Flavones: The Apoptosis in Prostate Cancer of Three Flavones Selected as Therapeutic Candidate Models
Abstract
1. Introduction
1.1. Cancer Causes and Features
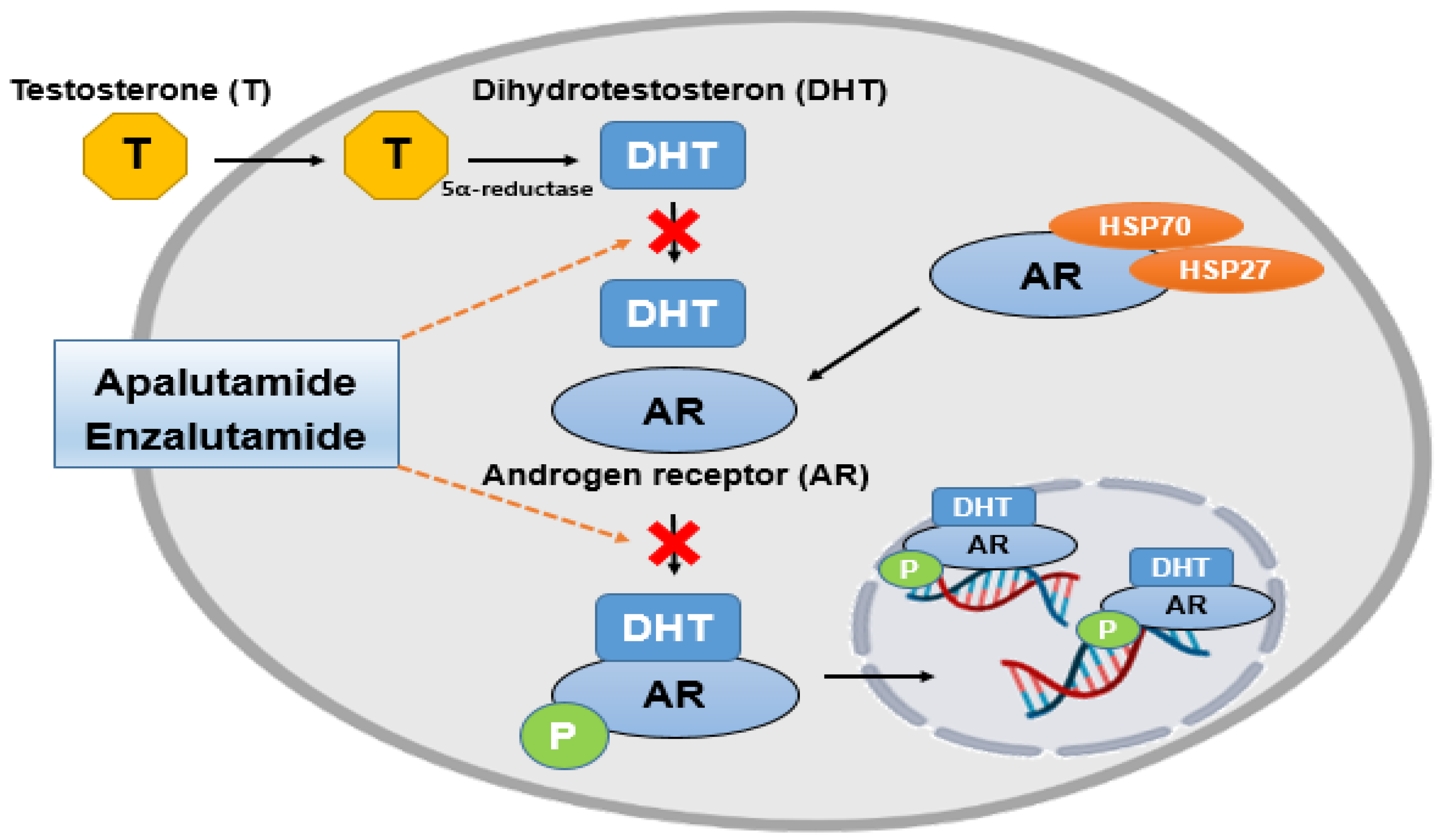
1.2. The Relationship between Causes of Prostate Cancer and Androgen Receptors
1.3. Flavonoids
1.4. Functions of Flavones and Their Effect on Prostate Cancer
2. Prostate Cancer
2.1. Prostate Cancer Epidemiology and Mortality in Humans
2.2. Classification of Prostate Cancer
2.3. Prostate Cancer Causes and Treatment
2.4. Side Effects of the Mentioned Chemical Drugs (Apalutamide and Enzalutamide)
3. The Mechanism of Apoptosis in Prostate Cancer
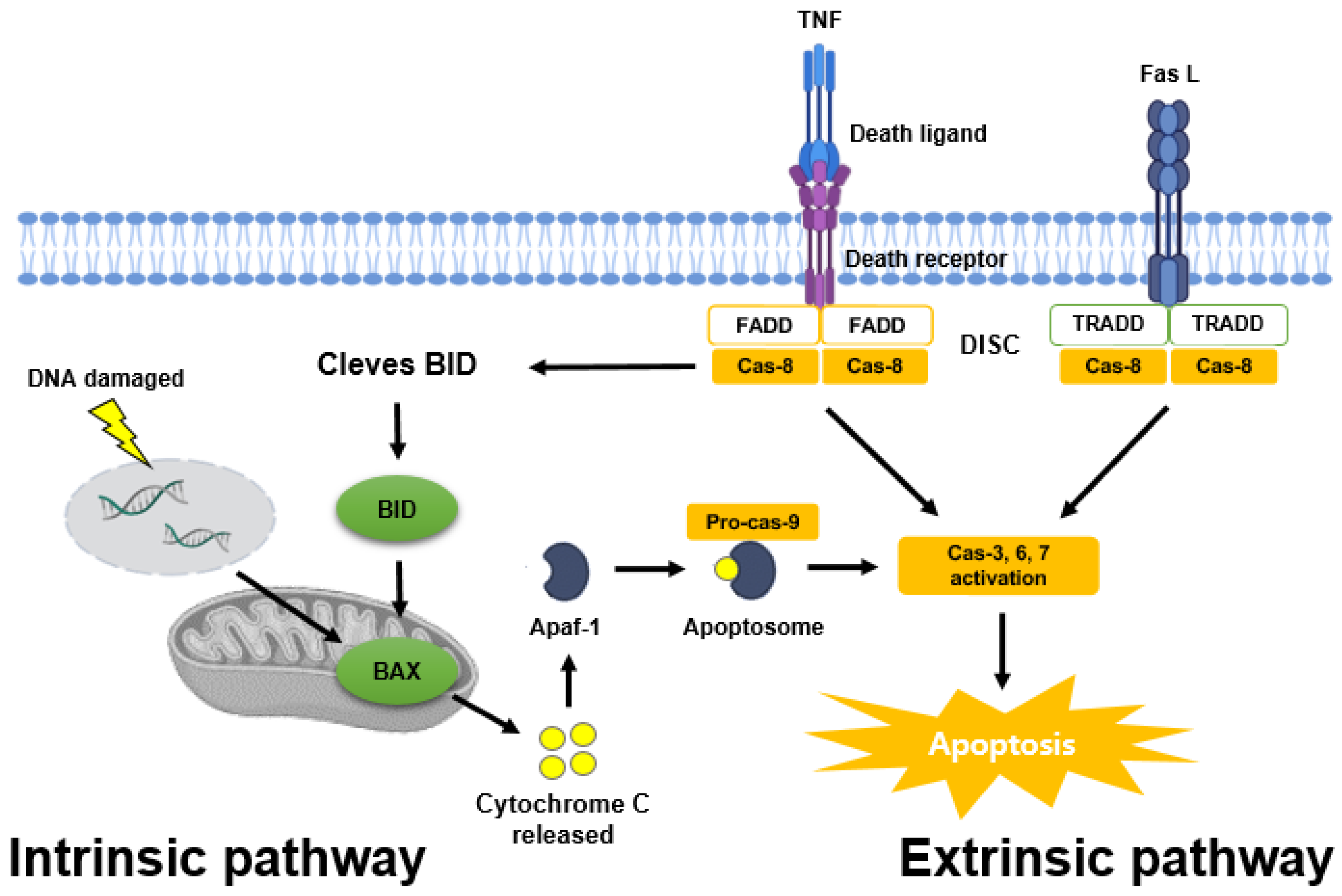
3.1. Apoptosis Pathways
3.2. Apoptosis in Prostate Cancer
3.3. The Genetic Mutations of Apoptosis in Prostate Cancer
4. Anticancer Effects of Three Selected Flavones in Prostate Cancer
4.1. Flavones as Promising Biochemical Agents for Anti-Prostate Cancer
4.2. Apigenin
4.3. Acacetin
4.4. Tangeretin
| Apigenin | |||
|---|---|---|---|
| Prostate Cancer Cell Line | Treatment Concentration | Anticancer Regulation Mechanism | Reference |
| PC-3 | 1, 2.5, 5, 10, 20, 25 μM |
| [32] |
| DU145 | 1, 5, 10, 20 μM |
| [98] |
| 22Rv1 | 2.5, 5, 10, 20 μM |
| [99] |
| LNCaP | 1, 5, 10, 20 μM |
| [98] |
| CSC | 25 μM |
| [100] |
| Mouse Model | Dosages | In Vivo Function | |
| 8-week-old male TRAMP mice | 20 and 50 μg/mouse/day (wt/vol)/gavage for 20 weeks |
| [101] |
| Transplanted mice PC-3 and 22Rv1 cells | 20 and 50 μg/mouse/day (wt/vol)/gavage for 20 weeks |
| [99] |
| Acacetin | |||
|---|---|---|---|
| Prostate Cancer Cell Line | Treatment Concentration | Anticancer Regulation Mechanism | Reference |
| DU145 | 5, 12.5, 20, 30, 50, 100 μM |
| [29,30] |
| LNCaP | 25, 50, 100 μM |
| [104] |
| Mouse Model | Dosages | In Vivo Function | |
| DU145 xenografted BALB/c mouse model | 50 (mg/kg)/5 days/intraperitoneally injected/week for 30 days |
| [30] |
| Tangeretin | |||
|---|---|---|---|
| Prostate Cancer Cell Line | Treatment Concentration | Anticancer Regulation Mechanism | Reference |
| PC-3 | 20, 50, 75, 100 μM |
| [107,108] |
| LNCaP | 20, 50, 75, 100 μM |
| [107,108] |
| DU145 | 25, 50, 100 μM |
| [109] |
| Mouse Model | Dosages | In Vivo Function | |
| PC3 xenografted immunodeficient mice | 25 μL and 50 μL/intraperitoneally injected/five times a week for 23 days |
| [110] |
| Oral administration of 50 μL and 100 μL |
| ||
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Wu, S.; Zhu, W.; Thompson, P.; Hannun, Y.A. Evaluating intrinsic and non-intrinsic cancer risk factors. Nat. Commun. 2018, 9, 3490. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Fernald, K.; Kurokawa, M. Evading apoptosis in cancer. Trends Cell Biol. 2013, 12, 620–633. [Google Scholar] [CrossRef]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Cancer statistics for the year 2020: An overview. Int. J. Cancer 2021, 149, 778–789. [Google Scholar] [CrossRef]
- Stangelberger, A.; Waldert, M.; Djavan, B. Prostate cancer in elderly men. Rev. Urol. 2008, 10, 111–119. [Google Scholar]
- Gathirua-Mwangi, W.G.; Zhang, J. Dietary factors and risk for advanced prostate cancer. Eur. J. Cancer Prev. 2014, 23, 96–109. [Google Scholar] [CrossRef]
- Heinlein, C.A.; Chang, C. Androgen receptor in prostate cancer. Endocr. Rev. 2004, 25, 276–308. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.N.; Baruah, M.M.; Sharma, N. Structure Based docking studies towards exploring potential anti-androgen activity of selected phytochemicals against Prostate Cancer. Sci. Rep. 2017, 7, 1955. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.Z.; Zhao, X.K. Prostate cancer: Current treatment and prevention strategies. Iran Red. Crescent. Med. J. 2013, 15, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Karantanos, T.; Corn, P.G.; Thompson, T.C. Prostate cancer progression after androgen deprivation therapy: Mechanisms of castrate resistance and novel therapeutic approaches. Oncogene 2013, 32, 5501–5511. [Google Scholar] [CrossRef] [PubMed]
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Busselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2018, 11, 28. [Google Scholar] [CrossRef]
- Khan, A.U.; Dagur, H.S.; Khan, M.; Malik, N.; Alam, M.; Mushtaque, M. Therapeutic role of flavonoids and flavones in cancer prevention: Current trends and future perspectives. Eur. J. Med. Chem. Rep. 2021, 3, 100010. [Google Scholar] [CrossRef]
- Xu, R.; Zhang, Y.; Ye, X.; Xue, S.; Shi, J.; Pan, J.; Chen, Q. Inhibition effects and induction of apoptosis of flavonoids on the prostate cancer cell line PC-3 in vitro. Food Chem. 2013, 138, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Raffa, D.; Maggio, B.; Raimondi, M.V.; Plescia, F.; Daidone, G. Recent discoveries of anticancer flavonoids. Eur. J. Med. Chem. 2017, 142, 213–228. [Google Scholar] [CrossRef]
- Kopustinskiene, D.M.; Jakstas, V.; Savickas, A.; Bernatoniene, J. Flavonoids as Anticancer Agents. Nutrients 2020, 12, 457. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Liskova, A.; Samec, M.; Koklesova, L.; Brockmueller, A.; Zhai, K.; Abdellatif, B.; Siddiqui, M.; Biringer, K.; Kudela, E.; Pec, M.; et al. Flavonoids as an effective sensitizer for anti-cancer therapy: Insights into multi-faceted mechanisms and applicability towards individualized patient profiles. EPMA J. 2021, 12, 155–176. [Google Scholar] [CrossRef]
- Gürler, S.B.; Kiraz, Y.; Baran, Y. Chapter 21—Flavonoids in cancer therapy: Current and future trends. In Biodiversity and Biomedicine; Ozturk, M., Egamberdieva, D., Pešić, M., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 403–440. [Google Scholar]
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food Sources, Bioavailability, Metabolism, and Bioactivity. Adv. Nutr. 2017, 8, 423–435. [Google Scholar] [CrossRef]
- Ollila, F.; Halling, K.; Vuorela, P.; Vuorela, H.; Slotte, J.P. Characterization of flavonoid—Biomembrane interactions. Arch. Biochem. Biophys. 2002, 399, 103–108. [Google Scholar] [CrossRef]
- Verma, A.K.; Pratap, R. The biological potential of flavones. Nat. Prod. Rep. 2010, 27, 1571–1593. [Google Scholar] [CrossRef]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Flavonoids as antioxidants: Determination of radical-scavenging efficiencies. Methods Enzymol. 1990, 186, 343–355. [Google Scholar] [PubMed]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.L.; Huang, Y.; Chen, Z.Y. Isolation and elucidation of antioxidant constituents from acetone extract in root of Scutellaria rehderiana. Zhongguo Zhong Yao Za Zhi 2004, 29, 863–864. [Google Scholar] [PubMed]
- Ninfali, P.; Antonelli, A.; Magnani, M.; Scarpa, E.S. Antiviral Properties of Flavonoids and Delivery Strategies. Nutrients 2020, 12, 2534. [Google Scholar] [CrossRef]
- Cushnie, T.P.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Hieble, J.P.; Kolpak, D.C.; McCafferty, G.P.; Ruffolo, R.R., Jr.; Testa, R.; Leonardi, A. Effects of alpha1-adrenoceptor antagonists on agonist and tilt-induced changes in blood pressure: Relationships to uroselectivity. Eur. J. Pharmacol. 1999, 373, 51–62. [Google Scholar] [CrossRef]
- Testa, R.; Guarneri, L.; Taddei, C.; Poggesi, E.; Angelico, P.; Sartani, A.; Leonardi, A.; Gofrit, O.N.; Meretyk, S.; Caine, M. Functional antagonistic activity of Rec 15/2739, a novel alpha-1 antagonist selective for the lower urinary tract, on noradrenaline-induced contraction of human prostate and mesenteric artery. J. Pharmacol. Exp. Ther. 1996, 277, 1237–1246. [Google Scholar]
- Kim, H.R.; Park, C.G.; Jung, J.Y. Acacetin (5,7-dihydroxy-4′-methoxyflavone) exhibits in vitro and in vivo anticancer activity through the suppression of NF-kappaB/Akt signaling in prostate cancer cells. Int. J. Mol. Med. 2014, 33, 317–324. [Google Scholar] [CrossRef]
- Yun, S.; Lee, Y.J.; Choi, J.; Kim, N.D.; Han, D.C.; Kwon, B.M. Acacetin Inhibits the Growth of STAT3-Activated DU145 Prostate Cancer Cells by Directly Binding to Signal Transducer and Activator of Transcription 3 (STAT3). Molecules 2021, 26, 6204. [Google Scholar] [CrossRef] [PubMed]
- Raza, W.; Luqman, S.; Meena, A. Prospects of tangeretin as a modulator of cancer targets/pathways. Pharmacol. Res. 2020, 161, 105202. [Google Scholar] [CrossRef]
- Shukla, S.; Fu, P.; Gupta, S. Apigenin induces apoptosis by targeting inhibitor of apoptosis proteins and Ku70-Bax interaction in prostate cancer. Apoptosis 2014, 19, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Rawla, P. Epidemiology of Prostate Cancer. World J. Oncol. 2019, 10, 63–89. [Google Scholar] [CrossRef] [PubMed]
- Bostwick, D.G.; Burke, H.B.; Djakiew, D.; Euling, S.; Ho, S.M.; Landolph, J.; Morrison, H.; Sonawane, B.; Shifflett, T.; Waters, D.J.; et al. Human prostate cancer risk factors. Cancer 2004, 101 (Suppl. S10), 2371–2490. [Google Scholar] [CrossRef]
- Dagnelie, P.C.; Schuurman, A.G.; Goldbohm, R.A.; Van den Brandt, P.A. Diet, anthropometric measures and prostate cancer risk: A review of prospective cohort and intervention studies. BJU Int. 2004, 93, 1139–1150. [Google Scholar] [CrossRef]
- Aronson, W.J.; Barnard, R.J.; Freedland, S.J.; Henning, S.; Elashoff, D.; Jardack, P.M.; Cohen, P.; Heber, D.; Kobayashi, N. Growth inhibitory effect of low fat diet on prostate cancer cells: Results of a prospective, randomized dietary intervention trial in men with prostate cancer. J. Urol. 2010, 183, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Venkateswaran, V.; Klotz, L.H. Diet and prostate cancer: Mechanisms of action and implications for chemoprevention. Nat. Rev. Urol. 2010, 7, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.M.; Shui, I.M.; Mucci, L.A.; Giovannucci, E. Calcium and phosphorus intake and prostate cancer risk: A 24-y follow-up study. Am. J. Clin. Nutr. 2015, 101, 173–183. [Google Scholar] [CrossRef]
- Datta, M.; Schwartz, G.G. Calcium and vitamin D supplementation during androgen deprivation therapy for prostate cancer: A critical review. Oncologist 2012, 17, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Helzlsouer, K.J.; Huang, H.Y.; Alberg, A.J.; Hoffman, S.; Burke, A.; Norkus, E.P.; Morris, J.S.; Comstock, G.W. Association between alpha-tocopherol, gamma-tocopherol, selenium, and subsequent prostate cancer. J. Natl. Cancer Inst. 2000, 92, 2018–2023. [Google Scholar] [CrossRef]
- Giovannucci, E.; Rimm, E.B.; Ascherio, A.; Colditz, G.A.; Spiegelman, D.; Stampfer, M.J.; Willett, W.C. Smoking and risk of total and fatal prostate cancer in United States health professionals. Cancer Epidemiol. Biomark. Prev. 1999, 8 Pt 1, 277–282. [Google Scholar]
- Rizos, C.; Papassava, M.; Golias, C.; Charalabopoulos, K. Alcohol consumption and prostate cancer: A mini review. Exp. Oncol. 2010, 32, 66–70. [Google Scholar]
- Wilson, K.M.; Kasperzyk, J.L.; Rider, J.R.; Kenfield, S.; van Dam, R.M.; Stampfer, M.J.; Giovannucci, E.; Mucci, L.A. Coffee consumption and prostate cancer risk and progression in the Health Professionals Follow-up Study. J. Natl. Cancer Inst. 2011, 103, 876–884. [Google Scholar] [CrossRef]
- Allott, E.H.; Masko, E.M.; Freedland, S.J. Obesity and prostate cancer: Weighing the evidence. Eur. Urol. 2013, 63, 800–809. [Google Scholar] [CrossRef]
- Gallagher, R.P.; Fleshner, N. Prostate cancer: 3. Individual risk factors. CMAJ 1998, 159, 807–813. [Google Scholar] [PubMed]
- Ferris-i-Tortajada, J.; Garcia-i-Castell, J.; Berbel-Tornero, O.; Ortega-Garcia, J.A. Constitutional risk factors in prostate cancer. Actas Urol. Esp. 2011, 35, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.; Paranjape, J.; Brown, T.L.; Nie, H.; Naik, S.; Dong, B.; Chang, A.; Trapp, B.; Fairchild, R.; Colmenares, C.; et al. Interferon action and apoptosis are defective in mice devoid of 2′,5′-oligoadenylate-dependent RNase L. EMBO J. 1997, 16, 6355–6363. [Google Scholar] [CrossRef]
- Chen, H.; Griffin, A.R.; Wu, Y.Q.; Tomsho, L.P.; Zuhlke, K.A.; Lange, E.M.; Gruber, S.B.; Cooney, K.A. RNASEL mutations in hereditary prostate cancer. J. Med. Genet. 2003, 40, e21. [Google Scholar] [CrossRef]
- Erkko, H.; Xia, B.; Nikkila, J.; Schleutker, J.; Syrjakoski, K.; Mannermaa, A.; Kallioniemi, A.; Pylkas, K.; Karppinen, S.M.; Rapakko, K.; et al. A recurrent mutation in PALB2 in Finnish cancer families. Nature 2007, 446, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.J.; Gaudet, M.M.; Pal, P.; Kirchhoff, T.; Balistreri, L.; Vora, K.; Bhatia, J.; Stadler, Z.; Fine, S.W.; Reuter, V.; et al. Germline BRCA mutations denote a clinicopathologic subset of prostate cancer. Clin. Cancer Res. 2010, 16, 2115–2121. [Google Scholar] [CrossRef]
- Okobia, M.N.; Zmuda, J.M.; Ferrell, R.E.; Patrick, A.L.; Bunker, C.H. Chromosome 8q24 variants are associated with prostate cancer risk in a high risk population of African ancestry. Prostate 2011, 71, 1054–1063. [Google Scholar] [CrossRef]
- Robbins, C.M.; Hooker, S.; Kittles, R.A.; Carpten, J.D. EphB2 SNPs and sporadic prostate cancer risk in African American men. PLoS ONE 2011, 6, e19494. [Google Scholar] [CrossRef] [PubMed]
- Hatcher, D.; Daniels, G.; Osman, I.; Lee, P. Molecular mechanisms involving prostate cancer racial disparity. Am. J. Transl. Res. 2009, 1, 235–248. [Google Scholar]
- Vijayakumar, S.; Winter, K.; Sause, W.; Gallagher, M.J.; Michalski, J.; Roach, M.; Porter, A.; Bondy, M. Prostate-specific antigen levels are higher in African-American than in white patients in a multicenter registration study: Results of RTOG 94-12. Int. J. Radiat. Oncol. Biol. Phys. 1998, 40, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Morgan, T.M.; Welty, C.J.; Vakar-Lopez, F.; Lin, D.W.; Wright, J.L. Ductal adenocarcinoma of the prostate: Increased mortality risk and decreased serum prostate specific antigen. J. Urol. 2010, 184, 2303–2307. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, P.A.; Moch, H.; Cubilla, A.L.; Ulbright, T.M.; Reuter, V.E. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part B: Prostate and Bladder Tumours. Eur. Urol. 2016, 70, 106–119. [Google Scholar] [CrossRef]
- Gleason, D.F.; Mellinger, G.T. Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J. Urol. 1974, 111, 58–64. [Google Scholar] [CrossRef]
- McKenney, J.K.; Simko, J.; Bonham, M.; True, L.D.; Troyer, D.; Hawley, S.; Newcomb, L.F.; Fazli, L.; Kunju, L.P.; Nicolas, M.M.; et al. The potential impact of reproducibility of Gleason grading in men with early stage prostate cancer managed by active surveillance: A multi-institutional study. J. Urol. 2011, 186, 465–469. [Google Scholar] [CrossRef]
- Logothetis, C.J.; Gallick, G.E.; Maity, S.N.; Kim, J.; Aparicio, A.; Efstathiou, E.; Lin, S.H. Molecular classification of prostate cancer progression: Foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013, 3, 849–861. [Google Scholar] [CrossRef]
- Chang, A.J.; Autio, K.A.; Roach, M., III; Scher, H.I. High-risk prostate cancer-classification and therapy. Nat. Rev. Clin. Oncol. 2014, 11, 308–323. [Google Scholar] [CrossRef]
- Rittmaster, R.S. 5alpha-reductase inhibitors in benign prostatic hyperplasia and prostate cancer risk reduction. Best Pract. Res. Clin. Endocrinol. Metab. 2008, 22, 389–402. [Google Scholar] [CrossRef]
- Shankar, S.; Chen, X.; Srivastava, R.K. Effects of sequential treatments with chemotherapeutic drugs followed by TRAIL on prostate cancer in vitro and in vivo. Prostate 2005, 62, 165–186. [Google Scholar] [CrossRef] [PubMed]
- Eisenberger, M.A.; Blumenstein, B.A.; Crawford, E.D.; Miller, G.; McLeod, D.G.; Loehrer, P.J.; Wilding, G.; Sears, K.; Culkin, D.J.; Thompson, I.M., Jr.; et al. Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N. Engl. J. Med. 1998, 339, 1036–1042. [Google Scholar] [CrossRef]
- Fujita, K.; Nonomura, N. Role of Androgen Receptor in Prostate Cancer: A Review. World J. Mens Health 2019, 37, 288–295. [Google Scholar] [CrossRef]
- Conteduca, V.; Mosca, A.; Brighi, N.; de Giorgi, U.; Rescigno, P. New Prognostic Biomarkers in Metastatic Castration-Resistant Prostate Cancer. Cells 2021, 10, 193. [Google Scholar] [CrossRef]
- Grossmann, M.E.; Huang, H.; Tindall, D.J. Androgen receptor signaling in androgen-refractory prostate cancer. J. Natl. Cancer Inst. 2001, 93, 1687–1697. [Google Scholar] [CrossRef]
- Palmberg, C.; Koivisto, P.; Hyytinen, E.; Isola, J.; Visakorpi, T.; Kallioniemi, O.P.; Tammela, T. Androgen receptor gene amplification in a recurrent prostate cancer after monotherapy with the nonsteroidal potent antiandrogen Casodex (bicalutamide) with a subsequent favorable response to maximal androgen blockade. Eur. Urol. 1997, 31, 216–219. [Google Scholar] [CrossRef]
- Stanbrough, M.; Bubley, G.J.; Ross, K.; Golub, T.R.; Rubin, M.A.; Penning, T.M.; Febbo, P.G.; Balk, S.P. Increased expression of genes converting adrenal androgens to testosterone in androgen-independent prostate cancer. Cancer Res. 2006, 66, 2815–2825. [Google Scholar] [CrossRef]
- Mohler, J.L.; Gregory, C.W.; Ford, O.H., III; Kim, D.; Weaver, C.M.; Petrusz, P.; Wilson, E.M.; French, F.S. The androgen axis in recurrent prostate cancer. Clin. Cancer Res. 2004, 10, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Titus, M.A.; Schell, M.J.; Lih, F.B.; Tomer, K.B.; Mohler, J.L. Testosterone and dihydrotestosterone tissue levels in recurrent prostate cancer. Clin. Cancer Res. 2005, 11, 4653–4657. [Google Scholar] [CrossRef]
- Mottet, N.; Bellmunt, J.; Bolla, M.; Joniau, S.; Mason, M.; Matveev, V.; Schmid, H.P.; Van der Kwast, T.; Wiegel, T.; Zattoni, F.; et al. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur. Urol. 2011, 59, 572–583. [Google Scholar] [CrossRef] [PubMed]
- Crawford, E.D.; Schellhammer, P.F.; McLeod, D.G.; Moul, J.W.; Higano, C.S.; Shore, N.; Denis, L.; Iversen, P.; Eisenberger, M.A.; Labrie, F. Androgen Receptor Targeted Treatments of Prostate Cancer: 35 Years of Progress with Antiandrogens. J. Urol. 2018, 200, 956–966. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, K.E.; Kelly, W.K. Outsmarting androgen receptor: Creative approaches for targeting aberrant androgen signaling in advanced prostate cancer. Expert Rev. Endocrinol. Metab. 2011, 6, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.A.; Malhotra, S.V.; Stoyanova, T. Second-Generation Antiandrogens: From Discovery to Standard of Care in Castration Resistant Prostate Cancer. Front. Oncol. 2019, 9, 801. [Google Scholar] [CrossRef]
- Al-Salama, Z.T. Apalutamide: First Global Approval. Drugs 2018, 78, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Clegg, N.J.; Wongvipat, J.; Joseph, J.D.; Tran, C.; Ouk, S.; Dilhas, A.; Chen, Y.; Grillot, K.; Bischoff, E.D.; Cai, L.; et al. ARN-509: A novel antiandrogen for prostate cancer treatment. Cancer Res. 2012, 72, 1494–1503. [Google Scholar] [CrossRef]
- Barton, D. Oral agents in cancer treatment: The context for adherence. Semin. Oncol. Nurs. 2011, 27, 104–115. [Google Scholar] [CrossRef]
- Mann, J.E. Apalutamide (Erleada®). Oncol. Times 2018, 40, 16. [Google Scholar] [CrossRef]
- Tran, C.; Ouk, S.; Clegg, N.J.; Chen, Y.; Watson, P.A.; Arora, V.; Wongvipat, J.; Smith-Jones, P.M.; Yoo, D.; Kwon, A.; et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 2009, 324, 787–790. [Google Scholar] [CrossRef]
- Graff, J.N.; Gordon, M.J.; Beer, T.M. Safety and effectiveness of enzalutamide in men with metastatic, castration-resistant prostate cancer. Expert Opin. Pharmacother. 2015, 16, 749–754. [Google Scholar] [CrossRef]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A Target for Anticancer Therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef]
- Li, P.; Nijhawan, D.; Budihardjo, I.; Srinivasula, S.M.; Ahmad, M.; Alnemri, E.S.; Wang, X. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997, 91, 479–489. [Google Scholar] [CrossRef]
- Bruckheimer, E.M.; Cho, S.H.; Sarkiss, M.; Herrmann, J.; McDonnell, T.J. The Bcl-2 gene family and apoptosis. Adv. Biochem. Eng. Biotechnol. 1998, 62, 75–105. [Google Scholar]
- Fresno Vara, J.A.; Casado, E.; de Castro, J.; Cejas, P.; Belda-Iniesta, C.; Gonzalez-Baron, M. PI3K/Akt signalling pathway and cancer. Cancer Treat Rev. 2004, 30, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Oda, E.; Ohki, R.; Murasawa, H.; Nemoto, J.; Shibue, T.; Yamashita, T.; Tokino, T.; Taniguchi, T.; Tanaka, N. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 2000, 288, 1053–1058. [Google Scholar] [CrossRef]
- Fulda, S.; Debatin, K.M. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 2006, 25, 4798–4811. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y. Mechanisms of caspase activation and inhibition during apoptosis. Mol. Cell 2002, 9, 459–470. [Google Scholar] [CrossRef]
- Schneider, P.; Tschopp, J. Apoptosis induced by death receptors. Pharm. Acta Helv. 2000, 74, 281–286. [Google Scholar] [CrossRef]
- Reynolds, A.R.; Kyprianou, N. Growth factor signalling in prostatic growth: Significance in tumour development and therapeutic targeting. Br. J. Pharmacol. 2006, 147 (Suppl. S2), S144–S152. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Adhami, V.M.; Mukhtar, H. Apoptosis by dietary agents for prevention and treatment of prostate cancer. Endocr. Relat. Cancer 2010, 17, R39–R52. [Google Scholar] [CrossRef]
- Xiang, Y.; Wang, Z.; Murakami, J.; Plummer, S.; Klein, E.A.; Carpten, J.D.; Trent, J.M.; Isaacs, W.B.; Casey, G.; Silverman, R.H. Effects of RNase L mutations associated with prostate cancer on apoptosis induced by 2′,5′-oligoadenylates. Cancer Res. 2003, 63, 6795–6801. [Google Scholar]
- Hernandez, I.; Maddison, L.A.; Wei, Y.; DeMayo, F.; Petras, T.; Li, B.; Gingrich, J.R.; Rosen, J.M.; Greenberg, N.M. Prostate-specific expression of p53(R172L) differentially regulates p21, Bax, and mdm2 to inhibit prostate cancer progression and prolong survival. Mol. Cancer Res. 2003, 1, 1036–1047. [Google Scholar]
- Gurova, K.V.; Rokhlin, O.W.; Budanov, A.V.; Burdelya, L.G.; Chumakov, P.M.; Cohen, M.B.; Gudkov, A.V. Cooperation of two mutant p53 alleles contributes to Fas resistance of prostate carcinoma cells. Cancer Res. 2003, 63, 2905–2912. [Google Scholar]
- McKenzie, S.; Kyprianou, N. Apoptosis evasion: The role of survival pathways in prostate cancer progression and therapeutic resistance. J. Cell Biochem. 2006, 97, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.P.; Li, S.; Chen, Y.M.; Li, H.B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef]
- Yan, X.; Qi, M.; Li, P.; Zhan, Y.; Shao, H. Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell Biosci. 2017, 7, 50. [Google Scholar] [CrossRef]
- Gupta, S.; Afaq, F.; Mukhtar, H. Involvement of nuclear factor-kappa B, Bax and Bcl-2 in induction of cell cycle arrest and apoptosis by apigenin in human prostate carcinoma cells. Oncogene 2002, 21, 3727–3738. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Kanwal, R.; Shankar, E.; Datt, M.; Chance, M.R.; Fu, P.; MacLennan, G.T.; Gupta, S. Apigenin blocks IKKalpha activation and suppresses prostate cancer progression. Oncotarget 2015, 6, 31216–31232. [Google Scholar] [CrossRef] [PubMed]
- Erdogan, S.; Doganlar, O.; Doganlar, Z.B.; Serttas, R.; Turkekul, K.; Dibirdik, I.; Bilir, A. The flavonoid apigenin reduces prostate cancer CD44(+) stem cell survival and migration through PI3K/Akt/NF-kappaB signaling. Life Sci. 2016, 162, 77–86. [Google Scholar] [CrossRef]
- Shukla, S.; Bhaskaran, N.; Babcook, M.A.; Fu, P.; Maclennan, G.T.; Gupta, S. Apigenin inhibits prostate cancer progression in TRAMP mice via targeting PI3K/Akt/FoxO pathway. Carcinogenesis 2014, 35, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C.; Katan, M.B. Dietary flavonoids: Intake, health effects and bioavailability. Food Chem. Toxicol. 1999, 37, 937–942. [Google Scholar] [CrossRef]
- Singh, S.; Gupta, P.; Meena, A.; Luqman, S. Acacetin, a flavone with diverse therapeutic potential in cancer, inflammation, infections and other metabolic disorders. Food Chem. Toxicol. 2020, 145, 111708. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Agrawal, P.; Yim, D.; Agarwal, C.; Agarwal, R. Acacetin inhibits cell growth and cell cycle progression, and induces apoptosis in human prostate cancer cells: Structure-activity relationship with linarin and linarin acetate. Carcinogenesis 2005, 26, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tu, Y.; Zhou, Q.; Hua, A.; Geng, P.; Chen, F.; Han, A.; Liu, J.; Dai, D.; Wang, S.; et al. Evaluation of acacetin inhibition potential against cytochrome P450 in vitro and in vivo. Chem. Biol. Interact. 2020, 329, 109147. [Google Scholar] [CrossRef]
- Ko, Y.C.; Choi, H.S.; Liu, R.; Kim, J.H.; Kim, S.L.; Yun, B.S.; Lee, D.S. Inhibitory Effects of Tangeretin, A Citrus Peel-Derived Flavonoid, on Breast Cancer Stem Cell Formation through Suppression of Stat3 Signaling. Molecules 2020, 25, 2599. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.B.; Xiao, N.; Liu, X.J. Dietary flavonoid tangeretin induces reprogramming of epithelial to mesenchymal transition in prostate cancer cells by targeting the PI3K/Akt/mTOR signaling pathway. Oncol. Lett. 2018, 15, 433–440. [Google Scholar] [CrossRef]
- Gravdal, K.; Halvorsen, O.J.; Haukaas, S.A.; Akslen, L.A. A switch from E-cadherin to N-cadherin expression indicates epithelial to mesenchymal transition and is of strong and independent importance for the progress of prostate cancer. Clin. Cancer Res. 2007, 13, 7003–7011. [Google Scholar] [CrossRef]
- Guo, J.-J.; Li, Y.-J.; Xin, L.-L. Tangeretin prevents prostate cancer cell proliferation and induces apoptosis via activation of Notch signalling and regulating the androgen receptor (AR) pathway and the phosphoinositide 3-kinase (PI3k)/Akt/mTOR pathways. Bangladesh J. Pharmacol. 2015, 10, 937–947. [Google Scholar] [CrossRef]
- Rawson, N.; Ho, C.-T.; Li, S. Efficacious anti-cancer property of flavonoids from citrus peels. Food Sci. Hum. Wellness 2014, 3, 104–109. [Google Scholar] [CrossRef]
- Lai, C.S.; Li, S.; Miyauchi, Y.; Suzawa, M.; Ho, C.T.; Pan, M.H. Potent anti-cancer effects of citrus peel flavonoids in human prostate xenograft tumors. Food Funct. 2013, 4, 944–949. [Google Scholar] [CrossRef]
- Lipke, M.; Sundaram, C.P. Laparoscopic radical prostatectomy. J. Minim. Access Surg. 2005, 1, 196–201. [Google Scholar]
- Bolla, M.; Van Tienhoven, G.; Warde, P.; Dubois, J.B.; Mirimanoff, R.O.; Storme, G.; Bernier, J.; Kuten, A.; Sternberg, C.; Billiet, I.; et al. External irradiation with or without long-term androgen suppression for prostate cancer with high metastatic risk: 10-year results of an EORTC randomised study. Lancet Oncol. 2010, 11, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Attard, G.; Murphy, L.; Clarke, N.W.; Cross, W.; Jones, R.J.; Parker, C.C.; Gillessen, S.; Cook, A.; Brawley, C.; Amos, C.L.; et al. Systemic Therapy in Advancing or Metastatic Prostate cancer: Evaluation of Drug Efficacy (STAMPEDE) investigators. Abiraterone acetate and prednisolone with or without enzalutamide for high-risk non-metastatic prostate cancer: A meta-analysis of primary results from two randomised controlled phase 3 trials of the STAMPEDE platform protocol. Lancet 2022, 399, 447–460. [Google Scholar] [PubMed]
- Prostate Cancer: Diagnosis and Management; National Institute for Health and Care Excellence: London, UK, 2021.
- Gurumurthy, S.; Murthi Vasudevan, K.; Rangnekar, V.M. Regulation of Apoptosis in Prostate Cancer. Cancer Metastasis Rev. 2001, 20, 225–243. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.Y.; Zhang, H.X.; Mezei, M.; Cui, M. Molecular docking: A powerful approach for structure-based drug discovery. Curr. Comput. Aided Drug Des. 2011, 7, 146–157. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, S.H.; Kim, H.H.; Park, M.Y.; Bhosale, P.B.; Abusaliya, A.; Won, C.K.; Park, K.I.; Kim, E.; Heo, J.D.; Kim, H.W.; et al. Flavones: The Apoptosis in Prostate Cancer of Three Flavones Selected as Therapeutic Candidate Models. Int. J. Mol. Sci. 2023, 24, 9240. https://doi.org/10.3390/ijms24119240
Jeong SH, Kim HH, Park MY, Bhosale PB, Abusaliya A, Won CK, Park KI, Kim E, Heo JD, Kim HW, et al. Flavones: The Apoptosis in Prostate Cancer of Three Flavones Selected as Therapeutic Candidate Models. International Journal of Molecular Sciences. 2023; 24(11):9240. https://doi.org/10.3390/ijms24119240
Chicago/Turabian StyleJeong, Se Hyo, Hun Hwan Kim, Min Young Park, Pritam Bhagwan Bhosale, Abuyaseer Abusaliya, Chung Kil Won, Kwang Il Park, Eunhye Kim, Jeong Doo Heo, Hyun Wook Kim, and et al. 2023. "Flavones: The Apoptosis in Prostate Cancer of Three Flavones Selected as Therapeutic Candidate Models" International Journal of Molecular Sciences 24, no. 11: 9240. https://doi.org/10.3390/ijms24119240
APA StyleJeong, S. H., Kim, H. H., Park, M. Y., Bhosale, P. B., Abusaliya, A., Won, C. K., Park, K. I., Kim, E., Heo, J. D., Kim, H. W., Ahn, M., Seong, J. K., & Kim, G. S. (2023). Flavones: The Apoptosis in Prostate Cancer of Three Flavones Selected as Therapeutic Candidate Models. International Journal of Molecular Sciences, 24(11), 9240. https://doi.org/10.3390/ijms24119240








