Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub
Abstract
1. Introduction
1.1. MDD Pathophysiology
1.2. Stress, Resilience and the Amygdala
1.3. Mitochondria
1.4. Neurotransmitters and Antidepressants
1.5. Pro-Inflammatory Cytokines, Tryptophan and Kynurenine
1.6. Aryl Hydrocarbon Receptor
1.7. Gut Microbiome, LPS and Butyrate
1.8. Ceramide
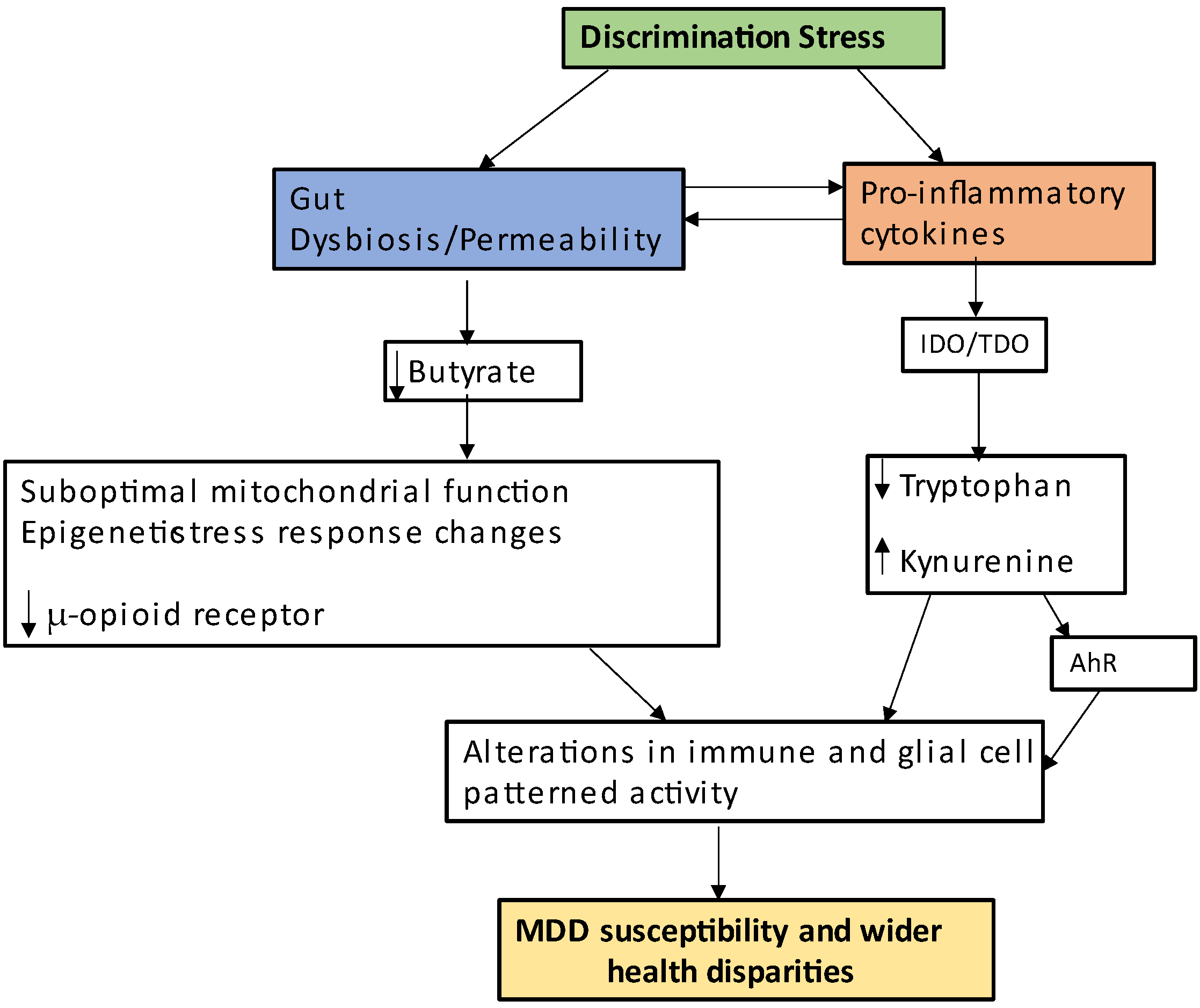
1.9. Social Processes and Discrimination
1.10. Circadian Dysruption
1.11. Summary
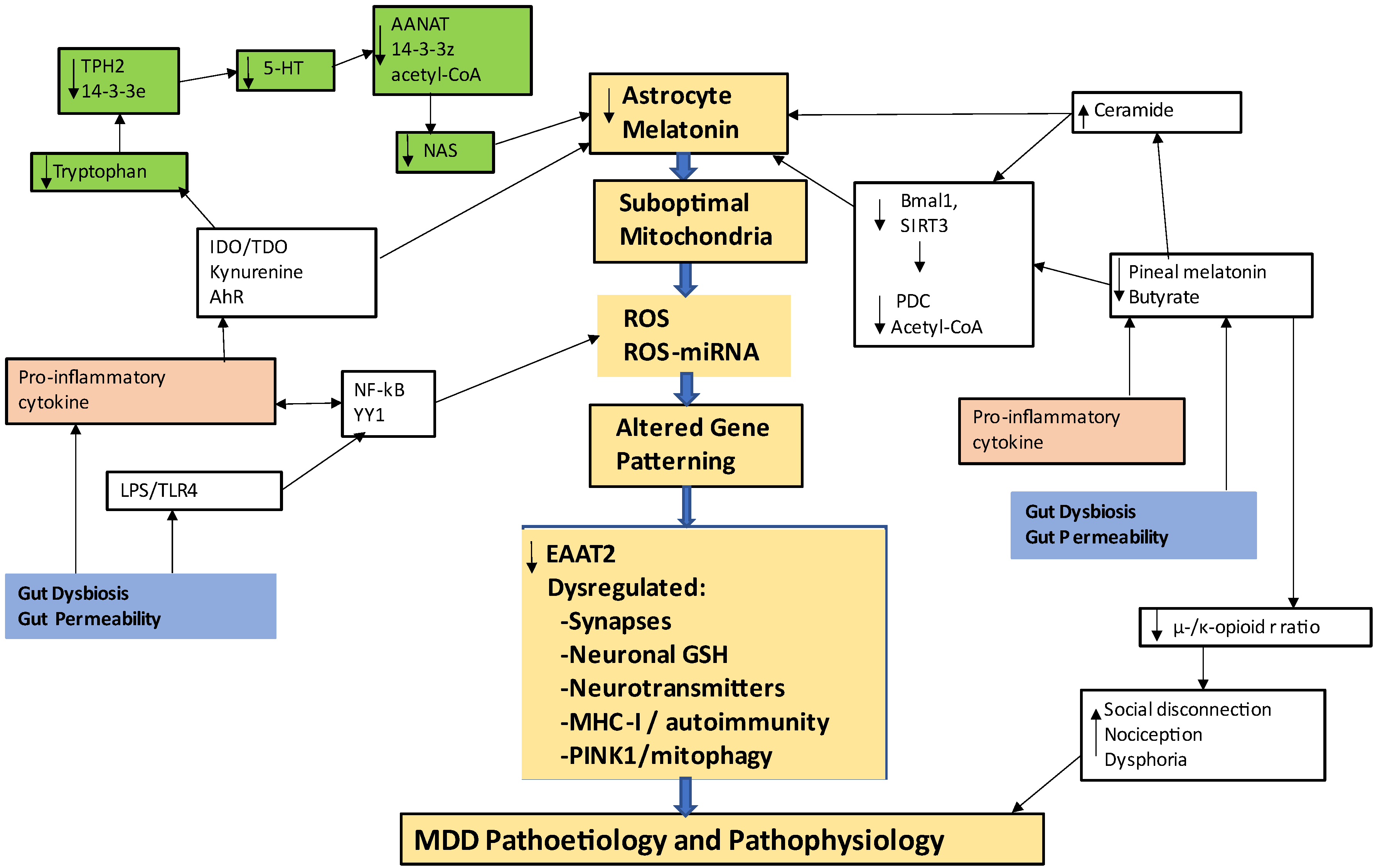
2. Integrating MDD Pathophysiology
2.1. Gut Microbiome, Butyrate and Mitochondria, ROS and microRNAs
2.2. Gut Microbiome, Amygdala and Opioidergic System
2.3. Gut Microbiome and Ceramide
2.4. Gut Microbiome, Amygdala and Personality
2.5. Neurogenesis and the Astrocyte Mitochondrial Melatonergic Pathway
2.6. Integrating Wider Bodies of Biomedical Data
3. Astrocytes as a Crucial Hub
Mitochondrial Melatonergic Pathway
4. Future Research Directions
- Does gut microbiome-derived butyrate require the capacity of the mitochondrial melatonergic pathway to be upregulated in order to mediate its extensive beneficial effects, especially in immune and glial cells?
- Does ceramide suppress 14-3-3ζ [196] and therefore the mitochondrial melatonergic pathway across different cell types, with consequences for the impact of gut microbiome-derived butyrate on mitochondrial and cellular function? As the over-expression of 14-3-3ζ prevents ceramide-induced autophagy [197], is this mediated via the upregulation of the melatonergic pathway?
- The importance of astrocytes, astrocytic mitochondrial melatonergic pathway, and the astrocyte network is paralleled in enteric glial cells. Enteric glial cells, like astrocytes, were classically conceptualized as providers of energy and antioxidants to enteric neurons. However, a growing body of data shows enteric glial cells to not only determine enteric neuron survival and function, but also to be an important interface with the gut microbiome, the mucosal immune system and vagal inputs to the gut. This may be important to MDD pathoetiology and its association with neurodegenerative conditions [198,199,200]. Do enteric glial cells have a mitochondrial melatonergic pathway, producing melatonin as shown in astrocytes, including regulation by apolipoprotein (Apo)E4 [170]? Are the effects of gut microbiome-derived butyrate in enteric glial cells dependent upon the capacity to upregulate the melatonergic pathway?
- Do NF-kB and YY1 induce the melatonergic pathway in astrocytes, leading to autocrine and paracrine effects that dampen inflammatory processes? Do endothelial cells, pericytes and systemic factors crossing the BBB regulate the astrocyte tryptophan-melatonergic pathway, thereby impacting on local neuronal regulation?
- Are the developmental regulatory effects of YY1 in astrocytes [175] dependent upon the capacity of YY1 to induce the melatonergic pathway?
- Are the detrimental effects of YY1 across a host of diverse medical conditions, including cancers, dementia, and type I diabetes, a consequence of melatonergic pathway suppression?
- Would the inhibition of the miRNAs that suppress 14-3-3 and the mitochondrial melatonergic pathway (e.g., miR-7, miR-375, miR-451 and miR-709) better optimize the beneficial effects of butyrate in MDD and related medical conditions?
- Is there an aging-associated increase in miRNAs that suppress the mitochondrial melatonergic pathway via the inhibition of the 14-3-3 isoforms, as with miR-709 in the murine liver [158]?
- Would the suppression of 14-3-3 isoforms and the mitochondrial melatonergic pathway have consequences for the regulation of the opioidergic system, e.g., the μ-/κ-opioid receptor ratio, and thereby impact on social processes as well as affective state? The integration of social processes, especially societal social stratification and discrimination, is challenging within a physiological framework. The above provides a basis for future investigations, including as to the relevance of a classical stress model, namely ‘stress-gut-dysbiosis-permeability-pro-inflammatory-cytokines--IDO-TDO-kynurenine-AhR’ pathway coupled to the differential opioidergic system regulation by butyrate and the mitochondrial melatonergic pathway. Would this be relevant to racial and sexual discrimination stress and its MDD and health disparities consequences?
- Some of the beneficial effects of melatonin are mediated via the alpha 7 nicotinic acetylcholine receptor (α7nAChR), including in the gut [201]. Are the gut barrier preserving effects of melatonin mediated via the α7nAChR on enteric glial cells?
- The α7nAChR, AhR and melatonin receptors are expressed on the mitochondrial membrane [87]. How do these receptors regulate mitochondrial function and how do they interact with the mitochondrial melatonergic pathway?
- Humans are unique in having duplicant α7nAChR (dupα7), which occludes α7nAChR effects. How relevant is this in cells linked to MDD pathophysiology, especially astrocytes, including in the regulation of cognition, given data showing its expression in distinct cortex regions in psychiatric conditions [202]?
- Future research investigating AhR effects in different cells, including astrocytes under different conditions and different AhR ligands, should better clarify the role of the AhR in MDD pathophysiology, including as to whether AhR effects vary according to the availability of the mitochondrial melatonergic pathway.
- Do early developmental impacts on the gut microbiome and gut permeability change the amygdala’s regulation of cortex and wider brain development, including inter-area interactions, with consequences for emotional and social processes? Does this involve alterations in the interactions of butyrate and the mitochondrial melatonergic pathway, including via the regulation of amygdala β-endorphin and μ-/κ-opioid receptor ratio, with consequent impacts on how the amygdala ‘affectively’ regulates other brain regions and their interaction.
5. Treatment Implications
- Given the powerful role of the gut in the pathoetiology and pathophysiology of MDD, the regulation of the gut microbiome and gut permeability has become a significant target for MDD treatment. As noted, enteric glial cells are significant determinants of enteric neuronal releases as well as providing an important interface for the gut microbiome and mucosal immune cells, platelets and vagal inputs. Recent work shows endocannabinoids to be significant inhibitors of enteric glial activation, with consequent benefits on the maintenance of the gut barrier. This would suggest the potential utilization of neutriceutical cannabinoids and cannabinoid-like molecules in the regulation of gut-linked MDD pathophysiology. The maintenance of the gut barrier will decrease levels of circulating LPS and therefore the activation of the LPS/NF-kB-YY1 pathway, which is problematic under conditions when neither NF-kB nor YY1 can induce the melatonergic pathway.
- Butyrate is one of the main beneficial factors produced by the gut microbiome, with butyrate producing bacteria being encouraged by pre- and pro-biotics as well as the dietary intake of foods that ferment in the large intestine, including complex carbohydrates. The nutriceutical, sodium butyrate, may also be useful as it provides an immediate increase in butyrate availability, whilst also encouraging the growth of butyrate producing bacteria [23]. As noted, such regulation of the gut microbiome/permeability has significant impacts on mitochondrial function across the body, with relevance to MDD pathophysiology.
- A number of studies have indicated the beneficial effects of α7nAChR agonists in MDD [203]. The α7nAChR is induced by melatonin and its utility in the gut, especially enteric glial cells and how they interface with vagal ACh inputs to the gut may be a relevant treatment target as well as the α7nAChR suppression of astrocytes and microglia reactivity [204].
- Clearly, early developmental processes are an important aspect of MDD pathoetiology. The above provides a research framework which should provide early development biomarkers and preventative interventions.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| α7nAChR | alpha 7 nicotinic acetylcholine receptor |
| AANAT | aralkylamine N-acetyltransferase |
| AhR | aryl hydrocarbon receptor |
| ASMT | acetylserotonin methytransferase |
| BDNF | brain-derived neurotrophic factor |
| CRH | corticotrophin releasing hormone |
| CYP | cytochrome P450 |
| dupα7 | duplicating alpha 7nicotinic receptor |
| EAAT | excitatory amino acid transporter |
| H2O2 | hydrogen peroxide |
| HDAC | histone deacetylase |
| HMGB | high-mobility group box |
| IDO | indoleamine 2,3-dioxygenase |
| KYNA | kynurenic acid |
| LPS | lipopolysaccharide |
| MDD | major depressive disorder |
| mPRC | medial prefrontal cortex |
| NAS | N-acetylserotonin |
| NF-kB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NMDA | N-methyl-d-aspartate |
| PDC | pyruvate dehydrogenase comple |
| SARS-CoV-2 | severe acute respiratory disease, coronavirus 2 |
| SMase | sphingomyelinase |
| TCA | tricarboxylic acid |
| TDO | tryptophan 2,3-dioxygenase |
| TLR | Toll-like receptor |
| TMAO | trimethylamine N-oxide |
| TPH2 | tryptophan hydroxylase |
| YY1 | yin yang 1 |
References
- Anderson, G.; Maes, M. Oxidative/nitrosative stress and immuno-inflammatory pathways in depression: Treatment implications. Curr. Pharm. Des. 2014, 20, 3812–3847. [Google Scholar] [CrossRef] [PubMed]
- Baldessarini, R.J. Epidemiology of suicide: Recent developments. Epidemiol. Psychiatr. Sci. 2019, 29, e71. [Google Scholar] [CrossRef] [PubMed]
- Perez-Caballero, L.; Torres-Sanchez, S.; Romero-López-Alberca, C.; González-Saiz, F.; Mico, J.A.; Berrocoso, E. Monoaminergic system and depression. Cell Tissue Res. 2019, 377, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Slyepchenko, A.; Maes, M.; Machado-Vieira, R.; Anderson, G.; Solmi, M.; Sanz, Y.; Berk, M.; Köhler, C.A.; Carvalho, A.F. Intestinal Dysbiosis, Gut Hyperpermeability and Bacterial Translocation: Missing Links Between Depression, Obesity and Type 2 Diabetes. Curr. Pharm. Des. 2016, 22, 6087–6106. [Google Scholar] [CrossRef] [PubMed]
- Somani, A.; Singh, A.K.; Gupta, B.; Nagarkoti, S.; Dalal, P.K.; Dikshit, M. Oxidative and Nitrosative Stress in Major Depressive Disorder: A Case Control Study. Brain Sci. 2022, 12, 144. [Google Scholar] [CrossRef] [PubMed]
- Scaini, G.; Mason, B.L.; Diaz, A.P.; Jha, M.K.; Soares, J.C.; Trivedi, M.H.; Quevedo, J. Dysregulation of mitochondrial dynamics, mitophagy and apoptosis in major depressive disorder: Does inflammation play a role? Mol. Psychiatry 2022, 27, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Sarno, E.; Moeser, A.J.; Robison, A.J. Neuroimmunology of depression. Adv. Pharmacol. 2021, 91, 259–292. [Google Scholar] [CrossRef]
- Gamradt, S.; Hasselmann, H.; Taenzer, A.; Brasanac, J.; Stiglbauer, V.; Sattler, A.; Sajitz-Hermstein, M.; Kierszniowska, S.; Ramien, C.; Nowacki, J.; et al. Reduced mitochondrial respiration in T cells of patients with major depressive disorder. iScience 2021, 24, 103312. [Google Scholar] [CrossRef]
- Park, S.C. Neurogenesis and antidepressant action. Cell Tissue Res. 2019, 377, 95–106. [Google Scholar] [CrossRef]
- Liu, J.; Fang, J.; Wang, Z.; Rong, P.; Hong, Y.; Fan, Y.; Wang, X.; Park, J.; Jin, Y.; Liu, C.; et al. Transcutaneous vagus nerve stimulation modulates amygdala functional connectivity in patients with depression. J. Affect. Disord. 2016, 205, 319–326. [Google Scholar] [CrossRef]
- Rodríguez-Soto, N.C.; Buxó, C.J.; Morou-Bermudez, E.; Pérez-Edgar, K.; Ocasio-Quiñones, I.T.; Surillo-González, M.B.; Martinez, K.G. The impact of prenatal maternal stress due to potentially traumatic events on child temperament: A systematic review. Dev. Psychobiol. 2021, 63, e22195. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. How Immune-inflammatory Processes Link CNS and Psychiatric Disorders: Classification and Treatment Implications. CNS Neurol. Disord. Drug Targets 2017, 16, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. Role of Opioidergic System in Regulating Depression Pathophysiology. Curr. Pharm. Des. 2020, 26, 5317–5334. [Google Scholar] [CrossRef] [PubMed]
- Al-Hakeim, H.K.; Al-Fadhel, S.Z.; Al-Dujaili, A.H.; Carvalho, A.; Sriswasdi, S.; Maes, M. Development of a Novel Neuro-immune and Opioid-Associated Fingerprint with a Cross-Validated Ability to Identify and Authenticate Unknown Patients with Major Depression: Far Beyond Differentiation, Discrimination, and Classification. Mol. Neurobiol. 2019, 56, 7822–7835. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhang, R.; Li, Z.; Yao, Z.; Xie, X.; Bai, R.; Li, L.; Zhang, X.; Zhang, S.; Shen, Y.; et al. Sini powder with paroxetine ameliorates major depressive disorder by modulating circadian rhythm: A randomized, double-blind, placebo-controlled trial. J. Pineal Res. 2022, 73, e12832. [Google Scholar] [CrossRef]
- Anderson, G. Linking the biological underpinnings of depression: Role of mitochondria interactions with melatonin, inflammation, sirtuins, tryptophan catabolites, DNA repair and oxidative and nitrosative stress, with consequences for classification and cognition. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 80 Pt C, 255–266. [Google Scholar] [CrossRef]
- Zajkowska, Z.; Gullett, N.; Walsh, A.; Zonca, V.; Pedersen, G.A.; Souza, L.; Kieling, C.; Fisher, H.L.; Kohrt, B.A.; Mondelli, V. Cortisol and development of depression in adolescence and young adulthood—A systematic review and meta-analysis. Psychoneuroendocrinology 2022, 136, 105625. [Google Scholar] [CrossRef]
- Vanuytsel, T.; van Wanrooy, S.; Vanheel, H.; Vanormelingen, C.; Verschueren, S.; Houben, E.; Salim Rasoel, S.; Tόth, J.; Holvoet, L.; Farré, R.; et al. Psychological stress and corticotropin-releasing hormone increase intestinal permeability in humans by a mast cell-dependent mechanism. Gut 2014, 63, 1293–1299. [Google Scholar] [CrossRef]
- Kanamori, A.; Tanaka, F.; Ominami, M.; Nadatani, Y.; Fukunaga, S.; Otani, K.; Hosomi, S.; Kamata, N.; Nagami, Y.; Taira, K.; et al. Psychological Stress Exacerbates Inflammation of the Ileum via the Corticotropin-Releasing Hormone-Mast Cell Axis in a Mouse Model of Eosinophilic Enteritis. Int. J. Mol. Sci. 2022, 23, 8538. [Google Scholar] [CrossRef]
- Samojedny, S.; Czechowska, E.; Pańczyszyn-Trzewik, P.; Sowa-Kućma, M. Postsynaptic Proteins at Excitatory Synapses in the Brain-Relationship with Depressive Disorders. Int. J. Mol. Sci. 2022, 23, 11423. [Google Scholar] [CrossRef]
- Kocamaz, D.; Franzke, C.; Gröger, N.; Braun, K.; Bock, J. Early Life Stress-Induced Epigenetic Programming of Hippocampal NPY-Y2 Receptor Gene Expression Changes in Response to Adult Stress. Front. Cell Neurosci. 2022, 16, 936979. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Liu, S.; Li, S.; Yang, J.; Hu, G.; Xu, C.; Song, W. Psychological stress in inflammatory bowel disease: Psychoneuroimmunological insights into bidirectional gut-brain communications. Front. Immunol. 2022, 13, 1016578. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. Gut Dysbiosis Dysregulates Central and Systemic Homeostasis via Suboptimal Mitochondrial Function: Assessment, Treatment and Classification Implications. Curr. Top. Med. Chem. 2020, 20, 524–539. [Google Scholar] [CrossRef] [PubMed]
- Galley, J.D.; Mashburn-Warren, L.; Blalock, L.C.; Lauber, C.L.; Carroll, J.E.; Ross, K.M.; Hobel, C.; Coussons-Read, M.; Dunkel Schetter, C.; Gur, T.L. Maternal Anxiety, Depression and Stress Affects Offspring Gut Microbiome Diversity and Bifidobacterial Abundances. Brain Behav. Immun. 2022, 107, S0889-159100410-X. [Google Scholar] [CrossRef]
- Eskandari, F.; Salimi, M.; Binayi, F.; Abdollahifar, M.A.; Eftekhary, M.; Hedayati, M.; Ghanbarian, H.; Zardooz, H. Investigating the Effects of Maternal Separation on Hypothalamic-Pituitary-Adrenal Axis and Glucose Homeostasis under Chronic Social Defeat Stress in Young Adult Male Rat Offspring. Neuroendocrinology 2022, 9, 1–9. [Google Scholar] [CrossRef]
- Hashimoto, S.; Ichiki, M.; Ishii, Y.; Morishita, C.; Shimura, A.; Kusumi, I.; Inoue, T.; Masuya, J. Victimization in Childhood Influences Presenteeism in Adulthood via Mediation by Neuroticism and Perceived Job Stressors. Neuropsychiatr. Dis. Treat. 2022, 18, 265–274. [Google Scholar] [CrossRef]
- Lin, J.; Su, Y.; Lv, X.; Liu, Q.; Wang, G.; Wei, J.; Zhu, G.; Chen, Q.; Tian, H.; Zhang, K.; et al. Childhood adversity, adulthood adversity and suicidal ideation in Chinese patients with major depressive disorder: In line with stress sensitization. Eur. Arch. Psychiatry Clin. Neurosci. 2022, 272, 887–896. [Google Scholar] [CrossRef]
- Kuzminskaite, E.; Penninx, B.W.J.H.; van Harmelen, A.L.; Elzinga, B.M.; Hovens, J.G.F.M.; Vinkers, C.H. Childhood Trauma in Adult Depressive and Anxiety Disorders: An Integrated Review on Psychological and Biological Mechanisms in the NESDA Cohort. J. Affect. Disord. 2021, 283, 179–191. [Google Scholar] [CrossRef]
- Cusack, S.E.; Sheerin, C.M.; Aggen, S.H.; Kendler, K.S.; Amstadter, A.B. Modeling the association between and predictors of two constructs of resilience. Soc. Psychiatry Psychiatr. Epidemiol. 2022, 57, 1471–1481. [Google Scholar] [CrossRef]
- Maes, M.; Anderson, G.; Betancort Medina, S.R.; Seo, M.; Ojala, J.O. Integrating Autism Spectrum Disorder Pathophysiology: Mitochondria, Vitamin A, CD38, Oxytocin, Serotonin and Melatonergic Alterations in the Placenta and Gut. Curr. Pharm. Des. 2019, 25, 4405–4420. [Google Scholar] [CrossRef]
- Eyles, D.W. How do established developmental risk-factors for schizophrenia change the way the brain develops? Transl. Psychiatry 2021, 11, 158. [Google Scholar] [CrossRef] [PubMed]
- Pinson, M.R.; Chung, D.D.; Adams, A.M.; Scopice, C.; Payne, E.A.; Sivakumar, M.; Miranda, R.C. Extracellular Vesicles in Premature Aging and Diseases in Adulthood Due to Developmental Exposures. Aging Dis. 2021, 12, 1516–1535. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Hu, S.; Zhao, Z.; Zhang, L.; Ma, Q. Antenatal Hypoxia Accelerates the Onset of Alzheimer’s Disease Pathology in 5xFAD Mouse Model. Front. Aging Neurosci. 2020, 12, 251. [Google Scholar] [CrossRef] [PubMed]
- Hans, L.K.M.; Dinga, R.; Hahn, T.; Ching, C.R.K.; Eyler, L.T.; Aftanas, L.; Aghajani, M.; Aleman, A.; Baune, B.T.; Berger, K.; et al. Brain aging in major depressive disorder: Results from the ENIGMA major depressive disorder working group. Mol. Psychiatry 2021, 26, 5124–5139. [Google Scholar] [CrossRef]
- Gao, W.; Salzwedel, A.P.; Carlson, A.L.; Xia, K.; Azcarate-Peril, M.A.; Styner, M.A.; Thompson, A.L.; Geng, X.; Goldman, B.D.; Gilmore, J.H.; et al. Gut microbiome and brain functional connectivity in infants-a preliminary study focusing on the amygdala. Psychopharmacology 2019, 236, 1641–1651. [Google Scholar] [CrossRef]
- Seo, M.; Anderson, G. Gut-Amygdala Interactions in Autism Spectrum Disorders: Developmental Roles via regulating Mitochondria, Exosomes, Immunity and microRNAs. Curr. Pharm. Des. 2019, 25, 4344–4356. [Google Scholar] [CrossRef]
- Cowan, C.S.M.; Hoban, A.E.; Ventura-Silva, A.P.; Dinan, T.G.; Clarke, G.; Cryan, J.F. Gutsy Moves: The Amygdala as a Critical Node in Microbiota to Brain Signaling. Bioessays 2018, 40, 1700172. [Google Scholar] [CrossRef]
- Anderson, G. Neuronal-immune interactions in mediating stress effects in the etiology and course of schizophrenia: Role of the amygdala in developmental co-ordination. Med. Hypotheses 2011, 76, 54–60. [Google Scholar] [CrossRef]
- Lei, Z.; Xie, L.; Li, C.H.; Lam, Y.Y.; Ramkrishnan, A.S.; Fu, Z.; Zeng, X.; Liu, S.; Iqbal, Z.; Li, Y. Chemogenetic Activation of Astrocytes in the Basolateral Amygdala Contributes to Fear Memory Formation by Modulating the Amygdala-Prefrontal Cortex Communication. Int. J. Mol. Sci. 2022, 23, 6092. [Google Scholar] [CrossRef]
- Liu, L.; Dai, L.; Xu, D.; Wang, Y.; Bai, L.; Chen, X.; Li, M.; Yang, S.; Tang, Y. Astrocyte secretes IL-6 to modulate PSD-95 palmitoylation in basolateral amygdala and depression-like behaviors induced by peripheral nerve injury. Brain Behav. Immun. 2022, 104, 139–154. [Google Scholar] [CrossRef]
- Jia, C.; Drew Gill, W.; Lovins, C.; Brown, R.W.; Hagg, T. Female-specific role of ciliary neurotrophic factor in the medial amygdala in promoting stress responses. Neurobiol. Stress. 2022, 17, 100435. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, M.L.; Wulf, H.A.; Browne, C.A.; Lucki, I. The kappa opioid receptor antagonist aticaprant reverses behavioral effects from unpredictable chronic mild stress in male mice. Psychopharmacology 2020, 237, 3715–3728. [Google Scholar] [CrossRef] [PubMed]
- Serra, I.; Esparza, J.; Delgado, L.; Martín-Monteagudo, C.; Puigròs, M.; Podlesniy, P.; Trullás, R.; Navarrete, M. Ca2+-modulated photoactivatable imaging reveals neuron-astrocyte glutamatergic circuitries within the nucleus accumbens. Nat. Commun. 2022, 13, 5272. [Google Scholar] [CrossRef] [PubMed]
- Grotle, A.K.; Darling, A.M.; Saunders, E.F.; Fadel, P.J.; Trott, D.W.; Greaney, J.L. Augmented T-cell mitochondrial reactive oxygen species in adults with major depressive disorder. Am. J. Physiol. Heart Circ. Physiol. 2022, 322, H568–H574. [Google Scholar] [CrossRef]
- Wang, Y.; Wei, J.; Chen, T.; Yang, X.; Zhao, L.; Wang, M.; Dou, Y.; Du, Y.; Ni, R.; Li, T.; et al. A Whole Transcriptome Analysis in Peripheral Blood Suggests That Energy Metabolism and Inflammation Are Involved in Major Depressive Disorder. Front. Psychiatry 2022, 13, 907034. [Google Scholar] [CrossRef]
- Huang, Y.C.; Lin, P.Y.; Lee, Y.; Lee, C.Y.; Lo, Y.C.; Hung, C.F.; Chen, C.S. Metabolic syndrome components and leukocyte telomere length in patients with major depressive disorder. World J. Biol. Psychiatry 2021, 16, 483–492. [Google Scholar] [CrossRef]
- Wang, Y.; Ni, J.; Gao, C.; Xie, L.; Zhai, L.; Cui, G.; Yin, X. Mitochondrial transplantation attenuates lipopolysaccharide- induced depression-like behaviors. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 93, 240–249. [Google Scholar] [CrossRef]
- Oldford, C.; Kuksal, N.; Gill, R.; Young, A.; Mailloux, R.J. Estimation of the hydrogen peroxide producing capacities of liver and cardiac mitochondria isolated from C57BL/6N and C57BL/6J mice. Free Radic. Biol. Med. 2019, 135, 15–27. [Google Scholar] [CrossRef]
- Fisher-Wellman, K.H.; Gilliam, L.A.A.; Lin, C.T.; Cathey, B.L.; Lark, D.S.; Darrell Neufer, P. Mitochondrial glutathione depletion reveals a novel role for the pyruvate dehydrogenase complex as a key H2O2-emitting source under conditions of nutrient overload. Free Radic. Biol. Med. 2013, 65, 1201–1208. [Google Scholar] [CrossRef]
- He, J.; Jiang, B.H. Interplay between Reactive oxygen Species and MicroRNAs in Cancer. Curr. Pharmacol. Rep. 2016, 2, 82–90. [Google Scholar] [CrossRef]
- Rasheed, M.; Asghar, R.; Firdoos, S.; Ahmad, N.; Nazir, A.; Ullah, K.M.; Li, N.; Zhuang, F.; Chen, Z.; Deng, Y. A Systematic Review of Circulatory microRNAs in Major Depressive Disorder: Potential Biomarkers for Disease Prognosis. Int. J. Mol. Sci. 2022, 23, 1294. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo-Pereira, C.; Villarejo-Zori, B.; Cipriano, P.C.; Tavares, D.; Ramírez-Pardo, I.; Boya, P.; Vieira, H.L.A. Carbon Monoxide Stimulates Both Mitophagy and Mitochondrial Biogenesis to Mediate Protection Against Oxidative Stress in Astrocytes. Mol. Neurobiol. 2022, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Ignatenko, O.; Malinen, S.; Rybas, S.; Vihinen, H.; Nikkanen, J.; Kononov, A.; Jokitalo, E.S.; Ince-Dunn, G.; Suomalainen, A. Mitochondrial dysfunction compromises ciliary homeostasis in astrocytes. J. Cell Biol. 2023, 222, e202203019. [Google Scholar] [CrossRef]
- Denker, N.; Harders, A.R.; Arend, C.; Dringen, R. Consumption and Metabolism of Extracellular Pyruvate by Cultured Rat Brain Astrocytes. Neurochem. Res. 2022, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Fernández-González, I.; Galea, E. Astrocyte strategies in the energy-efficient brain. Essays Biochem. 2022, 9, EBC20220077. [Google Scholar] [CrossRef] [PubMed]
- Moraes, J.B.; Maes, M.; Roomruangwong, C.; Bonifacio, K.L.; Barbosa, D.S.; Vargas, H.O.; Anderson, G.; Kubera, M.; Carvalho, A.F.; Nunes, S.O.V. In major affective disorders, early life trauma predict increased nitro-oxidative stress, lipid peroxidation and protein oxidation and recurrence of major affective disorders, suicidal behaviors and a lowered quality of life. Metab. Brain Dis. 2018, 33, 1081–1096. [Google Scholar] [CrossRef] [PubMed]
- Artigas, F.; Bortolozzi, A.; Celada, P. Can we increase speed and efficacy of antidepressant treatments? Part I: General aspects and monoamine-based strategies. Eur. Neuropsychopharmacol. 2018, 28, 445–456. [Google Scholar] [CrossRef]
- Patel, K.; Allen, S.; Haque, M.N.; Angelescu, I.; Baumeister, D.; Tracy, D.K. Bupropion: A systematic review and meta-analysis of effectiveness as an antidepressant. Ther. Adv. Psychopharmacol. 2016, 6, 99–144. [Google Scholar] [CrossRef]
- Vasiliu, O. Investigational Drugs for the Treatment of Depression (Part 1): Monoaminergic, Orexinergic, GABA-Ergic, and Anti-Inflammatory Agents. Front. Pharmacol. 2022, 13, 884143. [Google Scholar] [CrossRef]
- Khoodoruth, M.A.S.; Estudillo-Guerra, M.A.; Pacheco-Barrios, K.; Nyundo, A.; Chapa-Koloffon, G.; Ouanes, S. Glutamatergic System in Depression and Its Role in Neuromodulatory Techniques Optimization. Front. Psychiatry 2022, 13, 886918. [Google Scholar] [CrossRef]
- Bhatia, N.Y.; Ved, H.S.; Kale, P.P.; Doshi, G.M. Importance of Exploring N-Methyl-D-Aspartate (NMDA) as a Future Perspective Target in Depression. CNS Neurol. Disord. Drug Targets 2022, 21, 1004–1016. [Google Scholar] [CrossRef] [PubMed]
- Fries, G.R.; Saldana, V.A.; Finnstein, J.; Rein, T. Molecular pathways of major depressive disorder converge on the synapse. Mol. Psychiatry 2022, 6, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. Reconceptualizing adult neurogenesis: Role for sphingosine-1-phosphate and fibroblast growth factor-1 in co-ordinating astrocyte-neuronal precursor interactions. CNS Neurol. Disord. Drug Targets 2014, 13, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, S.; Qin, Q.; Xu, Z.; Qu, Y.; Wang, Y.; Wang, J.; Du, Z.; Yuan, S.; Hong, S.; et al. Genetic and Pharmacological Inhibition of Astrocytic Mysm1 Alleviates Depressive-Like Disorders by Promoting ATP Production. Adv. Sci. 2022, 22, e2204463. [Google Scholar] [CrossRef]
- Portal, B.; Vasile, F.; Zapata, J.; Lejards, C.; Ait Tayeb, A.E.K.; Colle, R.; Verstuyft, C.; Corruble, E.; Rouach, N.; Guiard, B.P. Astroglial Connexins Inactivation Increases Relapse of Depressive-like Phenotype after Antidepressant Withdrawal. Int. J. Mol. Sci. 2022, 23, 13227. [Google Scholar] [CrossRef]
- Zhong, Q.; Chen, J.J.; Wang, Y.; Shao, W.H.; Zhou, C.J.; Xie, P. Differential Gut Microbiota Compositions Related With the Severity of Major Depressive Disorder. Front. Cell Infect. Microbiol. 2022, 12, 907239. [Google Scholar] [CrossRef]
- Simon, M.S.; Schiweck, C.; Arteaga-Henríquez, G.; Poletti, S.; Haarman, B.C.M.; Dik, W.A.; Schwarz, M.; Vrieze, E.; Mikova, O.; Joergens, S.; et al. Monocyte mitochondrial dysfunction, inflammaging, and inflammatory pyroptosis in major depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 111, 110391. [Google Scholar] [CrossRef]
- Leite Dantas, R.; Freff, J.; Ambrée, O.; Beins, E.C.; Forstner, A.J.; Dannlowski, U.; Baune, B.T.; Scheu, S.; Alferink, J. Dendritic Cells: Neglected Modulators of Peripheral Immune Responses and Neuroinflammation in Mood Disorders? Cells 2021, 10, 941. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Gómez-Lahoz, A.M.; Orozco, A.; Lahera, G.; Diaz, D.; Ortega, M.A.; Albillos, A.; Quintero, J.; Aubá, E.; Monserrat, J.; et al. Expansion of CD4 T Lymphocytes Expressing Interleukin 17 and Tumor Necrosis Factor in Patients with Major Depressive Disorder. J. Pers. Med. 2021, 11, 220. [Google Scholar] [CrossRef]
- Seguella, L.; Pesce, M.; Capuano, R.; Casano, F.; Pesce, M.; Corpetti, C.; Vincenzi, M.; Maftei, D.; Lattanzi, R.; Del Re, A.; et al. High-fat diet impairs duodenal barrier function and elicits glia-dependent changes along the gut-brain axis that are required for anxiogenic and depressive-like behaviors. J. Neuroinflammation 2021, 18, 115. [Google Scholar] [CrossRef]
- Suzuki, H.; Savitz, J.; Kent Teague, T.; Gandhapudi, S.K.; Tan, C.; Misaki, M.; McKinney, B.A.; Irwin, M.R.; Drevets, W.C.; Bodurka, J.; et al. Altered populations of natural killer cells, cytotoxic T lymphocytes, and regulatory T cells in major depressive disorder: Association with sleep disturbance. Brain Behav. Immun. 2017, 66, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Grosse, L.; Hoogenboezem, T.; Ambrée, O.; Bellingrath, S.; Jörgens, S.; de Wit, H.J.; Wijkhuijs, A.M.; Arolt, V.; Drexhage, H.A. Deficiencies of the T and natural killer cell system in major depressive disorder: T regulatory cell defects are associated with inflammatory monocyte activation. Brain Behav. Immun. 2016, 54, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Lan, X.; Wang, C.; Li, W.; Chao, Z.; Lao, G.; Wu, K.; Li, G.; Ning, Y.; Zhou, Y. The association between overweight/obesity and poor cognitive function is mediated by inflammation in patients with major depressive disorder. J. Affect. Disord. 2022, 313, 118–125. [Google Scholar] [CrossRef] [PubMed]
- De Melo, L.G.P.; Nunes, S.O.V.; Anderson, G.; Vargas, H.O.; Barbosa, D.S.; Galecki, P.; Carvalho, A.F.; Maes, M. Shared metabolic and immune-inflammatory, oxidative and nitrosative stress pathways in the metabolic syndrome and mood disorders. Prog. Neuropsychopharmacol. Biol. Psychiatry 2017, 78, 34–50. [Google Scholar] [CrossRef]
- Gong, X.; Chang, R.; Zou, J.; Tan, S.; Huang, Z. The role and mechanism of tryptophan-kynurenine metabolic pathway in depression. Rev. Neurosci. 2022, 5, 47. [Google Scholar] [CrossRef]
- Paul, E.R.; Schwieler, L.; Erhardt, S.; Boda, S.; Trepci, A.; Kämpe, R.; Asratian, A.; Holm, L.; Yngve, A.; Dantzer, R.; et al. Peripheral and central kynurenine pathway abnormalities in major depression. Brain Behav. Immun. 2022, 101, 136–145. [Google Scholar] [CrossRef]
- Brown, S.J.; Brown, A.M.; Purves-Tyson, T.D.; Huang, X.F.; Shannon Weickert, C.; Newell, K.A. Alterations in the kynurenine pathway and excitatory amino acid transporter-2 in depression with and without psychosis: Evidence of a potential astrocyte pathology. J. Psychiatr. Res. 2022, 147, 203–211. [Google Scholar] [CrossRef]
- Steiner, J.; Walter, M.; Gos, T.; Guillemin, G.J.; Bernstein, H.G.; Sarnyai, Z.; Mawrin, C.; Brisch, R.; Bielau, H.; Meyer, Z.U.; et al. Severe depression is associated with increased microglial quinolinic acid in subregions of the anterior cingulate gyrus: Evidence for an immune-modulated glutamatergic neurotransmission? J. Neuroinflammation 2011, 8, 94. [Google Scholar] [CrossRef]
- Kholghi, G.; Eskandari, M.; Shokouhi Qare Saadlou, M.S.; Zarrindast, M.R.; Vaseghi, S. Night shift hormone: How does melatonin affect depression? Physiol. Behav. 2022, 252, 113835. [Google Scholar] [CrossRef]
- Xie, Q.E.; Du, X.; Wang, M.; Xie, F.; Zhang, Z.; Cao, Y.; Liu, Y. Identification of Serotonin as a Predictive Marker for Breast Cancer Patients. Int. J. Gen. Med. 2021, 14, 1939–1948. [Google Scholar] [CrossRef]
- Chen, Y.; Sha, R.; Xu, L.; Xia, Y.; Liu, Y.; Li, X.; Xie, H.Q.; Tang, N.; Zhao, B. 2,3,7,8-Tetrachlorodibenzo-p-dioxin promotes migration ability of primary cultured rat astrocytes via aryl hydrocarbon receptor. J. Environ. Sci. 2019, 76, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Lowery, R.L.; Latchney, S.E.; Peer, R.P.; Lamantia, C.E.; Lordy, K.A.; Opanashuk, L.A.; McCall, M.; Majewska, A.K. Gestational and lactational exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin primes cortical microglia to tissue injury. Brain Behav. Immun. 2022, 101, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Vrzalová, A.; Pečinková, P.; Illés, P.; Gurská, S.; Džubák, P.; Szotkowski, M.; Hajdúch, M.; Mani, S.; Dvořák, Z. Mixture Effects of Tryptophan Intestinal Microbial Metabolites on Aryl Hydrocarbon Receptor Activity. Int. J. Mol. Sci. 2022, 23, 10825. [Google Scholar] [CrossRef]
- Aguiniga, L.M.; Yang, W.; Yaggie, R.E.; Schaeffer, A.J.; Klumpp, D.J.; MAPP Research Network Study Group. Acyloxyacyl hydrolase modulates depressive-like behaviors through aryl hydrocarbon receptor. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 317, R289–R300. [Google Scholar] [CrossRef] [PubMed]
- Juricek, L.; Coumoul, X. The Aryl Hydrocarbon Receptor and the Nervous System. Int. J. Mol. Sci. 2018, 19, 2504. [Google Scholar] [CrossRef] [PubMed]
- Ren, R.; Fang, Y.; Sherchan, P.; Lu, Q.; Lenahan, C.; Zhang, J.H.; Zhang, J.; Tang, J. Kynurenine/Aryl Hydrocarbon Receptor Modulates Mitochondria-Mediated Oxidative Stress and Neuronal Apoptosis in Experimental Intracerebral Hemorrhage. Antioxid. Redox Signal. 2022, 28, 215. [Google Scholar] [CrossRef]
- Anderson, G.; Maes, M. Interactions of Tryptophan and Its Catabolites with Melatonin and the Alpha 7 Nicotinic Receptor in Central Nervous System and Psychiatric Disorders: Role of the Aryl Hydrocarbon Receptor and Direct Mitochondria Regulation. Int. J. Tryptophan Res. 2017, 10, 1178646917691738. [Google Scholar] [CrossRef]
- Hassan, L.; Peek, N.; Lovell, K.; Carvalho, A.F.; Solmi, M.; Stubbs, B.; Firth, J. Disparities in COVID-19 infection, hospitalisation and death in people with schizophrenia, bipolar disorder, and major depressive disorder: A cohort study of the UK Biobank. Mol. Psychiatry 2022, 27, 1248–1255. [Google Scholar] [CrossRef]
- Guo, L.; Schurink, B.; Roos, E.; Nossent, E.J.; Duitman, J.W.; Vlaar, A.P.; van der Valk, P.; Vaz, F.M.; Yeh, S.R.; Geeraerts, Z.; et al. Indoleamine 2,3-dioxygenase (IDO)-1 and IDO-2 activity and severe course of COVID-19. J. Pathol. 2022, 256, 256–261. [Google Scholar] [CrossRef]
- Anderson, G.; Carbone, A.; Mazzoccoli, G. Aryl Hydrocarbon Receptor Role in Co-Ordinating SARS-CoV-2 Entry and Symptomatology: Linking Cytotoxicity Changes in COVID-19 and Cancers; Modulation by Racial Discrimination Stress. Biology 2020, 9, 249. [Google Scholar] [CrossRef]
- Bhatt, S.; Kanoujia, J.; Mohanalakshmi, S.; Patil, C.R.; Gupta, G.; Chellappan, D.K.; Dua, K. Role of Brain-Gut-Microbiota Axis in Depression: Emerging Therapeutic Avenues. CNS Neurol. Disord. Drug Targets 2022, 29, 804. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Mon, M.A.; Gómez, A.M.; Orozco, A.; Lahera, G.; Sosa, M.D.; Diaz, D.; Auba, E.; Albillos, A.; Monserrat, J.; Alvarez-Mon, M. Abnormal Distribution and Function of Circulating Monocytes and Enhanced Bacterial Translocation in Major Depressive Disorder. Front. Psychiatry 2019, 10, 812. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, F.; Carpinteiro, A.; Edwards, M.J.; Wilson, G.C.; Keitsch, S.; Soddemann, M.; Wilker, B.; Kleuser, B.; Becker, K.A.; Müller, C.P.; et al. Stress induces major depressive disorder by a neutral sphingomyelinase 2-mediated accumulation of ceramide-enriched exosomes in the blood plasma. J. Mol. Med. 2022, 100, 1493–1508. [Google Scholar] [CrossRef]
- Edwards, M.J.; Wilson, G.C.; Keitsch, S.; Soddemann, M.; Wilker, B.; Müller, C.P.; Kornhuber, J.; Gulbins, E. Molecular targets of endothelial phosphatidic acid regulating major depressive disorder. J. Neurochem. 2022, 13, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, J.; Mao, G.; Zuo, J.; Li, S.; Yang, Y.; Thring, R.W.; Wu, M.; Tong, H. Sargassum fusiforme fucoidan alleviates diet-induced insulin resistance by inhibiting colon-derived ceramide biosynthesis. Food Funct. 2021, 12, 8440–8453. [Google Scholar] [CrossRef]
- Chen, W.F.; Lee, J.J.; Chang, C.C.; Lin, K.H.; Wang, S.H.; Sheu, J.R. Platelet protease-activated receptor (PAR)4, but not PAR1, associated with neutral sphingomyelinase responsible for thrombin-stimulated ceramide-NF-κB signaling in human platelets. Haematologica 2013, 98, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Murray, T.E.; Richards, C.M.; Robert-Gostlin, V.N.; Bernath, A.K.; Lindhout, I.A.; Klegeris, A. Potential neurotoxic activity of diverse molecules released by astrocytes. Brain Res. Bull. 2022, 189, 80–101. [Google Scholar] [CrossRef]
- Anderson, G.; Rodriguez, M.; Reiter, R.J. Multiple Sclerosis: Melatonin, Orexin, and Ceramide Interact with Platelet Activation Coagulation Factors and Gut-Microbiome-Derived Butyrate in the Circadian Dysregulation of Mitochondria in Glia and Immune Cells. Int. J. Mol. Sci. 2019, 20, 5500. [Google Scholar] [CrossRef]
- Eakin, C.M.; Lai, T.; Cohen, J.G. Alarming trends and disparities in high-risk endometrial cancer. Curr. Opin. Obstet. Gynecol. 2022, 14, 832. [Google Scholar] [CrossRef]
- Berger, Z.; Altiery, D.E.; Jesus, V.; Assoumou, S.A.; Greenhalgh, T. Long COVID and Health Inequities: The Role of Primary Care. Milbank Q. 2021, 99, 519–541. [Google Scholar] [CrossRef]
- Russell, D.W.; Clavél, F.D.; Cutrona, C.E.; Abraham, W.T.; Burzette, R.G. Neighborhood racial discrimination and the development of major depression. J. Abnorm. Psychol. 2018, 127, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Amato, K.R.; Arrieta, M.C.; Azad, M.B.; Bailey, M.T.; Broussard, J.L.; Bruggeling, C.E.; Claud, E.C.; Costello, E.K.; Davenport, E.R.; Dutilh, B.E.; et al. The human gut microbiome and health inequities. Proc. Natl. Acad. Sci. USA 2021, 118, e2017947118. [Google Scholar] [CrossRef] [PubMed]
- Caceres, B.A.; Barcelona, V.; Vo, D.; Suero-Tejeda, N.; Jackman, K.; Taylor, J.; Corwin, E. Investigating the Associations of Everyday Discrimination and Inflammation in Latina Women: A Pilot Study. Biol. Res. Nurs. 2021, 23, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Seaton, E.K.; Zeiders, K.H. Daily racial discrimination experiences, ethnic-racial identity, and diurnal cortisol patterns among Black adults. Cult. Divers. Ethn. Minor. Psychol. 2021, 27, 145–155. [Google Scholar] [CrossRef]
- Dasari, S.S.; Archer, M.; Mohamed, N.E.; Tewari, A.K.; Figueiro, M.G.; Kyprianou, N. Circadian Rhythm Disruption as a Contributor to Racial Disparities in Prostate Cancer. Cancers 2022, 14, 5116. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Fan, R.; Xie, L.; Shi, X.; Dong, K.; Zhang, S.; Tao, J.; Xu, W.; Ma, D.; Chen, J.; et al. A Growing Link between Circadian Rhythms, Type 2 Diabetes Mellitus and Alzheimer’s Disease. Int. J. Mol. Sci. 2022, 23, 504. [Google Scholar] [CrossRef]
- Tonon, A.C.; Constantino, D.B.; Amando, G.R.; Abreu, A.C.; Francisco, A.P.; de Oliveira, M.A.B.; Pilz, L.K.; Xavier, N.B.; Rohrsetzer, F.; Souza, L.; et al. Sleep disturbances, circadian activity, and nocturnal light exposure characterize high risk for and current depression in adolescence. Sleep 2022, 45, zsac104. [Google Scholar] [CrossRef]
- Beatty Moody, D.L.; Waldstein, S.R.; Tobin, J.N.; Cassells, A.; Schwartz, J.C.; Brondolo, E. Lifetime racial/ethnic discrimination and ambulatory blood pressure: The moderating effect of age. Health Psychol. 2016, 35, 333–342. [Google Scholar] [CrossRef]
- Lee, E.K.; Poon, P.; Yu, C.P.; Lee, V.W.; Chung, V.C.; Wong, S.Y. Controlled-release oral melatonin supplementation for hypertension and nocturnal hypertension: A systematic review and meta-analysis. J. Clin. Hypertens 2022, 24, 529–535. [Google Scholar] [CrossRef]
- Anderson, G. The effects of melatonin on signaling pathways and molecules involved in glioma: Melatonin and glioblastoma: Pathophysiology and treatment. Fundam. Clin. Pharmacol. 2020, 34, 189–191. [Google Scholar] [CrossRef]
- Rahman, S.; Alzarea, S. Glial mechanisms underlying major depressive disorder: Potential therapeutic opportunities. Prog. Mol. Biol. Transl. Sci. 2019, 167, 159–178. [Google Scholar] [CrossRef] [PubMed]
- Halassa, M.M.; Haydon, P.G. Integrated brain circuits: Astrocytic networks modulate neuronal activity and behavior. Annu. Rev. Physiol. 2010, 72, 335–355. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Bao, Y.; Chen, W.; He, Y. Melatonin exerts neuroprotective effects by attenuating astro- and microgliosis and suppressing inflammatory response following spinal cord injury. Neuropeptides 2020, 79, 102002. [Google Scholar] [CrossRef] [PubMed]
- Hartung, E.E.; Mukhtar, S.Z.; Shah, S.M.; Niles, L.P. 5-Azacytidine upregulates melatonin MT1 receptor expression in rat C6 glioma cells: Oncostatic implications. Mol. Biol. Rep. 2020, 47, 4867–4873. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.J.; Engstler, A.J.; Sellmann, C.; Ziegenhardt, D.; Landmann, M.; Kanuri, G.; Lounis, H.; Schröder, M.; Vetter, W.; Bergheim, I. Sodium butyrate protects mice from the development of the early signs of non-alcoholic fatty liver disease: Role of melatonin and lipid peroxidation. Br. J. Nutr. 2016, 116, 1682–1693. [Google Scholar] [CrossRef]
- Vučković, A.M.; Venerando, R.; Tibaldi, E.; Bosello Travain, V.; Roveri, A.; Bordin, L.; Miotto, G.; Cozza, G.; Toppo, S.; Maiorino, M.; et al. Aerobic pyruvate metabolism sensitizes cells to ferroptosis primed by GSH depletion. Free Radic. Biol. Med. 2021, 167, 45–53. [Google Scholar] [CrossRef]
- Magenta, A.; Dellambra, E.; Ciarapica, R.; Capogrossi, M.C. Oxidative stress, microRNAs and cytosolic calcium homeostasis. Cell Calcium 2016, 60, 207–217. [Google Scholar] [CrossRef]
- Jelen, L.A.; Stone, J.M.; Young, A.H.; Mehta, M.A. The opioid system in depression. Neurosci. Biobehav. Rev. 2022, 140, 104800. [Google Scholar] [CrossRef]
- Jindal, S.; Kumar, N.; Shah, A.A.; Shah, A.; Gourishetti, K.; Chamallamudi, M.R. Histone Deacetylase Inhibitors Prevented the Development of Morphine Tolerance by Decreasing IL6 Production and Upregulating μ-Opioid Receptors. CNS Neurol. Disord. Drug Targets 2021, 20, 190–198. [Google Scholar] [CrossRef]
- Meguro, Y.; Miyano, K.; Hirayama, S.; Yoshida, Y.; Ishibashi, N.; Ogino, T.; Fujii, Y.; Manabe, S.; Eto, M.; Nonaka, M.; et al. Neuropeptide oxytocin enhances μ opioid receptor signaling as a positive allosteric modulator. J. Pharm. Sci. 2018, 137, 67–75. [Google Scholar] [CrossRef]
- Shavali, S.; Ho, B.; Govitrapong, P.; Sawlom, S.; Ajjimaporn, A.; Klongpanichapak, S.; Ebadi, M. Melatonin exerts its analgesic actions not by binding to opioid receptor subtypes but by increasing the release of beta-endorphin an endogenous opioid. Brain Res. Bull. 2005, 64, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G. Pathoetiology and pathophysiology of borderline personality: Role of prenatal factors, gut microbiome, mu- and kappa-opioid receptors in amygdala-PFC interactions. Prog. Neuropsychopharmacol. Biol. Psychiatry 2020, 98, 109782. [Google Scholar] [CrossRef] [PubMed]
- Palomares-Castillo, E.; Hernández-Pérez, O.R.; Pérez-Carrera, D.; Crespo-Ramírez, M.; Fuxe, K.; Pérez de la Mora, M. The intercalated paracapsular islands as a module for integration of signals regulating anxiety in the amygdala. Brain Res. 2012, 1476, 211–234. [Google Scholar] [CrossRef] [PubMed]
- Mague, S.D.; Pliakas, A.M.; Todtenkopf, M.S.; Tomasiewicz, H.C.; Zhang, Y.; Stevens, W.C., Jr.; Jones, R.M.; Portoghese, P.S.; Carlezon, W.A., Jr. Antidepressant-like effects of kappa-opioid receptor antagonists in the forced swim test in rats. J. Pharm. Exp. Ther. 2003, 305, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.S.; Xu, W.J.; Zhu, H.Q.; Gao, L.; Lai, M.J.; Zhang, F.Q.; Zhou, W.H.; Liu, H.F. Effects of histone deacetylase inhibitor sodium butyrate on heroin seeking behavior in the nucleus accumbens in rats. Brain Res. 2016, 1652, 151–157. [Google Scholar] [CrossRef]
- Matuskey, D.; Dias, M.; Naganawa, M.; Pittman, B.; Henry, S.; Li, S.; Gao, H.; Ropchan, J.; Nabulsi, N.; Carson, R.E.; et al. Social status and demographic effects of the kappa opioid receptor: A PET imaging study with a novel agonist radiotracer in healthy volunteers. Neuropsychopharmacology 2019, 44, 1714–1719. [Google Scholar] [CrossRef]
- Volkova, A.; Ruggles, K.; Schulfer, A.; Gao, Z.; Ginsberg, S.D.; Blaser, M.J. Effects of early-life penicillin exposure on the gut microbiome and frontal cortex and amygdala gene expression. iScience 2021, 24, 102797. [Google Scholar] [CrossRef]
- Humphreys, K.L.; Camacho, M.C.; Roth, M.C.; Estes, E.C. Prenatal stress exposure and multimodal assessment of amygdala-medial prefrontal cortex connectivity in infants. Dev. Cogn. Neurosci. 2020, 46, 100877. [Google Scholar] [CrossRef]
- McGinty, V.B.; Grace, A.A. Activity-dependent depression of medial prefrontal cortex inputs to accumbens neurons by the basolateral amygdala. Neuroscience 2009, 162, 1429–1436. [Google Scholar] [CrossRef]
- Duan, K.; Gu, Q.; Petralia, R.S.; Wang, Y.X.; Panja, D.; Liu, X.; Lehmann, M.L.; Zhu, H.; Zhu, J.; Li, Z. Mitophagy in the basolateral amygdala mediates increased anxiety induced by aversive social experience. Neuron 2021, 109, 3793–3809.e8. [Google Scholar] [CrossRef]
- Tran, T.T.; Postal, B.G.; Demignot, S.; Ribeiro, A.; Osinski, C.; Pais de Barros, J.P.; Blachnio-Zabielska, A.; Leturque, A.; Rousset, M.; Carrière, V.; et al. Short Term Palmitate Supply Impairs Intestinal Insulin Signaling via Ceramide Production. J. Biol. Chem. 2016, 291, 16328–16338. [Google Scholar] [CrossRef]
- Ueda, N. A Rheostat of Ceramide and Sphingosine-1-Phosphate as a Determinant of Oxidative Stress-Mediated Kidney Injury. Int. J. Mol. Sci. 2022, 23, 4010. [Google Scholar] [CrossRef]
- Suda, K.; Matsuda, K. How Microbes Affect Depression: Underlying Mechanisms via the Gut-Brain Axis and the Modulating Role of Probiotics. Int. J. Mol. Sci. 2022, 23, 1172. [Google Scholar] [CrossRef] [PubMed]
- Meinitzer, S.; Baranyi, A.; Holasek, S.; Schnedl, W.J.; Zelzer, S.; Mangge, H.; Herrmann, M.; Meinitzer, A.; Enko, D. Sex-Specific Associations of Trimethylamine-N-Oxide and Zonulin with Signs of Depression in Carbohydrate Malabsorbers and Nonmalabsorbers. Dis. Markers 2020, 2020, 7897240. [Google Scholar] [CrossRef] [PubMed]
- Ochi, S.; Mori, T.; Iga, J.I.; Ueno, S.I. Prevalence of Comorbid Dementia in Late-life Depression and Bipolar Disorder: A Retrospective Inpatient Study. J. Alzheimers Dis. Rep. 2022, 6, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Yamane, M.; Yamane, S. The induction of colonocyte differentiation in CaCo-2 cells by sodium butyrate causes an increase in glucosylceramide synthesis in order to avoid apoptosis based on ceramide. Arch. Biochem. Biophys. 2007, 459, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Duan, C.; Li, Y.; Lu, H.; Guo, K.; Ge, X.; Chen, T.; Shang, Y.; Liu, H.; Zhang, D. Sodium butyrate reduces overnutrition-induced microglial activation and hypothalamic inflammation. Int. Immunopharmacol. 2022, 111, 109083. [Google Scholar] [CrossRef] [PubMed]
- Lobzhanidze, G.; Lordkipanidze, T.; Zhvania, M.; Japaridze, N.; MacFabe, D.F.; Pochkidze, N.; Gasimov, E.; Rzaev, F. Effect of propionic acid on the morphology of the amygdala in adolescent male rats and their behavior. Micron 2019, 125, 102732. [Google Scholar] [CrossRef]
- Calarco, C.A.; Lobo, M.K. Depression and substance use disorders: Clinical comorbidity and shared neurobiology. Int. Rev. Neurobiol. 2021, 157, 245–309. [Google Scholar] [CrossRef]
- Lyon, K.A.; Elliott, R.; Ware, K.; Juhasz, G.; Brown, L.J.E. Associations between Facets and Aspects of Big Five Personality and Affective Disorders:A Systematic Review and Best Evidence Synthesis. J. Affect. Disord. 2021, 288, 175–188, Erratum in: J. Affect. Disord. 2021, 294, 115. [Google Scholar] [CrossRef]
- Jylhä, P.; Melartin, T.; Rytsälä, H.; Isometsä, E. Neuroticism, introversion, and major depressive disorder—Traits, states, or scars? Depress. Anxiety 2009, 26, 325–334. [Google Scholar] [CrossRef]
- Jang, S.W.; Liu, X.; Pradoldej, S.; Tosini, G.; Chang, Q.; Iuvone, P.M.; Ye, K. N-acetylserotonin activates TrkB receptor in a circadian rhythm. Proc. Natl. Acad. Sci. USA 2010, 107, 3876–3881. [Google Scholar] [CrossRef]
- Anderson, G.; Mazzoccoli, G. Left Ventricular Hypertrophy: Roles of Mitochondria CYP1B1 and Melatonergic Pathways in Co-Ordinating Wider Pathophysiology. Int. J. Mol. Sci. 2019, 20, 4068. [Google Scholar] [CrossRef]
- Li, Q.; Wang, P.; Huang, C.; Chen, B.; Liu, J.; Zhao, M.; Zhao, J. N-Acetyl Serotonin Protects Neural Progenitor Cells Against Oxidative Stress-Induced Apoptosis and Improves Neurogenesis in Adult Mouse Hippocampus Following Traumatic Brain Injury. J. Mol. Neurosci. 2019, 67, 574–588. [Google Scholar] [CrossRef]
- Yoo, D.Y.; Nam, S.M.; Kim, W.; Lee, C.H.; Won, M.H.; Hwang, I.K.; Yoon, Y.S. N-acetylserotonin increases cell proliferation and differentiating neuroblasts with tertiary dendrites through upregulation of brain-derived neurotrophic factor in the mouse dentate gyrus. J. Vet. Med. Sci. 2011, 73, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Tran, A.A.; De Smet, M.; Grant, G.D.; Khoo, T.K.; Pountney, D.L. Investigating the Convergent Mechanisms between Major Depressive Disorder and Parkinson’s Disease. Complex. Psychiatry 2021, 6, 47–61. [Google Scholar] [CrossRef]
- Young, C.A.; Ealing, J.; McDermott, C.J.; Williams, T.L.; Al-Chalabi, A.; Majeed, T.; Talbot, K.; Harrower, T.; Faull, C.; Malaspina, A.; et al. Prevalence of depression in amyotrophic lateral sclerosis/motor neuron disease: Multi-attribute ascertainment and trajectories over 30 months. Amyotroph. Lateral Scler. Frontotemporal. Degener. 2022, 6, 1–9. [Google Scholar] [CrossRef]
- Wenger, A.; Calabrese, P. Comparing underlying mechanisms of depression in multiple sclerosis and rheumatoid arthritis. J. Integr. Neurosci. 2021, 20, 765–776. [Google Scholar] [CrossRef]
- do Sacramento, P.M.; Sales, M.; Kasahara, T.M.; Monteiro, C.; Oyamada, H.; Dias, A.S.O.; Lopes, L.; Castro, C.T.; Rossi, Á.D.; Milioni, L.M.; et al. Major depression favors the expansion of Th17-like cells and decrease the proportion of CD39+Treg cell subsets in response to myelin antigen in multiple sclerosis patients. Cell Mol. Life Sci. 2022, 79, 298. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Liu, X.; Rosales-Corral, S.A.; Acuna-Castroviejo, D.; Reiter, R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: A hypothesis related to melatonin’s primary function and evolution in eukaryotes. J. Pineal Res. 2013, 54, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G. Tumour Microenvironment: Roles of the Aryl Hydrocarbon Receptor, O-GlcNAcylation, Acetyl-CoA and Melatonergic Pathway in Regulating Dynamic Metabolic Interactions across Cell Types-Tumour Microenvironment and Metabolism. Int. J. Mol. Sci. 2020, 22, 141. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Carbone, A.; Mazzoccoli, G. Tryptophan Metabolites and Aryl Hydrocarbon Receptor in Severe Acute Respiratory Syndrome, Coronavirus-2 (SARS-CoV-2) Pathophysiology. Int. J. Mol. Sci. 2021, 22, 1597. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Xu, Z.; Zhang, Z.; Mo, Y.; Deng, Y.; Li, L.; Fei, S.; Wu, J.; Wang, K.; Zhang, Q.; et al. RNA-Seq approach to investigate the effects of melatonin on bone marrow-derived dendritic cells from dextran sodium sulfate-induced colitis mice. Toxicology 2022, 481, 153354. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Zhang, J.; Zhou, Y.; Li, X.; Li, H.; Liu, J.; Gou, K.; Zhao, J.; Cui, S. MicroRNA-7 inhibits melatonin synthesis by acting as a linking molecule between leptin and norepinephrine signaling pathways in pig pineal gland. J. Pineal Res. 2019, 66, e12552. [Google Scholar] [CrossRef] [PubMed]
- Pagan, C.; Goubran-Botros, H.; Delorme, R.; Benabou, M.; Lemière, N.; Murray, K.; Amsellem, F.; Callebert, J.; Chaste, P.; Jamain, S.; et al. Disruption of melatonin synthesis is associated with impaired 14-3-3 and miR-451 levels in patients with autism spectrum disorders. Sci. Rep. 2017, 7, 2096. [Google Scholar] [CrossRef]
- Guo, Y.; Ni, J.; Chen, S.; Bai, M.; Lin, J.; Ding, G.; Zhang, Y.; Sun, P.; Jia, Z.; Huang, S.; et al. MicroRNA-709 Mediates Acute Tubular Injury through Effects on Mitochondrial Function. J. Am. Soc. Nephrol. 2018, 29, 449–461. [Google Scholar] [CrossRef]
- Surendran, S.; Jideonwo, V.N.; Merchun, C.; Ahn, M.; Murray, J.; Ryan, J.; Dunn, K.W.; Kota, J.; Morral, N. Gene targets of mouse miR-709: Regulation of distinct pools. Sci. Rep. 2016, 6, 18958. [Google Scholar] [CrossRef]
- Maes, O.C.; An, J.; Sarojini, H.; Wang, E. Murine microRNAs implicated in liver functions and aging process. Mech. Ageing Dev. 2008, 129, 534–541. [Google Scholar] [CrossRef]
- Liu, J.; Fan, Y.; Ling-Li, Z.; Liu, B.; Ju, Y.; Wang, M.; Dong, Q.; Lu, X.; Sun, J.; Zhang, L.; et al. The neuroprogressive nature of major depressive disorder: Evidence from an intrinsic connectome analysis. Transl. Psychiatry 2021, 11, 102. [Google Scholar] [CrossRef]
- Araque, A.; Parpura, V.; Sanzgiri, R.P.; Haydon, P.G. Tripartite synapses: Glia, the unacknowledged partner. Trends Neurosci. 1999, 22, 208–215. [Google Scholar] [CrossRef]
- Priya, R.; Brutkiewicz, R.R. Brain astrocytes and microglia express functional MR1 molecules that present microbial antigens to mucosal-associated invariant T (MAIT) cells. J. Neuroimmunol. 2020, 349, 577428. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Quadri, Z.; Crivelli, S.M.; Elsherbini, A.; Zhang, L.; Tripathi, P.; Qin, H.; Roush, E.; Spassieva, S.D.; Nikolova-Karakashian, M.; et al. Neutral Sphingomyelinase 2 Mediates Oxidative Stress Effects on Astrocyte Senescence and Synaptic Plasticity Transcripts. Mol. Neurobiol. 2022, 59, 3233–3253. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Zhong, S.; Deng, Y.; Yao, X.; Liu, Q.; Wang, J.Z.; Xiao, S. HDAC7 Activates IKK/NF-κB Signaling to Regulate Astrocyte-Mediated Inflammation. Mol. Neurobiol. 2022, 59, 6141–6157. [Google Scholar] [CrossRef]
- Zhang, X.C.; Liang, H.F.; Luo, X.D.; Wang, H.J.; Gu, A.P.; Zheng, C.Y.; Su, Q.Z.; Cai, J. YY1 promotes IL-6 expression in LPS-stimulated BV2 microglial cells by interacting with p65 to promote transcriptional activation of IL-6. Biochem. Biophys. Res. Commun. 2018, 502, 269–275. [Google Scholar] [CrossRef]
- Bernard, M.; Voisin, P. Photoreceptor-specific expression, light-dependent localization, and transcriptional targets of the zinc-finger protein Yin Yang 1 in the chicken retina. J. Neurochem. 2008, 105, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Deng, Y.; Gao, J.M.; Lv, C.; Lang, L.H.; Shi, J.S.; Yu, C.Y.; Gong, Q.H. Icariside II inhibits lipopolysaccharide-induced inflammation and amyloid production in rat astrocytes by regulating IKK/IκB/NF-κB/BACE1 signaling pathway. Acta Pharm. Sin. 2020, 41, 154–162. [Google Scholar] [CrossRef]
- Zhai, K.; Huang, Z.; Huang, Q.; Tao, W.; Fang, X.; Zhang, A.; Li, X.; Stark, G.R.; Hamilton, T.A.; Bao, S. Pharmacological inhibition of BACE1 suppresses glioblastoma growth by stimulating macrophage phagocytosis of tumor cells. Nat. Cancer 2021, 2, 1136–1151. [Google Scholar] [CrossRef]
- Sayad, A.; Najafi, S.; Hussen, B.M.; Abdullah, S.T.; Movahedpour, A.; Taheri, M.; Hajiesmaeili, M. The Emerging Roles of the β-Secretase BACE1 and the Long Non-coding RNA BACE1-AS in Human Diseases: A Focus on Neurodegenerative Diseases and Cancer. Front. Aging Neurosci. 2022, 14, 853180. [Google Scholar] [CrossRef]
- Nunes, P.V.; Suemoto, C.K.; Rodriguez, R.D.; Paraizo Leite, R.E.; Nascimento, C.; Pasqualucci, C.A.; Nitrini, R.; Jacob-Filho, W.; Grinberg, L.T.; Lafer, B. Neuropathology of depression in non-demented older adults: A large postmortem study of 741 individuals. Neurobiol. Aging. 2022, 117, 107–116. [Google Scholar] [CrossRef]
- Liu, Y.J.; Zhuang, J.; Zhu, H.Y.; Shen, Y.X.; Tan, Z.L.; Zhou, J.N. Cultured rat cortical astrocytes synthesize melatonin: Absence of a diurnal rhythm. J. Pineal Res. 2007, 43, 232–238. [Google Scholar] [CrossRef]
- Markus, R.P.; Fernandes, P.A.; Kinker, G.S.; da Silveira Cruz-Machado, S.; Marçola, M. Immune-pineal axis—Acute inflammatory responses coordinate melatonin synthesis by pinealocytes and phagocytes. Br. J. Pharmacol. 2018, 175, 3239–3250. [Google Scholar] [CrossRef] [PubMed]
- Muxel, S.M.; Pires-Lapa, M.A.; Monteiro, A.W.; Cecon, E.; Tamura, E.K.; Floeter-Winter, L.M.; Markus, R.P. NF-κB drives the synthesis of melatonin in RAW 264.7 macrophages by inducing the transcription of the arylalkylamine-N-acetyltransferase (AA-NAT) gene. PLoS ONE 2012, 7, e52010. [Google Scholar] [CrossRef] [PubMed]
- Lwin, T.; Yang, J.L.; Ngampramuan, S.; Viwatpinyo, K.; Chancharoen, P.; Veschsanit, N.; Pinyomahakul, J.; Govitrapong, P.; Mukda, S. Melatonin ameliorates methamphetamine-induced cognitive impairments by inhibiting neuroinflammation via suppression of the TLR4/MyD88/NFκB signaling pathway in the mouse hippocampus. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 111, 110109. [Google Scholar] [CrossRef]
- Cucielo, M.S.; Cesário, R.C.; Silveira, H.S.; Gaiotte, L.B.; Dos Santos, S.A.A.; de Campos Zuccari, D.A.P.; Seiva, F.R.F.; Reiter, R.J.; de Almeida Chuffa, L.G. Melatonin Reverses the Warburg-Type Metabolism and Reduces Mitochondrial Membrane Potential of Ovarian Cancer Cells Independent of MT1 Receptor Activation. Molecules 2022, 27, 4350. [Google Scholar] [CrossRef] [PubMed]
- Pajarillo, E.; Nyarko-Danquah, I.; Digman, A.; Vied, C.; Son, D.S.; Lee, J.; Aschner, M.; Lee, E. Astrocytic Yin Yang 1 is critical for murine brain development and protection against apoptosis, oxidative stress, and inflammation. Glia 2022, 27, 24286. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, X.; Zheng, L.; Li, D.; Liu, Z.; Teng, L. The lncRNA NEAT1 Inhibits miRNA-216b and Promotes Colorectal Cancer Progression by Indirectly Activating YY1. J. Oncol. 2022, 2022, 8130132. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Sharma, R.; Rosales-Corral, S.; de Campos Zuccari, D.A.P.; de Almeida Chuffa, L.G. Melatonin: A mitochondrial resident with a diverse skill set. Life Sci. 2022, 301, 120612. [Google Scholar] [CrossRef]
- Tan, D.; and Reiter, R.J. Mitochondria: The birth place, battle ground and the site of melatonin metabolism in cells. Melatonin Res. 2019, 2, 44–66. [Google Scholar] [CrossRef]
- Winge, I.; McKinney, J.A.; Ying, M.; D’Santos, C.S.; Kleppe, R.; Knappskog, P.M.; Haavik, J. Activation and stabilization of human tryptophan hydroxylase 2 by phosphorylation and 14-3-3 binding. Biochem. J. 2008, 410, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Alaiyed, S.; McCann, M.; Mahajan, G.; Rajkowska, G.; Stockmeier, C.A.; Kellar, K.J.; Wu, J.Y.; Conant, K. Venlafaxine Stimulates an MMP-9-Dependent Increase in Excitatory/Inhibitory Balance in a Stress Model of Depression. J. Neurosci. 2020, 40, 4418–4431. [Google Scholar] [CrossRef]
- Pietropaolo, S.; Provenzano, G. Editorial: Targeting Excitation-Inhibition Imbalance in Neurodevelopmental and Autism Spectrum Disorders. Front. Neurosci. 2022, 16, 968115. [Google Scholar] [CrossRef] [PubMed]
- Foliaki, S.T.; Schwarz, B.; Groveman, B.R.; Walters, R.O.; Ferreira, N.C.; Orrù, C.D.; Smith, A.; Wood, A.; Schmit, O.M.; Freitag, P.; et al. Neuronal excitatory-to-inhibitory balance is altered in cerebral organoid models of genetic neurological diseases. Mol. Brain 2021, 14, 156. [Google Scholar] [CrossRef] [PubMed]
- Kiernan, M.C.; Ziemann, U.; Eisen, A. Amyotrophic lateral sclerosis: Origins traced to impaired balance between neural excitation and inhibition in the neonatal period. Muscle Nerve. 2019, 60, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Kehrer, C.; Maziashvili, N.; Dugladze, T.; Gloveli, T. Altered Excitatory-Inhibitory Balance in the NMDA-Hypofunction Model of Schizophrenia. Front. Mol. Neurosci. 2008, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Lauden, A.; Geishin, A.; Merzon, E.; Korobeinikov, A.; Green, I.; Golan-Cohen, A.; Vinker, S.; Manor, I.; Weizman, A.; Magen, E. Higher rates of allergies, autoimmune diseases and low-grade inflammation markers in treatment-resistant major depression. Brain Behav. Immun. Health 2021, 16, 100313. [Google Scholar] [CrossRef] [PubMed]
- Euesden, J.; Danese, A.; Lewis, C.M.; Maughan, B. A bidirectional relationship between depression and the autoimmune disorders—New perspectives from the National Child Development Study. PLoS ONE 2017, 12, e0173015. [Google Scholar] [CrossRef]
- Yang, H.; Liu, D.; Zhao, C.; Feng, B.; Lu, W.; Yang, X.; Xu, M.; Zhou, W.; Jing, H.; Yang, J. Mendelian randomization integrating GWAS and eQTL data revealed genes pleiotropically associated with major depressive disorder. Transl. Psychiatry 2021, 11, 225. [Google Scholar] [CrossRef]
- Howard, D.M.; Adams, M.J.; Clarke, T.K.; Hafferty, J.D.; Gibson, J.; Shirali, M.; Coleman, J.R.I.; Hagenaars, S.P.; Ward, J.; Wigmore, E.M.; et al. Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nat. Neurosci. 2019, 22, 343–352. [Google Scholar] [CrossRef]
- Nagel, M.; Jansen, P.R.; Stringer, S.; Watanabe, K.; de Leeuw, C.A.; Bryois, J.; Savage, J.E.; Hammerschlag, A.R.; Skene, N.G.; Muñoz-Manchado, A.B.; et al. Meta-analysis of genome-wide association studies for neuroticism in 449,484 individuals identifies novel genetic loci and pathways. Nat. Genet. 2018, 50, 920–927. [Google Scholar] [CrossRef]
- Wang, B.Y.; Ye, Y.Y.; Qian, C.; Zhang, H.B.; Mao, H.X.; Yao, L.P.; Sun, X.; Lu, G.H.; Zhang, S.Z. Stress increases MHC-I expression in dopaminergic neurons and induces autoimmune activation in Parkinson’s disease. Neural Regen. Res. 2021, 16, 2521–2527. [Google Scholar] [CrossRef]
- Agnihotri, S.K.; Shen, R.; Li, J.; Gao, X.; Büeler, H. Loss of PINK1 leads to metabolic deficits in adult neural stem cells and impedes differentiation of newborn neurons in the mouse hippocampus. FASEB J. 2017, 31, 2839–2853. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yan, J.; Sun, C.; Li, G.; Li, S.; Zhang, L.; Di, C.; Gan, L.; Wang, Y.; Zhou, R.; et al. Ameliorating mitochondrial dysfunction restores carbon ion-induced cognitive deficits via co-activation of NRF2 and PINK1 signaling pathway. Redox Biol. 2018, 17, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Gautier, C.A.; Kitada, T.; Shen, J. Loss of PINK1 causes mitochondrial functional defects and increased sensitivity to oxidative stress. Proc. Natl. Acad. Sci. USA 2008, 105, 11364–11369. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, R.S.; Bombeiro, A.L.; Chiarotto, G.B.; Cartarozzi, L.P.; Coser, L.O.; Ghizoni, E.; Tedeschi, H.; Cendes, F.; Lopes-Cendes, I.; Rogerio, F.; et al. Interferon-beta induces major histocompatibility complex of class I (MHC-I) expression and a proinflammatory phenotype in cultivated human astrocytes. Differentiation 2022, 128, 43–56. [Google Scholar] [CrossRef]
- Lemaître, F.; Farzam-Kia, N.; Carmena Moratalla, A.; Carpentier Solorio, Y.; Clenet, M.L.; Tastet, O.; Cleret-Buhot, A.; Guimond, J.V.; Haddad, E.; Duquette, P.; et al. IL-27 shapes the immune properties of human astrocytes and their impact on encountered human T lymphocytes. J. Neuroinflammation 2022, 19, 212. [Google Scholar] [CrossRef]
- Pozuelo-Rubio, M. Proteomic and biochemical analysis of 14-3-3-binding proteins during C2-ceramide-induced apoptosis. FEBS J. 2010, 277, 3321–3342. [Google Scholar] [CrossRef]
- Pozuelo-Rubio, M. Regulation of autophagic activity by 14-3-3ζ proteins associated with class III phosphatidylinositol-3-kinase. Cell Death Differ. 2011, 18, 479–492. [Google Scholar] [CrossRef]
- Cheng, B.; Du, M.; He, S.; Yang, L.; Wang, X.; Gao, H.; Chang, H.; Gao, W.; Li, Y.; Wang, Q.; et al. Inhibition of platelet activation suppresses reactive enteric glia and mitigates intestinal barrier dysfunction during sepsis. Mol. Med. 2022, 28, 127. [Google Scholar] [CrossRef]
- Gałecki, P.; Talarowska, M.; Anderson, G.; Berk, M.; Maes, M. Mechanisms underlying neurocognitive dysfunctions in recurrent major depression. Med. Sci. Monit. 2015, 21, 1535–1547. [Google Scholar] [CrossRef]
- Anderson, G.; Maes, M. Local melatonin regulates inflammation resolution: A common factor in neurodegenerative, psychiatric and systemic inflammatory disorders. CNS Neurol. Disord. Drug Targets 2014, 13, 817–827. [Google Scholar] [CrossRef]
- Sommansson, A.; Nylander, O.; Sjöblom, M. Melatonin decreases duodenal epithelial paracellular permeability via a nicotinic receptor-dependent pathway in rats in vivo. J. Pineal Res. 2013, 54, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Kunii, Y.; Zhang, W.; Xu, Q.; Hyde, T.M.; McFadden, W.; Shin, J.H.; Deep-Soboslay, A.; Ye, T.; Li, C.; Kleinman, J.E.; et al. CHRNA7 and CHRFAM7A mRNAs: Co-localized and their expression levels altered in the postmortem dorsolateral prefrontal cortex in major psychiatric disorders. Am. J. Psychiatry 2015, 172, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.; Levi, L.; Park, J.; Nastas, I.; Ford, L.; Rassnick, S.; Canuso, C.; Davis, J.M.; Weiser, M. The effects of JNJ-39393406 a positive allosteric nicotine modulator on mood and cognition in patients with unipolar depression: A double-blind, add-on, placebo-controlled trial. Eur. Neuropsychopharmacol. 2021, 51, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Zhang, W.; Bo, J.; Zhang, Z.; Lei, Y.; Huo, W.; Liu, Y.; Ma, Z.; Gu, X. Spinal activation of alpha7-nicotinic acetylcholine receptor attenuates posttraumatic stress disorder-related chronic pain via suppression of glial activation. Neuroscience 2017, 344, 243–254. [Google Scholar] [CrossRef]
- Fišar, Z.; Hansíková, H.; Křížová, J.; Jirák, R.; Kitzlerová, E.; Zvěřová, M.; Hroudová, J.; Wenchich, L.; Zeman, J.; Raboch, J. Activities of mitochondrial respiratory chain complexes in platelets of patients with Alzheimer’s disease and depressive disorder. Mitochondrion 2019, 48, 67–77. [Google Scholar] [CrossRef]
- Hroudová, J.; Fišar, Z.; Kitzlerová, E.; Zvěřová, M.; Raboch, J. Mitochondrial respiration in blood platelets of depressive patients. Mitochondrion 2013, 13, 795–800. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anderson, G. Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub. Int. J. Mol. Sci. 2023, 24, 350. https://doi.org/10.3390/ijms24010350
Anderson G. Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub. International Journal of Molecular Sciences. 2023; 24(1):350. https://doi.org/10.3390/ijms24010350
Chicago/Turabian StyleAnderson, George. 2023. "Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub" International Journal of Molecular Sciences 24, no. 1: 350. https://doi.org/10.3390/ijms24010350
APA StyleAnderson, G. (2023). Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub. International Journal of Molecular Sciences, 24(1), 350. https://doi.org/10.3390/ijms24010350





