The Genetic Diversity and Dysfunctionality of Catalase Associated with a Worse Outcome in Crohn’s Disease
Abstract
1. Introduction
2. Results
2.1. SNPs in the Catalase Gene Are Associated with the Risk of Crohn’s Disease in a Spanish Population
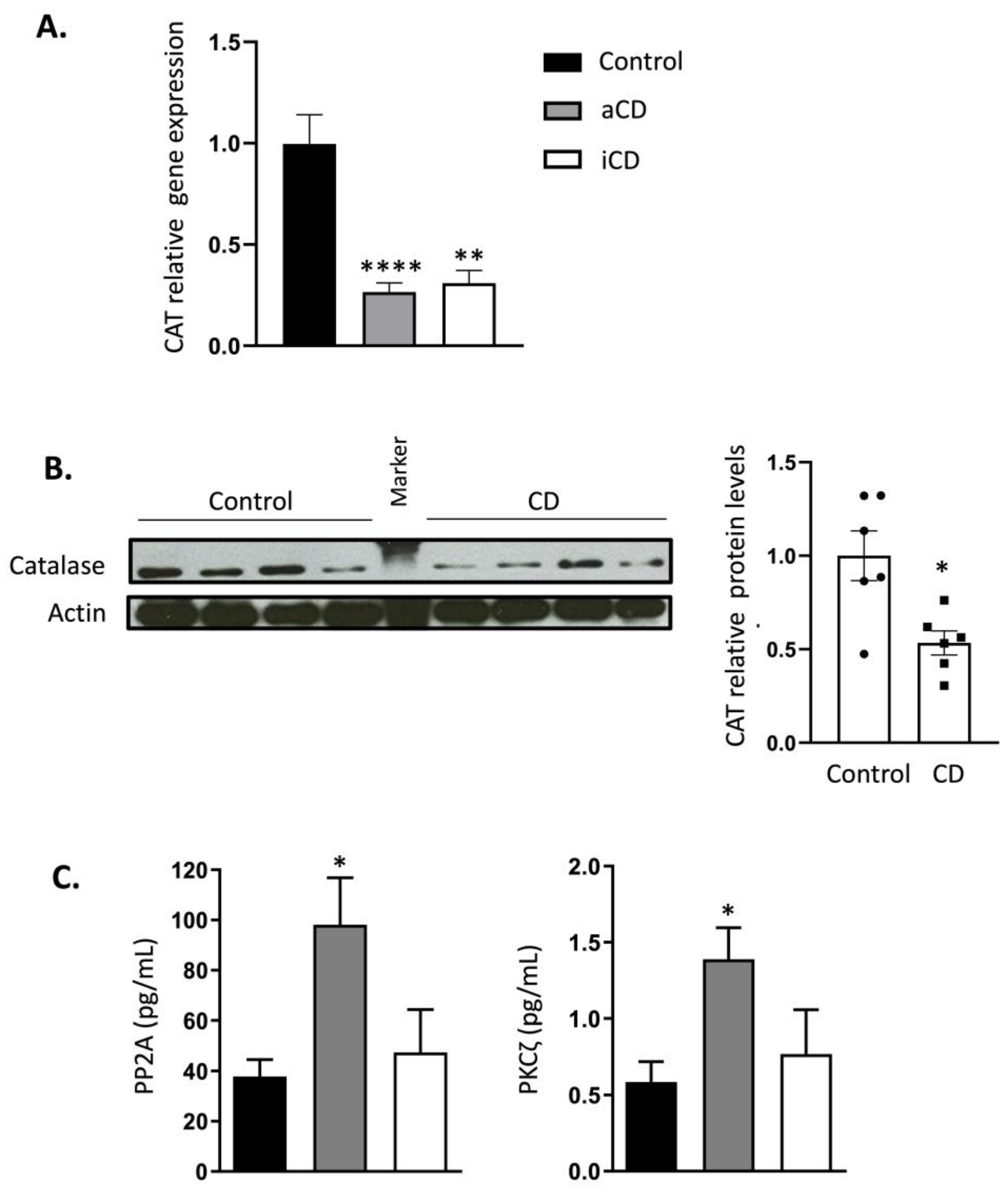
2.2. Catalase Expression Is Permanently Inhibited in Crohn’s Disease Patients
3. Discussion
4. Materials and Methods
4.1. Study Design
4.1.1. Discovery Cohort (Genetic Study)
4.1.2. Confirmatory Cohort (Functional and Regulatory Studies of CAT Gene)
4.2. Single Nucleotide Polymorphisms Analysis
4.3. RNA Isolation and Gene Expression Analysis
4.4. Isolation of Peripheral Leukocytes
4.5. Western Blotting
4.6. Protein Kinase C Zeta (PKCζ) and Protein Phosphatase 2A (PP2A) Levels
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rezaie, A.; Parker, R.D.; Abdollahi, M. Oxidative stress and pathogenesis of inflammatory bowel disease: An epiphenomenon or the cause? Dig. Dis. Sci. 2007, 52, 2015–2021. [Google Scholar] [CrossRef] [PubMed]
- Kruidenier, L.; Verspaget, H.W. Review article: Oxidative stress as a pathogenic factor in inflammatory bowel disease—radicals or ridiculous? Aliment. Pharmacol. Ther. 2002, 16, 1997–2015. [Google Scholar] [CrossRef] [PubMed]
- Bashir, S.; Harris, G.; Denman, M.A.; Blake, D.R.; Winyard, P.G. Oxidative DNA damage and cellular sensitivity to oxidative stress in human autoimmune diseases. Ann. Rheum. Dis. 1993, 52, 659–666. [Google Scholar] [CrossRef]
- Alemany-Cosme, E.; Sáez-González, E.; Moret, I.; Mateos, B.; Iborra, M.; Nos, P.; Sandoval, J.; Beltrán, B. Oxidative Stress in the Pathogenesis of Crohn’s Disease and the Interconnection with Immunological Response, Microbiota, External Environmental Factors, and Epigenetics. Antioxidants 2021, 10, 64. [Google Scholar] [CrossRef]
- Adler, V.; Yin, Z.; Tew, K.D.; Ronai, Z. Role of redox potential and reactive oxygen species in stress signaling. Oncogene 1999, 18, 6104–6111. [Google Scholar] [CrossRef] [PubMed]
- Herrlich, P.; Böhmer, F.D. Redox regulation of signal transduction in mammalian cells. Biochem. Pharmacol. 1999, 59, 35–41. [Google Scholar] [CrossRef]
- Boirivant, M.; Marini, M.; Di Felice, G.; Pronio, A.M.; Montesani, C.; Tersigni, R.; Strober, W. Lamina propria T cells in Crohn’s disease and other gastrointestinal inflammation show defective CD2 pathway-induced apoptosis. Gastroenterology 1999, 116, 557–565. [Google Scholar] [CrossRef]
- Kahl, R.; Kampkötter, A.; Wätjen, W.; Chovolou, Y. Antioxidant Enzymes and Apoptosis. Drug Metab. Rev. 2004, 36, 747–762. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Del Carmen, S.; Miyoshi, A.; Azevedo, V.; Sesma, F.; Langella, P.; Bermúdez-Humarán, L.G.; Watterlot, L.; Perdigon, G.; LeBlanc, A.D.M.D. Use of superoxide dismutase and catalase producing lactic acid bacteria in TNBS induced Crohn’s disease in mice. J. Biotechnol. 2011, 151, 287–293. [Google Scholar] [CrossRef]
- Parihar, A.K.S.; Srivastava, S.; Patel, S.; Singh, M.R.; Singh, D. Novel catalase loaded nanocores for the treatment of inflammatory bowel diseases. Artif. Cells. Nanomed. Biotechnol. 2016, 45, 981–989. [Google Scholar] [CrossRef]
- Beltrán, B.; Nos, P.; Dasí, F.; Iborra, M.; Bastida, G.; Martínez, M.; O’connor, J.-E.; Sáez, G.; Moret, I.; Ponce, J. Mitochondrial dysfunction, persistent oxidative damage, and catalase inhibition in immune cells of naïve and treated Crohn’s disease. Inflamm. Bowel Dis. 2010, 16, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Iborra, M.; Moret, I.; Rausell, F.; Bastida, G.; Aguas, M.; Cerrillo, E.; Nos, P.; Beltrán, B. Role of oxidative stress and antioxidant enzymes in Crohn’s disease. Biochem. Soc. Trans. 2011, 39, 1102–1106. [Google Scholar] [CrossRef]
- Hackenberg, T.; Juul, T.; Auzina, A.; Gwiżdż, S.; Małolepszy, A.; Van Der Kelen, K.; Dam, S.; Bressendorff, S.; Lorentzen, A.; Roepstorff, P.; et al. Catalase and NO CATALASE ACTIVITY1 Promote Autophagy-Dependent Cell Death in Arabidopsis. Plant Cell 2013, 25, 4616–4626. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.; Hofius, D.; Andersen, S.U. Signaling unmasked. Autophagy 2014, 10, 520–521. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lu, X.C.; Tao, Y.; Wu, C.; Zhao, P.L.; Li, K.; Zheng, J.Y.; Li, L.X. Association between Variants of the Autophagy Related Gene—IRGM and Susceptibility to Crohn’s Disease and Ulcerative Colitis: A Meta-Analysis. PLoS ONE 2013, 8, e80602. [Google Scholar] [CrossRef] [PubMed]
- Góth, L.; Rass, P.; Páy, A. Catalase enzyme mutations and their association with diseases. Mol. Diagn. 2004, 8, 141–149. [Google Scholar] [CrossRef]
- Ogata, M.; Wang, D.-H.; Ogino, K. Mammalian acatalasemia: The perspectives of bioinformatics and genetic toxicology. Acta Med. Okayama 2008, 62, 345–361. [Google Scholar]
- Moret-Tatay, I.; Iborra, M.; Cerrillo, E.; Tortosa, L.; Nos, P.; Beltrán, B. Possible Biomarkers in Blood for Crohn’s Disease: Oxidative Stress and MicroRNAs—Current Evidences and Further Aspects to Unravel. Oxidative Med. Cell Longev. 2015, 2016, 2325162. [Google Scholar] [CrossRef]
- Mrowicka, M.; Mrowicki, J.; Mik, M.; Wojtczak, R.; Dziki, L.; Dziki, A.; Majsterek, I. Association between SOD1, CAT, GSHPX1 polymorphisms and the risk of inflammatory bowel disease in the Polish population. Oncotarget 2017, 8, 109332–109339. [Google Scholar] [CrossRef]
- Yano, S.; Yano, N. Regulation of catalase enzyme activity by cell signaling molecules. Mol. Cell. Biochem. 2002, 240, 119–130. [Google Scholar] [CrossRef]
- Tüzün, A.; Erdil, A.; Inal, V.; Aydın, A.; Bağcı, S.; Yeşilova, Z.; Sayal, A.; Karaeren, N.; Dağalp, K. Oxidative stress and antioxidant capacity in patients with inflammatory bowel disease. Clin. Biochem. 2002, 35, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Kruidenier, L.; Kuiper, I.; Van Duijn, W.; Mieremet-Ooms, M.A.C.; Van Hogezand, R.A.; Lamers, C.B.H.W.; Verspaget, H.W. Imbalanced secondary mucosal antioxidant response in inflammatory bowel disease. J. Pathol. 2003, 201, 17–27. [Google Scholar] [CrossRef]
- Jahanshahi, G.; Motavasel, V.; Rezaie, A.; Hashtroudi, A.A.; Daryani, N.E.; Abdollahi, M. Alterations in Antioxidant Power and Levels of Epidermal Growth Factor and Nitric Oxide in Saliva of Patients with Inflammatory Bowel Diseases. Am. J. Dig. Dis. 2004, 49, 1752–1757. [Google Scholar] [CrossRef] [PubMed]
- Koutroubakis, I.E.; Malliaraki, N.; Dimoulios, P.D.; Karmiris, K.; Castanas, E.; Kouroumalis, E.A. Decreased Total and Corrected Antioxidant Capacity in Patients with Inflammatory Bowel Disease. Am. J. Dig. Dis. 2004, 49, 1433–1437. [Google Scholar] [CrossRef] [PubMed]
- Clerch, L.; Massaro, D. Oxidation-reduction-sensitive binding of lung protein to rat catalase mRNA. J. Biol. Chem. 1992, 267, 2853–2855. [Google Scholar] [CrossRef]
- Kodydková, J.; Vávrová, L.; Kocik, M.; Zak, A. Human catalase, its polymorphisms, regulation and changes of its activity in different diseases. Folia Biol. 2014, 60, 153–167. [Google Scholar]
- Shen, Y.; Li, D.; Tian, P.; Shen, K.; Zhu, J.; Feng, M.; Wan, C.; Yang, T.; Chen, L.; Wen, F. The Catalase C-262T Gene Polymorphism and Cancer Risk. Medicine 2015, 94, e679. [Google Scholar] [CrossRef]
- Yasmineh, W.G.; Parkin, J.L.; Caspers, J.I.; Theologides, A. Tumor necrosis factor/cachectin decreases catalase activity of rat liver. Cancer Res. 1991, 51, 3990–3995. [Google Scholar]
- Beier, K.; Völkl, A.; Fahimi, H. TNF-α downregulates the peroxisome proliferator activated receptor-α and the mRNAs encoding peroxisomal proteins in rat liver. FEBS Lett. 1997, 412, 385–387. [Google Scholar] [CrossRef]
- Durak, I.; Yasa, M.H.; Bektas, A.; Kaçmaz, M.; Cimen, M.Y.; Ozturk, H.S. Mucosal antioxidant defense is not impaired in ulcerative colitis. Hepatogastroenterology 2000, 47, 1015–1017. [Google Scholar]
- Bhaskar, L.; Ramakrishna, B.S.; Balasubramanian, K.A. Colonic mucosal antioxidant enzymes and lipid peroxide levels in normal subjects and patients with ulcerative colitis. J. Gastroenterol. Hepatol. 1995, 10, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Aimone-Gastin, I.; Cable, S.; Keller, J.M.; Bigard, M.A.; Champigneulle, B.; Gaucher, P.; Gueant, J.L.; Dauca, M. Studies on peroxisomes of colonic mucosa in Crohn’s disease. Dig. Dis. Sci. 1994, 39, 2177–2185. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Kindy, M.S.; Holtsberg, F.W.; Clair, D.K.S.; Yen, H.-C.; Germeyer, A.; Steiner, S.M.; Bruce-Keller, A.J.; Hutchins, J.B.; Mattson, M.P. Mitochondrial Manganese Superoxide Dismutase Prevents Neural Apoptosis and Reduces Ischemic Brain Injury: Suppression of Peroxynitrite Production, Lipid Peroxidation, and Mitochondrial Dysfunction. J. Neurosci. 1998, 18, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Nenoi, M.; Ichimura, S.; Mita, K.; Yukawa, O.; Cartwright, I.L. Regulation of the catalase gene promoter by Sp1, CCAAT-recognizing factors, and a WT1/Egr-related factor in hydrogen peroxide-resistant HP100 cells. Cancer Res. 2001, 61, 5885–5894. [Google Scholar] [PubMed]
- Chovolou, Y.; Watjen, W.; Kampkotter, A.; Kahl, R. Resistance to Tumor Necrosis Factor-α (TNF-α)-induced Apoptosis in Rat Hepatoma Cells Expressing TNF-α Is Linked to Low Antioxidant Enzyme Expression. J. Biol. Chem. 2003, 278, 29626–29632. [Google Scholar] [CrossRef]
- Kurien, B.T.; Scofield, R. Free radical mediated peroxidative damage in systemic lupus erythematosus. Life Sci. 2003, 73, 1655–1666. [Google Scholar] [CrossRef]
- Warchoł, T.; Lianeri, M.; Wudarski, M.; Łącki, J.K.; Jagodziński, P.P. Catalase −262C>T polymorphism in systemic lupus erythematosus in Poland. Rheumatol. Int. 2008, 28, 1035–1039. [Google Scholar] [CrossRef]
- Taysi, S.; Gul, M.; Sari, R.A.; Akcay, F.; Bakan, N. Serum Oxidant/Antioxidant Status in Patients with Systemic Lupus Erythematosus. Clin. Chem. Lab. Med. 2002, 40, 684–688. [Google Scholar] [CrossRef]
- Chistiakov, D.A.; Savost’Ianov, K.V.; Turakulov, R.I.; Shcherbacheva, L.N.; Mamaeva, G.G.; Balabolkin, M.I.; Nosikov, V.V. Nucleotide substitution C1167T in the catalase gene and position of nearby polymorphic markers DS11S907 and D11S2008 are connected with development of diabetes mellitus type 2. Mol. Biol. 2000, 34, 863–867. [Google Scholar]
- Casp, C.B.; She, J.X.; McCormack, W.T. Genetic association of the catalase gene (CAT) with vitiligo susceptibility. Pigment. Cell Res. 2002, 15, 62–66. [Google Scholar] [CrossRef]
- Jiang, Z.; Akey, J.M.; Shi, J.; Xiong, M.; Wang, Y.; Shen, Y.; Xu, X.; Chen, H.; Wu, H.; Xiao, J.; et al. A polymorphism in the promoter region of catalase is associated with blood pressure levels. Qual. Life Res. 2001, 109, 95–98. [Google Scholar] [CrossRef]
- Polonikov, A.V.; Ivanov, V.P.; Solodilova, M.A.; Kozhuhov, M.A.; Panfilov, V.I. Tobacco Smoking, Fruit and Vegetable Intake Modify Association Between -21A > T Polymorphism of Catalase Gene and Risk of Bronchial Asthma. J. Asthma 2009, 46, 217–224. [Google Scholar] [CrossRef]
- Bohanec Grabar, P.; Logar, D.; Tomsic, M.; Rozman, B.; Dolzan, V. Genetic polymorphisms modifying oxidative stress are associated with disease activity in rheumatoid arthritis patients. Dis. Markers 2009, 26, 41–48. [Google Scholar] [CrossRef]
- Em, S.; Laddha, N.C.; Chatterjee, S.; Gani, A.R.; Malek, R.A.; Shah, B.J.; Begum, R. Association of catalase T/C exon 9 and glutathione peroxidase codon 200 polymorphisms in relation to their activities and oxidative stress with vitiligo susceptibility in Gujarat population. Pigment Cell Res. 2007, 20, 405–407. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-H.; Hong, J.; Oh, B.; Cho, Y.; Lee, J.-Y.; Kim, H.-L.; Shin, E.-S.; Park, E.; Kim, S.-Y. Genetic association study of polymorphisms in the catalase gene with the risk of osteonecrosis of the femoral head in the Korean population. Osteoarthr. Cartil. 2008, 16, 1060–1066. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Konings, A.; Van Laer, L.; Pawelczyk, M.; Carlsson, P.-I.; Bondeson, M.-L.; Rajkowska, E.; Dudarewicz, A.; Vandevelde, A.; Fransen, E.; Huyghe, J.; et al. Association between variations in CAT and noise-induced hearing loss in two independent noise-exposed populations. Hum. Mol. Genet. 2007, 16, 1872–1883. [Google Scholar] [CrossRef] [PubMed]
- Marinko, T.; Konda, J.T.S.; Dolžan, V.; Goričar, K. Genetic Variability of Antioxidative Mechanisms and Cardiotoxicity after Adjuvant Radiotherapy in HER2-Positive Breast Cancer Patients. Dis. Markers 2020, 2020, 6645588. [Google Scholar] [CrossRef] [PubMed]
- Khodayari, S.; Salehi, Z.; Asl, S.F.; Aminian, K.; Gisomi, N.M.; Dalivandan, S.T. Catalase gene C-262T polymorphism: Importance in ulcerative colitis. J. Gastroenterol. Hepatol. 2013, 28, 819–822. [Google Scholar] [CrossRef]
- Bulatova, I.A.; Tretyakova, Y.I.; Shchekotov, V.V.; Shchekotova, A.P.; Ulitina, P.V.; Krivtsov, A.V.; Nenasheva, O.Y. Catalase gene rs1001179 polymorphism and oxidative stress in patients with chronic hepatitis C and ulcerative colitis. Ter. Arkhiv 2015, 87, 49–53. [Google Scholar] [CrossRef]
- Forsberg, L.; Lyrenäs, L.; Morgenstern, R.; De Faire, U. A common functional C-T substitution polymorphism in the promoter region of the human catalase gene influences transcription factor binding, reporter gene transcription and is correlated to blood catalase levels. Free Radic. Biol. Med. 2001, 30, 500–505. [Google Scholar] [CrossRef]
- Ahn, J.; Nowell, S.; McCann, S.E.; Yu, J.; Carter, L.; Lang, N.P.; Kadlubar, F.F.; Ratnasinghe, L.D.; Ambrosone, C.B. Associations between Catalase Phenotype and Genotype: Modification by Epidemiologic Factors. Cancer Epidemiol. Biomark. Prev. 2006, 15, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Bastaki, M.; Huen, K.; Manzanillo, P.; Chande, N.; Chen, C.; Balmes, J.R.; Tager, I.B.; Holland, N. Genotype-activity relationship for Mn-superoxide dismutase, glutathione peroxidase 1 and catalase in humans. Pharm. Genom. 2006, 16, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Nadif, R.; Mintz, M.; Jedlicka, A.; Bertrand, J.P.; Kleeberger, S.R.; Kauffmann, F. Association of CAT polymorphisms with catalase activity and exposure to environmental oxidative stimuli. Free Radic. Res. 2005, 39, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, H.; Chen, Y.; Kibriya, M.G.; Islam, M.N.; Slavkovich, V.N.; Graziano, J.H.; Santella, R.M. Susceptibility to arsenic-induced hyperkeratosis and oxidative stress genes myeloperoxidase and catalase. Cancer Lett. 2003, 201, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Goth, L.; Eaton, J.W. Hereditary catalase deficiencies and increased risk of diabetes. Lancet 2000, 356, 1820–1821. [Google Scholar] [CrossRef] [PubMed]
- Goulas, A.; Fidani, L.; Kotsis, A.; Mirtsou, V.; Petersen, R.C.; Tangalos, E.; Hardy, J. An association study of a functional catalase gene polymorphism, −262C→T, and patients with Alzheimer’s disease. Neurosci. Lett. 2002, 330, 210–212. [Google Scholar] [CrossRef]
- Chistiakov, D.; Zotova, E.; Savost’Anov, K.; Bursa, T.; Galeev, I.; Strokov, I.; Nosikov, V. The 262T>C promoter polymorphism of the catalase gene is associated with diabetic neuropathy in type 1 diabetic Russian patients. Diabetes Metab. 2006, 32, 63–68. [Google Scholar] [CrossRef]
- Christiansen, L.; Petersen, H.C.; Bathum, L.; Frederiksen, H.; McGue, M.; Christensen, K. The catalase -262C/T promoter polymorphism and aging phenotypes. J. Gerontol. Ser. A 2004, 59, B886–B887. [Google Scholar] [CrossRef]
- He, J.; Li, X.; Li, Y.; Ren, B.; Sun, J.; Zhang, W.; Li, W. Lack of association between the 389C>T polymorphism (rs769217) in the catalase (CAT) gene and the risk of vitiligo: An update by meta-analysis. Australas. J. Dermatol. 2014, 56, 180–185. [Google Scholar] [CrossRef]
- Lu, L.; Ji, Y.; Jin, H.; He, L.; Liu, L. Association of the 389 C/T polymorphism of the catalase gene with susceptibility to vitiligo: A meta-analysis. Clin. Exp. Dermatol. 2014, 39, 454–460. [Google Scholar] [CrossRef]
- Góth, L.; Vitai, M. Hypocatalasemia in hospital patients. Clin. Chem. 1996, 42, 341–342. [Google Scholar] [CrossRef] [PubMed]
- Brady, S.C.; Allan, L.A.; Clarke, P.R. Regulation of Caspase 9 through Phosphorylation by Protein Kinase C Zeta in Response to Hyperosmotic Stress. Mol. Cell Biol. 2005, 25, 10543–10555. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.E.W.; Lingen, M.W.; Zhu, B.; Zhu, H.; Straza, M.W.; Pierce, C.; Martin, L.E.; Rosner, M.R. Protein Kinase Cζ Mediates Epidermal Growth Factor–Induced Growth of Head and Neck Tumor Cells by Regulating Mitogen-Activated Protein Kinase. Cancer Res. 2006, 66, 6296–6303. [Google Scholar] [CrossRef]
- Pinton, P.; Rimessi, A.; Marchi, S.; Orsini, F.; Migliaccio, E.; Giorgio, M.; Contursi, C.; Minucci, S.; Mantovani, F.; Wieckowski, M.R.; et al. Protein Kinase C ß and Prolyl Isomerase 1 Regulate Mitochondrial Effects of the Life-Span Determinant p66 Shc. Science 2007, 315, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Janssens, V.; Goris, J. Protein phosphatase 2A: A highly regulated family of serine/threonine phosphatases implicated in cell growth and signalling. Biochem. J. 2001, 353, 417–439. [Google Scholar] [CrossRef]
- Jung, K.J.; Kim, D.H.; Lee, E.K.; Song, C.W.; Yu, B.P.; Chung, H.Y. Oxidative stress induces inactivation of protein phosphatase 2A, promoting proinflammatory NF-κB in aged rat kidney. Free Radic. Biol. Med. 2013, 61, 206–217. [Google Scholar] [CrossRef]
- Zabana, Y.; Panés, J.; Nos, P.; Gomollón, F.; Esteve, M.; García-Sánchez, V.; Gisbert, J.P.; Barreiro-De-Acosta, M.; Domènech, E. El registro ENEIDA (Estudio Nacional en Enfermedad Inflamatoria intestinal sobre Determinantes genéticos y Ambientales) de GETECCU: Diseño, monitorización y funciones. Gastroenterol. Hepatol. 2020, 43, 551–558. [Google Scholar] [CrossRef]
- Hampe, J.; Cuthbert, A.; Croucher, P.J.; Mirza, M.M.; Mascheretti, S.; Fisher, S.; Frenzel, H.; King, K.; Hasselmeyer, A.; MacPherson, A.J.; et al. Association between insertion mutation in NOD2 gene and Crohn’s disease in German and British populations. Lancet 2001, 357, 1925–1928. [Google Scholar] [CrossRef]
- Silverberg, M.S.; Satsangi, J.; Ahmad, T.; Arnott, I.D.R.; Bernstein, C.N.; Brant, S.R.; Caprilli, R.; Colombel, J.-F.; Gasche, C.; Geboes, K.; et al. Toward an Integrated Clinical, Molecular and Serological Classification of Inflammatory Bowel Disease: Report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can. J. Gastroenterol. 2005, 19, 5A–36A. [Google Scholar] [CrossRef]
- Harvey, R.F.; Bradshaw, M.J. Measuring Crohn’s disease activity. Lancet 1980, 1, 1134–1135. [Google Scholar] [CrossRef]
- Rimola, J.; Rodriguez, S.; Garcia-Bosch, O.; Ordas, I.; Ayala, E.; Aceituno, M.; Pellise, M.; Ayuso, C.; Ricart, E.; Donoso, L.; et al. Magnetic resonance for assessment of disease activity and severity in ileocolonic Crohn’s disease. Gut 2009, 58, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Mary, J.Y.; Modigliani, R. Development and validation of an endoscopic index of the severity for Crohn’s disease: A prospective multicentre study. Groupe d’Etudes Therapeutiques des Affections Inflammatoires du Tube Digestif (GETAID). Gut 1989, 30, 983–989. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, S.; Ziaugra, L.; Tabbaa, D. SNP Genotyping Using the Sequenom MassARRAY iPLEX Platform. Curr. Protoc. Hum. Genet. 2009, 60, 2.12.1–2.12.18. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
| Control (n = 625) | CD (n = 598) | ||
|---|---|---|---|
| Age | 47.34 ± 8.54 | 42.06 ± 15.17 | |
| Gender (M/F) | 389/236 | 281/317 | |
| Montreal Classification | |||
| A | A1: | 30 (5%) | |
| A2: | 401 (67%) | ||
| A3: | 167 (28%) | ||
| L | L1: | 227 (38%) | |
| L2: | 113 (19%) | ||
| L3: | 251 (42%) | ||
| L4: | 28 (4%) | ||
| B | B1: | 376 (63%) | |
| B2: | 132 (22%) | ||
| B3: | 90 (15%) | ||
| Perianal disease | 148 (24%) | ||
| Treatments: | 363 (82%) | ||
| Immunossupressors | 371 (62%) | ||
| Biologic therapy | |||
| Family history | 82 (15%) | ||
| Extraintestinal manifestations | 119 (20%) | ||
| Surgeries | 271 (45%) | ||
| Smokers | 185 (31%) | ||
| Nonsmokers | 269 (45%) | ||
| Exsmokers | 144 (24%) | ||
| CAT SNPs | Genotype | Significant Clinical Associations |
|---|---|---|
| rs1001179 | CC | Protect perianal disease |
| rs12273124 | AG | Increase inflammatory behavior in smokers |
| rs3758730 | TA | Protect stricturing behavior and ileocolonic location |
| TT | Increase stricturing behavior in smokers | |
| rs475043 | AG | Increase upper digestive location and penetrating behavior in smokers |
| GG | Protect upper digestive location | |
| rs494024 | GA | Protect penetrating behavior |
| GG | Protect inflammatory behavior | |
| rs564250 | TC | Increase inflammatory behavior in smokers |
| TT | Increase penetrating behavior |
| rs7943316 | Control | Patient | p Value |
|---|---|---|---|
| AA | 10 | 7 | 0.04 * |
| AT | 55 | 61 | |
| TT | 35 | 32 | |
| rs3758730 | |||
| AA | 76.5 | 76.5 | 0.23 |
| TA | 22 | 23 | |
| TT | 1.5 | 0.5 | |
| rs475043 | |||
| AA | 45 | 40 | 0.036 * |
| AG | 46 | 53 | |
| GG | 9 | 7 | |
| rs769217 | |||
| CC | 45 | 51 | 0.041 * |
| CT | 49 | 45 | |
| TT | 6 | 4 | |
| rs525938 | |||
| AA | 38 | 46 | 0.007 * |
| AG | 53 | 48 | |
| GG | 9 | 6 | |
| rs564250 | |||
| CC | 51 | 57 | 0.089 |
| CT | 46 | 41 | |
| TT | 3 | 2 | |
| rs1001179 | |||
| AA | 3 | 2 | 0.27 |
| GA | 36 | 36 | |
| GG | 31 | 62 |
| KERRYPNX | Control (n = 18) | aCD (n = 20) | iCD (n = 10) |
|---|---|---|---|
| Age | 27.83 ± 5.24 | 32.04 ± 9.14 | 32.1 ± 10.1 |
| Gender (M/F) | 4/14 | 7/13 | 6/4 |
| Harvey Index | -- | 7,31 ± 2,86 | <4 |
| CRP | 2.8 (2) | 70.31 ± 95.24 a,b | 3 ± 3.97 |
| Fibrinogen | 287 ± 69 | 531.42 ± 129.18 a | 349 ± 81.4 |
| ESR | 11.8 ± 12 | 57.87 ± 31.6 a,b | 10.25 ± 10.27 |
| S/nS/eS | 0/14/2 | 11/8/1 | 2/7/1 |
| Montreal Classification | |||
| A | |||
| A1: | - | 0 (0%) | 0 (0%) |
| A2: | - | 20 (100%) | 10 (100%) |
| A3: | - | 0 (0%) | 0 (0%) |
| L | |||
| L1: | - | 13 (65%) | 7 (70%) |
| L2: | - | 3 (15%) | 3 (30%) |
| L3: | - | 4 (20%) | 0 (0%) |
| L4: | - | 0 (0%) | 0 (0%) |
| B | |||
| B1 | - | 18 (90%) | 8 (80%) |
| B2: | - | 2 (10%) | 2 (20%) |
| B3: | - | 0 (0%) | 0 (0%) |
| p | |||
| Yes: | - | 4 (20%) | 0 (0%) |
| No: | - | 16 (80%) | 10 (100%) |
| No: | |||
| Time until remission (months) | 14.87 ± 10.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iborra, M.; Moret, I.; Busó, E.; García-Giménez, J.L.; Ricart, E.; Gisbert, J.P.; Cabré, E.; Esteve, M.; Márquez-Mosquera, L.; García-Planella, E.; et al. The Genetic Diversity and Dysfunctionality of Catalase Associated with a Worse Outcome in Crohn’s Disease. Int. J. Mol. Sci. 2022, 23, 15881. https://doi.org/10.3390/ijms232415881
Iborra M, Moret I, Busó E, García-Giménez JL, Ricart E, Gisbert JP, Cabré E, Esteve M, Márquez-Mosquera L, García-Planella E, et al. The Genetic Diversity and Dysfunctionality of Catalase Associated with a Worse Outcome in Crohn’s Disease. International Journal of Molecular Sciences. 2022; 23(24):15881. https://doi.org/10.3390/ijms232415881
Chicago/Turabian StyleIborra, Marisa, Inés Moret, Enrique Busó, José Luis García-Giménez, Elena Ricart, Javier P. Gisbert, Eduard Cabré, Maria Esteve, Lucía Márquez-Mosquera, Esther García-Planella, and et al. 2022. "The Genetic Diversity and Dysfunctionality of Catalase Associated with a Worse Outcome in Crohn’s Disease" International Journal of Molecular Sciences 23, no. 24: 15881. https://doi.org/10.3390/ijms232415881
APA StyleIborra, M., Moret, I., Busó, E., García-Giménez, J. L., Ricart, E., Gisbert, J. P., Cabré, E., Esteve, M., Márquez-Mosquera, L., García-Planella, E., Guardiola, J., Pallardó, F. V., Serena, C., Algaba-Chueca, F., Domenech, E., Nos, P., & Beltrán, B. (2022). The Genetic Diversity and Dysfunctionality of Catalase Associated with a Worse Outcome in Crohn’s Disease. International Journal of Molecular Sciences, 23(24), 15881. https://doi.org/10.3390/ijms232415881







