa, D.; Baiwir, D.; De Pauw, E.; Reenaers, C.; et al. Proteomics Highlights Common and Distinct Pathophysiological Processes Associated with Ileal and Colonic Ulcers in Crohn’s Disease. J. Crohns Colitis 2020, 14, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Arafah, K.; Kriegsmann, M.; Renner, M.; Lasitschka, F.; Fresnais, M.; Kriegsmann, K.; von Winterfeld, M.; Goeppert, B.; Kriegsmann, J.; Casadonte, R.; et al. Microproteomics and Immunohistochemistry Reveal Differences in Aldo-Keto Reductase Family 1 Member C3 in Tissue Specimens of Ulcerative Colitis and Crohn’s Disease. Proteom. Clin. Appl. 2020, 14, e1900110. [Google Scholar] [CrossRef] [PubMed]
- Merli, A.M.; Vieujean, S.; Massot, C.; Bletard, N.; Quesada Calvo, F.; Baiwir, D.; Mazzucchelli, G.; Servais, L.; Wera, O.; Oury, C.; et al. Solute carrier family 12 member 2 as a proteomic and histological biomarker of dysplasia and neoplasia in ulcerative colitis. J. Crohns Colitis 2020, 15, 287–298. [Google Scholar] [CrossRef]
- Pierre, N.; Baiwir, D.; Huynh-Thu, V.A.; Mazzucchelli, G.; Smargiasso, N.; De Pauw, E.; Bouhnik, Y.; Laharie, D.; Colombel, J.F.; Meuwis, M.A.; et al. Discovery of biomarker candidates associated with the risk of short-term and mid/long-term relapse after infliximab withdrawal in Crohn’s patients: A proteomics-based study. Gut 2020, 70, 1450–1457. [Google Scholar] [CrossRef]
- Liu, L.; Pu, D.; Wang, D.; Zhang, M.; Zhou, C.; Zhang, Z.; Feng, B. Proteomic Analysis of Potential Targets for Non-Response to Infliximab in Patients with Ulcerative Colitis. Front. Pharmacol. 2022, 13, 905133. [Google Scholar] [CrossRef]
- Gruver, A.M.; Westfall, M.D.; Ackermann, B.L.; Hill, S.; Morrison, R.D.; Bodo, J.; Lai, K.K.; Gemperline, D.C.; Hsi, E.D.; Liebler, D.C.; et al. Proteomic characterisations of ulcerative colitis endoscopic biopsies associate with clinically relevant histological measurements of disease severity. J. Clin. Pathol. 2022, 75, 636–642. [Google Scholar] [CrossRef]
- Vessby, J.; Wisniewski, J.R.; Lindskog, C.; Eriksson, N.; Gabrysch, K.; Zettl, K.; Wanders, A.; Carlson, M.; Rorsman, F.; Aberg, M. AGPAT1 as a Novel Colonic Biomarker for Discriminating between Ulcerative Colitis with and without Primary Sclerosing Cholangitis. Clin. Transl. Gastroenterol. 2022, 13, e00486. [Google Scholar] [CrossRef]
- Louis Sam Titus, A.S.C.; Vanarsa, K.; Soomro, S.; Patel, A.; Prince, J.; Kugathasan, S.; Mohan, C. Resistin, Elastase, and Lactoferrin as Potential Plasma Biomarkers of Pediatric Inflammatory Bowel Disease Based on Comprehensive Proteomic Screens. Mol. Cell. Proteom. 2023, 22, 100487. [Google Scholar] [CrossRef]
- Hsieh, S.Y.; Shih, T.C.; Yeh, C.Y.; Lin, C.J.; Chou, Y.Y.; Lee, Y.S. Comparative proteomic studies on the pathogenesis of human ulcerative colitis. Proteomics 2006, 6, 5322–5331. [Google Scholar] [CrossRef] [PubMed]
- Berndt, U.; Bartsch, S.; Philipsen, L.; Danese, S.; Wiedenmann, B.; Dignass, A.U.; Hammerle, M.; Sturm, A. Proteomic analysis of the inflamed intestinal mucosa reveals distinctive immune response profiles in Crohn’s disease and ulcerative colitis. J. Immunol. 2007, 179, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Nanni, P.; Parisi, D.; Roda, G.; Casale, M.; Belluzzi, A.; Roda, E.; Mayer, L.; Roda, A. Serum protein profiling in patients with inflammatory bowel diseases using selective solid-phase bulk extraction, matrix-assisted laser desorption/ionization time-of-flight mass spectrometry and chemometric data analysis. Rapid Commun. Mass Spectrom. 2007, 21, 4142–4148. [Google Scholar] [CrossRef] [PubMed]
- Fogt, F.; Jian, B.; Krieg, R.C.; Wellmann, A. Proteomic analysis of mucosal preparations from patients with ulcerative colitis. Mol. Med. Rep. 2008, 1, 51–54. [Google Scholar]
- Nanni, P.; Levander, F.; Roda, G.; Caponi, A.; James, P.; Roda, A. A label-free nano-liquid chromatography-mass spectrometry approach for quantitative serum peptidomics in Crohn’s disease patients. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2009, 877, 3127–3136. [Google Scholar] [CrossRef]
- Nanni, P.; Mezzanotte, L.; Roda, G.; Caponi, A.; Levander, F.; James, P.; Roda, A. Differential proteomic analysis of HT29 Cl.16E and intestinal epithelial cells by LC ESI/QTOF mass spectrometry. J. Proteom. 2009, 72, 865–873. [Google Scholar] [CrossRef]
- Hatsugai, M.; Kurokawa, M.S.; Kouro, T.; Nagai, K.; Arito, M.; Masuko, K.; Suematsu, N.; Okamoto, K.; Itoh, F.; Kato, T. Protein profiles of peripheral blood mononuclear cells are useful for differential diagnosis of ulcerative colitis and Crohn’s disease. J. Gastroenterol. 2010, 45, 488–500. [Google Scholar] [CrossRef]
- Li, N.; Wang, X.; Zhang, Y.; Zhai, J.; Zhang, T.; Wei, K. Comparative proteomics analysis of serum proteins in ulcerative colitis patients. Mol. Biol. Rep. 2012, 39, 5659–5667. [Google Scholar] [CrossRef]
- Erickson, A.R.; Cantarel, B.L.; Lamendella, R.; Darzi, Y.; Mongodin, E.F.; Pan, C.; Shah, M.; Halfvarson, J.; Tysk, C.; Henrissat, B.; et al. Integrated metagenomics/metaproteomics reveals human host-microbiota signatures of Crohn’s disease. PLoS ONE 2012, 7, e49138. [Google Scholar] [CrossRef]
- Presley, L.L.; Ye, J.; Li, X.; Leblanc, J.; Zhang, Z.; Ruegger, P.M.; Allard, J.; McGovern, D.; Ippoliti, A.; Roth, B.; et al. Host-microbe relationships in inflammatory bowel disease detected by bacterial and metaproteomic analysis of the mucosal-luminal interface. Inflamm. Bowel Dis. 2012, 18, 409–417. [Google Scholar] [CrossRef]
- Kwon, S.C.; Won, K.J.; Jung, S.H.; Lee, K.P.; Lee, D.Y.; Park, E.S.; Kim, B.; Cheon, G.J.; Han, K.H. Proteomic analysis of colonic mucosal tissue from tuberculous and ulcerative colitis patients. Korean J. Physiol. Pharmacol. 2012, 16, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Kohashi, M.; Nishiumi, S.; Ooi, M.; Yoshie, T.; Matsubara, A.; Suzuki, M.; Hoshi, N.; Kamikozuru, K.; Yokoyama, Y.; Fukunaga, K.; et al. A novel gas chromatography mass spectrometry-based serum diagnostic and assessment approach to ulcerative colitis. J. Crohns Colitis 2014, 8, 1010–1021. [Google Scholar] [CrossRef] [PubMed]
- Piras, C.S.A.; Grego, V.; Cassinotti, A.; Maconi, G.; Ardizzone, S.; Amoresano, A.; Bianchi Porro, G.; Bonizzi, L.; Roncada, P. Serum protein profiling of early and advanced stage Crohn’s disease. EuPA Open Proteom. 2014, 3, 48–59. [Google Scholar] [CrossRef]
- Miao, J.; Niu, J.; Wang, K.; Xiao, Y.; Du, Y.; Zhou, L.; Duan, L.; Li, S.; Yang, G.; Chen, L.; et al. Heat shock factor 2 levels are associated with the severity of ulcerative colitis. PLoS ONE 2014, 9, e88822. [Google Scholar] [CrossRef] [PubMed]
- Wasinger, V.C.; Yau, Y.; Duo, X.; Zeng, M.; Campbell, B.; Shin, S.; Luber, R.; Redmond, D.; Leong, R.W. Low Mass Blood Peptides Discriminative of Inflammatory Bowel Disease (IBD) Severity: A Quantitative Proteomic Perspective. Mol. Cell. Proteom. 2016, 15, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Xu, C.; Ning, L.; Hu, F.; Shan, G.; Chen, H.; Yang, M.; Chen, W.; Yu, J.; Xu, G. Exploration of Serum Proteomic Profiling and Diagnostic Model That Differentiate Crohn’s Disease and Intestinal Tuberculosis. PLoS ONE 2016, 11, e0167109. [Google Scholar] [CrossRef]
- Heier, C.R.; Fiorillo, A.A.; Chaisson, E.; Gordish-Dressman, H.; Hathout, Y.; Damsker, J.M.; Hoffman, E.P.; Conklin, L.S. Identification of Pathway-Specific Serum Biomarkers of Response to Glucocorticoid and Infliximab Treatment in Children with Inflammatory Bowel Disease. Clin. Transl. Gastroenterol. 2016, 7, e192. [Google Scholar] [CrossRef]
- Li, X.; LeBlanc, J.; Elashoff, D.; McHardy, I.; Tong, M.; Roth, B.; Ippoliti, A.; Barron, G.; McGovern, D.; McDonald, K.; et al. Microgeographic Proteomic Networks of the Human Colonic Mucosa and Their Association with Inflammatory Bowel Disease. Cell. Mol. Gastroenterol. Hepatol. 2016, 2, 567–583. [Google Scholar] [CrossRef]
- Di Narzo, A.F.; Telesco, S.E.; Brodmerkel, C.; Argmann, C.; Peters, L.A.; Li, K.; Kidd, B.; Dudley, J.; Cho, J.; Schadt, E.E.; et al. High-Throughput Characterization of Blood Serum Proteomics of IBD Patients with Respect to Aging and Genetic Factors. PLoS Genet. 2017, 13, e1006565. [Google Scholar] [CrossRef]
- Deeke, S.A.; Starr, A.E.; Ning, Z.; Ahmadi, S.; Zhang, X.; Mayne, J.; Chiang, C.K.; Singleton, R.; Benchimol, E.I.; Mack, D.R.; et al. Mucosal-luminal interface proteomics reveals biomarkers of pediatric inflammatory bowel disease-associated colitis. Am. J. Gastroenterol. 2018, 113, 713–724. [Google Scholar] [CrossRef]
- Denadai-Souza, A.; Bonnart, C.; Tapias, N.S.; Marcellin, M.; Gilmore, B.; Alric, L.; Bonnet, D.; Burlet-Schiltz, O.; Hollenberg, M.D.; Vergnolle, N.; et al. Functional Proteomic Profiling of Secreted Serine Proteases in Health and Inflammatory Bowel Disease. Sci. Rep. 2018, 8, 7834. [Google Scholar] [CrossRef] [PubMed]
- Di Narzo, A.F.; Brodmerkel, C.; Telesco, S.E.; Argmann, C.; Peters, L.A.; Li, K.; Kidd, B.; Dudley, J.; Cho, J.; Schadt, E.E.; et al. High-Throughput Identification of the Plasma Proteomic Signature of Inflammatory Bowel Disease. J. Crohns Colitis 2019, 13, 462–471. [Google Scholar] [CrossRef]
- Jin, L.; Li, L.; Hu, C.; Paez-Cortez, J.; Bi, Y.; Macoritto, M.; Cao, S.; Tian, Y. Integrative Analysis of Transcriptomic and Proteomic Profiling in Inflammatory Bowel Disease Colon Biopsies. Inflamm. Bowel Dis. 2019, 25, 1906–1918. [Google Scholar] [CrossRef] [PubMed]
- van der Post, S.; Jabbar, K.S.; Birchenough, G.; Arike, L.; Akhtar, N.; Sjovall, H.; Johansson, M.E.V.; Hansson, G.C. Structural weakening of the colonic mucus barrier is an early event in ulcerative colitis pathogenesis. Gut 2019, 68, 2142–2151. [Google Scholar] [CrossRef] [PubMed]
- Basso, D.; Padoan, A.; D’Inca, R.; Arrigoni, G.; Scapellato, M.L.; Contran, N.; Franchin, C.; Lorenzon, G.; Mescoli, C.; Moz, S.; et al. Peptidomic and proteomic analysis of stool for diagnosing IBD and deciphering disease pathogenesis. Clin. Chem. Lab. Med. 2020, 58, 968–979. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Chen, B.; Duan, Z.; Xia, Z.; Ding, Y.; Chen, T.; Liu, H.; Wang, B.; Yang, B.; Wang, X.; et al. Depression and anxiety in patients with active ulcerative colitis: Crosstalk of gut microbiota, metabolomics and proteomics. Gut Microbes 2021, 13, 1987779. [Google Scholar] [CrossRef]
- Sun, X.L.; Qiao, L.C.; Gong, J.; Wen, K.; Xu, Z.Z.; Yang, B.L. Proteomics identifies a novel role of fibrinogen-like protein 1 in Crohn’s disease. World J. Gastroenterol. 2021, 27, 5946–5957. [Google Scholar] [CrossRef]
- Park, J.; Jeong, D.; Chung, Y.W.; Han, S.; Kim, D.H.; Yu, J.; Cheon, J.H.; Ryu, J.H. Proteomic analysis-based discovery of a novel biomarker that differentiates intestinal Behcet’s disease from Crohn’s disease. Sci. Rep. 2021, 11, 11019. [Google Scholar] [CrossRef]
- Kalla, R.; Adams, A.T.; Bergemalm, D.; Vatn, S.; Kennedy, N.A.; Ricanek, P.; Lindstrom, J.; Ocklind, A.; Hjelm, F.; Ventham, N.T.; et al. Serum proteomic profiling at diagnosis predicts clinical course, and need for intensification of treatment in inflammatory bowel disease. J. Crohns Colitis 2021, 15, 699–708. [Google Scholar] [CrossRef]
- Bourgonje, A.R.; Wichers, S.J.; Hu, S.; van Dullemen, H.M.; Visschedijk, M.C.; Faber, K.N.; Festen, E.A.M.; Dijkstra, G.; Samsom, J.N.; Weersma, R.K.; et al. Proteomic analyses do not reveal subclinical inflammation in fatigued patients with clinically quiescent inflammatory bowel disease. Sci. Rep. 2022, 12, 14581. [Google Scholar] [CrossRef]
- Alfredsson, J.; Fabrik, I.; Gorreja, F.; Caer, C.; Sihlbom, C.; Block, M.; Borjesson, L.G.; Lindskog, E.B.; Wick, M.J. Isobaric labeling-based quantitative proteomics of FACS-purified immune cells and epithelial cells from the intestine of Crohn’s disease patients reveals proteome changes of potential importance in disease pathogenesis. Proteomics 2023, 23, e2200366. [Google Scholar] [CrossRef] [PubMed]
- Filipenko, N.R.; MacLeod, T.J.; Yoon, C.S.; Waisman, D.M. Annexin A2 is a novel RNA-binding protein. J. Biol. Chem. 2004, 279, 8723–8731. [Google Scholar] [CrossRef] [PubMed]
- Canto, C.; Menzies, K.J.; Auwerx, J. NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell. Metab. 2015, 22, 31–53. [Google Scholar] [CrossRef] [PubMed]
- Lund, F.E.; Cockayne, D.A.; Randall, T.D.; Solvason, N.; Schuber, F.; Howard, M.C. CD38: A new paradigm in lymphocyte activation and signal transduction. Immunol. Rev. 1998, 161, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.; Bonovas, S.; Doherty, G.; Kucharzik, T.; Gisbert, J.P.; Raine, T.; Adamina, M.; Armuzzi, A.; Bachmann, O.; Bager, P.; et al. ECCO Guidelines on Therapeutics in Crohn’s Disease: Medical Treatment. J. Crohns Colitis 2020, 14, 4–22. [Google Scholar] [CrossRef] [PubMed]
- Ruemmele, F.M.; Veres, G.; Kolho, K.L.; Griffiths, A.; Levine, A.; Escher, J.C.; Amil Dias, J.; Barabino, A.; Braegger, C.P.; Bronsky, J.; et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J. Crohns Colitis 2014, 8, 1179–1207. [Google Scholar] [CrossRef]
- Raine, T.; Bonovas, S.; Burisch, J.; Kucharzik, T.; Adamina, M.; Annese, V.; Bachmann, O.; Bettenworth, D.; Chaparro, M.; Czuber-Dochan, W.; et al. ECCO Guidelines on Therapeutics in Ulcerative Colitis: Medical Treatment. J. Crohns Colitis 2022, 16, 2–17. [Google Scholar] [CrossRef]
- Gouni-Berthold, I.; Baumeister, B.; Wegel, E.; Berthold, H.K.; Vetter, H.; Schmidt, C. Neutrophil-elastase in chronic inflammatory bowel disease: A marker of disease activity? Hepatogastroenterology 1999, 46, 2315–2320. [Google Scholar]
- Dabek, M.; Ferrier, L.; Roka, R.; Gecse, K.; Annahazi, A.; Moreau, J.; Escourrou, J.; Cartier, C.; Chaumaz, G.; Leveque, M.; et al. Luminal cathepsin g and protease-activated receptor 4: A duet involved in alterations of the colonic epithelial barrier in ulcerative colitis. Am. J. Pathol. 2009, 175, 207–214. [Google Scholar] [CrossRef]
- Boyapati, R.K.; Rossi, A.G.; Satsangi, J.; Ho, G.T. Gut mucosal DAMPs in IBD: From mechanisms to therapeutic implications. Mucosal. Immunol. 2016, 9, 567–582. [Google Scholar] [CrossRef]
- Mishra, S.; Ande, S.R.; Nyomba, B.L. The role of prohibitin in cell signaling. FEBS J. 2010, 277, 3937–3946. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.F.; Liang, Y.; Shi, S.L.; Liu, Q.R.; Xu, D.H.; Jing, G.J.; Wang, S.Y.; Kong, H.Y. Localization of prohibitin in the nuclear matrix and alteration of its expression during differentiation of human neuroblastoma SK-N-SH cells induced by retinoic acid. Cell. Mol. Neurobiol. 2011, 31, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Michalak, M.; Groenendyk, J.; Szabo, E.; Gold, L.I.; Opas, M. Calreticulin, a multi-process calcium-buffering chaperone of the endoplasmic reticulum. Biochem. J. 2009, 417, 651–666. [Google Scholar] [CrossRef] [PubMed]
- Bennett, T.A.; Edwards, B.S.; Sklar, L.A.; Rogelj, S. Sulfhydryl regulation of L-selectin shedding: Phenylarsine oxide promotes activation-independent L-selectin shedding from leukocytes. J. Immunol. 2000, 164, 4120–4129. [Google Scholar] [CrossRef]
- Datta, R.; deSchoolmeester, M.L.; Hedeler, C.; Paton, N.W.; Brass, A.M.; Else, K.J. Identification of novel genes in intestinal tissue that are regulated after infection with an intestinal nematode parasite. Infect. Immun. 2005, 73, 4025–4033. [Google Scholar] [CrossRef]
- Hayes, J.D.; Flanagan, J.U.; Jowsey, I.R. Glutathione transferases. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 51–88. [Google Scholar] [CrossRef]
- Silverberg, M.S.; Satsangi, J.; Ahmad, T.; Arnott, I.D.; Bernstein, C.N.; Brant, S.R.; Caprilli, R.; Colombel, J.F.; Gasche, C.; Geboes, K.; et al. Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: Report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can. J. Gastroenterol. 2005, 19 (Suppl. SA), 5A–36A. [Google Scholar] [CrossRef]
- Levine, A.; Griffiths, A.; Markowitz, J.; Wilson, D.C.; Turner, D.; Russell, R.K.; Fell, J.; Ruemmele, F.M.; Walters, T.; Sherlock, M.; et al. Pediatric modification of the Montreal classification for inflammatory bowel disease: The Paris classification. Inflamm. Bowel Dis. 2011, 17, 1314–1321. [Google Scholar] [CrossRef]
- Tan, K.; Lawler, J. The interaction of Thrombospondins with extracellular matrix proteins. J. Cell Commun. Signal. 2009, 3, 177–187. [Google Scholar] [CrossRef]
- Farina, G.; Lemaire, R.; Pancari, P.; Bayle, J.; Widom, R.L.; Lafyatis, R. Cartilage oligomeric matrix protein expression in systemic sclerosis reveals heterogeneity of dermal fibroblast responses to transforming growth factor beta. Ann. Rheum. Dis. 2009, 68, 435–441. [Google Scholar] [CrossRef]
- Kataoka, H.; Kawaguchi, M. Hepatocyte growth factor activator (HGFA): Pathophysiological functions in vivo. FEBS J. 2010, 277, 2230–2237. [Google Scholar] [CrossRef] [PubMed]
- Owen, K.A.; Qiu, D.; Alves, J.; Schumacher, A.M.; Kilpatrick, L.M.; Li, J.; Harris, J.L.; Ellis, V. Pericellular activation of hepatocyte growth factor by the transmembrane serine proteases matriptase and hepsin, but not by the membrane-associated protease uPA. Biochem. J. 2010, 426, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Cui, Q.; Fu, S.; Li, Z. Hepatocyte growth factor inhibits TGF-beta1-induced myofibroblast differentiation in tendon fibroblasts: Role of AMPK signaling pathway. J. Physiol. Sci. 2013, 63, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Gonczi, L.; Bessissow, T.; Lakatos, P.L. Disease monitoring strategies in inflammatory bowel diseases: What do we mean by “tight control”? World J. Gastroenterol. 2019, 25, 6172–6189. [Google Scholar] [CrossRef]
- Annese, V.; Daperno, M.; Rutter, M.D.; Amiot, A.; Bossuyt, P.; East, J.; Ferrante, M.; Gotz, M.; Katsanos, K.H.; Kiesslich, R.; et al. European evidence based consensus for endoscopy in inflammatory bowel disease. J. Crohns Colitis 2013, 7, 982–1018. [Google Scholar] [CrossRef]
- Travis, S.P.; Higgins, P.D.; Orchard, T.; Van Der Woude, C.J.; Panaccione, R.; Bitton, A.; O’Morain, C.; Panes, J.; Sturm, A.; Reinisch, W.; et al. Review article: Defining remission in ulcerative colitis. Aliment. Pharmacol. Ther. 2011, 34, 113–124. [Google Scholar] [CrossRef]
- Bryant, R.V.; Burger, D.C.; Delo, J.; Walsh, A.J.; Thomas, S.; von Herbay, A.; Buchel, O.C.; White, L.; Brain, O.; Keshav, S.; et al. Beyond endoscopic mucosal healing in UC: Histological remission better predicts corticosteroid use and hospitalisation over 6 years of follow-up. Gut 2016, 65, 408–414. [Google Scholar] [CrossRef]
- Christensen, B.; Hanauer, S.B.; Erlich, J.; Kassim, O.; Gibson, P.R.; Turner, J.R.; Hart, J.; Rubin, D.T. Histologic Normalization Occurs in Ulcerative Colitis and Is Associated with Improved Clinical Outcomes. Clin. Gastroenterol. Hepatol. 2017, 15, 1557–1564.e1551. [Google Scholar] [CrossRef]
- Christensen, B.; Erlich, J.; Gibson, P.R.; Turner, J.R.; Hart, J.; Rubin, D.T. Histologic Healing Is More Strongly Associated with Clinical Outcomes in Ileal Crohn’s Disease than Endoscopic Healing. Clin. Gastroenterol. Hepatol. 2020, 18, 2518–2525.e2511. [Google Scholar] [CrossRef]
- Rubin, D.T.; Huo, D.; Kinnucan, J.A.; Sedrak, M.S.; McCullom, N.E.; Bunnag, A.P.; Raun-Royer, E.P.; Cohen, R.D.; Hanauer, S.B.; Hart, J.; et al. Inflammation is an independent risk factor for colonic neoplasia in patients with ulcerative colitis: A case-control study. Clin. Gastroenterol. Hepatol. 2013, 11, 1601–1608.e4. [Google Scholar] [CrossRef]
- Gupta, R.B.; Harpaz, N.; Itzkowitz, S.; Hossain, S.; Matula, S.; Kornbluth, A.; Bodian, C.; Ullman, T. Histologic inflammation is a risk factor for progression to colorectal neoplasia in ulcerative colitis: A cohort study. Gastroenterology 2007, 133, 1099–1105, quiz 1340–1091. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Abdi, T.; Gentry, M.; Laine, L. Histological Disease Activity as a Predictor of Clinical Relapse Among Patients with Ulcerative Colitis: Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2016, 111, 1692–1701. [Google Scholar] [CrossRef] [PubMed]
- Peyrin-Biroulet, L.; Lemann, M. Review article: Remission rates achievable by current therapies for inflammatory bowel disease. Aliment. Pharmacol. Ther. 2011, 33, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Sandborn, W.J.; Khan, K.J.; Hanauer, S.B.; Talley, N.J.; Moayyedi, P. Efficacy of biological therapies in inflammatory bowel disease: Systematic review and meta-analysis. Am. J. Gastroenterol. 2011, 106, 644–659, quiz 660. [Google Scholar] [CrossRef]
- Lindsay, J.O.; Armuzzi, A.; Gisbert, J.P.; Bokemeyer, B.; Peyrin-Biroulet, L.; Nguyen, G.C.; Smyth, M.; Patel, H. Indicators of suboptimal tumor necrosis factor antagonist therapy in inflammatory bowel disease. Dig. Liver Dis. 2017, 49, 1086–1091. [Google Scholar] [CrossRef]
- Munkholm, P. Review article: The incidence and prevalence of colorectal cancer in inflammatory bowel disease. Aliment. Pharmacol. Ther. 2003, 18 (Suppl. S2), 1–5. [Google Scholar] [CrossRef]
- Ekbom, A.; Helmick, C.; Zack, M.; Adami, H.O. Ulcerative colitis and colorectal cancer. A population-based study. N. Engl. J. Med. 1990, 323, 1228–1233. [Google Scholar] [CrossRef]
- Eaden, J.A.; Abrams, K.R.; Mayberry, J.F. The risk of colorectal cancer in ulcerative colitis: A meta-analysis. Gut 2001, 48, 526–535. [Google Scholar] [CrossRef]
- Torres, J.; Pineton de Chambrun, G.; Itzkowitz, S.; Sachar, D.B.; Colombel, J.F. Review article: Colorectal neoplasia in patients with primary sclerosing cholangitis and inflammatory bowel disease. Aliment. Pharmacol. Ther. 2011, 34, 497–508. [Google Scholar] [CrossRef]
- Askling, J.; Dickman, P.W.; Karlen, P.; Brostrom, O.; Lapidus, A.; Lofberg, R.; Ekbom, A. Family history as a risk factor for colorectal cancer in inflammatory bowel disease. Gastroenterology 2001, 120, 1356–1362. [Google Scholar] [CrossRef]
- Rutter, M.; Saunders, B.; Wilkinson, K.; Rumbles, S.; Schofield, G.; Kamm, M.; Williams, C.; Price, A.; Talbot, I.; Forbes, A. Severity of inflammation is a risk factor for colorectal neoplasia in ulcerative colitis. Gastroenterology 2004, 126, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Salama, I.; Malone, P.S.; Mihaimeed, F.; Jones, J.L. A review of the S100 proteins in cancer. Eur. J. Surg. Oncol. 2008, 34, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.; Ellingson, C.; Coyne, T.; Hall, L.; Neill, M.; Christian, N.; Higham, C.; Dobrowolski, S.F.; Tuchman, M.; Summar, M.; et al. Genetic variation in the urea cycle: A model resource for investigating key candidate genes for common diseases. Hum. Mutat. 2009, 30, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Wink, D.A.; Ridnour, L.A.; Hussain, S.P.; Harris, C.C. The reemergence of nitric oxide and cancer. Nitric Oxide 2008, 19, 65–67. [Google Scholar] [CrossRef]
- Arroyo, J.P.; Kahle, K.T.; Gamba, G. The SLC12 family of electroneutral cation-coupled chloride cotransporters. Mol. Aspects Med. 2013, 34, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Van Limbergen, J.; Russell, R.K.; Drummond, H.E.; Aldhous, M.C.; Round, N.K.; Nimmo, E.R.; Smith, L.; Gillett, P.M.; McGrogan, P.; Weaver, L.T.; et al. Definition of phenotypic characteristics of childhood-onset inflammatory bowel disease. Gastroenterology 2008, 135, 1114–1122. [Google Scholar] [CrossRef]
- Levine, A.; de Bie, C.I.; Turner, D.; Cucchiara, S.; Sladek, M.; Murphy, M.S.; Escher, J.C.; The EUROKIDS Porto IBD Working Group of ESPGHAN. Atypical disease phenotypes in pediatric ulcerative colitis: 5-year analyses of the EUROKIDS Registry. Inflamm. Bowel Dis. 2013, 19, 370–377. [Google Scholar] [CrossRef]
- Fabian, O.; Hradsky, O.; Potuznikova, K.; Kalfusova, A.; Krskova, L.; Hornofova, L.; Zamecnik, J.; Bronsky, J. Low predictive value of histopathological scoring system for complications development in children with Crohn’s disease. Pathol. Res. Pract. 2017, 213, 353–358. [Google Scholar] [CrossRef]
- Fabian, O.; Hradsky, O.; Lerchova, T.; Mikus, F.; Zamecnik, J.; Bronsky, J. Limited clinical significance of tissue calprotectin levels in bowel mucosa for the prediction of complicated course of the disease in children with ulcerative colitis. Pathol. Res. Pract. 2019, 215, 152689. [Google Scholar] [CrossRef]
- Samygina, V.R.; Sokolov, A.V.; Bourenkov, G.; Petoukhov, M.V.; Pulina, M.O.; Zakharova, E.T.; Vasilyev, V.B.; Bartunik, H.; Svergun, D.I. Ceruloplasmin: Macromolecular assemblies with iron-containing acute phase proteins. PLoS ONE 2013, 8, e67145. [Google Scholar] [CrossRef]
- Zhang, H.; Kim, J.K.; Edwards, C.A.; Xu, Z.; Taichman, R.; Wang, C.Y. Clusterin inhibits apoptosis by interacting with activated Bax. Nat. Cell Biol. 2005, 7, 909–915. [Google Scholar] [CrossRef]
- Ignjatovic, V.; Lai, C.; Summerhayes, R.; Mathesius, U.; Tawfilis, S.; Perugini, M.A.; Monagle, P. Age-related differences in plasma proteins: How plasma proteins change from neonates to adults. PLoS ONE 2011, 6, e17213. [Google Scholar] [CrossRef] [PubMed]
- Bostrom, E.A.; Tarkowski, A.; Bokarewa, M. Resistin is stored in neutrophil granules being released upon challenge with inflammatory stimuli. Biochim. Biophys. Acta 2009, 1793, 1894–1900. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Liu, W.Z.; Zhao, Y.P.; Hu, Y.B.; Ge, Z.Z. Relationship between fecal lactoferrin and inflammatory bowel disease. Scand. J. Gastroenterol. 2007, 42, 1440–1444. [Google Scholar] [CrossRef] [PubMed]
- Alipour, M.; Zaidi, D.; Valcheva, R.; Jovel, J.; Martinez, I.; Sergi, C.; Walter, J.; Mason, A.L.; Wong, G.K.; Dieleman, L.A.; et al. Mucosal Barrier Depletion and Loss of Bacterial Diversity are Primary Abnormalities in Paediatric Ulcerative Colitis. J. Crohns Colitis 2016, 10, 462–471. [Google Scholar] [CrossRef]
- Schaeffer, D.F.; Win, L.L.; Hafezi-Bakhtiari, S.; Cino, M.; Hirschfield, G.M.; El-Zimaity, H. The phenotypic expression of inflammatory bowel disease in patients with primary sclerosing cholangitis differs in the distribution of colitis. Dig. Dis. Sci. 2013, 58, 2608–2614. [Google Scholar] [CrossRef]
- Sinakos, E.; Samuel, S.; Enders, F.; Loftus, E.V., Jr.; Sandborn, W.J.; Lindor, K.D. Inflammatory bowel disease in primary sclerosing cholangitis: A robust yet changing relationship. Inflamm. Bowel Dis. 2013, 19, 1004–1009. [Google Scholar] [CrossRef]
- Mertz, A.; Nguyen, N.A.; Katsanos, K.H.; Kwok, R.M. Primary sclerosing cholangitis and inflammatory bowel disease comorbidity: An update of the evidence. Ann. Gastroenterol. 2019, 32, 124–133. [Google Scholar] [CrossRef]
- Terg, R.; Sambuelli, A.; Coronel, E.; Mazzuco, J.; Cartier, M.; Negreira, S.; Munoz, A.; Gil, A.; Miguez, C.; Huernos, S.; et al. Prevalence of primary sclerosing cholangitis in patients with ulcerative colitis and the risk of developing malignancies. A large prospective study. Acta Gastroenterol. Latinoam. 2008, 38, 26–33. [Google Scholar]
- Fevery, J.; Henckaerts, L.; Van Oirbeek, R.; Vermeire, S.; Rutgeerts, P.; Nevens, F.; Van Steenbergen, W. Malignancies and mortality in 200 patients with primary sclerosering cholangitis: A long-term single-centre study. Liver Int. 2012, 32, 214–222. [Google Scholar] [CrossRef]
- Claessen, M.M.; Vleggaar, F.P.; Tytgat, K.M.; Siersema, P.D.; van Buuren, H.R. High lifetime risk of cancer in primary sclerosing cholangitis. J. Hepatol. 2009, 50, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Aguado, B.; Campbell, R.D. Characterization of a human lysophosphatidic acid acyltransferase that is encoded by a gene located in the class III region of the human major histocompatibility complex. J. Biol. Chem. 1998, 273, 4096–4105. [Google Scholar] [CrossRef]
- Titz, B.; Gadaleta, R.M.; Lo Sasso, G.; Elamin, A.; Ekroos, K.; Ivanov, N.V.; Peitsch, M.C.; Hoeng, J. Proteomics and Lipidomics in Inflammatory Bowel Disease Research: From Mechanistic Insights to Biomarker Identification. Int. J. Mol. Sci. 2018, 19, 2775. [Google Scholar] [CrossRef] [PubMed]
- Vargas, T.; Moreno-Rubio, J.; Herranz, J.; Cejas, P.; Molina, S.; Gonzalez-Vallinas, M.; Mendiola, M.; Burgos, E.; Aguayo, C.; Custodio, A.B.; et al. ColoLipidGene: Signature of lipid metabolism-related genes to predict prognosis in stage-II colon cancer patients. Oncotarget 2015, 6, 7348–7363. [Google Scholar] [CrossRef] [PubMed]
| Reference | Type of the Tissue | Cohort (n) | Proteomic Technique | Main Findings |
|---|---|---|---|---|
| Meuwis MA et al., 2007 [40] | Serum | CD (30), UC (30), inflammatory control (30), HC (30) | SELDI-TOF-MS | Identification of four proteins (PF4, MRP8, FIBA and Hpalpha2) that could serve as potential biomarkers of IBD. |
| Shkoda A et al., 2007 [42] | Isolated intestinal epithelia | CD (6), UC (6), CRC (6) | 2-DE and MALDI-TOF-MS | Identification of protein panels involved in signal transduction, stress response, and energy metabolism that are upregulated in IBD patients compared to controls. |
| Meuwis MA et al., 2008 [41] | Serum | CD (20) | SELDI-TOF-MS | Higher levels of PF4 were found in non-responders to infliximab therapy. |
| Brentnall TA et al., 2009 [43] | Intestinal samples | UC (15), HC (5) | iTRAQ and HPLC-TOF-MS/MS | Several proteins involved in UC neoplastic progression, including those related to mitochondria, oxidative activity, and calcium-binding, were identified. Two of them (CPS1 and S100P) were further confirmed via immuno-histochemistry. |
| Kanmura S et al., 2009 [44] | Serum | CD (22), UC (48), CRC (5), infectious colitis (6), HC (13) | SELDI-TOF-MS | Human neutrophil peptides 1, 2, and 3 are significantly higher in patients with active UC compared to patients with UC in remission or other diseases and decrease after successful corticosteroid therapy. The proteins may serve as biomarkers of active UC and predict treatment outcomes. |
| M’Koma AE et al., 2011 [45] | Intestinal samples | Colonic CD (24), UC (27) | Histology-directed MALDI-MS | Distinctive spectral peaks in submucosal layer were able to discriminate between colonic CD and UC. |
| May D et al., 2011 [46] | Intestinal samples | UC (15) | Label-free LTQ/Orbitrap hybrid MS coupled with nano-flow HPLC | Identification of several protein clusters differentially expressed in dysplastic and non-dysplastic mucosal regions in patients with UC-associated dysplasia or cancer. Two proteins (TRAP1 and CPS1) were detected in both dysplastic and non-dysplastic tissue via immuno-histochemistry. |
| Zhao X et al., 2011 [47] | Intestinal samples | UC (12), HC (12) | 2-DE and MALDI-TOF-MS | Increased expression of P-p38 and decrease in MAWBP and galektin-3 were found in UC patients compared to controls and correlated with disease progression. P38 MAPK pathway is suggested to be involved in patients with active UC. |
| Poulsen NA et al., 2012 [48] | Intestinal samples | UC (20), HC (4) | 2-DE and MALDI-TOF-MS | Forty-three proteins differentially expressed in inflamed colonic tissue in UC patients were identified. The proteins were mainly involved in energy metabolism (triosephosphate isomerase, glycerol-3-phosphate-dehydrogenase, alpha enolase and L-lactate dehydrogenase B-chain) and oxidative stress (superoxide dismutase, thioredoxins and selenium binding protein). |
| Seeley EH et al., 2013 [49] | Intestinal samples | Colonic CD (26), UC (26) | Histology-directed MALDI-MS | Based on 25 protein spectral peaks, a machine learning algorithm capable of differentiating between colonic CD and UC with 76.9% accuracy was constructed. |
| Zhou Z et al., 2013 [50] | Intestinal samples | CD (8) | 2-DE and MALDI-MS | Identification of six differentially expressed proteins in mucosal lesions compared to normal intestinal mucosa, including prohibitin, calreticulin, apolipoprotein A-I, intelectin-1, protein disulfide isomerase, and glutathione S transferase Pi. |
| Han NY et al., 2013 [51] | Intestinal samples | CD (3), UC (4), inflammatory polyps in UC (2), HC (3) | Label-free LC/MS | Identification of three proteins (PRG2, LCP1 and PSME1) serving as potential biomarkers of active CD. |
| Gazouli M et al., 2013 [52] | Serum | CD (18) | 2-DE and MALDI-TOF-MS | Identification of a panel of proteins (APOA1, APOE, CO4B, PLMN, TRFE, APOH and CLUS) that are upregulated in primary non-responders to infliximab therapy. |
| Vaiopoulou A et al., 2015 [53] | Serum | CD (12), pCD (12) | 2-DE and MALDI-TOF-MS | Ceruloplasmin and apolipoprotein B-100 are increased in children with CD, while clusterin is overexpressed in adult CD patients. |
| Bennike TB et al., 2015 [54] | Intestinal samples | UC (10), HC (10) | Label-free LC/MS | Identification of 46 proteins differentially expressed in UC compared to controls. The proteins were often associated with neutrophils and neutrophil extracellular traps formation, suggesting involvement of the innate immunity. |
| Townsend P et al., 2015 [55] | Serum | Stricturing CD (9), non-stricturing CD (9), UC (9) | LC-MS | Identification of the peptide/protein subset discriminating between the three cohorts with 70% accuracy for peptides and up to 80% for proteins. Several proteins distinguishing the stricturing CD were involved in complement activation, fibrinolysis, and lymphocyte adhesion. |
| Corfe BM et al., 2015 [56] | Intestinal samples | UC (45), PSC-UC (7), HC (10) | iTRAQ and HPLC-ESI/Q-TOF-MS/MS | Downregulation of keratins 8, 18, and 19 and vimentin in patients with acute distal UC compared to controls and samples from non-inflamed proximal mucosa. Upregulation of those proteins in patients with quiescent longstanding pancolitis in contrast to controls and patients with recent-onset remission. Decreased levels of several intermediate filament proteins in patients with PSC-UC and UC with dysplasia. |
| Starr AE et al., 2017 [57] | Intestinal samples | Discovery cohort: pCD (15), pUC (15), pHC (20) Validation cohort: pCD (15), pUC (15), pHC (19) | SILAC and HPLC-ESI-MS/MS | Two protein panels were identified that discriminate IBD from HC (FABP5, NAMPT, UGHD, LRPPRC and PPA1) and CD from UC (HADHB, SEC61A1, SND1, LAP3, LTA4H, MT2A, SLC25A1, HNRNP H3, TF, ECH1, TFRC and B2M), respectively. Application of the two panels to validation cohort differentiated 95,9% IBD patients from HC and 80% CD from UC. One hundred and sixteen proteins correlated with the severity of the disease. Visfatin and metallothionein-2 were subsequently confirmed via ELISA on an independent cohort. |
| Stidham WR et al., 2017 [58] | Serum | CD (40) | LC-MS | Identification of five glycoproteins showing ≥20% abundance change in ≥80% of the patients with inflammatory and fibrostenotic phenotype of CD. Among the glycoproteins, COMP and HGFA were elevated in the fibrostenotic group. |
| Moriggi M et al., 2017 [59] | Intestinal samples and isolated intestinal epithelia | CD (30), UC (30), controls (16) | 2-DE and MALDI-TOF-MS | Identification of proteins increased or decreased in inflamed and non-inflamed IBD patients, reflecting different patterns of extracellular matrix and cytoskeleton rearrangement and changes in cellular metabolism and autophagy. |
| Ning L et al., 2019 [60] | Intestinal samples | Discovery cohort: CD (9), UC (9), HC (6) Validation cohort: CD (3), UC (3), HC (3) | LC-MS/MS | Identification of protein spectra differentially expressed in CD, UC, and controls. Several novel proteins, including CD38, were introduced. CD38 expression was higher in IBD patients compared to controls, and higher in CD patients in comparison to UC. It was also more abundant in inflamed regions of the bowel. |
| Erdmann P et al., 2019 [61] | Intestinal samples | UC (10), HC (10) | LC-MS/MS | Identification of several genes for metabolizing enzymes (i.e., CYP2C9 or UGT1A1) and transporters (ABCB1, ABCG2 or MCT1) decreased during the inflammation. On the other hand, MRP4, OATP2B1, or ORCTL2 were significantly elevated in inflamed tissue. On the protein level, these results could be confirmed only for MCT1. |
| Schniers A et al., 2019 [62] | Intestinal samples | UC (17), HC (15) | LC-MS/MS | Downregulation of several proteins, including metallothioneins, PPAR-inducible proteins, fibrillar collagens, and proteins involved in bile acid transport and metabolic functions of nutrients, energy, steroids, xenobiotics, and carbonate, in patients with UC. Proteins involved in immune response and protein processing in endoplasmic reticulum were upregulated. |
| Pierre N et al., 2020 [63] | Intestinal samples | CD (16) | Label-free UPLC-ESI-MS/MS | Different proteins were expressed in ulcer edges from patients with ileal and colonic CD. The proteins were mainly associated with epithelial-mesenchymal transition, neutrophil degranulation, and ribosomes. |
| Arafah K et al., 2020 [64] | Intestinal samples | Discovery cohort: CD (9), UC (9); Validation cohort: CD (62), UC (51) | LC-MS/MS | Identification of several proteins upregulated in CD patients, including proteins related to neutrophilic activity and damage-associated molecular patterns. Aldo-keto reductase family 1 member C3 protein was found in 8/9 CD patients and no UC patients. |
| Merli AM et al., 2020 [65] | Intestinal samples | Discovery cohort: UC (5); Validation cohort: UC (74), sporadic lesions (174), HC (18) | Label-free LC-MS/MS | Proteomic analysis of dysplastic, inflammatory, and normal mucosal regions in UC patients. Eleven proteins were found to be more abundant in dysplastic foci, including solute carrier family 12 member 2, which was subsequently confirmedvia immuno-histochemistry. |
| Pierre N et al., 2020 [66] | Serum | CD (102) | UPLC-MS | Identification of several circulating biomarkers associated with risk of short-term and mid/long-term relapse after infliximab withdrawal. |
| Liu L et al., 2022 [67] | Intestinal samples | UC (12) | Label-free LC-MS | Identification of five proteins (ACTBL2, MBL2, BIP, EIF3D, and CR1) as potential predictive biomarkers of non-response to infliximab therapy. |
| Gruver AM et al., 2022 [68] | Intestinal samples | UC (19) | MS | Neutrophil-related proteins correlated with histological scoring indices of disease severity. A negative correlation was found between disease severity and cell junction proteins and β-catenin. |
| Vessby J et al., 2022 [69] | Intestinal samples | Discovery cohort: PSC-UC (9), UC (7), HC (7) Validation cohort: (PSC-UC (16), UC (21) | LC-MS/MS | 1-acetylglycerol-3-phosphate O-acyltransferase 1 was proved to be higher in PSC-UC patients compared to UC. The finding was confirmed via immuno-histochemistry. |
| Louis Sam Titus ASC et al., 2022 [70] | Serum | Discovery cohort: pUC (10), pCD (10), pHC (7) Validation cohort: pCD (30), pUC (30), pHC (16) | Aptamer-based targeted proteomic assay | Significant elevation of serum resistin, elastase, and lactoferrin in both pCD and pUC patients. The proteins (especially resistin) may serve as serum biomarkers of pIBD. |
| Candidate Biomarkers | Potential Utility |
|---|---|
| Bone marrow proteoglycan, calprotectin, carbonyl reductase, CD38, chitinase 3-like 1, collagen type VI alpha 1 chain, detyrosinated α-tubulin, FIBA, fibrinogen, focal adhesion kinase, GDP-forming subunit and periostin, hpalpha2, intelectin 1, keratin 19, L-lactate dehydrogenase, L-plastin, MRP8, myeloperoxidase, neutrophil defensin-1, olfactomedin 4, PF4, proteasome activator subunit 1, RHO-associated protein kinase 1, Rho-GDP, spectrin repeat containing nuclear envelope 2, succinate CoA ligase 2, tau tubulin kinase 2, tenascin C, vimentin, and vinculin. | Distinguishing IBD from other diseases |
| Aldo-keto reductase family 1 member C3 protein, cathepsin G, Ig mu chain C region protein group, Ig mu heavy chain disease protein, keratin 4, lactotransferrin, lysozyme C, neutrophil elastase, and proteins S100 A8 and A9. | Differentiating CD from UC |
| Apolipoprotein A-I, calreticulin, disulfide isomerase, glutathione S transferase Pi, heat-shock 70 kDa protein 5, hHeat-shock protein 90 kDa beta member 1, intelectin-1, and prohibitin. | Pathogenesis of CD |
| α-enolase, fibrillar collagens, galectin, glycerol-3-phosphate-dehydrogenase, L-lactate dehydrogenase B-chain, MAWD binding protein, metallothioneins, monocarboxylate transporter 1, P-p38, PPAR-inducible proteins, selenium binding protein, superoxide dismutase, thioredoxins, and triosephosphate isomerase. Spectrum of proteins associated with neutrophil extracellular traps and involved in bile acid transport and metabolic functions of nutrients, energy, steroids, xenobiotics, and carbonate. | Pathogenesis of UC |
| Cartilage oligomeric matrix protein and hepatocyte growth factor activator. | Stricturing phenotype of CD |
| β-catenin, cell junction proteins, human neutrophil peptides 1, 2, and 3, and neutrophil-associated proteins. | Assessment of the inflammatory activity in UC |
| α-1B-glycoprotein, apolipoprotein A-I, apolipoprotein E, β-actin-like protein 2, β-2-glycoprotein 1, bactericidal permeability-increasing protein, clusterin, complement C1r subcomponent, complement C4-B, complement receptor type 1, eukaryotic translation initiation factor 3 subunit D, leucine-rich alpha-2-glycoprotein, mannose binding protein C, plasminogen, protein platelet aggregation factor 4, serotransferrin, and vitamin D-binding protein. | Response to infliximab therapy |
| Carbamoyl-phosphate synthase 1, S100 calcium-binding protein P, solute carrier family 12 member 2, TNF receptor-associated protein 1, and isoform 1. | Neoplastic transformation in UC |
| Apolipoprotein B-100, ceruloplasmin, clusterin, elastase, lactoferrin, metallothionein-2, resistin, and visfatin. | Distinguishing pediatric IBD from adult IBD and other diseases |
| 1-acetylglycerol-3-phosphate O-acyltransferase 1, keratin 8, 18, and 19, and vimentin. | Pathogenesis of PSC-IBD (keratin 8 also involved in neoplastic transformation) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabian, O.; Bajer, L.; Drastich, P.; Harant, K.; Sticova, E.; Daskova, N.; Modos, I.; Tichanek, F.; Cahova, M. A Current State of Proteomics in Adult and Pediatric Inflammatory Bowel Diseases: A Systematic Search and Review. Int. J. Mol. Sci. 2023, 24, 9386. https://doi.org/10.3390/ijms24119386
Fabian O, Bajer L, Drastich P, Harant K, Sticova E, Daskova N, Modos I, Tichanek F, Cahova M. A Current State of Proteomics in Adult and Pediatric Inflammatory Bowel Diseases: A Systematic Search and Review. International Journal of Molecular Sciences. 2023; 24(11):9386. https://doi.org/10.3390/ijms24119386
Chicago/Turabian StyleFabian, Ondrej, Lukas Bajer, Pavel Drastich, Karel Harant, Eva Sticova, Nikola Daskova, Istvan Modos, Filip Tichanek, and Monika Cahova. 2023. "A Current State of Proteomics in Adult and Pediatric Inflammatory Bowel Diseases: A Systematic Search and Review" International Journal of Molecular Sciences 24, no. 11: 9386. https://doi.org/10.3390/ijms24119386
APA StyleFabian, O., Bajer, L., Drastich, P., Harant, K., Sticova, E., Daskova, N., Modos, I., Tichanek, F., & Cahova, M. (2023). A Current State of Proteomics in Adult and Pediatric Inflammatory Bowel Diseases: A Systematic Search and Review. International Journal of Molecular Sciences, 24(11), 9386. https://doi.org/10.3390/ijms24119386






