Current Advancements in the Molecular Mechanism of Plasma Treatment for Seed Germination and Plant Growth
Abstract
1. Introduction
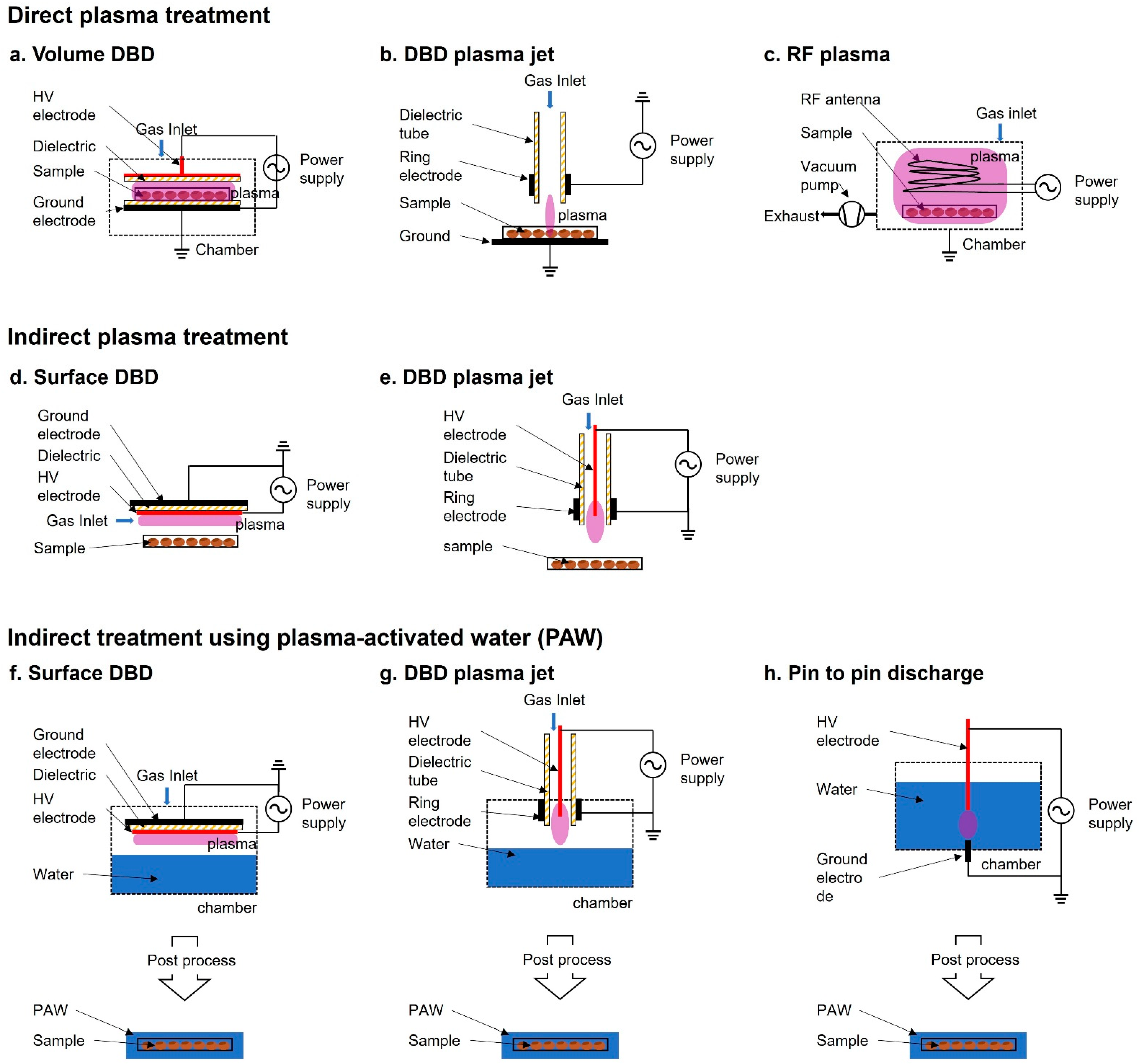
2. Plasma Treatment Method
3. Chemical and Physical Effects of Plasma Treatment
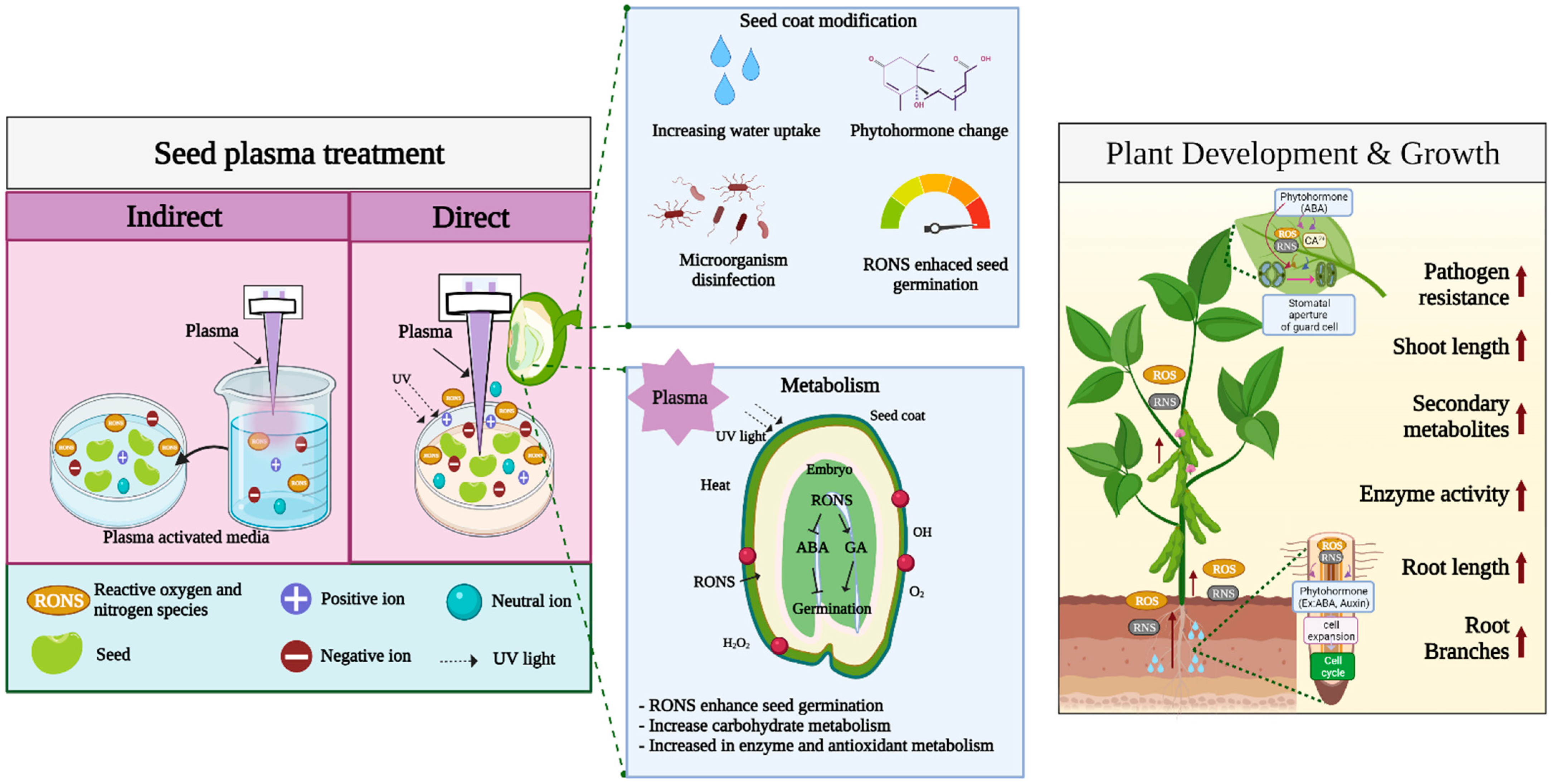
3.1. Chemical Effects of Plasma Treatment
3.2. Physical Effects of Plasma Treatment
4. Molecular Mechanism of Plasma Treatment
4.1. Regulation of Reactive Species
4.2. Effect of Plasma on Stress Response
4.3. Hormone and Metabolite Regulation
4.4. Transcriptome and Proteome Profiling
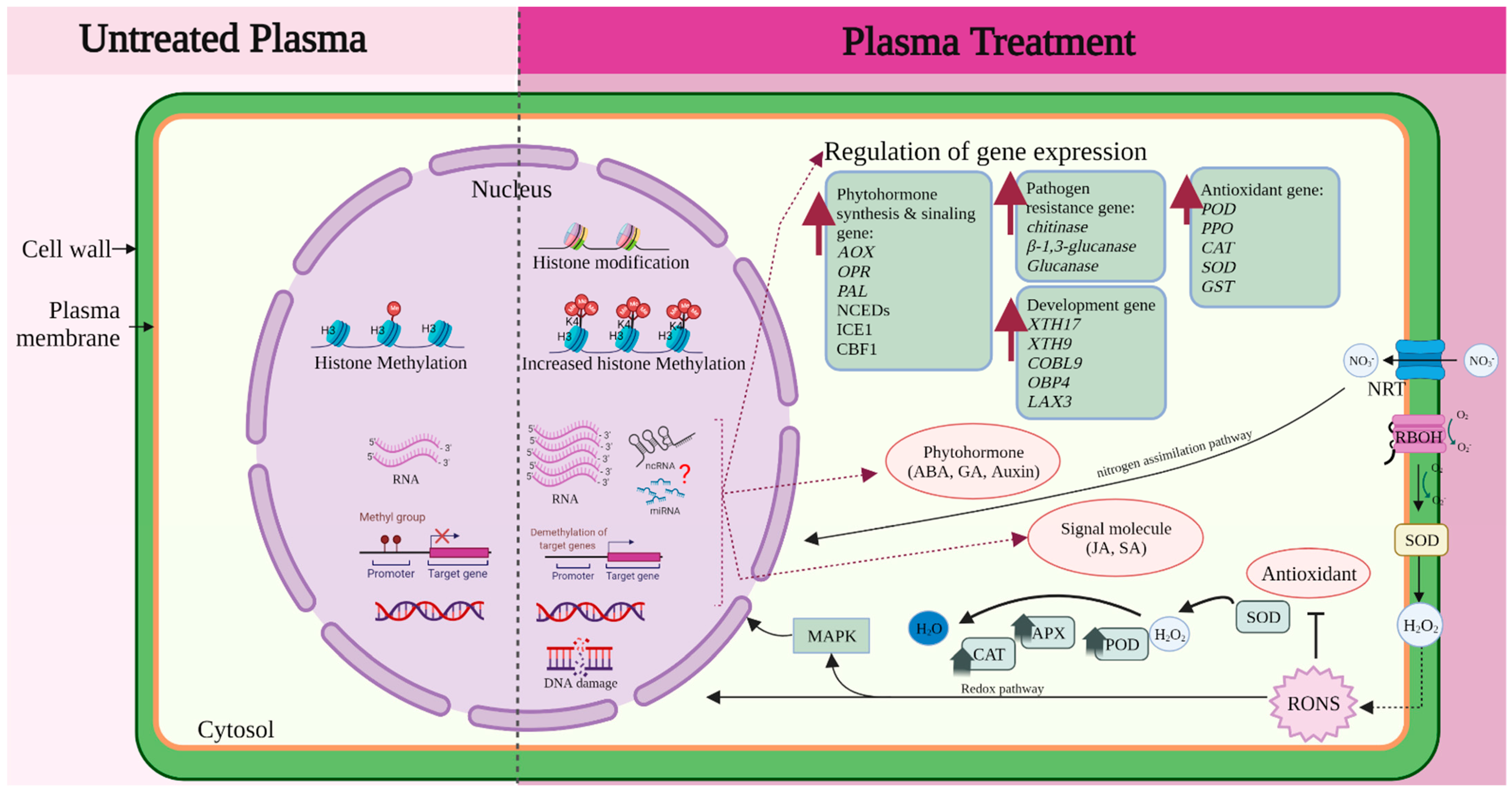
4.5. Epigenetic and Protein Expression
4.6. Beyond Gene Expression
5. Conclusions and Future Prospect
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Starič, P.; Vogel-Mikuš, K.; Mozetič, M.; Junkar, I. Effects of Nonthermal Plasma on Morphology, Genetics and Physiology of Seeds: A Review. Plants 2020, 9, 1736. [Google Scholar] [CrossRef]
- Yan, D.; Lin, L.; Zvansky, M.; Kohanzadeh, L.; Taban, S.; Chriqui, S.; Keidar, M. Improving Seed Germination by Cold Atmospheric Plasma. Plasma 2022, 5, 98–110. [Google Scholar] [CrossRef]
- Ikmal Misnal, M.F.; Redzuan, N.; Firdaus Zainal, M.N.; Raja Ibrahim, R.K.; Ahmad, N.; Agun, L. Emerging Cold Plasma Treatment on Rice Grains: A Mini Review. Chemosphere 2021, 274, 129972. [Google Scholar] [CrossRef] [PubMed]
- Nonogaki, H. Seed Dormancy and Germination—Emerging Mechanisms and New Hypotheses. Front. Plant Sci. 2014, 5, 233. [Google Scholar] [CrossRef] [PubMed]
- Tuan, P.A.; Sun, M.; Nguyen, T.-N.; Park, S.; Ayele, B.T. Molecular Mechanisms of Seed Germination. In Sprouted Grains; Feng, H., Nemzer, B., DeVries, J.W., Eds.; AACC International Press: St. Paul, MN, USA, 2019; pp. 1–24. ISBN 978-0-12-811525-1. [Google Scholar]
- Lutts, S.; Benincasa, P.; Wojtyla, L.; Kubala, S.; Pace, R.; Lechowska, K. Seed Priming: New Comprehensive Approaches for an Old Empirical Technique, New Challenges in Seed Biology-Basic and Translational Research Driving Seed Technology; InTechOpen: Rijeka, Croatia, 2016. [Google Scholar]
- Pawar, V.A.; Laware, S.L. Seed priming A critical review. Int. J. Sci. Res. Biol. Sci. 2018, 31, 94–101. [Google Scholar] [CrossRef]
- Zulfiqar, F. Effect of Seed Priming on Horticultural Crops. Sci. Hortic. 2021, 286, 110197. [Google Scholar] [CrossRef]
- Rasooli, Z.; Barzin, G.; Mahabadi, T.D.; Entezari, M. Stimulating Effects of Cold Plasma Seed Priming on Germination and Seedling Growth of Cumin Plant. South Afr. J. Bot. 2021, 142, 106–113. [Google Scholar] [CrossRef]
- Nalwa, C.; Thakur, A.K.; Vikram, A.; Rane, R.; Vaid, A. Studies on plasma treatment and priming of seeds of bell pepper (Capsicum annuum L.). J. Appl. Nat. Sci. 2017, 9, 1505–1509. [Google Scholar] [CrossRef]
- Darmanin, M.; Kozak, D.; de Oliveira Mallia, J.; Blundell, R.; Gatt, R.; Valdramidis, V.P. Generation of Plasma Functionalized Water: Antimicrobial Assessment and Impact on Seed Germination. Food Control 2020, 113, 107168. [Google Scholar] [CrossRef]
- Selcuk, M.; Oksuz, L.; Basaran, P. Decontamination of Grains and Legumes Infected with Aspergillus Spp. and Penicillum Spp. by Cold Plasma Treatment. Bioresour. Technol. 2008, 99, 5104–5109. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kabir, E.; Jahan, S.A. Exposure to Pesticides and the Associated Human Health Effects. Sci. Total Environ. 2017, 575, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Bafoil, M.; Jemmat, A.; Martinez, Y.; Merbahi, N.; Eichwald, O.; Dunand, C.; Yousfi, M. Effects of Low Temperature Plasmas and Plasma Activated Waters on Arabidopsis Thaliana Germination and Growth. PLoS ONE 2018, 13, e0195512. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.; Adhikari, M.; Ghimire, B.; Adhikari, B.C.; Park, G.; Choi, E.H. Cold Plasma Seed Priming Modulates Growth, Redox Homeostasis and Stress Response by Inducing Reactive Species in Tomato (Solanum lycopersicum). Free Radic. Biol. Med. 2020, 156, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.; Adhikari, M.; Park, G. The Effects of Plasma on Plant Growth, Development, and Sustainability. Appl. Sci. 2020, 10, 6045. [Google Scholar] [CrossRef]
- Adhikari, B.; Pangomm, K.; Veerana, M.; Mitra, S.; Park, G. Plant Disease Control by Non-Thermal Atmospheric-Pressure Plasma. Front. Plant Sci. 2020, 11, 77. [Google Scholar] [CrossRef] [PubMed]
- Koga, K.; Attri, P.; Kamataki, K.; Itagaki, N.; Shiratani, M.; Mildažiene, V. Impact of Radish Sprouts Seeds Coat Color on the Electron Paramagnetic Resonance Signals after Plasma Treatment. Jpn. J. Appl. Phys. 2020, 59, SHHF01. [Google Scholar] [CrossRef]
- Attri, P.; Ishikawa, K.; Okumura, T.; Koga, K.; Shiratani, M.; Mildaziene, V.; Ishikawa, K.; Okumura, T.; Koga, K.; Shiratani, M.; et al. Impact of Seed Color and Storage Time on the Radish Seed Germination and Sprout Growth in Plasma Agriculture. Sci. Rep. 2021, 11, 2539. [Google Scholar] [CrossRef]
- Sivachandiran, L.; Khacef, A. Enhanced Seed Germination and Plant Growth by Atmospheric Pressure Cold Air Plasma: Combined Effect of Seed and Water Treatment. RSC Adv. 2017, 7, 1822–1832. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, Y.Y.; Kim, Y.S.; Balaraju, K.; Mok, Y.S.; Yoo, S.J.; Jeon, Y. Enhancement of Seed Germination and Microbial Disinfection on Ginseng by Cold Plasma Treatment. J. Ginseng Res. 2021, 45, 519–526. [Google Scholar] [CrossRef]
- Song, J.-S.; Kim, S.B.; Ryu, S.; Oh, J.; Kim, D.-S. Emerging Plasma Technology That Alleviates Crop Stress During the Early Growth Stages of Plants: A Review. Front. Plant Sci. 2020, 11, 988. [Google Scholar] [CrossRef]
- Waskow, A.; Avino, F.; Howling, A.; Furno, I. Entering the Plasma Agriculture Field: An Attempt to Standardize Protocols for Plasma Treatment of Seeds. Plasma Process. Polym. 2022, 19, 2100152. [Google Scholar] [CrossRef]
- Nelson, S.O.; Stetson, L.E.; Stone, R.B.; Webb, J.C.; Pettibone, C.A.; Works, D.W.; Kehr, W.R.; VanRiper, G.E. Comparison of Infrared, Radiofrequency, and Gas-Plasma Treatments of Alfalfa Seed for Hard-Seed Reduction. Trans. ASAE 1964, 7, 276–280. [Google Scholar] [CrossRef]
- Alan, C. Pettibone Some Effects of Gas-Plasma Radiation on Seeds. Trans. ASAE 1965, 8, 319–321. [Google Scholar] [CrossRef]
- Webb, J.C.; Stone, B.; McDow, R.J.J. Response of Cottonseed to Audiofrequency Gas Plasma. Trans. ASAE 1966, 9, 872–874. [Google Scholar] [CrossRef]
- Nelson, S.O.; Kehr, W.R.; Stetson, L.E.; Stone, R.B.; Webb, J.C. Alfalfa Seed Germination Response to Electrical Treatments1. Crop Sci. 1977, 17, 863–866. [Google Scholar] [CrossRef]
- Ranieri, P.; Sponsel, N.; Kizer, J.; Rojas-Pierce, M.; Hernández, R.; Gatiboni, L.; Grunden, A.; Stapelmann, K. Plasma Agriculture: Review from the Perspective of the Plant and Its Ecosystem. Plasma Process. Polym. 2021, 18, 2000162. [Google Scholar] [CrossRef]
- Laroussi, M.; Bekeschus, S.; Keidar, M.; Bogaerts, A.; Lu, X.; Hori, M.; Stapelmann, K.; Miller, V.; Reuter, S.; Laux, C.; et al. Low Temperature Plasma for Biology, Hygiene, and Medicine: Perspective and Roadmap. In IEEE Transactions on Radiation and Plasma Medical Sciences; IEEE: Piscataway, NJ, USA, 2021. [Google Scholar]
- Bruggeman, P.; Brandenburg, R. Atmospheric Pressure Discharge Filaments and Microplasmas: Physics, Chemistry and Diagnostics. J. Phys. Appl. Phys. 2013, 46, 464001. [Google Scholar] [CrossRef]
- Gómez-Ramírez, A.; López-Santos, C.; Cantos, M.; García, J.L.; Molina, R.; Cotrino, J. Surface chemistry and germination improvement of Quinoa seeds subjected to plasma activation. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Meng, Y.; Qu, G.; Wang, T.; Sun, Q.; Liang, D.; Hu, S. Enhancement of Germination and Seedling Growth of Wheat Seed Using Dielectric Barrier Discharge Plasma with Various Gas Sources. Plasma Chem. Plasma Proc. 2017, 37, 1105–1119. [Google Scholar] [CrossRef]
- Rahman, M.M.; Sajib, S.A.; Rahi, M.S.; Tahura, S.; Roy, N.C.; Parvez, S.; Reza, M.A.; Talukder, M.R.; Kabir, A.H. Mechanisms and Signaling Associated with LPDBD Plasma Mediated Growth Improvement in Wheat. Sci. Rep. 2018, 8, 10498. [Google Scholar] [CrossRef]
- Li, Y.; Wang, T.; Meng, Y.; Qu, G.; Sun, Q.; Liang, D. Air Atmospheric Dielectric Barrier Discharge Plasma Induced Germination and Growth Enhancement of Wheat Seed. Plasma Chem. Plasma Proc. 2017, 37, 1621–1634. [Google Scholar] [CrossRef]
- Stolárik, T.; Henselová, M.; Martinka, M.; Novák, O.; Zahoranová, A.; Černák, M. Effect of Low-Temperature Plasma on the Structure of Seeds, Growth and Metabolism of Endogenous Phytohormones in Pea (Pisum sativum L.). Plasma Chem. Plasma Process. 2015, 35, 659–676. [Google Scholar] [CrossRef]
- Iranbakhsh, A.; Ardebili, N.O.; Ardebili, Z.O.; Shafaati, M.; Ghoranneviss, M. Non-Thermal Plasma Induced Expression of Heat Shock Factor A4A and Improved Wheat (Triticum aestivum L.) Growth and Resistance against Salt Stress. Plasma Chem. Plasma Proc. 2018, 38, 29–44. [Google Scholar] [CrossRef]
- Bafoil, M.; Le Ru, A.; Merbahi, N.; Eichwald, O.; Dunand, C.; Yousfi, M. New Insights of Low-Temperature Plasma Effects on Germination of Three Genotypes of Arabidopsis Thaliana Seeds under Osmotic and Saline Stresses. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, N.; Ono, R.; Nakano, R.; Shiratani, M.; Tashiro, K.; Kuhara, S.; Yasuda, K.; Hagiwara, H. DNA Microarray Analysis of Plant Seeds Irradiated by Active Oxygen Species in Oxygen Plasma. Plasma Med. 2016, 6. [Google Scholar] [CrossRef]
- Nakano, R.; Tashiro, K.; Aijima, R.; Hayashi, N. Effect of Oxygen Plasma Irradiation on Gene Expression in Plant Seeds Induced by Active Oxygen Species. Plasma Med. 2016, 6, 303–313. [Google Scholar] [CrossRef]
- Bormashenko, E.; Shapira, Y.; Grynyov, R.; Whyman, G.; Bormashenko, Y.; Drori, E. Interaction of Cold Radiofrequency Plasma with Seeds of Beans (Phaseolus vulgaris). J. Exp. Bot. 2015, 66, 4013–4021. [Google Scholar] [CrossRef]
- Li, L.; Li, J.; Shen, M.; Hou, J.; Shao, H.; Dong, Y. Improving Seed Germination and Peanut Yields by Cold Plasma Treatment. Plasma Sci. Technol. 2016, 18, 1027–1033. [Google Scholar] [CrossRef]
- Mildažiene, V.; Aleknavičiute, V.; Žukiene, R.; PauŽaite, G.; Naučiene, Z.; Filatova, I. Treatment of Common Sunflower (Helianthus annus L.) Seeds with Radio-Frequency Electromagnetic Field and Cold Plasma Induces Changes in Seed Phytohormone Balance, Seedling Development and Leaf Protein Expression. Sci. Rep. 2019, 9, 6437. [Google Scholar] [CrossRef]
- Saberi, M.; Modarres-Sanavy, S.A.M.; Zare, R.; Ghomi, H. Amelioration of Photosynthesis and Quality of Wheat under Non-Thermal Radio Frequency Plasma Treatment. Sci. Rep. 2018, 8, 11655. [Google Scholar] [CrossRef]
- Volkov, A.G.; Bookal, A.; Hairston, J.S.; Patel, D.; Volkov, A.G.; Bookal, A.; Hairston, J.S.; Patel, D. Radio Frequency Plasma Capacitor Can Increase Rates of Seeds Imbibition, Germination, and Radicle Growth. Funct. Plant Biol. 2020, 48, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Zhou, R.; Zhang, X.; Zhuang, J.; Yang, S.; Bazaka, K.; Ken Ostrikov, K. Effects of Atmospheric-Pressure N2, He, Air, and O2 Microplasmas on Mung Bean Seed Germination and Seedling Growth. Sci. Rep. 2016, 6, 32603. [Google Scholar] [CrossRef]
- De Groot, G.J.J.B.; Hundt, A.; Murphy, A.B.; Bange, M.P.; Mai-Prochnow, A. Cold Plasma Treatment for Cotton Seed Germination Improvement. Sci. Rep. 2018, 8, 14372. [Google Scholar] [CrossRef] [PubMed]
- Billah, M.; Sajib, S.A.; Roy, N.C.; Rashid, M.M.; Reza, M.A.; Hasan, M.M.; Talukder, M.R. Effects of DBD Air Plasma Treatment on the Enhancement of Black Gram (Vigna mungo L.) Seed Germination and Growth. Arch. Biochem. Biophys. 2020, 681, 108253. [Google Scholar] [CrossRef]
- Islam, S.; Omar, F.B.; Sajib, S.A.; Roy, N.C.; Reza, A.; Hasan, M. Effects of LPDBD Plasma and Plasma Activated Water on Germination and Growth in Rapeseed (Brassica napus). Gesunde Pflanz. 2019, 71, 175–185. [Google Scholar] [CrossRef]
- Tounekti, T.; Mujahid, Z.U.I.; Khemira, H. Non-Thermal Dielectric Barrier Discharge (DBD) Plasma Affects Germination of Coffee and Grape Seeds. In AIP Conference Proceedings; AIP Publishing LLC: Melville, NY, USA, 2018. [Google Scholar] [CrossRef]
- Pawłat, J.; Starek, A.; Sujak, A.; Terebun, P.; Kwiatkowski, M.; Budzeń, M. Effects of Atmospheric Pressure Plasma Jet Operating with DBD on Lavatera Thuringiaca L. Seeds’ Germination. PLoS ONE 2018, 13, e0194349. [Google Scholar] [CrossRef]
- Liu, B.; Honnorat, B.; Yang, H.; Arancibia, J.; Rajjou, L.; Rousseau, A. Non-Thermal DBD Plasma Array on Seed Germination of Different Plant Species. J. Phys. Appl. Phys. 2019, 52, 025401. [Google Scholar] [CrossRef]
- Bradu, C.; Kutasi, K.; Magureanu, M.; Puač, N.; Živković, S. Reactive Nitrogen Species in Plasma-Activated Water: Generation, Chemistry and Application in Agriculture. J. Phys. Appl. Phys. 2020, 53, 223001. [Google Scholar] [CrossRef]
- Al-Sharify, Z.T.; Al-Sharify, T.A.; al-Obaidy Baker, W.; al-Azawi, A.M. Investigative Study on the Interaction and Applications of Plasma Activated Water(PAW). IOP Conf. Ser. Mater. Sci. Eng. 2020, 870, 012042. [Google Scholar] [CrossRef]
- Thirumdas, R.; Kothakota, A.; Annapure, U.; Siliveru, K.; Blundell, R.; Gatt, R.; Valdramidis, V.P. Plasma Activated Water (PAW): Chemistry, Physico-Chemical Properties, Applications in Food and Agriculture. Trends Food Sci. Technol. 2018, 77, 21–31. [Google Scholar] [CrossRef]
- Šimečková, J.; Krčma, F.; Klofáč, D.; Dostál, L.; Kozáková, Z. Influence of Plasma-Activated Water on Physical and Physical–Chemical Soil Properties. Water 2020, 12, 2357. [Google Scholar] [CrossRef]
- Traylor, M.J.; Pavlovich, M.J.; Karim, S.; Hait, P.; Sakiyama, Y.; Clark, D.S.; Graves, D.B. Long-Term Antibacterial Efficacy of Air Plasma-Activated Water. J. Phys. Appl. Phys. 2011, 44, 472001. [Google Scholar] [CrossRef]
- Munekata, P.E.S.; Domínguez, R.; Pateiro, M.; Lorenzo, J.M. Influence of Plasma Treatment on the Polyphenols of Food Products—A Review. Foods 2020, 9, 929. [Google Scholar] [CrossRef]
- Graves, D.B.; Bakken, L.B.; Jensen, M.B.; Ingels, R. Plasma Activated Organic Fertilizer. Plasma Chem. Plasma Process. 2019, 39, 1–19. [Google Scholar] [CrossRef]
- Zhang, S.; Rousseau, A.; Dufour, T. Promoting Lentil Germination and Stem Growth by Plasma Activated Tap Water, Demineralized Water and Liquid Fertilizer. RSC Adv. 2017, 7, 31244–31251. [Google Scholar] [CrossRef]
- Misra, N.N.; Schlüter, O.; Cullen, P.J. Chapter 1—Plasma in Food and Agriculture. In Cold Plasma in Food and Agriculture; Misra, N.N., Schlüter, O., Cullen, P.J., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 1–16. ISBN 978-0-12-801365-6. [Google Scholar]
- Fan, L.; Liu, X.; Ma, Y.; Xiang, Q. Effects of Plasma-Activated Water Treatment on Seed Germination and Growth of Mung Bean Sprouts. J. Taibah Univ. Sci. 2020, 14, 823–830. [Google Scholar] [CrossRef]
- Song, J.-S.; Lee, M.J.; Ra, J.E.; Lee, K.S.; Eom, S.; Ham, H.M.; Kim, H.Y.; Kim, S.B.; Lim, J. Growth and Bioactive Phytochemicals in Barley (Hordeum vulgare L.) Sprouts Affected by Atmospheric Pressure Plasma during Seed Germination. J. Phys. Appl. Phys. 2020, 53, 314002. [Google Scholar] [CrossRef]
- Arjunan, K.P.; Sharma, V.K.; Ptasinska, S. Effects of Atmospheric Pressure Plasmas on Isolated and Cellular DNA—A Review. Int. J. Mol. Sci. 2015, 16, 2971–3016. [Google Scholar] [CrossRef]
- Halliwell, B. Reactive Species and Antioxidants. Redox Biology Is a Fundamental Theme of Aerobic Life. Plant Physiol. 2006, 141, 312–322. [Google Scholar] [CrossRef]
- Mhamdi, A.; Van Breusegem, F. Reactive Oxygen Species in Plant Development. Development 2018, 145, dev164376. [Google Scholar] [CrossRef]
- El-Maarouf-Bouteau, H.; Bailly, C. Oxidative Signaling in Seed Germination and Dormancy. Plant Signal. Behav. 2008, 3, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Sarinont, T.; Amano, T.; Attri, P.; Koga, K.; Hayashi, N.; Shiratani, M.; Biophys, L.A.B. Effects of Plasma Irradiation Using Various Feeding Gases on Growth of Raphanus Sativus. Arch. Biochem. Biophys. 2016, 605, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Waskow, A.; Howling, A.; Furno, I. Mechanisms of Plasma-Seed Treatments as a Potential Seed Processing Technology. Front. Phys. 2021, 9, 174. [Google Scholar] [CrossRef]
- Souza, F.H.; Marcos-Filho, J. The Seed Coat as a Modulator of Seed-Environment Relationships in Fabaceae. Braz. J. Bot. 2001, 24, 365–375. [Google Scholar] [CrossRef]
- Yusupov, M.; Razzokov, J.; Cordeiro, R.M.; Bogaerts, A. Transport of Reactive Oxygen and Nitrogen Species across Aquaporin: A Molecular Level Picture. Oxidative Med. Cell. Longev. 2019, 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Seol, Y.B.; Kim, J.; Park, S.H.; Chang, H.Y. Atmospheric Pressure Pulsed Plasma Induces Cell Death in Photosynthetic Organs via Intracellularly Generated ROS. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Ka, D.H.; Priatama, R.A.; Park, J.Y.; Park, S.J.; Kim, S.B.; Lee, I.A.; Lee, Y.K. Plasma-Activated Water Modulates Root Hair Cell Density via Root Developmental Genes in Arabidopsis Thaliana L. Appl. Sci. 2021, 11, 2240. [Google Scholar] [CrossRef]
- Lee, Y.K.; Lim, J.; Hong, E.J.; Kim, S.B. Plasma-Activated Water Regulates Root Hairs and Cotyledon Size Dependent on Cell Elongation in Nicotiana tabacum L. Plant Biotechnol. Rep. 2020, 14, 663–672. [Google Scholar] [CrossRef]
- Park, D.P.; Davis, K.; Gilani, S.; Alonzo, C.-A.; Dobrynin, D.; Friedman, G.; Fridman, A.; Rabinovich, A.; Fridman, G. Reactive Nitrogen Species Produced in Water by Non-Equilibrium Plasma Increase Plant Growth Rate and Nutritional Yield. Curr. Appl. Phys. 2013, 13, S19–S29. [Google Scholar] [CrossRef]
- Habibi, G. Hydrogen Peroxide (H2O2) Generation, Scavenging and Signaling in Plants. In Oxidative Damage to Plants; Ahmad, P., Ed.; Academic Press: San Diego, NJ, USA, 2014; pp. 557–584. ISBN 978-0-12-799963-0. [Google Scholar]
- Pei, Z.-M.; Murata, Y.; Benning, G.; Thomine, S.; Klüsener, B.; Allen, G.J.; Grill, E.; Schroeder, J.I. Calcium Channels Activated by Hydrogen Peroxide Mediate Abscisic Acid Signalling in Guard Cells. Nature 2000, 406, 731–734. [Google Scholar] [CrossRef]
- Liu, Y.; Ye, N.; Liu, R.; Chen, M.; Zhang, J. H2O2 Mediates the Regulation of ABA Catabolism and GA Biosynthesis in Arabidopsis Seed Dormancy and Germination. J. Exp. Bot. 2010, 61, 2979–2990. [Google Scholar] [CrossRef] [PubMed]
- Barba-Espín, G.; Diaz-Vivancos, P.; Job, D.; Belghazi, M.; Job, C.; Hernández, J.A. Understanding the Role of H2O2 during Pea Seed Germination: A Combined Proteomic and Hormone Profiling Approach. Plant Cell Environ. 2011, 34, 1907–1919. [Google Scholar] [CrossRef] [PubMed]
- Kurek, K.; Plitta-Michalak, B.; Ratajczak, E. Reactive Oxygen Species as Potential Drivers of the Seed Aging Process. Plants 2019, 8, 174. [Google Scholar] [CrossRef] [PubMed]
- Pandiselvam, R.; Mayookha, V.P.; Kothakota, A.; Sharmila, L.; Ramesh, S.V.; Bharathi, C.P. Impact of Ozone Treatment on Seed Germination-a Systematic Review. Ozone Sci. Eng. 2019, 42, 1–16. [Google Scholar] [CrossRef]
- Palou, L.; Crisosto, C.H.; Smilanick, J.L.; Adaskaveg, J.E.; Zoffoli, J.P. Effects of Continuous 0.3 Ppm Ozone Exposure on Decay Development and Physiological Responses of Peaches and Table Grapes in Cold Storage. Postharvest Biol. Technol. 2002, 24, 39–48. [Google Scholar] [CrossRef]
- Vlassi, E.; Vlachos, P.; Kornaros, M. Effect of Ozonation on Table Grapes Preservation in Cold Storage. J. Food Sci. Technol. 2018, 55, 2031–2038. [Google Scholar] [CrossRef] [PubMed]
- Misra, N.N.; Moiseev, T.; Patil, S.; Pankaj, S.K.; Bourke, P.; Mosnier, J.P.; Keener, K.M.; Cullen, P.J. Cold Plasma in Modified Atmospheres for Post-Harvest Treatment of Strawberries. Food Bioprocess Technol. 2014, 7, 3045–3054. [Google Scholar] [CrossRef]
- Waskow, A.; Howling, A.; Furno, I. Advantages and Limitations of Surface Analysis Techniques on Plasma-Treated Arabidopsis Thaliana Seeds. Front. Mater. 2021, 8, 123. [Google Scholar] [CrossRef]
- Ölmez, H.; Akbas, M.Y. Optimization of Ozone Treatment of Fresh-Cut Green Leaf Lettuce. J. Food Eng. 2009, 90, 487–494. [Google Scholar] [CrossRef]
- Mitsugi, F.; Abiru, T.; Ikegami, T.; Ebihara, K.; Aoqui, S.-I.; Nagahama, K. Influence of Ozone Generated by Surface Barrier Discharge on Nematode and Plant Growth. IEEE Trans. Plasma Sci. 2016, 44, 3071–3076. [Google Scholar] [CrossRef]
- Los, A.; Ziuzina, D.; Boehm, D.; Cullen, P.J.; Bourke, P. Investigation of Mechanisms Involved in Germination Enhancement of Wheat (Triticum Aestivum) by Cold Plasma: Effects on Seed Surface Chemistry and Characteristics. Plasma Proc. Polym. 2019, 16, 1800148. [Google Scholar] [CrossRef]
- Hasan, M.; Sohan, M.S.R.; Sajib, S.A.; Hossain, M.F.; Miah, M.; Maruf, M.M.H.; Khalid-Bin-Ferdaus, K.M.; Kabir, A.H.; Talukder, M.R.; Rashid, M.M.; et al. The Effect of Low-Pressure Dielectric Barrier Discharge (LPDBD) Plasma in Boosting Germination, Growth, and Nutritional Properties in Wheat. Plasma Chem. Plasma Process. 2022, 42, 339–362. [Google Scholar] [CrossRef]
- Lindsay, A.; Byrns, B.; King, W.; Andhvarapou, A.; Fields, J.; Knappe, D.; Fonteno, W.; Shannon, S. Fertilization of Radishes, Tomatoes, and Marigolds Using a Large-Volume Atmospheric Glow Discharge. Plasma Chem. Plasma Process. 2014, 34, 1271–1290. [Google Scholar] [CrossRef]
- Takahata, J.; Takaki, K.; Satta, N.; Takahashi, K.; Fujio, T.; Sasaki, Y. Improvement of Growth Rate of Plants by Bubble Discharge in Water. Jpn. J. Appl. Phys. 2014, 54, 01AG07. [Google Scholar] [CrossRef]
- Takano, K.; Takahata, J.; Takaki, K.; Satta, N.; Takahashi, K.; Fujio, T. Improvement of Growth Rate of Brassica Para Var. Perviridis by Discharge inside Bubble under Water in Hydroponic Cultivation. Electron. Commun. Jpn. 2016, 99, 72–79. [Google Scholar] [CrossRef]
- Kang, M.H.; Jeon, S.S.; Shin, S.M.; Veerana, M.; Ji, S.-H.; Uhm, H.-S.; Choi, E.-H.; Shin, J.H.; Park, G. Dynamics of Nitric Oxide Level in Liquids Treated with Microwave Plasma-Generated Gas and Their Effects on Spinach Development. Sci. Rep. 2019, 9, 1011. [Google Scholar] [CrossRef]
- Lamichhane, P.; Veerana, M.; Lim, J.S.; Mumtaz, S.; Shrestha, B.; Kaushik, N.K.; Park, G.; Choi, E.H. Low-Temperature Plasma-Assisted Nitrogen Fixation for Corn Plant Growth and Development. Int. J. Mol. Sci. 2021, 22, 5360. [Google Scholar] [CrossRef]
- Kidd, P.S.; Proctor, J. Why Plants Grow Poorly on Very Acid Soils: Are Ecologists Missing the Obvious? J. Exp. Bot. 2001, 52, 791–799. [Google Scholar] [CrossRef]
- Sakudo, A.; Yagyu, Y.; Onodera, T. Disinfection and Sterilization Using Plasma Technology: Fundamentals and Future Perspectives for Biological Applications. Int. J. Mol. Sci. 2019, 20, 5216. [Google Scholar] [CrossRef]
- Shintani, H.; Sakudo, A.; Burke, P.; McDonnell, G. Gas Plasma Sterilization of Microorganisms and Mechanisms of Action. Exp. Ther. Med. 2010, 1, 731–738. [Google Scholar] [CrossRef]
- Laroussi, M. Cold Plasma in Medicine and Healthcare: The New Frontier in Low Temperature Plasma Applications. Front. Phys. 2020, 8, 74. [Google Scholar] [CrossRef]
- Guo, J.; Wang, J.; Xie, H.; Jiang, J.; Li, C.; Li, W.; Li, L.; Liu, X.; Lin, F. Inactivation Effects of Plasma-Activated Water on Fusarium Graminearum. Food Control 2022, 134, 108683. [Google Scholar] [CrossRef]
- Khamsen, N.; Onwimol, D.; Teerakawanich, N.; Dechanupaprittha, S.; Kanokbannakorn, W.; Hongesombut, K. Rice (Oryza sativa L.) Seed Sterilization and Germination Enhancement via Atmospheric Hybrid Nonthermal Discharge Plasma. ACS Appl. Mater. Interfaces 2016, 8, 19268–19275. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.H.; Pengkit, A.; Choi, K.; Jeon, S.S.; Choi, H.W.; Shin, D.B.; Choi, E.H.; Uhm, H.S.; Park, G. Differential Inactivation of Fungal Spores in Water and on Seeds by Ozone and Arc Discharge Plasma. PLoS ONE 2015, 10, e0139263. [Google Scholar] [CrossRef] [PubMed]
- Świecimska, M.; Tulik, M.; Šerá, B.; Golińska, P.; Tomeková, J.; Medvecká, V.; Bujdáková, H.; Oszako, T.; Zahoranová, A.; Šerý, M. Non-Thermal Plasma Can Be Used in Disinfection of Scots Pine (Pinus sylvestris L.) Seeds Infected with Fusarium Oxysporum. Forests 2020, 11, 837. [Google Scholar] [CrossRef]
- ŠERÁ, B.; ŠERÝ, M. Non-Thermal Plasma Treatment as a New Biotechnology in Relation to Seeds, Dry Fruits, and Grains. Plasma Sci. Technol. 2018, 20, 044012. [Google Scholar] [CrossRef]
- Zahoranová, A.; Henselová, M.; Hudecová, D.; Kaliňáková, B.; Kováčik, D.; Medvecká, V.; Černák, M. Effect of Cold Atmospheric Pressure Plasma on the Wheat Seedlings Vigor and on the Inactivation of Microorganisms on the Seeds Surface. Plasma Chem. Plasma Process. 2016, 36, 397–414. [Google Scholar] [CrossRef]
- Masiello, M.; Somma, S.; Lo Porto, C.; Palumbo, F.; Favia, P.; Fracassi, F.; Logrieco, A.F.; Moretti, A. Plasma Technology Increases the Efficacy of Prothioconazole against Fusarium graminearum and Fusarium proliferatum Contamination of Maize (Zea mays) Seedlings. Int. J. Mol. Sci. 2021, 22, 9301. [Google Scholar] [CrossRef]
- Mravlje, J.; Regvar, M.; Vogel-Mikuš, K. Development of Cold Plasma Technologies for Surface Decontamination of Seed Fungal Pathogens: Present Status and Perspectives. J. Fungi 2021, 7, 650. [Google Scholar] [CrossRef]
- Cold Plasma in Food and Agriculture: Fundamentals and Applications; Misra, N.N., Schlüter, O.F.-K., Cullen, P.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 978-0-12-801365-6. [Google Scholar]
- Li, H.; Zhou, X.; Huang, Y.; Liao, B.; Cheng, L.; Ren, B. Reactive Oxygen Species in Pathogen Clearance: The Killing Mechanisms, the Adaption Response, and the Side Effects. Front. Microbiol. 2021, 11, 3610. [Google Scholar] [CrossRef]
- Vaishampayan, A.; Grohmann, E. Antimicrobials Functioning through ROS-Mediated Mechanisms: Current Insights. Microorganisms 2021, 10, 61. [Google Scholar] [CrossRef] [PubMed]
- Shaw, P.; Kumar, N.; Kwak, H.S.; Park, J.H.; Uhm, H.S.; Bogaerts, A.; Choi, E.H.; Attri, P. Bacterial Inactivation by Plasma Treated Water Enhanced by Reactive Nitrogen Species. Sci. Rep. 2018, 8, 11268. [Google Scholar] [CrossRef] [PubMed]
- Moisan, M.; Barbeau, J.; Moreau, S.; Pelletier, J.; Tabrizian, M.; Yahia, L. Low-Temperature Sterilization Using Gas Plasmas: A Review of the Experiments and an Analysis of the Inactivation Mechanisms. Int. J. Pharm. 2001, 226, 1–21. [Google Scholar] [CrossRef]
- Laroussi, M.; Leipold, F. Evaluation of the Roles of Reactive Species, Heat, and UV Radiation in the Inactivation of Bacterial Cells by Air Plasmas at Atmospheric Pressure. Int. J. Mass Spectrom. 2004, 233, 81–86. [Google Scholar] [CrossRef]
- Lackmann, J.-W.; Schneider, S.; Edengeiser, E.; Jarzina, F.; Brinckmann, S.; Steinborn, E.; Havenith, M.; Benedikt, J.; Bandow, J.E. Photons and Particles Emitted from Cold Atmospheric-Pressure Plasma Inactivate Bacteria and Biomolecules Independently and Synergistically. J. R. Soc. Interface 2013, 10, 20130591. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Garcia, G.; Arumugaswami, V.; Wirz, R.E. Cold Atmospheric Plasma for SARS-CoV-2 Inactivation. Phys. Fluids 2020, 32, 111702. [Google Scholar] [CrossRef]
- Cortázar, O.D.; Megía-Macías, A.; Moreno, S.; Brun, A.; Gómez-Casado, E. Vulnerability of SARS-CoV-2 and PR8 H1N1 Virus to Cold Atmospheric Plasma Activated Media. Sci. Rep. 2022, 12, 263. [Google Scholar] [CrossRef]
- Mohamed, H.; Nayak, G.; Rendine, N.; Wigdahl, B.; Krebs, F.C.; Bruggeman, P.J.; Miller, V. Non-Thermal Plasma as a Novel Strategy for Treating or Preventing Viral Infection and Associated Disease. Front. Phys. 2021, 9, 286. [Google Scholar] [CrossRef]
- Filipić, A.; Primc, G.; Zaplotnik, R.; Mehle, N.; Gutierrez-Aguirre, I.; Ravnikar, M.; Mozetič, M.; Žel, J.; Dobnik, D. Cold Atmospheric Plasma as a Novel Method for Inactivation of Potato Virus Y in Water Samples. Food Environ. Virol. 2019, 11, 220–228. [Google Scholar] [CrossRef]
- Filipić, A.; Dobnik, D.; Tušek Žnidarič, M.; Žegura, B.; Štern, A.; Primc, G.; Mozetič, M.; Ravnikar, M.; Žel, J.; Gutierrez Aguirre, I. Inactivation of Pepper Mild Mottle Virus in Water by Cold Atmospheric Plasma. Front. Microbiol. 2021, 12, 14. [Google Scholar] [CrossRef]
- Filipić, A.; Gutierrez-Aguirre, I.; Primc, G.; Mozetič, M.; Dobnik, D. Cold Plasma, a New Hope in the Field of Virus Inactivation. Trends Biotechnol. 2020, 38, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.; Mitra, S.; Baek, E.J.; Nguyen, L.N.; Bhartiya, P.; Kim, J.H.; Choi, E.H.; Kaushik, N.K. The Inactivation and Destruction of Viruses by Reactive Oxygen Species Generated through Physical and Cold Atmospheric Plasma Techniques: Current Status and Perspectives. J. Adv. Res. 2022. [Google Scholar] [CrossRef]
- Dauwe, R.; Roulard, R.; Ramos, M.; Thiombiano, B.; Mesnard, F.; Gontier, E.; Jamali, A. Etching of the Seed Cuticle by Cold Plasma Shortens Imbibitional Leakage in Linum usitatissimum L. Ind. Crops Prod. 2021, 167, 113536. [Google Scholar] [CrossRef]
- Kitazaki, S.; Sarinont, T.; Koga, K.; Hayashi, N.; Shiratani, M. Plasma Induced Long-Term Growth Enhancement of Raphanus sativus L. Using Combinatorial Atmospheric Air Dielectric Barrier Discharge Plasmas. Curr. Appl. Phys. 2014, 14, S149–S153. [Google Scholar] [CrossRef]
- Mildažiene, V.; Pauzaite, G.; Malakauskiene, A.; Zukiene, R.; Nauciene, Z.; Filatova, I. Response of Perennial Woody Plants to Seed Treatment by Electromagnetic Field and Low-Temperature Plasma. Bioelectromagnetics 2016, 37, 536–548. [Google Scholar] [CrossRef] [PubMed]
- Sera, B.; Spatenka, P.; Sery, M.; Vrchotova, N.; Hruskova, I. Influence of Plasma Treatment on Wheat and Oat Germination and Early Growth. IEEE Trans. Plasma Sci. 2010, 38, 2963–2968. [Google Scholar] [CrossRef]
- Bormashenko, E.; Grynyov, R.; Bormashenko, Y.; Drori, E. Cold Radiofrequency Plasma Treatment Modifies Wettability and Germination Speed of Plant Seeds. Sci. Rep. 2012, 2, 741. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, A.; Héroux, P.; Sand, W.; Sun, Z.; Zhan, J. Effect of dielectric barrier discharge cold plasma on pea seed growth. J. Agric. Food Chem. 2019, 67, 10813–10822. [Google Scholar] [CrossRef]
- Wang, X.Q.; Zhou, R.W.; De Groot, G.; Bazaka, K.; Murphy, A.B.; Ostrikov, K.K. Spectral Characteristics of Cotton Seeds Treated by a Dielectric Barrier Discharge Plasma. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Da Silva, A.R.M.; Farias, M.L.; da Silva, D.L.S.; Vitoriano, J.O.; de Sousa, R.C.; Alves-Junior, C. Using Atmospheric Plasma to Increase Wettability, Imbibition and Germination of Physically Dormant Seeds of Mimosa Caesalpiniafolia. Coll. Surf. Biointerfaces 2017, 157, 280–285. [Google Scholar] [CrossRef]
- Junior, C.A.; De Oliveira Vitoriano, J.; Da Silva, D.L.S.; De Lima Farias, M.; De Lima Dantas, N.B. Water Uptake Mechanism and Germination of Erythrina Velutina Seeds Treated with Atmospheric Plasma. Sci. Rep. 2016, 6, 1–7. [Google Scholar] [CrossRef]
- Terebun, P.; Kwiatkowski, M.; Starek, A.; Reuter, S.; Mok, Y.S.; Pawłat, J. Impact of Short Time Atmospheric Plasma Treatment on Onion Seeds. Plasma Chem. Plasma Process. 2021, 41, 559–571. [Google Scholar] [CrossRef]
- Sehrawat, R.; Thakur, A.K.; Vikram, A.; Vaid, A.; Rane, R. Effect of Cold Plasma Treatment on Physiological Quality of Okra Seed. J. Hill Agric. 2017, 8, 66–71. [Google Scholar] [CrossRef]
- Shapira, Y.; Multanen, V.; Whyman, G.; Bormashenko, Y.; Chaniel, G.; Barkay, Z.; Bormashenko, E. Plasma Treatment Switches the Regime of Wetting and Floating of Pepper Seeds. Coll. Surf. Biointerfaces 2017, 157, 417–423. [Google Scholar] [CrossRef]
- Molina, R.; Lalueza, A.; López-Santos, C.; Ghobeira, R.; Cools, P.; Morent, R.; de Geyter, N.; González-Elipe, A.R. Physicochemical Surface Analysis and Germination at Different Irrigation Conditions of DBD Plasma-Treated Wheat Seeds. Plasma Process. Polym. 2021, 18, 2000086. [Google Scholar] [CrossRef]
- Zahoranová, A.; Hoppanová, L.; Šimončicová, J.; Tučeková, Z.; Medvecká, V.; Hudecová, D.; Kaliňáková, B.; Kováčik, D.; Černák, M. Effect of Cold Atmospheric Pressure Plasma on Maize Seeds: Enhancement of Seedlings Growth and Surface Microorganisms Inactivation. Plasma Chem. Plasma Process. 2018, 38, 969–988. [Google Scholar] [CrossRef]
- Al-Whaibi, M.H. Plant heat-shock proteins: A mini review. J. King Saud. Univ. Sci. 2011, 23, 139–150. [Google Scholar] [CrossRef]
- Waters, E.R.; Vierling, E. Plant Small Heat Shock Proteins—Evolutionary and Functional Diversity. New Phytol. 2020, 227, 24–37. [Google Scholar] [CrossRef]
- Bourgine, B.; Guihur, A. Heat Shock Signaling in Land Plants: From Plasma Membrane Sensing to the Transcription of Small Heat Shock Proteins. Front. Plant Sci. 2021, 12, 1583. [Google Scholar] [CrossRef]
- Holubová, Ľ.; Švubová, R.; Slováková, Ľ.; Bokor, B.; Chobotová Kročková, V.; Renčko, J.; Uhrin, F.; Medvecká, V.; Zahoranová, A.; Gálová, E. Cold Atmospheric Pressure Plasma Treatment of Maize Grains—Induction of Growth, Enzyme Activities and Heat Shock Proteins. Int. J. Mol. Sci. 2021, 22, 8509. [Google Scholar] [CrossRef]
- Moisan, M.; Barbeau, J.; Crevier, M.-C.; Pelletier, J.; Philip, N.; Saoudi, B. Plasma Sterilization. Methods and Mechanisms. Pure Appl. Chem. 2002, 74, 349–358. [Google Scholar] [CrossRef]
- Ibrahim, M.; Imram, M.; Hussain, A.; Aslam, M.; Rehmani, F.S.; Ali, B. Phytochemical studies on Amberboa ramose. Pak. J. Chem. 2012, 2, 24–28. [Google Scholar] [CrossRef]
- Farokh, P.; Mahmoodzadeh, H.; Satari, T.N. Response of seed germination of safflower to UV-B radiation. Res. J. Env. Sci. 2010, 4, 70–74. [Google Scholar] [CrossRef][Green Version]
- Noble, R.E. Effects of UV-irradiation on seed germination. Sci. Total Env. 2002, 299, 173–176. [Google Scholar] [CrossRef]
- Sadeghianfar, P.; Nazari, M.; Backes, G. Exposure to Ultraviolet (UV-C) radiation increases germination rate of maize (Zea maize L.) and sugar beet (Beta vulgaris) seeds. Plants 2019, 8, 49. [Google Scholar] [CrossRef]
- Lotfy, K.; Al-Harbi, N.A.; Abd El-Raheem, H. Cold Atmospheric Pressure Nitrogen Plasma Jet for Enhancement Germination of Wheat Seeds. Plasma Chem. Plasma Process. 2019, 39, 897–912. [Google Scholar] [CrossRef]
- Caldwell, M.M.; Björn, L.O.; Bornman, J.F.; Flint, S.D.; Kulandaivelu, G.; Teramura, A.H. Effects of Increased Solar Ultraviolet Radiation on Terrestrial Ecosystems. J. Photochem. Photobiol. Biol. 1998, 46, 40–52. [Google Scholar] [CrossRef]
- Mildaziene, V.; Pauzaite, G.; Naucienė, Z.; Malakauskiene, A.; Zukiene, R.; Januskaitiene, I.; Jakstas, V.; Ivanauskas, L.; Filatova, I.; Lyushkevich, V. Pre-Sowing Seed Treatment with Cold Plasma and Electromagnetic Field Increases Secondary Metabolite Content in Purple Coneflower (Echinacea Purpurea) Leaves. Plasma Process. Polym. 2018, 15, 1700059. [Google Scholar] [CrossRef]
- Iranbakhsh, A.; Oraghi Ardebili, Z.; Molaei, H.; Oraghi Ardebili, N.; Amini, M. Cold Plasma Up-Regulated Expressions of WRKY1 Transcription Factor and Genes Involved in Biosynthesis of Cannabinoids in Hemp (Cannabis sativa L.). Plasma Chem. Plasma Process. 2020, 40, 527–537. [Google Scholar] [CrossRef]
- Mujahid, Z.; Tounekti, T.; Khemira, H. Cold Plasma Treatment to Release Dormancy and Improve Growth in Grape Buds: A Promising Alternative to Natural Chilling and Rest Breaking Chemicals. Sci. Rep. 2020, 10, 2667. [Google Scholar] [CrossRef]
- Filatova, I.; Lyushkevich, V.; Goncharik, S.; Zhukovsky, A.; Krupenko, N.; Kalatskaja, J. The Effect of Low-Pressure Plasma Treatment of Seeds on the Plant Resistance to Pathogens and Crop Yields. J. Phys. Appl. Phys. 2020, 53, 244001. [Google Scholar] [CrossRef]
- Cui, D.; Yin, Y.; Wang, J.; Wang, Z.; Ding, H.; Ma, R. Research on the Physio-Biochemical Mechanism of Non-Thermal Plasma-Regulated Seed Germination and Early Seedling Development in Arabidopsis. Front. Plant Sci. 2019, 10, 1322. [Google Scholar] [CrossRef] [PubMed]
- Moghanloo, M.; Iranbakhsh, A.; Ebadi, M.; Nejad Satari, T.; Oraghi Ardebili, Z. Seed Priming with Cold Plasma and Supplementation of Culture Medium with Silicon Nanoparticle Modified Growth, Physiology, and Anatomy in Astragalus Fridae as an Endangered Species. Acta Physiol. Plant 2019, 41, 1–13. [Google Scholar] [CrossRef]
- Pérez-Pizá, M.C.; Prevosto, L.; Grijalba, P.E.; Zilli, C.G.; Cejas, E.; Mancinelli, B. Improvement of Growth and Yield of Soybean Plants through the Application of Non-Thermal Plasmas to Seeds with Different Health Status. Heliyon 2019, 5, e01495. [Google Scholar] [CrossRef] [PubMed]
- Babajani, A.; Iranbakhsh, A.; Oraghi Ardebili, Z.; Eslami, B. Seed Priming with Non-Thermal Plasma Modified Plant Reactions to Selenium or Zinc Oxide Nanoparticles: Cold Plasma as a Novel Emerging Tool for Plant Science. Plasma Chem. Plasma Process. 2019, 39, 21–34. [Google Scholar] [CrossRef]
- Singh, R.; Prasad, P.; Mohan, R.; Verma, M.K. Kumar B Radiofrequency Cold Plasma Treatment Enhances Seed Germination and Seedling Growth in Variety CIM-Saumya of Sweet Basil (Ocimum basilicum L.). J. Appl. Res. Med. Aromat. Plants 2019, 12, 78–81. [Google Scholar] [CrossRef]
- Iqbal, T.; Farooq, M.; Afsheen, S.; Abrar, M.; Yousaf, M.; Ijaz, M. Cold Plasma Treatment and Laser Irradiation of Triticum Spp. Seeds for Sterilization and Germination. J. Laser Appl. 2019, 31, 042013. [Google Scholar] [CrossRef]
- Kabir, A.H.; Rahman, M.M.; Das, U.; Sarkar, U.; Roy, N.C.; Reza, M.A. Reduction of Cadmium Toxicity in Wheat through Plasma Technology. PLoS ONE 2019, 14, e0214509. [Google Scholar] [CrossRef]
- Afsheen, S.; Fatima, U.; Iqbal, T.; Abrar, M.; Muhammad, S.; Saeed, A. Influence of Cold Plasma Treatment on Insecticidal Properties of Wheat Seeds against Red Flour Beetles. Plasma Sci. Technol. 2019, 21, 085506. [Google Scholar] [CrossRef]
- Lo Porto, C.; Sergio, L.; Boari, F.; Logrieco, A.F.; Cantore, V. Cold Plasma Pretreatment Improves the Germination of Wild Asparagus (Asparagus acutifolius L.) Seeds. Sci. Hortic. 2019, 256, 108554. [Google Scholar] [CrossRef]
- Jiang, J.; Li, J.; Dong, Y. Effect of Cold Plasma Treatment on Seedling Growth and Nutrient Absorption of Tomato. Plasma Sci. Technol. 2018, 20, 044007. [Google Scholar] [CrossRef]
- Hosseini, S.I.; Mohsenimehr, S.; Hadian, J.; Ghorbanpour, M.; Shokri, B. Physico-Chemical Induced Modification of Seed Germination and Early Development in Artichoke (Cynara scolymus L.) Using Low Energy Plasma Technology. Phys. Plasmas 2018, 25, 013525. [Google Scholar] [CrossRef]
- Khatami, S.; Ahmadinia, A. Increased Germination and Growth Rates of Pea and Zucchini Seed by FSG Plasma. J. Theor. Appl. Phys. 2018, 12, 33–38. [Google Scholar] [CrossRef]
- Sohan, M.S.R.; Hasan, M.; Hossain, M.F.; Sajib, S.A.; Khalid-Bin-Ferdaus, K.M.; Kabir, A.H.; Rashid, M.M.; Talukder, M.R.; Elseehy, M.M.; El-Shehawi, A.M.; et al. Low-Frequency Glow Discharge (LFGD) Plasma Treatment Enhances Maize (Zea mays L.) Seed Germination, Agronomic Traits, Enzymatic Activities, and Nutritional Properties. Chem. Biol. Technol. Agric. 2022, 9, 18. [Google Scholar] [CrossRef]
- Suriyasak, C.; Hatanaka, K.; Tanaka, H.; Okumura, T.; Yamashita, D.; Attri, P.; Koga, K.; Shiratani, M.; Hamaoka, N.; Ishibashi, Y. Alterations of DNA Methylation Caused by Cold Plasma Treatment Restore Delayed Germination of Heat-Stressed Rice (Oryza sativa L.) Seeds. ACS Agric. Sci. Technol. 2021, 1, 5–10. [Google Scholar] [CrossRef]
- Pérez-Pizá, M.C.; Cejas, E.; Zilli, C.; Prevosto, L.; Mancinelli, B.; Santa-Cruz, D.; Yannarelli, G.; Balestrasse, K. Enhancement of Soybean Nodulation by Seed Treatment with Non–Thermal Plasmas. Sci. Rep. 2020, 10, 4917. [Google Scholar] [CrossRef]
- Seddighinia, F.S.; Iranbakhsh, A.; Oraghi Ardebili, Z.; Nejad Satari, T.; Soleimanpour, S. Seed Priming with Cold Plasma and Multi-Walled Carbon Nanotubes Modified Growth, Tissue Differentiation, Anatomy, and Yield in Bitter Melon (Momordica charantia). J. Plant Growth Regul. 2019, 39, 87–98. [Google Scholar] [CrossRef]
- Li, K.; Zhong, C.; Shi, Q.; Bi, H.; Gong, B. Cold Plasma Seed Treatment Improves Chilling Resistance of Tomato Plants through Hydrogen Peroxide and Abscisic Acid Signaling Pathway. Free Radic. Biol. Med. 2021, 172, 286–297. [Google Scholar] [CrossRef]
- Ghasempour, M.; Iranbakhsh, A.; Ebadi, M.; Oraghi Ardebili, Z. Seed Priming with Cold Plasma Improved Seedling Performance, Secondary Metabolism, and Expression of Deacetylvindoline O-Acetyltransferase Gene in Catharanthus roseus. Contrib. Plasma Phys. 2020, 60, e201900159. [Google Scholar] [CrossRef]
- Tong, J.; He, R.; Tang, X.; Li, M.; Wan, J. Transcriptomic Analysis of Seed Germination Improvement of Andrographis Paniculata Responding to Air Plasma Treatment. PLoS ONE 2020, 15, e0240939. [Google Scholar] [CrossRef]
- Li, Y.; Song, Z.; Zhang, T.; Ding, C.; Chen, H. Gene Expression Variation of Astragalus Adsurgens Pall. through Discharge Plasma and Its Activated Water. Free Radic. Biol. Med. 2022, 182, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.H.; Yoo, S.; Choi, E.H.; Kim, S.B.; Oh, J. Biochemical and Molecular Characterization of Enhanced Growth of Panax Ginseng C. A. Meyer Treated with Atmospheric Pressure Plasma. J. Phys. Appl. Phys. 2020, 53, 494001. [Google Scholar] [CrossRef]
- Han, B.; Yu, N.-N.; Zheng, W.; Zhang, L.-N.; Liu, Y.; Yu, J.-B.; Zhang, Y.-Q.; Park, G.; Sun, H.-N.; Kwon, T. Effect of Non-Thermal Plasma (NTP) on Common Sunflower (Helianthus annus L.) Seed Growth via Upregulation of Antioxidant Activity and Energy Metabolism-Related Gene Expression. Plant Growth Regul. 2021, 95, 271–281. [Google Scholar] [CrossRef]
- Waskow, A.; Guihur, A.; Howling, A.; Furno, I. RNA Sequencing of Arabidopsis Thaliana Seedlings after Non-Thermal Plasma-Seed Treatment Reveals Upregulation in Plant Stress and Defense Pathways. Int. J. Mol. Sci. 2022, 24, 3070. [Google Scholar] [CrossRef]
- Ebrahimibasabi, E.; Ebrahimi, A.; Momeni, M.; Amerian, M. reza Elevated Expression of Diosgenin-Related Genes and Stimulation of the Defense System in Trigonella Foenum-Graecum (Fenugreek) by Cold Plasma Treatment. Sci. Hortic. 2020, 271, 109494. [Google Scholar] [CrossRef]
- Perez, S.M.; Biondi, E.; Laurita, R.; Proto, M.; Sarti, F.; Gherardi, M. Plasma Activated Water as Resistance Inducer against Bacterial Leaf Spot of Tomato. PLoS ONE 2019, 14, e0217788. [Google Scholar] [CrossRef]
- Gomes, M.P.; Garcia, Q.S. Reactive Oxygen Species and Seed Germination. Biologia 2013, 68, 351–357. [Google Scholar] [CrossRef]
- Jeevan Kumar, S.P.; Rajendra Prasad, S.; Banerjee, R.; Thammineni, C. Seed Birth to Death: Dual Functions of Reactive Oxygen Species in Seed Physiology. Ann. Bot. 2015, 116, 663–668. [Google Scholar] [CrossRef]
- Grene, R. Oxidative Stress and Acclimation Mechanisms in Plants. Arab. Book 2002, 1, e0036. [Google Scholar] [CrossRef]
- Bailly, C. The Signalling Role of ROS in the Regulation of Seed Germination and Dormancy. Biochem. J. 2019, 476, 3019–3032. [Google Scholar] [CrossRef]
- Sun, C.; Zhang, Y.; Liu, L.; Liu, X.; Li, B.; Jin, C.; Lin, X. Molecular Functions of Nitric Oxide and Its Potential Applications in Horticultural Crops. Hortic. Res. 2021, 8, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, E.; Navascués, P.; Gómez-Ramírez, A.; Molina, R.; BRENES, A.P.; García, J.L.; Cotrino, J.; Cantos, M.; Gonzalez-Elipe, A.R.; Lopez-Santos, C. Triggering Factors of Germination in Plasma Activated Cotton Seeds: Water Imbibition vs. Reactive Species Formation. J. Phys. Appl. Phys. 2021, 54, 325205. [Google Scholar] [CrossRef]
- Adhikari, B.; Adhikari, M.; Ghimire, B.; Park, G.; Choi, E.H. Cold Atmospheric Plasma-Activated Water Irrigation Induces Defense Hormone and Gene Expression in Tomato Seedlings. Sci. Rep. 2019, 9, 16080. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.F.; Sohan, M.S.R.; Hasan, M.; Miah, M.M.; Sajib, S.A.; Karmakar, S.; Khalid-Bin-Ferdaus, K.M.; Kabir, A.H.; Rashid, M.M.; Talukder, M.R.; et al. Enhancement of Seed Germination Rate and Growth of Maize (Zea mays L.) Through LPDBD Ar/Air Plasma. J. Soil Sci. Plant Nutr. 2022, 1–14. [Google Scholar] [CrossRef]
- Takaki, K.; Takahata, J.; Watanabe, S.; Satta, N.; Yamada, O.; Fujio, T.; Sasaki, Y. Improvements in Plant Growth Rate Using Underwater Discharge. J. Phys. Conf. Ser. 2013, 418, 012140. [Google Scholar] [CrossRef]
- Guo, Q.; Wang, Y.; Zhang, H.; Qu, G.; Wang, T.; Sun, Q. Alleviation of Adverse Effects of Drought Stress on Wheat Seed Germination Using Atmospheric Dielectric Barrier Discharge Plasma Treatment. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Vishwakarma, K.; Upadhyay, N.; Kumar, N.; Yadav, G.; Singh, J.; Mishra, R.K.; Kumar, V.; Verma, R.; Upadhyay, R.G.; Pandey, M.; et al. Abscisic Acid Signaling and Abiotic Stress Tolerance in Plants: A Review on Current Knowledge and Future Prospects. Front. Plant Sci. 2017, 8, 1–14. [Google Scholar] [CrossRef]
- Degutyte-Fomins, L.; PauŽaite, G.; Žūkiene, R.; Mildažiene, V.; Koga, K.; Shiratani, M. Relationship between Cold Plasma Treatment-Induced Changes in Radish Seed Germination and Phytohormone Balance. Jpn. J. Appl. Phys. 2020, 59, SH1001. [Google Scholar] [CrossRef]
- Pérez Pizá, M.C.; Prevosto, L.; Zilli, C.; Cejas, E.; Kelly, H.; Balestrasse, K. Effects of Non–Thermal Plasmas on Seed-Borne Diaporthe/Phomopsis Complex and Germination Parameters of Soybean Seeds. Innov. Food Sci. Emerg. Technol. 2018, 49, 82–91. [Google Scholar] [CrossRef]
- Wang, J.; Cui, D.; Wang, L.; Du, M.; Yin, Y.; Ma, R.; Sun, H.; Jiao, Z. Atmospheric Pressure Plasma Treatment Induces Abscisic Acid Production, Reduces Stomatal Aperture and Improves Seedling Growth in Arabidopsis Thaliana. Plant Biol. 2021, 23, 564–573. [Google Scholar] [CrossRef]
- Šerá, B.; Vanková, R.; Roháček, K.; Šerý, M. Gliding Arc Plasma Treatment of Maize (Zea mays L.) Grains Promotes Seed Germination and Early Growth, Affecting Hormone Pools, but Not Significantly Photosynthetic Parameters. Agronomy 2021, 11, 2066. [Google Scholar] [CrossRef]
- Bigeard, J.; Hirt, H. Nuclear Signaling of Plant MAPKs. Front. Plant Sci. 2018, 9, 469. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Zhang, S. MAPK Cascades in Plant Disease Resistance Signaling. Annu. Rev. Phytopathol. 2013, 51, 245–266. [Google Scholar] [CrossRef] [PubMed]
- Jasim, S.F.; Al-zubaidi, L.A.; Abdulbaqi, N.J. The Effect of Cold Plasma on Gene Expression of Major Genes in the Biosynthesis of Phenylpropanoids and Essential Oil Contents in Ocimum basilicum L. Ann. Rom. Soc. Cell Biol. 2021, 25, 14996–15010. [Google Scholar]
- Guragain, R.P.; Baniya, H.B.; Pradhan, S.P.; Dhungana, S.; Chhetri, G.K.; Sedhai, B.; Basnet, N.; Panta, G.P.; Joshi, U.M.; Pandey, B.P.; et al. Impact of Non-Thermal Plasma Treatment on the Seed Germination and Seedling Development of Carrot (Daucus carota sativus L.). J. Phys. Commun. 2021, 5, 125011. [Google Scholar] [CrossRef]
- Wong, M.L.; Medrano, J.F. Real-Time PCR for MRNA Quantitation. BioTechniques 2005, 39, 75–85. [Google Scholar] [CrossRef]
- Hayashi, N.; Yamamoto, K. Variations in Plant Growth Characteristics Due to Oxygen Plasma Irradiation on Leaf and Seed. Agronomy 2022, 12, 259. [Google Scholar] [CrossRef]
- Cui, D.; Yin, Y.; Li, H.; Hu, X.; Zhuang, J.; Ma, R.; Jiao, Z. Comparative Transcriptome Analysis of Atmospheric Pressure Cold Plasma Enhanced Early Seedling Growth in Arabidopsis Thaliana. Plasma Sci. Technol. 2021, 23, 085502. [Google Scholar] [CrossRef]
- Watanabe, S.; Ono, R.; Hayashi, N.; Shiratani, M.; Tashiro, K.; Kuhara, S.; Inoue, A.; Yasuda, K.; Hagiwara, H. Growth Enhancement and Gene Expression of Arabidopsis Thaliana Irradiated with Active Oxygen Species. Jpn. J. Appl. Phys. 2016, 55, 07LG10. [Google Scholar] [CrossRef]
- Tamošiūnė, I.; Gelvonauskienė, D.; Haimi, P.; Mildažienė, V.; Koga, K.; Shiratani, M.; Baniulis, D. Cold Plasma Treatment of Sunflower Seeds Modulates Plant-Associated Microbiome and Stimulates Root and Lateral Organ Growth. Front. Plant Sci. 2020, 11, 1347. [Google Scholar] [CrossRef]
- Mira, S.; Pirredda, M.; Martín-Sánchez, M.; Marchessi, J.E.; Martín, C. DNA Methylation and Integrity in Aged Seeds and Regenerated Plants. Seed Sci. Res. 2020, 30, 1–9. [Google Scholar] [CrossRef]
- Zhang, J.J.; Jo, J.O.; Huynh, D.L.; Mongre, R.K.; Ghosh, M.; Singh, A.K. Growth-Inducing Effects of Argon Plasma on Soybean Sprouts via the Regulation of Demethylation Levels of Energy Metabolism-Related Genes. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-B.; Kim, B.; Bae, H.; Lee, H.; Lee, S.; Choi, E.H.; Kim, S.J. Differential Epigenetic Effects of Atmospheric Cold Plasma on MCF-7 and MDA-MB-231 Breast Cancer Cells. PLoS ONE 2015, 10, e0129931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Do, H.L.; Chandimali, N.; Lee, S.B.; Mok, Y.S.; Kim, N.; Kim, S.B.; Kwon, T.; Jeong, D.K. Non-Thermal Plasma Treatment Improves Chicken Sperm Motility via the Regulation of Demethylation Levels. Sci. Rep. 2018, 8, 7576. [Google Scholar] [CrossRef]
- Braný, D.; Dvorská, D.; Strnádel, J.; Matáková, T.; Halašová, E.; Škovierová, H. Effect of Cold Atmospheric Plasma on Epigenetic Changes, DNA Damage, and Possibilities for Its Use in Synergistic Cancer Therapy. Int. J. Mol. Sci. 2021, 22, 12252. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Pizá, M.C.; Ibañez, V.N.; Varela, A.; Cejas, E.; Ferreyra, M.; Chamorro-Garcés, J.C.; Zilli, C.; Vallecorsa, P.; Fina, B.; Prevosto, L.; et al. Non-Thermal Plasmas Affect Plant Growth and DNA Methylation Patterns in Glycine Max. J. Plant Growth Regul. 2021, 1–11. [Google Scholar] [CrossRef]
- Li, L.; Chen, Z.; Acheampong, A.; Huang, Q. Low-Temperature Plasma Promotes Growth of Haematococcus Pluvialis and Accumulation of Astaxanthin by Regulating Histone H3 Lysine 4 Tri-Methylation. Bioresour. Technol. 2022, 343, 126095. [Google Scholar] [CrossRef]
- Martin, C.; Zhang, Y. The Diverse Functions of Histone Lysine Methylation. Nat. Rev. Mol. Cell Biol. 2005, 6, 838–849. [Google Scholar] [CrossRef]
- Švubová, R.; Kyzek, S.; Medvecká, V.; Slováková, Ľ.; Gálová, E.; Zahoranová, A. Novel Insight at the Effect of Cold Atmospheric Pressure Plasma on the Activity of Enzymes Essential for the Germination of Pea (Pisum sativum L. Cv. Prophet) Seeds. Plasma Chem. Plasma Process. 2020, 40, 1221–1240. [Google Scholar] [CrossRef]
- Kyzek, S.; Holubová, Ľ.; Medvecká, V.; Zahoranová, A.; Ševčovičová, A.; Gálová, E. Genotoxic Effect of Low Temperature Plasma Treatment on Plant Seeds. Toxicol. Lett. 2017, 280, S119. [Google Scholar] [CrossRef]
- Peťková, M.; Švubová, R.; Kyzek, S.; Medvecká, V.; Slováková, Ľ.; Ševčovičová, A.; Gálová, E. The Effects of Cold Atmospheric Pressure Plasma on Germination Parameters, Enzyme Activities and Induction of DNA Damage in Barley. Int. J. Mol. Sci. 2021, 22, 2833. [Google Scholar] [CrossRef] [PubMed]
- Ndiffo Yemeli, G.B.; Švubová, R.; Kostolani, D.; Kyzek, S.; Machala, Z. The Effect of Water Activated by Nonthermal Air Plasma on the Growth of Farm Plants: Case of Maize and Barley. Plasma Process. Polym. 2021, 18, 2000205. [Google Scholar] [CrossRef]
- Alkawareek, M.Y.; Alshraiedeh, N.H.; Higginbotham, S.; Flynn, P.B.; Algwari, Q.T.; Gorman, S.P.; Graham, W.G.; Gilmore, B.F. Plasmid DNA Damage Following Exposure to Atmospheric Pressure Nonthermal Plasma: Kinetics and Influence of Oxygen Admixture. Plasma Med. 2014, 4, 211–219. [Google Scholar] [CrossRef]
- Liu, T.; Huang, Z.; Gui, X.; Xiang, W.; Jin, Y.; Chen, J.; Zhao, J. Multi-Omics Comparative Analysis of Streptomyces Mutants Obtained by Iterative Atmosphere and Room-Temperature Plasma Mutagenesis. Front. Microbiol. 2021, 11, 630309. [Google Scholar] [CrossRef]
- Ottenheim, C.; Nawrath, M.; Wu, J.C. Microbial Mutagenesis by Atmospheric and Room-Temperature Plasma (ARTP): The Latest Development. Bioresour. Bioprocess. 2018, 5, 12. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.-F.; Li, H.-P.; Wang, L.-Y.; Zhang, C.; Xing, X.-H.; Bao, C.-Y. Atmospheric and Room Temperature Plasma (ARTP) as a New Powerful Mutagenesis Tool. Appl. Microbiol. Biotechnol. 2014, 98, 5387–5396. [Google Scholar] [CrossRef]
- Li, J.; Wang, L.-Y.; Zhao, X.-T.; Fang, C.; Su, N.; Su, Y.-F.; Zhang, C.; Xu, Y.; Lv, M.; Li, H.-P.; et al. Investigations on Cold Atmospheric Plasma Jet for Plant Mutation. In Proceedings of the 4th Asia-Pacific Conference on Plasma Physics, Jeju, Korea, 26 October 2020; p. 1. [Google Scholar]
| Plasma Source | Gas Feeder | Seeds/Plants | Focus of the Study | Molecular and Physiological | Citation |
|---|---|---|---|---|---|
| CCP RF | Air | Echinacea purpurea (Coneflower) | Growth, | Vitamin C, Chlorogenic acid, Rutin | [145] |
| DBD | Argon (Ar) | Cannabis sativa (Hemp) | Growth, metabolism | WRKY1 transcription factor, Cannabinoid genes (CBDAS, THCAS, OAC, OLS) | [146] |
| DBD | Air | Vigna mungo L. (Black gram) | Germination and growth | Soluble protein, SOD, CAT | [47] |
| DBD | Helium (He) and O2 (Oxygen) | Vitis vinifera cv. Muscat of Alexandria | Seed dormancy | Proline, malondialdehyde (MDA), Catalase | [147] |
| CCP RF | Air | Maize Wheat Lupine | Growth, metabolism, disease resistance | Phenol, anthocyanin, proline, | [148] |
| CCP RF | Air | Helianthus annus L. (Sunflower) | Growth, metabolism | Phytohormone, protein expression (proteoforms), Protein interaction network | [42] |
| DBD | Air | Arabidopsis thaliana | Growth, Physio-biochemical | NO, nitrate, peroxynitrite, H2O2 Superoxide content in the seeds. MDA, proline, CAT, SOD, POD. Ca2+ expression, atomic ratio | [149] |
| DBD | Air | Wheat | Growth, metabolism | MDA, NO3− content in the seeds | [87] |
| DBD | Ar | Astragalus fridae | Growth, metabolism, nano particle effects, tissue differentiation | Chlorophyll, carotenoid, nitrate reductase, catalase | [150] |
| DBD | N2, O2 | Soybean | Growth, Biophysical, metabolism | Chlorophyll, MDA, CAT, GPOX, SOD | [151] |
| DBD | Ar | Melissa officinalis | Growth, metabolism, nano particle effects | Peroxidase, PAL | [152] |
| DBD | Air | Arabidopsis thaliana Col-0, gl2, and gpat5 | Biophysical, growth | GL2 and GPAT5 for the embryogenesis and Peroxidase 69 (AT5G64100) | [37] |
| RF | O and Ar | Ocimum basilicum L. (Basil) | Growth, metabolism | Carbohydrate and protein content, Catalase activity | [153] |
| Plasma enhanced chemical vapor deposition | Ar | Wheat | Biophysical, growth, disease resistance | Protein content | [154] |
| DBD | Air, Ar, O2 | Rapeseed, Mustard | Growth, metabolism | Soluble protein, Chlorophyll, SOD, APX, CAT. Gene expression of BnSOD, BnAPX, BnCAT in root | [48] |
| DBD | Air, Ar, O2 | Wheat | Biophysical, growth, metabolism | Chlorophyll content, cadmium, electrolyte, total protein, H2O2, NO, APX, SOD, CAT. Gene expression of TaLCT1, TaHMA2, TaSOD, TaAPX, TaCAT | [155] |
| DBD | Air, Ar, O2 | Wheat | Biophysical | - | [156] |
| RF | N2, O2 | Asparagus | Biophysical, growth | - | [157] |
| ICCP RF | He | Tomato | Growth | N, P, K content | [158] |
| CCP RF | N2 | Artichoke | Biophysical, growth, metabolism | POD, CAT content | [159] |
| DBD | He | Coffee, grape seeds | Growth | - | [49] |
| Gliding arc | Air | Pea Zucchini | Growth | - | [160] |
| DBD | Air | Thurigan Mallow | Biophysical, growth | - | [50] |
| LFGD | Ar and Air | maize (Zea mays L.) | Biophysical, growth | CAT, SOD, APX, GR content, total soluble protein, sugar, fat content, chlorophyll | [161] |
| DBD | Air | Rice | Gene expression of ABA catabolism (OsABA8′OH1−3), ABA biosynthesis genes (OsNCED1−5), α-amylase gene (OsAmy1–3), | [162] | |
| DBD | N2, O2 | Soybean | Biophysical, metabolism | Phytohormones (indole acetic acid/IAA and trans-zeatin riboside/tZR) glutathione, nitrogen activity, leghemoglobin content in nodule and GmEXP1 gene expression in roots | [163] |
| DBD | Air | Bitter melon (Momordica charantia) | Growth | - | [164] |
| DBD | Air | Tomato | Chilling resistance, metabolism | Phytohormone, hydrogen peroxide and abscisic acid signaling gene (RBOH1, NCED1, NCED2, ICE1, CBF1) | [165] |
| DBD | Ar | Astragalus fridae | Biophysical, metabolism | Chlorophyll, expression of phenylalanine ammonia lyase (PAL), and universal stress protein (USP). | [150] |
| DBD | Ar | Catharanthus roseus | Biophysical, metabolism | Chlorophyll, carotenoids, phenylalanine ammonia lyase (PAL) enzyme and deacetyl vindoline O-acetyltransferase (DAT) gene | [166] |
| DBD | Air | Andrographis paniculata | Biophysical, growth | Expression gene related to plants hormone (ACO, NCED5, CRF4, NRT1, RAP2-10, ERF098, and PRP3) | [167] |
| Needle array plate DBD | Air | Astragalus adsurgens Pall. | Biophysical, metabolism | - | [168] |
| Plasma Air-jet | Air | Tomato | Biophysical, metabolism | Antioxidants, phytohormones, and expression of defense genes (POD, CAT, PPO, SOD, GST, AOX, OPR, PAL, HAT, HFMET, MPK, and RBOH) | [15] |
| DBD | Air | Panax ginseng | Biophysical, metabolism, pathogenic defense system | Chlorophyll, total phenolic and pathogen-associated gene (PgPR2, PgPR5, PgPR10, and PgCAT) | [169] |
| DBD | Ar | Helianthus annus L. (Sunflower) | Biophysical, metabolism | Protein content, antioxidant enzyme activity, and RNA-seq | [170] |
| SDBD | Air | Arabidopsis thaliana | Plant defense | RNA-seq young seedling | [171] |
| SDBD | Air | Tobacco (Nicotiana tabacum) | Plant development, gene expression | Morphology, root and root hair development genes (NtCOBL, NtXTH5, NtXTH9, NtXTH15, NtXTH17, NtXTH27) | [73] |
| SDBD | Air | Arabidopsis | Plant development, gene expression | Morphology, root and root hair development genes (AtCOBL9, AtAUX1, AtLAX3, AtOBP4, AtXTH17) | [72] |
| DBD | Air | Wheat | Germination, growth, metabolism | CAT, SOD, gene expression (TaCAT, TaSOD) | [88] |
| DBD | Ar | Fenugreek (Trigonella foenum-graecum) | Metabolism, secondary metabolite, gene expression | Content of protein, carotenoid, chlorophyll, CAT, GPX, APX. Gene expression (SEP, SQS, CAS, SSR, SMT) | [172] |
| DBD | Air | Tomato | Plant defense, gene expression | Expression of defense genes (PAL, ERF1, PR1a, PR4, PR5) | [173] |
| Diffuse Coplanar Surface Barrier Discharge (DCSBD) | Air, O2, N2 | Maize | Plant growth, metabolism, gene expression | Enzyme activity (protease, glucanase, POX, SOD), expression of heat shock protein genes (HSP101, HSP70, HSF17) | [137] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Priatama, R.A.; Pervitasari, A.N.; Park, S.; Park, S.J.; Lee, Y.K. Current Advancements in the Molecular Mechanism of Plasma Treatment for Seed Germination and Plant Growth. Int. J. Mol. Sci. 2022, 23, 4609. https://doi.org/10.3390/ijms23094609
Priatama RA, Pervitasari AN, Park S, Park SJ, Lee YK. Current Advancements in the Molecular Mechanism of Plasma Treatment for Seed Germination and Plant Growth. International Journal of Molecular Sciences. 2022; 23(9):4609. https://doi.org/10.3390/ijms23094609
Chicago/Turabian StylePriatama, Ryza A., Aditya N. Pervitasari, Seungil Park, Soon Ju Park, and Young Koung Lee. 2022. "Current Advancements in the Molecular Mechanism of Plasma Treatment for Seed Germination and Plant Growth" International Journal of Molecular Sciences 23, no. 9: 4609. https://doi.org/10.3390/ijms23094609
APA StylePriatama, R. A., Pervitasari, A. N., Park, S., Park, S. J., & Lee, Y. K. (2022). Current Advancements in the Molecular Mechanism of Plasma Treatment for Seed Germination and Plant Growth. International Journal of Molecular Sciences, 23(9), 4609. https://doi.org/10.3390/ijms23094609







