Effects of Adrenomedullin on Atrial Electrophysiology and Pulmonary Vein Arrhythmogenesis
Abstract
1. Introduction
2. Results
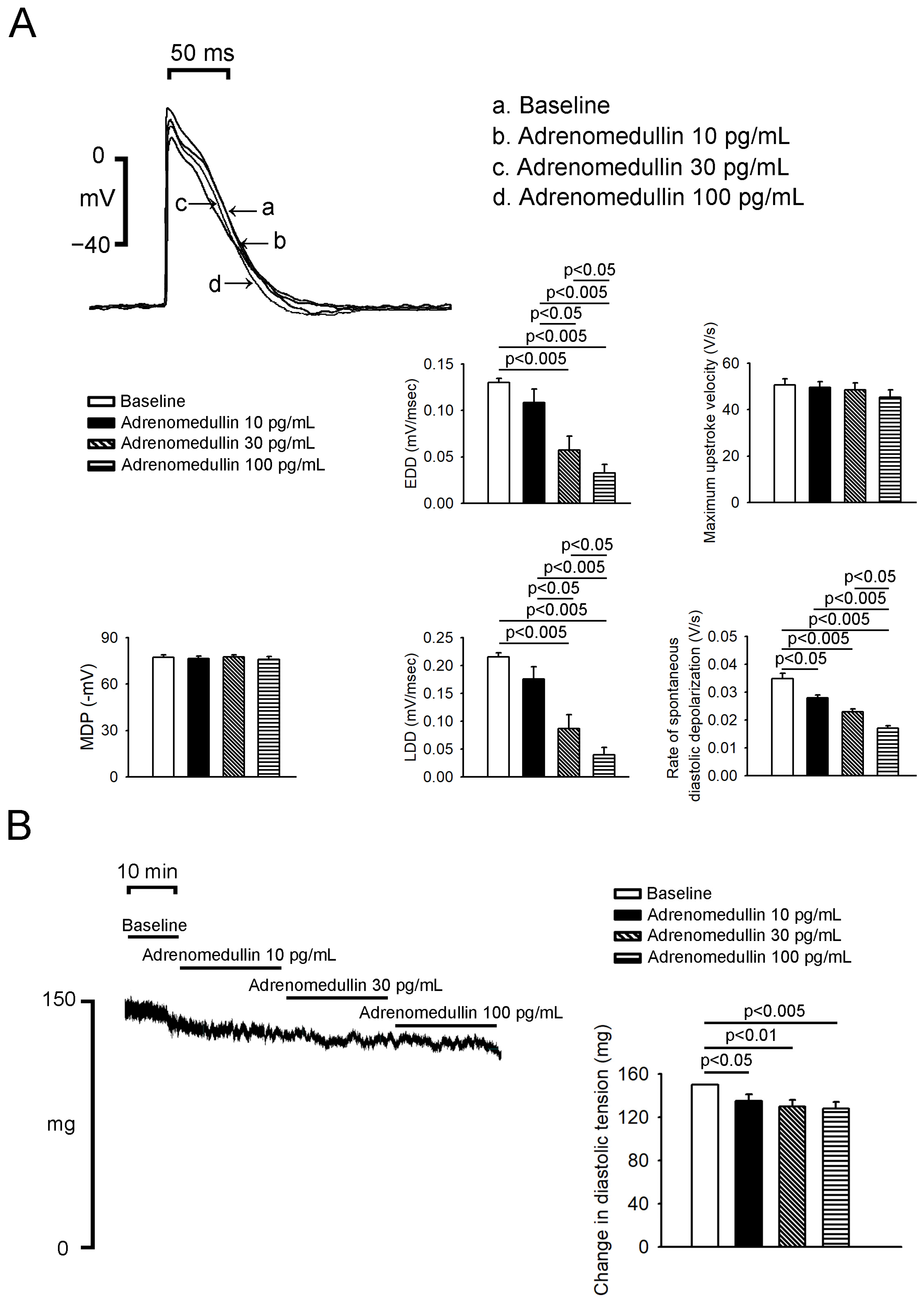
2.1. Effects of Adrenomedullin on PV and SAN (Sinoatrial Node) Electrical Activity
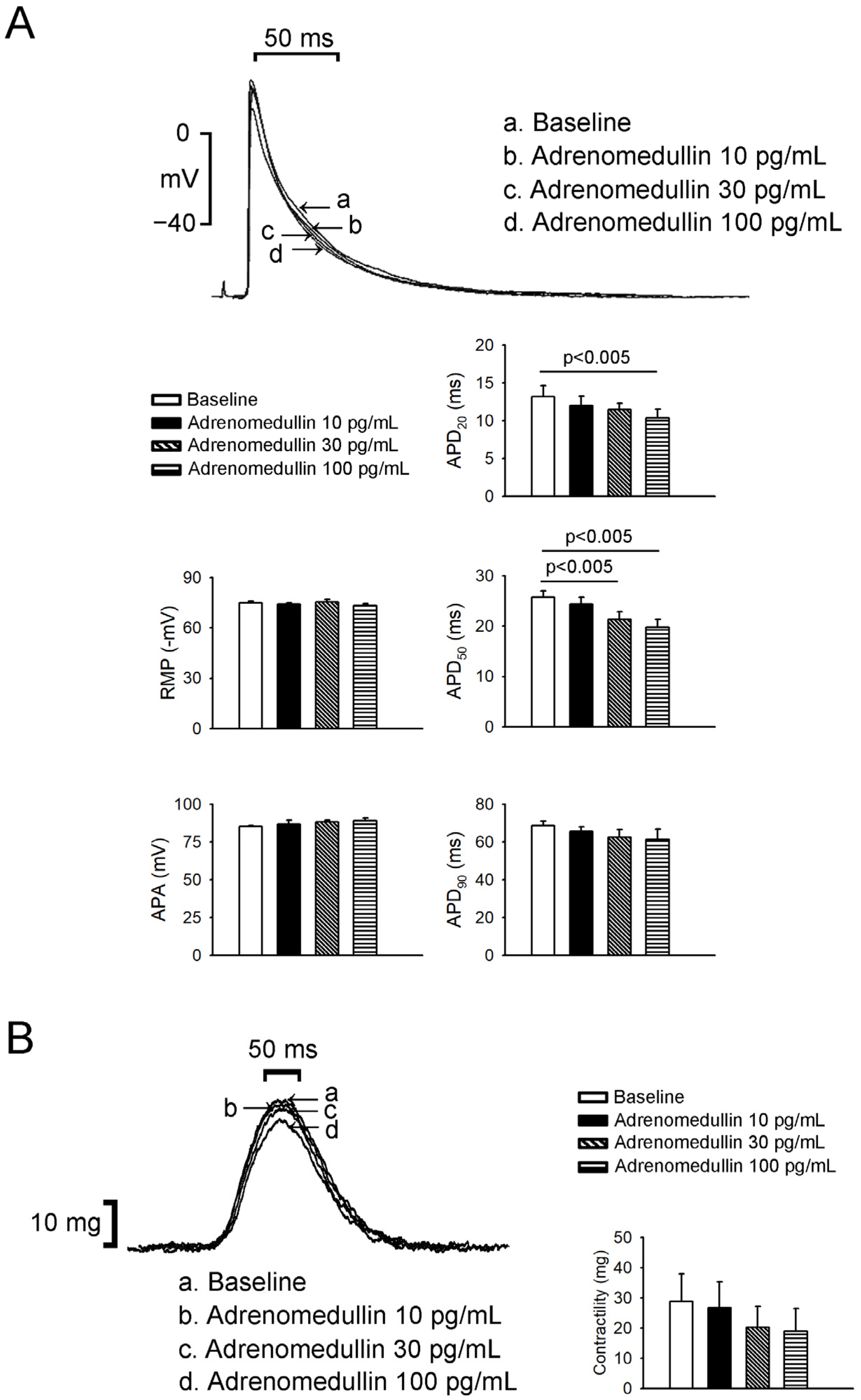
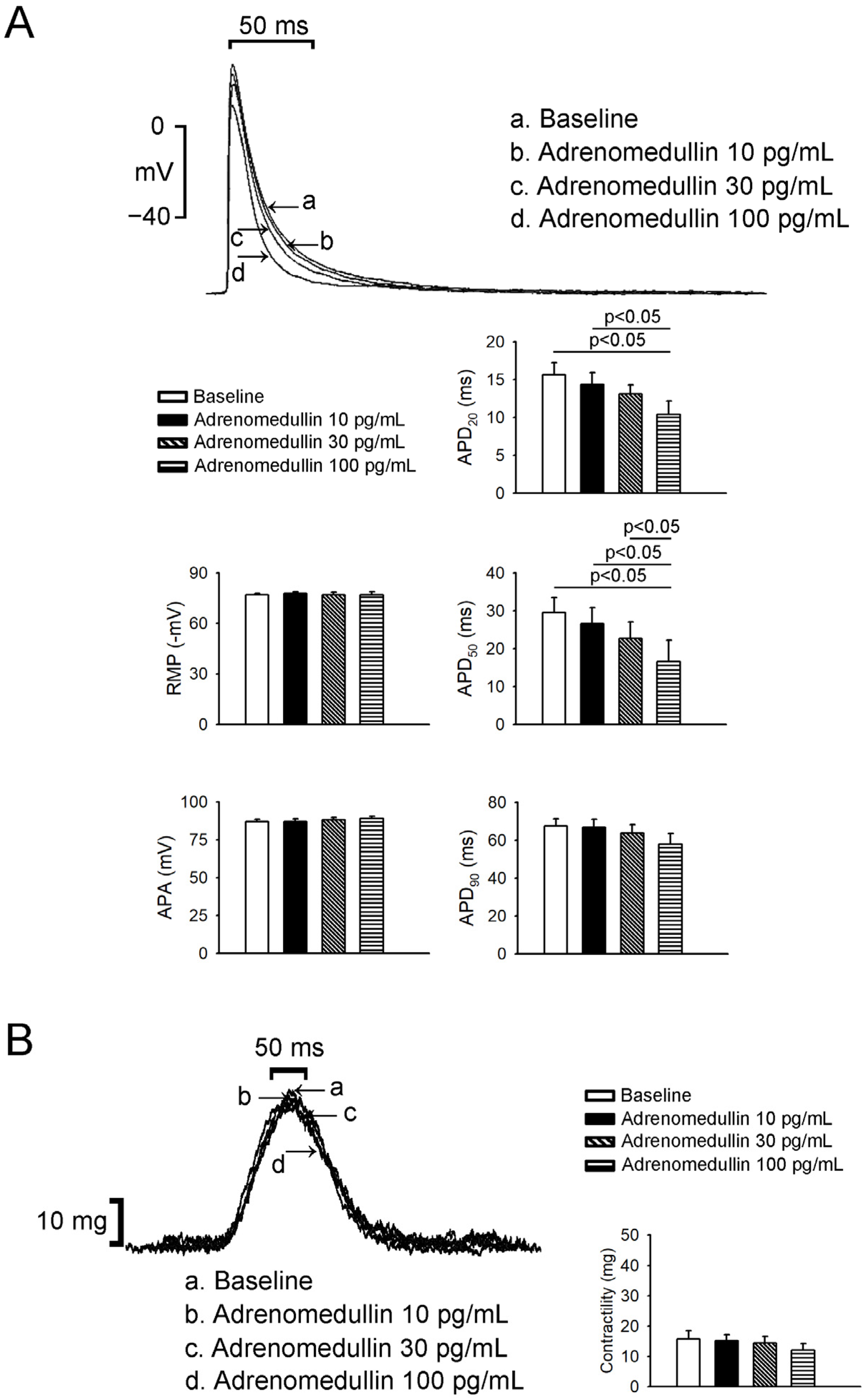
2.2. Effects of Adrenomedullin on Left Atrium (LA) and Right Atrium (RA) Electrophysical Characteristics and Contractility
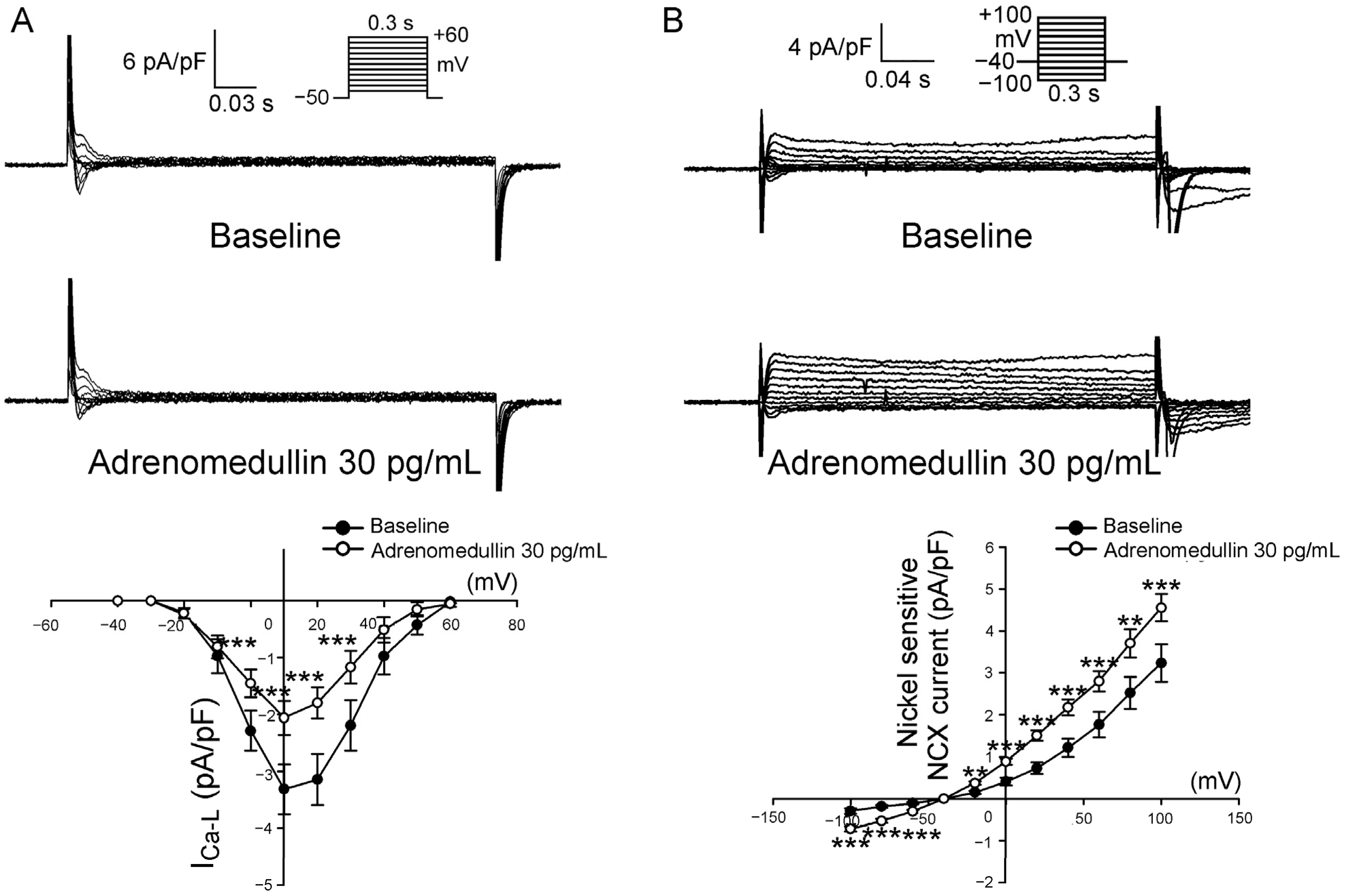
2.3. Effects of Adrenomedullin on ICa-L and NCX in PV Cardiomyocytes
3. Discussion
4. Materials and Methods
4.1. Electropharmacological Studies of PV and Sinoatrial Node Tissues
4.2. Isolation of Single PV Cardiomyocytes
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Samson, W.K. Adrenomedullin and the control of fluid and electrolyte homeostasis. Annu. Rev. Physiol. 1999, 61, 363–389. [Google Scholar] [CrossRef]
- Poyner, D.R.; Sexton, P.M.; Marshall, I.; Smith, D.M.; Quirion, R.; Born, W.; Muff, R.; Fischer, J.A.; Foord, S.M. International Union of Pharmacology. XXXII. The mammalian calcitonin gene-related peptides, adrenomedullin, amylin, and calcitonin receptors. Pharmacol. Rev. 2002, 54, 233–246. [Google Scholar] [CrossRef]
- Voors, A.A.; Kremer, D.; Geven, C.; Ter Maaten, J.M.; Struck, J.; Bergmann, A.; Pickkers, P.; Metra, M.; Mebazaa, A.; Dungen, H.D.; et al. Adrenomedullin in heart failure: Pathophysiology and therapeutic application. Eur. J. Heart Fail. 2019, 21, 163–171. [Google Scholar] [CrossRef]
- Ishimitsu, T.; Nishikimi, T.; Saito, Y.; Kitamura, K.; Eto, T.; Kangawa, K.; Matsuo, H.; Omae, T.; Matsuoka, H. Plasma levels of adrenomedullin, a newly identified hypotensive peptide, in patients with hypertension and renal failure. J. Clin. Investig. 1994, 94, 2158–2161. [Google Scholar] [CrossRef]
- Kohno, M.; Hanehira, T.; Kano, H.; Horio, T.; Yokokawa, K.; Ikeda, M.; Minami, M.; Yasunari, K.; Yoshikawa, J. Plasma adrenomedullin concentrations in essential hypertension. Hypertension 1996, 27, 102–107. [Google Scholar] [CrossRef]
- Martinez-Herrero, S.; Martinez, A. Adrenomedullin: Not Just Another Gastrointestinal Peptide. Biomolecules 2022, 12, 156. [Google Scholar] [CrossRef]
- Hinson, J.P.; Kapas, S.; Smith, D.M. Adrenomedullin, a multifunctional regulatory peptide. Endocr. Rev. 2000, 21, 138–167. [Google Scholar] [CrossRef]
- Ter Maaten, J.M.; Kremer, D.; Demissei, B.G.; Struck, J.; Bergmann, A.; Anker, S.D.; Ng, L.L.; Dickstein, K.; Metra, M.; Samani, N.J.; et al. Bio-adrenomedullin as a marker of congestion in patients with new-onset and worsening heart failure. Eur. J. Heart Fail. 2019, 21, 732–743. [Google Scholar] [CrossRef]
- Gaggin, H.K.; Januzzi, J.L., Jr. Biomarkers and diagnostics in heart failure. Biochim. Biophys. Acta 2013, 1832, 2442–2450. [Google Scholar] [CrossRef]
- Kita, T.; Kitamura, K. Translational studies of adrenomedullin and related peptides regarding cardiovascular diseases. Hypertens. Res. 2022, 45, 389–400. [Google Scholar] [CrossRef]
- Passaglia, P.; Gonzaga, N.A.; Tirapelli, D.P.; Tirapelli, L.F.; Tirapelli, C.R. Pharmacological characterisation of the mechanisms underlying the relaxant effect of adrenomedullin in the rat carotid artery. J. Pharm. Pharmacol. 2014, 66, 1734–1746. [Google Scholar] [CrossRef]
- Geven, C.; Kox, M.; Pickkers, P. Adrenomedullin and Adrenomedullin-Targeted Therapy As Treatment Strategies Relevant for Sepsis. Front. Immunol. 2018, 9, 292. [Google Scholar] [CrossRef]
- Lardizabal, J.A.; Deedwania, P.C. Atrial fibrillation in heart failure. Med. Clin. N. Am. 2012, 96, 987–1000. [Google Scholar] [CrossRef]
- Chen, S.A.; Hsieh, M.H.; Tai, C.T.; Tsai, C.F.; Prakash, V.S.; Yu, W.C.; Hsu, T.L.; Ding, Y.A.; Chang, M.S. Initiation of atrial fibrillation by ectopic beats originating from the pulmonary veins: Electrophysiological characteristics, pharmacological responses, and effects of radiofrequency ablation. Circulation 1999, 100, 1879–1886. [Google Scholar] [CrossRef]
- Kalifa, J.; Jalife, J.; Zaitsev, A.V.; Bagwe, S.; Warren, M.; Moreno, J.; Berenfeld, O.; Nattel, S. Intra-atrial pressure increases rate and organization of waves emanating from the superior pulmonary veins during atrial fibrillation. Circulation 2003, 108, 668–671. [Google Scholar] [CrossRef]
- Zhang, X.H.; Li, G.R.; Bourreau, J.P. The effect of adrenomedullin on the L-type calcium current in myocytes from septic shock rats: Signaling pathway. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H2888–H2893. [Google Scholar] [CrossRef]
- Khan, S.Q.; O′Brien, R.J.; Struck, J.; Quinn, P.; Morgenthaler, N.; Squire, I.; Davies, J.; Bergmann, A.; Ng, L.L. Prognostic value of midregional pro-adrenomedullin in patients with acute myocardial infarction: The LAMP (Leicester Acute Myocardial Infarction Peptide) study. J. Am. Coll. Cardiol. 2007, 49, 1525–1532. [Google Scholar] [CrossRef]
- Kerkela, R.; Ilves, M.; Pikkarainen, S.; Tokola, H.; Ronkainen, V.P.; Majalahti, T.; Leppaluoto, J.; Vuolteenaho, O.; Ruskoaho, H. Key roles of endothelin-1 and p38 MAPK in the regulation of atrial stretch response. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R140–R149. [Google Scholar] [CrossRef][Green Version]
- Latini, R.; Masson, S.; Pirelli, S.; Barlera, S.; Pulitano, G.; Carbonieri, E.; Gulizia, M.; Vago, T.; Favero, C.; Zdunek, D.; et al. Circulating cardiovascular biomarkers in recurrent atrial fibrillation: Data from the GISSI-atrial fibrillation trial. J. Intern. Med. 2011, 269, 160–171. [Google Scholar] [CrossRef]
- Smith, J.G.; Newton-Cheh, C.; Almgren, P.; Struck, J.; Morgenthaler, N.G.; Bergmann, A.; Platonov, P.G.; Hedblad, B.; Engstrom, G.; Wang, T.J.; et al. Assessment of conventional cardiovascular risk factors and multiple biomarkers for the prediction of incident heart failure and atrial fibrillation. J. Am. Coll. Cardiol. 2010, 56, 1712–1719. [Google Scholar] [CrossRef] [PubMed]
- Kita, T.; Kaji, Y.; Kitamura, K. Safety, Tolerability, and Pharmacokinetics of Adrenomedullin in Healthy Males: A Randomized, Double-Blind, Phase 1 Clinical Trial. Drug Des. Dev. Ther. 2020, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, N.; Satoh, T.; Nishikimi, T.; Uematsu, M.; Furuichi, S.; Sakamaki, F.; Oya, H.; Kyotani, S.; Nakanishi, N.; Goto, Y.; et al. Hemodynamic, renal, and hormonal effects of adrenomedullin infusion in patients with congestive heart failure. Circulation 2000, 101, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, H.; Hirata, Y.; Kakoki, M.; Suzuki, Y.; Nishimatsu, H.; Nagata, D.; Suzuki, E.; Kikuchi, K.; Nagano, T.; Kangawa, K.; et al. Role of nitric oxide–cGMP pathway in adrenomedullin-induced vasodilation in the rat. Hypertension 1999, 33, 689–693. [Google Scholar] [CrossRef]
- Kato, J.; Tsuruda, T.; Kita, T.; Kitamura, K.; Eto, T. Adrenomedullin: A protective factor for blood vessels. Arter. Thromb. Vasc. Biol. 2005, 25, 2480–2487. [Google Scholar] [CrossRef]
- Chang, S.L.; Chen, Y.C.; Chen, Y.J.; Wangcharoen, W.; Lee, S.H.; Lin, C.I.; Chen, S.A. Mechanoelectrical feedback regulates the arrhythmogenic activity of pulmonary veins. Heart 2007, 93, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Mittra, S.; Hyvelin, J.M.; Shan, Q.; Tang, F.; Bourreau, J.P. Role of cyclooxygenase in ventricular effects of adrenomedullin: Is adrenomedullin a double-edged sword in sepsis? Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H1034–H1042. [Google Scholar] [CrossRef]
- Mittra, S.; Bourreau, J.P. Gs and Gi coupling of adrenomedullin in adult rat ventricular myocytes. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H1842–H1847. [Google Scholar] [CrossRef]
- Ikenouchi, H.; Kangawa, K.; Matsuo, H.; Hirata, Y. Negative inotropic effect of adrenomedullin in isolated adult rabbit cardiac ventricular myocytes. Circulation 1997, 95, 2318–2324. [Google Scholar] [CrossRef]
- Chan, C.S.; Lin, Y.K.; Chen, Y.C.; Lu, Y.Y.; Chen, S.A.; Chen, Y.J. Heart Failure Differentially Modulates Natural (Sinoatrial Node) and Ectopic (Pulmonary Veins) Pacemakers: Mechanism and Therapeutic Implication for Atrial Fibrillation. Int. J. Mol. Sci. 2019, 20, 3224. [Google Scholar] [CrossRef]
- Chin, C.G.; Chen, Y.C.; Lin, Y.K.; Lu, Y.Y.; Cheng, W.L.; Chung, C.C.; Chen, S.A.; Chen, Y.J. Effect of macrophage migration inhibitory factor on pulmonary vein arrhythmogenesis through late sodium current. Europace 2022. [Google Scholar] [CrossRef]
- Hung, Y.; Chen, Y.C.; Huang, S.Y.; Lu, Y.Y.; Lin, Y.K.; Kao, Y.H.; Lin, W.S.; Chen, S.A.; Chen, Y.J. Klotho modulates electrical activity and calcium homeostasis in pulmonary vein cardiomyocytes via PI3K/Akt signalling. Europace 2020, 22, 1132–1141. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chin, C.-G.; Elimam, A.M.; Lin, F.-J.; Chen, Y.-C.; Lin, Y.-K.; Lu, Y.-Y.; Higa, S.; Chen, S.-A.; Hsieh, M.-H.; Chen, Y.-J. Effects of Adrenomedullin on Atrial Electrophysiology and Pulmonary Vein Arrhythmogenesis. Int. J. Mol. Sci. 2022, 23, 14064. https://doi.org/10.3390/ijms232214064
Chin C-G, Elimam AM, Lin F-J, Chen Y-C, Lin Y-K, Lu Y-Y, Higa S, Chen S-A, Hsieh M-H, Chen Y-J. Effects of Adrenomedullin on Atrial Electrophysiology and Pulmonary Vein Arrhythmogenesis. International Journal of Molecular Sciences. 2022; 23(22):14064. https://doi.org/10.3390/ijms232214064
Chicago/Turabian StyleChin, Chye-Gen, Ahmed Moustafa Elimam, Fong-Jhih Lin, Yao-Chang Chen, Yung-Kuo Lin, Yen-Yu Lu, Satoshi Higa, Shih-Ann Chen, Ming-Hsiung Hsieh, and Yi-Jen Chen. 2022. "Effects of Adrenomedullin on Atrial Electrophysiology and Pulmonary Vein Arrhythmogenesis" International Journal of Molecular Sciences 23, no. 22: 14064. https://doi.org/10.3390/ijms232214064
APA StyleChin, C.-G., Elimam, A. M., Lin, F.-J., Chen, Y.-C., Lin, Y.-K., Lu, Y.-Y., Higa, S., Chen, S.-A., Hsieh, M.-H., & Chen, Y.-J. (2022). Effects of Adrenomedullin on Atrial Electrophysiology and Pulmonary Vein Arrhythmogenesis. International Journal of Molecular Sciences, 23(22), 14064. https://doi.org/10.3390/ijms232214064






