Interactions between Nanoparticles and Intestine
Abstract
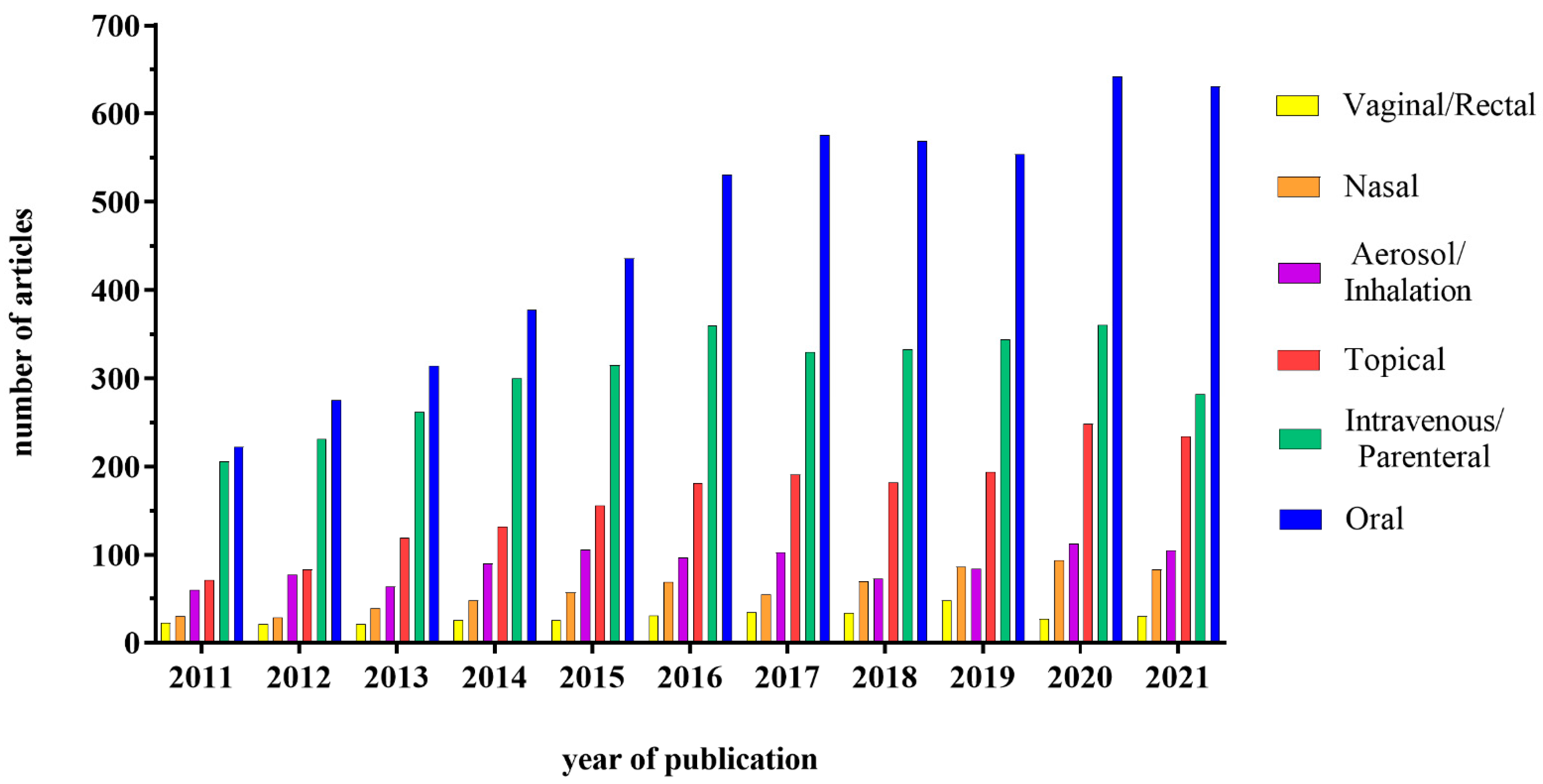
1. Introduction
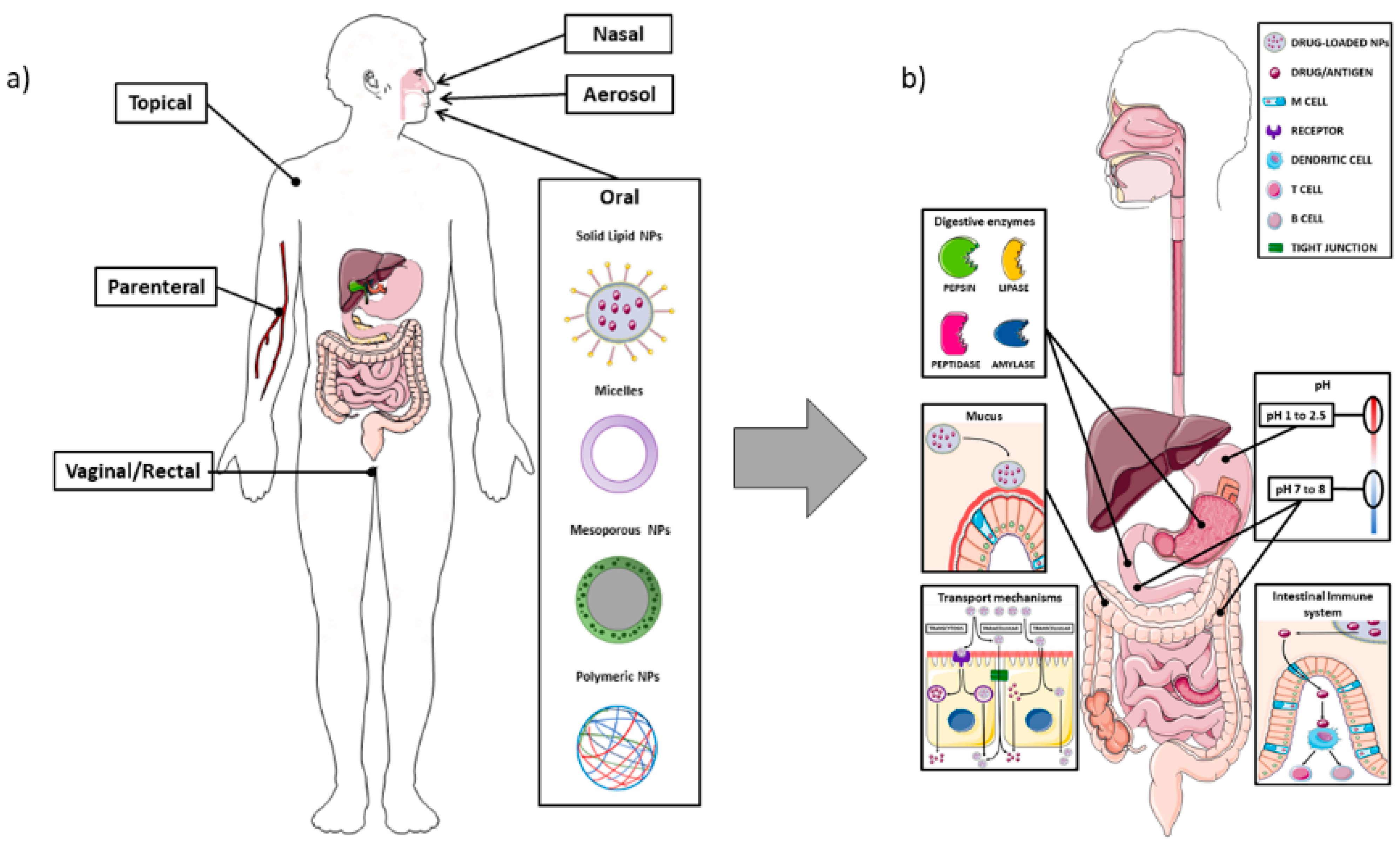
1.1. The Intestinal Barrier
1.2. Nanoparticles
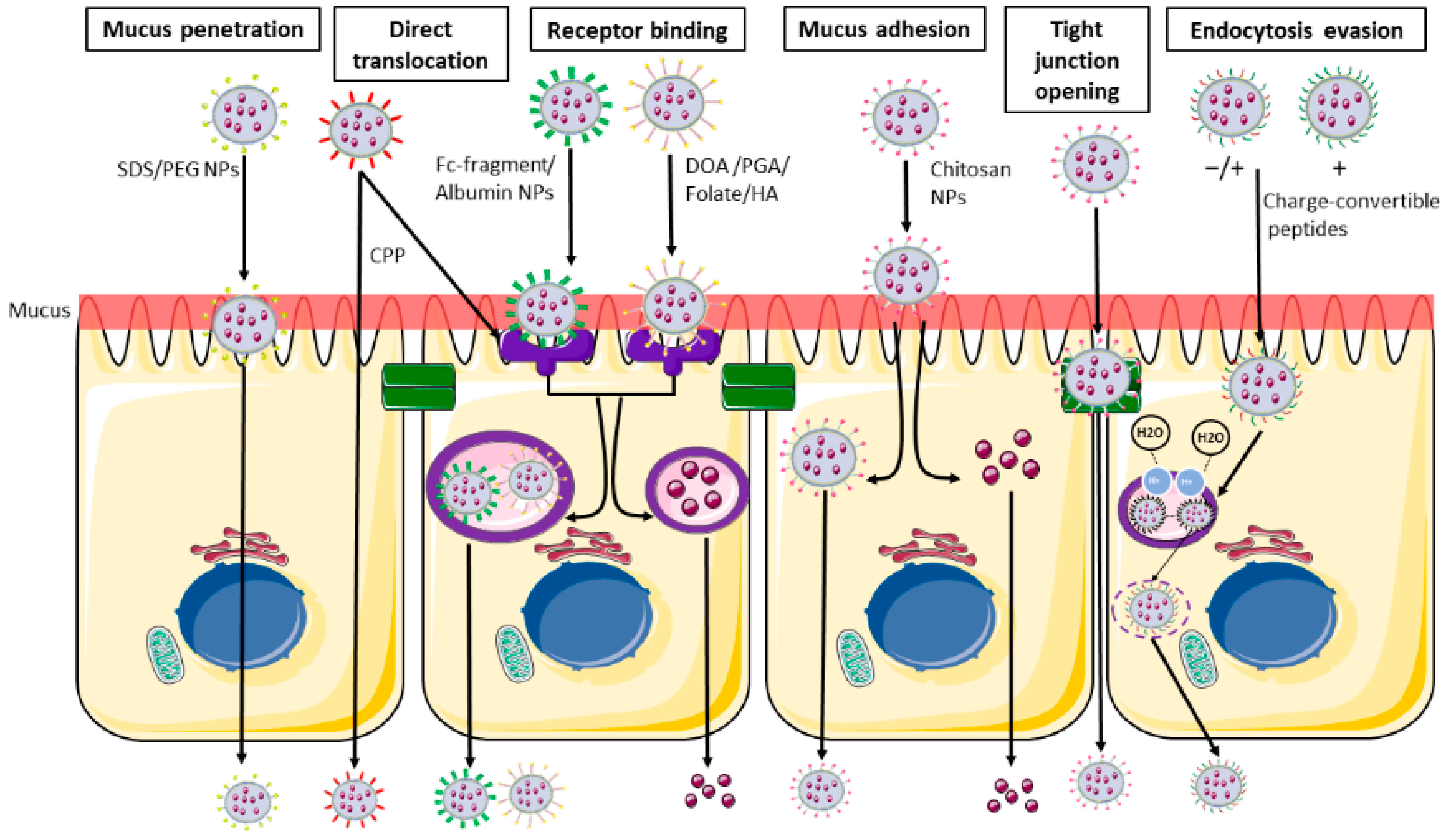
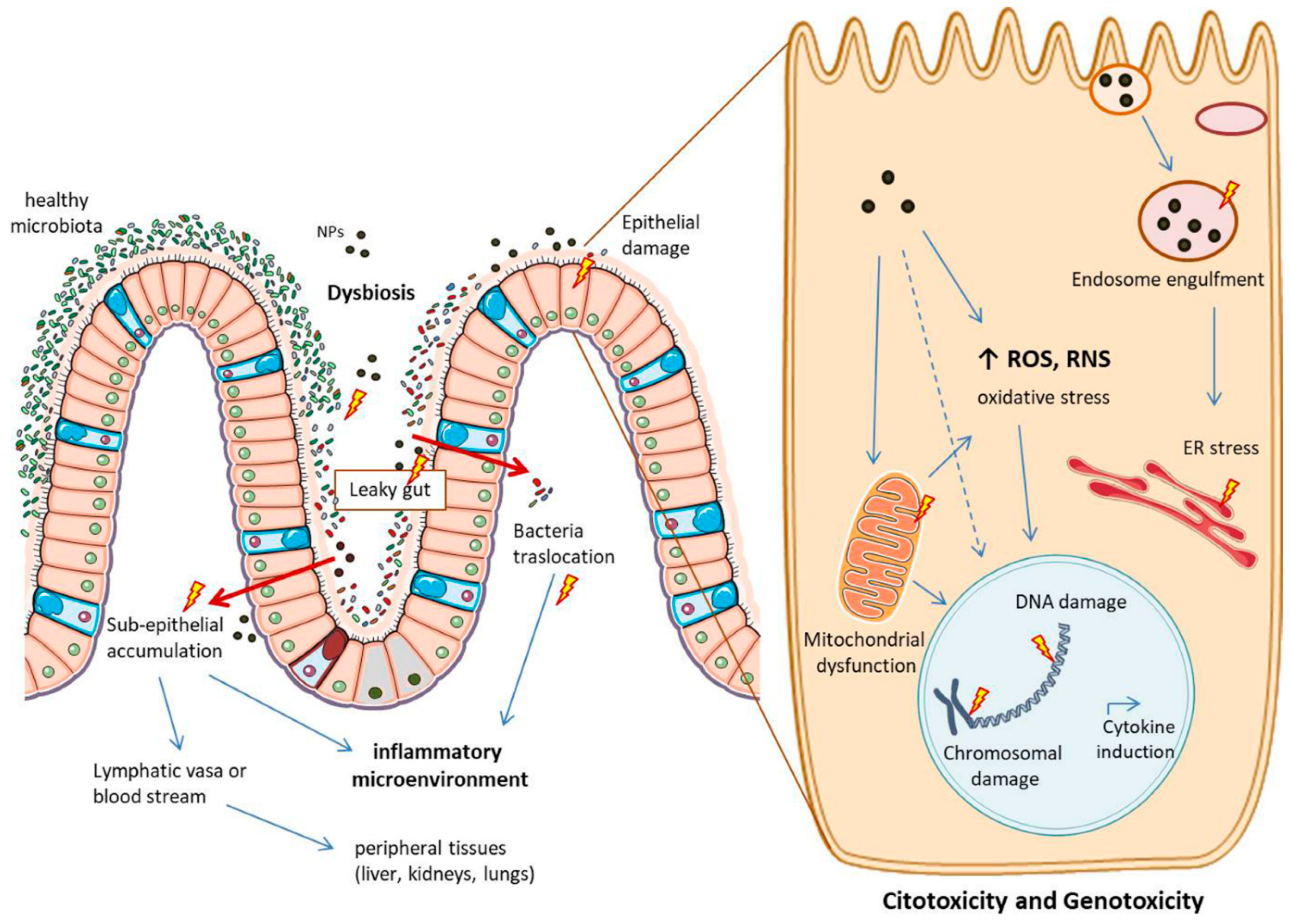
1.3. Nanoparticles—Intestine Interaction
2. Nanoparticles for Systemic Drug Delivery
3. Nanoparticles with Intestinal Targets
3.1. Inflammatory Bowel Diseases
3.2. Nanoparticles Loading Drugs
3.3. Nanoparticles Loading Biologics
3.4. Colon Cancer
4. Clinical Trials Status
5. Food Additives
5.1. E171—Titanium Dioxide
5.2. E174—Silver NPs
6. Summary
7. Future Perspective and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cueva, C.; Gil-Sánchez, I.; Tamargo, A.; Miralles, B.; Crespo, J.; Bartolomé, B.; Moreno-Arribas, M.V. Gastrointestinal digestion of food-use silver nanoparticles in the dynamic SIMulator of the GastroIntestinal tract (simgi®). Impact on human gut microbiota. Food Chem. Toxicol. 2019, 132, 110657. [Google Scholar] [CrossRef] [PubMed]
- Amara, S.; Bourlieu, C.; Humbert, L.; Rainteau, D.; Carrière, F. Variations in gastrointestinal lipases, pH and bile acid levels with food intake, age and diseases: Possible impact on oral lipid-based drug delivery systems. Adv. Drug Deliv. Rev. 2019, 142, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Ensign, L.M.; Cone, R.; Hanes, J. Oral drug delivery with polymeric nanoparticles: The gastrointestinal mucus barriers. Adv. Drug Deliv. Rev. 2012, 64, 557–570. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lu, Y.; Qi, J.; Wu, W. An update on oral drug delivery via intestinal lymphatic transport. Acta Pharm. Sin. B 2021, 11, 2449–2468. [Google Scholar] [CrossRef]
- Huang, X.; Tang, M. Review of gut nanotoxicology in mammals: Exposure, transformation, distribution and toxicity. Sci. Total Environ. 2021, 773, 145078. [Google Scholar] [CrossRef]
- Mancuso, C.; Re, F.; Rivolta, I.; Elli, L.; Gnodi, E.; Beaulieu, J.F.; Barisani, D. Dietary nanoparticles interact with gluten peptides and alter the intestinal homeostasis increasing the risk of celiac disease. Int. J. Mol. Sci. 2021, 22, 6102. [Google Scholar] [CrossRef]
- Ladaycia, A.; Passirani, C.; Lepeltier, E. Microbiota and nanoparticles: Description and interactions. Eur. J. Pharm. Biopharm. 2021, 169, 220–240. [Google Scholar] [CrossRef]
- Yu, R.; Ahmed, T.; Jiang, H.; Zhou, G.; Zhang, M.; Lv, L.; Li, B. Impact of zinc oxide nanoparticles on the composition of gut microbiota in healthy and autism spectrum disorder children. Materials 2021, 14, 5488. [Google Scholar] [CrossRef]
- Zhou, G.; Yu, R.; Ahmed, T.; Jiang, H.; Zhang, M.; Lv, L.; Alhumaydhi, F.A.; Allemailem, K.S.; Li, B. Biosynthesis and Characterization of Zinc Oxide Nanoparticles and Their Impact on the Composition of Gut Microbiota in Healthy and Attention-Deficit Hyperactivity Disorder Children. Front. Microbiol. 2021, 12, 2051. [Google Scholar] [CrossRef]
- Perez, L.; Scarcello, E.; Ibouraadaten, S.; Yakoub, Y.; Leinardi, R.; Ambroise, J.; Bearzatto, B.; Gala, J.L.; Paquot, A.; Muccioli, G.G.; et al. Dietary nanoparticles alter the composition and function of the gut microbiota in mice at dose levels relevant for human exposure. Food Chem. Toxicol. 2021, 154, 112352. [Google Scholar] [CrossRef]
- Ghebretatios, M.; Schaly, S.; Prakash, S. Nanoparticles in the food industry and their impact on human gut microbiome and diseases. Int. J. Mol. Sci. 2021, 22, 1942. [Google Scholar] [CrossRef] [PubMed]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.N.; Liu, X.T.; Liang, Z.H.; Wang, J.H. Gut microbiota in obesity. World J. Gastroenterol. 2021, 27, 3837–3850. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Price, J.; Arze, C.; Ananthakrishnan, A.N.; Schirmer, M.; Avila-Pacheco, J.; Poon, T.W.; Andrews, E.; Ajami, N.J.; Bonham, K.S.; Brislawn, C.J.; et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 2019, 569, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Trakman, G.L.; Fehily, S.; Basnayake, C.; Hamilton, A.L.; Russell, E.; Wilson-O’Brien, A.; Kamm, M.A. Diet and gut microbiome in gastrointestinal disease. J. Gastroenterol. Hepatol. 2022, 37, 237–245. [Google Scholar] [CrossRef]
- Zorraquín-Peña, I.; Cueva, C.; Bartolomé, B.; Moreno-Arribas, M.V. Silver nanoparticles against foodborne bacteria. Effects at intestinal level and health limitations. Microorganisms 2020, 8, 132. [Google Scholar] [CrossRef]
- Su, Y.; Wu, D.; Xia, H.; Zhang, C.; Shi, J.; Wilkinson, K.J.; Xie, B. Metallic nanoparticles induced antibiotic resistance genes attenuation of leachate culturable microbiota: The combined roles of growth inhibition, ion dissolution and oxidative stress. Environ. Int. 2019, 128, 407–416. [Google Scholar] [CrossRef]
- Fröhlich, E.E.; Fröhlich, E. Cytotoxicity of nanoparticles contained in food on intestinal cells and the gut microbiota. Int. J. Mol. Sci. 2016, 17, 509. [Google Scholar] [CrossRef]
- Cattò, C.; Garuglieri, E.; Borruso, L.; Erba, D.; Casiraghi, M.C.; Cappitelli, F.; Villa, F.; Zecchin, S.; Zanchi, R. Impacts of dietary silver nanoparticles and probiotic administration on the microbiota of an in-vitro gut model. Environ. Pollut. 2019, 245, 754–763. [Google Scholar] [CrossRef]
- Das, P.; McDonald, J.A.K.; Petrof, E.O.; Allen-Vercoe, E.; Walker, V.K. Nanosilver-Mediated Change in Human Intestinal Microbiota. J. Nanomed. Nanotechnol. 2014, 5, 5. [Google Scholar] [CrossRef]
- Swartzwelter, B.J.; Fux, A.C.; Johnson, L.; Swart, E.; Hofer, S.; Hofstätter, N.; Geppert, M.; Italiani, P.; Boraschi, D.; Duschl, A.; et al. The impact of nanoparticles on innate immune activation by live bacteria. Int. J. Mol. Sci. 2020, 21, 9695. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Yu, S.; Shi, C.; Gu, J.; Shao, Y.; Chen, Q.; Li, Y.; Mezzenga, R. Amyloid-Polyphenol Hybrid Nanofilaments Mitigate Colitis and Regulate Gut Microbial Dysbiosis. ACS Nano 2020, 14, 2760–2776. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Shen, H.; Zhao, S.; Zhang, Y.; Xu, H.; Wang, L.; Di, B.; Xu, L.; Hu, C. Orally administered mesoporous silica capped with the cucurbit[8]uril complex to combat colitis and improve intestinal homeostasis by targeting the gut microbiota. Nanoscale 2020, 12, 15348–15363. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Du, X.; Tian, J.; Kang, X.; Li, Y.; Dai, W.; Li, D.; Zhang, S.; Li, C. Berberine-Loaded Carboxylmethyl Chitosan Nanoparticles Ameliorate DSS-Induced Colitis and Remodel Gut Microbiota in Mice. Front. Pharmacol. 2021, 12, 553. [Google Scholar] [CrossRef]
- Yang, M.; Yang, C.; Zhang, Y.; Yan, X.; Ma, Y.; Zhang, Y.; Cao, Y.; Xu, Q.; Tu, K.; Zhang, M. An oral pH-activated “nano-bomb” carrier combined with berberine by regulating gene silencing and gut microbiota for site-specific treatment of ulcerative colitis. Biomater. Sci. 2022, 10, 1053–1067. [Google Scholar] [CrossRef]
- Chandrarathna, H.P.S.U.; Liyanage, T.D.; Edirisinghe, S.L.; Dananjaya, S.H.S. Modified Pectin and Modified Pectin Nanoparticles Modulate the Gut Microbiota and Trigger Immune Responses in Mice. Mar. Drugs 2020, 18, 175. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, Z.; Wang, R.; Jin, J.; Zhu, Y.Z. Short-term oral administration of mesoporous silica nanoparticles potentially induced colon inflammation in rats through alteration of gut microbiota. Int. J. Nanomed. 2021, 16, 881–893. [Google Scholar] [CrossRef]
- Landsiedel, R.; Hahn, D.; Ossig, R.; Ritz, S.; Sauer, L.; Buesen, R.; Rehm, S.; Wohlleben, W.; Groeters, S.; Strauss, V.; et al. Gut microbiome and plasma metabolome changes in rats after oral gavage of nanoparticles: Sensitive indicators of possible adverse health effects. Part. Fibre Toxicol. 2022, 19, 21. [Google Scholar] [CrossRef]
- Bredeck, G.; Kämpfer, A.A.M.; Sofranko, A.; Wahle, T.; Lison, D.; Ambroise, J.; Stahlmecke, B.; Albrecht, C.; Schins, R.P.F. Effects of dietary exposure to the engineered nanomaterials CeO2, SiO2, Ag, and TiO2 on the murine gut microbiome. Nanotoxicology 2021, 15, 934–950. [Google Scholar] [CrossRef]
- Diao, J.; Xia, Y.; Jiang, X.; Qiu, J.; Cheng, S.; Su, J.; Duan, X.; Gao, M.; Qin, X.; Zhang, J.; et al. Silicon dioxide nanoparticles induced neurobehavioral impairments by disrupting microbiota–gut–brain axis. J. Nanobiotechnol. 2021, 19, 174. [Google Scholar] [CrossRef]
- Chaplin, A.; Gao, H.; Asase, C.; Rengasamy, P.; Park, B.; Skander, D.; Bebek, G.; Rajagopalan, S.; Maiseyeu, A. Systemically-delivered biodegradable PLGA alters gut microbiota and induces transcriptomic reprogramming in the liver in an obesity mouse model. Sci. Rep. 2020, 10, 13786. [Google Scholar] [CrossRef] [PubMed]
- Halwani, A.A. Development of Pharmaceutical Nanomedicines: From the Bench to the Market. Pharmaceutics 2022, 14, 106. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K. Liposomes for enhanced bioavailability of water-insoluble drugs: In vivo evidence and recent approaches. Pharmaceutics 2020, 12, 264. [Google Scholar] [CrossRef] [PubMed]
- Salah, E.; Abouelfetouh, M.M.; Pan, Y.; Chen, D.; Xie, S. Solid lipid nanoparticles for enhanced oral absorption: A review. Colloids Surf. B Biointerfaces 2020, 196, 111305. [Google Scholar] [CrossRef] [PubMed]
- Anton, N.; Vandamme, T.F. Nano-emulsions and micro-emulsions: Clarifications of the critical differences. Pharm. Res. 2011, 28, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Shan, W.; Zhang, Z.; Huang, Y. Engineering nanomaterials to overcome the mucosal barrier by modulating surface properties. Adv. Drug Deliv. Rev. 2018, 124, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Younes, M.; Aquilina, G.; Castle, L.; Engel, K.H.; Fowler, P.; Frutos Fernandez, M.J.; Fürst, P.; Gundert-Remy, U.; Gürtler, R.; Husøy, T.; et al. Safety assessment of titanium dioxide (E171) as a food additive. EFSA J. 2021, 19, e06585. [Google Scholar] [CrossRef]
- Medina-Reyes, E.I.; Rodríguez-Ibarra, C.; Déciga-Alcaraz, A.; Díaz-Urbina, D.; Chirino, Y.I.; Pedraza-Chaverri, J. Food additives containing nanoparticles induce gastrotoxicity, hepatotoxicity and alterations in animal behavior: The unknown role of oxidative stress. Food Chem. Toxicol. 2020, 146, 111814. [Google Scholar] [CrossRef]
- Zhao, R.; Xiang, J.; Wang, B.; Chen, L.; Tan, S. Recent Advances in the Development of Noble Metal NPs for Cancer Therapy. Pharmacol. Ther. 2022, 2022, 2444516. [Google Scholar] [CrossRef]
- Mubeen, B.; Ansar, A.N.; Rasool, R.; Ullah, I.; Imam, S.S.; Alshehri, S.; Ghoneim, M.M.; Alzarea, S.I.; Nadeem, M.S.; Kazmi, I. Nanotechnology as a Novel Approach in Combating Microbes Providing an Alternative to Antibiotics. Antibiotics 2021, 10, 1473. [Google Scholar] [CrossRef]
- Madni, A.; Rehman, S.; Sultan, H.; Khan, M.M.; Ahmad, F.; Raza, M.R.; Rai, N.; Parveen, F. Mechanistic Approaches of Internalization, Subcellular Trafficking, and Cytotoxicity of Nanoparticles for Targeting the Small Intestine. AAPS PharmSciTech 2021, 22, 3. [Google Scholar] [CrossRef] [PubMed]
- Ways, T.M.M.; Lau, W.M.; Khutoryanskiy, V.V. Chitosan and its derivatives for application in mucoadhesive drug delivery systems. Polymers 2018, 10, 267. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Lin, L.Y.; Gao, M.Y.; Fang, Y.P. Mesoporous silica nanoparticles synthesized from liquid crystal display manufacturing extracts as a potential candidate for a drug delivery carrier: Evaluation of their safety and biocompatibility. Int. J. Nanomed. 2013, 8, 3833–3842. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Zhao, Q.; Hu, Y.; Sun, L.; Bai, L.; Jiang, T.; Wang, S. Ordered nanoporous silica as carriers for improved delivery of water insoluble drugs: A comparative study between three dimensional and two dimensional macroporous silica. Int. J. Nanomed. 2013, 8, 4015–4031. [Google Scholar] [CrossRef]
- González-Alvarez, M.; Coll, C.; Gonzalez-Alvarez, I.; Giménez, C.; Aznar, E.; Martínez-Bisbal, M.C.; Lozoya-Agulló, I.; Bermejo, M.; Martínez-Máñez, R.; Sancenón, F. Gated Mesoporous Silica Nanocarriers for a “two-Step” Targeted System to Colonic Tissue. Mol. Pharm. 2017, 14, 4442–4453. [Google Scholar] [CrossRef]
- Patra, C.N.; Priya, R.; Swain, S.; Kumar Jena, G.; Panigrahi, K.C.; Ghose, D. Pharmaceutical significance of Eudragit: A review. Futur. J. Pharm. Sci. 2017, 3, 33–45. [Google Scholar] [CrossRef]
- Szekalska, M.; Puciłowska, A.; Szymańska, E.; Ciosek, P.; Winnicka, K. Alginate: Current Use and Future Perspectives in Pharmaceutical and Biomedical Applications. Int. J. Polym. Sci. 2016, 2016, 7697031. [Google Scholar] [CrossRef]
- Okolie, C.L.; Mason, B.; Mohan, A.; Pitts, N.; Udenigwe, C.C. Extraction technology impacts on the structure-function relationship between sodium alginate extracts and their in vitro prebiotic activity. Food Biosci. 2020, 37, 100672. [Google Scholar] [CrossRef]
- Gómez-Mascaraque, L.G.; Martínez-Sanz, M.; Hogan, S.A.; López-Rubio, A.; Brodkorb, A. Nano- and microstructural evolution of alginate beads in simulated gastrointestinal fluids. Impact of M/G ratio, molecular weight and pH. Carbohydr. Polym. 2019, 223, 115121. [Google Scholar] [CrossRef]
- Qin, X.S.; Luo, Z.G.; Li, X.L. An enhanced pH-sensitive carrier based on alginate-Ca-EDTA in a set-type W1/O/W2 double emulsion model stabilized with WPI-EGCG covalent conjugates for probiotics colon-targeted release. Food Hydrocoll. 2021, 113, 106460. [Google Scholar] [CrossRef]
- Nilsen-Nygaard, J.; Strand, S.P.; Vårum, K.M.; Draget, K.I.; Nordgård, C.T. Chitosan: Gels and interfacial properties. Polymers 2015, 7, 552–579. [Google Scholar] [CrossRef]
- Vllasaliu, D.; Exposito-Harris, R.; Heras, A.; Casettari, L.; Garnett, M.; Illum, L.; Stolnik, S. Tight junction modulation by chitosan nanoparticles: Comparison with chitosan solution. Int. J. Pharm. 2010, 400, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, M.A.; Syeda, J.T.M.; Wasan, K.M.; Wasan, E.K. An overview of chitosan nanoparticles and its application in non-parenteral drug delivery. Pharmaceutics 2017, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Lin, Y.S.; Wu, S.J.; Mi, F.L. Mutlifunctional nanoparticles prepared from arginine-modified chitosan and thiolated fucoidan for oral delivery of hydrophobic and hydrophilic drugs. Carbohydr. Polym. 2018, 193, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Faralli, A.; Shekarforoush, E.; Ajalloueian, F.; Mendes, A.C.; Chronakis, I.S. In vitro permeability enhancement of curcumin across Caco-2 cells monolayers using electrospun xanthan-chitosan nanofibers. Carbohydr. Polym. 2019, 206, 38–47. [Google Scholar] [CrossRef]
- Yeh, T.H.; Hsu, L.W.; Tseng, M.T.; Lee, P.L.; Sonjae, K.; Ho, Y.C.; Sung, H.W. Mechanism and consequence of chitosan-mediated reversible epithelial tight junction opening. Biomaterials 2011, 32, 6164–6173. [Google Scholar] [CrossRef]
- Hsu, L.W.; Ho, Y.C.; Chuang, E.Y.; Chen, C.T.; Juang, J.H.; Su, F.Y.; Hwang, S.M.; Sung, H.W. Effects of pH on molecular mechanisms of chitosan-integrin interactions and resulting tight-junction disruptions. Biomaterials 2013, 34, 784–793. [Google Scholar] [CrossRef]
- Pritchard, K.; Lansley, A.B.; Martin, G.P.; Helliwell, M.; Marriott, C.; Benedetti, L.M. Evaluation of the bioadhesive properties of hyaluronan derivatives: Detachment weight and mucociliary transport rate studies. Int. J. Pharm. 1996, 129, 137–145. [Google Scholar] [CrossRef]
- Sandri, G.; Rossi, S.; Ferrari, F.; Bonferoni, M.C.; Zerrouk, N.; Caramella, C. Mucoadhesive and penetration enhancement properties of three grades of hyaluronic acid using porcine buccal and vaginal tissue, Caco-2 cell lines, and rat jejunum. J. Pharm. Pharmacol. 2004, 56, 1083–1090. [Google Scholar] [CrossRef]
- Xu, Q.; Ensign, L.M.; Boylan, N.J.; Schön, A.; Gong, X.; Yang, J.C.; Lamb, N.W.; Cai, S.; Yu, T.; Freire, E.; et al. Impact of Surface Polyethylene Glycol (PEG) Density on Biodegradable Nanoparticle Transport in Mucus ex Vivo and Distribution in Vivo. ACS Nano 2015, 9, 9217–9227. [Google Scholar] [CrossRef]
- Inchaurraga, L.; Martín-Arbella, N.; Zabaleta, V.; Quincoces, G.; Peñuelas, I.; Irache, J.M. In vivo study of the mucus-permeating properties of PEG-coated nanoparticles following oral administration. Eur. J. Pharm. Biopharm. 2015, 97, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhang, J.; Zhu, X.; Shan, W.; Li, L.; Zhong, J.; Zhang, Z.; Huang, Y. Efficient mucus permeation and tight junction opening by dissociable “mucus-inert” agent coated trimethyl chitosan nanoparticles for oral insulin delivery. J. Control. Release 2016, 222, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Kosińska, A.; Andlauer, W. Modulation of tight junction integrity by food components. Food Res. Int. 2013, 54, 951–960. [Google Scholar] [CrossRef]
- Haasbroek, A.; Willers, C.; Glyn, M.; Du Plessis, L.; Hamman, J. Intestinal drug absorption enhancement by Aloe vera gel and whole leaf extract: In vitro investigations into the mechanisms of action. Pharmaceutics 2019, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Butt, A.M.; Mohd Amin, M.C.I. Molecular Evaluation of Oral Immunogenicity of Hepatitis B Antigen Delivered by Hydrogel Microparticles. Mol. Pharm. 2019, 16, 3853–3872. [Google Scholar] [CrossRef] [PubMed]
- Di Pierro, M.; Lu, R.; Uzzau, S.; Wang, W.; Margaretten, K.; Pazzani, C.; Maimone, F.; Fasano, A. Zonula occludens toxin structure-function analysis: Identification of the fragment biologically active on tight junctions and of the zonulin receptor binding domain. J. Biol. Chem. 2001, 276, 19160–19165. [Google Scholar] [CrossRef]
- Lee, J.Y.J.H.; Sahu, A.; Choi, W.I.; Lee, J.Y.J.H.; Tae, G. ZOT-derived peptide and chitosan functionalized nanocarrier for oral delivery of protein drug. Biomaterials 2016, 103, 160–169. [Google Scholar] [CrossRef]
- Sonaje, K.; Lin, K.J.; Tseng, M.T.; Wey, S.P.; Su, F.Y.; Chuang, E.Y.; Hsu, C.W.; Chen, C.T.; Sung, H.W. Effects of chitosan-nanoparticle-mediated tight junction opening on the oral absorption of endotoxins. Biomaterials 2011, 32, 8712–8721. [Google Scholar] [CrossRef]
- Zhang, N.; Ping, Q.N.; Huang, G.H.; Xu, W.F. Investigation of lectin-modified insulin liposomes as carriers for oral administration. Int. J. Pharm. 2005, 294, 247–259. [Google Scholar] [CrossRef]
- Jia, Z.; Wignall, A.; Prestidge, C.; Thierry, B. An ex vivo investigation of the intestinal uptake and translocation of nanoparticles targeted to Peyer’s patches microfold cells. Int. J. Pharm. 2021, 594, 120167. [Google Scholar] [CrossRef]
- Garinot, M.; Fiévez, V.; Pourcelle, V.; Stoffelbach, F.; des Rieux, A.; Plapied, L.; Theate, I.; Freichels, H.; Jérôme, C.; Marchand-Brynaert, J.; et al. PEGylated PLGA-based nanoparticles targeting M cells for oral vaccination. J. Control. Release 2007, 120, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Berillo, D.; Yeskendir, A.; Zharkinbekov, Z.; Raziyeva, K.; Saparov, A. Peptide-Based Drug Delivery Systems. Medicina 2021, 57, 1209. [Google Scholar] [CrossRef] [PubMed]
- Hua, S. Advances in Oral Drug Delivery for Regional Targeting in the Gastrointestinal Tract—Influence of Physiological, Pathophysiological and Pharmaceutical Factors. Front. Pharmacol. 2020, 11, 524. [Google Scholar] [CrossRef]
- Tian, H.; He, Z.; Sun, C.; Yang, C.; Zhao, P.; Liu, L.; Leong, K.W.; Mao, H.Q.; Liu, Z.; Chen, Y. Uniform Core–Shell Nanoparticles with Thiolated Hyaluronic Acid Coating to Enhance Oral Delivery of Insulin. Adv. Healthc. Mater. 2018, 7, 285. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Viennois, E.; Chen, Q.; Wang, L.; Han, M.K.; Zhang, Y.; Zhang, Z.; Kang, Y.; Wan, Y.; Merlin, D. Silencing of Intestinal Glycoprotein CD98 by Orally Targeted Nanoparticles Enhances Chemosensitization of Colon Cancer. ACS Nano 2018, 12, 5253–5265. [Google Scholar] [CrossRef]
- Liu, P.; Gao, C.; Chen, H.; Vong, C.T.; Wu, X.; Tang, X.; Wang, S.; Wang, Y. Receptor-mediated targeted drug delivery systems for treatment of inflammatory bowel disease: Opportunities and emerging strategies. Acta Pharm. Sin. B 2021, 11, 2798–2818. [Google Scholar] [CrossRef]
- Liu, W.; Zhu, Y.; Wang, F.; Li, X.; Liu, X.; Pang, J.; Pan, W. Galactosylated chitosanfunctionalized mesoporous silica nanoparticles for efficient colon cancer cell-targeted drug delivery. R. Soc. Open Sci. 2018, 5, 181027. [Google Scholar] [CrossRef]
- Xi, Z.; Ahmad, E.; Zhang, W.; Li, J.; Wang, A.; Faridoon; Wang, N.; Zhu, C.; Huang, W.; Xu, L.; et al. Dual-modified nanoparticles overcome sequential absorption barriers for oral insulin delivery. J. Control. Release 2022, 342, 1–13. [Google Scholar] [CrossRef]
- Álvarez-González, B.; Rozalen, M.; Fernández-Perales, M.; Álvarez, M.A.; Sánchez-Polo, M. Methotrexate Gold Nanocarriers: Loading and Release Study: Its Activity in Colon and Lung Cancer Cells. Molecules 2020, 25, 6049. [Google Scholar] [CrossRef]
- Yong, J.M.; Mantaj, J.; Cheng, Y.; Vllasaliu, D. Delivery of nanoparticles across the intestinal epithelium via the transferrin transport pathway. Pharmaceutics 2019, 11, 298. [Google Scholar] [CrossRef]
- Zhang, W.; Michalowski, C.B.; Beloqui, A. Oral Delivery of Biologics in Inflammatory Bowel Disease Treatment. Front. Bioeng. Biotechnol. 2021, 9, 675194. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, Y.; Yu, S.; Yang, Z.; He, C.; Chen, X. Dual Stimuli-Responsive Nanoparticle-Incorporated Hydrogels as an Oral Insulin Carrier for Intestine-Targeted Delivery and Enhanced Paracellular Permeation. ACS Biomater. Sci. Eng. 2018, 4, 2889–2902. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, C.; Nilsen, J.; Grevys, A.; Nunes, R.; Andersen, J.T.; Sarmento, B. Engineered albumin-functionalized nanoparticles for improved FcRn binding enhance oral delivery of insulin. J. Control. Release 2020, 327, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Han, S.; Chen, Z.; Zhao, L.; Wang, C.; Guo, Q.; Li, Y.; Sun, Y. Layered double hydroxide modified with deoxycholic and hyaluronic acids for efficient oral insulin absorption. Int. J. Nanomed. 2021, 16, 7861–7873. [Google Scholar] [CrossRef]
- Urimi, D.; Agrawal, A.K.; Kushwah, V.; Jain, S. Polyglutamic Acid Functionalization of Chitosan Nanoparticles Enhances the Therapeutic Efficacy of Insulin Following Oral Administration. AAPS PharmSciTech 2019, 20, 131. [Google Scholar] [CrossRef]
- Li, R.; Laurent, F.; Taverner, A.; Mackay, J.; De Bank, P.A.; Mrsny, R.J. Intestinal transcytosis of a protein cargo and nanoparticles mediated by a non-toxic form of Pseudomonas aeruginosa exotoxin A. Pharmaceutics 2021, 13, 1171. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, D.; Ba, S.; Zhu, J.; Zhang, J.; Hong, W.; Zhao, X.; Hu, H.; Qiao, M. Poly(l-histidine) based triblock copolymers: PH induced reassembly of copolymer micelles and mechanism underlying endolysosomal escape for intracellular delivery. Biomacromolecules 2014, 15, 4032–4045. [Google Scholar] [CrossRef]
- Li, L.; Yang, L.; Li, M.; Zhang, L. A cell-penetrating peptide mediated chitosan nanocarriers for improving intestinal insulin delivery. Carbohydr. Polym. 2017, 174, 182–189. [Google Scholar] [CrossRef]
- Wu, J.Z.; Williams, G.R.; Li, H.Y.; Wang, D.; Wu, H.; De Li, S.; Zhu, L.M. Glucose- and temperature-sensitive nanoparticles for insulin delivery. Int. J. Nanomed. 2017, 12, 4037–4057. [Google Scholar] [CrossRef]
- Alfatama, M.; Lim, L.Y.; Wong, T.W. Alginate-C18 Conjugate Nanoparticles Loaded in Tripolyphosphate-Cross-Linked Chitosan-Oleic Acid Conjugate-Coated Calcium Alginate Beads as Oral Insulin Carrier. Mol. Pharm. 2018, 15, 3369–3382. [Google Scholar] [CrossRef]
- Czuba, E.; Diop, M.; Mura, C.; Schaschkow, A.; Langlois, A.; Bietiger, W.; Neidl, R.; Virciglio, A.; Auberval, N.; Julien-David, D.; et al. Oral insulin delivery, the challenge to increase insulin bioavailability: Influence of surface charge in nanoparticle system. Int. J. Pharm. 2018, 542, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Xia, D.; Zhu, Q.; Li, X.; He, S.; Zhu, C.; Guo, S.; Hovgaard, L.; Yang, M.; Gan, Y. Functional nanoparticles exploit the bile acid pathway to overcome multiple barriers of the intestinal epithelium for oral insulin delivery. Biomaterials 2018, 151, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Zheng, Y.; Wang, Y.; Hu, Y.; Shi, J.; Liu, Q.; Zhang, H.; Zhang, Z. Self-Regulated Carboxyphenylboronic Acid-Modified Mesoporous Silica Nanoparticles with “touch Switch” Releasing Property for Insulin Delivery. ACS Appl. Mater. Interfaces 2018, 10, 21927–21938. [Google Scholar] [CrossRef]
- Jamshidi, M.; Ziamajidi, N.; Khodadadi, I.; Dehghan, A.; Kalantarian, G.; Abbasalipourkabir, R. The effect of insulin-loaded trimethylchitosan nanoparticles on rats with diabetes type I. Biomed. Pharmacother. 2018, 97, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Ji, N.; Hong, Y.; Gu, Z.; Cheng, L.; Li, Z.; Li, C. Preparation and Characterization of Insulin-Loaded Zein/Carboxymethylated Short-Chain Amylose Complex Nanoparticles. J. Agric. Food Chem. 2018, 66, 9335–9343. [Google Scholar] [CrossRef]
- Song, M.; Wang, H.; Chen, K.; Zhang, S.; Yu, L.; Elshazly, E.H.; Ke, L.; Gong, R. Oral insulin delivery by carboxymethyl-β-cyclodextrin-grafted chitosan nanoparticles for improving diabetic treatment. Artif. Cells Nanomed. Biotechnol. 2018, 46, S774–S782. [Google Scholar] [CrossRef]
- Wang, W.; Liao, L.; Zhang, X.; Lei, F.; Zhang, Y.; Liu, G.; Xie, W. An intelligent nanoscale insulin delivery system. Molecules 2018, 23, 2945. [Google Scholar] [CrossRef]
- Xu, Y.; Zheng, Y.; Wu, L.; Zhu, X.; Zhang, Z.; Huang, Y. Novel Solid Lipid Nanoparticle with Endosomal Escape Function for Oral Delivery of Insulin. ACS Appl. Mater. Interfaces 2018, 10, 9315–9324. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, L.; Ban, Q.; Li, J.; Li, C.H.; Guan, Y.Q. Preparation and characterization of hydroxyapatite nanoparticles carrying insulin and gallic acid for insulin oral delivery. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 353–364. [Google Scholar] [CrossRef]
- Zhang, L.; Qin, H.; Li, J.; Qiu, J.N.; Huang, J.M.; Li, M.C.; Guan, Y.Q. Preparation and characterization of layer-by-layer hypoglycemic nanoparticles with pH-sensitivity for oral insulin delivery. J. Mater. Chem. B 2018, 6, 7451–7461. [Google Scholar] [CrossRef]
- Alsulays, B.B.; Anwer, M.K.; Soliman, G.A.; Alshehri, S.M.; Khafagy, E.S. Impact of penetratin stereochemistry on the oral bioavailability of insulin-loaded solid lipid nanoparticles. Int. J. Nanomed. 2019, 14, 9127–9138. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Ouyang, T.; Peng, T.; Zhang, X.; Xie, B.; Yang, X.; Liang, D.; Zhong, H. Enhanced oral absorption of insulin using colon-specific nanoparticles co-modified with amphiphilic chitosan derivatives and cell-penetrating peptides. Biomater. Sci. 2019, 7, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.B.; Tang, T.T.; Li, Y.J.; Wu, J.Y.; Wang, J.M.; Liu, X.Y.; Xiang, D.X. Phospholipid complex based nanoemulsion system for oral insulin delivery: Preparation, in vitro, and in vivo evaluations. Int. J. Nanomed. 2019, 14, 3055–3067. [Google Scholar] [CrossRef]
- Jamwal, S.; Ram, B.; Ranote, S.; Dharela, R.; Chauhan, G.S. New glucose oxidase-immobilized stimuli-responsive dextran nanoparticles for insulin delivery. Int. J. Biol. Macromol. 2019, 123, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Ji, N.; Hong, Y.; Gu, Z.; Cheng, L.; Li, Z.; Li, C. Chitosan coating of zein-carboxymethylated short-chain amylose nanocomposites improves oral bioavailability of insulin in vitro and in vivo. J. Control. Release 2019, 313, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mohammadpour, F.; Hadizadeh, F.; Tafaghodi, M.; Sadri, K.; Mohammadpour, A.H.; Kalani, M.R.; Gholami, L.; Mahmoudi, A.; Chamani, J. Preparation, in vitro and in vivo evaluation of PLGA/Chitosan based nano-complex as a novel insulin delivery formulation. Int. J. Pharm. 2019, 572, 118710. [Google Scholar] [CrossRef]
- Muntoni, E.; Marini, E.; Ahmadi, N.; Milla, P.; Ghè, C.; Bargoni, A.; Capucchio, M.T.; Biasibetti, E.; Battaglia, L. Lipid nanoparticles as vehicles for oral delivery of insulin and insulin analogs: Preliminary ex vivo and in vivo studies. Acta Diabetol. 2019, 56, 1283–1292. [Google Scholar] [CrossRef]
- Mudassir, J.; Darwis, Y.; Muhamad, S.; Khan, A.A. Self-assembled insulin and nanogels polyelectrolyte complex (Ins/NGs-PEC) for oral insulin delivery: Characterization, lyophilization and in-vivo evaluation. Int. J. Nanomed. 2019, 14, 4895–4909. [Google Scholar] [CrossRef]
- Tsai, L.C.; Chen, C.H.; Lin, C.W.; Ho, Y.C.; Mi, F.L. Development of mutlifunctional nanoparticles self-assembled from trimethyl chitosan and fucoidan for enhanced oral delivery of insulin. Int. J. Biol. Macromol. 2019, 126, 141–150. [Google Scholar] [CrossRef]
- Bai, Y.; Zhou, R.; Wu, L.; Zheng, Y.; Liu, X.; Wu, R.; Li, X.; Huang, Y. Nanoparticles with surface features of dendritic oligopeptides as potential oral drug delivery systems. J. Mater. Chem. B 2020, 8, 2636–2649. [Google Scholar] [CrossRef]
- Chai, Z.; Dong, H.; Sun, X.; Fan, Y.; Wang, Y.; Huang, F. Development of glucose oxidase-immobilized alginate nanoparticles for enhanced glucose-triggered insulin delivery in diabetic mice. Int. J. Biol. Macromol. 2020, 159, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Han, S.; Yang, X.; Xu, L.; Qi, H.; Hao, G.; Cao, J.; Liang, Y.; Ma, Q.; Zhang, G.; et al. Overcoming multiple absorption barrier for insulin oral delivery using multifunctional nanoparticles based on chitosan derivatives and hyaluronic acid. Int. J. Nanomed. 2020, 15, 4877–4898. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Zhang, X.; Qin, L.; Huo, Y.; Cui, Z.; Liu, C.; Sun, Y.; Guan, J.; Mao, S. Design of self-polymerized insulin loaded poly(n-butylcyanoacrylate) nanoparticles for tunable oral delivery. J. Control. Release 2020, 321, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wang, Q.; Liu, G.; Feng, Y.; Zhou, W. Cholesterol moieties as building blocks for assembling nanoparticles to achieve effective oral delivery of insulin. Biomater. Sci. 2020, 8, 3979–3993. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Lu, Y.; Xie, J.; Zhang, E.; Zhu, H.; Du, H.; Wang, K.; Song, B.; Yang, C.; Shi, Y.; et al. Zwitterionic micelles efficiently deliver oral insulin without opening tight junctions. Nat. Nanotechnol. 2020, 15, 605–614. [Google Scholar] [CrossRef]
- Jana, B.A.; Shinde, U.; Wadhwani, A. Synthetic enzyme-based nanoparticles act as smart catalyst for glucose responsive release of insulin. J. Biotechnol. 2020, 324, 1–6. [Google Scholar] [CrossRef]
- Mumuni, M.A.; Kenechukwu, F.C.; Ofokansi, K.C.; Attama, A.A.; Díaz, D.D. Insulin-loaded mucoadhesive nanoparticles based on mucin-chitosan complexes for oral delivery and diabetes treatment. Carbohydr. Polym. 2020, 229, 115506. [Google Scholar] [CrossRef]
- Sladek, S.; McCartney, F.; Eskander, M.; Dunne, D.J.; Santos-Martinez, M.J.; Benetti, F.; Tajber, L.; Brayden, D.J. An enteric-coated polyelectrolyte nanocomplex delivers insulin in rat intestinal instillations when combined with a permeation enhancer. Pharmaceutics 2020, 12, 259. [Google Scholar] [CrossRef]
- Sudhakar, S.; Chandran, S.V.; Selvamurugan, N.; Nazeer, R.A. Biodistribution and pharmacokinetics of thiolated chitosan nanoparticles for oral delivery of insulin in vivo. Int. J. Biol. Macromol. 2020, 150, 281–288. [Google Scholar] [CrossRef]
- Tan, X.; Yin, N.; Liu, Z.; Sun, R.; Gou, J.; Yin, T.; Zhang, Y.; He, H.; Tang, X. Hydrophilic and Electroneutral Nanoparticles to Overcome Mucus Trapping and Enhance Oral Delivery of Insulin. Mol. Pharm. 2020, 17, 3177–3191. [Google Scholar] [CrossRef]
- Wang, T.; Shen, L.; Zhang, Y.; Li, H.; Wang, Y.; Quan, D. “Oil-Soluble” Reversed Lipid Nanoparticles for Oral Insulin Delivery. J. Nanobiotechnol. 2020, 18, 98. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Deng, H.; Zhang, Y.; Wu, P.; He, B.; Dai, W.; Zhang, H.; Zhang, Q.; Zhao, R.; Wang, X. Thiolated Nanoparticles Overcome the Mucus Barrier and Epithelial Barrier for Oral Delivery of Insulin. Mol. Pharm. 2020, 17, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wu, H.; Long, R.; Wang, S.; Huang, H.; Xia, Y.; Wang, P.; Lei, Y.; Cai, Y.; Cai, D.; et al. Oral delivery of insulin with intelligent glucose-responsive switch for blood glucose regulation. J. Nanobiotechnol. 2020, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Liu, L.; Cao, Y.; Yu, S.; He, C.; Chen, X. A Nanocomposite Vehicle Based on Metal-Organic Framework Nanoparticle Incorporated Biodegradable Microspheres for Enhanced Oral Insulin Delivery. ACS Appl. Mater. Interfaces 2020, 12, 22581–22592. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Qian, K.; Yao, P. Insulin- And cholic acid-loaded zein/casein-dextran nanoparticles enhance the oral absorption and hypoglycemic effect of insulin. J. Mater. Chem. B 2021, 9, 6234–6245. [Google Scholar] [CrossRef] [PubMed]
- Benyettou, F.; Kaddour, N.; Prakasam, T.; Das, G.; Sharma, S.K.; Thomas, S.A.; Bekhti-Sari, F.; Whelan, J.; Alkhalifah, M.A.; Khair, M.; et al. In vivo oral insulin delivery via covalent organic frameworks. Chem. Sci. 2021, 12, 6037–6047. [Google Scholar] [CrossRef]
- Cui, Z.; Qin, L.; Guo, S.; Cheng, H.; Zhang, X.; Guan, J.; Mao, S. Design of biotin decorated enterocyte targeting muco-inert nanocomplexes for enhanced oral insulin delivery. Carbohydr. Polym. 2021, 261, 117873. [Google Scholar] [CrossRef]
- Kim, W.J.; Kwon, Y.J.; Cho, C.H.; Ye, S.K.; Kim, K.O. Insulin smart drug delivery nanoparticles of aminophenylboronic acid–POSS molecule at neutral pH. Sci. Rep. 2021, 11, 21894. [Google Scholar] [CrossRef]
- Li, H.; Zhou, R.; He, J.; Zhang, M.; Liu, J.; Sun, X.; Ni, P. Glucose-Sensitive Core-Cross-Linked Nanoparticles Constructed with Polyphosphoester Diblock Copolymer for Controlling Insulin Delivery. Bioconjug. Chem. 2021, 32, 2095–2107. [Google Scholar] [CrossRef]
- Li, J.; Wu, H.; Jiang, K.; Liu, Y.; Yang, L.; Park, H.J. Alginate Calcium Microbeads Containing Chitosan Nanoparticles for Controlled Insulin Release. Appl. Biochem. Biotechnol. 2021, 193, 463–478. [Google Scholar] [CrossRef]
- Liu, X.; Wu, R.; Li, Y.; Wang, L.; Zhou, R.; Li, L.; Xiang, Y.; Wu, J.; Xing, L.; Huang, Y. Angiopep-2-functionalized nanoparticles enhance transport of protein drugs across intestinal epithelia by self-regulation of targeted receptors. Biomater. Sci. 2021, 9, 2903–2916. [Google Scholar] [CrossRef] [PubMed]
- Qin, T.; Yan, L.; Wang, X.; Lin, S.; Zeng, Q. Glucose-Responsive Polyelectrolyte Complexes Based on Dendritic Mesoporous Silica for Oral Insulin Delivery. AAPS PharmSciTech 2021, 22, 226. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.; Liu, X.; Li, Y.; Tan, X.; Zhou, H.; Bai, X.; Yang, X.; Liu, W. Bioinspired zwitterionic polyphosphoester modified porous silicon nanoparticles for efficient oral insulin delivery. Biomater. Sci. 2021, 9, 685–699. [Google Scholar] [CrossRef] [PubMed]
- Volpatti, L.R.; Burns, D.M.; Basu, A.; Langer, R.; Anderson, D.G. Engineered insulin-polycation complexes for glucose-responsive delivery with high insulin loading. J. Control. Release 2021, 338, 71–79. [Google Scholar] [CrossRef]
- Wang, W.; Yu, C.; Zhang, F.; Li, Y.; Zhang, B.; Huang, J.; Zhang, Z.; Jin, L. Improved oral delivery of insulin by PLGA nanoparticles coated with 5β-cholanic acid conjugated glycol chitosan. Biomed. Mater. 2021, 16, 064103. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiong, M.; Ni, X.; Wang, J.; Rong, H.; Su, Y.; Yu, S.; Mohammad, I.S.; Leung, S.S.Y.; Hu, H. Virus-Mimicking Mesoporous Silica Nanoparticles with an Electrically Neutral and Hydrophilic Surface to Improve the Oral Absorption of Insulin by Breaking through Dual Barriers of the Mucus Layer and the Intestinal Epithelium. ACS Appl. Mater. Interfaces 2021, 13, 18077–18088. [Google Scholar] [CrossRef]
- Fu, Y.; Sun, Y.; Chen, M.; Xing, W.; Xu, Y.; Qian, X.; Zhu, W. Glycopolymer Nanoparticles with On-Demand Glucose-Responsive Insulin Delivery and Low-Hypoglycemia Risks for Type 1 Diabetic Treatment. Biomacromolecules 2022, 23, 1251–1258. [Google Scholar] [CrossRef]
- Li, J.; Qiang, H.; Yang, W.; Xu, Y.; Feng, T.; Cai, H.; Wang, S.; Liu, Z.; Zhang, Z.; Zhang, J. Oral insulin delivery by epithelium microenvironment-adaptive nanoparticles. J. Control. Release 2022, 341, 31–43. [Google Scholar] [CrossRef]
- Martins, J.P.; Figueiredo, P.; Wang, S.; Espo, E.; Celi, E.; Martins, B.; Kemell, M.; Moslova, K.; Mäkilä, E.; Salonen, J.; et al. Neonatal Fc receptor-targeted lignin-encapsulated porous silicon nanoparticles for enhanced cellular interactions and insulin permeation across the intestinal epithelium. Bioact. Mater. 2022, 9, 299–315. [Google Scholar] [CrossRef]
- Reboredo, C.; González-Navarro, C.J.; Martínez-López, A.L.; Martínez-Ohárriz, C.; Sarmento, B.; Irache, J.M. Zein-Based Nanoparticles as Oral Carriers for Insulin Delivery. Pharmaceutics 2022, 14, 39. [Google Scholar] [CrossRef]
- Rohra, N.; Gaikwad, G.; Dandekar, P.; Jain, R. Microfluidic Synthesis of a Bioactive Metal–Organic Framework for Glucose-Responsive Insulin Delivery. ACS Appl. Mater. Interfaces 2022, 14, 8251–8265. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Huang, J.; Jiang, S.; He, J.; Wang, Z.; Qin, H.; Guan, Y.Q. Glucose sensitive konjac glucomannan/concanavalin A nanoparticles as oral insulin delivery system. Int. J. Biol. Macromol. 2022, 202, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Pandita, D.; Kumar, S.; Lather, V. Hybrid poly(lactic-co-glycolic acid) nanoparticles: Design and delivery prospectives. Drug Discov. Today 2015, 20, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Yan, X.; Duan, L.; Cui, Y.; Yang, Y.; Li, J. Glucose-sensitive microcapsules from glutaraldehyde cross-linked hemoglobin and glucose oxidase. Biomacromolecules 2009, 10, 1212–1216. [Google Scholar] [CrossRef]
- Ma, Q.; Zhao, X.; Shi, A.; Wu, J. Bioresponsive functional phenylboronic acid-based delivery system as an emerging platform for diabetic therapy. Int. J. Nanomed. 2021, 16, 297–314. [Google Scholar] [CrossRef]
- Naeem, M.; Awan, U.A.; Subhan, F.; Cao, J.; Hlaing, S.P.; Lee, J.; Im, E.; Jung, Y.; Yoo, J.W. Advances in colon-targeted nano-drug delivery systems: Challenges and solutions. Arch. Pharm. Res. 2020, 43, 153–169. [Google Scholar] [CrossRef]
- Feng, Z.; Jiao, L.; Wu, Z.; Xu, J.; Gu, P.; Xu, S.; Liu, Z.; Hu, Y.; Liu, J.; Wu, Y.; et al. A Novel Nanomedicine Ameliorates Acute Inflammatory Bowel Disease by Regulating Macrophages and T-Cells. Mol. Pharm. 2021, 18, 3484–3495. [Google Scholar] [CrossRef]
- Nedelcu, A.; Mosteanu, O.; Pop, T.; Mocan, T.; Mocan, L. Recent advances in nanoparticle-mediated treatment of inflammatory bowel diseases. Appl. Sci. 2021, 11, 438. [Google Scholar] [CrossRef]
- Hartwig, O.; Shetab Boushehri, M.A.; Shalaby, K.S.; Loretz, B.; Lamprecht, A.; Lehr, C.M. Drug delivery to the inflamed intestinal mucosa—Targeting technologies and human cell culture models for better therapies of IBD. Adv. Drug Deliv. Rev. 2021, 175, 113828. [Google Scholar] [CrossRef]
- Naeem, M.; Lee, J.; Oshi, M.A.; Cao, J.; Hlaing, S.P.; Im, E.; Jung, Y.; Yoo, J.W. Colitis-targeted hybrid nanoparticles-in-microparticles system for the treatment of ulcerative colitis. Acta Biomater. 2020, 116, 368–382. [Google Scholar] [CrossRef]
- Zhou, H.; Ikeuchi-Takahashi, Y.; Hattori, Y.; Onishi, H. Nanogels of a succinylated glycol chitosan-succinyl prednisolone conjugate: Release behavior, gastrointestinal distribution, and systemic absorption. Int. J. Mol. Sci. 2020, 21, 2376. [Google Scholar] [CrossRef] [PubMed]
- Date, A.A.; Halpert, G.; Babu, T.; Ortiz, J.; Kanvinde, P.; Dimitrion, P.; Narayan, J.; Zierden, H.; Betageri, K.; Musmanno, O.; et al. Mucus-penetrating budesonide nanosuspension enema for local treatment of inflammatory bowel disease. Biomaterials 2018, 185, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Cho, W.J.; Jin, A.T.; Kok, L.Y.; Shi, Y.; Heller, D.E.; Lee, Y.A.L.; Zhou, Y.; Xie, X.; Korzenik, J.R.; et al. Heparin-Coated Albumin Nanoparticles for Drug Combination in Targeting Inflamed Intestine. Adv. Healthc. Mater. 2020, 9, 2000536. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; De Mei, C.; Fereira, M.; Marotta, R.; Yoon, H.Y.; Kim, K.; Kwon, I.C.; Decuzzi, P. Dexamethasone-loaded polymeric nanoconstructs for monitoring and treating inflammatory bowel disease. Theranostics 2017, 7, 3653–3666. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Gao, W.; Cai, X.; Xu, J.; Zou, D.; Li, Z.; Hu, B.; Zheng, Y. Nanozyme-mediated catalytic nanotherapy for inflammatory bowel disease. Theranostics 2019, 9, 2843–2855. [Google Scholar] [CrossRef]
- Asgharzadeh, F.; Hashemzadeh, A.; Rahmani, F.; Yaghoubi, A.; Nazari, S.E.; Avan, A.; Mehr, S.M.H.; Soleimanpour, S.; Khazaei, M. Cerium oxide nanoparticles acts as a novel therapeutic agent for ulcerative colitis through anti-oxidative mechanism. Life Sci. 2021, 278, 119500. [Google Scholar] [CrossRef]
- Ahmad, A.; Ansari, M.M.; Mishra, R.K.; Kumar, A.; Vyawahare, A.; Verma, R.K.; Raza, S.S.; Khan, R. Enteric-coated gelatin nanoparticles mediated oral delivery of 5-aminosalicylic acid alleviates severity of DSS-induced ulcerative colitis. Mater. Sci. Eng. C 2021, 119, 111582. [Google Scholar] [CrossRef]
- Davoudi, Z.; Peroutka-Bigus, N.; Bellaire, B.; Jergens, A.; Wannemuehler, M.; Wang, Q. Gut organoid as a new platform to study alginate and chitosan mediated plga nanoparticles for drug delivery. Mar. Drugs 2021, 19, 282. [Google Scholar] [CrossRef]
- Yaghoubi, A.; Davoodi, J.; Asgharzadeh, F.; Rezaie, S.; Nazari, E.; Khazaei, M.; Soleimanpour, S. Therapeutic effect of an anti-tuberculosis agent, isoniazid, and its nano-isoform in ulcerative colitis. Int. Immunopharmacol. 2021, 96, 107577. [Google Scholar] [CrossRef]
- Greish, K.; Taha, S.; Jasim, A.; Elghany, S.A.; Sultan, A.; AlKhateeb, A.; Othman, M.; Jun, F.; Taurin, S.; Bakhiet, M. Styrene maleic acid encapsulated raloxifene micelles for management of inflammatory bowel disease. Clin. Transl. Med. 2017, 6, 28. [Google Scholar] [CrossRef]
- Cai, X.; Wang, X.; He, M.; Wang, Y.; Lan, M.; Zhao, Y.; Gao, F. Colon-targeted delivery of tacrolimus using pH-responsive polymeric nanoparticles for murine colitis therapy. Int. J. Pharm. 2021, 606, 120836. [Google Scholar] [CrossRef] [PubMed]
- Amaldoss, M.J.N.; Najar, I.A.; Kumar, J.; Sharma, A. Therapeutic efficacy of rifaximin loaded tamarind gum polysaccharide nanoparticles in TNBS induced IBD model Wistar rats. Rep. Pract. Oncol. Radiother. 2021, 26, 712–729. [Google Scholar] [CrossRef] [PubMed]
- Eder, P.; Zielińska, A.; Karczewski, J.; Dobrowolska, A.; Słomski, R.; Souto, E.B. How could nanobiotechnology improve treatment outcomes of anti-TNF-α therapy in inflammatory bowel disease? Current knowledge, future directions. J. Nanobiotechnol. 2021, 19, 346. [Google Scholar] [CrossRef] [PubMed]
- Gareb, B.; Posthumus, S.; Beugeling, M.; Koopmans, P.; Touw, D.J.; Dijkstra, G.; Kosterink, J.G.W.; Frijlink, H.W. Towards the oral treatment of ileo-colonic inflammatory bowel disease with infliximab tablets: Development and validation of the production process. Pharmaceutics 2019, 11, 428. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, J.; Wang, L.; Pan, D.; Xu, Y.; Wang, F.; Sheng, J.; Li, X.; Yang, M. Oral delivery of anti-TNF antibody shielded by natural polyphenol-mediated supramolecular assembly for inflammatory bowel disease therapy. Theranostics 2020, 10, 10808–10822. [Google Scholar] [CrossRef] [PubMed]
- Knipe, J.M.; Strong, L.E.; Peppas, N.A. Enzyme- and pH-Responsive Microencapsulated Nanogels for Oral Delivery of siRNA to Induce TNF-α Knockdown in the Intestine. Biomacromolecules 2016, 17, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Xu, J.; Li, Y.; Zhao, R.; Du, S.; Lv, C.; Wu, W.; Liu, R.; Sheng, X.; Song, Y.; et al. MicroRNA-31 Reduces Inflammatory Signaling and Promotes Regeneration in Colon Epithelium, and Delivery of Mimics in Microspheres Reduces Colitis in Mice. Gastroenterology 2019, 156, 2281–2296.e6. [Google Scholar] [CrossRef]
- Neudecker, V.; Haneklaus, M.; Jensen, O.; Khailova, L.; Masterson, J.C.; Tye, H.; Biette, K.; Jedlicka, P.; Brodsky, K.S.; Gerich, M.E.; et al. Myeloid-derived miR-223 regulates intestinal inflammation via repression of the NLRP3 inflammasome. J. Exp. Med. 2017, 214, 1737–1752. [Google Scholar] [CrossRef]
- Gu, L.; Ren, F.; Fang, X.; Yuan, L.; Liu, G.; Wang, S. Exosomal MicroRNA-181a Derived From Mesenchymal Stem Cells Improves Gut Microbiota Composition, Barrier Function, and Inflammatory Status in an Experimental Colitis Model. Front. Med. 2021, 8, 898. [Google Scholar] [CrossRef]
- Attarwala, H.; Clausen, V.; Chaturvedi, P.; Amiji, M.M. Cosilencing Intestinal Transglutaminase-2 and Interleukin-15 Using Gelatin-Based Nanoparticles in an in Vitro Model of Celiac Disease. Mol. Pharm. 2017, 14, 3036–3044. [Google Scholar] [CrossRef]
- Attarwala, H.Z.; Suri, K.; Amiji, M.M. Co-Silencing of Tissue Transglutaminase-2 and Interleukin-15 Genes in a Celiac Disease Mimetic Mouse Model Using a Nanoparticle-in-Microsphere Oral System. Mol. Pharm. 2021, 18, 3099–3107. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Liu, D.; Zhang, T.; You, Q.; Huang, F.; Wu, J. Oral delivery of staphylococcal nuclease ameliorates DSS induced ulcerative colitis in mice via degrading intestinal neutrophil extracellular traps. Ecotoxicol. Environ. Saf. 2021, 215, 112161. [Google Scholar] [CrossRef] [PubMed]
- Gogineni, V.R.; Maddirela, D.R.; Park, W.; Jagtap, J.M.; Parchur, A.K.; Sharma, G.; Ibrahim, E.S.; Joshi, A.; Larson, A.C.; Kim, D.H.; et al. Localized and triggered release of oxaliplatin for the treatment of colorectal liver metastasis. J. Cancer 2020, 11, 6982–6991. [Google Scholar] [CrossRef] [PubMed]
- Saber, M.M.; Al-mahallawi, A.M.; Nassar, N.N.; Stork, B.; Shouman, S.A. Targeting colorectal cancer cell metabolism through development of cisplatin and metformin nano-cubosomes. BMC Cancer 2018, 18, 822. [Google Scholar] [CrossRef]
- Kotelevets, L.; Chastre, E.; Caron, J.; Mougin, J.; Bastian, G.; Pineau, A.; Walker, F.; Lehy, T.; Desmaële, D.; Couvreur, P. A squalene-based nanomedicine for oral treatment of colon cancer. Cancer Res. 2017, 77, 2964–2975. [Google Scholar] [CrossRef]
- Scheetz, L.M.; Yu, M.; Li, D.; Castro, M.G.; Moon, J.J.; Schwendeman, A. Synthetic HDL nanoparticles delivering docetaxel and cpg for chemoimmunotherapy of colon adenocarcinoma. Int. J. Mol. Sci. 2020, 21, 1777. [Google Scholar] [CrossRef]
- Li, T.; Deng, N.; Xu, R.; Fan, Z.; He, J.; Zheng, Z.; Deng, H.; Liao, R.; Lv, X.; Pang, C. NEAT1 siRNA Packed with Chitosan Nanoparticles Regulates the Development of Colon Cancer Cells via lncRNA NEAT1/miR-377-3p Axis. Biomed. Res. Int. 2021, 2021, 5528982. [Google Scholar] [CrossRef]
- Mundekkad, D.; Cho, W.C. Nanoparticles in Clinical Translation for Cancer Therapy. Int. J. Mol. Sci. 2022, 23, 1685. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.; Ramadan, H. Novel strategies to oral delivery of insulin: Current progress of nanocarriers for diabetes management. Drug Dev. Res. 2021. [Google Scholar] [CrossRef]
- Younis, M.A.; Tawfeek, H.M.; Abdellatif, A.A.H.; Abdel-Aleem, J.A.; Harashima, H. Clinical translation of nanomedicines: Challenges, opportunities, and keys. Adv. Drug Deliv. Rev. 2022, 181, 114083. [Google Scholar] [CrossRef]
- Baranowska-Wójcik, E. Factors Conditioning the Potential Effects TiO2 NPs Exposure on Human Microbiota: A Mini-Review. Biol. Trace Elem. Res. 2021, 199, 4458–4465. [Google Scholar] [CrossRef] [PubMed]
- Dorier, M.; Béal, D.; Marie-Desvergne, C.; Dubosson, M.; Barreau, F.; Houdeau, E.; Herlin-Boime, N.; Carriere, M. Continuous in vitro exposure of intestinal epithelial cells to E171 food additive causes oxidative stress, inducing oxidation of DNA bases but no endoplasmic reticulum stress. Nanotoxicology 2017, 11, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, C.; Barisani, D. Food additives can act as triggering factors in celiac disease: Current knowledge based on a critical review of the literature. World J. Clin. Cases 2019, 7, 917–927. [Google Scholar] [CrossRef]
- Food and Drug Administration. 21CFR.73.575 Titanium Dioxide. In Code of Federal Regulations Title 21; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2011; pp. 450–451. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm?fr=73.575 (accessed on 6 January 2022).
- More, S.; Bampidis, V.; Benford, D.; Bragard, C.; Halldorsson, T.; Hernández-Jerez, A.; Bennekou, S.H.; Koutsoumanis, K.; Lambré, C.; Machera, K.; et al. Guidance on technical requirements for regulated food and feed product applications to establish the presence of small particles including nanoparticles. EFSA J. 2021, 19, e06769. [Google Scholar] [CrossRef]
- EFSA Panel on Food Contact Materials Enzymes Flavourings and Processing Aids. Safety assessment of the substance zinc oxide, nanoparticles, for use in food contact materials. EFSA J. 2016, 14, 4408. [Google Scholar] [CrossRef]
- EFSA Panel on Food Contact Materials; Enzymes and Processing Aids (CEP); Lambré, C.; Barat Baviera, J.M.; Bolognesi, C.; Chesson, A.; Cocconcelli, P.S.; Rivière, G. Safety assessment of the substance silver nanoparticles for use in food contact materials. EFSA J. 2021, 19, e06790. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the re-evaluation of silver (E 174) as food additive. EFSA J. 2016, 14, 4364. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific Opinion on the re-evaluation of gold (E 175) as a food additive. EFSA J. 2016, 14, 4362. [Google Scholar] [CrossRef]
- European Food Safety Authority. Statement of EFSA on the Evaluation of a new study related to the bioavailability of aluminium in food. EFSA J. 2011, 9, 2157. [Google Scholar] [CrossRef]
- Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Dusemund, B.; Filipič, M.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; et al. Re-evaluation of silicon dioxide (E 551) as a food additive. EFSA J. 2018, 16, E05088. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific Opinion on the re-evaluation of iron oxides and hydroxides (E 172) as food additives. EFSA J. 2015, 13, 4317. [Google Scholar] [CrossRef]
- Murugadoss, S.; Brassinne, F.; Sebaihi, N.; Petry, J.; Cokic, S.M.; Van Landuyt, K.L.; Godderis, L.; Mast, J.; Lison, D.; Hoet, P.H.; et al. Agglomeration of titanium dioxide nanoparticles increases toxicological responses in vitro and in vivo. Part. Fibre Toxicol. 2020, 17, 10. [Google Scholar] [CrossRef]
- Marucco, A.; Prono, M.; Beal, D.; Alasonati, E.; Fisicaro, P.; Bergamaschi, E.; Carriere, M.; Fenoglio, I. Biotransformation of food-grade and nanometric tio2 in the oral-gastro-intestinal tract: Driving forces and effect on the toxicity toward intestinal epithelial cells. Nanomaterials 2020, 10, 2132. [Google Scholar] [CrossRef]
- Cao, X.; Zhang, T.; DeLoid, G.M.; Gaffrey, M.J.; Weitz, K.K.; Thrall, B.D.; Qian, W.J.; Demokritou, P. Evaluation of the cytotoxic and cellular proteome impacts of food-grade TiO2 (E171) using simulated gastrointestinal digestions and a tri-culture small intestinal epithelial model. NanoImpact 2020, 17, 100202. [Google Scholar] [CrossRef]
- Dudefoi, W.; Rabesona, H.; Rivard, C.; Mercier-Bonin, M.; Humbert, B.; Terrisse, H.; Ropers, M.H. In vitro digestion of food grade TiO2 (E171) and TiO2 nanoparticles: Physicochemical characterization and impact on the activity of digestive enzymes. Food Funct. 2021, 12, 5975–5988. [Google Scholar] [CrossRef]
- Benbow, N.L.; Rozenberga, L.; McQuillan, A.J.; Krasowska, M.; Beattie, D.A. ATR FTIR Study of the Interaction of TiO2 Nanoparticle Films with β-Lactoglobulin and Bile Salts. Langmuir 2021, 37, 13278–13290. [Google Scholar] [CrossRef]
- Coméra, C.; Cartier, C.; Gaultier, E.; Catrice, O.; Panouille, Q.; El Hamdi, S.; Tirez, K.; Nelissen, I.; Théodorou, V.; Houdeau, E. Jejunal villus absorption and paracellular tight junction permeability are major routes for early intestinal uptake of food-grade TiO2particles: An in vivo and ex vivo study in mice. Part. Fibre Toxicol. 2020, 17, 26. [Google Scholar] [CrossRef]
- Guillard, A.; Gaultier, E.; Cartier, C.; Devoille, L.; Noireaux, J.; Chevalier, L.; Morin, M.; Grandin, F.; Lacroix, M.Z.; Coméra, C.; et al. Basal Ti level in the human placenta and meconium and evidence of a materno-foetal transfer of food-grade TiO2nanoparticles in an ex vivo placental perfusion model. Part. Fibre Toxicol. 2020, 17, 51. [Google Scholar] [CrossRef]
- Su, J.; Duan, X.; Qiu, Y.; Zhou, L.; Zhang, H.; Gao, M.; Liu, Y.; Zou, Z.; Qiu, J.; Chen, C. Pregnancy exposure of titanium dioxide nanoparticles causes intestinal dysbiosis and neurobehavioral impairments that are not significant postnatally but emerge in adulthood of offspring. J. Nanobiotechnol. 2021, 19, 234. [Google Scholar] [CrossRef]
- Mortensen, N.P.; Moreno Caffaro, M.; Aravamudhan, S.; Beeravalli, L.; Prattipati, S.; Snyder, R.W.; Watson, S.L.; Patel, P.R.; Weber, F.X.; Montgomery, S.A.; et al. Simulated gastric digestion and in vivo intestinal uptake of orally administered cuo nanoparticles and tio2 e171 in male and female rat pups. Nanomaterials 2021, 11, 1487. [Google Scholar] [CrossRef]
- Bettini, S.; Boutet-Robinet, E.; Cartier, C.; Coméra, C.; Gaultier, E.; Dupuy, J.; Naud, N.; Taché, S.; Grysan, P.; Reguer, S.; et al. Food-grade TiO2 impairs intestinal and systemic immune homeostasis, initiates preneoplastic lesions and promotes aberrant crypt development in the rat colon. Sci. Rep. 2017, 7, 40373. [Google Scholar] [CrossRef]
- Hummel, T.Z.; Kindermann, A.; Stokkers, P.C.F.; Benninga, M.A.; Ten Kate, F.J.W. Exogenous pigment in peyer patches of children suspected of having IBD. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 477–480. [Google Scholar] [CrossRef]
- Barreau, F.; Tisseyre, C.; Ménard, S.; Ferrand, A.; Carriere, M. Titanium dioxide particles from the diet: Involvement in the genesis of inflammatory bowel diseases and colorectal cancer. Part. Fibre Toxicol. 2021, 18, 26. [Google Scholar] [CrossRef]
- Talbot, P.; Radziwill-Bienkowska, J.M.; Kamphuis, J.B.J.; Steenkeste, K.; Bettini, S.; Robert, V.; Noordine, M.L.; Mayeur, C.; Gaultier, E.; Langella, P.; et al. Food-grade TiO2 is trapped by intestinal mucus in vitro but does not impair mucin O-glycosylation and short-chain fatty acid synthesis in vivo: Implications for gut barrier protection. J. Nanobiotechnol. 2018, 16, 53. [Google Scholar] [CrossRef]
- Zhu, X.; Zhao, L.; Liu, Z.; Zhou, Q.; Zhu, Y.; Zhao, Y.; Yang, X. Long-term exposure to titanium dioxide nanoparticles promotes diet-induced obesity through exacerbating intestinal mucus layer damage and microbiota dysbiosis. Nano Res. 2021, 14, 1512–1522. [Google Scholar] [CrossRef]
- Cao, X.; Han, Y.; Gu, M.; Du, H.; Song, M.; Zhu, X.; Ma, G.; Pan, C.; Wang, W.; Zhao, E.; et al. Foodborne Titanium Dioxide Nanoparticles Induce Stronger Adverse Effects in Obese Mice than Non-Obese Mice: Gut Microbiota Dysbiosis, Colonic Inflammation, and Proteome Alterations. Small 2020, 16, 2001858. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, S.; Tang, Y.; You, T.; Xu, H. Lactobacillus rhamnosus GG Ameliorated Long-Term Exposure to TiO2Nanoparticles Induced Microbiota-Mediated Liver and Colon Inflammation and Fructose-Caused Metabolic Abnormality in Metabolism Syndrome Mice. J. Agric. Food Chem. 2021, 69, 9788–9799. [Google Scholar] [CrossRef]
- Zhao, Y.; Tang, Y.; Chen, L.; Lv, S.; Liu, S.; Nie, P.; Aguilar, Z.P.; Xu, H. Restraining the TiO2 nanoparticles-induced intestinal inflammation mediated by gut microbiota in juvenile rats via ingestion of Lactobacillus rhamnosus GG. Ecotoxicol. Environ. Saf. 2020, 206, 111393. [Google Scholar] [CrossRef]
- Corps Ricardo, A.I.; Rodríguez Fariñas, N.; Guzmán Bernardo, F.J.; Rodríguez Martín-Doimeadios, R.C.; Ríos, Á. Screening-confirmation strategy for nanomaterials involving spectroscopic analytical techniques and its application to the control of silver nanoparticles in pastry samples. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 246, 119015. [Google Scholar] [CrossRef]
- De Vos, S.; Waegeneers, N.; Verleysen, E.; Smeets, K.; Mast, J. Physico-chemical characterisation of the fraction of silver (nano)particles in pristine food additive E174 and in E174-containing confectionery. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2020, 37, 1831–1846. [Google Scholar] [CrossRef]
- Du, J.; Tang, J.; Xu, S.; Ge, J.; Dong, Y.; Li, H.; Jin, M. A review on silver nanoparticles-induced ecotoxicity and the underlying toxicity mechanisms. Regul. Toxicol. Pharmacol. 2018, 98, 231–239. [Google Scholar] [CrossRef]
- Grasso, A.; Ferrante, M.; Arena, G.; Salemi, R.; Zuccarello, P.; Fiore, M.; Copat, C. Chemical characterization and quantification of silver nanoparticles (Ag-NPs) and dissolved ag in seafood by single particle icp-ms: Assessment of dietary exposure. Int. J. Environ. Res. Public Health 2021, 18, 4076. [Google Scholar] [CrossRef]
- Stensberg, M.C.; Wei, Q.; McLamore, E.S.; Porterfield, D.M.; Wei, A.; Sepúlveda, M.S. Toxicological studies on silver nanoparticles: Challenges and opportunities in assessment, monitoring and imaging. Nanomedicine 2011, 6, 879–898. [Google Scholar] [CrossRef]
- Lamas, B.; Martins Breyner, N.; Houdeau, E. Impacts of foodborne inorganic nanoparticles on the gut microbiota-immune axis: Potential consequences for host health. Part. Fibre Toxicol. 2020, 17, 19. [Google Scholar] [CrossRef]
- Mao, B.H.; Chen, Z.Y.; Wang, Y.J.; Yan, S.J. Silver nanoparticles have lethal and sublethal adverse effects on development and longevity by inducing ROS-mediated stress responses. Sci. Rep. 2018, 8, 2445. [Google Scholar] [CrossRef]
- Mulenos, M.R.; Liu, J.; Lujan, H.; Guo, B.; Lichtfouse, E.; Sharma, V.K.; Sayes, C.M. Copper, silver, and titania nanoparticles do not release ions under anoxic conditions and release only minute ion levels under oxic conditions in water: Evidence for the low toxicity of nanoparticles. Environ. Chem. Lett. 2020, 18, 1319–1328. [Google Scholar] [CrossRef]
- Polet, M.; Laloux, L.; Cambier, S.; Ziebel, J.; Gutleb, A.C.; Schneider, Y.J. Soluble silver ions from silver nanoparticles induce a polarised secretion of interleukin-8 in differentiated Caco-2 cells. Toxicol. Lett. 2020, 325, 14–24. [Google Scholar] [CrossRef]
- Jia, M.; Zhang, W.; He, T.; Shu, M.; Deng, J.; Wang, J.; Li, W.; Bai, J.; Lin, Q.; Luo, F.; et al. Evaluation of the genotoxic and oxidative damage potential of silver nanoparticles in human NCM460 and HCT116 cells. Int. J. Mol. Sci. 2020, 21, 1618. [Google Scholar] [CrossRef]
- Rufino, A.T.; Ramalho, A.; Sousa, A.; de Oliveira, J.M.P.F.; Freitas, P.; Gonzalez Gómez, M.A.; Piñeiro-Redondo, Y.; Rivas, J.; Carvalho, F.; Fernandes, E.; et al. Protective role of flavonoids against intestinal pro-inflammatory effects of silver nanoparticles. Molecules 2021, 26, 6610. [Google Scholar] [CrossRef]
- Kämpfer, A.A.M.; Urbán, P.; La Spina, R.; Jiménez, I.O.; Kanase, N.; Stone, V.; Kinsner-Ovaskainen, A. Ongoing inflammation enhances the toxicity of engineered nanomaterials: Application of an in vitro co-culture model of the healthy and inflamed intestine. Toxicol. Vitr. 2020, 63, 104738. [Google Scholar] [CrossRef]
- van den Brule, S.; Ambroise, J.; Lecloux, H.; Levard, C.; Soulas, R.; De Temmerman, P.J.; Palmai-Pallag, M.; Marbaix, E.; Lison, D. Dietary silver nanoparticles can disturb the gut microbiota in mice. Part. Fibre Toxicol. 2016, 13, 38. [Google Scholar] [CrossRef]
- Agans, R.T.; Gordon, A.; Hussain, S.; Paliy, O. Titanium Dioxide Nanoparticles Elicit Lower Direct Inhibitory Effect on Human Gut Microbiota Than Silver Nanoparticles. Toxicol. Sci. 2019, 172, 411–416. [Google Scholar] [CrossRef]
- Gokulan, K.; Williams, K.; Orr, S.; Khare, S. Human intestinal tissue explant exposure to silver nanoparticles reveals sex dependent alterations in inflammatory responses and epithelial cell permeability. Int. J. Mol. Sci. 2020, 22, 9. [Google Scholar] [CrossRef]
- Siczek, K.; Zatorski, H.; Chmielowiec-Korzeniowska, A.; Pulit-Prociak, J.; Śmiech, M.; Kordek, R.; Tymczyna, L.; Banach, M.; Fichna, J. Synthesis and evaluation of anti-inflammatory properties of silver nanoparticle suspensions in experimental colitis in mice. Chem. Biol. Drug Des. 2017, 89, 538–547. [Google Scholar] [CrossRef]
- Gao, Y.; Li, T.; Duan, S.; Lyu, L.; Li, Y.; Xu, L.; Wang, Y. Impact of titanium dioxide nanoparticles on intestinal community in 2,4,6-trinitrobenzenesulfonic acid (TNBS)-induced acute colitis mice and the intervention effect of vitamin E. Nanoscale 2021, 13, 1842–1862. [Google Scholar] [CrossRef]
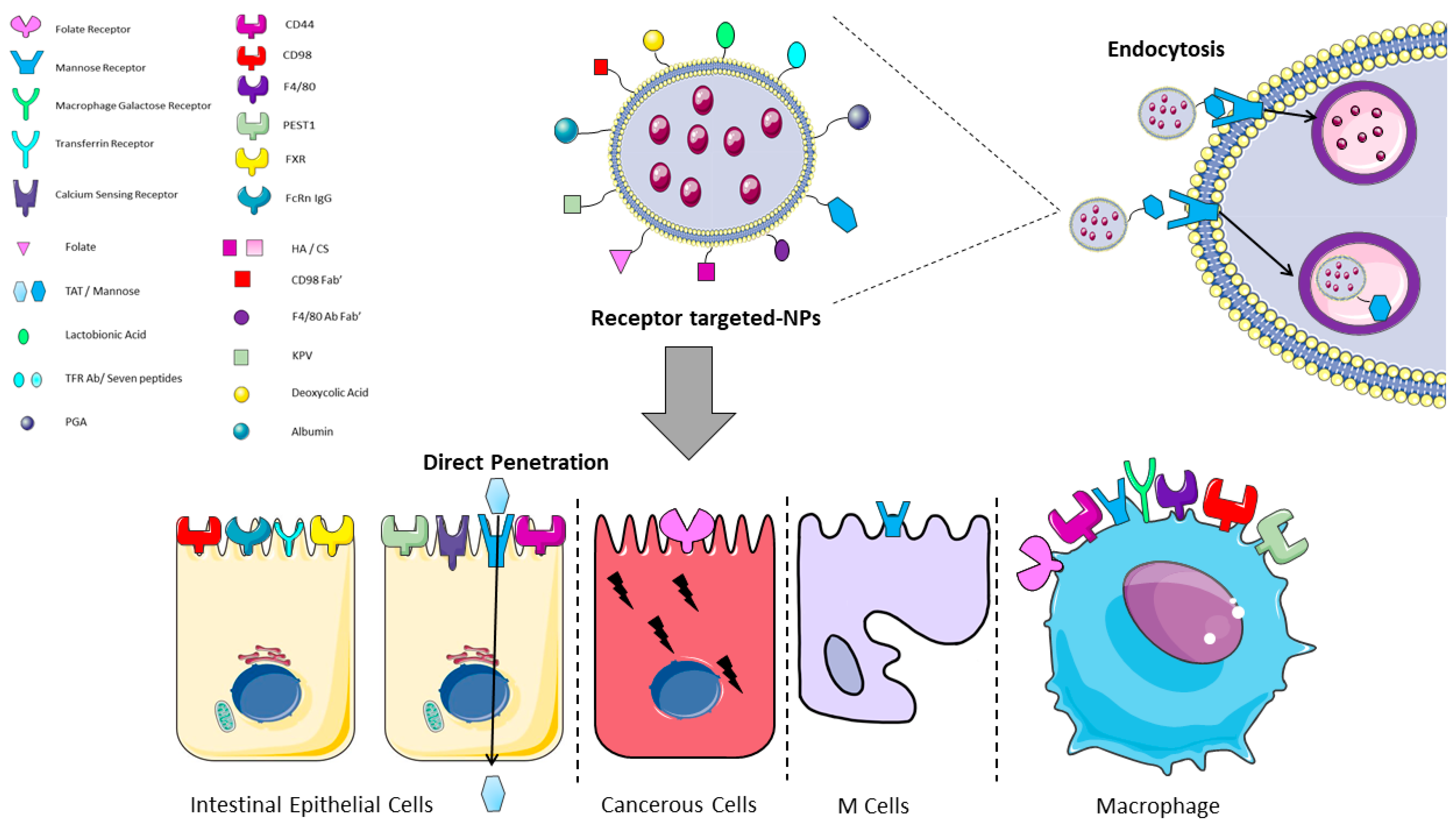
| Reference | Receptor | Ligand | Cell Type Expression | Direct Penetration | Endocytosis |
|---|---|---|---|---|---|
| Hua S 2020 [73] | Mannose Receptor | Mannose | Macrophages, Enterocytes, M cells | No | Yes |
| Tian 2018 [74] | CD44 | HA/CS | Macrophages, Intestinal Epithelial Cells | No | Yes |
| Xiao, 2018 [75] | CD98 | CD98 Fab’/single chain CD98 Ab | Intestinal Epithelial Cells, Macrophages | No | Yes |
| Peng L, 2021 [76] | F4/80 | F4/80 Ab Fab’ | Macrophages | No | Yes |
| Liu W, 2018 [77] | Macrophage Galactose Receptor | Lactobionic Acid | Macrophages | No | Yes |
| Xi Z 2022, Álvarez-González, 2020 [78,79] | Folate Receptor | Folate | Macrophages, Epithelial Cancer Cells | No | Yes |
| Yong, 2019 [80] | Transferrin Receptor | TFR Ab/Seven peptides | Intestinal Epithelial Cells | No | Yes |
| Zhang W, 2021 [81] | PEST1 | KPV | Macrophages, Intestinal Epithelial Cells | No | Yes |
| Liu L, 2018 [82] | Mannose Receptor | TAT | Intestinal Epithelial Cells, Macrophages | Yes | No |
| Azevedo, 2020 [83] | FcRn IgG | Albumin | Intestinal Epithelial Cells | No | Yes |
| Huang X, 2021 [84] | FXR | Deoxycolic Acid | Intestinal Epithelial Cells | No | Yes |
| Urimi, 2019 [85] | Calcium Sensing Receptor | PGA | Intestinal Epithelial Cells | No | Yes |
| Reference | Core of the NPs | Further Functionalization for Adhesion/Passage | Release Control | Reduces Glycaemia in Animal Model |
|---|---|---|---|---|
| Li L 2017 [88] | Chitosan | CPP | n/a | Yes |
| Wu J-Z 2017 [89] | diethylene glycol dimethacrylate | n/a | phenylboronic acid | Yes |
| Alfatama 2018 [90] | Alginate/Chitosan | n/a | n/a | Yes |
| Czuba 2018 [91] | PLGA | SDS | n/a | Yes |
| Fan 2018 [92] | Chitosan | Deoxycholic acid | n/a | Yes |
| Hou 2018 [93] | Mesoporous silica nanoparticle | n/a | phenylboronic acid | Yes |
| Jamshidi 2018 [94] | Chitosan | n/a | n/a | Yes |
| Ji N 2018 [95] | Zein + CSA | n/a | n/a | n/a |
| Liu L 2018 [82] | Chitosan + hydrogel | n/a | n/a | Yes |
| Song M 2018 [96] | Cyclodextrin/chitosan | n/a | n/a | Yes |
| Tian 2018 [74] | Chitosan/hyaluronic acid | n/a | n/a | Yes |
| Wang W 2018 [97] | Polyamidoamine/polyaspartic acid/phenylboronic acid/PEG | PEG | phenylboronic acid | Yes |
| Xu Y 2018 [98] | solid lipid nanoparticle + endosomal escape agent | n/a | n/a | Yes |
| Zhang Y 2018 [99] | hydroxyapatite | PEG | n/a | Yes |
| Zhang L. 2018 [100] | PLGA + chitosan + alginate | n/a | pH dependent | Yes |
| Alsulays 2019 [101] | Solid lipid nanoparticles | CPP | n/a | Yes |
| Guo 2019 [102] | Chitosan | CPP | n/a | yes |
| Hu 2019 [103] | phospholipids | n/a | n/a | Yes |
| Jamwal 2019 [104] | dextran | n/a | Glucose oxidase | n/a |
| Ji 2019 [105] | Chitosan/zein-carboxymethylated short-chain amylose | n/a | n/a | Yes |
| Mohammadpour 2019 [106] | PLGA + chitosan | n/a | Glucose oxidase | Yes |
| Muntoni 2019 [107] | Lipid nanoparticles | n/a | n/a | Yes |
| Mudassir 2019 [108] | Methyl methacrylate/itaconic acid nanogels | n/a | pH dependent | Yes |
| Tsai 2019 [109] | Chitosan + fucoidan | n/a | pH dependent | n/a |
| Urimi 2019 [85] | Chitosan | Polyglutamic acid | n/a | Yes |
| Azevedo 2020 [83] | Albumin | n/a | n/a | Yes |
| Bai 2020 [110] | PLGA + glutamic acid conjugated amphiphilic dendrimer | n/a | n/a | Yes |
| Chai 2020 [111] | Poly (acrylamido phenylboronic acid)/sodium alginate | n/a | Cicloborate (Glucose sensing) and glucose oxidase | Yes |
| Chen Z 2020 [112] | Chitosan/Hyaluronic acid | CPP | n/a | Yes |
| Cheng 2020 [113] | Poly (n-butylcyanoacrylate) | Ratio insulin/Poly (n-butylcyanoacrylate) | Ratio insulin/Poly (n-butylcyanoacrylate) | Yes |
| Ding 2020 [114] | amphiphilic cholesterol- phosphate conjugate | n/a | pH dependent | Yes |
| Han X 2020 [115] | Zwitterionic micelles | Betaine | n/a | Yes |
| Jana 2020 [116] | hyaluronic acid | n/a | Glucose oxidase | n/a |
| Mumuni 2020 [117] | Chitosan/mucin | n/a | n/a | yes |
| Sladek 2020 [118] | Hyaluronic acid/chitosan | Sucrose laurate | n/a | Yes |
| Sudhakar 2020 [119] | Chitosan | n/a | pH dependent | Yes |
| Tan X 2020 [120] | Mesoporous silica | PEG + CPP | n/a | Yes |
| Wang T 2020 [121] | Lipid nanoparticles | n/a | n/a | Yes |
| Zhou S 2020 [122] | Chitosan | PC6 | pH dependent | Yes |
| Zhou X 2020 [123] | Alginate | n/a | Glucose oxidase | Yes |
| Zhou Y 2020 [124] | FeCl3·6H2O + BTC | SDS | pH dependent | Yes |
| Bao X 2021 [125] | Zein/casein-dextran | Cholic acid | n/a | Yes |
| Benyettou 2021 [126] | Nanoscale imine-linked covalent organic frameworks | n/a | pH dependent | Yes |
| Cui 2021 [127] | Chitosan + Hyaluronic acid | Biotin | n/a | Yes |
| Huang X 2021 [84] | layered double hydroxide nanoparticle + hyaluronic acid | Deoxycholic acid | n/a | Yes |
| Kim WJ 2021 [128] | POSS-APBA | n/a | phenylboronic acid | n/a |
| Li H 2021 [129] | polyphosphoesters-based copolymer | n/a | phenylboronic acid | Yes |
| Li J 2021 [130] | Alginate/chitosan | n/a | pH dependent | Yes |
| Liu X 2021 [131] | PLGA/PEG | Angiopep-2 | n/a | Yes |
| Qin 2021 [132] | Mesoporous silica + Alginate + Boronic acid Mesoporous silica + Chitosan + boronic acid | n/a | phenylboronic acid | Yes |
| Rao 2021 [133] | Porous silicon nanoparticles | Zwitterionic dodecyl sulfobetaine | n/a | Yes |
| Volpatti 2021 [134] | Polycation | n/a | Glucose oxidase | Yes |
| Wang W 2021 [135] | PLGA | Chitosan + Cholanic acid | n/a | Yes |
| Zhang Y 2021 [136] | mesoporous silica nanoparticles | CPP | n/a | Yes |
| Fu 2022 [137] | Glycopolymer | n/a | phenylboronic acid | Yes |
| Li J 2022 [138] | PLGA-Hyd-PEG | PEG | n/a | Yes |
| Martins 2022 [139] | Lignin-encapsulated silicon | Fc fragment of IgG | pH dependent | n/a |
| Reboredo 2022 [140] | Zein | PEG | n/a | Yes |
| Rohra 2022 [141] | Gold nanoparticle-encapsulated zeolitic imidazolate framework-8 | n/a | Glucose oxidase | n/a |
| Xi Z 2022 [78] | PLGA/PEG | PEG, folate and charge-convertible tripeptide | n/a | Yes |
| Xu 2022 [142] | konjac glucomannan/concanavalin A | n/a | Glucose sensing | Yes |
| Drug Name | Company | Material Used | Delivery Route | NCT Number | Outcome |
|---|---|---|---|---|---|
| HDV-I | Diasome Pharmaceuticals and Integrium | Liposomal bilayer containing Hepatic directed vesicles (HDV)–insulin | Oral | NCT00814294 NCT00521378 | No results posted No results posted |
| Oshadi | Oshadi Drug Administration Ltd. | Silica-based NP with polysaccharides and oil combination of insulin, proinsulin, and C-peptide | Oral | NCT01120912 NCT01973920 NCT01772251 | No results posted No results posted No results posted |
| ORMD-0801 | Oramed Pharmaceuticals Ltd. | Human recombinant insulin contained in an enteric coated capsule with adjuvants | Oral | NCT02496000 | Positive: ORMD-0801 was well tolerated and had a significant anti-hyperglycaemic effect, not associated with any serious hypoglycaemia conditions. |
| NCT03467932 | No result posted | ||||
| NCT00867594 | No results posted | ||||
| IN-105 | Biocon Ltd. | PEGylated-Tregopil (modified form of human insulin) | Oral | NCT01035801 | No results posted |
| NCT04141423 | No results posted | ||||
| NCT03430856 | Positive: IN-105 is relatively well tolerated as compared to Insulin Aspart. | ||||
| GIPET® (insulin 338) | Novo Nordisk | Micelles-loaded human recombinant insulin | Oral | NCT01931137 NCT02470039 NCT02304627 NCT01809184 NCT01796366 | No results posted No results posted No results posted No results posted No results posted |
| NasulinTM | CPEX Pharmaceuticals | Formulation of CPE-215 (cyclopentadecalactone) recombinant human insulin | Intranasal | NCT00850096 | Positive: NasulinTM is relatively well tolerated and increased the absorption of insulin with repeated dosing on the same nostril. |
| Afrezza1 | Mannkind and Sanofi | Technosphere microparticles (fumaryl diketopiperazine (FDKP))of recombinant human insulin | Inhaled | NCT03143816 NCT02485327 | Positive: Afrezza1 improves post-prandial glucose without increasing hypoglycaemia. No results posted |
| Food Additive | Main Content | Properties | Current Recommendations (EFSA) | Estimated Intake in Toddlers | Estimated Intake in Children | Estimated Intake in Adults | Main Concerns |
|---|---|---|---|---|---|---|---|
| E171 [37] | TiO2 | Food colourant (white) | No longer considered safe when used as food additive | 0.9–12.8 mg/kg/day | 1.9–11.5 mg/kg/day | 0.7–6.7 mg/kg/day | Genotoxicity. DNA damage. Accumulation in tissues. Inflammation, dysbiosis, leaky gut worsened in pre-existing conditions. |
| E172 [192] | Fe oxides and hydroxides | Food colourant (yellow, red, black), Food supplements | No limitations at the moment. Need to distinguish the different compounds. Need for more studies on genotoxicity. | 0.4–10.5 mg/kg/day | 1.4–9.2 mg/kg/day | 0.3–2.4 mg/kg/day | Genotoxicity. Limited studies at the time of assessment (2015). |
| E173 * [190] | Al | Food colourant (grey) | TWI of 1mg/kg/week | n.a | n.a. | 0.2–1.5 mg/kg/week | Ions accumulation in tissues, including nervous system. Very few data at the moment. |
| E174 [188] | Ag | Food colourant (silver-grey), antimicrobial agent | Need for more data on E174 characterisation. Need for specifications of the mean particle size distribution and NPs %. | 0.003–0.08 mg/kg/day | 0.01–0.11 mg/kg/day | 0.001–0.03 mg/kg/day | Potential release of Ag ions. Potential: cytotoxicity, induction of oxidative stress, inflammatory response, dysbiosis. |
| - [187] | Ag | Antimicrobial agent in food packaging | Under the intended and tested condition of use do not give rise to toxicological concern. | <0.9 ug ion/kg/day (ADI) | <0.9 ug ion/kg/day (ADI) | <0.9 ug ion/kg/day (ADI) | Potential release of Ag ions. |
| E175 [189] | Au | Food colorant (yellowish gold) | Need for specifications of the mean particle size distribution and NPs percentage. Currently not enough data. | 0.01–0.26 μg/kg/day | 0.04–0.33 μg/kg/day | 0.01–0.09 μg/kg/day | Potential accumulation in tissues, but more data needed. |
| - [186] | ZnO | Antimicrobial agent and UV-light adsorber in food packaging | Migration only in the form of ions is observed, but lower than the specific migration limit → ok. Zinc upper limit 25mg/person/day | n.a. | n.a. | n.a. | Upper limit could be exceeded since Zn is present in more sources other than food packaging |
| E551 [191] | SiO2 | Texture-improving agent, flavour-carrying agent | More stringent limitations on metals present in E551 formulations. Need for more studies to define a proper ADI. | 18.5–39.4 mg/kg/day | 10.5–31.2 mg/kg/day | 4.9–13.2 mg/kg/day | Potential presence of metal contaminants. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitulo, M.; Gnodi, E.; Meneveri, R.; Barisani, D. Interactions between Nanoparticles and Intestine. Int. J. Mol. Sci. 2022, 23, 4339. https://doi.org/10.3390/ijms23084339
Vitulo M, Gnodi E, Meneveri R, Barisani D. Interactions between Nanoparticles and Intestine. International Journal of Molecular Sciences. 2022; 23(8):4339. https://doi.org/10.3390/ijms23084339
Chicago/Turabian StyleVitulo, Manuela, Elisa Gnodi, Raffaella Meneveri, and Donatella Barisani. 2022. "Interactions between Nanoparticles and Intestine" International Journal of Molecular Sciences 23, no. 8: 4339. https://doi.org/10.3390/ijms23084339
APA StyleVitulo, M., Gnodi, E., Meneveri, R., & Barisani, D. (2022). Interactions between Nanoparticles and Intestine. International Journal of Molecular Sciences, 23(8), 4339. https://doi.org/10.3390/ijms23084339






