Behavioral Phenotyping of Bbs6 and Bbs8 Knockout Mice Reveals Major Alterations in Communication and Anxiety
Abstract
1. Introduction
2. Results
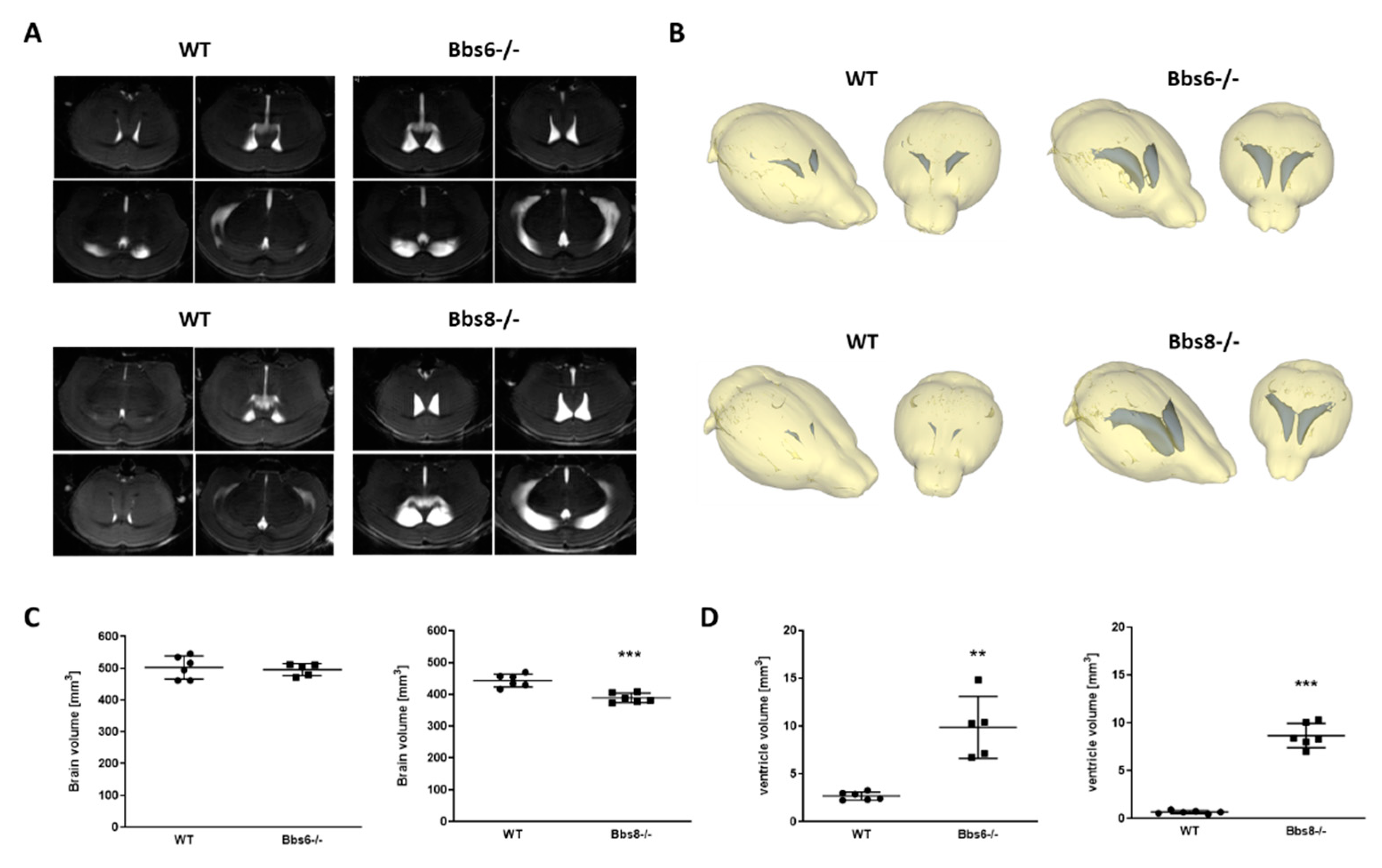
2.1. Enlarged Lateral Ventricles in Bbs6 and 8 Knockout Mice
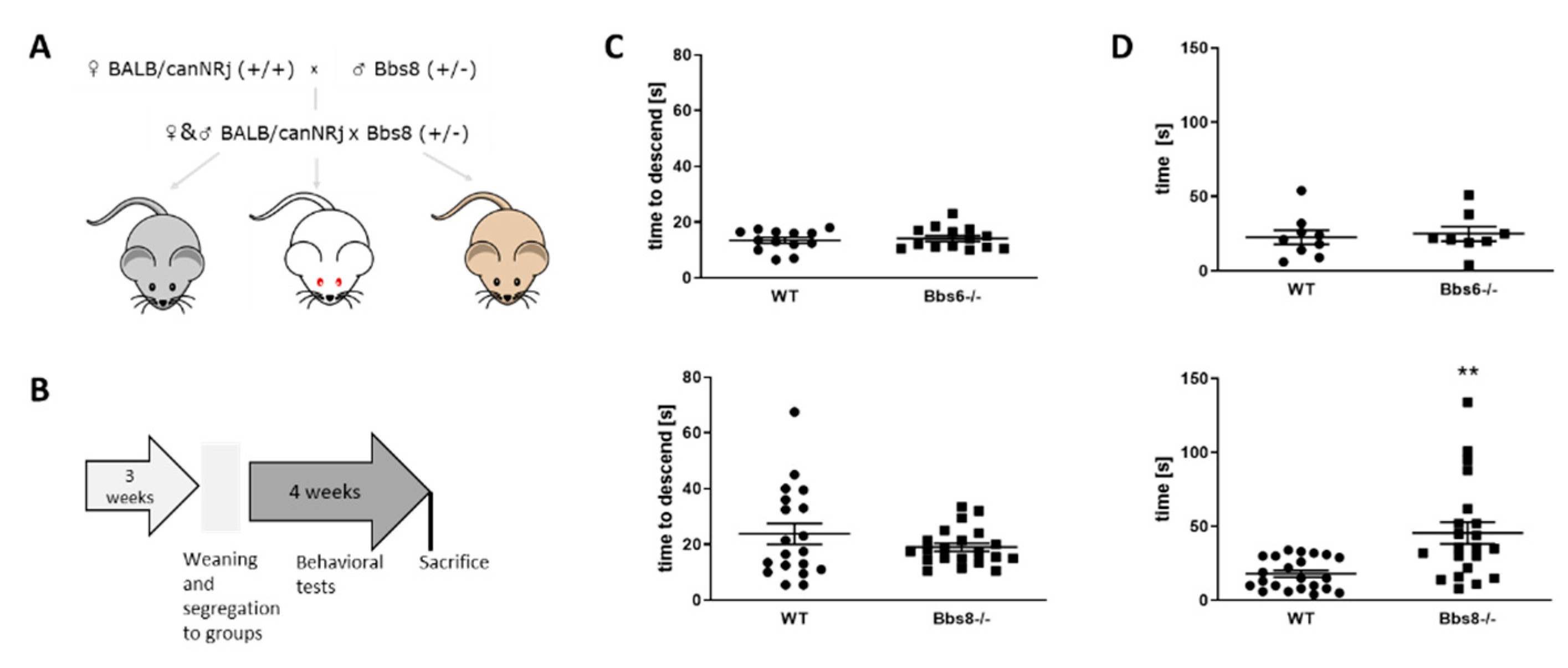
2.2. Behavioral Testing Reveals Early Olfactory Deficits in Bbs8 Knockout Mice
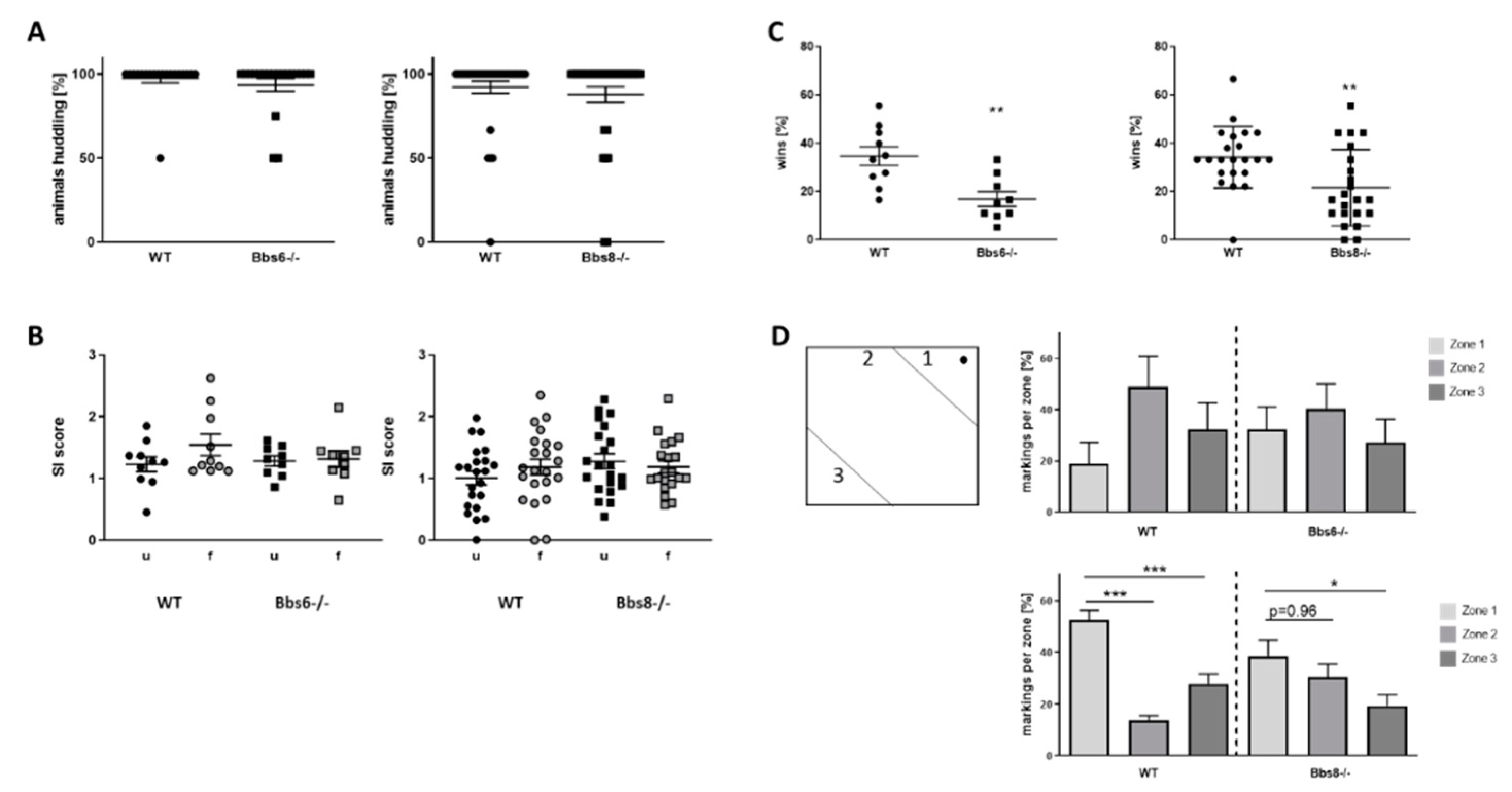
2.3. Socialization Is Unaffected in BBS Deficient Mice but Dominance-Related Behavior Is Reduced
2.4. Diminished Communicational Skills in BBS Knockout Mice
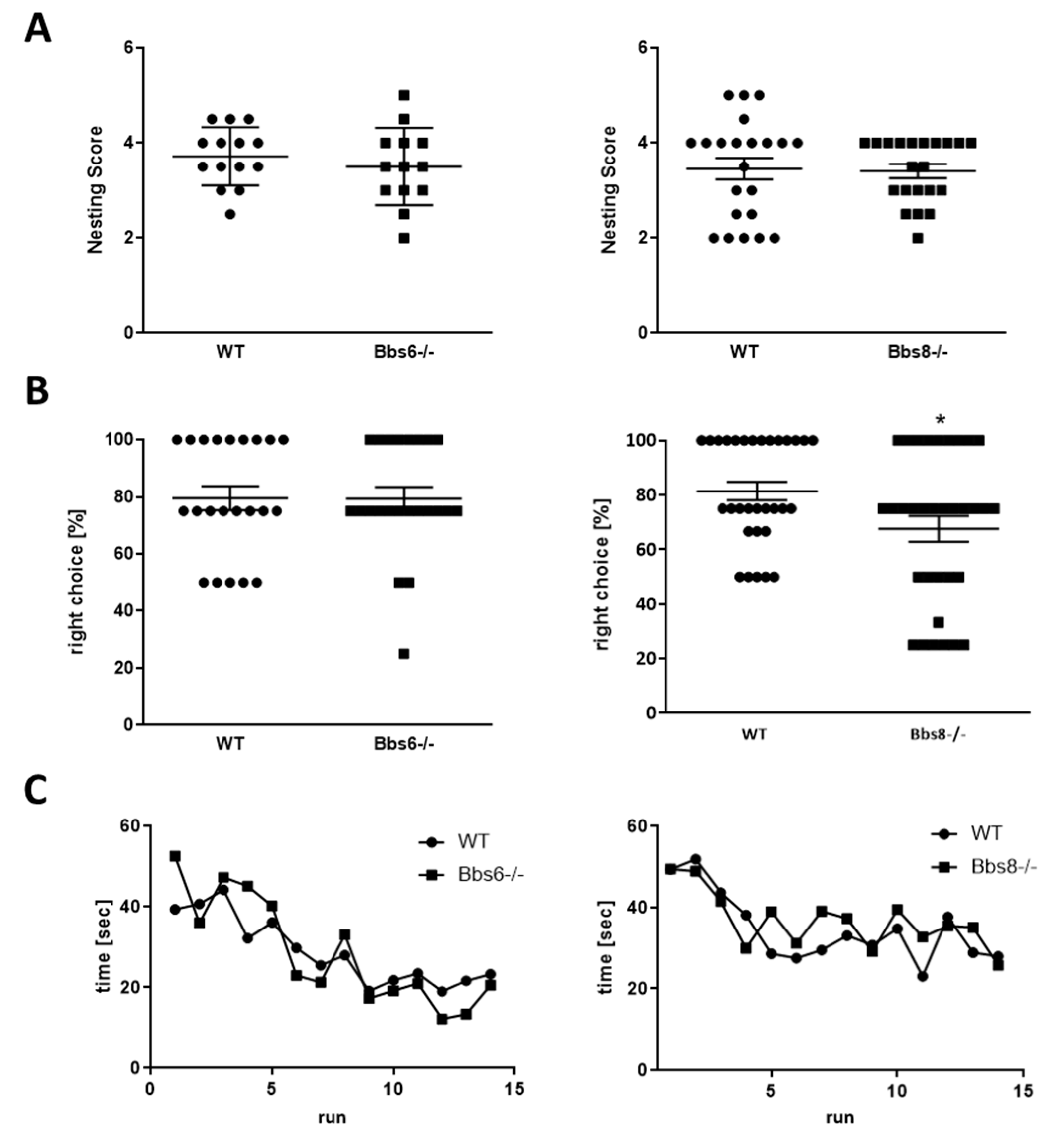
2.5. Cognitive Performance in Bbs6 and Bbs8 Knockout Mice Is Largely Unaffected
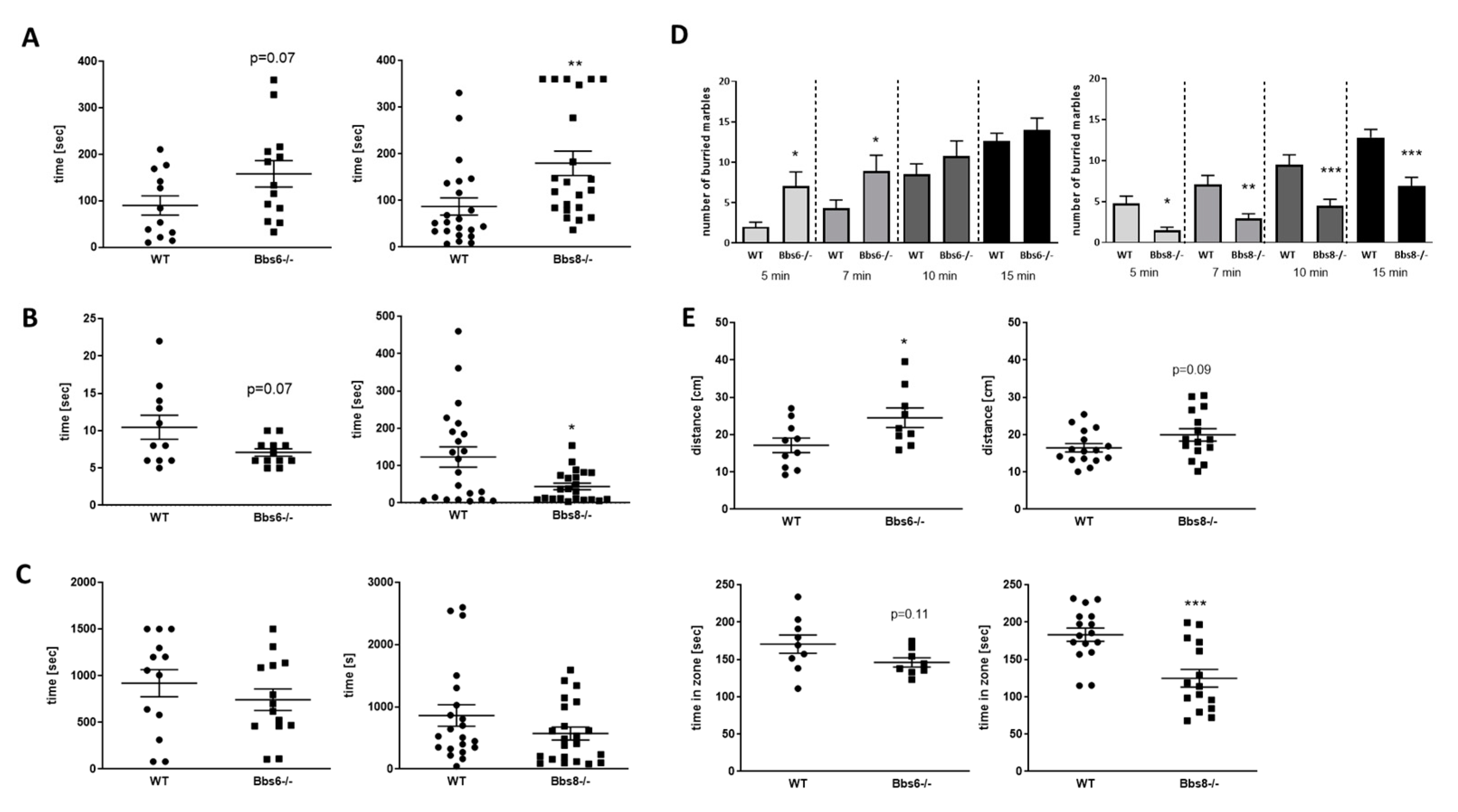
2.6. Bbs6 and Bbs8 Knockout Mice Display Reduced Anxiety in Non-Social Situations
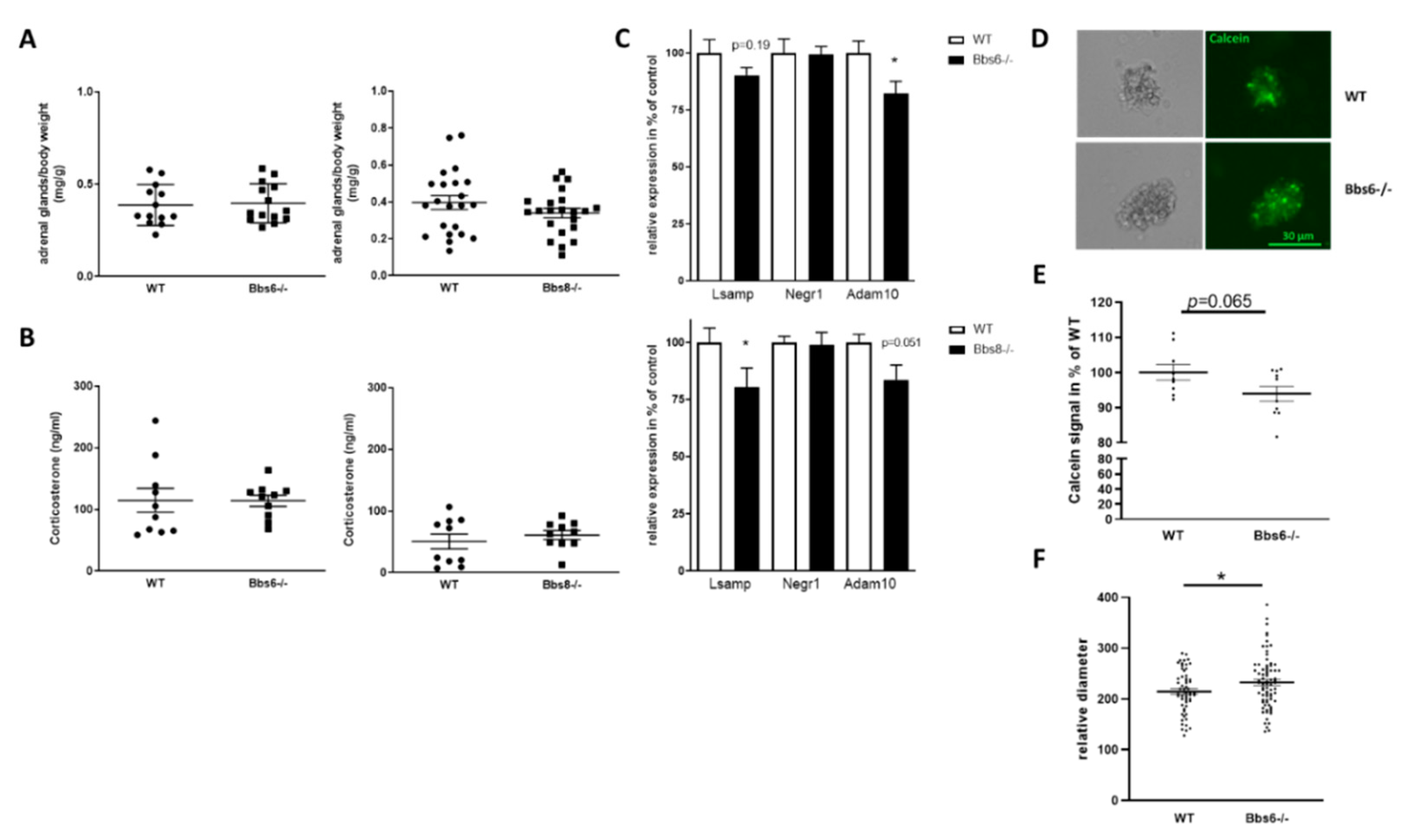
2.7. Impact of Bbs Protein Deficiency on the HPA Axis
3. Discussion
3.1. Bbs6 and 8 Knockout Mice Show Ventriculomegaly
3.2. Behavioral Phenotypes of Bbs6 and 8 Knockout Mice
3.3. Effect of BBS Deficiency on Hypothalamic Neurons
4. Materials and Methods
4.1. Animals
4.2. MRI and Volumetrics
4.3. Behavioral Tests
- Pole test
- Olfaction test
- Huddling test
- Social interaction
- Tube test
- Urine Scent Marking test
- Pasta gnawing test
- Ultra sonic vocalization assessment
- Nest building
- Radial Arm Water Maze
- T-Maze
- Forced Swim test
- Neophobia test
- Novelty induced repression of feeding test
- Marble burying test
- Open field-assessed parameters
4.4. Dissection
4.5. Corticosterone Measurement in Serum
4.6. RNA Preparation and qPCR
4.7. Neurosphere Culture
4.8. Calcein Staining and Neurosphere Size Determination
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Wallmeier, J.; Nielsen, K.G.; Kuehni, C.E.; Lucas, J.S.; Leigh, M.W.; Zariwala, M.A.; Omran, H. Motile ciliopathies. Nat. Rev. Dis. Primers 2020, 6, 77. [Google Scholar] [CrossRef] [PubMed]
- Anvarian, Z.; Mykytyn, K.; Mukhopadhyay, S.; Pedersen, L.B.; Christensen, S.T. Cellular signalling by primary cilia in development, organ function and disease. Nat. Rev. Nephrol. 2019, 15, 199–219. [Google Scholar] [CrossRef] [PubMed]
- Wheway, G.; Lord, J.; Baralle, D. Splicing in the pathogenesis, diagnosis and treatment of ciliopathies. Biochim. Biophys. Acta Gene Regul. Mech. 2019, 1862, 194433. [Google Scholar] [CrossRef]
- Bardet, G. On Congenital Obesity Syndrome with Polydactyly and Retinitis-Pigmentosa—(a Contribution to the Study of Clinical Forms of Hypophyseal Obesity) (Reprinted from Thesis, Faculte De Medecine De Paris, 1920). Obes. Res. 1995, 3, 387–399. [Google Scholar] [CrossRef]
- Biedl, A. A pair of siblings with adiposo-genital dystrophy. 1922. Obes. Res. 1995, 3, 404. [Google Scholar] [CrossRef] [PubMed]
- Beales, P.L.; Elcioglu, N.; Woolf, A.S.; Parker, D.; Flinter, F.A. New criteria for improved diagnosis of Bardet-Biedl syndrome: Results of a population survey. J. Med. Genet. 1999, 36, 437–446. [Google Scholar] [CrossRef]
- Forsythe, E.; Beales, P.L. Bardet-Biedl syndrome. Eur. J. Hum. Genet. 2013, 21, 8–13. [Google Scholar] [CrossRef]
- Fath, M.A.; Mullins, R.F.; Searby, C.; Nishimura, D.Y.; Wei, J.; Rahmouni, K.; Davis, R.E.; Tayeh, M.K.; Andrews, M.; Yang, B.; et al. Mkks-null mice have a phenotype resembling Bardet-Biedl syndrome. Hum. Mol. Genet. 2005, 14, 1109–1118. [Google Scholar] [CrossRef]
- Nishimura, D.Y.; Fath, M.; Mullins, R.F.; Searby, C.; Andrews, M.; Davis, R.; Andorf, J.L.; Mykytyn, K.; Swiderski, R.E.; Yang, B.; et al. Bbs2-null mice have neurosensory deficits, a defect in social dominance, and retinopathy associated with mislocalization of rhodopsin. Proc. Natl. Acad. Sci. USA 2004, 101, 16588–16593. [Google Scholar] [CrossRef]
- Ross, A.J.; May-Simera, H.; Eichers, E.R.; Kai, M.; Hill, J.; Jagger, D.J.; Leitch, C.C.; Chapple, J.P.; Munro, P.M.; Fisher, S.; et al. Disruption of Bardet-Biedl syndrome ciliary proteins perturbs planar cell polarity in vertebrates. Nat. Genet. 2005, 37, 1135–1140. [Google Scholar] [CrossRef]
- Tadenev, A.L.; Kulaga, H.M.; May-Simera, H.L.; Kelley, M.W.; Katsanis, N.; Reed, R.R. Loss of Bardet-Biedl syndrome protein-8 (BBS8) perturbs olfactory function, protein localization, and axon targeting. Proc. Natl. Acad. Sci. USA 2011, 108, 10320–10325. [Google Scholar] [CrossRef] [PubMed]
- Kretschmer, V.; Patnaik, S.R.; Kretschmer, F.; Chawda, M.M.; Hernandez-Hernandez, V.; May-Simera, H.L. Progressive Characterization of Visual Phenotype in Bardet-Biedl Syndrome Mutant Mice. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1132–1143. [Google Scholar] [CrossRef]
- Eichers, E.R.; Abd-El-Barr, M.M.; Paylor, R.; Lewis, R.A.; Bi, W.; Lin, X.; Meehan, T.P.; Stockton, D.W.; Wu, S.M.; Lindsay, E.; et al. Phenotypic characterization of Bbs4 null mice reveals age-dependent penetrance and variable expressivity. Hum. Genet. 2006, 120, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Pak, T.K.; Carter, C.S.; Zhang, Q.; Huang, S.C.; Searby, C.; Hsu, Y.; Taugher, R.J.; Vogel, T.; Cychosz, C.C.; Genova, R.; et al. A mouse model of Bardet-Biedl Syndrome has impaired fear memory, which is rescued by lithium treatment. PLoS Genet. 2021, 17, e1009484. [Google Scholar] [CrossRef]
- Barnett, S.; Reilly, S.; Carr, L.; Ojo, I.; Beales, P.L.; Charman, T. Behavioural phenotype of Bardet-Biedl syndrome. J. Med. Genet. 2002, 39, e76. [Google Scholar] [CrossRef] [PubMed]
- Nachury, M.V.; Loktev, A.V.; Zhang, Q.; Westlake, C.J.; Peranen, J.; Merdes, A.; Slusarski, D.C.; Scheller, R.H.; Bazan, J.F.; Sheffield, V.C.; et al. A core complex of BBS proteins cooperates with the GTPase Rab8 to promote ciliary membrane biogenesis. Cell 2007, 129, 1201–1213. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, S.R.; Kretschmer, V.; Brucker, L.; Schneider, S.; Volz, A.K.; Oancea-Castillo, L.D.R.; May-Simera, H.L. Bardet-Biedl Syndrome proteins regulate cilia disassembly during tissue maturation. Cell. Mol. Life Sci. 2019, 76, 757–775. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.; De Cegli, R.; Nagarajan, J.; Kretschmer, V.; Matthiessen, P.A.; Intartaglia, D.; Hotaling, N.; Ueffing, M.; Boldt, K.; Conte, I.; et al. Loss of Ciliary Gene Bbs8 Results in Physiological Defects in the Retinal Pigment Epithelium. Front. Cell Dev. Biol. 2021, 9, 607121. [Google Scholar] [CrossRef]
- Zhang, Q.; Nishimura, D.; Seo, S.; Vogel, T.; Morgan, D.A.; Searby, C.; Bugge, K.; Stone, E.M.; Rahmouni, K.; Sheffield, V.C. Bardet-Biedl syndrome 3 (Bbs3) knockout mouse model reveals common BBS-associated phenotypes and Bbs3 unique phenotypes. Proc. Natl. Acad. Sci. USA 2011, 108, 20678–20683. [Google Scholar] [CrossRef]
- Novielli-Kuntz, N.M.; Press, E.R.; Barr, K.; Prado, M.A.M.; Laird, D.W. Mutant Cx30-A88V mice exhibit hydrocephaly and sex-dependent behavioral abnormalities, implicating a functional role for Cx30 in the brain. Dis. Model. Mech. 2021, 14, dmm046235. [Google Scholar] [CrossRef]
- Minkeviciene, R.; Hlushchenko, I.; Virenque, A.; Lahti, L.; Khanal, P.; Rauramaa, T.; Koistinen, A.; Leinonen, V.; Noe, F.M.; Hotulainen, P. MIM-Deficient Mice Exhibit Anatomical Changes in Dendritic Spines, Cortex Volume and Brain Ventricles, and Functional Changes in Motor Coordination and Learning. Front. Mol. Neurosci. 2019, 12, 276. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Jayabal, S.; Ghorbani, M.; Legault, L.M.; McGraw, S.; Watt, A.J.; Yang, X.J. ATAT1 regulates forebrain development and stress-induced tubulin hyperacetylation. Cell. Mol. Life Sci. 2019, 76, 3621–3640. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, T.; Silversides, D.W.; Waymire, K.G.; Kwon, B.S.; Takeuchi, T.; Overbeek, P.A. Conserved cysteine to serine mutation in tyrosinase is responsible for the classical albino mutation in laboratory mice. Nucleic Acids Res. 1990, 18, 7293–7298. [Google Scholar] [CrossRef] [PubMed]
- Gadomsky, L.; Dos Santos Guilherme, M.; Winkler, J.; van der Kooij, M.A.; Hartmann, T.; Grimm, M.; Endres, K. Elevated Testosterone Level and Urine Scent Marking in Male 5xFAD Alzheimer Model Mice. Curr. Alzheimer Res. 2020, 17, 80–92. [Google Scholar] [CrossRef]
- Sangiamo, D.T.; Warren, M.R.; Neunuebel, J.P. Ultrasonic signals associated with different types of social behavior of mice. Nat. Neurosci. 2020, 23, 411–422. [Google Scholar] [CrossRef]
- Mockel, A.; Perdomo, Y.; Stutzmann, F.; Letsch, J.; Marion, V.; Dollfus, H. Retinal dystrophy in Bardet-Biedl syndrome and related syndromic ciliopathies. Prog. Retin. Eye Res. 2011, 30, 258–274. [Google Scholar] [CrossRef]
- Karamihalev, S.; Brivio, E.; Flachskamm, C.; Stoffel, R.; Schmidt, M.V.; Chen, A. Social dominance mediates behavioral adaptation to chronic stress in a sex-specific manner. eLife 2020, 9, e58723. [Google Scholar] [CrossRef]
- De Brouwer, G.; Fick, A.; Harvey, B.H.; Wolmarans, D. A critical inquiry into marble-burying as a preclinical screening paradigm of relevance for anxiety and obsessive-compulsive disorder: Mapping the way forward. Cogn. Affect. Behav. Neurosci. 2019, 19, 1–39. [Google Scholar] [CrossRef]
- Thomas, A.; Burant, A.; Bui, N.; Graham, D.; Yuva-Paylor, L.A.; Paylor, R. Marble burying reflects a repetitive and perseverative behavior more than novelty-induced anxiety. Psychopharmacology 2009, 204, 361–373. [Google Scholar] [CrossRef]
- Hoffman, K. Modeling Neuropsychiatric Disorders in Laboratory Animals; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Carboni, L.; Pischedda, F.; Piccoli, G.; Lauria, M.; Musazzi, L.; Popoli, M.; Mathe, A.A.; Domenici, E. Depression-Associated GeneNegr1-Fgfr2Pathway Is Altered by Antidepressant Treatment. Cells 2020, 9, 1818. [Google Scholar] [CrossRef]
- Nelovkov, A.; Philips, M.A.; Koks, S.; Vasar, E. Rats with low exploratory activity in the elevated plus-maze have the increased expression of limbic system-associated membrane protein gene in the periaqueductal grey. Neurosci. Lett. 2003, 352, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Sanz, R.L.; Ferraro, G.B.; Girouard, M.P.; Fournier, A.E. Ectodomain shedding of Limbic System-Associated Membrane Protein (LSAMP) by ADAM Metallopeptidases promotes neurite outgrowth in DRG neurons. Sci. Rep. 2017, 7, 7961. [Google Scholar] [CrossRef] [PubMed]
- Pischedda, F.; Piccoli, G. The IgLON Family Member Negri Promotes Neuronal Arborization Acting as Soluble Factor via FGFR2. Front. Mol. Neurosci. 2016, 8, 89. [Google Scholar] [CrossRef] [PubMed]
- Fearnley, S.; Raja, R.; Cloutier, J.F. Spatiotemporal expression of IgLON family members in the developing mouse nervous system. Sci. Rep. 2021, 11, 19536. [Google Scholar] [CrossRef]
- Singh, K.; Lillevali, K.; Gilbert, S.F.; Bregin, A.; Narvik, J.; Jayaram, M.; Rahi, M.; Innos, J.; Kaasik, A.; Vasar, E.; et al. The combined impact of IgLON family proteins Lsamp and Neurotrimin on developing neurons and behavioral profiles in mouse. Brain Res. Bull. 2018, 140, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Jorissen, E.; Prox, J.; Bernreuther, C.; Weber, S.; Schwanbeck, R.; Serneels, L.; Snellinx, A.; Craessaerts, K.; Thathiah, A.; Tesseur, I.; et al. The disintegrin/metalloproteinase ADAM10 is essential for the establishment of the brain cortex. J. Neurosci. 2010, 30, 4833–4844. [Google Scholar] [CrossRef]
- Zielinska, D.; Rajtar-Zembaty, A.; Starowicz-Filip, A. Cognitive disorders in children’s hydrocephalus. Neurol. Neurochir. Pol. 2017, 51, 234–239. [Google Scholar] [CrossRef]
- Kumar, V.; Umair, Z.; Kumar, S.; Goutam, R.S.; Park, S.; Kim, J. The regulatory roles of motile cilia in CSF circulation and hydrocephalus. Fluids Barriers CNS 2021, 18, 31. [Google Scholar] [CrossRef]
- Ji, W.Y.; Tang, Z.; Chen, Y.B.; Wang, C.S.; Tan, C.W.; Liao, J.B.; Tong, L.; Xiao, G.L. Ependymal Cilia: Physiology and Role in Hydrocephalus. Front. Mol. Neurosci. 2022, 15, 927479. [Google Scholar] [CrossRef]
- Carter, C.S.; Vogel, T.W.; Zhang, Q.H.; Seo, S.; Swiderski, R.E.; Moninger, T.O.; Cassell, M.D.; Thedens, D.R.; Keppler-Noreuil, K.M.; Nopoulos, P.; et al. Abnormal development of NG2(+)PDGFR-alpha(+) neural progenitor cells leads to neonatal hydrocephalus in a ciliopathy mouse model. Nat. Med. 2012, 18, 1797–1804. [Google Scholar] [CrossRef]
- Davis, R.E.; Swiderski, R.E.; Rahmouni, K.; Nishimura, D.Y.; Mullins, R.F.; Agassandian, K.; Philp, A.R.; Searby, C.C.; Andrews, M.P.; Thompson, S.; et al. A knockin mouse model of the Bardet-Biedl syndrome 1 M390R mutation has cilia defects, ventriculomegaly, retinopathy, and obesity. Proc. Natl. Acad. Sci. USA 2007, 104, 19422–19427. [Google Scholar] [CrossRef] [PubMed]
- Keppler-Noreuil, K.M.; Blumhorst, C.; Sapp, J.C.; Brinckman, D.; Johnston, J.; Nopoulos, P.C.; Biesecker, L.G. Brain tissue- and region-specific abnormalities on volumetric MRI scans in 21 patients with Bardet-Biedl syndrome (BBS). BMC Med. Genet. 2011, 12, 101. [Google Scholar] [CrossRef] [PubMed]
- Wapner, R.J.; Kurtz, A.B.; Ross, R.D.; Jackson, L.G. Ultrasonographic Parameters in the Prenatal-Diagnosis of Meckel Syndrome. Obstet. Gynecol. 1981, 57, 388–392. [Google Scholar] [PubMed]
- Swiderski, R.E.; Agassandian, K.; Ross, J.L.; Bugge, K.; Cassell, M.D.; Yeaman, C. Structural defects in cilia of the choroid plexus, subfornical organ and ventricular ependyma are associated with ventriculomegaly. Fluids Barriers CNS 2012, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Narita, K.; Kawate, T.; Kakinuma, N.; Takeda, S. Multiple primary cilia modulate the fluid transcytosis in choroid plexus epithelium. Traffic 2010, 11, 287–301. [Google Scholar] [CrossRef]
- Singh, M.; Garrison, J.E.; Wang, K.; Sheffield, V.C. Absence of BBSome function leads to astrocyte reactivity in the brain. Mol. Brain 2019, 12, 48. [Google Scholar] [CrossRef]
- Moy, S.S.; Nadler, J.J.; Young, N.B.; Nonneman, R.J.; Segall, S.K.; Andrade, G.M.; Crawley, J.N.; Magnuson, T.R. Social approach and repetitive behavior in eleven inbred mouse strains. Behav. Brain Res. 2008, 191, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, S.R.; Farag, A.; Brucker, L.; Volz, A.K.; Schneider, S.; Kretschmer, V.; May-Simera, H.L. Tissue-dependent differences in Bardet-Biedl syndrome gene expression. Biol. Cell 2020, 112, 39–52. [Google Scholar] [CrossRef]
- Dahmer-Heath, M.; Schriever, V.; Kollmann, S.; Schleithoff, C.; Titieni, A.; Cetiner, M.; Patzer, L.; Tonshoff, B.; Hansen, M.; Pennekamp, P.; et al. Systematic evaluation of olfaction in patients with hereditary cystic kidney diseases/renal ciliopathies. J. Med. Genet. 2021, 58, 629–636. [Google Scholar] [CrossRef]
- Iannaccone, A.; Mykytyn, K.; Persico, A.M.; Searby, C.C.; Baldi, A.; Jablonski, M.M.; Sheffield, V.C. Clinical evidence of decreased olfaction in Bardet-Biedl syndrome caused by a deletion in the BBS4 gene. Am. J. Med. Genet. A 2005, 132A, 343–346. [Google Scholar] [CrossRef]
- Kulaga, H.M.; Leitch, C.C.; Eichers, E.R.; Badano, J.L.; Lesemann, A.; Hoskins, B.E.; Lupski, J.R.; Beales, P.L.; Reed, R.R.; Katsanis, N. Loss of BBS proteins causes anosmia in humans and defects in olfactory cilia structure and function in the mouse. Nat. Genet. 2004, 36, 994–998. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.J.; Noblet, V.; Durand, M.; Scheidecker, S.; Zinetti-Bertschy, A.; Foucher, J.; Marion, V.; Muller, J.; Riehm, S.; Dollfus, H.; et al. Olfaction evaluation and correlation with brain atrophy in Bardet-Biedl syndrome. Clin. Genet. 2014, 86, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Cardenas-Rodriguez, M.; Irigoin, F.; Osborn, D.P.; Gascue, C.; Katsanis, N.; Beales, P.L.; Badano, J.L. The Bardet-Biedl syndrome-related protein CCDC28B modulates mTORC2 function and interacts with SIN1 to control cilia length independently of the mTOR complex. Hum. Mol. Genet. 2013, 22, 4031–4042. [Google Scholar] [CrossRef] [PubMed]
- Fabregat, M.; Nino-Rivero, S.; Pose, S.; Cardenas-Rodriguez, M.; Bresque, M.; Hernandez, K.; Prieto-Echague, V.; Schlapp, G.; Crispo, M.; Lagos, P.; et al. Generation and characterization of Ccdc28b mutant mice links the Bardet-Biedl associated gene with mild social behavioral phenotypes. PLoS Genet. 2022, 18, e1009896. [Google Scholar] [CrossRef] [PubMed]
- Brinckman, D.D.; Keppler-Noreuil, K.M.; Blumhorst, C.; Biesecker, L.G.; Sapp, J.C.; Johnston, J.J.; Wiggs, E.A. Cognitive, sensory, and psychosocial characteristics in patients with Bardet-Biedl syndrome. Am. J. Med. Genet. A 2013, 161A, 2964–2971. [Google Scholar] [CrossRef] [PubMed]
- Panny, A.; Glurich, I.; Haws, R.M.; Acharya, A. Oral and Craniofacial Anomalies of Bardet-Biedl Syndrome: Dental Management in the Context of a Rare Disease. J. Dent. Res. 2017, 96, 1361–1369. [Google Scholar] [CrossRef]
- Soliman, A.T.; Rajab, A.; AlSalmi, I.; Asfour, M.G. Empty sellae, impaired testosterone secretion, and defective hypothalamic-pituitary growth and gonadal axes in children with Bardet-Biedl syndrome. Metabolism 1996, 45, 1230–1234. [Google Scholar] [CrossRef]
- Reiter, J.F.; Leroux, M.R. Genes and molecular pathways underpinning ciliopathies. Nat. Rev. Mol. Cell Biol. 2017, 18, 533–547. [Google Scholar] [CrossRef]
- Bennouna-Greene, V.; Kremer, S.; Stoetzel, C.; Christmann, D.; Schuster, C.; Durand, M.; Verloes, A.; Sigaudy, S.; Holder-Espinasse, M.; Godet, J.; et al. Hippocampal dysgenesis and variable neuropsychiatric phenotypes in patients with Bardet-Biedl syndrome underline complex CNS impact of primary cilia. Clin. Genet. 2011, 80, 523–531. [Google Scholar] [CrossRef]
- Kerr, E.N.; Bhan, A.; Heon, E. Exploration of the cognitive, adaptive and behavioral functioning of patients affected with Bardet-Biedl syndrome. Clin. Genet. 2016, 89, 426–433. [Google Scholar] [CrossRef]
- Haq, N.; Schmidt-Hieber, C.; Sialana, F.J.; Ciani, L.; Heller, J.P.; Stewart, M.; Bentley, L.; Wells, S.; Rodenburg, R.J.; Nolan, P.M.; et al. Loss of Bardet-Biedl syndrome proteins causes synaptic aberrations in principal neurons. PLoS Biol. 2019, 17, e3000414. [Google Scholar] [CrossRef] [PubMed]
- Agassandian, K.; Patel, M.; Agassandian, M.; Steren, K.E.; Rahmouni, K.; Sheffield, V.C.; Card, J.P. Ciliopathy is differentially distributed in the brain of a Bardet-Biedl syndrome mouse model. PLoS ONE 2014, 9, e93484. [Google Scholar] [CrossRef] [PubMed]
- Hammelrath, L.; Skokic, S.; Khmelinskii, A.; Hess, A.; van der Knaap, N.; Staring, M.; Lelieveldt, B.P.F.; Wiedermann, D.; Hoehn, M. Morphological maturation of the mouse brain: An in vivo MRI and histology investigation. Neuroimage 2016, 125, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Blaess, S.; Wachten, D. The BBSome: A nexus controlling energy metabolism in the brain. J. Clin. Investig. 2021, 131, e148903. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Y.; Stratigopoulos, G.; Panigrahi, S.; Sui, L.; Zhang, Y.; Leduc, C.A.; Glover, H.J.; De Rosa, M.C.; Burnett, L.C.; et al. Bardet-Biedl syndrome proteins regulate intracellular signaling and neuronal function in patient-specific iPSC-derived neurons. J. Clin. Investig. 2021, 131, e146287. [Google Scholar] [CrossRef]
- Sanz, R.; Ferraro, G.B.; Fournier, A.E. IgLON cell adhesion molecules are shed from the cell surface of cortical neurons to promote neuronal growth. J. Biol. Chem. 2015, 290, 4330–4342. [Google Scholar] [CrossRef]
- Hashimoto, T.; Maekawa, S.; Miyata, S. IgLON cell adhesion molecules regulate synaptogenesis in hippocampal neurons. Cell Biochem. Funct. 2009, 27, 496–498. [Google Scholar] [CrossRef]
- Innos, J.; Philips, M.A.; Leidmaa, E.; Heinla, I.; Raud, S.; Reemann, P.; Plaas, M.; Nurk, K.; Kurrikoff, K.; Matto, V.; et al. Lower anxiety and a decrease in agonistic behaviour in Lsamp-deficient mice. Behav. Brain Res. 2011, 217, 21–31. [Google Scholar] [CrossRef]
- Innos, J.; Philips, M.A.; Raud, S.; Lillevali, K.; Koks, S.; Vasar, E. Deletion of the Lsamp gene lowers sensitivity to stressful environmental manipulations in mice. Behav. Brain Res. 2012, 228, 74–81. [Google Scholar] [CrossRef]
- Catania, E.H.; Pimenta, A.; Levitt, P. Genetic deletion of Lsamp causes exaggerated behavioral activation in novel environments. Behav Brain Res. 2008, 188, 380–390. [Google Scholar] [CrossRef]
- Philips, M.A.; Lillevali, K.; Heinla, I.; Luuk, H.; Hundahl, C.A.; Kongi, K.; Vanaveski, T.; Tekko, T.; Innos, J.; Vasar, E. Lsamp is implicated in the regulation of emotional and social behavior by use of alternative promoters in the brain. Brain Struct. Funct. 2015, 220, 1381–1393. [Google Scholar] [CrossRef] [PubMed]
- Behan, A.T.; Byrne, C.; Dunn, M.J.; Cagney, G.; Cotter, D.R. Proteomic analysis of membrane microdomain-associated proteins in the dorsolateral prefrontal cortex in schizophrenia and bipolar disorder reveals alterations in LAMP, STXBP1 and BASP1 protein expression. Mol. Psychiatr. 2009, 14, 601–613. [Google Scholar] [CrossRef]
- Koido, K.; Traks, T.; Balotsev, R.; Eller, T.; Must, A.; Koks, S.; Maron, E.; Toru, I.; Shlik, J.; Vasar, V.; et al. Associations between LSAMP gene polymorphisms and major depressive disorder and panic disorder. Transl. Psychiatry 2012, 2, e152. [Google Scholar] [CrossRef] [PubMed]
- Saftig, P.; Lichtenthaler, S.F. The alpha secretase ADAM10: A metalloprotease with multiple functions in the brain. Prog. Neurobiol. 2015, 135, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Pala, R.; Alomari, N.; Nauli, S.M. Primary Cilium-Dependent Signaling Mechanisms. Int J. Mol. Sci. 2017, 18, 2272. [Google Scholar] [CrossRef] [PubMed]
- Tippmann, F.; Hundt, J.; Schneider, A.; Endres, K.; Fahrenholz, F. Up-regulation of the alpha-secretase ADAM10 by retinoic acid receptors and acitretin. FASEB J. 2009, 23, 1643–1654. [Google Scholar] [CrossRef]
- Endres, K.; Fahrenholz, F.; Lotz, J.; Hiemke, C.; Teipel, S.; Lieb, K.; Tuscher, O.; Fellgiebel, A. Increased CSF APPs-alpha levels in patients with Alzheimer disease treated with acitretin. Neurology 2014, 83, 1930–1935. [Google Scholar] [CrossRef]
- Aedo-Jury, F.; Schwalm, M.; Hamzehpour, L.; Stroh, A. Brain states govern the spatio-temporal dynamics of resting-state functional connectivity. eLife 2020, 9, e53186. [Google Scholar] [CrossRef]
- Wilson, S.P. Modelling the emergence of rodent filial huddling from physiological huddling. R. Soc. Open Sci. 2017, 4, 170885. [Google Scholar] [CrossRef]
- Guilherme, M.D.; Tsoutsouli, T.; Todorov, H.; Teifel, S.; Nguyen, V.T.T.; Gerber, S.; Endres, K. N-6-Methyladenosine Modification in Chronic Stress Response Due to Social Hierarchy Positioning of Mice. Front. Cell Dev. Biol. 2021, 9, 705986. [Google Scholar] [CrossRef]
- Dos Santos Guilherme, M.; Valeri, F.; Winter, J.; Muller, M.B.; Schwiertz, A.; Endres, K. Resilience and the Gut Microbiome: Insights from Chronically Socially Stressed Wild-Type Mice. Microorganisms 2022, 10, 1077. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos Guilherme, M.; Stoye, N.M.; Rose-John, S.; Garbers, C.; Fellgiebel, A.; Endres, K. The Synthetic Retinoid Acitretin Increases IL-6 in the Central Nervous System of Alzheimer Disease Model. Mice and Human Patients. Front. Aging Neurosci. 2019, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Valeri, F.; Dos Santos Guilherme, M.; He, F.; Stoye, N.M.; Schwiertz, A.; Endres, K. Impact of the Age of Cecal Material Transfer Donors on Alzheimer’s Disease Pathology in 5xFAD Mice. Microorganisms 2021, 9, 2548. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.W.; Hengstler, H.; Schwald, K.; Berriel-Diaz, M.; Loreth, D.; Kirsch, M.; Kretz, O.; Haas, C.A.; de Angelis, M.H.; Herzig, S.; et al. Functional inactivation of the genome-wide association study obesity gene neuronal growth regulator 1 in mice causes a body mass phenotype. PLoS ONE 2012, 7, e41537. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rödig, N.; Sellmann, K.; dos Santos Guilherme, M.; Nguyen, V.T.T.; Cleppien, D.; Stroh, A.; May-Simera, H.L.; Endres, K. Behavioral Phenotyping of Bbs6 and Bbs8 Knockout Mice Reveals Major Alterations in Communication and Anxiety. Int. J. Mol. Sci. 2022, 23, 14506. https://doi.org/10.3390/ijms232314506
Rödig N, Sellmann K, dos Santos Guilherme M, Nguyen VTT, Cleppien D, Stroh A, May-Simera HL, Endres K. Behavioral Phenotyping of Bbs6 and Bbs8 Knockout Mice Reveals Major Alterations in Communication and Anxiety. International Journal of Molecular Sciences. 2022; 23(23):14506. https://doi.org/10.3390/ijms232314506
Chicago/Turabian StyleRödig, Nathalie, Kristin Sellmann, Malena dos Santos Guilherme, Vu Thu Thuy Nguyen, Dirk Cleppien, Albrecht Stroh, Helen Louise May-Simera, and Kristina Endres. 2022. "Behavioral Phenotyping of Bbs6 and Bbs8 Knockout Mice Reveals Major Alterations in Communication and Anxiety" International Journal of Molecular Sciences 23, no. 23: 14506. https://doi.org/10.3390/ijms232314506
APA StyleRödig, N., Sellmann, K., dos Santos Guilherme, M., Nguyen, V. T. T., Cleppien, D., Stroh, A., May-Simera, H. L., & Endres, K. (2022). Behavioral Phenotyping of Bbs6 and Bbs8 Knockout Mice Reveals Major Alterations in Communication and Anxiety. International Journal of Molecular Sciences, 23(23), 14506. https://doi.org/10.3390/ijms232314506








