Molecular Indicators of Blood-Brain Barrier Breakdown and Neuronal Injury in Pregnancy Complicated by Fetal Growth Restriction
Abstract
1. Introduction
2. Results
2.1. Group Characteristics
2.2. Serum and Placental Markers
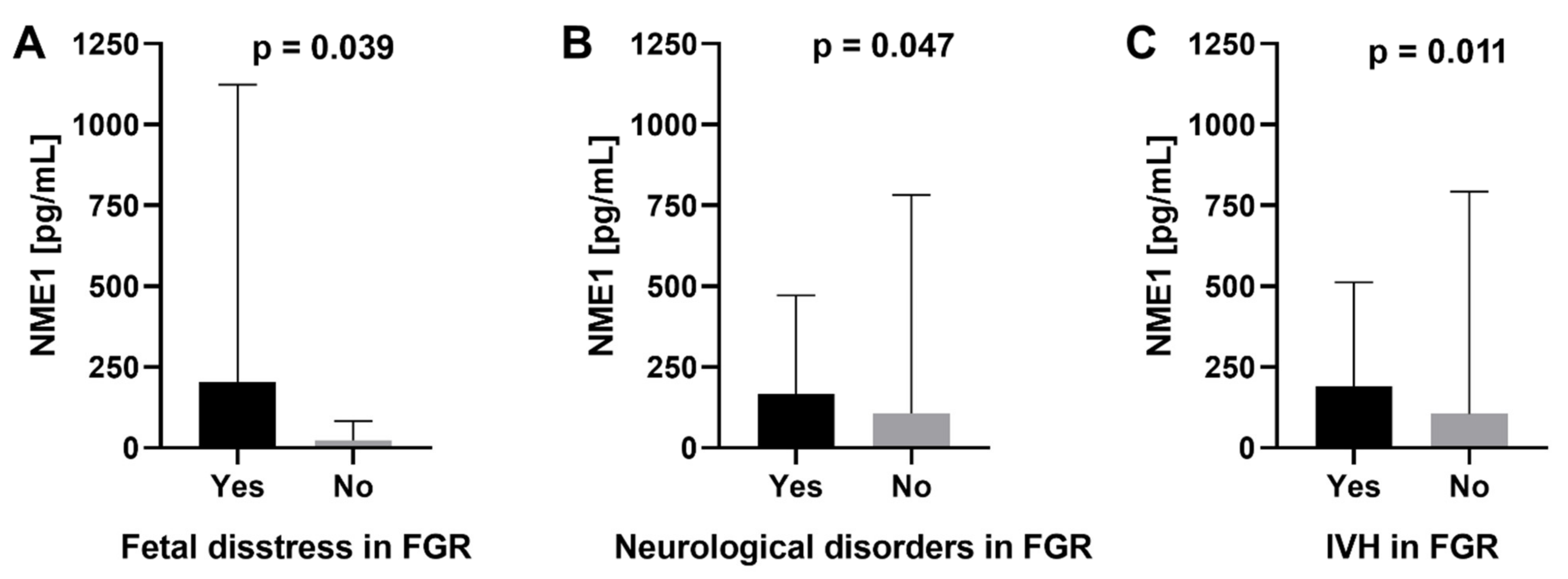
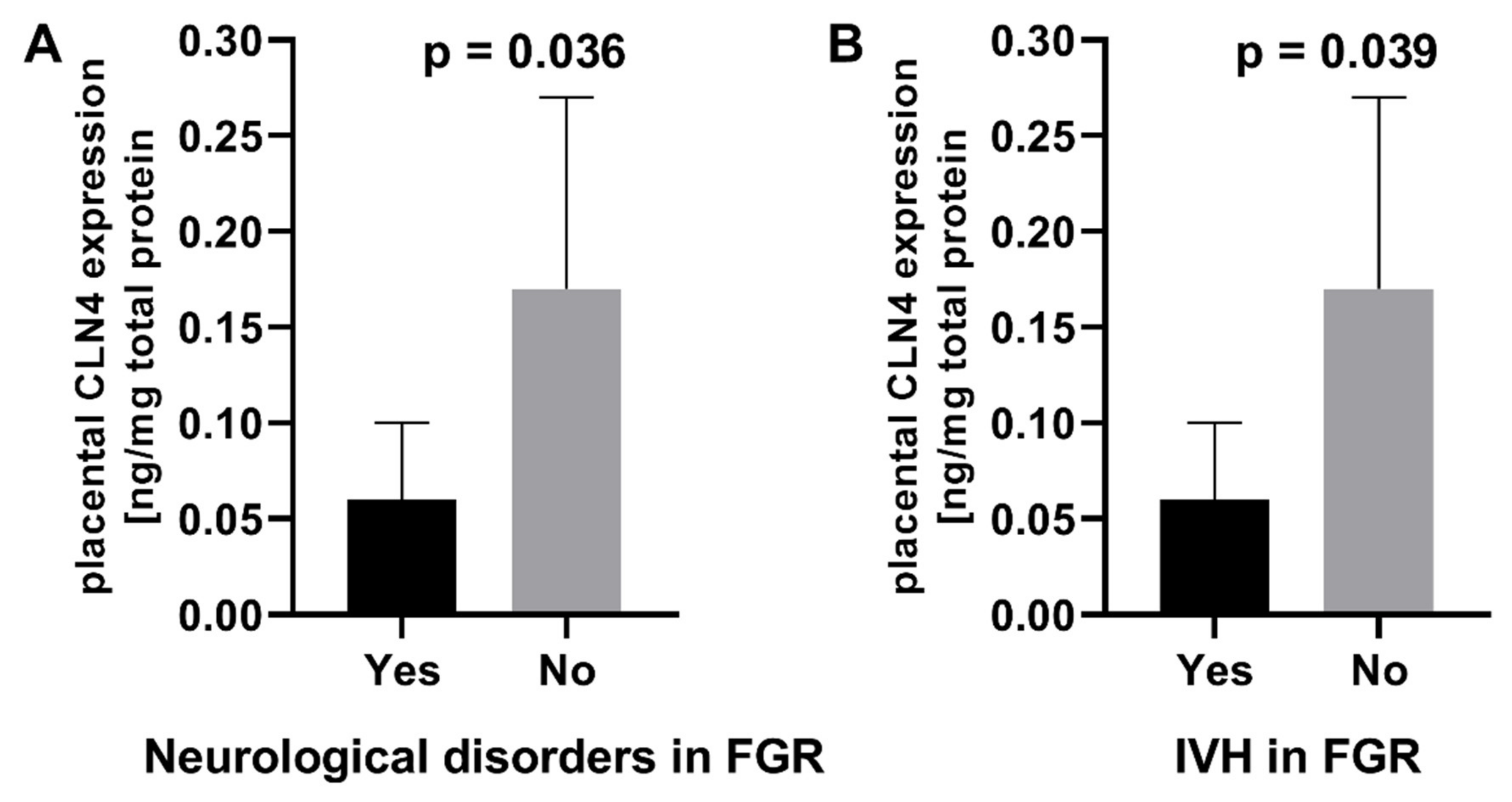
2.3. Association of Serum and Placental Markers and Neurological Outcomes
3. Discussion
4. Materials and Methods
4.1. The Studied and Control Groups
4.2. Collection of Blood and Placental Samples
4.3. Laboratory Serum Assays
4.4. Laboratory Placental Tests
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murki, S.; Sharma, D. Intrauterine growth retardation—A review article. J. Neonatal Biol. 2014, 3, 135. [Google Scholar] [CrossRef]
- Sharma, D.; Shastri, S.; Sharma, P. Intrauterine growth restriction: Antenatal and postnatal aspects. Clin. Med. Insights Pediatr. 2016, 10, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Mamelle, N.; Cochet, V.; Claris, O. Definition of fetal growth restriction according to constitutional growth potential. Biol. Neonate. 2001, 80, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Figueras, F.; Gratacós, E. Update on the diagnosis and classification of fetal growth restriction and proposal of a stage-based management protocol. Fetal Diagn. Ther. 2014, 36, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Nardozza, L.M.M.; Caetano, A.C.R.; Zamarian, A.C.P.; Mazzola, J.B.; Silva, C.P.; Marçal, V.; Lobo, T.F.; Peixoto, A.B.; Júnior, E.A. Fetal growth restriction: Current knowledge. Arch. Gynecol. Obstet. 2017, 295, 1061–1077. [Google Scholar] [CrossRef]
- Moh, W.; Graham, J.M.; Wadhawan, I.; Sanchez-Lara, P.A. Extrinsic factors influencing fetal deformations and intrauterine growth restriction. J. Pregnancy 2012, 2012, 750485. [Google Scholar] [CrossRef]
- Salafia, C.M.; Minior, V.K.; Pezzullo, J.C.; Popek, E.J.; Rosenkrantz, T.S.; Vintzileos, A.M. Intrauterine growth restriction in infants of less than thirty-two weeks’ gestation: Associated placental pathologic features. Am. J. Obstet. Gynecol. 1995, 173, 1049–1057. [Google Scholar] [CrossRef]
- Brosens, I.; Dixon, H.G.; Robertson, W.B. Fetal growth retardation and the arteries of the placental bed. Br. J. Obstet. Gynaecol. 1977, 84, 656–663. [Google Scholar] [CrossRef]
- Khong, T.Y.; De Wolf, F.; Robertson, W.B.; Brosens, I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. Br. J. Obstet. Gynaecol. 1986, 93, 1049–1059. [Google Scholar] [CrossRef]
- Giles, W.B.; Trudinger, B.J.; Baird, P.J. Fetal umbilical artery flow velocity waveforms and placental resistance: Pathological correlation. Br. J. Obstet. Gynaecol. 1985, 92, 31–38. [Google Scholar] [CrossRef]
- Barker, D.J.P. Fetal programming: Influences on development and disease in later life. In NIH Monograph Series; Marcel Dekker: New York, NY, USA, 2000. [Google Scholar]
- Baker, J.L.; Olsen, L.W.; Sørensen, T.I. Childhood body-mass index and the risk of coronary heart disease in adulthood. N. Engl. J. Med. 2007, 357, 2329–2337. [Google Scholar] [CrossRef]
- Barker, D.; Osmond, C.; Winter, P.; Margetts, B.; Simmonds, S. Weight in infancy and death from ischaemic heart disease. Lancet 1989, 2, 577–580. [Google Scholar] [CrossRef]
- Ramírez-Vélez, R. In utero fetal programming and its impact on health in adulthood. Endocrinol. Nutr. 2012, 59, 383–393. [Google Scholar] [CrossRef]
- Ego, A.; Zeitlin, J.; Batailler, P.; Cornec, S.; Fondeur, A.; Baran-Marszak, M.; Jouk, P.-S.; Debillon, T.; Cans, C. Stillbirth classification in population-based data and role of fetal growth restriction: The example of RECODE. BMC Pregnancy Childbirth 2013, 13, 182. [Google Scholar] [CrossRef]
- Unterscheider, J.; O’Donoghue, K.; Daly, S.; Geary, M.P.; Kennelly, M.M.; McAuliffe, F.M.; Hunter, A.; Morrison, J.J.; Burke, G.; Dicker, P.; et al. Fetal growth restriction and the risk of perinatal mortality-case studies from the multicentre PORTO study. BMC Pregnancy Childbirth 2014, 14, 63. [Google Scholar] [CrossRef]
- Im, D.H.; Kim, Y.N.; Cho, H.J.; Park, Y.H.; Kim, D.H.; Byun, J.M.; Jeong, D.H.; Lee, K.B.; Sung, M.S. Placental Pathologic Changes Associated with Fetal Growth Restriction and Consequent Neonatal Outcomes. Fetal Pediatr. Pathol. 2020, 40, 430–441. [Google Scholar] [CrossRef]
- Colson, A.; Sonveaux, P.; Debiève, F.; Sferruzzi-Perri, A.N. Adaptations of the human placenta to hypoxia: Opportunities for interventions in fetal growth restriction. Hum. Reprod. Update 2021, 27, 531–569. [Google Scholar] [CrossRef]
- Nowaczyk, J.; Poniedziałek, B.; Rzymski, P.; Sikora, D.; Ropacka-Lesiak, M. Platelets in Fetal Growth Restriction: Role of Reactive Oxygen Species, Oxygen Metabolism, and Aggregation. Cells 2022, 11, 724. [Google Scholar] [CrossRef]
- Verburg, B.O.; Jaddoe, V.W.; Wladimiroff, J.W.; Hofman, A.; Witteman, J.C.; Steegers, E.A. Fetal hemodynamic adaptive changes related to intrauterine growth: The Generation R Study. Circulation 2008, 117, 649–659. [Google Scholar] [CrossRef]
- Garcia-Canadilla, P.; Rudenick, P.A.; Crispi, F.; Cruz-Lemini, M.; Palau, G.; Camara, O.; Gratacos, E.; Bijens, B.H. A Computational Model of the Fetal Circulation to Quantify Blood Redistribution in Intrauterine Growth Restriction. PLOS Comput. Biol. 2014, 10, e1003667. [Google Scholar] [CrossRef]
- MacDonald, T.M.; Hui, L.; Tong, S.; Robinson, A.J.; Dane, K.M.; Middleton, A.L.; Walker, S.P. Reduced growth velocity across the third trimester is associated with placental insufficiency in fetuses born at a normal birthweight: A prospective cohort study. BMC Med. 2017, 15, 164. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.; Wong, F.Y.; Horne, R.S.; Yiallourou, S. Intrauterine growth restriction: Impact on cardiovascular development and function throughout infancy. Pediatr. Res. 2016, 79, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.; Crimmins, S.; Telugu, B.; Turan, S. Intrauterine hypoxia: Clinical consequences and therapeutic perspectives. Res. Rep. Neonatol. 2015, 5, 79–89. [Google Scholar] [CrossRef]
- Malhotra, A.; Ditchfield, M.; Fahey, M.C.; Castillo-Melendez, M.; Allison, B.; Polglase, G.; Wallace, E.; Hodges, R.; Jenkin, G.; Miller, S. detection and assessment of brain injury in the growth-restricted fetus and neonate. Pediatr. Res. 2017, 82, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Rees, S.; Harding, R.; Walker, D. The biological basis of injury and neuroprotection in the fetal and neonatal brain. Int. J. Dev. Neurosci. 2011, 29, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.L.; Huppi, P.S.; Mallard, C. The consequences of fetal growth restriction on brain structure and neurodevelopmental outcome. J. Physiol. 2016, 594, 807–823. [Google Scholar] [CrossRef]
- Polat, A.; Barlow, S.; Ber, R.; Achiron, R.; Katorza, E. Volumetric MRI study of the intrauterine growth restriction fetal brain. Eur. Radiol. 2017, 27, 2110–2118. [Google Scholar] [CrossRef]
- Padilla, N.; Junqué, C.; Figueras, F.; Sanz-Cortes, M.; Bargallo, N.; Arranz, A.; Donaire, A.; Figueras, J.; Gratacos, E. Differential vulnerability of gray matter and white matter to intrauterine growth restriction in preterm infants at 12 months corrected age. Brain Res. 2014, 1545, 1–11. [Google Scholar] [CrossRef]
- Morsing, E.; Malova, M.; Kahn, A.; Lätt, J.; Björkman-Burtscher, I.; Maršál, K.; Ley, D. Brain Volumes and Developmental Outcome in Childhood Following Fetal Growth Restriction Leading to Very Preterm Birth. Front. Physiol. 2018, 9, 1583. [Google Scholar] [CrossRef]
- Samuelsen, G.B.; Pakkenberg, B.; Bogdanović, N.; Gundersen, H.J.; Larsen, J.F.; Græm, N.; Laursen, H. Severe cell reduction in the future brain cortex in human growth-restricted fetuses and infants. Am. J. Obstet. Gynecol. 2007, 197, 56.e1–56.e7. [Google Scholar] [CrossRef]
- Dubois, J.; Benders, M.; Borradori-Tolsa, C.; Cachia, A.; Lazeyras, F.; Leuchter, R.H.-V.; Sizonenko, S.V.; Warfield, S.K.; Mangin, J.F.; Hüppi, P.S. Primary cortical folding in the human newborn: An early marker of later functional development. Brain 2008, 131 Pt 8, 2028–2041. [Google Scholar] [CrossRef]
- Batalle, D.; Eixarch, E.; Figueras, F.; Muñoz-Moreno, E.; Bargallo, N.; Illa, M.; Acosta-Rojas, R.; Amat-Roldan, I.; Gratacos, E. Altered small-world topology of structural brain networks in infants with intrauterine growth restriction and its association with later neurodevelopmental outcome. Neuroimage 2012, 60, 1352–1366. [Google Scholar] [CrossRef]
- Fischi-Gómez, E.; Vasung, L.; Meskaldji, D.-E.; Lazeyras, F.; Borradori-Tolsa, C.; Hagmann, P.; Barisnikov, K.; Thiran, J.-P.; Hüppi, P.S. Structural Brain Connectivity in School-Age Preterm Infants Provides Evidence for Impaired Networks Relevant for Higher Order Cognitive Skills and Social Cognition. Cereb. Cortex 2015, 25, 2793–2805. [Google Scholar] [CrossRef]
- Kazmierski, R.; Michalak, S.; Wencel-Warot, A.; Nowinski, W.L. Serum tight-junction proteins predict hemorrhagic transformation in ischemic stroke patients. Neurology 2012, 79, 1677–1685. [Google Scholar] [CrossRef]
- Kazmierski, R.; Michalak, S.; Wencel-Warot, A.; Nowinski, W.L. Blood-brain barrier dysfunction and recovery after ischemic stroke. Prog. Neurobiol. 2018, 163–164, 144–171. [Google Scholar] [CrossRef]
- Chow, B.W.; Gu, C. The molecular constituents of the blood-brain barrier. Trends Neurosci. 2015, 38, 598–608. [Google Scholar] [CrossRef]
- Persidsky, Y.; Ramirez, S.; Haorah, J.; Kanmogne, G.D. Blood-brain barrier: Structural components and function under physiologic and pathologic conditions. J. Neuroimmune Pharmacol. 2006, 1, 223–236. [Google Scholar] [CrossRef]
- Baeten, K.M.; Akassoglou, K. Extracellular matrix and matrix receptors in blood-brain barrier formation and stroke. Dev. Neurobiol. 2011, 71, 1018–1039. [Google Scholar] [CrossRef]
- Günzel, D.; Yu, A.S. Claudins and the modulation of tight junction permeability. Physiol. Rev. 2013, 93, 525–569. [Google Scholar] [CrossRef]
- Luissint, A.-C.; Artus, C.; Glacial, F.; Ganeshamoorthy, K.; Couraud, P.-O. Tight junctions at the blood brain barrier: Physiological architecture and disease-associated dysregulation. Fluids Barriers CNS 2012, 9, 23. [Google Scholar] [CrossRef]
- Greene, C.; Campbell, M. Tight junction modulation of the blood brain barrier: CNS delivery of small molecules. Tissue Barriers 2016, 4, e1138017. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Arrastia, R.; Wang, K.; Papa, L.; Sorani, M.D.; Yue, J.; Puccio, A.M.; McMahon, P.J.; Inoue, T.; Yuh, E.L.; Lingsma, H.F.; et al. Acute biomarkers of traumatic brain injury: Relationship between plasma levels of ubiquitin C-terminal hydrolase-L1 and glial fibrillary acidic protein. J. Neurotrauma 2014, 31, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Graham, E.M.; Burd, I.; Everett, A.D.; Northington, F.J. Blood Biomarkers for Evaluation of Perinatal Encephalopathy. Front. Pharmacol. 2016, 7, 196. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.-C.; Kwon, J.-Y.; Hwang, H.-S.; Kim, Y.-H.; Park, Y.-W. 488: Comparison of placental junctional proteins expression in preeclapsia with intrauterine growth restriction. Am. J. Obstet. Gynecol. 2007, 197, S142. [Google Scholar] [CrossRef]
- Scher, M.S. “The First Thousand Days” Define a Fetal/Neonatal Neurology Program. Front. Pediatr. 2021, 9, 683138. [Google Scholar] [CrossRef]
- Tam, S.J.; Watts, R.J. Connecting vascular and nervous system development: Angiogenesis and the blood-brain barrier. Annu. Rev. Neurosci. 2010, 33, 379–408. [Google Scholar] [CrossRef]
- Warrington, J.P.; Fan, F.; Murphy, S.R.; Roman, R.J.; Drummond, H.A.; Granger, J.P.; Ryan, M.J. Placental ischemia in pregnant rats impairs cerebral blood flow autoregulation and increases blood-brain barrier permeability. Physiol. Rep. 2014, 2, e12134. [Google Scholar] [CrossRef]
- Kuvacheva, N.V.; Morgun, A.; Malinovskaya, N.A.; Gorina, Y.V.; Khilazheva, E.D.; Pozhilenkova, E.A.; Panina, Y.; Boytsova, E.B.; Ruzaeva, V.A.; Trufanova, L.V.; et al. Tight junction proteins of cerebral endothelial cells in early postnatal development. Cell Tissue Biol. 2016, 10, 372–377. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, H.; Pan, Q.; Zhao, Y.; Chen, J.; Zhao, B.; Chen, Y. Hypoxia/Aglycemia-induced endothelial barrier dysfunction and tight junction protein downregulation can be ameliorated by citicoline. PLoS ONE 2013, 8, e82604. [Google Scholar] [CrossRef]
- Zehendner, C.M.; Librizzi, L.; Hedrich, J.; Bauer, N.M.; Angamo, E.A.; De Curtis, M.; Luhmann, H.J. Moderate hypoxia followed by reoxygenation results in blood-brain barrier breakdown via oxidative stress-dependent tight-junction protein disruption. PLoS ONE 2013, 8, e82823. [Google Scholar] [CrossRef]
- Mark, K.S.; Davis, T.P. Cerebral microvascular changes in permeability and tight junctions induced by hypoxia-reoxygenation. Am. J. Physiol. Heart Circ. Physiol. 2002, 282, H1485–H1494. [Google Scholar] [CrossRef]
- Yamagata, K.; Tagami, M.; Takenaga, F.; Yamori, Y.; Itoh, S. Hypoxia-induced changes in tight junction permeability of brain capillary endothelial cells are associated with IL-1beta and nitric oxide. Neurobiol. Dis. 2004, 17, 491–499. [Google Scholar] [CrossRef]
- Gazzolo, D.; Marinoni, E.; Di Iorio, R.; Lituania, M.; Marras, M.; Bruschettini, M.; Bruschettini, P.; Frulio, R.; Michetti, F.; Petraglia, F.; et al. High maternal blood S100B concentrations in pregnancies complicated by intrauterine growth restriction and intraventricular hemorrhage. Clin. Chem. 2006, 52, 819–826. [Google Scholar] [CrossRef]
- Velipaşaoğlu, M.; Yurdakök, M.; Özyüncü, Ö.; Portakal, O. Neural injury markers to predict neonatal complications in intrauterine growth restriction. J. Obstet. Gynaecol. 2015, 35, 555–560. [Google Scholar] [CrossRef]
- Gazzolo, D.; Di Iorio, R.; Marinoni, E.; Masetti, P.; Serra, G.; Giovannini, L.; Michetti, F. S100B protein is increased in asphyxiated term infants developing intraventricular hemorrhage. Crit. Care Med. 2002, 30, 1356–1360. [Google Scholar] [CrossRef]
- Mazarico, E.; Llurba, E.; Cumplido, R.; Valls, A.; Melchor, J.C.; Iglesias, M.; Cabero, L.; Gratacós, E.; Gómez-Roig, M.D. Neural injury markers in intrauterine growth restriction and their relation to perinatal outcomes. Pediatr. Res. 2017, 82, 452–457. [Google Scholar] [CrossRef]
- Boutsikou, T.; Mastorakos, G.; Kyriakakou, M.; Margeli, A.; Hassiakos, D.; Papassotiriou, I.; Kanaka-Gantenbein, C.; Malamitsi-Puchner, A. Circulating levels of inflammatory markers in intrauterine growth restriction. Mediat. Inflamm. 2010, 2010, 790605. [Google Scholar] [CrossRef]
- Kirmemiş, Ő.; Tatli, M.M.; Tayman, C.; Koca, C.; Karadağ, A.; Uras, N.; Dilmen, U.; Kafali, H. Subclinical hypoxia of infants with intrauterine growth retardation determined by increased serum S100B protein levels. Turk. J. Med. Sci. 2011, 41, 795–800. [Google Scholar] [CrossRef]
- Florio, P.; Marinoni, E.; Di Iorio, R.; Bashir, M.; Ciotti, S.; Sacchi, R.; Bruschettini, M.; Lituania, M.; Serra, G.; Michetti, F.; et al. Urinary S100B protein concentrations are increased in intrauterine growth-retarded newborns. Pediatrics 2006, 118, e747–e754. [Google Scholar] [CrossRef]
- Marinoni, E.; Di Iorio, R.; Gazzolo, D.; Lucchini, C.; Michetti, F.; Corvino, V.; Cosmi, E.V. Ontogenetic localization and distribution of S-100beta protein in human placental tissues. Obstet. Gynecol. 2002, 99, 1093–1099. [Google Scholar] [CrossRef]
- Schmidt, A.P.; Tort, A.; Amaral, O.; Schmidt, A.P.; Walz, R.; Vettorazzi, J.; Martins-Costa, S.H.; Ramos, J.G.L.; Souza, D.; Portela, L.V. Serum S100B in pregnancy-related hypertensive disorders: A case-control study. Clin. Chem. 2004, 50, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Tingley, W.G.; Ehlers, M.D.; Kameyama, K.; Doherty, C.; Ptak, J.B.; Riley, C.T.; Huganir, R.L. Characterization of protein kinase A and protein kinase C phosphorylation of the N-methyl-D-aspartate receptor NR1 subunit using phosphorylation site-specific antibodies. J. Biol. Chem. 1997, 272, 5157–5166. [Google Scholar] [CrossRef] [PubMed]
- Mishra, O.P.; Delivoria-Papadopoulos, M. Modification of modulatory sites of NMDA receptor in the fetal guinea pig brain during development. Neurochem. Res. 1992, 17, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Schober, M.E.; McKnight, R.A.; Yu, X.; Callaway, C.W.; Ke, X.; Lane, R.H. Intrauterine growth restriction due to uteroplacental insufficiency decreased white matter and altered NMDAR subunit composition in juvenile rat hippocampi. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R681–R692. [Google Scholar] [CrossRef] [PubMed]
- Phillips, T.J.; Scott, H.; Menassa, D.A.; Bignell, A.L.; Sood, A.; Morton, J.S.; Akagi, T.; Azuma, K.; Rogers, M.F.; Gilmore, C.E.; et al. Treating the placenta to prevent adverse effects of gestational hypoxia on fetal brain development. Sci. Rep. 2017, 7, 9079. [Google Scholar] [CrossRef] [PubMed]
- Liévano, S.; Alarcón, L.; Chávez–Munguía, B.; González–Mariscal, L. Endothelia of term human placentae display diminished expression of tight junction proteins during pre-eclampsia. Cell Tissue Res. 2006, 324, 433–448. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, H.-J.; Xia, X.-R.; Diao, F.-Y.; Ma, X.; Wang, J.; Gao, L.; Liu, J.; Gao, C.; Cui, Y.-G.; et al. Hypoxia-induced and HIF1α-VEGF-mediated tight junction dysfunction in choriocarcinoma cells: Implications for pre-eclampsia. Clin. Chim. Acta 2019, 489, 203–211. [Google Scholar] [CrossRef]
- Wang, Y.; Gu, Y.; Granger, D.; Roberts, J.M.; Alexander, J. Endothelial junctional protein redistribution and increased monolayer permeability in human umbilical vein endothelial cells isolated during pre-eclampsia. Am. J. Obstet. Gynecol. 2002, 186, 214–220. [Google Scholar] [CrossRef]
- Lööv, C.; Shevchenko, G.; Nadadhur, A.G.; Clausen, F.; Hillered, L.; Wetterhall, M.; Erlandsson, A. Identification of injury specific proteins in a cell culture model of traumatic brain injury. PLoS ONE 2013, 8, e55983. [Google Scholar] [CrossRef]
- Khong, T.Y.; Mooney, E.E.; Ariel, I.; Balmus, N.C.M.; Boyd, T.K.; Brundler, M.-A.; Derricott, H.; Evans, M.J.; Faye-Petersen, O.M.; Gillan, J.E.; et al. Sampling and Definitions of Placental Lesions: Amsterdam Placental Workshop Group Consensus Statement. Arch. Pathol. Lab. Med. 2016, 140, 698–713. [Google Scholar] [CrossRef]
- Papile, L.A.; Burstein, J.; Burstein, R.; Koffler, H. Incidence and evolution of subependymal and intraventricular hemorrhage: A study of infants with birth weights less than 1500 gm. J. Pediatr. 1978, 92, 529–534. [Google Scholar] [CrossRef]
- Bowerman, R.A.; Donn, S.M.; Silver, T.M.; Jaffe, M.H. Natural history of neonatal periventricular/intraventricular hemorrhage and its complications: Sonographic observations. AJR Am. J. Roentgenol. 1984, 143, 1041–1052. [Google Scholar] [CrossRef]
- De Vries, L.S.; Eken, P.; Dubowitz, L. The spectrum of leukomalacia using cranial ultrasound. Behav. Brain Res. 1992, 49, 1–6. [Google Scholar] [CrossRef]
- Ment, L.R.; Bada, H.S.; Barnes, P.; Grant, P.E.; Hirtz, D.; Papile, L.A.; Pinto–Martin, J.; Rivkin, M.; Slovis, T.L. Practice parameter: Neuroimaging of the neonate: Report of the quality standards subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2002, 58, 1726–1738. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Urząd Rejestracji Produktów Leczniczych, Wyrobów Medycznych i Produktów Biobójczych. Farmakopea Polska VII; t. 1, wyd. 7; Urząd Rejestracji Produktów Leczniczych, Wyrobów Medycznych i Produktów Biobójczych: Warszawa, Poland, 2006; 236p, ISBN 83-88157-37-X. [Google Scholar]
| Characteristics | FGR (n = 90) | Physiological Pregnancy (n = 70) | p-Value |
|---|---|---|---|
| Age (years, mean ± SD) | 29 ± 5 | 31 ± 5 | 0.0662 |
| BMI at the first prenatal visit (kg/m2) median, min–max) | 22.2 (15.2–42.0) | 21.9 (17.4–30.5) | 0.8443 |
| Gestational age at study eligibility (weeks, median, min–max) | 35 (24–41) | 36 (29–41) | 0.0677 |
| Estimated fetal weight (g, median, min–max) | 1932 (439–2920) | 3255 (756–4259) | <0.0001 |
| Percentile of estimated fetal weight (median, min–max) | 1.0 (0.1–9.0) | 41.0 (4.0–98.0) | <0.0001 |
| Mode of delivery (%) | |||
| Spontaneous | 20.5 | 41.8 | 0.0039 |
| Cesarean section | 75.0 | 41.8 | <0.0001 |
| Vacuum extractor | 4.5 | 16.4 | 0.0132 |
| Forceps | 0.0 | 0.0 | 1.0000 |
| Parameters | FGR (n = 90) | Physiological Pregnancy (n = 70) | p-Value |
|---|---|---|---|
| Delivery week (median, min–max) | 37 (26–41) | 39 (37–42) | <0.0001 |
| Premature delivery (%) | 42.2 | 0.0 | <0.0001 |
| Fetal distress (%) | 50.0 | 20.0 | 0.0001 |
| Birth weight (g) (median, min–max) <2500 overall (%) 1500–2500 (%) 1000–1500 (%) <1000 (%) | 2260 (420–3080) 70.5 46.6 8.0 15.9 | 3450 (2500–4600) 0.0 0.0 0.0 0.0 | <0.0001 |
| Anthropometric measurements (cm) (median, min–max) | |||
| Head circumference | 31.5 (22.0–35.0) | 34.0 (31.0–38.0) | <0.0001 |
| Thoracic circumference | 29.0 (17.5–33.0) | 34.0 (27.0–37.5) | <0.0001 |
| Body length | 49.0 (29.5–55.0) | 55.0 (45.0–61.0) | <0.0001 |
| Apgar score (points) (median, min–max) | |||
| 1st minute | 10 (0–10) | 10 (4–10) | 0.0017 |
| 3rd minute | 8 (2–10) | 9 (6–9) | 0.6209 |
| 5th minute | 10 (4–10) | 10 (9–10) | 0.0001 |
| pH (median, min–max) Venous Arterial | 7.33 (7.01–7.46) 7.27 (6.95–7.45) | 7.33 (7.15–7.48) 7.27 (7.06–7.40) | 0.5835 0.9758 |
| Base excess (mEq/L) (median, min–max) Venous Arterial | −2.6 (−11.3–3.2) −2.3 (−13.4–3.4) | −2.7 (−10.3–2.7) −2.6 (−11.8–3.4) | 0.2271 0.1345 |
| Metabolic acidosis (%) | |||
| Arterial pH < 7,30 overall | 56.7 | 42.9 | 0.1108 |
| 7.20–7.29 | 42.2 | 30.0 | 0.1376 |
| 7.10–7.19 | 12.2 | 10.0 | 0.8023 |
| 7.00–7.09 | 0.0 | 2.9 | 0.1899 |
| <7.00 | 2.2 | 0.0 | 0.5047 |
| Length of hospitalization (days) (median, min–max) | 6 (3–84) | 4 (2–22) | <0.0001 |
| >5 days (%) | 50.6 | 14.5 | <0.0001 |
| Neurological disorders overall (%) | 8.9 | 0.0 | 0.0098 |
| Intraventricular hemorrhage (%) | 7.8 | 0.0 | 0.0186 |
| First degree | 5.6 | 0.0 | 0.0683 |
| Second degree | 1.1 | 0.0 | 1.0000 |
| Third degree | 1.1 | 0.0 | 1.0000 |
| Fourth degree | 0.0 | 0.0 | - |
| Periventricular leucomalacia (%) | 2.2 | 0.0 | 0.5044 |
| Serum Concentrations | FGR (n = 90) | Physiological Pregnancy (n = 70) | p-Value |
|---|---|---|---|
| NR1 (pg/mL) mean ± SD (min–max) | 1295.2 ± 2852.6 (0.0–16,442.1) | 657.2 ± 1531.2 (0.0–9030.8) | 0.0799 |
| NME1 (pg/mL) mean ± SD (min–max) | 112.4 ± 650.7 (0.0–6085.6) | 77.1 ± 317.3 (0.0–2557.8) | 0.9246 |
| S100B (pg/mL) mean ± SD (min–max) | 29.6 ± 40.7 (0.0–165.9) | 14.9 ± 25.1 (0.0–72.8) | 0.0290 |
| OCLN (pg/mL) mean ± SD (min–max) | 32.8 ± 116.3 (0.0–676.0) | 8.4 ± 41.9 (0.0–284.0) | 0.0835 |
| CLN5 (pg/mL) mean ± SD (min–max) | 74.2 ± 167.6 (0.0–828.0) | 30.7 ± 150.6 (0.0–1115.0) | 0.0039 |
| zo-1 (RU/mL) mean ± SD (min–max) | 2.6 ± 4.6 (0.0–24.9) | 1.8 ± 4.2 (0.0–29.3) | 0.3122 |
| OCLN/zo-1 (pg/RU) mean ± SD (min–max) | 11.4 ± 50.5 (0.0–320.9) | 1.7 ± 8.9 (0.0–48.1) | 0.0612 |
| CLN5/zo-1 (pg/RU) mean ± SD (min–max) | 122.8 ± 6378.0 (0.0–4127.0) | 4.7 ± 12.2 (0.0–40.8) | 0.0181 |
| Placental Expression | FGR (n = 90) | Physiological Pregnancy (n = 70) | p-Value |
|---|---|---|---|
| OCLN (ng/mg total protein) mean ± SD (min–max) | 0.18 ± 0.16 (0.00–0.53) | 0.12 ± 0.13 (0.00–0.43) | 0.0791 |
| CLN5 (ng/mg total protein) mean ± SD (min–max) | 0.02 ± 0.02 (0.00–0.11) | 0.01 ± 0.02 (0.00–0.09) | 0.0119 |
| CLN4 (ng/mg total protein) mean ± SD (min–max) | 0.16 ± 0.10 (0.00–0.42) | 0.13 ± 0.10 (0.02–0.43) | 0.1806 |
| zo-1 (RU/mL) mean ± SD (min–max) | 0.24 ± 0.16 (0.02–0.66) | 0.20 ± 0.16 (0.02–0.54) | 0.2615 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Misan, N.; Michalak, S.; Rzymski, P.; Poniedziałek, B.; Kapska, K.; Osztynowicz, K.; Ropacka-Lesiak, M. Molecular Indicators of Blood-Brain Barrier Breakdown and Neuronal Injury in Pregnancy Complicated by Fetal Growth Restriction. Int. J. Mol. Sci. 2022, 23, 13798. https://doi.org/10.3390/ijms232213798
Misan N, Michalak S, Rzymski P, Poniedziałek B, Kapska K, Osztynowicz K, Ropacka-Lesiak M. Molecular Indicators of Blood-Brain Barrier Breakdown and Neuronal Injury in Pregnancy Complicated by Fetal Growth Restriction. International Journal of Molecular Sciences. 2022; 23(22):13798. https://doi.org/10.3390/ijms232213798
Chicago/Turabian StyleMisan, Natalia, Sławomir Michalak, Piotr Rzymski, Barbara Poniedziałek, Katarzyna Kapska, Krystyna Osztynowicz, and Mariola Ropacka-Lesiak. 2022. "Molecular Indicators of Blood-Brain Barrier Breakdown and Neuronal Injury in Pregnancy Complicated by Fetal Growth Restriction" International Journal of Molecular Sciences 23, no. 22: 13798. https://doi.org/10.3390/ijms232213798
APA StyleMisan, N., Michalak, S., Rzymski, P., Poniedziałek, B., Kapska, K., Osztynowicz, K., & Ropacka-Lesiak, M. (2022). Molecular Indicators of Blood-Brain Barrier Breakdown and Neuronal Injury in Pregnancy Complicated by Fetal Growth Restriction. International Journal of Molecular Sciences, 23(22), 13798. https://doi.org/10.3390/ijms232213798








