The Complete Loss of p53 Expression Uniquely Predicts Worse Prognosis in Colorectal Cancer
Abstract
:1. Introduction
2. Results
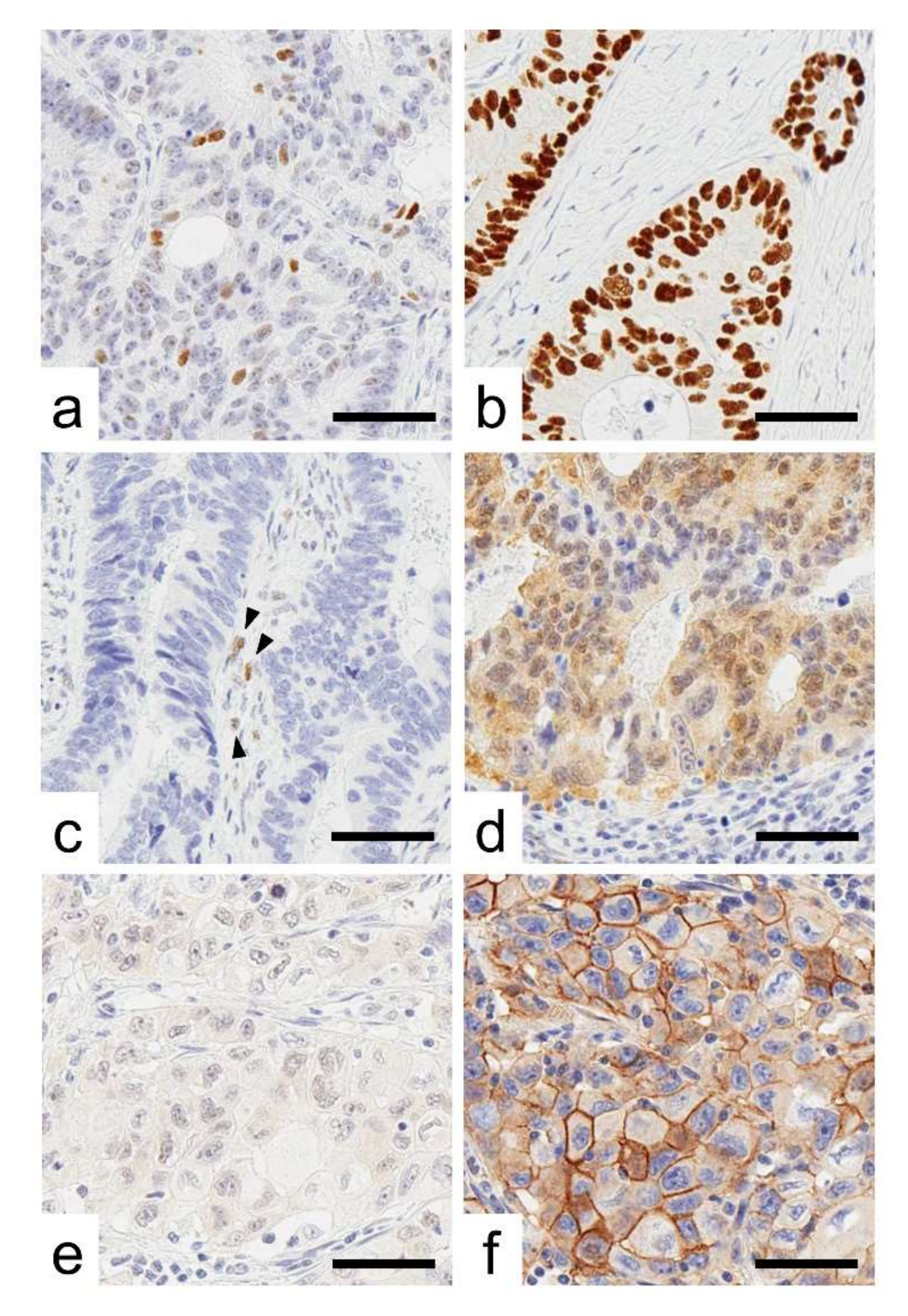
2.1. Expression Status of p53 in CRCs
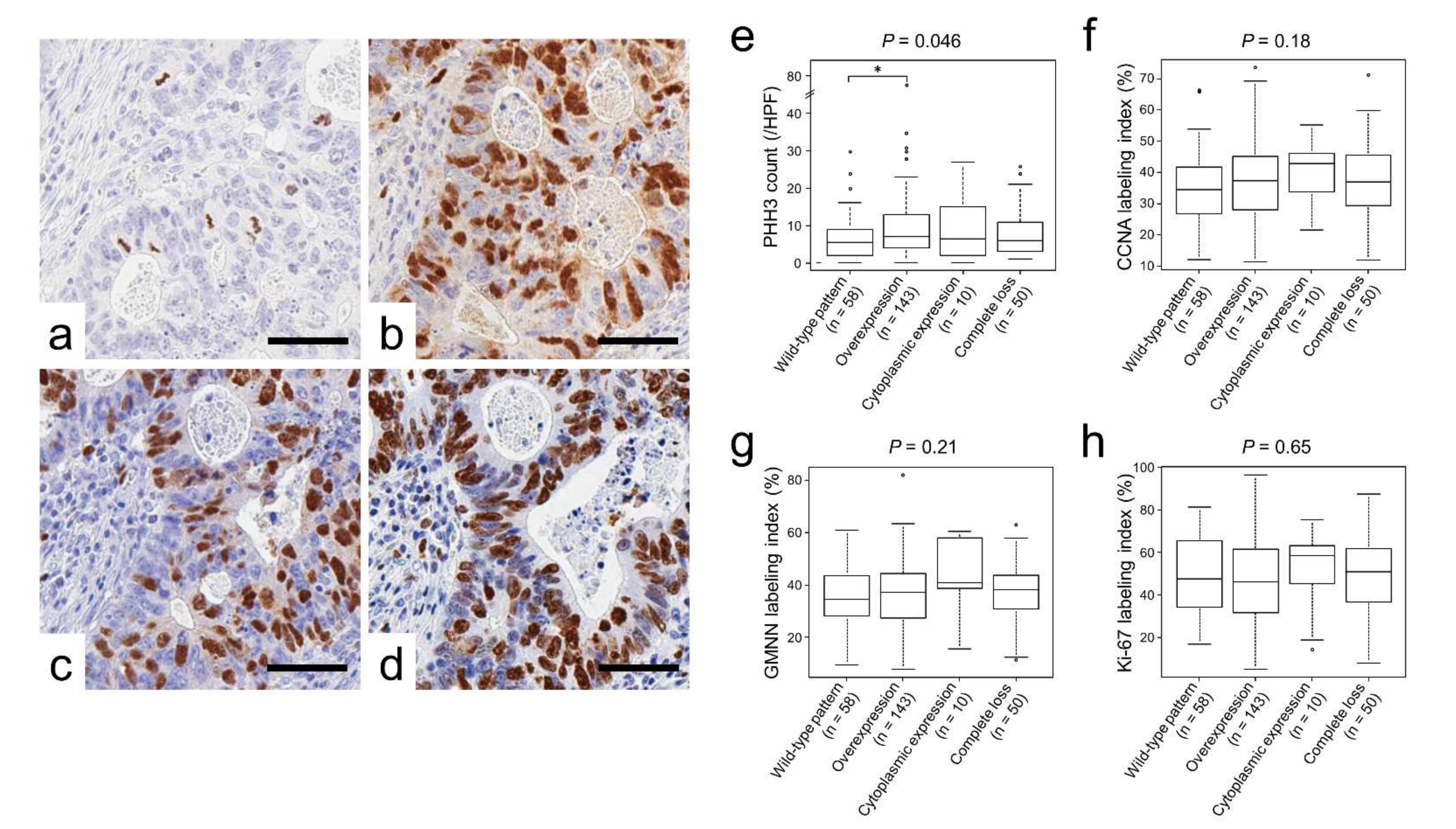
2.2. p53 Immunoreactivity Correlated with PHH3 Expression
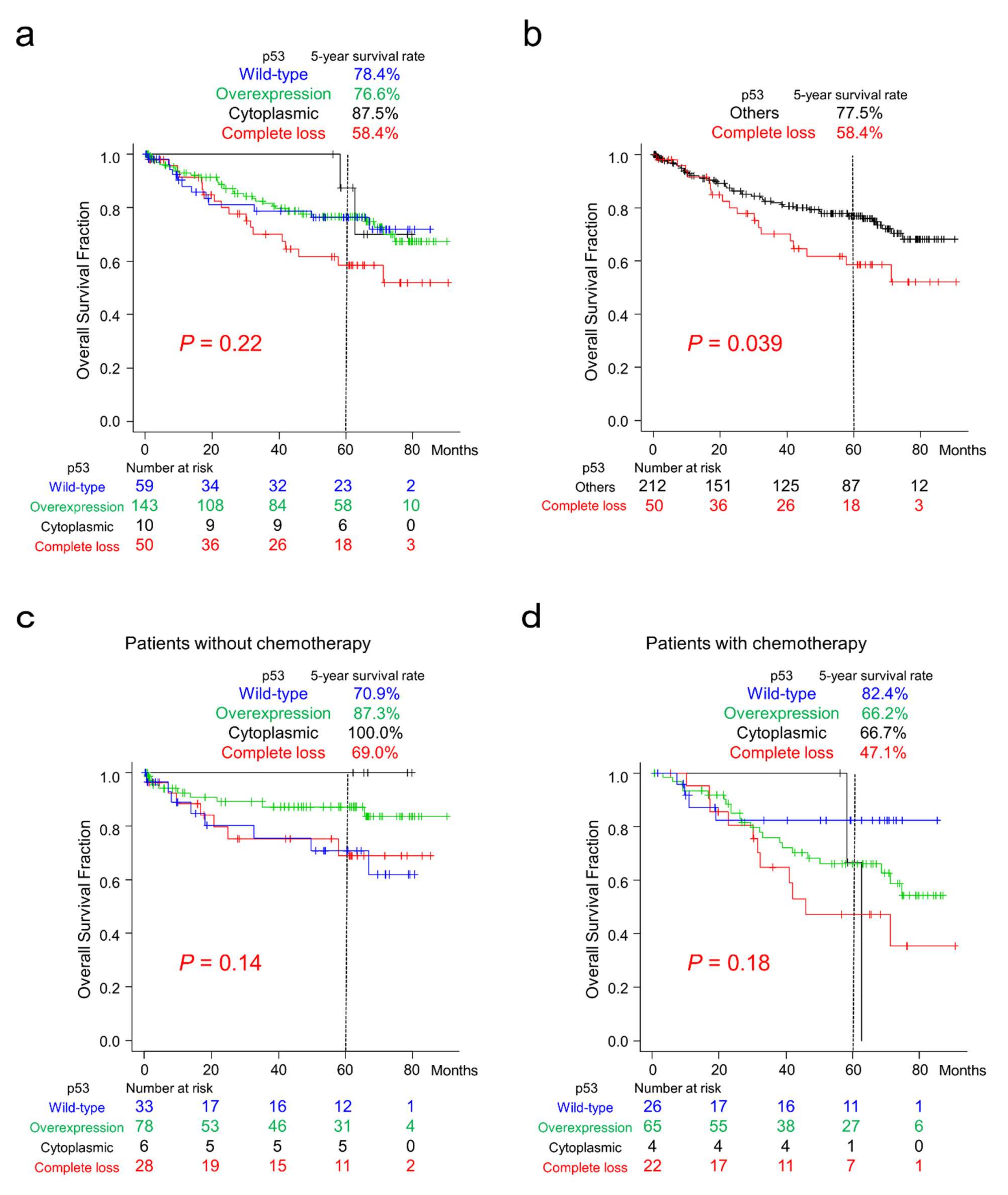
2.3. Survival Analyses of Patients with CRC According to p53 Immunoreactivity
2.4. Survival Analyses of Patients with CRC According to CDX2 and ALCAM Expression
3. Discussion
4. Materials and Methods
4.1. Tissue Samples
4.2. Immunohistochemistry
4.3. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oh, H.-H.; Joo, Y.-E. Novel biomarkers for the diagnosis and prognosis of colorectal cancer. Intest. Res. 2020, 18, 168–183. [Google Scholar] [CrossRef]
- Inoue, S.; Ito, H.; Tsunoda, T.; Murakami, H.; Ebi, M.; Ogasawara, N.; Kasugai, K.; Kasai, K.; Ikeda, H.; Inaguma, S. CD70 expression in tumor-associated fibroblasts predicts worse survival in colorectal cancer patients. Virchows Arch. 2019, 475, 425–434. [Google Scholar] [CrossRef]
- Inoue, S.; Tsunoda, T.; Riku, M.; Ito, H.; Inoko, A.; Murakami, H.; Ebi, M.; Ogasawara, N.; Pastan, I.; Kasugai, K.; et al. Diffuse mesothelin expression leads to worse prognosis through enhanced cellular proliferation in colorectal cancer. Oncol. Lett. 2020, 19, 1741–1750. [Google Scholar] [CrossRef]
- Koshino, A.; Inoue, S.; Sugimura-Nagata, A.; Nishiyama, T.; Murakami, H.; Ito, H.; Riku, M.; Inoko, A.; Ebi, M.; Ogasawara, N.; et al. High phospho-histone H3 expression uniquely predicts favorable survival among four markers of cellular proliferation in colorectal cancer. Pathol. Int. 2021, 71, 316–324. [Google Scholar] [CrossRef]
- Nagano-Matsuo, A.; Inoue, S.; Koshino, A.; Ota, A.; Nakao, K.; Komura, M.; Kato, H.; Naiki-Ito, A.; Watanabe, K.; Nagayasu, Y.; et al. PBK expression predicts favorable survival in colorectal cancer patients. Virchows Arch. 2021, 479, 277–284. [Google Scholar] [CrossRef]
- Sugimura-Nagata, A.; Koshino, A.; Inoue, S.; Matsuo-Nagano, A.; Komura, M.; Riku, M.; Ito, H.; Inoko, A.; Murakami, H.; Ebi, M.; et al. Expression and Prognostic Significance of CD47-SIRPA Macrophage Checkpoint Molecules in Colorectal Cancer. Int. J. Mol. Sci. 2021, 22, 2690–2701. [Google Scholar] [CrossRef]
- Lane, D.; Crawford, L.V. T antigen is bound to a host protein in SV40-transformed cells. Nature 1979, 278, 261–263. [Google Scholar] [CrossRef]
- Linzer, D.I.; Levine, A.J. Characterization of a 54K dalton cellular SV40 tumor antigen present in SV40-transformed cells and uninfected embryonal carcinoma cells. Cell 1979, 17, 43–52. [Google Scholar] [CrossRef]
- Kress, M.; May, E.; Cassingena, R.; May, P. Cassingena and May. Simian virus 40-transformed cells express new species of proteins precipitable by anti-simian virus 40 tumor serum. J. Virol. 1979, 31, 472–483. [Google Scholar] [CrossRef] [Green Version]
- Menendez, D.; Inga, A.; Resnick, M.A. The expanding universe of p53 targets. Nat. Rev. Cancer 2009, 9, 724–737. [Google Scholar] [CrossRef]
- Vogelstein, B.; Lane, D.; Levine, A.J. Surfing the p53 network. Nature 2000, 408, 307–310. [Google Scholar] [CrossRef]
- Jain, A.K.; Allton, K.; Iacovino, M.; Mahen, E.; Milczarek, R.J.; Zwaka, T.P.; Kyba, M.; Barton, M.C. p53 regulates cell cycle and microRNAs to promote differentiation of human embryonic stem cells. PLoS Biol. 2012, 10, e1001268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marión, R.M.; Strati, K.; Li, H.; Murga, M.; Blanco, R.; Ortega, S.; Fernandez-Capetillo, O.; Serrano, M.; Blasco, M.A. A p53-mediated DNA damage response limits reprogramming to ensure iPS cell genomic integrity. Nature 2009, 460, 1149–1153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hollstein, M.; Sidransky, D.; Vogelstein, B.; Harris, C.C. p53 mutations in human cancers. Science 1991, 253, 49–53. [Google Scholar] [CrossRef] [Green Version]
- Bullock, A.N.; Henckel, J.; DeDecker, B.S.; Johnson, C.M.; Nikolova, P.V.; Proctor, M.R.; Lane, D.; Fersht, A.R. Thermodynamic stability of wild-type and mutant p53 core domain. Proc. Natl. Acad. Sci. USA 1997, 94, 14338–14342. [Google Scholar] [CrossRef] [Green Version]
- Köbel, M.; Ronnett, B.M.; Singh, N.; Soslow, R.A.; Gilks, C.B.; McCluggage, W.G. Interpretation of P53 Immunohistochemistry in Endometrial Carcinomas: Toward Increased Reproducibility. Int. J. Gynecol. Pathol. 2019, 38 (Suppl. S1), S123–S131. [Google Scholar] [CrossRef] [PubMed]
- Bosari, S.; Viale, G.; Bossi, P.; Maggioni, M.; Coggi, G.; Murray, J.J.; Lee, A.K.C. Cytoplasmic accumulation of p53 protein: An independent prognostic indicator in colorectal adenocarcinomas. J. Natl. Cancer Inst. 1994, 86, 681–687. [Google Scholar] [CrossRef]
- Colomer, A.; Erill, N.; Verdú, M.; Roman, R.; Vidal, A.; Cordon-Cardo, C.; Puig, X. Lack of p53 nuclear immunostaining is not indicative of absence of TP53 gene mutations in colorectal adenocarcinomas. Appl. Immunohistochem. Mol. Morphol. 2003, 11, 130–137. [Google Scholar] [CrossRef]
- Levin, T.G.; Powell, A.E.; Davies, P.S.; Silk, A.D.; Dismuke, A.D.; Anderson, E.C.; Swain, J.R.; Wong, M.H. Characterization of the intestinal cancer stem cell marker CD166 in the human and mouse gastrointestinal tract. Gastroenterology 2010, 139, 2072–2082.e5. [Google Scholar] [CrossRef] [Green Version]
- Ginestier, C.; Hur, M.H.; Charafe-Jauffret, E.; Monville, F.; Dutcher, J.; Brown, M.; Jacquemier, J.; Viens, P.; Kleer, C.G.; Liu, S.; et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 2007, 1, 555–567. [Google Scholar] [CrossRef] [Green Version]
- Huang, E.; Hynes, M.J.; Zhang, T.; Ginestier, C.; Dontu, G.; Appelman, H.; Fields, J.Z.; Wicha, M.S.; Boman, B.M. Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res. 2009, 69, 3382–3389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, J.; Deng, R.; Zhang, P.; Wu, C.; Wu, K.; Shi, L.; Liu, X.; Bai, J.; Deng, M.; Shuai, X.; et al. miR-219-5p plays a tumor suppressive role in colon cancer by targeting oncogene Sall4. Oncol. Rep. 2015, 34, 1923–1932. [Google Scholar] [CrossRef] [PubMed]
- Dalerba, P.; Sahoo, D.; Paik, S.; Guo, X.; Yothers, G.; Song, N.; Wilcox-Fogel, N.; Forgó, E.; Rajendran, P.S.; Miranda, S.P.; et al. CDX2 as a Prognostic Biomarker in Stage II and Stage III Colon Cancer. N. Engl. J. Med. 2016, 374, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Inaguma, S.; Lasota, J.; Wang, Z.; Felisiak-Golabek, A.; Ikeda, H.; Miettinen, M. Clinicopathologic profile, immunophenotype, and genotype of CD274 (PD-L1)-positive colorectal carcinomas. Mod. Pathol. 2017, 30, 278–285. [Google Scholar] [CrossRef] [Green Version]
- Huncharek, M.; Kupelnick, B.; Geschwind, J.; Caubet, J. Prognostic significance of p53 mutations in non-small cell lung cancer: A meta-analysis of 829 cases from eight published studies. Cancer Lett. 2000, 153, 219–226. [Google Scholar] [CrossRef]
- Iacopetta, B. TP53 mutation in colorectal cancer. Hum. Mutat. 2003, 21, 271–276. [Google Scholar] [CrossRef]
- Mitsudomi, T.; Hamajima, N.; Ogawa, M.; Takahashi, T. Prognostic significance of p53 alterations in patients with non-small cell lung cancer: A meta-analysis. Clin. Cancer Res. 2000, 6, 4055–4063. [Google Scholar]
- Pharoah, P.D.P.; Day, N.E.; Caldas, C. Somatic mutations in the p53 gene and prognosis in breast cancer: A meta-analysis. Br. J. Cancer 1999, 80, 1968–1973. [Google Scholar] [CrossRef] [Green Version]
- Guinney, J.; Dienstmann, R.; Wang, X.; De Reyniès, A.; Schlicker, A.; Soneson, C.; Marisa, L.; Roepman, P.; Nyamundanda, G.; Angelino, P.; et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 2015, 21, 1350–1356. [Google Scholar] [CrossRef]
- Hwang, W.-L.; Yang, M.-H.; Tsai, M.-L.; Lan, H.-Y.; Su, S.-H.; Chang, S.-C.; Teng, H.-W.; Yang, S.-H.; Lan, Y.-T.; Chiou, S.-H.; et al. SNAIL regulates interleukin-8 expression, stem cell-like activity, and tumorigenicity of human colorectal carcinoma cells. Gastroenterology 2011, 141, 279–291.e5. [Google Scholar] [CrossRef] [Green Version]
- Ladstein, R.G.; Bachmann, I.M.; Straume, O.; Akslen, L.A. Prognostic importance of the mitotic marker phosphohistone H3 in cutaneous nodular melanoma. J. Investig. Dermatol. 2012, 132, 1247–1252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakashima, S.; Shiozaki, A.; Ichikawa, D.; Komatsu, S.; Konishi, H.; Iitaka, D.; Kubota, T.; Fujiwara, H.; Okamoto, K.; Kishimoto, M.; et al. Anti-phosphohistone H3 as an independent prognostic factor in human esophageal squamous cell carcinoma. Anticancer Res. 2013, 33, 461–467. [Google Scholar] [PubMed]
- Nowak, M.; Svensson, M.A.; Carlsson, J.; Vogel, W.; Kebschull, M.; Wernert, N.; Kristiansen, G.; Andren, O.; Braun, M.; Perner, S. Prognostic significance of phospho-histone H3 in prostate carcinoma. World J. Urol. 2014, 32, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Ramani, P.; Taylor, S.; Miller, E.; Sowa-Avugrah, E.; May, M.T. High phosphohistone H3 expression correlates with adverse clinical, biological, and pathological factors in neuroblastomas. J. Histochem. Cytochem. 2015, 63, 397–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribalta, T.; McCutcheon, I.E.; Aldape, K.D.; Bruner, J.M.; Fuller, G.N. The mitosis-specific antibody anti-phosphohistone-H3 (PHH3) facilitates rapid reliable grading of meningiomas according to WHO 2000 criteria. Am. J. Surg. Pathol. 2004, 28, 1532–1536. [Google Scholar] [CrossRef]
- Russo, A.; Russo, G. Ribosomal Proteins Control or Bypass p53 during Nucleolar Stress. Int. J. Mol. Sci. 2017, 18, 140. [Google Scholar] [CrossRef] [Green Version]
- Brierley, J.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours, 8th ed.; John Wiley & Sons, Inc.: Chichester, UK; Hoboken, NJ, USA, 2017. [Google Scholar]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl. 2013, 48, 452–458. [Google Scholar] [CrossRef] [Green Version]

| Total No. | p53 Immunoreactivity | |||||||
|---|---|---|---|---|---|---|---|---|
| 262 | (100%) | Wild-Type 59 (23%) | Overexpression 143 (55%) | Cytoplasmic Expression 10 (4%) | Complete Loss 50 (19%) | p-Value | ||
| Sex | 0.49 | a | ||||||
| Male | 139 | [53%] | 36 [61%] | 73 [51%] | 4 [40%] | 26 [52%] | ||
| Female | 123 | [47%] | 23 [39%] | 70 [49%] | 6 [60%] | 24 [48%] | ||
| Age, years (Mean ± S.D.) | 68.6 ± 12.6 | 68.5 ± 14.1 | 68.5 ± 12.0 | 70.8 ± 7.7 | 69.0 ± 12.9 | 0.79 | b | |
| Size, cm (Mean ± S.D.) | 5.0 ± 2.6 | 5.9 ± 2.6 † | 4.9 ± 2.7 | 4.5 ± 1.9 | 4.3 ± 1.8 | 0.0056 | b | |
| Tumor location | 0.10 | c | ||||||
| Right-sided colon | 120 | [46%] | 35 [59%] | 58 [40%] | 2 [20%] | 25 [50%] | ||
| Left-sided colon | 84 | [32%] | 16 [27%] | 51 [36%] | 5 [50%] | 12 [24%] | ||
| Rectum | 58 | [22%] | 8 [14%] | 34 [24%] | 3 [30%] | 13 [26%] | ||
| pT stage | 0.31 | a | ||||||
| pT2 | 36 | [13%] | 8 [14%] | 18 [13%] | 1 [10%] | 9 [18%] | ||
| pT3 | 182 | [70%] | 42 [71%] | 103 [72%] | 9 [90%] | 28 [56%] | ||
| pT4 | 44 | [16%] | 9 [15%] | 22 [15%] | 0 [0%] | 13 [26%] | ||
| Histological differentiation | 0.73 | a | ||||||
| Well to moderately | 235 | [90%] | 51 [86%] | 129 [90%] | 10 [100%] | 45 [90%] | ||
| Poorly | 27 | [10%] | 8 [14%] | 14 [10%] | 0 [0%] | 5 [10%] | ||
| Mucus production | 0.0015 | a | ||||||
| Positive | 14 | [5%] | 9 [15%] †† | 2 [1%] | 0 [0%] | 3 [6%] | ||
| Negative | 248 | [95%] | 50 [85%] | 141 [99%] | 10 [100%] | 47 [94%] | ||
| Lymph node metastasis | 0.19 | a | ||||||
| Positive | 122 | [49%] | 21 [38%] | 67 [51%] | 5 [50%] | 29 [59%] | ||
| Negative | 124 | [51%] | 34 [62%] | 65 [49%] | 5 [50%] | 20 [41%] | ||
| Peritoneal metastasis | a | |||||||
| Positive | 49 | [19%] | 7 [12%] | 32 [22%] | 0 [0%] | 10 [20%] | 0.15 | |
| Negative | 213 | [81%] | 52 [88%] | 111 [78%] | 10 [100%] | 40 [80%] | ||
| Distant organ metastasis | a | |||||||
| Positive | 43 | [16%] | 5 [8%] | 28 [20%] | 0 [0%] | 10 [20%] | 0.11 | |
| Negative | 219 | [84%] | 54 [92%] | 115 [80%] | 10 [100%] | 40 [80%] | ||
| Operation status | 0.23 | a | ||||||
| Complete resection | 230 | [88%] | 56 [95%] | 122 [85%] | 9 [90%] | 43 [86%] | ||
| Incomplete resection | 32 | [12%] | 3 [5%] | 21 [15%] | 1 [10%] | 7 [14%] | ||
| MMR system status | a | |||||||
| Deficient | 30 | [12%] | 19 [32%] ††† | 8 [6%] | 1 [10%] | 2 [4%] | <0.0001 | |
| Preserved | 232 | [88%] | 40 [68%] | 135 [94%] | 9 [90%] | 48 [96%] | ||
| Total No. | p53 Immunoreactivity | ||||||
|---|---|---|---|---|---|---|---|
| 262 | (100%) | Wild-Type 59 (23%) | Overexpression 143 (55%) | Cytoplasmic Expression 10 (4%) | Complete Loss 50 (19%) | p-Value | |
| CDX2 | 0.0031 | ||||||
| Positive | 233 | [89%] | 44 [75%] † | 132 [92%] | 10 [100%] | 47 [94%] | |
| Decreased or lost | 29 | [11%] | 15 [25%] | 11 [8%] | 0 [0%] | 3 [6%] | |
| ALCAM | 0.0016 | ||||||
| Positive | 76 | [29%] | 26 [44%] | 28 [21%] †† | 3 [30%] | 19 [38%] | |
| Negative | 186 | [71%] | 33 [56%] | 115 [79%] | 7 [70%] | 31 [62%] | |
| Stem-like immunophenotype | 0.19 | ||||||
| Positive | 10 | [4%] | 5 [8%] | 3 [2%] | 0 [0%] | 2 [4%] | |
| Negative | 252 | [96%] | 54 [92%] | 140 [98%] | 10 [100%] | 48 [96%] | |
| Total No. | p53 Immunoreactivity | |||||
|---|---|---|---|---|---|---|
| 262 (100%) | Wild-Type 59 (23%) | Overexpression 143 (55%) | Cytoplasmic Expression 10 (4%) | Complete Loss 50 (19%) | p-Value | |
| PHH3 (/HPF) | 7.0 (3, 12) | 5.0 (2.0, 9.0) † | 7.0 (4.0, 13.0) | 6.5 (2.3, 14.0) | 6.0 (3.3, 11.0) | 0.046 |
| CCNA (%) | 36.4 (28.3, 44.7) | 34.3 (26.8, 40.9) | 37.3 (28.0, 45.2) | 42.8 (35.7, 46.0) | 37.0 (29.6, 45.4) | 0.18 |
| GMNN (%) | 37.1 (28.4, 43.7) | 34.4 (27.7, 43.4) | 37.2 (27.3, 44.3) | 40.9 (38.7, 54.9) | 38.2 (30.0, 43.6) | 0.21 |
| Ki-67 (%) | 49.2 (34.2, 62.0) | 48.3 (34.9, 65.5) | 46.0 (31.6, 61.5) | 58.5 (47.6, 62.6) | 50.9 (36.5, 61.7) | 0.65 |
| Hazard | 95% CI | |||
|---|---|---|---|---|
| Ratio | Min | Max | p-Value | |
| Well to moderately differentiated histology | 0.17 | 0.09 | 0.33 | <0.0001 |
| Age (<70) | 0.52 | 0.30 | 0.91 | 0.021 |
| Sex (female) | 0.55 | 0.31 | 0.99 | 0.046 |
| p53 complete loss | 2.16 | 1.21 | 3.86 | 0.0087 |
| Incomplete resection | 2.65 | 1.31 | 5.35 | 0.0068 |
| Peritoneal metastasis | 5.32 | 2.97 | 9.54 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagao, K.; Koshino, A.; Sugimura-Nagata, A.; Nagano, A.; Komura, M.; Ueki, A.; Ebi, M.; Ogasawara, N.; Tsuzuki, T.; Kasai, K.; et al. The Complete Loss of p53 Expression Uniquely Predicts Worse Prognosis in Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 3252. https://doi.org/10.3390/ijms23063252
Nagao K, Koshino A, Sugimura-Nagata A, Nagano A, Komura M, Ueki A, Ebi M, Ogasawara N, Tsuzuki T, Kasai K, et al. The Complete Loss of p53 Expression Uniquely Predicts Worse Prognosis in Colorectal Cancer. International Journal of Molecular Sciences. 2022; 23(6):3252. https://doi.org/10.3390/ijms23063252
Chicago/Turabian StyleNagao, Kazuhiro, Akira Koshino, Akane Sugimura-Nagata, Aya Nagano, Masayuki Komura, Akane Ueki, Masahide Ebi, Naotaka Ogasawara, Toyonori Tsuzuki, Kenji Kasai, and et al. 2022. "The Complete Loss of p53 Expression Uniquely Predicts Worse Prognosis in Colorectal Cancer" International Journal of Molecular Sciences 23, no. 6: 3252. https://doi.org/10.3390/ijms23063252
APA StyleNagao, K., Koshino, A., Sugimura-Nagata, A., Nagano, A., Komura, M., Ueki, A., Ebi, M., Ogasawara, N., Tsuzuki, T., Kasai, K., Takahashi, S., Kasugai, K., & Inaguma, S. (2022). The Complete Loss of p53 Expression Uniquely Predicts Worse Prognosis in Colorectal Cancer. International Journal of Molecular Sciences, 23(6), 3252. https://doi.org/10.3390/ijms23063252






