Magnetic Fields and Cancer: Epidemiology, Cellular Biology, and Theranostics
Abstract
1. Introduction
1.1. Definition and Natural/Anthropogenic Sources of Magnetic Fields
1.2. Public Health Initiatives and Concern
2. Epidemiological Studies Evaluating MF and Cancer Relationships
2.1. Epidemiology of Residential/Domestic Exposure to MF
2.1.1. Brain Tumor
2.1.2. Breast Cancer
2.1.3. Leukemias
2.1.4. Other Cancers
2.2. Epidemiology of Occupational Exposure to MF
2.2.1. Brain Cancer
2.2.2. Breast Cancer
2.2.3. Leukemias
2.2.4. Other Cancers
3. In Vivo and In Vitro Effects of Magnetic Fields on Cancer
3.1. Studies on Human Cells (In Vitro Cellular Studies and Cell-Free Systems)
3.2. Studies on Animals
3.2.1. Studies on Mice
3.2.2. Studies on Rat
3.3. Involvement of Reactive Oxygen Species (ROS) and Reactive Nitrogen Species (RNS)
4. Magnetic Fields and Cancer Theranostics
4.1. Magnetic Fields and Cancer Diagnosis
4.1.1. Brain and Glioma Cancer
4.1.2. Head and Neck Cancer
4.1.3. Thyroid Cancer
4.1.4. Breast Cancer
4.1.5. Lung Cancer
4.1.6. Gastric Cancer
4.1.7. Pancreatic Cancer
4.1.8. Hepatocellular Carcinoma
4.1.9. Gallbladder Carcinoma
4.1.10. Renal Cancer
4.1.11. Bladder Cancer
4.1.12. Ovarian Cancer
4.1.13. Cervical Uterine Cancer
4.1.14. Endometrial Cancer
4.1.15. Prostate Cancer
4.1.16. Testicular Cancer
4.1.17. Colorectal Cancer
4.2. Magnetic Fields and Cancer Treatment
4.2.1. Delivery of Anticancer Agents via Magnetic Carrier Particles
4.2.2. Magnetomechanical Methods for Cancer Therapy
4.2.3. Magnetic Hyperthermia Ad Cancer Treatment
5. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| [F-18]-FDG | 18Fluor Desoxy Glucose |
| ADC | Apparent Diffusion Coefficient |
| AF | Alternating Field |
| AL | Acute Leukemia |
| ALL | Acute Lymphoblastic/lymphocytic Leukemia |
| AM | Amplitude Modulation |
| AMF | Alternating Magnetic Field |
| AMI | Acute Myocardial Infarction |
| AML | Acute Myeloid Leukemia |
| B-ALL | B-Lineage Acute Lymphoblastic Leukemia |
| C-11-MET | 11C–methyl-L-Methionine |
| CAD | Computer-Aided Diagnosis |
| CAT | Catalase |
| CCHD | Chronic Coronary Heart Disease |
| CLL | Chronic Lymphocytic Leukemia |
| CML | Chronic Myeloid Leukemia |
| CT | Computed Tomography |
| CYP-450 | Cytochrome P450 |
| DAB | Digital Audio Broadcasting |
| DCE | Dynamic Contrast-Enhanced |
| DGE | Dynamic Glucose-Enhanced |
| D-MRI | Diffusion Magnetic Resonance Imaging |
| Dox | Doxorubicin |
| DSC-MRI | Dynamic Susceptibility Contrast Magnetic Resonance Imaging |
| DVBT | Digital Video Broadcasting Terrestrial |
| DWI | Diffusion-Weighted Imaging |
| EHS | Electromagnetic Hypersensitivity |
| ELF | Extremely Low Frequency |
| EM | Electromagnetic |
| EMF | Electromagnetic Field |
| EMVI | Extramural Venous Invasion |
| ERK1/2 | Extracellular Signal-Regulated Kinases 1/2 |
| FM | Frequency Modulation |
| FMT | Fluorescence Molecular Tomography |
| GdEOB-DTPA | Gadolinium-Ethoxybenzyl-Diethylenetriamine Pentaacetic Acid |
| GfM | Gadofluorine-M |
| GIS | Geographical Information System |
| GMF | Geomagnetic Field |
| GPX | Glutathione Peroxidase |
| HR MRI | High-Resolution Magnetic Resonance Imaging |
| HVDC | High Voltage Direct Current |
| HVOTL | High-Voltage Overhead Transmission Line |
| IARC | International Agency for Research on Cancer |
| ICNIRP | International Commission on Non-Ionizing Radiation Protection |
| IDH | Isocitrate Dehydrogenase |
| IRPA | International Radiation Protection Association |
| LF | Low-Frequency |
| LF EMF | Low Frequency Electromagnetic Field |
| LL | Lymphatic Leukemia |
| MCE-MRI | Multiphase Contrast-Enhanced Magnetic Resonance Imaging |
| MDA | Malondialdehyde |
| MDCT | Multi Detector Computed Tomography |
| MF | Magnetic Field |
| MHT | Magnetic Hyperthermia Treatment |
| MION | Magnetic Iron Oxide Nanoparticles |
| MNP | Magnetic NanoParticle |
| Mp-MRI | Multi-Parametric Magnetic Resonance Imaging |
| MRI | Magnetic Resonance Imaging |
| Na-23-MRI | Na-23-Magnetic Resonance Imaging |
| NI | Non Ionizing |
| NI EMF | Non-Ionizing Electromagnetic Field |
| NMMA | Nano-Magnetomechanical Activation |
| NMR | Nuclear Magnetic Resonance |
| NPP | Nuclear Power Plants |
| OR | Odd Ratio |
| PET | Positron Emission Tomography |
| PFF | Power-Frequency Field |
| PKC | Protein Kinases C |
| RF | Radio Frequency |
| RFH | Radiofrequency Heat |
| RFID | Radio Frequency Range |
| RNS | Reactive Nitrogen Species |
| ROS | Reactive Oxygen Species |
| RP | Radical Pair |
| RPM | Radical Pair Mechanism |
| S | Singlet State |
| SAR | Specific Absorption Rate |
| SF | Static Field |
| SMF | Static Magnetic Field |
| SOD | Superoxide Dismutase |
| SPIO | Superparamagnetic Iron Oxide |
| SPION | Superparamagnetic Iron Oxide Nanoparticle |
| T | Triplet State |
| TAR | Tumor Area Ratio |
| TSMI | Tumor-Selective Molecular Imaging |
| TVR | Tumor Volume Ratio |
| ULF | Ultra-Low Frequency |
| USPIO | Ultrasmall Superparamagnetic Particles of Iron Oxide |
| WB-MRI | Whole Body Magnetic Resonance Imaging |
| WHO | World Health Organization |
| WLAN | Wireless Local Area Networks |
References
- Monadizadeh, S.; Kibert, C.J.; Li, J.X.; Woo, J.; Asutosh, A.; Roostaie, S.; Kouhirostami, M. A review of protocols and guidelines addressing the exposure of occupants to electromagnetic field radiation (EMFr) in buildings. J. Green Build. 2021, 16, 55–81. [Google Scholar] [CrossRef]
- Rathebe, P.C.; Modisane, D.S.; Rampedi, M.B.; Biddesay-Manilal, S.; Mbonane, T.P. A review on residential exposure to electromagnetic fields from overhead power lines: Electrification as a health burden in rural communities. In Proceedings of the 2019 Open Innovations (OI), Cape Town, South Africa, 2–4 October 2019; pp. 219–221. [Google Scholar]
- Gupta, S.; Sharma, R.S.; Singh, R. Non-ionizing radiation as possible carcinogen. Int. J. Environ. Health Res. 2020, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Mazzanti, G. Evaluation of continuous exposure to magnetic field from ac overhead transmission lines via historical load databases: Common procedures and innovative heuristic formulas. IEEE Trans. Power Deliv. 2010, 25, 238–247. [Google Scholar] [CrossRef]
- Landler, L.; Keays, D.A. Cryptochrome: The magnetosensor with a sinister side? PLoS Biol. 2018, 16, e3000018. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, J.I. Speculations on the influence of electromagnetism on genomic and associated structures. J. Int. Med. Res. 1996, 24, 1–11. [Google Scholar] [CrossRef]
- Repacholi, M. Concern that “EMF” magnetic fields from power lines cause cancer. Sci. Total Environ. 2012, 426, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Saliev, T.; Begimbetova, D.; Masoud, A.R.; Matkarimov, B. Biological effects of non-ionizing electromagnetic fields: Two sides of a coin. Prog. Biophys. Mol. Biol. 2019, 141, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Lednev, V.V. Possible mechanism for the influence of weak magnetic fields on biological systems. Bioelectromagnetics 1991, 12, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Occhipinti, A.; De Santis, A.; Maffei, M.E. Magnetoreception: An unavoidable step for plant evolution? Trends Plant Sci 2014, 19, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.E. Magnetic field effects on plant growth, development, and evolution. Front. Plant Sci. 2014, 5, 445. [Google Scholar] [CrossRef]
- Swanson, J.; Kheifets, L. Could the geomagnetic field be an effect modifier for studies of power-frequency magnetic fields and childhood leukaemia? J. Radiol. Prot. 2012, 32, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Amoon, A.T.; Swanson, J.; Vergara, X.; Kheifets, L. Relationship between distance to overhead power lines and calculated fields in two studies. J. Radiol. Prot. 2020, 40, 431–443. [Google Scholar] [CrossRef] [PubMed]
- Wyszkowska, J.; Szczygiel, M.; Trawinski, T. Static magnetic field and extremely low-frequency magnetic field in hybrid and electric vehicles. Prz. Elektrotech. 2020, 96, 60–62. [Google Scholar] [CrossRef]
- Vaitl, D.; Propson, N.; Stark, R.; Schienle, A. Natural very-low-frequency sferics and headache. Int. J. Biometeorol. 2001, 45, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Feychting, M.; Ahlborn, A.; Kheifets, L. EMF and health. Annu. Rev. Public Health 2005, 26, 165–189. [Google Scholar] [CrossRef] [PubMed]
- Repacholi, M.H. Who’s international EMF project. Radiat. Prot. Dosim. 1999, 83, 1–4. [Google Scholar] [CrossRef]
- Bowman, J.D.; Ray, T.K.; Park, R.M. Possible health benefits from reducing occupational magnetic fields. Am. J. Ind. Med. 2013, 56, 791–805. [Google Scholar] [CrossRef] [PubMed]
- Belyaev, I.; Dean, A.; Eger, H.; Hubmann, G.; Jandrisovits, R.; Kern, M.; Kundi, M.; Moshammer, H.; Lercher, P.; Muller, K.; et al. Europaem EMF guideline 2016 for the prevention, diagnosis and treatment of EMF-related health problems and illnesses. Rev. Environ. Health 2016, 31, 363–397. [Google Scholar] [CrossRef]
- Repacholi, M.H. A history of the international commission on non-ionizing radiation protection. Health Phys. 2017, 113, 282–300. [Google Scholar] [CrossRef] [PubMed]
- Soto Sumuano, J.L.; Abundis Gutierrez, E.; Tlacuilo-Parra, J.A.; Garibaldi Covarrubias, R.F.; Romo Rubio, H. Electromagnetic radiation, childhood leukemia and regulation. Rev. Int. Contam. Ambient. 2020, 36, 229–240. [Google Scholar]
- Knave, B. Electromagnetic fields and health outcomes. Ann. Acad. Med. Singap. 2001, 30, 489–493. [Google Scholar] [PubMed]
- Kelfkens, G.; Pruppers, M. Magnetic fields and childhood leukemia; science and policy in the Netherlands. In Proceedings of the Conference of the European Medical and Biological Engineering Conference (EMBEC) and the Nordic-Baltic Conference on Biomedical Engineering and Medical Physics (NBC), Tampere, Finland, 11–15 June 2017; Eskola, H., Vaisanen, O., Viik, J., Hyttinen, J., Eds.; Springer: Singapore, 2018; Volume 65, pp. 498–501. [Google Scholar]
- Teepen, J.C.; van Dijck, J. Impact of high electromagnetic field levels on childhood leukemia incidence. Int. J. Cancer 2012, 131, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Calvente, I.; Fernandez, M.F.; Villalba, J.; Olea, N.; Nunez, M.I. Exposure to electromagnetic fields (non-ionizing radiation) and its relationship with childhood leukemia: A systematic review. Sci. Total Environ. 2010, 408, 3062–3069. [Google Scholar] [CrossRef] [PubMed]
- Calvente, I.; Davila-Arias, C.; Ocon-Hernandez, O.; Perez-Lobato, R.; Ramos, R.; Artacho-Cordon, F.; Olea, N.; Nunez, M.I.; Fernandez, M.F. Characterization of indoor extremely low frequency and low frequency electromagnetic fields in the inma-granada cohort. PLoS ONE 2014, 9, e106666. [Google Scholar] [CrossRef]
- Miah, T.; Kamat, D. Current understanding of the health effects of electromagnetic fields. Pediatric Ann. 2017, 46, E172–E174. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bates, M.N. Extremely low-frequency electromagnetic-fields and cancer—The epidemiologic evidence. Environ. Health Perspect. 1991, 95, 147–156. [Google Scholar] [CrossRef]
- Wertheimer, N.; Leeper, E. Electrical wiring configurations and childhood cancer. Am. J. Epidemiol. 1979, 109, 273–284. [Google Scholar] [CrossRef] [PubMed]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Non-ionizing radiation, part 2: Radiofrequency electromagnetic fields. IARC Monogr. Eval. Carcinog. Risks Hum. 2013, 102 Pt 2, 1–460. [Google Scholar]
- Deutsch, S.; Wilkening, G.M. Electromagnetic field cancer scares. Health Phys. 1997, 73, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Karimi, A.; Moghaddam, F.G.; Valipour, M. Insights in the biology of extremely low-frequency magnetic fields exposure on human health. Mol. Biol. Rep. 2020, 47, 5621–5633. [Google Scholar] [CrossRef] [PubMed]
- Moulder, J.E.; Foster, K.R. Biological effects of power-frequency fields as they relate to carcinogenesis. Proc. Soc. Exp. Biol. Med. 1995, 209, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Dieudonne, M. Electromagnetic hypersensitivity: A critical review of explanatory hypotheses. Environ. Health 2020, 19, 48. [Google Scholar] [CrossRef] [PubMed]
- Elliott, P.; Shaddick, G.; Douglass, M.; de Hoogh, K.; Briggs, D.J.; Toledano, M.B. Adult cancers near high-voltage overhead power lines. Epidemiology 2013, 24, 184–190. [Google Scholar] [CrossRef]
- Lambrozo, J. Electric and magnetic fields with a frequency of 50–60 hz: Assessment of 20 years of research. Indoor Built Environ. 2001, 10, 299–305. [Google Scholar]
- Souques, M.; Lambrozo, J. 50–60 hz magnetic fields and health: What’s new? Radioprotection 2015, 50, 95–99. [Google Scholar] [CrossRef]
- Sheppard, A.R.; Kavet, R.; Renew, D.C. Exposure guidelines for low-frequency electric and magnetic fields: Report from the brussels workshop. Health Phys. 2002, 83, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Maes, A.; Verschaeve, L. Genetic damage in humans exposed to extremely low-frequency electromagnetic fields. Arch. Toxicol. 2016, 90, 2337–2348. [Google Scholar] [CrossRef] [PubMed]
- Lacy-Hulbert, A.; Metcalfe, J.C.; Hesketh, R. Biological responses to electromagnetic fields. FASEB J. 1998, 12, 395–420. [Google Scholar] [CrossRef] [PubMed]
- Kavet, R. EMF and current cancer concepts. Bioelectromagnetics 1996, 17, 339–357. [Google Scholar] [CrossRef]
- Rathebe, P.; Weyers, C.; Raphela, F. Exposure levels of elf magnetic fields in the residential areas of mangaung metropolitan municipality. Environ. Monit. Assess. 2018, 190, 544. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Camba, E.A.; Segura-Garcia, J.; Gomez-Perretta, C. Exposure to 50 hz magnetic fields in homes and areas surrounding urban transformer stations in silla (spain): Environmental impact assessment. Sustainability 2018, 10, 2641. [Google Scholar] [CrossRef]
- Sadeghi, T.; Ahmadi, A.; Javadian, M.; Gholamian, S.A.; Delavar, M.A.; Esmailzadeh, S.; Ahmadi, B.; Hadighi, M.S.H. Preterm birth among women living within 600 meters of high voltage overhead power lines: A case-control study. Rom. J. Intern. Med. 2017, 55, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Ahlbom, A.; Cardis, E.; Green, A.; Linet, M.; Savitz, D.; Swerdlow, A.; Epidemiology, I.S.C. Review of the epidemiologic literature on EMF and health. Environ. Health Perspect. 2001, 109, 911–933. [Google Scholar] [PubMed]
- Alonso, A.; Bahillo, A.; de la Rosa, R.; Carrera, A.; Duran, R.J.; Fernandez, P. Measurement procedure to assess exposure to extremely low-frequency fields: A primary school case study. Radiat. Prot. Dosim. 2012, 151, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Sung, F.C.; Chen, F.L.; Lee, P.C.; Silva, M.; Mezei, G. Extremely-low-frequency magnetic field exposure of children at schools near high voltage transmission lines. Sci. Total Environ. 2007, 376, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Tognola, G.; Chiaramello, E.; Bonato, M.; Magne, I.; Souques, M.; Fiocchi, S.; Parazzini, M.; Ravazzani, P. Cluster analysis of residential personal exposure to elf magnetic field in children: Effect of environmental variables. Int. J. Environ. Res. Public Health 2019, 16, 4363. [Google Scholar] [CrossRef] [PubMed]
- Huss, A.; Goris, K.; Vermeulen, R.; Kromhout, H. Does apartment’s distance to an in-built transformer room predict magnetic field exposure levels? J. Expo. Sci. Environ. Epidemiol. 2013, 23, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Roosli, M.; Jenni, D.; Kheifets, L.; Mezei, G. Extremely low frequency magnetic field measurements in buildings with transformer stations in switzerland. Sci. Total Environ. 2011, 409, 3364–3369. [Google Scholar] [CrossRef] [PubMed]
- Ilonen, K.; Markkanen, A.; Mezei, G.; Juutilainen, J. Indoor transformer stations as predictors of residential elf magnetic field exposure. Bioelectromagnetics 2008, 29, 213–218. [Google Scholar] [CrossRef]
- Vergara, X.P.; Kavet, R.; Crespi, C.M.; Hooper, C.; Silva, J.M.; Kheifets, L. Estimating magnetic fields of homes near transmission lines in the california power line study. Environ. Res. 2015, 140, 514–523. [Google Scholar] [CrossRef]
- Aldrich, T.E.; Andrews, K.W.; Liboff, A.R. Brain cancer risk and electromagnetic fields (EMFs): Assessing the geomagnetic component. Arch. Environ. Health 2001, 56, 314–319. [Google Scholar] [CrossRef]
- Nikkila, A.; Kendall, G.; Raitanen, J.; Spycher, B.; Lohi, O.; Auvinen, A. Effects of incomplete residential histories on studies of environmental exposure with application to childhood leukaemia and background radiation. Environ. Res. 2018, 166, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S.; Kheifets, L.; Zafanella, L.E.; Kalton, G.W. Leukemia attributable to residential magnetic fields: Results from analyses allowing for study biases. Risk Anal. 2006, 26, 471–482. [Google Scholar] [CrossRef]
- Miller, K.D.; Ostrom, Q.T.; Kruchko, C.; Patil, N.; Tihan, T.; Cioffi, G.; Fuchs, H.E.; Waite, K.A.; Jemal, A.; Siegel, R.L.; et al. Brain and other central nervous system tumor statistics, 2021. CA Cancer J. Clin. 2021, 71, 381–406. [Google Scholar] [CrossRef]
- Farmanfarma, K.K.; Mohammadian, M.; Shahabinia, Z.; Hassanipour, S.; Salehiniya, H. Brain cancer in the world: An epidemiological review. World Cancer Res. J. 2019, 6, 5. [Google Scholar]
- Pourreza, R.; Zhuge, Y.; Ning, H.; Miller, R. Brain tumor segmentation in MRI scans using deeply-supervised neural networks. In Proceedings of the 3rd International Workshop on Brain-Lesion (BrainLes) Held Jointly at the Conference on Medical Image Computing for Computer Assisted Intervention (MICCAI), Quebec City, QC, Canada, 14 September 2017; Springer International Publishing Ag: Quebec City, QC, Canada, 2018; pp. 320–331. [Google Scholar]
- Kheifets, L.I. Electric and magnetic field exposure and brain cancer: A review. Bioelectromagnetics 2001, 22, S120–S131. [Google Scholar] [CrossRef]
- Schoenfeld, E.R.; Henderson, K.; O’Leary, E.; Grimson, R.; Kaune, W.; Leske, M.C. Magnetic field exposure assessment: A comparison of various methods. Bioelectromagnetics 1999, 20, 487–496. [Google Scholar] [CrossRef]
- Eskelinen, T.; Keinänen, J.; Salonen, H.; Juutilainen, J. Use of spot measurements for assessing residential elf magnetic field exposure: A validity study. Bioelectromagnetics 2002, 23, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, H.; Mohseni, S.; Akmal, A.A.S. Electromagnetic fields near transmission lines—Problems and solutions. Iran. J. Environ. Health Sci. Eng. 2010, 7, 181–188. [Google Scholar]
- Kheifets, L.; Crespi, C.M.; Hooper, C.; Oksuzyan, S.; Cockburn, M.; Ly, T.; Mezei, G. Epidemiologic study of residential proximity to transmission lines and childhood cancer in california: Description of design, epidemiologic methods and study population. J. Expo. Sci. Environ. Epidemiol. 2015, 25, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Blaasaas, K.G.; Tynes, T. Comparison of three different ways of measuring distances between residences and high voltage power lines. Bioelectromagnetics 2002, 23, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Carles, C.; Esquirol, Y.; Turuban, M.; Piel, C.; Migault, L.; Pouchieu, C.; Bouvier, G.; Fabbro-Peray, P.; Lebailly, P.; Baldi, I. Residential proximity to power lines and risk of brain tumor in the general population. Environ. Res. 2020, 185, 109473. [Google Scholar] [CrossRef]
- Marcilio, I.; Gouveia, N.; Pereira Filho, M.L.; Kheifets, L. Adult mortality from leukemia, brain cancer, amyotrophic lateral sclerosis and magnetic fields from power lines: A case-control study in Brazil. Rev. Bras. Epidemiol. 2011, 14, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Baldi, I.; Coureau, G.; Jaffré, A.; Gruber, A.; Ducamp, S.; Provost, D.; Lebailly, P.; Vital, A.; Loiseau, H.; Salamon, R. Occupational and residential exposure to electromagnetic fields and risk of brain tumors in adults: A case–control study in Gironde, France. Int. J. Cancer 2011, 129, 1477–1484. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Lin, R.S.; Sung, F.C. Elevated residential exposure to power frequency magnetic field associated with greater average age at diagnosis for patients with brain tumors. Bioelectromagnetics 2003, 24, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, D.O. Extremely low frequency electromagnetic fields and cancer: How source of funding affects results. Environ. Res 2019, 178, 108688. [Google Scholar] [CrossRef] [PubMed]
- Mezei, G.; Gadallah, M.; Kheifets, L. Residential magnetic field exposure and childhood brain cancer—A meta-analysis. Epidemiology 2008, 19, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Kheifets, L.; Ahlbom, A.; Crespi, C.M.; Feychting, M.; Johansen, C.; Monroe, J.; Murphy, M.F.G.; Oksuzyan, S.; Preston-Martin, S.; Roman, E.; et al. A pooled analysis of extremely low-frequency magnetic fields and childhood brain tumors. Am. J. Epidemiol. 2010, 172, 752–761. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Nitta, H.; Kubo, O.; Yamamoto, S.; Yamaguchi, N.; Akiba, S.; Honda, Y.; Hagihara, J.; Isaka, K.; Ojima, T.; et al. Power-frequency magnetic fields and childhood brain tumors: A case-control study in Japan. J. Epidemiol. 2010, 20, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.E.; Swanson, J.; Vincent, T.J.; Draper, G.J. Childhood cancer and magnetic fields from high-voltage power lines in england and wales: A case-control study. Br. J. Cancer 2010, 103, 1122–1127. [Google Scholar] [CrossRef]
- Seomun, G.; Lee, J.; Park, J. Exposure to extremely low-frequency magnetic fields and childhood cancer: A systematic review and meta-analysis. PLoS ONE 2021, 16, e0251628. [Google Scholar] [CrossRef]
- Ozen, S. Low-frequency transient electric and magnetic fields coupling to child body. Radiat. Prot. Dosim. 2008, 128, 62–67. [Google Scholar] [CrossRef]
- Miller, K.D.; Nogueira, L.; Mariotto, A.B.; Rowland, J.H.; Yabroff, K.R.; Alfano, C.M.; Jemal, A.; Kramer, J.L.; Siegel, R.L. Cancer treatment and survivorship statistics, 2019. CA Cancer J. Clin. 2019, 69, 363–385. [Google Scholar] [CrossRef]
- Davis, S.; Mirick, D.K.; Chen, C.; Stanczyk, F.Z. Effects of 60-hz magnetic field exposure on nocturnal 6-sulfatoxymelatonin, estrogens, luteinizing hormone, and follicle, stimulating hormone in healthy reproductive-age women: Results of a crossover trial. Ann. Epidemiol. 2006, 16, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Graham, C.; Cook, M.R.; Gerkovich, M.M.; Sastre, A. Melatonin and 6-ohms in high-intensity magnetic fields. J. Pineal Res. 2001, 31, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Youngstedt, S.D.; Kripke, D.F.; Elliott, J.A.; Assmus, J.D. No association of 6-sulfatoxymelatonin with in-bed 60-hz magnetic field exposure or illumination level among older adults. Environ. Res. 2002, 89, 201–209. [Google Scholar] [CrossRef]
- Levallois, P.; Dumont, M.; Touitou, Y.; Gingras, S.; Masse, B.; Gauvin, D.; Kroger, E.; Bourdages, M.; Douville, P. Effects of electric and magnetic fields from high-power lines on female urinary excretion of 6-sulfatoxymelatonin. Am. J. Epidemiol. 2001, 154, 601–609. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Feychting, M. Invited commentary: Extremely low-frequency magnetic fields and breast cancerunow it is enough! Am. J. Epidemiol. 2013, 178, 1046–1050. [Google Scholar] [CrossRef] [PubMed]
- Touitou, Y.; Lambrozo, J.; Camus, F.O.; Charbuy, H. Magnetic fields and the melatonin hypothesis: A study of workers chronically exposed to 50-hz magnetic fields. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 284, R1529–R1535. [Google Scholar] [CrossRef] [PubMed]
- Touitou, Y.; Selmaoui, B.; Lambrozo, J.; Auzeby, A. Assessment of the effects of magnetic fields (50 hz) on melatonin secretion in humans and rats. A circadian study. Bull. Acad. Natl. Med. 2002, 186, 1625–1639. [Google Scholar] [PubMed]
- Davis, S.; Mirick, D.K. Residential magnetic fields, medication use, and the risk of breast cancer. Epidemiology 2007, 18, 266–269. [Google Scholar] [CrossRef] [PubMed]
- de Bruyn, L.; de Jager, L.; Kuyl, J.M. The influence of long-term exposure of mice to randomly varied power frequency magnetic fields on their nocturnal melatonin secretion patterns. Environ. Res. 2001, 85, 115–121. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, E.S.; Schoenfeld, E.R.; Henderson, K.; Grimson, R.; Kabat, G.C.; Kaune, W.T.; Gammon, M.D.; Leske, C.; Grp, E. Wire coding in the EMF and breast cancer on long island study: Relationship to magnetic fields. J. Expo. Anal. Environ. Epidemiol. 2003, 13, 283–293. [Google Scholar] [CrossRef][Green Version]
- London, S.J.; Pogoda, J.M.; Hwang, K.L.; Langholz, B.; Monroe, K.R.; Kolonel, L.N.; Kaune, W.T.; Peters, J.M.; Henderson, B.E. Residential magnetic field exposure and breast cancer risk: A nested case-control study from a multiethnic cohort in los angeles county, california. Am. J. Epidemiol. 2003, 158, 969–980. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Haugsdal, B.; Tynes, T.; Rotnes, J.S.; Griffiths, D. A single nocturnal exposure to 2–7 millitesla static magnetic fields does not inhibit the excretion of 6-sulfatoxymelatonin in healthy young men. Bioelectromagnetics 2001, 22, 1–6. [Google Scholar] [CrossRef]
- Fedrowitz, M.; Westermann, J.; Loscher, W. Magnetic field exposure increases cell proliferation but does not affect melatonin levels in the mammary gland of female sprague dawley rats. Cancer Res. 2002, 62, 1356–1363. [Google Scholar] [PubMed]
- Vanderstraeten, J.; Verschaeve, L.; Burda, H.; Bouland, C.; Brouwer, C. Health effects of extremely low-frequency magnetic fields: Reconsidering the melatonin hypothesis in the light of current data on magnetoreception. J. Appl. Toxicol. 2012, 32, 952–958. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Lai, J.S.; Ruan, G.R.; Chen, C.; Wang, D.W. Meta-analysis of extremely low frequency electromagnetic fields and cancer risk: A pooled analysis of epidemiologic studies. Environ. Int. 2016, 88, 36–43. [Google Scholar] [CrossRef]
- Zhu, K.M.; Hunter, S.; Payne-Wilks, K.; Roland, C.L.; Forbes, D.S. Use of electric bedding devices and risk of breast cancer in african-american women. Am. J. Epidemiol. 2003, 158, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Laden, F.; Hunter, D.J. Environmental risk factors and female breast cancer. Annu. Rev. Public Health 1998, 19, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Amoon, A.T.; Swanson, J.; Magnani, C.; Johansen, C.; Kheifets, L. Pooled analysis of recent studies of magnetic fields and childhood leukemia. Environ. Res. 2022, 204, 7. [Google Scholar] [CrossRef] [PubMed]
- Elwood, J.M. Childhood leukemia and residential magnetic fields: Are pooled analyses more valid than the original studies? Bioelectromagnetics 2006, 27, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Schuz, J. Exposure to extremely low-frequency magnetic fields and the risk of childhood cancer: Update of the epidemiological evidence. Prog. Biophys. Mol. Biol. 2011, 107, 339–342. [Google Scholar] [CrossRef]
- Ghahremani, S.; Shiroudbakhshi, K.; Kordasiabi, A.H.S.; FiroozBakht, M.; Hosseinzadegan, M.; Ashrafinia, F.; Rahafard, S. Exposure to magnetic fields and childhood leukemia: An overview of meta-analysis. Int. J. Pediatr.-Mashhad 2020, 8, 11361–11365. [Google Scholar]
- Tafrishi, R.; Seyfari, B.; Rahimi, R.; Chaichi, Z.; Tarazjani, A.D.; Marvi, N.; Maazallahi, M.; Dolatian, Z.; Ashrafinia, F. Modifiable and non-modifiable factors affecting the risk of childhood leukemia: An overview of meta-analysis. Int. J. Pediatr. 2021, 9, 13243–13248. [Google Scholar]
- Zhao, L.Y.; Liu, X.D.; Wang, C.P.; Yan, K.K.; Lin, X.J.; Li, S.; Bao, H.H.; Liu, X. Magnetic fields exposure and childhood leukemia risk: A meta-analysis based on 11,699 cases and 13,194 controls. Leuk. Res. 2014, 38, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Amoon, A.T.; Crespi, C.M.; Ahlbom, A.; Bhatnagar, M.; Bray, I.; Bunch, K.J.; Clavel, J.; Feychting, M.; Hemon, D.; Johansen, C.; et al. Proximity to overhead power lines and childhood leukaemia: An international pooled analysis. Br. J. Cancer 2018, 119, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, C.; Brauner, E.V.; Rod, N.H.; Albieri, V.; Andersen, C.E.; Ulbak, K.; Hertel, O.; Johansen, C.; Schuz, J.; Raaschou-Nielsen, O. Distance to high-voltage power lines and risk of childhood leukemia—An analysis of confounding by and interaction with other potential risk factors. PLoS ONE 2014, 9, e107096. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, C.; Johansen, C.; Schuz, J.; Olsen, J.H.; Raaschou-Nielsen, O. Residential exposure to extremely low-frequency magnetic fields and risk of childhood leukaemia, cns tumour and lymphoma in denmark. Br. J. Cancer 2015, 113, 1370–1374. [Google Scholar] [CrossRef]
- Nunez-Enriquez, J.C.; Correa-Correa, V.; Flores-Lujano, J.; Perez-Saldivar, M.L.; Jimenez-Hernandez, E.; Martin-Trejo, J.A.; Espinoza-Hernandez, L.E.; Medina-Sanson, A.; Cardenas-Cardos, R.; Flores-Villegas, L.V.; et al. Extremely low-frequency magnetic fields and the risk of childhood b-lineage acute lymphoblastic leukemia in a city with high incidence of leukemia and elevated exposure to elf magnetic fields. Bioelectromagnetics 2020, 41, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Schuz, J.; Grigat, J.P.; Brinkmann, K.; Michaelis, J. Residential magnetic fields as a risk factor for childhood acute leukaemia: Results from a german population-based case-control study. Int. J. Cancer 2001, 91, 728–735. [Google Scholar] [CrossRef]
- Kheifets, L.; Crespi, C.M.; Hooper, C.; Cockburn, M.; Amoon, A.T.; Vergara, X.P. Residential magnetic fields exposure and childhood leukemia: A population-based case-control study in california. Cancer Causes Control 2017, 28, 1117–1123. [Google Scholar] [CrossRef]
- Crespi, C.M.; Swanson, J.; Vergara, X.P.; Kheifets, L. Childhood leukemia risk in the california power line study: Magnetic fields versus distance from power lines. Environ. Res. 2019, 171, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Crespi, C.M.; Vergara, X.P.; Hooper, C.; Oksuzyan, S.; Wu, S.; Cockburn, M.; Kheifets, L. Childhood leukaemia and distance from power lines in california: A population-based case-control study. Br. J. Cancer 2016, 115, 122–128. [Google Scholar] [CrossRef]
- Sermage-Faure, C.; Demoury, C.; Rudant, J.; Goujon-Bellec, S.; Guyot-Goubin, A.; Deschamps, F.; Hemon, D.; Clavel, J. Childhood leukaemia close to high-voltage power lines—The geocap study, 2002–2007. Br. J. Cancer 2013, 108, 1899–1906. [Google Scholar] [CrossRef]
- Tabrizi, M.M.; Bidgoli, S.-A. Increased risk of childhood acute lymphoblastic leukemia (all) by prenatal and postnatal exposure to high voltage power lines: A case control study in isfahan, iran. Asian Pac. J. Cancer Prev. 2015, 16, 2347–2350. [Google Scholar] [CrossRef] [PubMed]
- Schuz, J.; Dasenbrock, C.; Ravazzani, P.; Roosli, M.; Schar, P.; Bounds, P.L.; Erdmann, F.; Borkhardt, A.; Cobaleda, C.; Fedrowitz, M.; et al. Extremely low-frequency magnetic fields and risk of childhood leukemia: A risk assessment by the arimmora consortium. Bioelectromagnetics 2016, 37, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Sermage-Faure, C.; Laurier, D.; Goujon-Bellec, S.; Chartier, M.; Guyot-Goubin, A.; Rudant, J.; Hemon, D.; Clavel, J. Childhood leukemia around french nuclear power plantsuthe geocap study, 2002–2007. Int. J. Cancer 2012, 131, E769–E780. [Google Scholar] [CrossRef] [PubMed]
- Ziegelberger, G.; Dehos, A.; Grosche, B.; Hornhardt, S.; Jung, T.; Weiss, W. Childhood leukemia—Risk factors and the need for an interdisciplinary research agenda. Prog. Biophys. Mol. Biol. 2011, 107, 312–314. [Google Scholar] [CrossRef]
- Wünsch, V.; Pelissari, D.M.; Barbieri, F.E.; Sant’Anna, L.; de Oliveira, C.T.; de Mata, J.F.; Tone, L.G.; Lee, M.L.D.; de Andrea, M.L.M.; Bruniera, P.; et al. Exposure to magnetic fields and childhood acute lymphocytic leukemia in Sao Paulo, Brazil. Cancer Epidemiol. 2011, 35, 534–539. [Google Scholar] [CrossRef]
- Jirik, V.; Pekarek, L.; Janout, V.; Tomaskova, H. Association between childhood leukaemia and exposure to power-frequency magnetic fields in Middle Europe. Biomed. Environ. Sci. 2012, 25, 597–601. [Google Scholar] [PubMed]
- Hakim, A.S.B.; Abd Rahman, N.B.; Mokhtar, M.Z.; Bin Said, I.; Hussain, H. ELF-EMF correlation study on distance from overhead transmission lines and acute leukemia among children in Klang Valley, Malaysia. In Proceedings of the 2014 IEEE Conference on Biomedical Engineering and Sciences (IECBES), Kuala Lumpur, Malaysia, 8–10 December 2014; pp. 710–714. [Google Scholar]
- Karipidis, K.K. Survey of residential power-frequency magnetic fields in Melbourne, Australia. Radiat. Prot. Dosim. 2015, 163, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Jirik, V.; Pekarek, L.; Janout, V. Assessment of population exposure to extremely low frequency magnetic fields and its possible childhood health risk in the Czech Republic. Indoor Built Environ. 2011, 20, 362–368. [Google Scholar] [CrossRef]
- Kokate, P.A.; Mishra, A.K.; Lokhande, S.K.; Bodhe, G.L. Extremely low frequency electromagnetic field (ELF-EMF) and childhood leukemia (cl) near transmission lines: A review. Adv. Electromagn. 2016, 5, 30–40. [Google Scholar] [CrossRef][Green Version]
- Zaki, A.M.; Abd Rahim, M.A.; Zaidun, Z.; Ramdzan, A.R.; Isa, Z.M. Exposure to non-ionizing radiation and childhood cancer: A meta-analysis. Middle East J. Cancer 2020, 11, 1–11. [Google Scholar]
- Swanson, J.; Bunch, K.J. Reanalysis of risks of childhood leukaemia with distance from overhead power lines in the UK. J. Radiol. Prot. 2018, 38, N30–N35. [Google Scholar] [CrossRef] [PubMed]
- Liorni, I.; Parazzini, M.; Struchen, B.; Fiocchi, S.; Roosli, M.; Ravazzani, P. Children’s personal exposure measurements to extremely low frequency magnetic fields in Italy. Int. J. Environ. Res. Public Health 2016, 13, 549. [Google Scholar] [CrossRef]
- Amoon, A.T.; Arah, O.A.; Kheifets, L. The sensitivity of reported effects of EMF on childhood leukemia to uncontrolled confounding by residential mobility: A hybrid simulation study and an empirical analysis using caps data. Cancer Causes Control 2019, 30, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Amoon, A.T.; Oksuzyan, S.; Crespi, C.M.; Arah, O.A.; Cockburn, M.; Vergara, X.; Kheifets, L. Residential mobility and childhood leukemia. Environ. Res. 2018, 164, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Bunch, K.J.; Swanson, J.; Vincent, T.J.; Murphy, M.F.G. Magnetic fields and childhood cancer: An epidemiological investigation of the effects of high-voltage underground cables. J. Radiol. Prot. 2015, 35, 695–705. [Google Scholar] [CrossRef] [PubMed]
- Auger, N.; Bilodeau-Bertrand, M.; Marcoux, S.; Kosatsky, T. Residential exposure to electromagnetic fields during pregnancy and risk of child cancer: A longitudinal cohort study. Environ. Res. 2019, 176, 108524. [Google Scholar] [CrossRef] [PubMed]
- Soderberg, K.C.; Naumburg, E.; Anger, G.; Cnattingius, S.; Ekbom, A.; Feychting, M. Childhood leukemia and magnetic fields in infant incubators. Epidemiology 2002, 13, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Slusky, D.A.; Does, M.; Metayer, C.; Mezei, G.; Selvin, S.; Buffler, P.A. Potential role of selection bias in the association between childhood leukemia and residential magnetic fields exposure: A population-based assessment. Cancer Epidemiol. 2014, 38, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Leitgeb, N. Synoptic analysis clarifies childhood leukemia risk from elf magnetic field exposure. J. Electromagn. Anal. Appl. 2015, 7, 10. [Google Scholar] [CrossRef][Green Version]
- Schuz, J.; Erdmann, F. Environmental exposure and risk of childhood leukemia: An overview. Arch. Med. Res. 2016, 47, 607–614. [Google Scholar] [CrossRef]
- Salvan, A.; Ranucci, A.; Lagorio, S.; Magnani, C.; Grp, S.R. Childhood leukemia and 50 hz magnetic fields: Findings from the italian setil case-control study. Int. J. Environ. Res. Public Health 2015, 12, 2184–2204. [Google Scholar] [CrossRef]
- Omura, Y.; Losco, M. Electromagnetic-fields in the home-environment (color tv, computer monitor, microwave-oven, cellular phone, etc) as potential contributing factors for the induction of oncogen c-fos ab1, oncogen c-fos ab2, integrin alpha-5-beta-1 and development of cancer, as well as effects of microwave on amino-acid-composition of food and living human brain. Acupunct. Electro-Ther. Res. 1993, 18, 33–73. [Google Scholar]
- Verreault, R.; Weiss, N.S.; Hollenbach, K.A.; Strader, C.H.; Daling, J.R. Use of electric blankets and risk of testicular cancer. Am. J. Epidemiol. 1990, 131, 759–762. [Google Scholar] [CrossRef]
- Zhu, K.; Weiss, N.S.; Stanford, J.L.; Daling, J.R.; Stergachis, A.; McKnight, B.; Brawer, M.K.; Levine, R.S. Prostate cancer in relation to the use of electric blanket or heated water bed. Epidemiology 1999, 10, 83–85. [Google Scholar] [CrossRef]
- McElroy, J.A.; Newcomb, P.A.; Trentham-Dietz, A.; Hampton, J.M.; Kanarek, M.S.; Remington, P.L. Endometrial cancer incidence in relation to electric blanket use. Am. J. Epidemiol. 2002, 156, 262–267. [Google Scholar] [CrossRef][Green Version]
- Gudina, M.V.; Borodin, A.S.; Tuzhilkin, D.A.; Pikalova, L.V. Malignant neoplasms on territories with different levels of magnetic fields of industrial frequency. In Proceedings of the 24th International Symposium on Atmospheric and Ocean Optics: Atmospheric Physics, Tomsk, Russia, 2–5 July 2018; Matvienko, G.G., Romanovskii, O.A., Eds.; SPIE—International Society for Optics and Photonics: Bellingham, WA, USA, 2018; Volume 10833. [Google Scholar]
- Hallberg, O.; Johansson, O. Melanoma incidence and frequency modulation (FM) broadcasting. Arch. Environ. Health 2002, 57, 32–40. [Google Scholar] [CrossRef]
- Mahram, M.; Ghazavi, M. The effect of extremely low frequency electromagnetic fields on pregnancy and fetal growth, and development. Arch. Iran. Med. 2013, 16, 221–224. [Google Scholar]
- Deadman, J.E.; Infante-Rivard, C. Individual estimation of exposures to extremely low frequency magnetic fields in jobs commonly held by women. Am. J. Epidemiol. 2002, 155, 368–378. [Google Scholar] [CrossRef]
- Ptitsyna, N.G.; Villoresi, G.; Kopytenko, Y.A. Railway-generated magnetic field: Environmental aspects. Railw. Transp. Policies Technol. Perspect. 2009, 87–140. [Google Scholar]
- Bowman, J.D.; Methner, M.M. Hazard surveillance for industrial magnetic fields: II. Field characteristics from waveform measurements. Ann. Occup. Hyg. 2000, 44, 615–633. [Google Scholar] [CrossRef]
- Methner, M.M.; Bowman, J.D. Hazard surveillance for industrial magnetic fields: I. Walkthrough survey of ambient fields and sources. Ann. Occup. Hyg. 2000, 44, 603–614. [Google Scholar] [CrossRef]
- ILO. Radiation Protection of Workers–Ionizing Radiation, ILO Code of Practice; International Labour Office: Geneva, Switzerland, 1987. [Google Scholar]
- ICNIRP. Guidelines for limiting exposure to time-varying electric and magnetic fields (1 hz–100 khz). Health Phys. 2010, 99, 818–836. [Google Scholar] [CrossRef]
- Engel, C.L.; Rasanayagam, M.S.; Gray, J.M.; Rizzo, J. Work and female breast cancer: The state of the evidence, 2002–2017. New Solut. 2018, 28, 55–78. [Google Scholar] [CrossRef]
- McCurdy, A.L.; Wijnberg, L.; Loomis, D.; Savitz, D.; Nylander-French, L.A. Exposure to extremely low frequency magnetic fields among working women and homemakers. Ann. Occup. Hyg. 2001, 45, 643–650. [Google Scholar] [CrossRef]
- Li, P.; McLaughlin, J.; Infante-Rivard, C. Maternal occupational exposure to extremely low frequency magnetic fields and the risk of brain cancer in the offspring. Cancer Causes Control 2009, 20, 945–955. [Google Scholar] [CrossRef]
- Sorahan, T. Magnetic fields and brain tumour risks in uk electricity supply workers. Occup. Med. 2014, 64, 157–165. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Turner, M.C.; Benke, G.; Bowman, J.D.; Figuerola, J.; Fleming, S.; Hours, M.; Kincl, L.; Krewski, D.; McLean, D.; Parent, M.E.; et al. Occupational exposure to extremely low-frequency magnetic fields and brain tumor risks in the interocc study. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1863–1872. [Google Scholar] [CrossRef] [PubMed]
- Oraby, T.; Sivaganesan, S.; Bowman, J.D.; Kincl, L.; Richardson, L.; McBride, M.; Siemiatycki, J.; Cardis, E.; Krewski, D.; On behalf of the INTEROCC Study Group. Berkson error adjustment and other exposure surrogates in occupational case-control studies, with application to the canadian interocc study. J. Expo. Sci. Environ. Epidemiol. 2018, 28, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, M.; Koppel, T.; Ahonen, M.; Hardell, L. Case-control study on occupational exposure to extremely low-frequency electromagnetic fields and the association with meningioma. BioMed Res. Int. 2018, 2018, 5912394. [Google Scholar] [CrossRef]
- Goodman, M.; Kelsh, M.; Ebi, K.; Iannuzzi, J.; Langholz, B. Evaluation of potential confounders in planning a study of occupational magnetic field exposure and female breast cancer. Epidemiology 2002, 13, 50–58. [Google Scholar] [CrossRef]
- Kelsh, M.A.; Bracken, T.D.; Sahl, J.D.; Shum, M.; Ebi, K.L. Occupational magnetic field exposures of garment workers: Results of personal and survey measurements. Bioelectromagnetics 2003, 24, 316–326. [Google Scholar] [CrossRef]
- Juutilainen, J.; Kumlin, T. Occupational magnetic field exposure and melatonin: Interaction with light-at-night. Bioelectromagnetics 2006, 27, 423–426. [Google Scholar] [CrossRef]
- Hardell, L.; Holmberg, B.; Malker, H.; Paulsson, L.E. Exposure to extremely-low-frequency electromagnetic-fields and the risk of malignant diseases—An evaluation of epidemiologic and experimental findings. Eur. J. Cancer Prev. 1995, 4, 3–107. [Google Scholar] [CrossRef] [PubMed]
- McElroy, J.A.; Egan, K.M.; Titus-Ernstoff, L.; Anderson, H.A.; Trentham-Dietz, A.; Hampton, J.M.; Newcomb, P.A. Occupational exposure to electromagnetic field and breast cancer risk in a large, population based, case-control study in the united states. J. Occup. Environ. Med. 2007, 49, 266–274. [Google Scholar] [CrossRef]
- Grundy, A.; Harris, S.A.; Demers, P.A.; Johnson, K.C.; Agnew, D.A.; Villeneuve, P.J.; Canadian Canc Registries, E. Occupational exposure to magnetic fields and breast cancer among canadian men. Cancer Med. 2016, 5, 586–596. [Google Scholar] [CrossRef]
- Minder, C.E.; Pfluger, D.H. Leukemia, brain tumors, and exposure to extremely low frequency electromagnetic fields in swiss railway employees. Am. J. Epidemiol. 2001, 153, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Cam, S.T.; Firlarer, A.; Ozden, S.; Canseven, A.G.; Seyhan, N. Occupational exposure to magnetic fields from transformer stations and electric enclosures in Turkey. Electromagn. Biol. Med. 2011, 30, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Bethwaite, P.; Cook, A.; Kennedy, J.; Pearce, N. Acute leukemia in electrical workers: A new zealand case-control study. Cancer Causes Control 2001, 12, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Kheifets, L.; Monroe, J.; Vergara, X.; Mezei, G.; Afifi, A.A. Occupational electromagnetic fields and leukemia and brain cancer: An update to two meta-analyses. J. Occup. Environ. Med. 2008, 50, 677–688. [Google Scholar] [CrossRef]
- Roosli, M.; Lortscher, M.; Egger, M.; Pfluger, D.; Schreier, N.; Lortscher, E.; Locher, P.; Spoerri, A.; Minder, C. Leukaemia, brain tumours and exposure to extremely low frequency magnetic fields: Cohort study of Swiss railway employees. Occup. Environ. Med. 2007, 64, 553–559. [Google Scholar] [CrossRef]
- Huss, A.; Spoerri, A.; Egger, M.; Kromhout, H.; Vermeulen, R.; Swiss Natl, C. Occupational extremely low frequency magnetic fields (elf-mf) exposure and hematolymphopoietic cancers—Swiss national cohort analysis and updated meta-analysis. Environ. Res. 2018, 164, 467–474. [Google Scholar] [CrossRef]
- Nordenson, I.; Mild, K.H.; Jarventaus, H.; Hirvonen, A.; Sandstrom, M.; Wilen, J.; Blix, N.; Norppa, H. Chromosomal aberrations in peripheral lymphocytes of train engine drivers. Bioelectromagnetics 2001, 22, 306–315. [Google Scholar] [CrossRef]
- Talibov, M.; Guxens, M.; Pukkala, E.; Huss, A.; Kromhout, H.; Slottje, P.; Martinsen, J.I.; Kjaerheim, K.; Sparen, P.; Weiderpass, E.; et al. Occupational exposure to extremely low-frequency magnetic fields and electrical shocks and acute myeloid leukemia in four nordic countries. Cancer Causes Control 2015, 26, 1079–1085. [Google Scholar] [CrossRef]
- Tynes, T.; Haldorsen, T. Residential and occupational exposure to 50 hz magnetic fields and hematological cancers in Norway. Cancer Causes Control 2003, 14, 715–720. [Google Scholar] [CrossRef]
- Harrington, J.M.; Nichols, L.; Sorahan, T.; van Tongeren, M. Leukaemia mortality in relation to magnetic field exposure: Findings from a study of united kingdom electricity generation and transmission workers, 1973–1997. Occup. Environ. Med. 2001, 58, 307–314. [Google Scholar] [CrossRef]
- Willett, E.V.; McKinney, P.A.; Fear, N.T.; Cartwright, R.A.; Roman, E. Occupational exposure to electromagnetic fields and acute leukaemia: Analysis of a case-control study. Occup. Environ. Med. 2003, 60, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Hug, K.; Grize, L.; Seidler, A.; Kaatsch, P.; Schuz, J. Parental occupational exposure to extremely low frequency magnetic fields and childhood cancer: A german case-control study. Am. J. Epidemiol. 2010, 171, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Talibov, M.; Olsson, A.; Bailey, H.; Erdmann, F.; Metayer, C.; Magnani, C.; Petridou, E.; Auvinen, A.; Spector, L.; Clavel, J.; et al. Parental occupational exposure to low-frequency magnetic fields and risk of leukaemia in the offspring: Findings from the childhood leukaemia international consortium (CLIC). Occup. Environ. Med. 2019, 76, 746–753. [Google Scholar] [CrossRef]
- Su, L.L.; Fei, Y.; Wei, X.X.; Guo, J.; Jiang, X.G.; Lu, L.Q.; Chen, G.D. Associations of parental occupational exposure to extremely low-frequency magnetic fields with childhood leukemia risk. Leuk. Lymphoma 2016, 57, 2855–2862. [Google Scholar] [CrossRef]
- Hakansson, N.; Floderus, B.; Gustavsson, P.; Johansen, C.; Olsen, J.H. Cancer incidence and magnetic field exposure in industries using resistance welding in Sweden. Occup. Environ. Med. 2002, 59, 481–486. [Google Scholar] [CrossRef]
- Yousif, L.; Blettner, M.; Hammer, G.P.; Zeeb, H. Testicular cancer risk associated with occupational radiation exposure: A systematic literature review. J. Radiol. Prot. 2010, 30, 389–406. [Google Scholar] [CrossRef]
- Karipidis, K.; Benke, G.; Sim, M.; Fritschi, L.; Yost, M.; Armstrong, B.; Hughes, A.M.; Grulich, A.; Vajdic, C.M.; Kaldor, J.M.; et al. Occupational exposure to power frequency magnetic fields and risk of non-hodgkin lymphoma. Occup. Environ. Med. 2007, 64, 25–29. [Google Scholar] [CrossRef]
- Forssen, U.M.; Lonn, S.; Ahlbom, A.; Savitz, D.A.; Feychting, M. Occupational magnetic field exposure and the risk of acoustic neuroma. Am. J. Ind. Med. 2006, 49, 112–118. [Google Scholar] [CrossRef]
- Carlberg, M.; Koppel, T.; Ahonen, M.; Hardell, L. Case-control study on occupational exposure to extremely low-frequency electromagnetic fields and the association with acoustic neuroma. Environ. Res. 2020, 187, 109621. [Google Scholar] [CrossRef]
- Lope, V.; Perez-Gomez, B.; Aragones, N.; Lopez-Abentes, G.; Gustavsson, P.; Floderus, B.; Dosemeci, M.; Silva, A.; Pollan, M. Occupational exposure to ionizing radiation and electromagnetic fields in relation to the risk of thyroid cancer in Sweden. Scand. J. Work Environ. Health 2006, 32, 276–284. [Google Scholar] [CrossRef]
- De Roos, A.J.; Teschke, K.; Savitz, D.A.; Poole, C.; Grufferman, S.; Pollock, B.H.; Olshan, A.F. Parental occupational exposures to electromagnetic fields and radiation and the incidence of neuroblastoma in offspring. Epidemiology 2001, 12, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Sahl, J.; Mezei, G.; Kavet, R.; McMillan, A.; Silvers, A.; Sastre, A.; Kheifets, L. Occupational magnetic field exposure and cardiovascular mortality in a cohort of electric utility workers. Am. J. Epidemiol. 2002, 156, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Lange, S.; Viergutz, T.; Simko, M. Modifications in cell cycle kinetics and in expression of g(1) phase-regulating proteins in human amniotic cells after exposure to electromagnetic fields and ionizing radiation. Cell Prolif. 2004, 37, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.Z.; Dong, L.A.; Chen, Y.T.; Qi, H.S.; Xiao, D.M. Effects of sinusoidal magnetic field observed on cell proliferation, ion concentration, and osmolarity in two human cancer cell lines. Electromagn. Biol. Med. 2006, 25, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Buemi, M.; Marino, D.; Di Pasquale, G.; Floccari, F.; Senatore, M.; Aloisi, C.; Grasso, F.; Mondio, G.; Perillo, P.; Frisina, N.; et al. Cell proliferation/cell death balance in renal cell cultures after exposure to a static magnetic field. Nephron 2001, 87, 269–273. [Google Scholar] [CrossRef]
- Sztafrowski, D.; Jazwiec, B.; Gumiela, J.; Kuliczkowski, K. Influence of north and south poles of static magnetic field (SMF) on apoptosis of hl60 cell line. Prz. Elektrotech. 2018, 94, 182–185. [Google Scholar]
- Tenuzzo, B.; Chionna, A.; Panzarini, E.; Lanubile, R.; Tarantino, P.; Di Jeso, B.; Dwikat, M.; Dini, L. Biological effects of 6 mt static magnetic fields: A comparative study in different cell types. Bioelectromagnetics 2006, 27, 560–577. [Google Scholar] [CrossRef]
- Naziroglu, M.; Clg, B.; Dogan, S.; Uguz, A.C.; Dilek, S.; Faouzi, D. 2.45-gz wireless devices induce oxidative stress and proliferation through cytosolic ca2+ influx in human leukemia cancer cells. Int. J. Radiat. Biol. 2012, 88, 449–456. [Google Scholar] [CrossRef]
- Barati, M.; Javidi, M.A.; Darvishi, B.; Shariatpanahi, S.P.; Moosavi, Z.S.M.; Ghadirian, R.; Khani, T.; Sanati, H.; Simaee, H.; Barough, M.S.; et al. Necroptosis triggered by ros accumulation and Ca2+ overload, partly explains the inflammatory responses and anti-cancer effects associated with 1 hz, 100 mt elf-mf in vivo. Free Radic. Biol. Med. 2021, 169, 84–98. [Google Scholar] [CrossRef]
- Rosen, A.D. Mechanism of action of moderate-intensity static magnetic fields on biological systems. Cell Biochem. Biophys. 2003, 39, 163–173. [Google Scholar] [CrossRef]
- Wei, J.; Sun, J.; Xu, H.; Shi, L.; Sun, L.; Zhang, J. Effects of extremely low frequency electromagnetic fields on intracellular calcium transients in cardiomyocytes. Electromagn. Biol. Med. 2015, 34, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Chen, C.C.; Tu, W.; Cheng, Y.T.; Tseng, F.G. Design and fabrication of a microplatform for the proximity effect study of localized elf-EMF on the growth of in vitro hela and pc-12 cells. J. Micromech. Microeng. 2010, 20, 125023. [Google Scholar] [CrossRef]
- Bae, J.E.; Do, J.Y.; Kwon, S.H.; Lee, S.D.; Jung, Y.W.; Kim, S.C.; Chae, K.S. Electromagnetic field-induced converse cell growth during a long-term observation. Int. J. Radiat. Biol. 2013, 89, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.; Lange, S.; Viergutz, T.; Kriehuber, R.; Weiss, D.G.; Simko, M. Influence of 50 hz electromagnetic fields in combination with a tumour promoting phorbol ester on protein kinase c and cell cycle in human cells. Mol. Cell. Biochem. 2002, 232, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Kapri-Pardes, E.; Hanoch, T.; Maik-Rachline, G.; Murbach, M.; Bounds, P.L.; Kuster, N.; Seger, R. Activation of signaling cascades by weak extremely low frequency electromagnetic fields. Cell. Physiol. Biochem. 2017, 43, 1533–1546. [Google Scholar] [CrossRef] [PubMed]
- Kirschenlohr, H.; Ellis, P.; Hesketh, R.; Metcalfe, J. Gene expression profiles in white blood cells of volunteers exposed to a 50 hz electromagnetic field. Radiat. Res. 2012, 178, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Kabacik, S.; Kirschenlohr, H.; Raffy, C.; Whitehill, K.; Coster, M.; Abe, M.; Brindle, K.; Badie, C.; Sienkiewicz, Z.; Bouffler, S. Investigation of transcriptional responses of juvenile mouse bone marrow to power frequency magnetic fields. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 2013, 745, 40–45. [Google Scholar] [CrossRef]
- Panagopoulos, D.J.; Karabarbounis, A.; Lioliousis, C. Elf alternating magnetic field decreases reproduction by DNA damage induction. Cell Biochem. Biophys. 2013, 67, 703–716. [Google Scholar] [CrossRef]
- Pardo, J.C.T.; Grimaldi, S.; Taranta, M.; Naldi, I.; Cinti, C. Microwave electromagnetic field regulates gene expression in t-lymphoblastoid leukemia ccrf-cem cell line exposed to 900 mhz. Electromagn. Biol. Med. 2012, 31, 1–18. [Google Scholar] [CrossRef]
- Zhou, J.L.; Li, C.L.; Yao, G.D.; Chiang, H.A.; Chang, Z.L. Gene expression of cytokine receptors in hl60 cells exposed to a 50 hz magnetic field. Bioelectromagnetics 2002, 23, 339–346. [Google Scholar] [CrossRef]
- Diab, K.A. The impact of the low frequency of the electromagnetic field on human. In Cell Biology and Translational Medicine, Volume 7: Stem Cells and Therapy: Emerging Approaches; Turksen, K., Ed.; Springer: Berlin/Heidelberg, Germany, 2020; Volume 1237, pp. 135–149. [Google Scholar]
- Giorgi, G.; Del Re, B. Epigenetic dysregulation in various types of cells exposed to extremely low-frequency magnetic fields. Cell Tissue Res 2021, 386, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Harada, S.; Yamada, S.; Kuramata, O.; Gunji, Y.; Kawasaki, M.; Miyakawa, T.; Yonekura, H.; Sakurai, S.; Bessho, K.; Hosono, R.; et al. Effects of high elf magnetic fields on enzyme-catalyzed DNA and RNA synthesis in vitro and on a cell-free DNA mismatch repair. Bioelectromagnetics 2001, 22, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Li, H.L.; Lin, L.; Li, L.; Zhou, L.; Zhang, Y.; Hao, S.; Ding, Z.H. Exosomal small rna sequencing uncovers the microrna dose markers for power frequency electromagnetic field exposure. Biomarkers 2018, 23, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Manser, M.; Sater, M.R.A.; Schmid, C.D.; Noreen, F.; Murbach, M.; Kuster, N.; Schuermann, D.; Schar, P. ELF-MF exposure affects the robustness of epigenetic programming during granulopoiesis. Sci. Rep. 2017, 7, 43345. [Google Scholar] [CrossRef]
- Sun, W.J.; Gan, Y.P.; Fu, Y.T.; Lu, D.Q.; Chiang, H. An incoherent magnetic field inhibited EGF receptor clustering and phosphorylation induced by a 50-hz magnetic field in cultured FL cells. Cell. Physiol. Biochem. 2008, 22, 507–514. [Google Scholar] [CrossRef]
- Ke, X.Q.; Sun, W.J.; Lu, D.Q.; Fu, Y.T.; Chiang, H. 50-hz magnetic field induces EGF-receptor clustering and activates RAS. Int. J. Radiat. Biol. 2008, 84, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Eleuteri, A.M.; Amici, M.; Bonfili, L.; Cecarini, V.; Cuccioloni, M.; Grimaldi, S.; Giuliani, L.; Angeletti, M.; Fioretti, E. 50 hz extremely low frequency electromagnetic fields enhance protein carbonyl groups content in cancer cells: Effects on proteasomal systems. J. Biomed. Biotechnol. 2009, 2009, 834239. [Google Scholar] [CrossRef]
- Testa, A.; Cordelli, E.; Stronati, L.; Marino, C.; Lovisolo, G.A.; Fresegna, A.M.; Conti, D.; Villani, P. Evaluation of genotoxic effect of low level 50 hz magnetic fields on human blood cells using different cytogenetic assays. Bioelectromagnetics 2004, 25, 613–619. [Google Scholar] [CrossRef]
- Stronati, L.; Testa, A.; Villani, R.; Marino, C.; Lovisolo, G.A.; Conti, D.; Russo, F.; Fresegna, A.M.; Cordelli, E. Absence of genotoxicity in human blood cells exposed to 50 hz magnetic fields as assessed by comet assay, chromosome aberration, micronucleus, and sister chromatid exchange analyses. Bioelectromagnetics 2004, 25, 41–48. [Google Scholar] [CrossRef]
- Kayhan, H.; Erdebilli, B.; Gonen, S.; Esmekaya, M.A.; Ertekin, E.; Canseven, A.G. Effects of extremely low-frequency magnetic field on healthy fibroblasts and breast cancer cells. J. Istanb. Fac. Med. 2020, 83, 384–389. [Google Scholar] [CrossRef]
- Kim, J.; Ha, C.S.; Lee, H.J.; Song, K. RePETitive exposure to a 60-hz time-varying magnetic field induces DNA double-strand breaks and apoptosis in human cells. Biochem. Biophys. Res. Commun. 2010, 400, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Supino, R.; Bottone, M.G.; Pellicciari, C.; Caserini, C.; Bottiroli, G.; Belleri, M.; Veicsteinas, A. Sinusoidal 50 hz magnetic fields do not affect structural morphology and proliferation of human cells in vitro. Histol. Histopathol. 2001, 16, 719–726. [Google Scholar]
- Savage, R.E.; Kanitz, M.H.; Lotz, W.G.; Conover, D.; Hennessey, E.M.; Hanneman, W.H.; Witzmann, F.A. Changes in gene and protein expression in magnetic field-treated human glioma cells. Toxicol. Mech. Methods 2005, 15, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Kanitz, M.H.; Witzmann, F.A.; Lotz, W.G.; Conover, D.; Savage, R.E. Investigation of protein expression in magnetic field-treated human glioma cells. Bioelectromagnetics 2007, 28, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, H.; Tsuchiya, T.; Mizoe, H.; Ozeki, H.; Kanao, S.; Yomori, H.; Sakane, C.; Hasebe, S.; Motomura, T.; Yamakawa, T.; et al. No effect of extremely low-frequency magnetic field observed on cell growth or initial response of cell proliferation in human cancer cell lines. Bioelectromagnetics 2002, 23, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Calcabrini, C.; Mancini, U.; De Bellis, R.; Diaz, A.R.; Martinelli, M.; Cucchiarini, L.; Sestili, P.; Stocchi, V.; Potenza, L. Effect of extremely low-frequency electromagnetic fields on antioxidant activity in the human keratinocyte cell line nctc 2544. Biotechnol. Appl. Biochem. 2017, 64, 415–422. [Google Scholar] [CrossRef]
- Kim, K.; Lee, Y.S.; Kim, N.; Choi, H.D.; Kang, D.J.; Kim, H.R.; Lim, K.M. Effects of electromagnetic waves with LTE and 5g bandwidth on the skin pigmentation in vitro. Int. J. Mol. Sci. 2021, 22, 170. [Google Scholar] [CrossRef]
- Jandova, A.; Pokorny, J.; Cocek, A.; Trojan, S.; Nedbalova, M.; Dohnalova, A. Effects of sinusoidal 0.5 mt magnetic field on leukocyte adherence inhibition. Electromagn. Biol. Med. 2004, 23, 81–96. [Google Scholar]
- Jandova, A.; Hurych, J.; Pokorny, J.; Cocek, A.; Trojan, S.; Nedbalova, M.; Dohnalova, A. Effects of sinusoidal magnetic field on adherence inhibition of leukocytes. Electro- Magn. 2001, 20, 397–413. [Google Scholar] [CrossRef]
- Kaszuba-Zwoinska, J.; Zdzilowska, E.; Chorobik, P.; Slodowska-Hajduk, Z.; Juszczak, K.; Zaraska, W.; Thor, P.J. Pulsing electromagnetic field and death of proliferating peripheral blood mononuclear cells from patients with acute myelogenic leukemia. Adv. Clin. Exp. Med. 2011, 20, 721–727. [Google Scholar]
- Mangiacasale, R.; Tritarelli, A.; Sciamanna, I.; Cannone, M.; Lavia, P.; Barberis, M.C.; Lorenzini, R.; Cundari, E. Normal and cancer-prone human cells respond differently to extremely low frequency magnetic fields. FEBS Lett. 2001, 487, 397–403. [Google Scholar] [CrossRef]
- Kaszuba-Zwoinska, J.; Wojcik, K.; Bereta, M.; Ziomber, A.; Pierzchalski, P.; Rokita, E.; Marcinkiewicz, J.; Zaraska, W.; Thor, P. Pulsating electromagnetic field stimulation prevents cell death of puromycin treated u937 cell line. J. Physiol. Pharmacol. 2010, 61, 201–205. [Google Scholar] [PubMed]
- Hirose, H.; Nakahara, T.; Zhang, Q.M.; Yonei, S.; Miyakoshi, J. Static magnetic field with a strong magnetic field gradient (41.7 t/m) induces c-jun expression in hl-60 cells. In Vitro Cell. Dev. Biol.-Anim. 2003, 39, 348–352. [Google Scholar] [CrossRef]
- Golbach, L.A.; Philippi, J.G.M.; Cuppen, J.J.M.; Savelkoul, H.F.J.; Verburg-van Kemenade, B.M.L. Calcium signalling in human neutrophil cell lines is not affected by low-frequency electromagnetic fields. Bioelectromagnetics 2015, 36, 430–443. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.H.; Dong, L.A.; Zhang, B.H.; Qi, N.M. Effects of extremely low-frequency magnetic field on growth and differentiation of human mesenchymal stem cells. Electromagn. Biol. Med. 2010, 29, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Santini, M.T.; Rainaldi, G.; Ferrante, A.; Indovina, P.L.; Vecchia, P.; Donelli, G. Effects of a 50 hz sinusoidal magnetic field on cell adhesion molecule expression in two human osteosarcoma cell lines (mg-63 and saos-2). Bioelectromagnetics 2003, 24, 327–338. [Google Scholar] [CrossRef]
- Saunders, R. Static magnetic fields: Animal studies. Prog. Biophys. Mol. Biol. 2005, 87, 225–239. [Google Scholar] [CrossRef]
- Mild, K.H.; Mattsson, M.O. Elf noise fields: A review. Electromagn. Biol. Med. 2010, 29, 72–97. [Google Scholar] [CrossRef]
- Marino, A.A.; Wolcott, R.M.; Chervenak, R.; Jourd’heuil, F.; Nilsen, E.; Frilot, C. Nonlinear dynamical law governs magnetic field induced changes in lymphoid phenotype. Bioelectromagnetics 2001, 22, 529–546. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.E.; Morris, J.E.; Sasser, L.B.; Loscher, W. Effects of 50-or 60-hertz, 100 mu t magnetic field exposure in the dmba mammary cancer model in sprague-dawley rats: Possible explanations for different results from two laboratories. Environ. Health Perspect. 2000, 108, 797–802. [Google Scholar]
- Maruvada, P.S.; Harvey, S.M.; Jutras, P.; Goulet, D.; Mandeville, R. A magnetic field exposure facility for evaluation of animal carcinogenicity. Bioelectromagnetics 2000, 21, 432–438. [Google Scholar] [CrossRef]
- Loscher, W. Do cocarcinogenic effects of elf electromagnetic fields require repeated long-term interaction with carcinogens? Characteristics of positive studies using the dmba breast cancer model in rats. Bioelectromagnetics 2001, 22, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Fedrowitz, M.; Kamino, K.; Loscher, W. Significant differences in the effects of magnetic field exposure on 7,12-dimethylbenz(a)anthracene-induced mammary carcinogenesis in two substrains of sprague-dawley rats. Cancer Res. 2004, 64, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Galloni, P.; Marino, C. Effects of 50 hz magnetic field exposure on tumor experimental models. Bioelectromagnetics 2000, 21, 608–614. [Google Scholar] [CrossRef]
- McLean, J.R.; Thansandote, A.; McNamee, J.P.; Tryphonas, L.; Lecuyer, D.; Gajda, G. A 60 hz magnetic field does not affect the incidence of squamous cell carcinomas in sencar mice. Bioelectromagnetics 2003, 24, 75–81. [Google Scholar] [CrossRef]
- McNamee, J.P.; Bellier, P.V.; McLean, J.R.N.; Marro, L.; Gajda, G.B.; Thansandote, A. DNA damage and apoptosis in the immature mouse cerebellum after acute exposure to a 1 mt, 60 hz magnetic field. Mutat. Res. Genet. Toxicol. Environ. Mutagenesis 2002, 513, 121–133. [Google Scholar] [CrossRef]
- McNamee, J.P.; Bellier, P.V.; Chauhan, V.; Gajda, G.B.; Lemay, E.; Thansandote, A. Evaluating DNA damage in rodent brain after acute 60 hz magnetic-field exposure. Radiat. Res. 2005, 164, 791–797. [Google Scholar] [CrossRef]
- Luo, X.; Jia, S.J.; Li, R.Y.; Gao, P.; Zhang, Y.W. Occupational exposure to 50 hz magnetic fields does not alter responses of inflammatory genes and activation of splenic lymphocytes in mice. Int. J. Occup. Med. Environ. Health 2016, 29, 277–291. [Google Scholar] [CrossRef]
- Sommer, A.M.; Lerchl, A. 50 hz magnetic fields of 1 mt do not promote lymphoma development in AKR/J mice. Radiat. Res. 2006, 165, 343–349. [Google Scholar] [CrossRef]
- Tatarov, I.; Panda, A.; Petkov, D.; Kolappaswamy, K.; Thompson, K.; Kavirayani, A.; Lipsky, M.M.; Elson, E.; Davis, C.C.; Martin, S.S.; et al. Effect of magnetic fields on tumor growth and viability. Comp. Med. 2011, 61, 339–345. [Google Scholar]
- Vallejo, D.; Hidalgo, M.A.; Hernandez, J.M. Effects of long-term exposure to an extremely low frequency magnetic field (15 microt) on selected blood coagulation variables in of1 mice. Electromagn. Biol. Med. 2019, 38, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Cabrales, L.B.; Ciria, H.C.; Bruzon, R.P.; Quevedo, M.S.; Cespedes, M.C.; Salas, M.F. Elf magnetic field effects on some hematological and biochemical parameters of peripheral blood in mice. Electro- Magn. 2001, 20, 185–191. [Google Scholar] [CrossRef]
- Qi, G.Y.; Zuo, X.X.; Zhou, L.H.; Aoki, E.; Okamula, A.; Watanebe, M.; Wang, H.P.; Wu, Q.H.; Lu, H.L.; Tuncel, H.; et al. Effects of extremely low-frequency electromagnetic fields (elf-EMF) exposure on b6c3f1 mice. Environ. Health Prev. Med. 2015, 20, 287–293. [Google Scholar] [CrossRef]
- Salim, E.I.; Omar, K.M.; Abou-Hattab, H.A.; Abou-Zaid, F.A. Pituitary toxicity but lack of rat colon carcinogenicity of a dc-magnetic field in a medium-term bioassay. Asian Pac. J. Cancer Prev. 2008, 9, 131–140. [Google Scholar]
- Anderson, L.E.; Morris, J.E.; Miller, D.L.; Rafferty, C.N.; Ebi, K.L.; Sasser, L.B. Large granular lymphocytic (lgl) leukemia in rats exposed to intermittent 60 hz magnetic fields. Bioelectromagnetics 2001, 22, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Choi, S.Y.; Jang, J.J.; Gimm, Y.M.; Pack, J.K.; Choi, H.D.; Kim, N.; Lee, Y.S. Lack of promotion of mammary, lung and skin tumorigenesis by 20 khz triangular magnetic fields. Bioelectromagnetics 2007, 28, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Tuncel, H.; Shimamoto, F.; Cagatay, P.; Kalkan, M.T. Variable e-cadherin expression in a mnu-induced colon tumor model in rats which exposed with 50 hz frequency sinusoidal magnetic field. Tohoku J. Exp. Med. 2002, 198, 245–249. [Google Scholar] [CrossRef][Green Version]
- Fedrowitz, M.; Loscher, W. Exposure of fischer 344 rats to a weak power frequency magnetic field facilitates mammary tumorigenesis in the dmba model of breast cancer. Carcinogenesis 2008, 29, 186–193. [Google Scholar] [CrossRef]
- Ushio-Fukai, M.; Ash, D.; Nagarkoti, S.; de Chantemele, E.J.B.; Fulton, D.J.R.; Fukai, T. Interplay between reactive oxygen/reactive nitrogen species and metabolism in vascular biology and disease. Antioxid. Redox Signal. 2021, 34, 1319–1354. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef]
- Azab, A.E.; Ebrahim, S.A. Exposure to electromagnetic fields induces oxidative stress and pathophysiological changes in the cardiovascular system. J. Appl. Biotechnol. Bioeng. 2017, 4, 96–102. [Google Scholar] [CrossRef]
- Schuermann, D.; Mevissen, M. Manmade electromagnetic fields and oxidative stress-biological effects and consequences for health. Int. J. Mol. Sci. 2021, 22, 3772. [Google Scholar] [CrossRef] [PubMed]
- Havas, M. When theory and observation collide: Can non-ionizing radiation cause cancer? Environ. Pollut. 2017, 221, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Hore, P.J.; Ivanov, K.L.; Wasielewski, M.R. Spin chemistry. J. Chem. Phys. 2020, 152, 120401. [Google Scholar] [CrossRef]
- Jones, A.R. Magnetic field effects in proteins. Mol. Phys. 2016, 114, 1691–1702. [Google Scholar] [CrossRef]
- Mattsson, M.O.; Simkó, M. Grouping of experimental conditions as an approach to evaluate effects of extremely low-frequency magnetic fields on oxidative response in in vitro studies. Front. Public Health 2014, 2, 132. [Google Scholar] [CrossRef]
- Juutilainen, J.; Herrala, M.; Luukkonen, J.; Naarala, J.; Hore, P.J. Magnetocarcinogenesis: Is there a mechanism for carcinogenic effects of weak magnetic fields? Proc. Biol. Sci. 2018, 285, 20180590. [Google Scholar] [CrossRef]
- Höytö, A.; Herrala, M.; Luukkonen, J.; Juutilainen, J.; Naarala, J. Cellular detection of 50 hz magnetic fields and weak blue light: Effects on superoxide levels and genotoxicity. Int. J. Radiat. Biol. 2017, 93, 646–652. [Google Scholar] [CrossRef]
- Orel, V.E.; Krotevych, M.; Dasyukevich, O.; Rykhalskyi, O.; Syvak, L.; Tsvir, H.; Tsvir, D.; Garmanchuk, L.; Orel Vcapital Ve, C.; Sheina, I.; et al. Effects induced by a 50 hz electromagnetic field and doxorubicin on walker-256 carcinosarcoma growth and hepatic redox state in rats. Electromagn. Biol. Med. 2021, 40, 475–487. [Google Scholar] [CrossRef]
- Mannerling, A.C.; Simko, M.; Mild, K.H.; Mattsson, M.O. Effects of 50-hz magnetic field exposure on superoxide radical anion formation and hsp70 induction in human k562 cells. Radiat. Environ. Biophys. 2010, 49, 731–741. [Google Scholar] [CrossRef]
- Bułdak, R.J.; Polaniak, R.; Bułdak, Ł.; Żwirska-Korczala, K.; Skonieczna, M.; Monsiol, A.; Kukla, M.; Duława-Bułdak, A.; Birkner, E. Short-term exposure to 50 hz elf-EMF alters the cisplatin-induced oxidative response in at478 murine squamous cell carcinoma cells. Bioelectromagnetics 2012, 33, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Luukkonen, J.; Liimatainen, A.; Juutilainen, J.; Naarala, J. Induction of genomic instability, oxidative processes, and mitochondrial activity by 50hz magnetic fields in human sh-sy5y neuroblastoma cells. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 2014, 760, 33–41. [Google Scholar] [CrossRef]
- Xu, A.; Wang, Q.; Lin, T. Low-frequency magnetic fields (lf-mfs) inhibit proliferation by triggering apoptosis and altering cell cycle distribution in breast cancer cells. Int. J. Mol. Sci. 2020, 21, 2952. [Google Scholar] [CrossRef] [PubMed]
- Cios, A.; Ciepielak, M.; Stankiewicz, W.; Szymanski, L. The influence of the extremely low frequency electromagnetic field on clear cell renal carcinoma. Int. J. Mol. Sci. 2021, 22, 1342. [Google Scholar] [CrossRef] [PubMed]
- Amara, S.; Abdelmelek, H.; Garrel, C.; Guiraud, P.; Douki, T.; Ravanat, J.L.; Favier, A.; Sakly, M.; Ben Rhouma, K. Zinc supplementation ameliorates static magnetic field-induced oxidative stress in rat tissues. Environ. Toxicol. Pharmacol. 2007, 23, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Amara, S.; Abdelmelek, H.; Garrel, C.; Guiraud, P.; Douki, T.; Ravanat, J.L.; Favier, A.; Sakly, M.; Ben Rhouma, K. Influence of static magnetic field on cadmium toxicity: Study of oxidative stress and DNA damage in rat tissues. J. Trace Elem. Med. Biol. 2006, 20, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Amara, S.; Douki, T.; Garel, C.; Favier, A.; Sakly, M.; Rhouma, K.B.; Abdelmelek, H. Effects of static magnetic field exposure on antioxidative enzymes activity and DNA in rat brain. Gen. Physiol. Biophys. 2009, 28, 260–265. [Google Scholar] [CrossRef]
- Amara, S.; Douki, T.; Ravanat, J.L.; Garrel, C.; Guiraud, P.; Favier, A.; Sakly, M.; Ben Rhouma, K.; Abdelmelek, H. Influence of a static magnetic field (250 mt) on the antioxidant response and DNA integrity in thp1 cells. Phys. Med. Biol. 2007, 52, 889–898. [Google Scholar] [CrossRef]
- Reale, M.; Kamal, M.A.; Patruno, A.; Costantini, E.; D’Angelo, C.; Pesce, M.; Greig, N.H. Neuronal cellular responses to extremely low frequency electromagnetic field exposure: Implications regarding oxidative stress and neurodegeneration. PLoS ONE 2014, 9, e104973. [Google Scholar] [CrossRef]
- Patruno, A.; Tabrez, S.; Pesce, M.; Shakil, S.; Kamal, M.A.; Reale, M. Effects of extremely low frequency electromagnetic field (elf-EMF) on catalase, cytochrome p450 and nitric oxide synthase in erythro-leukemic cells. Life Sci. 2015, 121, 117–123. [Google Scholar] [CrossRef]
- Lee, B.C.; Johng, H.M.; Lim, J.K.; Jeong, J.H.; Baik, K.Y.; Nam, T.J.; Lee, J.H.; Kim, J.; Sohn, U.D.; Yoon, G.; et al. Effects of extremely low frequency magnetic field on the antioxidant defense system in mouse brain: A chemiluminescence study. J. Photochem. Photobiol. B Biol. 2004, 73, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Lewicka, M.; Henrykowska, G.A.; Pacholski, K.; Smigielski, J.; Rutkowski, M.; Dziedziczak-Buczynska, M.; Buczynski, A. The effect of electromagnetic radiation emitted by display screens on cell oxygen metabolism—In vitro studies. Arch. Med. Sci. 2015, 11, 1330–1339. [Google Scholar] [CrossRef] [PubMed]
- Zwirska-Korczala, K.; Adamczyk-Sowa, M.; Polaniak, R.; Sowa, P.; Birkner, E.; Drzazga, Z.; Brzozowski, T.; Konturek, S.J. Influence of extremely-low-frequency magnetic field on antioxidative melatonin properties in at478 murine squamous cell carcinoma culture. Biol. Trace Elem. Res. 2004, 102, 227–243. [Google Scholar] [CrossRef]
- Zhao, B.; Yu, T.; Wang, S.; Che, J.; Zhou, L.; Shang, P. Static magnetic field (0.2–0.4 t) stimulates the self-renewal ability of osteosarcoma stem cells through autophagic degradation of ferritin. Bioelectromagnetics 2021, 42, 371–383. [Google Scholar] [CrossRef]
- Kesari, K.K.; Juutilainen, J.; Luukkonen, J.; Naarala, J. Induction of micronuclei and superoxide production in neuroblastoma and glioma cell lines exposed to weak 50 hz magnetic fields. J. R. Soc. Interface 2016, 13, 20150995. [Google Scholar] [CrossRef]
- Erdal, N.; Gürgül, S.; Tamer, L.; Ayaz, L. Effects of long-term exposure of extremely low frequency magnetic field on oxidative/nitrosative stress in rat liver. J. Radiat. Res. 2008, 49, 181–187. [Google Scholar] [CrossRef][Green Version]
- Koh, E.K.; Ryu, B.K.; Jeong, D.Y.; Bang, I.S.; Nam, M.H.; Chae, K.S. A 60-hz sinusoidal magnetic field induces apoptosis of prostate cancer cells through reactive oxygen species. Int. J. Radiat. Biol. 2008, 84, 945–955. [Google Scholar] [CrossRef]
- Kahya, M.C.; Nazıroğlu, M.; Çiğ, B. Selenium reduces mobile phone (900 mhz)-induced oxidative stress, mitochondrial function, and apoptosis in breast cancer cells. Biol. Trace Elem. Res. 2014, 160, 285–293. [Google Scholar] [CrossRef]
- Polaniak, R.; Buldak, R.J.; Karon, M.; Birkner, K.; Kukla, M.; Zwirska-Korczala, K.; Birkner, E. Influence of an extremely low frequency magnetic field (elf-EMF) on antioxidative vitamin e properties in at478 murine squamous cell carcinoma culture in vitro. Int. J. Toxicol. 2010, 29, 221–230. [Google Scholar] [CrossRef]
- Bekhite, M.M.; Finkensieper, A.; Abou-Zaid, F.A.; El-Shourbagy, I.K.; El-Fiky, N.K.; Omar, K.M.; Sauer, H.; Wartenberg, M. Differential effects of high and low strength magnetic fields on mouse embryonic development and vasculogenesis of embryonic stem cells. Reprod. Toxicol. 2016, 65, 46–58. [Google Scholar] [CrossRef]
- Kang, S.K. Measuring the value of MRI: Comparative effectiveness & outcomes research. J. Magn. Reson. Imaging 2019, 49, e78–e84. [Google Scholar] [PubMed]
- van der Heide, U.A.; Frantzen-Steneker, M.; Astreinidou, E.; Nowee, M.E.; van Houdt, P.J. MRI basics for radiation oncologists. Clin. Transl. Radiat. Oncol. 2019, 18, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Raaijmakers, A.J.E.; Raaymakers, B.W.; Lagendijk, J.J.W. Magnetic-field-induced dose effects in mr-guided radiotherapy systems: Dependence on the magnetic field strength. Phys. Med. Biol. 2008, 53, 909–923. [Google Scholar] [CrossRef] [PubMed]
- Morsing, A.; Hildebrandt, M.G.; Vilstrup, M.H.; Wallenius, S.E.; Gerke, O.; Petersen, H.; Johansen, A.; Andersen, T.L.; Hoilund-Carlsen, P.F. Hybrid PET/MRI in major cancers: A scoping review. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2138–2151. [Google Scholar] [CrossRef]
- Scheenen, T.W.J.; Zamecnik, P. The role of magnetic resonance imaging in (future) cancer staging note the nodes. Investig. Radiol. 2021, 56, 42–49. [Google Scholar] [CrossRef]
- Avasthi, A.; Caro, C.; Pozo-Torres, E.; Leal, M.P.; Garcia-Martin, M.L. Magnetic nanoparticles as MRI contrast agents. Top. Curr. Chem. 2020, 378, 43. [Google Scholar]
- Poku, L.O.; Cheng, Y.N.; Wang, K.; Sun, X.L. Na-23-MRI as a noninvasive biomarker for cancer diagnosis and prognosis. J. Magn. Reson. Imaging 2021, 53, 995–1014. [Google Scholar] [CrossRef]
- Visser, M.; Muller, D.M.J.; van Duijn, J.M.; Smits, M.; Verburg, N.; Hendriks, E.J.; Nabuurs, R.J.A.; Bot, J.C.J.; Eijgelaar, R.S.; Witte, M.; et al. Inter-rater agreement in glioma segmentations on longitudinal MRI. NeuroImage Clin. 2019, 22, 101727. [Google Scholar] [CrossRef]
- Patil, R.; Ljubimov, A.V.; Gangalum, P.R.; Ding, H.; Portilla-Arias, J.; Wagner, S.; Inoue, S.; Konda, B.; Rekechenetskiy, A.; Chesnokova, A.; et al. MRI virtual biopsy and treatment of brain metastatic tumors with targeted nanobioconjugates: Nanoclinic in the brain. ACS Nano 2015, 9, 5594–5608. [Google Scholar] [CrossRef]
- Lasocki, A.; Anjari, M.; Kokurcan, S.O.; Thust, S.C. Conventional MRI features of adult diffuse glioma molecular subtypes: A systematic review. Neuroradiology 2021, 63, 353–362. [Google Scholar] [CrossRef]
- Machhale, K.; Nandpuru, H.B.; Kapur, V.; Kosta, L. MRI brain cancer classification using hybrid classifier (SVM-KNN). In Proceedings of the 2015 International Conference on Industrial Instrumentation and Control (ICIC), Pune, India, 28–30 May 2015; pp. 60–65. [Google Scholar]
- Nandpuru, H.B.; Salankar, S.S.; Bora, V.R. MRI brain cancer classification using support vector machine. In Proceedings of the 2014 IEEE Students’ Conference on Electrical, Electronics and Computer Science, Bhopal, India, 1–2 March 2014. [Google Scholar]
- Naser, M.A.; Deen, M.J. Brain tumor segmentation and grading of lower-grade glioma using deep learning in MRI images. Comput. Biol. Med. 2020, 121, 8. [Google Scholar] [CrossRef] [PubMed]
- Clement, P.; Booth, T.; Borovecki, F.; Emblem, K.E.; Figueiredo, P.; Hirschler, L.; Jancalek, R.; Keil, V.C.; Maumet, C.; Ozsunar, Y.; et al. Glimr: Cross-border collaborations to promote advanced MRI biomarkers for glioma. J. Med. Biol. Eng. 2021, 41, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Zhuge, Y.; Ning, H.; Mathen, P.; Cheng, J.Y.; Krauze, A.V.; Camphausen, K.; Miller, R.W. Automated glioma grading on conventional MRI images using deep convolutional neural networks. Med. Phys. 2020, 47, 3044–3053. [Google Scholar] [CrossRef] [PubMed]
- Wietelmann, D.; Schumacher, M.; Muendel, J. Brain-stem glioma. Radiologe 1998, 38, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Porcari, P.; Hegi, M.E.; Lei, H.; Hamou, M.F.; Vassallo, I.; Capuani, S.; Gruetter, R.; Mlynarik, V. Early detection of human glioma sphere xenografts in mouse brain using diffusion MRI at 14.1t. NMR Biomed. 2016, 29, 1577–1589. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rogers, C.M.; Jones, P.S.; Weinberg, J.S. Intraoperative MRI for brain tumors. J. Neuro-Oncol. 2021, 151, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Poussaint, T.Y.; Kocak, M.; Vajapeyam, S.; Packer, R.I.; Robertson, R.L.; Geyer, R.; Haas-Kogan, D.; Pollack, I.F.; Vezina, G.; Zimmerman, R.; et al. MRI as a central component of clinical trials analysis in brainstem glioma: A report from the pediatric brain tumor consortium (pbtc). Neuro-Oncology 2011, 13, 417–427. [Google Scholar] [CrossRef]
- Cabaj, A.; Bekiesinska-Figatowska, M.; Duczkowska, A.; Duczkowski, M. Brain MRI findings in neurological complications of cancer treatment. Adv. Clin. Exp. Med. 2016, 25, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Kjaer, A.; Loft, A.; Law, I.; Berthelsen, A.K.; Borgwardt, L.; Lofgren, J.; Johnbeck, C.B.; Hansen, A.E.; Keller, S.; Holm, S.; et al. Pet/MRI in cancer patients: First experiences and vision from copenhagen. Magn. Reson. Mat. Phys. Biol. Med. 2013, 26, 37–47. [Google Scholar] [CrossRef]
- Tsiouris, S.; Bougias, C.; Fotopoulos, A. Principles and current trends in the correlative evaluation of glioma with advanced MRI techniques and PET. Hell. J. Nucl. Med. 2019, 22, 206–219. [Google Scholar]
- Puttick, S.; Bell, C.; Dowson, N.; Rose, S.; Fay, M. PET, MRI, and simultaneous PET/MRI in the development of diagnostic and therapeutic strategies for glioma. Drug Discov. Today 2015, 20, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Shankar, A.; Bomanji, J.; Hyare, H. Hybrid PET-MRI imaging in paediatric and TYA brain tumours: Clinical applications and challenges. J. Pers. Med. 2020, 10, 218. [Google Scholar] [CrossRef] [PubMed]
- Deuschl, C.; Kirchner, J.; Poeppel, T.D.; Schaarschmidt, B.; Kebir, S.; El Hindy, N.; Hense, J.; Quick, H.H.; Glas, M.; Herrmann, K.; et al. C-11-met PET/MRI for detection of recurrent glioma. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Kebir, S.; Weber, M.; Lazaridis, L.; Deuschl, C.; Schmidt, T.; Monninghoff, C.; Keyvani, K.; Umutlu, L.; Pierscianek, D.; Forsting, M.; et al. Hybrid c-11-met PET/MRI combined with “machine learning” in glioma diagnosis according to the revised glioma who classification 2016. Clin. Nucl. Med. 2019, 44, 214–220. [Google Scholar] [CrossRef]
- Suh, C.H.; Kim, H.S.; Jung, S.C.; Choi, C.G.; Kim, S.J. Perfusion MRI as a diagnostic biomarker for differentiating glioma from brain metastasis: A systematic review and meta-analysis. Eur. Radiol. 2018, 28, 3819–3831. [Google Scholar] [CrossRef]
- Xu, X.; Chan, K.W.Y.; Knutsson, L.; Artemov, D.; Xu, J.D.; Liu, G.; Kato, Y.; Lal, B.; Laterra, J.; McMahon, M.T.; et al. Dynamic glucose enhanced (dge) MRI for combined imaging of blood-brain barrier break down and increased blood volume in brain cancer. Magn. Reson. Med. 2015, 74, 1556–1563. [Google Scholar] [CrossRef]
- Davis, S.C.; Samkoe, K.S.; O’Hara, J.A.; Gibbs-Strauss, S.L.; Payne, H.L.; Hoopes, P.J.; Paulsen, K.D.; Pogue, B.W. MRI-coupled fluorescence tomography quantifies EGFR activity in brain tumors. Acad. Radiol. 2010, 17, 271–276. [Google Scholar] [CrossRef]
- Aquino, D.; Gioppo, A.; Finocchiaro, G.; Bruzzone, M.G.; Cuccarini, V. MRI in glioma immunotherapy: Evidence, pitfalls, and perspectives. J. Immunol. Res. 2017, 2017, 5813951. [Google Scholar] [CrossRef]
- Jenkinson, M.D.; Du Plessis, D.G.; Walker, C.; Smith, T.S. Advanced MRI in the management of adult gliomas. Br. J. Neurosurg. 2007, 21, 550–561. [Google Scholar] [CrossRef]
- Jestaedt, L.; Lemke, D.; Weiler, M.; Pfenning, P.N.; Heiland, S.; Wick, W.; Bendszus, M. Gadofluorine m enhanced MRI in experimental glioma: Superior and persistent intracellular tumor enhancement compared with conventional MRI. J. Magn. Reson. Imaging 2012, 35, 551–560. [Google Scholar] [CrossRef]
- Liao, J.C.; Xia, R.; Liu, T.; Feng, H.; Ai, H.; Song, B.; Gao, F.B. In vivo dynamic monitoring of the biological behavior of labeled c6 glioma by MRI. Mol. Med. Rep. 2013, 7, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Zaccagna, F.; Riemer, F.; Priest, A.N.; McLean, M.A.; Allinson, K.; Grist, J.T.; Dragos, C.; Matys, T.; Gillard, J.H.; Watts, C.; et al. Non-invasive assessment of glioma microstructure using verdict MRI: Correlation with histology. Eur. Radiol. 2019, 29, 5559–5566. [Google Scholar] [CrossRef] [PubMed]
- Anka, A.; Thompson, P.; Mott, E.; Sharma, R.; Zhang, R.Z.; Cross, N.; Sun, J.Y.; Flask, C.A.; Oleinick, N.L.; Dean, D. Dynamic contrast enhanced-magnetic resonance imaging (DCE-MRI) for the assessment of pc 4-sensitized photodynamic therapy of a u87-derived glioma model in the athymic nude rat. In Proceedings of the Conference on Photonic Therapeutics and Diagnostics VI, San Francisco, CA, USA, 23–25 January 2010; SPIE—Society of Photo-Optical Instrumentation Engineers: San Francisco, CA, USA, 2010. [Google Scholar]
- Belle, V.; Anka, A.; Cross, N.; Thompson, P.; Mott, E.; Sharma, R.; Gray, K.; Zhang, R.Z.; Xu, Y.S.; Sun, J.Y.; et al. Dynamic contrast enhanced-magnetic resonance imaging (DCE-MRI) of photodynamic therapy (PDT) outcome and associated changes in the blood-brain barrier following PC 4-PDT of glioma in an athymic nude rat model. In Proceedings of the Conference on Photonic Therapeutics and Diagnostics VIII, San Francisco, CA, USA, 21–24 January 2012; SPIE-Society of Photo-Optical Instrumentation Engineers: San Francisco, CA, USA, 2012. [Google Scholar]
- Young, J.R.; Ressler, J.A.; Mortimer, J.E.; Schmolze, D.; Fitzgibbons, M.; Chen, B.H.T. Performance of enhancement on brain MRI for identifying her2 overexpression in breast cancer brain metastases. Eur. J. Radiol. 2021, 144, 6. [Google Scholar] [CrossRef]
- Papadimitrakopoulou, V.A. Carcinogenesis of head and neck cancer and the role of chemoprevention in its reversal. Curr. Opin. Oncol. 2000, 12, 240–245. [Google Scholar] [CrossRef]
- Loeffelbein, D.J.; Souvatzoglou, M.; Wankerl, V.; Dinges, J.; Ritschl, L.M.; Mucke, T.; Pickhard, A.; Eiber, M.; Schwaiger, M.; Beer, A.J. Diagnostic value of retrospective PET-MRI fusion in head-and-neck cancer. BMC Cancer 2014, 14, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Mullins, B.T.; Falchook, A.D.; Lian, J.; He, K.L.; Shen, D.G.; Dance, M.; Lin, W.L.; Sills, T.M.; Das, S.K.; et al. Evaluation of PET/MRI for tumor volume delineation for head and neck cancer. Front. Oncol. 2017, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Grosse, J.; Hellwig, D. Pet/CT and PET/MRI in head and neck cancer. Laryngorhinootologie 2020, 99, 12–21. [Google Scholar]
- Hermans, R. Diffusion-weighted MRI in head and neck cancer. Curr. Opin. Otolaryngol. Head Neck Surg. 2010, 18, 72–78. [Google Scholar] [CrossRef]
- Thoeny, H.C. Diffusion-weighted MRI in head and neck radiology: Applications in oncology. Cancer Imaging 2011, 10, 209–214. [Google Scholar] [CrossRef]
- Schakel, T.; Hoogduin, J.M.; Terhaard, C.H.J.; Philippens, M.E.P. Diffusion weighted MRI in head-and-neck cancer: Geometrical accuracy. Radiother. Oncol. 2013, 109, 394–397. [Google Scholar] [CrossRef]
- El Beltagi, A.H.; Elsotouhy, A.H.; Own, A.M.; Abdelfattah, W.; Nair, K.; Vattoth, S. Functional magnetic resonance imaging of head and neck cancer: Performance and potential. Neuradiol. J. 2019, 32, 36–52. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.X.; Soraghan, J.; Di-caterina, G.; Grose, D.; Doshi, T. Automatic 3D detection and segmentation of head and neck cancer from MRI data. In Proceedings of the 2018 7th European Workshop on Visual Information Processing (EUVIP), Tampere, Finland, 26–28 November 2018. [Google Scholar]
- Chen, A.M.; Hsu, S.; Lamb, J.; Yang, Y.; Agazaryan, N.; Steinberg, M.L.; Low, D.A.; Cao, M. MRI-guided radiotherapy for head and neck cancer: Initial clinical experience. Clin. Transl. Oncol. 2018, 20, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Baulch, J.; Gandhi, M.; Sommerville, J.; Panizza, B. 3t MRI evaluation of large nerve perineural spread of head and neck cancers. J. Med. Imaging Radiat. Oncol. 2015, 59, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Kanda, T.; Kitajima, K.; Suenaga, Y.; Konishi, J.; Sasaki, R.; Morimoto, K.; Saito, M.; Otsuki, N.; Nibu, K.; Sugimura, K. Value of retrospective image fusion of 18F-FDG PET and MRI for preoperative staging of head and neck cancer: Comparison with PET/CT and contrast-enhanced neck MRI. Eur. J. Radiol. 2013, 82, 2005–2010. [Google Scholar] [CrossRef] [PubMed]
- Baghi, M.; Mack, M.G.; Hambek, M.; Rieger, J.; Vogl, T.; Gstoettner, W.; Knecht, R. The efficacy of MRI with ultrasmall superparamagnetic iron oxide particles (uspio) in head and neck cancers. Anticancer Res. 2005, 25, 3665–3670. [Google Scholar] [PubMed]
- Rahbari, R.; Zhang, L.S.; Kebebew, E. Thyroid cancer gender disparity. Future Oncol. 2010, 6, 1771–1779. [Google Scholar] [CrossRef] [PubMed]
- Dinauer, C.A.; Breuer, C.; Rivkees, S.A. Differentiated thyroid cancer in children: Diagnosis and management. Curr. Opin. Oncol. 2008, 20, 59–65. [Google Scholar] [CrossRef]
- Massimino, M.; Evans, D.B.; Podda, M.; Spinelli, C.; Collini, P.; Pizzi, N.; Bleyer, A. Thyroid cancer in adolescents and young adults. Pediatr. Blood Cancer 2018, 65, 9. [Google Scholar] [CrossRef]
- Regalbuto, C.; Frasca, F.; Pellegriti, G.; Malandrino, P.; Marturano, I.; Di Carlo, I.; Pezzino, V. Update on thyroid cancer treatment. Future Oncol. 2012, 8, 1331–1348. [Google Scholar] [CrossRef]
- CynaGorse, F.; Toubert, M.E.; Zagdanski, A.M.; deKerviler, E.; Feger, C.; Benchaib, N.; Attal, P.; Frija, J.; LavalJeantet, M. Recurrence of differentiated thyroid carcinomas: Value of MRI. J. Radiol. 1996, 77, 1195–1200. [Google Scholar]
- Chen, Q.H.; Raghavan, P.; Mukherjee, S.; Jameson, M.J.; Patrie, J.; Xin, W.J.; Xian, J.F.; Wang, Z.C.; Levine, P.A.; Wintermark, M. Accuracy of MRI for the diagnosis of metastatic cervical lymphadenopathy in patients with thyroid cancer. Radiol. Med. 2015, 120, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Gross, N.D.; Weissman, J.L.; Talbot, J.M.; Andersen, P.E.; Wax, M.K.; Cohen, J.I. MRI detection of cervical metastasis from differentiated thyroid carcinoma. Laryngoscope 2001, 111, 1905–1909. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.M.; Xun, X.Q.; Wang, Y.Z.; Mei, L.; He, L.; Zeng, W.; Wang, C.Y.; Tao, H. MRI and ultrasonography detection of cervical lymph node metastases in differentiated thyroid carcinoma before reoperation. Am. J. Transl. Res. 2014, 6, 147–154. [Google Scholar] [PubMed]
- Samanci, C.; Onal, Y.; Sager, S.; Asa, S.; Ustabasioglu, F.E.; Alis, D.; Akman, C.; Sonmezoglu, K. Diagnostic capabilities of MRI versus 18F FDG PET-ct in postoperative patients with thyroglobulin positive, 131I-negative local recurrent or metastatic thyroid cancer. Curr. Med. Imaging 2019, 15, 956–964. [Google Scholar] [CrossRef]
- Wang, J.C.; Takashima, S.; Matsushita, T.; Takayama, F.; Kobayashi, T.; Kadoya, M. Esophageal invasion by thyroid carcinomas: Prediction using magnetic resonance imaging. J. Comput. Assist. Tomogr. 2003, 27, 18–25. [Google Scholar] [CrossRef]
- Brown, A.M.; Nagala, S.; McLean, M.A.; Lu, Y.G.; Scoffings, D.; Apte, A.; Gonen, M.; Stambuk, H.E.; Shaha, A.R.; Tuttle, R.M.; et al. Multi-institutional validation of a novel textural analysis tool for preoperative stratification of suspected thyroid tumors on diffusion-weighted MRI. Magn. Reson. Med. 2016, 75, 1708–1716. [Google Scholar] [CrossRef]
- Naglah, A.; Khalifa, F.; Khaled, R.; Razek, A.; Ghazal, M.; Giridharan, G.; El-Baz, A. Novel MRI-based cad system for early detection of thyroid cancer using multi-input CNN. Sensors 2021, 21, 3878. [Google Scholar] [CrossRef]
- Abd el Aziz, L.M.; Hamisa, M.; Badwy, M.E. Differentiation of thyroid nodules using diffusion-weighted MRI. Alex. J. Med. 2015, 51, 305–309. [Google Scholar] [CrossRef]
- Taha, M.S.; Hassan, O.; Amir, M.; Taha, T.; Riad, M.A. Diffusion-weighted MRI in diagnosing thyroid cartilage invasion in laryngeal carcinoma. Eur. Arch. Oto-Rhino-Laryngol. 2014, 271, 2511–2516. [Google Scholar] [CrossRef]
- Binse, I.; Poeppel, T.D.; Ruhlmann, M.; Gomez, B.; Umutlu, L.; Bockisch, A.; Rosenbaum-Krumme, S.J. Imaging with i-124 in differentiated thyroid carcinoma: Is PET/MRI superior to PET/CT? Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1011–1017. [Google Scholar] [CrossRef]
- Seiboth, L.; Van Nostrand, D.; Wartofsky, L.; Ousman, Y.; Jonklaas, J.; Butler, C.; Atkins, F.; Burman, K. Utility of PET/neck MRI digital fusion images in the management of recurrent or persistent thyroid cancer. Thyroid 2008, 18, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Teller, P.; Jefford, V.J.; Gabram, S.G.A.; Newell, M.; Carlson, G.W. The utility of breast MRI in the management of breast cancer. Breast J. 2010, 16, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Mann, R.M.; Cho, N.; Moy, L. Breast MRI: State of the art. Radiology 2019, 292, 520–536. [Google Scholar] [CrossRef] [PubMed]
- Militello, C.; Rundo, L.; Dimarco, M.; Orlando, A.; Conti, V.; Woitek, R.; D’Angelo, I.; Bartolotta, T.V.; Russo, G. Semi-automated and interactive segmentation of contrast-enhancing masses on breast DCE-MRI using spatial fuzzy clustering. Biomed. Signal Process. Control 2022, 71, 12. [Google Scholar] [CrossRef]
- Vogel, W.V.; Nestle, U.; Valli, M.C. Pet/MRI in breast cancer. Clin. Transl. Imaging 2017, 5, 71–78. [Google Scholar] [CrossRef]
- Heacock, L.; Reig, B.; Lewin, A.A.; Toth, H.K.; Moy, L.; Lee, C.S. Abbreviated breast MRI: Road to clinical implementation. J. Breast Imaging 2020, 2, 201–214. [Google Scholar] [CrossRef]
- Mann, R.M.; Kuhl, C.K.; Moy, L. Contrast-enhanced MRI for breast cancer screening. J. Magn. Reson. Imaging 2019, 50, 377–390. [Google Scholar] [CrossRef]
- van Zandwijk, N. Chemoprevention of lung cancer. Lung Cancer 2001, 34, S91–S94. [Google Scholar] [CrossRef]
- Hintze, C.; Dinkel, J.; Biederer, J.; Heussel, C.P.; Puderbach, M. New procedures—Comprehensive staging of lung cancer with MRI. Radiologe 2010, 50, 699–705. [Google Scholar] [CrossRef]
- Khalil, A.; Bouhela, T.; Carette, M.F. Contribution of MRI in lung cancer staging. JBR-BTR 2013, 96, 132–141. [Google Scholar] [CrossRef][Green Version]
- Biederer, J.; Ohno, Y.; Hatabu, H.; Schiebler, M.L.; van Beek, E.J.R.; Vogel-Claussen, J.; Kauczor, H.U. Screening for lung cancer: Does MRI have a role? Eur. J. Radiol. 2017, 86, 353–360. [Google Scholar] [CrossRef]
- Meier-Schroers, M.; Homsi, R.; Schild, H.H.; Thomas, D. Lung cancer screening with MRI: Characterization of nodules with different non-enhanced MRI sequences. Acta Radiol. 2019, 60, 168–176. [Google Scholar] [CrossRef]
- Meier-Schroers, M.; Homsi, R.; Skowasch, D.; Buermann, J.; Zipfel, M.; Schild, H.H.; Thomas, D. Lung cancer screening with MRI: Results of the first screening round. J. Cancer Res. Clin. Oncol. 2018, 144, 117–125. [Google Scholar] [CrossRef]
- Cobben, D.C.P.; de Boer, H.C.J.; Tijssen, R.H.; Rutten, E.; van Vulpen, M.; Peerlings, J.; Troost, E.G.C.; Hoffmann, A.L.; van Lier, A. Emerging role of MRI for radiation treatment planning in lung cancer. Technol. Cancer Res. Treat. 2016, 15, NP47–NP60. [Google Scholar] [CrossRef]
- Weller, A.; Papoutsaki, M.V.; Waterton, J.C.; Chiti, A.; Stroobants, S.; Kuijer, J.; Blackledge, M.; Morgan, V.; deSouza, N.M. Diffusion-weighted (dw) MRI in lung cancers: Adc test-retest repeatability. Eur. Radiol. 2017, 27, 4552–4562. [Google Scholar] [CrossRef]
- Kim, H.S.; Lee, K.S.; Ohno, Y.; van Beek, E.J.R.; Biederer, J.; Int Workshop Pulm, F. PET/CT versus MRI for diagnosis, staging, and follow-up of lung cancer. J. Magn. Reson. Imaging 2015, 42, 247–260. [Google Scholar] [CrossRef]
- Ohno, Y.; Koyama, H.; Lee, H.Y.; Yoshikawa, T.; Sugimura, K. Magnetic resonance imaging (MRI) and positron emission tomography (PET)/MRI for lung cancer staging. J. Thorac. Imaging 2016, 31, 215–227. [Google Scholar] [CrossRef]
- Pasechnikov, V.; Chukov, S.; Fedorov, E.; Kikuste, I.; Leja, M. Gastric cancer: Prevention, screening and early diagnosis. World J. Gastroenterol. 2014, 20, 13842–13862. [Google Scholar] [CrossRef]
- Renzulli, M.; Clemente, A.; Spinelli, D.; Ierardi, A.M.; Marasco, G.; Farina, D.; Brocchi, S.; Ravaioli, M.; Pettinari, I.; Cescon, M.; et al. Gastric cancer staging: Is it time for magnetic resonance imaging? Cancers 2020, 12, 1402. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Yu, J.C. The role of MRI in the diagnosis and treatment of gastric cancer. Diagn. Interv. Radiol. 2020, 26, 176–182. [Google Scholar] [CrossRef]
- De Vuysere, S.; Vandecaveye, V.; De Bruecker, Y.; Carton, S.; Vermeiren, K.; Tollens, T.; De Keyzer, F.; Dresen, R.C. Accuracy of whole-body diffusion-weighted MRI (wb-dwi/MRI) in diagnosis, staging and follow-up of gastric cancer, in comparison to CT: A pilot study. BMC Med. Imaging 2021, 21, 9. [Google Scholar] [CrossRef]
- Caivano, R.; Rabasco, P.; Lotumolo, A.; D’ Antuono, F.; Zandolino, A.; Villonio, A.; Macarini, L.; Guglielmi, G.; Salvatore, M.; Cammarota, A. Gastric cancer: The role of diffusion weighted imaging in the preoperative staging. Cancer Investig. 2014, 32, 184–190. [Google Scholar] [CrossRef]
- Hasbahceci, M.; Akcakaya, A.; Memmi, N.; Turkmen, I.; Cipe, G.; Yildiz, P.; Arici, D.S.; Muslumanoglu, M. Diffusion MRI on lymph node staging of gastric adenocarcinoma. Quant. Imaging Med. Surg. 2015, 5, 392–400. [Google Scholar]
- Takahashi, H.; Yano, H.; Matsushita, M.; Monden, T.; Kinuta, M.; Tateishi, H.; Nakano, Y.; Matsui, S.; Iwazawa, T.; Kanoh, T.; et al. Preoperative staging of gastric cancer: Diagnosing the depth of invasion in the gastric wall with MRI. In Proceedings of the 3rd International Gastric Cancer Congress, Seoul, Korea, 27–30 April 1999; pp. 411–414. [Google Scholar]
- Tokuhara, T.; Tanigawa, N.; Matsuki, M.; Nomura, E.; Mabuchi, H.; Lee, S.W.; Tatsumi, Y.; Nishimura, H.; Yoshinaka, R.; Kurisu, Y.; et al. Evaluation of lymph node metastases in gastric cancer using magnetic resonance imaging with ultrasmall superparamagnetic iron oxide (uspio): Diagnostic performance in post-contrast images using new diagnostic criteria. Gastric Cancer 2008, 11, 194–200. [Google Scholar] [CrossRef]
- Arocena, M.G.; Barturen, A.; Bujanda, L.; Casado, O.; Ramirez, M.M.; Oleagoitia, J.M.; Iturri, M.G.; Mugica, P.; Cosme, A.; Gutierrez-Stampa, M.A.; et al. MRI and endoscopic ultrasonography in the staging of gastric cancer. Rev. Esp. Enferm. Dig. 2006, 98, 582–586. [Google Scholar] [CrossRef][Green Version]
- Malaj, A.; Bilaj, F.; Shahini, A.; Miraka, M. Ct/MRI accuracy in detecting and determining preoperative stage of gastric adenocarcinoma in albania. Wspolczesna Onkol. 2017, 21, 168–173. [Google Scholar] [CrossRef]
- Heye, T.; Kuntz, C.; Dux, M.; Encke, J.; Palmowski, M.; Autschbach, F.; Volke, F.; Kauffmann, G.W.; Grenacher, L. Ct and endoscopic ultrasound in comparison to endoluminal MRI-preliminary results in staging gastric carcinoma. Eur. J. Radiol. 2009, 70, 336–341. [Google Scholar] [CrossRef]
- Kim, A.Y.; Han, J.K.; Seong, C.K.; Kim, T.K.; Choi, B.I. MRI in staging advanced gastric cancer: Is it useful compared with spiral CT? J. Comput. Assist. Tomogr. 2000, 24, 389–394. [Google Scholar] [CrossRef]
- Maccioni, F.; Marcelli, G.; Al Ansari, N.; Zippi, M.; De Marco, V.; Kagarmanova, A.; Vestri, A.; Marcheg-giano-Clarke, L.; Marini, M. Preoperative t and n staging of gastric cancer: Magnetic resonance imaging (MRI) versus multi detector computed tomography (MDCT). Clin. Ter. 2010, 161, e57–e62. [Google Scholar]
- Ma, L.; Xu, X.W.; Zhang, M.; Zheng, S.Q.; Zhang, B.; Zhang, W.; Wang, P.J. Dynamic contrast-enhanced MRI of gastric cancer: Correlations of the pharmacokinetic parameters with histological type, lauren classification, and angiogenesis. Magn. Reson. Imaging 2017, 37, 27–32. [Google Scholar] [CrossRef]
- Joo, I.; Lee, J.M.; Han, J.K.; Yang, H.K.; Lee, H.J.; Choi, B.I. Dynamic contrast-enhanced MRI of gastric cancer: Correlation of the perfusion parameters with pathological prognostic factors. J. Magn. Reson. Imaging 2015, 41, 1608–1614. [Google Scholar] [CrossRef]
- Bae, S.W.; Berlth, F.; Jeong, K.Y.; Suh, Y.S.; Kong, S.H.; Lee, H.J.; Kim, W.H.; Chung, J.K.; Yang, H.K. Establishment of a 18F FDG-PET/MRI imaging protocol for gastric cancer pdx as a preclinical research tool. J. Gasric Cancer 2020, 20, 60–71. [Google Scholar] [CrossRef]
- Lee, D.H.; Kim, S.H.; Joo, I.; Hur, B.Y.; Han, J.K. Comparison between 18F-FDG PET/MRI and mdct for the assessment of preoperative staging and resectability of gastric cancer. Eur. J. Radiol. 2016, 85, 1085–1091. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, D.; Liu, J.J.; Cui, J.X.; Xi, H.Q.; Zhang, K.C.; Huang, X.H.; Wei, B.; Wang, X.X.; Xu, B.X.; et al. Comparing PET/MRI with PET/CT for pretreatment staging of gastric cancer. Gastroenterol. Res. Pract. 2019, 2019, 9564627. [Google Scholar] [CrossRef]
- Yuan, Y.; Chen, L.G.; Ren, S.N.; Wang, Z.; Chen, Y.K.; Jin, A.G.; Li, S.; Fang, X.; Wang, T.G.; Bian, Y.; et al. Diagnostic performance in t staging for patients with esophagogastric junction cancer using high-resolution MRI: A comparison with conventional MRI at 3 tesla. Cancer Imaging 2019, 19, 9. [Google Scholar] [CrossRef]
- Pang, L.; Wang, J.; Fan, Y.; Xu, R.; Bai, Y.P.; Bai, L.C. Correlations of tnm staging and lymph node metastasis of gastric cancer with MRI features and vegf expression. Cancer Biomark. 2018, 23, 53–59. [Google Scholar] [CrossRef]
- Giganti, F.; Orsenigo, E.; Arcidiacono, P.G.; Nicoletti, R.; Albarello, L.; Ambrosi, A.; Salerno, A.; Esposito, A.; Petrone, M.C.; Chiari, D.; et al. Preoperative locoregional staging of gastric cancer: Is there a place for magnetic resonance imaging? Prospective comparison with eus and multidetector computed tomography. Gastric Cancer 2016, 19, 216–225. [Google Scholar] [CrossRef]
- Garrido-Laguna, I.; Hidalgo, M. Pancreatic cancer: From state-of-the-art treatments to promising novel therapies. Nat. Rev. Clin. Oncol. 2015, 12, 319–334. [Google Scholar] [CrossRef]
- Barnes, C.A.; Krzywda, E.; Lahiff, S.; McDowell, D.; Christians, K.K.; Knechtges, P.; Tolat, P.; Hohenwalter, M.; Dua, K.; Khan, A.H.; et al. Development of a high risk pancreatic screening clinic using 3.0 t MRI. Fam. Cancer 2018, 17, 101–111. [Google Scholar] [CrossRef]
- Gaa, J.; Fingerle, A.A.; Holzapfel, K.; Rummeny, E.J. MRI for malignant pancreatic tumors. Radiologe 2009, 49, 124–130. [Google Scholar] [CrossRef]
- Ueno, M.; Niwa, T.; Ohkawa, S.; Amano, A.; Masaki, T.; Miyakawa, K.; Yoshida, T. The usefulness of perfusion-weighted magnetic resonance imaging in advanced pancreatic cancer. Pancreas 2009, 38, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Gassert, F.G.; Ziegelmayer, S.; Luitjens, J.; Gassert, F.T.; Tollens, F.; Rink, J.; Makowski, M.R.; Rubenthaler, J.; Froelich, M.F. Additional MRI for initial m-staging in pancreatic cancer: A cost-effectiveness analysis. Eur. Radiol. 2021, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Visser, B.C.; Yeh, B.M.; Qayyum, A.; Way, L.W.; McCulloch, C.E.; Coakley, F.V. Characterization of cystic pancreatic masses: Relative accuracy of CT and MRI. Am. J. Roentgenol. 2007, 189, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.M.; Ni, J.M.; Zhang, Z.Y.; Zhang, L.; Li, B.; Jiang, C.J. Presurgical evaluation of pancreatic cancer: A comprehensive imaging comparison of CT versus MRI. Am. J. Roentgenol. 2016, 206, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Ming, B.; Wu, J.L.; Zhou, T.; Zhang, S.Y.; Chen, Y.; Lan, C.; Zhang, X.M. Magnetic resonance imaging for preoperative staging of pancreatic cancer based on the 8th edition of ajcc guidelines. J. Gastrointest. Oncol. 2020, 11, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Litjens, G.; Riviere, D.M.; van Geenen, E.J.M.; Radema, S.A.; Brosens, L.A.A.; Prokop, M.; van Laarhoven, C.; Hermans, J.J. Diagnostic accuracy of contrast-enhanced diffusion-weighted MRI for liver metastases of pancreatic cancer: Towards adequate staging and follow-up of pancreatic cancer—DIA-PANC study: Study protocol for an international, multicenter, diagnostic trial. BMC Cancer 2020, 20, 8. [Google Scholar] [CrossRef]
- Jhaveri, K.S.; Jandaghi, A.B.; Thipphavong, S.; Espin-Garcia, O.; Dodd, A.; Hutchinson, S.; Reichman, T.W.; Moulton, C.A.; McGilvary, I.D.; Gallinger, S. Can preoperative liver MRI with gadoxetic acid help reduce open-close laparotomies for curative intent pancreatic cancer surgery? Cancer Imaging 2021, 21, 12. [Google Scholar] [CrossRef]
- Riviere, D.M.; van Geenen, E.J.M.; van der Kolk, B.M.; Nagtegaal, I.D.; Radema, S.A.; van Laarhoven, C.; Hermans, J.J. Improving preoperative detection of synchronous liver metastases in pancreatic cancer with combined contrast-enhanced and diffusion-weighted MRI. Abdom. Radiol. 2019, 44, 1756–1765. [Google Scholar] [CrossRef]
- Yang, M.; Hopp, A.C.; Bekaii-Saab, T.S.; Collins, J.M. Sister mary joseph nodule in advanced pancreatic adenocarcinoma identified on 18F-FDG PET/MRI. J. Nucl. Med. Technol. 2019, 47, 341–342. [Google Scholar] [CrossRef]
- Sandrasegaran, K.; Nutakki, K.; Tahir, B.; Dhanabal, A.; Tann, M.; Cote, G.A. Use of diffusion-weighted MRI to differentiate chronic pancreatitis from pancreatic cancer. Am. J. Roentgenol. 2013, 201, 1002–1008. [Google Scholar] [CrossRef]
- Kim, J.K.; Altun, E.; Elias, J.; Pamuklar, E.; Rivero, H.; Semelka, R.C. Focal pancreatic mass: Distinction of pancreatic cancer from chronic pancreatitis using gadolinium-enhanced 3d-gradient-echo MRI. J. Magn. Reson. Imaging 2007, 26, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.A.; Fu, W.W.; Liu, Z.J.; Liu, M.; Ren, R.M.; Zhai, H.X.; Li, C.L. MRI-guided celiac plexus neurolysis for pancreatic cancer pain: Efficacy and safety. J. Magn. Reson. Imaging 2016, 44, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T.; Ueno, M.; Shinya, N.; Gotoh, T.; Kwee, T.C.; Takahara, T.; Yoshida, T.; Ohkawa, S.; Doiuchi, T.; Inoue, T. Dynamic susceptibility contrast MRI in advanced pancreatic cancer: Semi-automated analysis to predict response to chemotherapy. NMR Biomed. 2010, 23, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, F.; Meng, Y.F.; Wang, H.; Le, T.; Wei, B.J.; Lee, D.; Willis, P.; Shen, B.Z.; Yang, X.M. Diffusion-weighted MRI monitoring of pancreatic cancer response to radiofrequency heat-enhanced intratumor chemotherapy. NMR Biomed. 2013, 26, 1762–1767. [Google Scholar] [CrossRef]
- Grandhi, M.S.; Kim, A.K.; Ronnekleiv-Kelly, S.M.; Kamel, I.R.; Ghasebeh, M.A.; Pawlik, T.M. Hepatocellular carcinoma: From diagnosis to treatment. Surg. Oncol. 2016, 25, 74–85. [Google Scholar] [CrossRef]
- Wang, G.B.; Zhu, S.C.; Li, X.K. Comparison of values of CT and MRI imaging in the diagnosis of hepatocellular carcinoma and analysis of prognostic factors. Oncol. Lett. 2019, 17, 1184–1188. [Google Scholar] [CrossRef]
- Kloeckner, R.; dos Santos, D.P.; Kreitner, K.F.; Leicher-Duber, A.; Weinmann, A.; Mittler, J.; Duber, C. Quantitative assessment of washout in hepatocellular carcinoma using MRI. BMC Cancer 2016, 16, 8. [Google Scholar] [CrossRef][Green Version]
- Sanghvi, T.; Boyum, J.; Spilseth, B.; Schat, R.; Estby, H.; Taylor, A. MRI for hepatocellular carcinoma: A primer for magnetic resonance imaging interpretation. Abdom. Radiol. 2018, 43, 1143–1151. [Google Scholar] [CrossRef]
- Hectors, S.J.; Lewis, S.; Besa, C.; King, M.J.; Said, D.; Putra, J.; Ward, S.; Higashi, T.; Thung, S.; Yao, S.; et al. MRI radiomics features predict immuno-oncological characteristics of hepatocellular carcinoma. Eur. Radiol. 2020, 30, 3759–3769. [Google Scholar] [CrossRef]
- Low, H.M.; Choi, J.Y.; Tan, C.H. Pathological variants of hepatocellular carcinoma on MRI: Emphasis on histopathologic correlation. Abdom. Radiol. 2019, 44, 493–508. [Google Scholar] [CrossRef]
- Kim, Y.N.; Song, J.S.; Moon, W.S.; Hwang, H.P.; Kim, Y.K. Intra-individual comparison of hepatocellular carcinoma imaging computed tomconstrast-enhancedtte computed tomography, gadopentetate dimeglumine-enhanced MRI, and gadoxetic acid-enhanced MRI. Acta Radiol. 2018, 59, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Gabr, A.E.; Mikhael, H.S.W.; El-Maadawy, S.M. Comparison between subtraction and dynamic MRI in assessing treatment response following radiofrequency ablation in patients with hepatocellular carcinoma. Egypt. J. Radiol. Nucl. Med. 2021, 52, 10. [Google Scholar] [CrossRef]
- Jha, R.C.; Zanello, P.A.; Nguyen, X.M.; Pehlivanova, M.; Johnson, L.B.; Fishbein, T.; Shetty, K. Small hepatocellular carcinoma: MRI findings for predicting tumor growth rates. Acad. Radiol. 2014, 21, 1455–1464. [Google Scholar] [PubMed]
- Abdullah, S.S.; Pialat, J.B.; Wiart, M.; Duboeuf, F.; Mabrut, J.Y.; Bancel, B.; Rode, A.; Ducerf, C.; Baulieux, J.; Berthezene, Y. Characterization of hepatocellular carcinoma and colorectal liver metastasis by means of perfusion MRI. J. Magn. Reson. Imaging 2008, 28, 390–395. [Google Scholar] [CrossRef]
- Gong, X.Q.; Tao, Y.Y.; Wu, Y.K.; Liu, N.; Yu, X.; Wang, R.; Zheng, J.; Liu, N.; Huang, X.H.; Li, J.D.; et al. Progress of MRI radiomics in hepatocellular carcinoma. Front. Oncol. 2021, 11, 13. [Google Scholar] [CrossRef]
- Carbonell, G.; Kennedy, P.; Bane, O.; Kirmani, A.; El Homsi, M.; Stocker, D.; Said, D.; Mukherjee, P.; Gevaert, O.; Lewis, S.; et al. Precision of MRI radiomics features in the liver and hepatocellular carcinoma. Eur. Radiol. 2021, 1–11. [Google Scholar] [CrossRef]
- Kim, H.; Park, M.S.; Choi, J.Y.; Park, Y.N.; Kim, M.J.; Kim, K.S.; Choi, J.S.; Han, K.H.; Kim, E.; Kim, K.W. Can microvessel invasion of hepatocellular carcinoma be predicted by pre-operative MRI? Eur. Radiol. 2009, 19, 1744–1751. [Google Scholar] [CrossRef]
- Hennedige, T.; Venkatesh, S.K. Imaging of hepatocellular carcinoma: Diagnosis, staging and treatment monitoring. Cancer Imaging 2013, 12, 530–547. [Google Scholar] [CrossRef]
- Taneja, S.; Taneja, R.; Kashyap, V.; Jha, A.; Jena, A. Ga-68-psma uptake in hepatocellular carcinoma. Clin. Nucl. Med. 2017, 42, E69–E70. [Google Scholar] [CrossRef]
- Dondi, F.; Albano, D.; Cerudelli, E.; Gazzilli, M.; Giubbini, R.; Treglia, G.; Bertagna, F. Radiolabelled psma PET/CT or PET/MRI in hepatocellular carcinoma (HCC): A systematic review. Clin. Transl. Imaging 2020, 8, 461–467. [Google Scholar] [CrossRef]
- Qiu, Z.L.; Pan, Y.S.; Wei, J.; Wu, D.J.; Xia, Y.; Shen, D.G. Predicting symptoms from multiphasic MRI via multi-instance attention learning for hepatocellular carcinoma grading. In Proceedings of the International Conference on Medical Image Computing and Computer Assisted Intervention (MICCAI), Strasbourg, France, 27 September–1 October 2021; Springer: Cham, Switzerland, 2021; pp. 439–448. [Google Scholar]
- Saito, K.; Ledsam, J.; Sugimoto, K.; Sourbron, S.; Araki, Y.; Tokuuye, K. Dce-MRI for early prediction of response in hepatocellular carcinoma after tace and sorafenib therapy: A pilot study. J. Belg. Soc. Radiol. 2018, 102, 9. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.F.; Yu, Y.X.; Wang, X.M.; Hu, M.J.; Du, M.Z.; Guo, L.C.A.; Sun, S.F.; Hu, C.H. Texture analysis based on gd-eob-dtpa-enhanced MRI for identifying vessels encapsulating tumor clusters (vetc)-positive hepatocellular carcinoma. J. Hepatocell. Carcinoma 2021, 8, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Q.; Wang, X.; Zhao, D.W.; Sun, J.; Liu, J.J.; Lin, D.D.; Yang, G.; Liu, H.; Xia, Z.Y.; Jia, C.Y.; et al. Application of gd-eob-dtpa-enhanced magnetic resonance imaging (MRI) in hepatocellular carcinoma. World J. Surg. Oncol. 2020, 18, 219. [Google Scholar] [CrossRef] [PubMed]
- Hanna, R.F.; Kased, N.; Kwan, S.W.; Gamst, A.C.; Santosa, A.C.; Hassanein, T.; Sirlin, C.B. Double-contrast MRI for accurate staging of hepatocellular carcinoma in patients with cirrhosis. Am. J. Roentgenol. 2008, 190, 47–57. [Google Scholar] [CrossRef]
- Wu, Y.N.; Huang, L.N.; Li, B.S.; Li, H.G. The diagnostic value of gd-eob-dtpa-enhanced MRI scans in small hepatocellular carcinoma in patients with liver cirrhosis. Int. J. Clin. Exp. Med. 2020, 13, 6909–6915. [Google Scholar]
- Taouli, B.; Johnson, R.S.; Hajdu, C.H.; Oei, M.T.H.; Merad, M.; Yee, H.; Rusinek, H. Hepatocellular carcinoma: Perfusion quantification with dynamic contrast-enhanced MRI. Am. J. Roentgenol. 2013, 201, 795–800. [Google Scholar] [CrossRef]
- Akhtar, S.; Hussain, M.; Ali, S.; Maqsood, S.; Akram, S.; Abbas, N. Comparison of positive predictive value of multiphasic dynamic contrast enhanced MRI with dynamic contrast enhanced ct for the detection of hepatocellular carcinoma. Pak. J. Med Health Sci. 2020, 14, 562–564. [Google Scholar]
- Gluskin, J.S.; Chegai, F.; Monti, S.; Squillaci, E.; Mannelli, L. Hepatocellular carcinoma and diffusion-weighted MRI: Detection and evaluation of treatment response. J. Cancer 2016, 7, 1565–1570. [Google Scholar] [CrossRef]
- Chen, H.Y.; Hou, Y.L.; Ma, X.M.; Xie, H.Y.; Ye, M.; Bai, Y.R. Ct and MRI in target delineation in primary hepatocellular carcinoma. Cancer Radiother. 2013, 17, 750–754. [Google Scholar] [CrossRef]
- Zemour, J.; Marty, M.; Lapuyade, B.; Collet, D.; Chiche, L. Gallbladder tumor and pseudotumor: Diagnosis and management. J. Visc. Surg. 2014, 151, 289–300. [Google Scholar] [CrossRef]
- Tseng, J.H.; Wan, Y.L.; Hung, C.F.; Ng, K.K.; Pan, K.T.; Chou, A.S.B.; Liu, N.J. Diagnosis and staging of gallbladder carcinoma—Evaluation with dynamic MR imaging. Clin. Imaging 2002, 26, 177–182. [Google Scholar] [CrossRef]
- Vendrami, C.L.; Magnetta, M.J.; Mittal, P.K.; Moreno, C.C.; Miller, F.H. Gallbladder carcinoma and its differential diagnosis at MRI: What radiologists should know. Radiographics 2021, 41, 78–95. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.S.; Zou, X.L.; Wang, Q.X.; Hu, D.Y.; Li, Z.; Xu, C.O. Gallbladder carcinoma: An initial clinical experience of reduced field-of-view diffusion-weighted MRI. Cancer Imaging 2020, 20, 8. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, H.; Hoogwater, F.J.H.; Holtman, G.A.; van der Hoorn, A.; de Boer, M.T.; de Haas, R.J. Clinical value of diffusion-weighted MRI for differentiation between benign and malignant gallbladder disease: A systematic review and meta-analysis. Acta Radiol. 2021, 62, 987–996. [Google Scholar] [CrossRef]
- Corgna, E.; Betti, M.; Gatta, G.; Roila, F.; De Mulder, P.H.M. Renal cancer. Crit. Rev. Oncol./Hematol. 2007, 64, 247–261. [Google Scholar] [CrossRef]
- Bensalah, K.; Albiges, L.; Bernhard, J.C.; Bigot, P.; Bodin, T.; Boissier, R.; Correas, J.M.; Gimel, P.; Hetet, J.F.; Long, J.A.; et al. French ccafu guidelines—Update 2018–2020: Management of kidney cancer. Prog. Urol. 2018, 28, R5–R33. [Google Scholar] [CrossRef]
- Couvidat, C.; Eiss, D.; Verkarre, V.; Merran, S.; Correas, J.M.; Mejean, A.; Helenon, O. Renal papillary carcinoma: CT and MRI features. Diagn. Interv. Imaging 2014, 95, 1055–1063. [Google Scholar] [CrossRef]
- Shinagare, A.B.; Davenport, M.S.; Park, H.; Pedrosa, I.; Remer, E.M.; Chandarana, H.; Doshi, A.M.; Schieda, N.; Smith, A.D.; Vikram, R.; et al. Lexicon for renal mass terms at CT and MRI: A consensus of the society of abdominal radiology disease-focused panel on renal cell carcinoma. Abdom. Radiol. 2021, 46, 703–722. [Google Scholar] [CrossRef]
- Vannier, M.W. Imaging Renal Carcinoma; Demos Medical Publications: New York, NY, USA, 2011; Volume 2, pp. 37–56. [Google Scholar]
- Chiarello, M.A.; Mali, R.D.; Kang, S.K. Diagnostic accuracy of MRI for detection of papillary renal cell carcinoma: A systematic review and meta-analysis. Am. J. Roentgenol. 2018, 211, 812–821. [Google Scholar] [CrossRef]
- Wehrli, N.E.; Kim, M.J.; Matza, B.W.; Melamed, J.; Taneja, S.S.; Rosenkrantz, A.B. Utility of MRI features in differentiation of central renal cell carcinoma and renal pelvic urothelial carcinoma. Am. J. Roentgenol. 2013, 201, 1260–1267. [Google Scholar] [CrossRef]
- Palmowski, M.; Schifferdecker, I.; Zwick, S.; Macher-Goeppinger, S.; Laue, H.; Haferkamp, A.; Kauczor, H.U.; Kiessling, F.; Hallscheidt, P. Tumor perfusion assessed by dynamic contrast-enhanced MRI correlates to the grading of renal cell carcinoma: Initial results. Eur. J. Radiol. 2010, 74, e177–e181. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Wang, H.; Li, H.F.; Rao, Y.W.; Liu, J.H.; Tian, S.F.; Ju, Y.; Li, Y.; Chen, A.L.; Chen, L.H.; et al. Diagnostic significance of diffusion-weighted MRI in renal cancer. BioMed Res. Int. 2015, 2015, 172165. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef]
- Saito, W.; Amanuma, M.; Tanaka, J.; Heshiki, A. Histopathological analysis of a bladder cancer stalk observed on MRI. Magn. Reson. Imaging 2000, 18, 411–415. [Google Scholar] [CrossRef]
- Tillou, X.; Grardel, E.; Fourmarier, M.; Bernasconi, T.; Demailly, M.; Hakami, F.; Saint, F.; Petit, J. Can MRI be used to distinguish between superficial and invasive transitional cell bladder cancer? Prog. Urol. 2008, 18, 440–444. [Google Scholar] [CrossRef]
- Dolz, J.; Xu, X.P.; Rony, J.; Yuan, J.; Liu, Y.; Granger, E.; Desrosiers, C.; Zhang, X.; Ben Ayed, I.; Lu, H.B. Multiregion segmentation of bladder cancer structures in MRI with progressive dilated convolutional networks. Med. Phys. 2018, 45, 5482–5493. [Google Scholar] [CrossRef] [PubMed]
- Barentsz, J.O.; Jager, G.J.; Witjes, J.A.; Ruijs, J.H.J. Primary staging of urinary bladder carcinoma: The role of MRI and a comparison with ct. Eur. Radiol. 1996, 6, 129–133. [Google Scholar] [CrossRef]
- Mehraj, A.; Hameed, A.; Nanda, S.; Wazir, B.S.; Khan, M.A. Role of MRI in staging urinary bladder tumors. Ann. Med. Health Sci. Res. 2017, 7, 116–121. [Google Scholar]
- Abouelkheir, R.T.; Abdelhamid, A.; Abou El-Ghar, M.; El-Diasty, T. Imaging of bladder cancer: Standard applications and future trends. Med. Lith. 2021, 57, 220. [Google Scholar] [CrossRef]
- Wong, V.C.K.; Ganeshan, D.; Jensen, C.T.; Devine, C.E. Imaging and management of bladder cancer. Cancers 2021, 13, 1396. [Google Scholar] [CrossRef]
- Woo, S.; Suh, C.H.; Kim, S.Y.; Cho, J.Y.; Kim, S.H. Diagnostic performance of MRI for prediction of muscle-invasiveness of bladder cancer: A systematic review and meta-analysis. Eur. J. Radiol. 2017, 95, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Ghafoori, M.; Shakiba, M.; Ghiasi, A.; Asvadi, N.; Hosseini, K.; Alavi, M. Value of MRI in local staging of bladder cancer. Urol. J. 2013, 10, 866–872. [Google Scholar]
- Rosenkrantz, A.B.; Mussi, T.C.; Melamed, J.; Taneja, S.S.; Huang, W.C. Bladder cancer: Utility of MRI in detection of occult muscle-invasive disease. Acta Radiol. 2012, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Badawy, M.; Farg, H.; Gadelhak, B.; ElGhar, M.A.; Sadeq, A.G.; Borg, M. Diagnostic performance of 3-tesla multiparametric MRI for assessment of the bladder cancer t stage and histologic grade. Egypt. J. Radiol. Nucl. Med. 2020, 51, 11. [Google Scholar] [CrossRef]
- Juri, H.; Narumi, Y.; Panebianco, V.; Osuga, K. Staging of bladder cancer with multiparametric MRI. Br. J. Radiol. 2020, 93, 7. [Google Scholar] [CrossRef] [PubMed]
- Naish, J.H.; McGrath, D.M.; Bains, L.J.; Passera, K.; Roberts, C.; Watson, Y.; Cheung, S.; Taylor, M.B.; Logue, J.P.; Buckley, D.L.; et al. Comparison of dynamic contrast-enhanced MRI and dynamic contrast-enhanced CT biomarkers in bladder cancer. Magn. Reson. Med. 2011, 66, 219–226. [Google Scholar] [CrossRef]
- Hijab, A.; Tocco, B.; Hanson, I.; Meijer, H.; Nyborg, C.J.; Bertelsen, A.S.; Smeenk, R.J.; Smith, G.; Michalski, J.; Baumann, B.C.; et al. Mr-guided adaptive radiotherapy for bladder cancer. Front. Oncol. 2021, 11, 12. [Google Scholar] [CrossRef]
- Wollin, D.A.; Deng, F.M.; Huang, W.C.; Babb, J.S.; Rosenkrantz, A.B. Conventional and diffusion-weighted MRI features in diagnosis of metastatic lymphadenopathy in bladder cancer. Can. J. Urol. 2014, 21, 7454–7459. [Google Scholar]
- Watanabe, H.; Kanematsu, M.; Kondo, H.; Goshima, S.; Tsuge, Y.; Onozuka, M.; Moriyama, N. Preoperative t staging of urinary bladder cancer: Does diffusion-weighted MRI have supplementary value? Am. J. Roentgenol. 2009, 192, 1361–1366. [Google Scholar] [CrossRef]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Miller, D.; Bishop, K.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; et al. Seer Cancer Statistics Review, 1975–2014, April 2017 ed.; National Cancer Institute: Bethesda, MD, USA, 2017.
- Sharma, S.K.; Nemieboka, B.; Sala, E.; Lewis, J.S.; Zeglis, B.M. Molecular imaging of ovarian cancer. J. Nucl. Med. 2016, 57, 827–833. [Google Scholar] [CrossRef]
- Engbersen, M.P.; van’ t Sant, I.; Lok, C.; Lambregts, D.M.J.; Sonke, G.S.; Beets-Tan, R.G.H.; van Driel, W.J.; Lahaye, M.J. MRI with diffusion-weighted imaging to predict feasibility of complete cytoreduction with the peritoneal cancer index (PCI) in advanced stage ovarian cancer patients. Eur. J. Radiol. 2019, 114, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Michielsen, K.; Dresen, R.; Vanslembrouck, R.; De Keyzer, F.; Amant, F.; Mussen, E.; Leunen, K.; Berteloot, P.; Moerman, P.; Vergote, I.; et al. Diagnostic value of whole body diffusion-weighted MRI compared to computed tomography for pre-operative assessment of patients suspected for ovarian cancer. Eur. J. Cancer 2017, 83, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Rockall, A.G. Diffusion weighted MRI in ovarian cancer. Curr. Opin. Oncol. 2014, 26, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.S.; Koopmeiners, J.S.; Kuehn-Hajder, J.E.; Metzger, G.J.; Lakkadi, N.; Downs, L.S.; Bolan, P.J. Quantitative multiparametric MRI of ovarian cancer. J. Magn. Reson. Imaging 2013, 38, 1501–1509. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, M.S.; Petersen, L.K.; Blaakaer, J.; Marinovskij, E.; Rosenkilde, M.; Andersen, G.; Bouchelouche, K.; Iversen, L.H. Assessment of peritoneal metastases with DW-MRI, CT, and FDG PET/CT before cytoreductive surgery for advanced stage epithelial ovarian cancer. Eur. J. Surg. Oncol. 2021, 47, 2134–2141. [Google Scholar] [CrossRef]
- Khiewvan, B.; Torigian, D.A.; Emamzadehfard, S.; Paydary, K.; Salavati, A.; Houshmand, S.; Werner, T.J.; Alavi, A. An update on the role of PET/CT and PET/MRI in ovarian cancer. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Tsuyoshi, H.; Tsujikawa, T.; Yamada, S.; Okazawa, H.; Yoshida, Y. Diagnostic value of [18F]FDG PET/MRI for staging in patients with ovarian cancer. EJNMMI Res. 2020, 10, 117. [Google Scholar] [CrossRef]
- Kim, C.K.; Park, B.K.; Choi, J.Y.; Kim, B.G.; Han, H. Detection of recurrent ovarian cancer at MRI: Comparison with integrated PET/CT. J. Comput. Assist. Tomogr. 2007, 31, 868–875. [Google Scholar] [CrossRef]
- Schmidt, S.; Meuli, R.A.; Achtari, C.; Prior, J.O. Peritoneal carcinomatosis in primary ovarian cancer staging comparison between mdct, MRI, and 18F-FDG PET/CT. Clin. Nucl. Med. 2015, 40, 371–377. [Google Scholar] [CrossRef]
- Sanli, Y.; Turkmen, C.; Bakir, B.; Iyibozkurt, C.; Ozel, S.; Has, D.; Yilmaz, E.; Topuz, S.; Yavuz, E.; Unal, S.N.; et al. Diagnostic value of PET/CT is similar to that of conventional MRI and even better for detecting small peritoneal implants in patients with recurrent ovarian cancer. Nucl. Med. Commun. 2012, 33, 509–515. [Google Scholar] [CrossRef]
- Nam, E.J.; Yun, M.J.; Oh, Y.T.; Kim, J.W.; Kim, J.H.; Kim, S.; Jung, Y.W.; Kim, S.W.; Kim, Y.T. Diagnosis and staging of primary ovarian cancer: Correlation between PET/CT, doppler us, and CT or MRI. Gynecol. Oncol. 2010, 116, 389–394. [Google Scholar] [PubMed]
- Reinhold, C.; Rockall, A.; Sadowski, E.A.; Siegelman, E.S.; Maturen, K.E.; Vargas, H.A.; Forstner, R.; Glanc, P.; Andreotti, R.F.; Thomassin-Naggara, I. Ovarian-adnexal reporting lexicon for MRI: A white paper of the acr ovarian-adnexal reporting and data systems MRI committee. J. Am. Coll. Radiol. 2021, 18, 713–729. [Google Scholar] [CrossRef] [PubMed]
- Murakami, N.; Ando, K.; Murata, M.; Murata, K.; Ohno, T.; Aoshika, T.; Kato, S.; Okonogi, N.; Saito, A.I.; Kim, J.Y.; et al. Why not de-intensification for uterine cervical cancer? Gynecol. Oncol. 2021, 163, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Novellas, S.; Fournol, M.; Marcotte-Bloch, C.; Mondot, L.; Caramella, T.; Bongain, A.; Chevallier, P. Magnetic resonance staging of uterine cervix carcinoma. Feuill. Rad. 2008, 48, 147–154. [Google Scholar] [CrossRef]
- Bourgioti, C.; Chatoupis, K.; Moulopoulos, L.A. Current imaging strategies for the evaluation of uterine cervical cancer. World J. Radiol. 2016, 8, 342–354. [Google Scholar] [CrossRef]
- Theodore, C.; Levaillant, J.M.; Capmas, P.; Chabi, N.; Skalli, D.; Vienet-Legue, L.; Haddad, B.; Fernandez, H.; Touboul, C. MRI and ultrasound fusion imaging for cervical cancer. Anticancer Res. 2017, 37, 5079–5085. [Google Scholar]
- Marzouk, F.; Jalaguier-Coudray, A.; Villard-Mahjoub, R. Uterine cervical cancer: The new figo classification. Imaging Femme 2021, 31, 30–39. [Google Scholar]
- Kasuya, G.; Toita, T.; Furutani, K.; Kodaira, T.; Ohno, T.; Kaneyasu, Y.; Yoshimura, R.; Uno, T.; Yogi, A.; Ishikura, S.; et al. Distribution patterns of metastatic pelvic lymph nodes assessed by ct/MRI in patients with uterine cervical cancer. Radiat. Oncol. 2013, 8, 6. [Google Scholar] [CrossRef]
- Zhu, Y.X.; Shen, B.M.; Pei, X.; Liu, H.X.; Li, G.Y. Ct, MRI, and PET imaging features in cervical cancer staging and lymph node metastasis. Am. J. Transl. Res. 2021, 13, 10536–10544. [Google Scholar]
- Grueneisen, J.; Schaarschmidt, B.M.; Heubner, M.; Aktas, B.; Kinner, S.; Forsting, M.; Lauenstein, T.; Ruhlmann, V.; Umutlu, L. Integrated PET/MRI for whole-body staging of patients with primary cervical cancer: Preliminary results. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1814–1824. [Google Scholar] [CrossRef]
- Monteil, J.; Maubon, A.; Leobon, S.; Roux, S.; Marin, B.; Renaudie, J.; Genet, D.; Fermeaux, V.; Aubard, Y.; Tubiana-Mathieu, N. Lymph node assessment with 18F-FDG-PET and MRI in uterine cervical cancer. Anticancer Res. 2011, 31, 3865–3871. [Google Scholar] [PubMed]
- de Boer, P.; Adam, J.A.; Buist, M.R.; van de Vijver, M.J.; Rasch, C.R.; Stoker, J.; Bipat, S.; Stalpers, L.J.A. Role of MRI in detecting involvement of the uterine internal os in uterine cervical cancer: Systematic review of diagnostic test accuracy. Eur. J. Radiol. 2013, 82, E422–E428. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, K.; Suenaga, Y.; Ueno, Y.; Kanda, T.; Maeda, T.; Deguchi, M.; Ebina, Y.; Yamada, H.; Takahashi, S.; Sugimura, K. Fusion of PET and MRI for staging of uterine cervical cancer: Comparison with contrast-enhanced 18F-FDG PET/CT and pelvic MRI. Clin. Imaging 2014, 38, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Balleyguier, C.; Fournet, C.; Ben Hassen, W.; Zareski, E.; Morice, P.; Haie-Meder, C.; Uzan, C.; Gouy, S.; Duvillard, P.; Lhomme, C. Management of cervical cancer detected during pregnancy: Role of magnetic resonance imaging. Clin. Imaging 2013, 37, 70–76. [Google Scholar] [CrossRef]
- Surov, A.; Meyer, H.J.; Schob, S.; Hohn, A.K.; Bremicker, K.; Exner, M.; Stumpp, P.; Purz, S. Parameters of simultaneous 18F-FDG-PET/MRI predict tumor stage and several histopathological features in uterine cervical cancer. Oncotarget 2017, 8, 28285–28296. [Google Scholar] [CrossRef][Green Version]
- Lee, J.; Kim, C.K.; Gu, K.W.; Park, W. Value of blood oxygenation level-dependent MRI for predicting clinical outcomes in uterine cervical cancer treated with concurrent chemoradiotherapy. Eur. Radiol. 2019, 29, 6256–6265. [Google Scholar] [CrossRef]
- Saida, T.; Sakata, A.; Tanaka, Y.O.; Ochi, H.; Ishiguro, T.; Sakai, M.; Takahashi, H.; Satoh, T.; Minami, M. Clinical and MRI characteristics of uterine cervical adenocarcinoma: Its variants and mimics. Korean J. Radiol. 2019, 20, 14. [Google Scholar] [CrossRef]
- Steiner, A.; Narva, S.; Rinta-Kiikka, I.; Hietanen, S.; Hynninen, J.; Virtanen, J. Diagnostic efficiency of whole-body 18F-FDG PET/MRI, MRI alone, and suv and adc values in staging of primary uterine cervical cancer. Cancer Imaging 2021, 21, 11. [Google Scholar] [CrossRef]
- Ran, C.; Sun, J.; Qu, Y.H.; Long, N. Clinical value of MRI, serum scca, and ca125 levels in the diagnosis of lymph node metastasis and para-uterine infiltration in cervical cancer. World J. Surg. Oncol. 2021, 19, 11. [Google Scholar] [CrossRef]
- Park, B.K.; Kim, T.J. Useful MRI findings for minimally invasive surgery for early cervical cancer. Cancers 2021, 13, 4078. [Google Scholar] [CrossRef]
- Hauge, A.; Wegner, C.S.; Gaustad, J.V.; Simonsen, T.G.; Andersen, L.M.K.; Rofstad, E.K. Diffusion-weighted MRI-derived adc values reflect collagen i content in pdx models of uterine cervical cancer. Oncotarget 2017, 8, 105682–105691. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ye, Z.X.; Sun, H.R.; Bai, R.J. Clinical application of diffusion-weighted magnetic resonance imaging in uterine cervical cancer. Int. J. Gynecol. Cancer 2015, 25, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, H.D.; Bai, X.; Ye, Z.X.; Sun, H.R.; Bai, R.J.; Wang, D.H. Differentiation of metastatic from non-metastatic lymph nodes in patients with uterine cervical cancer using diffusion-weighted imaging. Gynecol. Oncol. 2011, 122, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bai, R.J.; Sun, H.R.; Liu, H.D.; Wang, D.H. Diffusion-weighted magnetic resonance imaging of uterine cervical cancer. J. Comput. Assist. Tomogr. 2009, 33, 858–862. [Google Scholar]
- Hallac, R.R.; Ding, Y.; Yuan, Q.; McColl, R.W.; Lea, J.; Sims, R.D.; Weatherall, P.T.; Mason, R.P. Oxygenation in cervical cancer and normal uterine cervix assessed using blood oxygenation level-dependent (bold) MRI at 3t. Nmr Biomed. 2012, 25, 1321–1330. [Google Scholar] [CrossRef]
- Ryan, A.J.; Susil, B.; Jobling, T.W.; Oehler, M.K. Endometrial cancer. Cell Tissue Res. 2005, 322, 53–61. [Google Scholar] [CrossRef]
- Soneji, N.D.; Bharwani, N.; Ferri, A.; Stewart, V.; Rockall, A. Pre-operative MRI staging of endometrial cancer in a multicentre cancer network: Can we match single centre study results? Eur. Radiol. 2018, 28, 4725–4734. [Google Scholar] [CrossRef]
- Taieb, S.; Rocourt, N.; Narducci, F.; Ceugnart, L. Endometrial cancer imaging. Bull. Cancer 2012, 99, 13–20. [Google Scholar]
- Nougaret, S.; Horta, M.; Sala, E.; Lakhman, Y.; Thomassin-Naggara, I.; Kido, A.; Masselli, G.; Bharwani, N.; Sadowski, E.; Ertmer, A.; et al. Endometrial cancer MRI staging: Updated guidelines of the european society of urogenital radiology. Eur. Radiol. 2019, 29, 792–805. [Google Scholar] [CrossRef]
- Meissnitzer, M.; Forstner, R. MRI of endometrium cancer—How we do it. Cancer Imaging 2016, 16, 9. [Google Scholar] [CrossRef]
- Ortoft, G.; Dueholm, M.; Mathiesen, O.; Hansen, E.S.; Lundorf, E.; Moller, C.; Marinovskij, E.; Petersen, L.K. Preoperative staging of endometrial cancer using tvs, MRI, and hysteroscopy. Acta Obstet. Gynecol. Scand. 2013, 92, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Sadowski, E.A.; Robbins, J.B.; Guite, K.; Patel-Lippmann, K.; del Rio, A.M.; Kushner, D.M.; Al-Niaimi, A. Preoperative pelvic MRI and serum cancer antigen-125: Selecting women with grade 1 endometrial cancer for lymphadenectomy. Am. J. Roentgenol. 2015, 205, W556–W564. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Terai, Y.; Ono, Y.J.; Fujiwara, S.; Tanaka, Y.; Sasaki, H.; Tsunetoh, S.; Kanemura, M.; Yamamoto, K.; Yamada, T.; et al. Preoperative MRI and intraoperative frozen section diagnosis of myometrial invasion in patients with endometrial cancer. Int. J. Gynecol. Cancer 2015, 25, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Abu Freij, M.; Saleh, H.; Rawlins, H.; Duncan, T.; Nieto, J. The use of MRI for selecting patients with endometrial cancer and significant co-morbidities for vaginal hysterectomy. Arch. Gynecol. Obstet. 2011, 283, 1097–1101. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shachar, I.; Vitellas, K.M.; Cohn, D.E. The role of MRI in the conservative management of endometrial cancer. Gynecol. Oncol. 2004, 93, 233–237. [Google Scholar] [CrossRef]
- Bakir, B.; Sanli, S.; Bakir, V.L.; Ayas, S.; Yildiz, S.O.; Iyibozkurt, A.C.; Kartal, M.G.; Yavuz, E. Role of diffusion weighted MRI in the differential diagnosis of endometrial cancer, polyp, hyperplasia, and physiological thickening. Clin. Imaging 2017, 41, 86–94. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, L.; Nuerlan, T. Application of diffusion-weighted MRI for endometrial cancer diagnosis. Eur. J. Gynaecol. Oncol. 2018, 39, 655–658. [Google Scholar]
- Ma, Q.L.; Wang, H.Y.; Tao, C.; Duan, F.; Li, X.L. Evaluation of 3.0t multimodal MRI in patients with endometrial cancer. Acta Med. Mediterr. 2021, 37, 1887–1892. [Google Scholar]
- Chen, J.Y.; Gu, H.L.; Fan, W.M.; Wang, Y.H.; Chen, S.; Chen, X.; Wang, Z.Q. MRI-based radiomic model for preoperative risk stratification in stage I endometrial cancer. J. Cancer 2021, 12, 726–734. [Google Scholar] [CrossRef]
- Fasmer, K.E.; Hodneland, E.; Dybvik, J.A.; Wagner-Larsen, K.; Trovik, J.; Salvesen, O.; Krakstad, C.; Haldorsen, I.H.S. Whole-volume tumor MRI radiomics for prognostic modeling in endometrial cancer. J. Magn. Reson. Imaging 2021, 53, 928–937. [Google Scholar] [CrossRef]
- Keles, D.K.; Evrimler, S.; Merd, N.; Erdemoglu, E. Endometrial cancer: The role of MRI quantitative assessment in preoperative staging and risk stratification. Acta Radiol 2021. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Y.; Fan, W.M.; Gu, H.L.; Zhang, W.; Liu, Y.T.; Wang, Y.J.; Pan, Z.C.; Wang, Z.Q. Preoperative MRI and immunohistochemical examination for the prediction of high-risk endometrial cancer. Gland Surg. 2021, 10, 2180–2191. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Cho, A.; Yun, M.; Kim, Y.T.; Kang, W. Comparison of FDG PET/CT and MRI in lymph node staging of endometrial cancer. Ann. Nucl. Med. 2016, 30, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Tsuyoshi, H.; Tsujikawa, T.; Yamada, S.; Okazawa, H.; Yoshida, Y. Diagnostic value of 18F-FDG PET/MRI for staging in patients with endometrial cancer. Cancer Imaging 2020, 20, 9. [Google Scholar] [CrossRef] [PubMed]
- Bezzi, C.; Zambella, E.; Ghezzo, S.; Fallanca, F.; Samanes Gajate, A.M.; Franchini, A.; Ironi, G.; Bergamini, A.; Monaco, L.; Evangelista, L.; et al. 18F-FDG PET/MRI in endometrial cancer: Systematic review and meta-analysis. Clin. Transl. Imaging 2021, 1–14. [Google Scholar] [CrossRef]
- Kitajima, K.; Suenaga, Y.; Ueno, Y.; Kanda, T.; Maeda, T.; Takahashi, S.; Ebina, Y.; Miyahara, Y.; Yamada, H.; Sugimura, K. Value of fusion of PET and MRI for staging of endometrial cancer: Comparison with 18F-FDG contrast-enhanced PET/CT and dynamic contrast-enhanced pelvic MRI. Eur. J. Radiol. 2013, 82, 1672–1676. [Google Scholar] [CrossRef]
- Bian, L.H.; Wang, M.; Gong, J.; Liu, H.H.; Wang, N.; Wen, N.; Fan, W.S.; Xu, B.X.; Wang, M.Y.; Ye, M.X.; et al. Comparison of integrated PET/MRI with PET/ct in evaluation of endometrial cancer: A retrospective analysis of 81 cases. PeerJ 2019, 7, 10. [Google Scholar] [CrossRef]
- Hama, Y.; Tate, E. Palliative MRI-guided intensity modulated radiation therapy (imrt) for locally recurrent endometrial cancer. Aktualni Gynekol. Porod. 2020, 12, 70–74. [Google Scholar]
- Whitaker, H.; Tam, J.O.; Connor, M.J.; Grey, A. Prostate cancer biology & genomics. Transl. Androl. Urol. 2020, 9, 1481–1491. [Google Scholar]
- Shukla-Dave, A.; Hricak, H. Role of MRI in prostate cancer detection. NMR Biomed. 2014, 27, 16–24. [Google Scholar] [CrossRef]
- Hotker, A.M.; Dappa, E.; Mazaheri, Y.; Ehdaie, B.; Zheng, J.T.; Capanu, M.; Hricak, H.; Akin, O. The influence of background signal intensity changes on cancer detection in prostate MRI. Am. J. Roentgenol. 2019, 212, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Ventrella, E.; Eusebi, L.; Carpagnano, F.A.; Bartelli, F.; Cormio, L.; Guglielmi, G. Multiparametric MRI of prostate cancer: Recent advances. Curr. Radiol. Rep. 2020, 8, 14. [Google Scholar] [CrossRef]
- Sun, Y.; Reynolds, H.M.; Parameswaran, B.; Wraith, D.; Finnegan, M.E.; Williams, S.; Haworth, A. Multiparametric MRI and radiomics in prostate cancer: A review. Australas. Phys. Eng. Sci. Med. 2019, 42, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Bora, G.S.; Kumar, R.; Jagannathan, N.R. Multiparametric (MP) MRI of prostate cancer. Prog. Nucl. Magn. Reson. Spectrosc. 2018, 105, 23–40. [Google Scholar] [CrossRef] [PubMed]
- Ghai, S.; Haider, M.A. Multiparametric-MRI in diagnosis of prostate cancer. Indian J. Urol. 2015, 31, 194–201. [Google Scholar] [CrossRef]
- Wu, R.C.; Lebastchi, A.H.; Hadaschik, B.A.; Emberton, M.; Moore, C.; Laguna, P.; Futterer, J.J.; George, A.K. Role of MRI for the detection of prostate cancer. World J. Urol. 2021, 39, 637–649. [Google Scholar] [CrossRef]
- Piert, M.; El Naqa, I.; Davenport, M.S.; Incerti, E.; Mapelli, P.; Picchio, M. Pet/MRI and prostate cancer. Clin. Transl. Imaging 2016, 4, 473–485. [Google Scholar] [CrossRef]
- Park, H.; Wood, D.; Hussain, H.; Meyer, C.R.; Shah, R.B.; Johnson, T.D.; Chenevert, T.; Piert, M. Introducing parametric fusion PET/MRI of primary prostate cancer. J. Nucl. Med. 2012, 53, 546–551. [Google Scholar] [CrossRef]
- Lindenberg, L.; Ahlman, M.; Turkbey, B.; Mena, E.; Choyke, P. Evaluation of prostate cancer with PET/MRI. J. Nucl. Med. 2016, 57, 111S–116S. [Google Scholar] [CrossRef][Green Version]
- Kreydin, E.I.; Barrisford, G.W.; Feldman, A.S.; Preston, M.A. Testicular cancer: What the radiologist needs to know. Am. J. Roentgenol. 2013, 200, 1215–1225. [Google Scholar] [CrossRef]
- Nestler, T.; Baunacke, M.; Drager, D.; von Landenberg, N.; Groeben, C.; Huber, J. Testicular cancer guideline adherence and patterns of care in germany: A nationwide survey. Eur. J. Cancer Care 2019, 28, 9. [Google Scholar] [CrossRef] [PubMed]
- Hale, G.R.; Teplitsky, S.; Truong, H.; Gold, S.A.; Bloom, J.B.; Agarwal, P.K. Lymph node imaging in testicular cancer. Transl. Androl. Urol. 2018, 7, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Dulal, M.; Lei, J.; Dou, S.W.; Zhu, S.C. Role of imaging in testicular cancer. Curr. Med. Imaging Rev. 2017, 13, 38–49. [Google Scholar] [CrossRef]
- Rud, E.; Langberg, C.W.; Baco, E.; Lauritzen, P.; Sandbaek, G. MRI in the follow-up of testicular cancer: Less is more. Anticancer Res. 2019, 39, 2963–2968. [Google Scholar] [CrossRef] [PubMed]
- Tsili, A.C.; Sofikitis, N.; Stiliara, E.; Argyropoulou, M.I. MRI of testicular malignancies. Abdom. Radiol. 2019, 44, 1070–1082. [Google Scholar] [CrossRef] [PubMed]
- Larsen, S.K.A.; Agerbaek, M.; Jurik, A.G.; Pedersen, E.M. Ten years of experience with MRI follow-up of testicular cancer stage i: A retrospective study and an MRI protocol with dwi. Acta Oncol. 2020, 59, 1374–1381. [Google Scholar] [CrossRef]
- Manganaro, L.; Saldari, M.; Pozza, C.; Vinci, V.; Gianfrilli, D.; Greco, E.; Franco, G.; Sergi, M.E.; Scialpi, M.; Catalano, C.; et al. Dynamic contrast-enhanced and diffusion-weighted mr imaging in the characterisation of small, non-palpable solid testicular tumours. Eur. Radiol. 2018, 28, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Laukka, M.; Mannisto, S.; Beule, A.; Kouri, M.; Blomqvist, C. Comparison between ct and MRI in detection of metastasis of the retroperitoneum in testicular germ cell tumors: A prospective trial. Acta Oncol. 2020, 59, 660–665. [Google Scholar] [CrossRef] [PubMed]
- Reinges, M.H.T.; Kaiser, W.A.; Miersch, W.D.; Vogel, J.; Reiser, M. Dynamic MRI of benign and malignant testicular lesions—Preliminary-observations. Eur. Radiol. 1995, 5, 615–622. [Google Scholar] [CrossRef]
- Rocher, L.; Ksouri, A.; Maxwell, F.; Bresson, B.; Hindawi, G.; Balasa, C.; Bellin, M.F.; Albiges, L. Testicular tumors: A diagnostic challenge of imaging. Bull. Cancer 2019, 106, 875–886. [Google Scholar] [CrossRef]
- Khanna, M.; Abualruz, A.R.; Yadav, S.K.; Mafraji, M.; Al-Rumaihi, K.; Al-Bozom, I.; Kumar, D.; Tsili, A.C.; Schieda, N. Diagnostic performance of multi-parametric MRI to differentiate benign sex cord stromal tumors from malignant (non-stromal and stromal) testicular neoplasms. Abdom. Radiol. 2021, 46, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Sohaib, S.A.; Koh, D.M.; Barbachano, Y.; Parikh, J.; Husband, J.E.S.; Dearnaley, D.P.; Horwich, A.; Huddart, R. Prospective assessment of MRI for imaging retroperitoneal metastases from testicular germ cell tumours. Clin. Radiol. 2009, 64, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.F.H.; Beaujon, L.J.F.; Villamizar, S.Y.G.; Lopez, F.F.F. Magnetic resonance versus computed tomography for the detection of retroperitoneal lymph node metastasis due to testicular cancer: A systematic literature review. Eur. J. Radiol. Open 2021, 8, 7. [Google Scholar]
- Kubik-Huch, R.A.; Hailemariam, S.; Hamm, B. Ct and MRI of the male genital tract: Radiologic-pathologic correlation. Eur. Radiol. 1999, 9, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Bleiberg, H. Colorectal cancer: The challenge. Eur. J. Cancer 1996, 32A, S2–S6. [Google Scholar] [CrossRef]
- de Souza, G.D.; Souza, L.R.Q.; Cuenca, R.M.; Vilela, V.M.; Santos, B.; de Aguiar, F.S. Pre- and postoperative imaging methods in colorectal cancer. ABCD-Arq. Bras. Cir. Dig.-Braz. Arch. Dig. Surg. 2018, 31, 5. [Google Scholar] [CrossRef]
- Georgiou, P.A.; Tekkis, P.P.; Constantinides, V.A.; Patel, U.; Goldin, R.D.; Darzi, A.W.; Nicholls, R.J.; Brown, G. Diagnostic accuracy and value of magnetic resonance imaging (MRI) in planning exenterative pelvic surgery for advanced colorectal cancer. Eur. J. Cancer 2013, 49, 72–81. [Google Scholar] [CrossRef]
- Annemans, L.; Lencioni, R.; Warie, H.; Bartolozzi, C.; Ciceri, M.; Muller, U. Health economic evaluation of ferucarbotran-enhanced MRI in the diagnosis of liver metastases in colorectal cancer patients. Int. J. Colorectal Dis. 2008, 23, 77–83. [Google Scholar] [CrossRef]
- Engbersen, M.P.; Rijsemus, C.J.V.; Nederend, J.; Aalbers, A.G.J.; de Hingh, I.; Retel, V.; Lambregts, D.M.J.; Van der Hoeven, E.; Boerma, D.; Wiezer, M.J.; et al. Dedicated MRI staging versus surgical staging of peritoneal metastases in colorectal cancer patients considered for crs-hipec; the disco randomized multicenter trial. BMC Cancer 2021, 21, 8. [Google Scholar] [CrossRef]
- Westberg, K.; Othman, B.; Suzuki, C.; Blomqvist, L.; Martling, A.; Iversen, H. Magnetic resonance imaging as a predictor of surgical outcome in patients with local pelvic recurrence of colorectal cancer. Eur. J. Surg. Oncol. 2021, 47, 2119–2124. [Google Scholar] [CrossRef]
- Nasseri, Y.; Langenfeld, S.J. Imaging for colorectal cancer. Surg. Clin. 2017, 97, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Dresen, R.C.; De Vuysere, S.; De Keyzer, F.; Van Cutsem, E.; Prenen, H.; Vanslembrouck, R.; De Hertogh, G.; Wolthuis, A.; D’Hoore, A.; Vandecaveye, V. Whole-body diffusion-weighted MRI for operability assessment in patients with colorectal cancer and peritoneal metastases. Cancer Imaging 2019, 19, 10. [Google Scholar] [CrossRef] [PubMed]
- Sabry, M.S.; Rady, A.E.E.; Niazi, G.E.M.; Ali, S.A. Role of diffusion-weighted MRI in diagnosis and post therapeutic follow-up of colorectal cancer. Egypt. J. Radiol. Nucl. Med. 2021, 52, 11. [Google Scholar] [CrossRef]
- Ono, K.; Ochiai, R.; Yoshida, T.; Kitagawa, M.; Omagari, J.; Kobayashi, H.; Yamashita, Y. Comparison of diffusion-weighted MRI and 2- fluorine-18 -fluoro-2-deoxy-d-glucose positron emission tomography (FDG-PET) for detecting primary colorectal cancer and regional lymph node metastases. J. Magn. Reson. Imaging 2009, 29, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Woo, S.; Han, S.; Suh, C.H.; Vargas, H.A. The diagnostic performance of MRI for detection of extramural venous invasion in colorectal cancer: A systematic review and meta-analysis of the literature. Am. J. Roentgenol. 2019, 213, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Smith, N.J.; Shihab, O.; Arnaout, A.; Swift, R.I.; Brown, G. MRI for detection of extramural vascular invasion in rectal cancer. Am. J. Roentgenol. 2008, 191, 1517–1522. [Google Scholar] [CrossRef]
- Soomro, M.H.; De Cola, G.; Conforto, S.; Schmid, M.; Giunta, G.; Guidi, E.; Neri, E.; Caruso, D.; Ciolina, M.; Laghi, A.; et al. Automatic segmentation of colorectal cancer in 3D MRI by combining deep learning and 3D level-set algorithm-a preliminary study. In Proceedings of the 2018 IEEE 4th Middle East Conference on Biomedical Engineering (MECBME), Tunis, Tunisia, 28–30 March 2018; pp. 198–203. [Google Scholar]
- Ogawa, M.; Ichiba, N.; Watanabe, M.; Yanaga, K. The usefulness of diffusion MRI in detection of lymph node metastases of colorectal cancer. Anticancer Res. 2016, 36, 815–819. [Google Scholar]
- Ichikawa, T.; Erturk, S.M.; Motosugi, U.; Sou, H.; Iino, H.; Araki, T.; Fujii, H. High-b-value diffusion-weighted MRI in colorectal cancer. Am. J. Roentgenol. 2006, 187, 181–184. [Google Scholar] [CrossRef]
- García-Figueiras, R.; Baleato-González, S.; Canedo-Antelo, M.; Alcalá, L.; Marhuenda, A. Imaging advances on ct and MRI in colorectal cancer. Curr. Colorectal Cancer Rep. 2021, 17, 113–130. [Google Scholar] [CrossRef]
- Kijima, S.; Sasaki, T.; Nagata, K.; Utano, K.; Lefor, A.T.; Sugimoto, H. Preoperative evaluation of colorectal cancer using ct colonography, MRI, and PET/CT. World J. Gastroenterol. 2014, 20, 16964–16975. [Google Scholar] [CrossRef]
- Kulemann, V.; Schima, W.; Tamandl, D.; Kaczirek, K.; Gruenberger, T.; Wrba, F.; Weber, M.; Ba-Ssalamah, A. Preoperative detection of colorectal liver metastases in fatty liver: MDCT or MRI? Eur. J. Radiol. 2011, 79, E1–E6. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.; Lee, J.M.; Song, Y.S.; Woo, S.; Hur, B.Y.; Jeon, J.H.; Paeng, J.C. Added value of integrated whole-body PET/MRI for evaluation of colorectal cancer: Comparison with contrast-enhanced mdct. Am. J. Roentgenol. 2016, 206, W10–W20. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, G. Importance of whole body MRI for staging of colorectal cancer. Radiologe 2012, 52, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Datta, N.R.; Krishnan, S.; Speiser, D.E.; Neufeld, E.; Kuster, N.; Bodis, S.; Hofmann, H. Magnetic nanoparticle-induced hyperthermia with appropriate payloads: Paul ehrlich’s "magic (nano)bullet" for cancer theranostics? Cancer Treat. Rev. 2016, 50, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Douziech-Eyrolles, L.; Marchais, H.; Herve, K.; Munnier, E.; Souce, M.; Linassier, C.; Dubois, P.; Chourpa, I. Nanovectors for anticancer agents based on superparamagnetic iron oxide nanoparticles. Int. J. Nanomed. 2007, 2, 541–550. [Google Scholar]
- Balaita, L.; Popa, M. Polymer magnetic particles in biomedical applications. Rev. Roum. Chim. 2009, 54, 185–199. [Google Scholar]
- Rotariu, O.; Udrea, L.E.; Strachan, N.J.C.; Badescu, V. The guidance of magnetic colloids in simulated tissues for targeted drug delivery. J. Optoelectron. Adv. Mater. 2007, 9, 942–945. [Google Scholar]
- Alexiou, C.; Arnold, W.; Klein, R.J.; Parak, F.G.; Hulin, P.; Bergemann, C.; Erhardt, W.; Wagenpfeil, S.; Lubbe, A.S. Locoregional cancer treatment with magnetic drug targeting. Cancer Res. 2000, 60, 6641–6648. [Google Scholar]
- Wahajuddin; Arora, S. Superparamagnetic iron oxide nanoparticles: Magnetic nanoplatforms as drug carriers. Int. J. Nanomed. 2012, 7, 3445–3471. [Google Scholar] [CrossRef]
- Mansouri, M.; Nazarpak, M.H.; Solouk, A.; Akbari, S.; Hasani-Sadrabadi, M.M. Magnetic responsive of paclitaxel delivery system based on spion and palmitoyl chitosan. J. Magn. Magn. Mater. 2017, 421, 316–325. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, Q.F.; Peng, J.R.; Su, J.; Lu, X.L.; Zhao, Y.X.; Qian, Z.Y. Doxorubicin-conjugated heparin-coated superparamagnetic iron oxide nanoparticles for combined anticancer drug delivery and magnetic resonance imaging. J. Biomed. Nanotechnol. 2016, 12, 1963–1974. [Google Scholar] [CrossRef] [PubMed]
- Mattingly, S.J.; O’Toole, M.G.; James, K.T.; Clark, G.J.; Nantz, M.H. Magnetic nanoparticle-supported lipid bilayers for drug delivery. Langmuir 2015, 31, 3326–3332. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, M.; Hamishehkar, H.; Arsalani, N.; Entezami, A.A. Preparation of thermo and ph-responsive polymer@au/fe3o4 core/shell nanoparticles as a carrier for delivery of anticancer agent. J. Nanopart. Res. 2015, 17, 13. [Google Scholar] [CrossRef]
- Song, W.X.; Muthana, M.; Mukherjee, J.; Falconer, R.J.; Biggs, C.A.; Zhao, X.B. Magnetic-silk core-shell nanoparticles as potential carriers for targeted delivery of curcumin into human breast cancer cells. ACS Biomater. Sci. Eng. 2017, 3, 1027–1038. [Google Scholar] [CrossRef]
- Rotariu, O.; Udrea, L.E.; Strachan, N.J.C.; Badescu, V. Targeting magnetic carrier particles in tumour microvasculature—A numerical study. J. Optoelectron. Adv. Mater. 2005, 7, 3209–3218. [Google Scholar]
- Norris, M.D.; Seidel, K.; Kirschning, A. Externally induced drug release systems with magnetic nanoparticle carriers: An emerging field in nanomedicine. Adv. Ther. 2019, 2, 1800092. [Google Scholar] [CrossRef]
- Ahmad, R.S.; Ali, Z.S.; Mou, X.B.; Wang, J.H.; Yi, H.; He, N.Y. Recent advances in magnetic nanoparticle design for cancer therapy. J. Nanosci. Nanotechnol. 2016, 16, 9393–9403. [Google Scholar] [CrossRef]
- Ramazanov, M.; Karimova, A.; Shirinova, H. Magnetism for drug delivery, MRI and hyperthermia applications: A review. Biointerface Res. Appl. Chem. 2021, 11, 8654–8668. [Google Scholar]
- Yang, H.Y.; Li, Y.; Lee, D.S. Multifunctional and stimuli-responsive magnetic nanoparticle-based delivery systems for biomedical applications. Adv. Ther. 2018, 1, 1800011. [Google Scholar] [CrossRef]
- Schneider-Futschik, E.K.; Reyes-Ortega, F. Advantages and disadvantages of using magnetic nanoparticles for the treatment of complicated ocular disorders. Pharmaceutics 2021, 13, 1157. [Google Scholar] [CrossRef] [PubMed]
- Zamay, T.N.; Prokopenko, V.S.; Zamay, S.S.; Lukyanenko, K.A.; Kolovskaya, O.S.; Orlov, V.A.; Zamay, G.S.; Galeev, R.G.; Narodov, A.A.; Kichkailo, A.S. Magnetic nanodiscs-a new promising tool for microsurgery of malignant neoplasms. Nanomaterials 2021, 11, 1459. [Google Scholar] [CrossRef]
- Golovin, Y.I.; Golovin, D.Y.; Vlasova, K.Y.; Veselov, M.M.; Usvaliev, A.D.; Kabanov, A.V.; Klyachko, N.L. Non-heating alternating magnetic field nanomechanical stimulation of biomolecule structures via magnetic nanoparticles as the basis for future low-toxic biomedical applications. Nanomaterials 2021, 11, 2255. [Google Scholar] [CrossRef]
- Chen, M.W.; Wu, J.J.; Ning, P.; Wang, J.J.; Ma, Z.; Huang, L.Q.; Plaza, G.R.; Shen, Y.J.; Xu, C.; Han, Y.; et al. Remote control of mechanical forces via mitochondrial-targeted magnetic nanospinners for efficient cancer treatment. Small 2020, 16, 14. [Google Scholar] [CrossRef] [PubMed]
- Alsharif, N.A.; Aleisa, F.A.; Liu, G.Y.; Ooi, B.S.; Patel, N.; Ravasi, T.; Merzaban, J.S.; Kosel, J. Functionalization of magnetic nanowires for active targeting and enhanced cell-killing efficacy. ACS Appl. Bio Mater. 2020, 3, 4789–4797. [Google Scholar] [CrossRef]
- Chen, Y.; Han, P.; Wu, Y.; Zhang, Z.F.; Yue, Y.; Li, W.H.; Chu, M.Q. Hedgehog-like gold-coated magnetic microspheres that strongly inhibit tumor growth through magnetomechanical force and photothermal effects. Small 2018, 14, 13. [Google Scholar] [CrossRef]
- Goiriena-Goikoetxea, M.; Munoz, D.; Orue, I.; Fernandez-Gubieda, M.L.; Bokor, J.; Muela, A.; Garcia-Arribas, A. Disk-shaped magnetic particles for cancer therapy. Appl. Phys. Rev. 2020, 7, 15. [Google Scholar] [CrossRef]
- Rahban, D.; Doostan, M.; Salimi, A. Cancer therapy; prospects for application of nanoparticles for magnetic-based hyperthermia. Cancer Investig. 2020, 38, 507–521. [Google Scholar] [CrossRef]
- Kok, H.P.; Cressman, E.N.K.; Ceelen, W.; Brace, C.L.; Ivkov, R.; Grull, H.; ter Haar, G.; Wust, P.; Crezee, J. Heating technology for malignant tumors: A review. Int. J. Hyperth. 2020, 37, 711–741. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Wang, Y.; Zhu, W.; Li, G.; Ma, X.; Zhang, Y.; Chen, S.; Tiwari, S.; Shi, K.; et al. Comprehensive understanding of magnetic hyperthermia for improving antitumor therapeutic efficacy. Theranostics 2020, 10, 3793–3815. [Google Scholar] [CrossRef]
- Chang, D.; Lim, M.; Goos, J.; Qiao, R.; Ng, Y.Y.; Mansfeld, F.M.; Jackson, M.; Davis, T.P.; Kavallaris, M. Biologically targeted magnetic hyperthermia: Potential and limitations. Front. Pharm. 2018, 9, 831. [Google Scholar] [CrossRef]
- Frazier, N.; Ghandehari, H. Hyperthermia approaches for enhanced delivery of nanomedicines to solid tumors. Biotechnol. Bioeng. 2015, 112, 1967–1983. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Lee, S.B.; Kim, Y.U.; Kim, K.N.; Choi, S.Y.; Lee, K.H.; Shim, I.B.; Kim, C.S. Effect of ferrite thermoseeds on destruction of carcinoma cells under alternating magnetic field. J. Mater. Sci. 2003, 38, 4221–4233. [Google Scholar] [CrossRef]
- Shido, Y.; Nishida, Y.; Suzuki, Y.; Kobayashi, T.; Ishiguro, N. Targeted hyperthermia using magnetite cationic liposomes and an alternating magnetic field in a mouse osteosarcoma model. J. Bone Jt. Surg. Br. Vol. 2010, 92B, 580–585. [Google Scholar] [CrossRef]
- Manohar, A.; Vijayakanth, V.; Pallavolu, M.R.; Kim, K.H. Effects of Ni—Substitution on structural, magnetic hyperthermia, photocatalytic and cytotoxicity study of MgFe2O4 nanoparticles. J. Alloys Compd. 2021, 879, 160515. [Google Scholar] [CrossRef]
- Chen, S.Z.; Han, F.S.; Huang, D.D.; Meng, J.Q.; Chu, J.P.; Wang, M.; Wang, P.J. Fe3O4 magnetic nanoparticle-enhanced radiotherapy for lung adenocarcinoma via delivery of siBIRC5 and AS-ODN. J. Transl. Med. 2021, 19, 1–14. [Google Scholar] [CrossRef]
- Jiang, K.Y.; Zhang, Q.B.; Hinojosa, D.T.; Zhang, L.L.; Xiao, Z.; Yin, Y.; Tong, S.; Colvin, V.L.; Bao, G. Controlled oxidation and surface modification increase heating capacity of magnetic iron oxide nanoparticles. Appl. Phys. Rev. 2021, 8, 031407. [Google Scholar] [CrossRef]
- Simeonidis, K.; Kaprara, E.; Rivera-Gil, P.; Xu, R.X.; Teran, F.J.; Kokkinos, E.; Mitropoulos, A.; Maniotis, N.; Balcells, L. Hydrotalcite-embedded magnetite nanoparticles for hyperthermia-triggered chemotherapy. Nanomaterials 2021, 11, 1796. [Google Scholar] [CrossRef]
- Kawashita, M.; Domi, S.; Saito, Y.; Aoki, M.; Ebisawa, Y.; Kokubo, T.; Saito, T.; Takano, M.; Araki, N.; Hiraoka, M. In vitro heat generation by ferrimagnetic maghemite microspheres for hyperthermic treatment of cancer under an alternating magnetic field. J. Mater. Sci. Mater. Med. 2008, 19, 1897–1903. [Google Scholar] [CrossRef]
- Tietze, R.; Zaloga, J.; Unterweger, H.; Lyer, S.; Friedrich, R.P.; Janko, C.; Pottler, M.; Durr, S.; Alexiou, C. Magnetic nanoparticle-based drug delivery for cancer therapy. Biochem. Biophys. Res. Commun. 2015, 468, 463–470. [Google Scholar] [CrossRef]
- Zhi, D.F.; Yang, T.; Yang, J.; Fu, S.; Zhang, S.B. Targeting strategies for superparamagnetic iron oxide nanoparticles in cancer therapy. Acta Biomater. 2020, 102, 13–34. [Google Scholar] [CrossRef]
- Uskokovic, V.; Drofenik, M. Gold-embellished mixed-valence manganite as a smart, self-regulating magnetoplasmonic nanomaterial. Mater. Chem. Phys. 2021, 271, 124870. [Google Scholar] [CrossRef]
- Rodrigues, H.F.; Capistrano, G.; Bakuzis, A.F. In vivo magnetic nanoparticle hyperthermia: A review on preclinical studies, low-field nano-heaters, noninvasive thermometry and computer simulations for treatment planning. Int. J. Hyperth. 2020, 37, 76–99. [Google Scholar] [CrossRef] [PubMed]
- Rotariu, O.; Iacob, G.; Strachan, N.J.C.; Chiriac, H. Simulating the embolization of blood vessels using magnetic microparticles and acupuncture needle in a magnetic field. Biotechnol. Prog. 2004, 20, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Anani, T.; Rahmati, S.; Sultana, N.; David, A.E. MRI-traceable theranostic nanoparticles for targeted cancer treatment. Theranostics 2021, 11, 579–601. [Google Scholar] [CrossRef]
- Kozissnik, B.; Bohorquez, A.C.; Dobson, J.; Rinaldi, C. Magnetic fluid hyperthermia: Advances, challenges, and opportunity. Int. J. Hyperth. 2013, 29, 706–714. [Google Scholar] [CrossRef]
- Xie, J.; Yan, C.; Yan, Y.; Chen, L.; Song, L.; Zang, F.; An, Y.; Teng, G.; Gu, N.; Zhang, Y. Multi-modal mn–zn ferrite nanocrystals for magnetically-induced cancer targeted hyperthermia: A comparison of passive and active targeting effects. Nanoscale 2016, 8, 16902–16915. [Google Scholar] [CrossRef]
- Angelakeris, M. Magnetic nanoparticles: A multifunctional vehicle for modern theranostics. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 1642–1651. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.W.; Tsao, H.Y.; Chiang, C.S.; Chen, S.Y. Advances in magnetic nanoparticle-mediated cancer immune-theranostics. Adv. Healthc. Mater. 2021, 10, 2001451. [Google Scholar] [CrossRef]
- Rajan, A.; Sahu, N.K. Review on magnetic nanoparticle-mediated hyperthermia for cancer therapy. J. Nanopart. Res. 2020, 22, 1–25. [Google Scholar] [CrossRef]
- Soetaert, F.; Korangath, P.; Serantes, D.; Fiering, S.; Ivkov, R. Cancer therapy with iron oxide nanoparticles: Agents of thermal and immune therapies. Adv. Drug Deliv. Rev. 2020, 163, 65–83. [Google Scholar] [CrossRef]
- Li, X.X.; Li, W.Y.; Wang, M.N.; Liao, Z.H. Magnetic nanoparticles for cancer theranostics: Advances and prospects. J. Control. Release 2021, 335, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, L.; Gomaa, H.G.; Ragab, D.; Zhu, J. Magnetic nanoparticles for environmental and biomedical applications: A review. Particuology 2017, 30, 1–14. [Google Scholar] [CrossRef]
- Li, Y.H.; Wang, N.; Huang, X.M.; Li, F.Y.; Davis, T.P.; Qiao, R.R.; Ling, D.S. Polymer-assisted magnetic nanoparticle assemblies for biomedical applications. ACS Appl. Bio Mater. 2020, 3, 121–142. [Google Scholar] [CrossRef] [PubMed]
- Pooam, M.; Jourdan, N.; El Esawi, M.; Sherrard, R.M.; Ahmad, M. Hek293 cell response to static magnetic fields via the radical pair mechanism may explain therapeutic effects of pulsed electromagnetic fields. PLoS ONE 2020, 15, e0243038. [Google Scholar] [CrossRef]
- Chatterjee, R.; Chatterjee, J. Ros and oncogenesis with special reference to emt and stemness. Eur. J. Cell Biol. 2020, 99, 8. [Google Scholar] [CrossRef]
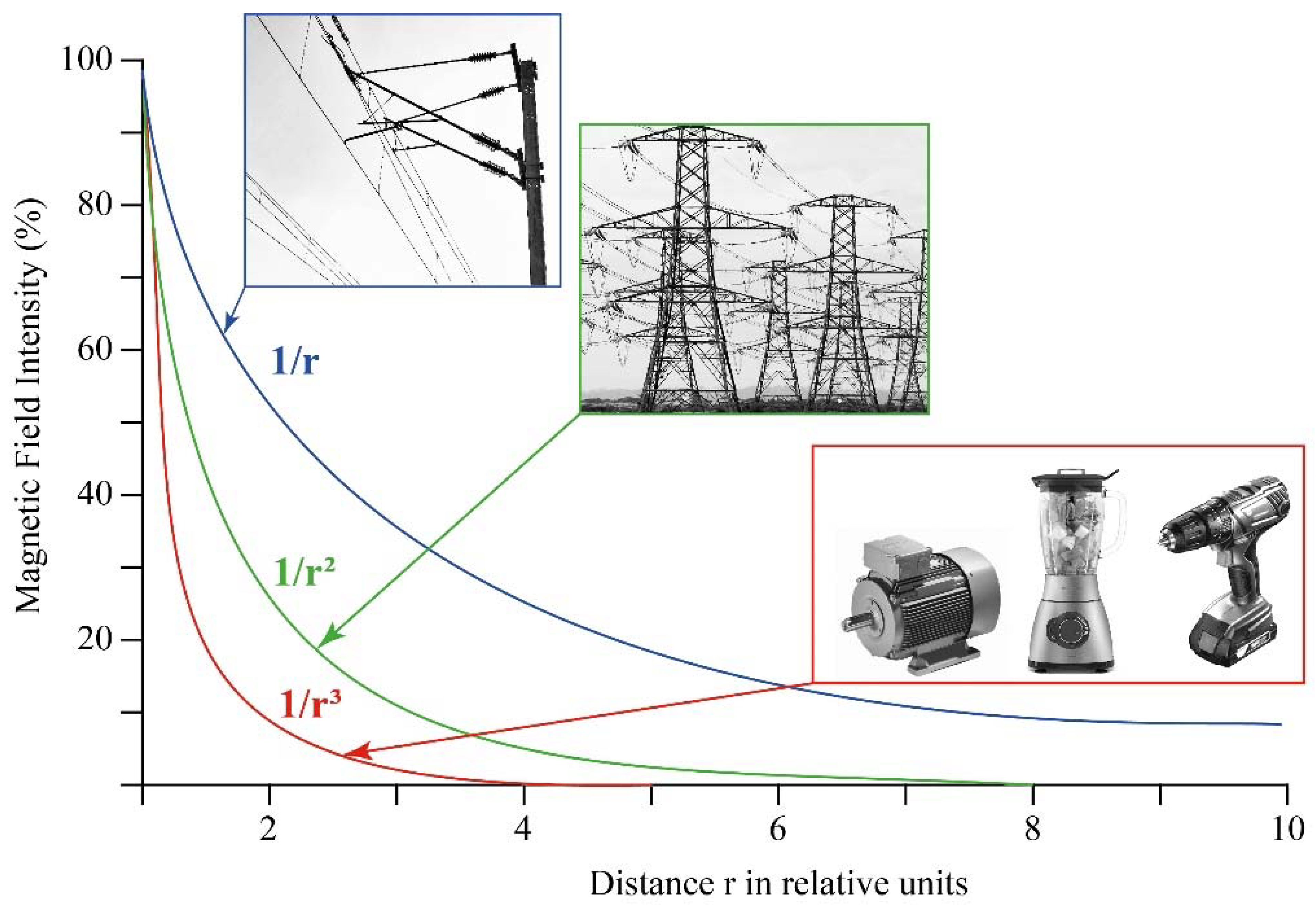
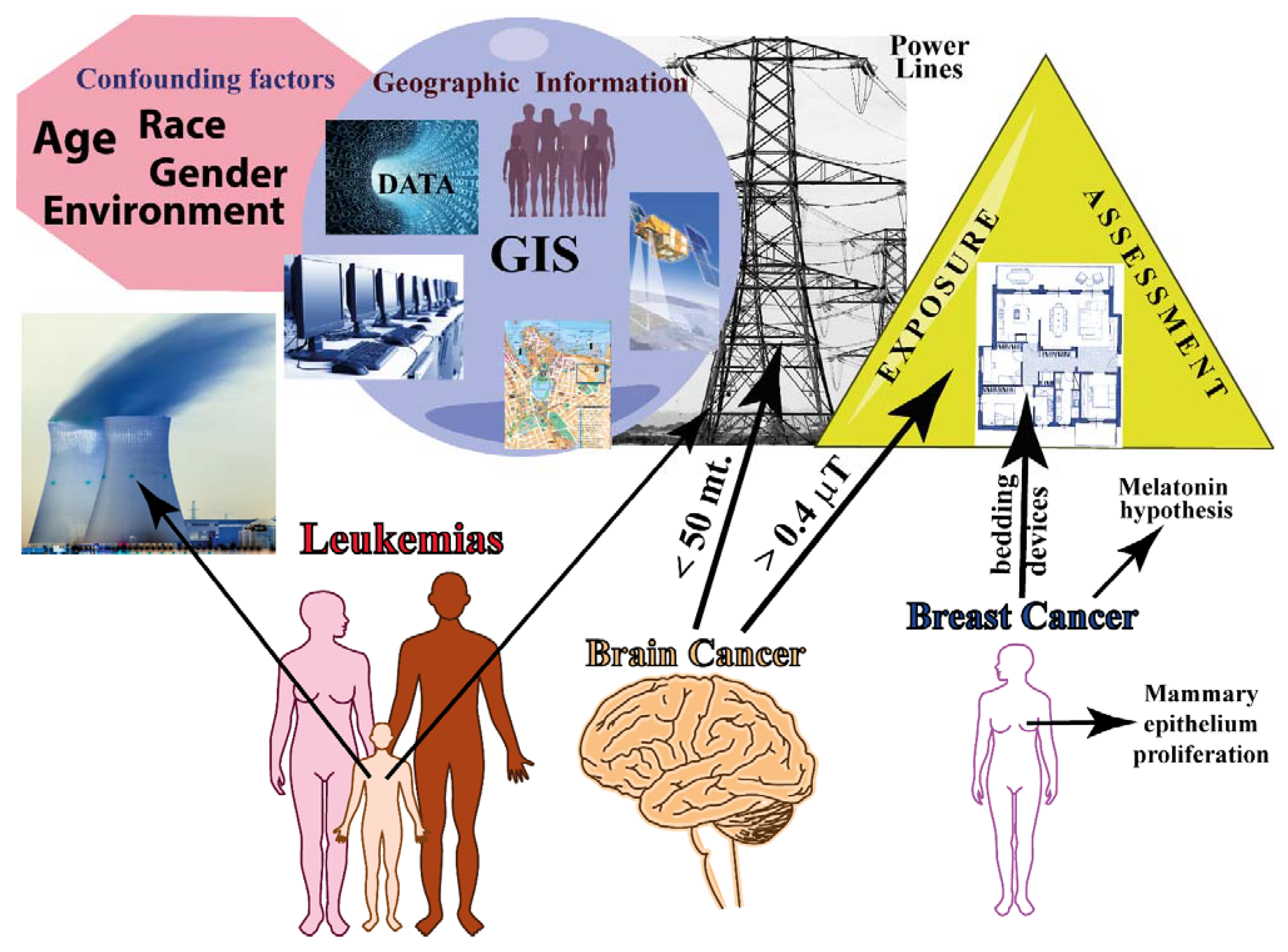
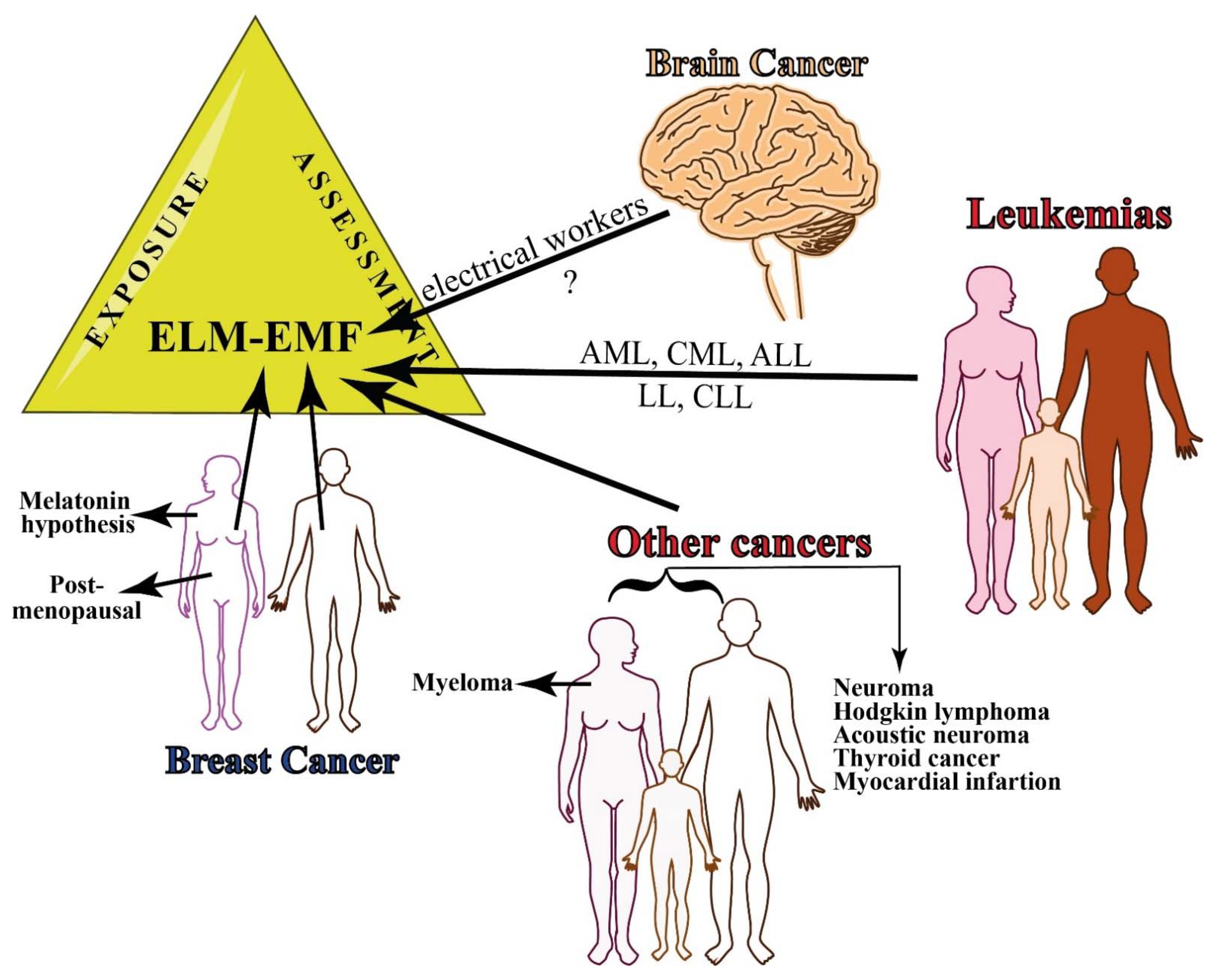
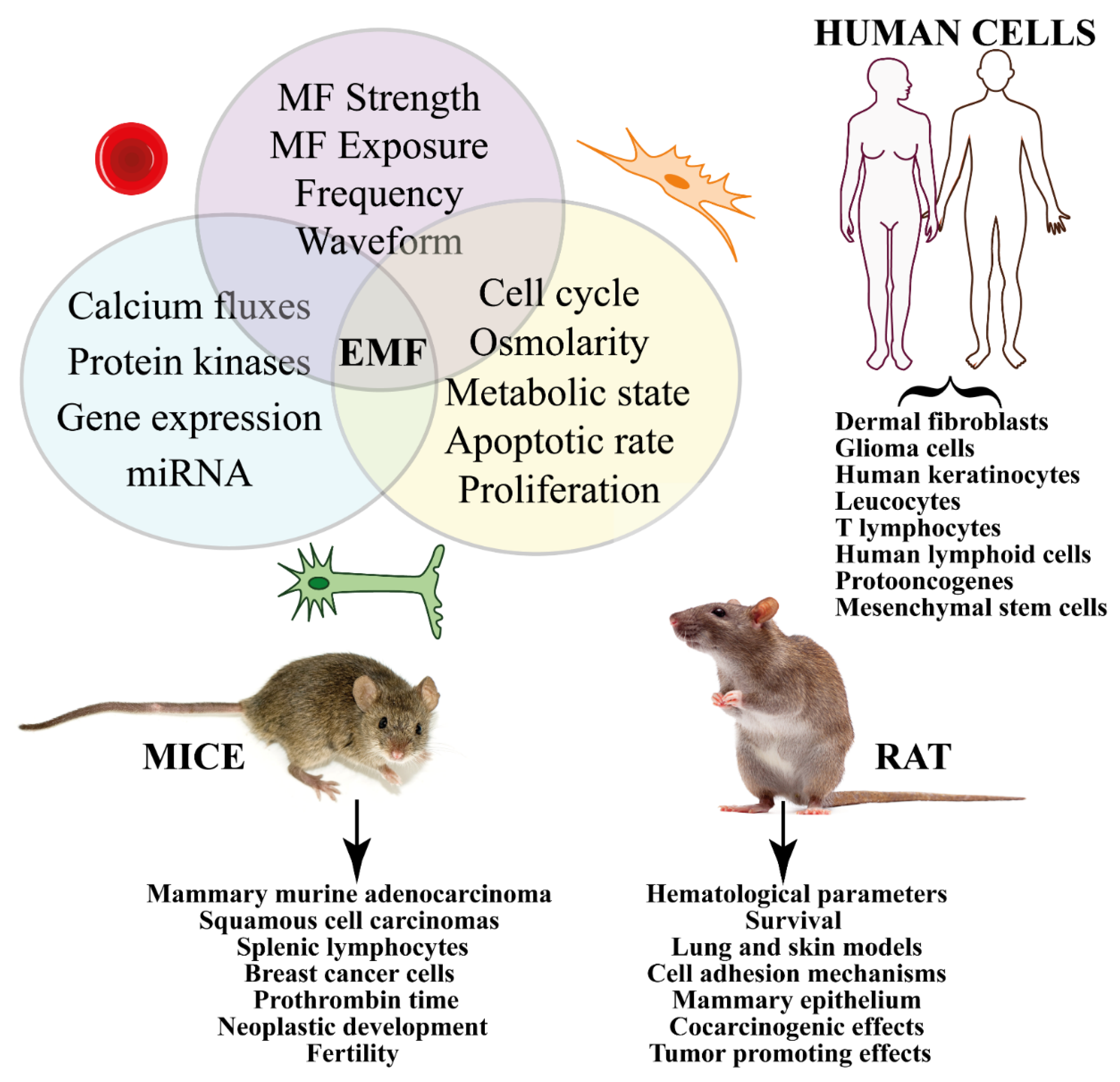
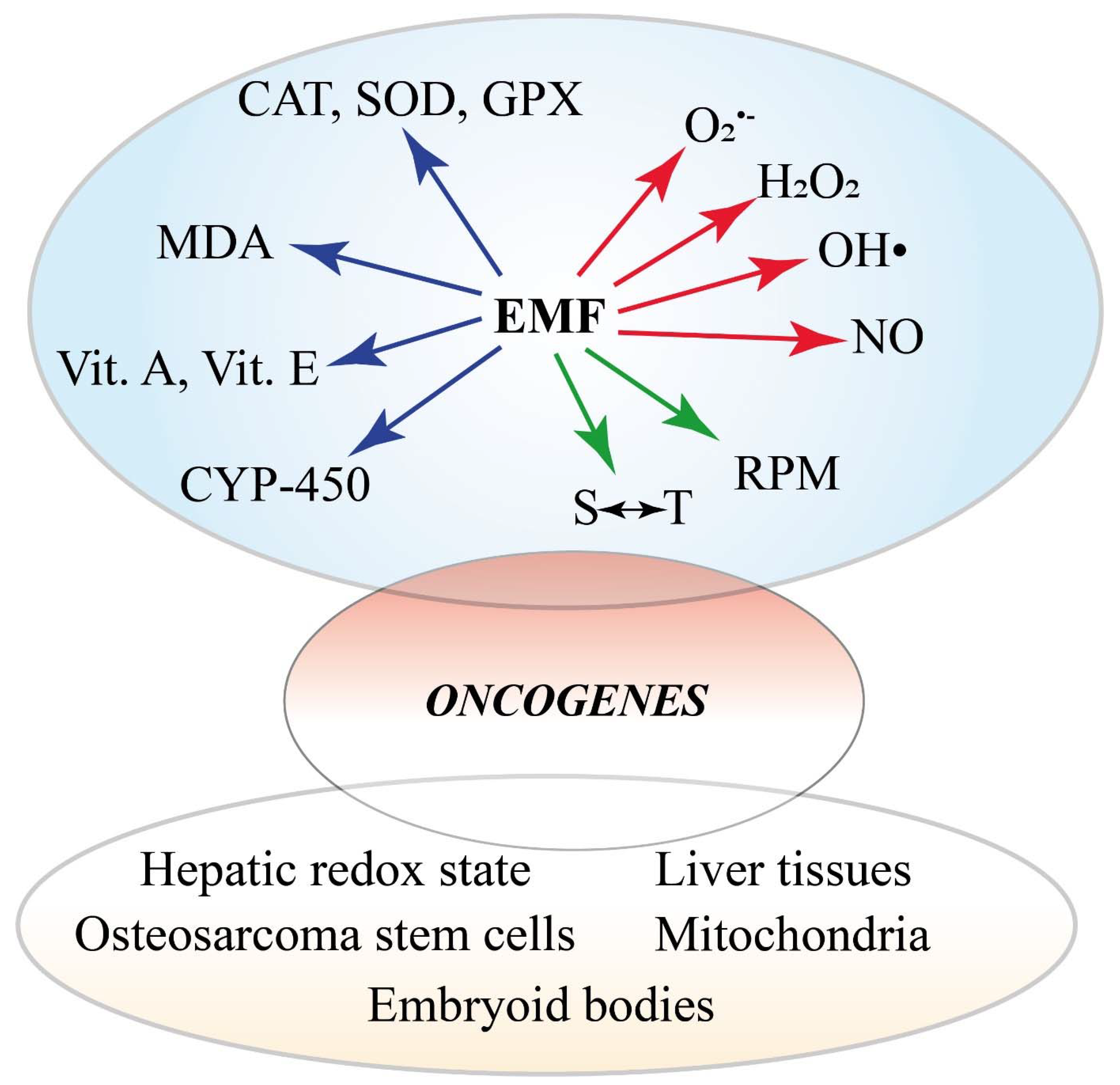
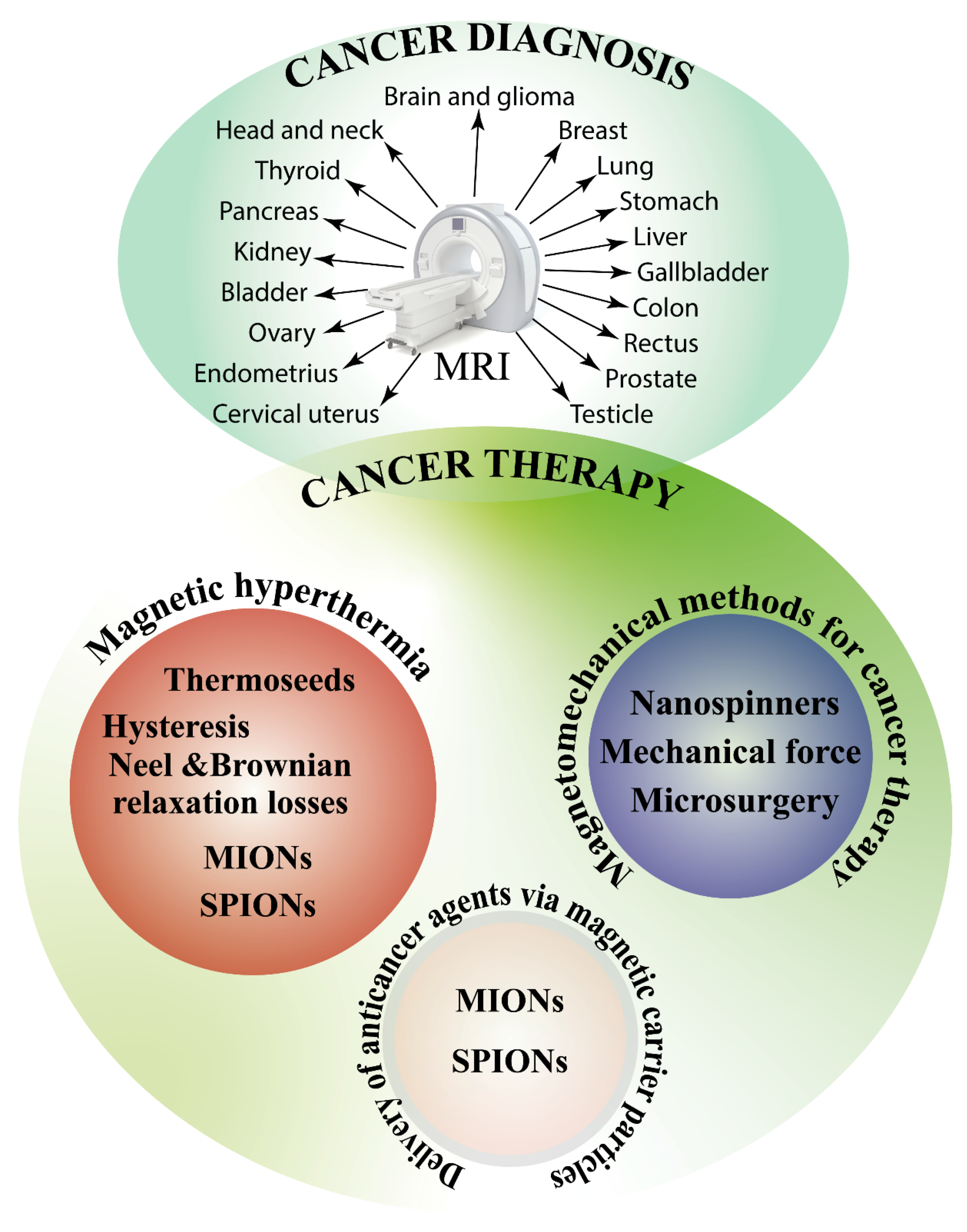
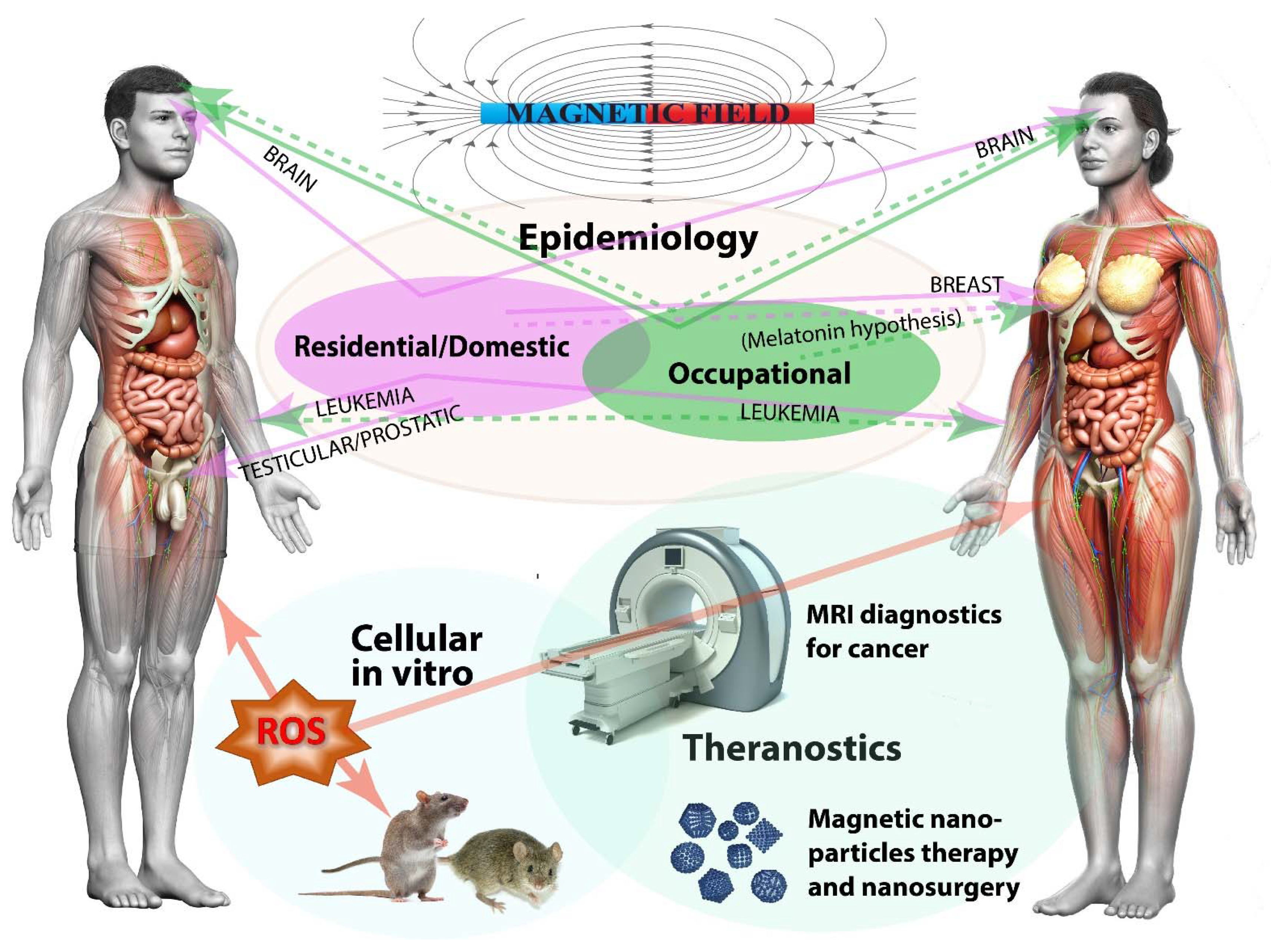
| Type of Radiation | Type of Field | Frequency | Wavelength | Use | Examples | Effect |
|---|---|---|---|---|---|---|
| NI | SMF | 0 Hz | N.A. | GMF, permanent magnets, transmission lines, HVDC lines, batteries, between objects with different electrical charges, MRI | Action of force | |
| NI | AMF | 0.3 Hz 3 Hz 16 2/3 Hz 50 Hz 300 Hz 3 kHz 30 kHz | 106 km 100,000 km 18,000 km 6000 km 1000 km 100 km 10 km | Low Frequency traction current and three phase alternating current | Technical appliances such as power lines, wiring and household appliances such as appliances for heating (e.g., electric cooker, electric heating, washing machine, electric water heater, iron), appliances with a transformer or magnetic coils (e.g., radio clock, low-voltage halogen lamps, television set, WiFi) and appliances with an electric motor (e.g., vacuum cleaner, drill, hand blender, hair dryer, electric cars) | Stimulation/irritation |
| NI | AMF | 300 kHz 3 MHz 30 MHz 300 MHz 3 GHz 30 GHz 300 GHz 3 THz | 1 km 100 m 10 m 1 m 10 mm 10 mm 1 mm 100 µm | Radio frequency. Radio/television, microwaves, terahertz waves | Induction cookers and electronic article surveillance systems in stores, as well as many industrial and medical applications, PC monitors, mobile phone, microwave ovens, radar stations. Broadcasting frequencies (short wave, AM, and FM radio), digital television (digital video broadcasting-terrestrial, DVBT) and digital radio (digital audio broadcasting, DAB). Wireless local area networks (WiFi, WLAN), cordless telephones, Bluetooth devices, baby monitors, electronic article surveillance systems and RFID (radio frequency range), radar systems, radio relay systems, satellite TV and satellite Internet, radio solutions for stationary Internet | Thermal effect |
| NI | AMF | 30 THz 300 THz | 10 µm 1 µm | Infrared | Bulb lamps, heaters, body scanners for security control | Thermal effect |
| NI | AMF | 380 THz 789 THz | 780 nm 380 nm | Visible Light | ||
| Ionizing | AMF | 3 × 1015 Hz 3 × 1016 Hz 3 × 1017 Hz 3 × 1018 Hz 3 × 1019 Hz 3 × 1020 Hz | 100 nm 10 nm 1 nm 100 pm 10 pm 1 pm | UV-light, X-rays, gamma rays | Nuclear power plants, X-ray machines, radioactive material. | Ionization |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maffei, M.E. Magnetic Fields and Cancer: Epidemiology, Cellular Biology, and Theranostics. Int. J. Mol. Sci. 2022, 23, 1339. https://doi.org/10.3390/ijms23031339
Maffei ME. Magnetic Fields and Cancer: Epidemiology, Cellular Biology, and Theranostics. International Journal of Molecular Sciences. 2022; 23(3):1339. https://doi.org/10.3390/ijms23031339
Chicago/Turabian StyleMaffei, Massimo E. 2022. "Magnetic Fields and Cancer: Epidemiology, Cellular Biology, and Theranostics" International Journal of Molecular Sciences 23, no. 3: 1339. https://doi.org/10.3390/ijms23031339
APA StyleMaffei, M. E. (2022). Magnetic Fields and Cancer: Epidemiology, Cellular Biology, and Theranostics. International Journal of Molecular Sciences, 23(3), 1339. https://doi.org/10.3390/ijms23031339