A Pleomorphic Puzzle: Heterogeneous Pulmonary Vascular Occlusions in Patients with COVID-19
Abstract
1. Introduction
2. Results
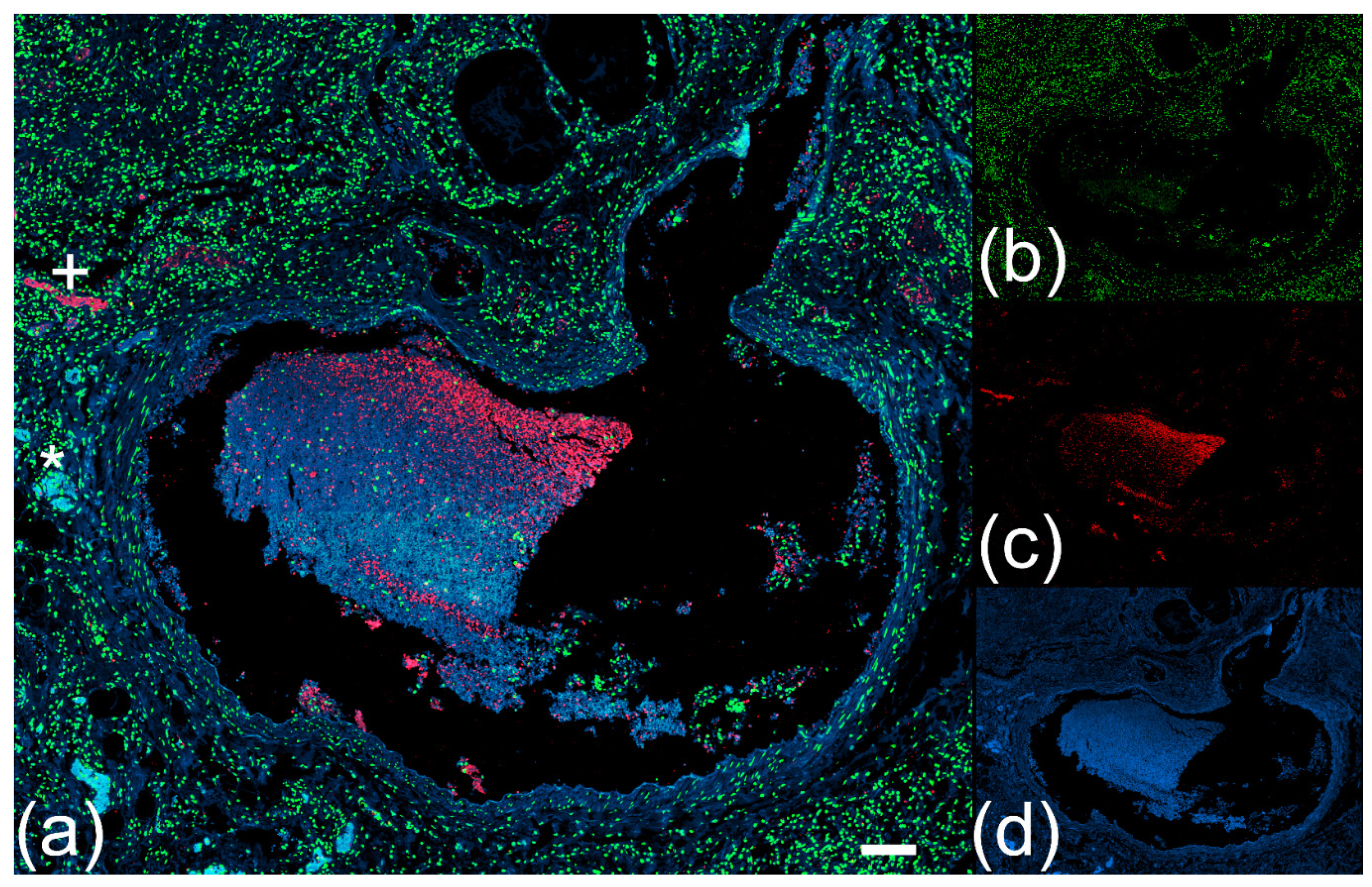
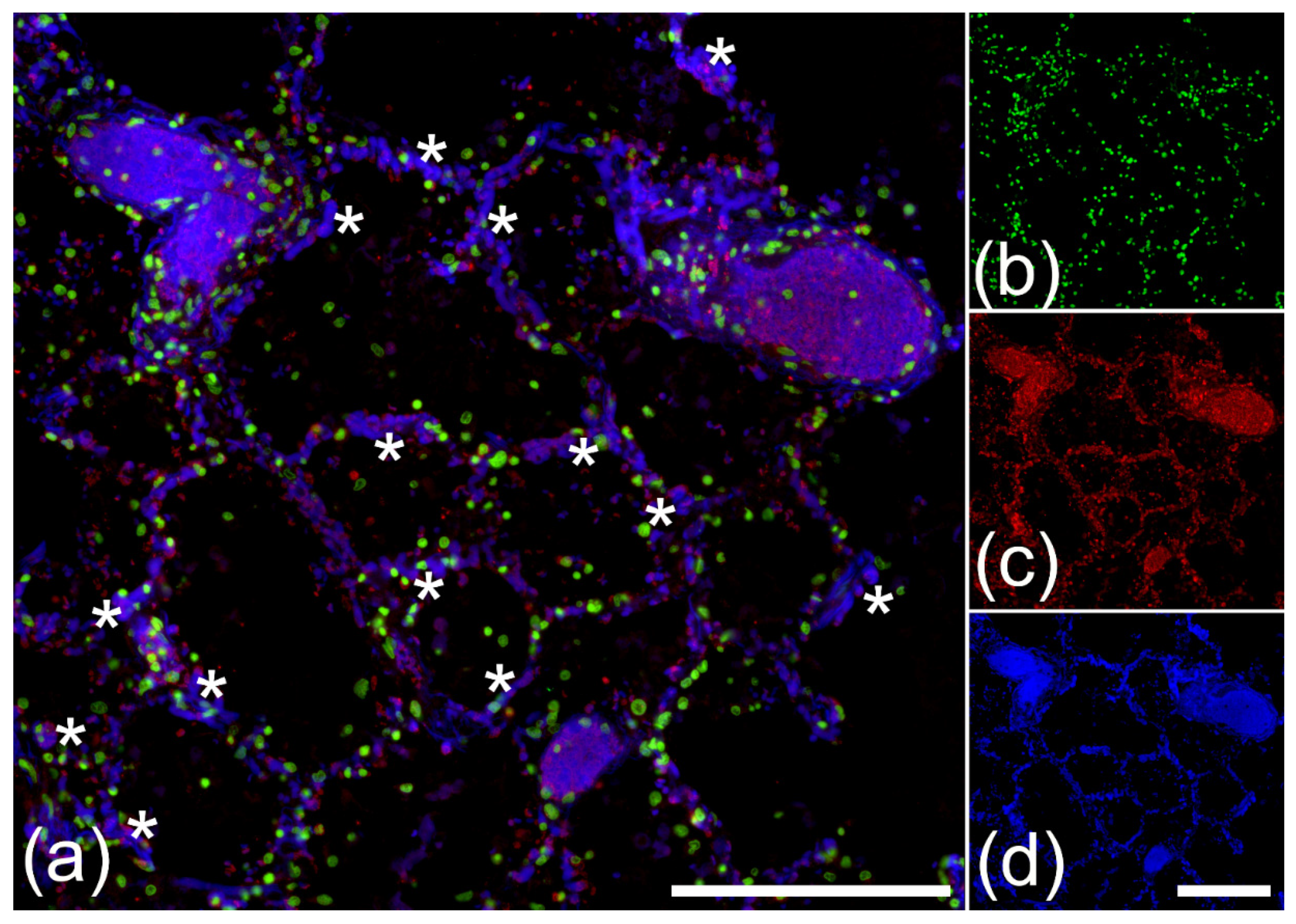
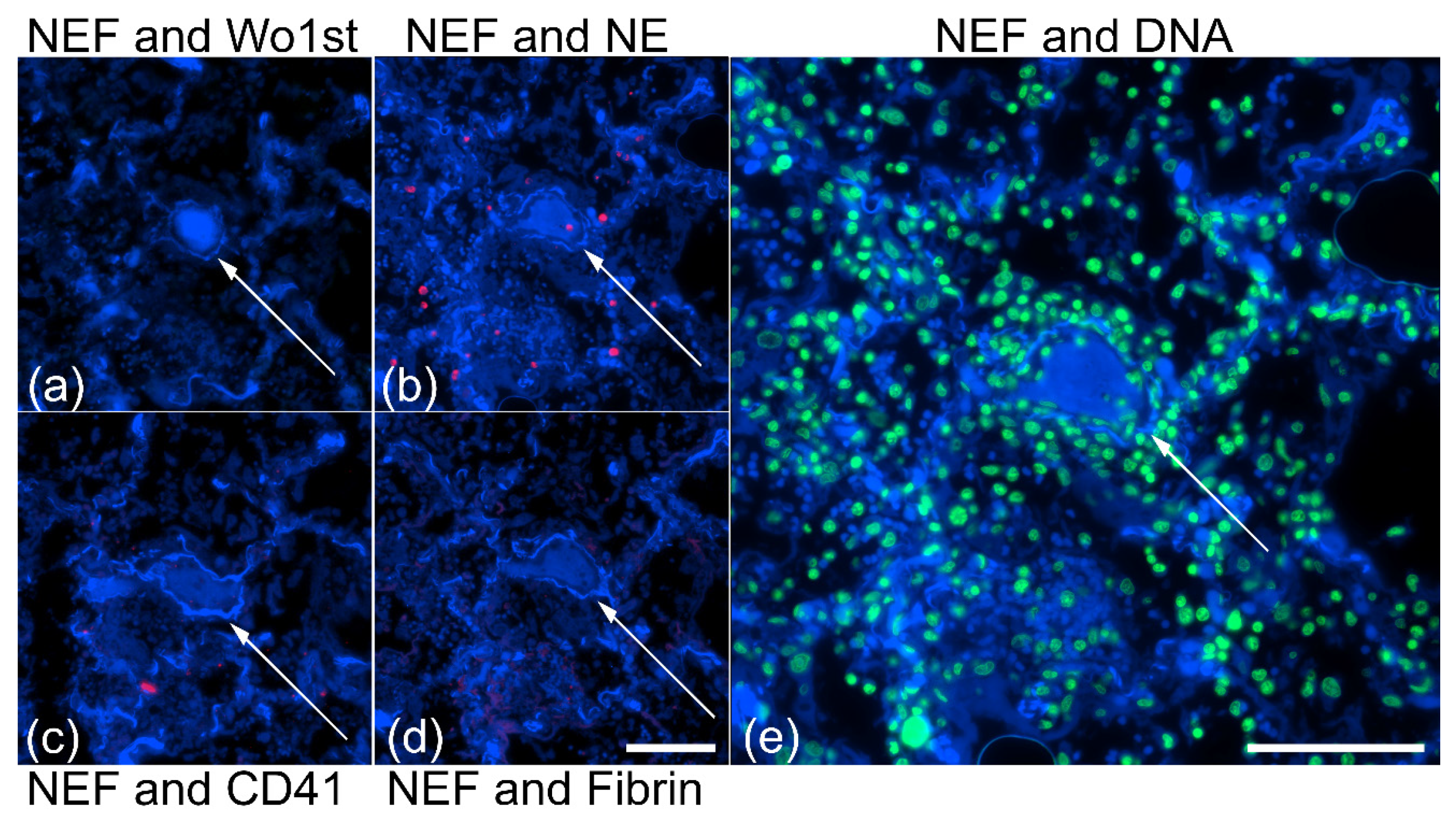
2.1. NEF of Vascular Occlusion Shows Size Heterogeneity
2.2. NEF from Vascular Occlusions Is Associated with Hemoglobin-α
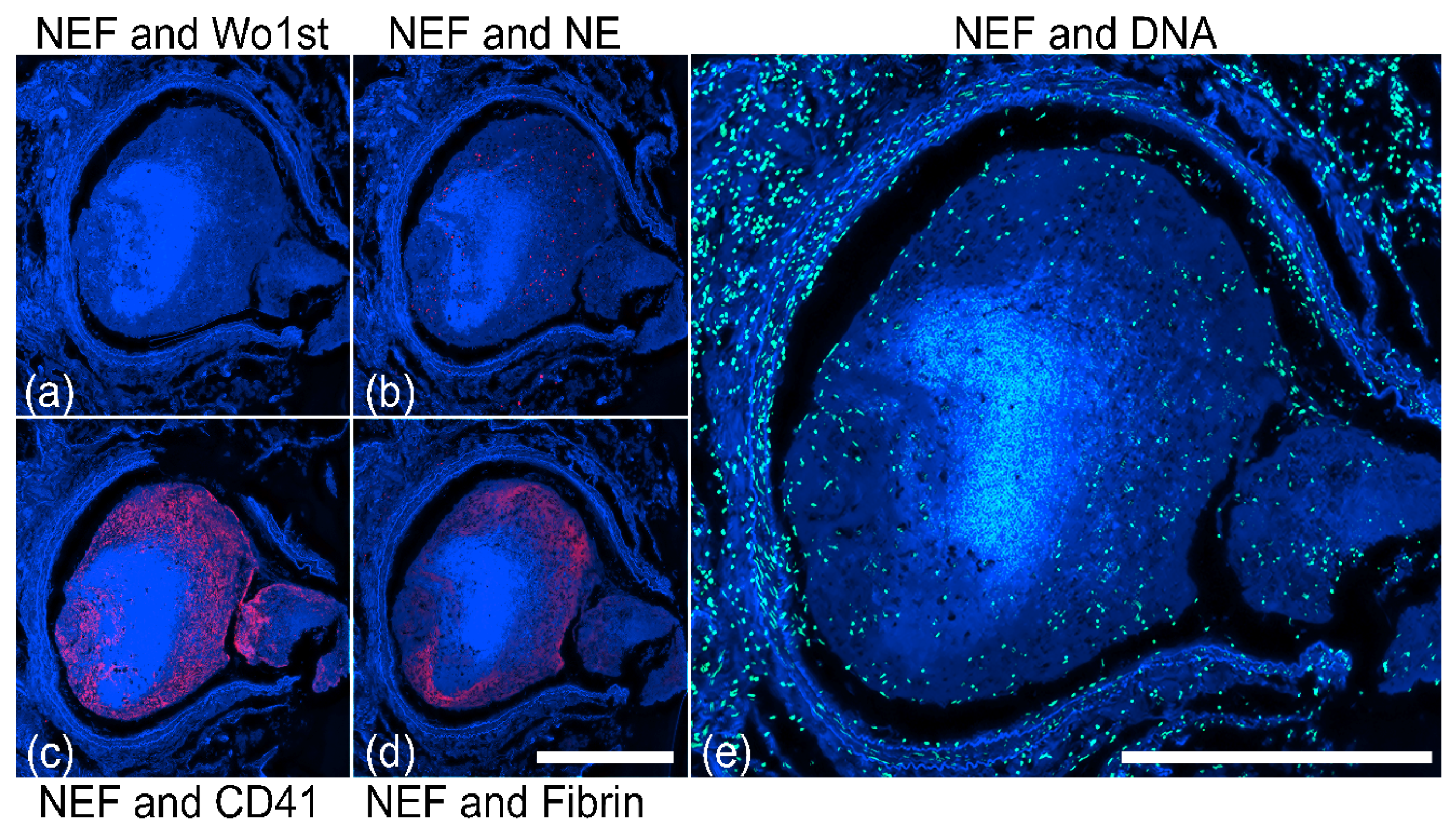
2.3. NEF-Detected Vascular Occlusions Exhibit Pleomorphic Composition of Platelets and Fibrin
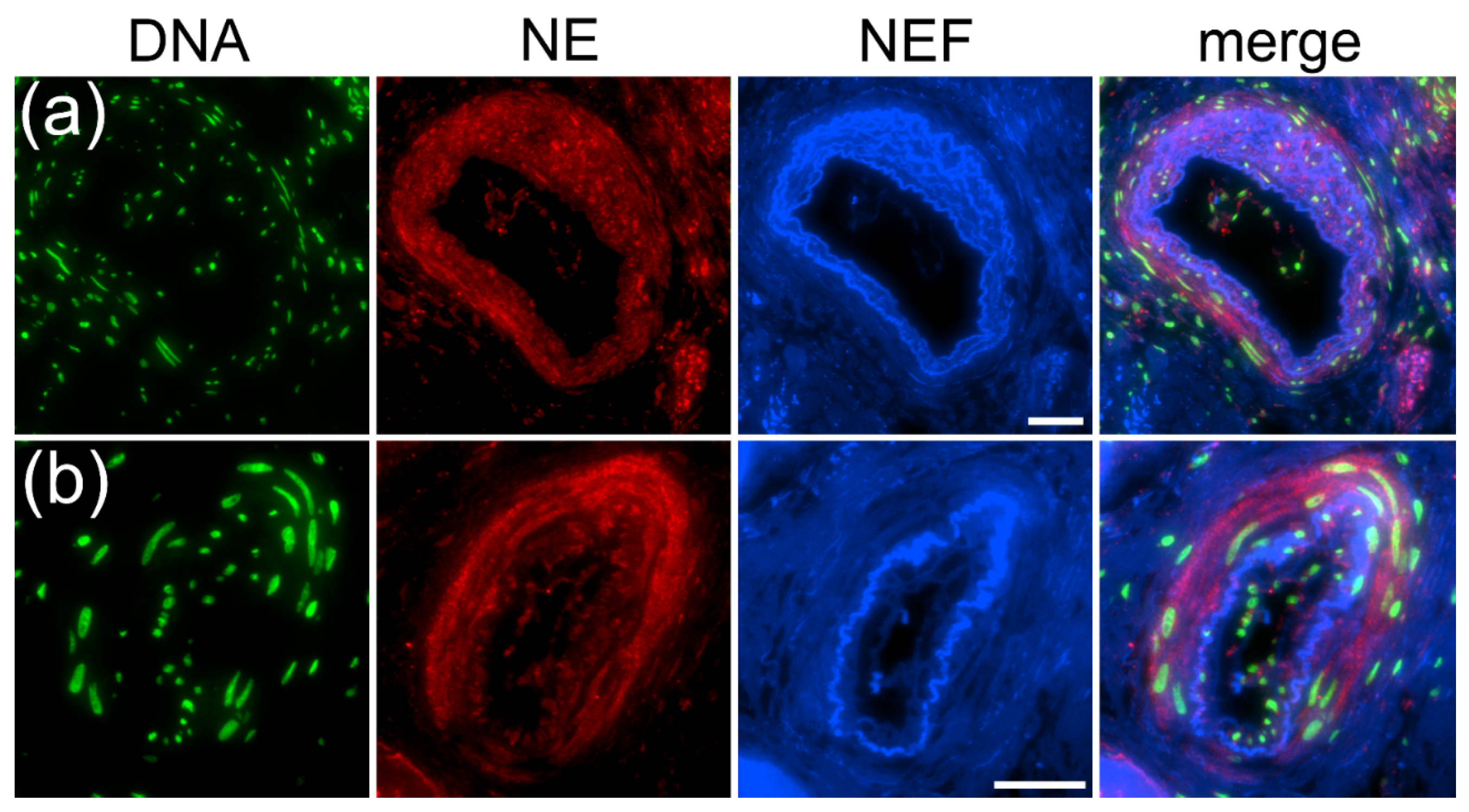
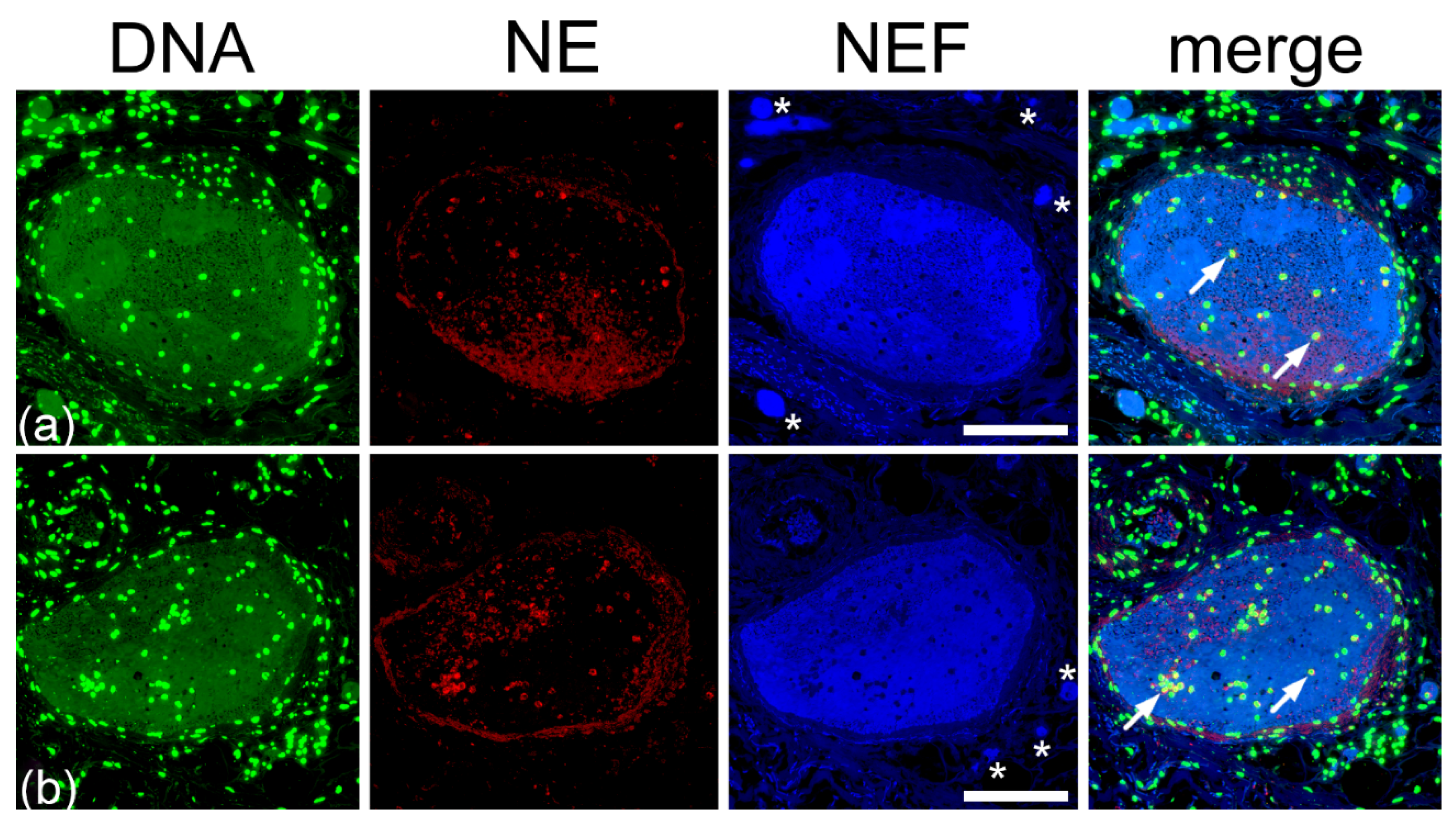
2.4. Type I Pleomorphism: DNA-Rich and NE-Poor
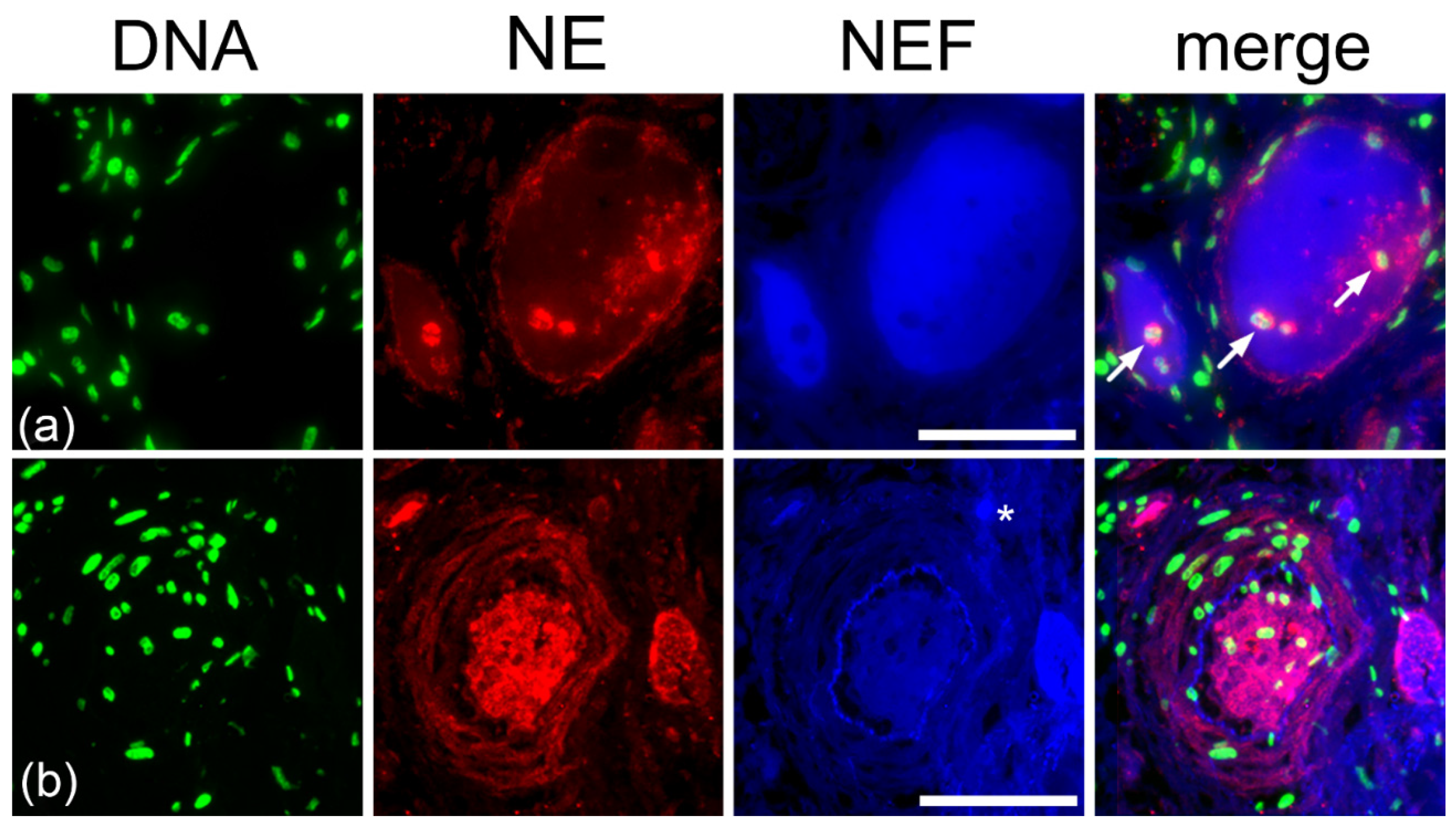
2.5. Type II Pleomorphism: NE-Rich and DNA-Poor
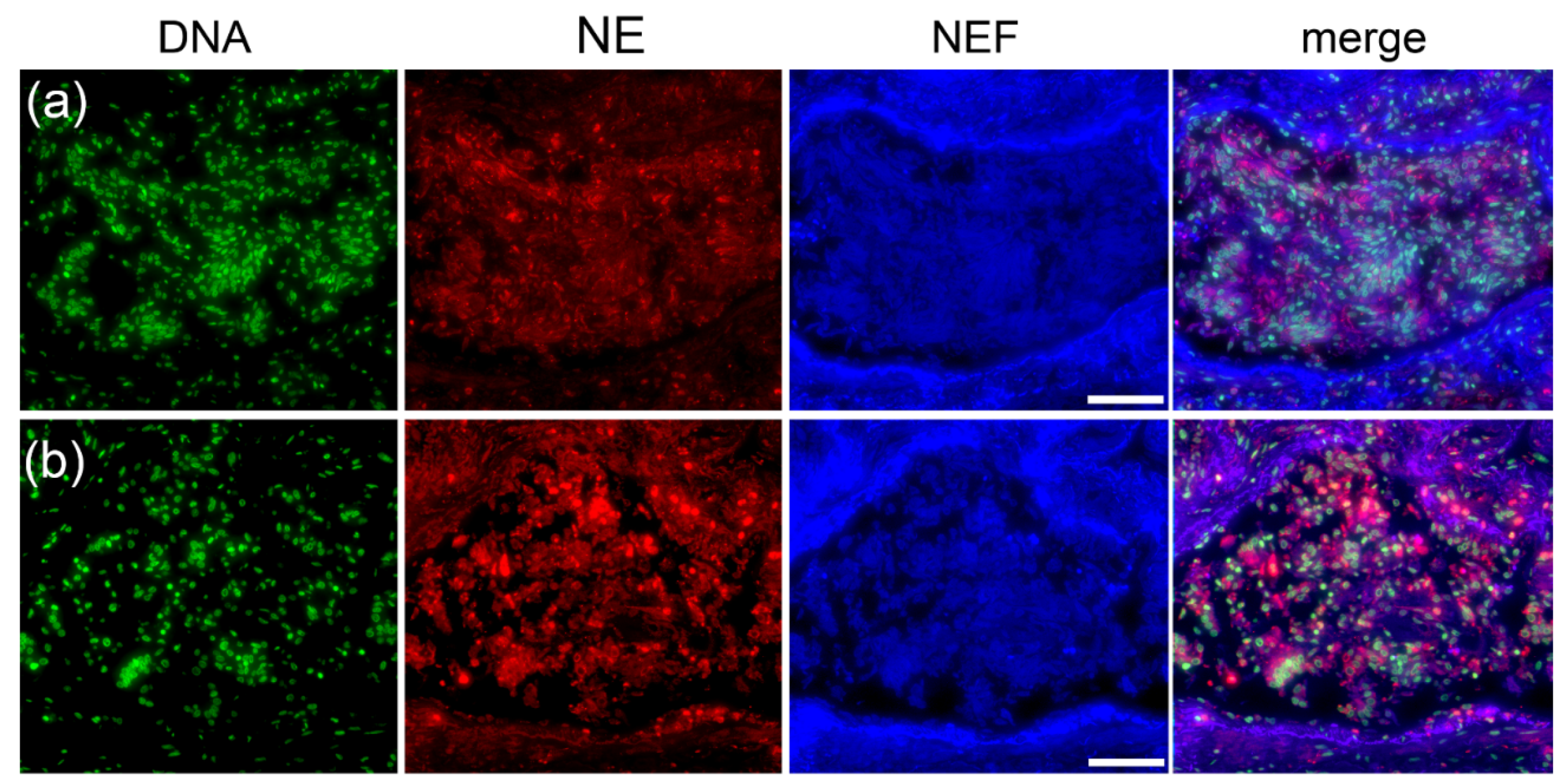
2.6. Type III Pleomorphism: DNA- and NE-Rich
2.7. Age and Gender Influence the Occurrence of Type of Pleomorphic Occlusion
3. Discussion
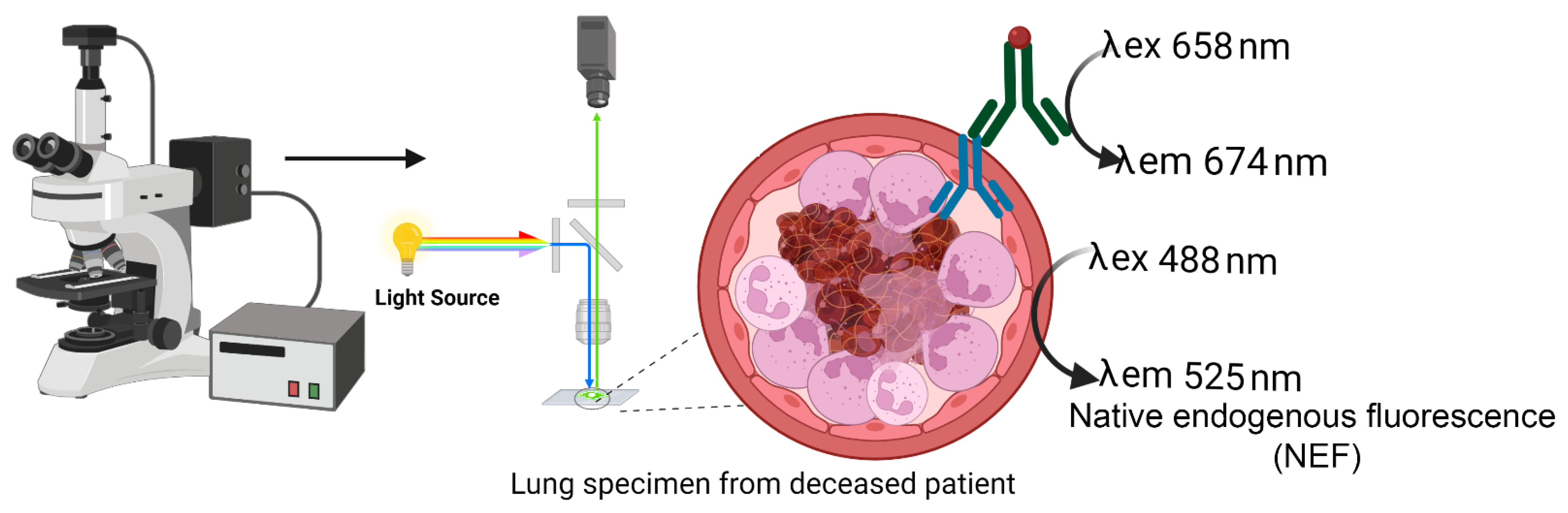
4. Materials and Methods
4.1. Patients
4.2. Ethics Statement
4.3. Immunofluorescence Staining
4.4. Hematoxylin and Eosin (H&E) Staining
4.5. CD31 Staining
4.6. Native Endogenous Fluorescence (NEF)
4.7. Morphometry
4.8. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Hornuss, D.; Lange, B.; Schroter, N.; Rieg, S.; Kern, W.V.; Wagner, D. Anosmia in COVID-19 patients. Clin. Microbiol. Infect 2020, 26, 1426–1427. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Lin, M.; Wei, L.; Xie, L.; Zhu, G.; Dela Cruz, C.S.; Sharma, L. Epidemiologic and Clinical Characteristics of Novel Coronavirus Infections Involving 13 Patients Outside Wuhan, China. JAMA 2020, 323, 1092–1093. [Google Scholar] [CrossRef] [PubMed]
- Ai, J.W.; Zi, H.; Wang, Y.; Huang, Q.; Wang, N.; Li, L.Y.; Pei, B.; Ji, J.; Zeng, X.T. Clinical Characteristics of COVID-19 Patients With Gastrointestinal Symptoms: An Analysis of Seven Patients in China. Front. Med. (Lausanne) 2020, 7, 308. [Google Scholar] [CrossRef]
- Fox, S.E.; Akmatbekov, A.; Harbert, J.L.; Li, G.; Quincy Brown, J.; Vander Heide, R.S. Pulmonary and cardiac pathology in African American patients with COVID-19: An autopsy series from New Orleans. Lancet Respir. Med. 2020, 8, 681–686. [Google Scholar] [CrossRef]
- Lorini, F.L.; Di Matteo, M.; Gritti, P.; Grazioli, L.; Benigni, A.; Zacchetti, L.; Bianchi, I.; Fabretti, F.; Longhi, L. Coagulopathy and COVID-19. Eur. Heart J. Suppl. 2021, 23, E95–E98. [Google Scholar] [CrossRef]
- Loo, J.; Spittle, D.A.; Newnham, M. COVID-19, immunothrombosis and venous thromboembolism: Biological mechanisms. Thorax 2021, 76, 412–420. [Google Scholar] [CrossRef]
- Martinod, K.; Wagner, D.D. Thrombosis: Tangled up in NETs. Blood 2014, 123, 2768–2776. [Google Scholar] [CrossRef]
- Mahajan, A.; Hasikova, L.; Hampel, U.; Gruneboom, A.; Shan, X.; Herrmann, I.; Garreis, F.; Bock, F.; Knopf, J.; Singh, J.; et al. Aggregated neutrophil extracellular traps occlude Meibomian glands during ocular surface inflammation. Ocul. Surf. 2021. [Google Scholar] [CrossRef]
- Munoz, L.E.; Boeltz, S.; Bilyy, R.; Schauer, C.; Mahajan, A.; Widulin, N.; Gruneboom, A.; Herrmann, I.; Boada, E.; Rauh, M.; et al. Neutrophil Extracellular Traps Initiate Gallstone Formation. Immunity 2019, 51, 443–450.e444. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.B.; Wright, H.J.; Roberts, A.; Ling-Mountford, N.; Cooper, P.R.; Chapple, I.L. Neutrophil hyper-responsiveness in periodontitis. J. Dent. Res. 2007, 86, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Listgarten, M.A.; Rosenberg, S.; Lerner, S. Progressive replacement of epithelial attachment by a connective tissue junction after experimental periodontal surgery in rats. J. Periodontol. 1982, 53, 659–670. [Google Scholar] [CrossRef]
- Vitkov, L.; Minnich, B.; Knopf, J.; Schauer, C.; Hannig, M.; Herrmann, M. NETs Are Double-Edged Swords with the Potential to Aggravate or Resolve Periodontal Inflammation. Cells 2020, 9, 2614. [Google Scholar] [CrossRef] [PubMed]
- Leppkes, M.; Knopf, J.; Naschberger, E.; Lindemann, A.; Singh, J.; Herrmann, I.; Sturzl, M.; Staats, L.; Mahajan, A.; Schauer, C.; et al. Vascular occlusion by neutrophil extracellular traps in COVID-19. EBioMedicine 2020, 58, 102925. [Google Scholar] [CrossRef] [PubMed]
- Leppkes, M.; Maueroder, C.; Hirth, S.; Nowecki, S.; Gunther, C.; Billmeier, U.; Paulus, S.; Biermann, M.; Munoz, L.E.; Hoffmann, M.; et al. Externalized decondensed neutrophil chromatin occludes pancreatic ducts and drives pancreatitis. Nat. Commun. 2016, 7, 10973. [Google Scholar] [CrossRef]
- Yaykasli, K.O.; Schauer, C.; Munoz, L.E.; Mahajan, A.; Knopf, J.; Schett, G.; Herrmann, M. Neutrophil Extracellular Trap-Driven Occlusive Diseases. Cells 2021, 10, 2208. [Google Scholar] [CrossRef] [PubMed]
- Behzadifard, M.; Soleimani, M. NETosis and SARS-COV-2 infection related thrombosis: A narrative review. Thromb. J. 2022, 20, 13. [Google Scholar] [CrossRef] [PubMed]
- Perico, L.; Benigni, A.; Casiraghi, F.; Ng, L.F.P.; Renia, L.; Remuzzi, G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat. Rev. Nephrol. 2021, 17, 46–64. [Google Scholar] [CrossRef]
- Middleton, E.A.; He, X.Y.; Denorme, F.; Campbell, R.A.; Ng, D.; Salvatore, S.P.; Mostyka, M.; Baxter-Stoltzfus, A.; Borczuk, A.C.; Loda, M.; et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood 2020, 136, 1169–1179. [Google Scholar] [CrossRef]
- Abou-Ismail, M.Y.; Diamond, A.; Kapoor, S.; Arafah, Y.; Nayak, L. The hypercoagulable state in COVID-19: Incidence, pathophysiology, and management. Thromb. Res. 2020, 194, 101–115. [Google Scholar] [CrossRef] [PubMed]
- Barnes, B.J.; Adrover, J.M.; Baxter-Stoltzfus, A.; Borczuk, A.; Cools-Lartigue, J.; Crawford, J.M.; Dassler-Plenker, J.; Guerci, P.; Huynh, C.; Knight, J.S.; et al. Targeting potential drivers of COVID-19: Neutrophil extracellular traps. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef] [PubMed]
- Veras, F.P.; Pontelli, M.C.; Silva, C.M.; Toller-Kawahisa, J.E.; de Lima, M.; Nascimento, D.C.; Schneider, A.H.; Caetite, D.; Tavares, L.A.; Paiva, I.M.; et al. SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, A. A NET-thrombosis axis in COVID-19. Blood 2020, 136, 1118–1119. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Yan, M.; Yu, J.; Xu, Q.; Xia, X.; Liao, J.; Zheng, W. In vivo identification of arteries and veins using two-photon excitation elastin autofluorescence. J. Anat. 2020, 236, 171–179. [Google Scholar] [CrossRef]
- Shrirao, A.B.; Schloss, R.S.; Fritz, Z.; Shrirao, M.V.; Rosen, R.; Yarmush, M.L. Autofluorescence of blood and its application in biomedical and clinical research. Biotechnol. Bioeng. 2021, 118, 4550–4576. [Google Scholar] [CrossRef]
- Croce, A.C.; Bottiroli, G. Autofluorescence spectroscopy and imaging: A tool for biomedical research and diagnosis. Eur. J. Histochem. 2014, 58, 2461. [Google Scholar] [CrossRef]
- Valdebenito, S.; Bessis, S.; Annane, D.; Lorin de la Grandmaison, G.; Cramer-Borde, E.; Prideaux, B.; Eugenin, E.A.; Bomsel, M. COVID-19 Lung Pathogenesis in SARS-CoV-2 Autopsy Cases. Front Immunol. 2021, 12, 735922. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef]
- Bosmuller, H.; Matter, M.; Fend, F.; Tzankov, A. The pulmonary pathology of COVID-19. Virchows. Arch. 2021, 478, 137–150. [Google Scholar] [CrossRef]
- Ogawa, M.; Doo, F.X.; Somwaru, A.S.; Roudenko, A.; Kamath, A.; Friedman, B. Peripheral arterial occlusion due to COVID-19: CT angiography findings of nine patients. Clin. Imaging. 2021, 73, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Evert, K.; Dienemann, T.; Brochhausen, C.; Lunz, D.; Lubnow, M.; Ritzka, M.; Keil, F.; Trummer, M.; Scheiter, A.; Salzberger, B.; et al. Autopsy findings after long-term treatment of COVID-19 patients with microbiological correlation. Virchows. Arch. 2021. [Google Scholar] [CrossRef] [PubMed]
- Blasco, A.; Coronado, M.J.; Hernandez-Terciado, F.; Martin, P.; Royuela, A.; Ramil, E.; Garcia, D.; Goicolea, J.; Del Trigo, M.; Ortega, J.; et al. Assessment of Neutrophil Extracellular Traps in Coronary Thrombus of a Case Series of Patients With COVID-19 and Myocardial Infarction. JAMA Cardiol 2020. [Google Scholar] [CrossRef] [PubMed]
- Staats, L.A.N.; Pfeiffer, H.; Knopf, J.; Lindemann, A.; Furst, J.; Kremer, A.E.; Hackstein, H.; Neurath, M.F.; Munoz, L.E.; Achenbach, S.; et al. IgA2 Antibodies against SARS-CoV-2 Correlate with NET Formation and Fatal Outcome in Severely Diseased COVID-19 Patients. Cells 2020, 9, 2676. [Google Scholar] [CrossRef] [PubMed]
- Narasaraju, T.; Tang, B.M.; Herrmann, M.; Muller, S.; Chow, V.T.K.; Radic, M. Neutrophilia and NETopathy as Key Pathologic Drivers of Progressive Lung Impairment in Patients With COVID-19. Front. Pharmacol. 2020, 11, 870. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, S.M.; Geller, A.E.; Hu, X.; Tieri, D.; Ding, C.; Klaes, C.K.; Cooke, E.A.; Woeste, M.R.; Martin, Z.C.; Chen, O.; et al. A specific low-density neutrophil population correlates with hypercoagulation and disease severity in hospitalized COVID-19 patients. JCI Insight 2021, 6. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, L.E.; Pekkarinen, P.T.; Alander, M.; Nowlan, K.H.A.; Nguyen, N.A.; Jokiranta, S.; Kuivanen, S.; Patjas, A.; Mero, S.; Pakkanen, S.H.; et al. Characterization of low-density granulocytes in COVID-19. PLoS Pathog. 2021, 17, e1009721. [Google Scholar] [CrossRef]
- Kaplan, M.J.; Radic, M. Neutrophil extracellular traps: Double-edged swords of innate immunity. J. Immunol. 2012, 189, 2689–2695. [Google Scholar] [CrossRef]
- Bonaventura, A.; Vecchie, A.; Dagna, L.; Martinod, K.; Dixon, D.L.; Van Tassell, B.W.; Dentali, F.; Montecucco, F.; Massberg, S.; Levi, M.; et al. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat. Rev. Immunol. 2021, 21, 319–329. [Google Scholar] [CrossRef]
- Giagulli, V.A.; Guastamacchia, E.; Magrone, T.; Jirillo, E.; Lisco, G.; De Pergola, G.; Triggiani, V. Worse progression of COVID-19 in men: Is testosterone a key factor? Andrology 2021, 9, 53–64. [Google Scholar] [CrossRef]
- Castle, S.C.; Uyemura, K.; Rafi, A.; Akande, O.; Makinodan, T. Comorbidity is a better predictor of impaired immunity than chronological age in older adults. J. Am. Geriatr. Soc. 2005, 53, 1565–1569. [Google Scholar] [CrossRef] [PubMed]
- Linehan, E.; Fitzgerald, D.C. Ageing and the immune system: Focus on macrophages. Eur. J. Microbiol. Immunol. (Bp) 2015, 5, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Agrawal, S.; Gupta, S. Role of Dendritic Cells in Inflammation and Loss of Tolerance in the Elderly. Front. Immunol. 2017, 8, 896. [Google Scholar] [CrossRef]
- Wenisch, C.; Patruta, S.; Daxbock, F.; Krause, R.; Horl, W. Effect of age on human neutrophil function. J. Leukoc. Biol. 2000, 67, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Tschan, S.L.; Bolliger, D. Coagulation and Aging: Implications for the Anesthesiologist. Currt. Anesthesiol. Reports 2021, 11, 387–395. [Google Scholar] [CrossRef]
- Zuo, Y.; Yalavarthi, S.; Shi, H.; Gockman, K.; Zuo, M.; Madison, J.A.; Blair, C.; Weber, A.; Barnes, B.J.; Egeblad, M.; et al. Neutrophil extracellular traps in COVID-19. JCI Insight 2020, 5. [Google Scholar] [CrossRef] [PubMed]
- Schonrich, G.; Raftery, M.J. Neutrophil Extracellular Traps Go Viral. Front. Immunol. 2016, 7, 366. [Google Scholar] [CrossRef]
- Schroeder, M.; Schaumburg, B.; Mueller, Z.; Parplys, A.; Jarczak, D.; Roedl, K.; Nierhaus, A.; de Heer, G.; Grensemann, J.; Schneider, B.; et al. High estradiol and low testosterone levels are associated with critical illness in male but not in female COVID-19 patients: A retrospective cohort study. Emerg. Microbes Infect. 2021, 10, 1807–1818. [Google Scholar] [CrossRef]
- Abd El-Lateef, A.E.; Alghamdi, S.; Ebid, G.; Khalil, K.; Kabrah, S.; Abdel Ghafar, M.T. Coagulation Profile in COVID-19 Patients and its Relation to Disease Severity and Overall Survival: A Single-Center Study. British J. Biomed. Sci. 2022, 79. [Google Scholar] [CrossRef]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef]
- Zhao, Y.; Nie, H.X.; Hu, K.; Wu, X.J.; Zhang, Y.T.; Wang, M.M.; Wang, T.; Zheng, Z.S.; Li, X.C.; Zeng, S.L. Abnormal immunity of non-survivors with COVID-19: Predictors for mortality. Infect. Dis. Poverty 2020, 9, 108. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wang, K.; Zuo, P.; Liu, Y.; Zhang, M.; Xie, S.; Zhang, H.; Chen, X.; Liu, C. Early decrease in blood platelet count is associated with poor prognosis in COVID-19 patients-indications for predictive, preventive, and personalized medical approach. EPMA J. 2020, 11, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]



| Pleomorphism in Occlusion | ||||||
|---|---|---|---|---|---|---|
| Patient | Age (in Range) | Gender | Weight (in kgs) | Type I | Type II | Type III |
| 1 | 58–62 | Female | N/A | 356 | 94 | 2 |
| 2 | 66–71 | Female | 100 | 46 | 34 | 12 |
| 3 | 71–75 | Male | 85 | 281 | 4 | 13 |
| 4 | 70–74 | Female | 120 | 35 | 79 | 5 |
| 5 | 81–85 | Male | N/A | 334 | 124 | 0 |
| 6 | 56–60 | Male | N/A | 496 | 259 | 5 |
| 7 | 74–78 | Male | 80 | 447 | 491 | 9 |
| 8 | 64–68 | Male | 97 | 32 | 87 | 12 |
| 9 | 71–75 | Female | N/A | 419 | 326 | 5 |
| 10 | 60–64 | Female | N/A | 394 | 521 | 9 |
| 11 | 53–57 | Male | 64 | 16 | 42 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, J.; Herrmann, I.; Mahajan, A.; Schauer, C.; Shan, X.; Hartmann, A.; Rieker, R.J.; Evert, K.; Falkeis, C.; Naschberger, E.; et al. A Pleomorphic Puzzle: Heterogeneous Pulmonary Vascular Occlusions in Patients with COVID-19. Int. J. Mol. Sci. 2022, 23, 15126. https://doi.org/10.3390/ijms232315126
Singh J, Herrmann I, Mahajan A, Schauer C, Shan X, Hartmann A, Rieker RJ, Evert K, Falkeis C, Naschberger E, et al. A Pleomorphic Puzzle: Heterogeneous Pulmonary Vascular Occlusions in Patients with COVID-19. International Journal of Molecular Sciences. 2022; 23(23):15126. https://doi.org/10.3390/ijms232315126
Chicago/Turabian StyleSingh, Jeeshan, Irmgard Herrmann, Aparna Mahajan, Christine Schauer, Xiaomei Shan, Arndt Hartmann, Ralf J. Rieker, Katja Evert, Christina Falkeis, Elisabeth Naschberger, and et al. 2022. "A Pleomorphic Puzzle: Heterogeneous Pulmonary Vascular Occlusions in Patients with COVID-19" International Journal of Molecular Sciences 23, no. 23: 15126. https://doi.org/10.3390/ijms232315126
APA StyleSingh, J., Herrmann, I., Mahajan, A., Schauer, C., Shan, X., Hartmann, A., Rieker, R. J., Evert, K., Falkeis, C., Naschberger, E., von Stillfried, S., Boor, P., Muñoz, L. E., Schett, G., Herrmann, M., & Knopf, J. (2022). A Pleomorphic Puzzle: Heterogeneous Pulmonary Vascular Occlusions in Patients with COVID-19. International Journal of Molecular Sciences, 23(23), 15126. https://doi.org/10.3390/ijms232315126











