TERT Promoter Mutations as Simple and Non-Invasive Urinary Biomarkers for the Detection of Urothelial Bladder Cancer in a High-Risk Region
Abstract
1. Introduction
2. Results
2.1. Descriptive Statistics
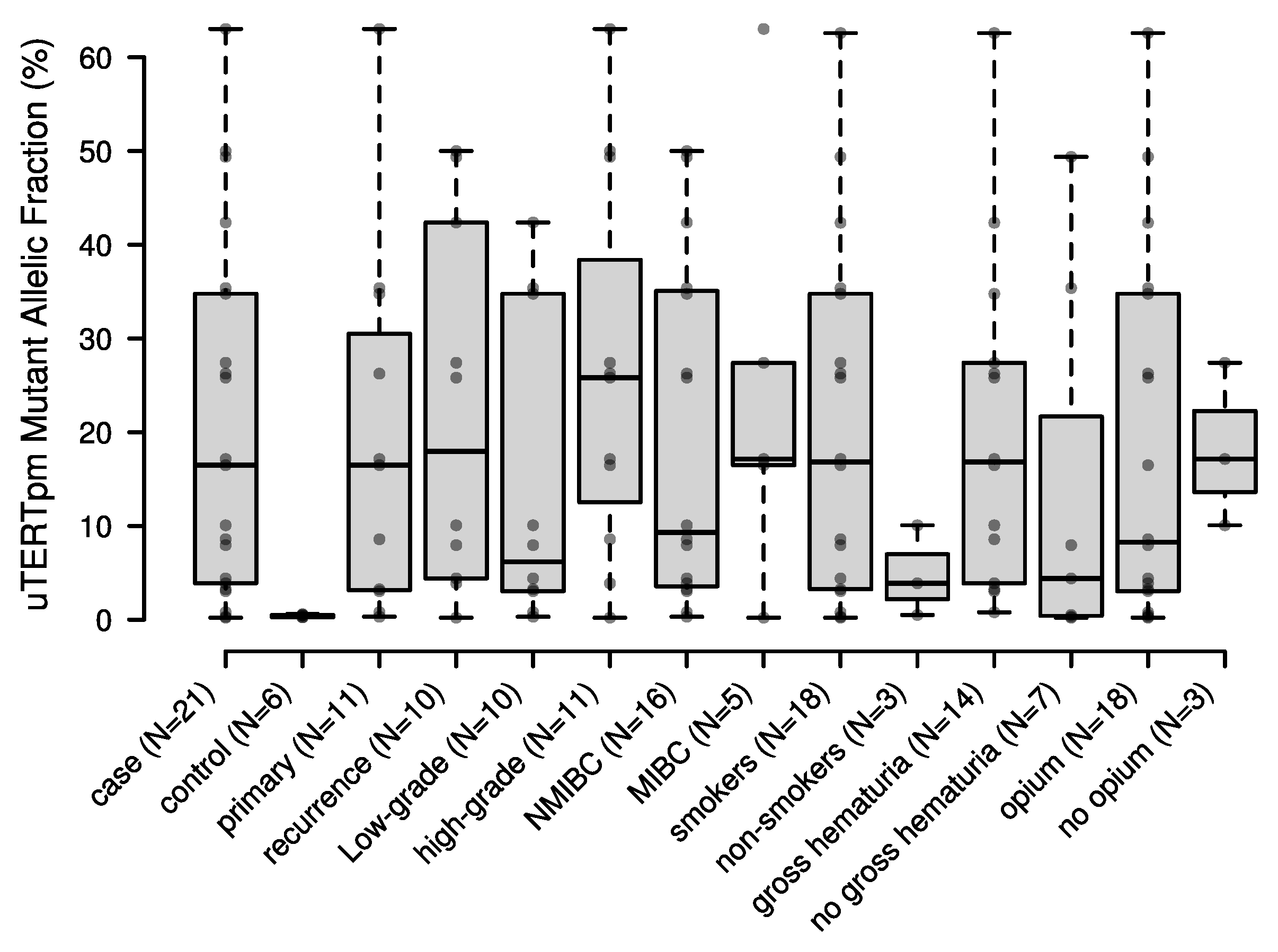
2.2. Performance of Urinary TERT Promoter Mutations (uTERTpm) in Detecting BC
2.3. Performance of Urine Cytology Alone and in Combination of uTERTpm in Detecting BC
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Study Design
4.3. Preparation and Storage of Urine Samples
4.4. Detection of TERT Promoter Mutations Using ddPCR Assay
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Goodarzi, E.; Sohrabivafa, M.; Dehkordi, A.H.; Khazaei, Z. Worldwide incidence and mortality of bladder cancer and human development index: An ecological study. Indian J. Med. Spéc. 2020, 11, 88–97. [Google Scholar] [CrossRef]
- Salehiniya, H.; Rafiemanesh, H.; Lotfi, Z.; Bakhtazad, S.; Ghoncheh, M. The epidemiological and histological trend of bladder cancer in Iran. J. Cancer Res. Ther. 2018, 14, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Brennan, P.; Bogillot, O.; Cordier, S.; Greiser, E.; Schill, W.; Vineis, P.; Lopez-Abente, G.; Tzonou, A.; Chang-Claude, J.; Bolm-Audorff, U.; et al. Cigarette smoking and bladder cancer in men: A pooled analysis of 11 case-control studies. Int. J. Cancer 2000, 86, 289–294. [Google Scholar] [CrossRef]
- Zaghloul, M.S. Bladder cancer and schistosomiasis. J. Egypt. Natl. Cancer Inst. 2012, 24, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Abdolahinia, Z.; Pakmanesh, H.; Mirzaee, M.; Bazrafshan, A.; Bafti, M.S.; Shahesmaeili, A. Opium and Cigarette Smoking are Independently Associated with Bladder Cancer: The Findings of a Matched Case—Control Study. Asian Pac. J. Cancer Prev. 2021, 22, 3385–3391. [Google Scholar] [CrossRef]
- Hadji, M.; Rashidian, H.; Marzban, M.; Naghibzadeh-Tahami, A.; Gholipour, M.; Mohebbi, E.; Safari-Faramani, R.; Seyyedsalehi, M.S.; Hosseini, B.; Bakhshi, M.; et al. Opium use and risk of bladder cancer: A multicentre case-referent study in Iran. Int. J. Epidemiol. 2022, 51, 830–838. [Google Scholar] [CrossRef]
- Boffetta, P.; Autier, P.; Boniol, M.; Boyle, P.; Hill, C.; Aurengo, A.; Masse, R.; de Thé, G.; Valleron, A.-J.; Monier, R.; et al. An Estimate of Cancers Attributable to Occupational Exposures in France. J. Occup. Environ. Med. 2010, 52, 399–406. [Google Scholar] [CrossRef]
- Leal, J.; Luengo-Fernandez, R.; Sullivan, R.; Witjes, J.A. Economic Burden of Bladder Cancer Across the European Union. Eur. Urol. 2016, 69, 438–447. [Google Scholar] [CrossRef]
- Moch, H.; Cubilla, A.L.; Humphrey, P.A.; Reuter, V.E.; Ulbright, T.M. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs—Part A: Renal, Penile, and Testicular Tumours. Eur. Urol. 2016, 70, 93–105. [Google Scholar] [CrossRef]
- Bladder cancer: Diagnosis and management of bladder cancer: © NICE (2015). Bladder cancer: Diagnosis and management of bladder cancer. BJU Int. 2017, 120, 755–765. [CrossRef] [PubMed]
- Van Rhijn, B.W.; Burger, M.; Lotan, Y.; Solsona, E.; Stief, C.G.; Sylvester, R.J.; Witjes, J.A.; Zlotta, A.R. Recurrence and progression of disease in non-muscle-invasive bladder cancer: From epidemiology to treatment strategy. Eur. Urol. 2009, 56, 430–442. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.P.; Lieskovsky, G.; Cote, R.; Groshen, S.; Feng, A.C.; Boyd, S.; Skinner, E.; Bochner, B.; Thangathurai, D.; Mikhail, M.; et al. Radical cystectomy in the treatment of invasive bladder cancer: Long-term results in 1,054 patients. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2001, 19, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Witjes, J.A.; Bruins, H.M.; Cathomas, R.; Compérat, E.M.; Cowan, N.C.; Gakis, G.; Hernández, V.; Espinós, E.L.; Lorch, A.; Neuzillet, Y.; et al. European Association of Urology Guidelines on Muscle-invasive and Metastatic Bladder Cancer: Summary of the 2020 Guidelines. Eur. Urol. 2020, 79, 82–104. [Google Scholar] [CrossRef]
- Hollenbeck, B.K.; Ms, R.L.D.; Ye, Z.; Hollingsworth, J.M.; Skolarus, T.A.; Kim, S.P.; Montie, J.E.; Lee, C.T.; Wood, D.P.; Miller, D.C. Delays in diagnosis and bladder cancer mortality. Cancer 2010, 116, 5235–5242. [Google Scholar] [CrossRef] [PubMed]
- Allory, Y.; Beukers, W.; Sagrera, A.; Flández, M.; Marqués, M.; Márquez, M.; van der Keur, K.A.; Dyrskjot, L.; Lurkin, I.; Vermeij, M.; et al. Telomerase Reverse Transcriptase Promoter Mutations in Bladder Cancer: High Frequency Across Stages, Detection in Urine, and Lack of Association with Outcome. Eur. Urol. 2013, 65, 360–366. [Google Scholar] [CrossRef]
- Hurst, C.D.; Platt, F.M.; Knowles, M.A. Comprehensive Mutation Analysis of the TERT Promoter in Bladder Cancer and Detection of Mutations in Voided Urine. Eur. Urol. 2014, 65, 367–369. [Google Scholar] [CrossRef]
- Tilki, D.; Burger, M.; Dalbagni, G.; Grossman, H.B.; Hakenberg, O.W.; Palou, J.; Reich, O.; Rouprêt, M.; Shariat, S.F.; Zlotta, A.R. Urine Markers for Detection and Surveillance of Non–Muscle-Invasive Bladder Cancer. Eur. Urol. 2011, 60, 484–492. [Google Scholar] [CrossRef]
- Vinagre, J.; Pinto, V.; Celestino, R.; Reis, M.; Pópulo, H.; Boaventura, P.; Melo, M.; Catarino, T.; Lima, J.; Lopes, J.M.; et al. Telomerase promoter mutations in cancer: An emerging molecular biomarker? Virchows Arch. Int. J. Pathol. 2014, 465, 119–133. [Google Scholar] [CrossRef]
- Zvereva, M.; Pisarev, E.; Hosen, I.; Kisil, O.; Matskeplishvili, S.; Kubareva, E.; Kamalov, D.; Tivtikyan, A.; Manel, A.; Vian, E.; et al. Activating Telomerase TERT Promoter Mutations and Their Application for the Detection of Bladder Cancer. Int. J. Mol. Sci. 2020, 21, 6034. [Google Scholar] [CrossRef]
- Kinde, I.; Munari, E.; Faraj, S.F.; Hruban, R.H.; Schoenberg, M.; Bivalacqua, T.; Allaf, M.; Springer, S.; Wang, Y.; Diaz, L.A., Jr.; et al. TERT Promoter Mutations Occur Early in Urothelial Neoplasia and Are Biomarkers of Early Disease and Disease Recurrence in Urine. Cancer Res. 2013, 73, 7162–7167. [Google Scholar] [CrossRef] [PubMed]
- Dahmcke, C.M.; Steven, K.E.; Larsen, L.K.; Poulsen, A.L.; Abdul-Al, A.; Dahl, C.; Guldberg, P. A Prospective Blinded Evaluation of Urine-DNA Testing for Detection of Urothelial Bladder Carcinoma in Patients with Gross Hematuria. Eur. Urol. 2016, 70, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Ward, D.G.; Baxter, L.; Gordon, N.S.; Ott, S.; Savage, R.S.; Beggs, A.; James, J.D.; Lickiss, J.; Green, S.; Wallis, Y.; et al. Multiplex PCR and Next Generation Sequencing for the Non-Invasive Detection of Bladder Cancer. PLoS ONE 2016, 11, e0149756. [Google Scholar] [CrossRef] [PubMed]
- Critelli, R.; Fasanelli, F.; Oderda, M.; Polidoro, S.; Assumma, M.B.; Viberti, C.; Preto, M.; Gontero, P.; Cucchiarale, G.; Lurkin, I.; et al. Detection of multiple mutations in urinary exfoliated cells from male bladder cancer patients at diagnosis and during follow-up. Oncotarget 2016, 7, 67435–67448. [Google Scholar] [CrossRef] [PubMed]
- Springer, S.U.; Chen, C.H.; Pena, M.D.C.R.; Li, L.; Douville, C.; Wang, Y.; Cohen, J.D.; Taheri, D.; Silliman, N.; Schaefer, J.; et al. Non-invasive detection of urothelial cancer through the analysis of driver gene mutations and aneuploidy. Elife 2018, 7, e32143. [Google Scholar] [CrossRef]
- Shahesmaeili, A.; Afshar, R.M.; Sadeghi, A.; Bazrafshan, A. Cancer Incidence in Kerman Province, Southeast of Iran: Report of an ongoing Population-Based Cancer Registry, 2014. Asian Pac. J. Cancer Prev. 2018, 19, 1533–1541. [Google Scholar] [CrossRef]
- Sanli, O.; Dobruch, J.; Knowles, M.A.; Burger, M.; Alemozaffar, M.; Nielsen, M.E.; Lotan, Y. Bladder cancer. Nat. Rev. Dis. Prim. 2017, 3, 17022. [Google Scholar] [CrossRef]
- Rachakonda, P.S.; Hosen, I.; de Verdier, P.J.; Fallah, M.; Heidenreich, B.; Ryk, C.; Wiklund, N.P.; Steineck, G.; Schadendorf, D.; Hemminki, K.; et al. TERT promoter mutations in bladder cancer affect patient survival and disease recurrence through modification by a common polymorphism. Proc. Natl. Acad. Sci. USA 2013, 110, 17426–17431. [Google Scholar] [CrossRef]
- Borah, S.; Xi, L.; Zaug, A.J.; Powell, N.M.; Dancik, G.M.; Cohen, S.B.; Costello, J.C.; Theodorescu, D.; Cech, T.R. TERTpromoter mutations and telomerase reactivation in urothelial cancer. Science 2015, 347, 1006–1010. [Google Scholar] [CrossRef]
- Huang, D.-S.; Wang, Z.; He, X.-J.; Diplas, B.H.; Yang, R.; Killela, P.J.; Meng, Q.; Ye, Z.-Y.; Wang, W.; Jiang, X.-T.; et al. Recurrent TERT promoter mutations identified in a large-scale study of multiple tumour types are associated with increased TERT expression and telomerase activation. Eur. J. Cancer 2015, 51, 969–976. [Google Scholar] [CrossRef]
- Avogbe, P.H.; Manel, A.; Vian, E.; Durand, G.; Forey, N.; Voegele, C.; Zvereva, M.; Hosen, I.; Meziani, S.; De Tilly, B.; et al. Urinary TERT promoter mutations as non-invasive biomarkers for the comprehensive detection of urothelial cancer. eBioMedicine 2019, 44, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Hosen, I.; Sheikh, M.; Zvereva, M.; Scelo, G.; Forey, N.; Durand, G.; Voegele, C.; Poustchi, H.; Khoshnia, M.; Roshandel, G.; et al. Urinary TERT promoter mutations are detectable up to 10 years prior to clinical diagnosis of bladder cancer: Evidence from the Golestan Cohort Study. eBioMedicine 2020, 53, 102643. [Google Scholar] [CrossRef]
- Ward, D.G.; Gordon, N.S.; Boucher, R.H.; Pirrie, S.J.; Baxter, L.; Ott, S.; Silcock, L.; Whalley, C.M.; Stockton, J.D.; Beggs, A.D.; et al. Targeted deep sequencing of urothelial bladder cancers and associated urinary DNA: A 23-gene panel with utility for non-invasive diagnosis and risk stratification. Br. J. Urol. 2019, 124, 532–544. [Google Scholar] [CrossRef] [PubMed]
- Stasik, S.; Salomo, K.; Heberling, U.; Froehner, M.; Sommer, U.; Baretton, G.B.; Ehninger, G.; Wirth, M.P.; Thiede, C.; Fuessel, S. Evaluation of TERT promoter mutations in urinary cell-free DNA and sediment DNA for detection of bladder cancer. Clin. Biochem. 2018, 64, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Descotes, F.; Kara, N.; Decaussin-Petrucci, M.; Piaton, E.; Geiguer, F.; Rodriguez-Lafrasse, C.; Terrier, J.E.; Lopez, J.; Ruffion, A. Non-invasive prediction of recurrence in bladder cancer by detecting somatic TERT promoter mutations in urine. Br. J. Cancer 2017, 117, 583–587. [Google Scholar] [CrossRef]
- Safiri, S.; Kolahi, A.-A.; Naghavi, M. Global, regional and national burden of bladder cancer and its attributable risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease study 2019. BMJ Glob. Health 2021, 6, e004128. [Google Scholar] [CrossRef]
- Zhu, C.-Z.; Ting, H.-N.; Ng, K.-H.; Ong, T.-A. A review on the accuracy of bladder cancer detection methods. J. Cancer 2019, 10, 4038–4044. [Google Scholar] [CrossRef]
- Lotan, Y.; Roehrborn, C.G. Sensitivity and specificity of commonly available bladder tumor markers versus cytology: Results of a comprehensive literature review and meta-analyses. Urology 2003, 61, 109–118. [Google Scholar] [CrossRef]
- Barkan, G.A.; Wojcik, E.M.; Nayar, R.; Savic-Prince, S.; Quek, M.L.; Kurtycz, D.F.; Rosenthal, D.L. The Paris System for Reporting Urinary Cytology: The Quest to Develop a Standardized Terminology. Adv. Anat. Pathol. 2016, 23, 193–201. [Google Scholar] [CrossRef]
- Available online: https://ethics.research.ac.ir/ProposalCertificateEn.php?id=81331&Print=true&NoPrintHeader=true&NoPrintFooter=true&NoPrintPageBorder=true&LetterPrint=true (accessed on 8 November 2022).
- Hadji, M.; Rashidian, H.; Marzban, M.; Gholipour, M.; Naghibzadeh-Tahami, A.; Mohebbi, E.; Ebrahimi, E.; Hosseini, B.; Haghdoost, A.A.; Rezaianzadeh, A.; et al. The Iranian Study of Opium and Cancer (IROPICAN): Rationale, Design, and Initial Findings. Arch. Iran. Med. 2021, 24, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Hosen, I.; Forey, N.; Durand, G.; Voegele, C.; Bilici, S.; Avogbe, P.H.; Delhomme, T.M.; Foll, M.; Manel, A.; Vian, E.; et al. Development of Sensitive Droplet Digital PCR Assays for Detecting Urinary TERT Promoter Mutations as Non-Invasive Biomarkers for Detection of Urothelial Cancer. Cancers 2020, 12, 3541. [Google Scholar] [CrossRef] [PubMed]
- Zieliński, W. The Shortest Clopper–Pearson Confidence Interval for Binomial Probability. Commun. Stat. Simul. Comput. 2009, 39, 188–193. [Google Scholar] [CrossRef]
- Mercaldo, N.D.; Lau, K.F.; Zhou, X.H. Confidence intervals for predictive values with an emphasis to case–control studies. Stat. Med. 2007, 26, 2170–2183. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, N.; Rinaldetti, S.; Cheikh, B.B.; Zhou, Q.; Hass, E.P.; Jones, R.T.; Joshi, M.; LaBarbera, D.V.; Knott, S.R.V.; Cech, T.R.; et al. TRIM28 is a transcriptional activator of the mutant TERT promoter in human bladder cancer. Proc. Natl. Acad. Sci. USA 2021, 118, 5235–5242. [Google Scholar] [CrossRef]
| Characteristic | Case Group (Total n = 31) N (%) | Control Group (Total n = 50) N (%) | p-Value |
|---|---|---|---|
| Age (years) 1 | 64.9 ± 9.4 | 59.6 ± 13.4 | 0.058 |
| Sex | |||
| Male | 26/31 (83.8%) | 34/50 (74%) | p = 0.11 |
| Female | 5/31 (16.1%) | 16/50 (26%) | |
| Smoking | 26/31 (83%) | 8/50 (16%) | p < 0.001 |
| Opium use | 24/31 (77%) | 7/50 (14%) | p < 0.001 |
| Hematuria | |||
| Gross Microscopic | 19/31 (61%) 9/31 (29%) | - - | - |
| Tumor stage | |||
| MIBC 2 | 7/31 (22%) | - | - |
| NMIBC 3 | 24/31 (77%) | - | |
| Tumor grade | |||
| Low-grade | 16/31 (52%) | - | - |
| High-grade | 15/31 (48%) | - |
| Characteristics | All BC Cases (n = 31) | Primary BC (n = 11) | Recurrent BC (n = 20) |
|---|---|---|---|
| True positive (n) | 21 | 11 | 10 |
| True negative (n) | 44 | 44 | 44 |
| False positive (n) | 6 | 6 | 6 |
| False negative (n) | 10 | 0 | 10 |
| Sensitivity (95% CI) (%) | 67.7 (48.6–83.3) | 100.0 (71.5–100.0) | 50.0 (27.2–72.8) |
| Specificity (95% CI) (%) | 88.0 (75.6–95.4) | 88.0 (75.6–95.4) | 88.0 (75.6–95.4) |
| Positive likelihood ratio (95% CI) | 5.6 (2.5–12.4) | 8.3 (3.9–17.6) | 4.1 (1.7–9.9) |
| Negative likelihood ratio (95% CI) | 0.3 (0.2–0.6) | 0.0 | 0.5 (0.3–0.8) |
| Positive predictive value (95% CI) (%) * | 70.7 (52.3–84.1) | 78.1 (62.7–88.3) | 64.1 (42.8–80.9) |
| Negative predictive value (95% CI) (%)* | 86.4 (79.0–91.4) | 100.0 | 80.4 (72.3–86.5) |
| Accuracy (95% CI) (%) * | 81.9 (71.8–89.6) | 91.6 (81.6–97.1) | 76.6 (64.9–85.9) |
| Subgroups | uTERTpm | Urine Cytology | Combined uTERTpm/Urine Cytology | |||
|---|---|---|---|---|---|---|
| Sensitivity % (95% CI) | Specificity % (95% CI) | Sensitivity % (95% CI) | Specificity% (95% CI) | Sensitivity % (95% CI) | Specificity% (95% CI) | |
| All patients | 67.7 (48.6–83.3) | 88.0 (75.6–95.4) | 67.7 (48.6–83.3) | 62.0 (47.1–75.3) | 83.8 (66.2–94.5) | 52.0 (37.4–66.3) |
| Tumor stage | ||||||
| NMIBC (n = 24) | 66.7 (44.7–84.4) | - | 58.3 (36.7–77.9) | - | 79.2 (57.9–92.9) | - |
| MIBC (n = 7) | 71.4 (29.0–96.3) | - | 100.0 (59.0–100.0) | - | 100.0 (59.0–100.0) | - |
| Grade | ||||||
| Low-grade (n = 16) | 62.5 (35.4–84.8) | - | 56.2 (29.9–80.3) | - | 75.0 (47.6–92.7) | - |
| High-grade (n = 15) | 73.3 (44.9–92.2) | - | 80.0 (51.9–95.7) | - | 93.3 (68.1–99.9) | - |
| Gross Hematuria | ||||||
| Yes (n = 19) | 73.6 (48.8–90.8) | - | 57.8 (33.5–79.6) | - | 84.2 (60.4–96.6) | - |
| No (n = 12) | 58.3 (27.6–84.8) | - | 83.3 (51.5–97.9) | - | 83.3 (51.5–97.9) | - |
| Smoking | ||||||
| Yes (n = 25) | 69.2 (48.2–85.6) | 77.7 (39.9–97.1) | 76.9 (56.3–91.0) | 66.6 (29.9–92.5) | 88.4 (69.8–97.5) | 44.4 (13.7–78.8) |
| No (n = 6) | 60.0 (14.6–94.7) | 90.2 (76.8–97.2) | 20.0 (0.5–71.6) | 60.9 (44.5–75.8) | 60.0 (14.6–94.7) | 53.0 (37.4–69.3) |
| Opium use | ||||||
| Yes (n = 23) | 75.0 (53.2–90.2) | 85.7 (42.1–99.6) | 70.8 (48.9–87.3) | 57.1 (18.4–90.1) | 87.5 (67.6–97.3) | 42.8 (9.9–81.5) |
| No (n = 8) | 42.8 (9.9–81.5) | 88.3 (74.9–96.1) | 57.1 (18.4–90.1) | 62.7 (46.7–77.0) | 71.4 (29.0–96.3) | 53.4 (37.6–68.8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pakmanesh, H.; Anvari, O.; Forey, N.; Weiderpass, E.; Malekpourafshar, R.; Iranpour, M.; Shahesmaeili, A.; Ahmadi, N.; Bazrafshan, A.; Zendehdel, K.; et al. TERT Promoter Mutations as Simple and Non-Invasive Urinary Biomarkers for the Detection of Urothelial Bladder Cancer in a High-Risk Region. Int. J. Mol. Sci. 2022, 23, 14319. https://doi.org/10.3390/ijms232214319
Pakmanesh H, Anvari O, Forey N, Weiderpass E, Malekpourafshar R, Iranpour M, Shahesmaeili A, Ahmadi N, Bazrafshan A, Zendehdel K, et al. TERT Promoter Mutations as Simple and Non-Invasive Urinary Biomarkers for the Detection of Urothelial Bladder Cancer in a High-Risk Region. International Journal of Molecular Sciences. 2022; 23(22):14319. https://doi.org/10.3390/ijms232214319
Chicago/Turabian StylePakmanesh, Hamid, Omid Anvari, Nathalie Forey, Elisabete Weiderpass, Reza Malekpourafshar, Maryam Iranpour, Armita Shahesmaeili, Nahid Ahmadi, Azam Bazrafshan, Kazem Zendehdel, and et al. 2022. "TERT Promoter Mutations as Simple and Non-Invasive Urinary Biomarkers for the Detection of Urothelial Bladder Cancer in a High-Risk Region" International Journal of Molecular Sciences 23, no. 22: 14319. https://doi.org/10.3390/ijms232214319
APA StylePakmanesh, H., Anvari, O., Forey, N., Weiderpass, E., Malekpourafshar, R., Iranpour, M., Shahesmaeili, A., Ahmadi, N., Bazrafshan, A., Zendehdel, K., Kannengiesser, C., Ba, I., McKay, J., Zvereva, M., Hosen, M. I., Sheikh, M., & Calvez-Kelm, F. L. (2022). TERT Promoter Mutations as Simple and Non-Invasive Urinary Biomarkers for the Detection of Urothelial Bladder Cancer in a High-Risk Region. International Journal of Molecular Sciences, 23(22), 14319. https://doi.org/10.3390/ijms232214319






