Osteoarthritis Pain
Abstract
1. Introduction
2. Materials and Methods
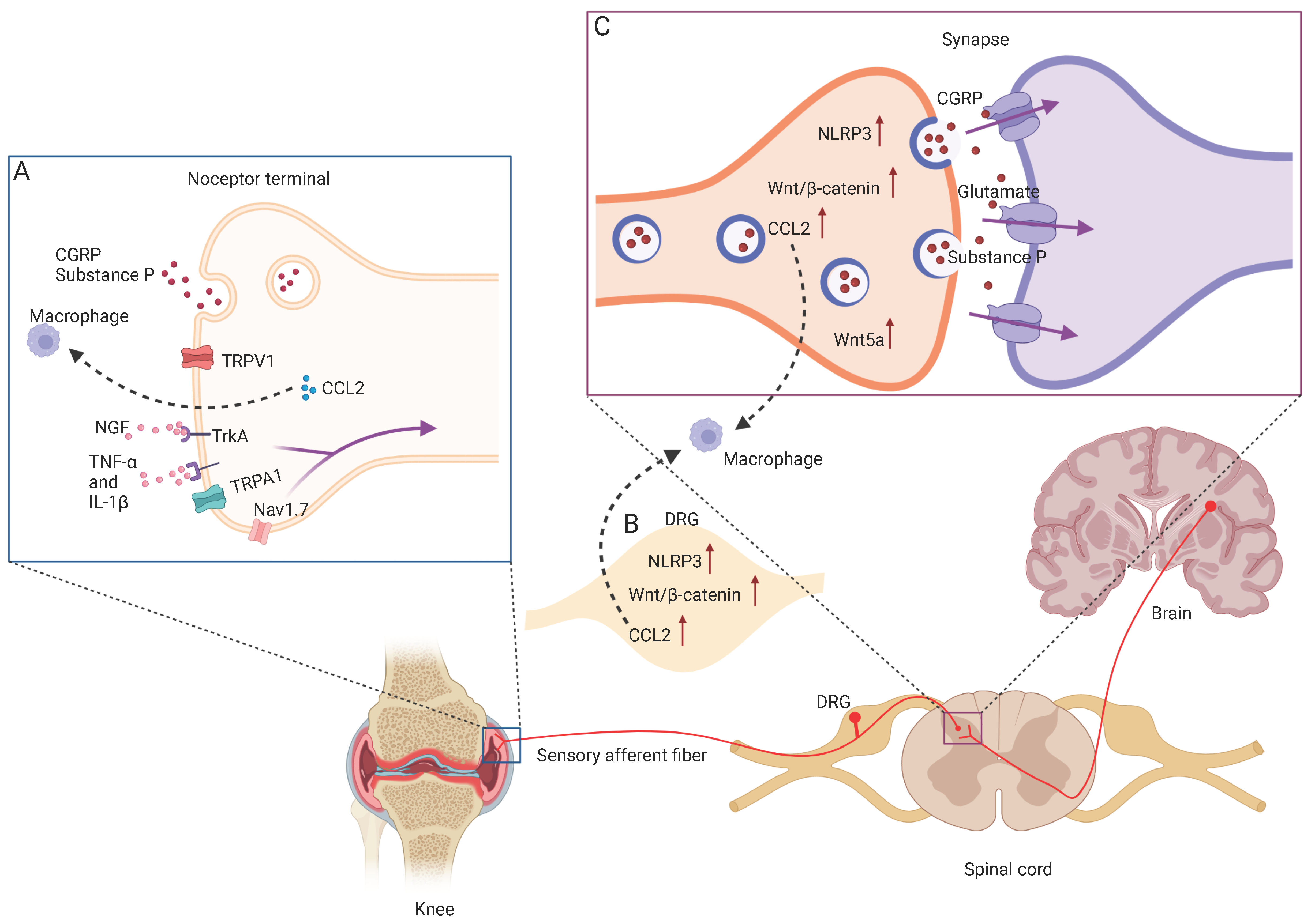
3. Characteristics and Structural Correlation of OA Pain
4. Mechanisms of OA Pain
4.1. NGF/TrkA Signaling
4.2. CGRP
4.3. CCL2/CCR2
4.4. TNF-α and IL-1β
4.5. NLRP3 Inflammasome
4.6. Wnt/β-Catenin
5. Common Animal Models for OA Pain Research
6. Emerging Preclinical Drugs Targeting OA Pain
6.1. Nerve Growth Factor Inhibitor
6.2. TNF Antibody
6.3. IL-1β Antibody
6.4. Wnt Inhibitor
6.5. NLRP3 Inhibitor
6.6. Ion Channels Modulator
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar] [CrossRef]
- Safiri, S.; Kolahi, A.A.; Smith, E.; Hill, C.; Bettampadi, D.; Mansournia, M.A.; Hoy, D.; Ashrafi-Asgarabad, A.; Sepidarkish, M.; Almasi-Hashiani, A.; et al. Global, regional and national burden of osteoarthritis 1990–2017: A systematic analysis of the Global Burden of Disease Study 2017. Ann. Rheum. Dis. 2020, 79, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Shen, J.; Zhao, W.; Wang, T.; Han, L.; Hamilton, J.L.; Im, H.J. Osteoarthritis: Toward a comprehensive understanding of pathological mechanism. Bone Res. 2017, 5, 16044. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Ma, F.; Yi, D.; Yu, H.; Tong, L.; Chen, D. Molecular signaling in temporomandibular joint osteoarthritis. J. Orthop. Transl. 2022, 32, 21–27. [Google Scholar] [CrossRef]
- Yao, X.; Sun, K.; Yu, S.; Luo, J.; Guo, J.; Lin, J.; Wang, G.; Guo, Z.; Ye, Y.; Guo, F. Chondrocyte ferroptosis contribute to the progression of osteoarthritis. J. Orthop. Transl. 2021, 27, 33–43. [Google Scholar] [CrossRef]
- Chen, D.; Kim, D.J.; Shen, J.; Zou, Z.; O’Keefe, R.J. Runx2 plays a central role in Osteoarthritis development. J. Orthop. Transl. 2020, 23, 132–139. [Google Scholar] [CrossRef]
- Groenewald, C.B.; Murray, C.B.; Battaglia, M.; Scaini, M.S.; Quinn, P.D. Prevalence of pain management techniques among adults with chronic pain in the United States, 2019. JAMA Netw. Open 2022, 5, e2146697. [Google Scholar] [CrossRef]
- Pitcher, M.H.; Von Korff, M.; Bushnell, M.C.; Porter, L. Prevalence and profile of high-impact chronic pain in the United States. J. Pain 2019, 20, 146–160. [Google Scholar] [CrossRef]
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.S.; Ringkamp, M.; Sluka, K.A.; et al. The revised International Association for the Study of Pain definition of pain: Concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097. [Google Scholar] [CrossRef]
- Chen, S.; Chen, M.; Wu, X.; Lin, S.; Tao, C.; Cao, H.; Shao, Z.; Xiao, G. Global, regional and national burden of low back pain 1990–2019: A systematic analysis of the Global Burden of Disease study 2019. J. Orthop. Transl. 2022, 32, 49–58. [Google Scholar] [CrossRef]
- Song, J.; Chang, A.H.; Chang, R.W.; Lee, J.; Pinto, D.; Hawker, G.; Nevitt, M.; Dunlop, D.D. Relationship of knee pain to time in moderate and light physical activities: Data from Osteoarthritis Initiative. Semin. Arthritis Rheum. 2018, 47, 683–688. [Google Scholar] [CrossRef]
- Belluzzi, E.; Stocco, E.; Pozzuoli, A.; Granzotto, M.; Porzionato, A.; Vettor, R.; De Caro, R.; Ruggieri, P.; Ramonda, R.; Rossato, M.; et al. Contribution of infrapatellar fat pad and synovial membrane to knee osteoarthritis pain. Biomed Res. Int. 2019, 2019, 6390182. [Google Scholar] [CrossRef]
- Miller, R.J.; Malfait, A.M.; Miller, R.E. The innate immune response as a mediator of osteoarthritis pain. Osteoarthr. Cartil. 2020, 28, 562–571. [Google Scholar] [CrossRef]
- Neogi, T. The epidemiology and impact of pain in osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1145–1153. [Google Scholar] [CrossRef]
- Mezey, G.A.; Mate, Z.; Paulik, E. Factors influencing pain management of patients with osteoarthritis: A cross-sectional study. J. Clin. Med. 2022, 11, 1352. [Google Scholar] [CrossRef]
- Crow, J.A.; Fillingim, R.B. Working toward mechanistic pain phenotyping in osteoarthritis. Osteoarthr. Cartil. 2022, 30, 495–497. [Google Scholar] [CrossRef]
- Salaffi, F.; Ciapetti, A.; Carotti, M. The sources of pain in osteoarthritis: A pathophysiological review. Reumatismo 2014, 66, 57–71. [Google Scholar] [CrossRef]
- Zhang, Y.; Nevitt, M.; Niu, J.; Lewis, C.; Torner, J.; Guermazi, A.; Roemer, F.; McCulloch, C.; Felson, D.T. Fluctuation of knee pain and changes in bone marrow lesions, effusions, and synovitis on magnetic resonance imaging. Arthritis Rheum. 2011, 63, 691–699. [Google Scholar] [CrossRef]
- Morgan, M.; Thai, J.; Nazemian, V.; Song, R.; Ivanusic, J.J. Changes to the activity and sensitivity of nerves innervating subchondral bone contribute to pain in late-stage osteoarthritis. Pain 2022, 163, 390–402. [Google Scholar] [CrossRef]
- Zhu, S.; Zhu, J.; Zhen, G.; Hu, Y.; An, S.; Li, Y.; Zheng, Q.; Chen, Z.; Yang, Y.; Wan, M.; et al. Subchondral bone osteoclasts induce sensory innervation and osteoarthritis pain. J. Clin. Investig. 2019, 129, 1076–1093. [Google Scholar] [CrossRef]
- Davidson, E.N.B.; van Caam, A.P.; Vitters, E.L.; Bennink, M.B.; Thijssen, E.; van den Berg, W.B.; Koenders, M.I.; van Lent, P.L.; van de Loo, F.A.; van der Kraan, P.M. TGF-beta is a potent inducer of Nerve Growth Factor in articular cartilage via the ALK5-Smad2/3 pathway. Potential role in OA related pain? Osteoarthr. Cartil. 2015, 23, 478–486. [Google Scholar] [CrossRef]
- Hill, C.L.; Gale, D.G.; Chaisson, C.E.; Skinner, K.; Kazis, L.; Gale, M.E.; Felson, D.T. Knee effusions, popliteal cysts, and synovial thickening: Association with knee pain in osteoarthritis. J. Rheumatol. 2001, 28, 1330–1337. [Google Scholar]
- Simkin, P.A. Bone pain and pressure in osteoarthritic joints. Novartis Found. Symp. 2004, 260, 179–186; discussion 186–190, 277–279. [Google Scholar]
- Torres, L.; Dunlop, D.D.; Peterfy, C.; Guermazi, A.; Prasad, P.; Hayes, K.W.; Song, J.; Cahue, S.; Chang, A.; Marshall, M.; et al. The relationship between specific tissue lesions and pain severity in persons with knee osteoarthritis. Osteoarthr. Cartil. 2006, 14, 1033–1040. [Google Scholar] [CrossRef]
- Belluzzi, E.; Macchi, V.; Fontanella, C.G.; Carniel, E.L.; Olivotto, E.; Filardo, G.; Sarasin, G.; Porzionato, A.; Granzotto, M.; Pozzuoli, A.; et al. Infrapatellar fat pad gene expression and protein production in patients with and without osteoarthritis. Int. J. Mol. Sci. 2020, 21, 6016. [Google Scholar] [CrossRef]
- Bamps, D.; Vriens, J.; de Hoon, J.; Voets, T. TRP channel cooperation for nociception: Therapeutic opportunities. Annu. Rev. Pharmacol. Toxicol. 2021, 61, 655–677. [Google Scholar] [CrossRef]
- Vincent, T.L. Peripheral pain mechanisms in osteoarthritis. Pain 2020, 161, S138–S146. [Google Scholar] [CrossRef]
- Stockl, S.; Eitner, A.; Bauer, R.J.; Konig, M.; Johnstone, B.; Grassel, S. Substance P and Alpha-calcitonin gene-related peptide differentially affect human osteoarthritic and healthy chondrocytes. Front. Immunol. 2021, 12, 722884. [Google Scholar] [CrossRef]
- Soni, A.; Wanigasekera, V.; Mezue, M.; Cooper, C.; Javaid, M.K.; Andrew, J.P.; Tracey, I. Central sensitization in knee osteoarthritis: Relating presurgical brainstem neuroimaging and PainDETECT-based patient stratification to arthroplasty outcome. Arthritis Rheumatol. 2019, 71, 550–560. [Google Scholar] [CrossRef]
- Fu, K.; Robbins, S.R.; McDougall, J.J. Osteoarthritis: The genesis of pain. Rheumatology 2018, 57, iv43–iv50. [Google Scholar] [CrossRef]
- Hochman, J.R.; French, M.R.; Bermingham, S.L.; Hawker, G.A. The nerve of osteoarthritis pain. Arthritis Care Res. 2010, 62, 1019–1023. [Google Scholar] [CrossRef]
- Eitner, A.; Hofmann, G.O.; Schaible, H.G. Mechanisms of osteoarthritic pain. Studies in humans and experimental models. Front. Mol. Neurosci. 2017, 10, 349. [Google Scholar] [CrossRef]
- Dainese, P.; Wyngaert, K.V.; De Mits, S.; Wittoek, R.; Van Ginckel, A.; Calders, P. Association between knee inflammation and knee pain in patients with knee osteoarthritis: A systematic review. Osteoarthr. Cartil. 2021, 30, 516–534. [Google Scholar] [CrossRef]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef]
- Miller, R.E.; Miller, R.J.; Malfait, A.M. Osteoarthritis joint pain: The cytokine connection. Cytokine 2014, 70, 185–193. [Google Scholar] [CrossRef]
- Hattori, T.; Shimo, K.; Niwa, Y.; Tokiwa, Y.; Matsubara, T. Association of chronic pain with radiologic severity and central sensitization in hip osteoarthritis patients. J. Pain Res. 2021, 14, 1153–1160. [Google Scholar] [CrossRef]
- Bjurstrom, M.F.; Blennow, K.; Zetterberg, H.; Bodelsson, M.; Walden, M.; Dietz, N.; Hall, S.; Hansson, O.; Irwin, M.R.; Mattsson-Carlgren, N. Central nervous system monoaminergic activity in hip osteoarthritis patients with disabling pain: Associations with pain severity and central sensitization. Pain Rep. 2022, 7, e988. [Google Scholar] [CrossRef]
- Ohashi, Y.; Fukushima, K.; Inoue, G.; Uchida, K.; Koyama, T.; Tsuchiya, M.; Uchiyama, K.; Takahira, N.; Takaso, M. Central sensitization inventory scores correlate with pain at rest in patients with hip osteoarthritis: A retrospective study. BMC Musculoskelet. Disord. 2020, 21, 595. [Google Scholar] [CrossRef]
- Ohashi, Y.; Fukushima, K.; Uchida, K.; Koyama, T.; Tsuchiya, M.; Saito, H.; Uchiyama, K.; Takahira, N.; Inoue, G.; Takaso, M. Adverse effects of higher preoperative pain at rest, a central sensitization-related symptom, on outcomes after total hip arthroplasty in patients with osteoarthritis. J. Pain Res. 2021, 14, 3345–3352. [Google Scholar] [CrossRef]
- Hempstead, B.L.; Martin-Zanca, D.; Kaplan, D.R.; Parada, L.F.; Chao, M.V. High-affinity NGF binding requires coexpression of the trk proto-oncogene and the low-affinity NGF receptor. Nature 1991, 350, 678–683. [Google Scholar] [CrossRef]
- Malfait, A.M.; Miller, R.E.; Miller, R.J. Basic mechanisms of pain in osteoarthritis: Experimental observations and new perspectives. Rheum. Dis. Clin. N. Am. 2021, 47, 165–180. [Google Scholar] [CrossRef]
- Ohashi, Y.; Uchida, K.; Fukushima, K.; Satoh, M.; Koyama, T.; Tsuchiya, M.; Saito, H.; Takahira, N.; Inoue, G.; Takaso, M. NGF expression and elevation in hip osteoarthritis patients with pain and central sensitization. Biomed Res. Int. 2021, 2021, 9212585. [Google Scholar] [CrossRef]
- Skaper, S.D. Nerve growth factor: A neuroimmune crosstalk mediator for all seasons. Immunology 2017, 151, 1–15. [Google Scholar] [CrossRef]
- McNamee, K.E.; Burleigh, A.; Gompels, L.L.; Feldmann, M.; Allen, S.J.; Williams, R.O.; Dawbarn, D.; Vincent, T.L.; Inglis, J.J. Treatment of murine osteoarthritis with TrkAd5 reveals a pivotal role for nerve growth factor in non-inflammatory joint pain. Pain 2010, 149, 386–392. [Google Scholar] [CrossRef]
- Ohashi, Y.; Uchida, K.; Fukushima, K.; Satoh, M.; Koyama, T.; Tsuchiya, M.; Saito, H.; Uchiyama, K.; Takahira, N.; Inoue, G.; et al. Correlation between CD163 expression and resting pain in patients with hip osteoarthritis: Possible contribution of CD163+ monocytes/macrophages to pain pathogenesis. J. Orthop. Res. 2021. [Google Scholar] [CrossRef]
- Kc, R.; Li, X.; Kroin, J.S.; Liu, Z.; Chen, D.; Xiao, G.; Levine, B.; Li, J.; Hamilton, J.L.; van Wijnen, A.J.; et al. PKCdelta null mutations in a mouse model of osteoarthritis alter osteoarthritic pain independently of joint pathology by augmenting NGF/TrkA-induced axonal outgrowth. Ann. Rheum. Dis. 2016, 75, 2133–2141. [Google Scholar] [CrossRef]
- Zhao, L.; Huang, J.; Fan, Y.; Li, J.; You, T.; He, S.; Xiao, G.; Chen, D. Exploration of CRISPR/Cas9-based gene editing as therapy for osteoarthritis. Ann. Rheum. Dis. 2019, 78, 676–682. [Google Scholar] [CrossRef]
- Dirmeier, M.; Capellino, S.; Schubert, T.; Angele, P.; Anders, S.; Straub, R.H. Lower density of synovial nerve fibres positive for calcitonin gene-related peptide relative to substance P in rheumatoid arthritis but not in osteoarthritis. Rheumatology 2008, 47, 36–40. [Google Scholar] [CrossRef][Green Version]
- Iyengar, S.; Ossipov, M.H.; Johnson, K.W. The role of calcitonin gene-related peptide in peripheral and central pain mechanisms including migraine. Pain 2017, 158, 543–559. [Google Scholar] [CrossRef]
- Russo, A.F. Calcitonin gene-related peptide (CGRP): A new target for migraine. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 533–552. [Google Scholar] [CrossRef]
- Walsh, D.A.; Mapp, P.I.; Kelly, S. Calcitonin gene-related peptide in the joint: Contributions to pain and inflammation. Br. J. Clin. Pharmacol. 2015, 80, 965–978. [Google Scholar] [CrossRef]
- Bullock, C.M.; Wookey, P.; Bennett, A.; Mobasheri, A.; Dickerson, I.; Kelly, S. Peripheral calcitonin gene-related peptide receptor activation and mechanical sensitization of the joint in rat models of osteoarthritis pain. Arthritis Rheumatol. 2014, 66, 2188–2200. [Google Scholar] [CrossRef]
- Hershey, A.D. CGRP—The next frontier for migraine. N. Engl. J. Med. 2017, 377, 2190–2191. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Ruan, Y.C.; Yu, M.K.; O’Laughlin, M.; Wise, H.; Chen, D.; Tian, L.; Shi, D.; Wang, J.; et al. Implant-derived magnesium induces local neuronal production of CGRP to improve bone-fracture healing in rats. Nat. Med. 2016, 22, 1160–1169. [Google Scholar] [CrossRef]
- Dong, T.; Chang, H.; Zhang, F.; Chen, W.; Zhu, Y.; Wu, T.; Zhang, Y. Calcitonin gene-related peptide can be selected as a predictive biomarker on progression and prognosis of knee osteoarthritis. Int. Orthop. 2015, 39, 1237–1243. [Google Scholar] [CrossRef]
- Benschop, R.J.; Collins, E.C.; Darling, R.J.; Allan, B.W.; Leung, D.; Conner, E.M.; Nelson, J.; Gaynor, B.; Xu, J.; Wang, X.F.; et al. Development of a novel antibody to calcitonin gene-related peptide for the treatment of osteoarthritis-related pain. Osteoarthr. Cartil. 2014, 22, 578–585. [Google Scholar] [CrossRef]
- Powell, R.; Young, V.A.; Pryce, K.D.; Sheehan, G.D.; Bonsu, K.; Ahmed, A.; Bhattacharjee, A. Inhibiting endocytosis in CGRP+ nociceptors attenuates inflammatory pain-like behavior. Nat. Commun. 2021, 12, 5812. [Google Scholar] [CrossRef]
- De Logu, F.; Nassini, R.; Hegron, A.; Landini, L.; Jensen, D.D.; Latorre, R.; Ding, J.; Marini, M.; de Araujo, D.S.M.; Ramirez-Garcia, P.; et al. Schwann cell endosome CGRP signals elicit periorbital mechanical allodynia in mice. Nat. Commun. 2022, 13, 646. [Google Scholar] [CrossRef]
- Jin, Y.; Smith, C.; Monteith, D.; Brown, R.; Camporeale, A.; McNearney, T.A.; Deeg, M.A.; Raddad, E.; Xiao, N.; de la Pena, A.; et al. CGRP blockade by galcanezumab was not associated with reductions in signs and symptoms of knee osteoarthritis in a randomized clinical trial. Osteoarthr. Cartil. 2018, 26, 1609–1618. [Google Scholar] [CrossRef]
- Uchida, K.; Takano, S.; Takata, K.; Mukai, M.; Koyama, T.; Ohashi, Y.; Saito, H.; Takaso, M.; Miyagi, M.; Inoue, G. Differential synovial CGRP/RAMP1 expression in men and women with knee osteoarthritis. Cureus 2021, 13, e15483. [Google Scholar] [CrossRef]
- Yoshimura, T.; Robinson, E.A.; Tanaka, S.; Appella, E.; Kuratsu, J.; Leonard, E.J. Purification and amino acid analysis of two human glioma-derived monocyte chemoattractants. J. Exp. Med. 1989, 169, 1449–1459. [Google Scholar] [CrossRef]
- White, G.E.; Iqbal, A.J.; Greaves, D.R. CC chemokine receptors and chronic inflammation—Therapeutic opportunities and pharmacological challenges. Pharmacol. Rev. 2013, 65, 47–89. [Google Scholar] [CrossRef]
- Zhu, S.; Liu, M.; Bennett, S.; Wang, Z.; Pfleger, K.D.G.; Xu, J. The molecular structure and role of CCL2 (MCP-1) and C-C chemokine receptor CCR2 in skeletal biology and diseases. J. Cell. Physiol. 2021, 236, 7211–7222. [Google Scholar] [CrossRef]
- Miller, R.E.; Tran, P.B.; Das, R.; Ghoreishi-Haack, N.; Ren, D.; Miller, R.J.; Malfait, A.M. CCR2 chemokine receptor signaling mediates pain in experimental osteoarthritis. Proc. Natl. Acad. Sci. USA 2012, 109, 20602–20607. [Google Scholar] [CrossRef]
- Raghu, H.; Lepus, C.M.; Wang, Q.; Wong, H.H.; Lingampalli, N.; Oliviero, F.; Punzi, L.; Giori, N.J.; Goodman, S.B.; Chu, C.R.; et al. CCL2/CCR2, but not CCL5/CCR5, mediates monocyte recruitment, inflammation and cartilage destruction in osteoarthritis. Ann. Rheum. Dis. 2017, 76, 914–922. [Google Scholar] [CrossRef]
- Ishihara, S.; Obeidat, A.M.; Wokosin, D.L.; Ren, D.; Miller, R.J.; Malfait, A.M.; Miller, R.E. The role of intra-articular neuronal CCR2 receptors in knee joint pain associated with experimental osteoarthritis in mice. Arthritis Res. Ther. 2021, 23, 103. [Google Scholar] [CrossRef]
- Leung, Y.Y.; Huebner, J.L.; Haaland, B.; Wong, S.B.S.; Kraus, V.B. Synovial fluid pro-inflammatory profile differs according to the characteristics of knee pain. Osteoarthr. Cartil. 2017, 25, 1420–1427. [Google Scholar] [CrossRef]
- Robinson, W.H.; Lepus, C.M.; Wang, Q.; Raghu, H.; Mao, R.; Lindstrom, T.M.; Sokolove, J. Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 2016, 12, 580–592. [Google Scholar] [CrossRef]
- Kalaitzoglou, E.; Griffin, T.M.; Humphrey, M.B. Innate immune responses and osteoarthritis. Curr. Rheumatol. Rep. 2017, 19, 45. [Google Scholar] [CrossRef]
- Woodell-May, J.E.; Sommerfeld, S.D. Role of inflammation and the immune system in the progression of osteoarthritis. J. Orthop. Res. 2020, 38, 253–257. [Google Scholar] [CrossRef]
- Chevalier, X.; Goupille, P.; Beaulieu, A.D.; Burch, F.X.; Bensen, W.G.; Conrozier, T.; Loeuille, D.; Kivitz, A.J.; Silver, D.; Appleton, B.E. Intraarticular injection of anakinra in osteoarthritis of the knee: A multicenter, randomized, double-blind, placebo-controlled study. Arthritis Rheum. 2009, 61, 344–352. [Google Scholar] [CrossRef]
- Maksymowych, W.P.; Russell, A.S.; Chiu, P.; Yan, A.; Jones, N.; Clare, T.; Lambert, R.G. Targeting tumour necrosis factor alleviates signs and symptoms of inflammatory osteoarthritis of the knee. Arthritis Res. Ther. 2012, 14, R206. [Google Scholar] [CrossRef]
- Li, L.; Li, Z.; Li, Y.; Hu, X.; Zhang, Y.; Fan, P. Profiling of inflammatory mediators in the synovial fluid related to pain in knee osteoarthritis. BMC Musculoskelet. Disord. 2020, 21, 99. [Google Scholar] [CrossRef]
- Orita, S.; Ishikawa, T.; Miyagi, M.; Ochiai, N.; Inoue, G.; Eguchi, Y.; Kamoda, H.; Arai, G.; Suzuki, M.; Sakuma, Y.; et al. Percutaneously absorbed NSAIDs attenuate local production of proinflammatory cytokines and suppress the expression of c-Fos in the spinal cord of a rodent model of knee osteoarthritis. J. Orthop. Sci. 2012, 17, 77–86. [Google Scholar] [CrossRef]
- Chen, J.L.; Zhou, X.; Liu, B.L.; Wei, X.H.; Ding, H.L.; Lin, Z.J.; Zhan, H.L.; Yang, F.; Li, W.B.; Xie, J.C.; et al. Normalization of magnesium deficiency attenuated mechanical allodynia, depressive-like behaviors, and memory deficits associated with cyclophosphamide-induced cystitis by inhibiting TNF-alpha/NF-kappaB signaling in female rats. J. Neuroinflamm. 2020, 17, 99. [Google Scholar] [CrossRef]
- Richter, F.; Natura, G.; Loser, S.; Schmidt, K.; Viisanen, H.; Schaible, H.G. Tumor necrosis factor causes persistent sensitization of joint nociceptors to mechanical stimuli in rats. Arthritis Rheum. 2010, 62, 3806–3814. [Google Scholar] [CrossRef]
- Levescot, A.; Chang, M.H.; Schnell, J.; Nelson-Maney, N.; Yan, J.; Martinez-Bonet, M.; Grieshaber-Bouyer, R.; Lee, P.Y.; Wei, K.; Blaustein, R.B.; et al. IL-1beta-driven osteoclastogenic Tregs accelerate bone erosion in arthritis. J. Clin. Investig. 2021, 131, e141008. [Google Scholar] [CrossRef]
- Kusakabe, T.; Sawaji, Y.; Endo, K.; Suzuki, H.; Konishi, T.; Maekawa, A.; Murata, K.; Yamamoto, K. DUSP-1 induced by PGE2 and PGE1 attenuates IL-1beta-activated MAPK signaling, leading to suppression of NGF expression in human intervertebral disc cells. Int. J. Mol. Sci. 2021, 23, 371. [Google Scholar] [CrossRef]
- Eitner, L.; Ozgul, O.S.; Enax-Krumova, E.K.; Vollert, J.; Maier, C.; Hoffken, O. Conditioned pain modulation using painful cutaneous electrical stimulation or simply habituation? Eur. J. Pain. 2018, 22, 1281–1290. [Google Scholar] [CrossRef]
- Swanson, K.V.; Deng, M.; Ting, J.P. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef]
- Mangan, M.S.J.; Olhava, E.J.; Roush, W.R.; Seidel, H.M.; Glick, G.D.; Latz, E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 2018, 17, 588–606. [Google Scholar] [CrossRef]
- Yu, H.; Yao, S.; Zhou, C.; Fu, F.; Luo, H.; Du, W.; Jin, H.; Tong, P.; Chen, D.; Wu, C.; et al. Morroniside attenuates apoptosis and pyroptosis of chondrocytes and ameliorates osteoarthritic development by inhibiting NF-kappaB signaling. J. Ethnopharmacol. 2021, 266, 113447. [Google Scholar] [CrossRef]
- Liu, C.C.; Huang, Z.X.; Li, X.; Shen, K.F.; Liu, M.; Ouyang, H.D.; Zhang, S.B.; Ruan, Y.T.; Zhang, X.L.; Wu, S.L.; et al. Upregulation of NLRP3 via STAT3-dependent histone acetylation contributes to painful neuropathy induced by bortezomib. Exp. Neurol. 2018, 302, 104–111. [Google Scholar] [CrossRef]
- Grace, P.M.; Strand, K.A.; Galer, E.L.; Urban, D.J.; Wang, X.; Baratta, M.V.; Fabisiak, T.J.; Anderson, N.D.; Cheng, K.; Greene, L.I.; et al. Morphine paradoxically prolongs neuropathic pain in rats by amplifying spinal NLRP3 inflammasome activation. Proc. Natl. Acad. Sci. USA 2016, 113, E3441–E3450. [Google Scholar] [CrossRef]
- Cheng, F.; Yan, F.F.; Liu, Y.P.; Cong, Y.; Sun, K.F.; He, X.M. Dexmedetomidine inhibits the NF-kappaB pathway and NLRP3 inflammasome to attenuate papain-induced osteoarthritis in rats. Pharm. Biol. 2019, 57, 649–659. [Google Scholar] [CrossRef]
- Yuasa, T.; Otani, T.; Koike, T.; Iwamoto, M.; Enomoto-Iwamoto, M. Wnt/beta-catenin signaling stimulates matrix catabolic genes and activity in articular chondrocytes: Its possible role in joint degeneration. Lab. Investig. 2008, 88, 264–274. [Google Scholar] [CrossRef]
- Tamamura, Y.; Otani, T.; Kanatani, N.; Koyama, E.; Kitagaki, J.; Komori, T.; Yamada, Y.; Costantini, F.; Wakisaka, S.; Pacifici, M.; et al. Developmental regulation of Wnt/beta-catenin signals is required for growth plate assembly, cartilage integrity, and endochondral ossification. J. Biol. Chem. 2005, 280, 19185–19195. [Google Scholar] [CrossRef]
- Zhu, M.; Tang, D.; Wu, Q.; Hao, S.; Chen, M.; Xie, C.; Rosier, R.N.; O’Keefe, R.J.; Zuscik, M.; Chen, D. Activation of beta-catenin signaling in articular chondrocytes leads to osteoarthritis-like phenotype in adult beta-catenin conditional activation mice. J. Bone Miner. Res. 2009, 24, 12–21. [Google Scholar] [CrossRef]
- Palomer, E.; Martin-Flores, N.; Jolly, S.; Pascual-Vargas, P.; Benvegnu, S.; Podpolny, M.; Teo, S.; Vaher, K.; Saito, T.; Saido, T.C.; et al. Epigenetic repression of Wnt receptors in AD: A role for Sirtuin2-induced H4K16ac deacetylation of Frizzled1 and Frizzled7 promoters. Mol. Psychiatry 2022. [Google Scholar] [CrossRef]
- van Vliet, A.C.; Lee, J.; van der Poel, M.; Mason, M.R.J.; Noordermeer, J.N.; Fradkin, L.G.; Tannemaat, M.R.; Malessy, M.J.A.; Verhaagen, J.; De Winter, F. Coordinated changes in the expression of Wnt pathway genes following human and rat peripheral nerve injury. PLoS ONE 2021, 16, e0249748. [Google Scholar] [CrossRef]
- Patel, A.K.; Park, K.K.; Hackam, A.S. Wnt signaling promotes axonal regeneration following optic nerve injury in the mouse. Neuroscience 2017, 343, 372–383. [Google Scholar] [CrossRef]
- Kurimoto, S.; Jung, J.; Tapadia, M.; Lengfeld, J.; Agalliu, D.; Waterman, M.; Mozaffar, T.; Gupta, R. Activation of the Wnt/beta-catenin signaling cascade after traumatic nerve injury. Neuroscience 2015, 294, 101–108. [Google Scholar] [CrossRef]
- Zhou, Y.Q.; Tian, X.B.; Tian, Y.K.; Mei, W.; Liu, D.Q.; Ye, D.W. Wnt signaling: A prospective therapeutic target for chronic pain. Pharmacol. Ther. 2022, 231, 107984. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, Z. Effect of Wnt signaling pathway on pathogenesis and intervention of neuropathic pain. Exp. Ther. Med. 2018, 16, 3082–3088. [Google Scholar] [CrossRef]
- Simonetti, M.; Kuner, R. Spinal Wnt5a plays a key role in spinal dendritic spine remodeling in neuropathic and inflammatory pain models and in the proalgesic effects of peripheral Wnt3a. J. Neurosci. 2020, 40, 6664–6677. [Google Scholar] [CrossRef]
- Zhang, Z.L.; Yu, G.; Peng, J.; Wang, H.B.; Li, Y.L.; Liang, X.N.; Su, R.B.; Gong, Z.H. Wnt1/beta-catenin signaling upregulates spinal VGLUT2 expression to control neuropathic pain in mice. Neuropharmacology 2020, 164, 107869. [Google Scholar] [CrossRef]
- Kim, H.K.; Bae, J.; Lee, S.H.; Hwang, S.H.; Kim, M.S.; Kim, M.J.; Jun, S.; Cervantes, C.L.; Jung, Y.S.; Back, S.; et al. Blockers of Wnt3a, Wnt10a, or beta-catenin prevent chemotherapy-induced neuropathic pain in vivo. Neurotherapeutics 2021, 18, 601–614. [Google Scholar] [CrossRef]
- Resham, K.; Sharma, S.S. Pharmacologic inhibition of porcupine, disheveled, and beta-catenin in Wnt signaling pathway ameliorates diabetic peripheral neuropathy in rats. J. Pain 2019, 20, 1338–1352. [Google Scholar] [CrossRef]
- Zhang, Y.K.; Huang, Z.J.; Liu, S.; Liu, Y.P.; Song, A.A.; Song, X.J. WNT signaling underlies the pathogenesis of neuropathic pain in rodents. J. Clin. Investig. 2013, 123, 2268–2286. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, D.; Li, X.; Gao, B.; Sun, C.; Zhou, S.; Ma, Y.; Chen, X.; Xu, D. The Wnt/beta-catenin pathway regulated cytokines for pathological neuropathic pain in chronic compression of dorsal root ganglion model. Neural Plast. 2021, 2021, 6680192. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Chen, Y.; Yu, J.; Yin, D.; Liu, C.; Chen, X.; Zhang, D. TCF4 mediates the maintenance of neuropathic pain through Wnt/beta-catenin signaling following peripheral nerve injury in rats. J. Mol. Neurosci. 2015, 56, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Chen, Y.; Liu, R.; Li, W.; Hua, B.; Bao, Y. Wnt signaling pathways: A role in pain processing. Neuromolecular Med. 2022. [Google Scholar] [CrossRef]
- Tang, S.-J. Synaptic activity-regulated Wnt signaling in synaptic plasticity, glial function and chronic pain. CNS Neurol. Disord. Drug Targets 2014, 13, 737–744. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Y.P.; Huang, Z.J.; Zhang, Y.K.; Song, A.A.; Ma, P.C.; Song, X.J. Wnt/Ryk signaling contributes to neuropathic pain by regulating sensory neuron excitability and spinal synaptic plasticity in rats. Pain 2015, 156, 2572–2584. [Google Scholar] [CrossRef]
- Boakye, P.A.; Tang, S.J.; Smith, P.A. Mediators of neuropathic pain; focus on spinal microglia, CSF-1, BDNF, CCL21, TNF-alpha, Wnt ligands, and interleukin 1beta. Front. Pain Res. 2021, 2, 698157. [Google Scholar] [CrossRef]
- Alves, C.J.; Couto, M.; Sousa, D.M.; Magalhaes, A.; Neto, E.; Leitao, L.; Conceicao, F.; Monteiro, A.C.; Ribeiro-da-Silva, M.; Lamghari, M. Nociceptive mechanisms driving pain in a post-traumatic osteoarthritis mouse model. Sci. Rep. 2020, 10, 15271. [Google Scholar] [CrossRef]
- Miller, R.E.; Malfait, A.M. Osteoarthritis pain: What are we learning from animal models? Best Pract. Res. Clin. Rheumatol. 2017, 31, 676–687. [Google Scholar] [CrossRef]
- Xu, J.; Yan, L.; Yan, B.; Zhou, L.; Tong, P.; Shan, L. Osteoarthritis pain model induced by intra-articular injection of mono-iodoacetate in rats. J. Vis. Exp. 2020, 159, e60649. [Google Scholar] [CrossRef]
- Pitcher, T.; Sousa-Valente, J.; Malcangio, M. The monoiodoacetate model of osteoarthritis pain in the mouse. J. Vis. Exp. 2016, 111, 53746. [Google Scholar] [CrossRef]
- Liu, P.; Okun, A.; Ren, J.; Guo, R.C.; Ossipov, M.H.; Xie, J.; King, T.; Porreca, F. Ongoing pain in the MIA model of osteoarthritis. Neurosci. Lett. 2011, 493, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Blanchet, T.J.; Morris, E.A. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthr. Cartil. 2007, 15, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Fu, F.; Yao, S.; Luo, H.; Xu, T.; Jin, H.; Tong, P.; Chen, D.; Wu, C.; Ruan, H. Biomechanical, histologic, and molecular characteristics of graft-tunnel healing in a murine modified ACL reconstruction model. J. Orthop. Transl. 2020, 24, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Ruan, M.Z.; Patel, R.M.; Dawson, B.C.; Jiang, M.M.; Lee, B.H. Pain, motor and gait assessment of murine osteoarthritis in a cruciate ligament transection model. Osteoarthr. Cartil. 2013, 21, 1355–1364. [Google Scholar] [CrossRef]
- Zhang, L.; Li, M.; Li, X.; Liao, T.; Ma, Z.; Zhang, L.; Xing, R.; Wang, P.; Mao, J. Characteristics of sensory innervation in synovium of rats within different knee osteoarthritis models and the correlation between synovial fibrosis and hyperalgesia. J. Adv. Res. 2022, 35, 141–151. [Google Scholar] [CrossRef]
- Hwang, H.S.; Park, I.Y.; Hong, J.I.; Kim, J.R.; Kim, H.A. Comparison of joint degeneration and pain in male and female mice in DMM model of osteoarthritis. Osteoarthr. Cartil. 2021, 29, 728–738. [Google Scholar] [CrossRef]
- Li, J.; Zhang, B.; Liu, W.X.; Lu, K.; Pan, H.; Wang, T.; Oh, C.D.; Yi, D.; Huang, J.; Zhao, L.; et al. Metformin limits osteoarthritis development and progression through activation of AMPK signalling. Ann. Rheum. Dis. 2020, 79, 635–645. [Google Scholar] [CrossRef]
- Lampropoulou-Adamidou, K.; Lelovas, P.; Karadimas, E.V.; Liakou, C.; Triantafillopoulos, I.K.; Dontas, I.; Papaioannou, N.A. Useful animal models for the research of osteoarthritis. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 263–271. [Google Scholar] [CrossRef]
- Inglis, J.J.; McNamee, K.E.; Chia, S.L.; Essex, D.; Feldmann, M.; Williams, R.O.; Hunt, S.P.; Vincent, T. Regulation of pain sensitivity in experimental osteoarthritis by the endogenous peripheral opioid system. Arthritis Rheum. 2008, 58, 3110–3119. [Google Scholar] [CrossRef]
- Lane, N.E.; Schnitzer, T.J.; Birbara, C.A.; Mokhtarani, M.; Shelton, D.L.; Smith, M.D.; Brown, M.T. Tanezumab for the treatment of pain from osteoarthritis of the knee. N. Engl. J. Med. 2010, 363, 1521–1531. [Google Scholar] [CrossRef]
- Schnitzer, T.J.; Easton, R.; Pang, S.; Levinson, D.J.; Pixton, G.; Viktrup, L.; Davignon, I.; Brown, M.T.; West, C.R.; Verburg, K.M. Effect of tanezumab on joint pain, physical function, and patient global assessment of osteoarthritis among patients with osteoarthritis of the hip or knee: A randomized clinical trial. JAMA 2019, 322, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, F.; Blanco, F.J.; Guermazi, A.; Miki, K.; Yamabe, T.; Viktrup, L.; Junor, R.; Carey, W.; Brown, M.T.; West, C.R.; et al. Subcutaneous tanezumab for osteoarthritis of the hip or knee: Efficacy and safety results from a 24-week randomised phase III study with a 24-week follow-up period. Ann. Rheum. Dis. 2020, 79, 800–810. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, M.C.; Carrino, J.A.; Schnitzer, T.J.; Guermazi, A.; Walsh, D.A.; White, A.; Nakajo, S.; Fountaine, R.J.; Hickman, A.; Pixton, G.; et al. Long-term safety and efficacy of subcutaneous tanezumab versus nonsteroidal antiinflammatory drugs for hip or knee osteoarthritis: A randomized trial. Arthritis Rheumatol. 2021, 73, 1167–1177. [Google Scholar] [CrossRef] [PubMed]
- Dakin, P.; DiMartino, S.J.; Gao, H.; Maloney, J.; Kivitz, A.J.; Schnitzer, T.J.; Stahl, N.; Yancopoulos, G.D.; Geba, G.P. The efficacy, tolerability, and joint safety of fasinumab in osteoarthritis pain: A phase IIb/III double-blind, placebo-controlled, randomized clinical trial. Arthritis Rheumatol. 2019, 71, 1824–1834. [Google Scholar] [CrossRef] [PubMed]
- Ekman, E.F.; Gimbel, J.S.; Bello, A.E.; Smith, M.D.; Keller, D.S.; Annis, K.M.; Brown, M.T.; West, C.R.; Verburg, K.M. Efficacy and safety of intravenous tanezumab for the symptomatic treatment of osteoarthritis: 2 randomized controlled trials versus naproxen. J. Rheumatol. 2014, 41, 2249–2259. [Google Scholar] [CrossRef] [PubMed]
- Tive, L.; Bello, A.E.; Radin, D.; Schnitzer, T.J.; Nguyen, H.; Brown, M.T.; West, C.R. Pooled analysis of tanezumab efficacy and safety with subgroup analyses of phase III clinical trials in patients with osteoarthritis pain of the knee or hip. J. Pain Res. 2019, 12, 975–995. [Google Scholar] [CrossRef]
- Dakin, P.; Kivitz, A.J.; Gimbel, J.S.; Skrepnik, N.; DiMartino, S.J.; Emeremni, C.A.; Gao, H.; Stahl, N.; Weinreich, D.M.; Yancopoulos, G.D.; et al. Efficacy and safety of fasinumab in patients with chronic low back pain: A phase II/III randomised clinical trial. Ann. Rheum. Dis. 2020, 80, 509–517. [Google Scholar] [CrossRef]
- Watt, F.E.; Blauwet, M.B.; Fakhoury, A.; Jacobs, H.; Smulders, R.; Lane, N.E. Tropomyosin-related kinase A (TrkA) inhibition for the treatment of painful knee osteoarthritis: Results from a randomized controlled phase 2a trial. Osteoarthr. Cartil. 2019, 27, 1590–1598. [Google Scholar] [CrossRef]
- Aitken, D.; Laslett, L.L.; Pan, F.; Haugen, I.K.; Otahal, P.; Bellamy, N.; Bird, P.; Jones, G. A randomised double-blind placebo-controlled crossover trial of HUMira (adalimumab) for erosive hand OsteoaRthritis—The HUMOR trial. Osteoarthr. Cartil. 2018, 26, 880–887. [Google Scholar] [CrossRef]
- Chevalier, X.; Ravaud, P.; Maheu, E.; Baron, G.; Rialland, A.; Vergnaud, P.; Roux, C.; Maugars, Y.; Mulleman, D.; Lukas, C.; et al. Adalimumab in patients with hand osteoarthritis refractory to analgesics and NSAIDs: A randomised, multicentre, double-blind, placebo-controlled trial. Ann. Rheum. Dis. 2015, 74, 1697–1705. [Google Scholar] [CrossRef]
- Kloppenburg, M.; Peterfy, C.; Haugen, I.K.; Kroon, F.; Chen, S.; Wang, L.; Liu, W.; Levy, G.; Fleischmann, R.M.; Berenbaum, F.; et al. Phase IIa, placebo-controlled, randomised study of lutikizumab, an anti-interleukin-1alpha and anti-interleukin-1beta dual variable domain immunoglobulin, in patients with erosive hand osteoarthritis. Ann. Rheum Dis 2019, 78, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.B.; Proudman, S.; Kivitz, A.J.; Burch, F.X.; Donohue, J.P.; Burstein, D.; Sun, Y.N.; Banfield, C.; Vincent, M.S.; Ni, L.; et al. A randomized, double-blind study of AMG 108 (a fully human monoclonal antibody to IL-1R1) in patients with osteoarthritis of the knee. Arthritis Res. Ther. 2011, 13, R125. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, R.; Bliddal, H.; Blanco, F.; Schnitzer, T.; Peterfy, C.; Chen, S.; Wang, L.; Conaghan, P.; Berenbaum, F.; Pelletier, J.-P.; et al. SAT0575 Safety and efficacy of lutikizumab (ABT-981), an anti–interleukin-1 alpha/beta dual variable domain (DVD) immunoglobulin, in subjects with knee osteoarthritis: Results from the randomised, double-blind, placebo-controlled, parallel-group phase 2 trial. Ann. Rheum. Dis. 2018, 77, 1141–1142. [Google Scholar]
- Yazici, Y.; McAlindon, T.E.; Fleischmann, R.; Gibofsky, A.; Lane, N.E.; Kivitz, A.J.; Skrepnik, N.; Armas, E.; Swearingen, C.J.; DiFrancesco, A.; et al. A novel Wnt pathway inhibitor, SM04690, for the treatment of moderate to severe osteoarthritis of the knee: Results of a 24-week, randomized, controlled, phase 1 study. Osteoarthr. Cartil. 2017, 25, 1598–1606. [Google Scholar] [CrossRef]
- Hochberg, M.C. Serious joint-related adverse events in randomized controlled trials of anti-nerve growth factor monoclonal antibodies. Osteoarthr. Cartil. 2015, 23, S18–S21. [Google Scholar] [CrossRef]
- Krupka, E.; Jiang, G.L.; Jan, C. Efficacy and safety of intra-articular injection of tropomyosin receptor kinase A inhibitor in painful knee osteoarthritis: A randomized, double-blind and placebo-controlled study. Osteoarthr. Cartil. 2019, 27, 1599–1607. [Google Scholar] [CrossRef]
- Favero, M.; Belluzzi, E.; Ortolan, A.; Lorenzin, M.; Oliviero, F.; Doria, A.; Scanzello, C.R.; Ramonda, R. Erosive hand osteoarthritis: Latest findings and outlook. Nat. Rev. Rheumatol. 2022, 18, 171–183. [Google Scholar] [CrossRef]
- Wang, J. Efficacy and safety of adalimumab by intra-articular injection for moderate to severe knee osteoarthritis: An open-label randomized controlled trial. J. Int. Med. Res. 2018, 46, 326–334. [Google Scholar] [CrossRef]
- Varan, O.; Babaoglu, H.; Atas, N.; Satis, H.; Salman, R.B.; Tufan, A.; Goker, B. Response to anakinra in 3 patients with erosive osteoarthritis. J. Clin. Rheumatol. 2021, 27, e52–e53. [Google Scholar] [CrossRef]
- Bacconnier, L.; Jorgensen, C.; Fabre, S. Erosive osteoarthritis of the hand: Clinical experience with anakinra. Ann. Rheum Dis 2009, 68, 1078–1079. [Google Scholar] [CrossRef]
- Kosloski, M.P.; Goss, S.; Wang, S.X.; Liu, J.; Loebbert, R.; Medema, J.K.; Liu, W.; Dutta, S. Pharmacokinetics and tolerability of a dual variable domain immunoglobulin ABT-981 against IL-1alpha and IL-1beta in healthy subjects and patients with osteoarthritis of the knee. J. Clin. Pharmacol. 2016, 56, 1582–1590. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.X.; Abramson, S.B.; Attur, M.; Karsdal, M.A.; Preston, R.A.; Lozada, C.J.; Kosloski, M.P.; Hong, F.; Jiang, P.; Saltarelli, M.J.; et al. Safety, tolerability, and pharmacodynamics of an anti-interleukin-1alpha/beta dual variable domain immunoglobulin in patients with osteoarthritis of the knee: A randomized phase 1 study. Osteoarthr. Cartil. 2017, 25, 1952–1961. [Google Scholar] [CrossRef] [PubMed]
- Schieker, M.; Conaghan, P.G.; Mindeholm, L.; Praestgaard, J.; Solomon, D.H.; Scotti, C.; Gram, H.; Thuren, T.; Roubenoff, R.; Ridker, P.M. Effects of interleukin-1beta inhibition on incident hip and knee replacement: Exploratory analyses from a randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 2020, 173, 509–515. [Google Scholar] [CrossRef]
- Yazici, Y.; McAlindon, T.E.; Gibofsky, A.; Lane, N.E.; Clauw, D.; Jones, M.; Bergfeld, J.; Swearingen, C.J.; DiFrancesco, A.; Simsek, I.; et al. Lorecivivint, a novel intraarticular CDC-like kinase 2 and dual-specificity tyrosine phosphorylation-regulated kinase 1A inhibitor and Wnt pathway modulator for the treatment of knee osteoarthritis: A phase II randomized trial. Arthritis Rheumatol. 2020, 72, 1694–1706. [Google Scholar] [CrossRef] [PubMed]
- Yazici, Y.; McAlindon, T.E.; Gibofsky, A.; Lane, N.E.; Lattermann, C.; Skrepnik, N.; Swearingen, C.J.; Simsek, I.; Ghandehari, H.; DiFrancesco, A.; et al. A Phase 2b randomized trial of lorecivivint, a novel intra-articular CLK2/DYRK1A inhibitor and Wnt pathway modulator for knee osteoarthritis. Osteoarthr. Cartil. 2021, 29, 654–666. [Google Scholar] [CrossRef] [PubMed]
- Jansen, T.L.; Klück, V.; Janssen, M.; Comarniceanu, A.; Efdé, M.; Scribner, C.L.; Barrow, R.B.; Skouras, D.B.; Dinarello, C.A.; Joosten, L.A. P160 The first phase 2A proof-of-concept study of a selective NLRP3 inflammasome inhibitor, dapansutrile™ (OLT1177™), in acute gout. Ann. Rheum. Dis. 2019, 78, A70–A71. [Google Scholar]
- Stevens, R.M.; Ervin, J.; Nezzer, J.; Nieves, Y.; Guedes, K.; Burges, R.; Hanson, P.D.; Campbell, J.N. Randomized, double-blind, placebo-controlled trial of intraarticular trans-capsaicin for pain associated with osteoarthritis of the knee. Arthritis Rheumatol. 2019, 71, 1524–1533. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, L.; Liu, Z.; Zhang, L.; Xing, R.; Yin, S.; Li, X.; Zhang, N.; Wang, P. sanse powder essential oil nanoemulsion negatively regulates TRPA1 by AMPK/mTOR signaling in synovitis: Knee osteoarthritis rat model and fibroblast-like synoviocyte isolates. Mediat. Inflamm. 2021, 2021, 4736670. [Google Scholar] [CrossRef]
- Chen, J.; Joshi, S.K.; DiDomenico, S.; Perner, R.J.; Mikusa, J.P.; Gauvin, D.M.; Segreti, J.A.; Han, P.; Zhang, X.F.; Niforatos, W.; et al. Selective blockade of TRPA1 channel attenuates pathological pain without altering noxious cold sensation or body temperature regulation. Pain 2011, 152, 1165–1172. [Google Scholar] [CrossRef]
| Model | Species | Procedure | Mechanism of Model | Disease Onset | Advantages | Disadvantages | References |
|---|---|---|---|---|---|---|---|
| MIA | Rat, mouse | Intra-articular injection of MIA | Disrupt chondrocyte glycolysis via inhibiting glyceraldehyde-3-phosphatase dehydrogenase | 1 week | Rapid, reproducible, robust pain-like behaviour and peripheral/central sensitization partially characterized. | Extensive and rapidly developing pathology does not mimic human OA. | [109,110,111,112] |
| ACLT | Dog, Rat | Transection of ACL | Surgical destabilization of the knee | 2–3 weeks | Severe OA and subchondral bone destruction, though less rapidly than MIA model. | Technically difficult and time consuming. | [113,114,115] |
| DMM | Mouse | Transection of medial menisco-tibial ligament | Surgical destabilization of the knee | 4–8 weeks | Modest OA, less rapidly than ACLT and MIA model. DMM model amenable to genetic manipulation. | Less difficult than ACLT | [107,112,116,117] |
| Type of Drug | Drug Name | Route of Administration | ClinicalTrials.gov Identifier | Clinical Phase and Status | Affected OA Joint | Primary Measures or Results | References |
|---|---|---|---|---|---|---|---|
| CGRP antibody | Galcanezumab | S.C. | NCT02192190 | Phase 2, completed | knee | No improvement in WOMAC pain | [60] |
| IL-1 receptor antagonist | Anakinra | I.A. | NCT00110916 | Phase 2, completed | Knee | No improvements on OA symptoms | [72] |
| TNFα antibody | Adalimumab | S.C. | NCT00686439 | Phase 2, completed | knee | Improvement in WOMAC pain. | [73] |
| NGF inhibitor | Tanezumab | I.V. | NCT00394563 | Phase 2, completed | Knee | Reduction in joint pain and improvement in function | [120] |
| NGF inhibitor | Fasinumab | S.C. | NCT02709486, NCT02528188, NCT02697773 | Phase 3, completed | Knee, hip | Improvement in pain within the first week, and pain and function were improved throughout 24 weeks | [121,122,123] |
| S.C. | NCT02447276 | Phase 3, completed | Knee, hip | Improvements in OA pain and function | [124] | ||
| S.C. | NCT02683239, NCT03161093, NCT03304379 | Phase 3, completed | Knee, hip | WOMAC pain subscale score | |||
| Tanezumab | I.V. | NCT00863304, NCT00830063, NCT00744471 | Phase 3, completed | Knee, hip | Improvement of pain, physical function, and patient global assessment of OA | [125,126] | |
| TrkA inhibitor | ASP7962 | P.O. | NCT02611466 | Phase 2, completed | Knee | No improvement in WOMAC pain | [127] |
| GZ389988A | I.A. | NCT02845271 | Phase 2, completed | Knee | Improvement in WOMAC pain | [128] | |
| TNFα antibody | Adalimumab | S.C. | ACTRN12612000791831 | Phase 2, completed | Hand | No improvements on symptoms or bone marrow lesions | [129] |
| S.C. | NCT00597623 | Phase 3, completed | Hand | No improvement in WOMAC pain | [130] | ||
| CCR2 antagonist | CNTX-6970 | P.O. | NCT05025787 | Phase 2, recruiting | Knee | WOMAC pain | |
| NGF/TNF-α bispecific antibody | MEDI7352 | P.O. | NCT04675034 | Phase 2b, recruiting | Knee | NRS | |
| IL-1α/β antibody | Lutikizumab | S.C. | NCT02384538 | Phase 2, completed | Hand | No improvement in pain score | [131] |
| IL-1R1 antibody | AMG 108 | I.V. | NCT00110942 | Phase 2, completed | Knee | Minimal clinical benefit | [132] |
| IL-1α/β antibody | Lutikizumab | S.C. | NCT02087904 | Phase 2, completed | Knee | No improvement in WOMAC pain | [133] |
| NLRP3 inhibitor | DVF890 | P.O. | NCT04886258 | Phase 2a, recruiting | Knee | KOOS pain sub-scale | |
| Wnt inhibitor | Lorecivivint (SM04690) | I.A. | NCT04385303 (NCT03928184 *) | Phase 3, active, not recruiting | Knee | Improvement in NRS Pain | [134] |
| TRPV1 inhibitor | CNTX-4975 | I.A. | NCT03660943, NCT03661996 | Phase 3, completed | Knee | WOMAC | |
| NE06860 | P.O. | NCT02712957 | Phase 2, completed | Knee | NRS | ||
| TRPA1 antagonist | LY3526318 | P.O. | NCT05080660 | Phase 2, recruiting | Knee | NRS and WOMAC pain | |
| SCN9A # antisense drug | OLP1002 | S.C. | NCT05216341 | Phase 2, recruiting | Knee, hip | WOMAC and VAS | |
| Central analgesic | Cannabinoid | P.O. | NCT04992962 | Phase 2, recruiting | Knee | NRS and KOOS | |
| Tubulin inhibitor | Colchicine | P.O. | NCT03913442 | Phase 4, recruiting | Knee | VAS pain scores | |
| Aryl hydrocarbon receptor antagonist | Resvertrol | P.O. | NCT02905799 | Phase 3, recruiting | Knee | NRS | |
| Peroxynitrite decomposer | ACP044 | P.O. | NCT05008835 | Phase 2, recruiting | Knee | NRS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, H.; Huang, T.; Lu, W.W.; Tong, L.; Chen, D. Osteoarthritis Pain. Int. J. Mol. Sci. 2022, 23, 4642. https://doi.org/10.3390/ijms23094642
Yu H, Huang T, Lu WW, Tong L, Chen D. Osteoarthritis Pain. International Journal of Molecular Sciences. 2022; 23(9):4642. https://doi.org/10.3390/ijms23094642
Chicago/Turabian StyleYu, Huan, Tianwen Huang, William Weijia Lu, Liping Tong, and Di Chen. 2022. "Osteoarthritis Pain" International Journal of Molecular Sciences 23, no. 9: 4642. https://doi.org/10.3390/ijms23094642
APA StyleYu, H., Huang, T., Lu, W. W., Tong, L., & Chen, D. (2022). Osteoarthritis Pain. International Journal of Molecular Sciences, 23(9), 4642. https://doi.org/10.3390/ijms23094642







