Effect of Shading on the Morphological, Physiological, and Biochemical Characteristics as Well as the Transcriptome of Matcha Green Tea
Abstract
1. Introduction
2. Results
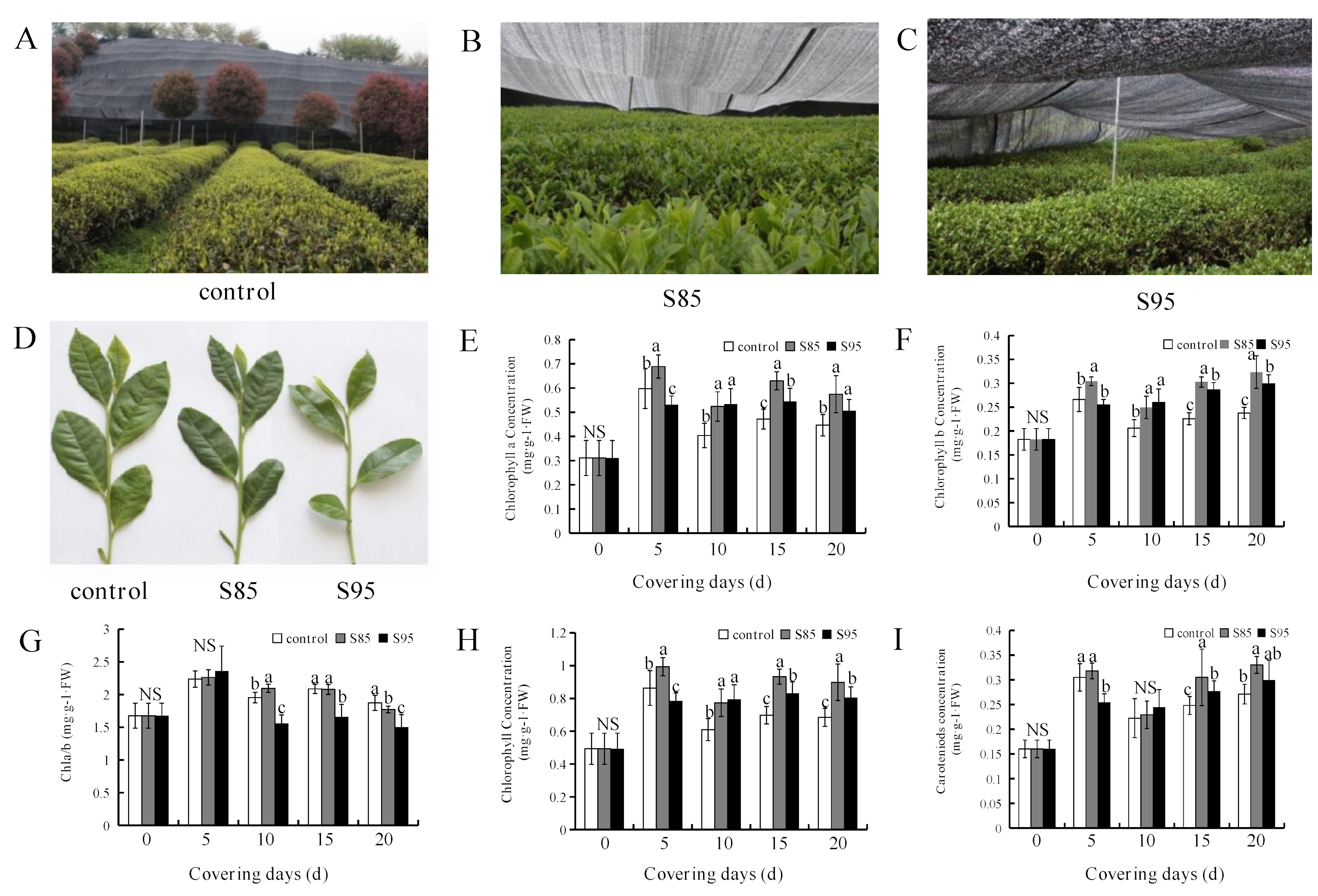
2.1. Effect of Shading on Chlorophyll and Carotenoid Contents
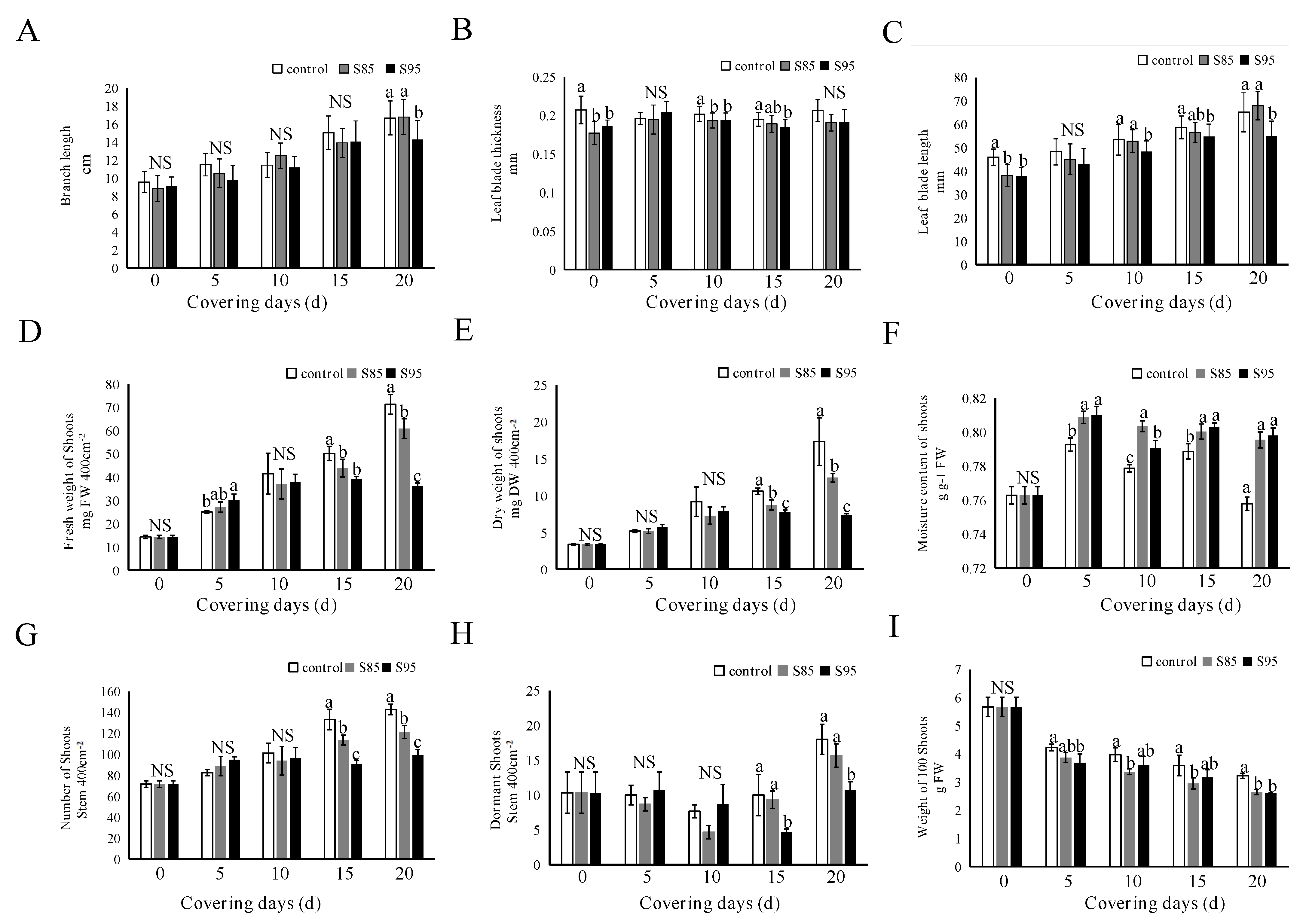
2.2. Influence of Shading on Tea Morphology and Growth Parameters
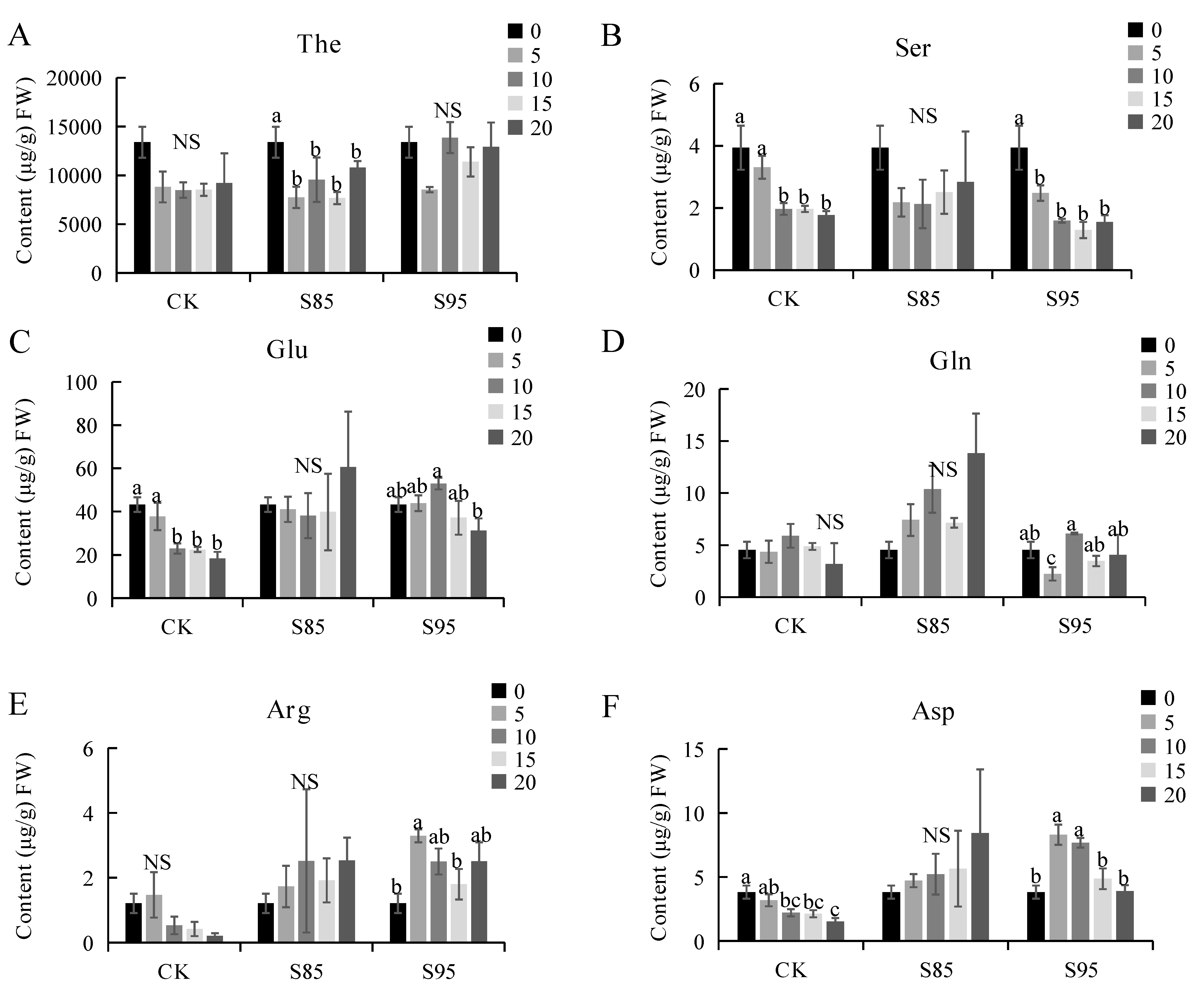
2.3. Effect of Shading on the Amino Acid Content
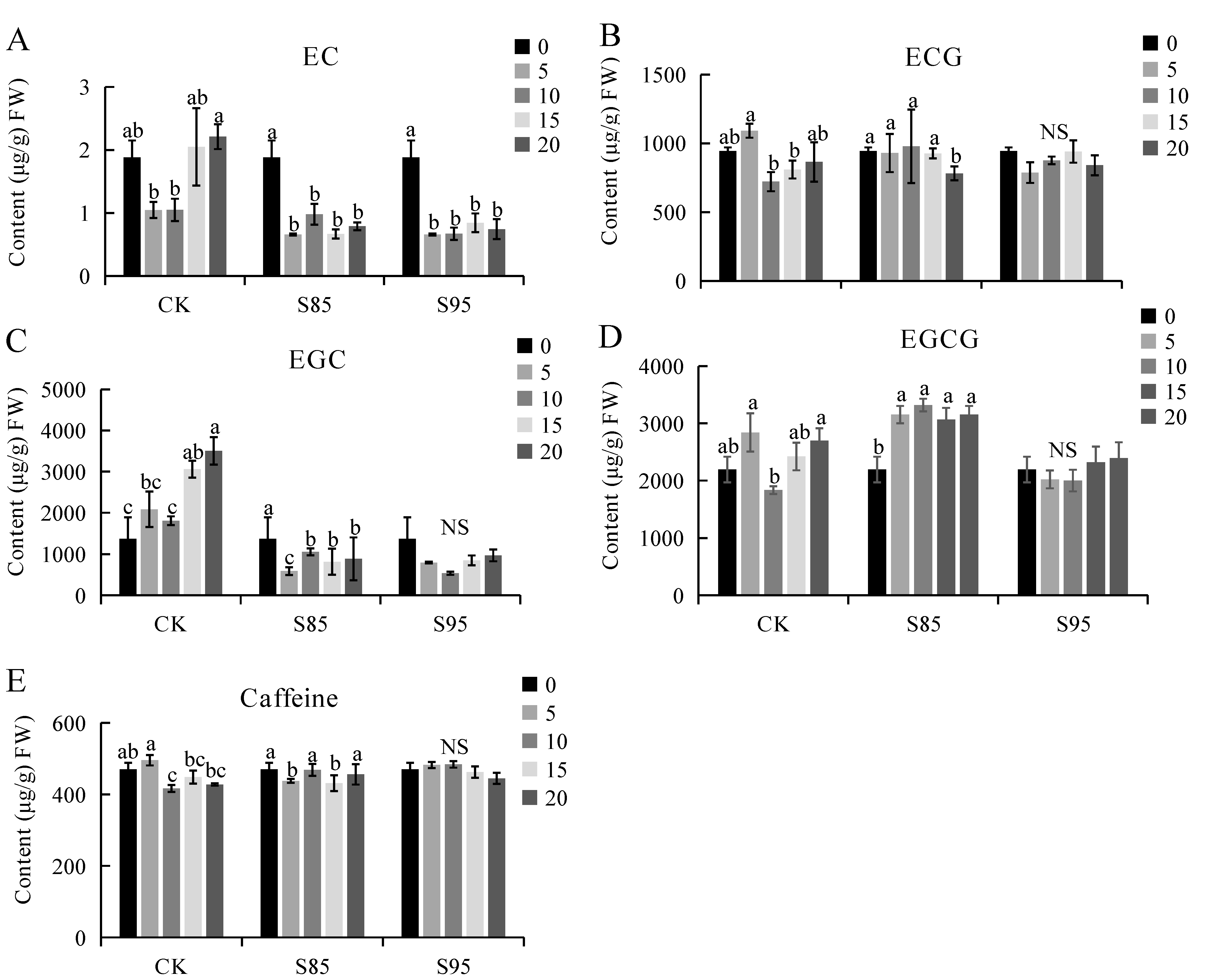
2.4. Effect of Shading on Catechin and Caffeine Contents
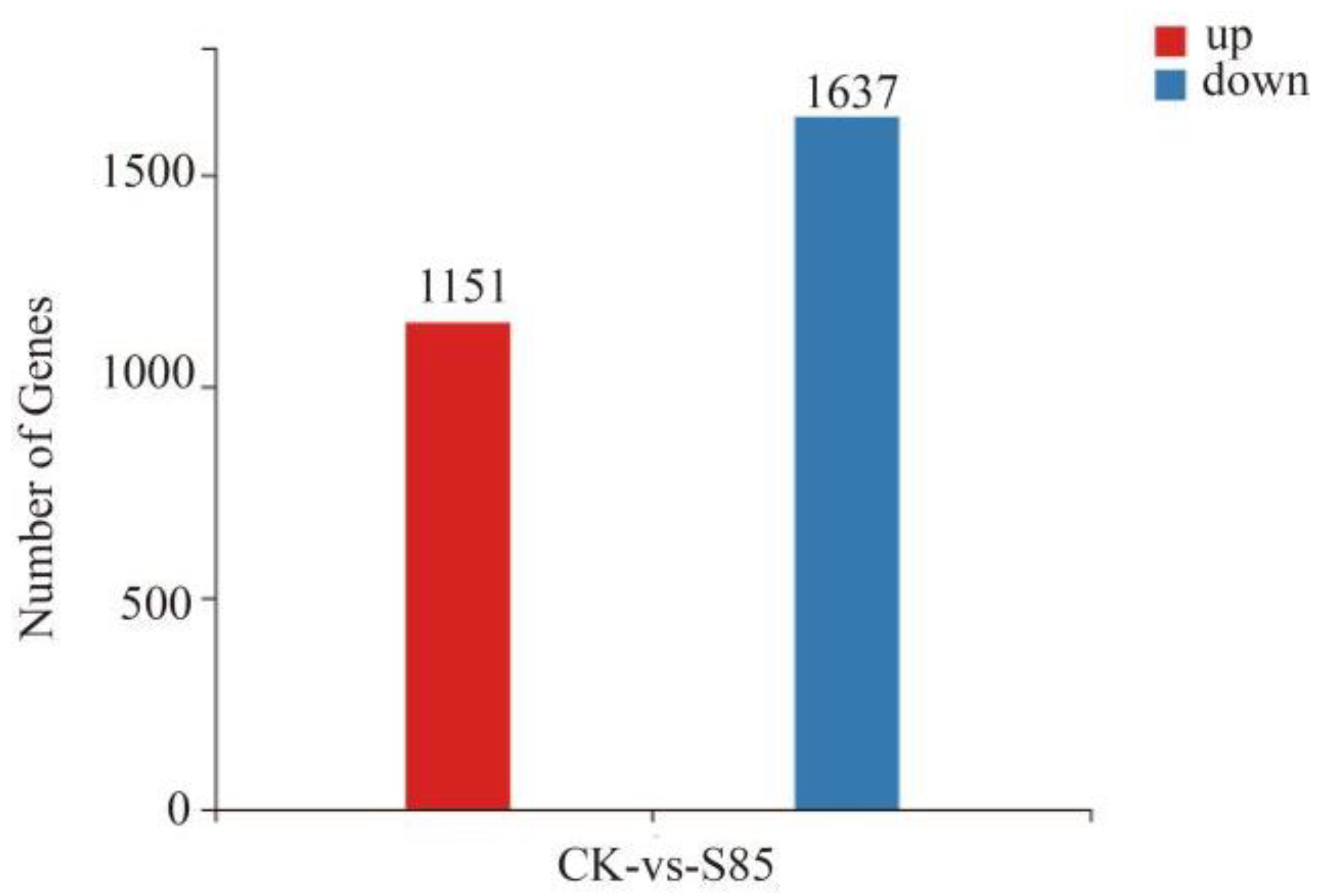
2.5. Transcriptome Sequencing, Unigene Assembly, and Annotation
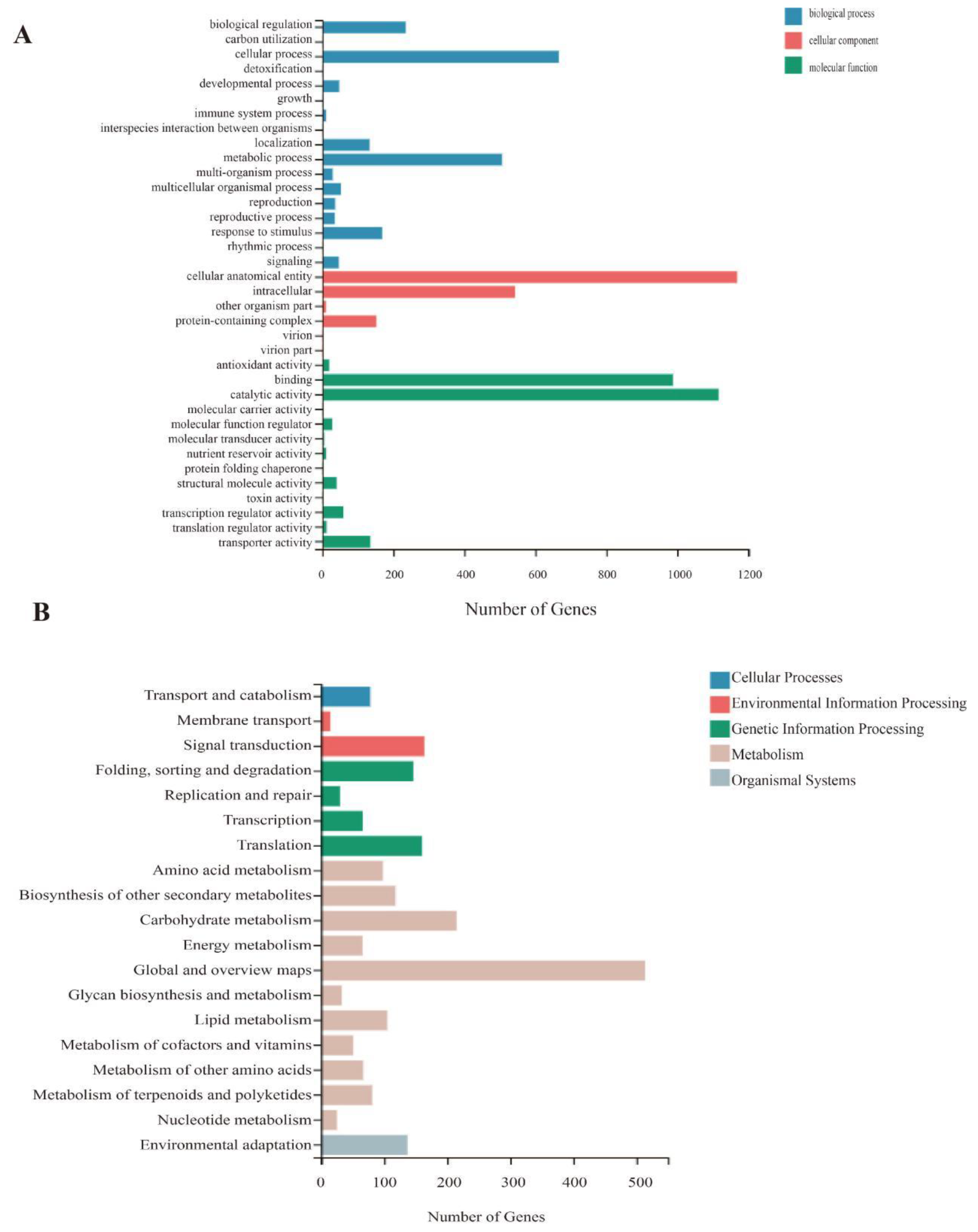
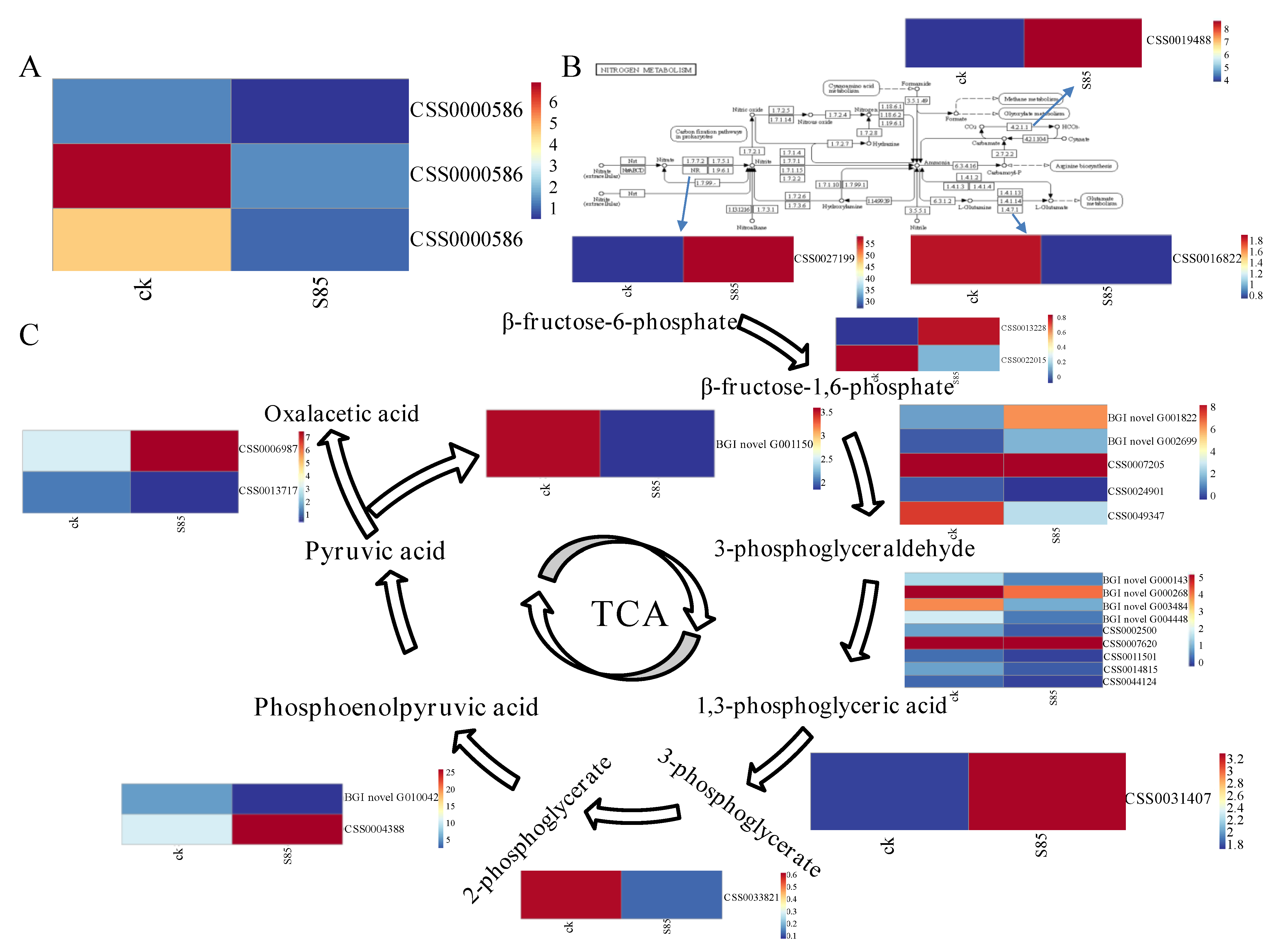
2.6. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analyses of the Differentially Expressed Genes
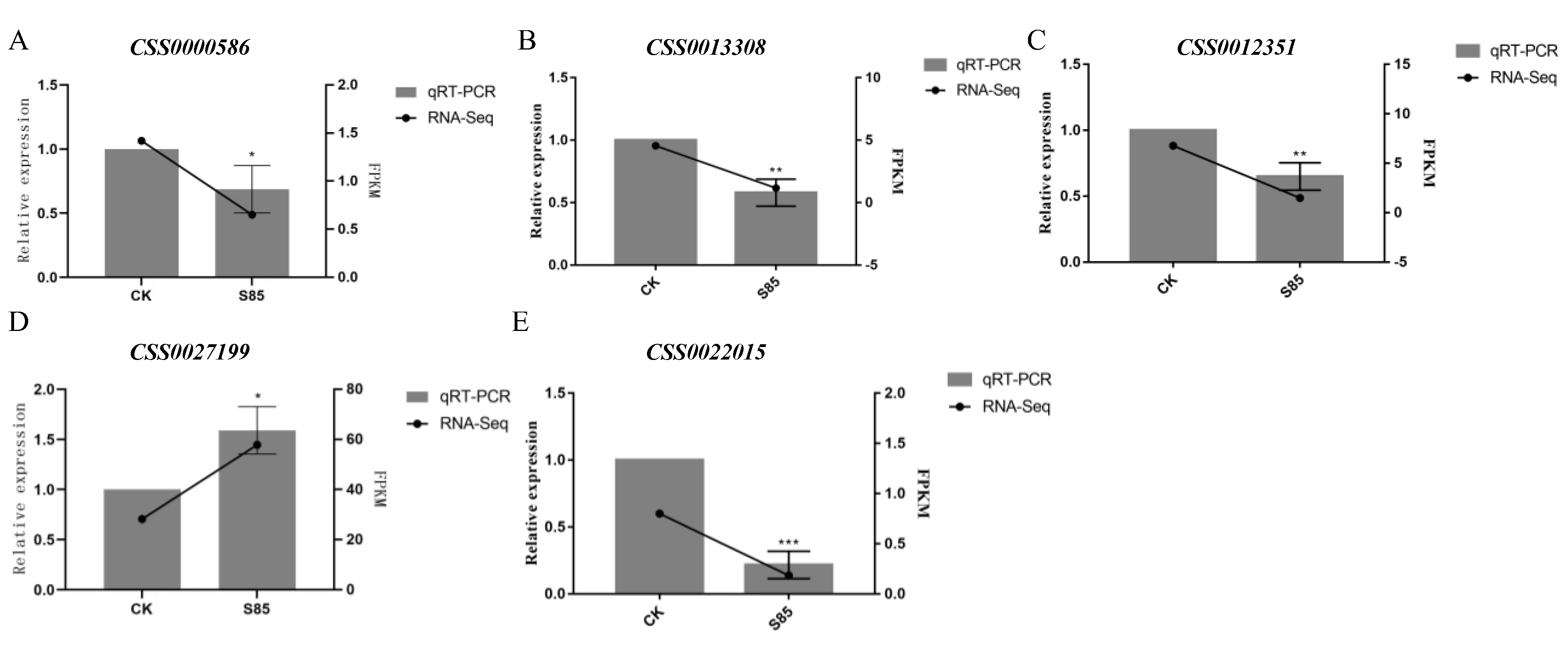
2.7. Differential Gene Expression Levels Were Verified by Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Treatments
4.2. Analysis of Physiological and Biochemical Indicators
4.2.1. Examination of Tea Morphology
4.2.2. Determination of Free Amino Acid, Catechin, and Caffeine Contents
4.2.3. Determination of the Chlorophyll Content
4.2.4. RNA Extraction, cDNA Library Construction, and Sequencing
4.2.5. Unigene Assembly and Annotation
4.2.6. Gene Expression Analysis and Detection of Differentially Expressed Genes
4.2.7. Gene Ontology and Kyoto Encyclopedia of Genes and Genomes Enrichment Analyses
4.2.8. Quantitative Real-Time Polymerase Chain Reaction Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Namita, P.; Mukesh, R.; Vijay, K.J. Camellia sinensis (green tea): A review. Glob. J. Pharmacol. 2012, 6, 52–59. [Google Scholar]
- Fujiki, H.; Sueoka, E.; Watanabe, T.; Suganuma, M. Synergistic enhancement of anticancer effects on numerous human cancer cell lines treated with the combination of EGCG, other green tea catechins, and anticancer compounds. J. Cancer Res. Clin. Oncol. 2015, 141, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Kochman, J.; Jakubczyk, K.; Antoniewicz, J.; Mruk, H.; Janda, K. Health Benefits and Chemical Composition of Matcha Green Tea: A Review. Molecules 2020, 26, 85. [Google Scholar] [CrossRef] [PubMed]
- Ekborg-Ott, K.H.; Taylor, A.; Armstrong, D.W. Varietal differences in the total and enantiomeric composition of theanine in tea. J. Agric. Food Chem. 1997, 45, 353–363. [Google Scholar] [CrossRef]
- McDowell, I.; Owuor, P. Taste of tea. New Sci. 1992, 133, 30–33. [Google Scholar]
- Yang, Z.; Baldermann, S.; Watanabe, N. Recent studies of the volatile compounds in tea. Food Res. Int. 2013, 53, 585–599. [Google Scholar] [CrossRef]
- Yu, Z.; Yang, Z. Understanding different regulatory mechanisms of proteinaceous and non-proteinaceous amino acid formation in tea (Camellia sinensis) provides new insights into the safe and effective alteration of tea flavor and function. Crit. Rev. Food Sci. 2020, 60, 844–858. [Google Scholar] [CrossRef]
- Zeng, L.; Watanabe, N.; Yang, Z. Understanding the biosyntheses and stress response mechanisms of aroma compounds in tea (Camellia sinensis) to safely and effectively improve tea aroma. Crit. Rev. Food Sci. 2019, 59, 2321–2334. [Google Scholar] [CrossRef]
- Sano, T.; Horie, H.; Hirono, Y. Effect of shading intensity on morphological and color traits and on chemical components of new tea (Camellia sinensis L.) shoots under direct covering cultivation. J. Sci. Food Agric. 2018, 98, 5666–5676. [Google Scholar]
- Kobayashi, E.; Nakamura, Y.; Suzuki, T.; Oishi, T.; Inaba, K. Influence of light intensities on the color and ingredients of new shoots in tea plants. Res. J. 2011, 111, 39–49. [Google Scholar]
- Matsunaga, A.; Sano, T.; Hirono, Y.; Horie, H. Effect of various directly covered shading levels on chemical components in the new shoots of the first flush. Tea Res. J. 2016, 122, 1–7, (In Japanese with English abstract). [Google Scholar] [CrossRef]
- Mukai, T.; Horie, H.; Goto, T. Differences in free amino acids and total nitrogen contents among various prices of green tea. Tea Res. J. 1992, 76, 45–50. [Google Scholar] [CrossRef]
- Wang, L.; Feng, Z.; Wang, X.; Zhang, X. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Li, F.; Yang, T.; Feng, L.; Zhang, S.; Li, F.; Li, W.; Xu, G.; Bao, S.; Wan, X.; et al. Theanine transporters identified in tea plants (Camellia sinensis L.). Plant J. 2020, 101, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Wu, W.; Liu, X.; Wang, Y.; Xu, P. Transcription factor CsWRKY40 regulates L-theanine hydrolysis by activating the CsPDX2.1 promoter in tea leaves during withering. Hortic. Res. 2022, 9, uhac025. [Google Scholar]
- Ashihara, H. Occurrence, biosynthesis and metabolism of L-theanine (gamma-glutamyl-L-ethylamide) in plants: A comprehensive review. Nat. Prod. Commun. 2015, 10, 803–810. [Google Scholar]
- Yang, T.; Xie, Y.; Lu, X.; Yan, X.; Wang, Y.; Ma, J.; Cheng, X.; Lin, S.; Bao, S.; Wan, X.; et al. Shading Promoted Theanine Biosynthesis in the Roots and Allocation in the Shoots of the Tea Plant (Camellia sinensis L.) Cultivar Shuchazao. J. Agric. Food Chem. 2021, 69, 4795–4803. [Google Scholar] [CrossRef]
- Carr, M.; Stephens, W. Climate, weather and the yield of tea. In Tea—Cultivation to Consumption; Wilson, K.C., Clifford, M.N., Eds.; Chapman and Hall: London, UK, 1992; pp. 87–135. [Google Scholar]
- Lee, L.S.; Choi, J.H.; Son, N.; Kim, S.H.; Park, J.D.; Jang, D.J.; Jeong, Y.; Kim, H.J. Metabolomic analysis of the effect of shade treatment on the nutritional and sensory qualities of green tea. J. Agric. Food Chem. 2013, 61, 332–338. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, L.; Shan, Y.; Liu, Y.; Tian, Y.; Xia, T. Influence of shade on flavonoid biosynthesis in tea (Camellia sinensis (L.) O. Kuntze). Sci. Hortic. 2012, 141, 7–16. [Google Scholar] [CrossRef]
- Ku, K.M.; Choi, J.N.; Kim, J.; Kim, J.K.; Yoo, L.G.; Lee, S.J.; Hong, Y.-S.; Lee, C.H. Metabolomics analysis reveals the compositional differences of shade grown tea (Camellia sinensis L.). J. Agric. Food Chem. 2010, 58, 418–426. [Google Scholar] [CrossRef]
- Bhagat, A.R.; Delgado, A.M.; Issaoui, M.; Chammem, N.; Fiorino, M.; Pellerito, A.; Natalello, S. Review of the Role of Fluid Dairy in Delivery of Polyphenolic Compounds in the Diet: Chocolate Milk, Coffee Beverages, Matcha Green Tea, and Beyond. J. AOAC Int. 2019, 102, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.L.; Peng, J.Y.; Liu, F.; He, Y. Fast Identification of Matcha and Green Tea Powder with Laser-Induced Breakdown Spectroscopy. Spectrosc. Spect. Anal. 2017, 37, 1908–1911. [Google Scholar]
- Liu, L.; Lin, N.; Liu, X.; Yang, S.; Wang, W.; Wan, X. From Chloroplast biogenesis to chlorophyll accumulation: The interplay of light and hormones on gene expression in Camellia sinensis cv. Shuchazao Leaves. Front. Plant Sci. 2020, 11, 256. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, H.; Tanaka, Y.; Umetsu, K.; Morita, S.; Ono, Y.; Suzuki, T.; Takemoto, T.; Morita, A.; Ikka, T. Phenotypic Markers Reflecting the Status of Overstressed Tea Plants Subjected to Repeated Shade Cultivation. Front. Plant Sci. 2020, 11, 556476. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Zhao, H.; Guo, F.; Shi, X.; Ye, C.; Yang, P.; Liu, B.; Ni, D. Transcriptomic analysis reveals mechanism of light-sensitive albinism in tea plant Camellia sinensis ‘Huangjinju’. BMC Plant Biol. 2020, 20, 216. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Su, H.; Jin, R.; Mao, Y.; Xu, A.; Cheng, H.; Wang, Y.; Meng, Q. Shading Effects on Leaf Color Conversion and Biosynthesis of the Major Secondary Metabolites in the Albino Tea Cultivar “Yujinxiang”. J. Agric. Food Chem. 2020, 68, 2528–2538. [Google Scholar] [CrossRef]
- Saijo, R. Effect of shade treatment on biosynthesis of catechins in tea plant. Plant Cell Physiol. 1980, 21, 989–998. [Google Scholar]
- Yu, Z.; Liao, Y.; Zeng, L.; Dong, F.; Watanabe, N.; Yang, Z. Transformation of catechins into theaflavins by upregulation of CsPPO3 in preharvest tea (Camellia sinensis) leaves exposed to shading treatment. Food Res. Int. 2020, 129, 108842. [Google Scholar] [CrossRef]
- Sasaoka, K.; Kito, M.; Inagaki, H. Studies on the Biosynthesis of Theanine in Tea Seedlings Synthesis of Theanine by the Homogenate of Tea Seedlings. Agric. Biol. Chem. 1963, 27, 467–468. [Google Scholar] [CrossRef]
- Kito, M.; Kokura, H.; Izaki, J.; Sasaoka, K. Fate of the radioactive carbon of theanine labeled with ethylamine-1-14C in tea seedlings. Agric. Biol. Chem. 1966, 30, 623–624. [Google Scholar] [CrossRef]
- Oh, K.; Kato, T.; Xu, H.L. Transport of nitrogen assimilation in xylem vessels of green tea plants fed with NH4-N and NO3-N. Pedosphere 2008, 18, 222–226. [Google Scholar] [CrossRef]
- Ruan, J.; Ma, L.; Yang, Y. Magnesium nutrition on accumulation and transport of amino acids in tea plants. J. Sci. Food Agric. 2012, 92, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef]
- Buchfink, B.; Xie, C.; Huson, D.H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 2015, 12, 59–60. [Google Scholar] [CrossRef]
- Sanseverino, W.; Roma, G. PRGdb: A bioinformatics platform for plant resistance gene analysis. Nucleic Acids Res. 2010, 38, 814–821. [Google Scholar] [CrossRef]
- Zhou, X.; Bao, S.; Liu, J.; Zhuang, Y. De novo sequencing and analysis of the transcriptome of the wild eggplant species Solanum aculeatissimum in response to Verticillium dahliae. Plant Mol. Biol. Rep. 2016, 34, 1193–1203. [Google Scholar] [CrossRef]
| Sample | Raw Reads | Clean Reads | Q20/% | Q30/% | Clean Reads Ratio/% | Total Map/% | Unique Map/% |
|---|---|---|---|---|---|---|---|
| Control | 45.28 × 106 | 42.01 × 106 | 95.98 | 93.38 | 92.78 | 74.04 | 36.88 |
| S85 | 46.27 × 106 | 42 × 106 | 95.86 | 90.27 | 90.82 | 73.68 | 36.61 |
| Primer Name | Primer Sequences (5′-3′) | Sequence Length |
|---|---|---|
| CSS0000586-F | AGCAGTGATTGGGTCAGGAG | 123 bp |
| CSS0000586-R | GCGAGGAGAGTAGAAGTGTAGT | |
| CSS0012351-F | CGCCTGAAGAGAACTGGAAC | 177 bp |
| CSS0012351-R | CTGTGAGAGGAGATTGGAAGTG | |
| CSS0013308-F | CGCCTGAAGAGAACTGGAACT | 180 bp |
| CSS0013308-R | GCACTGTGAGAGGAGATTGGAA | |
| CSS0027199-F | ACCCTTCACAAACACCACCTC | 167 bp |
| CSS0027199-R | CGGTTCCAGCGTTGATGAGA | |
| CSS0022015-F | GACCCAAGCCAAATACTTCGT | 112 bp |
| CSS0022015-R | GGCATAGCAATCCACCTTCTT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Ye, K.; Xu, Y.; Zhao, Y.; Zhao, D. Effect of Shading on the Morphological, Physiological, and Biochemical Characteristics as Well as the Transcriptome of Matcha Green Tea. Int. J. Mol. Sci. 2022, 23, 14169. https://doi.org/10.3390/ijms232214169
Chen X, Ye K, Xu Y, Zhao Y, Zhao D. Effect of Shading on the Morphological, Physiological, and Biochemical Characteristics as Well as the Transcriptome of Matcha Green Tea. International Journal of Molecular Sciences. 2022; 23(22):14169. https://doi.org/10.3390/ijms232214169
Chicago/Turabian StyleChen, Xi, Kun Ye, Yan Xu, Yichen Zhao, and Degang Zhao. 2022. "Effect of Shading on the Morphological, Physiological, and Biochemical Characteristics as Well as the Transcriptome of Matcha Green Tea" International Journal of Molecular Sciences 23, no. 22: 14169. https://doi.org/10.3390/ijms232214169
APA StyleChen, X., Ye, K., Xu, Y., Zhao, Y., & Zhao, D. (2022). Effect of Shading on the Morphological, Physiological, and Biochemical Characteristics as Well as the Transcriptome of Matcha Green Tea. International Journal of Molecular Sciences, 23(22), 14169. https://doi.org/10.3390/ijms232214169






