Change in the Electronic Structure of the Cobalt(II) Ion in a One-Dimensional Polymer with Flexible Linkers Induced by a Structural Phase Transition †
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization
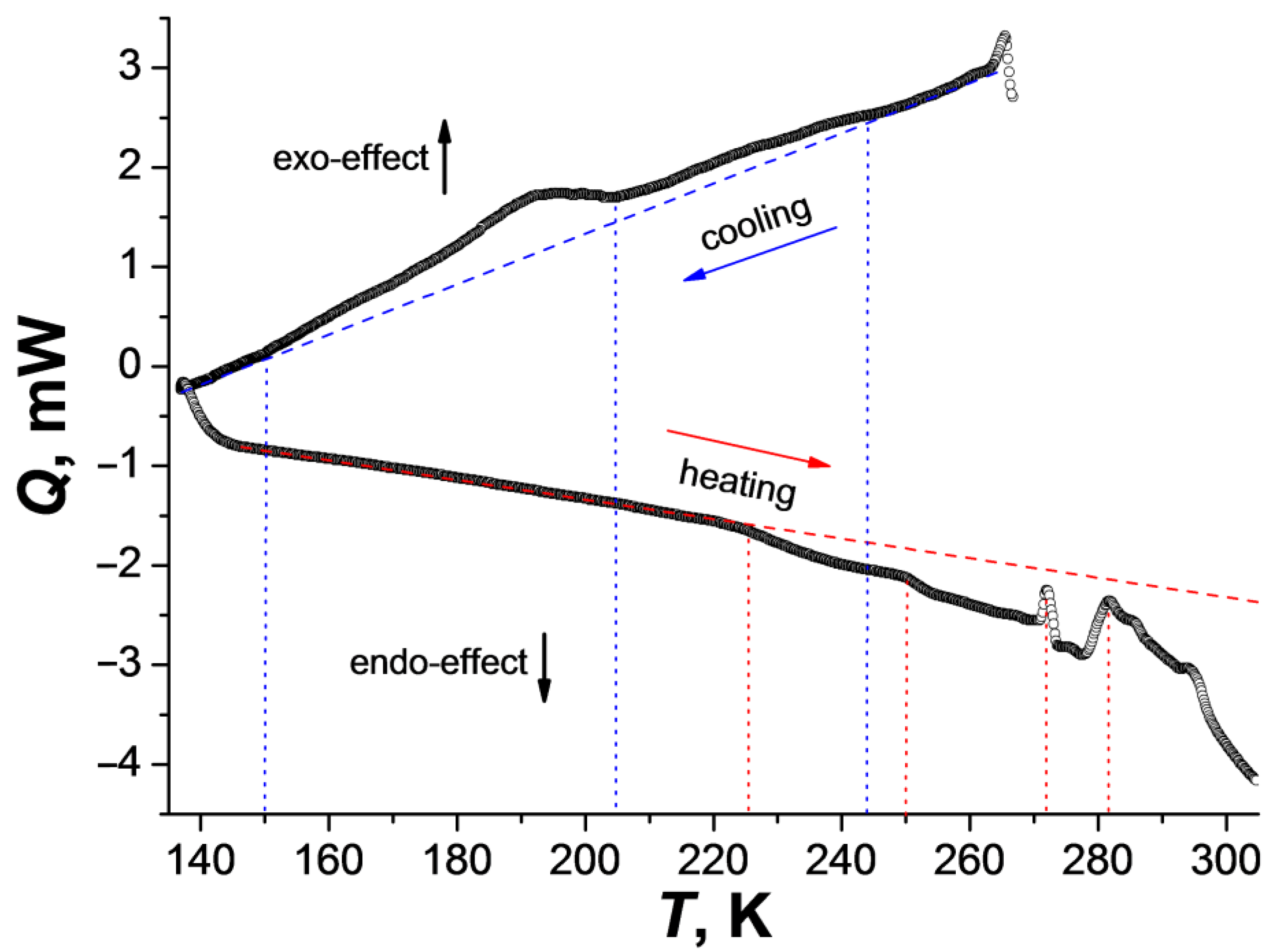
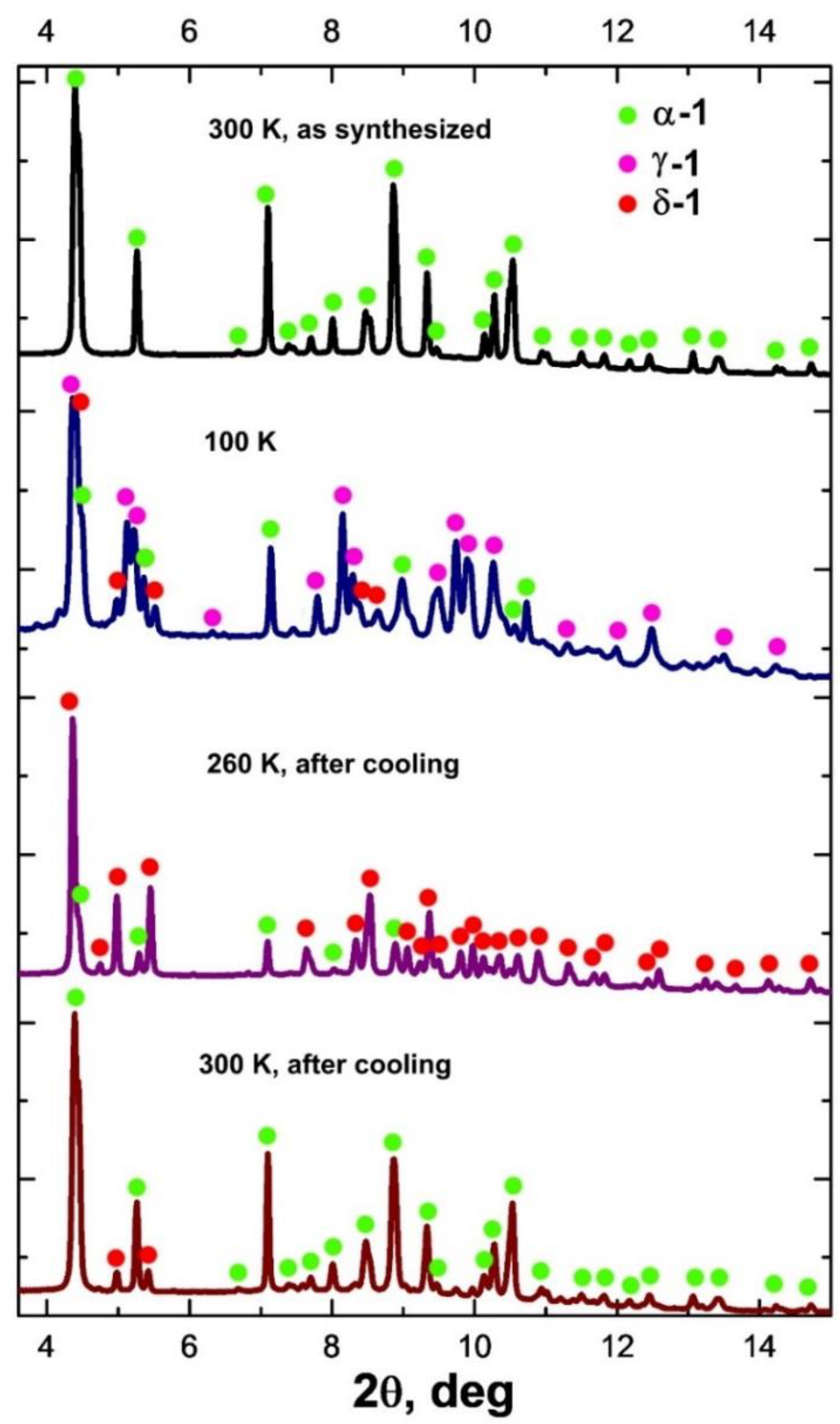
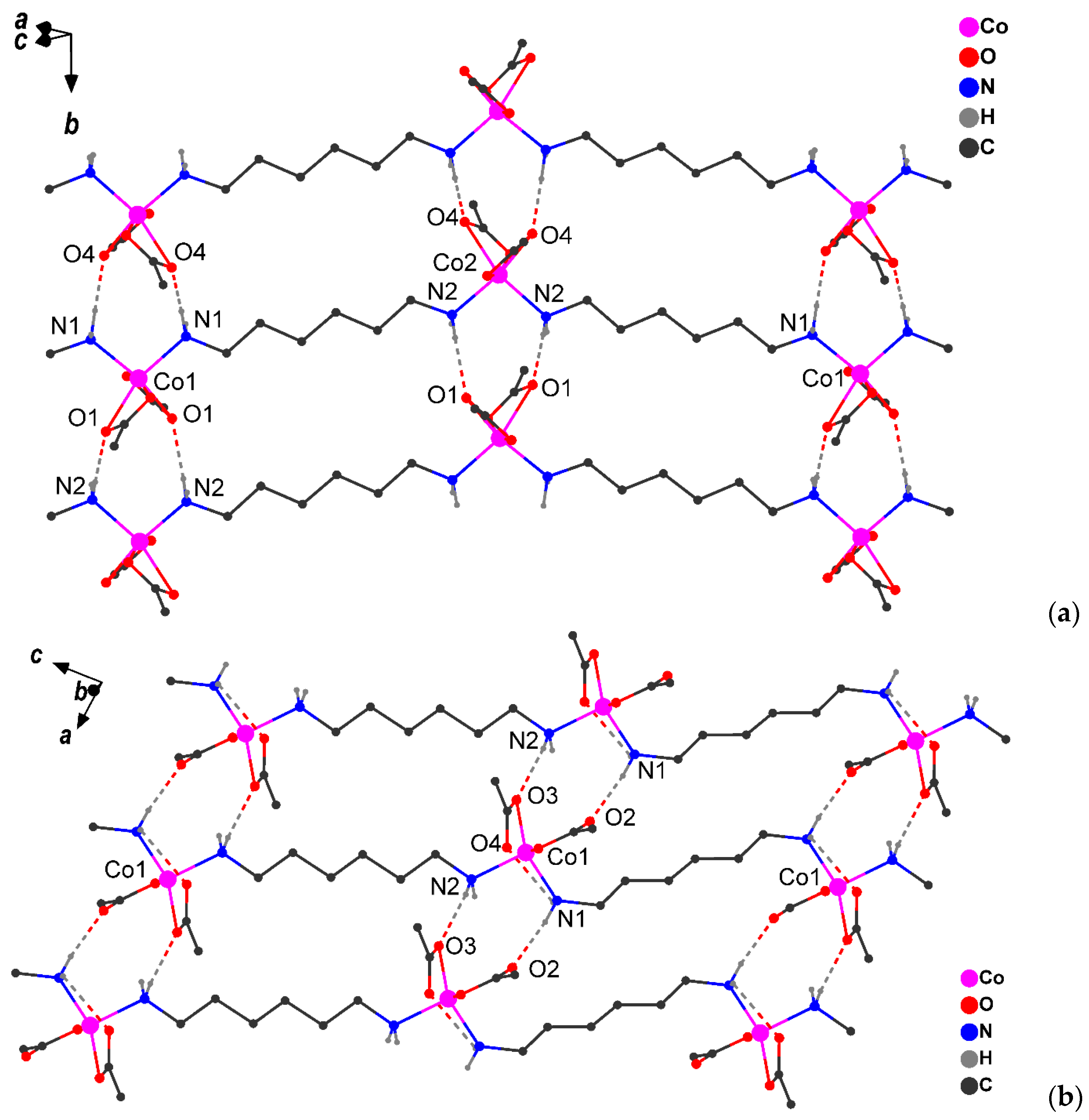
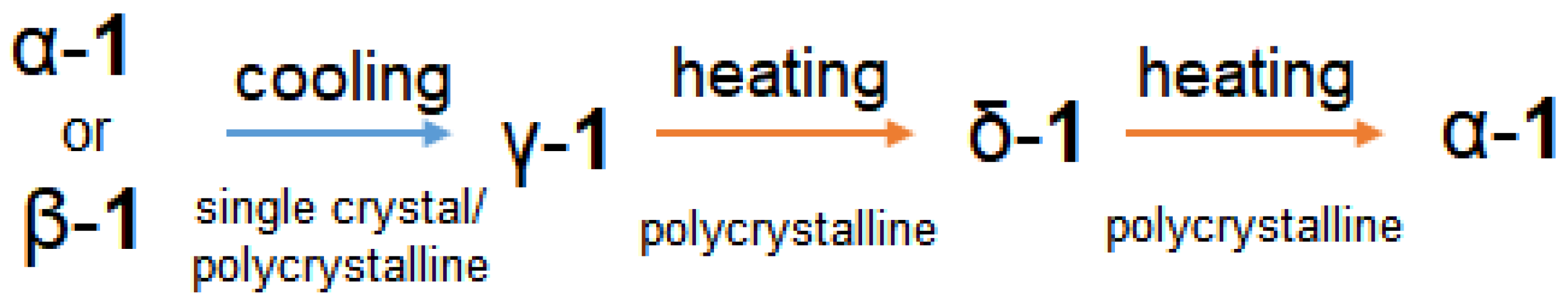
2.2. Diffraction and DSC Studies of Polycrystals and Single Crystals
2.3. Spectral Properties
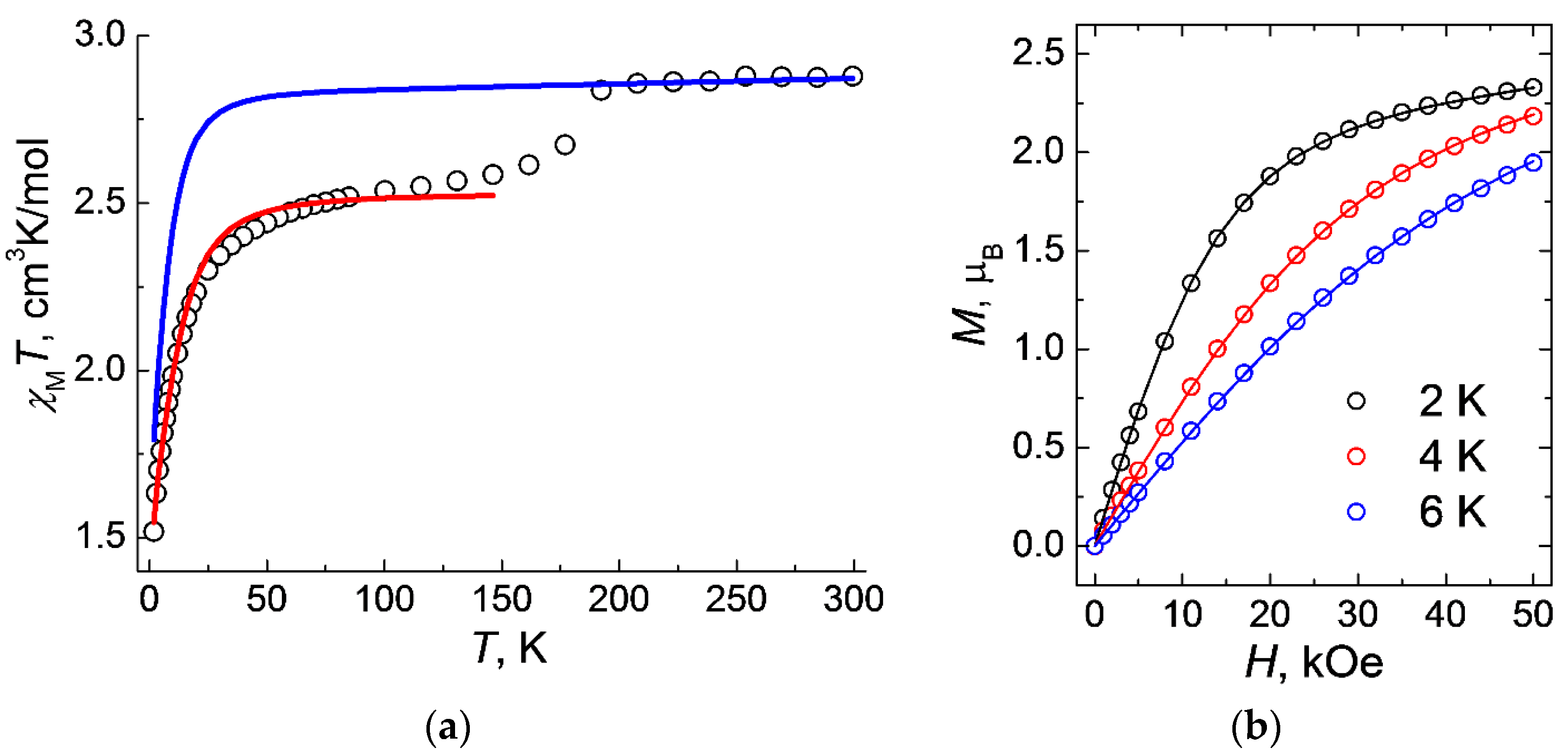
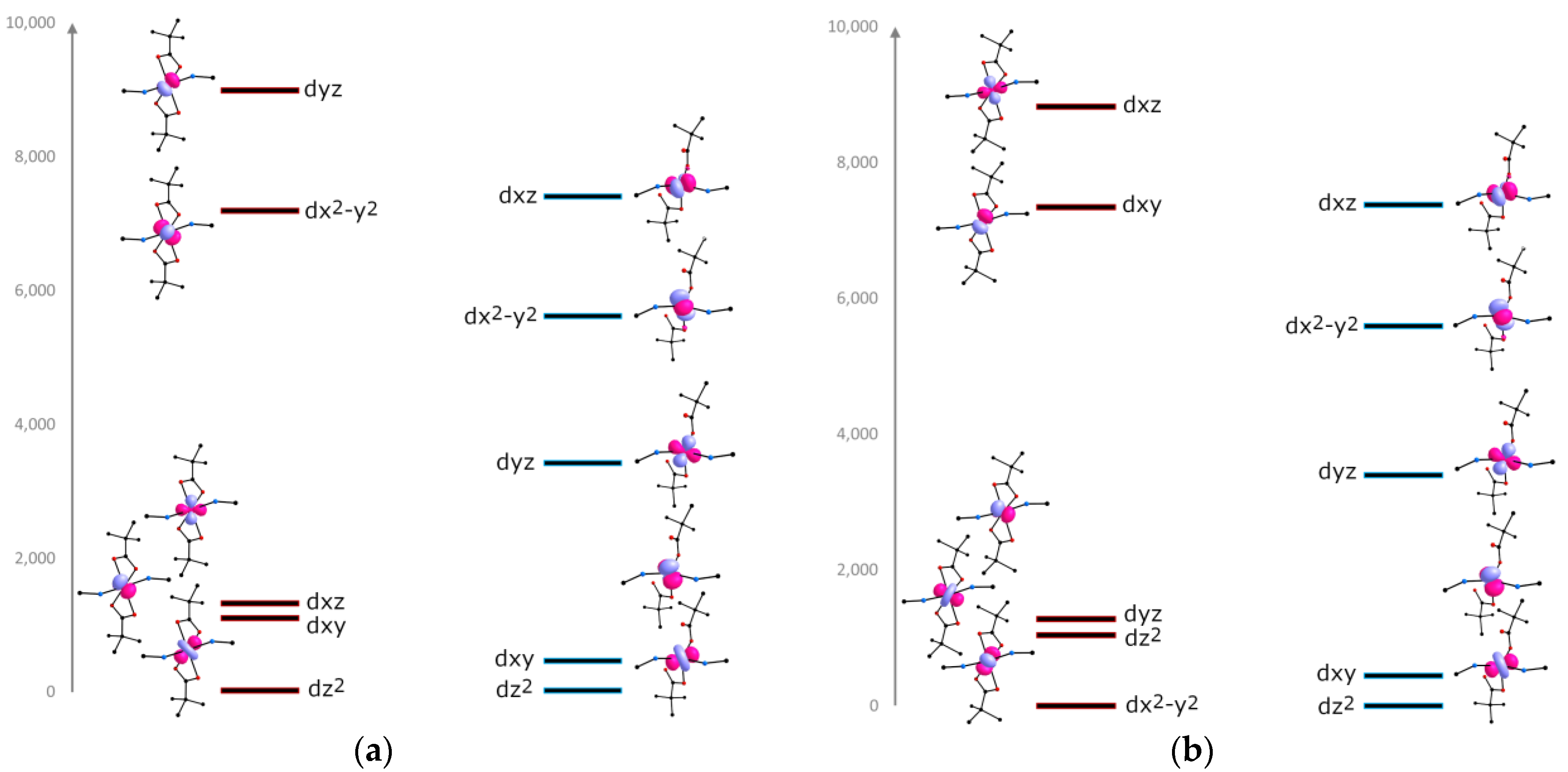
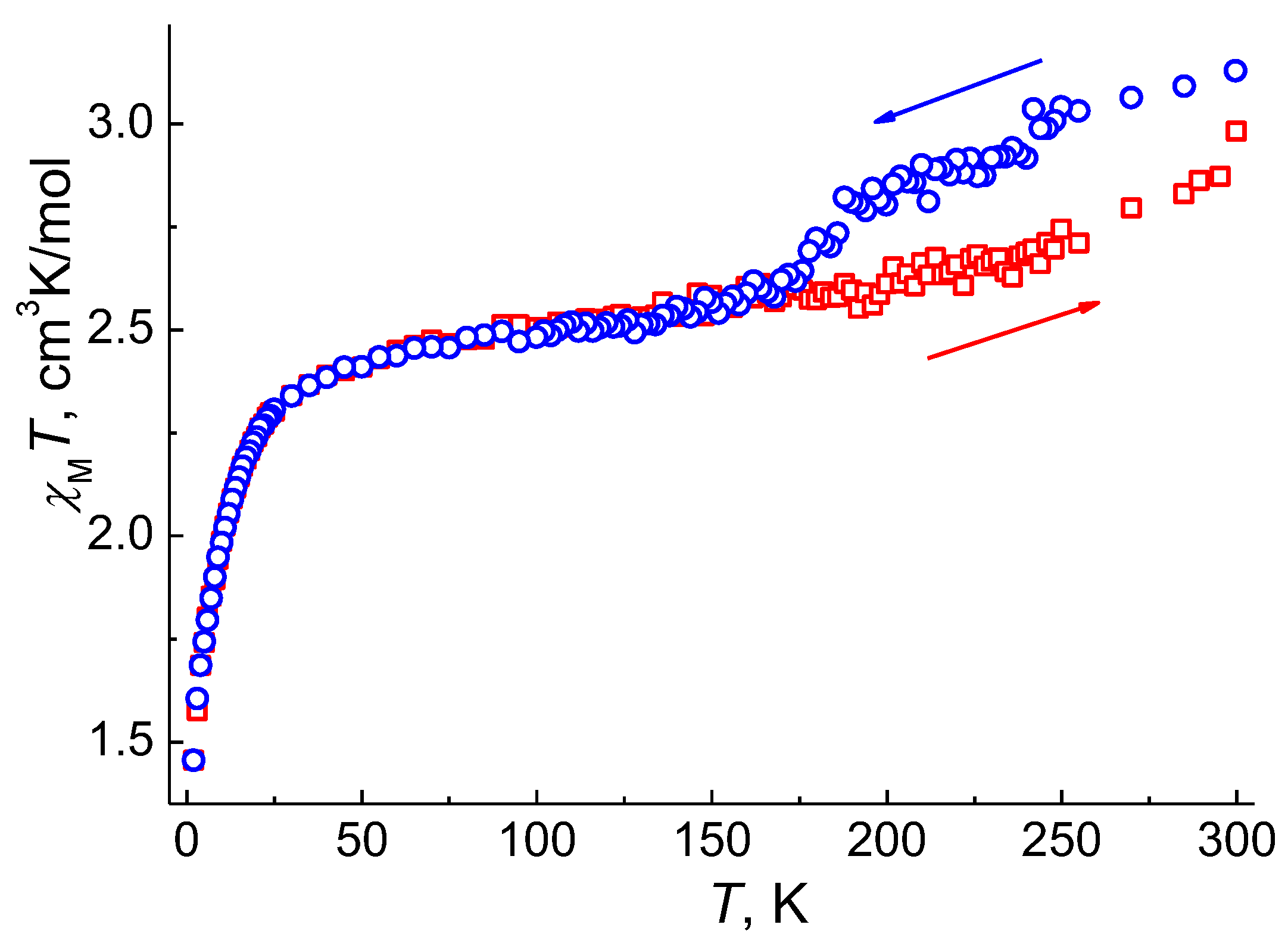
2.4. EPR Spectroscopy, DC Magnetic Data and Theoretical Calculations
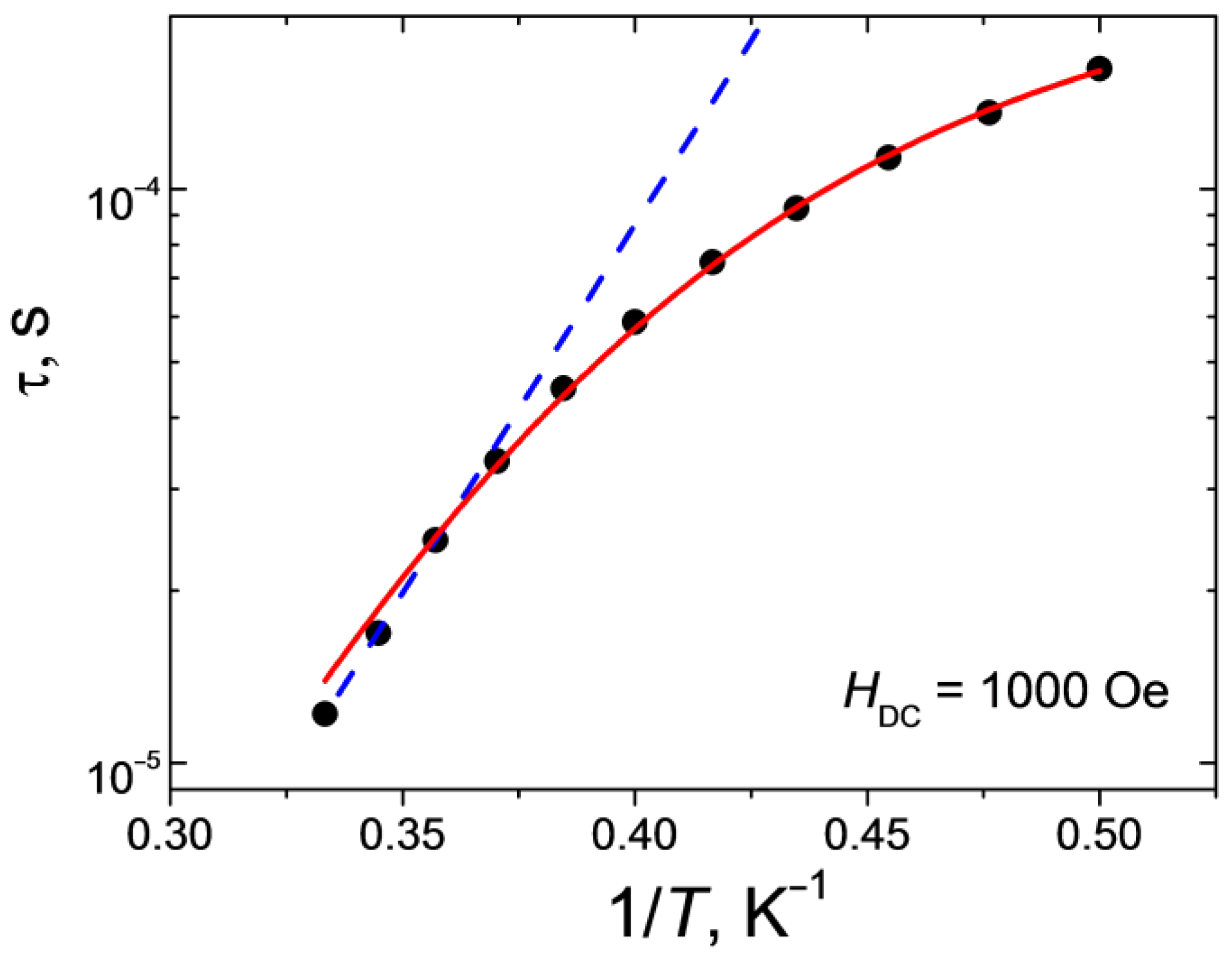
2.5. AC Magnetic Data
2.6. Discussions
2.7. Conclusions
3. Methods and Materials
3.1. Main Methods
3.2. Materials and Synthesis
Synthesis of [Co2(Piv)2(NH2(CH2)6NH2)]n (1)
3.3. Cooling Sample 1
3.4. PXRD
3.5. Single Crystal X-ray
3.6. Quantum Chemical Calculation
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bogani, L.; Wernsdorfer, W. Molecular Spintronics Using Single-Molecule Magnets. Nat. Mater. 2008, 7, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Li, B.; He, H.; Zhou, W.; Chen, B.; Qian, G. Metal–Organic Frameworks as Platforms for Functional Materials. Acc. Chem. Res. 2016, 49, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Westerström, R.; Dreiser, J.; Piamonteze, C.; Muntwiler, M.; Weyeneth, S.; Krämer, K.; Liu, S.-X.; Decurtins, S.; Popov, A.; Yang, S.; et al. Tunneling, Remanence, and Frustration in Dysprosium-Based Endohedral Single-Molecule Magnets. Phys. Rev. B 2014, 89, 60406. [Google Scholar] [CrossRef]
- Sato, O.; Tao, J.; Zhang, Y.-Z. Control of Magnetic Properties through External Stimuli. Angew. Chemie Int. Ed. 2007, 46, 2152–2187. [Google Scholar] [CrossRef] [PubMed]
- Pankratova, Y.; Aleshin, D.; Nikovskiy, I.; Novikov, V.; Nelyubina, Y. In Situ NMR Search for Spin-Crossover in Heteroleptic Cobalt(II) Complexes. Inorg. Chem. 2020, 59, 7700–7709. [Google Scholar] [CrossRef] [PubMed]
- Akiyoshi, R.; Ohtani, R.; Lindoy, L.F.; Hayami, S. Spin Crossover Phenomena in Long Chain Alkylated Complexes. Dalton Trans. 2021, 50, 5065–5079. [Google Scholar] [CrossRef]
- Hayami, S.; Holmes, S.M.; Halcrow, M.A. Spin-State Switches in Molecular Materials Chemistry. J. Mater. Chem. C 2015, 3, 7775–7778. [Google Scholar] [CrossRef]
- Tezgerevska, T.; Alley, K.G.; Boskovic, C. Valence Tautomerism in Metal Complexes: Stimulated and Reversible Intramolecular Electron Transfer between Metal Centers and Organic Ligands. Coord. Chem. Rev. 2014, 268, 23–40. [Google Scholar] [CrossRef]
- Aquino, M.A.S.; Lee, F.L.; Gabe, E.J.; Bensimon, C.; Greedan, J.E.; Crutchley, R.J. Superexchange Metal-Metal Coupling in Dinuclear Pentaammineruthenium Complexes Incorporating a 1,4-Dicyanamidobenzene Dianion Bridging Ligand. J. Am. Chem. Soc. 1992, 114, 5130–5140. [Google Scholar] [CrossRef]
- Sessoli, R.; Gatteschi, D.; Caneschi, A.; Novak, M.A. Magnetic Bistability in a Metal-Ion Cluster. Nature 1993, 365, 141–143. [Google Scholar] [CrossRef]
- Gütlich, P.; Goodwin, H.A. (Eds.) Spin Crossover in Transition Metal Compounds, I–III; Topics in Current Chemistry; Springer: Berlin/Heidelberg, Germany, 2004; Volume 233–235, ISBN 978-3-642-07326-7. [Google Scholar]
- Gaspar, A.B.; Seredyuk, M. Spin Crossover in Soft Matter. Coord. Chem. Rev. 2014, 268, 41–58. [Google Scholar] [CrossRef]
- Frost, M.J.; Harriman, K.; Murugesu, M. The Rise of 3-d Single-Ion Magnets in Molecular Magnetism: Towards Materials from Molecules? Chem. Sci. 2016, 7, 2470–2491. [Google Scholar] [CrossRef] [PubMed]
- Gatteschi, D.; Sessoli, R.; Villain, J. Molecular Nanomagnets; Oxford University Press on Demand: Oxford, UK, 2006; Volume 5, ISBN 0198567537. [Google Scholar]
- Winpenny, R.E.P. (Ed.) Single-Molecule Magnets and Related Phenomena; Springer: New York, NY, USA, 2006; ISBN 3540332391. [Google Scholar]
- Zenno, H.; Akiyoshi, R.; Nakamura, M.; Sekine, Y.; Hayami, S. Crystal Structures and Spin Crossover of Iron(III) Cocrystal Formed via Halogen Bonding. Chem. Lett. 2021, 50, 1259–1262. [Google Scholar] [CrossRef]
- Pfrunder, M.C.; Whittaker, J.J.; Parsons, S.; Moubaraki, B.; Murray, K.S.; Moggach, S.A.; Sharma, N.; Micallef, A.S.; Clegg, J.K.; McMurtrie, J.C. Controlling Spin Switching with Anionic Supramolecular Frameworks. Chem. Mater. 2020, 32, 3229–3234. [Google Scholar] [CrossRef]
- Murata, S.; Takahashi, K.; Mochida, T.; Sakurai, T.; Ohta, H.; Yamamoto, T.; Einaga, Y. Cooperative Spin-Crossover Transition from Three-Dimensional Purely π-Stacking Interactions in a Neutral Heteroleptic Azobisphenolate FeIII Complex with a N3O3 Coordination Sphere. Dalton Trans. 2017, 46, 5786–5789. [Google Scholar] [CrossRef]
- Milocco, F.; de Vries, F.; Siebe, H.S.; Engbers, S.; Demeshko, S.; Meyer, F.; Otten, E. Widening the Window of Spin-Crossover Temperatures in Bis(Formazanate)Iron(II) Complexes via Steric and Noncovalent Interactions. Inorg. Chem. 2021, 60, 2045–2055. [Google Scholar] [CrossRef]
- Zenno, H.; Kobayashi, F.; Nakamura, M.; Sekine, Y.; Lindoy, L.F.; Hayami, S. Hydrogen Bond-Induced Abrupt Spin Crossover Behaviour in 1-D Cobalt(II) Complexes—The Key Role of Solvate Water Molecules. Dalton Trans. 2021, 50, 7843–7853. [Google Scholar] [CrossRef]
- Kanetomo, T.; Ni, Z.; Enomoto, M. Hydrogen-Bonded Cobalt(II)-Organic Framework: Normal and Reverse Spin-Crossover Behaviours. Dalton Trans. 2022, 51, 5034–5040. [Google Scholar] [CrossRef]
- Juhász, G.; Matsuda, R.; Kanegawa, S.; Inoue, K.; Sato, O.; Yoshizawa, K. Bistability of Magnetization without Spin-Transition in a High-Spin Cobalt(II) Complex Due to Angular Momentum Quenching. J. Am. Chem. Soc. 2009, 131, 4560–4561. [Google Scholar] [CrossRef]
- Su, S.-Q.; Wu, S.-Q.; Baker, M.L.; Bencok, P.; Azuma, N.; Miyazaki, Y.; Nakano, M.; Kang, S.; Shiota, Y.; Yoshizawa, K.; et al. Quenching and Restoration of Orbital Angular Momentum through a Dynamic Bond in a Cobalt(II) Complex. J. Am. Chem. Soc. 2020, 142, 11434–11441. [Google Scholar] [CrossRef]
- Baran, E.J.; Piro, O.E.; Zinczuk, J. A New Supramolecular Assembly Obtained by Reaction Between Thiosaccharin and Hexamethylenediamine. Z. Nat. B 2007, 62, 1530–1534. [Google Scholar] [CrossRef][Green Version]
- Stewart, J.E. Vibrational Spectra of Primary and Secondary Aliphatic Amines. J. Chem. Phys. 1959, 30, 1259–1265. [Google Scholar] [CrossRef]
- Nakamoto, K. Recent Progress in Inorganic Vibrational Spectroscopy. J. Spectrosc. Soc. Japan 1981, 30, 437–454. [Google Scholar] [CrossRef]
- Ghosh, I.; Chakraborty, B.; Bera, A.; Paul, S.; Paine, T.K. Selective Oxygenation of C–H and C=C Bonds with H2O2 by High-Spin Cobalt(II)-Carboxylate Complexes. Dalton Trans. 2022, 51, 2480–2492. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, L.-Y.; Bao, Y.-M.; Zhang, N.; Zhang, J.-Y.; Xing, Y.-Y.; Deng, W.; Liu, Z.-J. Structures and Catalytic Oxidative Coupling Reaction of Four Co-MOFs Modified with R-Isophthalic Acid (R-H, OH and COOH) and Trigonal Ligands. CrystEngComm 2021, 23, 7590–7601. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, J.; Sun, H.X.; He, M.; Li, W.; Du, L.; Xie, M.; Zhao, Q.-H. R-Substituent-Induced Structural Diversity and Single-Crystal to Single-Crystal Transformation of Coordination Polymers: Synthesis, Luminescence, and Magnetic Behaviors. Cryst. Growth Des. 2021, 21, 5086–5099. [Google Scholar] [CrossRef]
- Nikolić, M.A.; Szécsényi, K.M.; Dražić, B.; Rodić, M.V.; Stanić, V.; Tanasković, S. Binuclear Co(II) Complexes with Macrocycle and Carboxylato Ligands: Structure, Cytotoxicity and Thermal Behavior. J. Mol. Struct. 2021, 1236, 130133. [Google Scholar] [CrossRef]
- Chen, K.-J.; Madden, D.G.; Pham, T.; Forrest, K.A.; Kumar, A.; Yang, Q.-Y.; Xue, W.; Space, B.; Perry IV, J.J.; Zhang, J.-P.; et al. Tuning Pore Size in Square-Lattice Coordination Networks for Size-Selective Sieving of CO2. Angew. Chemie Int. Ed. 2016, 55, 10268–10272. [Google Scholar] [CrossRef]
- White, A.H.; Willis, A.C. Structural Studies in Metal–Purpurate Complexes. Part 7. Crystal Structures of Diaquanitratopurpurato-Cobalt(II) and -Zinc(II) Dihydrat. J. Chem. Soc. Dalt. Trans. 1977, 14, 1377–1381. [Google Scholar] [CrossRef]
- Głowiak, T.; Kurdziel, K. Crystal Structure and Physico-Chemical Properties of Hepta-Coordinate Isomorphic Cobalt(II) and Nickel(II) Complexes of 1-Allylimidazole. J. Mol. Struct. 2000, 516, 1–5. [Google Scholar] [CrossRef]
- Pérez-Lourido, P.; Madarasi, E.; Antal, F.; Esteban-Gómez, D.; Wang, G.; Angelovski, G.; Platas-Iglesias, C.; Tircsó, G.; Valencia, L. Stable and Inert Macrocyclic Cobalt(Ii) and Nickel(Ii) Complexes with ParaCEST Response. Dalton Trans. 2022, 51, 1580–1593. [Google Scholar] [CrossRef] [PubMed]
- Stoll, S.; Schweiger, A. EasySpin, a Comprehensive Software Package for Spectral Simulation and Analysis in EPR. J. Magn. Reson. 2006, 178, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Polunin, R.A.; Kolotilov, S.V.; Kiskin, M.A.; Cador, O.; Mikhalyova, E.A.; Lytvynenko, A.S.; Golhen, S.; Ouahab, L.; Ovcharenko, V.I.; Eremenko, I.L.; et al. Topology Control of Porous Coordination Polymers by Building Block Symmetry. Eur. J. Inorg. Chem. 2010, 2010, 5055–5057. [Google Scholar] [CrossRef]
- Litvinenko, A.S.; Mikhaleva, E.A.; Kolotilov, S.V.; Pavlishchuk, V.V. Effect of Spin–Orbit Coupling on the Magnetic Susceptibility of Polynuclear Complexes of 3d Metals Containing a Co2+ Ion. Theor. Exp. Chem. 2011, 46, 422–428. [Google Scholar] [CrossRef]
- Chilton, N.F.; Anderson, R.P.; Turner, L.D.; Soncini, A.; Murray, K.S. PHI: A Powerful New Program for the Analysis of Anisotropic Monomeric and Exchange-Coupled Polynuclear d- and f-Block Complexes. J. Comput. Chem. 2013, 34, 1164–1175. [Google Scholar] [CrossRef] [PubMed]
- Griffith, J.S. The Theory of Transition-Metal Ions; Cambridge University Press: Cambridge, UK, 1964; ISBN 0521051509. [Google Scholar]
- Lloret, F.; Julve, M.; Cano, J.; Ruiz-García, R.; Pardo, E. Magnetic Properties of Six-Coordinated High-Spin Cobalt(II) Complexes: Theoretical Background and Its Application. Inorg. Chim. Acta 2008, 361, 3432–3445. [Google Scholar] [CrossRef]
- Palii, A.V.; Korchagin, D.V.; Yureva, E.A.; Akimov, A.V.; Misochko, E.Y.; Shilov, G.V.; Talantsev, A.D.; Morgunov, R.B.; Aldoshin, S.M.; Tsukerblat, B.S. Single-Ion Magnet Et4N[CoII(Hfac)3] with Nonuniaxial Anisotropy: Synthesis, Experimental Characterization, and Theoretical Modeling. Inorg. Chem. 2016, 55, 9696–9706. [Google Scholar] [CrossRef]
- Korchagin, D.V.; Gureev, Y.E.; Yureva, E.A.; Shilov, G.V.; Akimov, A.V.; Misochko, E.Y.; Morgunov, R.B.; Zakharov, K.V.; Vasiliev, A.N.; Palii, A.V.; et al. Field-induced single-ion magnet based on a quasi-octahedral Co(II) complex with mixed sulfur–oxygen coordination environment. Dalton Trans. 2021, 50, 13815–13822. [Google Scholar] [CrossRef]
- Palii, A.; Tsukerblat, B.; Klokishner, S.; Dunbar, K.R.; Clemente-Juan, J.M.; Coronado, E. Beyond the Spin Model: Exchange Coupling in Molecular Magnets with Unquenched Orbital Angular Momenta. Chem. Soc. Rev. 2011, 40, 3130–3156. [Google Scholar] [CrossRef]
- Matyukhina, A.K.; Zorina-Tikhonova, E.N.; Goloveshkin, A.S.; Babeshkin, K.A.; Efimov, N.N.; Kiskin, M.A.; Eremenko, I.L. Field-Induced Slow Magnetic Relaxation in CoII Cyclopropane-1,1-Dicarboxylates. Molecules 2022, 27, 6537. [Google Scholar] [CrossRef]
- Zorina-Tikhonova, E.N.; Matyukhina, A.K.; Chistyakov, A.S.; Vologzhanina, A.V.; Korlyukov, A.A.; Gogoleva, N.V.; Novikova, V.A.; Belova, E.V.; Ugolkova, E.A.; Starikova, A.A.; et al. Synthesis, Structure, Magnetic Properties and Thermal Behaviour of Ba–MII (MII = Mn, Co, Cu, and Zn) Allylmalonates. N. J. Chem. 2022, 46, 21245–21256. [Google Scholar] [CrossRef]
- Tupolova, Y.P.; Shcherbakov, I.N.; Popov, L.D.; Lebedev, V.E.; Tkachev, V.V.; Zakharov, K.V.; Vasiliev, A.N.; Korchagin, D.V.; Palii, A.V.; Aldoshin, S.M. Field-Induced Single-Ion Magnet Behaviour of a Hexacoordinated Co(Ii) Complex with Easy-Axis-Type Magnetic Anisotropy. Dalton Trans. 2019, 48, 6960–6970. [Google Scholar] [CrossRef] [PubMed]
- Rigamonti, L.; Bridonneau, N.; Poneti, G.; Tesi, L.; Sorace, L.; Pinkowicz, D.; Jover, J.; Ruiz, E.; Sessoli, R.; Cornia, A. A Pseudo-Octahedral Cobalt(II) Complex with Bispyrazolylpyridine Ligands Acting as a Zero-Field Single-Molecule Magnet with Easy Axis Anisotropy. Chem. Eur. J. 2018, 24, 8857–8868. [Google Scholar] [CrossRef] [PubMed]
- Matos, C.R.M.O.; Sarmiento, C.V.; Silva, H.C.; Ferreira, G.B.; Guedes, G.P.; Nunes, W.C.; Ronconi, C.M. Field-Induced Single-Ion Magnets Exhibiting Tri-Axial Anisotropy in a 1D Co(Ii) Coordination Polymer with a Rigid Ligand 4,4′-(Buta-1,3-Diyne-1,4-Diyl)Dibenzoate. Dalton Trans. 2021, 50, 15003–15014. [Google Scholar] [CrossRef] [PubMed]
- Nemec, I.; Herchel, R.; Trávníček, Z. Two Polymorphic Co(Ii) Field-Induced Single-Ion Magnets with Enormous Angular Distortion from the Ideal Octahedron. Dalton Trans. 2018, 47, 1614–1623. [Google Scholar] [CrossRef] [PubMed]
- Świtlicka, A.; Machura, B.; Penkala, M.; Bieńko, A.; Bieńko, D.C.; Titiš, J.; Rajnák, C.; Boča, R.; Ozarowski, A. Slow Magnetic Relaxation in Hexacoordinated Cobalt(Ii) Field-Induced Single-Ion Magnets. Inorg. Chem. Front. 2020, 7, 2637–2650. [Google Scholar] [CrossRef]
- Huang, X.-C.; Xu, R.; Chen, Y.-Z.; Zhang, Y.-Q.; Shao, D. Two Four-Coordinate and Seven-Coordinate CoII Complexes Based on the Bidentate Ligand 1, 8-Naphthyridine Showing Slow Magnetic Relaxation Behavior. Chem. Asian J. 2020, 15, 279–286. [Google Scholar] [CrossRef]
- Saber, M.R.; Dunbar, K.R. Ligands Effects on the Magnetic Anisotropy of Tetrahedral Cobalt Complexes. Chem. Commun. 2014, 50, 12266–12269. [Google Scholar] [CrossRef]
- Smolko, L.; Černák, J.; Dušek, M.; Miklovič, J.; Titiš, J.; Boča, R. Three Tetracoordinate Co(II) Complexes [Co(Biq)X2] (X = Cl, Br, I) with Easy-Plane Magnetic Anisotropy as Field-Induced Single-Molecule Magnets. Dalton Trans. 2015, 44, 17565–17571. [Google Scholar] [CrossRef]
- Suturina, E.A.; Maganas, D.; Bill, E.; Atanasov, M.; Neese, F. Magneto-Structural Correlations in a Series of Pseudotetrahedral [CoII(XR)4]2– Single Molecule Magnets: An Ab Initio Ligand Field Study. Inorg. Chem. 2015, 54, 9948–9961. [Google Scholar] [CrossRef]
- Neese, F. The ORCA Program System. WIREs Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Boča, R. Zero-Field Splitting in Metal Complexes. Coord. Chem. Rev. 2004, 248, 757–815. [Google Scholar] [CrossRef]
- Damgaard-Møller, E.; Krause, L.; Tolborg, K.; Macetti, G.; Genoni, A.; Overgaard, J. Quantification of the Magnetic Anisotropy of a Single-Molecule Magnet from the Experimental Electron Density. Angew. Chemie Int. Ed. 2020, 59, 21203–21209. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zhai, Y.-Q.; Deng, Y.-F.; Chen, W.-P.; Zhang, T.; Zheng, Y.-Z. Correlating Magnetic Anisotropy with the Subtle Coordination Geometry Variation of a Series of Cobalt(Ii)-Sulfonamide Complexes. Dalton Trans. 2019, 48, 15419–15426. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.-Q.; Deng, Y.-F.; Zheng, Y.-Z. Pseudotetrahedral Cobalt(Ii) Complexes with PNP-Ligands Showing Uniaxial Magnetic Anisotropy. Dalton Trans. 2018, 47, 8874–8878. [Google Scholar] [CrossRef]
- Lang, L.; Atanasov, M.; Neese, F. Improvement of Ab Initio Ligand Field Theory by Means of Multistate Perturbation Theory. J. Phys. Chem. A 2020, 124, 1025–1037. [Google Scholar] [CrossRef]
- Sertphon, D.; Murray, K.S.; Phonsri, W.; Jover, J.; Ruiz, E.; Telfer, S.G.; Alkaş, A.; Harding, P.; Harding, D.J. Slow Relaxation of Magnetization in a Bis-Mer-Tridentate Octahedral Co(II) Complex. Dalton Trans. 2018, 47, 859–867. [Google Scholar] [CrossRef]
- Nemec, I.; Fellner, O.F.; Indruchová, B.; Herchel, R. Trigonally Distorted Hexacoordinate Co(II) Single-Ion Magnets. Materials 2022, 15, 1064. [Google Scholar] [CrossRef]
- Sundararajan, M.; Ganyushin, D.; Ye, S.; Neese, F. Multireference Ab Initio Studies of Zero-Field Splitting and Magnetic Circular Dichroism Spectra of Tetrahedral Co(II) Complexes. Dalton Trans. 2009, 30, 6021–6036. [Google Scholar] [CrossRef]
- Damgaard-Møller, E.; Krause, L.; Lassen, H.; Malaspina, L.A.; Grabowsky, S.; Bamberger, H.; McGuire, J.; Miras, H.N.; Sproules, S.; Overgaard, J. Investigating Complex Magnetic Anisotropy in a Co(II) Molecular Compound: A Charge Density and Correlated Ab Initio Electronic Structure Study. Inorg. Chem. 2020, 59, 13190–13200. [Google Scholar] [CrossRef]
- Tripathi, S.; Vaidya, S.; Ansari, K.U.; Ahmed, N.; Rivière, E.; Spillecke, L.; Koo, C.; Klingeler, R.; Mallah, T.; Rajaraman, G.; et al. Influence of a Counteranion on the Zero-Field Splitting of Tetrahedral Cobalt(II) Thiourea Complexes. Inorg. Chem. 2019, 58, 9085–9100. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, J.; Castro, I.; Ruiz-García, R.; Cano, J.; Julve, M.; Lloret, F.; De Munno, G.; Wernsdorfer, W.; Pardo, E. Field-Induced Slow Magnetic Relaxation in a Six-Coordinate Mononuclear Cobalt(II) Complex with a Positive Anisotropy. J. Am. Chem. Soc. 2012, 134, 15704–15707. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, S.; Singh, S.K.; Shukla, P.; Ansari, K.; Rajaraman, G.; Shanmugam, M. Role of Halide Ions in the Nature of the Magnetic Anisotropy in Tetrahedral CoII Complexes. Chem. Eur. J. 2017, 23, 9546–9559. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, S.; Upadhyay, A.; Singh, S.K.; Gupta, T.; Tewary, S.; Langley, S.K.; Walsh, J.P.S.; Murray, K.S.; Rajaraman, G.; Shanmugam, M. A Synthetic Strategy for Switching the Single Ion Anisotropy in Tetrahedral Co(Ii) Complexes. Chem. Commun. 2015, 51, 3739–3742. [Google Scholar] [CrossRef]
- Zadrozny, J.M.; Liu, J.; Piro, N.A.; Chang, C.J.; Hill, S.; Long, J.R. Slow Magnetic Relaxation in a Pseudotetrahedral Cobalt(Ii) Complex with Easy-Plane Anisotropy. Chem. Commun. 2012, 48, 3927–3929. [Google Scholar] [CrossRef]
- Yambulatov, D.S.; Nikolaevskii, S.A.; Shmelev, M.A.; Babeshkin, K.A.; Korchagin, D.V.; Efimov, N.N.; Goloveshkin, A.S.; Petrov, P.A.; Kiskin, M.A.; Sokolov, M.N.; et al. Heterometallic Coii-Lii Carboxylate Complexes with N-Heterocyclic Carbene, Triphenylphosphine and Pyridine: A Comparative Study of Magnetic Properties. Mendeleev Commun. 2021, 31, 624–627. [Google Scholar] [CrossRef]
- Svetogorov, R.D.; Dorovatovskii, P.V.; Lazarenko, V.A. Belok/XSA Diffraction Beamline for Studying Crystalline Samples at Kurchatov Synchrotron Radiation Source. Cryst. Res. Technol. 2020, 55, 1900184. [Google Scholar] [CrossRef]
- Svetogorov, R.D. Dionis—Diffraction Open Integration Software, Certificate of Registration № 2018660965; State Registration Certificate of a Computer Program; National Research Center “Kurchatov Institute”: Moscow, Russia, 2018.
- Sheldrick, G.M. SADABS. In Program for Empirical Absorption Correction of Area Detector Data; Bruker AXS Inc.: Madiso, WI, USA, 1997. [Google Scholar]
- Sheldrick, G.M. SHELXT Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Malmqvist, P.-Å.; Roos, B.O. The CASSCF State Interaction Method. Chem. Phys. Lett. 1989, 155, 189–194. [Google Scholar] [CrossRef]
- Neese, F. Calculation of the Zero-Field Splitting Tensor on the Basis of Hybrid Density Functional and Hartree-Fock Theory. J. Chem. Phys. 2007, 127, 164112. [Google Scholar] [CrossRef] [PubMed]
- Angeli, C.; Borini, S.; Cestari, M.; Cimiraglia, R. A Quasidegenerate Formulation of the Second Order N-Electron Valence State Perturbation Theory Approach. J. Chem. Phys. 2004, 121, 4043–4049. [Google Scholar] [CrossRef] [PubMed]
- Angeli, C.; Cimiraglia, R.; Evangelisti, S.; Leininger, T.; Malrieu, J.-P. Introduction of N-Electron Valence States for Multireference Perturbation Theory. J. Chem. Phys. 2001, 114, 10252–10264. [Google Scholar] [CrossRef]
- Angeli, C.; Cimiraglia, R.; Malrieu, J.-P. N-Electron Valence State Perturbation Theory: A Spinless Formulation and an Efficient Implementation of the Strongly Contracted and of the Partially Contracted Variants. J. Chem. Phys. 2002, 117, 9138–9153. [Google Scholar] [CrossRef]
- Ganyushin, D.; Neese, F. First-Principles Calculations of Zero-Field Splitting Parameters. J. Chem. Phys. 2006, 125, 24103. [Google Scholar] [CrossRef]
- Neese, F. Efficient and Accurate Approximations to the Molecular Spin-Orbit Coupling Operator and Their Use in Molecular g-Tensor Calculations. J. Chem. Phys. 2005, 122, 34107. [Google Scholar] [CrossRef]
- Maurice, R.; Bastardis, R.; de Graaf, C.; Suaud, N.; Mallah, T.; Guihéry, N. Universal Theoretical Approach to Extract Anisotropic Spin Hamiltonians. J. Chem. Theory Comput. 2009, 5, 2977–2984. [Google Scholar] [CrossRef]
- Schäfer, A.; Huber, C.; Ahlrichs, R. Fully Optimized Contracted Gaussian Basis Sets of Triple Zeta Valence Quality for Atoms Li to Kr. J. Chem. Phys. 1994, 100, 5829–5835. [Google Scholar] [CrossRef]
- Pantazis, D.A.; Chen, X.-Y.; Landis, C.R.; Neese, F. All-Electron Scalar Relativistic Basis Sets for Third-Row Transition Metal Atoms. J. Chem. Theory Comput. 2008, 4, 908–919. [Google Scholar] [CrossRef]
- Neese, F. An Improvement of the Resolution of the Identity Approximation for the Formation of the Coulomb Matrix. J. Comput. Chem. 2003, 24, 1740–1747. [Google Scholar] [CrossRef]
- Atanasov, M.; Ganyushin, D.; Sivalingam, K.; Neese, F. A Modern First-Principles View on Ligand Field Theory Through the Eyes of Correlated Multireference Wavefunctions BT. In Molecular Electronic Structures of Transition Metal Complexes II; Mingos, D.M.P., Day, P., Dahl, J.P., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 149–220. ISBN 978-3-642-27378-0. [Google Scholar]
- Singh, S.K.; Eng, J.; Atanasov, M.; Neese, F. Covalency and Chemical Bonding in Transition Metal Complexes: An Ab Initio Based Ligand Field Perspective. Coord. Chem. Rev. 2017, 344, 2–25. [Google Scholar] [CrossRef]


| Parameter | α-1 | β-1 | γ-1 | δ-1 |
|---|---|---|---|---|
| Chemical formula | C16H34CoN2O4 | C16H34CoN2O4 | C16H34CoN2O4 | |
| Mr (g mol−1) | 377.38 | 377.38 | 377.38 | |
| Crystal system, space group | Monoclinic, C2/c | Monoclinic, P2/n | Triclinic, P-1 | Triclinic, P1 |
| Temperature (K) | 293 | 293 | 150 | 260 |
| a, b, c (Å) | a = 19.892(2) b = 11.0060(15) c = 20.188(3) | a = 9.9569(16) b = 5.5100(9) c = 19.766(3) | a = 5.628(3) b = 9.297(5) c = 19.601(11) | a = 5.69977(16) b = 9.7985(3) c = 19.6559(8) |
| α, β, γ (°) | β = 106.753(4) | β = 102.015(7) | α = 89.746(14) β = 82.975(13) γ = 78.253(14) | α = 98.411(2) β = 95.424(3) γ = 66.597(2) |
| V (Å3) | 4232.1(9) | 1060.7(3) | 996.4(10) | 995.82(6) |
| Z | 8 | 2 | 2 | |
| µ (mm−1) | 0.83 | 0.83 | 0.88 | |
| Dcalc (g cm−3) | 1.185 | 1.182 | 1.258 | |
| Crystal size (mm) | 0.12 × 0.12 × 0.02 | 0.12 × 0.11 × 0.1 | 0.1 × 0.09 × 0.01 | |
| Absorption correction | Multi-scan | |||
| Tmin, Tmax | 0.243, 0.299 | 0.282, 0.381 | 0.253, 0.381 | |
| No. of measured reflections | 13275 | 7374 | 3142 | |
| No. of independent reflections | 4154 | 2078 | 2710 | |
| No. of observed [I > 2σ(I)] reflections | 2003 | 1512 | 817 | |
| Rint | 0.075 | 0.076 | 0.061 | |
| (sin θ/λ)max (Å−1) | 0.617 | 0.617 | 0.595 | |
| R1/wR(F2), [I > 2σ(I)] | 0.0569/0.1212 | 0.0864/0.2208 | 0.1109/0.2235 | |
| R1/wR(F2), (all data) | 0.1317/0.1604 | 0.1194/0.2416 | 0.2770/0.2821 | |
| GOOF | 1.00 | 1.08 | 0.87 | |
| No. of parameters | 252 | 126 | 213 | |
| Δρmax, Δρmin (e Å−3) | 0.43/−0.25 | 0.63/−0.83 | 0.88/−0.42 | |
| Temperature, K | Phase Content (%) | ||
|---|---|---|---|
| α-1 | γ-1 | δ-1 | |
| 300 (cooling) | 100 | - | - |
| 260 (cooling) | 100 | - | - |
| 220 (cooling) | 100 | - | - |
| 180 (cooling) | 54.3(1.3) | 34.7(6) | 11.0(8) |
| 140 (cooling) | 28(3) | 57.9(1.2) | 13.7(1.8) |
| 100 (cooling) | 27.2(1.3) | 60.5(1.0) | 12.2(1.1) |
| 140 (heating) | 27.7(1.4) | 61.5(9) | 10.8(1.2) |
| 180 (heating) | 27(5) | 55(3) | 18(4) |
| 220 (heating) | 22(3) | - | 78(3) |
| 260 (heating) | 23.4(1.4) | - | 76.6(1.4) |
| 300 (heating) | 80.9(6) | - | 19.1(6) |
| H-Bond | Symmetry Equivalent | D-H, Å | H…A, Å | D…A, Å | D-H…A, deg. |
|---|---|---|---|---|---|
| α-1 | |||||
| N1-H1A…O4 | 1/2-x, 1/2-y, 1-z | 0.89 | 2.10 | 2.975(6) | 170 |
| N2-H2A…O1 | 1/2-x, 3/2-y, 1-z | 0.89 | 2.11 | 2.951(6) | 159 |
| β-1 | |||||
| N1-H1A…O2 | x, 1 + y, z | 0.89 | 2.18 | 2.879(13) | 135 |
| N1-H1B…O2′ | 3/2-x, 1 + y, 1/2-z | 0.89 | 2.07 | 2.845(13) | 144 |
| γ-1 | |||||
| N1-H1A…O2 | 1 + x, y, z | 0.91 | 2.04 | 2.944(16) | 172 |
| N1-H1B…O4 | x, y, z | 0.91 | 2.55 | 2.991(15) | 110 |
| N2-H2A…O3 | 1 + x, y, z | 0.91 | 2.19 | 3.025(16) | 152 |
| Parameter | α-1 | γ-1 |
|---|---|---|
| Δax | −1316.9 | - |
| Δrh | 153.7 | - |
| D, cm−1 | - | 17.555 |
| |E/D| | - | 0.015 |
| gx, gy, gz | 2.135, 3.123, 1.845 | 2.287, 2.303, 2.125 |
| giso | 2.368 | 2.238 |
| ζ, cm−1 | 523.61 | 520.43 |
| λ, cm−1 | −174.54 | −173.5 |
| Phase | Term | Initial State, cm−1 | KD, cm−1 |
|---|---|---|---|
| α-1 | 4Eg 4A2g | 0 307.4 1470.6 | 0 232.6 621.3 917.8 1846.7 1915.9 |
| γ-1 | 4B2 4E | 3688.5 4552.6 | 0 35.1 3685.3 3786.1 4564.4 4658.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yambulatov, D.S.; Voronina, J.K.; Goloveshkin, A.S.; Svetogorov, R.D.; Veber, S.L.; Efimov, N.N.; Matyukhina, A.K.; Nikolaevskii, S.A.; Eremenko, I.L.; Kiskin, M.A. Change in the Electronic Structure of the Cobalt(II) Ion in a One-Dimensional Polymer with Flexible Linkers Induced by a Structural Phase Transition. Int. J. Mol. Sci. 2023, 24, 215. https://doi.org/10.3390/ijms24010215
Yambulatov DS, Voronina JK, Goloveshkin AS, Svetogorov RD, Veber SL, Efimov NN, Matyukhina AK, Nikolaevskii SA, Eremenko IL, Kiskin MA. Change in the Electronic Structure of the Cobalt(II) Ion in a One-Dimensional Polymer with Flexible Linkers Induced by a Structural Phase Transition. International Journal of Molecular Sciences. 2023; 24(1):215. https://doi.org/10.3390/ijms24010215
Chicago/Turabian StyleYambulatov, Dmitriy S., Julia K. Voronina, Alexander S. Goloveshkin, Roman D. Svetogorov, Sergey L. Veber, Nikolay N. Efimov, Anna K. Matyukhina, Stanislav A. Nikolaevskii, Igor L. Eremenko, and Mikhail A. Kiskin. 2023. "Change in the Electronic Structure of the Cobalt(II) Ion in a One-Dimensional Polymer with Flexible Linkers Induced by a Structural Phase Transition" International Journal of Molecular Sciences 24, no. 1: 215. https://doi.org/10.3390/ijms24010215
APA StyleYambulatov, D. S., Voronina, J. K., Goloveshkin, A. S., Svetogorov, R. D., Veber, S. L., Efimov, N. N., Matyukhina, A. K., Nikolaevskii, S. A., Eremenko, I. L., & Kiskin, M. A. (2023). Change in the Electronic Structure of the Cobalt(II) Ion in a One-Dimensional Polymer with Flexible Linkers Induced by a Structural Phase Transition. International Journal of Molecular Sciences, 24(1), 215. https://doi.org/10.3390/ijms24010215









