Actions of Bisphenol A on Different Feto-Maternal Compartments Contributing to Preterm Birth
Abstract
1. Introduction
2. Results and Discussion
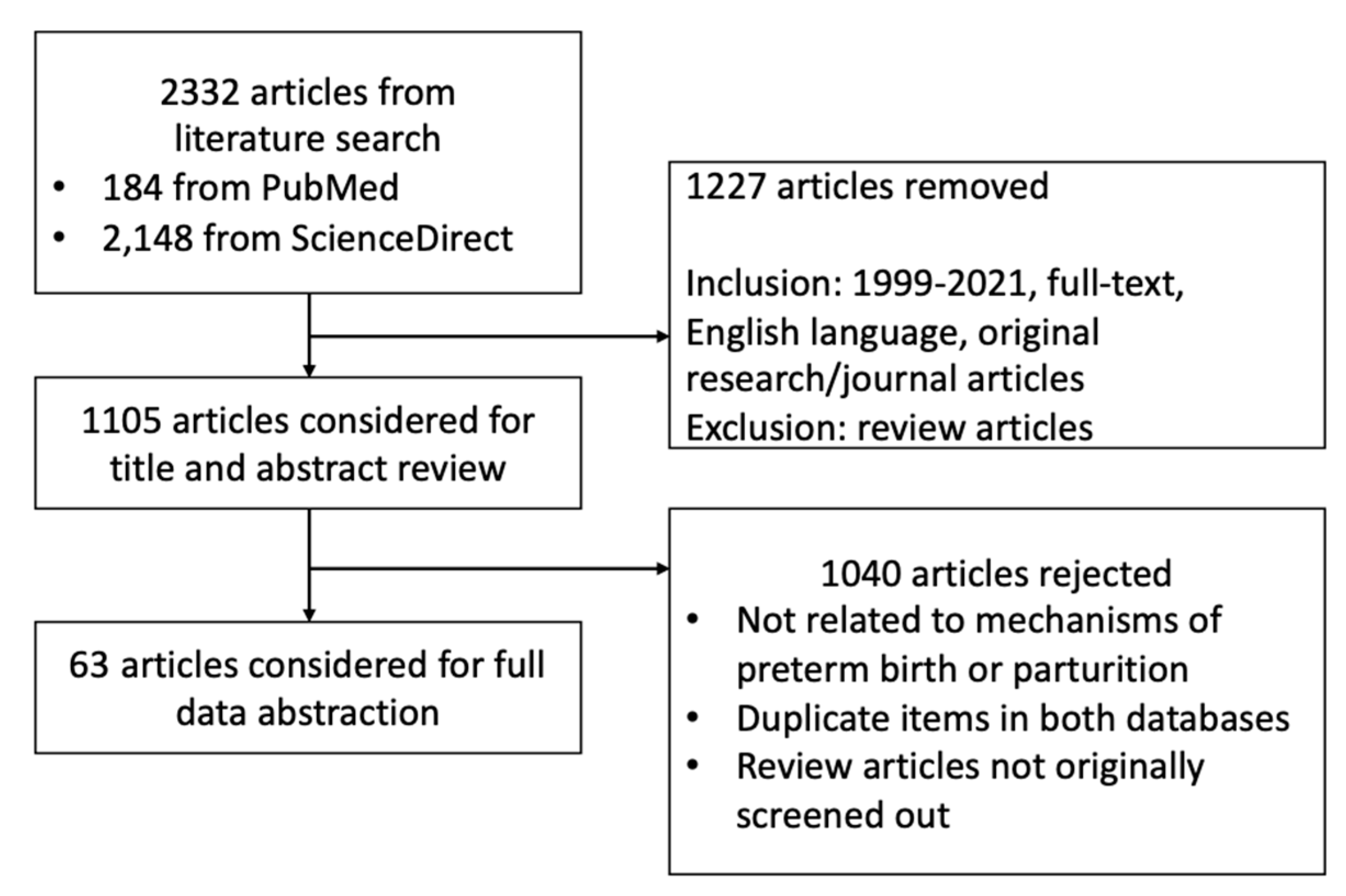
2.1. Search Strategy Employed
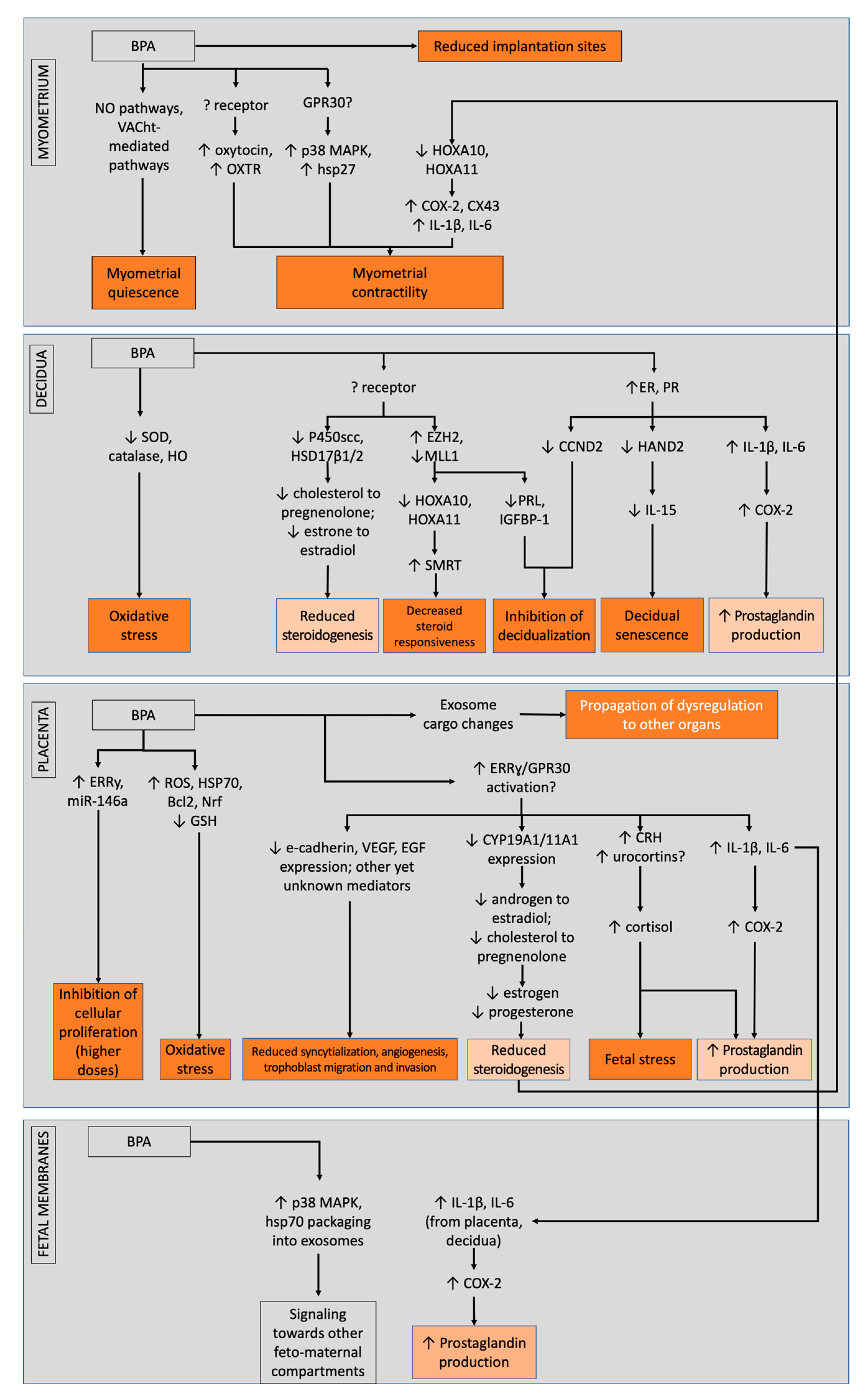
2.2. Effects of Bisphenols on the Placenta
2.3. Effects of Bisphenols on the Decidua
2.4. Effects of Bisphenols on the Myometrium
2.5. Effects of Bisphenols on Fetal Membranes
2.6. Limitations of the Study
3. Conclusions
4. Materials and Methods
4.1. Search Strategy
4.2. Inclusion Criteria
4.3. Exclusion Criteria
4.4. Article Strength Determination
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Preterm Birth. 19 February 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/preterm-birth (accessed on 14 March 2021).
- Blencowe, H.; Cousens, S.; Chou, D.; Oestergaard, M.; Say, L.; Moller, A.-B.; Kinney, M.; Lawn, J.; the Born Too Soon Preterm Birth Action Group. Born Too Soon: The global epidemiology of 15 million preterm births. Reprod. Health 2013, 10 (Suppl. S1), S2. [Google Scholar] [CrossRef] [PubMed]
- Saigal, S.; Doyle, L. An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet 2008, 371, 261–269. [Google Scholar] [CrossRef]
- Menon, R. Spontaneous preterm birth, a clinical dilemma: Etiologic, pathophysiologic and genetic heterogeneities and racial disparity. Acta Obstet. Gynecol. Scand. 2008, 87, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Committee on Understanding Premature Birth and Assuring Healthy Outcomes; Behrman, R.E.; Butler, A.S. (Eds.) The Role of Environmental Toxicants in Preterm Birth. In Preterm Birth: Causes, Consequences, and Prevention; National Academies Press: Washington, DC, USA, 2007. Available online: https://www.ncbi.nlm.nih.gov/books/NBK11368/ (accessed on 1 August 2021).
- Porpora, M.G.; Piacenti, I.; Scaramuzzino, S.; Masciullo, L.; Rech, F.; Panici, P.B. Environmental Contaminants Exposure and Preterm Birth: A Systematic Review. Toxics 2019, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Li, J.; Xu, S.; Zhao, H.; Li, Y.; Zhou, Y.; Fang, J.; Liao, J.; Cai, Z.; Xia, W. Bisphenol A and bisphenol S exposures during pregnancy and gestational age—A longitudinal study in China. Chemosphere 2019, 237, 124426. [Google Scholar] [CrossRef]
- Mustieles, V.; Zhang, Y.; Yland, J.; Braun, J.M.; Williams, P.L.; Wylie, B.J.; Attaman, J.A.; Ford, J.B.; Azevedo, A.; Calafat, A.M.; et al. Maternal and paternal preconception exposure to phenols and preterm birth. Environ. Int. 2020, 137, 105523. [Google Scholar] [CrossRef]
- Cantonwine, D.; Meeker, J.D.; Hu, H.; Sánchez, B.N.; Lamadrid-Figueroa, H.; Mercado-García, A.; Fortenberry, G.Z.; Calafat, A.M.; Téllez-Rojo, M.M. Bisphenol a exposure in Mexico City and risk of prematurity: A pilot nested case control study. Environ. Health 2010, 9, 62. [Google Scholar] [CrossRef]
- Aung, M.T.; Ferguson, K.; Cantonwine, D.E.; McElrath, T.F.; Meeker, J.D. Preterm birth in relation to the bisphenol A replacement, bisphenol S, and other phenols and parabens. Environ. Res. 2018, 169, 131–138. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef]
- Staples, C.A.; Dome, P.B.; Klecka, G.M.; Oblock, S.T.; Harris, L.R. A review of the environmental fate, effects, and exposures of bisphenol A. Chemosphere 1998, 36, 2149–2173. [Google Scholar] [CrossRef]
- Vom Saal, F.S.; Hughes, C. An Extensive New Literature Concerning Low-Dose Effects of Bisphenol A Shows the Need for a New Risk Assessment. Environ. Health Perspect. 2005, 113, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Clark, E. Sulfolane and sulfones. In Kirk-Othmer Encyclopedia of Chemical Technology; Wiley: New York, NY, USA, 2000. [Google Scholar]
- Liao, C.; Kannan, K. Concentrations and Profiles of Bisphenol A and Other Bisphenol Analogues in Foodstuffs from the United States and Their Implications for Human Exposure. J. Agric. Food Chem. 2013, 61, 4655–4662. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Liu, F.; Kannan, K. Bisphenol S, a New Bisphenol Analogue, in Paper Products and Currency Bills and Its Association with Bisphenol A Residues. Environ. Sci. Technol. 2012, 46, 6515–6522. [Google Scholar] [CrossRef]
- Liao, C.; Kannan, K. A Survey of Alkylphenols, Bisphenols, and Triclosan in Personal Care Products from China and the United States. Arch. Environ. Contam. Toxicol. 2014, 67, 50–59. [Google Scholar] [CrossRef]
- Namat, A.; Xia, W.; Xiong, C.; Xu, S.; Wu, C.; Wang, A.; Li, Y.; Wu, Y.; Li, J. Association of BPA exposure during pregnancy with risk of preterm birth and changes in gestational age: A meta-analysis and systematic review. Ecotoxicol. Environ. Saf. 2021, 220, 112400. [Google Scholar] [CrossRef]
- Bukovsky, A.; Caudle, M.R.; Cekanova, M.; Fernando, R.I.; Wimalasena, J.; Foster, J.S.; Henley, D.C.; Elder, R.F. Placental expression of estrogen receptor beta and its hormone binding variant—Comparison with estrogen receptor alpha and a role for estrogen receptors in asymmetric division and differentiation of estrogen-dependent cells. Reprod. Biol. Endocrinol. 2003, 1, 36. [Google Scholar] [CrossRef]
- De Aguiar Greca, S.-C.; Kyrou, I.; Pink, R.; Randeva, H.; Grammatopoulos, D.; Silva, E.; Karteris, E. Effects of the Endocrine Disrupting Chemical Bisphenol A (Bpa) in Human Placentation in vitro. EJEA. Available online: http://www.endocrine-abstracts.org/ea/0065/ea0065p358.htm (accessed on 19 July 2021).
- Morice, L.; Benaîtreau, D.; Dieudonné, M.-N.; Morvan, C.; Serazin, V.; de Mazancourt, P.; Pecquery, R.; Dos Santos, E. Antiproliferative and proapoptotic effects of bisphenol A on human trophoblastic JEG-3 cells. Reprod. Toxicol. 2011, 32, 69–76. [Google Scholar] [CrossRef]
- Profita, M.; Fabbri, E.; Spisni, E.; Valbonesi, P. Comparing effects and action mechanisms of BPA and BPS on HTR-8/Svneo placental cells. Biol. Reprod. 2021, 105, 1355–1364. [Google Scholar] [CrossRef]
- Spagnoletti, A.; Paulesu, L.; Mannelli, C.; Ermini, L.; Romagnoli, R.; Cintorino, M.; Ietta, F. Low concentrations of Bisphenol A and para-Nonylphenol affect extravillous pathway of human trophoblast cells. Mol. Cell. Endocrinol. 2015, 412, 56–64. [Google Scholar] [CrossRef]
- Benachour, N.; Aris, A. Toxic effects of low doses of Bisphenol-A on human placental cells. Toxicol. Appl. Pharmacol. 2009, 241, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-Y.; Lu, J.; Zhang, Y.-Z.; Zhang, M.; Liu, T.; Qu, X.-L. Effect of Bisphenol A on invasion ability of human trophoblastic cell line BeWo. Int. J. Clin. Exp. Pathol. 2015, 8, 14355–14364. [Google Scholar] [PubMed]
- Mørck, T.J.; Sorda, G.; Bechi, N.; Rasmussen, B.S.; Nielsen, J.B.; Ietta, F.; Rytting, E.; Mathiesen, L.; Paulesu, L.; Knudsen, L.E. Placental transport and in vitro effects of Bisphenol A. Reprod. Toxicol. 2010, 30, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Chu, P.-W.; Yang, Z.-J.; Huang, H.-S.; Chang, A.-A.; Cheng, Y.-C.; Wu, G.-J.; Lan, H.-C. Low-dose bisphenol A activates the ERK signaling pathway and attenuates steroidogenic gene expression in human placental cells. Biol. Reprod. 2017, 98, 250–258. [Google Scholar] [CrossRef]
- Pérez-Albaladejo, E.; Fernandes, D.; Lacorte, S.; Porte, C. Comparative toxicity, oxidative stress and endocrine disruption potential of plasticizers in JEG-3 human placental cells. Toxicol. In Vitro 2017, 38, 41–48. [Google Scholar] [CrossRef]
- Takeda, Y.; Liu, X.; Sumiyoshi, M.; Matsushima, A.; Shimohigashi, M.; Shimohigashi, Y. Placenta expressing the greatest quantity of bisphenol A receptor ERR{gamma} among the human reproductive tissues: Predominant expression of type-1 ERRgamma isoform. J. Biochem. 2009, 146, 113–122. [Google Scholar] [CrossRef]
- Avissar-Whiting, M.; Veiga, K.R.; Uhl, K.M.; Maccani, M.A.; Gagne, L.A.; Moen, E.L.; Marsit, C.J. Bisphenol A exposure leads to specific microRNA alterations in placental cells. Reprod. Toxicol. 2010, 29, 401–406. [Google Scholar] [CrossRef]
- De Felice, B.; Manfellotto, F.; Palumbo, A.R.; Troisi, J.; Zullo, F.; Di Carlo, C.; Sardo, A.D.S.; De Stefano, N.; Ferbo, U.; Guida, M.; et al. Genome–wide microRNA expression profiling in placentas from pregnant women exposed to BPA. BMC Med. Genom. 2015, 8, 56. [Google Scholar] [CrossRef]
- Narciso, L.; Ietta, F.; Romagnoli, R.; Paulesu, L.; Mantovani, A.; Tait, S. Effects of Bisphenol A on endogenous retroviral envelopes expression and trophoblast fusion in BeWo cells. Reprod. Toxicol. 2019, 89, 35–44. [Google Scholar] [CrossRef]
- Mao, J.; Jain, A.; Denslow, N.D.; Nouri, M.-Z.; Chen, S.; Wang, T.; Zhu, N.; Koh, J.; Sarma, S.J.; Sumner, B.W.; et al. Bisphenol A and bisphenol S disruptions of the mouse placenta and potential effects on the placenta–brain axis. Proc. Natl. Acad. Sci. USA 2020, 117, 4642–4652. [Google Scholar] [CrossRef]
- Lan, X.; Fu, L.-J.; Zhang, J.; Liu, X.-Q.; Zhang, H.-J.; Zhang, X.; Ma, M.-F.; Chen, X.-M.; He, J.-L.; Li, L.-B.; et al. Bisphenol A exposure promotes HTR-8/Svneo cell migration and impairs mouse placentation involving upregulation of integrin-β1 and MMP-9 and stimulation of MAPK and PI3K signaling pathways. Oncotarget 2017, 8, 51507–51521. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, T.; Wakimoto, Y.; Nakamuta, N.; Phichitraslip, T.; Wakitani, S.; Kusakabe, K.; Hondo, E.; Kiso, Y. Effects of Bisphenol A (BPA) on Placentation and Survival of the Neonates in Mice. J. Reprod. Dev. 2007, 53, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Tait, S.; Tassinari, R.; Maranghi, F.; Mantovani, A. Bisphenol A affects placental layers morphology and angiogenesis during early pregnancy phase in mice: BPA exposure affects placenta angiogenesis. J. Appl. Toxicol. 2015, 35, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Song, T.; Che, D.; Li, C.; Jiang, J.; Pang, J.; Yang, Y.; Goma; Li, P. The effect of bisphenol a exposure onto endothelial and decidualized stromal cells on regulation of the invasion ability of trophoblastic spheroids in in vitro co-culture model. Biochem. Biophys. Res. Commun. 2019, 516, 506–514. [Google Scholar] [CrossRef]
- Wei, P.; Ru, D.; Li, X.; Shi, D.; Zhang, M.; Xu, Q.; Zhou, H.; Wen, S. Exposure to environmental bisphenol A inhibits HTR-8/Svneo cell migration and invasion. J. Biomed. Res. 2020, 34, 369–378. [Google Scholar] [CrossRef]
- Barberio, L.; Paulesu, L.; Canesi, L.; Grasselli, E.; Mandalà, M. Bisphenol a Interferes with Uterine Artery Features and Impairs Rat Feto-Placental Growth. Int. J. Mol. Sci. 2021, 22, 6912. [Google Scholar] [CrossRef]
- Müller, J.E.; Meyer, N.; Santamaria, C.G.; Schumacher, A.; Luque, E.H.; Zenclussen, M.L.; Rodriguez, H.A.; Zenclussen, A.C. Bisphenol A exposure during early pregnancy impairs uterine spiral artery remodeling and provokes intrauterine growth restriction in mice. Sci. Rep. 2018, 8, 9196. [Google Scholar] [CrossRef]
- Basak, S.; Srinivas, V.; Duttaroy, A.K. Bisphenol-A impairs cellular function and alters DNA methylation of stress pathway genes in first trimester trophoblast cells. Reprod. Toxicol. 2018, 82, 72–79. [Google Scholar] [CrossRef]
- Gwon, L.W.; Park, S.G.; Lin, C.; Lee, B.J.; Nam, S.-Y. The effects of caffeine and bisphenol A singularly or in combination on cultured mouse embryos and yolk sac placenta. Reprod. Toxicol. 2019, 91, 92–100. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, X.; Ye, Y.; Li, X. Bisphenol A affects estradiol metabolism by targeting CYP1A1 and CYP19A1 in human placental JEG-3 cells. Toxicol. In Vitro 2019, 61, 104615. [Google Scholar] [CrossRef]
- Huang, H.; Leung, L.K. Bisphenol A downregulates CYP19 transcription in JEG-3 cells. Toxicol. Lett. 2009, 189, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Nativelle-Serpentini, C.; Richard, S.; Séralini, G.-E.; Sourdaine, P. Aromatase activity modulation by lindane and bisphenol-A in human placental JEG-3 and transfected kidney E293 cells. Toxicol. In Vitro 2003, 17, 413–422. [Google Scholar] [CrossRef]
- Benachour, N.; Moslemi, S.; Sipahutar, H.; Seralini, G. Cytotoxic effects and aromatase inhibition by xenobiotic endocrine disrupters alone and in combination. Toxicol. Appl. Pharmacol. 2007, 222, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Diao, H.; Smith, M.A.; Song, X.; Ye, X. Preimplantation exposure to bisphenol A (BPA) affects embryo transport, preimplantation embryo development, and uterine receptivity in mice. Reprod. Toxicol. 2011, 32, 434–441. [Google Scholar] [CrossRef]
- Park, C.; Song, H.; Choi, J.; Sim, S.; Kojima, H.; Park, J.; Iida, M.; Lee, Y. The mixture effects of bisphenol derivatives on estrogen receptor and androgen receptor. Environ. Pollut. 2020, 260, 114036. [Google Scholar] [CrossRef]
- Imanishi, S.; Manabe, N.; Nishizawa, H.; Morita, M.; Sugimoto, M.; Iwahori, M.; Miyamoto, H. Effects of Oral Exposure of Bisphenol A on mRNA Expression of Nuclear Receptors in Murine Placentae Assessed by DNA Microarray. J. Reprod. Dev. 2003, 49, 329–336. [Google Scholar] [CrossRef]
- Tong, C.; Feng, X.; Chen, J.; Qi, X.; Zhou, L.; Shi, S.; Kc, K.; Stanley, J.L.; Baker, P.N.; Zhang, H. G protein-coupled receptor 30 regulates trophoblast invasion and its deficiency is associated with preeclampsia. J. Hypertens. 2016, 34, 710–718. [Google Scholar] [CrossRef]
- Greca, S.-C.D.A.; Kyrou, I.; Pink, R.; Randeva, H.; Grammatopoulos, D.; Silva, E.; Karteris, E. Involvement of the Endocrine-Disrupting Chemical Bisphenol A (BPA) in Human Placentation. J. Clin. Med. 2020, 9, 405. [Google Scholar] [CrossRef]
- Okamoto, E.; Takagi, T.; Azuma, C.; Kimura, T.; Tokugawa, Y.; Mitsuda, N.; Saji, F.; Tanizawa, O. Expression of the Corticotropin-Releasing Hormone (CRH) Gene in Human Placenta and Amniotic Membrane. Horm. Metab. Res. 1990, 22, 394–397. [Google Scholar] [CrossRef]
- Challis, J.R.G. CRH, a placental clock and preterm labour. Nat. Med. 1995, 1, 416. [Google Scholar] [CrossRef]
- Huang, H.; Tan, W.; Wang, C.; Leung, L.K. Bisphenol A induces corticotropin-releasing hormone expression in the placental cells JEG-3. Reprod. Toxicol. 2012, 34, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Huang, H.; Wang, Y.; Wong, T.Y.; Wang, C.; Leung, L.K. Bisphenol A differentially activates protein kinase C isoforms in murine placental tissue. Toxicol. Appl. Pharmacol. 2013, 269, 163–168. [Google Scholar] [CrossRef] [PubMed]
- You, X.; Yang, R.; Tang, X.; Gao, L.; Ni, X. Corticotropin-Releasing Hormone Stimulates Estrogen Biosynthesis in Cultured Human Placental Trophoblasts1. Biol. Reprod. 2006, 74, 1067–1072. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, R.; You, X.; Tang, X.; Gao, L.; Ni, X. Corticotropin-releasing hormone inhibits progesterone production in cultured human placental trophoblasts. J. Mol. Endocrinol. 2006, 37, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Arita, Y.; Park, H.J.; Cantillon, A.; Getahun, D.; Menon, R.; Peltier, M.R. Effect of bisphenol-A (BPA) on placental biomarkers for inflammation, neurodevelopment and oxidative stress. J. Périnat. Med. 2019, 47, 741–749. [Google Scholar] [CrossRef]
- Song, W.; Puttabyatappa, M.; Zeng, L.; Vazquez, D.; Pennathur, S.; Padmanabhan, V. Developmental programming: Prenatal bisphenol A treatment disrupts mediators of placental function in sheep. Chemosphere 2019, 243, 125301. [Google Scholar] [CrossRef]
- Kallapur, S.G.; Presicce, P.; Senthamaraikannan, P.; Alvarez, M.; Tarantal, A.F.; Miller, L.M.; Jobe, A.H.; Chougnet, C.A. Intra-Amniotic IL-1β Induces Fetal Inflammation in Rhesus Monkeys and Alters the Regulatory T Cell/IL-17 Balance. J. Immunol. 2013, 191, 1102–1109. [Google Scholar] [CrossRef] [PubMed]
- Sadowsky, D.W.; Adams, K.M.; Gravett, M.G.; Witkin, S.S.; Novy, M.J. Preterm labor is induced by intraamniotic infusions of interleukin-1β and tumor necrosis factor-α but not by interleukin-6 or interleukin-8 in a nonhuman primate model. Am. J. Obstet. Gynecol. 2006, 195, 1578–1589. [Google Scholar] [CrossRef]
- Wakabayashi, A.; Sawada, K.; Nakayama, M.; Toda, A.; Kimoto, A.; Mabuchi, S.; Kinose, Y.; Nakamura, K.; Takahashi, K.; Kurachi, H.; et al. Targeting interleukin-6 receptor inhibits preterm delivery induced by inflammation. Mol. Hum. Reprod. 2013, 19, 718–726. [Google Scholar] [CrossRef]
- Ponniah, M.; Billett, E.E.; De Girolamo, L.A. Bisphenol A Increases BeWo Trophoblast Survival in Stress-Induced Paradigms through Regulation of Oxidative Stress and Apoptosis. Chem. Res. Toxicol. 2015, 28, 1693–1703. [Google Scholar] [CrossRef]
- Sheller-Miller, S.; Radnaa, E.; Arita, Y.; Getahun, D.; Jones, R.J.; Peltier, M.R.; Menon, R. Environmental pollutant induced cellular injury is reflected in exosomes from placental explants. Placenta 2020, 89, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Menon, R.; Sheller, S.; Saade, G.; Salomon, C. Oxidative stress induced amnion cell derived exosomes produce inflammatory changes in myometrial cells: A feto-maternal signaling in human parturition. Placenta 2016, 45, 111–112. [Google Scholar] [CrossRef]
- Marinić, M.; Mika, K.; Chigurupati, S.; Lynch, V.J. Evolutionary transcriptomics implicates HAND2 in the origins of implantation and regulation of gestation length. eLife 2021, 10, e61257. [Google Scholar] [CrossRef]
- Brighton, P.J.; Maruyama, Y.; Fishwick, K.; Vrljicak, P.; Tewary, S.; Fujihara, R.; Muter, J.; Lucas, E.S.; Yamada, T.; Woods, L.; et al. Clearance of senescent decidual cells by uterine natural killer cells in cycling human endometrium. eLife 2017, 6, e31274. [Google Scholar] [CrossRef]
- Cha, J.M.; Aronoff, D.M. A role for cellular senescence in birth timing. Cell Cycle 2017, 16, 2023–2031. [Google Scholar] [CrossRef]
- Cho, Y.J.; Park, S.B.; Park, J.W.; Oh, S.R.; Han, M. Bisphenol A modulates inflammation and proliferation pathway in human endometrial stromal cells by inducing oxidative stress. Reprod. Toxicol. 2018, 81, 41–49. [Google Scholar] [CrossRef]
- Sarno, J.L.; Schatz, F.; Lockwood, C.J.; Huang, S.-T.J.; Taylor, H.S. Thrombin and Interleukin-1β Regulate HOXA10 Expression in Human Term Decidual Cells: Implications for Preterm Labor. J. Clin. Endocrinol. Metab. 2006, 91, 2366–2372. [Google Scholar] [CrossRef]
- Oner, C.; Schatz, F.; Kizilay, G.; Murk, W.; Buchwalder, L.F.; Kayisli, U.A.; Arici, A.; Lockwood, C.J. Progestin-Inflammatory Cytokine Interactions Affect Matrix Metalloproteinase-1 and -3 Expression in Term Decidual Cells: Implications for Treatment of Chorioamnionitis-Induced Preterm Delivery. J. Clin. Endocrinol. Metab. 2008, 93, 252–259. [Google Scholar] [CrossRef]
- Lockwood, C.J.; Murk, W.K.; Kayisli, U.A.; Buchwalder, L.F.; Huang, S.J.; Arcuri, F.; Li, M.; Gopinath, A.; Schatz, F. Regulation of Interleukin-6 Expression in Human Decidual Cells and Its Potential Role in Chorioamnionitis. Am. J. Pathol. 2010, 177, 1755–1764. [Google Scholar] [CrossRef]
- Mannelli, C.; Szóstek, A.Z.; Lukasik, K.; Carotenuto, C.; Ietta, F.; Romagnoli, R.; Ferretti, C.; Paulesu, L.; Wołczynski, S.; Skarzynski, D.J. Bisphenol A modulates receptivity and secretory function of human decidual cells: An in vitro study. Reproduction 2015, 150, 115–125. [Google Scholar] [CrossRef][Green Version]
- Aghajanova, L.; Giudice, L.C. Effect of bisphenol A on human endometrial stromal fibroblasts in vitro. Reprod. BioMed. Online 2011, 22, 249–256. [Google Scholar] [CrossRef]
- Berger, R.G.; Foster, W.G.; Decatanzaro, D. Bisphenol-A exposure during the period of blastocyst implantation alters uterine morphology and perturbs measures of estrogen and progesterone receptor expression in mice. Reprod. Toxicol. 2010, 30, 393–400. [Google Scholar] [CrossRef]
- Aldad, T.S.; Rahmani, N.; Leranth, C.; Taylor, H.S. Bisphenol-A exposure alters endometrial progesterone receptor expression in the nonhuman primate. Fertil. Steril. 2011, 96, 175–179. [Google Scholar] [CrossRef]
- Varayoud, J.; Ramos, J.G.; Bosquiazzo, V.L.; Muñoz-De-Toro, M.; Luque, E.H. Developmental Exposure to Bisphenol A Impairs the Uterine Response to Ovarian Steroids in the Adult. Endocrinology 2008, 149, 5848–5860. [Google Scholar] [CrossRef]
- Xiong, Y.; Wen, X.; Liu, H.; Zhang, M.; Zhang, Y. Bisphenol a affects endometrial stromal cells decidualization, involvement of epigenetic regulation. J. Steroid Biochem. Mol. Biol. 2020, 200, 105640. [Google Scholar] [CrossRef]
- Matsumoto, H.; Sakai, K.; Iwashita, M. Insulin-like growth factor binding protein-1 induces decidualization of human endometrial stromal cells via 5 1 integrin. Mol. Hum. Reprod. 2008, 14, 485–489. [Google Scholar] [CrossRef]
- Rhee, H.S.; Oh, S.H.; Ko, B.J.; Han, D.M.; Jeon, B.H.; Park, H.; Moon, H.B.; Kim, W.S. Expression of 3b-Hydroxysteroid dehydrogenase and P450 side chain cleavage enzyme in the human uterine endometrium. Exp. Mol. Med. 2003, 35, 160–166. [Google Scholar] [CrossRef]
- Forte, M.; Mita, L.; Cobellis, L.; Merafina, V.; Specchio, R.; Rossi, S.; Mita, D.G.; Mosca, L.; Castaldi, M.A.; De Falco, M.; et al. Triclosan and bisphenol a affect decidualization of human endometrial stromal cells. Mol. Cell. Endocrinol. 2016, 422, 74–83. [Google Scholar] [CrossRef]
- Koyama-Nasu, R.; Nasu-Nishimura, Y.; Todo, T.; Ino, Y.; Saito, N.; Aburatani, H.; Funato, K.; Echizen, K.; Sugano, H.; Haruta, R.; et al. The critical role of cyclin D2 in cell cycle progression and tumorigenicity of glioblastoma stem cells. Oncogene 2012, 32, 3840–3845. [Google Scholar] [CrossRef]
- Olson, M.R.; Su, R.; Flaws, J.; Fazleabas, A.T. Bisphenol A impairs decidualization of human uterine stromal fibroblasts. Reprod. Toxicol. 2017, 73, 339–344. [Google Scholar] [CrossRef]
- Yuan, M.; Hu, M.; Lou, Y.; Wang, Q.; Mao, L.; Zhan, Q.; Jin, F. Environmentally relevant levels of bisphenol A affect uterine decidualization and embryo implantation through the estrogen receptor/serum and glucocorticoid-regulated kinase 1/epithelial sodium ion channel α-subunit pathway in a mouse model. Fertil. Steril. 2018, 109, 735–744.e1. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Jiang, L.; Lee, Y.-L.; Wong, C.K.C.; Ng, E.H.Y.; Yeung, W.S.B.; Lee, K.-F. Bisphenol compounds regulate decidualized stromal cells in modulating trophoblastic spheroid outgrowth and invasion in vitro†. Biol. Reprod. 2019, 102, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-R.; Kim, Y.S.; Yoon, J.A.; Lyu, S.W.; Shin, H.; Lim, H.J.; Hong, S.-H.; Lee, D.R.; Song, H. Egr1 is rapidly and transiently induced by estrogen and bisphenol A via activation of nuclear estrogen receptor-dependent ERK1/2 pathway in the uterus. Reprod. Toxicol. 2014, 50, 60–67. [Google Scholar] [CrossRef]
- Guo, B.; Tian, X.-C.; Li, D.-D.; Yang, Z.-Q.; Cao, H.; Zhang, Q.-L.; Liu, J.-X.; Yue, Z.-P. Expression, regulation and function of Egr1 during implantation and decidualization in mice. Cell Cycle 2014, 13, 2626–2640. [Google Scholar] [CrossRef]
- Enquobahrie, D.A.; Williams, M.A.; Qiu, C.; Muhie, S.Y.; Slentz-Kesler, K.; Ge, Z.; Sorenson, T. Early pregnancy peripheral blood gene expression and risk of preterm delivery: A nested case control study. BMC Pregnancy Childbirth 2009, 9, 56. [Google Scholar] [CrossRef]
- Shin, S.Y.; Kim, J.H.; Baker, A.; Lim, Y.; Lee, Y.H. Transcription Factor Egr-1 Is Essential for Maximal Matrix Metalloproteinase-9 Transcription by Tumor Necrosis Factor α. Mol. Cancer Res. 2010, 8, 507–519. [Google Scholar] [CrossRef]
- Godbole, G.; Suman, P.; Malik, A.; Galvankar, M.; Joshi, N.; Fazleabas, A.; Gupta, S.K.; Modi, D. Decrease in Expression of HOXA10 in the Decidua After Embryo Implantation Promotes Trophoblast Invasion. Endocrinology 2017, 158, 2618–2633. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Wei, P.; Shi, D.; Wen, S.; Wu, F.; Liu, L.; Ye, N.; Zhou, H. Bisphenol A affects trophoblast invasion by inhibiting CXCL8 expression in decidual stromal cells. Mol. Cell. Endocrinol. 2017, 470, 38–47. [Google Scholar] [CrossRef]
- Okuda, K.; Takiguchi, M.; Yoshihara, S. In vivo estrogenic potential of 4-methyl-2,4-bis(4-hydroxyphenyl)pent-1-ene, an active metabolite of bisphenol A, in uterus of ovariectomized rat. Toxicol. Lett. 2010, 197, 7–11. [Google Scholar] [CrossRef]
- Cook, J.C.; Kaplan, A.M.; Davis, L.G.; O’Connor, J.C. Development of a Tier I Screening Battery for Detecting Endocrine-Active Compounds (EACs). Regul. Toxicol. Pharmacol. 1997, 26 Pt 1, 60–68. [Google Scholar] [CrossRef]
- Gray, L.E., Jr.; Ostby, J. Effects of Pesticides and Toxic Substances On Behavioral and Morphological Reproductive Development: Endocrine Versus Nonendocrine Mechanisms. Toxicol. Ind. Health 1998, 14, 159–184. [Google Scholar]
- Dodge, J.A.; Glasebrook, A.L.; Magee, D.E.; Phillips, D.L.; Sato, M.; Short, L.L.; Bryant, H.U. Environmental estrogens: Effects on cholesterol lowering and bone in the ovariectomized rat. J. Steroid Biochem. Mol. Biol. 1996, 59, 155–161. [Google Scholar] [CrossRef]
- Ashby, J.; Tinwell, H. Uterotrophic activity of bisphenol A in the immature rat. Environ. Health Perspect. 1998, 106, 719–720. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morrison, A.; Callanan, J.; Evans, N.; Aldridge, T.; Sweeney, T. Effects of endocrine disrupting compounds on the pathology and oestrogen receptor α and β distribution in the uterus and cervix of ewe lambs. Domest. Anim. Endocrinol. 2003, 25, 329–343. [Google Scholar] [CrossRef] [PubMed]
- Othman, E.R.; Al-Adly, D.M.M.; Elgamal, D.A.; Ghandour, N.; El-Sharkawy, S. Bisphenol A Concentrates Preferentially in Human Uterine Leiomyoma and Induces Proliferation in Rat Myometrium. Reprod. Sci. 2016, 23, 508–514. [Google Scholar] [CrossRef]
- Papaconstantinou, A.D.; Umbreit, T.H.; Fisher, B.R.; Goering, P.L.; Lappas, N.T.; Brown, K.M. Bisphenol A-Induced Increase in Uterine Weight and Alterations in Uterine Morphology in Ovariectomized B6C3F1 Mice: Role of the Estrogen Receptor. Toxicol. Sci. 2000, 56, 332–339. [Google Scholar] [CrossRef]
- Papaconstantinou, A.D.; Fisher, B.R.; Umbreit, T.H.; Goering, P.L.; Lappas, N.T.; Brown, K.M. Effects of beta-estradiol and bisphenol A on heat shock protein levels and localization in the mouse uterus are antagonized by the antiestrogen ICI 182,780. Toxicol. Sci. 2001, 63, 173–180. [Google Scholar] [CrossRef]
- An, B.-S.; Ahn, H.-J.; Kang, H.-S.; Jung, E.-M.; Yang, H.; Hong, E.-J.; Jeung, E.-B. Effects of estrogen and estrogenic compounds, 4-tert-octylphenol, and bisphenol A on the uterine contraction and contraction-associated proteins in rats. Mol. Cell. Endocrinol. 2013, 375, 27–34. [Google Scholar] [CrossRef]
- Salleh, N.; Giribabu, N.; Feng, A.O.M.; Myint, K. Bisphenol A, Dichlorodiphenyltrichloroethane (DDT) and Vinclozolin Affect ex-vivo Uterine Contraction in Rats via Uterotonin (Prostaglandin F2α, Acetylcholine and Oxytocin) Related Pathways. Int. J. Med. Sci. 2015, 12, 914–925. [Google Scholar] [CrossRef]
- Gupta, H.; Deshpande, S.B. Bisphenol A decreases the spontaneous contractions of rat uterus in vitro through a nitrergic mechanism. J. Basic Clin. Physiol. Pharmacol. 2018, 29, 593–598. [Google Scholar] [CrossRef]
- Liliana, R.; Slawomir, G.; Tomasz, J.; Joanna, W.; Andrzej, P. The Neurochemical Characterization of Parasympathetic Nerve Fibers in the Porcine Uterine Wall Under Physiological Conditions and After Exposure to Bisphenol A (BPA). Neurotox. Res. 2019, 35, 867–882. [Google Scholar] [CrossRef] [PubMed]
- Liliana, R.; Slawomir, G.; Tomasz, J.; Joanna, W.; Andrzej, P. The effects of Bisphenol A (BPA) on sympathetic nerve fibers in the uterine wall of the domestic pig. Reprod. Toxicol. 2018, 84, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Maiti, K.; Paul, J.; Read, M.; Chan, E.C.; Riley, S.; Nahar, P.; Smith, R. G-1-Activated Membrane Estrogen Receptors Mediate Increased Contractility of the Human Myometrium. Endocrinology 2011, 152, 2448–2455. [Google Scholar] [CrossRef] [PubMed]
- Kita, K.; Jin, Y.-H.; Sun, Z.; Chen, S.-P.; Sumiya, Y.; Hongo, T.; Suzuki, N. Increase in the levels of chaperone proteins by exposure to β-estradiol, bisphenol A and 4-methoxyphenol in human cells transfected with estrogen receptor α cDNA. Toxicol. Vitr. 2009, 23, 728–735. [Google Scholar] [CrossRef]
- Kang, S.Y.; Kim, M.-R.; Kim, J.H.; Cho, H.H. Gene expression profiles of the normal myometrium after 48 and 96 hours of exposure to BPA. BioChip J. 2015, 9, 293–299. [Google Scholar] [CrossRef]
- Kang, S.Y.; Song, J.Y.; Cho, H.H. Gene expression analysis of uterine smooth muscle cells exposed to bisphenol A. Toxicol. Environ. Health Sci. 2014, 6, 261–267. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.; Xie, N.; Chen, R.; Lee, A.R.; Slater, D.; Lye, S.; Dong, X. HoxA10 and HoxA11 Regulate the Expression of Contraction-Associated Proteins and Contribute to Regionalized Myometrium Phenotypes in Women. Reprod. Sci. 2017, 25, 44–50. [Google Scholar] [CrossRef]
- Cermik, D.; Karaca, M.; Taylor, H.S. HOXA10 Expression Is Repressed by Progesterone in the Myometrium: Differential Tissue-Specific Regulation of HOX Gene Expression in the Reproductive Tract1. J. Clin. Endocrinol. Metab. 2001, 86, 3387–3392. [Google Scholar]
- Edlow, A.G.; Chen, M.; Smith, N.A.; Lu, C.; McElrath, T.F. Fetal bisphenol A exposure: Concentration of conjugated and unconjugated bisphenol A in amniotic fluid in the second and third trimesters. Reprod. Toxicol. 2012, 34, 1–7. [Google Scholar] [CrossRef]
- Sheller-Miller, S.; Papaconstantinou, J.; Urrabaz-Garza, R.; Richardson, L.; Saade, G.; Salomon, C.; Menon, R. Amnion-Epithelial-Cell-Derived Exosomes Demonstrate Physiologic State of Cell under Oxidative Stress. PLoS ONE 2016, 11, e0157614. [Google Scholar]
- Menon, R.; Papaconstantinou, J. p38 Mitogen activated protein kinase (MAPK): A new therapeutic target for reducing the risk of adverse pregnancy outcomes. Expert Opin. Ther. Targets 2016, 20, 1397–1412. [Google Scholar] [CrossRef] [PubMed]
- Menon, R.; Boldogh, I.; Urrabaz-Garza, R.; Polettini, J.; Syed, T.A.; Saade, G.R.; Papaconstantinou, J.; Taylor, R.N. Senescence of Primary Amniotic Cells via Oxidative DNA Damage. PLoS ONE 2013, 8, e83416. [Google Scholar] [CrossRef] [PubMed]
- Omere, C.; Richardson, L.; Saade, G.R.; Bonney, E.A.; Kechichian, T.; Menon, R. Interleukin (IL)-6: A Friend or Foe of Pregnancy and Parturition? Evidence From Functional Studies in Fetal Membrane Cells. Front. Physiol. 2020, 11, 891. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, M.D.; Dudley, D.J.; Edwin, S.S.; Schiller, S.L. Interleukin-6 stimulates prostaglandin production by human amnion and decidual cells. Eur. J. Pharmacol. 1991, 192, 189–191. [Google Scholar] [CrossRef]
- Gingrich, J.; Pu, Y.; Roberts, J.; Karthikraj, R.; Kannan, K.; Ehrhardt, R.; Veiga-Lopez, A. Gestational bisphenol S impairs placental endocrine function and the fusogenic trophoblast signaling pathway. Arch. Toxicol. 2018, 92, 1861–1876. [Google Scholar] [CrossRef] [PubMed]
- Ticiani, E.; Gingrich, J.; Pu, Y.; Vettathu, M.; Davis, J.; Martin, D.; Petroff, M.G.; Veiga-Lopez, A. Bisphenol S and Epidermal Growth Factor Receptor Signaling in Human Placental Cytotrophoblasts. Environ. Health Perspect. 2021, 129, 027005. [Google Scholar] [CrossRef]
- Aker, A.M.; Ferguson, K.K.; Rosario, Z.Y.; Mukherjee, B.; Alshawabkeh, A.N.; Calafat, A.M.; Cordero, J.F.; Meeker, J.D. A repeated measures study of phenol, paraben and Triclocarban urinary biomarkers and circulating maternal hormones during gestation in the Puerto Rico PROTECT cohort. Environ. Health 2019, 18, 28. [Google Scholar] [CrossRef]
- Pepels, P.P.L.M.; Spaanderman, M.E.A.; Hermus, A.R.M.M.; Lotgering, F.K.; Sweep, C.G.J. Placental urocortin-2 and -3: Endocrine or paracrine functioning during healthy pregnancy? Placenta 2010, 31, 475–481. [Google Scholar] [CrossRef]
- Peretz, J.; Vrooman, L.; Ricke, W.; Hunt, P.A.; Ehrlich, S.; Hauser, R.; Padmanabhan, V.; Taylor, H.S.; Swan, S.; Vandevoort, C.A.; et al. Bisphenol A and Reproductive Health: Update of Experimental and Human Evidence, 2007–2013. Environ. Health Perspect. 2014, 122, 775–786. [Google Scholar] [CrossRef]
| Summary of Effects | References | Models Used | Strength | Notes | |
|---|---|---|---|---|---|
| Proliferation | Increased—10−5–10 μM No effect—0.1–50 μM; Decreased—10−4–1 μM; ≥100 μM | Benachour 2007 Spagnoletti 2015 Morice 2011 Morck 2010 Basak 2018 Greca 2019 Greca 2020 Profita 2021 | Embryonic 293 cells HTR-8/SVneo cells JEG-3, BeWo cells BeWo cells HTR-8/SVneo cells BeWo cells BeWo cells HTR-8/SVneo cells | STRONG | Different cell lines vary in the concentrations wherein increased proliferation occurs, but higher concentrations ≥ 100 μM are consistently cytotoxic Cell number of BPA-treated cells was significantly decreased when treated with G15, a GPr30 inhibitor |
| Trophoblast motility | Decreased migration—10−5–50 μM Decreased attachment—10–100 μM Decreased integrin, vimentin—0.1–10 μM Decreased E-cadherin—10−3–100 μM | Spagnoletti 2015 Lan 2017 Wang 2015 Gao 2019 Wei 2020 | HTR-8/SVneo cells HTR-8/SVneo cells BeWo cells JEG-3 cells HTR-8/SVneo cells | STRONG | In HTR/SV8neo cells, membrane ER GPR30 antagonist, G15, partially restored HTR-8/SVneo trophoblast migration |
| Placenta morphology | Decreased labyrinthine zones, thinner spongiotrophoblasts—200 μg–50 mg/kg/day Decreased ratio of spongiotrophoblast zone to giant cell area—200 μg/kg Increased total vessel area—50 mg/kg/day | Tachibana 2007 Tait 2015 Lan 2017 Chu 2018 Mao 2020 Gwon 2020 | Mouse placenta Mouse placenta HTR-8/SVneo cells JEG-3 cells Mouse placenta Mouse placenta | STRONG | |
| Trophoblast morphology | Increased syncytin and cell fusion—0.05 μM Decreased villous outgrowth—0.1 to 50 μM Increased villous outgrowth—10 μM Reduced syncytialization—0.5 mg/kg/day, 1 uM | Tait 2015 Narciso 2019 Gao 2019 | Mouse placenta BeWo cells JEG-3 cells | STRONG | Increased villous outgrowth observed for trophoblast spheroid cell models; decreased villous outgrowth in 2D trophoblast cell cultures |
| CRH production | Increased CRH expression—200 mg/kg, 25–50 μM | Huang 2012 Tan 2013 | JEG-3 cells Mouse placenta | STRONG | At 1–50 μM, increasing p-PKA, p-PKCalpha, which may mediate CRH expression |
| Steroidogenesis | Decreased CYP11A1, CYP19—10−3–100 μM Decreased progesterone, estradiol—10−3–1 μM Decreased aromatase—5–1000 μM Increased CYP1A1—10–50 μM No effect on serum progesterone in sheep—0.5 mg/kg/day | Benachour 2007 Nativelle-Serpentini 2003 Huang and Leung 2009 Chu 2018 Xu 2019 | Embryonic 293 cells JEG-3 cells JEG-3 cells JEG-3 cells JEG-3 cells | LIMITED | In JEG-3 cells, no difference with CYP19 production |
| MMPs | Decreased MMP2, MMP9—10−3–100 μM Increased MMP9—0.1–10 μM | Wang 2015 Wei 2020 | BeWo cells HTR-8/SVneo cells | LIMITED | |
| TIMP | Increased TIMP1, TIMP2—10−3–100 μM Decreased TIMP3—10–50 μM | Lan 2017 Wang 2015 Wei 2020 | HTR-8/SVneo cells BeWo cells HTR-8/SVneo cells | LIMITED | |
| miR-146a | Increased—25 ng/μL | Avissar-Whiting 2010 de Felice 2015 | SV40 cells Human placenta | LIMITED | miR-146a was the only miRNA validated by qRT-PCR as significantly upregulated in both 3A and HTR-8 cells with BPA treatment at 25 ng/μL |
| Other hormone production | Decreased serotonin—200 μg/kg Increased dopamine—200 μg/kg Decreased B-hCG—103–108 μM Increased testosterone concentrations—20–200 mg/kg/day | Morck 2010 Tan 2013 Mao 2020 | BeWo cells Mouse placenta Mouse placenta | LIMITED | Not necessarily concluding towards preterm birth |
| Apoptosis-related | Increased apoptosis—0.01–0.1 μM Increase caspase-3 production—10−6 μM Decreased caspase-3 production—9 μM | Morice 2011 Narciso 2019 | JEG-3, BeWo cells BeWo cells | INCONCLUSIVE | |
| Oxidative stress | Increased ROS production—50–500 μM Increased HSP70, Bcl2—0.9 to 9.0 μM Decreased GSH—0.9 to 9.0 μM Increased Nrf—9.0 μM | Ponniah 2015 Perez-Albaladejo 2017 | BeWo cells JEG-3 cells | INCONCLUSIVE | Decrease in GSH + increased ROS production increases oxidative damage, while Nrf, HSP70, and Bcl2 production may be counterregulatory measures |
| Inflammation | Decreased TNF-alpha—1.4 × 10−6 to 0.04 μM Increased IL-6—1–10 μM Increased IL-1B—0.001–10 μM | Benachour and Aziz 2009 Arita 2019 | Human primary cytotrophoblasts Placental explants | INCONCLUSIVE | |
| PR receptor | Decrease in PR expression—2 μg/kg/day | Imanishi 2003 | Mouse placenta | INCONCLUSIVE | Decrease in PR expression was embryo sex-selective |
| ER receptor | BPA binds to ERRg about 100 times greater than to ERa and Erb | Takeda 2009 | Human placenta | INCONCLUSIVE | May provide alternative hypothesis for a pathway for other BPA actions other than classical steroid receptors |
| ERK1/2 | Increased p-ERK—10−3–1 μM | Chu 2018 Lan 2017 | JEG-3 cells HTR-8/SVneo cells | INCONCLUSIVE | treatment with the extracellular signal-regulated kinase (ERK1/2) inhibitor U0126 or the PI3K inhibitor LY294002 for 24 h abolished the BPA-induced phosphorylation of ERK and Akt |
| Akt | Increased p-Akt—0.01 μM | Lan 2017 Greca 2020 | HTR-8/SVneo cells BeWo cells | INCONCLUSIVE | treatment with the extracellular signal-regulated kinase (ERK1/2) inhibitor U0126 or the PI3K inhibitor LY294002 for 24 h abolished the BPA-induced phosphorylation of ERK and Akt |
| PAG1 and PSPB | Decreased serum concentration—0.5 mg/kg/day | Gingrich 2018 | Sheep placenta | INCONCLUSIVE | |
| Exosomes | Increased placental exosome expression of HMGB1, caspase 4, MAPK14 expression—20 μM | Sheller-Miller 2020 | Human placental exosomes | INCONCLUSIVE | |
| Summary of Effects | References | Model Used | Strength | Notes | |
|---|---|---|---|---|---|
| Inflammation | Increased LIF, IL-10—10 μM Decreased PAI-1, TNFa—10 μM Decreased CXCL8, IL-6, CCL11—1–10 μM Increased CXCL6—1–10 μM Increased IL-6—10−6–0.001 μmol Increased I-kB, nF-kB, IL-1B—10−6–0.001 μmol | Li 2017 Cho 2018 Fan 2020 Xiong 2020 | Human decidual stromal cells Human endometrial (EM) cells Human EM cells, JEG-3 cells Human EM cells | STRONG | Effects on IL-6 may be discordant due to two different laboratory setups; additionally, total concentration of BPA was not noted in Cho 2018, precluding us from identifying at what exact concentration is the IL-6, I-kB, nF-kB, and IL-1B increase noted. |
| ER expression | Decreased—50–100 μM, 0.5 mg/kg/day–20 mg/kg/day Increased ERa—10−6–1 μM Increased ERB—0.001 μM | Varayoud 2008 Aghajanova and Giudice 2011 Mannelli 2015 Cho 2018 | Human uterine stromal cells Human EM cells Placental explant Human EM cells | STRONG | Total concentration of BPA was not noted in Cho 2018, precluding us from identifying at what exact concentration is the ERa increase noted. SMRT corepressor decreases ER and PR receptivity as per Varayoud 2008 |
| PR expression | Increased—50 μg/kg/day, 1 μM | Aldad 2011 | Human EM cells, Ishikawa cells | LIMITED | SMRT corepressor decreases ER and PR receptivity as per Varayoud 2008 No effect as per Aghajanova and Giudice 2011 |
| Decidual proliferation | Decreased—50–100 μM, 0.5 mg/kg/day to 20 mg/kg/day Increased—0.01 μM | Varayoud 2008 Aghajanova and Giudice 2011 | Human uterine stromal cells Human EM cells | LIMITED | No effect on viability as per Mannelli et al.; may be due to differences in setups |
| Steroidogenesis | Decreased P450scc—50–100 μM Decreased 17BHSD1—50–100 μM Increased SMRT—0.5 mg/kg/day and 20 mg/kg/day | Aghajanova and Giudice 2011 Varayoud 2008 | Human EM cells Human uterine stromal cells | LIMITED | |
| IGFBP-1 | Increased—10−8 to 10−11 μM, 50 μM | Aghajanova and Giudice 2011 Forte 2015 Fan 2020 | Human EM cells Human EM cells Human EM cells, Ishikawa cells | LIMITED | No effect on IGFBP-1 secretion as per Mannelli et al.; may be due to differences in setups |
| PRL | Increased—10−8–10−11 μM, 0.1–10 uM Decreased—0.1–88 μM | Forte 2015 Mannelli 2015 Olson 2017 Fan 2020 Xiong 2020 | Human EM cells Placental explants HuF cells Human EM cells, Ishikawa cells Human EM cells | LIMITED | Same endometrial cells, but different effects, perhaps due to different laboratory setups At lower concentrations, PRL appears to be increased, while for higher concentrations, PRL appears to be decreased |
| ENaC | Decreased—0.01–1 μg/mL | Yuan 2018 | Mouse EM cells, Ishikawa cells | INCONCLUSIVE | |
| SGK1 | Decreased—100 μg/kg | Yuan 2018 | Mouse EM cells, Ishikawa cells | INCONCLUSIVE | |
| Cell cycle | Lower G0/G1 cycles, Higher G2/M cycles—10−11–10−8 μM Decreased CCND2—44 μM | Forte 2015 Olson 2017 | Human EM cells HuF cells | INCONCLUSIVE | |
| Hoxa10/11 | Decreased—0.5–20 mg/kg/day | Varayoud 2008 | Human uterine stromal cells | INCONCLUSIVE | |
| Egr1 | Increased—20–500 mg/kg | Kim 2017 | Mouse uterus | INCONCLUSIVE | ERK1/2 and AKT were rapidly activated by BPA, BPA-induced Egr1 expression is mediated by ERK1/2, but not AKT phosphorylation |
| p-ERK | Increased—10−6–0.001 μmol | Cho 2018 | Human EM cells | INCONCLUSIVE | Both appear to increase similarly, but ERK may be more involved in the signaling process than Akt (Kim 2017) |
| p-Akt | Increased—10−6–0.001 μmol | Cho 2018 | Human EM cells | INCONCLUSIVE | |
| p-JNK | Increased—10−6–0.001 μmol | Cho 2018 | Human EM cells | INCONCLUSIVE | |
| Histone methylation | Decreased MLL1—0.1–10 μM Increased EZH2—0.1–10 μM Increased H3K4me3—0.1–10 μM Increased H3K27me3—10−5–10 μM | Xiong 2020 | Human EM cells | INCONCLUSIVE | |
| Oxidative stress | Decreased SOD, catalase, HO—10−6–0.001 μmol | Cho 2018 | Human EM cells | INCONCLUSIVE | Total concentration of BPA was not noted in this experiment; only picomolar values were given |
| Summary of Effects | References | Model Used | Strength | Notes | |
|---|---|---|---|---|---|
| Uterine weight and thickness | Increased—2–30 mg/kg/day, 5 mL/kg Increased thickness—50 mg/kg/day Increased endometrial area—3.5 mg/kg Increased luminal epithelium height—2 mg/day | Dodge 1996 Ashby and Tinwell 1998 Papaconstantinou 2000 Okuda 2010 | Mouse uterus Mouse uterus Mouse uterus Human myometrial cells | STRONG | No effect on uterus weight and histological parameters as per Okuda; differences may be due to difference in lab setups EM thickness increase in ewe lambs; luminal height in mice |
| Uterine contractility | Decreased—10−6–10−4 μM Decreased amplitude—1–10 μM | An 2013 Gupta and Deshpande 2018 Salleh 2015 | Rat uterus Rat uterus Rat uterus | STRONG | |
| VACht fibers | Increased—0.05 and 0.5 mg/kg/day | Liliana 2019 | Pig uterus | LIMITED | |
| DBH fibers | Increased—0.05 and 0.5 mg/kg/day | Liliana 2019 | Pig uterus | LIMITED | |
| Implantation rates | Decreased—100 mg/kg/day Decreased—implantation sites—6.75 and 10.125 mg/kg/day Delayed embryo reception—40 mg/kg/day | Xiao 2011 Berger 2010 | Mouse myometrial cells Mouse myometrial cells | LIMITED | |
| hsp27 | Increased—100 mg/kg/day | Papaconstantinou 2001 | Mouse uterus | INCONCLUSIVE | |
| grp94 | Increased—100 mg/kg/day | Papaconstantinou 2002 | Mouse uterus | INCONCLUSIVE | |
| oxytocin and oxytocin receptor | Increased oxytocin—500 mg/kg/day Increased oxytocin receptor—100 mg/kg/day | An 2013 | Rat uterus | INCONCLUSIVE | |
| PR expression | Decreased—100 mg/kg/day Decreased—10.125 mg/kg/day | Xiao 2011 Berger 2010 | Mouse myometrial cells Mouse myometrial cells | INCONCLUSIVE | |
| ER expression | Decreased—10.125 mg/kg/day | Berger 2010 | Mouse myometrial cells | INCONCLUSIVE | |
| Gene expression profile | Increased genes for PI3K-Akt signaling, metabolic pathways, cancer pathways, actin regulation, ECM interaction, focal adhesion—0.01 μM | Kang 2014 | Human myometrial cells | INCONCLUSIVE | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vidal, M.S., Jr.; Menon, R.; Yu, G.F.B.; Amosco, M.D. Actions of Bisphenol A on Different Feto-Maternal Compartments Contributing to Preterm Birth. Int. J. Mol. Sci. 2022, 23, 2411. https://doi.org/10.3390/ijms23052411
Vidal MS Jr., Menon R, Yu GFB, Amosco MD. Actions of Bisphenol A on Different Feto-Maternal Compartments Contributing to Preterm Birth. International Journal of Molecular Sciences. 2022; 23(5):2411. https://doi.org/10.3390/ijms23052411
Chicago/Turabian StyleVidal, Manuel S., Jr., Ramkumar Menon, Gracia Fe B. Yu, and Melissa D. Amosco. 2022. "Actions of Bisphenol A on Different Feto-Maternal Compartments Contributing to Preterm Birth" International Journal of Molecular Sciences 23, no. 5: 2411. https://doi.org/10.3390/ijms23052411
APA StyleVidal, M. S., Jr., Menon, R., Yu, G. F. B., & Amosco, M. D. (2022). Actions of Bisphenol A on Different Feto-Maternal Compartments Contributing to Preterm Birth. International Journal of Molecular Sciences, 23(5), 2411. https://doi.org/10.3390/ijms23052411







