Effects of Fe and Mn Deficiencies on the Root Protein Profiles of Tomato (Solanum lycopersicum) Using Two-Dimensional Electrophoresis and Label-Free Shotgun Analyses
Abstract
:1. Introduction
2. Results
2.1. Effects of Fe and Mn Deficiencies on Leaf Pigments and Metal Concentrations
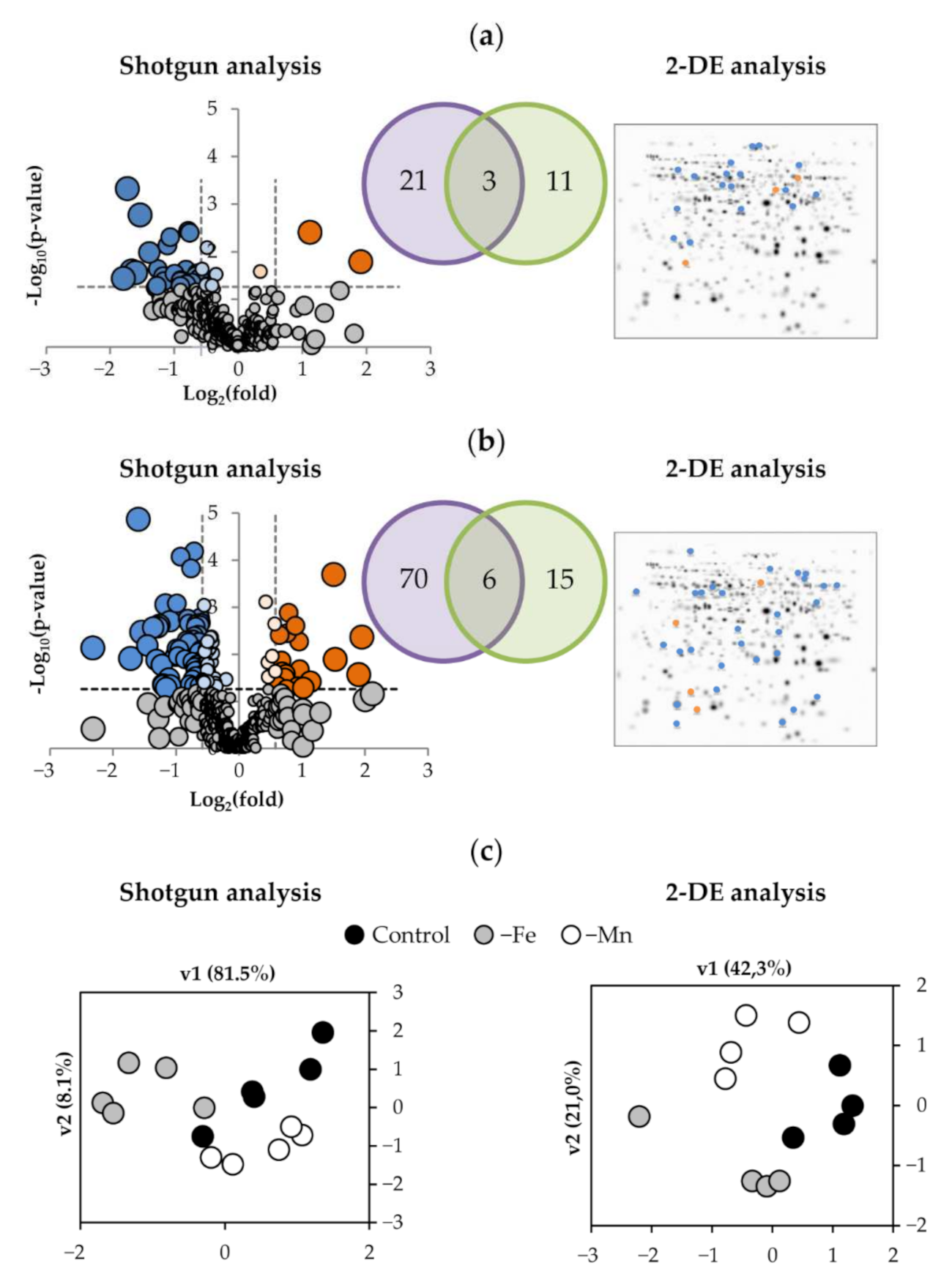
2.2. Identification of Root Proteins by Shotgun and 2-DE
2.3. Effect of Fe-Deficiency and Mn-Deficiency on the Root Proteome
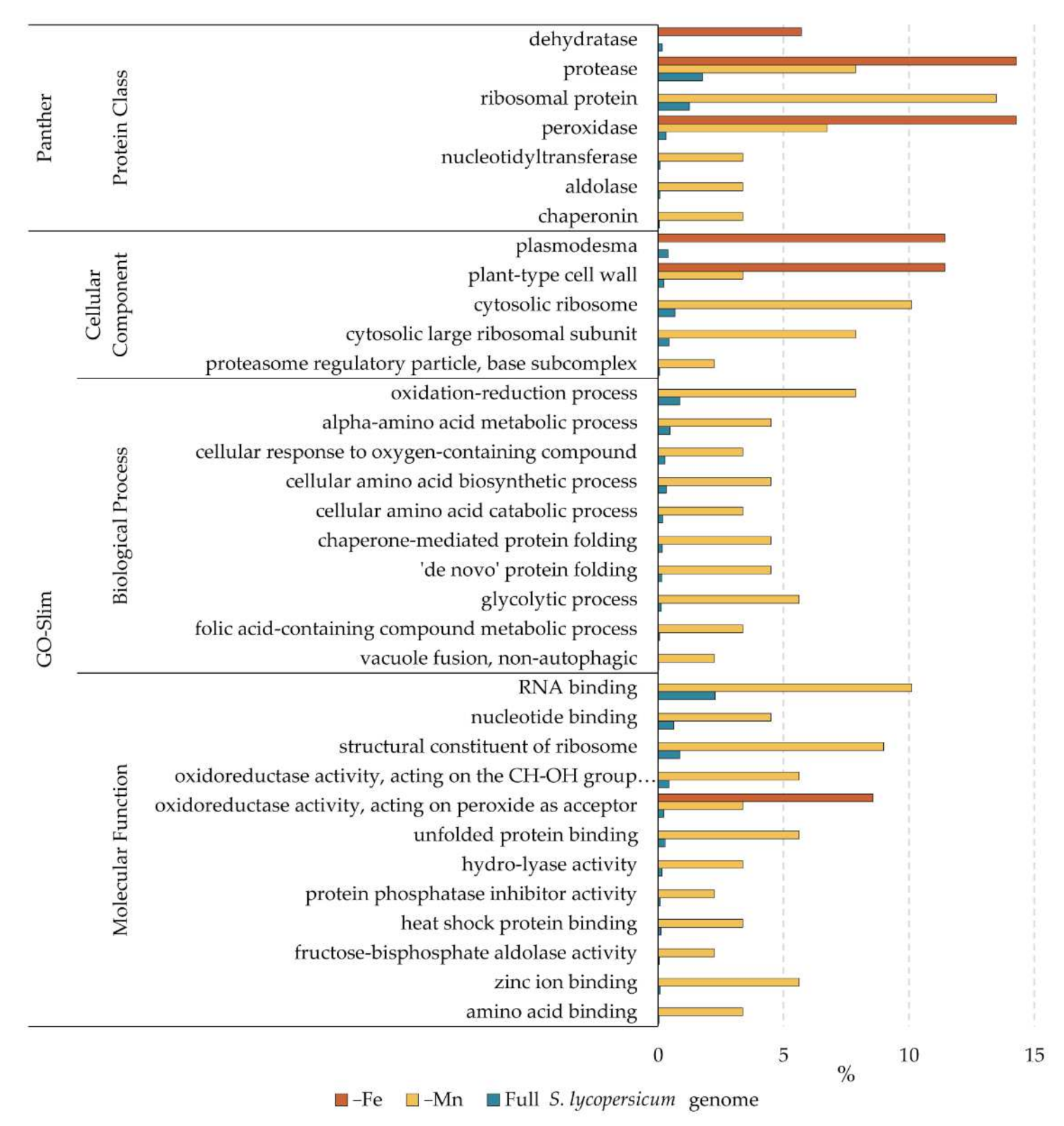
2.4. GO-Enrichment Analysis of Differential Proteins
3. Discussion
3.1. The Cell Wall Is a Key Component to Fe and Mn Deficiencies
3.2. Contrasting Changes in Protein Profiles with Fe and Mn Deficiencies
3.2.1. Oxidoreductases and Oxidative Stress Responses
3.2.2. Coumarin Production
3.2.3. Protein and N Metabolism
3.2.4. Different Ways to Cope with a Common Problem: The Lack of Energy
3.3. A Possible Role of Extracellular Proteins in the Fe Deficiency Response
3.4. Mn Deficiency and Pathogen Response
4. Materials and Methods
4.1. Plant Material and Sampling
4.2. Experimental Design
4.3. Mineral Analysis, Chlorophyll Estimation and Photosynthetic Pigment Analysis
4.4. Root Iron Reductase Activity
4.5. Protein Extraction
4.6. Label-Free Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS)
4.7. Protein 2-DE Separation of Root Samples
4.8. Gel Image and Statistical Analysis
4.9. Protein in Gel Digestion
4.10. Protein Identification by Nanoliquid Chromatography-Tandem Mass Spectrometry (nLC−ESI−MS/MS)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marschner, P. (Ed.) Mineral Nutrition of Higher Plants; Academic Press: Cambridge, MA, USA, 2012; ISBN 978-0-12-384905-2. [Google Scholar]
- Goussias, C.; Boussac, A.; Rutherford, A.W. Photosystem II and photosynthetic oxidation of water: An overview. Philos. Trans. R. Soc. B Biol. Sci. 2002, 357, 1369–1381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grotz, N.; Guerinot, M.L. Molecular aspects of Cu, Fe and Zn homeostasis in plants. Biochim. Biophys. Acta BBA-Mol. Cell Res. 2006, 1763, 595–608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vert, G.; Grotz, N.; Dédaldéchamp, F.; Gaymard, F.; Guerinot, M.L.; Briat, J.-F.; Curie, C. IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 2002, 14, 1223–1233. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, T.; Nishizawa, N.K. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Biol. 2012, 63, 131–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, M.D.; Kropat, J.; Tottey, S.; Del Campo, J.A.; Merchant, S.S. Manganese deficiency in Chlamydomonas results in loss of photosystem II and MnSOD function, sensitivity to peroxides, and secondary phosphorus and iron deficiency. Plant Physiol. 2007, 143, 263–277. [Google Scholar] [CrossRef] [Green Version]
- Millaleo, R.; Reyes-Diaz, M.; Ivanov, A.; Mora, M.; Alberdi, M. Manganese as essential and toxic element for plants: Transport, accumulation and resistance mechanisms. J. Soil Sci. Plant Nutr. 2010, 10, 470–481. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.A.; Guerinot, M.L. Mining iron: Iron uptake and transport in plants. FEBS Lett. 2007, 581, 2273–2280. [Google Scholar] [CrossRef] [Green Version]
- Pallotta, M.A.; Graham, R.D.; Langridge, P.; Sparrow, D.H.B.; Barker, S.J. RFLP mapping of manganese efficiency in barley. Theor. Appl. Genet. 2000, 101, 1100–1108. [Google Scholar] [CrossRef]
- Hebbern, C.A.; Pedas, P.; Schjoerring, J.K.; Knudsen, L.; Husted, S. Genotypic differences in manganese efficiency: Field experiments with winter barley (Hordeum vulgare L.). Plant Soil 2005, 272, 233–244. [Google Scholar] [CrossRef]
- George, T.S.; French, A.; Brown, L.K.; Karley, A.J.; White, P.; Ramsay, L.; Daniell, T. Genotypic variation in the ability of landraces and commercial cereal varieties to avoid manganese deficiency in soils with limited manganese availability: Is there a role for root-exuded phytases? Physiol. Plant. 2014, 151, 243–256. [Google Scholar] [CrossRef]
- Korcak, R. Iron deficiency chlorosis. In Horticultural Reviews; John and Wiley and Sons: Hoboken, NJ, USA, 2011; Volume 9, pp. 133–186. ISBN 9781118060827. [Google Scholar]
- Sanz, M.; Cavero, J.; Abadia, J. Iron chlorosis in the Ebro River basin, Spain. J. Plant Nutr. 1992, 15, 1971–1981. [Google Scholar] [CrossRef]
- Tagliavini, M.; Zavalloni, C.; Rombolà, A.D.; Quartieri, M.; Malaguti, D.; Mazzanti, F.; Millard, P.; Marangoni, B. Mineral nutrient partitioning to fruits of deciduous trees. Acta Hortic. 2000, 512, 131–140. [Google Scholar] [CrossRef]
- Hansen, N.C.; Hopkins, B.G.; Ellsworth, J.W.; Jolley, V.D. Iron Nutrition in Field Crops BT-Iron Nutrition in Plants and Rhizospheric Microorganisms; Barton, L.L., Abadia, J., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 23–59. ISBN 978-1-4020-4743-5. [Google Scholar]
- Colangelo, E.P.; Guerinot, M.L. The essential basic helix-loop-helix protein FIT1 is required for the iron deficiency response. Plant Cell 2004, 16, 3400–3412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Connorton, J.M.; Balk, J.; Rodríguez-Celma, J. Iron homeostasis in plants—A brief overview. Metallomics 2017, 9, 813–823. [Google Scholar] [CrossRef] [Green Version]
- Ling, H.-Q.; Bauer, P.; Bereczky, Z.; Keller, B.; Ganal, M. The tomato fer gene encoding a bHLH protein controls iron-uptake responses in roots. Proc. Natl. Acad. Sci. USA 2002, 99, 13938–13943. [Google Scholar] [CrossRef] [Green Version]
- Brumbarova, T.; Bauer, P. Iron-mediated control of the basic helix-loop-helix protein FER, A regulator of iron uptake in tomato. Plant Physiol. 2005, 137, 1018–1026. [Google Scholar] [CrossRef] [Green Version]
- Yuan, Y.X.; Zhang, J.; Wang, D.W.; Ling, H.Q. AtbHLH29 of Arabidopsis thaliana is a functional ortholog of tomato FER involved in controlling iron acquisition in strategy I plants. Cell Res. 2005, 15, 613–621. [Google Scholar] [CrossRef] [Green Version]
- Bauer, P.; Ling, H.-Q.; Guerinot, M.L. FIT, the FER-LIKE IRON DEFICIENCY INDUCED TRANSCRIPTION FACTOR in Arabidopsis. Plant Physiol. Biochem. 2007, 45, 260–261. [Google Scholar] [CrossRef]
- Connolly, E.L.; Campbell, N.H.; Grotz, N.; Prichard, C.L.; Guerinot, M.L. Overexpression of the FRO2 ferric chelate reductase confers tolerance to growth on low iron and uncovers posttranscriptional control. Plant Physiol. 2003, 133, 1102–1110. [Google Scholar] [CrossRef] [Green Version]
- Henriques, R.; Jásik, J.; Klein, M.; Martinoia, E.; Feller, U.; Schell, J.; Pais, M.S.; Koncz, C. Knock-out of Arabidopsis metal transporter gene IRT1 results in iron deficiency accompanied by cell differentiation defects. Plant Mol. Biol. 2002, 50, 587–597. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Celma, J.; Lin, W.-D.; Fu, G.-M.; Abadía, J.; López-Millán, A.-F.; Schmidt, W. Mutually exclusive alterations in secondary metabolism are critical for the uptake of insoluble iron compounds by Arabidopsis and Medicago truncatula. Plant Physiol. 2013, 162, 1473–1485. [Google Scholar] [CrossRef] [PubMed]
- Fourcroy, P.; Sisó-Terraza, P.; Sudre, D.; Savirón, M.; Reyt, G.; Gaymard, F.; Abadía, A.; Abadia, J.; Álvarez-Fernández, A.; Briat, J. Involvement of the ABCG 37 transporter in secretion of scopoletin and derivatives by Arabidopsis roots in response to iron deficiency. New Phytol. 2014, 201, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Schmid, N.B.; Giehl, R.F.; Döll, S.; Mock, H.-P.; Strehmel, N.; Scheel, D.; Kong, X.; Hider, R.C.; von Wirén, N. Feruloyl-CoA 6’-Hydroxylase1-dependent coumarins mediate iron acquisition from alkaline substrates in Arabidopsis. Plant Physiol. 2014, 164, 160–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, T.J.W.; Perry, P.J.; Ciani, S.; Pandian, S.; Schmidt, W. Manganese deficiency alters the patterning and development of root hairs in Arabidopsis. J. Exp. Bot. 2008, 59, 3453–3464. [Google Scholar] [CrossRef] [Green Version]
- Yamaji, N.; Sasaki, A.; Xia, J.X.; Yokosho, K.; Ma, J.F. A node-based switch for preferential distribution of manganese in rice. Nat. Commun. 2013, 4, 2442. [Google Scholar] [CrossRef]
- Cailliatte, R.; Schikora, A.; Briat, J.-F.; Mari, S.; Curie, C. High-affinity manganese uptake by the metal transporter NRAMP1 is essential for Arabidopsis growth in low manganese conditions. Plant Cell 2010, 22, 904–917. [Google Scholar] [CrossRef] [Green Version]
- Socha, A.L.; Guerinot, M.L. Mn-euvering manganese: The role of transporter gene family members in manganese uptake and mobilization in plants. Front. Plant Sci. 2014, 5, 106. [Google Scholar] [CrossRef] [Green Version]
- Alejandro, S.; Höller, S.; Meier, B.; Peiter, E. Manganese in plants: From acquisition to subcellular allocation. Front. Plant Sci. 2020, 11, 300. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Celma, J.; Tsai, Y.-H.; Wen, T.-N.; Wu, Y.-C.; Curie, C.; Schmidt, W. Systems-wide analysis of manganese deficiency-induced changes in gene activity of Arabidopsis roots. Sci. Rep. 2016, 6, 35846. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.-Q.; Chen, J.-G.; Chen, Z.-R.; An, D.; Lv, Q.-Y.; Han, M.-L.; Wang, Y.-L.; Salt, D.E.; Chao, D.-Y. A new vesicle trafficking regulator CTL1 plays a crucial role in ion homeostasis. PLoS Biol. 2017, 15, e2002978. [Google Scholar] [CrossRef]
- Agorio, A.; Giraudat, J.; Bianchi, M.W.; Marion, J.; Espagne, C.; Castaings, L.; Lelièvre, F.; Curie, C.; Thomine, S.; Merlot, S. Phosphatidylinositol 3-phosphate-binding protein AtPH1 controls the localization of the metal transporter NRAMP1 in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, E3354–E3363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rai, S.; Singh, P.K.; Mankotia, S.; Swain, J.; Satbhai, S.B. Iron homeostasis in plants and its crosstalk with copper, zinc, and manganese. Plant Stress 2021, 1, 100008. [Google Scholar] [CrossRef]
- Gayomba, S.R.; Ezhai, Z.; Ejung, H.-I.; Vatamaniuk, O.K. Local and systemic signaling of iron status and its interactions with homeostasis of other essential elements. Front. Plant Sci. 2015, 6, 716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers, E.E.; Eide, D.J.; Guerinot, M.L. Altered selectivity in an Arabidopsis metal transporter. Proc. Natl. Acad. Sci. USA 2000, 97, 12356–12360. [Google Scholar] [CrossRef] [Green Version]
- Pedas, P.; Ytting, C.K.; Fuglsang, A.T.; Jahn, T.P.; Schjoerring, J.K.; Husted, S. Manganese efficiency in barley: Identification and characterization of the metal ion transporter HvIRT1. Plant Physiol. 2008, 148, 455–466. [Google Scholar] [CrossRef] [Green Version]
- Lanquar, V.; Ramos, M.S.; Lelièvre, F.; Barbier-Brygoo, H.; Krieger-Liszkay, A.; Kraemer, U.; Thomine, S. Export of vacuolar manganese by AtNRAMP3 and AtNRAMP4 is required for optimal photosynthesis and growth under manganese deficiency. Plant Physiol. 2010, 152, 1986–1999. [Google Scholar] [CrossRef] [Green Version]
- Ceballos-Laita, L.; Gutierrez-Carbonell, E.; Takahashi, D.; Abadía, A.; Uemura, M.; Abadia, J.; López-Millán, A.F. Effects of Fe and Mn deficiencies on the protein profiles of tomato (Solanum lycopersicum) xylem sap as revealed by shotgun analyses. J. Proteom. 2018, 170, 117–129. [Google Scholar] [CrossRef] [Green Version]
- Carrasco-Gil, S.; Rios, J.J.; Álvarez-Fernández, A.; Abadía, A.; García-Mina, J.M.; Abadia, J. Effects of individual and combined metal foliar fertilisers on iron—and manganese-deficient Solanum lycopersicum plants. Plant Soil 2016, 402, 27–45. [Google Scholar] [CrossRef] [Green Version]
- Nishizono, H.; Ichikawa, H.; Suziki, S.; Ishii, F. The role of the root cell wall in the heavy metal tolerance of Athyrium yokoscense. Plant Soil 1987, 101, 15–20. [Google Scholar] [CrossRef]
- Parrotta, L.; Guerriero, G.; Sergeant, K.; Cai, G.; Hausman, J.-F. Target or barrier? The cell wall of early- and later-diverging plants vs cadmium toxicity: Differences in the response mechanisms. Front. Plant Sci. 2015, 6, 133. [Google Scholar] [CrossRef] [Green Version]
- Krzesłowska, M. The cell wall in plant cell response to trace metals: Polysaccharide remodeling and its role in defense strategy. Acta Physiol. Plant. 2011, 33, 35–51. [Google Scholar] [CrossRef] [Green Version]
- Celus, M.; Kyomugasho, C.; Van Loey, A.M.; Grauwet, T.; Hendrickx, M.E. Influence of pectin structural properties on interactions with divalent cations and its associated functionalities. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1576–1594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Celma, J.; Lattanzio, G.; Grusak, M.A.; Abadía, A.; Abadía, J.; López-Millán, A.-F. Root responses of Medicago truncatula plants grown in two different iron deficiency conditions: Changes in root protein profile and riboflavin biosynthesis. J. Proteome Res. 2011, 10, 2590–2601. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Celma, J.; Lattanzio, G.; Jiménez, S.; Briat, J.-F.; Abadía, J.; Abadía, A.; Gogorcena, Y.; López-Millán, A.-F. Changes induced by Fe deficiency and Fe resupply in the root protein profile of a peach-almond hybrid rootstock. J. Proteome Res. 2013, 12, 1162–1172. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wu, X.-D.; Hao, S.-T.; Wang, X.-J.; Ling, H.-Q. Proteomic response to iron deficiency in tomato root. Proteomics 2008, 8, 2299–2311. [Google Scholar] [CrossRef]
- Rellán-Álvarez, R.; Andaluz, S.; Rodríguez-Celma, J.; Wohlgemuth, G.; Zocchi, G.; Álvarez-Fernández, A.; Fiehn, O.; López-Millán, A.F.; Abadía, J. Changes in the proteomic and metabolic profiles of Beta vulgaris root tips in response to iron deficiency and resupply. BMC Plant Biol. 2010, 10, 120. [Google Scholar] [CrossRef] [Green Version]
- Santos, C.S.; Ozgur, R.; Uzilday, B.; Turkan, I.; Roriz, M.; Rangel, A.O.; Carvalho, S.M.; Vasconcelos, M.W. Understanding the role of the antioxidant system and the tetrapyrrole cycle in iron deficiency chlorosis. Plants 2019, 8, 348. [Google Scholar] [CrossRef] [Green Version]
- Babalakova, N.; Salama, Z.; Rocheva, S.; Fouly, M. El Root redox-system activities and H2O2–metabolizing enzymes in cucumber cultivars grown in conditions of iron deficit. J. Food Agric. Environ. 2011, 9, 533–537. [Google Scholar]
- Tsai, H.-H.; Rodríguez-Celma, J.; Lan, P.; Wu, Y.-C.; Vélez-Bermúdez, I.C.; Schmidt, W. Scopoletin 8-Hydroxylase-mediated fraxetin production is crucial for iron mobilization. Plant Physiol. 2018, 177, 194–207. [Google Scholar] [CrossRef] [Green Version]
- Queiroz, C.; Lopes, M.L.M.; Fialho, E.; Valente-Mesquita, V.L. Polyphenol oxidase: Characteristics and mechanisms of browning control. Food Rev. Int. 2008, 24, 361–375. [Google Scholar] [CrossRef]
- Rodríguez-Celma, J.; Pan, I.C.; Li, W.D.; Lan, P.D.; Buckhout, T.J.; Schmidt, W. The transcriptional response of Arabidopsis leaves to Fe deficiency. Front. Plant Sci. 2013, 4, 276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rengel, Z.; Graham, R.D.; Pedler, J.F. Manganese nutrition and accumulation of phenolics and lignin as related to differential resistance of wheat genotypes to the take-all fungus. Plant Soil 1993, 151, 255–263. [Google Scholar] [CrossRef]
- Abadía, J.; Abadía, A. Iron and Plant Pigments. In Iron Chelation in Plants and Soil Microorganisms; Barton, L.L., Hemming, B.C., Eds.; Academic Press: San Diego, CA, USA, 1993; pp. 327–343. ISBN 978-0-12-079870-4. [Google Scholar]
- Larbi, A.; Abadía, A.; Morales, F.; Abadia, J. Fe resupply to Fe-deficient sugar beet plants leads to rapid changes in the violaxanthin cycle and other photosynthetic characteristics without significant de novo chlorophyll synthesis. Photosynth. Res. 2004, 79, 59–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bienfait, H.F.; Bino, R.J.; van der Bliek, A.M.; Duivenvoorden, J.F.; Fontaine, J.M. Characterization of ferric reducing activity in roots of Fe-deficient Phaseolus vulgaris. Physiol. Plant. 1983, 59, 196–202. [Google Scholar] [CrossRef]
- Li, B.; Takahashi, D.; Kawamura, Y.; Uemura, M. Comparison of Plasma membrane proteomic changes of Arabidopsis suspension-cultured cells (T87 Line) after cold and ABA treatment in association with freezing tolerance development. Plant Cell Physiol. 2012, 53, 543–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, D.; Li, B.; Nakayama, T.; Kawamura, Y.; Uemura, M. Shotgun Proteomics of Plant Plasma Membrane and Mi-Crodomain Proteins Using nano-LC-MS/MS. BT-Plant Proteomics: Methods and Protocols; Jorrin-Novo, J.V., Komatsu, S., Weckwerth, W., Wienkoop, S., Eds.; Humana Press: Totowa, NJ, USA, 2014; pp. 481–498. ISBN 978-1-62703-631-3. [Google Scholar]
- Ceballos-Laita, L.; Gutierrez-Carbonell, E.; Imai, H.; Abadía, A.; Uemura, M.; Abadía, J.; López-Millán, A.F. Effects of manganese toxicity on the protein profile of tomato (Solanum lycopersicum) roots as revealed by two complementary proteomic approaches, two-dimensional electrophoresis and shotgun analysis. J. Proteom. 2018, 185, 51–63. [Google Scholar] [CrossRef]
- Gutierrez-Carbonell, E.; Takahashi, D.; Lüthje, S.; González-Reyes, J.A.; Mongrand, S.; Contreras-Moreira, B.; Abadía, A.; Uemura, M.; Abadia, J.; López-Millán, A.F. A shotgun proteomic approach reveals that Fe deficiency causes marked changes in the protein profiles of plasma membrane and detergent-resistant microdomain preparations from Beta vulgaris roots. J. Proteome Res. 2016, 15, 2510–2524. [Google Scholar] [CrossRef] [Green Version]
- Vizcaíno, J.A.; Csordas, A.; del-Toro, N.; Dianes, J.A.; Griss, J.; Lavidas, I.; Mayer, G.; Perez-Riverol, Y.; Reisinger, F.; Ternent, T.; et al. 2016 update of the PRIDE database and its related tools. Nucleic Acids Res. 2016, 44, D447–D456. [Google Scholar] [CrossRef]


| Control | -Fe | -Mn | |
|---|---|---|---|
| SPAD | 42.4 ± 0.7 a | 13.6 ± 0.5 b | 29.6 ± 0.8 c |
| Total Chl (Chl a + Chl b) | 323.9 ± 14.1 a | 88.0 ± 13.5 b | 219.5 ± 11.3 c |
| Neoxanthin | 12.0 ± 0.6 a | 2.4 ± 0.2 b | 6.7 ± 0.4 c |
| Violaxanthin (V) | 17.6 ± 0.9 a | 4.6 ± 0.5 b | 10.0 ± 0.7 c |
| Taraxanthin | 0.2 ± 0.1 a | 0 a | 1.6 ± 0.2 b |
| Antheraxanthin (A) | 0.8 ± 0.2 a | 1.9 ± 0.1 b | 1.3 ± 0.2 c |
| Lutein | 44.8 ± 1.6 a | 15.3 ± 0.7 b | 24.5 ± 1.3 c |
| Zeaxanthin (Z) | 0.2 ± 0.1 a | 3.1 ± 0.4 b | 0.3 ± 0.1 a |
| (V + A + Z) | 18.6 ± 0.8 a | 9.5 ± 0.4 b | 11.5 ± 0.6 c |
| Chl a/Chl b ratio | 3.0 ± 0.1 a | 2.8 ± 0.1 a | 3.0 ± 0.1 a |
| (A + Z)/(V + A + Z) ratio | 0.06 ± 0.01 a | 0.53 ± 0.04 b | 0.14 ± 0.03 c |
| # | Accession | UniProtKB | Description | Fold SG | Fold 2D | Mn |
|---|---|---|---|---|---|---|
| Oxidoreductases (6) | ||||||
| 1 | Solyc07g052510.4.1 | A0A3Q7HDZ4 | peroxidase (TPX1) | −0.76 | −3.00 | - |
| 2 | Solyc12g005790.2.1 | A0A3Q7JR84 | peroxidase 27 | −0.82 | + | |
| 3 | Solyc04g071890.3.1 | A0A3Q7G7T0 * | peroxidase | −1.06 | ||
| 4 | Solyc10g076245.1.1 | A0A3Q7IJN4 * | peroxidase 70 | −1.40 | ||
| 5 | Solyc05g046000.4.1 | A0A3Q7GKW8 | peroxidase 27-like | −1.73 | ||
| 6 | Solyc10g076210.2.1 | A0A3Q7IJN4 * | peroxidase 1 | −3.00 | ||
| Carbohydrate Metabolism (4) | ||||||
| 7 | Solyc05g050800.3.1 | A0A3Q7GLU0 | phosphoglycerate mutase family | −0.65 | ||
| 8 | Solyc08g080140.4.1 | A0A3Q7HT77 | bifunctional dTDP-4-dehydrorhamnose 3,5-epimerase/reductase | −0.81 | - | |
| 9 | Solyc07g052350.3.1 | A0A3Q7HBK4 | aconitate hydratase | −1.17 | −3.00 | |
| 10 | Solyc06g073190.3.1 | Q42896 | fructokinase-like | −2.30 | ||
| Polysaccharide Metabolism (3) | ||||||
| 11 | Solyc12g098540.2.1 | A0A3Q7JEE6 * | apyrase | 1.91 | ||
| 12 | Solyc01g104950.4.1 | A0A3Q7EQN7 | beta-D-xylosidase 2 precursor | −0.90 | −1.14 | - |
| 13 | Solyc01g107590.3.1 | A0A3Q7ESC5 | cinnamyl alcohol dehydrogenase | −1.54 | - | |
| Protein Metabolism (10) | ||||||
| 14 | Solyc08g067100.2.1 | K4CLT6 | eukaryotic aspartyl protease family | −0.61 | ||
| 15 | Solyc05g013820.4.1 | A0A3Q7GHJ1 | proteasome subunit beta type-7-A | −0.73 | - | |
| 16 | Solyc08g079920.2.1 | A0A3Q7HVI4 * | P69f protein | −1.16 | ||
| 17 | Solyc03g019690.1.1 | A0A3Q7FGU5 | Kunitz-type protease inhibitor | −1.60 | ||
| 18 | Solyc08g079930.2.1 | A0A3Q7HVI4 * | subtilisin-like protease | −1.79 | ||
| 19 | Solyc02g081700.1.1 | A0A3Q7F6F6 | proteasome subunit alpha type | −3.00 | ||
| 20 | Solyc08g082820.4.1 | A0A3Q7HX02 | glucose-regulated protein 78 | 1.69 | ||
| 21 | Solyc01g028810.3.1 | A0A3Q7ECG0 | chaperonin | −0.75 | ||
| 22 | Solyc01g099900.4.1 | A0A3Q7ENE9 | ribosomal protein L18 | 1.51 | ||
| 23 | Solyc12g008630.2.1 | A0A3Q7J3G4 | mitochondrial processing peptidase alpha subunit | −3.00 | ||
| Aminoacid Metabolism (5) | ||||||
| 24 | Solyc01g080280.3.1 | A0A3Q7EI59 | chloroplast glutamine synthetase | −0.79 | ||
| 25 | Solyc06g060790.1.1 | A0A3Q7GXH5 | 3-isopropylmalate dehydratase | −1.11 | ||
| 26 | Solyc11g011380.2.1 | A0A3Q7IRF8 * | glutamine synthetase | −1.26 | ||
| 27 | Solyc12g005080.2.1 | A0A3Q7J1A5 | dihydrolipoyllysine-residue succinyltransferase component of 2-oxoglutarate dehydrogenase | −0.84 | ||
| 28 | Solyc09g008280.2.1 | P43282 | S-adenosylmethionine synthase | −3.00 | - | |
| Signaling/Regulation (3) | ||||||
| 29 | Solyc08g076960.1.1 | A0A3Q7HTY9 | abscisic acid receptor PYL1 | −1.00 | ||
| 30 | Solyc07g062110.3.1 | A0A3Q7IAM5 | protein FLX-like 1 | −0.59 | ||
| 31 | Solyc02g093340.3.1 | A0A3Q7FDP2 | heterogeneous nuclear ribonucleoprotein A3 | −3.00 | ||
| Miscellaneous (4) | ||||||
| 32 | Solyc01g102390.4.1 | A0A3Q7EP74 * | germin-like protein 5-1 | 1.12 | ||
| 33 | Solyc03g025850.3.1 | Q9XEX8 | remorin 1 | −0.66 | ||
| 34 | Solyc03g113570.1.1 | A0A3Q7FQV4 * | germin-like protein | −0.91 | ||
| 35 | Solyc05g008460.4.1 | A0A3Q7GD18 | ATP synthase subunit beta | −1.66 | ||
| # | Accession | UniProtKB | Description | Fold SG | Fold 2D | Fe |
|---|---|---|---|---|---|---|
| Oxidoreductases (18) | ||||||
| 36 | Solyc02g084780.4.1 | A0A3Q7F7C4 * | peroxidase superfamily protein | 1.53 | ||
| 37 | Solyc03g032000.4.1 | A0A3Q7G4N1 | thioredoxin reductase 2-like | 1.21 | ||
| 38 | Solyc08g081530.3.1 | A0A3Q7HU15 | monodehydroascorbate reductase | 0.96 | ||
| 2 | Solyc12g005790.2.1 | A0A3Q7JR84 | peroxidase 27 | 0.93 | - | |
| 39 | Solyc02g062510.3.1 | A0A3Q7F0H1 | peroxidase | 0.89 | ||
| 40 | Solyc10g076240.3.1 | A0A3Q7IJN4 | cationic peroxidase 1 | 0.79 | ||
| 41 | Solyc05g056540.4.1 | A0A3Q7HI60 | alcohol dehydrogenase 1B | 0.76 | ||
| 42 | Solyc02g084800.4.1 | A0A3Q7F7C4 | peroxidase 72 | 0.73 | ||
| 43 | Solyc10g050890.2.1 | A0A3Q7IH89 | nitrite reductase 2 | 0.68 | ||
| 44 | Solyc02g078650.4.1 | A0A3Q7F625 | polyphenol oxidase | 0.62 | ||
| 45 | Solyc11g072550.2.1 | A0A3Q7J0V5 | 4,5-dioxygenase-like protein | −0.63 | ||
| 46 | Solyc06g005150.3.1 | Q52QQ4 | ascorbate peroxidase | −0.64 | ||
| 47 | Solyc01g100360.4.1 | A0A3Q7ENY7 | dihydrolipoyl dehydrogenase 2 | −0.68 | ||
| 48 | Solyc09g007520.3.1 | A0A3Q7HVX4 | peroxidase | −0.78 | −0.74 | |
| 49 | Solyc09g011240.3.1 | A0A3Q7I069 | aldo-keto reductase 4B-like | −0.84 | ||
| 50 | Solyc06g059740.4.1 | A0A3Q7GTE7 | alcohol dehydrogenase 2 | −1.10 | ||
| 51 | Solyc07g043420.3.1 | Q40131 | 2-oxoglutarate-dependent dioxygenase 2 | −2.32 | ||
| 1 | Solyc07g052510.4.1 | A0A3Q7HDZ4 | peroxidase | −3.64 | - | |
| Carbohydrate Metabolism (10) | ||||||
| 52 | Solyc05g005490.4.1 | Q5NE21 | carbonic anh isoform 1 | 1.95 | ||
| 53 | Solyc03g115990.3.1 | A0A3Q7GH43 | malate dehydrogenase | −0.66 | ||
| 54 | Solyc09g075450.3.1 | A0A3Q7I742 | fumarate hydratase | −0.69 | ||
| 55 | Solyc09g009260.3.1 | A0A3Q7HX95 | fructose-1,6-bisphosphate aldolase | −0.71 | ||
| 56 | Solyc04g011400.3.1 | A0A3Q7FZG5 | UDP-glucuronate decarboxylase 1 | −0.71 | ||
| 57 | Solyc10g083570.3.1 | A0A3Q7ILY0 | fructose-bisphosphate aldolase | −0.99 | ||
| 8 | Solyc08g080140.4.1 | A0A3Q7HT77 | bifunctional dTDP-4-dehydrorhamnose 3,5-epimerase/reductase | −1.72 | −1.65 | - |
| 58 | Solyc09g009020.3.1 | P26300 | enolase | −1.15 | ||
| 59 | Solyc04g011510.4.1 | A0A3Q7FZI5 | triosephosphate isomerase | −1.28 | ||
| 60 | Solyc10g085550.3.1 | A0A3Q7IN81 * | enolase | −2.06 | ||
| Polysaccaride Metabolism (5) | ||||||
| 61 | Solyc03g123630.4.1 | A0A3Q7GM93 * | pectinesterase/pectinesterase inhibitor U1 precursor | 0.61 | ||
| 12 | Solyc01g104950.4.1 | A0A3Q7EQN7 | beta-D-xylosidase 2 precursor | −0.74 | −0.62 | - |
| 13 | Solyc01g107590.3.1 | A0A3Q7ESC5 | cinnamyl alcohol dehydrogenase | −1.37 | - | |
| 62 | Solyc08g079080.5.1 | A0A3Q7HT63 | acid beta-fructofuranosidase | −2.56 | ||
| 63 | Solyc01g111230.3.1 | A0A3Q7EVU4 | dirigent protein | −3.00 | ||
| Protein Metabolism (35) | ||||||
| 64 | Solyc06g072220.1.1 | A0A3Q7GYL5 | Kunitz trypsin inhibitor | 1.50 | ||
| 65 | Solyc01g080010.2.1 | A0A3Q7EHP2 | xyloglucan endoglucanase inhibitor | 0.95 | ||
| 66 | Solyc12g088670.2.1 | O49877 | cysteine protease CYP1 | 0.74 | ||
| 67 | Solyc09g007640.4.1 | A0A3Q7HWG9 | serine carboxypeptidase-like 50 | 0.67 | ||
| 68 | Solyc02g068740.3.1 | A0A3Q7F1L3 | glycine cleavage system H family | −0.58 | ||
| 69 | Solyc01g099760.3.1 | A0A3Q7F894 | LeMA-1 putatve Mg-dependent ATPase 1 | −0.74 | −0.74 | |
| 70 | Solyc02g083710.3.1 | A0A3Q7F6N7 | 26S proteasome non-ATPase regulatory subunit 4 | −0.80 | ||
| 71 | Solyc04g076190.1.1 | A0A3Q7G5A8 | aspartic proteinase nepenthesin-1 | −1.20 | ||
| 72 | Solyc04g080960.4.1 | A0A3Q7GB74 | pre-pro-cysteine proteinase | −1.23 | ||
| 73 | Solyc01g100320.3.1 | A0A3Q7ENV3 | disulfide-isomerase-like protein | −0.57 | ||
| 74 | Solyc07g049450.3.1 | A0A3Q7HDI6 | protein disulfide isomerase family | −0.61 | ||
| 75 | Solyc08g079170.3.1 | A0A3Q7HV43 | heat shock protein STI | −0.61 | ||
| 76 | Solyc08g079260.3.1 | A0A3Q7HV74 | tetratricopeptide repeat-containing | −0.63 | ||
| 77 | Solyc01g106260.3.1 | A0A3Q7FBU5 | heat shock protein 70 | −0.64 | ||
| 78 | Solyc07g042250.3.1 | Q9M5A8 | chaperonin 21 precursor | −0.65 | ||
| 79 | Solyc01g088610.4.1 | A0A3Q7F3J4 | 10 kDa chaperonin 1 | −0.95 | ||
| 80 | Solyc06g065520.3.1 | A0A3Q7GYZ2 | T-complex protein eta subunit | −0.89 | ||
| 81 | Solyc03g121330.3.1 | A0A3Q7FVJ9 | 60S ribosomal protein L28, putative | −0.61 | ||
| 82 | Solyc01g096580.3.1 | A0A3Q7F6F2 | ribosomal protein S10p/S20e | −0.62 | ||
| 83 | Solyc09g010100.3.1 | Q2MI68 * | 30S ribosomal protein S11 | −0.63 | ||
| 84 | Solyc09g005720.3.1 | A0A3Q7HVY6 | 60S ribosomal protein L23A | −0.69 | ||
| 85 | Solyc12g044720.2.1 | A0A3Q7JA54 | 60S ribosomal L28-like protein | −0.69 | ||
| 86 | Solyc03g096360.4.1 | A0A3Q7FPS6 | 60S ribosomal protein L35a-2 | −0.70 | ||
| 87 | Solyc06g073430.4.1 | A0A3Q7ITW7 * | 40S ribosomal protein S29 | −0.72 | ||
| 88 | Solyc11g017070.2.1 | A0A3Q7JK86 | eukaryotic translation initiation factor 3 subunit I | −0.73 | ||
| 89 | Solyc06g008170.3.1 | K4CUW3 | 50S ribosomal protein L14 | −0.80 | ||
| 90 | Solyc03g112360.1.1 | A0A3Q7FRG1 | 60S ribosomal protein L27A | −0.82 | ||
| 91 | Solyc10g086010.2.1 | A0A3Q7IMU3 | 60S ribosomal L4 | −0.97 | ||
| 22 | Solyc01g099900.4.1 | A0A3Q7ENE9 | 60S ribosomal protein L18-2 | −0.99 | + | |
| 92 | Solyc01g099890.2.1 | A0A3Q7ENE9 * | PUA domain-containing protein | −1.27 | ||
| 93 | Solyc12g096300.2.1 | A0A3Q7JEJ5 | 40S ribosomal protein S6 | −1.55 | ||
| 94 | Solyc07g005560.3.1 | Q9AXQ5 | eukaryotic translation initiation factor 5A | −0.84 | ||
| 95 | Solyc03g083390.4.1 | A0A3Q7FN48 | protein BOBBER 1 | −0.66 | ||
| 96 | Solyc08g074290.3.1 | A0A3Q7HSI3 | myosin heavy chain-like protein | −0.86 | ||
| 97 | Solyc02g087300.1.1 | A0A3Q7FW84 | transducin/WD40 repeat-like | −1.00 | ||
| Aminoacid Metabolism (9) | ||||||
| 98 | Solyc01g112280.3.1 | A0A3Q7EV39 | N-acyl-L-amino-acid amidohydrolase | 0.61 | ||
| 99 | Solyc05g053810.3.1 | A0A3Q7GNF6 | serine hydroxymethyltransferase | −0.76 | −0.76 | |
| 100 | Solyc02g082830.3.1 | A0A3Q7F688 | phosphoserine aminotransferase 2 | −0.93 | ||
| 101 | Solyc04g074480.3.1 | A0A3Q7H097 | 3-deoxy-D-arabinoheptulosonate 7-phosphate (DAHP) synthase 2 | −1.17 | ||
| 28 | Solyc09g008280.2.1 | P43282 | S-adenosyl-L-methionine synthase | −1.29 | - | |
| 102 | Solyc12g098490.2.1 | A0A3Q7JE99 | serine hydroxymethyltransferase | −1.32 | ||
| 103 | Solyc10g083970.1.1 | A0A3Q7IMD9 | S-adenosylmethionine synthase | −1.60 | −1.00 | |
| 104 | Solyc12g099000.3.1 | A0A3Q7JEH3 | S-adenosylmethionine synthase | −0.94 | ||
| 105 | Solyc04g076790.3.1 | A0A3Q7G863 | serine hydroxymethyltransferase | −1.84 | ||
| Signaling/Regulation (11) | ||||||
| 106 | Solyc09g091000.4.1 | A0A3Q7I801 | pathogenesis-related protein STH-2 | 1.90 | ||
| 107 | Solyc09g082780.3.1 | A0A3Q7I7U0 | stem-specific protein TSJT1 | 0.64 | ||
| 108 | Solyc09g090990.2.1 | A0A3Q7I9H4 * | major allergen Mal d 1 | 0.66 | ||
| 109 | Solyc12g088720.2.1 | A0A3Q7JBV0 | polyadenylate-binding protein | −0.62 | ||
| 110 | Solyc12g014210.3.1 | A0A3Q7J5F6 | UBP1-associated protein 2C-like | −0.62 | ||
| 111 | Solyc03g096460.4.1 | Q672Q3 | wound/stress protein precursor | −1.10 | ||
| 112 | Solyc04g074040.3.1 | A0A3Q7H017 | serine/arginine-rich splicing factor | −1.12 | ||
| 113 | Solyc02g071180.3.1 | A0A3Q7F2T0 | RNA polymerase II degradation factor-like protein (DUF1296) | −1.21 | ||
| 114 | Solyc09g009030.4.1 | A0A3Q7HYW7 | histone deacetylase HDT1 | −1.46 | ||
| 115 | Solyc01g109660.2.1 | A0A3Q7IGH7 * | glycine-rich RNA-binding protein | −0.62 | ||
| 116 | Solyc12g095960.3.1 | A0A3Q7JDH7 | Insulin-like growth factor 2 mRNA-binding protein 2 | −0.92 | ||
| Miscellaneous (3) | ||||||
| 117 | Solyc03g115110.4.1 | A0A3Q7FRU0 | ATP synthase subunit gamma | 1.13 | ||
| 118 | Solyc06g062380.3.1 | A0A3Q7GUF9 | acid phosphatase-like protein 1 | 0.72 | ||
| 119 | Solyc11g039980.3.1 | A0A3Q7JLN4 | ATP synthase subunit alpha | 0.63 | ||
| # | Accession | UniProtKB | Description | −Fe SG | −Fe 2D | −Mn SG | −Mn 2D |
|---|---|---|---|---|---|---|---|
| 22 | Solyc01g099900.4.1 | A0A3Q7ENE9 | ribosomal protein L18 | 1.51 | −0.99 | ||
| 2 | Solyc12g005790.2.1 | A0A3Q7JR84 | peroxidase 27 | −0.82 | 0.93 | ||
| 1 | Solyc07g052510.4.1 | A0A3Q7HDZ4 | peroxidase (TPX1) | −0.76 | −3.00 | −3.64 | |
| 13 | Solyc01g107590.3.1 | A0A3Q7ESC5 | cinnamyl alcohol dehydrogenase | −1.54 | −1.37 | ||
| 8 | Solyc08g080140.4.1 | A0A3Q7HT77 | bifunctional dTDP-4-dehydrorhamnose 3,5-epimerase/reductase | −0.81 | −1.72 | −1.65 | |
| 12 | Solyc01g104950.4.1 | A0A3Q7EQN7 | beta-D-xylosidase 2 | −0.90 | −1.14 | −0.74 | −0.62 |
| 28 | Solyc09g008280.2.1 | P43282 | S-adenosylmethionine synthase | −3.00 | −1.29 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ceballos-Laita, L.; Takahashi, D.; Uemura, M.; Abadía, J.; López-Millán, A.F.; Rodríguez-Celma, J. Effects of Fe and Mn Deficiencies on the Root Protein Profiles of Tomato (Solanum lycopersicum) Using Two-Dimensional Electrophoresis and Label-Free Shotgun Analyses. Int. J. Mol. Sci. 2022, 23, 3719. https://doi.org/10.3390/ijms23073719
Ceballos-Laita L, Takahashi D, Uemura M, Abadía J, López-Millán AF, Rodríguez-Celma J. Effects of Fe and Mn Deficiencies on the Root Protein Profiles of Tomato (Solanum lycopersicum) Using Two-Dimensional Electrophoresis and Label-Free Shotgun Analyses. International Journal of Molecular Sciences. 2022; 23(7):3719. https://doi.org/10.3390/ijms23073719
Chicago/Turabian StyleCeballos-Laita, Laura, Daisuke Takahashi, Matsuo Uemura, Javier Abadía, Ana Flor López-Millán, and Jorge Rodríguez-Celma. 2022. "Effects of Fe and Mn Deficiencies on the Root Protein Profiles of Tomato (Solanum lycopersicum) Using Two-Dimensional Electrophoresis and Label-Free Shotgun Analyses" International Journal of Molecular Sciences 23, no. 7: 3719. https://doi.org/10.3390/ijms23073719
APA StyleCeballos-Laita, L., Takahashi, D., Uemura, M., Abadía, J., López-Millán, A. F., & Rodríguez-Celma, J. (2022). Effects of Fe and Mn Deficiencies on the Root Protein Profiles of Tomato (Solanum lycopersicum) Using Two-Dimensional Electrophoresis and Label-Free Shotgun Analyses. International Journal of Molecular Sciences, 23(7), 3719. https://doi.org/10.3390/ijms23073719