Effect of Humic Acid on Soil Physical and Chemical Properties, Microbial Community Structure, and Metabolites of Decline Diseased Bayberry
Abstract
1. Introduction
2. Results
2.1. Effect of Humic Acid on Vegetative Growth and Fruit Quality
2.2. Effect of Humic Acid in Microbial Community Diversity
2.3. Effect of Humic Acid in Soil Microbial Community Structure
2.4. Effect of Humic Acid on Soil Nutrient Status
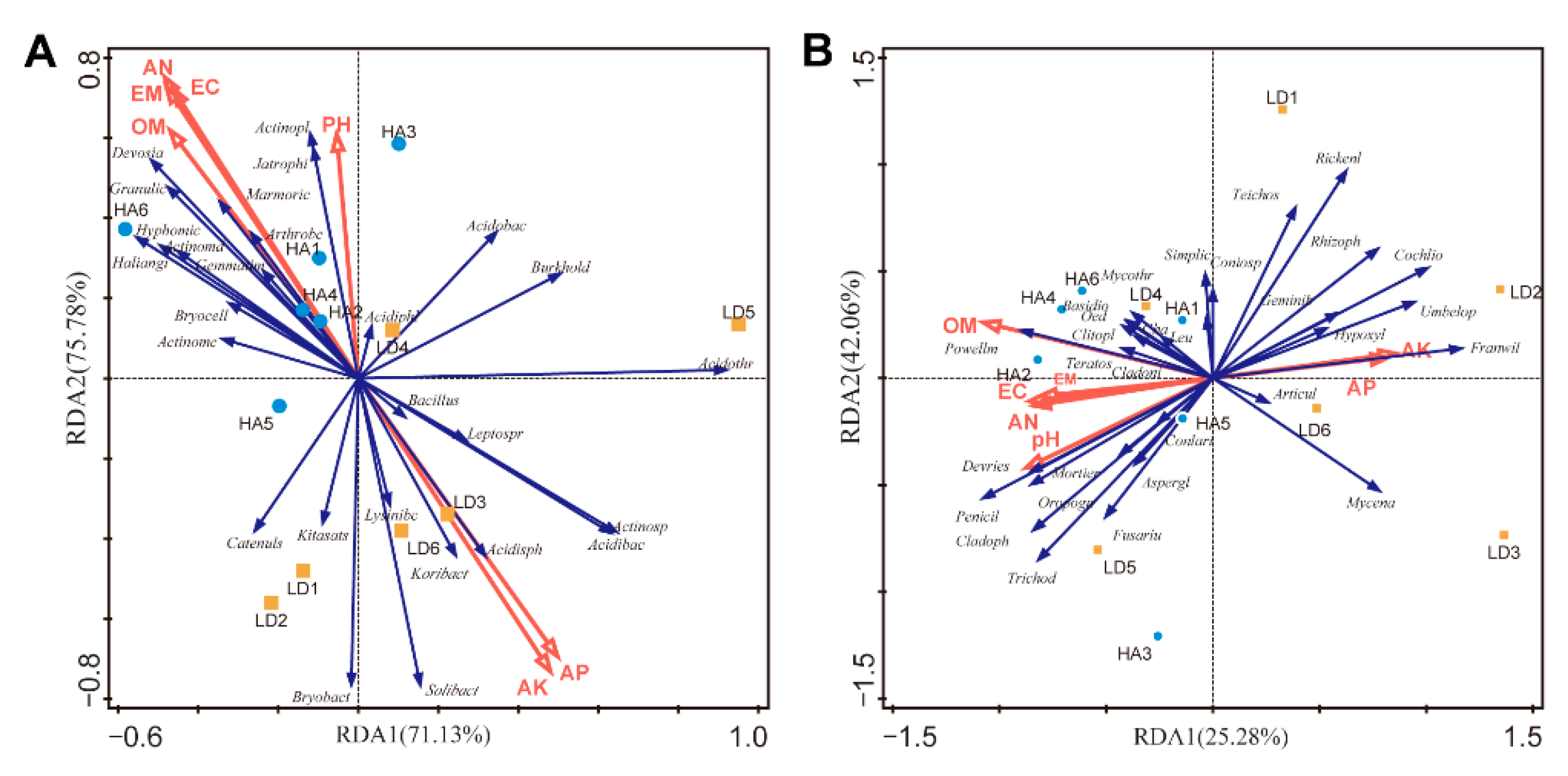
2.5. Effect of Humic Acid on RDA of Soil Properties and Microbial Communities
2.6. Change in Rhizosphere Soil Metabolomics
2.7. Analysis of Differential Metabolites
2.8. Effect of Humic Acid on Metabolic Pathways
2.9. Correlation of Soil Microorganisms with Metabolites
3. Materials and Methods
3.1. Experimental Design
3.2. Measurement of Vegetative Growth Parameters
3.3. Measurement of Fruit Economic Characters
3.4. Soil Sample Collection and Physical and Chemical Property Measurements
3.5. Soil Genome Sequencing
3.6. Gas Chromatography-Mass Spectrometry (GC-MS) Metabolomics Analysis
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ren, H.; Yu, H.; Zhang, S.; Liang, S.; Zheng, X.; Zhang, S.; Yao, P.; Zheng, H.; Qi, X. Genome Sequencing Provides Insights into the Evolution and Antioxidant Activity of Chinese Bayberry. BMC Genom. 2019, 20, 458. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Huang, H.; Xu, C.; Li, X.; Chen, K. Biological Activities of Extracts from Chinese Bayberry (Myrica Rubra Sieb. et Zucc.): A Review. Plant Foods Hum. Nutr. 2013, 68, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Wang, H.; Qi, X.; Yu, Z.; Zheng, X.; Zhang, S.; Wang, Z.; Zhang, M.; Ahmed, T.; Li, B. The Damage Caused by Decline Disease in Bayberry Plants through Changes in Soil Properties, Rhizosphere Microbial Community Structure and Metabolites. Plants 2021, 10, 2083. [Google Scholar] [CrossRef]
- Nunes, R.O.; Domiciano, G.A.; Alves, W.S.; Melo, A.C.A.; Nogueira, F.C.S.; Canellas, L.P.; Olivares, F.L.; Zingali, R.B.; Soares, M.R. Evaluation of the Effects of Humic Acids on Maize Root Architecture by Label-Free Proteomics Analysis. Sci. Rep. 2019, 9, 12019. [Google Scholar] [CrossRef]
- Lian, X.; Liao, S.; Yang, Y.; Zhang, X.; Wang, Y. Effect of PH or Metal Ions on the Oil/Water Interfacial Behavior of Humic Acid Based Surfactant. Langmuir 2020, 36, 10838–10845. [Google Scholar] [CrossRef] [PubMed]
- Suh, H.Y.; Yoo, K.S.; Suh, S.G. Tuber Growth and Quality of Potato (Solanum Tuberosum L.) as Affected by Foliar or Soil Application of Fulvic and Humic Acids. Hortic. Environ. Biotechnol. 2014, 55, 183–189. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Y.; Yang, F. Critical Review on Soil Phosphorus Migration and Transformation under Freezing-Thawing Cycles and Typical Regulatory Measurements. Sci. Total Environ. 2021, 751, 141614. [Google Scholar] [CrossRef] [PubMed]
- Man-hong, Y.; Lei, Z.; Sheng-tao, X.; McLaughlin, N.B.; Jing-hui, L. Effect of Water Soluble Humic Acid Applied to Potato Foliage on Plant Growth, Photosynthesis Characteristics and Fresh Tuber Yield under Different Water Deficits. Sci. Rep. 2020, 10, 7854. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Tong, J.; Xu, Q.; Dai, L.; Ye, J.; Wu, H.; Xu, C.; Shi, J. Regulation Mechanisms of Humic Acid on Pb Stress in Tea Plant (Camellia Sinensis L.). Environ. Pollut. 2020, 267, 115546. [Google Scholar] [CrossRef]
- Pang, L.; Song, F.; Song, X.; Guo, X.; Lu, Y.; Chen, S.; Zhu, F.; Zhang, N.; Zou, J.; Zhang, P. Effects of Different Types of Humic Acid Isolated from Coal on Soil NH3 Volatilization and CO2 Emissions. Environ. Res. 2021, 194, 110711. [Google Scholar] [CrossRef]
- Nardi, S.; Pizzeghello, D.; Muscolo, A.; Vianello, A. Physiological Effects of Humic Substances on Higher Plants. Soil Biol. Biochem. 2002, 34, 1527–1536. [Google Scholar] [CrossRef]
- Naidu, Y.; Meon, S.; Siddiqui, Y. Foliar Application of Microbial-Enriched Compost Tea Enhances Growth, Yield and Quality of Muskmelon (Cucumis Melo L.) Cultivated under Fertigation System. Sci. Hortic. 2013, 159, 33–40. [Google Scholar] [CrossRef]
- Olivares, F.L.; Aguiar, N.O.; Rosa, R.C.C.; Canellas, L.P. Substrate Biofortification in Combination with Foliar Sprays of Plant Growth Promoting Bacteria and Humic Substances Boosts Production of Organic Tomatoes. Sci. Hortic. 2015, 183, 100–108. [Google Scholar] [CrossRef]
- Ren, H.-Y.; Wang, H.-Y.; Wang, C.-A.; Zheng, X.-L.; Qi, X.-J. Effects of Humic Acid on Vegetative Growth and Fruit Quality of Bayberry with Decline Disease. Hunan Agr. Sci. 2022, 2022, 50–52. [Google Scholar]
- Tang, H.; Xiao, X.; Xu, Y.; Li, C.; Cheng, K.; Pan, X.; Li, W. Utilization of Carbon Sources in the Rice Rhizosphere and Nonrhizosphere Soils with Different Long-Term Fertilization Management. J. Basic Microbiol. 2019, 59, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tao, Y.; Sun, G.; Wang, L. Effects of Di-n-Butyl Phthalate on the Physiology and Ultrastructure of Cucumber Seedling Roots. Environ. Sci. Pollut. Res. Int. 2014, 21, 6662–6670. [Google Scholar] [CrossRef][Green Version]
- Zhao, L.; Zhang, H.; White, J.C.; Chen, X.; Li, H.; Qu, X.; Ji, R. Metabolomics Reveals That Engineered Nanomaterial Exposure in Soil Alters Both Soil Rhizosphere Metabolite Profiles and Maize Metabolic Pathways. Environ. Sci. Nano 2019, 6, 1716–1727. [Google Scholar] [CrossRef]
- Tian, L.; Shen, J.; Sun, G.; Wang, B.; Ji, R.; Zhao, L. Foliar Application of SiO2 Nanoparticles Alters Soil Metabolite Profiles and Microbial Community Composition in the Pakchoi (Brassica Chinensis L.) Rhizosphere Grown in Contaminated Mine Soil. Environ. Sci. Technol. 2020, 54, 13137–13146. [Google Scholar] [CrossRef]
- Ghimire, B.K.; Ghimire, B.; Yu, C.Y.; Chung, I.-M. Allelopathic and Autotoxic Effects of Medicago Sativa—Derived Allelochemicals. Plants 2019, 8, 233. [Google Scholar] [CrossRef]
- Neal, A.L.; Ahmad, S.; Gordon-Weeks, R.; Ton, J. Benzoxazinoids in Root Exudates of Maize Attract Pseudomonas Putida to the Rhizosphere. PLoS ONE 2012, 7, e35498. [Google Scholar] [CrossRef]
- Pan, L.; Lei, D.; Jin, L.; He, Y.; Yang, Q. Promising Fungicides from Allelochemicals: Synthesis of Umbelliferone Derivatives and Their Structure–Activity Relationships. Molecules 2018, 23, 3002. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Wang, H.; Wang, Q.; Qi, X.; Zhang, S.; Yu, Z.; Ijaz, M.; Zhang, M.; Ahmed, T.; El-Sharnouby, M.; et al. Effect of Fungicides on Bayberry Decline Disease by Modulating Rhizosphere Soil Properties, Microflora, and Metabolites. Agronomy 2022, 12, 677. [Google Scholar] [CrossRef]
- Gu, D.Y.; Wang, X.F.; Yang, F.J.; Jiao, J.; Wei, M.; Shi, Q.H. Effects of purified humic acid on growth and nutrient absorption of cucumber (Cucumis sativus)seedlings under low nitrogen stress. Ying Yong Sheng Tai Xue Bao 2016, 27, 2535–2542. [Google Scholar] [PubMed]
- Ren, H.; Wang, H.; Yu, Z.; Zhang, S.; Qi, X.; Sun, L.; Wang, Z.; Zhang, M.; Ahmed, T.; Li, B. Effect of Two Kinds of Fertilizers on Growth and Rhizosphere Soil Properties of Bayberry with Decline Disease. Plants 2021, 10, 2386. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Pimentel, J.L.; Dominguez-Moñino, I.; Jurado, V.; Laiz, L.; Caldeira, A.T.; Saiz-Jimenez, C. The Rare Actinobacterium Crossiella Sp. Is a Potential Source of New Bioactive Compounds with Activity against Bacteria and Fungi. Microorganisms 2022, 10, 1575. [Google Scholar] [CrossRef]
- Pan, L.; Chen, J.; Ren, S.; Shen, H.; Rong, B.; Liu, W.; Yang, Z. Complete Genome Sequence of Mycobacterium Mya-Zh01, an Endophytic Bacterium, Promotes Plant Growth and Seed Germination Isolated from Flower Stalk of Doritaenopsis. Arch. Microbiol. 2020, 202, 1965–1976. [Google Scholar] [CrossRef]
- Wang, J.L.; Liu, K.L.; Zhao, X.Q.; Gao, G.-F.; Wu, Y.H.; Shen, R.F. Microbial Keystone Taxa Drive Crop Productivity through Shifting Aboveground-Belowground Mineral Element Flows. Sci. Total Environ. 2022, 811, 152342. [Google Scholar] [CrossRef]
- Zuriegat, Q.; Zheng, Y.; Liu, H.; Wang, Z.; Yun, Y. Current Progress on Pathogenicity-Related Transcription Factors in Fusarium Oxysporum. Mol. Plant Pathol. 2021, 22, 882–895. [Google Scholar] [CrossRef]
- Shan, T.; Zhou, L.; Li, B.; Chen, X.; Guo, S.; Wang, A.; Tian, L.; Liu, J. The Plant Growth-Promoting Fungus MF23 (Mycena Sp.) Increases Production of Dendrobium Officinale (Orchidaceae) by Affecting Nitrogen Uptake and NH4+ Assimilation. Front. Plant Sci. 2021, 12, 6935561. [Google Scholar] [CrossRef]
- de Morais, E.G.; Silva, C.A.; Jindo, K. Humic Acid Improves Zn Fertilization in Oxisols Successively Cultivated with Maize–Brachiaria. Molecules 2021, 26, 4588. [Google Scholar] [CrossRef]
- Adamiano, A.; Fellet, G.; Vuerich, M.; Scarpin, D.; Carella, F.; Piccirillo, C.; Jeon, J.-R.; Pizzutti, A.; Marchiol, L.; Iafisco, M. Calcium Phosphate Particles Coated with Humic Substances: A Potential Plant Biostimulant from Circular Economy. Molecules 2021, 26, 2810. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.Y.; Wu, Y.C.; Ahmed, T.; Qi, X.J.; Li, B. Response of resistant and susceptible bayberry cultivars to infection of twig blight pathogen by histological observation and gibberellin related genes expression. Pathogens 2021, 10, 402. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Su, Y.; Ahmed, T.; Ren, H.; Javed, M.R.; Yao, Y.; An, Q.; Yan, J.; Li, B. Effects of Different Organic Fertilizers on Improving Soil from Newly Reclaimed Land to Crop Soil. Agriculture 2021, 11, 560. [Google Scholar] [CrossRef]
- Oney-Birol, S. Exogenous L-Carnitine Promotes Plant Growth and Cell Division by Mitigating Genotoxic Damage of Salt Stress. Sci. Rep. 2019, 9, 17229. [Google Scholar] [CrossRef]
- Ben Rejeb, K.; Abdelly, C.; Savouré, A. Proline, a multifunctional amino-acid involved in plant adaptation to environmental constraints. Biol. Aujourdhui. 2012, 206, 291–299. [Google Scholar] [CrossRef]
- Ni, D.; Xu, W.; Zhu, Y.; Pang, X.; Lv, J.; Mu, W. Insight into the Effects and Biotechnological Production of Kestoses, the Smallest Fructooligosaccharides. Crit. Rev. Biotechnol. 2021, 41, 34–46. [Google Scholar] [CrossRef]
- Corneo, P.E.; Nesler, A.; Lotti, C.; Chahed, A.; Vrhovsek, U.; Pertot, I.; Perazzolli, M. Interactions of Tagatose with the Sugar Metabolism Are Responsible for Phytophthora Infestans Growth Inhibition. Microbiol. Res. 2021, 247, 126724. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.; Kim, K. Diclofenac Modified the Root System Architecture of Arabidopsis via Interfering with the Hormonal Activities of Auxin. J. Hazard. Mater. 2021, 413, 125402. [Google Scholar] [CrossRef]
- Li, Z.; Cheng, B.; Yong, B.; Liu, T.; Peng, Y.; Zhang, X.; Ma, X.; Huang, L.; Liu, W.; Nie, G. Metabolomics and physiological analyses reveal β-sitosterol as an important plant growth regulator inducing tolerance to water stress in white clover. Planta 2019, 250, 2033–2046. [Google Scholar] [CrossRef]
- Chun, H.J.; Baek, D.; Cho, H.M.; Jung, H.S.; Jeong, M.S.; Jung, W.H.; Choi, C.W.; Lee, S.H.; Jin, B.J.; Park, M.S.; et al. Metabolic Adjustment of Arabidopsis Root Suspension Cells During Adaptation to Salt Stress and Mitotic Stress Memory. Plant Cell Physiol. 2019, 60, 612–625. [Google Scholar] [CrossRef]
- Bruni, G.O.; Klasson, K.T. Aconitic Acid Recovery from Renewable Feedstock and Review of Chemical and Biological Applications. Foods 2022, 11, 573. [Google Scholar] [CrossRef] [PubMed]
- Leandro, L.; Cardoso, M.; Silva, S.; Souza, M.; Veneziani, R.; Ambrosio, S.; Martins, C.H. Antibacterial Activity Pinus Elliottii and Its Major Compound, Dehydroabietic Acid, against Multidrug-Resistant Strains. J. Med. Microbiol. 2014, 63, 1649–1653. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Li, M.; Wei, G.; Tian, R.; Li, C.; Wang, B.; Lin, R.; Shi, C.; Chi, X.; Zhou, B.; et al. The Occurrence of Potato Common Scab Correlates with the Community Composition and Function of the Geocaulosphere Soil Microbiome. Microbiome 2019, 7, 14. [Google Scholar] [CrossRef]
- Appleton, J. Arginine: Clinical Potential of a Semi-Essential Amino Acid. (Arginine). Altern. Med. Rev. 2002, 7, 512–523. [Google Scholar]
- Liang, W.; Ma, X.; Wan, P.; Liu, L. Plant Salt-Tolerance Mechanism: A Review. Biochem. Biophys Res. Commuun. 2018, 495, 286–291. [Google Scholar] [CrossRef]
- Shibl, A.A.; Isaac, A.; Ochsenkühn, M.A.; Cárdenas, A.; Fei, C.; Behringer, G.; Arnoux, M.; Drou, N.; Santos, M.P.; Gunsalus, K.C.; et al. Diatom Modulation of Select Bacteria through Use of Two Unique Secondary Metabolites. Proc. Natl. Acad. Sci. USA 2020, 117, 27445–27455. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Zhong, S.; Wang, Y.; Wang, B.; Mei, X.; Li, R.; Ruan, Y.; Shen, Q. Induced Soil Microbial Suppression of Banana Fusarium Wilt Disease Using Compost and Biofertilizers to Improve Yield and Quality. Eur. J. Soil Biol. 2013, 57, 1–8. [Google Scholar] [CrossRef]
- Wu, L.; Wen, C.; Qin, Y.; Yin, H.; Tu, Q.; Van Nostrand, J.D.; Yuan, T.; Yuan, M.; Deng, Y.; Zhou, J. Phasing Amplicon Sequencing on Illumina Miseq for Robust Environmental Microbial Community Analysis. BMC Microbiol. 2015, 15, 125. [Google Scholar] [CrossRef]








| Parameters | Value | Parameters | Value |
|---|---|---|---|
| Branch length (cm) | Branch diameter (mm) | ||
| LD | 63.34 ± 4.76 | LD | 2.63 ± 0.09 |
| HA | 81.47 ± 0.93 * | HA | 2.92 ± 0.03 * |
| Leaf length (mm) | Leaf width (mm) | ||
| LD | 107.79 ± 1.57 | LD | 32.84 ± 0.45 |
| HA | 120.87 ± 0.55 * | HA | 37.72 ± 2.27 * |
| Leaf thickness (10 pieces/mm) | Chlorophyll/(SPAD) | ||
| LD | 4.68 ± 0.05 | LD | 41.06 ± 0.30 |
| HA | 5.18 ± 0.14 * | HA | 48.35 ± 0.14 * |
| Parameters | Value | Parameters | Value |
|---|---|---|---|
| Single fruit weight (g) | Total soluble solids (%) | ||
| LD | 26.86 ± 0.74 | LD | 13.71 ± 0.69 |
| HA | 36.61 ± 0.13 * | HA | 15.93 ± 0.20 * |
| Total sugar | Vitamin C (mg/100 g) | ||
| LD | 12.70 ± 0.03 | LD | 2.16 ± 0.00 |
| HA | 15.84 ± 0.78 * | HA | 5.90 ± 0.06 * |
| Titratable acid (%) | |||
| LD | 0.04 ± 0.00 | ||
| HA | 0.02 ± 0.00 # |
| Physical and Chemical Properties | LD | HA |
|---|---|---|
| pH | 4.57 ± 0.27 | 4.92 * ± 0.34 |
| Organic matter (%) | 1.84 ± 0.34 | 2.37 * ± 0.13 |
| Alkali hydrolyzed nitrogen (mg/kg) | 45.71 ± 1.82 | 151.72 * ± 24.78 |
| Available phosphorus (mg/kg) | 205.70 ± 8.63 | 27.81 # ± 3.97 |
| Available potassium (mg/kg) | 432.20 ± 3.89 | 116.87 # ± 11.25 |
| Exchangeable calcium (mg/kg) | 137.32 ± 16.43 | 598.61 * ± 16.19 |
| Exchangeable magnesium (mg/kg) | 24.41 ± 2.36 | 101.55* ± 0.94 |
| Soil Environment | Contribution at the Bacterial Genus Level (%) | Contribution at the Fungal Genus Level (%) |
|---|---|---|
| pH | 10.0 | 14.7 |
| Organic matter | 4.2 | 32.6 |
| Available nitrogen | 13.0 | 7.9 |
| Available phosphorus | 25.0 | 18.5 |
| Available potassium | 16.5 | 9.4 |
| Exchangeable calcium | 28.8 | 8.4 |
| Exchangeable magnesium | 2.6 | 8.4 |
| Metabolite Name | Relative Content | Metabolite Name | Relative Content |
|---|---|---|---|
| Putrescine | 2-Hydroxypyrazinyl-2-Propenoic Acid | ||
| LD | 223.48 ± 12.81 | LD | 124.87 ± 3.48 |
| HA | 260.99 * ± 32.39 | HA | 145.17 * ± 14.46 |
| Metharbital | Alanine-Alanine | ||
| LD | 6.03 ± 0.47 | LD | 132.12 ± 6.12 |
| HA | 7.70 * ± 1.26 | HA | 147.06 * ± 13.59 |
| Lyxose | Urea | ||
| D | 0.42 ± 0.09 | LD | 170.82 ± 1.40 |
| HA | 9.97 * ± 5.43 | HA | 204.50 * ± 16.49 |
| Glucose-1,2,3,4,5,6,6 Deuterated | 2-Monoolein | ||
| LD | 0.37 ± 0.03 | LD | 0.48 ± 0.06 |
| HA | 0.44 * ± 0.04 | HA | 7.94 * ± 4.22 |
| 1-Hexadecanol | Beta-Sitosterol | ||
| LD | 1.25 ± 0.16 | LD | 2.33 ± 0.08 |
| HA | 2.48 * ± 0.77 | HA | 4.07 * ± 1.16 |
| Dehydroabietic Acid | Glucoheptulose | ||
| LD | 2.00 ± 0.21 | LD | 4.88 ± 0.37 |
| HA | 3.91 * ± 1.51 | HA | 6.49 * ± 0.50 |
| Pentonolactone | Guanidinosuccinate | ||
| LD | 1.91 ± 0.03 | LD | 0.30 ± 0.13 |
| HA | 41.36 * ± 18.97 | HA | 3.58 * ± 2.17 |
| Diclofenac | Hydroxylamine | ||
| LD | 4.61 ± 0.54 | LD | 22.48 ± 2.52 |
| HA | 7.05 * ± 0.79 | HA | 30.40 * ± 3.09 |
| 5-Methoxytryptamine | Butylamine | ||
| LD | 58.12 ± 10.05 | LD | 1.05 ± 0.02 |
| HA | 108.26 * ± 8.43 | HA | 1.29 * ± 0.10 |
| Aconitic Acid | Tbs Compound | ||
| LD | 0.77 ± 0.11 | LD | 3.92 ± 0.18 |
| HA | 28.17 * ± 20.37 | HA | 4.44 * ± 0.46 |
| Epicatechin | Coniferin | ||
| LD | 0.35 ± 0.05 | LD | 0.41 ± 0.09 |
| HA | 9.50 * ± 4.72 | HA | 0.70 * ± 0.19 |
| Myo-Inositol | Palatinitol | ||
| LD | 51.82 ± 5.53 | LD | 12.43 ± 0.74 |
| HA | 27.57 # ± 5.15 | HA | 4.92 # ± 1.45 |
| Hexitol | Glucosamine | ||
| LD | 4.61 ± 0.69 | LD | 1.02 ± 0.06 |
| HA | 1.76 # ± 0.55 | HA | 0.73 # ± 0.14 |
| Benzoic Acid | Tyrosine | ||
| LD | 6.30 ± 0.94 | LD | 1.69 ± 0.32 |
| HA | 4.24 # ± 1.61 | HA | 0.86 # ± 0.19 |
| Isohexonic Acid | 2-Deoxytetronic acid | ||
| LD | 5.88 ± 1.28 | LD | 2.02 ± 0.28 |
| HA | 2.51 # ± 0.41 | HA | 1.36 # ± 0.19 |
| Isothreonic Acid | Agmatine | ||
| LD | 2.82 ± 0.64 | LD | 1.26 ± 0.20 |
| HA | 1.75 # ± 0.32 | HA | 0.68 # ± 0.25 |
| Oxoproline | Threonine | ||
| LD | 41.30 ± 4.88 | LD | 1.23 ± 0.29 |
| HA | 23.33 # ± 9.17 | HA | 0.80 # ± 0.24 |
| Proline | Serine | ||
| LD | 5.97 ± 2.37 | LD | 1.28 ± 0.39 |
| HA | 2.27 # ± 1.06 | HA | 0.66 # ± 0.25 |
| Ribose | Beta-Gentiobiose | ||
| LD | 246.68 ± 38.36 | LD | 0.97 ± 0.43 |
| HA | 192.67 # ± 20.11 | HA | 0.48 # ± 0.06 |
| Ribonic Acid | Tagatose | ||
| LD | 8.23 ± 2.78 | LD | 47.61 ± 22.07 |
| HA | 5.30 # ± 0.87 | HA | 20.05 # ± 8.06 |
| Lactobionic Acid | Conduritol-Beta-Expoxide | ||
| LD | 15.30 ± 4.80 | LD | 2.05 ± 1.15 |
| HA | 7.40 # ± 1.69 | HA | 0.63 # ± 0.28 |
| Glyceric Acid | Hexadecylglycerol | ||
| LD | 6.83 ± 1.18 | LD | 2.68 ± 0.13 |
| HA | 5.05 # ± 1.27 | HA | 1.60 # ± 0.50 |
| Glucose | Vanillic Acid | ||
| LD | 13.14 ± 1.23 | LD | 1.81 ± 0.15 |
| HA | 7.62 # ± 1.76 | HA | 1.27 # ± 0.18 |
| Chenodeoxycholic Acid | Zymosterol | ||
| LD | 0.44 ± 0.06 | LD | 8.71 ± 1.22 |
| HA | 0.24 # ± 0.08 | HA | 2.11 # ± 1.46 |
| Deoxycholic Acid | 2-Picolinic acid | ||
| LD | 14.16 ± 2.26 | LD | 2.46 ± 0.35 |
| HA | 2.90 # ± 2.65 | HA | 1.42 # ± 0.41 |
| Galacturonic Acid | Butyrolactam | ||
| LD | 1.13 ± 0.09 | LD | 3.09 ± 0.20 |
| HA | 0.74 # ± 0.27 | HA | 2.61 # ± 0.39 |
| Glycerol | N-Acetyl-D-Mannosamine | ||
| LD | 160.69 ± 8.38 | LD | 0.90 ± 0.03 |
| HA | 132.88 # ± 26.61 | HA | 0.64 # ± 0.20 |
| 2-Ketoadipic Acid | 4-Hydroxybutyric acid | ||
| LD | 3.53 ± 0.12 | LD | 8.08 ± 0.47 |
| HA | 2.32 # ± 0.60 | HA | 4.04 # ± 2.28 |
| Glutamate | Erythronic Acid | ||
| LD | 0.22 ± 0.04 | LD | 0.29 ± 0.03 |
| HA | 0.14 # ± 0.05 | HA | 0.24 # ± 0.04 |
| Xylose | Xylonolactone | ||
| LD | 0.04 ± 0.03 | LD | 0.11 ± 0.09 |
| HA | 0.80 # ± 0.15 | HA | 0.86 # ± 0.14 |
| Adipic Acid | 6-Deoxyglucose | ||
| LD | 1.04 ± 0.05 | LD | 13.40 ± 0.38 |
| HA | 0.76 # ± 0.19 | HA | 8.40 # ± 1.17 |
| Fructose | N-Acetylgalactosamine | ||
| LD | 3.71 ± 0.15 | LD | 4.24 ± 0.23 |
| HA | 2.70 # ± 0.38 | HA | 2.21 # ± 0.77 |
| 2-Methylglyceric acid | Carinitine | ||
| LD | 1.00 ± 0.05 | LD | 5.33 ± 0.94 |
| HA | 0.69 # ± 0.13 | HA | 2.25 # ± 0.98 |
| Inositol-4-Monophosphate | 1-Monoolein | ||
| LD | 2.13 ± 0.24 | LD | 1.91 ± 0.25 |
| HA | 1.18 # ± 0.31 | HA | 0.69 # ± 0.22 |
| Maltotriose | 2-Monopalmitin | ||
| LD | 2.20 ± 0.31 | LD | 2.05 ± 0.06 |
| HA | 0.88 # ± 0.32 | HA | 1.50 # ± 0.22 |
| Phytol | 3-Hydroxybenzoic acid | ||
| LD | 0.92 ± 0.13 | LD | 8.23 ± 1.48 |
| HA | 0.51 # ± 0.17 | HA | 4.53 # ± 1.11 |
| 2-Ketoglucose Dimethylacetal | 3-Hydroxybutyric acid | ||
| LD | 4.44 ± 1.15 | LD | 30.44 ± 9.12 |
| HA | 1.99 # ± 0.78 | HA | 13.47 # ± 7.17 |
| Melezitose | Glutamic Acid | ||
| LD | 1.14 ± 0.22 | LD | 1.76 ± 0.24 |
| HA | 0.42 # ± 0.15 | HA | 0.93 # ± 0.21 |
| Galactitol | 1,5-Anhydroglucitol | ||
| LD | 21.08 ± 3.78 | LD | 6.71 ± 3.06 |
| HA | 12.55 # ± 2.63 | HA | 2.11 # ± 0.23 |
| 1-Kestose | Oleic Acid | ||
| LD | 1.14 ± 0.22 | LD | 0.77 ± 0.18 |
| HA | 366.82 # ± 33.77 | HA | 0.51 # ± 0.12 |
| Phenol | |||
| LD | 23.03 ± 0.64 | ||
| HA | 26.95 * ± 3.18 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, H.; Islam, M.S.; Wang, H.; Guo, H.; Wang, Z.; Qi, X.; Zhang, S.; Guo, J.; Wang, Q.; Li, B. Effect of Humic Acid on Soil Physical and Chemical Properties, Microbial Community Structure, and Metabolites of Decline Diseased Bayberry. Int. J. Mol. Sci. 2022, 23, 14707. https://doi.org/10.3390/ijms232314707
Ren H, Islam MS, Wang H, Guo H, Wang Z, Qi X, Zhang S, Guo J, Wang Q, Li B. Effect of Humic Acid on Soil Physical and Chemical Properties, Microbial Community Structure, and Metabolites of Decline Diseased Bayberry. International Journal of Molecular Sciences. 2022; 23(23):14707. https://doi.org/10.3390/ijms232314707
Chicago/Turabian StyleRen, Haiying, Mohammad Shafiqul Islam, Hongyan Wang, Hao Guo, Zhenshuo Wang, Xingjiang Qi, Shuwen Zhang, Junning Guo, Qi Wang, and Bin Li. 2022. "Effect of Humic Acid on Soil Physical and Chemical Properties, Microbial Community Structure, and Metabolites of Decline Diseased Bayberry" International Journal of Molecular Sciences 23, no. 23: 14707. https://doi.org/10.3390/ijms232314707
APA StyleRen, H., Islam, M. S., Wang, H., Guo, H., Wang, Z., Qi, X., Zhang, S., Guo, J., Wang, Q., & Li, B. (2022). Effect of Humic Acid on Soil Physical and Chemical Properties, Microbial Community Structure, and Metabolites of Decline Diseased Bayberry. International Journal of Molecular Sciences, 23(23), 14707. https://doi.org/10.3390/ijms232314707







