Cancer Stem Cells and Their Possible Implications in Cervical Cancer: A Short Review
Abstract
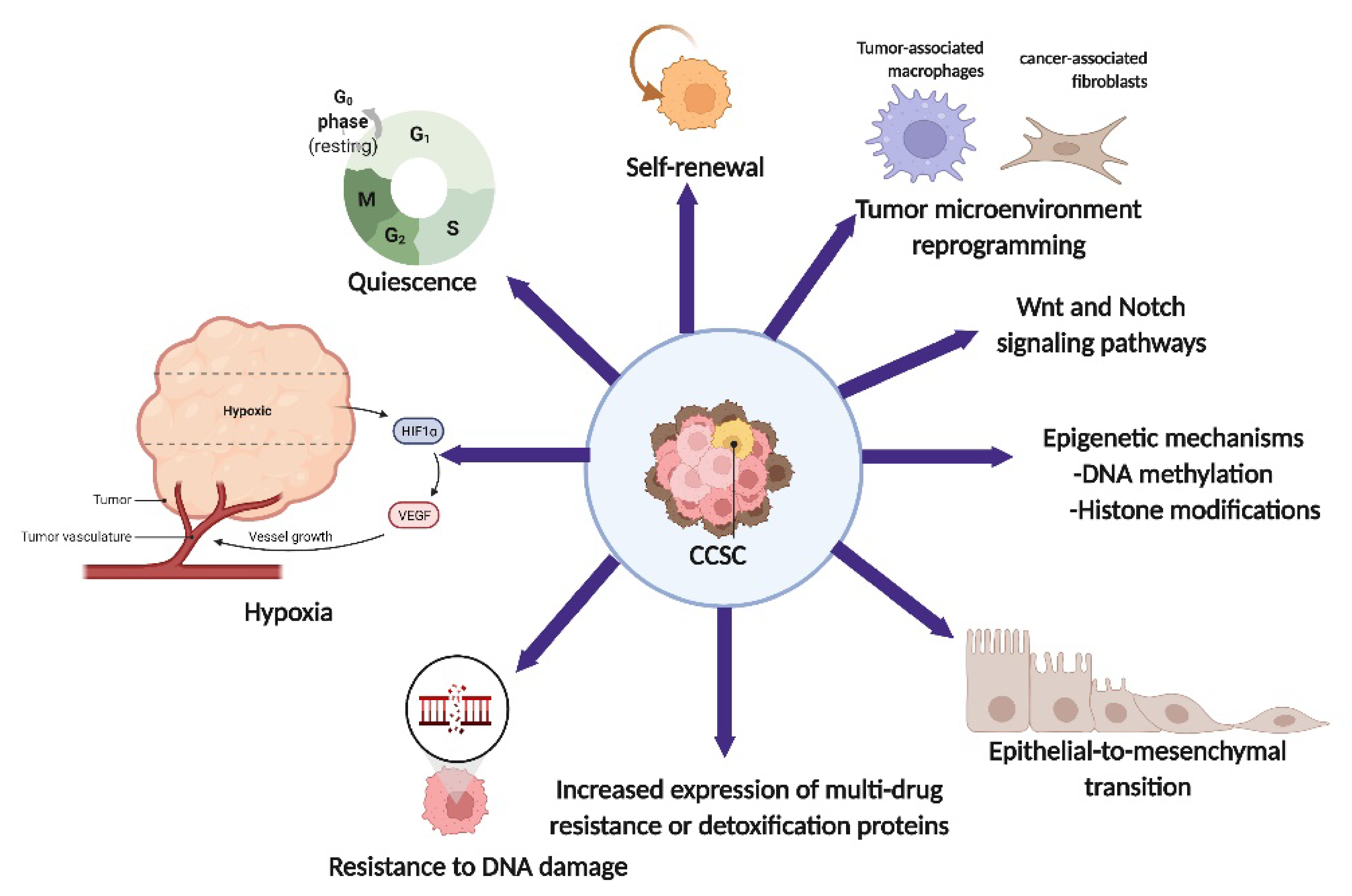
1. Introduction
2. Cancer Stem Cells in Cervical Cancer
3. Cancer Stem Cells and Therapeutic Implication
4. The Role of CSCs in Resistance to Cytotoxic Therapies: Chemo- and Radiotherapy
5. Resistance to DNA Damage-Induced Cell Death
6. CSCs’ Quiescence
7. Epithelial-Mesenchymal Transition (EMT)
8. Tumor Microenvironment
9. Hypoxia
10. Multidrug Resistance (MDR) and ALDH-Associated Resistance
11. Epigenetic Programming
12. Anti-CCSC Therapeutic Strategies
13. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADCs | antibody-drug conjugates |
| ALDH | aldehyde dehydrogenase |
| ATM | ataxia telangiectasia mutated |
| ATR | ataxia telangiectasia and Rad3-related |
| BRCA1 | breast cancer 1 |
| CAFs | cancer-associated fibroblasts |
| CC | cervical cancer |
| CCSC | cervical cancer stem cell |
| CHK1 and 2 | checkpoint kinase 1 and 2 |
| cPLA2α | cytosolic phospholipase A2alpha |
| CSC | cancer stem cell |
| DDR | DNA damage response |
| DNMTs | DNA methyltransferases |
| EMT | epithelial-mesenchymal transition |
| HBO | hyperbaric oxygen |
| HDACs | histone deacetylases |
| HPV | human papillomavirus |
| MDR | multidrug resistance |
| miRNA/miR | micro-RNA |
| MSI1 | musashi RNA binding protein 1 |
| NPs | nanoparticles |
| PARP | poly (ADP-ribose) polymerase |
| SP | Side population |
| TAMs | tumor-associated macrophages |
| TME | tumor microenvironment |
References
- Arbyn, M.; Weiderpass, E.; Bruni, L.; de Sanjosé, S.; Saraiya, M.; Ferlay, J.; Bray, F. Estimates of incidence and mortality of cervical cancer in 2018: A worldwide analysis. Lancet Glob. Health 2020, 8, e191–e203. [Google Scholar] [CrossRef]
- Mosmann, J.P.; Zayas, S.; Kiguen, A.X.; Venezuela, R.F.; Rosato, O.; Cuffini, C.G. Human papillomavirus and Chlamydia trachomatis in oral and genital mucosa of women with normal and abnormal cervical cytology. BMC Infect. Dis. 2021, 21, 422. [Google Scholar] [CrossRef] [PubMed]
- AACR Congress Progress Report 2021 (p.30), Cancerprogressreport.org. Philadelphia: American Association for Cancer Research. 2021. Available online: https://cancerprogressreport.aacr.org/wp-content/uploads/sites/2/2021/10/AACR_CPR_2021.pdf (accessed on 30 March 2022).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Luo, H.; Zhang, W.; Shen, Z.; Hu, X.; Zhu, X. Molecular mechanisms of cisplatin resistance in cervical cancer. Drug Des. Devel Ther. 2016, 10, 1885–1895. [Google Scholar] [CrossRef]
- Maneo, A.; Chiari, S.; Bonazzi, C.; Mangioni, C. Neoadjuvant chemotherapy and conservative surgery for stage IB1 cervical cancer. Gynecol. Oncol. 2008, 111, 438–443. [Google Scholar] [CrossRef]
- Duenas-Gonzalez, A.; Gonzalez-Fierro, A. Pharmacodynamics of current and emerging treatments for cervical cancer. Expert Opin. Drug Metab. Toxicol. 2019, 15, 671–682. [Google Scholar] [CrossRef]
- Wang, Q.; Peng, H.; Qi, X.; Wu, M.; Zhao, X. Targeted therapies in gynecological cancers: A comprehensive review of clinical evidence. Signal. Transduct. Target. Ther. 2020, 5, 137. [Google Scholar] [CrossRef]
- Áyen, Á.; Jiménez Martínez, Y.; Boulaiz, H. Targeted gene delivery therapies for cervical cancer. Cancers 2020, 12, 1301. [Google Scholar] [CrossRef]
- Liu, L.; Wang, M.; Li, X.; Yin, S.; Wang, B. An overview of novel agents for cervical cancer treatment by inducing apoptosis: Emerging drugs ongoing clinical trials and preclinical studies. Front. Med. 2021, 8, 682366. [Google Scholar] [CrossRef]
- Lathia, J.D.; Liu, H. Overview of cancer stem cells and stemness for community oncologists. Target. Oncol. 2017, 12, 387–399. [Google Scholar] [CrossRef]
- Valent, P.; Bonnet, D.; De Maria, R.; Lapidot, T.; Copland, M.; Melo, J.V.; Chomienne, C.; Ishikawa, F.; Schuringa, J.J.; Stassi, G.; et al. Cancer stem cell definitions and terminology: The devil is in the details. Nat. Rev. Cancer 2012, 12, 767–775. [Google Scholar] [CrossRef]
- Huang, R.; Rofstad, E.K. Cancer stem cells (CSCs), cervical CSCs and targeted therapies. Oncotarget 2017, 8, 35351–35367. [Google Scholar] [CrossRef]
- Messerschmidt, J.L.; Bhattacharya, P.; Messerschmidt, G.L. Cancer Clonal theory, immune escape, and their evolving roles in cancer multi-agent therapeutics. Curr. Oncol. Rep. 2017, 19, 66. [Google Scholar] [CrossRef]
- Sudhalkar, N.; Rathod, N.P.; Mathews, A.; Chopra, S.; Sriram, H.; Shrivastava, S.K.; Goda, J.S. Potential role of cancer stem cells as biomarkers and therapeutic targets in cervical cancer. Cancer Rep. (Hoboken) 2019, 2, e1144. [Google Scholar] [CrossRef]
- Adiga, D.; Eswaran, S.; Pandey, D.; Sharan, K.; Kabekkodu, S.P. Molecular landscape of recurrent cervical cancer. Crit. Rev. Oncol. Hematol. 2021, 157, 103178. [Google Scholar] [CrossRef]
- Huang, Z.; Wu, T.; Liu, A.Y.; Ouyang, G. Differentiation and transdifferentiation potentials of cancer stem cells. Oncotarget 2015, 6, 39550–39563. [Google Scholar] [CrossRef]
- Di Felice, E.; Caroli, S.; Paterlini, L.; Campari, C.; Prandi, S.; Giorgi Rossi, P. Cervical cancer epidemiology in foreign women in Northern Italy: Role of human papillomavirus prevalence in country of origin. Eur. J. Cancer Prev. 2015, 24, 223–230. [Google Scholar] [CrossRef]
- Yuan, Y.; Cai, X.; Shen, F.; Ma, F. HPV post-infection microenvironment and cervical cancer. Cancer Lett. 2021, 497, 243–254. [Google Scholar] [CrossRef]
- Hu, Q.; Wang, Y.; Zhang, Y.; Ge, Y.; Yin, Y.; Zhu, H. A new HPV score system predicts the survival of patients with cervical cancers. Front. Genet. 2021, 12, 747090. [Google Scholar] [CrossRef]
- Herfs, M.; Crum, C.P. Cervical cancer: Squamocolumnar junction ablation—tying up loose ends? Nat. Rev. Clin. Oncol. 2015, 12, 378–380. [Google Scholar] [CrossRef]
- Herfs, M.; Yamamoto, Y.; Laury, A.; Wang, X.; Nucci, M.R.; McLaughlin-Drubin, M.E.; Münger, K.; Feldman, S.; McKeon, F.D.; Xian, W.; et al. A discrete population of squamocolumnar junction cells implicated in the pathogenesis of cervical cancer. Proc. Natl. Acad. Sci. USA 2012, 109, 10516–10521. [Google Scholar] [CrossRef]
- Herfs, M.; Parra-Herran, C.; Howitt, B.E.; Laury, A.R.; Nucci, M.R.; Feldman, S.; Jimenez, C.A.; McKeon, F.D.; Xian, W.; Crum, C.P. Cervical squamocolumnar junction-specific markers define distinct, clinically relevant subsets of low-grade squamous intraepithelial lesions. Am. J. Surg. Pathol. 2013, 37, 1311–1318. [Google Scholar] [CrossRef]
- Thankamony, A.P.; Saxena, K.; Murali, R.; Jolly, M.K.; Nair, R. Cancer stem cell plasticity-a deadly deal. Front. Mol. Biosci. 2020, 7, 79. [Google Scholar] [CrossRef]
- Mendoza-Almanza, G.; Ortíz-Sánchez, E.; Rocha-Zavaleta, L.; Rivas-Santiago, C.; Esparza-Ibarra, E.; Olmos, J. Cervical cancer stem cells and other leading factors associated with cervical cancer development. Oncol. Lett. 2019, 18, 3423–3432. [Google Scholar] [CrossRef]
- Feng, D.; Peng, C.; Li, C.; Zhou, Y.; Li, M.; Ling, B.; Wei, H.; Tian, Z. Identification and characterization of cancer stem-like cells from primary carcinoma of the cervix uteri. Oncol. Rep. 2009, 22, 1129–1134. [Google Scholar]
- Bortolomai, I.; Canevari, S.; Facetti, I.; De Cecco, L.; Castellano, G.; Zacchetti, A.; Alison, M.R.; Miotti, S. Tumor initiating cells: Development and critical characterization of a model derived from the A431 carcinoma cell line forming spheres in suspension. Cell Cycle 2010, 9, 1194–1206. [Google Scholar] [CrossRef]
- López, J.; Poitevin, A.; Mendoza-Martínez, V.; Pérez-Plasencia, C.; García-Carrancá, A. Cancer-initiating cells derived from established cervical cell lines exhibit stem-cell markers and increased radioresistance. BMC Cancer 2012, 12, 48. [Google Scholar] [CrossRef]
- Zhang, S.L.; Wang, Y.S.; Zhou, T.; Yu, X.W.; Wei, Z.T.; Li, Y.L. Isolation and characterization of cancer stem cells from cervical cancer HeLa cells. Cytotechnology 2012, 64, 477–484. [Google Scholar] [CrossRef]
- Wang, K.; Zeng, J.; Luo, L.; Yang, J.; Chen, J.; Li, B.; Shen, K. Identification of a cancer stem cell-like side population in the HeLa human cervical carcinoma cell line. Oncol. Lett. 2013, 6, 1673–1680. [Google Scholar] [CrossRef]
- Liu, S.Y.; Zheng, P.S. High aldehyde dehydrogenase activity identifies cancer stem cells in human cervical cancer. Oncotarget 2013, 4, 2462–2475. [Google Scholar] [CrossRef]
- Qi, W.; Zhao, C.; Zhao, L.; Liu, N.; Li, X.; Yu, W.; Wei, L. Sorting and identification of side population cells in the human cervical cancer cell line HeLa. Cancer Cell Int. 2014, 14, 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Guo, H.; Lin, C.; Yang, L.; Wang, X. Enrichment and characterization of cancer stemlike cells from a cervical cancer cell line. Mol. Med. Rep. 2014, 9, 2117–2123. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Toledo, J.; Ponciano-Gómez, A.; Ortiz-Sánchez, E.; Garrido, E. Side populations from cervical-cancer-derived cell lines have stem-cell-like properties. Mol. Biol. Rep. 2014, 41, 1993–2004. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Yang, W.T.; Xu, R.; Liu, J.T.; Zheng, P.S. Cervical cancer cells with positive Sox2 expression exhibit the properties of cancer stem cells. PLoS ONE 2014, 9, e87092. [Google Scholar] [CrossRef]
- Kumazawa, S.; Kajiyama, H.; Umezu, T.; Mizuno, M.; Suzuki, S.; Yamamoto, E.; Mitsui, H.; Sekiya, R.; Shibata, K.; Kikkawa, F. Possible association between stem-like hallmark and radioresistance in human cervical carcinoma cells. J. Obstet. Gynaecol. Res. 2014, 40, 1389–1398. [Google Scholar] [CrossRef]
- Hou, T.; Zhang, W.; Tong, C.; Kazobinka, G.; Huang, X.; Huang, Y.; Zhang, Y. Putative stem cell markers in cervical squamous cell carcinoma are correlated with poor clinical outcome. BMC Cancer 2015, 15, 785. [Google Scholar] [CrossRef]
- Liu, H.; Wang, Y.J.; Bian, L.; Fang, Z.H.; Zhang, Q.Y.; Cheng, J.X. CD44+/CD24+ cervical cancer cells resist radiotherapy and exhibit properties of cancer stem cells. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1745–1754. [Google Scholar]
- Ortiz-Sánchez, E.; Santiago-López, L.; Cruz-Domínguez, V.B.; Toledo-Guzmán, M.E.; Hernández-Cueto, D.; Muñiz-Hernández, S.; Garrido, E.; Cantú De León, D.; García-Carrancá, A. Characterization of cervical cancer stem cell-like cells: Phenotyping, stemness, and human papilloma virus co-receptor expression. Oncotarget 2016, 7, 31943–31954. [Google Scholar] [CrossRef]
- Xie, Q.; Liang, J.; Rao, Q.; Xie, X.; Li, R.; Liu, Y.; Zhou, H.; Han, J.; Yao, T.; Lin, Z. Aldehyde dehydrogenase 1 expression predicts chemoresistance and poor clinical outcomes in patients with locally advanced cervical cancer treated with neoadjuvant chemotherapy prior to radical hysterectomy. Ann. Surg. Oncol. 2016, 23, 163–170. [Google Scholar] [CrossRef]
- Wei, Z.T.; Yu, X.W.; He, J.X.; Liu, Y.; Zhang, S.L. Characteristics of primary side population cervical cancer cells. Oncol. Lett. 2017, 14, 3536–3544. [Google Scholar] [CrossRef]
- Javed, S.; Sharma, B.K.; Sood, S.; Sharma, S.; Bagga, R.; Bhattacharyya, S.; Rayat, C.S.; Dhaliwal, L.; Srinivasan, R. Significance of CD133 positive cells in four novel HPV-16 positive cervical cancer-derived cell lines and biopsies of invasive cervical cancer. BMC Cancer 2018, 18, 357. [Google Scholar] [CrossRef]
- Li, H.; Zhang, W.; Yan, M.; Qiu, J.; Chen, J.; Sun, X.; Chen, X.; Song, L.; Zhang, Y. Nucleolar and spindle associated protein 1 promotes metastasis of cervical carcinoma cells by activating Wnt/β-catenin signaling. J. Exp. Clin. Cancer Res. 2019, 38, 33. [Google Scholar] [CrossRef]
- Yao, T.; Weng, X.; Yao, Y.; Huang, C.; Li, J.; Peng, Y.; Lin, R.; Lin, Z. ALDH-1-positive cells exhibited a radioresistant phenotype that was enhanced with hypoxia in cervical cancer. BMC Cancer 2020, 20, 891. [Google Scholar] [CrossRef]
- Zhou, H.M.; Zhang, J.G.; Zhang, X.; Li, Q. Targeting cancer stem cells for reversing therapy resistance: Mechanism, signaling, and prospective agents. Signal. Transduct. Target. Ther. 2021, 6, 62. [Google Scholar] [CrossRef]
- Marzagalli, M.; Fontana, F.; Raimondi, M.; Limonta, P. Cancer stem cells-key players in tumor relapse. Cancers 2021, 13, 376. [Google Scholar] [CrossRef]
- Ngwa, W.; Irabor, O.C.; Schoenfeld, J.D.; Hesser, J.; Demaria, S.; Formenti, S.C. Using immunotherapy to boost the abscopal effect. Nat. Rev. Cancer 2018, 18, 313–322. [Google Scholar] [CrossRef]
- Batlle, E.; Clevers, H. Cancer stem cells revisited. Nat. Med. 2017, 23, 1124–1134. [Google Scholar] [CrossRef]
- Lytle, N.K.; Barber, A.G.; Reya, T. Stem cells fate in cancer growth, progression and therapy resistance. Nat. Rev. Cancer 2018, 18, 669–680. [Google Scholar] [CrossRef]
- Chopra, S.; Deodhar, K.; Pai, V.; Pant, S.; Rathod, N.; Goda, J.S.; Sudhalkar, N.; Pandey, P.; Waghmare, S.; Engineer, R.; et al. Cancer stem cells, CD44, and outcomes following chemoradiation in locally advanced cervical cancer: Results from a prospective study. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 161–168. [Google Scholar] [CrossRef]
- Zhao, J. Cancer stem cells and chemoresistance: The smartest survives the raid. Pharmacol. Ther. 2016, 160, 145–158. [Google Scholar] [CrossRef]
- Zuccherato, L.W.; Machado, C.; Magalhães, W.; Martins, P.R.; Campos, L.S.; Braga, L.C.; Teixeira-Carvalho, A.; Martins-Filho, O.A.; Franco, T.; Paula, S.; et al. Cervical cancer stem-like cell transcriptome profiles predict response to chemoradiotherapy. Front. Oncol. 2021, 11, 639339. [Google Scholar] [CrossRef]
- Bao, S.; Wu, Q.; McLendon, R.E.; Hao, Y.; Shi, Q.; Hjelmeland, A.B.; Dewhirst, M.W.; Bigner, D.D.; Rich, J.N. Glioma stem cells promote radioresistance bypreferential activation of the DNA damage response. Nature 2006, 444, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Venkatesha, V.A.; Parsels, L.A.; Parsels, J.D.; Zhao, L.; Zabludoff, S.D.; Simeone, D.M.; Maybaum, J.; Lawrence, T.S.; Morgan, M.A. Sensitization of pancreatic cancer stem cells to gemcitabine by Chk1 inhibition. Neoplasia 2012, 14, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, X.; Wang, T.; Guo, Q.; Xi, T.; Zheng, L. Emerging agents that target signaling pathways in cancer stem cells. J. Hematol. Oncol. 2020, 13, 60. [Google Scholar] [CrossRef]
- Yang, L.; Shi, P.; Zhao, G.; Xu, J.; Peng, W.; Zhang, J.; Zhang, G.; Wang, X.; Dong, Z.; Chen, F.; et al. Targeting cancer stem cell pathways for cancer therapy. Signal. Transduct Target. Ther. 2020, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Tomao, F.; Santangelo, G.; Musacchio, L.; Di Donato, V.; Fischetti, M.; Giancotti, A.; Perniola, G.; Petrella, M.C.; Monti, M.; Palaia, I.; et al. Targeting cervical cancer: Is there a role for poly (ADP-ribose) polymerase inhibition? J. Cell Physiol. 2020, 235, 5050–5058. [Google Scholar] [CrossRef]
- Zhang, J.; Si, J.; Gan, L.; Di, C.; Xie, Y.; Sun, C.; Li, H.; Guo, M.; Zhang, H. Research progress on therapeutic targeting of quiescent cancer cells. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2810–2820. [Google Scholar] [CrossRef]
- Xu, H.; Sun, Y.; Zeng, L.; Li, Y.; Hu, S.; He, S.; Chen, H.; Zou, Q.; Luo, B. Inhibition of cytosolic phospholipase A2 alpha increases chemosensitivity in cervical carcinoma through suppressing β-catenin signaling. Cancer Biol. Ther. 2019, 20, 912–921. [Google Scholar] [CrossRef]
- Prieto-Vila, M.; Takahashi, R.U.; Usuba, W.; Kohama, I.; Ochiya, T. Drug resistance driven by cancer stem cells and their niche. Int. J. Mol. Sci. 2017, 18, 2574. [Google Scholar] [CrossRef]
- Phi, L.; Sari, I.N.; Yang, Y.G.; Lee, S.H.; Jun, N.; Kim, K.S.; Lee, Y.K.; Kwon, H.Y. Cancer Stem Cells (CSCs) in drug resistance and their therapeutic implications in cancer treatment. Stem Cells Int. 2018, 2018, 5416923. [Google Scholar] [CrossRef]
- Qureshi, R.; Arora, H.; Rizvi, M.A. EMT in cervical cancer: Its role in tumour progression and response to therapy. Cancer Lett. 2015, 356 (2 Pt B), 321–331. [Google Scholar] [CrossRef]
- Roma-Rodrigues, C.; Mendes, R.; Baptista, P.V.; Fernandes, A.R. Targeting Tumor Microenvironment for Cancer Therapy. Int J. Mol. Sci. 2019, 20, 840. [Google Scholar] [CrossRef]
- Fan, Z.; Cui, H.; Yu, H.; Ji, Q.; Kang, L.; Han, B.; Wang, J.; Dong, Q.; Li, Y.; Yan, Z.; et al. MiR-125a promotes paclitaxel sensitivity in cervical cancer through altering STAT3 expression. Oncogenesis 2016, 5, e197. [Google Scholar] [CrossRef]
- Graham, K.; Unger, E. Overcoming tumor hypoxia as a barrier to radiotherapy, chemotherapy and immunotherapy in cancer treatment. Int. J. Nanomed. 2018, 13, 6049–6058. [Google Scholar] [CrossRef]
- Hughes, V.S.; Wiggins, J.M.; Siemann, D.W. Tumor oxygenation and cancer therapy-then and now. Br. J. Radiol. 2019, 92, 20170955. [Google Scholar] [CrossRef]
- Terraneo, N.; Jacob, F.; Dubrovska, A.; Grünberg, J. Novel therapeutic strategies for ovarian cancer stem cells. Front. Oncol. 2020, 10, 319. [Google Scholar] [CrossRef]
- Watson, E.R.; Halnan, K.E.; Dische, S.; Saunders, M.I.; Cade, I.S.; McEwen, J.B.; Wiernik, G.; Perrins, D.J.; Sutherland, I. Hyperbaric oxygen and radiotherapy: A Medical Research Council trial in carcinoma of the cervix. Br. J. Radiol. 1978, 51, 879–887. [Google Scholar] [CrossRef]
- Höckel, M.; Knoop, C.; Schlenger, K.; Vorndran, B.; Baussmann, E.; Mitze, M.; Knapstein, P.G.; Vaupel, P. Intratumoral pO2 predicts survival in advanced cancer of the uterine cervix. Radiother. Oncol. 1993, 26, 45–50. [Google Scholar] [CrossRef]
- Hockel, M.; Schlenger, K.; Aral, B.; Mitze, M.; Schaffer, U.; Vaupel, P. Association between tumor hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res. 1996, 56, 4509–4515. [Google Scholar]
- Boesch, M.; Zeimet, A.G.; Reimer, D.; Schmidt, S.; Gastl, G.; Parson, W.; Spoeck, F.; Hatina, J.; Wolf, D.; Sopper, S. The side population of ovarian cancer cells defines a heterogeneous compartment exhibiting stem cell characteristics. Oncotarget 2014, 5, 7027–7039. [Google Scholar] [CrossRef]
- Choi, Y.H.; Yu, A.M. ABC transporters in multidrug resistance and pharmacokinetics, and strategies for drug development. Curr. Pharm. Des. 2014, 20, 793–807. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Zheng, Y.; Ma, L.; Tian, L.; Sun, Q. Clinically-Relevant ABC Transporter for Anti-Cancer Drug Resistance. Front. Pharmacol. 2021, 12, 648407. [Google Scholar] [CrossRef] [PubMed]
- Chanmahasathien, W.; Ampasavate, C.; Greger, H.; Limtrakul, P. Stemona alkaloids, from traditional Thai medicine, increase chemosensitivity via P-glycoprotein-mediated multidrug resistance. Phytomedicine 2011, 18, 199–204. [Google Scholar] [CrossRef]
- Kuroda, T.; Hirohashi, Y.; Torigoe, T.; Yasuda, K.; Takahashi, A.; Asanuma, H.; Morita, R.; Mariya, T.; Asano, T.; Mizuuchi, M.; et al. ALDH1-high ovarian cancer stem-like cells can be isolated from serous and clear cell adenocarcinoma cells, and ALDH1 high expression is associated with poor prognosis. PLoS ONE 2013, 8, e65158. [Google Scholar] [CrossRef] [PubMed]
- Ayub, T.H.; Keyver-Paik, M.D.; Debald, M.; Rostamzadeh, B.; Thiesler, T.; Schroder, L.; Barchet, W.; Abramian, A.; Kaiser, C.; Kristiansen, G.; et al. Accumulation of ALDH1-positive cells after neoadjuvant chemotherapy predicts treatment resistance and prognosticates poor outcome in ovarian cancer. Oncotarget 2015, 6, 16437–16448. [Google Scholar] [CrossRef]
- Sun, Y.; Jia, X.; Wu, X. High expressions of Lgr5 and ALDH1 in primary epithelial ovarian cancer correlate with advanced tumor stage and grade as well as poor prognosis of the patients. Gynecol. Obstet. Investig. 2015, 81, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.H.; Wang, Y.C.; Chou, Y.C.; Yu, M.H.; Chao, T.K. The combination of aldehyde dehydrogenase 1 (ALDH1) and CD44 is associated with poor outcomes in endometrial cancer. PLoS ONE 2018, 13, e0206685. [Google Scholar] [CrossRef]
- Rao, Q.X.; Yao, T.T.; Zhang, B.Z.; Lin, R.C.; Chen, Z.L.; Zhou, H.; Wang, L.J.; Lu, H.W.; Chen, Q.; Di, N.; et al. Expression and functional role of ALDH1 in cervical carcinoma cells. Asian Pac. J. Cancer Prev. 2012, 13, 1325–1331. [Google Scholar] [CrossRef][Green Version]
- Yao, T.; Wu, Z.; Liu, Y.; Rao, Q.; Lin, Z. Aldehyde dehydrogenase 1 (ALDH1) positivity correlates with poor prognosis in cervical cancer. J. Int. Med. Res. 2014, 42, 1038–1042. [Google Scholar] [CrossRef]
- Tulake, W.; Yuemaier, R.; Sheng, L.; Ru, M.; Lidifu, D.; Abudula, A. Upregulation of stem cell markers ALDH1A1 and OCT4 as potential biomarkers for the early detection of cervical carcinoma. Oncol. Lett. 2018, 16, 5525–5534. [Google Scholar] [CrossRef]
- Muralikrishnan, V.; Hurley, T.D.; Nephew, K.P. Targeting aldehyde dehydrogenases to eliminate cancer stem cells in gynecologic malignancies. Cancers 2020, 12, 961. [Google Scholar] [CrossRef]
- Boyd, P.; Major, I.; Wang, W.; McConville, C. Development of disulfiram-loaded vaginal rings for the localised treatment of cervical cancer. Eur. J. Pharm. Biopharm. 2014, 88, 945–953. [Google Scholar] [CrossRef]
- Wongtrakoongate, P. Epigenetic therapy of cancer stem and progenitor cells by targeting DNA methylation machineries. World J. Stem Cells 2015, 7, 137–148. [Google Scholar] [CrossRef]
- Hu, C.; Liu, X.; Zeng, Y.; Liu, J.; Wu, F. DNA methyltransferase inhibitors combination therapy for the treatment of solid tumor: Mechanism and clinical application. Clin. Epigenetics 2021, 13, 166. [Google Scholar] [CrossRef]
- Liu, S.; Chang, W.; Jin, Y.; Feng, C.; Wu, S.; He, J.; Xu, T. The function of histone acetylation in cervical cancer development. Biosci. Rep. 2019, 39, BSR20190527. [Google Scholar] [CrossRef]
- Bayat, S.; Shekari Khaniani, M.; Choupani, J.; Alivand, M.R.; Mansoori Derakhshan, S. HDACis (class I), cancer stem cell, and phytochemicals: Cancer therapy and prevention implications. Biomed. Pharmacother. 2018, 97, 1445–1453. [Google Scholar] [CrossRef]
- Pathania, R.; Ramachandran, S.; Mariappan, G.; Thakur, P.; Shi, H.; Choi, J.H.; Manicassamy, S.; Kolhe, R.; Prasad, P.D.; Sharma, S.; et al. Combined inhibition of DNMT and HDAC blocks the tumorigenicity of cancer stem-like cells and attenuates mammary tumor growth. Cancer Res. 2016, 76, 3224–3235. [Google Scholar] [CrossRef]
- Azad, N.S.; El-Khoueiry, A.; Yin, J.; Oberg, A.L.; Flynn, P.; Adkins, D.; Sharma, A.; Weisenberger, D.J.; Brown, T.; Medvari, P.; et al. Combination epigenetic therapy in metastatic colorectal cancer (mCRC) with subcutaneous 5-azacitidine and entinostat: A phase 2 consortium/stand Up 2 cancer study. Oncotarget 2017, 8, 35326–35338. [Google Scholar] [CrossRef]
- Huang, Y.; Davidson, N.E. Targeting tumorigenicity of breast cancer stemlike cells using combination epigenetic therapy: Something old and something new. J. Thorac. Dis. 2016, 8, 2971–2974. [Google Scholar] [CrossRef]
- Cai, N.; Wang, Y.D.; Zheng, P.S. The microrna-302–367 cluster suppresses the proliferation of cervical carcinoma cells through the novel target akt1. RNA 2013, 19, 85–95. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.; Liu, N.; Gao, Y.; Li, L. MiR-23b controls ALDH1A1 expression in cervical cancer stem cells. BMC Cancer 2017, 17, 292. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yue, Y.; Wang, R.; Gong, B.; Duan, Z. MicroRNA-145 inhibits tumorigenesis and invasion of cervical cancer stem cells. Int. J. Oncol. 2017, 50, 853–862. [Google Scholar] [CrossRef] [PubMed]
- Blackford, A.N.; Jackson, S.P. ATM, ATR, and DNA-PK: The trinity at the heart of the DNA damage response. Mol. Cell. 2017, 66, 801–817. [Google Scholar] [CrossRef] [PubMed]
- Chao, H.X.; Poovey, C.E.; Privette, A.A.; Grant, G.D.; Chao, H.Y.; Cook, J.G.; Purvis, J.E. Orchestration of DNA damage checkpoint dynamics across the human cell cycle. Cell Syst. 2017, 5, 445–459.e5. [Google Scholar] [CrossRef]
- Brinkman, J.A.; Liu, Y.; Kron, S.J. Small-molecule drug repurposing to target DNA damage repair and response pathways. Semin. Cancer Biol. 2021, 68, 230–241. [Google Scholar] [CrossRef]
- Calcinotto, A.; Kohli, J.; Zagato, E.; Pellegrini, L.; Demaria, M.; Alimonti, A. Cellular senescence: Aging, cancer, and injury. Physiol. Rev. 2019, 99, 1047–1078. [Google Scholar] [CrossRef]
- Wang, W.J.; Wu, S.P.; Liu, J.B.; Shi, Y.S.; Huang, X.; Zhang, Q.B.; Yao, K.T. MYC regulation of CHK1 and CHK2 promotes radioresistance in a stem cell-like population of nasopharyngeal carcinoma cells. Cancer Res. 2013, 73, 1219–1231. [Google Scholar] [CrossRef]
- De Angelis, M.L.; Francescangeli, F.; La Torre, F.; Zeuner, A. Stem cell plasticity and dormancy in the development of cancer therapy resistance. Front. Oncol. 2019, 9, 626. [Google Scholar] [CrossRef]
- Ayob, A.Z.; Ramasamy, T.S. Cancer stem cells as key drivers of tumour progression. J. Biomed. Sci. 2018, 25, 20. [Google Scholar] [CrossRef]
- Luo, M.; Li, J.F.; Yang, Q.; Zhang, K.; Wang, Z.W.; Zheng, S.; Zhou, J.J. Stem cell quiescence and its clinical relevance. World J. Stem Cells 2020, 12, 1307–1326. [Google Scholar] [CrossRef]
- Roca, M.S.; Di Gennaro, E.; Budillon, A. Implication for cancer stem cells in solid cancer chemo-resistance: Promising therapeutic strategies based on the use of HDAC inhibitors. J. Clin. Med. 2019, 8, 912. [Google Scholar] [CrossRef]
- Oshimori, N.; Oristian, D.; Fuchs, E. TGF-β promotes heterogeneity and drug resistance in squamous cell carcinoma. Cell 2015, 160, 963–976. [Google Scholar] [CrossRef]
- Creighton, C.J.; Li, X.; Landis, M.; Dixon, J.M.; Neumeister, V.M.; Sjolund, A.; Rimm, D.L.; Wong, H.; Rodriguez, A.; Herschkowitz, J.I.; et al. Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proc. Natl. Acad. Sci. USA 2009, 106, 13820–13825. [Google Scholar] [CrossRef]
- Oskarsson, T.; Batlle, E.; Massagué, J. Metastatic stem cells: Sources, niches, and vital pathways. Cell Stem Cell 2014, 14, 306–321. [Google Scholar] [CrossRef]
- Puisieux, A.; Brabletz, T.; Caramel, J. Oncogenic roles of EMT-inducing transcription factors. Nat. Cell Biol. 2014, 16, 488–494. [Google Scholar] [CrossRef]
- Carnero, A.; Garcia-Mayea, Y.; Mir, C.; Lorente, J.; Rubio, I.T.; LLeonart, M.E. The cancer stem-cell signalling network and resistance to therapy. Cancer Treat. Rev. 2016, 49, 25–36. [Google Scholar] [CrossRef]
- Zhu, L.F.; Hu, Y.; Yang, C.C.; Xu, X.H.; Ning, T.Y.; Wang, Z.L.; Ye, J.H.; Liu, L.K. Snail overexpression induces an epithelial to mesenchymal transition and cancer stem cell-like properties in SCC9 cells. Lab. Investig. 2012, 92, 744–752. [Google Scholar] [CrossRef]
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun. Signal. 2020, 18, 59. [Google Scholar] [CrossRef]
- Plaks, V.; Kong, N.; Werb, Z. The cancer stem cell niche: How essential is the niche in regulating stemness of tumor cells? Cell Stem Cell 2015, 16, 225–238. [Google Scholar] [CrossRef]
- Liu, Z.; Qi, L.; Li, Y.; Zhao, X.; Sun, B. VEGFR2 regulates endothelial differentiation of colon cancer cells. BMC Cancer 2017, 17, 593. [Google Scholar] [CrossRef]
- Shangguan, W.; Fan, C.; Chen, X.; Lu, R.; Liu, Y.; Li, Y.; Shang, Y.; Yin, D.; Zhang, S.; Huang, Q.; et al. Endothelium originated from colorectal cancer stem cells constitute cancer blood vessels. Cancer Sci. 2017, 108, 1357–1367. [Google Scholar] [CrossRef]
- Eskander, R.N.; Tewari, K.S. Beyond angiogenesis blockade: Targeted therapy for advanced cervical cancer. J. Gynecol Oncol. 2014, 25, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Xu, J.; Liu, S. Cancer Stem Cells and Neovascularization. Cells 2021, 10, 1070. [Google Scholar] [CrossRef] [PubMed]
- Pasquier, J.; Ghiabi, P.; Chouchane, L.; Razzouk, K.; Rafii, S.; Rafii, A. Angiocrine endothelium: From physiology to cancer. J. Transl. Med. 2020, 18, 52. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Farhood, B.; Mortezaee, K. Cancer stem cells (CSCs) in cancer progression and therapy. J. Cell Physiol. 2019, 234, 8381–8395. [Google Scholar] [CrossRef]
- Balsat, C.; Signolle, N.; Goffin, F.; Delbecque, K.; Plancoulaine, B.; Sauthier, P.; Samouëlian, V.; Béliard, A.; Munaut, C.; Foidart, J.M.; et al. Improved computer-assisted analysis of the global lymphatic network in human cervical tissues. Mod. Pathol. 2014, 27, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Daverey, A.; Drain, A.P.; Kidambi, S. Physical Intimacy of Breast Cancer Cells with Mesenchymal Stem Cells Elicits Trastuzumab Resistance through Src Activation. Sci. Rep. 2015, 5, 13744. [Google Scholar] [CrossRef] [PubMed]
- Yao, T.; Lu, R.; Zhang, Y.; Zhang, Y.; Zhao, C.; Lin, R.; Lin, Z. Cervical cancer stem cells. Cell Prolif. 2015, 48, 611–625. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, B.; Xiao, S.; Li, Y.; Chen, Q. miR-125a/b inhibits tumor-associated macrophages mediated in cancer stem cells of hepatocellular carcinoma by targeting CD90. J. Cell Biochem. 2019, 120, 3046–3055. [Google Scholar] [CrossRef]
- Heddleston, J.M.; Li, Z.; Lathia, J.D.; Bao, S.; Hjelmeland, A.B.; Rich, J.N. Hypoxia inducible factors in cancer stem cells. Br. J. Cancer 2010, 102, 789–795. [Google Scholar] [CrossRef]
- Sonveaux, P. ROS and radiotherapy: More we care. Oncotarget 2017, 8, 35482–35483. [Google Scholar] [CrossRef]
- Pattabiraman, D.R.; Weinberg, R.A. Tackling the cancer stem cells—What challenges do they pose? Nat. Rev. Drug Discov. 2014, 13, 497–512. [Google Scholar] [CrossRef]
- Alisi, A.; Cho, W.C.; Locatelli, F.; Fruci, D. Multidrug resistance and cancer stem cells in neuroblastoma and hepatoblastoma. Int. J. Mol. Sci. 2013, 14, 24706–24725. [Google Scholar] [CrossRef]
- Seigel, G.M.; Campbell, L.M.; Narayan, M.; Gonzalez-Fernandez, F. Cancer stem cell characteristics in retinoblastoma. Mol. Vis. 2005, 11, 729–737. [Google Scholar]
- Hirschmann-Jax, C.; Foster, A.E.; Wulf, G.G.; Nuchtern, J.G.; Jax, T.W.; Gobel, U.; Goodell, M.A.; Brenner, M.K. A distinct “side population” of cells with high drug efflux capacity in human tumor cells. Proc. Natl. Acad. Sci. USA 2004, 101, 14228–14233. [Google Scholar] [CrossRef]
- Haraguchi, N.; Utsunomiya, T.; Inoue, H.; Tanaka, F.; Mimori, K.; Barnard, G.F.; Mori, M. Characterization of a side population of cancer cells from human gastrointestinal system. Stem Cells 2006, 24, 506–513. [Google Scholar] [CrossRef]
- Singh, S.; Brocker, C.; Koppaka, V.; Chen, Y.; Jackson, B.C.; Matsumoto, A.; Thompson, D.C.; Vasiliou, V. Aldehyde dehydrogenases in cellular responses to oxidative/electrophilic stress. Free Radic. Biol. Med. 2013, 56, 89–101. [Google Scholar] [CrossRef]
- Raha, D.; Wilson, T.R.; Peng, J.; Peterson, D.; Yue, P.; Evangelista, M.; Wilson, C.; Merchant, M.; Settleman, J. The cancer stem cell marker aldehyde dehydrogenase is required to maintain a drug-tolerant tumor cell subpopulation. Cancer Res. 2014, 74, 3579–3590. [Google Scholar] [CrossRef]
- Pors, K.; Moreb, J.S. Aldehyde dehydrogenases in cancer: An opportunity for biomarker and drug development? Drug Discov. Today 2014, 19, 1953–1963. [Google Scholar] [CrossRef]
- Keyvani-Ghamsari, S.; Khorsandi, K.; Rasul, A.; Zaman, M.K. Current understanding of epigenetics mechanism as a novel target in reducing cancer stem cells resistance. Clin. Epigenetics 2021, 13, 120. [Google Scholar] [CrossRef]
- Shukla, S.; Meeran, S.M. Epigenetics of cancer stem cells: Pathways and therapeutics. Biochim. Biophys. Acta 2014, 1840, 3494–3502. [Google Scholar] [CrossRef]
- Talukdar, S.; Emdad, L.; Das, S.K.; Sarkar, D.; Fisher, P.B. Evolving strategies for therapeutically targeting cancer stem cells. Adv. Cancer Res. 2016, 131, 159–191. [Google Scholar]
- Eun, K.; Ham, S.W.; Kim, H. Cancer stem cell heterogeneity: Origin and new perspectives on CSC targeting. BMB Rep. 2017, 50, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Dragomir, M.P.; Knutsen, E.; Calin, G.A. Classical and noncanonical functions of miRNAs in cancers. Trends Genet. 2022, 38, 379–394. [Google Scholar] [CrossRef]
- Di Fiore, R.; Suleiman, S.; Pentimalli, F.; O’Toole, S.A.; O’Leary, J.J.; Ward, M.P.; Conlon, N.T.; Sabol, M.; Ozretić, P.; Erson-Bensan, A.E.; et al. Could MicroRNAs be useful tools to improve the diagnosis and treatment of rare gynecological cancers? A brief overview. Int. J. Mol. Sci. 2021, 22, 3822. [Google Scholar] [CrossRef]
- Huang, T.; Alvarez, A.; Hu, B.; Cheng, S.Y. Noncoding RNAs in cancer and cancer stem cells. Chin. J. Cancer. 2013, 32, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Lou, W.; Liu, J.; Gao, Y.; Zhong, G.; Ding, B.; Xu, L. MicroRNA regulation of liver cancer stem cells. Am. J. Cancer Res. 2018, 8, 1141. [Google Scholar]
- Sun, H.R.; Wang, S.; Yan, S.C.; Zhang, Y.; Nelson, P.J.; Jia, H.L.; Qin, L.X.; Dong, Q.Z. Therapeutic strategies targeting cancer stem cells and their microenvironment. Front. Oncol. 2019, 9, 1104. [Google Scholar] [CrossRef]
- Rahman, M.; Jamil, H.; Akhtar, N.; Rahman, K.; Islam, R.; Asaduzzaman, S. Stem cell and cancer stem cell: A tale of two cells. Prog. Stem Cell 2016, 3, 97–108. [Google Scholar] [CrossRef]
- Vidal, S.J.; Rodriguez-Bravo, V.; Galsky, M.; Cordon-Cardo, C.; Domingo-Domenech, J. Targeting cancer stem cells to suppress acquired chemotherapy resistance. Oncogene 2014, 33, 4451–4463. [Google Scholar] [CrossRef] [PubMed]
- Organista-Nava, J.; Illades-Aguiar, B. Role of Oct3/4 in cervical cancer tumorigenesis. Front. Oncol. 2020, 10, 247. [Google Scholar]
- Lou, D.; Wang, B.; Wang, Y.; Cao, Y. Selective killing of cancer stem cells by a novel dual-targeting strategy. Med. Hypotheses. 2012, 79, 430–432. [Google Scholar] [CrossRef]
- Burke, A.R.; Singh, R.N.; Carroll, D.L.; Wood, J.C.; D’Agostino, R.B.; Jr Ajayan, P.M.; Torti, F.M.; Torti, S.V. The resistance of breast cancer stem cells to conventional hyperthermia and their sensitivity to nanoparticle-mediated photothermal therapy. Biomaterials 2012, 33, 2961–2970. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, F.; Wang, L.; Xie, C.; Wu, P.; Du, S.; Zhou, S.; Sun, Z.; Liu, Q.; Yu, L.; et al. Enhanced and prolonged antitumor effect of Salinomycin-loaded gelatinase-responsive nanoparticles via targeted drug delivery and inhibition of cervical cancer stem cells. Int. J. Nanomed. 2020, 15, 1283–1295. [Google Scholar] [CrossRef]
- Wang, Q.; Yen, Y.T.; Xie, C.; Liu, F.; Liu, Q.; Wei, J.; Yu, L.; Wang, L.; Meng, F.; Li, R.; et al. Combined delivery of salinomycin and docetaxel by dual-targeting gelatinase nanoparticles effectively inhibits cervical cancer cells and cancer stem cells. Drug Deliv. 2021, 28, 510–519. [Google Scholar] [CrossRef]
- Duan, H.; Liu, Y.; Gao, Z.; Huang, W. Recent advances in drug delivery systems for targeting cancer stem cells. Acta Pharm. Sin. B 2021, 11, 55–70. [Google Scholar] [CrossRef]
- Deonarain, M.P.; Kousparou, C.A.; Epenetos, A.A. Antibodies targeting cancer stem cells: A new paradigm in immunotherapy? MAbs 2009, 1, 12–25. [Google Scholar] [CrossRef]
| Study | Sample | CSC Marker(s) and/or Phenotype(s) | CSC Characteristics and/or Clinical Significance |
|---|---|---|---|
| Feng et al., 2009 [26] | Primary tumor sphere culture | CD44+/CK17+ | Chemoresistance; tumorigenicity |
| Bortolomai et al., 2010 [27] | 3 cell lines; xenografts | ALDHhigh; SP | Sphere formation; tumorigenicity |
| López et al., 2012 [28] | 4 cell lines; xenografts | CD49f+ | Sphere formation; radioresistance; tumorigenicity |
| Zhang et al., 2012 [29] | HeLa cells; xenografts | SP | Increased invasiveness; tumorigenicity |
| Wang et al., 2013 [30] | HeLa cells; xenografts | SP | Colony formation; radio- and chemo-resistance; tumorigenicity |
| Liu & Zheng, 2013 [31] | 4 cell lines and 5 primary tumor xenografts | ALDHhigh | Chemoresistance; tumorigenicity |
| Qi et al., 2014 [32] | HeLa cells; xenografts | SP | Radio- and chemo-resistance; tumorigenicity |
| Wang et al., 2014 [33] | HeLa cells | OCT4, SOX2 and ALDH | Colony formation; sphere formation; chemoresistance |
| Villanueva-Toledo et al., 2014 [34] | 3 cell lines | SP | Colony formation; sphere formation |
| Liu et al., 2014 [35] | 2 cell lines; xenografts | SOX2 | Sphere formation; tumorigenicity |
| Kumazawa et al., 2014 [36] | HeLa cells; xenografts | CXCR4, Oct3/4, CD133, and SOX2 | Sphere formation; radioresistance; tumorigenicity |
| Hou et al., 2015 [37] | 179 tissue specimens | MSI1, ALDH1, SOX2 and CD49f | High expression of MSI1, ALDH1, and SOX2, and low expression of CD49f predict poor prognosis inspite ofpostoperative chemotherapy |
| Liu et al., 2016 [38] | SiHa cells; xenografts | CD44+/CD24+ | Sphere formation; radioresistance; tumorigenicity |
| Ortiz-Sánchez et al., 2016 [39] | 4 cell lines; xenografts | CK-17+, p63+, CD49f+, ALDHhigh | Sphere formation; tumorigenicity |
| Xie et al., 2016 [40] | 52 tumor samples | ALDH1 | ALDH1 expression predicts chemoresistance and poor clinical outcomes in patients with LACC receiving NAC prior to radical hysterectomy |
| Wei et al., 2017 [41] | Primary cell cultures | SP | Colony formation; tumorigenicity |
| Javed et al., 2018 [42] | Primary cell cultures | CD133+ | Sphere formation; EMT and radioresistance |
| Li et al., 2019 [43] | 6 cell lines; xenografts; 233 tissue specimens | NUSAP1 | Sphere formation; EMT and tumorigenicity. High expression of NUSAP1 positively correlated with lymph node metastasis. Patients with high NUSAP1 expression have shorter 5-year metastasis-free survival |
| Yao et al, 2020 [44] | 2 cell lines; xenografts | ALDHhigh | Sphere formation; radioresistance; tumorigenicity |
| Cellular Mechanism | Cancer Therapeutic Resistance | Therapeutic Approach | Example of Therapeutic Approach to CC |
|---|---|---|---|
| High DNA repair capacity and activation of anti-apoptotic pathways | Chemo- and radioresistance | Inhibition of the DNA damage checkpoints CHK1 and CHK2; targeting self-renewal and survival-related pathways (e.g. WNT/β-catenin, Hedgehog, Notch and PI3K/AKT/mTOR pathways); anti-apoptotic Bcl-2 family proteins; PARP family of enzymes [11,45,46,53,54,55,56] | PARP inhibitors (e.g. veliparib, olaparib, niraparib and rucaparib) are currently being studied [57] |
| Cell quiescence | Chemo- and radioresistance | Allowing cells to remain dormant indefinitely; reactivating dormant cells; eradicating dormant cells [58] | Inhibition of cytosolic phospholipase A2 alpha (cPLA2α) with efipladib improves chemosensitivity [59] |
| EMT | Chemo- and radioresistance | Targeting factors (e.g. cytokines, proteins, miRNAs, transcription factors, miRNA) and signaling pathways involved in EMT [60,61] | Plant products (e.g. anthocyanins, morusin and curcumin) inhibit EMT [62] |
| Tumor environment | Chemo- and radioresistance | Targeting the components of the tumor microenvironment (e.g. CAFs or TAMs) [61,63] | The upregulation of miR-125a sensitized to paclitaxel and cisplatin [64] |
| Hypoxia | Chemo- and radioresistance | Tumor oxygenation and oxygen therapeutics [65,66,67] | Hyperbaric oxygen and radiotherapy [68,69,70] |
| Multidrug resistance (MDR) | Chemoresistance | Inhibiting ABC transporters [71,72,73] | Stemofoline increases chemosensitivity by inhibiting P-glycoprotein [74] |
| ALDH-associated resistance | Chemo- and radioresistance | Inhibiting ALDHs [75,76,77,78,79,80,81,82] | Disulfiram-loaded vaginal ring potentially used for the localised treatment of CC [83] |
| Epigenetic Programming (e.g. epigenetic mechanisms, abnormal expression of miRNAs) | Chemo- and radioresistance | Inhibiting DNMTs and HDACs; manipulating miRNAs [84,85,86,87,88,89,90] | SGI-1027, a DNMT1 inhibitor, impairs CC cell propagation [85]. HDAC inhibitors (e.g. vorinostat, valproic acid, oxamflatin, 2-Oxo-1,3-thiazolidine, etc) may add to the efficiency of CSC therapy [86,87]. The miR-302–367 cluster [91], miR-23b [92] and miR-145 may serve as potential therapeutic reagents [93] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Fiore, R.; Suleiman, S.; Drago-Ferrante, R.; Subbannayya, Y.; Pentimalli, F.; Giordano, A.; Calleja-Agius, J. Cancer Stem Cells and Their Possible Implications in Cervical Cancer: A Short Review. Int. J. Mol. Sci. 2022, 23, 5167. https://doi.org/10.3390/ijms23095167
Di Fiore R, Suleiman S, Drago-Ferrante R, Subbannayya Y, Pentimalli F, Giordano A, Calleja-Agius J. Cancer Stem Cells and Their Possible Implications in Cervical Cancer: A Short Review. International Journal of Molecular Sciences. 2022; 23(9):5167. https://doi.org/10.3390/ijms23095167
Chicago/Turabian StyleDi Fiore, Riccardo, Sherif Suleiman, Rosa Drago-Ferrante, Yashwanth Subbannayya, Francesca Pentimalli, Antonio Giordano, and Jean Calleja-Agius. 2022. "Cancer Stem Cells and Their Possible Implications in Cervical Cancer: A Short Review" International Journal of Molecular Sciences 23, no. 9: 5167. https://doi.org/10.3390/ijms23095167
APA StyleDi Fiore, R., Suleiman, S., Drago-Ferrante, R., Subbannayya, Y., Pentimalli, F., Giordano, A., & Calleja-Agius, J. (2022). Cancer Stem Cells and Their Possible Implications in Cervical Cancer: A Short Review. International Journal of Molecular Sciences, 23(9), 5167. https://doi.org/10.3390/ijms23095167









