Inflammation: A New Look at an Old Problem
Abstract
:1. Introduction
2. Cellular Stress as a Functional Unit of Pro-Inflammatory Tissue Stress
2.1. General Characteristics of Cellular and Tissue Stress
2.2. Triggers of Cellular and Tissue Stress, and Response Regulation
- Any damage to macromolecules (in cells and extracellular matrix) that is recognizable by CS sensors [11].
- Potentially dangerous disturbances of key homeostasis parameters: acid–base balance, temperature, osmotic and hydrostatic pressure, changes in cytoplasmic and mitochondrial levels of calcium cations and other electrolytes, and decrease in cellular concentrations of ATP, oxygen (hypoxia), and some metabolites [4].
- Of special note are the lipotoxicity factors that act on mitochondria and other cellular structures. These include: excessive contents of saturated free fatty acids (FFA), diacylglycerol, and ceramides, as well as modified carnitine, non-esterified cholesterol, and some other hydrophobic molecules [4,12,13,14].
- Recognition of alarm signals from pathogens and damaged tissues by the pattern-recognizing receptors (PRRs) of cells directly associated with inflammation: immunocytes, epitheliocytes, connective tissue cells, and endotheliocytes [15]. PRR ligands are represented by conserved microbial structures—pathogen-associated molecular patterns (PAMPs)—and endogenous, damage-associated molecular patterns (DAMPs). Receiving signals via PAMPs and DAMPs, cells can rapidly enter into a state of stress prior to being damaged and realize their pro-inflammatory and immunocompetent functions. Particularly noteworthy among the PRRs are two families: toll-like receptors (TLRs) and intracellular NOD-like receptors (NLRs) [16].
- Antigen recognition by antibodies (with subsequent action of immune complexes on cells) and by T-cell receptors (TCR), leading to a strong activation effect on both the T-lymphocytes and the cells interacting with them, above all the antigen-presenting cells.
- The action of various activators of the complement, hemostasis, and kallikrein–kinin systems followed by the effect of the activation products of these systems on various cells.
- Excitotoxicity in the ‘narrow sense’ is the toxic effect of high doses of glutamate [17,18] and some other neurotransmitters and their catabolic products [19] on neurons; in the ‘broad sense’, it is the pathological hyperactivation of cells by various regulatory molecules, primarily pro-inflammatory cytokines such as TNF-α and IL-1β. The latter manifestation of excitotoxicity is most prominent in the cytokine storm syndrome [20], including severe COVID-19 [21,22]. The phenomenon of cytokine excitotoxicity makes the development of CS ‘contagious’.
2.3. Particular Typical Processes of Cellular Stress
- Oxidative stress—Oxidative stress develops in a cell when the accumulation of pro-oxidants disturbs the redox equilibrium and causes an imbalance between the oxidants and antioxidants in favor of the oxidants [37,38]. The accumulation of ROS in the nucleus contributes to DNA damage, so the redox equilibrium in this cellular compartment is relatively stable. The main site of ROS formation under CS is the mitochondria [39]. In the cytoplasm, ROS generation occurs with the participation of cytochrome C released from the mitochondria as well as NADPH-oxidases of microsomal oxidation, 5-lipoxygenase, xanthine oxidase, and cytochrome P-450. They directly or indirectly activate many of the receptors, transcription factors (TFs), and protein kinases associated with CS development. Both excessive and insufficient development of oxidative stress in response to damage can be a trigger for the onset and progression of a wide range of human diseases [40,41,42].
- DNA-damage response (DDR)—Cells have developed the capacity for DDR to be able to control genotoxic stress and maintain accurate transmission of genetic information to subsequent generations. The accumulation of DNA damage in the cell leads to a number of alternative outcomes of DDR, including cell cycle arrest, senescence, malignization, or apoptosis [43]. In human cells, more than 1000 proteins are involved in the DDR process. These are primarily nuclear chaperones (ubiquitin, nucleophosmin and SUMO protein), nuclear protein kinases (ATM, ATR, DNA-PKcs and Chk1/2), various nucleases, polymerases, ligases and DNA glycosylases, and many TFs, especially p53. The main function of DDR is to stop the cell cycle to enable DNA repair and cell survival [35]. At the same time, the process of apoptosis is an extreme variant of DDR aimed at preventing malignization and making it impossible to transmit genetic abnormalities to daughter cells.
- Mitochondrial stress, including mitochondrial unfolded protein response (UPRmt)—Mitochondria are the main donors of ATP and ROS, and the end point of catabolism and the starting point for anabolism. There are approximately 1500 proteins functioning in human mitochondria, of which only 13 are encoded in mitochondrial DNA (mtDNA) [44]. These are mainly the most important proteins of the mitochondrial respiratory complexes. The mitochondrial proteome is tuned to the functional status of its cell and depends on the action of activating and damaging factors on the mitochondrion itself. The extreme connection between the mitochondria and the cell nucleus is a well-established phenomenon that occurs in response to mitochondrial dysfunction. Various injuries in the proteome and mtDNA, including the accumulation of denatured proteins in the mitochondria, cause UPRmt development. UPRmt involves multidirectional changes in the biosynthesis of various mitochondrial proteins (reduction in potentially toxic proteins); and increased production and transport into the mitochondria of chaperones capable of repairing damaged mitochondrial proteins [45]. Integrative mitochondrial stress is primarily associated with the activation of ATF4 (activating transcription factor 4) [46] and the production of heat shock proteins (HSPs) and many kinases that integrate mitochondria into the CS system. Mitochondrial stress is aimed at eliminating mitochondrial damage and dysfunction. However, under certain scenarios, this program complex may fail to perform effectively, because individual mechanisms of mitochondrial stress may themselves become involved in the vicious pathogenetic circle that is characteristic of many diseases [18,47].
- Stress of the endoplasmic reticulum (ER), including calcium-dependent mechanisms and UPRER—The disruption of ER integrity or accumulation of misfolded proteins in these cellular compartments initiates ER stress, primarily in the form of UPRER [33]. The UPRER process aims to restore an altered ER homeostasis by pursuing the following main objectives: (1) suspension of the synthesis and excretion of secretory proteins from the cell; (2) increased transcription of chaperones and other proteins involved in protein folding and protein maturation; (3) induction of denatured protein degradation via the ER-associated degradation complex (ERAD) [48]. UPRER is mediated by three main transmembrane sensors: (1) the inositol-requiring enzyme 1 (IRE1), (2) the protein kinase PERK, and (3) the transcription factor ATF6. They are all preserved in an inactive state, mainly by virtue of the BiP/GRP78 chaperone coupled to them [49]. Under ER stress, this chaperone binds to and is blocked by various unfolded proteins and thereby releases UPRER inducers in the active state. Thus, signal transduction via PERK, IRE1, and ATF6 provides a coordinated response that contributes to overcoming the impaired ER proteostasis. Prolonged or intensity-critical UPRER, in turn, induces apoptosis through several pathways, including excess Ca2+ release into the cytoplasm from the ER. However, because ER stress activates the anti-apoptotic pathways (anti-apoptotic proteins of the Bcl-2 family) as well, apoptosis is an extreme and far from the only variant of the ER stress outcome [33].
- Response of inducible HSPs, including their participation in the UPR [50]. The HSPs response is an evolutionarily ancient and highly conserved molecular response of the cell to disturbances in its protein homeostasis (proteostasis) [51]. HSPs are the main chaperones of UPR. In addition, they perform numerous regulatory functions that influence almost all major CS processes [52].
- Inhibition (during cell growth) or intensification of autophagy processes (utilization of altered organelles and macromolecules) and other manifestations of lysosomal stress—Autophagy is a catabolic process involving a lysosomal phase, which is conserved in the evolution of all eukaryotes and runs (or occurs) in all human cells. Autophagy is part of many physiological or pathological processes, and its severity increases significantly during starvation and severe CS [53]. In these cases, autophagy usually promotes cell survival. Normally, most damaged and short-lived proteins degrade by the proteasome pathway after they have been marked with ubiquitin. In CS, the ubiquitin–proteasome pathway is overloaded and could act as an additional autophagy activating mechanism [54]. Besides, many long-lived proteins, large protein aggregates, and individual organelles can only be utilized by the process of autophagy with the participation of lysosomes and numerous supporting protein factors. In particular, mitophagy is the only mechanism for the physiological recycling of mitochondria [55]. Thus, autophagy largely determines the balance between protein biosynthesis, organelle biogenesis, and organelle degradation. Moreover, autophagy can also be crucial in preventing cell apoptosis or necrosis by removing damaged and pathologically activated mitochondria as well as various protein complexes and intracellular parasites. Autophagy is broadly divided into three main types and has several levels of regulation by CS mechanisms [56]. As cells age, autophagy regulation and realization may be disbalanced [57]. In particular, in normal aging and neurodegenerative diseases, the balance between the number of mitochondria (directly dependent on mitophagy intensity) and their degree of dysfunction (also dependent on mitophagy, but in the opposite way) may be maintained or disturbed [58].
- Inflammasome formation—Inflammasome is a multimeric cytosolic protein complex with sensory molecules in the form of intracellular PRRs of two families: NLRs (mostly) or absent in melanoma 2-like receptors (ALRs). During protein complex assembly, these receptors bind to procaspase-1, after which procaspase-1 is converted to caspase-1. Caspase-1 then induces the processing of IL-1β and IL-18 and, under certain conditions, the development of pyroptosis (programmed necrosis) [59]. Inflammasome formation is a sign of a relatively pronounced pro-inflammatory stress of immunocytes, epitheliocytes, endotheliocytes, and some other cells. Several additional conditions are necessary for the formation of an inflammasome, such as the activation of pro-inflammatory signaling pathways associated with the transcription factor NF-κB, oxidative stress buildup, and a decrease in the K+ concentration of the cytoplasm. The NLRP3 inflammasome assembly process is activated by the greatest variety of factors—namely: PAMP, DAMP, ROS, lysosomal proteinases, cholesterol crystals, β-amyloid, uric acid (metabolic DAMP), calcium phosphates, many exogenous irritants (e.g., asbestos and silicon), mtDNA release from mitochondria into cytoplasm, and recognition of internal and external PRRs signals [60,61,62]. The biological role of inflammasome formation is to enhance the development of inflammation and the immune response through pyroptosis, IL-1β production, and other factors [63]. However, the disruption of restrictive control over inflammasome formation can cause severe complications, especially in genetically determined autoinflammatory diseases [64].
- Formation of stress, non-coding RNAs, microRNAs—MicroRNAs (miRNAs) are small non-coding RNAs that, like long non-coding RNAs, have the ability to modulate gene expression at the post-transcriptional level either by inhibiting matrix RNA (mRNA) translation or by stimulating mRNA degradation [65]. The involvement of both types of non-coding RNAs in the pathologies associated with the development of CS has now become evident [40,66]. Moreover, miRNAs can also regulate CS development through intercellular communication, through the effects of extracellular vesicles containing non-coding RNAs [67].
- Formation of stress granules – At the post-transcriptional stage, RNA-binding proteins (RBPs) are a key contributor to the stress-induced regulation of the destiny and function of various RNAs [68]. At the same time, the function of stress granules down to a few microns in size, consisting of RNA and protein, is not yet fully understood [69]. Additionally, CS can induce in cells the formation of gel-like structures, including ones involving amyloid and prion-like proteins [70]. The formation of these structures is dynamic; they condense or dissolve quickly and are therefore ideal for participating in urgent cellular adaptation to stresses.
- Formation of an intracellular network of cellular stress signaling pathways—At the cell level, stress development is mediated by complex programs of epigenetic control and intertwining of signaling pathways whose protein elements are continually undergoing multiple posttranslational modifications [71]. Along with that, various extracellular and intracellular stress signals can activate common collector-type protein kinases (e.g., MAPK, Akt, PI3K, PKC, ATM, ATR, AMPK, PKA, PKR, mTOR) and key universal cellular stress transcription factors (e.g., NF-κB, p53, AP-1, HIF, HSF, NRF2, ATF4) in different cells. The same signaling molecules can be activated in different ways and participate in differently directed processes. However, in general, the polyfunctional factors may feature certain functional preferences. Thus, the key role in the development of CS in hypoxia is attributed to HIF-1 (hypoxia-inducible factor-1) [72]; in HSP production, to HSF1 (heat shock factor 1) [73]; in oxidative stress, to NRF2 which triggers antioxidant production through a negative feedback mechanism [74]; while ATF4, as already noted, plays a determining role in UPRmt development. The dynamic network of signaling pathways integrates the different elements of the CS into a single whole, including the receptor and secretory phenotype of pro-inflammatory cells (Figure 3).
- Formation of pro-inflammatory receptor and secretory cell phenotype—Almost all nucleated cells, when activated, express inducible receptors and secrete a spectrum of inflammatory mediators, including cytokines [4,75,76]. This fact determines the possibility of cytokine network formation in all possible variants of TS. Thus, the emergence of a pro-inflammatory phenotype in a large number of cells at once causes a network effect with TS development [77].
2.4. Outcomes of Cellular Stress
- Cell adaptation to a damaging factor. By virtue of their ability to respond to CS, cells can become resistant to the damaging factor and recover intracellular and tissue homeostasis. Having achieved partial adaptation to the prolonged action of damaging factors, cells can sustain their pro-inflammatory status, forming a state of tissue allostasis.
- Apoptosis is an essential component of various processes, including normal cell turnover, proper development and functioning of the immune system, hormone-dependent atrophy of unnecessary tissues, embryonic development, and death of damaged or malignant cells without pro-inflammatory response [78]. Meanwhile, a level of apoptosis that exceeds the regenerative capacity of the organ promotes tissue atrophy and, consequently, sclerosis of the parenchyma [79,80]. The process of apoptosis is induced by many signaling pathways which can be subdivided into ‘extrinsic’ and ‘intrinsic’ depending to varying degrees on caspase engagement. External proapoptotic signals are directed towards receptors for cytokines of the TNF family, which are involved in the activation of proapoptotic caspases. The main intrinsic pathway of apoptosis is the result of increased mitochondrial permeability and the release of pro-apoptotic molecules into the cytoplasm, primarily of cytochrome C (activates caspase 9 and then other pro-apoptotic caspases) [81]. The mitochondrial response, in turn, is triggered and controlled by pro-apoptotic and anti-apoptotic proteins of the Bcl-2 family, IAP, and many other factors [82,83]. Dead, fragmented cells produced by apoptosis are rapidly taken up by stromal macrophages for final degradation without significant DAMP formation. Caspases that are involved in apoptosis are divided into initiators (2, 8, 9, 10, 12) and effectors (3, 6, 7) [81,84]. The complexity of the mechanisms of apoptosis regulation [85] determines the fact that this process may manifest itself in the development of different variants and stages of CS.
- Programmed cell necrosis: necroptosis, pyroptosis, NETosis, parthanatos, autophagia, “cornification”, oxytosis, ferroptosis, secondary necrosis, oncosis, sarmoptosis, autosis, autolysis, paraptosis, and “mitotic crash” [4,86,87,88,89,90,91,92,93,94,95]. The numerous designations of this process reflect differences in the signaling pathways and in the biochemical and morphological features of the process, including the activation of necrosis-specific caspase types [86]. Thus, we can distinguish several variants of programmed necrosis, which are associated with high pro-inflammatory levels of CS and the formation of high concentrations of DAMP: pyroptosis (associated with inflammasome formation) [63]; NETosis (neutrophil extracellular traps formation), which was originally associated only with neutrophils, but was later discovered in other professional phagocytes [90,91,92]; autophagic cell death is a term widely used to describe cases of cell death accompanied by massive cytoplasmic vacuolization [93]; necroptosis, associated with the activation of receptor-interacting protein kinase 1 and 3 (RIPK1, RIPK3) and formation of a protein complex known as necrosome [94]; secondary necrosis, which occurs when apoptotic cells are not cleared in a timely manner and the process progresses to a “late apoptosis” phenomenon [95].
- Metaplasia is associated with the development of CS and TS, e.g., in the metaplasia of airway epithelium [96], endometrium [97], or connective tissue [98]. Gastric epithelium metaplasia occurs against the background of inflammation and atrophic changes (especially of the glands) of the gastric mucosa [99]. The process of metaplasia tends to be progressive and presents a risk of malignization of the relevant tissue [99].
- Cell malignancy and malignant tumor formation are associated with failure of the DDR mechanisms, retention of multiple mutations useful for tumor cell survival but harmful to the organism, and formation of a ‘parasitic’ genome in tumor cells.
- Cell aging is caused by the stochastic accumulation of damage in biomolecules (in the genome, transcriptome, proteome) that are vital for proper cellular function. These changes provoke CS with ROS accumulation, cell cycle blockade, and cellular and tissue allostasis formation [100,101]. The aging process affects all cell types, including stem cells [102]. Cell aging entails a state of irreversible arrest of proliferation in which cells remain metabolically active and secrete a number of pro-inflammatory and proteolytic factors and other components of the senescence-associated secretory phenotype (SASP) [103]. Cell aging is characterized by morphological transformations, namely: high level of β-galactosidase (SA-β-gal) expression, accumulation of cyclin-dependent kinase 2A inhibitor protein p16INK4a, SASP, formation of heterochromatin foci (SAHF), accumulation of aberrant protein aggregates and granules in cells, telomere shortening, and oxidative stress [43,104,105]. Cell aging is an alternative (to malignancy) means of CS development, in which the cell continues to accumulate sublethal damage, which may finally lead it to some variant of cell death or persistent dysfunction. SASP includes growth factors, cytokines, and extracellular proteases that modulate most of the both beneficial and detrimental microenvironment phenotypes caused by ageing cells [106]. In this case, cell aging and pro-inflammatory SASP may form a vicious pathogenetic circle involved in the formation of aging tissue allostasis [107].
2.5. Stages of Cellular and Tissue Stress
2.5.1. Stage 1
2.5.2. Stage 2
2.5.3. Stage 3
2.6. The Physiological Role of Cellular and Tissue Stress
3. Tissue Stress as a Common Pathogenetic Platform for Modeling Basic General Pathological Processes in Humans
3.1. Tissue Stress Variants in the Focus of a Classical Inflammation
3.2. Typical Patterns of Autoimmune Pathologies Can Be Considered as a Special Form of General Pathological Inflammatory Process
- Molecular mimicry of microbial proteins.
- Bystander activation—the release of autoantigens from tissue damaged by inflammation.
- Breakdown of biological barriers in immunoprivileged organs (central nervous system, eyes, testes, placenta), opening access to potential autoantigens for adaptive immunity.
- Polyclonal activation of lymphocytes in response to microbial super-antigens, or other factors activating potentially autoreactive T- and B-lymphocyte clones.
- Epitope spreading, a situation where autoimmune response targets do not remain the same but can diversify to include other epitopes on the same protein or on other proteins in the same tissue.
- Deficiency of the immunosuppressor vector i-reg in the processes of immune inflammation development.
3.3. Tumor Tissue Is under Tissue Pro-Inflammatory Stress
3.4. Chronic Systemic Low-Grade Inflammation
- ChLGI is a manifestation of tissue stress in response to local or systemic damage at sub-threshold levels for the development of classical and systemic inflammation, respectively.
- The key triggers of ChSLGI are metabolic factors including: modified proteins (denatured, oxidized, glycated), high concentrations of saturated FFA and oxidized low-density lipoproteins (oxLDL), homocysteine, and many other metabolites. The progressive accumulation of genome, proteome, and metabolome injuries during aging contributes to the body’s pro-inflammatory status and the development of ChSLGI. Of particular importance in the development of ChSLGI are scavenger receptors of stromal macrophages, endotheliocytes, and some other cells, with these receptors being associated with metabolism, immunity, and inflammation [26].
- ChSLGI is characterized by moderate manifestations of SIR, namely: the elevation of C-reactive protein in the blood is usually in the borderline range of 3–10 mg/mL (a criterion for metabolic syndrome), and the elevation of pro-inflammatory cytokines is usually no more than 2–4 times over the upper normal range; the signs of significant tissue decay and systemic coagulopathy are not characteristic; the signs of organ dysfunction develop slowly as part of allostasis; and there is no direct association of these changes with systemic manifestations of infections and autoimmune diseases, i.e., with systemic manifestations of classical inflammation [221,222].
- The differentiation of local ChLGI from ChSLGI makes sense in the presence of a clinical presentation of these local abnormalities, for example in diabetic kidney disease [123].
- ChLGI involves a large number of parenchymatous and stromal cells of various organs, with relatively little involvement of inflammatory ‘professional cells’ (leukocytes and their progeny characteristic of the inflammatory focus). Therefore, ChLGI has no barrier function and no visible signs of classical inflammation.
- A key and integrating pathogenetic phenomenon of ChSLGI is endotheliosis, more specifically the pathological activation and dysfunction of endotheliocytes with the disruption of endothelial glycocalyx integrity in different parts of the vascular network [223].
- In ChSLGI, interrelated changes occur in key facultatively glycolating tissues (fat, muscle, liver), which leads to the development of insulin resistance and additional disturbance of metabolic homeostasis [57,224,225,226,227]. Therefore, the clinical presentation of ChSLGI is associated with morbid obesity, metabolic syndrome, sarcopenia, and type 2 diabetes mellitus. At the same time, the role of cellular and tissue aging is evident in the pathogenesis of these pathologies [227,228]. Moreover, atherosclerosis, osteoarthritis, neurodegeneration, hypertension, and chronic heart failure are typical local phenomena in aging and ChSLGI [229,230,231].
3.5. Para-Inflammatory Neurodegeneration
- The brain does not use higher fatty acids for energy generation, which reduces the effects of lipotoxicity factors on it. However, the brain depends critically for its energy production on aerobic glycolysis (the brain consumes ~20% oxygen under normal conditions having a mass of ~2%). Therefore, neurons are highly sensitive to glucose transport, hypoxia, and mitochondrial stress [244,245], and cognitive disorders are characteristic companions of vascular pathologies [246].
- Most neurons are postmitotic cells, for which the typical outcomes of cellular stress are ageing, apoptosis, or programmed necrosis [251]. These processes depend not only on age [252], but also on genetic and environmental risk factors for neurodegeneration [253,254]. Therefore, neurodegenerations, for example, in normal ageing, Alzheimer’s or Parkinson’s disease display the specific characteristics of proteinopathies and their localizations [255].
3.6. Atherosclerosis
- An important role in the pathogenesis of atherosclerosis belongs to scavenger receptors, in particular: SR-E1 (LOX-1) and SR-B2 (CD36) are involved in endotheliocyte activation, whereas SR-A1 (CD204) and SR-B2 are involved in the uptake of modified LDL by atherogenic macrophages [292,293,294,295]. In addition, endotheliocytes and macrophages are atherogenically activated by SR-J1 receptors (RAGE), which recognize advanced glycationend-products (AGEs) [26,296]. In contrast, some macrophage SRs (SR-B1 (SCARB1), SR-L1 (CD91, LRP1), SR-I1 (CD163)) and vascular myocytes SR-L display antiatherogenic activity [26,297,298].
- As for endothelial dysfunction associated with atherosclerosis, cardiovascular diseases, tissue ischemia, and hypoxia, it increases the pro-inflammatory status of various organs and the organism as a whole [123].
3.7. Systemic Inflammation as a General Pathological Process
4. Evolutionary Trends in the Development of Inflammation
- The development of tissue pro-inflammatory stress based on non-adaptive, innate immunity mechanisms is characteristic of all metazoans. Thus, invertebrates have all the basic protective mechanisms of phagocytes, including: a variety of PRRs, hydrolases, free radicals, cationic proteins, extracellular DNA traps, etc. [310]. For example, compared to mammals, some echinoderm species have about an order of magnitude greater variety of extracellular and intracellular PRRs of the two most important families, TLR and NLR [311]. Invertebrates, as well as vertebrates, also have the problem of immune system ageing related, among other things, to cellular stress mechanisms [312]. Another variant of the cell/tissue stress outcome—i.e., tumorigenesis—is also characteristic of invertebrates [313].
- Highly organized invertebrates that have hemocytes, hemolymph, and a neuroendocrine system are capable of responding to damage and infection by developing SIR, which consists in the accumulation of stress hormones and neurotransmitters [314], some hemocyte populations and a variety of bactericidal and pro-inflammatory molecules, including cytokine-like factors in hemolymph [315,316,317,318,319,320,321]. The hemostasis system in invertebrates is not specialized and is mainly represented by cells (hemocytes) and adhesive molecules of the immune system [322,323]. In some invertebrates, such as insects, the innate immune system is capable of adaptive responses that usually provide a short-term acquired resistance to viral and extracellular infections [324,325,326].
- The development of classical inflammation and the emergence of the lymphocytic adaptive immune system and the progressive hemostasis system became possible only in vertebrates due to the emergence of an elementary basis of microcirculation in them—microcirculatory units including vascular (precapillary arterioles, capillaries, capillary sphincters, postcapillary venules) and extravascular transport communications that ensure exchange processes between blood and a particular tissue area [327,328]. This determined not only the possibility of directed and selective leukocyte migration, but also the appearance of interrelated components of the exudative vascular complex (EVC), including the microvascular network, mast cells, and complement, kininogenesis, and hemostasis plasma systems (Table 3). Vertebrates starting with bony fish reveal orthologues of major TFs and cytokines that are specific to different T-cell immune response vectors (i) [329,330] and provide the development of specific productive inflammation directed towards a particular infectious factor [329,330].
- 4.
- The EVC and immune system of a more advanced level in higher vertebrates (reptiles, birds, and mammals) are responsible for the possibility of development of the most traumatic variants of exudative–destructive inflammation such as caseous necrosis and, in mammals, purulent inflammation (Table 3). At present, it can be confidently stated that systemic inflammation (systemic ‘inflammatory microcirculation’) can occur only in mammals and isolated manifestations of systemic inflammation may be found in birds.
5. Discussion
- The use of clinical criteria alone is insufficient for the verification of complex pathological processes, e.g., metabolic syndrome criteria for the verification of ChSLGI, or Sepsis-3 and SIR criteria for the verification of systemic inflammation as a general pathological process.
- In molecular research in an in vitro system, there will be a need for a more fundamental characterization of the cellular and tissue system of which the molecular mechanisms under study are a part.
- The development and practical use of clinical models will need to be harmonized with models of general pathological processes, which will objectively determine stricter requirements for theoretical training not only for scientific researchers, but also for practitioners.
- New scientific disciplines will probably need to be created, or existing disciplines—such as systems biology and integrative medicine—may need to be substantially modernized.
- The key objective of modern medicine, according to many specialists, should be the prevention of diseases and their complications, which will require a theoretical substantiation of the relationship between physiological and pathological processes, and characterization of transition zones between qualitatively different human pathological states.
- Pathology assessment methods will require additional sophistication along with broader use of computer network information technologies, including clinical decision support software.
- The distance between the avalanche-like accumulation of primary research data and their synthesis between analytical and synthetic approaches in medicine is increasing.
- In etiopathogenetic therapy, the famous Hippocratic postulate “Primum non nocere” (“First, do no harm”) is often violated. Applying the principles of evidence-based medicine limits these negative effects. However, this problem, too, will inevitably require a revision of many theoretical concepts, primarily through wider use of the systems approach in medicine.
- At first glance, a systematic approach based on the use of models of general patterns of pathology contradicts the principles of a personalized approach in medicine, which is not true. On the contrary, it is impossible to describe a specific clinical situation and propose a patient-centered treatment protocol without separating the general from the particular. Thus, the use of a personalized approach will, over time, increasingly often ‘stumble’ over the unresolved general problems of pathology.
- The ever-increasing specialization of clinicians makes it difficult for them to assess the patient’s body as a holistic system. Often, there is a lack of cooperation among the various practitioners providing care for a particular patient at the same time.
- The prognostic, diagnostic, and outcome monitoring criteria used in clinical protocols already lag behind the capabilities of modern technology, including data from molecular research, and instrumental and information technologies. The idea that this problem can only be solved by using mathematical methods, ignoring heuristic approaches to the modeling of complex systems is, in our opinion, erroneous for many reasons. The widespread use of the terms: ‘inflammation’, ‘neuroinflammation’, ‘systemic inflammation’, ‘systemic inflammatory response’, etc. without their necessary characterization has essentially turned these terms into ‘vague’ concepts that require specification in each study.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AGE | advanced glycation end-products |
| ALRs | absent in melanoma 2-like receptors |
| AMPK | 5’AMP-activated protein kinase |
| ATF | activating transcription factor |
| ATP | adenosine triphosphate |
| ChLGI | chronic low-grade inflammation |
| ChSLGI | chronic systemic low-grade inflammation |
| cNOS | constitutive NO synthase |
| COX | cyclooxygenase |
| CRP | C-reactive protein |
| CS | cellular stress |
| CTL | cytotoxic T lymphocytes |
| DAMPs | damage-associated molecular patterns |
| DDR | DNA-damage response |
| DNA | deoxyribonucleic acid |
| ER | endoplasmic reticulum |
| ERAD | ER-associated degradation complex |
| ERK | extracellular signal-regulated kinase |
| EVC | exudative vascular complex |
| FFA | free fatty acids |
| GLUT-4 | glucose transporter type 4 |
| HIF | hypoxia-inducible factor |
| HLA | human leukocyte antigens |
| HSF | heat shock factor |
| HSP | heat shock protein |
| IAP | inhibitor of apoptosis proteins |
| IFN | interferon |
| IL | interleukin |
| ILC | innate lymphoid cells |
| iNOS | inducible NO synthase |
| IRE | inositol-requiring enzyme |
| JAK | Janus kinase |
| JNK | c-Jun N-terminal kinase |
| M | macrophage |
| MAPK | of mitogen-activated protein kinase |
| miRNA | microRNA |
| MS | multiple sclerosis |
| mtDNA | mitochondrial DNA |
| mTORC1 | mammalian target of rapamycin complex 1 |
| NES | neuroendocrine system |
| NF-κB | nuclear factor-κB |
| NK | natural killer cells |
| NLRP3/NLRP6 | NLR family pyrin domain containing 3/NLR family pyrin domain containing 6 |
| NLR | NOD-like receptor |
| NRF | nuclear respiratory factor |
| oxLDL | oxidized low-density lipoproteins |
| PAMP | pathogen-associated molecular pattern |
| PERK | protein kinase RNA-like endoplasmic reticulum kinase |
| PI3K | phosphoinositide 3-kinases |
| PRR | pattern-recognizing receptor |
| RBP | RNA-binding protein |
| RIPK | receptor-interacting protein kinase |
| RNA | ribonucleic acid |
| ROS | reactive oxygen species |
| SASP | senescence-associated secretory phenotype |
| SI | systemic inflammation |
| SIR | systemic inflammatory response |
| SR | scavenger receptor |
| STAT | signal transducer and activator of transcription |
| TAMs | tumor-associated macrophages |
| TCR | T-cell receptor |
| TF | transcription factor |
| TGF-β | transforming growth factor beta |
| Th | T helper cells |
| TLR | Toll-like receptor |
| TNF | tumor necrosis factor |
| Tr1 | Type 1 regulatory T cells (CD4+) |
| Treg | CD4+ regulatory T cells |
| TS | tissue stress |
| UPRER | endoplasmic reticulum unfolded protein response |
| UPRmt | mitochondrial unfolded protein response |
References
- Ransom, W.H. The Inflammation Idea in General Pathology; Williams&Norgate: London, UK, 1905; p. 354. [Google Scholar]
- Bennett, J.M.; Reeves, G.; Billman, G.E.; Sturmberg, J.P. Inflammation-nature’s way to efficiently respond to all types of challenges: Implications for understanding and managing "the epidemic" of chronic diseases. Front. Med. (Lausanne) 2018, 5, 316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granger, D.N.; Senchenkova, E. Inflammation and the Microcirculation; Morgan & Claypool Life Sciences: San Rafael, CA, USA, 2010. [Google Scholar]
- Gusev, E.Y.; Zotova, N.V. Cellular stress and general pathological processes. Curr. Pharm. Des. 2019, 25, 251–297. [Google Scholar] [CrossRef] [PubMed]
- Plytycz, B.; Seljelid, R. From inflammation to sickness: Historical perspective. Arch. Immunol. Ther. Exp. (Warsz) 2003, 51, 105–109. [Google Scholar] [PubMed]
- Bone, R.C.; Balk, R.A.; Cerra, F.B.; Dellinger, R.P.; Fein, A.M.; Knaus, W.A.; Schein, R.M.; Sibbald, W.J. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 1992, 101, 1644–1655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- van Greevenbroek, M.M.J.; Schalkwijk, C.G.; Stehouwer, C.D. Dysfunctional adipose tissue and low-grade inflammation in the management of the metabolic syndrome: Current practices and future advances. F1000Research 2016, 5, 2515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zafar, U.; Khaliq, S.; Ahmad, H.U.; Manzoor, S.; Lone, K.P. Metabolic syndrome: An update on diagnostic criteria, pathogenesis, and genetic links. Hormones (Athens) 2018, 17, 299–313. [Google Scholar] [CrossRef]
- Muralidharan, S.; Mandrekar, P. Cellular stress response and innate immune signaling: Integrating pathways in host defense and inflammation. J. Leukoc. Biol. 2013, 94, 1167–1184. [Google Scholar] [CrossRef] [Green Version]
- Kültz, D. Evolution of the cellular stress proteome: From monophyletic origin to ubiquitous function. J. Exp. Biol. 2003, 206, 3119–3124. [Google Scholar] [CrossRef] [Green Version]
- Farrell, G.C.; Haczeyni, F.; Chitturi, S. Pathogenesis of NASH: How metabolic complications of overnutrition favor lipotoxicity and pro-inflammatory fatty liver disease. Adv. Exp. Med. Biol. 2018, 1061, 19–44. [Google Scholar] [CrossRef]
- Ježek, P.; Jaburek, M.; Holendová, B.; Plecitá-Hlavatá, L. Fatty acid-stimulated insulin secretion vs. lipotoxicity. Molecules 2018, 23, 1483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langlois, A.; Forterre, A.; Pinget, M.; Bouzakri, K. Impact of Moderate Exercise on Fatty Acid Oxidation in Pancreatic β-Cells and Skeletal Muscle. J. Endocrinol. Investig. 2021, 44, 1815–1825. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wu, M. Pattern recognition receptors in health and diseases. Signal Transduct. Target. Ther. 2021, 6, 291. [Google Scholar] [CrossRef] [PubMed]
- Wicherska-Pawłowska, K.; Wróbel, T.; Rybka, J. Toll-Like Receptors (TLRs), NOD-Like Receptors (NLRs), and RIG-I-Like Receptors (RLRs) in innate immunity. TLRs, NLRs, and RLRs ligands as immunotherapeutic agents for hematopoietic diseases. Int. J. Mol. Sci. 2021, 22, 13397. [Google Scholar] [CrossRef]
- Granzotto, A.; Weiss, J.H.; Sensi, S.L. Editorial: Excitotoxicity turns 50. The death that never dies. Front. Neurosci. 2022, 15, 831809. [Google Scholar] [CrossRef]
- Verma, M.; Lizama, B.N.; Chu, C.T. Excitotoxicity, calcium and mitochondria: A triad in synaptic neurodegeneration. Transl. Neurodegener. 2022, 11, 3. [Google Scholar] [CrossRef]
- Segura-Aguilar, J.; Mannervik, B.; Inzunza, J.; Varshney, M.; Nalvarte, I.; Muñoz, P. Astrocytes protect dopaminergic neurons against aminochrome neurotoxicity. Neural. Regen. Res. 2022, 17, 1861–1866. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; June, C.H. Cytokine storm. N. Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef]
- Gusev, E.; Sarapultsev, A.; Hu, D.; Chereshnev, V. Problems of pathogenesis and pathogenetic therapy of COVID-19 from the perspective of the general theory of pathological systems (general pathological processes). Int. J. Mol. Sci. 2021, 22, 7582. [Google Scholar] [CrossRef]
- Gusev, E.; Sarapultsev, A.; Solomatina, L.; Chereshnev, V. SARS-CoV-2-Specific immune response and the pathogenesis of COVID-19. Int. J. Mol. Sci. 2022, 23, 1716. [Google Scholar] [CrossRef]
- Checa, J.; Aran, J.M. Reactive oxygen species: Drivers of physiological and pathological processes. J. Inflamm. Res. 2020, 13, 1057–1073. [Google Scholar] [CrossRef] [PubMed]
- Sazonova, M.A.; Sinyov, V.V.; Ryzhkova, A.I.; Sazonova, M.D.; Kirichenko, T.V.; Khotina, V.A.; Khasanova, Z.B.; Doroschuk, N.A.; Karagodin, V.P.; Orekhov, A.N.; et al. Some molecular and cellular stress mechanisms associated with neurodegenerative diseases and atherosclerosis. Int. J. Mol. Sci. 2021, 22, 699. [Google Scholar] [CrossRef] [PubMed]
- Chovatiya, R.; Medzhitov, R. Stress, inflammation, and defense of homeostasis. Mol. Cell. 2014, 54, 281–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gusev, E.Y.; Zotova, N.V.; Zhuravleva, Y.A.; Chereshnev, V.A. Physiological and Pathogenic Role of Scavenger Receptors in Humans. Med. Immunol. (Russia) 2020, 22, 7–48. [Google Scholar] [CrossRef]
- Komai, K.; Shichita, T.; Ito, M.; Kanamori, M.; Chikuma, S.; Yoshimura, A. Role of scavenger receptors as damage-associated molecular pattern receptors in Toll-like receptor activation. Int. Immunol. 2017, 29, 59–70. [Google Scholar] [CrossRef]
- Gulati, A.; Kaur, D.; Prasad, G.V.R.K.; Mukhopadhaya, A. PRR function of innate immune receptors in recognition of bacteria or bacterial ligands. Adv. Exp. Med. Biol. 2018, 1112, 255–280. [Google Scholar] [CrossRef]
- Czerkies, M.; Borzęcka, K.; Zdioruk, M.I.; Płóciennikowska, A.; Sobota, A.; Kwiatkowska, K. An interplay between scavenger receptor A and CD14 during activation of J774 cells by high concentrations of LPS. Immunobiology 2013, 218, 1217–1226. [Google Scholar] [CrossRef]
- Triantafilou, M.; Gamper, F.G.; Haston, R.M.; Mouratis, M.A.; Morath, S.; Hartung, T.; Triantafilou, K. Membrane sorting of Toll-Like Receptor (TLR)-2/6 and TLR2/1 heterodimers at the cell surface determines heterotypic associations with CD36 and intracellular targeting. J. Biol. Chem. 2006, 281, 31002–31011. [Google Scholar] [CrossRef] [Green Version]
- Fulda, S.; Gorman, A.M.; Hori, O.; Samali, A. Cellular stress responses: Cell survival and cell death. Int. J. Cell Biol. 2010, 2010, 214074. [Google Scholar] [CrossRef] [Green Version]
- Galluzzi, L.; Yamazaki, T.; Kroemer, G. Linking cellular stress responses to systemic homeostasis. Nat. Rev. Mol. Cell Biol. 2018, 19, 731–745. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Leli, N.M.; Koumenis, C.; Amaravadi, R.K. Regulation of autophagy by canonical and non-canonical ER stress responses. Semin. Cancer Biol. 2020, 66, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Hanawalt, P.; Sweasy, J. Mechanistic understanding of cellular responses to genomic stress. Environ. Mol. Mutagen. 2020, 61, 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshida, K.; Fujita, M. DNA damage responses that enhance resilience to replication stress. Cell. Mol. Life Sci. 2021, 78, 6763–6773. [Google Scholar] [CrossRef] [PubMed]
- Morshed, S.A.; Davies, T.F. Understanding thyroid cell stress. J. Clin. Endocrinol. Metab. 2020, 105, e66–e69. [Google Scholar] [CrossRef]
- Jelic, M.D.; Mandic, A.D.; Maricic, S.M.; Srdjenovic, B.U. Oxidative stress and its role in cancer. J. Cancer Res. Ther. 2021, 17, 22–28. [Google Scholar] [CrossRef]
- Wigner, P.; Grębowski, R.; Bijak, M.; Saluk-Bijak, J.; Szemraj, J. The interplay between oxidative stress, inflammation and angiogenesis in bladder cancer development. Int. J. Mol. Sci. 2021, 22, 4483. [Google Scholar] [CrossRef]
- Vargas-Mendoza, N.; Angeles-Valencia, M.; Morales-González, Á.; Madrigal-Santillán, E.O.; Morales-Martínez, M.; Madrigal-Bujaidar, E.; Álvarez-González, I.; Gutiérrez-Salinas, J.; Esquivel-Chirino, C.; Chamorro-Cevallos, G.; et al. oxidative stress, mitochondrial function and adaptation to exercise: New perspectives in nutrition. Life 2021, 11, 1269. [Google Scholar] [CrossRef]
- Konovalova, J.; Gerasymchuk, D.; Parkkinen, I.; Chmielarz, P.; Domanskyi, A. Interplay between MicroRNAs and oxidative stress in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 6055. [Google Scholar] [CrossRef] [Green Version]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative stress in cancer. Cancer Cell 2020, 38, 167–197. [Google Scholar] [CrossRef]
- Sławińska, N.; Krupa, R. Molecular aspects of senescence and organismal ageing-DNA damage response, telomeres, inflammation and chromatin. Int. J. Mol. Sci. 2021, 22, 590. [Google Scholar] [CrossRef] [PubMed]
- Priesnitz, C.; Becker, T. Pathways to balance mitochondrial translation and protein import. Genes Dev. 2018, 32, 1285–1296. [Google Scholar] [CrossRef] [PubMed]
- Eckl, E.-M.; Ziegemann, O.; Krumwiede, L.; Fessler, E.; Jae, L.T. Sensing, signaling and surviving mitochondrial stress. Cell. Mol. Life Sci. 2021, 78, 5925–5951. [Google Scholar] [CrossRef] [PubMed]
- Quirós, P.M.; Prado, M.A.; Zamboni, N.; D’Amico, D.; Williams, R.W.; Finley, D.; Gygi, S.P.; Auwerx, J. Multi-omics analysis identifies ATF4 as a key regulator of the mitochondrial stress response in mammals. J. Cell Biol. 2017, 216, 2027–2045. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Miao, W.; Ma, J.; Xv, Z.; Bo, H.; Li, J.; Zhang, Y.; Ji, L.L. Acute exercise-induced mitochondrial stress triggers an inflammatory response in the myocardium via NLRP3 inflammasome activation with mitophagy. Oxid. Med. Cell. Longev. 2016, 2016, 1987149. [Google Scholar] [CrossRef] [Green Version]
- Schröder, M. Endoplasmic reticulum stress responses. Cell. Mol. Life Sci. 2008, 65, 862–894. [Google Scholar] [CrossRef]
- Walter, P.; Ron, D. The unfolded protein response: From stress pathway to homeostatic regulation. Science 2011, 334, 1081–1086. [Google Scholar] [CrossRef] [Green Version]
- Garbuz, D.G. Regulation of heat shock gene expression in response to stress. Mol. Biol. (Mosk) 2017, 51, 400–417. [Google Scholar] [CrossRef]
- Morimoto, R.I. The heat shock response: Systems biology of proteotoxic stress in aging and disease. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 91–99. [Google Scholar] [CrossRef] [Green Version]
- Lang, B.J.; Guerrero, M.E.; Prince, T.L.; Okusha, Y.; Bonorino, C.; Calderwood, S.K. The functions and regulation of heat shock proteins; key orchestrators of proteostasis and the heat shock response. Arch. Toxicol. 2021, 95, 1943–1970. [Google Scholar] [CrossRef]
- Ravanan, P.; Srikumar, I.F.; Talwar, P. Autophagy: The spotlight for cellular stress responses. Life Sci. 2017, 188, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yamashita, T.; Shang, J.; Shi, X.; Morihara, R.; Huang, Y.; Sato, K.; Takemoto, M.; Hishikawa, N.; Ohta, Y.; et al. Molecular switching from ubiquitin–proteasome to autophagy pathways in mice stroke model. J. Cereb. Blood Flow Metab. 2020, 40, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, G.; Schwarz, T.L. The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ. 2013, 20, 31–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Klionsky, D.J. The regulation of autophagy—Unanswered questions. J. Cell Sci. 2011, 124, 161–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitada, M.; Koya, D. Autophagy in metabolic disease and ageing. Nat. Rev. Endocrinol. 2021, 17, 647–661. [Google Scholar] [CrossRef]
- Eshraghi, M.; Adlimoghaddam, A.; Mahmoodzadeh, A.; Sharifzad, F.; Yasavoli-Sharahi, H.; Lorzadeh, S.; Albensi, B.C.; Ghavami, S. Alzheimer’s disease pathogenesis: Role of autophagy and mitophagy focusing in microglia. Int. J. Mol. Sci. 2021, 22, 3330. [Google Scholar] [CrossRef]
- Man, S.M.; Karki, R.; Kanneganti, T.-D. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol. Rev. 2017, 277, 61–75. [Google Scholar] [CrossRef] [Green Version]
- Holley, C.L.; Schroder, K. The rOX-stars of inflammation: Links between the inflammasome and mitochondrial meltdown. Clin. Transl. Immunol. 2020, 9, e01109. [Google Scholar] [CrossRef] [Green Version]
- Lee, G.-S.; Subramanian, N.; Kim, A.I.; Aksentijevich, I.; Goldbach-Mansky, R.; Sacks, D.B.; Germain, R.N.; Kastner, D.L.; Chae, J.J. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature 2012, 492, 123–127. [Google Scholar] [CrossRef] [Green Version]
- Savage, C.D.; Lopez-Castejon, G.; Denes, A.; Brough, D. NLRP3-inflammasome activating DAMPs stimulate an inflammatory response in glia in the absence of priming which contributes to brain inflammation after injury. Front. Immunol. 2012, 3, 288. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Hauenstein, A.V. The NLRP3 inflammasome: Mechanism of action, role in disease and therapies. Mol. Asp. Med. 2020, 76, 100889. [Google Scholar] [CrossRef]
- Masumoto, J.; Zhou, W.; Morikawa, S.; Hosokawa, S.; Taguchi, H.; Yamamoto, T.; Kurata, M.; Kaneko, N. Molecular biology of autoinflammatory diseases. Inflamm. Regen. 2021, 41, 33. [Google Scholar] [CrossRef] [PubMed]
- de Sousa, M.C.; Gjorgjieva, M.; Dolicka, D.; Sobolewski, C.; Foti, M. Deciphering miRNAs’ action through miRNA editing. Int. J. Mol. Sci. 2019, 20, 6249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Xu, W.; Feng, Y.; Zhou, X. Non-coding RNA involvement in the pathogenesis of diabetic cardiomyopathy. J. Cell. Mol. Med. 2019, 23, 5859–5867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrell, C.R.; Jovicic, N.; Djonov, V.; Arsenijevic, N.; Volarevic, V. Mesenchymal stem cell-derived exosomes and other extracellular vesicles as new remedies in the therapy of inflammatory diseases. Cells 2019, 8, 1605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Backlund, M.; Stein, F.; Rettel, M.; Schwarzl, T.; Perez-Perri, J.I.; Brosig, A.; Zhou, Y.; Neu-Yilik, G.; Hentze, M.W.; Kulozik, A.E. Plasticity of nuclear and cytoplasmic stress responses of RNA-binding proteins. Nucleic Acids Res. 2020, 48, 4725–4740. [Google Scholar] [CrossRef]
- Sidibé, H.; Vande Velde, C. Collective learnings of studies of stress granule assembly and composition. Methods Mol. Biol. 2022, 2428, 199–228. [Google Scholar] [CrossRef]
- Lau, Y.; Oamen, H.P.; Caudron, F. Protein phase separation during stress adaptation and cellular memory. Cells 2020, 9, 1302. [Google Scholar] [CrossRef]
- Bardwell, L.; Zou, X.; Nie, Q.; Komarova, N.L. Mathematical models of specificity in cell signaling. Biophys. J. 2007, 92, 3425–3441. [Google Scholar] [CrossRef] [Green Version]
- Pan, Z.; Ma, G.; Kong, L.; Du, G. Hypoxia-inducible factor-1: Regulatory mechanisms and drug development in stroke. Pharmacol. Res. 2021, 170, 105742. [Google Scholar] [CrossRef]
- Zhang, B.; Fan, Y.; Cao, P.; Tan, K. Multifaceted roles of HSF1 in cell death: A state-of-the-art review. Biochim. Biophys. Acta Rev. Cancer 2021, 1876, 188591. [Google Scholar] [CrossRef] [PubMed]
- Robledinos-Antón, N.; Fernández-Ginés, R.; Manda, G.; Cuadrado, A. Activators and inhibitors of NRF2: A review of their potential for clinical development. Oxid. Med. Cell. Longev. 2019, 2019, 9372182. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.; Jung, M.; Kim, C.; Kang, H.; Han, S.; Cha, S.; Jeong, S.M.; Lee, E.K. Loss of RNA binding protein HuD facilitates the production of the senescence-associated secretory phenotype. Cell Death Dis. 2022, 13, 329. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Deng, Z.; Barkema, H.W.; Xu, M.; Gao, J.; Liu, G.; Lin, Y.; Kastelic, J.P.; Han, B. Nrf2 and NF-κB/NLRP3 inflammasome pathways are involved in Prototheca bovis infections of mouse mammary gland tissue and mammary epithelial cells. Free Radic. Biol. Med. 2022, 184, 148–157. [Google Scholar] [CrossRef]
- Henderson, B.; Kaiser, F. Do reciprocal interactions between cell stress proteins and cytokines create a new intra-/extra-cellular signaling nexus? Cell. Stress Chaperones 2013, 18, 685–701. [Google Scholar] [CrossRef] [Green Version]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Yu, S.; Gu, Y.; Wang, T.; Mu, L.; Wang, H.; Yan, S.; Wang, A.; Wang, J.; Liu, L.; Shen, H.; et al. Study of neuronal apoptosis ceRNA network in hippocampal sclerosis of human temporal lobe epilepsy by RNA-Seq. Front. Neurosci. 2021, 15, 770627. [Google Scholar] [CrossRef]
- Dupont-Versteegden, E.E. Apoptosis in skeletal muscle and its relevance to atrophy. World J. Gastroenterol. 2006, 12, 7463–7466. [Google Scholar] [CrossRef]
- Arya, R.; White, K. Cell death in development: Signaling pathways and core mechanisms. Semin. Cell. Dev. Biol. 2015, 39, 12–19. [Google Scholar] [CrossRef] [Green Version]
- Rosa, N.; Ivanova, H.; Wagner, L.E.; Kale, J.; Rovere, R.L.; Welkenhuyzen, K.; Louros, N.; Karamanou, S.; Shabardina, V.; Lemmens, I.; et al. Bcl-xL acts as an inhibitor of IP3R channels, thereby antagonizing Ca2+-driven apoptosis. Cell Death Differ. 2021, 29, 788–805. [Google Scholar] [CrossRef]
- Bahatyrevich-Kharitonik, B.; Medina-Guzman, R.; Flores-Cortes, A.; García-Cruzado, M.; Kavanagh, E.; Burguillos, M.A. Cell death related proteins beyond apoptosis in the CNS. Front. Cell Dev. Biol. 2022, 9, 825747. [Google Scholar] [CrossRef] [PubMed]
- Bíliková, P.; Švandová, E.; Veselá, B.; Doubek, J.; Poliard, A.; Matalová, E. Coupling activation of pro-apoptotic caspases with autophagy in the Meckel´s cartilage. Physiol. Res. 2019, 68, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, M.; Derry, W.B. Your neighbours matter—Non-autonomous control of apoptosis in development and disease. Cell Death Differ. 2016, 23, 1110–1118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, E.H.; Wong, S.-W.; Martinez, J. Programmed necrosis and disease: We interrupt your regular programming to bring you necroinflammation. Cell Death Differ. 2019, 26, 25–40. [Google Scholar] [CrossRef] [Green Version]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef]
- Fricker, M.; Tolkovsky, A.M.; Borutaite, V.; Coleman, M.; Brown, G.C. Neuronal cell death. Physiol. Rev. 2018, 98, 813–880. [Google Scholar] [CrossRef]
- Miao, E.A.; Rajan, J.V.; Aderem, A. Caspase-1-induced pyroptotic cell death. Immunol. Rev. 2011, 243, 206–214. [Google Scholar] [CrossRef]
- Echevarría, L.U.; Leimgruber, C.; González, J.G.; Nevado, A.; Álvarez, R.; García, L.N.; Quintar, A.A.; Maldonado, C.A. Evidence of eosinophil extracellular trap cell death in COPD: Does it represent the trigger that switches on the disease? Int. J. Chronic Obstr. Pulm. Dis. 2017, 12, 885–896. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.; Reilly, B.; Tan, C.; Wang, P.; Aziz, M. Extracellular CIRP induces macrophage extracellular trap formation via gasdermin D activation. Front. Immunol. 2021, 12, 780210. [Google Scholar] [CrossRef]
- Nija, R.J.; Sanju, S.; Sidharthan, N.; Mony, U. Extracellular trap by blood cells: Clinical implications. Tissue Eng. Regen. Med. 2020, 17, 141–153. [Google Scholar] [CrossRef]
- Jung, S.; Choe, S.; Woo, H.; Jeong, H.; An, H.-K.; Moon, H.; Ryu, H.Y.; Yeo, B.K.; Lee, Y.W.; Choi, H.; et al. Autophagic death of neural stem cells mediates chronic stress-induced decline of adult hippocampal neurogenesis and cognitive deficits. Autophagy 2020, 16, 512–530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.; Lv, X.; Hu, B.; Shao, Z.; Wang, B.; Ma, K.; Lin, H.; Cui, M. RIPK1/RIPK3/MLKL-mediated necroptosis contributes to compression-induced rat nucleus pulposus cells death. Apoptosis 2017, 22, 626–638. [Google Scholar] [CrossRef] [PubMed]
- De Schutter, E.; Ramon, J.; Pfeuty, B.; De Tender, C.; Stremersch, S.; Remdonck, K.; de Beeck, K.O.; Declercq, W.; Riquet, F.B.; Braeckmans, K.; et al. Plasma membrane perforation by GSDME during apoptosis-driven secondary necrosis. Cell. Mol. Life Sci. 2021, 79, 19. [Google Scholar] [CrossRef] [PubMed]
- Cassandras, M.; Wang, C.; Kathiriya, J.; Tsukui, T.; Matatia, P.; Matthay, M.; Wolters, P.; Molofsky, A.; Sheppard, D.; Chapman, H.; et al. Gli1+ mesenchymal stromal cells form a pathological niche to promote airway progenitor metaplasia in the fibrotic lung. Nat. Cell Biol. 2020, 22, 1295–1306. [Google Scholar] [CrossRef]
- Scutiero, G.; Iannone, P.; Bernardi, G.; Bonaccorsi, G.; Spadaro, S.; Volta, C.A.; Greco, P.; Nappi, L. Oxidative stress and endometriosis: A systematic review of the literature. Oxid. Med. Cell. Longev. 2017, 2017, 7265238. [Google Scholar] [CrossRef]
- Komro, J.; Gonzales, J.; Marberry, K.; Main, D.C.; Cramberg, M.; Kondrashov, P. Fibrocartilaginous metaplasia and neovascularization of the anterior cruciate ligament in patients with osteoarthritis. Clin. Anat. 2020, 33, 899–905. [Google Scholar] [CrossRef]
- Park, Y.H.; Kim, N. Review of atrophic gastritis and intestinal metaplasia as a premalignant lesion of gastric cancer. J. Cancer Prev. 2015, 20, 25–40. [Google Scholar] [CrossRef]
- Sikora, E.; Bielak-Zmijewska, A.; Mosieniak, G. A common signature of cellular senescence; does it exist? Ageing Res. Rev. 2021, 71, 101458. [Google Scholar] [CrossRef]
- Uyar, B.; Palmer, D.; Kowald, A.; Escobar, H.M.; Barrantes, I.; Möller, S.; Akalin, A.; Fuellen, G. Single-cell analyses of aging, inflammation and senescence. Ageing Res. Rev. 2020, 64, 101156. [Google Scholar] [CrossRef]
- Rudolph, K.L. Stem cell aging. Mech. Ageing Dev. 2021, 193, 111394. [Google Scholar] [CrossRef]
- Roger, L.; Tomas, F.; Gire, V. Mechanisms and regulation of cellular senescence. Int. J. Mol. Sci. 2021, 22, 13173. [Google Scholar] [CrossRef] [PubMed]
- Sikora, E.; Bielak-Żmijewska, A.; Mosieniak, G. What is and what is not cell senescence. Postepy Biochem. 2018, 64, 110–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erusalimsky, J.D. Oxidative stress, telomeres and cellular senescence: What non-drug interventions might break the link? Free Radic. Biol. Med. 2020, 150, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Malaquin, N.; Martinez, A.; Rodier, F. Keeping the senescence secretome under control: Molecular reins on the senescence-associated secretory phenotype. Exp. Gerontol. 2016, 82, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Kandhaya-Pillai, R.; Miro-Mur, F.A.; Alijotas-Reig, J.; Tchkonia, T.; Kirkland, J.L.; Schwartz, S. TNFα-senescence initiates a STAT-dependent positive feedback loop, leading to a sustained interferon signature, DNA damage, and cytokine secretion. Aging (Albany N. Y.) 2017, 9, 2411–2435. [Google Scholar] [CrossRef] [Green Version]
- Su, K.-H.; Cao, J.; Tang, Z.; Dai, S.; He, Y.; Sampson, S.B.; Benjamin, I.J.; Dai, C. HSF1 critically attunes proteotoxic stress sensing by mTORC1 to combat stress and promote growth. Nat. Cell Biol. 2016, 18, 527–539. [Google Scholar] [CrossRef]
- Rathinaswamy, M.K.; Burke, J.E. Class I phosphoinositide 3-kinase (PI3K) regulatory subunits and their roles in signaling and disease. Adv. Biol. Regul. 2020, 75, 100657. [Google Scholar] [CrossRef]
- Tian, T.; Li, X.; Zhang, J. mTOR signaling in cancer and mTOR inhibitors in solid tumor targeting therapy. Int. J. Mol. Sci. 2019, 20, 755. [Google Scholar] [CrossRef] [Green Version]
- Valdés, F.Z.; Luna, V.Z.; Arévalo, B.R.; Brown, N.V.; Gutiérrez, M.C. Adenosine: Synthetic methods of its derivatives and antitumor activity. Mini Rev. Med. Chem. 2018, 18, 1684–1701. [Google Scholar] [CrossRef]
- Borea, P.A.; Gessi, S.; Merighi, S.; Vincenzi, F.; Varani, K. Pharmacology of adenosine receptors: The state of the art. Physiol. Rev. 2018, 98, 1591–1625. [Google Scholar] [CrossRef]
- Sakamoto, K.; Holman, G.D. Emerging role for AS160/TBC1D4 and TBC1D1 in the regulation of GLUT4 traffic. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E29–E37. [Google Scholar] [CrossRef] [PubMed]
- Haider, N.; Lebastchi, J.; Jayavelu, A.K.; Batista, T.M.; Pan, H.; Dreyfuss, J.M.; Carcamo-Orive, I.; Knowles, J.W.; Mann, M.; Kahn, C.R. Signaling defects associated with insulin resistance in nondiabetic and diabetic individuals and modification by sex. J. Clin. Investig. 2021, 131, e151818. [Google Scholar] [CrossRef] [PubMed]
- Mihaylova, M.M.; Shaw, R.J. The AMPK signaling pathway coordinates cell growth, autophagy and metabolism. Nat. Cell Biol. 2011, 13, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Farkhondeh, T.; Mehrpour, O.; Buhrmann, C.; Pourbagher-Shahri, A.M.; Shakibaei, M.; Samarghandian, S. Organophosphorus compounds and MAPK signaling pathways. Int. J. Mol. Sci. 2020, 21, 4258. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Tsuchiya, R.; Hozumi, Y.; Nakano, T.; Okada, M.; Goto, K. Reciprocal regulation of p53 and NF-κB by diacylglycerol kinase &zeta. Adv. Biol. Regul. 2016, 60, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Chittiboyina, S.; Bai, Y.; Lelièvre, S.A. Microenvironment-cell nucleus relationship in the context of oxidative stress. Front. Cell Dev. Biol. 2018, 6, 23. [Google Scholar] [CrossRef]
- Bautista, L.E. Inflammation, endothelial dysfunction, and the risk of high blood pressure: Epidemiologic and biological evidence. J. Hum. Hypertens. 2003, 17, 223–230. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Zhou, Y.; Fan, Y.; Gong, Y.; Yang, J.; Yang, R.; Li, L.; Zou, L.; Xu, X.; Li, G.; et al. Involvement of purinergic 2X 4 receptor in glycoprotein 120-induced pyroptosis in dorsal root ganglia. J. Neurochem. 2019, 151, 584–594. [Google Scholar] [CrossRef]
- Maik-Rachline, G.; Wortzel, I.; Seger, R. Alternative splicing of MAPKs in the regulation of signaling specificity. Cells 2021, 10, 3466. [Google Scholar] [CrossRef]
- Jiang, M.; Wang, H.; Liu, Z.; Lin, L.; Wang, L.; Xie, M.; Li, D.; Zhang, J.; Zhang, R. Endoplasmic reticulum stress-dependent activation of iNOS/NO-NF-κB signaling and NLRP3 inflammasome contributes to endothelial inflammation and apoptosis associated with microgravity. FASEB J. 2020, 34, 10835–10849. [Google Scholar] [CrossRef]
- Gusev, E.; Solomatina, L.; Zhuravleva, Y.; Sarapultsev, A. The pathogenesis of end-stage renal disease from the standpoint of the theory of general pathological processes of inflammation. Int. J. Mol. Sci. 2021, 22, 11453. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Letai, A.; Sarosiek, K. Regulation of apoptosis in health and disease: The balancing act of BCL-2 family proteins. Nat. Rev. Mol. Cell Biol. 2019, 20, 175–193. [Google Scholar] [CrossRef] [PubMed]
- Nyasavajjala, S.M.; Phillips, B.E.; Lund, J.N.; Williams, J.P. Creatinine and myoglobin are poor predictors of anaerobic threshold in colorectal cancer and health. J. Cachexia Sarcopenia Muscle 2015, 6, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Yeung, Y.T.; Aziz, F.; Guerrero-Castilla, A.; Arguelles, S. Signaling pathways in inflammation and anti-inflammatory therapies. Curr. Pharm. Des. 2018, 24, 1449–1484. [Google Scholar] [CrossRef] [PubMed]
- Elchaninov, A.V.; Fatkhudinov, T.K.; Vishnyakova, P.A.; Lokhonina, A.V.; Sukhikh, G.T. Phenotypical and functional polymorphism of liver resident macrophages. Cells 2019, 8, 1032. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Nardo, G.; Gilardi, G. Natural compounds as pharmaceuticals: The key role of cytochromes P450 reactivity. Trends Biochem. Sci. 2020, 45, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Xia, Q.; Huang, X.; Huang, J.; Zheng, Y.; March, M.E.; Li, J.; Wei, Y. The role of autophagy in skeletal muscle diseases. Front. Physiol. 2021, 12, 638983. [Google Scholar] [CrossRef]
- Krüger, K.; Reichel, T.; Zeilinger, C. Role of heat shock proteins 70/90 in exercise physiology and exercise immunology and their diagnostic potential in sports. J. Appl. Physiol. (1985) 2019, 126, 916–927. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [Green Version]
- Dubuisson, N.; Versele, R.; de Carrizosa, M.A.D.-L.; Selvais, C.M.; Brichard, S.M.; Abou-Samra, M. Walking down skeletal muscle lane: From inflammasome to disease. Cells 2021, 10, 3023. [Google Scholar] [CrossRef]
- Wang, J.; Dong, R.; Zheng, S. Roles of the inflammasome in the gut-liver axis (Review). Mol. Med. Rep. 2019, 19, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Scieglinska, D.; Krawczyk, Z.; Sojka, D.R.; Gogler-Pigłowska, A. Heat shock proteins in the physiology and pathophysiology of epidermal keratinocytes. Cell Stress Chaperones 2019, 24, 1027–1044. [Google Scholar] [CrossRef] [Green Version]
- Angel, P.; Szabowski, A.; Schorpp-Kistner, M. Function and regulation of AP-1 subunits in skin physiology and pathology. Oncogene 2001, 20, 2413–2423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thapa, P.; Farber, D.L. The role of the thymus in the immune response. Thorac. Surg. Clin. 2019, 29, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Jedlicka, J.; Becker, B.F.; Chappell, D. Endothelial glycocalyx. Crit. Care Clin. 2020, 36, 217–232. [Google Scholar] [CrossRef] [PubMed]
- Poto, R.; Cristinziano, L.; Modestino, L.; de Paulis, A.; Marone, G.; Loffredo, S.; Galdiero, M.R.; Varricchi, G. Neutrophil extracellular traps, angiogenesis and cancer. Biomedicines 2022, 10, 431. [Google Scholar] [CrossRef]
- Manjili, S.H.; Isbell, M.; Ghochaghi, N.; Perkinson, T.; Manjili, M.H. Multifaceted functions of chronic inflammation in regulating tumor dormancy and relapse. Semin. Cancer Biol. 2022, 78, 17–22. [Google Scholar] [CrossRef]
- Cristinziano, L.; Modestino, L.; Antonelli, A.; Marone, G.; Simon, H.-U.; Varricchi, G.; Galdiero, M.R. Neutrophil extracellular traps in cancer. Semin. Cancer Biol. 2022, 79, 91–104. [Google Scholar] [CrossRef]
- Maiorino, L.; Daßler-Plenker, J.; Sun, L.; Egeblad, M. Innate immunity and cancer pathophysiology. Annu. Rev. Pathol. Mech. Dis. 2022, 17, 425–457. [Google Scholar] [CrossRef]
- Gutierrez, M.J.; Lapidus, S.K. Systemic autoinflammatory diseases: A growing family of disorders of overlapping immune dysfunction. Rheum. Dis. Clin. N. Am. 2022, 48, 371–395. [Google Scholar] [CrossRef]
- Chen, M.; Xu, H. Parainflammation, chronic inflammation, and age-related macular degeneration. J. Leukoc. Biol. 2015, 98, 713–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butler, D.C.; Lewin, D.N.; Batalis, N.I. Differential diagnosis of hepatic necrosis encountered at autopsy. Acad. Forensic Pathol. 2018, 8, 256–295. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-Y.; Yeh, M.M. Non-alcoholic fatty liver disease: A review with clinical and pathological correlation. J. Formos. Med. Assoc. 2021, 120, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Yeager, M.P.; Guyre, C.A.; Sites, B.D.; Collins, J.E.; Pioli, P.A.; Guyre, P.M. The stress hormone cortisol enhances interferon-υ-mediated proinflammatory responses of human immune cells. Anesth. Analg. 2018, 127, 556–563. [Google Scholar] [CrossRef]
- Longstreth, G.F.; Iyer, R.L.; Chu, L.-H.; Chen, W.; Yen, L.S.; Hodgkins, P.; Kawatkar, A.A. Acute diverticulitis: Demographic, clinical and laboratory features associated with computed tomography findings in 741 patients. Aliment. Pharmacol. Ther. 2012, 36, 886–894. [Google Scholar] [CrossRef]
- de Jonge, J.; Scheijmans, J.C.G.; van Rossem, C.C.; van Geloven, A.A.W.; Boermeester, M.A.; Bemelman, W.A.; Snapshot Appendicitis Collaborative Study group. Normal inflammatory markers and acute appendicitis: A national multicentre prospective cohort analysis. Int. J. Colorectal Dis. 2021, 36, 1507–1513. [Google Scholar] [CrossRef]
- Polepalle, T.; Moogala, S.; Boggarapu, S.; Pesala, D.S.; Palagi, F.B. Acute phase proteins and their role in periodontitis: A review. J. Clin. Diagn. Res. 2015, 9, ZE01–ZE05. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Cellular and Molecular Immunology, 9th ed.; Elsevier: Philadelphia, PA, USA, 2018; 608p. [Google Scholar]
- Thomer, L.; Schneewind, O.; Missiakas, D. Pathogenesis of Staphylococcus aureus Bloodstream Infections. Annu. Rev. Pathol. Mech. Dis. 2016, 11, 343–364. [Google Scholar] [CrossRef] [Green Version]
- Magán-Fernández, A.; Al-Bakri, S.M.R.; O’Valle, F.; Benavides-Reyes, C.; Abadía-Molina, F.; Mesa, F. neutrophil extracellular traps in periodontitis. Cells 2020, 9, 1494. [Google Scholar] [CrossRef]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage activation and polarization: Nomenclature and experimental guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef] [Green Version]
- Gaus, H.; Miller, C.M.; Seth, P.P.; Harris, E.N. Structural determinants for the interactions of chemically modified nucleic acids with the stabilin-2 clearance receptor. Biochemistry 2018, 57, 2061–2064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luckheeram, R.V.; Zhou, R.; Verma, A.D.; Xia, B. CD4+T cells: Differentiation and functions. Clin. Dev. Immunol. 2012, 2012, 925135. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Souabni, A.; Flavell, R.A.; Wan, Y.Y. An intrinsic mechanism predisposes Foxp3-expressing regulatory T cells to Th2 conversion in vivo. J. Immunol. 2010, 185, 5983–5992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazzoni, A.; Maggi, L.; Liotta, F.; Cosmi, L.; Annunziato, F. Biological and clinical significance of T helper 17 cell plasticity. Immunology 2019, 158, 287–295. [Google Scholar] [CrossRef] [Green Version]
- Hirahara, K.; Nakayama, T. CD4+T-cell subsets in inflammatory diseases: Beyond the Th1/Th2 paradigm. Int. Immunol. 2016, 28, 163–171. [Google Scholar] [CrossRef] [Green Version]
- Shimizu, J.; Takai, K.; Fujiwara, N.; Arimitsu, N.; Ueda, Y.; Wakisaka, S.; Yoshikawa, H.; Kaneko, F.; Suzuki, T.; Suzuki, N. Excessive CD4+ T cells co-expressing interleukin-17 and interferon-γ in patients with behçet’s disease. Clin. Exp. Immunol. 2012, 168, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Bart, V.M.T.; Pickering, R.J.; Taylor, P.R.; Ipseiz, N. Macrophage reprogramming for therapy. Immunology 2021, 163, 128–144. [Google Scholar] [CrossRef]
- Ross, E.A.; Devitt, A.; Johnson, J.R. Macrophages: The good, the bad, and the gluttony. Front. Immunol. 2021, 12, 708186. [Google Scholar] [CrossRef]
- Nakagawa, M.; Karim, M.R.; Izawa, T.; Kuwamura, M.; Yamate, J. Immunophenotypical characterization of M1/M2 macrophages and lymphocytes in cisplatin-induced rat progressive renal fibrosis. Cells 2021, 10, 257. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J. T Helper cell differentiation, heterogeneity, and plasticity. Cold Spring Harb. Perspect. Biol. 2018, 10, a030338. [Google Scholar] [CrossRef] [Green Version]
- Wen, T.-H.; Tsai, K.-W.; Wu, Y.-J.; Liao, M.-T.; Lu, K.-C.; Hu, W.-C. The framework for human host immune responses to four types of parasitic infections and relevant key JAK/STAT signaling. Int. J. Mol. Sci. 2021, 22, 13310. [Google Scholar] [CrossRef] [PubMed]
- Castro, G.; Liu, X.; Ngo, K.; De Leon-Tabaldo, A.; Zhao, S.; Luna-Roman, R.; Yu, J.; Cao, T.; Kuhn, R.; Wilkinson, P.; et al. RORγt and RORα signature genes in human Th17 cells. PLoS ONE 2017, 12, e0181868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujita, H.; Nograles, K.E.; Kikuchi, T.; Gonzalez, J.; Carucci, J.A.; Krueger, J.G. Human Langerhans cells induce distinct IL-22-producing CD4+ T cells lacking IL-17 production. Proc. Natl. Acad. Sci. USA 2009, 106, 21795–21800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paul, W.E.; Zhu, J. How are TH2-type immune responses initiated and amplified? Nat. Rev. Immunol. 2010, 10, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Wu, J.; Ma, M. Implications of macrophage polarization in corneal transplantation rejection. Transpl. Immunol. 2021, 64, 101353. [Google Scholar] [CrossRef]
- Loo, T.T.; Gao, Y.; Lazarevic, V. Transcriptional regulation of CD4+TH cells that mediate tissue inflammation. J. Leukoc. Biol. 2018, 104, 1069–1085. [Google Scholar] [CrossRef]
- Tindemans, I.; Joosse, M.E.; Samsom, J.N. Dissecting the heterogeneity in T-Cell mediated inflammation in IBD. Cells 2020, 9, 110. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Guan, L.; Tang, L.; Liu, S.; Zhou, Y.; Chen, C.; He, Z.; Xu, L. T Helper 9 cells: A new player in immune-related diseases. DNA Cell Biol. 2019, 38, 1040–1047. [Google Scholar] [CrossRef] [Green Version]
- Jin, T.; Mohammad, M.; Pullerits, R.; Ali, A. Bacteria and host interplay in Staphylococcus aureus septic arthritis and sepsis. Pathogens 2021, 10, 158. [Google Scholar] [CrossRef]
- Rood, J.E.; Behrens, E.M. Inherited autoinflammatory syndromes. Annu. Rev. Pathol. 2022, 17, 227–249. [Google Scholar] [CrossRef]
- Doria, A.; Zen, M.; Bettio, S.; Gatto, M.; Bassi, N.; Nalotto, L.; Ghirardello, A.; Iaccarino, L.; Punzi, L. Autoinflammation and autoimmunity: Bridging the divide. Autoimmun. Rev. 2012, 12, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Kant, S.; Kronbichler, A.; Sharma, P.; Geetha, D. Advances in understanding of pathogenesis and treatment of immune-mediated kidney disease: A review. Am. J. Kidney Dis. 2022, 79, 582–600. [Google Scholar] [CrossRef] [PubMed]
- Zucchi, D.; Elefante, E.; Schilirò, D.; Signorini, V.; Trentin, F.; Bortoluzzi, A.; Tani, C. One year in review 2022: Systemic lupus erythematosus. Clin. Exp. Rheumatol. 2022, 40, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Sun, W.; Xu, H. Pathogenesis of concanavalin a induced autoimmune hepatitis in mice. Int. Immunopharmacol. 2022, 102, 108411. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, M.D.; Gratz, I.K.; Paw, J.S.; Abbas, A.K. Treating human autoimmunity: Current practice and future prospects. Sci. Transl. Med. 2012, 4, 125sr1. [Google Scholar] [CrossRef] [Green Version]
- Arleevskaya, M.I.; Manukyan, G.; Inoue, R.; Aminov, R. Editorial: Microbial and environmental factors in autoimmune and inflammatory diseases. Front. Immunol. 2017, 8, 243. [Google Scholar] [CrossRef] [Green Version]
- Costenbader, K.H.; Gay, S.; Alarcón-Riquelme, M.E.; Iaccarino, L.; Doria, A. Genes, epigenetic regulation and environmental factors: Which is the most relevant in developing autoimmune diseases? Autoimmun. Rev. 2012, 11, 604–609. [Google Scholar] [CrossRef]
- Smatti, M.K.; Cyprian, F.S.; Nasrallah, G.K.; Al Thani, A.A.; Almishal, R.O.; Yassine, H.M. Viruses and autoimmunity: A review on the potential interaction and molecular mechanisms. Viruses 2019, 11, 762. [Google Scholar] [CrossRef] [Green Version]
- Chan, L.S.; Vanderlugt, C.J.; Hashimoto, T.; Nishikawa, T.; Zone, J.J.; Black, M.M.; Wojnarowska, F.; Stevens, S.R.; Chen, M.; Fairley, J.A.; et al. Epitope spreading: Lessons from autoimmune skin diseases. J. Investig. Dermatol. 1998, 110, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Tuohy, V.K.; Kinkel, R.P. Epitope spreading: A mechanism for progression of autoimmune disease. Arch. Immunol. Ther. Exp. (Warsz). 2000, 48, 347–351. [Google Scholar]
- Siloşi, I.; Siloşi, C.A.; Boldeanu, M.V.; Cojocaru, M.; Biciuşcă, V.; Avrămescu, C.S.; Cojocaru, I.M.; Bogdan, M.; FolcuŢi, R.M. The role of autoantibodies in health and disease. Rom. J. Morphol. Embryol. 2016, 57, 633–638. [Google Scholar] [PubMed]
- Yazdanpanah, N.; Rezaei, N. Autoimmune complications of COVID-19. J. Med. Virol. 2022, 94, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Song, X.-D.; Wang, Y.-N.; Zhang, A.-L.; Liu, B. Advances in research on the interaction between inflammation and cancer. J. Int. Med. Res. 2020, 48, 300060519895347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fishbein, A.; Hammock, B.D.; Serhan, C.N.; Panigrahy, D. Carcinogenesis: Failure of resolution of inflammation? Pharmacol. Ther. 2021, 218, 107670. [Google Scholar] [CrossRef]
- Vendramin, R.; Litchfield, K.; Swanton, C. Cancer evolution: Darwin and beyond. EMBO J. 2021, 40, e108389. [Google Scholar] [CrossRef]
- Brabletz, S.; Schuhwerk, H.; Brabletz, T.; Stemmler, M.P. Dynamic EMT: A multi-tool for tumor progression. EMBO J. 2021, 40, e108647. [Google Scholar] [CrossRef]
- Hinshaw, D.C.; Shevde, L.A. The tumor microenvironment innately modulates cancer progression. Cancer Res. 2019, 79, 4557–4566. [Google Scholar] [CrossRef] [Green Version]
- Mantovani, A.; Ponzetta, A.; Inforzato, A.; Jaillon, S. Innate immunity, inflammation and tumour progression: Double-edged swords. J. Intern. Med. 2019, 285, 524–532. [Google Scholar] [CrossRef] [Green Version]
- Turajlic, S.; Sottoriva, A.; Graham, T.; Swanton, C. Resolving genetic heterogeneity in cancer. Nat. Rev. Genet. 2019, 20, 404–416. [Google Scholar] [CrossRef]
- Soongsathitanon, J.; Jamjuntra, P.; Sumransub, N.; Yangngam, S.; De la Fuente, M.; Landskron, G.; Thuwajit, P.; Hermoso, M.A.; Thuwajit, C. Crosstalk between tumor-infiltrating immune cells and cancer-associated fibroblasts in tumor growth and immunosuppression of breast cancer. J Immunol. Res. 2021, 2021, 8840066. [Google Scholar] [CrossRef]
- Nejad, A.E.; Najafgholian, S.; Rostami, A.; Sistani, A.; Shojaeifar, S.; Esparvarinha, M.; Nedaeinia, R.; Javanmard, S.H.; Taherian, M.; Ahmadlou, M.; et al. The role of hypoxia in the tumor microenvironment and development of cancer stem cell: A novel approach to developing treatment. Cancer Cell. Int. 2021, 21, 62. [Google Scholar] [CrossRef] [PubMed]
- Greten, F.R.; Grivennikov, S.I. Inflammation and cancer: Triggers, mechanisms, and consequences. Immunity 2019, 51, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Wu, L.; Yan, G.; Chen, Y.; Zhou, M.; Wu, Y.; Li, Y. Inflammation and tumor progression: Signaling pathways and targeted intervention. Signal Transduct. Target. Ther. 2021, 6, 263. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, N.M.; Gucalp, A.; Dannenberg, A.J.; Hudis, C.A. Obesity and cancer mechanisms: Tumor microenvironment and inflammation. J. Clin. Oncol. 2016, 34, 4270–4276. [Google Scholar] [CrossRef]
- Mirlekar, B. Tumor promoting roles of IL-10, TGF-β, IL-4, and IL-35: Its implications in cancer immunotherapy. SAGE Open. Med. 2022, 10, 20503121211069012. [Google Scholar] [CrossRef] [PubMed]
- Setrerrahmane, S.; Xu, H. Tumor-related interleukins: Old validated targets for new anti-cancer drug development. Mol. Cancer 2017, 16, 153. [Google Scholar] [CrossRef]
- Edwardson, D.W.; Boudreau, J.; Mapletoft, J.; Lanner, C.; Kovala, A.T.; Parissenti, A.M. Inflammatory cytokine production in tumor cells upon chemotherapy drug exposure or upon selection for drug resistance. PLoS ONE 2017, 12, e0183662. [Google Scholar] [CrossRef] [Green Version]
- Paccagnella, M.; Abbona, A.; Michelotti, A.; Geuna, E.; Ruatta, F.; Landucci, E.; Denaro, N.; Vanella, P.; Lo Nigro, C.; Galizia, D.; et al. Circulating cytokines in metastatic breast cancer patients select different prognostic groups and patients who might benefit from treatment beyond progression. Vaccines 2022, 10, 78. [Google Scholar] [CrossRef]
- Cook, D.P.; Vanderhyden, B.C. Transcriptional census of epithelial-mesenchymal plasticity in cancer. Sci. Adv. 2022, 8, 7640. [Google Scholar] [CrossRef]
- Jun, J.C.; Rathore, A.; Younas, H.; Gilkes, D.; Polotsky, V.Y. Hypoxia-inducible factors and cancer. Curr. Sleep Med. Rep. 2017, 3, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Hoesel, B.; Schmid, J.A. The complexity of NF-κB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibrahim, S.A.E.-F.; Abudu, A.; Johnson, E.; Aftab, N.; Conrad, S.; Fluck, M. The role of AP-1 in self-sufficient proliferation and migration of cancer cells and its potential impact on an autocrine/paracrine loop. Oncotarget 2018, 9, 34259–34278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uhlitz, F.; Bischoff, P.; Peidli, S.; Sieber, A.; Trinks, A.; Lüthen, M.; Obermayer, B.; Blanc, E.; Ruchiy, Y.; Sell, T.; et al. Mitogen-activated protein kinase activity drives cell trajectories in colorectal cancer. EMBO Mol. Med. 2021, 13, e14123. [Google Scholar] [CrossRef] [PubMed]
- Markopoulos, G.S.; Roupakia, E.; Marcu, K.B.; Kolettas, E. Epigenetic regulation of inflammatory cytokine-induced epithelial-to-mesenchymal cell transition and cancer stem cell generation. Cells 2019, 8, 1143. [Google Scholar] [CrossRef] [Green Version]
- Huang, R.-X.; Zhou, P.-K. DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct. Target. Ther. 2020, 5, 60. [Google Scholar] [CrossRef]
- Pu, C.; Tao, S.; Xu, J.; Huang, M. Harnessing genomic stress for antitumor immunity. Antioxid. Redox Signal. 2021, 34, 1128–1150. [Google Scholar] [CrossRef]
- Sriram, G.; Milling, L.E.; Chen, J.-K.; Kong, Y.W.; Joughin, B.A.; Abraham, W.; Swartwout, S.; Handly, E.D.; Irvine, D.J.; Yaffe, M.B. The injury response to DNA damage in live tumor cells promotes antitumor immunity. Sci. Signal 2021, 14, 4764. [Google Scholar] [CrossRef]
- Marei, H.E.; Althani, A.; Afifi, N.; Hasan, A.; Caceci, T.; Pozzoli, G.; Morrione, A.; Giordano, A.; Cenciarelli, C. p53 signaling in cancer progression and therapy. Cancer Cell Int. 2021, 21, 703. [Google Scholar] [CrossRef]
- Chiang, Y.-T.; Chien, Y.-C.; Lin, Y.-H.; Wu, H.-H.; Lee, D.-F.; Yu, Y.-L. the function of the mutant p53-R175H in cancer. Cancers 2021, 13, 4088. [Google Scholar] [CrossRef]
- Turgeon, M.-O.; Perry, N.J.S.; Poulogiannis, G. DNA damage, repair, and cancer metabolism. Front. Oncol. 2018, 8, 15. [Google Scholar] [CrossRef] [Green Version]
- Arfin, S.; Jha, N.K.; Jha, S.K.; Kesari, K.K.; Ruokolainen, J.; Roychoudhury, S.; Rathi, B.; Kumar, D. Oxidative stress in cancer cell metabolism. Antioxidants 2021, 10, 642. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.K.; Chae, S.-W.; Kim, H.-R.; Chae, H.J. Endoplasmic reticulum stress and cancer. J. Cancer Prev. 2014, 19, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Cubillos-Ruiz, J.R. Endoplasmic reticulum stress signals in the tumour and its microenvironment. Nat. Rev. Cancer 2021, 21, 71–88. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, J.; Kumar, R.; Inigo, J.; Yadava, N.; Chandra, D. Mitochondrial stress response and cancer. Trends Cancer 2020, 6, 688–701. [Google Scholar] [CrossRef]
- Jang, J.-H.; Kim, D.-H.; Surh, Y.-J. Dynamic roles of inflammasomes in inflammatory tumor microenvironment. NPJ Precis. Oncol. 2021, 5, 18. [Google Scholar] [CrossRef]
- He, Q.; Fu, Y.; Tian, D.; Yan, W. The contrasting roles of inflammasomes in cancer. Am. J. Cancer Res. 2018, 8, 566–583. [Google Scholar]
- Agostini, M.; Ganini, C.; Candi, E.; Melino, G. The role of noncoding RNAs in epithelial cancer. Cell Death Discov. 2020, 6, 13. [Google Scholar] [CrossRef] [Green Version]
- Devaraj, S.; Singh, U.; Jialal, I. Human C-reactive protein and the metabolic syndrome. Curr. Opin. Lipidol. 2009, 20, 182–189. [Google Scholar] [CrossRef] [Green Version]
- Hassannejad, R.; Sharrouf, H.; Haghighatdoost, F.; Kirk, B.; Amirabdollahian, F. Diagnostic power of circulatory metabolic biomarkers as metabolic syndrome risk predictors in community-dwelling older adults in northwest of England (A feasibility study). Nutrients. 2021, 13, 2275. [Google Scholar] [CrossRef]
- Goligorsky, M.S. Vascular endothelium in diabetes. Am. J. Physiol. Renal. Physiol. 2017, 312, F266–F275. [Google Scholar] [CrossRef] [Green Version]
- Ye, J. Mechanisms of insulin resistance in obesity. Front. Med. 2013, 7, 14–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daryabor, G.; Atashzar, M.R.; Kabelitz, D.; Meri, S.; Kalantar, K. The effects of type 2 diabetes mellitus on organ metabolism and the immune system. Front. Immunol. 2020, 11, 1582. [Google Scholar] [CrossRef] [PubMed]
- Polyzos, S.A.; Margioris, A.N. Sarcopenic obesity. Hormones (Athens) 2018, 17, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wu, K.K.L.; Jiang, X.; Xu, A.; Cheng, K.K.Y. The role of adipose tissue senescence in obesity- and ageing-related metabolic disorders. Clin. Sci. (Lond). 2020, 134, 315–330. [Google Scholar] [CrossRef]
- Narasimhan, A.; Flores, R.R.; Robbins, P.D.; Niedernhofer, L.J. Role of cellular senescence in Type II diabetes. Endocrinology 2021, 162, 136. [Google Scholar] [CrossRef]
- Veronese, N.; Cooper, C.; Reginster, J.-Y.; Hochberg, M.; Branco, J.; Bruyère, O.; Chapurlat, R.; Al-Daghri, N.; Dennison, E.; Herrero-Beaumont, G.; et al. Type 2 diabetes mellitus and osteoarthritis. Semin. Arthritis Rheum. 2019, 49, 9–19. [Google Scholar] [CrossRef]
- Maliszewska, K.; Adamska-Patruno, E.; Krętowski, A. The interplay between muscle mass decline, obesity, and type 2 diabetes. Pol. Arch. Intern. Med. 2019, 129, 809–816. [Google Scholar] [CrossRef] [Green Version]
- Pugazhenthi, S.; Qin, L.; Reddy, P.H. Common neurodegenerative pathways in obesity, diabetes, and Alzheimer’s disease. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1037–1045. [Google Scholar] [CrossRef]
- Powell, E.E.; Wong, V.W.; Rinella, M. Non-alcoholic fatty liver disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef]
- Herculano-Houzel, S. The human brain in numbers: A linearly scaled-up primate brain. Front. Hum. Neurosci. 2009, 3, 31. [Google Scholar] [CrossRef] [Green Version]
- Azam, S.; Haque, M.E.; Balakrishnan, R.; Kim, I.-S.; Choi, D.-K. The Ageing Brain: Molecular and Cellular Basis of Neurodegeneration. Front. Cell Dev. Biol. 2021, 9, 683459. [Google Scholar] [CrossRef] [PubMed]
- Dugger, B.N.; Dickson, D.W. Pathology of Neurodegenerative Diseases. Cold Spring Harb. Perspect. Biol. 2017, 9, a028035. [Google Scholar] [CrossRef] [PubMed]
- Varderidou-Minasian, S.; Verheijen, B.M.; Schätzle, P.; Hoogenraad, C.C.; Pasterkamp, R.J.; Altelaar, M. Deciphering the proteome dynamics during development of neurons derived from induced pluripotent stem cells. J. Proteome Res. 2020, 19, 2391–2403. [Google Scholar] [CrossRef] [PubMed]
- Murao, N.; Nishitoh, H. Role of the unfolded protein response in the development of central nervous system. J. Biochem. 2017, 162, 155–162. [Google Scholar] [CrossRef] [Green Version]
- Hughes, D.; Mallucci, G.R. The unfolded protein response in neurodegenerative disorders—Therapeutic modulation of the perk pathway. FEBS J. 2019, 286, 342–355. [Google Scholar] [CrossRef]
- Kampinga, H.H.; Bergink, S. Heat shock proteins as potential targets for protective strategies in neurodegeneration. Lancet Neurol. 2016, 15, 748–759. [Google Scholar] [CrossRef]
- Webster, J.M.; Darling, A.L.; Uversky, V.N.; Blair, L.J. Small heat shock proteins, big impact on protein aggregation in neurodegenerative disease. Front. Pharmacol. 2019, 10, 1047. [Google Scholar] [CrossRef]
- Armada-Moreira, A.; Gomes, J.I.; Pina, C.C.; Savchak, O.K.; Gonçalves-Ribeiro, J.; Rei, N.; Pinto, S.; Morais, T.P.; Martins, R.S.; Ribeiro, F.F.; et al. Going the extra (synaptic) mile: Excitotoxicity as the road toward neurodegenerative diseases. Front. Cell. Neurosci. 2020, 14, 90. [Google Scholar] [CrossRef]
- Rossi, S.; Motta, C.; Studer, V.; Macchiarulo, G.; Volpe, E.; Barbieri, F.; Ruocco, G.; Buttari, F.; Finardi, A.; Mancino, R.; et al. Interleukin-1β causes excitotoxic neurodegeneration and multiple sclerosis disease progression by activating the apoptotic protein p53. Mol. Neurodegener. 2014, 9, 56. [Google Scholar] [CrossRef] [Green Version]
- Makhija, K.; Karunakaran, S. The role of inflammatory cytokines on the aetiopathogenesis of depression. Aust. N. Z. J. Psychiatry 2013, 47, 828–839. [Google Scholar] [CrossRef]
- Cobley, J.N.; Fiorello, M.L.; Bailey, D.M. 13 reasons why the brain is susceptible to oxidative stress. Redox. Biol. 2018, 15, 490–503. [Google Scholar] [CrossRef] [PubMed]
- Jha, M.K.; Morrison, B.M. Glia-neuron energy metabolism in health and diseases: New insights into the role of nervous system metabolic transporters. Exp. Neurol. 2018, 309, 23–31. [Google Scholar] [CrossRef]
- Graff-Radford, J. Vascular cognitive impairment. Continuum (Minneap. Minn.) 2019, 25, 147–164. [Google Scholar] [CrossRef] [PubMed]
- Daneman, R.; Prat, A. The blood–brain barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, a020412. [Google Scholar] [CrossRef] [Green Version]
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 2018, 14, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Harry, G.J. Microglia in neurodegenerative events—An initiator or a significant other? Int. J. Mol. Sci. 2021, 22, 5818. [Google Scholar] [CrossRef] [PubMed]
- Labandeira-Garcia, J.L.; Rodríguez-Perez, A.I.; Garrido-Gil, P.; Rodriguez-Pallares, J.; Lanciego, J.L.; Guerra, M.J. Brain renin-angiotensin system and microglial polarization: Implications for aging and neurodegeneration. Front. Aging Neurosci. 2017, 9, 129. [Google Scholar] [CrossRef] [Green Version]
- Chi, H.; Chang, H.Y.; Sang, T.K. Neuronal cell death mechanisms in major neurodegenerative diseases. Int. J. Mol. Sci. 2018, 19, 3082. [Google Scholar] [CrossRef] [Green Version]
- Hou, Y.; Dan, X.; Babbar, M.; Wei, Y.; Hasselbalch, S.G.; Croteau, D.L.; Bohr, V.A. Ageing as a risk factor for neurodegenerative disease. Nat. Rev. Neurol. 2019, 15, 565–581. [Google Scholar] [CrossRef]
- Breijyeh, Z.; Karaman, R. Comprehensive review on Alzheimer’s disease: Causes and treatment. Molecules 2020, 25, 5789. [Google Scholar] [CrossRef]
- Hirschberg, S.; Gisevius, B.; Duscha, A.; Haghikia, A. Implications of diet and the gut microbiome in neuroinflammatory and neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 3109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, H.; Bagarinao, E.; Maesawa, S.; Hara, K.; Kawabata, K.; Ogura, A.; Ohdake, R.; Shima, S.; Mizutani, Y.; Ueda, A.; et al. Characteristics of neural network changes in normal aging and early dementia. Front. Aging Neurosci. 2021, 13, 747359. [Google Scholar] [CrossRef] [PubMed]
- Garwood, C.J.; Ratcliffe, L.E.; Simpson, J.E.; Heath, P.R.; Ince, P.G.; Wharton, S.B. Review: Astrocytes in Alzheimer’s disease and other age-associated dementias: A supporting player with a central role. Neuropathol. Appl. Neurobiol. 2017, 43, 281–298. [Google Scholar] [CrossRef] [PubMed]
- Molnar, M.J.; Kovacs, G.G. Mitochondrial Diseases. Handb. Clin. Neurol. 2017, 145, 147–155. [Google Scholar] [CrossRef]
- Cohen, J.; Torres, C. Astrocyte senescence: Evidence and significance. Aging Cell. 2019, 18, e12937. [Google Scholar] [CrossRef] [Green Version]
- Han, X.; Zhang, T.; Liu, H.; Mi, Y.; Gou, X. Astrocyte senescence and Alzheimer’s disease: A review. Front. Aging Neurosci. 2020, 12, 148. [Google Scholar] [CrossRef] [PubMed]
- Freeman, L.; Guo, H.; David, C.N.; Brickey, W.J.; Jha, S.; Ting, J.P. NLR members NLRC4 and NLRP3 mediate sterile inflammasome activation in microglia and astrocytes. J. Exp. Med. 2017, 214, 1351–1370. [Google Scholar] [CrossRef] [Green Version]
- Duan, Y.; Kelley, N.; He, Y. Role of the NLRP3 inflammasome in neurodegenerative diseases and therapeutic implications. Neural Regen. Res. 2020, 15, 1249–1250. [Google Scholar] [CrossRef]
- Jin, X.; Liu, M.-Y.; Zhang, D.-F.; Zhong, X.; Du, K.; Qian, P.; Yao, W.-F.; Gao, H.; Wei, M.-J. Baicalin mitigates cognitive impairment and protects neurons from microglia-mediated neuroinflammation via suppressing NLRP3 inflammasomes and TLR4/NF-κB signaling pathway. CNS Neurosci. Ther. 2019, 25, 575–590. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Y.; Zhang, J.; Yang, G. Mechanisms of NLRP3 inflammasome activation: Its role in the treatment of Alzheimer’s disease. Neurochem. Res. 2020, 45, 2560–2572. [Google Scholar] [CrossRef]
- Lillis, A.P.; Van Duyn, L.B.; Murphy-Ullrich, J.E.; Strickland, D.K. LDL receptor-related protein 1: Unique tissue-specific functions revealed by selective gene knockout studies. Physiol. Rev. 2008, 88, 887–918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilkinson, K.; El Khoury, J. Microglial scavenger receptors and their roles in the pathogenesis of Alzheimer’s disease. Int. J. Alzheimers Dis. 2012, 2012, 489456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cornejo, F.; von Bernhardi, R. Role of scavenger receptors in glia-mediated neuroinflammatory response associated with Alzheimer’s disease. Mediat. Inflamm. 2013, 2013, 895651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iram, T.; Ramirez-Ortiz, Z.; Byrne, M.H.; Coleman, U.A.; Kingery, N.D.; Means, T.K.; Frenkel, D.; El Khoury, J. Megf10 is a receptor for C1Q that mediates clearance of apoptotic cells by astrocytes. J. Neurosci. 2016, 36, 5185–5192. [Google Scholar] [CrossRef] [Green Version]
- Eugenín, J.; Vecchiola, A.; Murgas, P.; Arroyo, P.; Cornejo, F.; von Bernhardi, R. Expression pattern of scavenger receptors and Amyloid-β phagocytosis of astrocytes and microglia in culture are modified by acidosis: Implications for Alzheimer’s disease. J. Alzheimers Dis. 2016, 53, 857–873. [Google Scholar] [CrossRef]
- Wang, J.-Y.; Lai, C.-L.; Lee, C.-T.; Lin, C.-Y. Electronegative low-density lipoprotein L5 impairs viability and NGF-induced neuronal differentiation of PC12 cells via LOX-1. Int. J. Mol. Sci. 2017, 18, 1744. [Google Scholar] [CrossRef] [Green Version]
- Wei, W.; Ma, D.; Li, L.; Zhang, L. Progress in the application of drugs for the treatment of multiple sclerosis. Front. Pharmacol. 2021, 12, 724718. [Google Scholar] [CrossRef]
- Lemus, H.N.; Warrington, A.E.; Rodriguez, M. Multiple sclerosis: Mechanisms of disease and strategies for myelin and axonal repair. Neurol. Clin. 2018, 36, 1–11. [Google Scholar] [CrossRef]
- Ghasemi, N.; Razavi, S.; Nikzad, E. Multiple sclerosis: Pathogenesis, symptoms, diagnoses and cell-based therapy. Cell J. 2017, 19, 1–10. [Google Scholar] [CrossRef]
- Poznyak, A.V.; Nikiforov, N.G.; Markin, A.M.; Kashirskikh, D.A.; Myasoedova, V.A.; Gerasimova, E.V.; Orekhov, A.N. Overview of OxLDL and its impact on cardiovascular health: Focus on atherosclerosis. Front. Pharmacol. 2021, 11, 613780. [Google Scholar] [CrossRef]
- Wysoczynski, M.; Kim, J.; Moore, J.B., 4th; Uchida, S. Macrophage long non-coding RNAs in pathogenesis of cardiovascular disease. Noncoding RNA 2020, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, C.J.; Lao, K.H.; Zeng, L. The salient role of micrornas in atherogenesis. J. Mol. Cell Cardiol. 2018, 122, 98–113. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Yang, C.; Qu, S.-L.; Shao, Y.-D.; Zhou, C.-Y.; Chao, R.; Huang, L.; Zhang, C. S100 proteins in atherosclerosis. Clin. Chim. Acta 2020, 502, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Jinnouchi, H.; Guo, L.; Sakamoto, A.; Torii, S.; Sato, Y.; Cornelissen, A.; Kuntz, S.; Paek, K.H.; Fernandez, R.; Fuller, D.; et al. Diversity of macrophage phenotypes and responses in atherosclerosis. Cell. Mol. Life Sci. 2020, 77, 1919–1932. [Google Scholar] [CrossRef] [PubMed]
- Bobryshev, Y.V.; Ivanova, E.A.; Chistiakov, D.A.; Nikiforov, N.G.; Orekhov, A.N. Macrophages and their role in atherosclerosis: Pathophysiology and transcriptome analysis. Biomed. Res. Int. 2016, 2016, 9582430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, P.; Ji, H.H.; Li, Y.J.; Guo, S.D. Macrophage plasticity and atherosclerosis therapy. Front. Mol. Biosci. 2021, 8, 679797. [Google Scholar] [CrossRef]
- Kyaw, T.; Peter, K.; Li, Y.; Tipping, P.; Toh, B.H.; Bobik, A. Cytotoxic lymphocytes and atherosclerosis: Significance, mechanisms and therapeutic challenges. Br. J. Pharmacol. 2017, 174, 3956–3972. [Google Scholar] [CrossRef] [Green Version]
- Depuydt, M.A.C.; Prange, K.H.M.; Slenders, L.; Örd, T.; Elbersen, D.; Boltjes, A.; de Jager, S.C.A.; Asselbergs, F.W.; de Borst, G.J.; Aavik, E.; et al. Microanatomy of the human atherosclerotic plaque by single-cell transcriptomics. Circ. Res. 2020, 127, 1437–1455. [Google Scholar] [CrossRef]
- Winkels, H.; Wolf, D. Heterogeneity of T cells in atherosclerosis defined by single-cell RNA-sequencing and cytometry by time of flight. Arter. Thromb. Vasc. Biol. 2021, 41, 549–563. [Google Scholar] [CrossRef]
- Kaiser, M.; Younge, B.; Björnsson, J.; Goronzy, J.J.; Weyand, C.M. Formation of new vasa vasorum in vasculitis: Production of angiogenic cytokines by multinucleated giant cells. Am. J. Pathol. 1999, 155, 765–774. [Google Scholar] [CrossRef]
- Numano, F. Vasa Vasoritis, Vasculitis and Atherosclerosis. Int. J. Cardiol. 2000, 75, S1–S19. [Google Scholar] [CrossRef]
- Gomez-Delgado, F.; Katsiki, N.; Lopez-Miranda, J.; Perez-Martinez, P. Dietary habits, lipoprotein metabolism and cardiovascular disease: From individual foods to dietary patterns. Crit. Rev. Food. Sci. Nutr. 2021, 61, 1651–1669. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, L.; Chen, W.; Xu, S.; Feng, X.; Zhang, L. Natural products: The role and mechanism in low-density lipoprotein oxidation and atherosclerosis. Phytother. Res. 2021, 35, 2945–2967. [Google Scholar] [CrossRef] [PubMed]
- Shibabaw, T. Omega-3 polyunsaturated fatty acids: Anti-inflammatory and anti-hypertriglyceridemia mechanisms in cardiovascular disease. Mol. Cell. Biochem. 2021, 476, 993–1003. [Google Scholar] [CrossRef]
- Steenbeke, M.; De Bruyne, S.; De Buyzere, M.; Lapauw, B.; Speeckaert, R.; Petrovic, M.; Delanghe, J.R.; Speeckaert, M.M. The role of soluble receptor for advanced glycation end-products (sRAGE) in the general population and patients with diabetes mellitus with a focus on renal function and overall outcome. Crit. Rev. Clin. Lab. Sci. 2021, 58, 113–130. [Google Scholar] [CrossRef]
- Gimbrone, M.A., Jr.; García-Cardeña, G. Vascular endothelium, hemodynamics, and the pathobiology of atherosclerosis. Cardiovasc. Pathol. 2013, 22, 9–15. [Google Scholar] [CrossRef] [Green Version]
- Mitra, R.; O’Neil, G.L.; Harding, I.C.; Cheng, M.J.; Mensah, S.A.; Ebong, E.E. Glycocalyx in atherosclerosis-relevant endothelium function and as a therapeutic target. Curr. Atheroscler. Rep. 2017, 19, 63. [Google Scholar] [CrossRef] [Green Version]
- Qu, J.; Cheng, Y.; Wu, W.; Yuan, L.; Liu, X. Glycocalyx impairment in vascular disease: Focus on inflammation. Front. Cell. Dev. Biol. 2021, 9, 730621. [Google Scholar] [CrossRef]
- De Siqueira, J.; Zani, I.A.; Russell, D.A.; Wheatcroft, S.B.; Ponnambalam, S.; Homer-Vanniasinkam, S. Clinical and preclinical use of LOX-1-Specific antibodies in diagnostics and therapeutics. J. Cardiovasc. Transl. Res. 2015, 8, 458–465. [Google Scholar] [CrossRef]
- Dai, Y.; Condorelli, G.; Mehta, J.L. Scavenger receptors and non-coding RNAs: Relevance in atherogenesis. Cardiovasc. Res. 2016, 109, 24–33. [Google Scholar] [CrossRef]
- Ben, J.; Zhu, X.; Zhang, H.; Chen, Q. Class A1 scavenger receptors in cardiovascular diseases. Br. J. Pharmacol. 2015, 172, 5523–5530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, K.; Xu, Y.; Sahebkar, A.; Xu, S. CD36 in atherosclerosis: Pathophysiological mechanisms and therapeutic implications. Curr. Atheroscler. Rep. 2020, 22, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Cheng, M.; Tong, F.; Su, X. Association between RAGE variants and the susceptibility to atherosclerotic lesions in Chinese Han population. Exp. Ther. Med. 2019, 17, 2019–2030. [Google Scholar] [CrossRef] [PubMed]
- Lenahan, C.; Huang, L.; Travis, Z.D.; Zhang, J.H. Scavenger receptor Class B type 1 (SR-B1) and the modifiable risk factors of stroke. Chin. Neurosurg. J. 2019, 5, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wild, J.B.; Stather, P.W.; Sylvius, N.; Choke, E.; Sayers, R.D.; Bown, M.J. Low density lipoprotein receptor related protein 1 and abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. 2012, 44, 127–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocha, V.Z.; Libby, P. Obesity, inflammation, and atherosclerosis. Nat. Rev. Cardiol. 2009, 6, 399–409. [Google Scholar] [CrossRef]
- Aboonabi, A.; Meyer, R.R.; Singh, I. The association between metabolic syndrome components and the development of atherosclerosis. J. Hum. Hypertens. 2019, 33, 844–855. [Google Scholar] [CrossRef]
- Low Wang, C.C.; Hess, C.N.; Hiatt, W.R.; Goldfine, A.B. Clinical update: Cardiovascular disease in diabetes mellitus: Atherosclerotic cardiovascular disease and heart failure in type 2 diabetes mellitus—Mechanisms, management, and clinical considerations. Circulation 2016, 133, 2459–2502. [Google Scholar] [CrossRef]
- Geng, S.; Chen, K.; Yuan, R.; Peng, L.; Maitra, U.; Diao, N.; Chen, C.; Zhang, Y.; Hu, Y.; Qi, C.F.; et al. The persistence of low-grade inflammatory monocytes contributes to aggravated atherosclerosis. Nat. Commun. 2016, 7, 13436. [Google Scholar] [CrossRef]
- Gusev, E.Y.; Zotova, N.V. Pathogenesis and prediction of critical complications of polytrauma from the position of common pathological processes. Polytrauma 2021, 1, 97–105. [Google Scholar]
- Bochkarev, P.Y.; Berdyugina, O.V.; Zhidkova, V.S.; Zubova, T.E.; Gusev, E.Y. The role of systemic inflammation in the pathogenesis of hemorrhagic stroke in the presence or absence of effective brain blood flow. Z. Nevrol. Psikhiatr. Im. S. S. Korsakova 2020, 120, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Iba, T.; Levi, M.; Levy, J.H. Sepsis-induced coagulopathy and disseminated intravascular coagulation. Semin. Thromb. Hemost. 2020, 46, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Pool, R.; Gomez, H.; Kellum, J.A. Mechanisms of organ dysfunction in sepsis. Crit. Care Clin. 2018, 34, 63–80. [Google Scholar] [CrossRef] [PubMed]
- Zotova, N.V.; Zhuravleva, Y.A.; Zubova, T.E.; Gusev, E.Y. Integral estimation of systemic inflammatory response under sepsis. Gen. Physiol. Biophys. 2020, 39, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Zotova, N.V.; Chereshnev, V.A.; Gusev, E.Y. Systemic inflammation: Methodological approaches to identification of the common pathological process. PLoS ONE 2016, 11, e0155138. [Google Scholar] [CrossRef] [PubMed]
- Gusev, E.Y.; Zotova, N.V.; Chereshnev, V.A. Sepsis-3: New edition—Old problems. Analysis from the perspective of general pathology. Russ. J. Infect. Immun. 2021, 11, 649–662. [Google Scholar] [CrossRef]
- Krautz, R.; Arefin, B.; Theopold, U. Damage signals in the insect immune response. Front. Plant. Sci. 2014, 5, 342. [Google Scholar] [CrossRef] [Green Version]
- Hibino, T.; Loza-Coll, M.; Messier, C.; Majeske, A.J.; Cohen, A.H.; Terwilliger, D.P.; Buckley, K.M.; Brockton, V.; Nair, S.V.; Berney, K.; et al. The immune gene repertoire encoded in the purple sea urchin genome. Dev. Biol. 2006, 300, 349–365. [Google Scholar] [CrossRef] [Green Version]
- Müller, L.; Fülöp, T.; Pawelec, G. Immunosenescence in vertebrates and invertebrates. Immun. Ageing 2013, 10, 12. [Google Scholar] [CrossRef] [Green Version]
- Aktipis, C.A.; Boddy, A.M.; Jansen, G.; Hibner, U.; Hochberg, M.E.; Maley, C.C.; Wilkinson, G.S. Cancer across the tree of life: Cooperation and cheating in multicellularity. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140219. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Zhou, Z.; Jiang, Q.; Wang, L.; Yi, Q.; Qiu, L.; Song, L. The neuroendocrine immunomodulatory axis-like pathway mediated by circulating hemocytes in pacific oyster crassostrea gigas. Open Biol. 2017, 7, 160289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gianazza, E.; Eberini, I.; Palazzolo, L.; Miller, I. Hemolymph proteins: An overview across marine arthropods and molluscs. J. Proteom. 2021, 245, 104294. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Yan, D.; Wang, X.; Zhang, L.; Chen, P. Hemocyte changes during immune melanization in bombyx mori infected with escherichia coli. Insects 2019, 10, 301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stączek, S.; Zdybicka-Barabas, A.; Wiater, A.; Pleszczyńska, M.; Cytryńska, M. Activation of cellular immune response in insect model host galleria mellonella by fungal α-1,3-glucan. Pathog. Dis. 2020, 78, 62. [Google Scholar] [CrossRef] [PubMed]
- Mao, F.; Wong, N.K.; Lin, Y.; Zhang, X.; Liu, K.; Huang, M.; Xu, D.; Xiang, Z.; Li, J.; Zhang, Y.; et al. transcriptomic evidence reveals the molecular basis for functional differentiation of hemocytes in a marine invertebrate, crassostrea gigas. Front. Immunol. 2020, 11, 911. [Google Scholar] [CrossRef]
- Xin, L.; Zhang, H.; Zhang, R.; Li, H.; Wang, W.; Wang, L.; Wang, H.; Qiu, L.; Song, L. CgIL17-5, an ancient inflammatory cytokine in crassostrea gigas exhibiting the heterogeneity functions compared with vertebrate interleukin17 molecules. Dev. Comp. Immunol. 2015, 53, 339–348. [Google Scholar] [CrossRef]
- Zheng, Y.; Liu, Z.; Wang, L.; Li, M.; Zhang, Y.; Zong, Y.; Li, Y.; Song, L. A novel tumor necrosis factor in the pacific oyster crassostrea gigas mediates the antibacterial response by triggering the synthesis of lysozyme and nitric oxide. Fish Shellfish Immunol. 2020, 98, 334–341. [Google Scholar] [CrossRef]
- Zhao, Y.H.; Li, H.; Zhao, H.; Sun, W.K.; Wang, Q.; Li, W.W. An ancient interleukin-16-like molecule regulates hemocyte proliferation via integrin β1 in invertebrates. J. Biol. Chem. 2021, 297, 100943. [Google Scholar] [CrossRef]
- Herwald, H.; Theopold, U. Hemostasis in Invertebrates and Vertebrates: An Evolutionary Excursion. J. Innate Immun. 2011, 3, 1–2. [Google Scholar] [CrossRef]
- Loof, T.G.; Schmidt, O.; Herwald, H.; Theopold, U. Coagulation systems of invertebrates and vertebrates and their roles in innate immunity: The same side of two coins? J. Innate Immun. 2011, 3, 34–40. [Google Scholar] [CrossRef]
- Melillo, D.; Marino, R.; Italiani, P.; Boraschi, D. Innate immune memory in invertebrate metazoans: A critical appraisal. Front. Immunol. 2018, 9, 1915. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.; Eleftherianos, I. Memory and specificity in the insect immune system: Current Perspectives and future challenges. Front. Immunol. 2017, 8, 539. [Google Scholar] [CrossRef] [PubMed]
- Tassetto, M.; Kunitomi, M.; Andino, R. Circulating immune cells mediate a systemic RNAi-based adaptive antiviral response in drosophila. Cell 2017, 169, 314–325.e13. [Google Scholar] [CrossRef] [Green Version]
- Russell, M.J.; Dombkowski, R.A.; Olson, K.R. Effects of hypoxia on vertebrate blood vessels. J. Exp. Zool. A Ecol. Genet. Physiol. 2008, 309, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.; Sethi, A.K.; Girotra, G.; Mohta, M. The microcirculation in sepsis. Indian J. Anaesth. 2009, 53, 281–293. [Google Scholar]
- Yamaguchi, T.; Takizawa, F.; Fischer, U.; Dijkstra, J.M. Along the axis between type 1 and type 2 immunity; principles conserved in evolution from fish to mammals. Biology 2015, 4, 814–859. [Google Scholar] [CrossRef] [Green Version]
- Zou, J.; Secombes, C.J. The function of fish cytokines. Biology 2016, 5, 23. [Google Scholar] [CrossRef]
- Gourbal, B.; Pinaud, S.; Beckers, G.J.M.; Van Der Meer, J.W.M.; Conrath, U.; Netea, M.G. Innate immune memory: An evolutionary perspective. Immunol. Rev. 2018, 283, 21–40. [Google Scholar] [CrossRef]
- Gusev, E.Y.; Zhuravleva, Y.A.; Zotova, N.V. Correlation of the evolution of immunity and inflammation in vertebrates. Biol. Bull. Rev. 2019, 9, 358–372. [Google Scholar] [CrossRef]
- Mulero, I.; Sepulcre, M.P.; Meseguer, J.; García-Ayala, A.; Mulero, V. Histamine is stored in mast cells of most evolutionarily advanced fish and regulates the fish inflammatory response. Proc. Natl. Acad. Sci. USA 2007, 104, 19434–19439. [Google Scholar] [CrossRef] [Green Version]
- Nonaka, M.; Yoshizaki, F. Evolution of the complement system. Mol. Immunol. 2004, 40, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Doolittle, R.F. Coagulation in vertebrates with a focus on evolution and inflammation. J. Innate Immun. 2011, 3, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Viertlboeck, B.C.; Schweinsberg, S.; Hanczaruk, M.A.; Schmitt, R.; Du Pasquier, L.; Herberg, F.W.; Göbel, T.W. The chicken leukocyte receptor complex encodes a primordial, activating, high-affinity IgY Fc receptor. Proc. Natl. Acad. Sci. USA 2007, 104, 11718–11723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Selye, H. Experimental evidence supporting the conception of “Adaptation Energy”. Am. J. Physiol. 1938, 123, 758–765. [Google Scholar] [CrossRef] [Green Version]
- Selye, H. The general adaptation syndrome and the diseases of adaptation. J. Clin. Endocrinol. Metab. 1946, 6, 117–230. [Google Scholar] [CrossRef]
- Sudheesh, P.S.; Al-Ghabshi, A.; Al-Mazrooei, N.; Al-Habsi, S. Comparative pathogenomics of bacteria causing infectious diseases in fish. Int. J. Evol. Biol. 2012, 2012, 457264. [Google Scholar] [CrossRef]
- Parto, P.; Haghighi, Z.M.S.; Vaissi, S.; Sharifi, M. Microbiological and histological examinations of endangered neurergus kaiseri tissues displaying red-leg syndrome. Asian Herpetol. Res. 2014, 5, 204–208. [Google Scholar] [CrossRef]
- Loch, T.P.; Faisal, M. Emerging flavobacterial infections in fish: A review. J. Adv. Res. 2015, 6, 283–300. [Google Scholar] [CrossRef]
- Hill, W.A.; Newman, S.J.; Craig, L.; Carter, C.; Czarra, J.; Brown, J.P. Diagnosis of aeromonas hydrophila, mycobacterium species, and batrachochytrium dendrobatidis in an African clawed frog (Xenopus laevis). J. Am. Assoc. Lab. Anim. Sci. 2010, 49, 215–220. [Google Scholar]
- Montali, R.J. Comparative pathology of inflammation in the higher vertebrates (reptiles, birds and mammals). J. Comp. Pathol. 1988, 99, 1–26. [Google Scholar] [CrossRef]
- Woo, P.C.; Lau, S.K.; Martelli, P.; Hui, S.W.; Lau, C.C.; Fan, R.Y.; Groff, J.M.; Tam, E.W.; Chan, K.H.; Yuen, K.Y. Fatal systemic necrotizing infections associated with a novel paramyxovirus, anaconda paramyxovirus, in green anaconda juveniles. J. Clin. Microbiol. 2014, 52, 3614–3623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lemon, M.J.; Pack, L.; Forzán, M.J. Valvular endocarditis and septic thrombosis associated with a radial fracture in a red-tailed hawk (Buteo jamaicensis). Can. Vet. J. 2012, 53, 79–82. [Google Scholar] [PubMed]
- Saumya, D.; Wijetunge, S.; Dunn, P.; Wallner-Pendleton, E.; Lintner, V.; Matthews, T.; Pierre, T.; Kariyawasam, S. Acute septicemia caused by streptococcus gallolyticus subsp. pasteurianus in Turkey poults. Avian Dis. 2014, 58, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Munford, R.S. Severe sepsis and septic shock: The role of gram-negative bacteremia. Annu. Rev. Pathol. 2006, 1, 467–496. [Google Scholar] [CrossRef] [PubMed]
- Rhee, C.; Murphy, M.V.; Li, L.; Platt, R.; Klompas, M. Centers for disease control and prevention epicenters program. comparison of trends in sepsis incidence and coding using administrative claims versus objective clinical data. Clin. Infect. Dis. 2015, 60, 88–95. [Google Scholar] [CrossRef] [Green Version]
- Hurley, J.C.; Nowak, P.; Öhrmalm, L.; Gogos, C.; Armaganidis, A.; Giamarellos-Bourboulis, E.J. Endotoxemia as a diagnostic tool for patients with suspected bacteremia caused by gram-negative organisms: A meta-analysis of 4 decades of studies. J. Clin. Microbiol. 2015, 53, 1183–1191. [Google Scholar] [CrossRef] [Green Version]
- Osterbur, K.; Mann, F.A.; Kuroki, K.; De Clue, A. Multiple organ dysfunction syndrome in humans and animals. J. Vet. Intern. Med. 2014, 28, 1141–1151. [Google Scholar] [CrossRef] [Green Version]
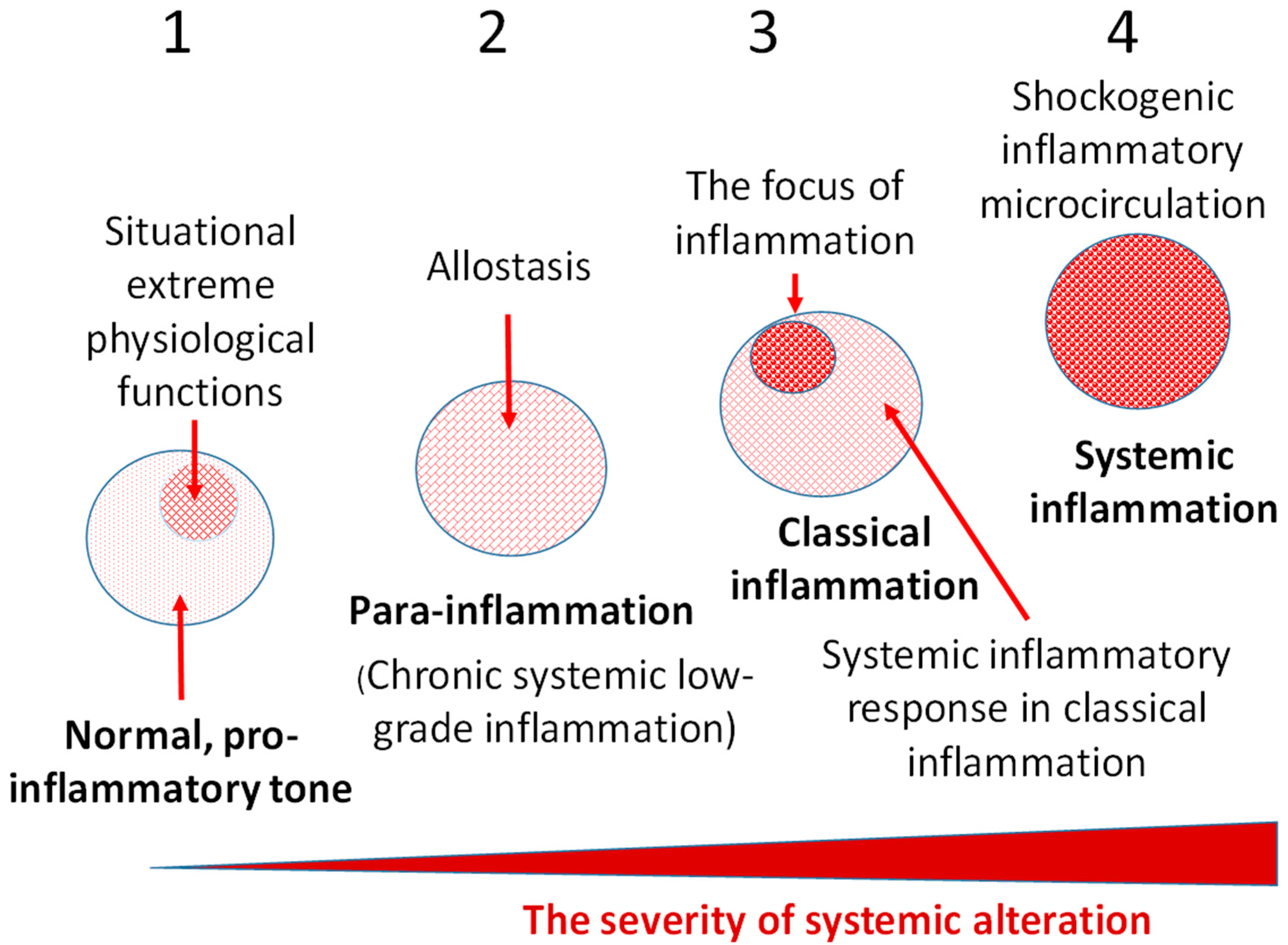
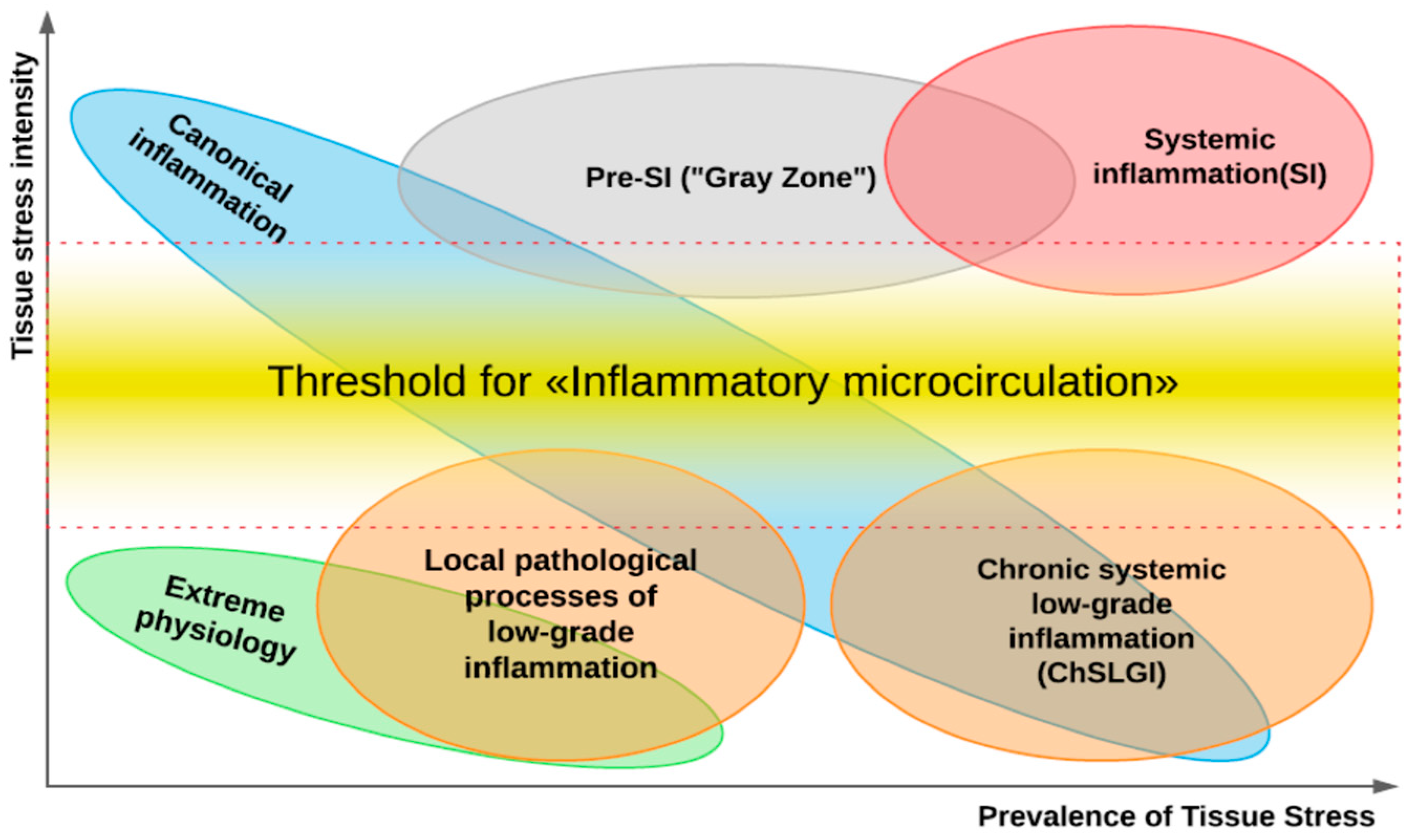

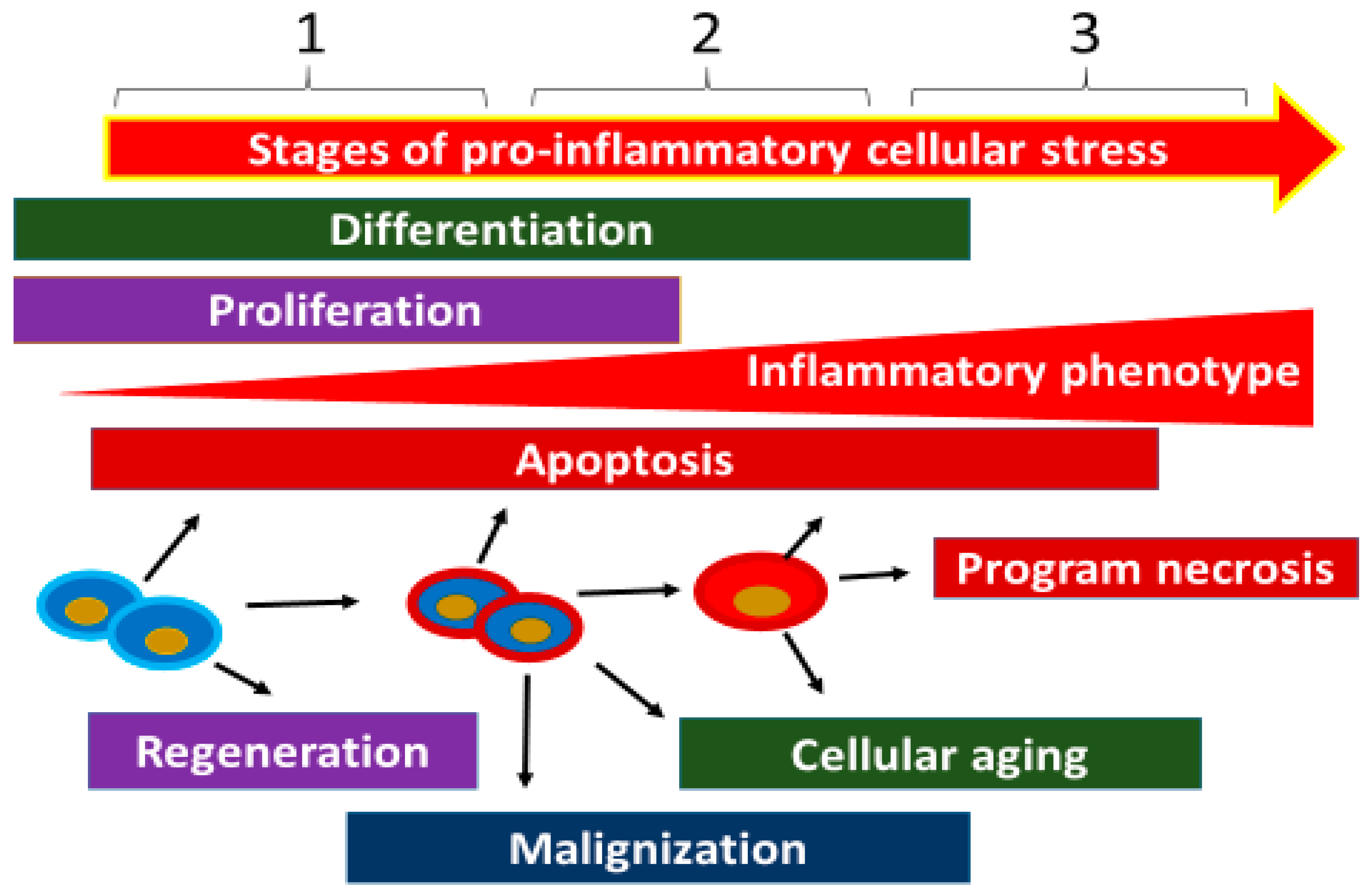
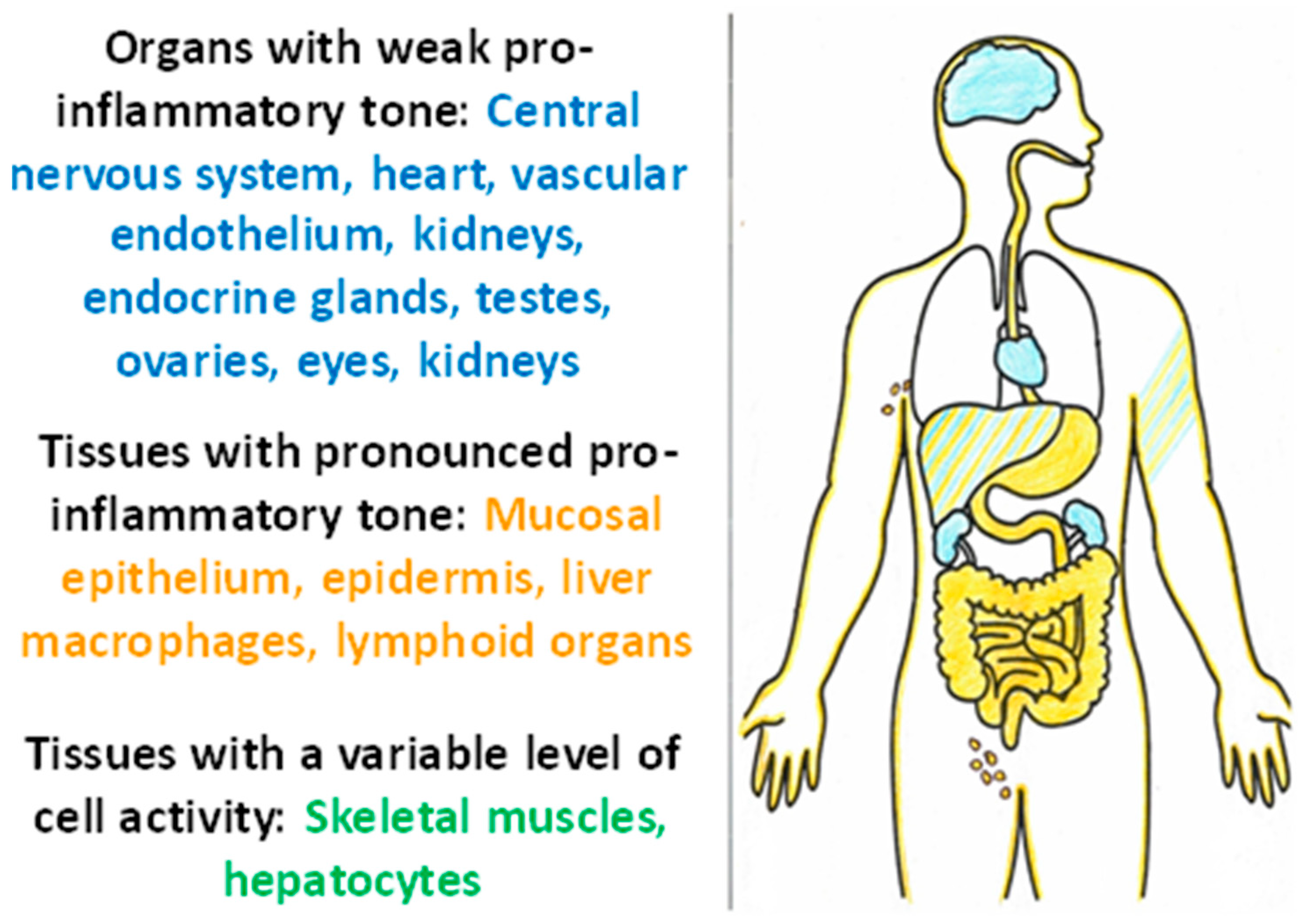
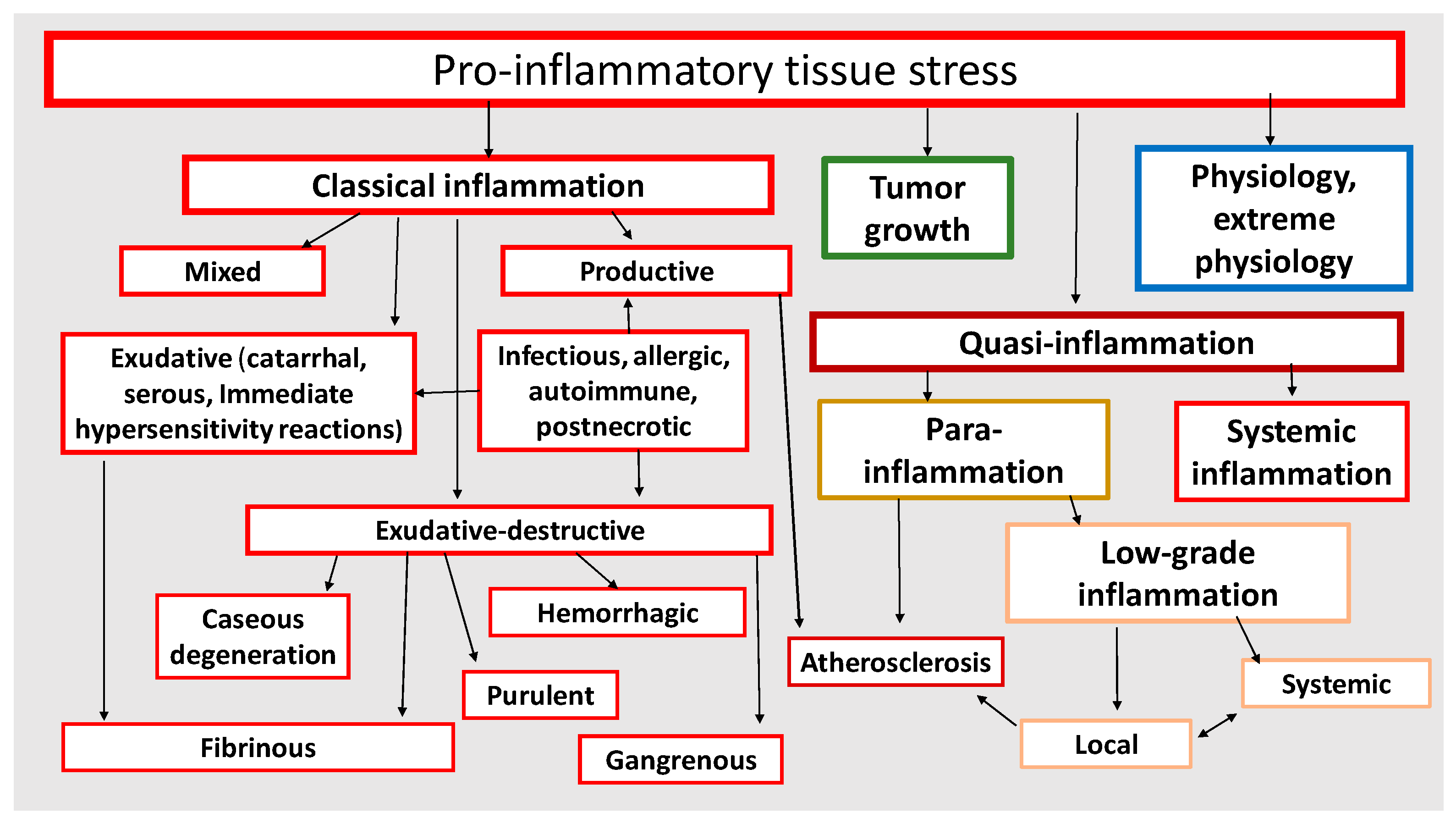
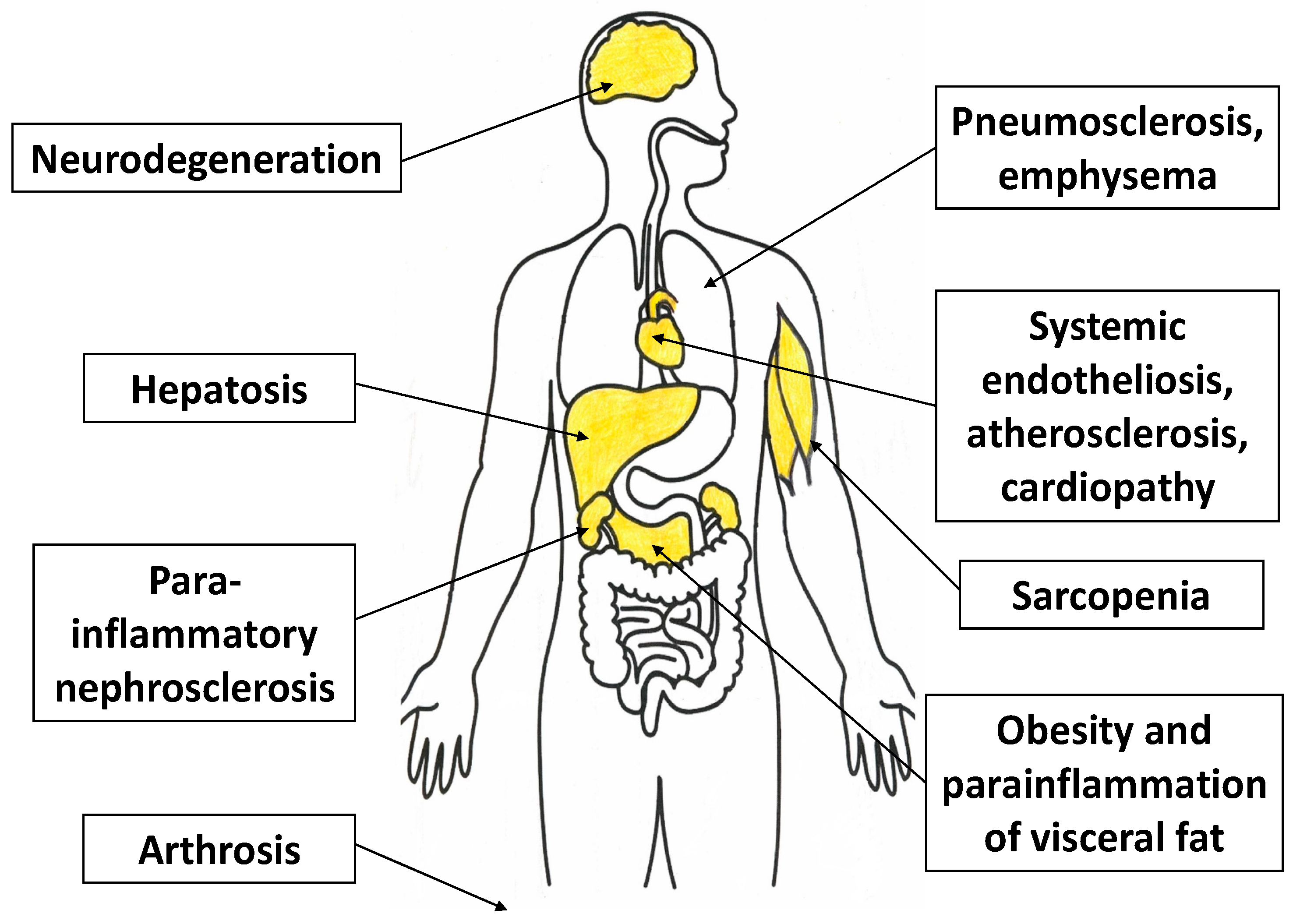

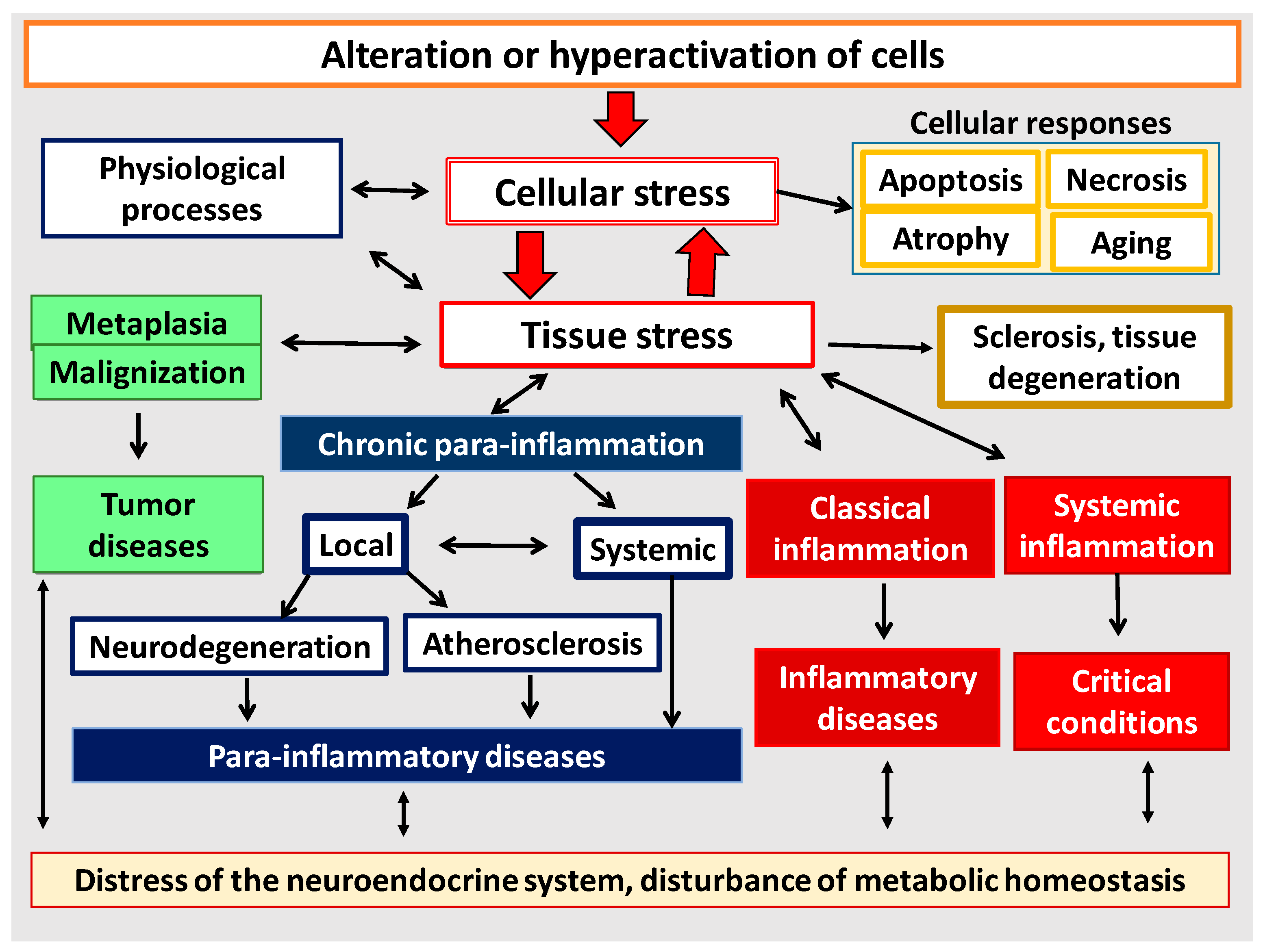
| Phenomena | Stage 1 | Stage 2 | Stage 3 |
|---|---|---|---|
| Proliferation | activated | variable | suppressed |
| Dominance 1 of growth factors | yes | no | no |
| Insulin resistance | no | possible | yes |
| Phosphoinositide 3-kinases (PI3K) 2 | activation | not typical | not typical |
| mTORC1 expression | high | variable | variable or low |
| Autophagy | low | elevated | high |
| Inflammasomes | low | NLRP3 activation in various cells | |
| Apoptosis | possible | possible | possible |
| Programmed necrosis 3 | not typical | unlikely | possible |
| Effects of SR on PRR | suppressed | variable | activated |
| Purinergic receptors 4 | P1 | P2X and P2Y | P2X and P2Y |
| p53/NF-κB ratio | ↑/↓ | ↓/↑ | ↓/↑ |
| Mitogen activated protein kinases | ERK > JNK and p38 | ERK < JNK and p38 | ERK < JNK and p38 |
| Production and reception of pro-inflammatory cytokines | moderate | high | Unstable 4 |
| iNOS endotheliocytes | inactive | inactive | active |
| cNOS endotheliocytes | ? | inhibited | ? |
| Unfolded protein response | progression | ||
| ROS formation | progression | ||
| NF-κB, AP-1, HIF-1α, HSFs, Egr | progression of expression of these transcription factors | ||
| The role of non-coding RNA | depends on cell type and formation of extracellular vesicles | ||
| I | Th (TFs), Cytokines: Activators and *-Inhibitors | Main Cytokines Th | Other Cells (TFs; Cytokines Production // Reception; *-Inhibitors) | Major Role in Inflammation | Complications |
|---|---|---|---|---|---|
| i1 | Th1 (T-bet, STAT4, STAT1); IL-12, IFN-γ; IL-4 *, IL-10 * | IFN-γ, IL-2, CXCL10, CXCL11 | M1 (STAT1, NF-κB; TNF-α, IL-1β, IL-6, IL-12, IL-15, IL-23 // IFN-γ, TNF-α; IL-10 *, TGF-β *), CTL, NK, ILC1 (IFN-γ) | Response to intracellular infection, antitumor immunity | Autoimmune processes, allograft rejection |
| i2 | Th2 1 (GATA3, STAT5, STAT6); IL-4, IL-25, IL-33; IFN-γ *, TGF-β *, IL-12 * | IL-4, IL-5, IL-13, IL-25, CCL17, CCL22 | M2a (STAT6, STAT1, GATA3; IL-6, IL-10 // IL-4, IL-13, IL-33), Tc2 (IL-5, IL-13), mast cells, basophils, ILC2 (IL-4), epithelial cells, eosinophils | Antimetazoan immunity, chronic inflammation, inflammation in damage-sensitive tissues | Allergic processes, i1 suppression, tissue fibrosis |
| i3 | Th17 (RORγt, RORα, STAT3, STAT5); IL-1β, IL-6, IL-23, TGFβ; IL-10* | IL-17A/F, IL-21, IL-22, CCL20, CXCL-1,7,20 | M2b (TNF-α, IL-1β, IL-6, IL-10 // IL-17A/F, TNF-α, IL-1, IL-6, IL-23; IL-10 *), Tc17 (IL-17), neutrophils, ILC3 | Response to extracellular infection | Autoimmune processes, allograft rejection |
| Th22 (RUNX3, AHR, STAT3); IL-6, IL-1β, TNF-α | IL-22, CCL-2, 20, CXCL-9, 10, 11, FGF | Epithelial cells, langerhans cells | Protection of the epidermis against extracellular infection | Autoimmune skin processes | |
| i-reg | Treg (FOXP3, STAT3/5, SMAD2/3, RORγt GATA3,); IL-2, IL-10, TGF-β | TGFβ, IL-10, CCL4 | M2c (SMAD2, SMAD3, STAT3; IL-10, TGFβ // IL-10, TGF-β), Tr1 (IL-10, IFN-γ), Tc-reg (TGFβ, IL-10), ILC10 (IL-10) | Limiting the expression of i1 and i3, inhibition of the autoimmune process | i1 and i3 immunosup- pression, infections, tumor growth |
| Immune and Inflammatory Mechanisms | Taxa | ||||
|---|---|---|---|---|---|
| Invertebrates | Bony Fishes | Reptiles | Birds | Mammals | |
| The reaction of phagocytes | Yes 1 | yes | yes | yes | yes |
| PRR in phagocytes | yes | yes | yes | yes | yes |
| Lymph formation 2 | no | yes | yes | yes | yes |
| The lymph nodes | no | no | no | yes/no | yes |
| Vessels, hearts | yes/no | yes | yes | yes | yes |
| Blood microcirculation | No 3 | yes | yes | yes | yes |
| Exudative reactions | no | yes | yes | yes | yes |
| Histamine in mast cells | no | yes/no 4 | yes | yes | yes |
| Anaphylatoxins(C3a, C5a) | no | yes | yes | yes | Yes 5 |
| Kinins | no | yes | yes | yes | yes |
| Kallikrein–kinins | no | no | yes | yes | yes |
| Hemostasis system | no | yes | yes | yes | Yes 6 |
| Non-nucleated platelets | no | no | no | no | yes |
| Adaptive immunity | yes/no | yes | yes | yes | yes |
| Lymphoid system | no | yes | yes | yes | yes |
| Cytokine network | No 7 | yes | yes | yes | yes |
| Main classes Ig | no | IgM | IgM, IgY | IgY, IgM | IgG, IgM |
| IgE | no | no | no | no | yes |
| Delayed-type hypersensitivity | no | no | no | yes/no 8 | yes |
| Autoimmune processes | no | yes | yes | yes | yes |
| Para-inflammation | yes | yes | yes | yes | yes |
| Classical inflammation | no | yes | yes | yes | yes |
| Purulent inflammation | no | no | no | no | yes |
| SIR | yes/no | yes | yes | yes | yes |
| Systemic inflammation 9 | no | no | no | ? | yes |
| NES distress reaction 10 | ? | ? | ? | yes | yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gusev, E.; Zhuravleva, Y. Inflammation: A New Look at an Old Problem. Int. J. Mol. Sci. 2022, 23, 4596. https://doi.org/10.3390/ijms23094596
Gusev E, Zhuravleva Y. Inflammation: A New Look at an Old Problem. International Journal of Molecular Sciences. 2022; 23(9):4596. https://doi.org/10.3390/ijms23094596
Chicago/Turabian StyleGusev, Evgenii, and Yulia Zhuravleva. 2022. "Inflammation: A New Look at an Old Problem" International Journal of Molecular Sciences 23, no. 9: 4596. https://doi.org/10.3390/ijms23094596
APA StyleGusev, E., & Zhuravleva, Y. (2022). Inflammation: A New Look at an Old Problem. International Journal of Molecular Sciences, 23(9), 4596. https://doi.org/10.3390/ijms23094596







