Roles and Regulation of BCL-xL in Hematological Malignancies
Abstract
1. Introduction
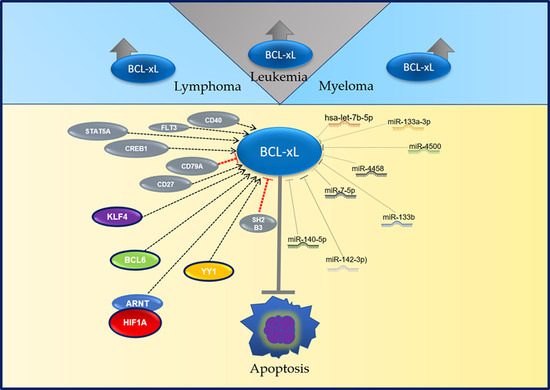
2. BCL-xL in Hematological Malignancies
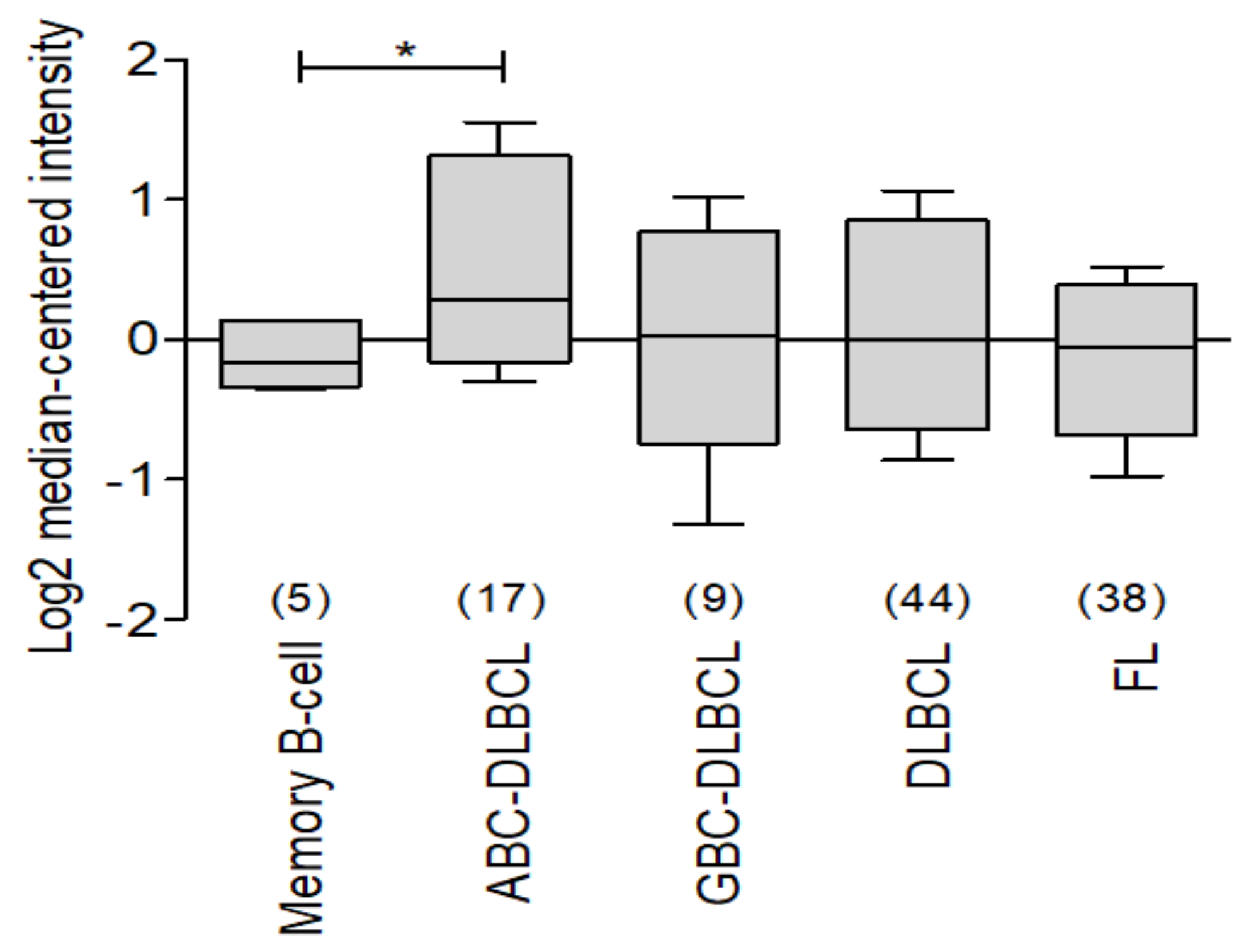
2.1. BCL-xL in Non-Hodgkin Lymphoma
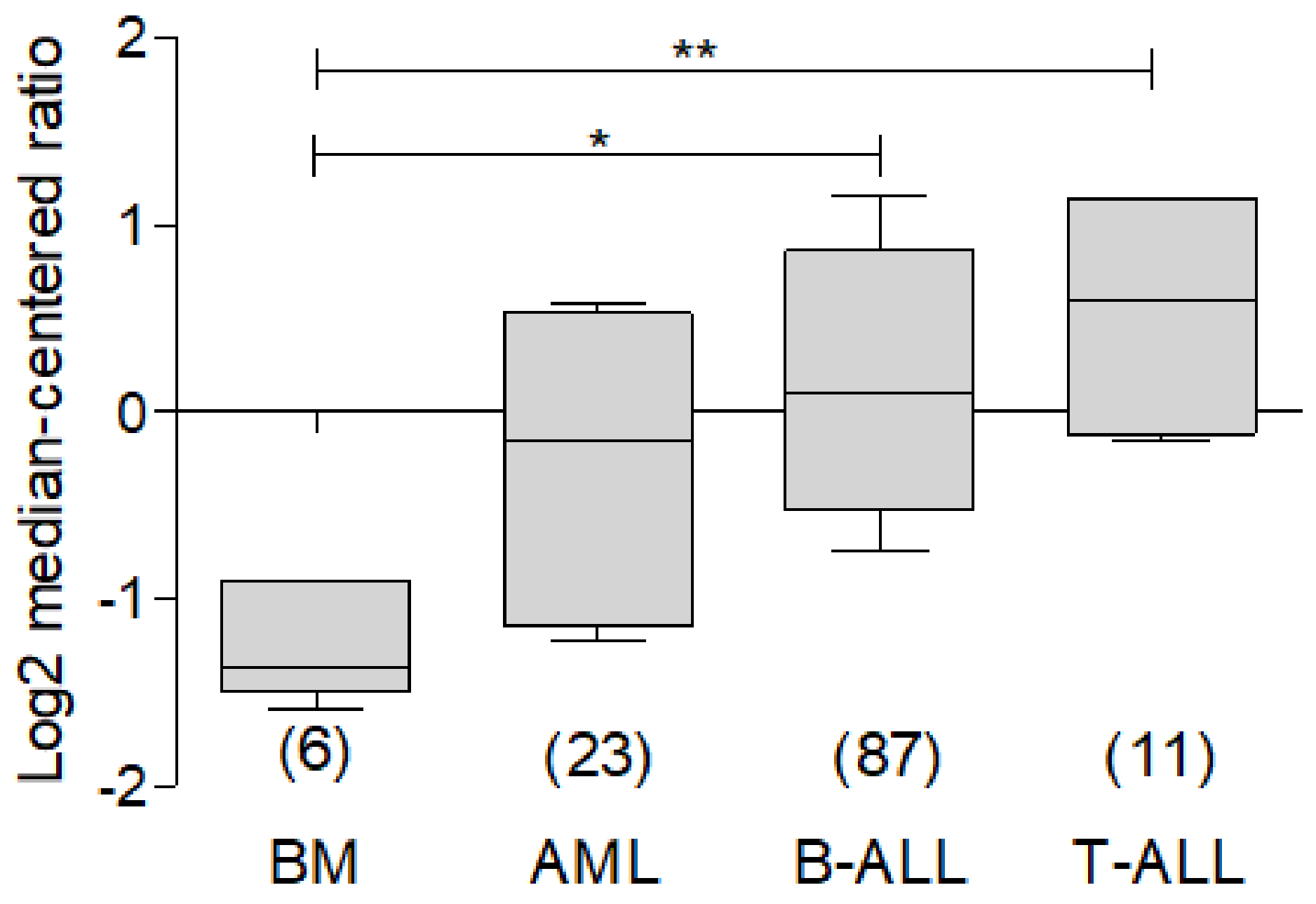
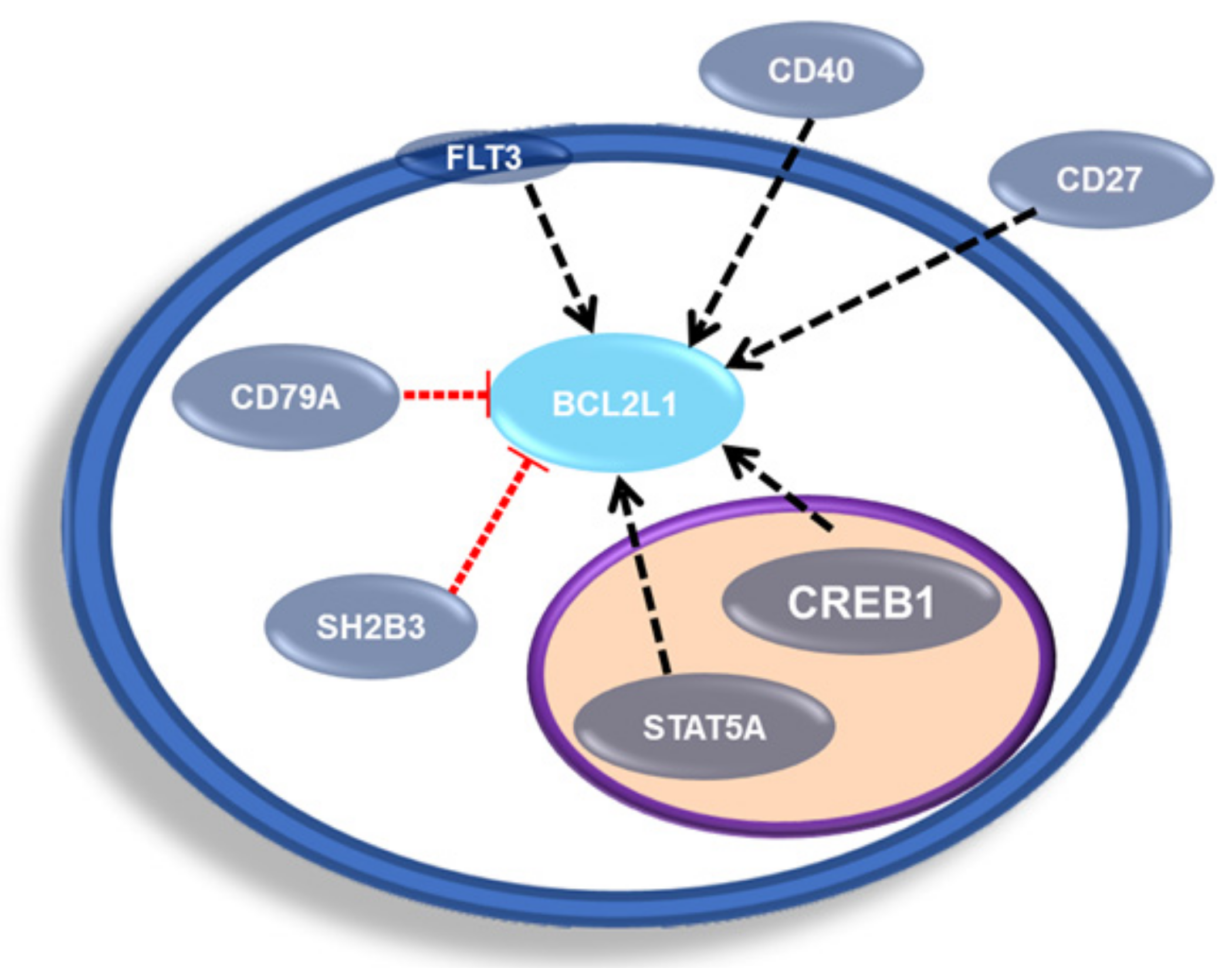
2.2. BCL-xL in Leukemia
2.3. BCL-xL in Multiple Myeloma
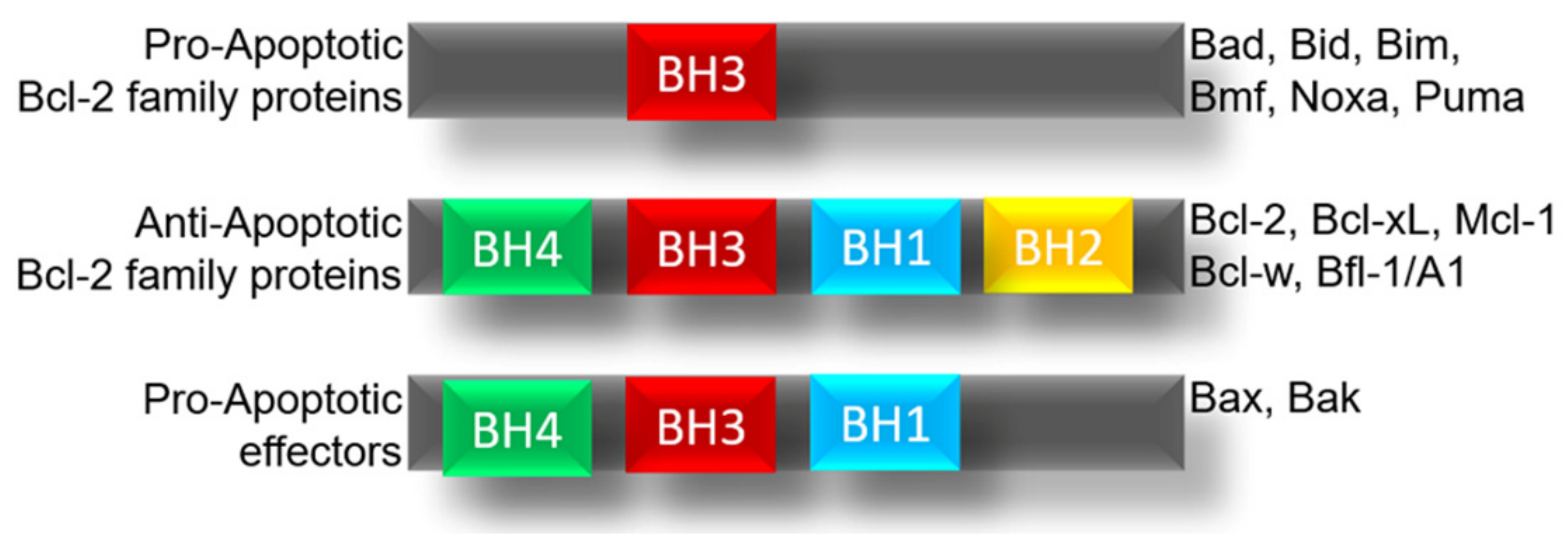
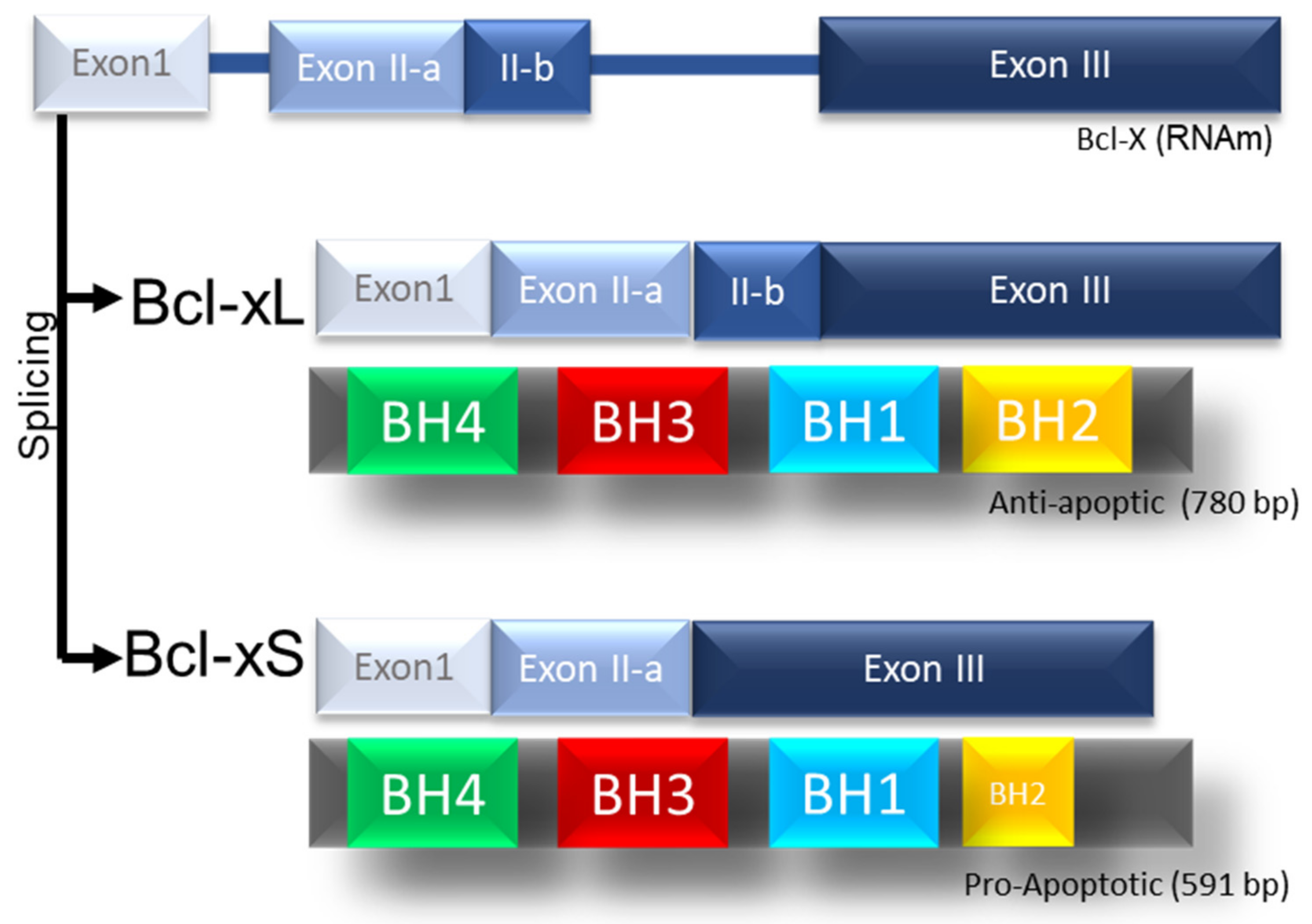
3. Regulation of BCL-xL
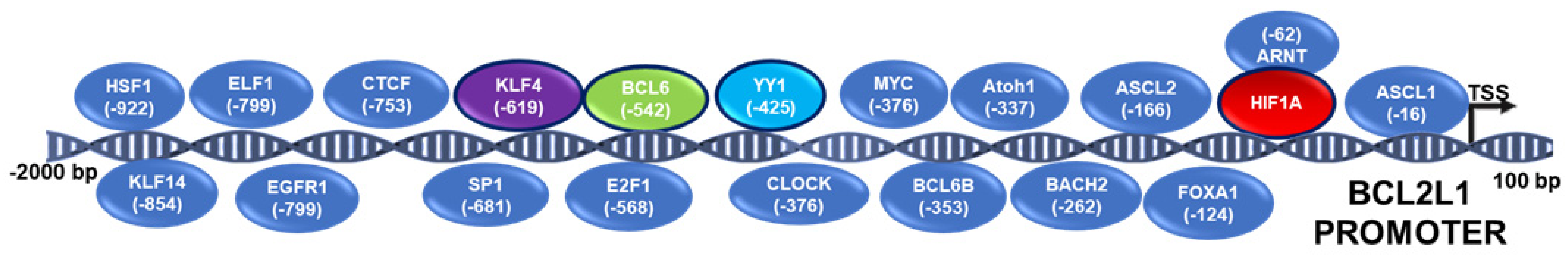
3.1. BCL-xL Regulation by Transcription Factors
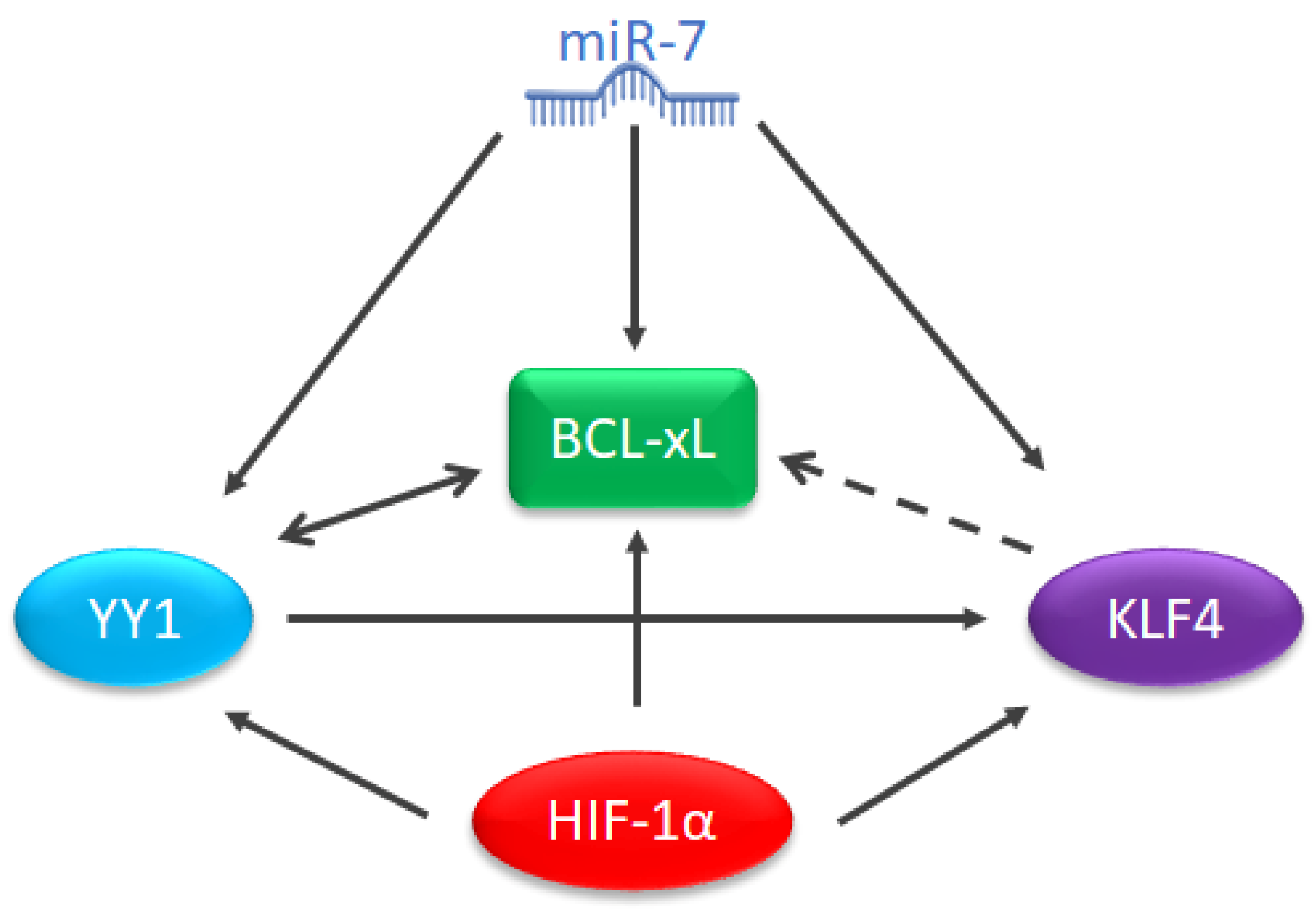
3.2. MicroRNAs and BCL-xL
3.3. LeukemiRs and LymphomiRs in the Regulation of BCL-xL
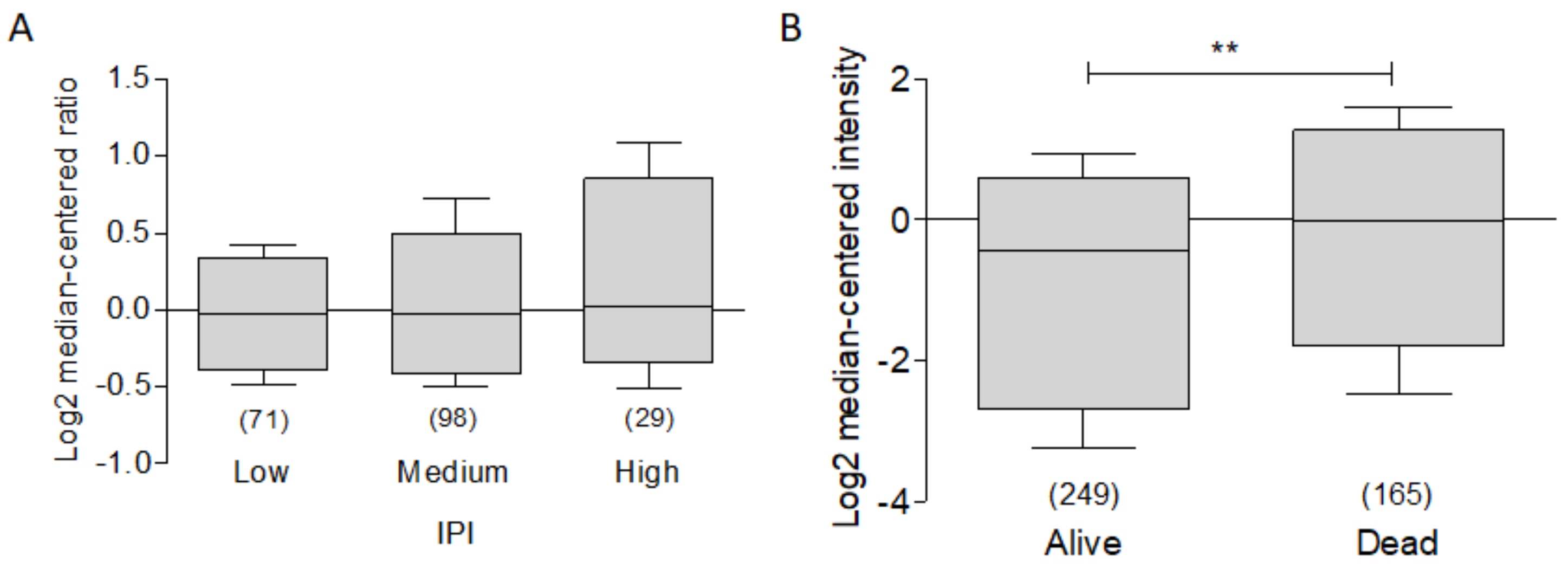
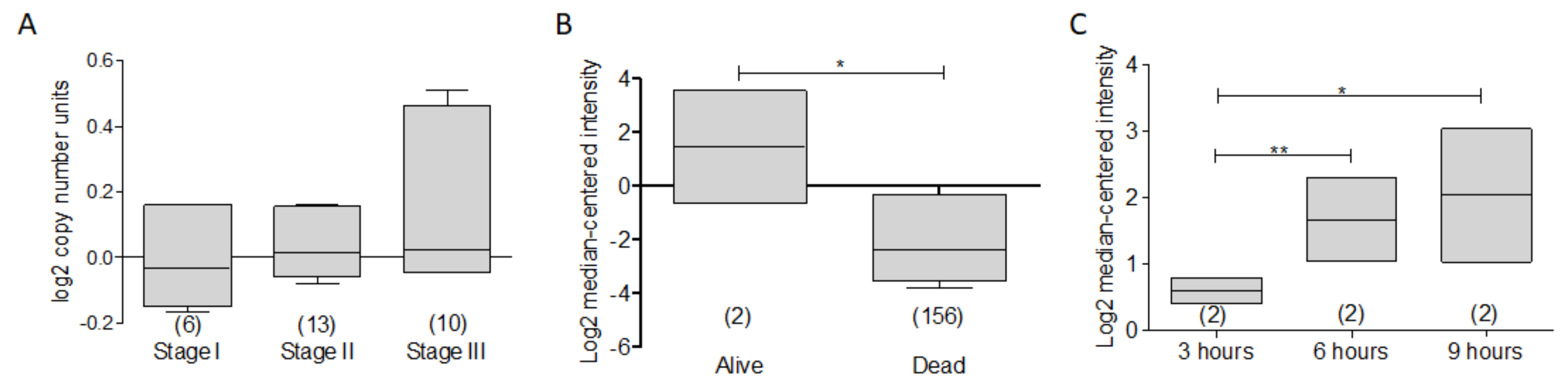
3.4. BCL-XL as a Potential Biomarker in Hematological Malignancies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| B-ALL | B-cell Acute lymphocytic leukemia |
| B-NHL | B non-Hodgkin lymphoma |
| DLBCL | diffuse large B cell lymphoma |
| FL | Follicular Lymphoma |
| IHC | immunohistochemistry |
| IPI | International prognostic index |
| MGUS | Monoclonal gammopathy of undetermined significance |
| MM | Multiple Myeloma |
| MZBCL | marginal zone b-cell lymphoma |
| SM | Smoldering Myeloma |
| T-ALL | T-cell Acute lymphocytic leukemia |
References
- Morales-Martinez, M.; Vega, M.I. Participation of different miRNAs in the regulation of YY1: Their role in pathogenesis, chemoresistance, and therapeutic implication in hematologic malignancies. YY1 Control. Pathog. Drug Resist. Cancer. 2021, 171–198. [Google Scholar] [CrossRef]
- Xerri, L.; Parc, P.; Brousset, P.; Schlaifer, D.; Hassoun, J.; Reed, J.C.; Krajewski, S.; Birnbaum, D. Predominant expression of the long isoform of Bcl-x (Bcl-xL) in human lymphomas. Br. J. Haematol. 1996, 92, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Shangary, S.; E Johnson, D. Recent advances in the development of anticancer agents targeting cell death inhibitors in the Bcl-2 protein family. Leukemia 2003, 17, 1470–1481. [Google Scholar] [CrossRef]
- Chapman, M.A.; Lawrence, M.S.; Keats, J.; Cibulskis, K.; Sougnez, C.; Schinzel, A.C.; Harview, C.; Brunet, J.-P.; Ahmann, G.J.; Adli, M.; et al. Initial genome sequencing and analysis of multiple myeloma. Nature 2011, 471, 467–472. [Google Scholar] [CrossRef]
- Minn, A.; Rudin, C.; Boise, L.; Thompson, C. Expression of bcl-xL can confer a multidrug resistance phenotype. Blood 1995, 86, 1903–1910. [Google Scholar] [CrossRef] [PubMed]
- Youle, R.J.; Strasser, A. The BCL-2 protein family: Opposing activities that mediate cell death. Nat. Rev. Mol. Cell Biol. 2008, 9, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Czabotar, P.E.; Lessene, G.; Strasser, A.; Adams, J.M. Control of apoptosis by the BCL-2 protein family: Implications for physiology and therapy. Nat. Rev. Mol. Cell Biol. 2014, 15, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Boise, L.; González-García, M.; Postema, C.E.; Ding, L.; Lindsten, T.; Turka, L.A.; Mao, X.; Nuñez, G.; Thompson, C.B. bcl-x, a bcl-2-related gene that functions as a dominant regulator of apoptotic cell death. Cell 1993, 74, 597–608. [Google Scholar] [CrossRef]
- Tsujimoto, Y.; Shimizu, S. Bcl-2 family: Life-or-death switch. FEBS Lett. 2000, 466, 6–10. [Google Scholar] [CrossRef]
- Nakamura, A.; Swahari, V.; Plestant, C.; Smith, I.; McCoy, E.; Smith, S.; Moy, S.S.; Anton, E.S.; Deshmukh, M. Bcl-xL Is Essential for the Survival and Function of Differentiated Neurons in the Cortex That Control Complex Behaviors. J. Neurosci. 2016, 36, 5448–5461. [Google Scholar] [CrossRef] [PubMed]
- Borrás, C.; Mas-Bargues, C.; Román-Domínguez, A.; Sanz-Ros, J.; Gimeno-Mallench, L.; Inglés, M.; Gambini, J.; Viña, J. BCL-xL, a Mitochondrial Protein Involved in Successful Aging: From C. elegans to Human Centenarians. Int. J. Mol. Sci. 2020, 21, 418. [Google Scholar] [CrossRef] [PubMed]
- Muchmore, S.W.; Sattler, M.; Liang, H.; Meadows, R.P.; Harlan, J.E.; Yoon, H.S.; Nettesheim, D.; Chang, B.S.; Thompson, C.B.; Wong, S.-L.; et al. X-ray and NMR structure of human Bcl-xL, an inhibitor of programmed cell death. Nature 1996, 381, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Trisciuoglio, D.; Tupone, M.G.; Desideri, M.; Di Martile, M.; Gabellini, C.; Buglioni, S.; Pallocca, M.; Alessandrini, G.; D’Aguanno, S.; Del Bufalo, D. BCL-XL overexpression promotes tumor progression-associated properties. Cell Death Dis. 2017, 8, 3216. [Google Scholar] [CrossRef] [PubMed]
- Hager, J.H.; Ulanet, D.B.; Hennighausen, L.; Hanahan, D. Genetic Ablation of Bcl-x Attenuates Invasiveness without Affecting Apoptosis or Tumor Growth in a Mouse Model of Pancreatic Neuroendocrine Cancer. PLoS ONE 2009, 4, e4455. [Google Scholar] [CrossRef]
- Choi, S.; Chen, Z.; Tang, L.H.; Fang, Y.; Shin, S.J.; Panarelli, N.C.; Chen, Y.-T.; Li, Y.; Jiang, X.; Du, Y.-C.N. Bcl-xL promotes metastasis independent of its anti-apoptotic activity. Nat. Commun. 2016, 7, 10384. [Google Scholar] [CrossRef] [PubMed]
- Cho, A.A.; Bonavida, B. Targeting the Overexpressed YY1 in Cancer Inhibits EMT and Metastasis. Crit. Rev. Oncog. 2017, 22, 49–61. [Google Scholar] [CrossRef]
- Su, R.-J.; Baylink, D.J.; Neises, A.; Kiroyan, J.B.; Meng, X.; Payne, K.J.; Tschudy-Seney, B.; Duan, Y.; Appleby, N.; Kearns-Jonker, M.; et al. Efficient Generation of Integration-Free iPS Cells from Human Adult Peripheral Blood Using BCL-XL Together with Yamanaka Factors. PLoS ONE 2013, 8, e64496. [Google Scholar] [CrossRef]
- Gabellini, C.; Castellini, L.; Trisciuoglio, D.; Kracht, M.; Zupi, G.; Del Bufalo, D. Involvement of nuclear factor-kappa B in bcl-xL-induced interleukin 8 expression in glioblastoma. J. Neurochem. 2008, 107, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Gabellini, C.; Gómez-Abenza, E.; Ibáñez-Molero, S.; Tupone, M.G.; Pérez-Oliva, A.B.; de Oliveira, S.; Del Bufalo, D.; Mulero, V. Interleukin 8 mediates bcl-xL-induced enhancement of human melanoma cell dissemination and angiogenesis in a zebrafish xenograft model. Int. J. Cancer 2017, 142, 584–596. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, Y.; Song, T.; Xue, Z.; Shen, X.; Liang, F.; Zhao, Y.; Li, Z.; Sheng, H. An antiapoptotic Bcl-2 family protein index predicts the response of leukaemic cells to the pan-Bcl-2 inhibitor S1. Br. J. Cancer 2013, 108, 1870–1878. [Google Scholar] [CrossRef][Green Version]
- Scheffold, A.; Jebaraj, B.M.C.; Stilgenbauer, S. Venetoclax: Targeting BCL2 in Hematological Cancers. In Small Molecules in Hematology. Recent Results in Cancer Research; Martens, U., Ed.; Springer: Cham, Switzerland, 2018; Volume 212. [Google Scholar]
- Yalniz, F.F.; Wierda, W.G. Targeting BCL2 in Chronic Lymphocytic Leukemia and Other Hematologic Malignancies. Drugs 2019, 79, 1287–1304. [Google Scholar] [CrossRef]
- Zhu, Y.; Doornebal, E.; Pirtskhalava, T.; Giorgadze, N.; Wentworth, M.; Fuhrmann-Stroissnigg, H.; Niedernhofer, L.; Robbins, P.; Tchkonia, T.; Kirkland, J.L. New agents that target senescent cells: The flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463. Aging 2017, 9, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Boehm, A.L.; Miranda, M.B.; Shangary, S.; Grandis, J.R.; E Johnson, D. Targeting Antiapoptotic Bcl-2 Family Members with Cell-Permeable BH3 Peptides Induces Apoptosis Signaling, Death in Head, Neck Squamous Cell Carcinoma Cells. Neoplasia 2007, 9, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Mateos-Chávez, A.A.; Muñoz-López, P.; Becerra-Báez, E.I.; Flores-Martínez, L.F.; Prada-Gracia, D.; Moreno-Vargas, L.M.; Baay-Guzmán, G.J.; Juárez-Hernández, U.; Chávez-Munguía, B.; Cabrera-Muñóz, L.; et al. Live Attenuated Salmonella enterica Expressing and Releasing Cell-Permeable Bax BH3 Peptide Through the MisL Autotransporter System Elicits Antitumor Activity in a Murine Xenograft Model of Human B Non-hodgkin’s Lymphoma. Front. Immunol. 2019, 10, 2562. [Google Scholar] [CrossRef]
- Rhodes, D.R.; Yu, J.; Shanker, K.; Deshpande, N.; Varambally, R.; Ghosh, D.; Barrette, T.; Pander, A.; Chinnaiyan, A.M. ONCOMINE: A Cancer Microarray Database and Integrated Data-Mining Platform. Neoplasia 2004, 6, 533–545. [Google Scholar] [CrossRef]
- Basso, K.; A Margolin, A.; Stolovitzky, G.; Klein, U.; Dalla-Favera, R.; Califano, A. Reverse engineering of regulatory networks in human B cells. Nat. Genet. 2005, 37, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Storz, M.N.; van de Rijn, M.; Kim, Y.H.; Mraz-Gernhard, S.; Kohler, S.; Hoppe, R.T. Gene Expression Profiles of Cutaneous B Cell Lymphoma. J. Investig. Dermatol. 2003, 120, 865–870. [Google Scholar] [CrossRef]
- McDonnell, T.J.; Deane, N.; Platt, F.; Nunez, G.; Jaeger, U.; McKearn, J.P.; Korsmeyer, S.J. bcl-2-Immunoglobulin transgenic mice demonstrate extended B cell survival and follicular lymphoproliferation. Cell 1989, 57, 79–88. [Google Scholar] [CrossRef]
- Huang, J.Z.; Sanger, W.G.; Greiner, T.C.; Staudt, L.M.; Weisenburger, D.D.; Pickering, D.L.; Lynch, J.C.; Armitage, J.O.; Warnke, R.A.; Alizadeh, A.A.; et al. The t(14;18) defines a unique subset of diffuse large B-cell lymphoma with a germinal center B-cell gene expression profile. Blood 2002, 99, 2285–2290. [Google Scholar] [CrossRef]
- Kang, M.H.; Reynolds, C.P. Bcl-2 Inhibitors: Targeting Mitochondrial Apoptotic Pathways in Cancer Therapy. Clin. Cancer Res. 2009, 15, 1126–1132. [Google Scholar] [CrossRef]
- Martínez-Paniagua, M.A.; Baritaki, S.; Huerta-Yepez, S.; Ortiz-Navarrete, V.F.; González-Bonilla, C.; Bonavida, B.; Vega, M.I. Mcl-1 and YY1 inhibition and induction of DR5 by the BH3-mimetic Obatoclax (GX15-070) contribute in the sensitization of B-NHL cells to TRAIL apoptosis. Cell Cycle 2011, 10, 2792–2805. [Google Scholar] [CrossRef] [PubMed]
- Compagno, M.; Lim, W.K.; Grunn, A.; Nandula, S.V.; Brahmachary, M.; Shen, Q.; Bertoni, F.; Ponzoni, M.; Scandurra, M.; Califano, A.; et al. Mutations of multiple genes cause deregulation of NF-κB in diffuse large B-cell lymphoma. Nature 2009, 459, 717–721. [Google Scholar] [CrossRef]
- Perri, M.; Yap, J.L.; Yu, J.; Cione, E.; Fletcher, S.; Kane, M.A.; Perri, M.; Yap, J.L.; Yu, J.; Cione, E.; et al. BCL-xL/MCL-1 inhibition and RARγ antagonism work cooperatively in human HL60 leukemia cells. Exp. Cell Res. 2014, 327, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Mirjolet, J.-F.; Barberi-Heyob, M.; Didelot, C.M.; Peyrat, J.-P.; Abecassis, J.; Millon, R.; Merlin, J.-L. Bcl-2/Bax protein ratio predicts 5-fluorouracil sensitivity independently of p53 status. Br. J. Cancer 2000, 83, 1380–1386. [Google Scholar] [CrossRef]
- Kogan, S.C.; Brown, D.E.; Shultz, D.; Truong, B.-T.H.; Lallemand, V.; Guillemin, M.-C.; Lagasse, E.; Weissman, I.L.; Bishop, J.M. Bcl-2 Cooperates with Promyelocytic Leukemia Retinoic Acid Receptor α Chimeric Protein (Pmlrarα) to Block Neutrophil Differentiation and Initiate Acute Leukemia. J. Exp. Med. 2001, 193, 531–544. [Google Scholar] [CrossRef]
- Zou, J.; Li, P.; Lu, F.; Liu, N.; Dai, J.; Ye, J.; Qu, X.; Sun, X.; Ma, D.; Park, J.; et al. Notch1 is required for hypoxia-induced proliferation, invasion and chemoresistance of T-cell acute lymphoblastic leukemia cells. J. Hematol. Oncol. 2013, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Chen, Y.; Li, C.; Qiao, Z.; Zhou, M.; Findley, H.W.; Wang, B. [Relationship between bcl-X(L) expression and sensitivity to chemotherapy in acute myeloid leukemic cells]. Zhonghua Xue Ye Xue Za Zhi 1997, 18, 584–587. [Google Scholar]
- Souers, A.J.; Leverson, J.D.; Boghaert, E.R.; Ackler, S.L.; Catron, N.D.; Chen, J.; Dayton, B.D.; Ding, H.; Enschede, S.H.; Fairbrother, W.J.; et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat. Med. 2013, 19, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Poeta, G.; Bruno, A.; Del Principe, M.; Venditti, A.; Maurillo, L.; Buccisano, F.; Stasi, R.; Neri, B.; Luciano, F.; Siniscalchi, A.; et al. Deregulation of the Mitochondrial Apoptotic Machinery and Development of Molecular Targeted Drugs in Acute Myeloid Leukemia. Curr. Cancer Drug Targets 2008, 8, 207–222. [Google Scholar] [CrossRef]
- Rahmani, M.; Aust, M.M.; Hawkins, E.; Parker, R.E.; Ross, M.; Kmieciak, M.; Reshko, L.B.; Rizzo, K.A.; Dumur, C.I.; Ferreira-Gonzalez, A.; et al. Co-administration of the mTORC1/TORC2 inhibitor INK128 and the Bcl-2/Bcl-xL antagonist ABT-737 kills human myeloid leukemia cells through Mcl-1 down-regulation and AKT inactivation. Haematologica 2015, 100, 1553–1563. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.; Ritz, C.; Lindgren, D.; Edén, P.; Lassen, C.; Heldrup, J.; Olofsson, T.; Råde, J.; Fontes, M.; Porwit-MacDonald, A.; et al. Microarray-based classification of a consecutive series of 121 childhood acute leukemias: Prediction of leukemic and genetic subtype as well as of minimal residual disease status. Leukemia 2007, 21, 1198–1203. [Google Scholar] [CrossRef]
- Tu, Y.; Renner, S.; Xu, F.; Fleishman, A.; Taylor, J.; Weisz, J.; Vescio, R.; Rettig, M.; Berenson, J.; Krajewski, S.; et al. BCL-X expression in multiple myeloma: Possible indicator of chemoresistance. Cancer Res. 1998, 58, 256–262. [Google Scholar] [PubMed]
- Punnoose, E.A.; Leverson, J.D.; Peale, F.; Boghaert, E.R.; Belmont, L.D.; Tan, N.; Young, A.; Mitten, M.; Ingalla, E.; Darbonne, W.C.; et al. Expression Profile of BCL-2, BCL-XL, and MCL-1 Predicts Pharmacological Response to the BCL-2 Selective Antagonist Venetoclax in Multiple Myeloma Models. Mol. Cancer Ther. 2016, 15, 1132–1144. [Google Scholar] [CrossRef]
- Badros, A.Z.; Goloubeva, O.; Rapoport, A.P.; Ratterree, B.; Gahres, N.; Meisenberg, B.; Takebe, N.; Heyman, M.; Zwiebel, J.; Streicher, H.; et al. Phase II Study of G3139, a Bcl-2 Antisense Oligonucleotide, in Combination with Dexamethasone and Thalidomide in Relapsed Multiple Myeloma Patients. J. Clin. Oncol. 2005, 23, 4089–4099. [Google Scholar] [CrossRef]
- Lernoux, M.; Schnekenburger, M.; Dicato, M.; Diederich, M. Susceptibility of multiple myeloma to B-cell lymphoma 2 family inhibitors. Biochem. Pharmacol. 2021, 188, 114526. [Google Scholar] [CrossRef]
- Kline, M.P.; Rajkumar, S.V.; Timm, M.M.; Kimlinger, T.K.; Haug, J.L.; Lust, J.A.; Greipp, P.R.; Kumar, S. ABT-737, an inhibitor of Bcl-2 family proteins, is a potent inducer of apoptosis in multiple myeloma cells. Leukemia 2007, 21, 1549–1560. [Google Scholar] [CrossRef] [PubMed]
- Ackler, S.; Mitten, M.J.; Foster, K.; Oleksijew, A.; Refici, M.; Tahir, S.K.; Xiao, Y.; Tse, C.; Frost, D.J.; Fesik, S.W.; et al. The Bcl-2 inhibitor ABT-263 enhances the response of multiple chemotherapeutic regimens in hematologic tumors in vivo. Cancer Chemother. Pharmacol. 2010, 66, 869–880. [Google Scholar] [CrossRef]
- Vaxman, I.; Sidiqi, M.H.; Gertz, M. Venetoclax for the treatment of multiple myeloma. Expert Rev. Hematol. 2018, 11, 915–920. [Google Scholar] [CrossRef]
- Chen, N.; Chen, X.; Huang, R.; Zeng, H.; Gong, J.; Meng, W.; Lu, Y.; Zhao, F.; Wang, L.; Zhou, Q. BCL-xL Is a Target Gene Regulated by Hypoxia-inducible Factor-1α. J. Biol. Chem. 2009, 284, 10004–10012. [Google Scholar] [CrossRef]
- Packham, G.; White, E.L.; Eischen, C.M.; Yang, H.; Parganas, E.; Ihle, J.N.; Grillot, D.A.; Zambetti, G.P.; Nuñez, G.; Cleveland, J.L. Selective regulation of Bcl-XL by a Jak kinase-dependent pathway is bypassed in murine hematopoietic malignancies. Genes Dev. 1998, 12, 2475–2487. [Google Scholar] [CrossRef] [PubMed]
- Puthier, D.; Derenne, S.; Barillé, S.; Moreau, P.; Harousseau, J.-L.; Bataille, R.; Amiot, M. Mcl-1 and Bcl-xL are co-regulated by IL-6 in human myeloma cells. Br. J. Haematol. 1999, 107, 392–395. [Google Scholar] [CrossRef]
- Grad, J.M.; Zeng, X.-R.; Boise, L. Regulation of Bcl-xL: A little bit of this and a little bit of STAT. Curr. Opin. Oncol. 2000, 12, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Licata, L.; Lo Surdo, P.; Iannuccelli, M.; Palma, A.; Micarelli, E.; Perfetto, L.; Peluso, D.; Calderone, A.; Castagnoli, L.; Cesareni, G. SIGNOR 2.0, the SIGnaling Network Open Resource 2.0: 2019 update. Nucleic Acids Res. 2020, 48, D504–D510. [Google Scholar] [CrossRef] [PubMed]
- Dreos, R.; Ambrosini, G.; Groux, R.; Cavin Périer, R.; Bucher, P. The eukaryotic promoter database in its 30th year: Focus on non-vertebrate organisms. Nucleic Acids Res. 2017, 45, D51–D55. [Google Scholar] [CrossRef]
- Huerta-Yepez, S.; Baritaki, S.; Baay-Guzman, G.; Hernandez-Luna, M.; Hernandez-Cueto, A.; I Vega, M.; Bonavida, B. Contribution of either YY1 or BclXL-induced inhibition by the NO-donor DETANONOate in the reversal of drug resistance, both in vitro and in vivo. YY1 and BclXL are overexpressed in prostate cancer. Nitric Oxide 2013, 29, 17–24. [Google Scholar] [CrossRef]
- Morales-Martinez, M.; Valencia, A.; Vega, G.; Neri, N.; Nambo, M.J.; Alvarado, I.; Cuadra, I.; Duran-Padilla, M.A.; Martinez-Maza, O.; Huerta-Yepez, S.; et al. Regulation of Krüppel-Like Factor 4 (KLF4) expression through the transcription factor Yin-Yang 1 (YY1) in non-Hodgkin B-cell lymphoma. Oncotarget 2019, 10, 2173–2188. [Google Scholar] [CrossRef]
- Wu, S.; Kasim, V.; Kano, M.; Tanaka, S.; Ohba, S.; Miura, Y.; Miyata, K.; Liu, X.; Matsuhashi, A.; Chung, U.-I.; et al. Transcription Factor YY1 Contributes to Tumor Growth by Stabilizing Hypoxia Factor HIF-1α in a p53-Independent Manner. Cancer Res. 2013, 73, 1787–1799. [Google Scholar] [CrossRef]
- Morales-Martinez, M.; Vega, G.G.; Neri, N.; Nambo, M.J.; Alvarado, I.; Cuadra, I.; Duran-Padilla, M.A.; Huerta-Yepez, S.; Vega, M.I. MicroRNA-7 Regulates Migration and Chemoresistance in Non-Hodgkin Lymphoma Cells Through Regulation of KLF4 and YY1. Front. Oncol. 2020, 10, 588893. [Google Scholar] [CrossRef]
- Xiong, S.; Zheng, Y.; Jiang, P.; Liu, R.; Liu, X.; Chu, Y. MicroRNA-7 Inhibits the Growth of Human Non-Small Cell Lung Cancer A549 Cells through Targeting BCL-2. Int. J. Biol. Sci. 2011, 7, 805–814. [Google Scholar] [CrossRef]
- Ambros, V. MicroRNA Pathways in Flies and Worms: Growth, Death, Fat, Stress, and Timing. Cell 2003, 113, 673–676. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, Biogenesis, Mechanism, and Function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Calin, G.A.; Dumitru, C.D.; Shimizu, M.; Bichi, R.; Zupo, S.; Noch, E.; Aldler, H.; Rattan, S.; Keating, M.; Rai, K.; et al. Frequent deletions and down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 2002, 99, 15524–15529. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, C.C.; Cheng, H.H.; Tewari, M. MicroRNA profiling: Approaches and considerations. Nat. Rev. Genet. 2012, 13, 358–369. [Google Scholar] [CrossRef]
- Lawrie, C.H. MicroRNAs in hematological malignancies. Blood Rev. 2013, 27, 143–154. [Google Scholar] [CrossRef]
- Yendamuri, S.; A Calin, G. The role of microRNA in human leukemia: A review. Leukemia 2009, 23, 1257–1263. [Google Scholar] [CrossRef]
- Luna-Aguirre, C.M.; Martinez-Fierro, M.D.L.L.; Mar-Aguilar, F.; Garza-Veloz, I.; Treviño-Alvarado, V.; Rojas-Martinez, A.; Jaime-Perez, J.C.; Malagon-Santiago, G.I.; Gutierrez-Aguirre, C.H.; Gonzalez-Llano, O.; et al. Circulating microRNA expression profile in B-cell acute lymphoblastic leukemia. Cancer Biomark. 2015, 15, 299–310. [Google Scholar] [CrossRef]
- Bonavida, B. (Ed.) YY1 in the Control of the Pathogenesis and Drug Resistance of Cancer; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar] [CrossRef]
- The Prognostic Value of Bcl-XL Gene Expression for Remission Induction Is Influenced by Cytogenetics in Adult Acute Myeloid Leukemia|Haematologica. Available online: https://www.haematologica.org/article/view/2019 (accessed on 30 November 2021).
- Agnelli, L.; Mosca, L.; Fabris, S.; Lionetti, M.; Andronache, A.; Kwee, I.; Todoerti, K.; Verdelli, D.; Battaglia, C.; Bertoni, F.; et al. A SNP microarray and FISH-based procedure to detect allelic imbalances in multiple myeloma: An integrated genomics approach reveals a wide gene dosage effect. Genes Chromosom. Cancer 2009, 48, 603–614. [Google Scholar] [CrossRef]
- Zhan, F.; Huang, Y.; Colla, S.; Stewart, J.P.; Hanamura, I.; Gupta, S.; Epstein, J.; Yaccoby, S.; Sawyer, J.; Burington, B.; et al. The molecular classification of multiple myeloma. Blood 2006, 108, 2020–2028. [Google Scholar] [CrossRef]
- Raab, M.S.; Breitkreutz, I.; Tonon, G.; Zhang, J.; Hayden, P.J.; Nguyen, T.; Fruehauf, J.H.; Lin, B.K.; Chauhan, D.; Hideshima, T.; et al. Targeting PKC: A novel role for beta-catenin in ER stress and apoptotic signaling. Blood 2009, 113, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Neri, A.; Marmiroli, S.; Tassone, P.; Lombardi, L.; Nobili, L.; Verdelli, D.; Civallero, M.; Cosenza, M.; Bertacchini, J.; Federico, M.; et al. The oral protein-kinase Cβinhibitor enzastaurin (LY317615) suppresses signalling through the AKT pathway, inhibits proliferation and induces apoptosis in multiple myeloma cell lines. Leuk. Lymphoma 2008, 49, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Rao, D.; O’Connell, R.M.; Chaudhuri, A.A.; Garcia-Flores, Y.; Geiger, T.L.; Baltimore, D. MicroRNA-34a Perturbs B Lymphocyte Development by Repressing the Forkhead Box Transcription Factor Foxp1. Immunity 2010, 33, 48–59. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, K.A.; Wentzel, E.A.; Zeller, K.I.; Dang, C.; Mendell, J.T. c-Myc-regulated microRNAs modulate E2F1 expression. Nature 2005, 435, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Ivanovska, I.; Ball, A.S.; Diaz, R.L.; Magnus, J.F.; Kibukawa, M.; Schelter, J.M.; Kobayashi, S.V.; Lim, L.; Burchard, J.; Jackson, A.L.; et al. MicroRNAs in the miR-106b Family Regulate p21/CDKN1A and Promote Cell Cycle Progression. Mol. Cell. Biol. 2008, 28, 2167–2174. [Google Scholar] [CrossRef]
- Hernandez-Luna, M.; Rocha-Zavaleta, L.; Vega, M.I.; Huerta-Yepez, S. Hypoxia inducible factor-1? induces chemoresistance phenotype in non-Hodgkin lymphoma cell line via up-regulation of Bcl-xL. Leuk. Lymphoma 2012, 54, 1048–1055. [Google Scholar] [CrossRef]
- Witten, L.; Slack, F.J. miR-155 as a novel clinical target for hematological malignancies. Carcinogenesis 2019, 41, 2–7. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, L.; Barlogie, B.; Stephens, O.; Wu, X.; Williams, D.R.; Cartron, M.-A.; van Rhee, F.; Nair, B.; Waheed, S.; et al. High-risk myeloma is associated with global elevation of miRNAs and overexpression ofEIF2C2/AGO2. Proc. Natl. Acad. Sci. USA 2010, 107, 7904–7909. [Google Scholar] [CrossRef]
- Agnelli, L.; Bisognin, A.; Todoerti, K.; Manzoni, M.; Taiana, E.; Galletti, S.; Cutrona, G.; Gaffo, E.; Bortoluzzi, S.; Neri, A. Expanding the repertoire of miRNAs and miRNA-offset RNAs expressed in multiple myeloma by small RNA deep sequencing. Blood Cancer J. 2019, 9, 21. [Google Scholar] [CrossRef]
- Van Roosbroeck, K.; Calin, G.A. Cancer Hallmarks and MicroRNAs: The Therapeutic Connection. Adv. Cancer Res. 2017, 135, 119–149. [Google Scholar] [CrossRef]
- Al-Harbi, S.; Choudhary, G.S.; Ebron, J.S.; Hill, B.T.; Vivekanathan, N.; Ting, A.H.; Radivoyevitch, T.; Smith, M.R.; Shukla, G.C.; Almasan, A. miR-377-dependent BCL-xL regulation drives chemotherapeutic resistance in B-cell lymphoid malignancies. Mol. Cancer 2015, 14, 185. [Google Scholar] [CrossRef]
- Shen, M.Y.; Wang, Y.; Cui, S.Y.; Wu, X.L.; Guo, Y.; Xu, R.R. MicroRNA-125a regulates proliferation and apoptosis of acute myeloid leukemia through targeting NF-κB pathway. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 3594–3601. [Google Scholar] [CrossRef]
- Gong, J.; Zhang, J.-P.; Li, B.; Zeng, C.; You, K.; Chen, M.-X.; Yuan, Y.; Zhuang, S.-M. MicroRNA-125b promotes apoptosis by regulating the expression of Mcl-1, Bcl-w and IL-6R. Oncogene 2012, 32, 3071–3079. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. miRBase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef]
- Farrugia, M.K.; Sharma, S.B.; Lin, C.-C.; McLaughlin, S.; Vanderbilt, D.B.; Ammer, A.G.; Salkeni, M.; Stoilov, P.; Agazie, Y.M.; Creighton, C.J.; et al. Regulation of anti-apoptotic signaling by Kruppel-like factors 4 and 5 mediates lapatinib resistance in breast cancer. Cell Death Dis. 2015, 6, e1699. [Google Scholar] [CrossRef]
- Gao, J.; Gu, J.; Pan, X.; Gan, X.; Ju, Z.; Zhang, S.; Xiaojie, G.; Lu, L.; Wang, X. Blockade of miR-142-3p promotes anti-apoptotic and suppressive function by inducing KDM6A-mediated H3K27me3 demethylation in induced regulatory T cells. Cell Death Dis. 2019, 10, 332. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Zhang, H.; Wang, Y.; Li, M.; Xu, W.; Kang, Y.; Wang, Z.; Wang, Z.; Cheng, P.; Tong, D.; et al. MicroRNA-133a, downregulated in osteosarcoma, suppresses proliferation and promotes apoptosis by targeting Bcl-xL and Mcl-1. Bone 2013, 56, 220–226. [Google Scholar] [CrossRef]
- Shimizu, S.; Takehara, T.; Hikita, H.; Kodama, T.; Miyagi, T.; Hosui, A.; Tatsumi, T.; Ishida, H.; Noda, T.; Nagano, H.; et al. The let-7 family of microRNAs inhibits Bcl-xL expression and potentiates sorafenib-induced apoptosis in human hepatocellular carcinoma. J. Hepatol. 2010, 52, 698–704. [Google Scholar] [CrossRef]
- Rosenwald, A.; Wright, G.; Chan, W.C.; Connors, J.M.; Campo, E.; Fisher, R.I.; Gascoyne, R.D.; Muller-Hermelink, H.K.; Smeland, E.B.; Giltnane, J.M.; et al. The Use of Molecular Profiling to Predict Survival after Chemotherapy for Diffuse Large-B-Cell Lymphoma. N. Engl. J. Med. 2002, 346, 1937–1947. [Google Scholar] [CrossRef]
- Lenz, G.; Wright, G.; Dave, S.S.; Xiao, W.; Powell, J.; Zhao, H.; Xu, W.; Tan, B.; Goldschmidt, N.; Iqbal, J.; et al. Stromal Gene Signatures in Large-B-Cell Lymphomas. N. Engl. J. Med. 2008, 359, 2313–2323. [Google Scholar] [CrossRef]
- Heuser, M.; Wingen, L.U.; Steinemann, D.; Cario, G.; Von Neuhoff, N.; Tauscher, M.; Bullinger, L.; Krauter, J.; Heil, G.; Döhner, H.; et al. Gene-expression profiles and their association with drug resistance in adult acute myeloid leukemia. Haematologica 2005, 90, 1484–1492. [Google Scholar] [PubMed]
- Bullinger, L.; Döhner, K.; Bair, E.; Fröhling, S.; Schlenk, R.F.; Tibshirani, R.; Döhner, H.; Pollack, J.R. Use of Gene-Expression Profiling to Identify Prognostic Subclasses in Adult Acute Myeloid Leukemia. N. Engl. J. Med. 2004, 350, 1605–1616. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-S.; Wang, Z.-X.; Lv, C.-Y.; Liang, X.-D.; Sun, M.; Guo, Y.-Y.; De, W. Prognostic significance of Bcl-xL gene expression in human colorectal cancer. Acta Histochem. 2011, 113, 810–814. [Google Scholar] [CrossRef]




| Transcription Factor | Potential Sites |
|---|---|
| ARNT::HIF1A | −62, −377, −532, −842 |
| ASCL1 | −16, −17, −112, −342, −383, −522, −523, −628, −835, −867 |
| Ahr::Arnt | −62, −842 |
| Ar | −96, −670, −856 |
| Arnt | −377, −532 |
| Arntl | −377 |
| Ascl2 | −166, −383, −523 |
| Atoh1 | −337 |
| BACH2 | 63, −262 |
| BCL6B | −353, −362, −540 |
| Bcl6 | −542, −734 |
| CLOCK | −337, −376, −377, −532, −533 |
| CTCF | 70, −36, −83, −112, −293, −336, −574, −577, −645, −705, −710, −753 |
| E2F1 | −435, −568, −569, −715, −716, −944 |
| EGR1 | 34, −49, −506, −799 |
| ELF1 | 60, 22, −37, −488, −538, −799 |
| FOXA1 | −124 |
| HSF1 | −922 |
| IRF1 | −140 |
| KLF4 | −64, −175, −459, −619, −898 |
| KLF14 | 37, −22, −46, −64, −79, −294, −602, −681, −714, −717, −738, −749, −763, −827, −854 |
| MYC | −376, −533 |
| SP1 | 20, −45, −78, −181, −247, −285, −412, −460, −492, −591, −620, −681, −704, −739, −748, −762, −803, −855 |
| YY1 | −425 |
| Hematological Malignancies | miRNAs Related |
|---|---|
| B-Cell Lymphoma | miR-17-92, miR-34a, miR-21. |
| DLBCL | miR-155, miR-125, |
| FL | miR-17-92, |
| AML | miR-155, miR-125, miR-181, miR-221/222, miR-29a, miR-124a. |
| ALL | miR-17-92, miR-125, miR-15a/16-1, miR-124a. |
| CLL | miR-181, miR-221/222, miR-29a, miR-15a/16-1. |
| CML | miR-17-92, |
| MM | miR-17-92, miR-15a/16-1, miR-21 |
| LeukermiRs | Leukemia Type | Expression | Role |
|---|---|---|---|
| miR-511, miR-222, miR-34 | ALL | High | Leukemogenesis |
| miR-199a-3p, miR-223, miR-221, miR-26a. | ALL | Down | Leukemogenesis |
| miR-181b-5p, miR-181a-3p, miR-181a-5p, miR-342-3p | -- | Dysregulated | Differentiate subtypes of leukemia. |
| miR-450a-5p, miR-1225a, | -- | Dysregulated | Dysregulated in all subtypes of leukemia |
| miR-128, miR-146a, miR-155, miR-181a, miR-195 | ALL | High | High expression vs. healthy control. |
| Let-7, miR-223 | ALL | Lower | Lower expression in ALL vs. AML |
| miR-29c-5p | ALL | High | Increased expression allows one to difference between B and T ALL. |
| LymphomiR | Target | Roll | Refs. |
|---|---|---|---|
| miR-34 | FOXP3, SIRT1 | Media transition pro-B a pre-B | [70] |
| miR-7 | KLF4, YY1 | Chemoresistance & migration | [61] |
| miR-17-5p | E2F1 | Cell Cycle Regulation | [71] |
| miR-106b | P21/CDK1 | Cell Cycle Regulation | [72] |
| miR-17-92 cluster | HIF-1a | Chemoresistance | [73] |
| miR-155 | TP53INP | Tumor growth | [74] |
| Upregulated in DLBCL by small-RNA-seq [65] | Downregulated in DLBCL by small-RNA-seq [65] | Downregulated in DLBCL and BL [41] | Upregulated in FL [63] | Downregulated in FL [63] | ||
|---|---|---|---|---|---|---|
| miR-124 | miR-425 | miR-150 | miR-193a-5p | miR-1295 | miR-17 * | miR-222 |
| miR-532-5p | miR-141 | miR-189 | miR-193b * | miR-1471 | miR-30a | miR-301b |
| miR-145 | miR-223 | miR-345 | miR-33a | miR-431 * | ||
| miR-197 | miR-768-3p | miR-513b | miR-106a * | |||
| miR-345 | miR-15 | miR-574-3p | miR-141 | |||
| miR-424 | miR-584 | miR-202 | ||||
| miR-128 | miR-663 | miR-205 | ||||
| miR-122 | miR-1287 | miR-570 | ||||
| Target and microRNA | Position |
|---|---|
| Position 85–92 of BCL2L1 3’ UTR hsa-miR-140-5p | 5’ ...CAUUGCCACCAGGAG--AACCACUA... ||| ||||||| 3’ GAUGGUAUCCCAUUUUGGUGAC |
| Position 88–94 of BCL2L1 3’ UTR hsa-miR-142-3p.1 | 5’ ...UGCCACCAGGAGAACCACUACAU... ||||||| 3’ AGGUAUUUCAUCCUUUGUGAUGU |
| Position 764–771 of BCL2L1 3’ UTR hsa-miR-133a-3p.1 | 5’ ...CCAUGACCAUACUGAGGGACCAA... ||||||| 3’ GUCGACCAACUUCCCCUGGUU |
| Position 765–771 of BCL2L1 3’ UTR hsa-miR-133a-3p.2 | 5’ ...CAUGACCAUACUGAGGGACCAAC... ||||||| 3’ GUCGACCAACUUCCCCUGGUUU |
| Position 765–771 of BCL2L1 3’ UTR hsa-miR-133b | 5’ ...CAUGACCAUACUGAGGGACCAAC... ||||||| 3’ AUCGACCAACUUCCCCUGGUUU |
| Position 938–944 of BCL2L1 3’ UTR hsa-miR-7-5p | 5’ ...UAUGGGAGCCCCAGGGUCUUCCC... ||||||| 3’ UGUUGUUUUAGUGAUCAGAAGGU |
| Position 945–952 of BCL2L1 3’ UTR hsa-miR-4458 | 5’ ...GCCCCAGGGUCUUCC----CUACCUCA... |||||| ||||||| 3’ AAGAAGGUGUGGAUGGAGA |
| Position 945–952 of BCL2L1 3’ UTR hsa-miR-4500 | 5’ ...GCCCCAGGGUCUUCCCUACCUCA... ||||||| 3’ UUCUUUGAUGAUGGAGU |
| Position 945–952 of BCL2L1 3’ UTR hsa-let-7b-5p | 5’ ...GCCCCAGGGUCUUCCCUACCUCA... ||||||| 3’ UUGGUGUGUUGGAUGAUGGAGU |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morales-Martínez, M.; Vega, M.I. Roles and Regulation of BCL-xL in Hematological Malignancies. Int. J. Mol. Sci. 2022, 23, 2193. https://doi.org/10.3390/ijms23042193
Morales-Martínez M, Vega MI. Roles and Regulation of BCL-xL in Hematological Malignancies. International Journal of Molecular Sciences. 2022; 23(4):2193. https://doi.org/10.3390/ijms23042193
Chicago/Turabian StyleMorales-Martínez, Mario, and Mario I. Vega. 2022. "Roles and Regulation of BCL-xL in Hematological Malignancies" International Journal of Molecular Sciences 23, no. 4: 2193. https://doi.org/10.3390/ijms23042193
APA StyleMorales-Martínez, M., & Vega, M. I. (2022). Roles and Regulation of BCL-xL in Hematological Malignancies. International Journal of Molecular Sciences, 23(4), 2193. https://doi.org/10.3390/ijms23042193