A Systematic Review of Atypical Endometriosis-Associated Biomarkers
Abstract
:1. Introduction
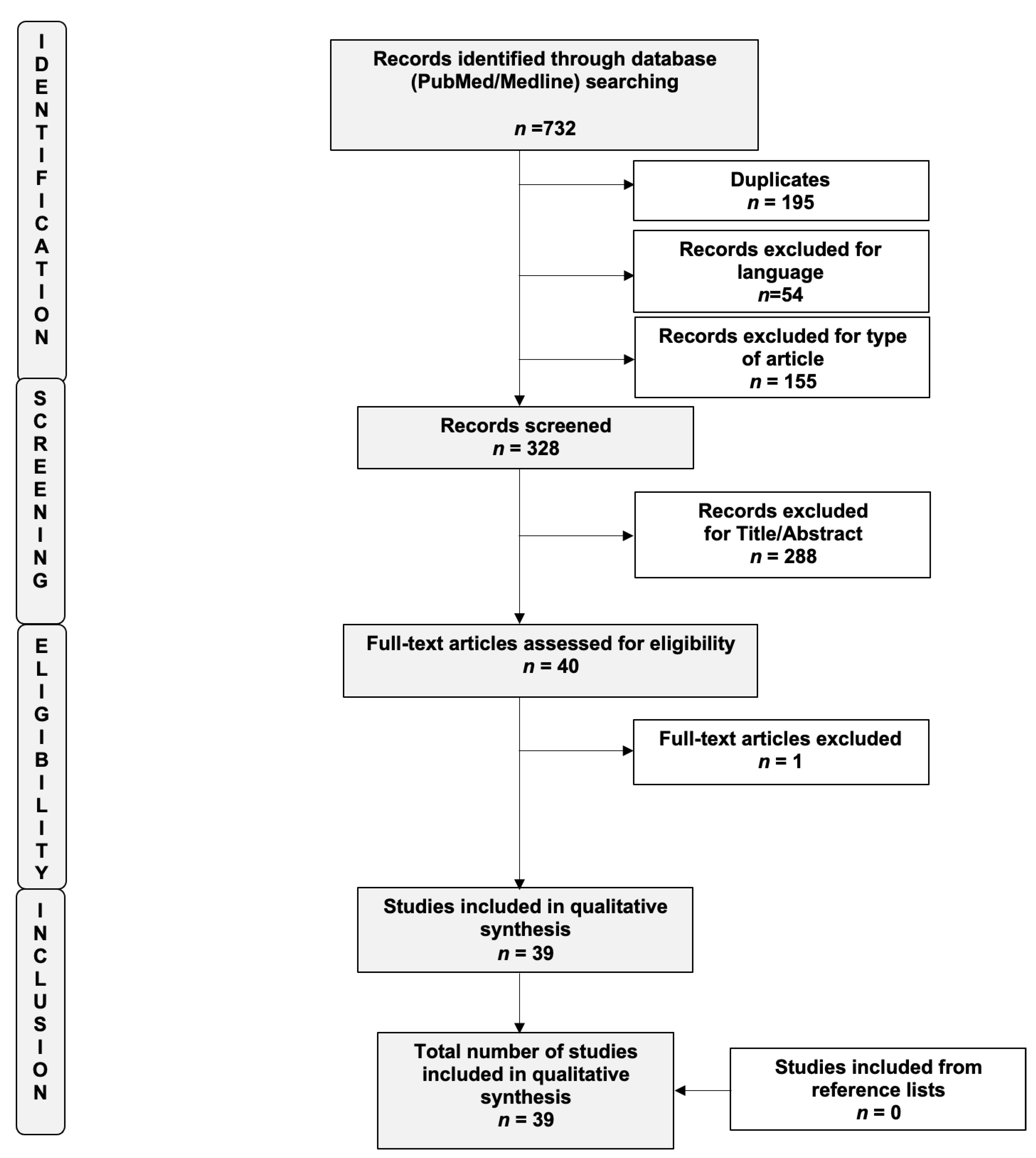
2. Materials and Methods
3. Results
3.1. PI3K/AKT/mTOR Pathway
3.2. ARID1a and BAF250
3.3. Estrogen Receptors and Progesterone Receptors
3.4. Transcriptional and Nuclear Factors
3.5. Growth Factors and Their Receptors
3.6. Others
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Parazzini, F.; Esposito, G.; Tozzi, L.; Noli, S.; Bianchi, S. Epidemiology of endometriosis and its comorbidities. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 209, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Sampson, J.A. Endometrial carcinoma of the ovary, arising in endometrial tissue in that organ. Arch. Surg. 1925, 10, 1–72. [Google Scholar] [CrossRef]
- Scott, R.B. Malignant changes in endometriosis. Obstet. Gynecol. 1953, 2, 283–289. [Google Scholar] [PubMed]
- Matias-Guiu, X.; Stewart, C.J. Endometriosis-associated ovarian neoplasia. Pathology 2018, 50, 190–204. [Google Scholar] [CrossRef]
- Pearce, C.L.; Templeman, C.; Rossing, M.A.; Lee, A.; Near, A.M.; Webb, P.M.; Nagle, C.M.; A Doherty, J.; Cushing-Haugen, K.L.; Wicklund, K.G.; et al. Association between endometriosis and risk of histological subtypes of ovarian cancer: A pooled analysis of case–control studies. Lancet Oncol. 2012, 13, 385–394. [Google Scholar] [CrossRef] [Green Version]
- Anglesio, M.S.; Yong, P. Endometriosis-associated Ovarian Cancers. Clin. Obstet. Gynecol. 2017, 60, 711–727. [Google Scholar] [CrossRef]
- Wilbur, M.A.; Shih, I.-M.; Segars, J.H.; Fader, A.N. Cancer Implications for Patients with Endometriosis. Semin. Reprod. Med. 2017, 35, 110–116. [Google Scholar] [CrossRef]
- Mikami, Y. Endometriosis-related ovarian neoplasms: Pathogenesis and histopathologic features. Diagn. Histopathol. 2014, 20, 357–363. [Google Scholar] [CrossRef]
- Králíčková, M.; Laganà, A.S.; Ghezzi, F.; Vetvicka, V. Endometriosis and risk of ovarian cancer: What do we know? Arch. Gynecol. Obstet. 2020, 301, 1–10. [Google Scholar] [CrossRef]
- Vetvicka, V.; Fiala, L.; Garzon, S.; Buzzaccarini, G.; Terzic, M.; Laganà, A.S. Endometriosis and gynaecological cancers: Molecular insights behind a complex machinery. Prz. Menopauzalny 2021, 20, 201–206. [Google Scholar] [CrossRef]
- Wiegand, K.C.; Shah, S.P.; Al-Agha, O.M.; Zhao, Y.; Tse, K.; Zeng, T.; Senz, J.; McConechy, M.K.; Anglesio, M.S.; Kalloger, S.E.; et al. ARID1AMutations in Endometriosis-Associated Ovarian Carcinomas. N. Engl. J. Med. 2010, 363, 1532–1543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiegand, K.C.; Lee, A.; Al-Agha, O.M.; Chow, C.; E Kalloger, S.; Scott, D.W.; Steidl, C.; Wiseman, S.M.; Gascoyne, R.D.; Gilks, B.; et al. Loss of BAF250a (ARID1A) is frequent in high-grade endometrial carcinomas. J. Pathol. 2011, 224, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Tsunoda, H.; Nishida, M.; Morishita, Y.; Takimoto, Y.; Kubo, T.; Noguchi, M. Loss of het- erozygosity on 10q23.3 and mutation of the tumor suppressor gene PTEN in benign endo- metrial cyst of the ovary: Possible sequence progression from benign endometrial cyst to endometrioid carcinoma and clear cell carcinoma of the ovary. Cancer Res. 2000, 60, 7052–7056. [Google Scholar] [PubMed]
- Yamamoto, S.; Tsuda, H.; Takano, M.; Iwaya, K.; Tamai, S.; Matsubara, O. PIK3CA mutation is an early event in the development of endometriosis-associated ovarian clear cell adenocarcinoma. J. Pathol. 2011, 225, 189–194. [Google Scholar] [CrossRef]
- Kato, N.; Sasou, S.-I.; Motoyama, T. Expression of hepatocyte nuclear factor-1beta (HNF-1beta) in clear cell tumors and endometriosis of the ovary. Mod. Pathol. 2005, 19, 83–89. [Google Scholar] [CrossRef] [Green Version]
- Fujimura, M.; Hidaka, T.; Kataoka, K.; Yamakawa, Y.; Akada, S.; Teranishi, A.; Saito, S. Absence of Estrogen Receptor-α Expression in Human Ovarian Clear Cell Adenocarcinoma Compared with Ovarian Serous, Endometrioid, and Mucinous Adenocarcinoma. Am. J. Surg. Pathol. 2001, 25, 667–672. [Google Scholar] [CrossRef]
- Akahane, T.; Sekizawa, A.; Purwosunu, Y.; Nagatsuka, M.; Okai, T. The Role of p53 Mutation in the Carcinomas Arising from Endometriosis. Int. J. Gynecol. Pathol. 2007, 26, 345–351. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef]
- EFSA Scientific Committee; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; Ricci, A.; et al. The principles and methods behind EFSA’s Guidance on Uncertainty Analysis in Scientific Assessment. EFSA J. 2018, 16, e05122. [Google Scholar] [CrossRef] [Green Version]
- Chalas, E.; ChumasS, J.; Barbieri, R.; Mann, W.J. Nucleolar organizer regions in endometriosis, atypical endometriosis, and clear cell and endometrioid carcinomas. Gynecol. Oncol. 1990, 40, 260–263. [Google Scholar] [CrossRef]
- Ogawa, S.; Kaku, T.; Amada, S.; Kobayashi, H.; Hirakawa, T.; Ariyoshi, K.; Kamura, T.; Nakano, H. Ovarian Endometriosis Associated with Ovarian Carcinoma: A Clinicopathological and Immunohistochemical Study. Gynecol. Oncol. 2000, 77, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Bayramođlu, H.; Duzcan, E. Atypical epithelial changes and mutant p53 gene expression in ovarian endometriosis. Pathol. Oncol. Res. 2001, 7, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Nezhat, F.; Cohen, C.; Rahaman, J.; Gretz, H.; Cole, P.; Kalir, T. Comparative immunohistochemical studies ofbcl-2 and p53 proteins in benign and malignant ovarian endometriotic cysts. Cancer 2002, 94, 2935–2940. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen, M.G.; Sehdev, A.E.S.; Fader, A.N.; Zahurak, M.L.; Richardson, M.; Fruehauf, J.P.; Montz, F.J.; Bristow, R.E. Endometriosis-associated ovarian carcinoma: Differential expression of vascular endothelial growth factor and estrogen/progesterone receptors. Cancer 2003, 98, 1658–1663. [Google Scholar] [CrossRef]
- Amemiya, S.; Sekizawa, A.; Otsuka, J.; Tachikawa, T.; Saito, H.; Okai, T. Malignant transformation of endometriosis and genetic alterations of K-ras and microsatellite instability. Int. J. Gynecol. Obstet. 2004, 86, 371–376. [Google Scholar] [CrossRef]
- de la Cuesta, R.S.; Izquierdo, M.; Cañamero, M.; Granizo, J.J.; Manzarbeitia, F. Increased prevalence of p53 overexpression from typical endometriosis to atypical endometriosis and ovarian cancer associated with endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2004, 113, 87–93. [Google Scholar] [CrossRef]
- Akahane, T.; Sekizawa, A.; Okuda, T.; Kushima, M.; Saito, H.; Okai, T. Disappearance of Steroid Hormone Dependency During Malignant Transformation of Ovarian Clear Cell Cancer. Int. J. Gynecol. Pathol. 2005, 24, 369–376. [Google Scholar] [CrossRef]
- Ali-Fehmi, R.; Khalifeh, I.; Bandyopadhyay, S.; Lawrence, W.D.; Silva, E.; Liao, D.; Sarkar, F.H.; Munkarah, A.R. Patterns of Loss of Heterozygosity at 10q23.3 and Microsatellite Instability in Endometriosis, Atypical Endometriosis, and Ovarian Carcinoma Arising in Association with Endometriosis. Int. J. Gynecol. Pathol. 2006, 25, 223–229. [Google Scholar] [CrossRef]
- Finas, D.; Huszar, M.; Agic, A.; Dogan, S.; Kiefel, H.; Riedle, S.; Gast, D.; Marcovich, R.; Noack, F.; Altevogt, P.; et al. L1 cell adhesion molecule (L1CAM) as a pathogenetic factor in endometriosis. Hum. Reprod. 2008, 23, 1053–1062. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, S.; Tsuda, H.; Takano, M.; Kita, T.; Kudoh, K.; Furuya, K.; Tamai, S.; Matsubara, O. Expression of platelet-derived growth factors and their receptors in ovarian clear-cell carcinoma and its putative precursors. Mod. Pathol. 2007, 21, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, S.; Tsuda, H.; Miyai, K.; Takano, M.; Tamai, S.; Matsubara, O. Cumulative alterations of p27Kip1-related cell-cycle regulators in the development of endometriosis-associated ovarian clear cell adenocarcinoma. Histopathology 2010, 56, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Yamamoto, S.; Takano, M.; Matsubara, O.; Furuya, K. Aberrant Expression of the Mammalian Target of Rapamycin, Hypoxia-inducible Factor-1α, and Glucose Transporter 1 in the Development of Ovarian Clear-cell Adenocarcinoma. Int. J. Gynecol. Pathol. 2012, 31, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.; Awadallah, A.; Xin, W. Loss of ARID1A/BAF250a expression in ovarian endometriosis and clear cell carcinoma. Int. J. Clin. Exp. Pathol. 2012, 5, 642–650. [Google Scholar] [PubMed]
- Yamamoto, S.; Tsuda, H.; Takano, M.; Tamai, S.; Matsubara, O. Loss of ARID1A protein expression occurs as an early event in ovarian clear-cell carcinoma development and frequently coexists with PIK3CA mutations. Mod. Pathol. 2011, 25, 615–624. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, S.; Tsuda, H.; Miyai, K.; Takano, M.; Tamai, S.; Matsubara, O. Accumulative copy number increase of MET drives tumor development and histological progression in a subset of ovarian clear-cell adenocarcinomas. Mod. Pathol. 2011, 25, 122–130. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, S.; Tsuda, H.; Honda, K.; Takano, M.; Tamai, S.; Imoto, I.; Inazawa, J.; Yamada, T.; Matsubara, O. ACTN4 gene amplification and actinin-4 protein overexpression drive tumour development and histological progression in a high-grade subset of ovarian clear-cell adenocarcinomas. Histopathology 2012, 60, 1073–1083. [Google Scholar] [CrossRef]
- Lai, C.-R.; Hsu, C.-Y.; Chen, Y.-J.; Yen, M.-S.; Chao, K.-C.; Li, A.F.-Y. Ovarian cancers arising from endometriosis: A microenvironmental biomarker study including ER, HNF1ß, p53, PTEN, BAF250a, and COX-2. J. Chin. Med. Assoc. 2013, 76, 629–634. [Google Scholar] [CrossRef] [Green Version]
- Vercellini, P.; Cribiù, F.M.; Del Gobbo, A.; Carcangiu, M.L.; Somigliana, E.; Bòsari, S. The oncofetal protein IMP3: A novel biomarker and triage tool for premalignant atypical endometriotic lesions. Fertil. Steril. 2013, 99, 1974–1979. [Google Scholar] [CrossRef]
- Lin, K.; Zhan, H.; Ma, J.; Xu, K.; Wu, R.; Zhou, C.; Lin, J. Increased steroid receptor RNA activator protein (SRAP) accompanied by decreased estrogen receptor-beta (ER-β) levels during the malignant transformation of endometriosis associated ovarian clear cell carcinoma. Acta Histochem. 2014, 116, 878–882. [Google Scholar] [CrossRef]
- Suryawanshi, S.; Huang, X.; Elishaev, E.; Budiu, R.A.; Zhang, L.; Kim, S.; Donnellan, N.; Mantia-Smaldone, G.; Ma, T.; Tseng, G.; et al. Complement Pathway Is Frequently Altered in Endometriosis and Endometriosis-Associated Ovarian Cancer. Clin. Cancer Res. 2014, 20, 6163–6174. [Google Scholar] [CrossRef] [Green Version]
- Anglesio, M.S.; Bashashati, A.; Wang, Y.K.; Senz, J.; Ha, G.; Yang, W.; Aniba, M.R.; Prentice, L.M.; Farahani, H.; Chang, H.L.; et al. Multifocal endometriotic lesions associated with cancer are clonal and carry a high mutation burden. J. Pathol. 2015, 236, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Yamazaki, M.; Takahashi, H.; Kajita, S.; Suzuki, E.; Tsuruta, T.; Saegusa, M. Distinct β-Catenin and PIK3CA Mutation Profiles in Endometriosis-Associated Ovarian Endometrioid and Clear Cell Carcinomas. Am. J. Clin. Pathol. 2015, 144, 452–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Er, T.-K.; Su, Y.-F.; Wu, C.-C.; Chen, C.-C.; Wang, J.; Hsieh, T.-H.; Herreros-Villanueva, M.; Chen, W.-T.; Chen, Y.-T.; Liu, T.-C.; et al. Targeted next-generation sequencing for molecular diagnosis of endometriosis-associated ovarian cancer. J. Mol. Med. 2016, 94, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Takano, M.; Miyamoto, M.; Sasaki, N.; Goto, T.; Suzuki, A.; Hirata, J.; Sasa, H.; Tsuda, H.; Furuya, K. Effect of ARID1A/BAF250a expression on carcinogenesis and clinicopathological factors in pure-type clear cell adenocarcinoma of the ovary. Mol. Clin. Oncol. 2016, 5, 395–401. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Hui, Y.; Lin, L.; Wu, Y.; Zhang, X.; Qin, X. Possible relevance of tumor-related genes mutation to malignant transformation of endometriosis. Eur. J. Gynaecol. Oncol. 2016, 37, 89–94. [Google Scholar]
- Stamp, J.P.; Gilks, C.B.; Wesseling, M.; Eshragh, S.; Ceballos, K.; Anglesio, M.S.; Kwon, J.S.; Tone, A.; Huntsman, D.G.; Carey, M.S. BAF250a Expression in Atypical Endometriosis and Endometriosis-Associated Ovarian Cancer. Int. J. Gynecol. Cancer 2016, 26, 825–832. [Google Scholar] [CrossRef]
- Andersen, C.L.; Boisen, M.M.; Sikora, M.J.; Ma, T.; Tseng, G.; Suryawanshi, S.; Vlad, A.; Elishaev, E.; Edwards, R.P.; Oesterreich, S. The Evolution of Estrogen Receptor Signaling in the Progression of Endometriosis to Endometriosis-Associated Ovarian Cancer. Horm. Cancer 2018, 9, 399–407. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, X.; Anaya, Y.; Parodi, L.; Cheng, L.; Anderson, M.L.; Hawkins, S.M. Distinct molecular pathways in ovarian endometrioid adenocarcinoma with concurrent endometriosis. Int. J. Cancer 2018, 143, 2505–2515. [Google Scholar] [CrossRef]
- Sevilla, I.; Linde, F.M.; Sánchez, M.D.P.M.; Arense, J.J.; Torroba, A.; Díaz, A.N.; Ferrer, M.L.S. Prognostic importance of atypical endometriosis with architectural hyperplasia versus cytologic atypia in endometriosis-associated ovarian cancer. J. Gynecol. Oncol. 2019, 30, e63. [Google Scholar] [CrossRef]
- Jiao, Y.; Lu, B. Poorly differentiated mucinous carcinoma with signet ring cells in an ovarian endometriotic cyst: A case report. Diagn. Pathol. 2019, 14, 73. [Google Scholar] [CrossRef] [Green Version]
- Păvăleanu, I.; Lozneanu, L.; Balan, R.A.; Giuşcă, S.E.; Avădănei, E.-R.; Căruntu, I.-D.; Amălinei, C. Insights into molecular pathways of endometriosis and endometriosis-related ovarian carcinoma. Rom. J. Morphol. Embryol. 2021, 61, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Penciu, R.-C.; Postolache, I.; Steriu, L.; Izvoranu, S.; Tica, A.-A.; Mocanu, I.-D.; Sârbu, V.; Deacu, M.; Tica, I.; Bălţătescu, G.-I.; et al. Is there a relationship in-between ovarian endometriosis and ovarian cancer? Immunohistochemical profile of four cases with coexisting ovarian endometriosis and cancer. Rom. J. Morphol. Embryol. 2020, 61, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Lenz, J.; Chvatal, R.; Fiala, L.; Konecna, P.; Lenz, D. Comparative immunohistochemical study of deep infiltrating endometriosis, lymph node endometriosis and atypical ovarian endometriosis including description of a perineural invasion. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech Repub. 2021, 165, 69–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, H.Y.; Yang, W.; Chay, D.B.; Lee, E.J.; Chung, J.Y.; Kim, H.S.; Kim, J.H. Tetraspanin 1 promotes endometriosis leading to ovarian clear cell carcinoma. Mol. Oncol. 2020, 15, 987–1004. [Google Scholar] [CrossRef]
- Varma, R.; Rollason, T.; Gupta, J.K.; Maher, E.R. Endometriosis and the neoplastic process. Reproduction 2004, 127, 293–304. [Google Scholar] [CrossRef] [Green Version]
- Fukunaga, M.; Nomura, K.; Ishikawa, E.; Ushigome, S. Ovarian atypical endometriosis: Its close association with malignant epithelial tumours. Histopathology 1997, 30, 249–255. [Google Scholar] [CrossRef]
- Berman, J.J.; Albores-Saavedra, J.; Bostwick, D.; DeLellis, R.; Eble, J.; Hamilton, S.R.; Hruban, R.H.; Mutter, G.L.; Page, D.; Rohan, T.; et al. Precancer: A conceptual working definition: Results of a Consensus Conference. Cancer Detect. Prev. 2006, 30, 387–394. [Google Scholar] [CrossRef]
- Mikhaleva, L.M.; Davydov, A.; Patsap, O.I.; Mikhaylenko, E.V.; Nikolenko, V.; Neganova, M.E.; Klochkov, S.; Somasundaram, S.; Kirkland, C.E.; Aliev, G. Malignant Transformation and Associated Biomarkers of Ovarian Endometriosis: A Narrative Review. Adv. Ther. 2020, 37, 2580–2603. [Google Scholar] [CrossRef]
- Anglesio, M.S.; Papadopoulos, N.; Ayhan, A.; Nazeran, T.M.; Noë, M.; Horlings, H.M.; Lum, A.; Jones, S.; Senz, J.; Seckin, T.; et al. Cancer-Associated Mutations in Endometriosis without Cancer. N. Engl. J. Med. 2017, 376, 1835–1848. [Google Scholar] [CrossRef] [Green Version]
- Cochrane, D.R.; Tessier-Cloutier, B.; Lawrence, K.M.; Nazeran, T.; Karnezis, A.N.; Salamanca, C.; Cheng, A.S.; McAlpine, J.N.; Hoang, L.N.; Gilks, C.B.; et al. Clear cell and endometrioid carcinomas: Are their differences attributable to distinct cells of 772 origin? J. Pathol. 2017, 243, 26–36. [Google Scholar] [CrossRef] [Green Version]
- Karnezis, A.N.; Cho, K.; Gilks, C.B.; Pearce, C.L.; Huntsman, D.G. The disparate origins of ovarian cancers: Pathogenesis and prevention strategies. Nat. Cancer 2016, 17, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Sieh, W.; Köbel, M.; A Longacre, T.; Bowtell, D.D.; Defazio, A.; Goodman, M.T.; Høgdall, E.; Deen, S.; Wentzensen, N.; Moysich, K.B.; et al. Hormone-receptor expression and ovarian cancer survival: An Ovarian Tumor Tissue Analysis consortium study. Lancet Oncol. 2013, 14, 853–862. [Google Scholar] [CrossRef] [Green Version]
- Lowery, W.J.; Schildkraut, J.M.; Akushevich, L.; Bentley, R.; Marks, J.R.; Huntsman, D.; Berchuck, A. Loss of ARID1A-Associated Protein Expression is a Frequent Event in Clear Cell and Endometrioid Ovarian Cancers. Int. J. Gynecol. Cancer 2012, 22, 9–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tkalia, I.G.; I Vorobyova, L.; Svintsitsky, V.S.; Nespryadko, S.V.; Goncharuk, I.V.; Lukyanova, N.Y.; Chekhun, V.F. Clinical significance of hormonal receptor status of malignant ovarian tumors. Exp. Oncol. 2014, 36, 125–133. [Google Scholar]
- Chen, S.; Dai, X.; Gao, Y.; Shen, F.; Ding, J.; Chen, Q. The positivity of estrogen receptor and progesterone receptor may not be associated with metastasis and recurrence in epithelial ovarian cancer. Sci. Rep. 2017, 7, 16922. [Google Scholar] [CrossRef] [Green Version]
- Chan, K.K.L.; Siu, M.K.Y.; Jiang, Y.X.; Wang, J.J.; Wang, Y.; Leung, T.H.Y.; Liu, S.S.; Cheung, A.N.Y.; Ngan, H.Y.S. Differential expression of estrogen receptor subtypes and variants in ovarian cancer: Effects on cell invasion, proliferation and prognosis. BMC Cancer 2017, 17, 606. [Google Scholar] [CrossRef]
- Jiang, X.; Morland, S.J.; Hitchcock, A.; Thomas, E.J.; Campbell, I.G. Allelo- typing of endometriosis with adjacent ovarian carcinoma reveals evidence of a common lineage. Cancer Res. 1998, 58, 1707–1712. [Google Scholar]
- Guan, B.; Rahmanto, Y.S.; Wu, R.-C.; Wang, Y.; Wang, Z.; Wang, T.-L.; Shih, I.-M. Roles of Deletion of Arid1a, a Tumor Suppressor, in Mouse Ovarian Tumorigenesis. JNCI J. Natl. Cancer Inst. 2014, 106, dju146. [Google Scholar] [CrossRef] [Green Version]
- Ghai, V.; Jan, H.; Shakir, F.; Haines, P.; Kent, A. Diagnostic delay for superficial and deep endometriosis in the United Kingdom. J. Obstet. Gynaecol. 2020, 40, 83–89. [Google Scholar] [CrossRef]
- Hudson, Q.J.; Ashjaei, K.; Perricos, A.; Kuessel, L.; Husslein, H.; Wenzl, R.; Yotova, I. Endometriosis Patients Show an Increased M2 Response in the Peritoneal CD14+low/CD68+low Macrophage Subpopulation Coupled with an Increase in the T-helper 2 and T-regulatory Cells. Reprod. Sci. 2020, 27, 1920–1931. [Google Scholar] [CrossRef]
- Seckin, B.; Ates, M.C.; Kirbas, A.; Yesilyurt, H. Usefulness of hematological parameters for differential diagnosis of endometriomas in adolescents/young adults and older women. Int. J. Adolesc. Med. Health 2018, 33. [Google Scholar] [CrossRef] [PubMed]
- Králíčková, M.; Vetvicka, V.; Fiala, L.; Laganà, A.S.; Garzon, S. The Search for Biomarkers in Endometriosis: A Long and Windy Road. Reprod. Sci. 2021, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Vanhie, A.; Dorien, O.; Peterse, D.; Beckers, A.; Cuéllar, A.; Fassbender, A.; Meuleman, C.; Mestdagh, P.; D’Hooghe, T. Plasma miRNAs as biomarkers for endometriosis. Hum. Reprod. 2019, 34, 1650–1660. [Google Scholar] [CrossRef] [PubMed]
- ETIC Endometriosis Treatment Italian Club; Alio, L.; Angioni, S.; Arena, S.; Bartiromo, L.; Bergamini, V.; Berlanda, N.; Bonin, C.; Busacca, M.; Candiani, M.; et al. When more is not better: 10 ’don’ts’ in endometriosis management. An ETIC* position statement. Hum. Reprod. Open 2019, 2019, hoz009. [Google Scholar] [CrossRef]
- Jing, X.; Li, C.; Sun, J.; Peng, J.; Dou, Y.; Xu, X.; Ma, C.; Dong, Z.; Liu, Y.; Zhang, H.; et al. Systemic Inflammatory Response Markers Associated with Infertility and Endometrioma or Uterine Leiomyoma in Endometriosis. Ther. Clin. Risk Manag. 2020, 16, 403–412. [Google Scholar] [CrossRef]
- Ottolina, J.; Bartiromo, L.; Dolci, C.; Salmeri, N.; Schimberni, M.; Villanacci, R.; Viganò, P.; Candiani, M. Assessment of Coagulation Parameters in Women Affected by Endometriosis: Validation Study and Systematic Review of the Literature. Diagnostics 2020, 10, 567. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhao, X.; Lu, J.; Xue, J.; Liu, P.; Mao, H. Prognostic roles of neutrophil to lymphocyte ratio and platelet to lymphocyte ratio in ovarian cancer: A meta-analysis of retrospective studies. Arch. Gynecol. Obstet. 2018, 297, 849–857. [Google Scholar] [CrossRef]
- Hainaut, P.; Plymoth, A. Targeting the hallmarks of can-cer: Towards a rational approach to next-generation cancer therapy. Curr Opin Oncol 2013, 25, 50–51. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, T.H.; Chung, H.H.; Song, Y.S. Risk and prognosis of ovarian cancer in women with endometriosis: A meta-analysis. Br. J. Cancer 2014, 110, 1878–1890. [Google Scholar] [CrossRef]
| Authors | Date | Type of Study | Immunohistochemical Analysis | Study Period | AE (n), Mean Age ± ds (yo) | EAOC (n), Mean Age ± ds (yo) | Endometrios (n), Mean Age ± ds (yo) | Results |
|---|---|---|---|---|---|---|---|---|
| Chalas et al. [20] | 1990 | Retrospective Study | Average AgNORs * per nucleus | NR | 10, NR | 10, NR | 10, NR |
|
| Ogawa et al. [21] | 2000 | Retrospective Study | Ki-67 | 1980–1995 | 29, NR | 30 CCC, 3 EnOC, 4 OSC, 51.4 yo (range, 22–80 years) | 33, NR |
|
| Bayramoglu et al. [22] | 2001 | Retrospective Study | p53 | NR | 7, 34.1 ± NR | 10, NR | 137, 36.1 ± NR |
|
| Nezhat et al. [23] | 2002 | Comparative Study | bcl-2, p53 | NR | -- | 24 EnOC, 19 CCC, 40 OSC; NR | 30; NR |
|
| Del Carmen et al. [24] | 2003 | Retrospective study | VEGF, Ki-67, ER, PR | NR | 17, NR | 8, NR | -- |
|
| Amemiya et al. [25] | 2004 | Case series | K-ras mutation MSI | 1987–1999 | -- | 12 EnOC, 56 ± 7.3 (range 43–68) | -- |
|
| Sáinz de la Cuesta et al. [26] | 2004 | Retrospective study | p53, c-erbB-2, MIB1, and Bcl-2 | 1948–1999 | 6 45.5 yo (range 33–78) | 17 45.5 yo (range 33–78) | 17 45.5 yo (range 33–78) |
|
| Akahane et al. [27] | 2005 | Case Series | ERα, ERβ, PRA, PRB p53, Ki-67, c-erbB-2, EGFR | 1993–2000 | -- | 4 CCC, 56,5 ± 1.11 4 EnOC, 56,8 ± 7.8 | -- | Both CCC and EnOC:
|
| Kato N et al. [15] | 2006 | Retrospective Study | HNF-1beta | NR | 4, 32–79 | 30 (17 CCC), 32–79 | 40, NR |
|
| Ali-Fehmi et al. [28] | 2006 | Retrospective Study | LOH and MSI at PTEN (10q23.3) in loci D10S215, D10S608, and D10S541 | NR | 12, NR | 20, NR 5 CCC 7 EnOC 8 OSC | 23, NR |
|
| Akahane et al. [17] | 2007 | Retrospective Study | p53 | 1993–2002 | -- | 13 CCC, 55 ± NR (range 29–62) 9 EnOC, 51 ± NR (range 42–76) | 7, 34 ± NR (range 30–43) |
|
| Finas et al. [29] | 2008 | Retrospective Study | L1CAM | NR | 14, NR | -- | 17, NR |
|
| Yamamoto et al. [30] | 2008 | Retrospective Study | PDGFR-a, PDGFR-b, PDGF-AB | 1987–2005 | 19, NR | 31 CCC, NR | 17, NR |
|
| Wiegand et al. [11] | 2010 | Case reports of 2 patients | BAF250a | NR | 2 AE contiguous to CCC | 2 CCC | 2 distant endometriosis to CCC |
|
| Yamamoto et al. [31] | 2010 | Retrospective Study | p27Kip1, Skp2, Cks1, cyclin A, and cyclin E, and Ki67 labeling index | 1988–2007 | 15, NR | 23 CCC, NR | 31, NR |
|
| Yamamoto et al. [14] | 2011 | Retrospective Study | PIK3CA | 1986–2007 | 8, NR | 23 CCC, NR | 8, NR |
|
| Kato et al. [32] | 2012 | Retrospective Study | p-mTOR, HIF-1, Glut1 | 1987–2005 | 16, NR | 36 CCC, NR | 21, NR |
|
| Xiao et al. [33] | 2012 | Retrospective study | BAF250a, HNF-1b, ER and PR | 1995–2010 | 13 | 26 CCC | 36 |
|
| Yamamoto et al. [34] | 2012 | Retrospective Study | ARID1A, PIK3CA | 1986–2007 | 22, NR | 28 CCC, NR | 22, NR |
|
| Yamamoto et al. [35] | 2012 | Retrospective Study | MET | 1987–2006 | 10, NR | 5 CCC, NR | 10, NR |
|
| Yamamoto et al. [36] | 2012 | Retrospective study | ACTN4 gene (encoding for Actinin-4) | 1986–2007 | 12 adjacent to tumor, of which:-9 both AE and BE -3 only AE | 16 adjacent to tumor, of -9 both AE and BE which:-7 only BE | 19 CCC |
|
| Lai et al. [37] | 2013 | Retrospective Study | ER, HNF-1 beta, p53, PTEN, BAF250a, COX-2 | 2001–2011 | -- | 79 ((33 EnOC; 40 CCC; 4 OSC; 1 ASC; 1 AS), NR | -- |
|
| Vercellini et al. [38] | 2013 | Retrospective study | IMP3 | 2004-2009 | 9 (35.1 ± 8.1) | NR | 508 |
|
| Lin et al. [39] | 2014 | Retrospective Study | SRAP, ERs | 2003–2012 | 12, 32–62 yo | 12 CCC, 32–62 yo | 24, NR |
|
| Suryawanshi et al. [40] | 2014 | Retrospective Study | Immune gene expression analysis | NR | 15 (48 ± 6.5) | 28 (54.8 yo ± 11.6) | NE: 32 (46.5 yo ± 6) BE:30 (40 yo ± 10) |
|
| Anglesio et al. [41] | 2015 | Whole-genome shotgun sequencing (WGSS) | Overall pattern of somatic mutations within EAOC (CCC and EnOC) | NR | - | 7 CCC | 7 |
|
| Matsumoto et al. [42] | 2015 | Retrospective Study | HIF-1a, iNOS, PIK3CA, pAkt, p65, and HNF-1β, Mutations of the β-Catenin and PIK3CA Genes | 2000–2014 | -- | 28 CCC, 35 EnOC, 54.1 (range 22–28) | -- |
|
| Er et al. [43] | 2016 | Case series | DNA extraction and sequencing | 2006–2012 | -- | 8 CCC 2 EnOC | -- |
|
| Kato et al. [44] | 2016 | Retrospective Study | BAF250a | 1984–2007 | 38, NR | 38 CCC, NR | 18, NR |
|
| Ma et al. [45] | 2016 | Retrospective Study | PTEN and p53 | 10, NR | 23, NR | 20, NR |
| |
| Stamp et al. [46] | 2016 | Retrospective study | BAF250a | 2005–2008 | 23 associated with cancer 8 not associated with cancer 45 yo (33–59) | 21 EnOC, 50 yo (30-70) 11 CCC, 57 yo (34–68) 3 mixed |
| |
| Andersen et al. [47] | 2018 | Retrospective Study | E2sig, ERα, ERβ, PR | NR | 11, 47 (range 34–20) | 19, 57.5 (range 47–77) 5 CCC 14 EnOC | 11, 39 (range 25–74) |
|
| Zhang et al. [48] | 2018 | Retrospective Study | genome-wide transcriptomic profiling | NR | 4, NR | 5 EnOC, NR | 4, NR |
|
| Niguez-Sevilla et al. [49] | 2019 | Prospective Study | Ki-67, BAF250a, COX-2 | 2014–2017 | 23 | 26 | 159 |
|
| Jiao et al. [50] | 2019 | Case Report | CK7, CEA, p16, CA 125, MUC-6, p53, PTEN, ER, PR, CK20, PAX-8, CDX-2 | NR | 46 yo | 1 OMC | NR |
|
| Păvăleanu et al. [51] | 2020 | Retrospective Study | E-cadherin,β-catenin, CK18, Bcl-2/Bax, ER, PR | 2005-2017 | -- | 19 (8 EnOC; 11 non EnOC), 59.10 yo | 31, 36.61 yo |
|
| Penciu et al. [52] | 2020 | Case Series | ER, PR, Ki67, p53, p16, WT1, CD 34, CD10 | 2015–2017 | -- | 2 EnOC, 2 OSC; 30–60 yo | -- |
|
| Lenz et al. [53] | 2021 | Retrospective Study | ER, PR, Ki67, p53 | NR | 5, NR | -- | 40, 33.1 yo (22.0–47.0) |
|
| Shin et al. [54] | 2021 | Case control study | TSPAN1 | NR | 18 (40 ± 6.6) | 7 AdjEm (44.5 ± 11) 17 CCC (43.6 ± 10.6) 12 EnOC (48.5 + 10) | 9 (27.7 ± 5.4) |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartiromo, L.; Schimberni, M.; Villanacci, R.; Mangili, G.; Ferrari, S.; Ottolina, J.; Salmeri, N.; Dolci, C.; Tandoi, I.; Candiani, M. A Systematic Review of Atypical Endometriosis-Associated Biomarkers. Int. J. Mol. Sci. 2022, 23, 4425. https://doi.org/10.3390/ijms23084425
Bartiromo L, Schimberni M, Villanacci R, Mangili G, Ferrari S, Ottolina J, Salmeri N, Dolci C, Tandoi I, Candiani M. A Systematic Review of Atypical Endometriosis-Associated Biomarkers. International Journal of Molecular Sciences. 2022; 23(8):4425. https://doi.org/10.3390/ijms23084425
Chicago/Turabian StyleBartiromo, Ludovica, Matteo Schimberni, Roberta Villanacci, Giorgia Mangili, Stefano Ferrari, Jessica Ottolina, Noemi Salmeri, Carolina Dolci, Iacopo Tandoi, and Massimo Candiani. 2022. "A Systematic Review of Atypical Endometriosis-Associated Biomarkers" International Journal of Molecular Sciences 23, no. 8: 4425. https://doi.org/10.3390/ijms23084425
APA StyleBartiromo, L., Schimberni, M., Villanacci, R., Mangili, G., Ferrari, S., Ottolina, J., Salmeri, N., Dolci, C., Tandoi, I., & Candiani, M. (2022). A Systematic Review of Atypical Endometriosis-Associated Biomarkers. International Journal of Molecular Sciences, 23(8), 4425. https://doi.org/10.3390/ijms23084425








