EPH/Ephrin-Targeting Treatment in Breast Cancer: A New Chapter in Breast Cancer Therapy
Abstract
1. Introduction
2. EPH/Ephrin-Targeting Therapy in BC
3. EPHA2-Targeting Therapeutic Agents
4. EPHA10-Targeting Therapeutic Agents
5. EPHB4-Targeting Therapeutic Agents
6. Ephrin-A2-Targeting Therapeutic Agents
7. Ephrin-A4-Targeting Therapeutic Agents
8. Ephrin-B2-Targeting Therapeutic Agents
9. Multiple EPH/ephrin-Targeting Therapeutic Agents
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Key Statistics for Breast Cancer; American Cancer Society: Atlanta, GA, USA, 2022.
- Breast Cancer Signs and Symptoms; American Cancer Society: Atlanta, GA, USA, 2022.
- Breast Cancer Early Detection and Diagnosis; American Cancer Society: Atlanta, GA, USA, 2022.
- Treatment of Breast Cancer by Stage; American Cancer Society: Atlanta, GA, USA, 2019.
- Eph Nomenclature Committee. Unified nomenclature for Eph family receptors and their ligands, the ephrins. Cell 1997, 90, 403–404. [Google Scholar] [CrossRef]
- Wei, Q.; Liu, J.; Wang, N.; Zhang, X.; Jin, J.; Chin-Sang, I.; Zheng, J.; Jia, Z. Structures of an Eph receptor tyrosine kinase and its potential activation mechanism. Acta Crystallogr. Sect. D Biol. Crystallogr. 2014, 70, 3135–3143. [Google Scholar] [CrossRef]
- Kania, A.; Klein, R. Mechanisms of ephrin-Eph signalling in development, physiology and disease. Nat. Rev. Mol. Cell Biol. 2016, 17, 240–256. [Google Scholar] [CrossRef]
- Shiuan, E.; Chen, J. Eph Receptor Tyrosine Kinases in Tumor Immunity. Cancer Res. 2016, 76, 6452–6457. [Google Scholar] [CrossRef]
- Yamada, T.; Yuasa, M.; Masaoka, T.; Taniyama, T.; Maehara, H.; Torigoe, I.; Yoshii, T.; Shinomiya, K.; Okawa, A.; Sotome, S. After repeated division, bone marrow stromal cells express inhibitory factors with osteogenic capabilities, and EphA5 is a primary candidate. Bone 2013, 57, 343–354. [Google Scholar] [CrossRef]
- Coulthard, M.G.; Duffy, S.; Down, M.; Evans, B.; Power, M.; Smith, F.; Stylianou, C.; Kleikamp, S.; Oates, A.; Lackmann, M.; et al. The role of the Eph-ephrin signalling system in the regulation of developmental pat-terning. Int. J. Dev. Biol. 2002, 46, 375–384. [Google Scholar]
- Rudno-Rudzińska, J.; 2-nd Department of General and Oncological Surgery; Kielan, W.; Frejlich, E.; Kotulski, K.; Hap, W.; Kurnol, K.; Dzierżek, P.; Zawadzki, M.; Hałoń, A. A review on Eph/ephrin, angiogenesis and lymphangiogenesis in gastric, colorectal and pancreatic cancers. Chin. J. Cancer Res. 2017, 29, 303–312. [Google Scholar] [CrossRef]
- Pergaris, A.; Danas, E.; Goutas, D.; Sykaras, A.; Soranidis, A.; Theocharis, S. The Clinical Impact of the EPH/Ephrin System in Cancer: Unwinding the Thread. Int. J. Mol. Sci. 2021, 22, 8412. [Google Scholar] [CrossRef]
- Hadjimichael, A.C.; Pergaris, A.; Kaspiris, A.; Foukas, A.F.; Kokkali, S.; Tsourouflis, G.; Theocharis, S. The EPH/Ephrin System in Bone and Soft Tissue Sarcomas’ Pathogenesis and Therapy: New Advancements and a Literature Review. Int. J. Mol. Sci. 2022, 23, 5171. [Google Scholar] [CrossRef]
- Masaoutis, C.; Georgantzoglou, N.; Sarantis, P.; Theochari, I.; Tsoukalas, N.; Bobos, M.; Alexandrou, P.; Pergaris, A.; Rontogianni, D.; Theocharis, S. Ephrin Receptors (Ephs) Expression in Thymic Epithelial Tumors: Prognostic Implications and Future Therapeutic Approaches. Diagnostics 2021, 11, 2265. [Google Scholar] [CrossRef]
- Papadakos, S.P.; Petrogiannopoulos, L.; Pergaris, A.; Theocharis, S. The EPH/Ephrin System in Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 2761. [Google Scholar] [CrossRef]
- Psilopatis, I.; Pergaris, A.; Vrettou, K.; Tsourouflis, G.; Theocharis, S. The EPH/Ephrin System in Gynecological Cancers: Focusing on the Roots of Carcinogenesis for Better Patient Management. Int. J. Mol. Sci. 2022, 23, 3249. [Google Scholar] [CrossRef]
- Kaenel, P.; Mosimann, M.; Andres, A.-C. The multifaceted roles of Eph/ephrin signaling in breast cancer. Cell Adhes. Migr. 2012, 6, 138–147. [Google Scholar] [CrossRef]
- Perez White, B.E.; Getsios, S. Eph receptor and ephrin function in breast, gut, and skin epithelia. Cell Adhes. Migr. 2014, 8, 327–338. [Google Scholar] [CrossRef]
- Vaught, D.; Brantley-Sieders, D.M.; Chen, J. Eph receptors in breast cancer: Roles in tumor promotion and tumor suppression. Breast Cancer Res. 2008, 10, 217. [Google Scholar] [CrossRef]
- Nikas, I.; Ryu, H.S.; Theocharis, S. Viewing the Eph receptors with a focus on breast cancer heterogeneity. Cancer Lett. 2018, 434, 160–171. [Google Scholar] [CrossRef]
- Anderton, M.; van der Meulen, E.; Blumenthal, M.J.; Schäfer, G. The Role of the Eph Receptor Family in Tumorigenesis. Cancers 2021, 13, 206. [Google Scholar] [CrossRef]
- Zhao, P.; Jiang, D.; Huang, Y.; Chen, C. EphA2: A promising therapeutic target in breast cancer. J. Genet. Genom. 2021, 48, 261–267. [Google Scholar] [CrossRef]
- Huang, F.; Reeves, K.; Han, X.; Fairchild, C.; Platero, S.; Wong, T.W.; Lee, F.; Shaw, P.; Clark, E. Identification of candidate molecular markers predicting sensitivity in solid tumors to da-satinib: Rationale for patient selection. Cancer Res. 2007, 67, 2226–2238. [Google Scholar] [CrossRef]
- Torres-Adorno, A.M.; Vitrac, H.; Qi, Y.; Tan, L.; Levental, K.R.; Fan, Y.-Y.; Yang, P.; Chapkin, R.S.; Eckhardt, B.L.; Ueno, N.T. Eicosapentaenoic acid in combination with EPHA2 inhibition shows efficacy in preclinical models of triple-negative breast cancer by disrupting cellular cholesterol efflux. Oncogene 2019, 38, 2135–2150. [Google Scholar] [CrossRef]
- Cha, J.-H.; Chan, L.-C.; Wang, Y.-N.; Chu, Y.-Y.; Wang, C.-H.; Lee, H.-H.; Xia, W.; Shyu, W.-C.; Liu, S.-P.; Yao, J.; et al. Ephrin receptor A10 monoclonal antibodies and the derived chimeric antigen receptor T cells exert an antitumor response in mouse models of triple-negative breast cancer. J. Biol. Chem. 2022, 298. [Google Scholar] [CrossRef]
- Nagano, K.; Maeda, Y.; Kanasaki, S.-I.; Watanabe, T.; Yamashita, T.; Inoue, M.; Higashisaka, K.; Yoshioka, Y.; Abe, Y.; Mukai, Y.; et al. Ephrin receptor A10 is a promising drug target potentially useful for breast cancers including triple negative breast cancers. J. Control. Release 2014, 189, 72–79. [Google Scholar] [CrossRef]
- Taki, S.; Kamada, H.; Inoue, M.; Nagano, K.; Mukai, Y.; Higashisaka, K.; Yoshioka, Y.; Tsutsumi, Y.; Tsunoda, S.-I. A Novel Bispecific Antibody against Human CD3 and Ephrin Receptor A10 for Breast Cancer Therapy. PLoS ONE 2015, 10, e0144712. [Google Scholar] [CrossRef]
- Damelin, M.; Bankovich, A.; Park, A.; Aguilar, J.; Anderson, W.; Santaguida, M.; Aujay, M.; Fong, S.; Khandke, K.; Pulito, V.; et al. Anti-EFNA4 Calicheamicin Conjugates Effectively Target Triple-Negative Breast and Ovarian Tumor-Initiating Cells to Result in Sustained Tumor Regressions. Clin. Cancer Res. 2015, 21, 4165–4173. [Google Scholar] [CrossRef]
- Garrido-Laguna, I.; Krop, I.; Burris, H.A., 3rd; Hamilton, E.; Braiteh, F.; Weise, A.M.; Abu-Khalaf, M.; Werner, T.L.; Pirie-Shepherd, S.; Zopf, C.J.; et al. First-in-human, phase I study of PF-06647263, an anti-EFNA4 calicheamicin antibody-drug conjugate, in patients with advanced solid tumors. Int. J. Cancer 2019, 145, 1798–1808. [Google Scholar] [CrossRef]
- Huang, Z.R.; Tipparaju, S.K.; Kirpotin, D.B.; Christine, P.; Tad, K.; Noble, C.O.; Alexander, K.; Jimmy, T.; Kamoun, W.S.; Drummond, D.C. Formulation optimization of an ephrin A2 targeted immunoliposome encapsu-lating reversibly modified taxane prodrugs. J. Control Release 2019, 310, 47–57. [Google Scholar] [CrossRef]
- Noblitt, L.W.; Bangari, D.S.; Shukla, S.; Knapp, D.W.; Mohammed, S.I.; Kinch, M.S.; Mittal, S.K. Decreased tumorigenic potential of EphA2-overexpressing breast cancer cells following treatment with adenoviral vectors that express EphrinA1. Cancer Gene Ther. 2004, 11, 757–766. [Google Scholar] [CrossRef]
- Tandon, M.; Vemula, S.V.; Sharma, A.; Ahi, Y.; Mittal, S.; Bangari, D.S.; Mittal, S.K. EphrinA1-EphA2 interaction-mediated apoptosis and FMS-like tyrosine kinase 3 receptor ligand-induced immunotherapy inhibit tumor growth in a breast cancer mouse model. J. Gene Med. 2012, 14, 77–89. [Google Scholar] [CrossRef]
- Wykosky, J.; Gibo, D.M.; Debinski, W. A novel, potent, and specific ephrinA1-based cytotoxin against EphA2 receptor expressing tumor cells. Mol. Cancer Ther. 2007, 6, 3208–3218. [Google Scholar] [CrossRef]
- Su, Q.; Wang, J.; Wu, Q.; Ullah, A.; Ghauri, M.A.; Sarwar, A.; Chen, L.; Liu, F.; Zhang, Y. Sanguinarine combats hypoxia-induced activation of EphB4 and HIF-1α pathways in breast cancer. Phytomedicine 2021, 84, 153503. [Google Scholar] [CrossRef]
- Ma, W.; Zhu, M.; Zhang, D.; Yang, L.; Yang, T.; Li, X.; Zhang, Y. Berberine inhibits the proliferation and migration of breast cancer ZR-75-30 cells by targeting Ephrin-B2. Phytomedicine 2017, 25, 45–51. [Google Scholar] [CrossRef]
- Zadeh, T.; Lucero, M.; Kandpal, R.P. Artesunate-induced Cellular Effects Are Mediated by Specific EPH Receptors and Ephrin Ligands in Breast Carcinoma Cells. Cancer Genom.-Proteom. 2021, 19, 19–26. [Google Scholar] [CrossRef]
- Levêque, D.; Becker, G.; Bilger, K.; Natarajan-Amé, S. Clinical Pharmacokinetics and Pharmacodynamics of Dasatinib. Clin. Pharmacokinet. 2020, 59, 849–856. [Google Scholar] [CrossRef]
- Chang, Q.; Jorgensen, C.; Pawson, T.; Hedley, D.W. Effects of dasatinib on EphA2 receptor tyrosine kinase activity and downstream signalling in pancreatic cancer. Br. J. Cancer 2008, 99, 1074–1082. [Google Scholar] [CrossRef]
- Curiel, D.T. The development of conditionally replicative adenoviruses for cancer therapy. Clin. Cancer Res. 2000, 6, 3395–3399. [Google Scholar]
- Hitt, M.M.; Graham, F.L. Adenovirus vectors for human gene therapy. Adv. Virus Res. 2000, 55, 479–505. [Google Scholar] [CrossRef]
- St George, J.A. Gene therapy progress and prospects: Adenoviral vectors. Gene Ther. 2003, 10, 1135–1141. [Google Scholar] [CrossRef]
- Udompholkul, P.; Baggio, C.; Gambini, L.; Sun, Y.; Zhao, M.; Hoffman, R.; Pellecchia, M. Effective Tumor Targeting by EphA2-Agonist-Biotin-Streptavidin Conjugates. Molecules 2021, 26, 3687. [Google Scholar] [CrossRef]
- Zahavi, D.; Weiner, L. Monoclonal Antibodies in Cancer Therapy. Antibodies 2020, 9, 34. [Google Scholar] [CrossRef]
- Basu, P.; Kumar, G.S. Sanguinarine and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016, 928, 155–172. [Google Scholar] [CrossRef]
- Paszko, E.; Senge, M.O. Immunoliposomes. Curr. Med. Chem. 2012, 19, 5239–5277. [Google Scholar] [CrossRef]
- Barneh, F.; Moshayedi, M.; Sadeghi, H.M.-M.; Haghjooy-Javanmard, S.; Sabzghabaee, A.M.; Badri, S. EphB4 Tyrosine Kinase Stimulation Inhibits Growth of MDA-MB-231 Breast Cancer Cells in a Dose and Time Dependent Manner. Dis. Mark. 2013, 35, 933–938. [Google Scholar] [CrossRef]
- Noren, N.K.; Foos, G.; Hauser, C.A.; Pasquale, E.B. The EphB4 receptor suppresses breast cancer cell tumorigenicity through an Abl–Crk pathway. Nat. Cell Biol. 2006, 8, 815–825. [Google Scholar] [CrossRef]
- Ye, Y.; Liu, X.; Wu, N.; Han, Y.; Wang, J.; Yu, Y.; Chen, Q. Efficacy and Safety of Berberine Alone for Several Metabolic Disorders: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Front. Pharmacol. 2021, 12, 653887. [Google Scholar] [CrossRef]
- Kouakou, Y.I.; Tod, M.; Leboucher, G.; Lavoignat, A.; Bonnot, G.; Bienvenu, A.L.; Picot, S. Systematic review of artesunate pharmacokinetics: Implication for treatment of re-sistant malaria. Int. J. Infect. Dis. 2019, 89, 30–44. [Google Scholar] [CrossRef]
- Xi, H.-Q.; Wu, X.-S.; Wei, B.; Chen, L. Eph receptors and ephrins as targets for cancer therapy. J. Cell. Mol. Med. 2012, 16, 2894–2909. [Google Scholar] [CrossRef]
- Buckens, O.J.; El Hassouni, B.; Giovannetti, E.; Peters, G.J. The role of Eph receptors in cancer and how to target them: Novel ap-proaches in cancer treatment. Expert Opin. Investig. Drugs 2020, 29, 567–582. [Google Scholar] [CrossRef]
- Lodola, A.; Giorgio, C.; Incerti, M.; Zanotti, I.; Tognolini, M. Targeting Eph/ephrin system in cancer therapy. Eur. J. Med. Chem. 2017, 142, 152–162. [Google Scholar] [CrossRef]
- Survival Rates for Breast Cancer; American Cancer Society: Atlanta, GA, USA, 2022.
| Targeting Strategy | Mechanism of Action | References |
|---|---|---|
| Small molecule drugs | Tyrosine kinase inhibitors | [23,24] |
| Monoclonal antibodies | Anti-EPH/ephrin monoclonal antibodies, Antibody-drug conjugations | [25,26,27,28,29] |
| Immunoliposomes | Antibodies coupled to the liposomal surface | [30] |
| Adenoviral-based cancer therapy | Human adenovirus-based vectors | [31,32] |
| Natural compounds | Cytotoxins, Alkaloids, Artemisinin | [33,34,35,36] |
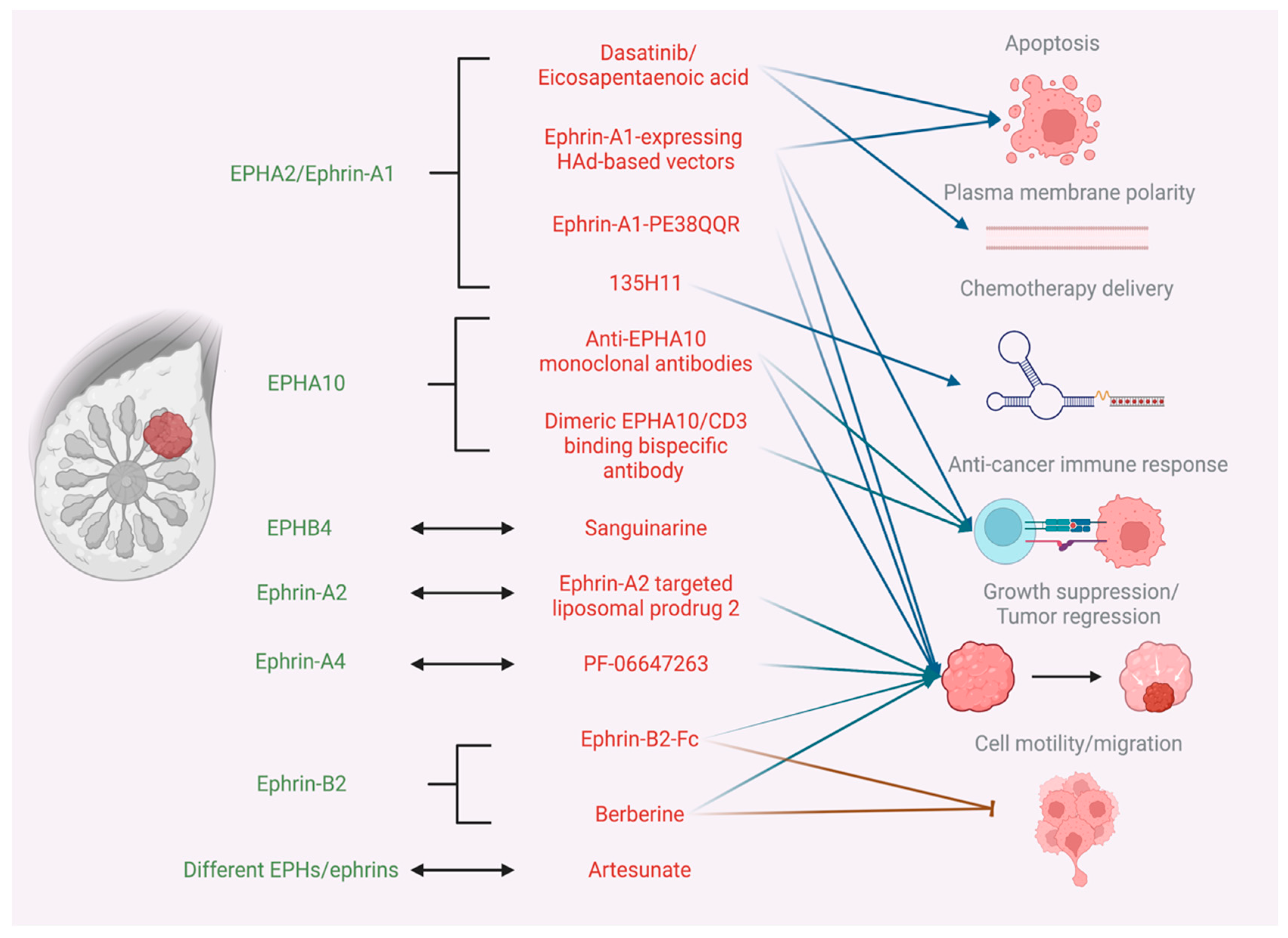
| Therapeutic Agent | Targeted EPH/Ephrin | Mechanism of Action | References |
|---|---|---|---|
| Dasatinib/ Eicosapentaenoic acid | EPHA2 | • Reduction in EPHA2 expression, phosphorylation, and kinase activity • Plasma membrane polarity increase through ABCA1-dependent cholesterol accumulation • Apoptosis induction | [23,24] |
| Ephrin-A1-expressing HAd-based vectors | Ephrin-A1/EPHA2 | • EPHA2 upregulation • Decreased BC cell viability • Inhibition of tumor formation • Apoptosis induction • Anticancer adaptive immune response activation | [31,32] |
| Ephrin-A1-PE38QQR | Ephrin-A1/EPHA2 | • Cytotoxic effects on BC cells | [33] |
| 135H11 | EPHA2 | • Agonistic activity only after successful dimerization or clustering • Selective chemotherapy delivery | [42] |
| Anti-EPHA10 monoclonal antibodies | EPHA10 | • Tumor regression induction • BC growth suppression • CD8+ tumor-infiltrating CTL activation • EPHA10 internalization | [25,26] |
| Dimeric EPHA10/CD3 binding bispecific antibody | EPHA10 | • Anticancer T-cell stimulation | [27] |
| Sanguinarine | EPHB4 | • Down regulation of hypoxia- induced EPHB4 • STAT3 activation | [34] |
| Ephrin-A2 targeted liposomal prodrug 2 | Ephrin-A2 | • BC tumor regression | [30] |
| PF-06647263 | Ephrin-A4 | • BC tumor regression | [28,29] |
| Ephrin-B2-Fc | Ephrin-B2/EPHB4 | • Dose- dependent, EPHB4- mediated BC growth inhibition • Inhibition of EPHB4- mediated, Abl-Crk -dependent BC cell growth, motility, and invasion | [46,47] |
| Berberine | Ephrin-B2 | • Reduced ZR-75-30 BC cell proliferation and migration | [35] |
| Artesunate | EPHA3, EPHA7, EPHA8, EPHA10, EPHB6, ephrin-A2, ephrin-A3 | • EPHA8, EPHA10, EPHB6 and ephrin-A2 upregulation in MCF7 cells • EPHA3 and EPHA10 upregulation in MDA-MB-231 cells • EPHA7 and ephrin-A3 down regulation in MDA-MB-231 cells | [36] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Psilopatis, I.; Souferi-Chronopoulou, E.; Vrettou, K.; Troungos, C.; Theocharis, S. EPH/Ephrin-Targeting Treatment in Breast Cancer: A New Chapter in Breast Cancer Therapy. Int. J. Mol. Sci. 2022, 23, 15275. https://doi.org/10.3390/ijms232315275
Psilopatis I, Souferi-Chronopoulou E, Vrettou K, Troungos C, Theocharis S. EPH/Ephrin-Targeting Treatment in Breast Cancer: A New Chapter in Breast Cancer Therapy. International Journal of Molecular Sciences. 2022; 23(23):15275. https://doi.org/10.3390/ijms232315275
Chicago/Turabian StylePsilopatis, Iason, Eleni Souferi-Chronopoulou, Kleio Vrettou, Constantinos Troungos, and Stamatios Theocharis. 2022. "EPH/Ephrin-Targeting Treatment in Breast Cancer: A New Chapter in Breast Cancer Therapy" International Journal of Molecular Sciences 23, no. 23: 15275. https://doi.org/10.3390/ijms232315275
APA StylePsilopatis, I., Souferi-Chronopoulou, E., Vrettou, K., Troungos, C., & Theocharis, S. (2022). EPH/Ephrin-Targeting Treatment in Breast Cancer: A New Chapter in Breast Cancer Therapy. International Journal of Molecular Sciences, 23(23), 15275. https://doi.org/10.3390/ijms232315275









