Stabilized Double-Stranded RNA Strategy Improves Cotton Resistance to CBW (Anthonomus grandis)
Abstract
1. Introduction
2. Results
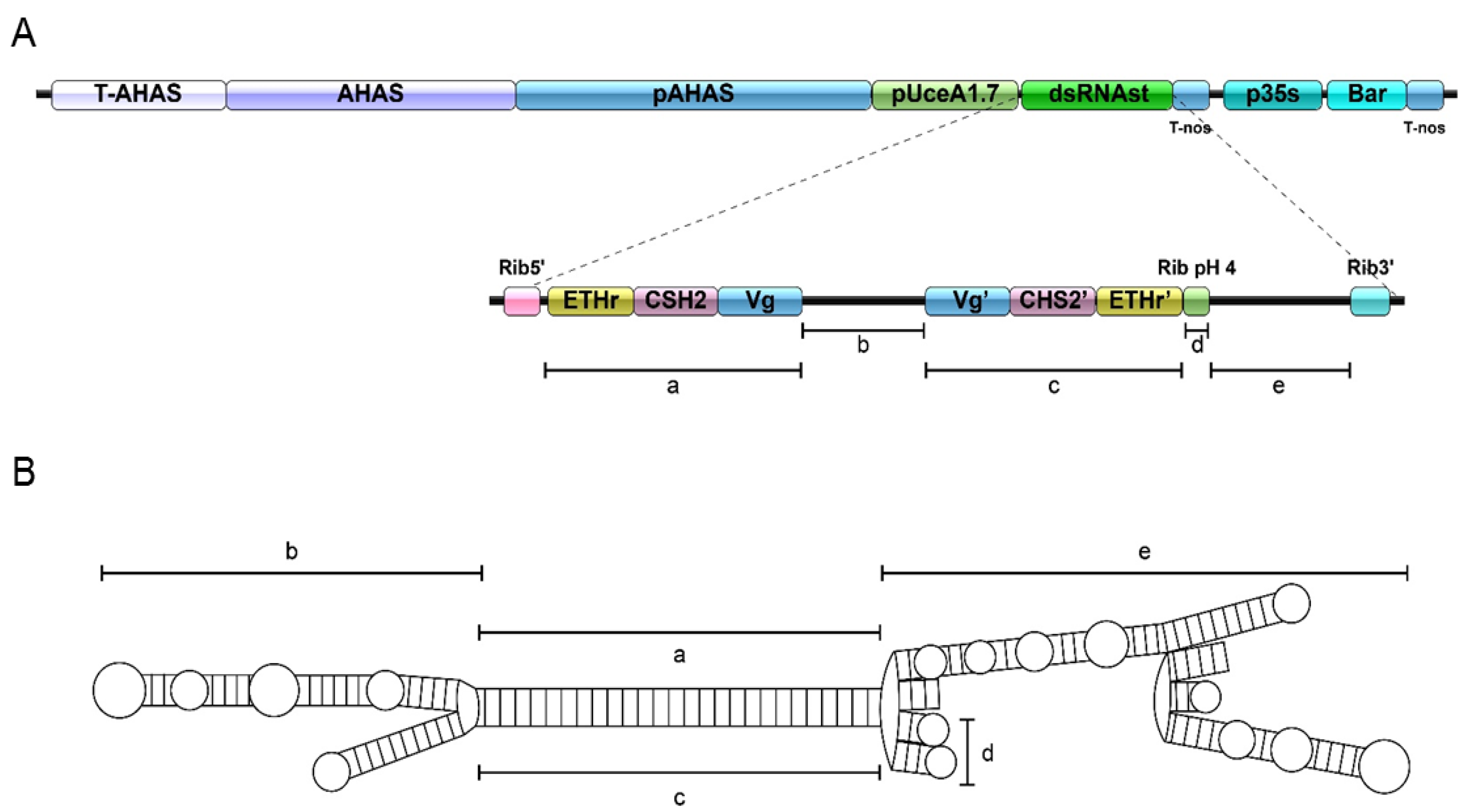
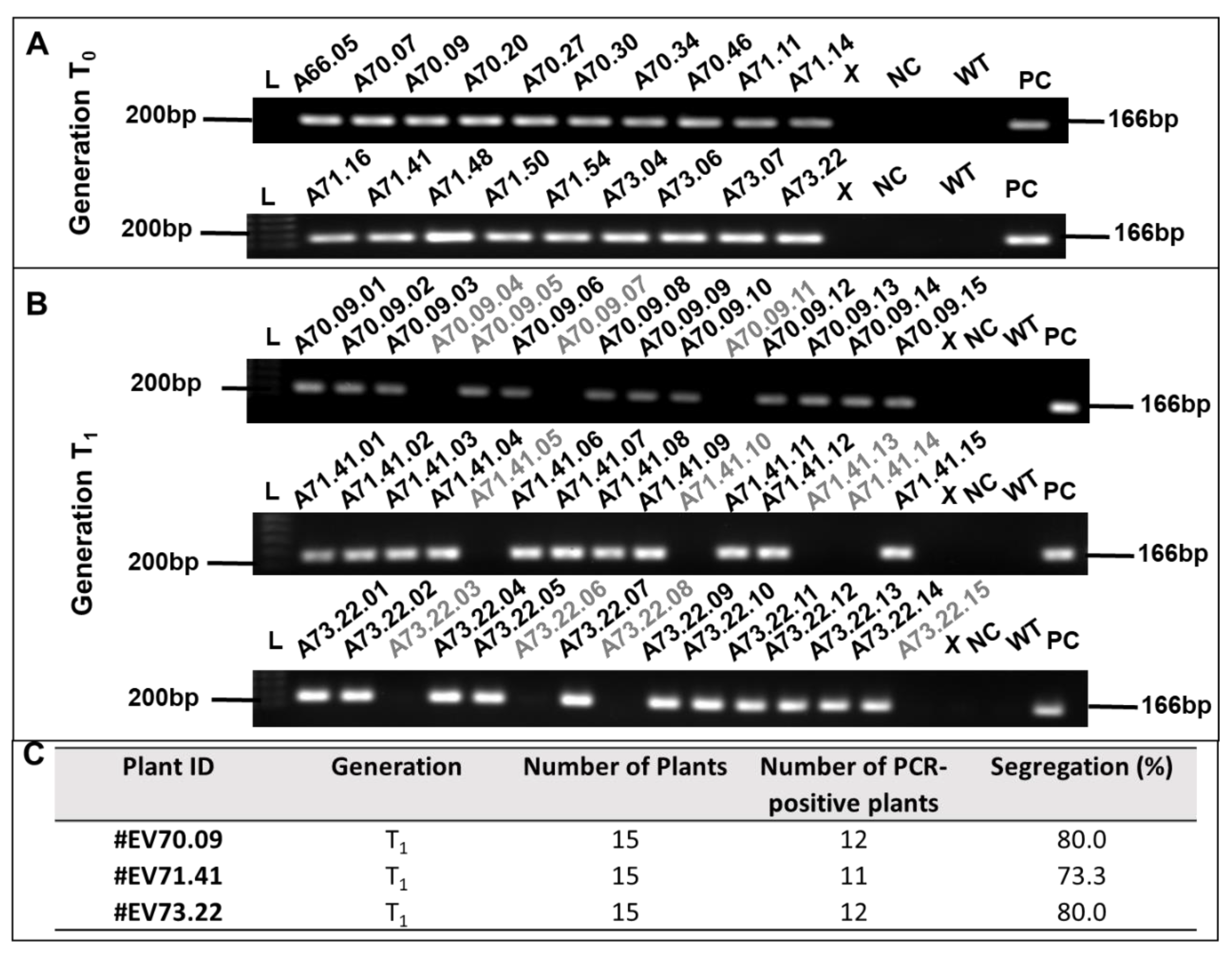
2.1. Generation of GM Cotton Lines
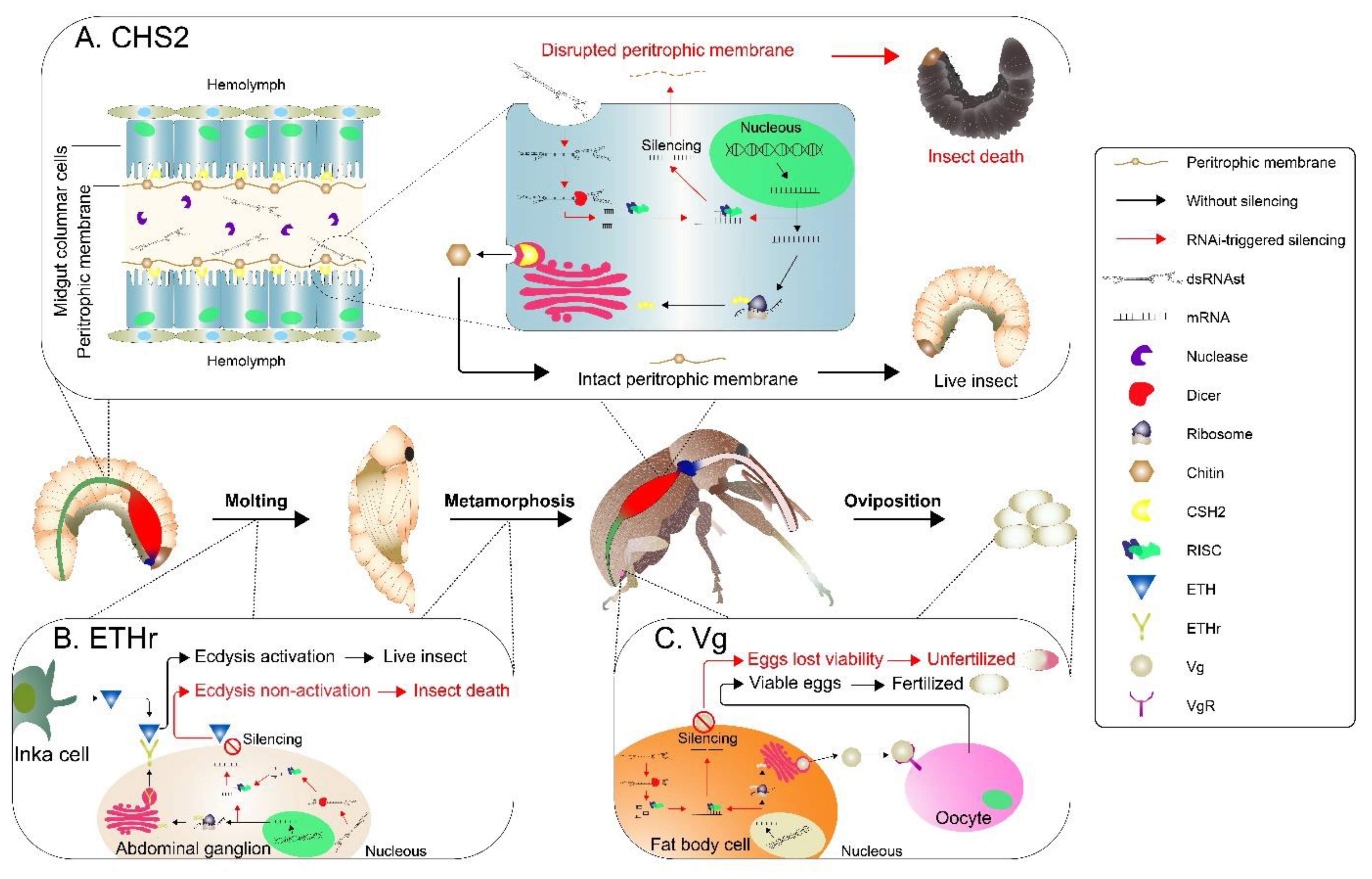
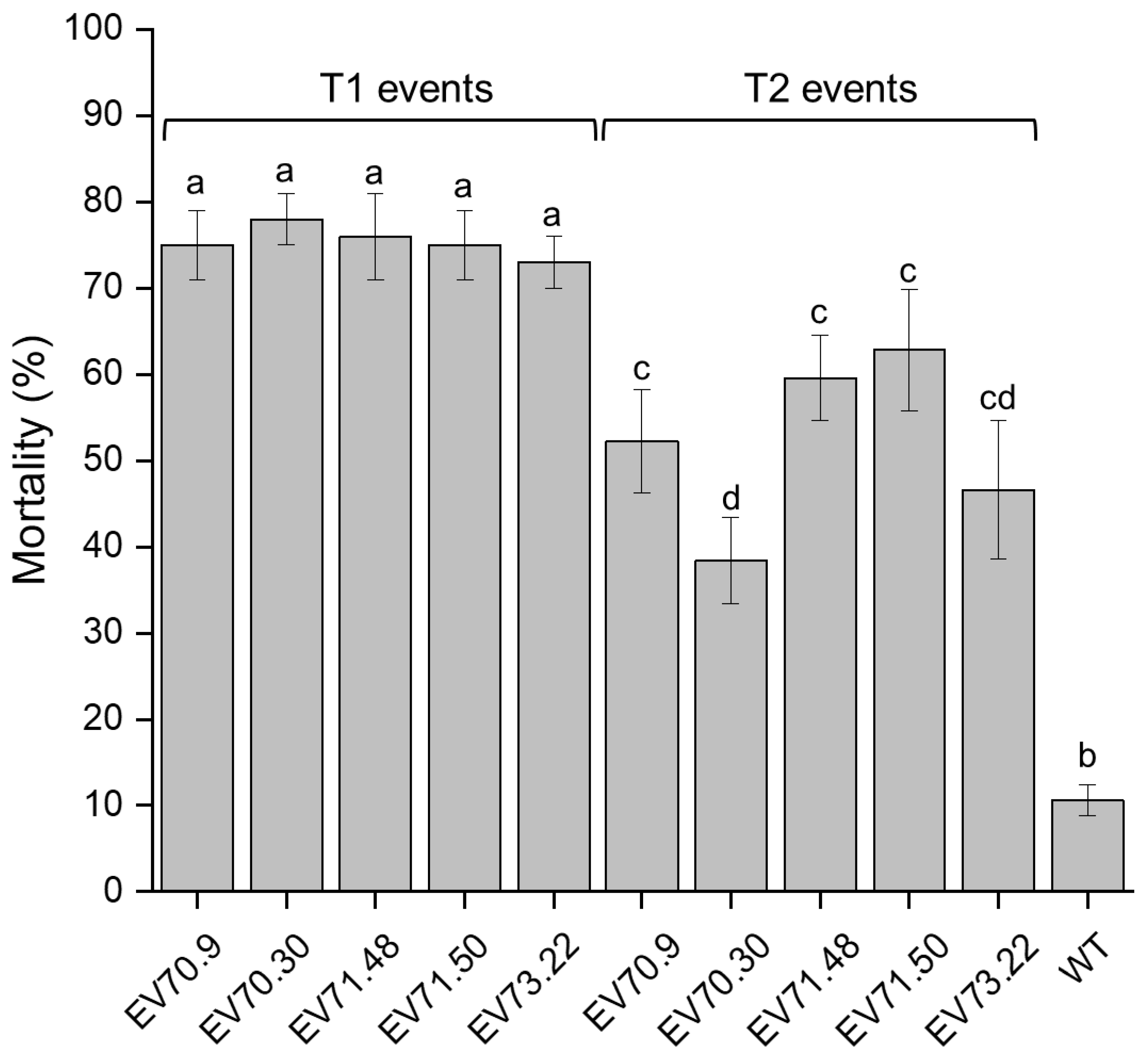
2.2. Knockdown of RNAi-Targeted Genes and Effects on Development
3. Discussion
4. Material and Methods
4.1. Construction of Transgenic Cotton
4.2. In Vitro Cotton Regeneration and Plant Acclimatization
4.3. DNA Extraction and PCR
4.4. Transgene Copy Number Estimation
4.5. Evaluation of the CBW’s Survival in Transgenic Cotton Plants
4.6. Expression Analysis of dsRNA Molecules
4.7. Validation of RNAi-Mediated Knockdown
4.8. Phenotypic Effects of dsRNA-Expressing Plants on CBW
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, M.A.; Wahid, A.; Ahmad, M.; Tahir, M.T.; Ahmed, M.; Ahmad, S.; Hasanuzzaman, M. World cotton production and consumption: An overview. In Cotton Production and Uses; Ahmad, S., Hasanuzzaman, M., Eds.; Springer: Singapore, 2020; pp. 1–7. [Google Scholar] [CrossRef]
- Hoffmann, L.V.; Kresic, I.B.; Paz, J.G.; Bela, D.A.; Tcach, N.E.; Lamas, F.M.; Sofiatti, V. Cotton production in Brazil and other South American countries. In Cotton Production; Jabran, K., Singh, B., Eds.; Wiley: Singapore, 2019; pp. 277–295. [Google Scholar] [CrossRef]
- Arruda, L.S.; Torres, J.B.; Rolim, G.G.; Silva-Torres, C.S.A. Dispersal of boll weevil toward and within the cotton plant and implications for insecticide exposure. Pest Manag. Sci. 2021, 77, 1339–1347. [Google Scholar] [CrossRef]
- Ribeiro, T.P.; Arraes, F.B.M.; Lourenço-Tessutti, I.T.; Silva, M.S.; Lisei-de-Sá, M.E.; Lucena, W.A.; Macedo, L.L.P.; Lima, J.N.; Santos Amorim, R.M.; Artico, S.; et al. Transgenic cotton expressing Cry10Aa toxin confers high resistance to the cotton boll weevil. Plant Biotechnol. J. 2017, 15, 997–1009. [Google Scholar] [CrossRef]
- Ribeiro, T.P.; Lourenço-Tessutti, I.T.; de Melo, B.P.; Morgante, C.V.; Filho, A.S.; Lins, C.B.J.; Ferreira, G.F.; Mello, G.N.; Macedo, L.L.P.; Lucena, W.A.; et al. Improved cotton transformation protocol mediated by Agrobacterium and biolistic combined-methods. Planta 2021, 254, 20. [Google Scholar] [CrossRef]
- Ni, M.; Ma, W.; Wang, X.; Gao, M.; Dai, Y.; Wei, X.; Zhang, L.; Peng, Y.; Chen, S.; Ding, L.; et al. Next-generation transgenic cotton: Pyramiding RNAi and Bt counters insect resistance. Plant Biotechnol. J. 2017, 15, 1204–1213. [Google Scholar] [CrossRef] [PubMed]
- Mezzetti, B.; Smagghe, G.; Arpaia, S.; Christiaens, O.; Dietz-Pfeilstetter, A.; Jones, H.; Kostov, K.; Sabbadini, S.; Opsahl-Sorteberg, H.-G.; Ventura, V.; et al. RNAi: What is its position in agriculture? J. Pest Sci. 2020, 93, 1125–1130. [Google Scholar] [CrossRef]
- Price, D.R.G.; Gatehouse, J.A. RNAi-mediated crop protection against insects. Trends Biotechnol. 2008, 26, 393–400. [Google Scholar] [CrossRef]
- Baum, J.A.; Bogaert, T.; Clinton, W.; Heck, G.R.; Feldmann, P.; Ilagan, O.; Johnson, S.; Plaetinck, G.; Munyikwa, T.; Pleau, M.; et al. Control of coleopteran insect pests through RNA interference. Nat. Biotechnol. 2007, 25, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- Huvenne, H.; Smagghe, G. Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: A review. J. Insect Physiol. 2010, 56, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Arraes, F.B.M.; Martins-de-Sa, D.; Noriega Vasquez, D.D.; Melo, B.P.; Faheem, M.; de Macedo, L.L.P.; Morgante, C.V.; Barbosa, J.A.R.G.; Togawa, R.C.; Moreira, V.J.V.; et al. Dissecting protein domain variability in the core RNA interference machinery of five insect orders. RNA Biol. 2020, 18, 1653–1681. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Richtman, N.M.; Zhao, J.-Z.; Duncan, K.E.; Niu, X.; Procyk, L.A.; Oneal, M.A.; Kernodle, B.M.; Steimel, J.P.; Crane, V.C.; et al. Discovery of midgut genes for the RNA interference control of corn rootworm. Sci. Rep. 2016, 6, 30542. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Kassa, A.; Hu, X.; Robeson, J.; McMahon, M.; Richtman, N.M.; Steimel, J.P.; Kernodle, B.M.; Crane, V.C.; Sandahl, G.; et al. Control of western corn rootworm (Diabrotica virgifera virgifera) reproduction through plant-mediated RNA interference. Sci. Rep. 2017, 7, 12591. [Google Scholar] [CrossRef] [PubMed]
- Knorr, E.; Fishilevich, E.; Tenbusch, L.; Frey, M.L.F.; Rangasamy, M.; Billion, A.; Worden, S.E.; Gandra, P.; Arora, K.; Lo, W.; et al. Gene silencing in Tribolium castaneum as a tool for the targeted identification of candidate RNAi targets in crop pests. Sci. Rep. 2018, 8, 2061. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Aksoy, E.; Çalışkan, M.E.; Bakhsh, A. Transgenic potato lines expressing hairpin RNAi construct of molting-associated EcR gene exhibit enhanced resistance against Colorado potato beetle (Leptinotarsa decemlineata, Say). Transgenic Res. 2019, 28, 151–164. [Google Scholar] [CrossRef]
- Mohan, C.; Shibao, P.Y.T.; de Paula, F.F.P.; Toyama, D.; Vieira, M.A.S.; Figueira, A.; Scotton, D.; Soares-Costa, A.; Henrique-Silva, F. hRNAi-mediated knock-down of Sphenophorus levis V-ATPase E in transgenic sugarcane (Saccharum spp. interspecific hybrid) affects the insect growth and survival. Plant Cell Rep. 2021, 40, 507–516. [Google Scholar] [CrossRef]
- Bonfim, K.; Faria, J.C.; Nogueira, E.O.; Mendes, É.A.; Aragão, F.J. RNAi-mediated resistance to Bean golden mosaic virus in genetically engineered common bean (Phaseolus vulgaris). Mol. Plant-Microbe Interact. 2007, 20, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.B.; Monteiro, T.R.; Cabral, G.B.; Aragão, F.J. RNAi-mediated resistance to whitefly (Bemisia tabaci) in genetically engineered lettuce (Lactuca sativa). Transgenic Res. 2017, 26, 613–624. [Google Scholar] [CrossRef] [PubMed]
- Cagliari, D.; Dias, N.P.; Galdeano, D.M.; Dos Santos, E.A.; Smagghe, G.; Zotti, M.J. Management of pest insects and plant diseases by non-transformative RNAi. Front. Plant Sci. 2019, 10, 1319. [Google Scholar] [CrossRef] [PubMed]
- Bento, F.M.; Marques, R.N.; Campana, F.B.; Demétrio, C.G.; Leandro, R.A.; Parra, J.R.P.; Figueira, A. Gene silencing by RNAi via oral delivery of dsRNA by bacteria in the South American tomato pinworm, Tuta absoluta. Pest Manag. Sci. 2020, 76, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Camargo, R.A.; Barbosa, G.O.; Possignolo, I.P.; Peres, L.E.; Lam, E.; Lima, J.E.; Figueira, A.; Marques-Souza, H. RNA interference as a gene silencing tool to control Tuta absoluta in tomato (Solanum lycopersicum). PeerJ 2016, 4, e2673. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Pinto, C.E.; Coelho, R.R.; Leite, A.G.; Silveira, D.A.; de Souza, D.A.; Lopes, R.B.; Macedo, L.L.; Silva, M.C.; Ribeiro, T.P.; Morgante, C.V.; et al. Increasing Anthonomus grandis susceptibility to Metarhizium anisopliae through RNAi-induced AgraRelish knockdown: A perspective to combine biocontrol and biotechnology. Pest Manag. Sci. 2021, 77, 4054–4063. [Google Scholar] [CrossRef] [PubMed]
- Noriega, D.D.; Arraes, F.; Antonino, J.D.; Macedo, L.L.; Fonseca, F.C.; Togawa, R.C.; Grynberg, P.; Silva, M.; Negrisoli, A.S.; Grossi-de-Sa, M.F. Transcriptome analysis and knockdown of the juvenile hormone esterase gene reveal abnormal feeding behavior in the sugarcane giant borer. Front. Physiol. 2020, 11, 588450. [Google Scholar] [CrossRef]
- Firmino, A.A.P.; Fonseca, F.C.d.A.; de Macedo, L.L.P.; Coelho, R.R.; Antonino de Souza, J.D., Jr.; Togawa, R.C.; Silva-Junior, O.B.; Pappas, G.J., Jr.; da Silva, M.C.M.; Engler, G.; et al. Transcriptome analysis in cotton boll weevil (Anthonomus grandis) and RNA interference in insect pests. PLoS ONE 2013, 8, e85079. [Google Scholar] [CrossRef] [PubMed]
- Firmino, A.A.P.; Pinheiro, D.H.; Moreira-Pinto, C.E.; Antonino, J.D.; Macedo, L.L.P.; Martins-de-Sa, D.; Arraes, F.B.M.; Coelho, R.R.; Fonseca, F.C.d.A.; Silva, M.C.M.; et al. RNAi-mediated suppression of Laccase2 impairs cuticle tanning and molting in the cotton boll weevil (Anthonomus grandis). Front. Physiol. 2020, 11, 591569. [Google Scholar] [CrossRef] [PubMed]
- Gillet, F.-X.; Garcia, R.A.; Macedo, L.L.; Albuquerque, E.V.; Silva, M.; Grossi-de-Sa, M.F. Investigating engineered ribonucleoprotein particles to improve oral RNAi delivery in crop insect pests. Front. Physiol. 2017, 8, 256. [Google Scholar] [CrossRef] [PubMed]
- Alamalakala, L.; Parimi, S.; Patel, N.; Char, B. Insect RNAi: Integrating a new tool in the crop protection toolkit. In Trends in Insect Molecular Biology and Biotechnology; Kumar, D., Gong, C., Eds.; Springer: Cham, Switzerland, 2017; pp. 193–232. [Google Scholar] [CrossRef]
- Liu, S.; Jaouannet, M.; Dempsey, D.M.A.; Imani, J.; Coustau, C.; Kogel, K.-H. RNA-based technologies for insect control in plant production. Biotechnol. Adv. 2020, 39, 107463. [Google Scholar] [CrossRef]
- Almeida Garcia, R.; Lima Pepino Macedo, L.; do Nascimento, D.C.; Gillet, F.-X.; Moreira-Pinto, C.E.; Faheem, M.; Moreschi Basso, A.M.; Mattar Silva, M.C.; Grossi-de-Sa, M.F. Nucleases as a barrier to gene silencing in the cotton boll weevil, Anthonomus grandis. PLoS ONE 2017, 12, e0189600. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Khan, S.A.; Heckel, D.G.; Bock, R. Next-generation insect-resistant plants: RNAi-mediated crop protection. Trends Biotechnol. 2017, 35, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Gordon, K.H.J.; Waterhouse, P.M. RNAi for insect-proof plants. Nat. Biotechnol. 2007, 25, 1231–1232. [Google Scholar] [CrossRef] [PubMed]
- Ding, B. The biology of viroid-host interactions. Annu. Rev. Phytopathol. 2009, 47, 105–131. [Google Scholar] [CrossRef] [PubMed]
- Macedo, L.L.P.; Grossi-de-Sa, M.F.; Mattar Silva, M.C.; Almeida Garcia, R.; Godinho, R.A.M.; Albuquerque, E.V.S. Aumento da Eficácia de Supressão de Expressão de Genes por meio do uso de Moléculas de RNA com Estrutura. Estabilizada. Patent Number: BR 10 2017 006904 4, 4 March 2017. [Google Scholar]
- Jayasena, V.K.; Gold, L. In vitro selection of self-cleaving RNAs with a low pH optimum. Proc. Natl. Acad. Sci. USA 1997, 94, 10612–10617. [Google Scholar] [CrossRef] [PubMed]
- Hammann, C.; Luptak, A.; Perreault, J.; de la Peña, M. The ubiquitous hammerhead ribozyme. RNA 2012, 18, 871–885. [Google Scholar] [CrossRef]
- Silver, K.; Cooper, A.M.W.; Zhu, K.Y. Strategies for enhancing the efficiency of RNA interference in insects. Pest Manag. Sci. 2021, 77, 2645–2658. [Google Scholar] [CrossRef] [PubMed]
- Coelho, R.R.; de Souza Júnior, J.D.A.; Firmino, A.A.P.; de Macedo, L.L.P.; Fonseca, F.C.A.; Terra, W.R.; Engler, G.; de Almeida Engler, J.; da Silva, M.C.M.; Grossi-de-Sa, M.F. Vitellogenin knockdown strongly affects cotton boll weevil egg viability but not the number of eggs laid by females. Meta Gene 2016, 9, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Macedo, L.L.P.; Antonino de Souza Junior, J.D.; Coelho, R.R.; Fonseca, F.C.A.; Firmino, A.A.P.; Silva, M.C.M.; Fragoso, R.R.; Albuquerque, E.V.S.; Silva, M.S.; de Almeida Engler, J.; et al. Knocking down chitin synthase 2 by RNAi is lethal to the cotton boll weevil. Biotechnol. Res. Innov. 2017, 1, 72–86. [Google Scholar] [CrossRef]
- Merzendorfer, H. Insect chitin synthases: A review. J. Comp. Physiol. B 2006, 176, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Merzendorfer, H. Chitin synthesis inhibitors: Old molecules and new developments. Insect Sci. 2012, 20, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Mysore, K.; Flannery, E.; Michel, K.; Severson, D.W.; Zhu, K.Y.; Duman-Scheel, M. Chitosan/interfering RNA nanoparticle mediated gene silencing in disease vector mosquito larvae. J. Vis. Exp. JoVE 2015, 97, e52523. [Google Scholar] [CrossRef] [PubMed]
- Christiaens, O.; Tardajos, M.G.; Martinez Reyna, Z.L.; Dash, M.; Dubruel, P.; Smagghe, G. Increased RNAi efficacy in Spodoptera exigua via the formulation of dsRNA with guanylated polymers. Front. Physiol. 2018, 9, 316. [Google Scholar] [CrossRef]
- Shao, Z.-M.; Li, Y.-J.; Ding, J.-H.; Liu, Z.-X.; Zhang, X.-R.; Wang, J.; Sheng, S.; Wu, F.-A. Identification, characterization, and functional analysis of chitin synthase genes in Glyphodes pyloalis Walker (Lepidoptera: Pyralidae). Int. J. Mol. Sci. 2020, 21, 4656. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Du, M.-y.; Wang, G.-x.; Wang, Z.-y.; Lu, Y.-j. Identification, mRNA expression, and functional analysis of chitin synthase 2 gene in the rusty grain beetle, Cryptolestes ferrugineus. J. Stored Prod. Res. 2020, 87, 101622. [Google Scholar] [CrossRef]
- Zhang, Z.-J.; Xia, L.; Du, J.; Li, S.-W.; Zhao, F. Cloning, characterization, and RNAi effect of the chitin synthase B gene in Cnaphalocrocis medinalis. J. Asia-Pac. Entomol. 2021, 24, 486–492. [Google Scholar] [CrossRef]
- Hagedorn, H.H.; Kunkel, J.G. Vitellogenin and vitellin in insects. Annu. Rev. Entomol. 1979, 24, 475–505. [Google Scholar] [CrossRef]
- Sappington, T.W.; Raikhel, A.S. Molecular characteristics of insect vitellogenins and vitellogenin receptors. Insect Biochem. Mol. Biol. 1998, 28, 277–300. [Google Scholar] [CrossRef]
- Veerana, M.; Kubera, A.; Ngernsiri, L. Analysis of the vitellogenin gene of rice moth, Corcyra cephalonica Stainton. Arch. Insect Biochem. Physiol. 2014, 87, 126–147. [Google Scholar] [CrossRef] [PubMed]
- Shang, F.; Niu, J.Z.; Ding, B.Y.; Zhang, Q.; Ye, C.; Zhang, W.; Smagghe, G.; Wang, J.J. Vitellogenin and its receptor play essential roles in the development and reproduction of the brown citrus aphid, Aphis (Toxoptera) citricidus. Insect Mol. Biol. 2018, 27, 221–233. [Google Scholar] [CrossRef]
- Liang, C.; Liu, T.-h.; Han, S.-p.; He, Y.-z. Molecular cloning, expression profiling and RNA interference of a vitellogenin gene from Harmonia axyridis (Coleoptera: Coccinellidae). J. Integr. Agric. 2019, 18, 2311–2320. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.; Liu, Y.; Zhao, M.; Jin, J.; Zhou, Z.; Guo, J. Identification and expression patterns of three vitellogenin genes and their roles in reproduction of the alligatorweed flea beetle Agasicles hygrophila (Coleoptera: Chrysomelidae). Front. Physiol. 2019, 10, 368. [Google Scholar] [CrossRef] [PubMed]
- Husain, M.; Rasool, K.G.; Tufail, M.; Alwaneen, W.S.; Aldawood, A.S. RNAi-mediated silencing of vitellogenin gene curtails oogenesis in the almond moth Cadra cautella. PLoS ONE 2021, 16, e0245928. [Google Scholar] [CrossRef]
- Roller, L.; Zitnanová, I.; Dai, L.; Simo, L.; Park, Y.; Satake, H.; Tanaka, Y.; Adams, M.E.; Zitnan, D. Ecdysis triggering hormone signaling in arthropods. Peptides 2010, 31, 429–441. [Google Scholar] [CrossRef] [PubMed]
- Arakane, Y.; Li, B.; Muthukrishnan, S.; Beeman, R.W.; Kramer, K.J.; Park, Y. Functional analysis of four neuropeptides, EH, ETH, CCAP and bursicon, and their receptors in adult ecdysis behavior of the red flour beetle, Tribolium castaneum. Mech. Dev. 2008, 125, 984–995. [Google Scholar] [CrossRef]
- Shi, Y.; Jiang, H.-B.; Gui, S.-H.; Liu, X.-Q.; Pei, Y.-X.; Xu, L.; Smagghe, G.; Wang, J.-J. Ecdysis triggering hormone signaling (ETH/ETHR-A) is required for the larva-larva ecdysis in Bactrocera dorsalis (Diptera: Tephritidae). Front. Physiol. 2017, 8, 587. [Google Scholar] [CrossRef]
- Shi, Y.; Liu, T.-Y.; Jiang, H.-B.; Liu, X.-Q.; Dou, W.; Park, Y.; Smagghe, G.; Wang, J.-J. The ecdysis triggering hormone system, via ETH/ETHR-B, is essential for successful reproduction of a major pest insect, Bactrocera dorsalis (Hendel). Front. Physiol. 2019, 10, 151. [Google Scholar] [CrossRef]
- Shen, C.-H.; Xu, Q.-Y.; Fu, K.-Y.; Guo, W.-C.; Jin, L.; Li, G.-Q. Two splice isoforms of Leptinotarsa ecdysis triggering hormone receptor have distinct roles in larva-pupa transition. Front. Physiol. 2020, 11, 593962. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.H.; Xu, Q.Y.; Fu, K.Y.; Guo, W.C.; Jin, L.; Li, G.Q. Ecdysis triggering hormone is essential for larva-pupa-adult transformation in Leptinotarsa decemlineata. Insect Mol. Biol. 2021, 30, 241–252. [Google Scholar] [CrossRef]
- Borges, A.G. Novas Biomoléculas Potencialmente Aplicadas No Controle de Anthonomus grandis, via RNA Interferente. Ph.D. Thesis, University of Brasilia, Brasilia, Brazil, 2020. [Google Scholar]
- Vatanparast, M.; Kazzazi, M.; Mirzaie-asl, A.; Hosseininaveh, V. RNA interference-mediated knockdown of some genes involved in digestion and development of Helicoverpa armigera. Bull. Entomol. Res. 2017, 107, 777–790. [Google Scholar] [CrossRef]
- Bai-Zhong, Z.; Xu, S.; Cong-Ai, Z.; Liu-Yang, L.; Ya-She, L.; Xing, G.; Dong-Mei, C.; Zhang, P.; Ming-Wang, S.; Xi-Ling, C. Silencing of cytochrome P450 in Spodoptera frugiperda (Lepidoptera: Noctuidae) by RNA interference enhances susceptibility to Chlorantraniliprole. J. Insect Sci. 2020, 20, 12. [Google Scholar] [CrossRef] [PubMed]
- Lü, J.; Liu, Z.Q.; Guo, W.; Guo, M.J.; Chen, S.M.; Yang, C.X.; Zhang, Y.J.; Pan, H.P. Oral delivery of dsHvlwr is a feasible method for managing the pest Henosepilachna vigintioctopunctata (Coleoptera: Coccinellidae). Insect Sci. 2021, 28, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-F.; Chen, Z.; Wang, X.-B.; Xu, J.; Chen, P.; Ye, H. Bacterial-mediated RNAi and functional analysis of Natalisin in a moth. Sci. Rep. 2021, 11, 4662. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Liu, Z.; Chen, J.; Sun, G.; Jiang, Y.; Li, M.; Xiong, L.; Chen, S.; Zhou, Y.; Asad, M.; et al. Silencing arginine kinase/integrin β1 subunit by transgenic plant expressing dsRNA inhibits the development and survival of Plutella xylostella. Pest Manag. Sci. 2020, 76, 1761–1771. [Google Scholar] [CrossRef] [PubMed]
- Arya, S.K.; Singh, S.; Upadhyay, S.K.; Tiwari, V.; Saxena, G.; Verma, P.C. RNAi-based gene silencing in Phenacoccus solenopsis and its validation by in planta expression of a double-stranded RNA. Pest Manag. Sci. 2021, 77, 1796–1805. [Google Scholar] [CrossRef] [PubMed]
- Bao, W.; Li, A.; Zhang, Y.; Diao, P.; Zhao, Q.; Yan, T.; Zhou, Z.; Duan, H.; Li, X.; Wuriyanghan, H. Improvement of host-induced gene silencing efficiency via polycistronic-tRNA-amiR expression for multiple target genes and characterization of RNAi mechanism in Mythimna separata. Plant Biotechnol. J. 2021, 19, 1370–1385. [Google Scholar] [CrossRef]
- Tian, G.; Cheng, L.; Qi, X.; Ge, Z.; Niu, C.; Zhang, X.; Jin, S. Transgenic cotton plants expressing double-stranded RNAs target HMG-CoA reductase (HMGR) gene inhibits the growth, development and survival of cotton bollworms. Int. J. Biol. Sci. 2015, 11, 1296–1305. [Google Scholar] [CrossRef]
- Han, Q.; Wang, Z.; He, Y.; Xiong, Y.; Lv, S.; Li, S.; Zhang, Z.; Qiu, D.; Zeng, H. Transgenic cotton plants expressing the HaHR3 gene conferred enhanced resistance to Helicoverpa armigera and improved cotton yield. Int. J. Mol. Sci. 2017, 18, 1874. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.-B.; Tao, X.-Y.; Xue, X.-Y.; Wang, L.-J.; Chen, X.-Y. Cotton plants expressing CYP6AE14 double-stranded RNA show enhanced resistance to bollworms. Transgenic Res. 2011, 20, 665–673. [Google Scholar] [CrossRef]
- Yoon, J.-S.; Gurusamy, D.; Palli, S.R. Accumulation of dsRNA in endosomes contributes to inefficient RNA interference in the fall armyworm, Spodoptera frugiperda. Insect Biochem. Mol. Biol. 2017, 90, 53–60. [Google Scholar] [CrossRef]
- Hu, Y.-B.; Dammer, E.B.; Ren, R.-J.; Wang, G. The endosomal-lysosomal system: From acidification and cargo sorting to neurodegeneration. Transl. Neurodegener. 2015, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bally, J.; Fishilevich, E.; Bowling, A.J.; Pence, H.E.; Narva, K.E.; Waterhouse, P.M. Improved insect-proofing: Expressing double-stranded RNA in chloroplasts. Pest Manag. Sci. 2018, 74, 1751–1758. [Google Scholar] [CrossRef]
- Bolognesi, R.; Ramaseshadri, P.; Anderson, J.; Bachman, P.; Clinton, W.; Flannagan, R.; Ilagan, O.; Lawrence, C.; Levine, S.; Moar, W. Characterizing the mechanism of action of double-stranded RNA activity against western corn rootworm (Diabrotica virgifera virgifera LeConte). PLoS ONE 2012, 7, e47534. [Google Scholar] [CrossRef]
- Li, H.; Khajuria, C.; Rangasamy, M.; Gandra, P.; Fitter, M.; Geng, C.; Woosely, A.; Hasler, J.; Schulenberg, G.; Worden, S. Long ds RNA but not si RNA initiates RNA i in western corn rootworm larvae and adults. J. Appl. Entomol. 2015, 139, 432–445. [Google Scholar] [CrossRef]
- Wang, Y. Current view and perspectives in viroid replication. Curr. Opin. Virol. 2021, 47, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.-F.; Mu, L.-L.; Chen, X.; Guo, W.-C.; Li, G.-Q. RNA interference of chitin synthase genes inhibits chitin biosynthesis and affects larval performance in Leptinotarsa decemlineata (Say). Int. J. Biol. Sci. 2016, 12, 1319–1331. [Google Scholar] [CrossRef]
- Ye, C.; Jiang, Y.-D.; An, X.; Yang, L.; Shang, F.; Niu, J.; Wang, J.-J. Effects of RNAi-based silencing of chitin synthase gene on moulting and fecundity in pea aphids (Acyrthosiphon pisum). Sci. Rep. 2019, 9, 3694. [Google Scholar] [CrossRef]
- Kato, N.; Mueller, C.R.; Fuchs, J.F.; Wessely, V.; Lan, Q.; Christensen, B.M. Regulatory mechanisms of chitin biosynthesis and roles of chitin in peritrophic matrix formation in the midgut of adult Aedes aegypti. Insect Biochem. Mol. Biol. 2006, 36, 1–9. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, H.; Li, S.; Zhu, K.Y.; Ma, E.; Zhang, J. Characterization of a midgut-specific chitin synthase gene (LmCHS2) responsible for biosynthesis of chitin of peritrophic matrix in Locusta migratoria. Insect Biochem. Mol. Biol. 2012, 42, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Mu, L.L.; Kang, W.N.; Ze, L.J.; Shen, C.H.; Jin, L.; Anjum, A.A.; Li, G.Q. RNA interference targeting ecdysone receptor blocks the larval–pupal transition in Henosepilachna vigintioctopunctata. Insect Sci. 2021, 28, 419–429. [Google Scholar] [CrossRef]
- Alagia, A.; Eritja, R. siRNA and RNAi optimization. WIREs RNA 2016, 7, 316–329. [Google Scholar] [CrossRef]
- Harwood, G.; Amdam, G. Vitellogenin in the honey bee midgut. Apidologie 2021, 52, 837–847. [Google Scholar] [CrossRef]
- Lee, J.B.; Park, K.-E.; Lee, S.A.; Jang, S.H.; Eo, H.J.; Am Jang, H.; Kim, C.-H.; Ohbayashi, T.; Matsuura, Y.; Kikuchi, Y. Gut symbiotic bacteria stimulate insect growth and egg production by modulating hexamerin and vitellogenin gene expression. Dev. Comp. Immunol. 2017, 69, 12–22. [Google Scholar] [CrossRef] [PubMed]
- He, Y.-Z.; Wang, Y.-M.; Yin, T.-Y.; Cuellar, W.J.; Liu, S.-S.; Wang, X.-W. Gut-expressed vitellogenin facilitates the movement of a plant virus across the midgut wall in its insect vector. mSystems 2021, 6, e0058121. [Google Scholar] [CrossRef]
- Kumar, P.; Pandit, S.S.; Baldwin, I.T. Tobacco rattle virus vector: A rapid and transient means of silencing Manduca sexta genes by plant mediated RNA interference. PLoS ONE 2012, 7, e31347. [Google Scholar] [CrossRef]
- Jin, S.; Singh, N.D.; Li, L.; Zhang, X.; Daniell, H. Engineered chloroplast dsRNA silences cytochrome p450 monooxygenase, V-ATPase and chitin synthase genes in the insect gut and disrupts Helicoverpa zea larval development and pupation. Plant Biotechnol. J. 2015, 13, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Zhang, P.; Liu, C.; Zeng, F. Co-silence of the coatomer β and V-ATPase A genes by siRNA feeding reduces larval survival rate and weight gain of cotton bollworm, Helicoverpa armigera. Pestic. Biochem. Physiol. 2015, 118, 71–76. [Google Scholar] [CrossRef]
- Sharath Chandra, G.; Asokan, R.; Manamohan, M.; Ellango, R.; Sharma, H.C.; Akbar, S.M.D.; Krishna Kumar, N.K. Double-stranded RNA-mediated suppression of trypsin-like serine protease (t-SP) triggers over-expression of another t-SP isoform in Helicoverpa armigera. Appl. Biochem. Biotechnol. 2018, 184, 746–761. [Google Scholar] [CrossRef]
- Meng, F.; Yang, M.; Li, Y.; Li, T.; Liu, X.; Wang, G.; Wang, Z.; Jin, X.; Li, W. Functional analysis of RNA interference-related soybean pod borer (Lepidoptera) genes based on transcriptome sequences. Front. Physiol. 2018, 9, 383. [Google Scholar] [CrossRef]
- Yu, X.; Killiny, N. RNA interference of two glutathione S-transferase genes, Diaphorina citri DcGSTe2 and DcGSTd1, increases the susceptibility of Asian citrus psyllid (Hemiptera: Liviidae) to the pesticides fenpropathrin and thiamethoxam. Pest Manag. Sci. 2018, 74, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Rankin, S.M.; Dossat, H.B.; Garcia, K.M. Effects of diet and mating status upon corpus allatum activity, oocyte growth, and salivary gland size in the ring-legged earwig. Entomol. Exp. Appl. 1997, 83, 31–40. [Google Scholar] [CrossRef]
- Parthasarathy, R.; Sun, Z.; Bai, H.; Palli, S.R. Juvenile hormone regulation of vitellogenin synthesis in the red flour beetle, Tribolium castaneum. Insect Biochem. Mol. Biol. 2010, 40, 405–414. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, L.; He, Q.; Zhou, S. Regulatory mechanisms of vitellogenesis in insects. Front. Cell Dev. Biol. 2021, 8, 593613. [Google Scholar] [CrossRef]
- Heard, E.; Martienssen, R.A. Transgenerational epigenetic inheritance: Myths and mechanisms. Cell 2014, 157, 95–109. [Google Scholar] [CrossRef]
- Mette, M.; Aufsatz, W.; Van der Winden, J.; Matzke, M.; Matzke, A. Transcriptional silencing and promoter methylation triggered by double-stranded RNA. EMBO J. 2000, 19, 5194–5201. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, T.; Miyasaka, H.; Ohama, T. Unstable RNAi effects through epigenetic silencing of an inverted repeat transgene in Chlamydomonas reinhardtii. Genetics 2008, 180, 1927–1944. [Google Scholar] [CrossRef] [PubMed]
- Aragão, F.J.L.; Sarokin, L.; Vianna, G.R.; Rech, E.L. Selection of transgenic meristematic cells utilizing a herbicidal molecule results in the recovery of fertile transgenic soybean [Glycine max (L.) Merril] plants at a high frequency. Theor. Appl. Genet. 2000, 101, 1–6. [Google Scholar] [CrossRef]
- Viana, A.A.B.; Fragoso, R.R.; Guimarães, L.M.; Pontes, N.; Oliveira-Neto, O.B.; Artico, S.; Nardeli, S.M.; Alves-Ferreira, M.; Batista, J.A.N.; Silva, M.C.M.; et al. Isolation and functional characterization of a cotton ubiquitination-related promoter and 5’UTR that drives high levels of expression in root and flower tissues. BMC Biotechnol. 2011, 11, 115. [Google Scholar] [CrossRef] [PubMed]
- Yi, C.; Hong, Y. Estimating the copy number of transgenes in transformed cotton by real-time quantitative PCR. Methods Mol. Biol. 2019, 1902, 137–157. [Google Scholar] [CrossRef]
- Paes de Melo, B.; Lourenço-Tessutti, I.T.; Morgante, C.V.; Santos, N.C.; Pinheiro, L.B.; de Jesus Lins, C.B.; Silva, M.C.M.; Macedo, L.L.P.; Fontes, E.P.B.; Grossi-de-Sa, M.F. Soybean embryonic axis transformation: Combining biolistic and Agrobacterium-mediated protocols to overcome typical complications of in vitro plant regeneration. Front. Plant Sci. 2020, 11, 1228. [Google Scholar] [CrossRef]
- Zhao, S.; Fernald, R.D. Comprehensive algorithm for quantitative real-time polymerase chain reaction. J. Comput. Biol. A J. Comput. Mol. Cell Biol. 2005, 12, 1047–1064. [Google Scholar] [CrossRef]
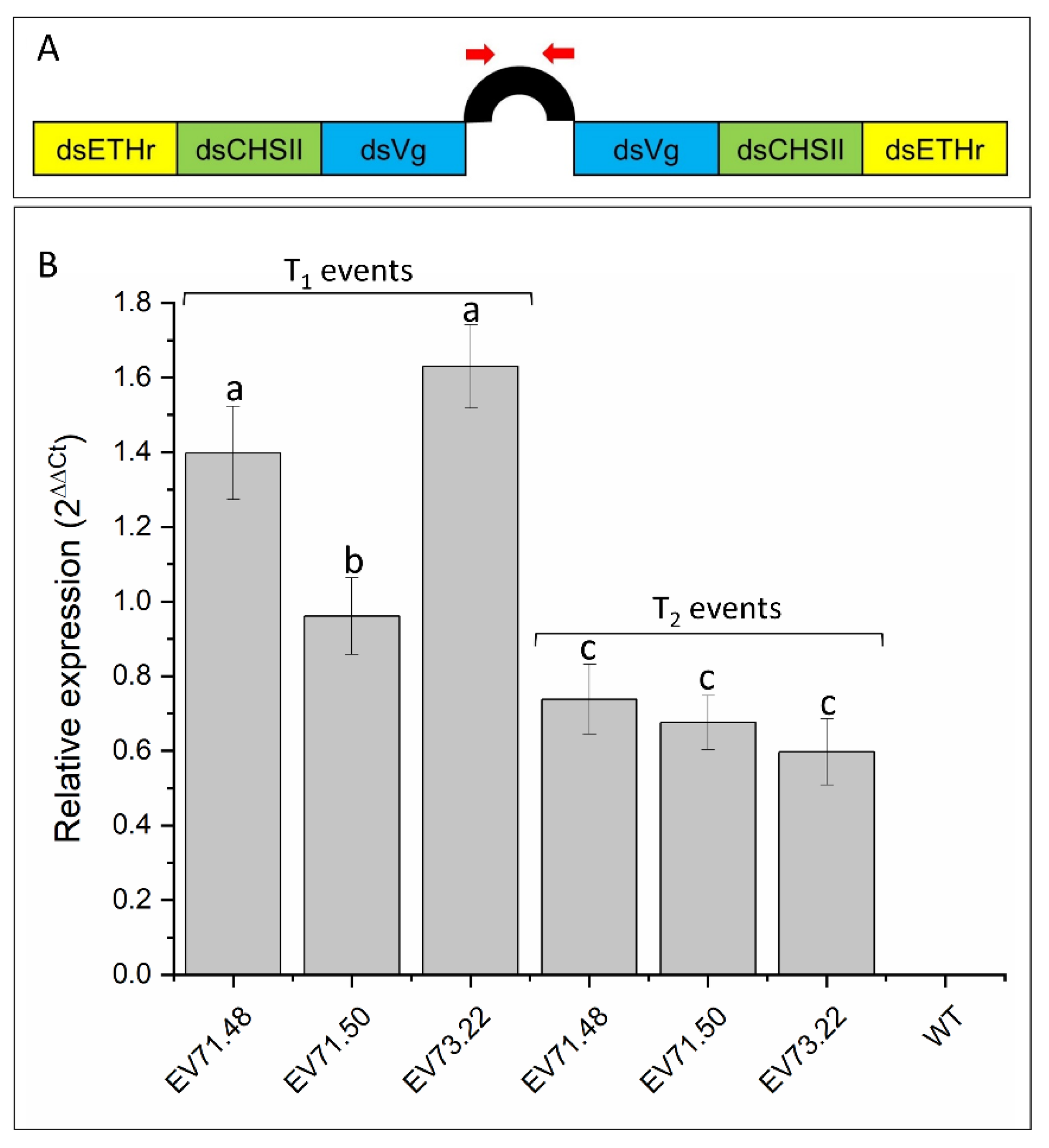
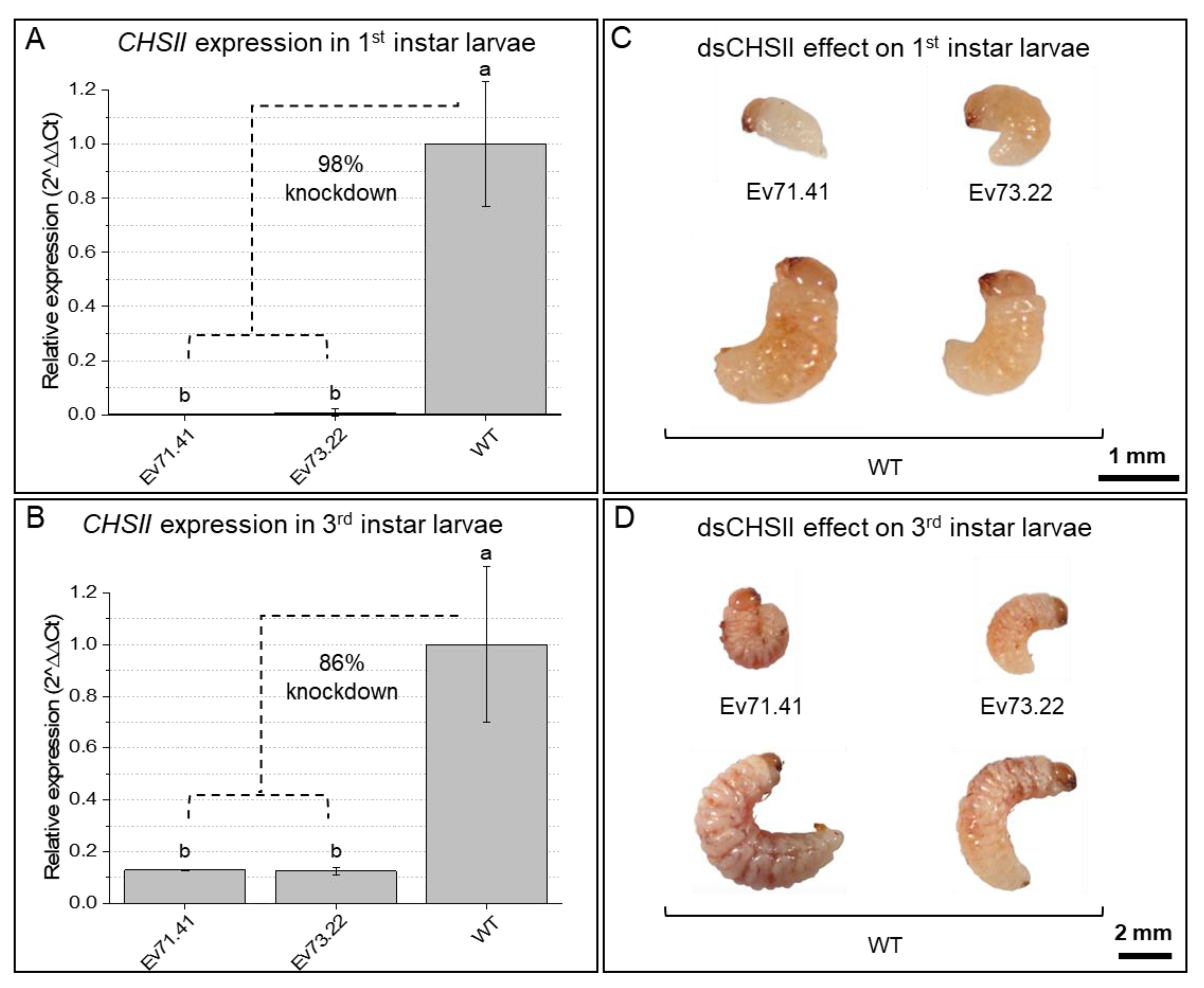

| #Transformation Experiments ID | Number of Inoculated Embryos | Number of Transformed Plants | Regeneration Efficiency | Number of PCR-Positive T0 Plants | Transformation Efficiency (PCR-Positive T0) |
|---|---|---|---|---|---|
| A66 | 550 | 10 | 1.82 | 1 | 10.0 |
| A70 | 550 | 50 | 9.09 | 7 | 14.0 |
| A71 | 550 | 55 | 10.00 | 7 | 12.7 |
| A73 | 550 | 35 | 6.36 | 4 | 11.4 |
| Total | 2200 | 150 | 6.82 (±3.67) | 19 | 12.07 (±1.71) |
| Plant ID | 2xAHAS/UBC1 | Estimated Copy Number |
|---|---|---|
| Ev70.09.18.02 | 1.53 | 2 |
| Ev70.09.18.05 | 1.49 | 2 |
| Ev70.09.18.08 | 1.52 | 2 |
| Ev70.09.18.13 | 1.52 | 2 |
| Ev70.09.18.17 | 1.49 | 2 |
| Ev71.41.01.01 | 1.36 | 1 |
| Ev71.41.01.03 | 1.23 | 1 |
| Ev71.41.01.08 | 1.37 | 1 |
| Ev71.41.01.10 | 1.40 | 1 |
| Ev71.41.01.12 | 0.79 | 1 |
| Ev71.48.07.01 | 1.54 | 2 |
| Ev71.48.07.02 | 1.64 | 2 |
| Ev71.48.07.08 | 1.49 | 2 |
| Ev71.48.07.11 | 1.54 | 2 |
| Ev71.48.07.14 | 2.11 | 2 |
| Ev73.22.12.02 | 1.51 | 2 |
| Ev73.22.12.05 | 1.50 | 2 |
| Ev73.22.12.08 | 1.54 | 2 |
| Ev73.22.12.16 | 1.55 | 2 |
| Ev73.22.12.19 | 1.70 | 2 |
| WT | −0.16 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, T.P.; Vasquez, D.D.N.; Macedo, L.L.P.; Lourenço-Tessutti, I.T.; Valença, D.C.; Oliveira-Neto, O.B.; Paes-de-Melo, B.; Rodrigues-Silva, P.L.; Firmino, A.A.P.; Basso, M.F.; et al. Stabilized Double-Stranded RNA Strategy Improves Cotton Resistance to CBW (Anthonomus grandis). Int. J. Mol. Sci. 2022, 23, 13713. https://doi.org/10.3390/ijms232213713
Ribeiro TP, Vasquez DDN, Macedo LLP, Lourenço-Tessutti IT, Valença DC, Oliveira-Neto OB, Paes-de-Melo B, Rodrigues-Silva PL, Firmino AAP, Basso MF, et al. Stabilized Double-Stranded RNA Strategy Improves Cotton Resistance to CBW (Anthonomus grandis). International Journal of Molecular Sciences. 2022; 23(22):13713. https://doi.org/10.3390/ijms232213713
Chicago/Turabian StyleRibeiro, Thuanne P., Daniel D. N. Vasquez, Leonardo L. P. Macedo, Isabela T. Lourenço-Tessutti, David C. Valença, Osmundo B. Oliveira-Neto, Bruno Paes-de-Melo, Paolo L. Rodrigues-Silva, Alexandre A. P. Firmino, Marcos F. Basso, and et al. 2022. "Stabilized Double-Stranded RNA Strategy Improves Cotton Resistance to CBW (Anthonomus grandis)" International Journal of Molecular Sciences 23, no. 22: 13713. https://doi.org/10.3390/ijms232213713
APA StyleRibeiro, T. P., Vasquez, D. D. N., Macedo, L. L. P., Lourenço-Tessutti, I. T., Valença, D. C., Oliveira-Neto, O. B., Paes-de-Melo, B., Rodrigues-Silva, P. L., Firmino, A. A. P., Basso, M. F., Lins, C. B. J., Neves, M. R., Moura, S. M., Tripode, B. M. D., Miranda, J. E., Silva, M. C. M., & Grossi-de-Sa, M. F. (2022). Stabilized Double-Stranded RNA Strategy Improves Cotton Resistance to CBW (Anthonomus grandis). International Journal of Molecular Sciences, 23(22), 13713. https://doi.org/10.3390/ijms232213713









