Molecular Traits and Functional Exploration of BES1 Gene Family in Plants
Abstract
:1. Introduction
2. Results
2.1. Identification of BES1 Gene Family in Plantae
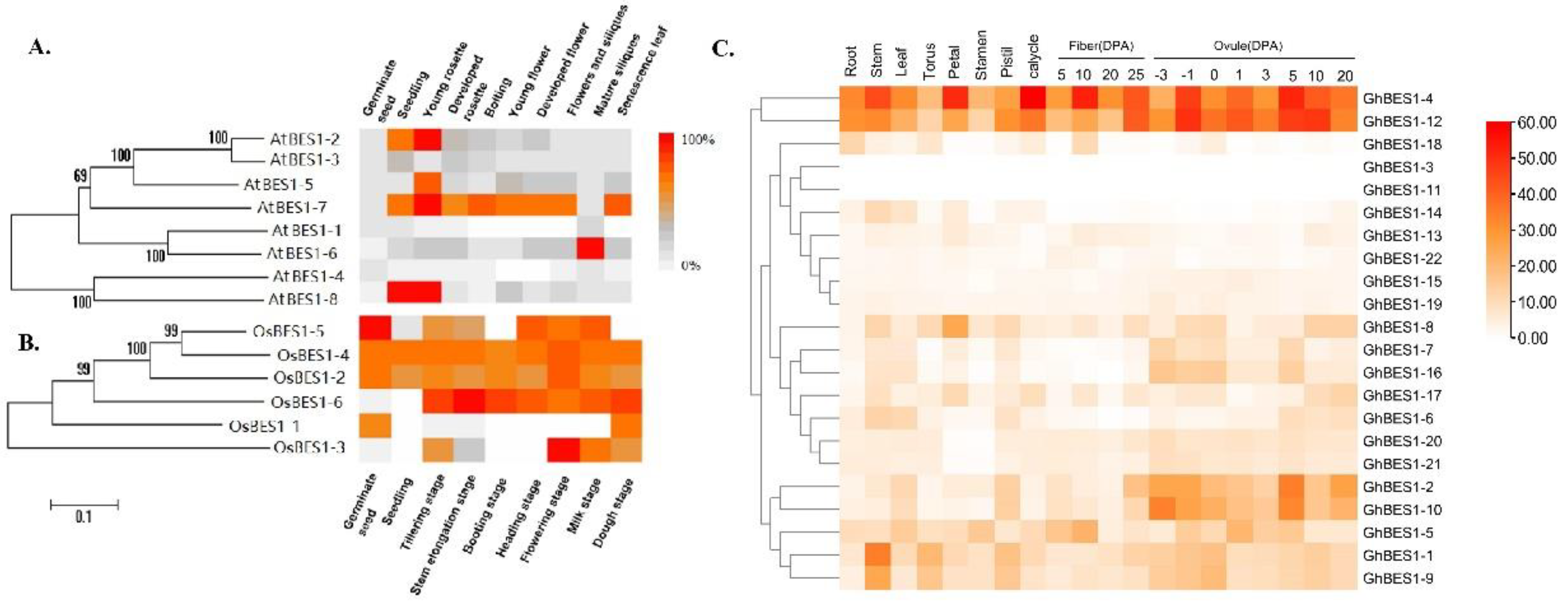
2.2. Phylogenetic Analysis, Conserved Motif, and Protein Characteristics
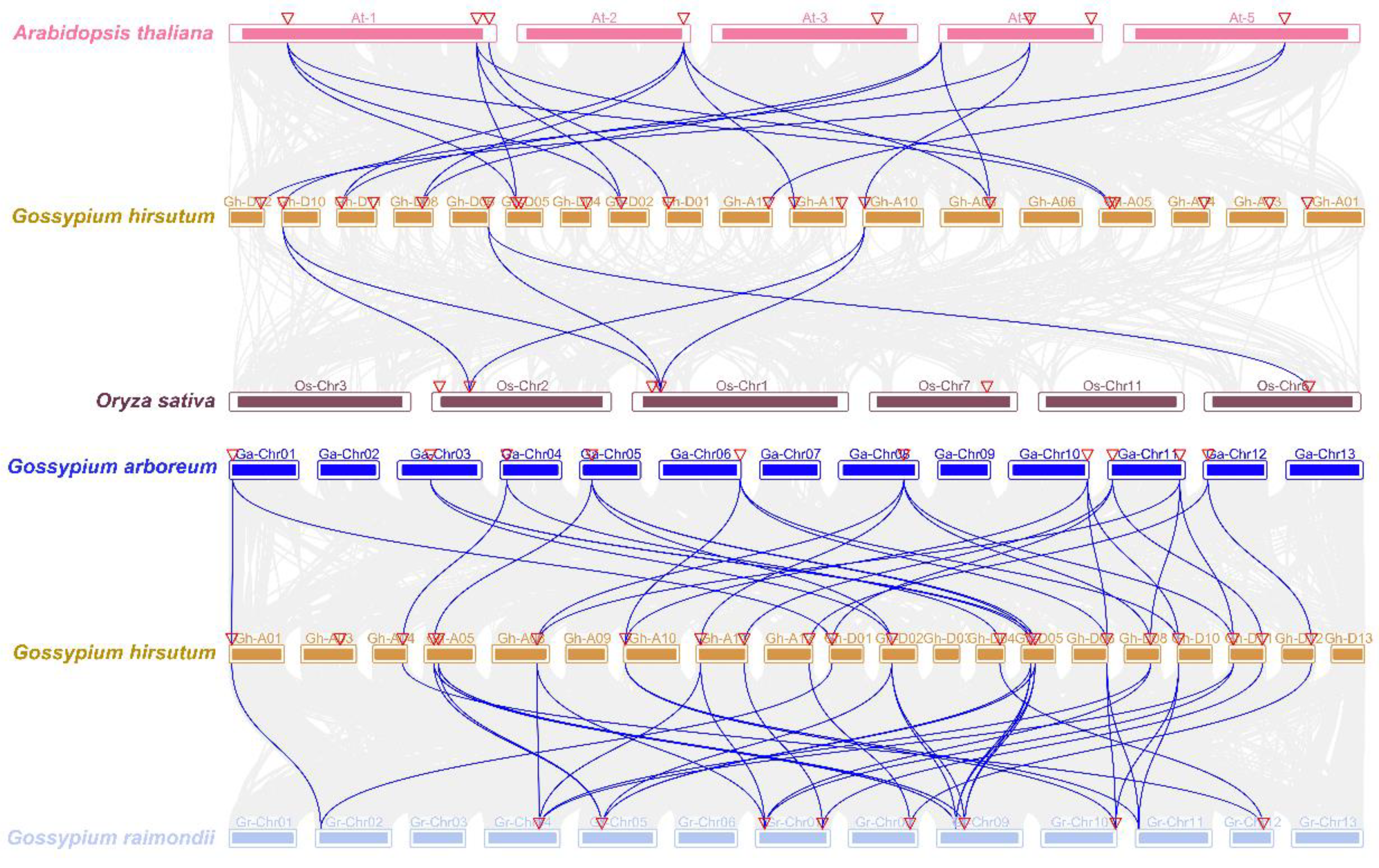
2.3. Structure, Collinearity, and Cis-Acting Elements Analysis
2.4. Expression Analysis of BES1 Family Genes in Arabidopsis, Rice, and Cotton
2.5. Expression Profiles of BES1 Family Genes under Abiotic Stresses
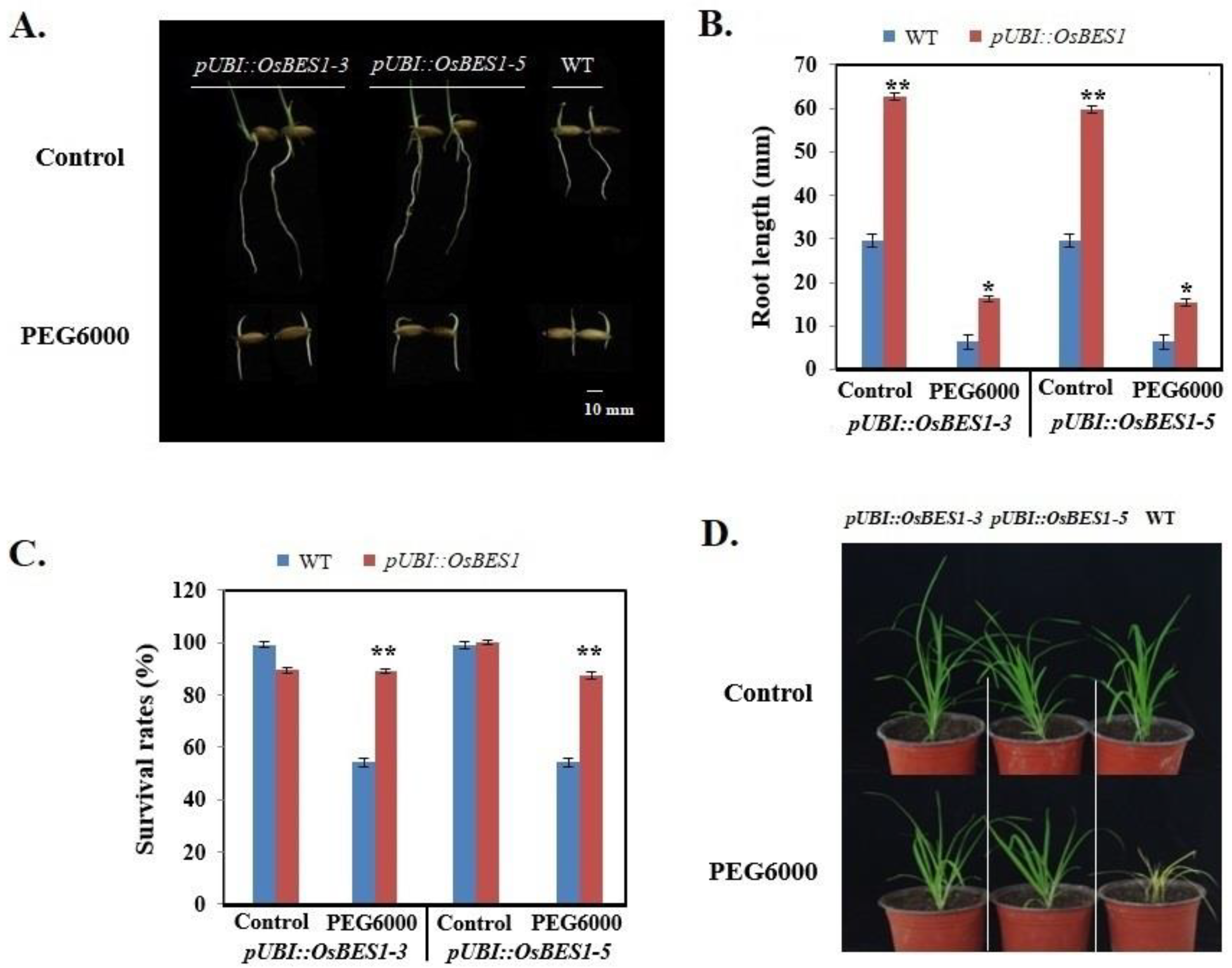
2.6. Functional Verification of Transgenic Plants
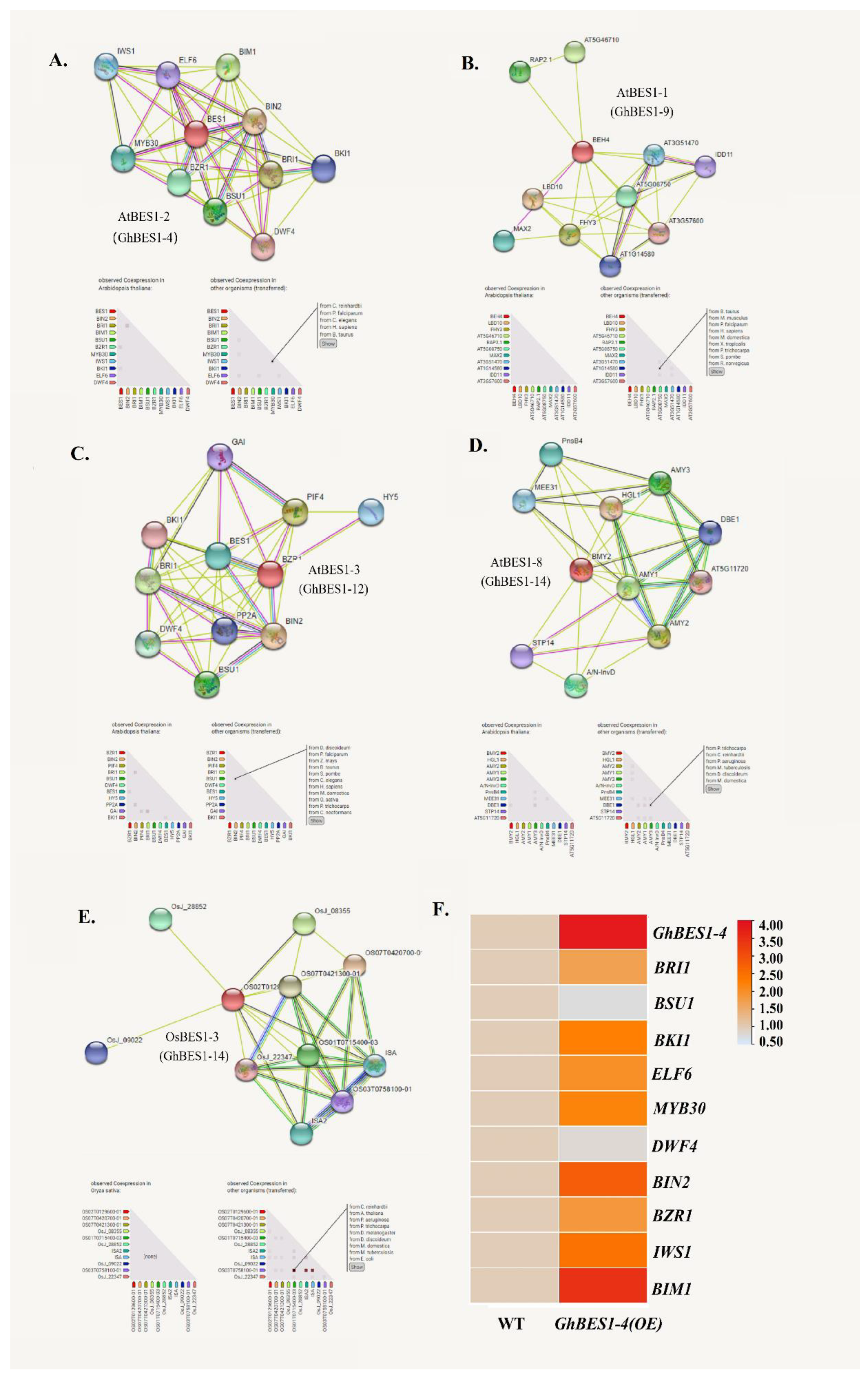
2.7. Construction of Protein Interaction Network and Gene Co-Expression
3. Discussion
4. Materials and Methods
4.1. Sequence Retrieval and Gene Identification
4.2. Bioinformatics and Protein Interaction Analysis
4.3. Expression Data, Plant Materials, and Abiotic Stress Treatment
4.4. Functional Verification of Transgenic Plants
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yin, Y.; Wang, Z.-Y.; Mora-Garcia, S.; Li, J.; Yoshida, S.; Asami, T.; Chory, J. BES1 Accumulates in the Nucleus in Response to Brassinosteroids to Regulate Gene Expression and Promote Stem Elongation Extensive Loss-of-Function Genetic Screens for BR-Insensitive Dwarf Mutants Have Identified Mutations; Elsevier: Amsterdam, The Netherlands, 2002; Volume 109. [Google Scholar]
- Kono, A.; Yin, Y. Updates on BES1/BZR1 regulatory networks coordinating plant growth and stress responses. Front. Plant Sci. 2020, 11, 617162. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Peng, P.; Schmitz, R.J.; Decker, A.D.; Tax, F.E.; Li, J. Two putative BIN2 substrates are nuclear components of brassinosteroid signaling. Plant Physiol. 2002, 130, 1221–1229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Tang, J.; Hu, R.; Wu, P.; Hou, X.-L.; Song, X.-M.; Xiong, A.-S. Genome-wide analysis of the BES1 transcription factor family in Chinese cabbage (Brassica rapa ssp. pekinensis). Plant Growth Regul. 2016, 80, 291–301. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, Z.T.; Li, M.; Wei, X.Z.; Li, X.J.; Li, B.Y.; Li, X.-B. Cotton (Gossypium hirsutum) 14-3-3 proteins participate in regulation of fibre initiation and elongation by modulating brassinosteroid signalling. Plant Biotechnol. J. 2015, 13, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Lian, H. Phyb interacts with BES1 to regulate brassinosteroid signaling in Arabidopsis. In Phyb-BES1 Interaction Represses BR Signaling Pathway N.D.; Oxford Academic: Oxford, UK, 2019. [Google Scholar] [CrossRef]
- Zhang, D.W. Sumoylation of BRI1-EMS-SUPPRESSOR 1 (BES1) by the SUMO E3 ligase SIZ1 negatively regulates brassinosteroids signaling in Arabidopsis thaliana. In Sumoylation Negatively Regulates BR Signaling N.D.; Oxford Academic: Oxford, UK, 2019. [Google Scholar] [CrossRef]
- Ruan, J.; Chen, H.; Zhu, T.; Yu, Y.; Lei, Y.; Yuan, L.; Liu, J.; Wang, Z.-Y.; Kuang, J.-F.; Lu, W.-J.; et al. Brassinosteroids repress the seed maturation program during the seed-to-seedling transition. Plant Physiol. 2021, 186, 534–548. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.G.; Won, J.H.; Choi, Y.R.; Lee, K.; Seo, P.J. Brassinosteroids regulate circadian oscillation via the BES1/TPL-CCA1/LHY module in Arabidopsisthaliana. IScience 2020, 23, 101528. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhao, C.; Gao, Y.; Xu, Y.; Wang, S.; Li, C.; Xie, Y.; Chen, P.; Yang, P.; Yuan, L.; et al. A multifaceted module of BRI1 Ethylmethane Sulfonate Supressor1 (BES1)-MYB88 in growth and stress tolerance of apple. Plant Physiol. 2021, 185, 1903–1923. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Dou, L.; Gong, Z.; Wang, X.; Mao, T. BES1 hinders ABSCISIC Acid Insensitive5 and promotes seed germination in Arabidopsis. New Phytol. 2019, 221, 908–918. [Google Scholar] [CrossRef] [Green Version]
- Sun, F.; Ding, L.; Feng, W.; Cao, Y.; Lu, F.; Yang, Q.; Li, W.; Lu, Y.; Shabek, N.; Fu, F.; et al. Maize transcription factor ZmBES1/BZR1-5 positively regulates kernel size. J. Exp. Bot. 2021, 72, 1714–1726. [Google Scholar] [CrossRef]
- Xie, Z.; Nolan, T.; Jiang, H.; Tang, B.; Zhang, M.; Li, Z.; Yin, Y. The AP2/ERF transcription factor TINY modulates brassinosteroid-regulated plant growth and drought responses in arabidopsis. Plant Cell 2019, 31, 1788–1806. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Sun, S.; Wang, X. Regulation of shoot branching by strigolactones and brassinosteroids: Conserved and specific functions of Arabidopsis BES1 and rice BZR1. Mol. Plant 2020, 13, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Khaliq, A.; Lu, S.; Xie, M.; Ma, Z.; Mao, J.; Chen, B. Genome-wide identification and characterization of the BES1 gene family in apple (Malus domestica). Plant Biol. 2020, 22, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Q.; Sun, F.A.; Feng, W.Q.; Lu, F.Z.; Li, W.C.; Fu, F.L. The BES1/BZR1 transcription factors regulate growth, development and stress resistance in plants. Yi Chuan Hered. 2019, 41, 206–214. [Google Scholar] [CrossRef]
- Song, X.; Ma, X.; Li, C.; Hu, J.; Yang, Q.; Wang, T.; Wang, L.; Wang, J.; Guo, D.; Ge, W.; et al. Comprehensive analyses of the BES1 gene family in Brassica napus and examination of their evolutionary pattern in representative species. BMC Genom. 2018, 19, 346. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Bai, M.Y.; Oh, E.; Zhu, J.Y. Brassinosteroid signaling network and regulation of photomorphogenesis. Annu. Rev. Genet. 2012, 46, 701–724. [Google Scholar] [CrossRef]
- Iwamoto, M.; Maekawa, M.; Saito, A.; Higo, H.; Higo, K. Evolutionary relationship of plant catalase genes inferred from exon-intron structures: Isozyme divergence after the separation of monocots and dicots. Theor. Appl. Genet. 1998, 97, 9–19. [Google Scholar] [CrossRef]
- Lv, B.; Tian, H.; Zhang, F.; Liu, J.; Lu, S.; Bai, M.; Li, C.; Ding, Z. Brassinosteroids regulate root growth by controlling reactive oxygen species homeostasis and dual effect on ethylene synthesis in Arabidopsis. PLoS Genet. 2018, 14, e1007144. [Google Scholar] [CrossRef]
- Martins, S.; Montiel-Jorda, A.; Cayrel, A.; Huguet, S.; Roux, C.P.L.; Ljung, K.; Vert, G. Brassinosteroid signaling-dependent root responses to prolonged elevated ambient temperature. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.-Y.; Nakano, T.; Gendron, J.; He, J.; Chen, M.; Vafeados, D.; Yang, Y.; Fujioka, S.; Yoshida, S.; Asami, T.; et al. Nuclear-Localized BZR1 Mediates Brassinosteroid-Induced Growth and Feedback Suppression of Brassinosteroid Biosynthesis; dwarfism, Sterility, and Photomorphogenesis in the Dark (Li et al Molecular Genetic Studies of BR-Insensitive Mutants in Arabidopsis Have Led to the Identification of a Receptor Kinase, BRI1, as a Key Signaling Component in the BR. Vol. 2. Li and Chory; Elsevier: Amsterdam, The Netherlands, 2002. [Google Scholar]
- Mei, Y.; Yang, X.; Huang, C.; Zhang, X.; Zhou, X. Tomato leaf curl Yunnan virus-encoded C4 induces cell division through enhancing stability of Cyclin D 1.1 via impairing NbSKη-mediated phosphorylation in Nicotiana benthamiana. PLoS Pathog. 2018, 14, e1006789. [Google Scholar] [CrossRef] [Green Version]
- Tian, X.; Xuan, L.; Liu, B.; Hu, T.; Wang, C.; Wang, X. Effects of heterologous expression of Populus euphratica brassinosteroids biosynthetic enzyme genes CPD (PeCPD) and DWF4 (PeDWF4) on tissue dedifferentiation and growth of Arabidopsis thaliana seedlings. Plant Cell Tissue Organ Cult. 2018, 132, 111–121. [Google Scholar] [CrossRef]
- Sahni, S.; Prasad, B.D.; Liu, Q.; Grbic, V.; Sharpe, A.; Singh, S.P.; Krishna, P. Overexpression of the brassinosteroid biosynthetic gene DWF4 in Brassica napus simultaneously increases seed yield and stress tolerance. Sci. Rep. 2016, 6, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yu, X.; Thompson, A.; Guo, M.; Yoshida, S.; Asami, T.; Chory, J.; Yin, Y. Arabidopsis MYB30 is a direct target of BES1 and cooperates with BES1 to regulate brassinosteroid-induced gene expression. Plant J. Cell Mol. Biol. 2009, 58, 275–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raffaele, S.; Vailleau, F.; Léger, A.; Joubès, J.; Miersch, O.; Huard, C.; Blée, E.; Mongrand, S.; Domergue, F.; Roby, D. A MYB transcription factor regulates very-long-chain fatty acid biosynthesis for activation of the hypersensitive cell death response in Arabidopsis. Plant Cell 2008, 20, 752–767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raffaele, S.; Rivas, S.; Roby, D. An essential role for salicylic acid in AtMYB30-mediated control of the hypersensitive cell death program in Arabidopsis. FEBS Lett. 2006, 580, 3498–3504. [Google Scholar] [CrossRef] [PubMed]
- Daniel, X.; Lacomme, C.; Morel, J.; Roby, D. A novel myb oncogene homologue in Arabidopsis thaliana related to hypersensitive cell death. Plant J. Cell Mol. Biol. 1999, 20, 57–66. [Google Scholar] [CrossRef] [Green Version]
- Hu, B.; Jin, J.; Guo, A.Y.; Zhang, H.; Luo, J.; Gao, G. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics 2015, 31, 1296–1297. [Google Scholar] [CrossRef] [Green Version]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef] [Green Version]
- Winter, D.; Vinegar, B.; Nahal, H.; Ammar, R.; Wilson, G.V.; Provart, N.J. An “electronic fluorescent pictograph” browser for exploring and analyzing large-scale biological data sets. PLoS ONE 2007, 2, e718. [Google Scholar] [CrossRef]
- Zhuang, X.; Jiang, J.; Li, J.; Ma, Q.; Xu, Y.; Xue, Y.; Xu, Z.; Chong, K. Over-expression of OsAGAP, an ARF-GAP, interferes with auxin influx, vesicle trafficking and root development. Plant J. 2006, 48, 581–591. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.H.; Wang, Y.G.; Wang, S.P.; Li, H.J.; Xin, Q.G. Improved zinc tolerance of tobacco by transgenic expression of an allene oxide synthase gene from hexaploid wheat. Acta Physiol. Plant. 2014, 36, 2433–2440. [Google Scholar] [CrossRef]
- Shabala, S.N.; Shabala, S.I.; Martynenko, A.I.; Babourina, O.; Newman, I.A. Salinity effect on bioelectric activity, growth, Na+ accumulation and chlorophyll fluorescence of maize leaves: A comparative survey and prospects for screening. Aust. J. Plant Physiol. 1998, 25, 609–616. [Google Scholar] [CrossRef]



| Lineage | Organism | Genome Size (Mb) | Gene Member | Number/Mb |
|---|---|---|---|---|
| Glaucophytes | Cyanophora paradoxa | 0.14 | 0 | 0 |
| Rhodophytes | Galdieria sulphuraria | 13.7 | 0 | 0 |
| Cyanidioschyzon merolae | 16 | 0 | 0 | |
| Chlorophytes | Chlamydomonas reinhardtii | 121 | 0 | 0 |
| Volvox carteri | 120 | 0 | 0 | |
| Coccomyxa subellipsoidea C-169 | 48.8 | 0 | 0 | |
| Chlorella variabilis NC64A | 46.2 | 0 | 0 | |
| Micromonas pusilla RCC299 | 21 | 0 | 0 | |
| Ostreococcus lucimarinus | 13.2 | 0 | 0 | |
| Ostreococcus tauri | 12 | 0 | 0 | |
| Bryophytes | Physcomitrella patens | 511 | 6 | 0.0059 |
| Marchantia polymorpha | 216.2 | 4 | 0.003 | |
| Lycophytes | Selaginella moellendorffii | 100 | 4 | 0.0400 |
| Gymnosperms | Picea abies | 19,600 | 5 | 0.003 |
| Picea sitchensis | 5500 | 4 | 0.002 | |
| Bacal angiosperm | Amborella trichopoda | 699.2 | 9 | 0.005 |
| Nymphaea colorata | 404.533 | 9 | 0.005 | |
| Monocots | Zea mays | 2365 | 8 | 0.0034 |
| Sorghum bicolor | 732.2 | 8 | 0.0109 | |
| Oryza sativa | 430 | 6 | 0.003 | |
| Brachypodium distachyum | 272 | 8 | 0.004 | |
| Eudicots | Arabidopsis thaliana | 125 | 8 | 0.0640 |
| Aquilegia coerulea | 306.5 | 4 | 0.0131 | |
| Gossypium raimondii | 775 | 11 | 0.0142 | |
| Mimulus guttatus | 430 | 8 | 0.0186 | |
| Gossypium hirsutum | 2430 | 22 | 0.0273 | |
| Gossypium arboretum | 752 | 11 | 0.0160 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Z.; Liu, X.; Zhu, W.; Lin, H.; Chen, X.; Li, Y.; Ye, W.; Yin, Z. Molecular Traits and Functional Exploration of BES1 Gene Family in Plants. Int. J. Mol. Sci. 2022, 23, 4242. https://doi.org/10.3390/ijms23084242
Sun Z, Liu X, Zhu W, Lin H, Chen X, Li Y, Ye W, Yin Z. Molecular Traits and Functional Exploration of BES1 Gene Family in Plants. International Journal of Molecular Sciences. 2022; 23(8):4242. https://doi.org/10.3390/ijms23084242
Chicago/Turabian StyleSun, Zhenting, Xingzhou Liu, Weidong Zhu, Huan Lin, Xiugui Chen, Yan Li, Wuwei Ye, and Zujun Yin. 2022. "Molecular Traits and Functional Exploration of BES1 Gene Family in Plants" International Journal of Molecular Sciences 23, no. 8: 4242. https://doi.org/10.3390/ijms23084242
APA StyleSun, Z., Liu, X., Zhu, W., Lin, H., Chen, X., Li, Y., Ye, W., & Yin, Z. (2022). Molecular Traits and Functional Exploration of BES1 Gene Family in Plants. International Journal of Molecular Sciences, 23(8), 4242. https://doi.org/10.3390/ijms23084242






