Acetylation Enhances the Anticancer Activity and Oral Bioavailability of 5-Demethyltangeretin
Abstract
1. Introduction
2. Results and Discussion
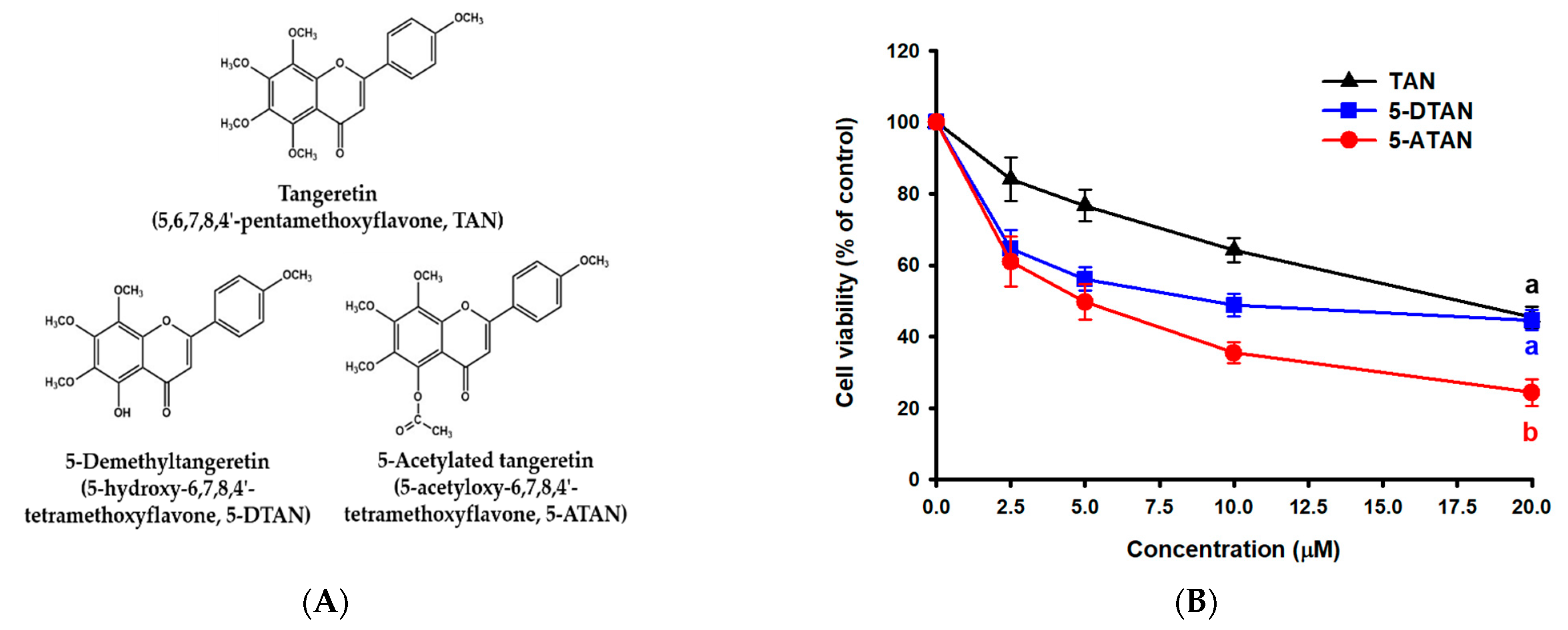
2.1. Effects of TAN, 5-DTAN, and 5-ATAN on Cell Proliferation of PC-3 Human Prostate Cancer Cells
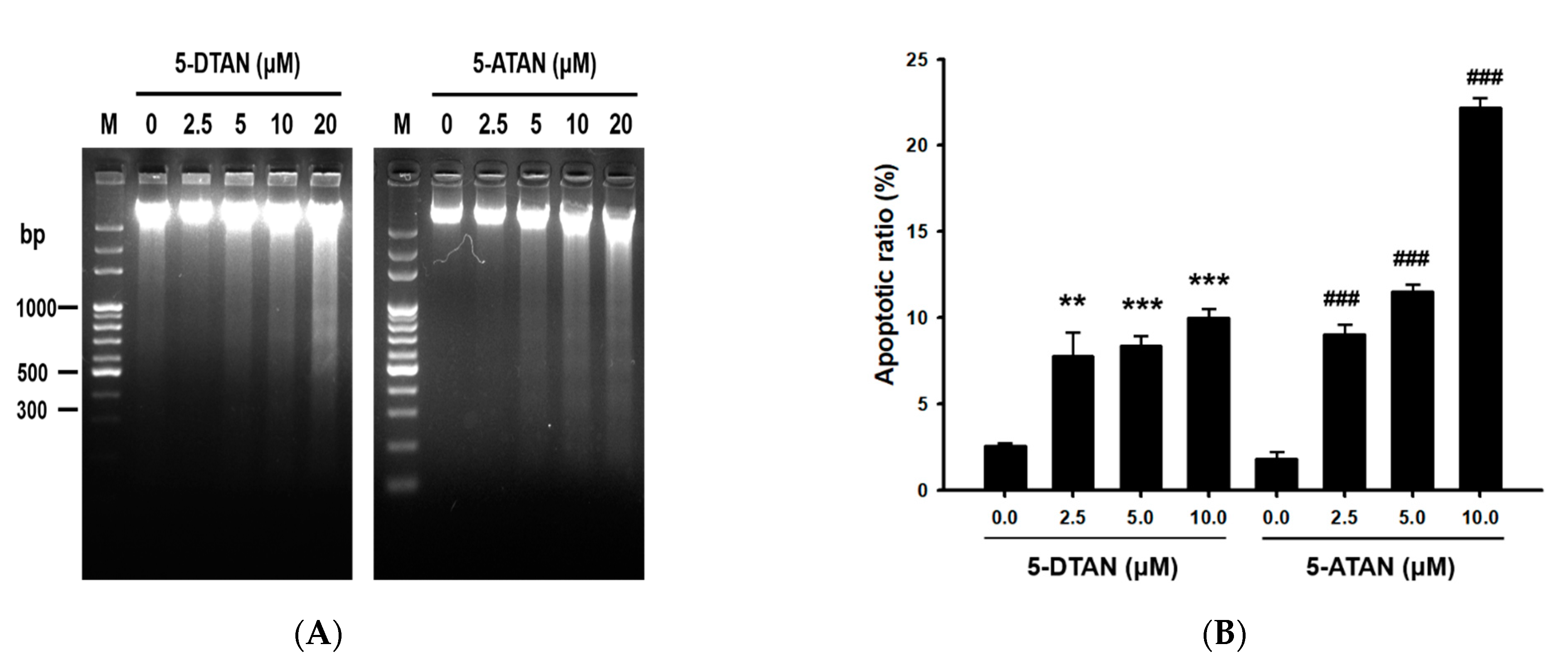
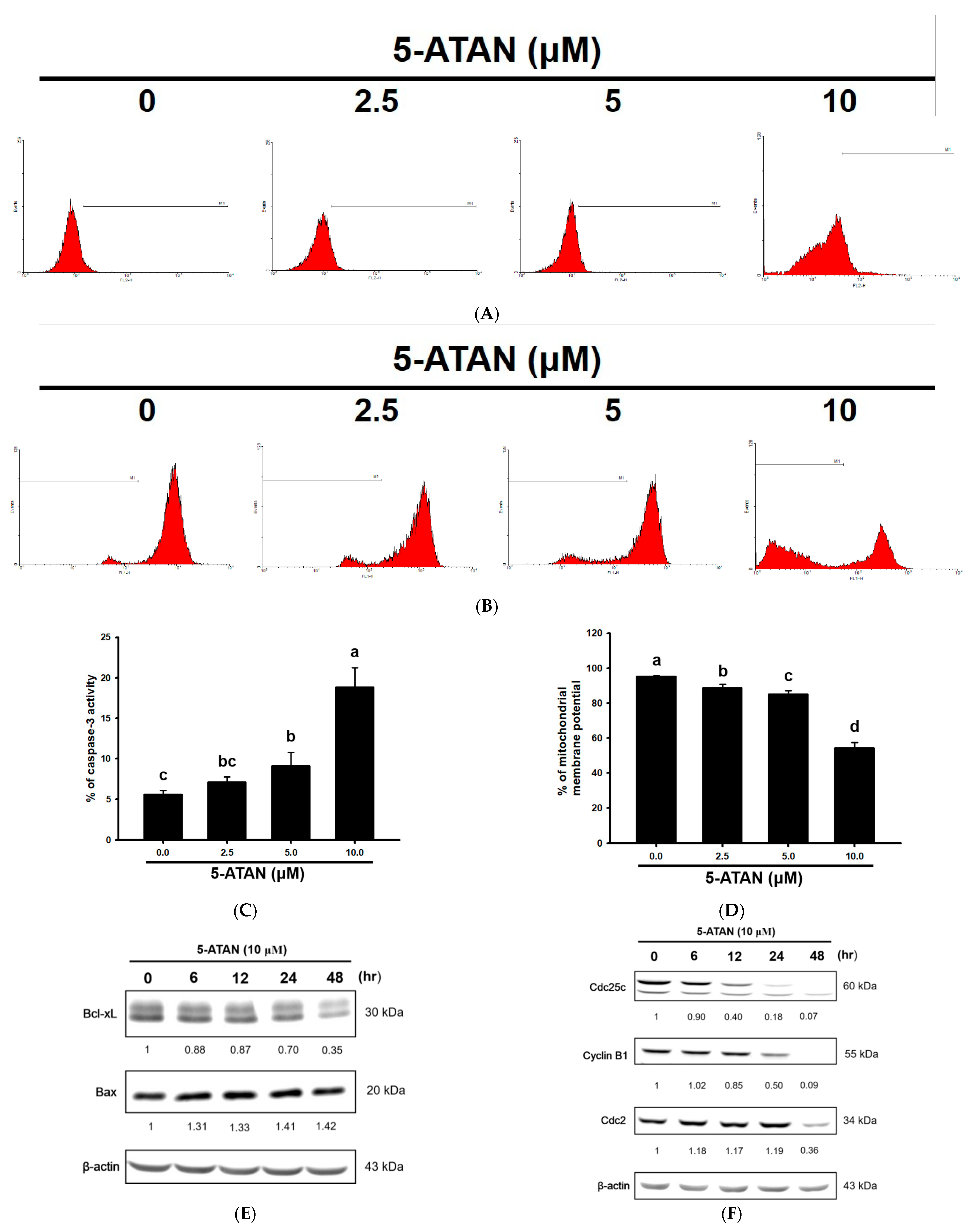
2.2. The 5-ATAN-Induced Cell Cycle Arrest and Apoptosis in PC-3 Human Prostate Cancer Cells
2.3. Effects of 5-ATAN on Intrinsic Mitochondrial Pathway and Cell Cycle Checkpoint Proteins in PC-3 Human Prostate Cancer Cells
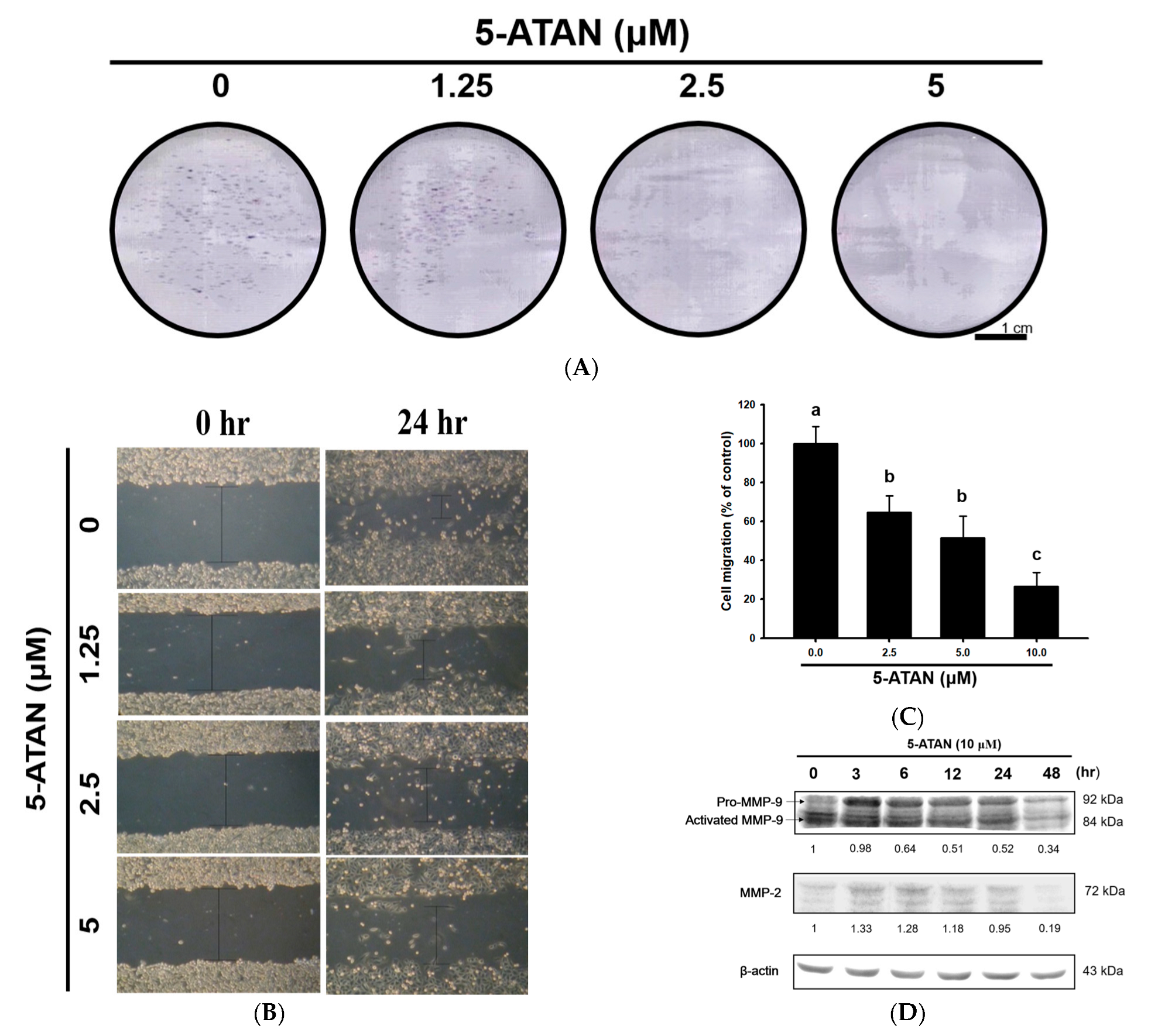
2.4. The 5-ATAN Inhibited Colony Formation and Cell migration in PC-3 Human Prostate Cancer Cells
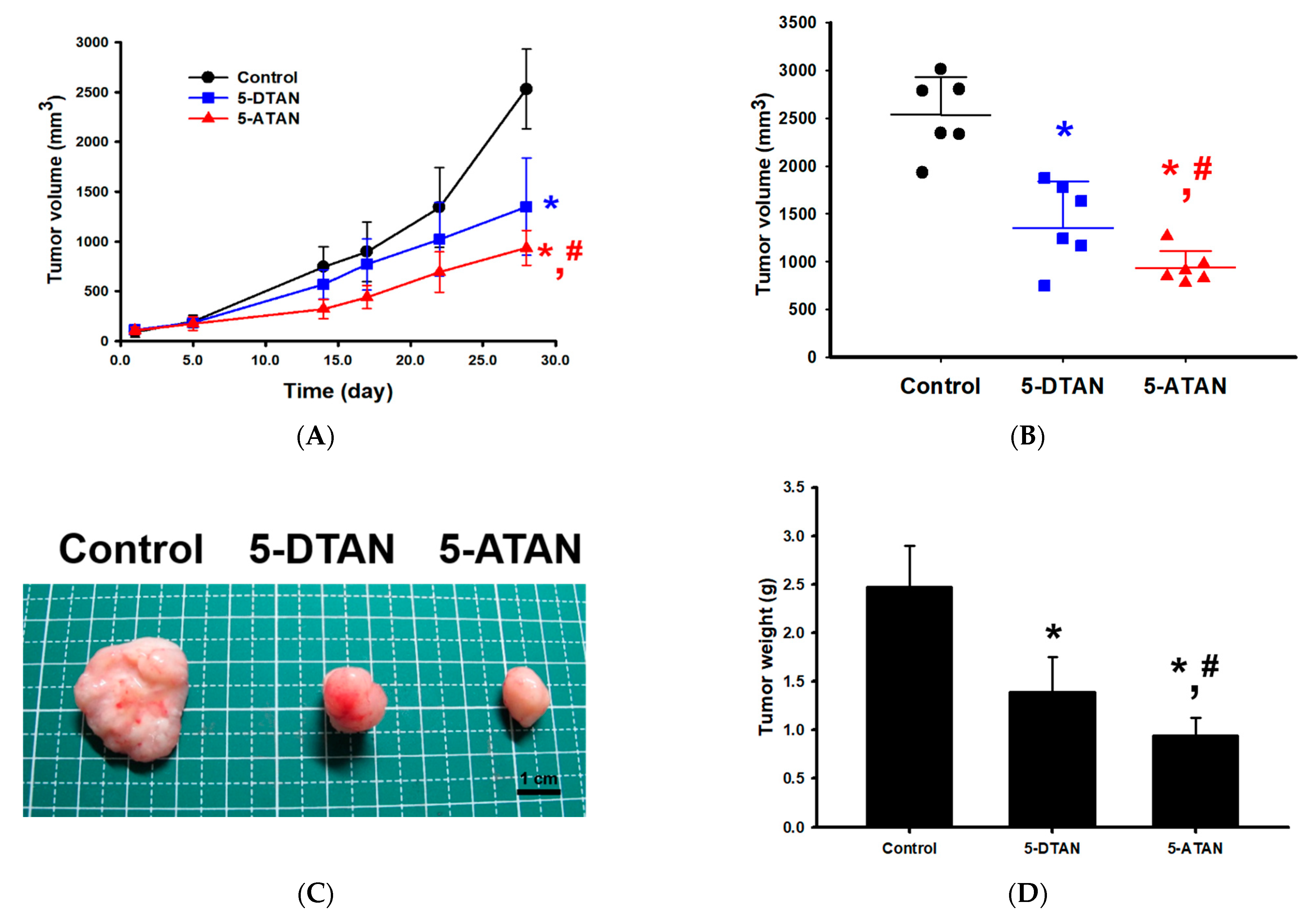
2.5. Effects of 5-DTAN and 5-ATAN on Tumor Size and Weight of PC-3 Cell Xenografts in Nude Mice
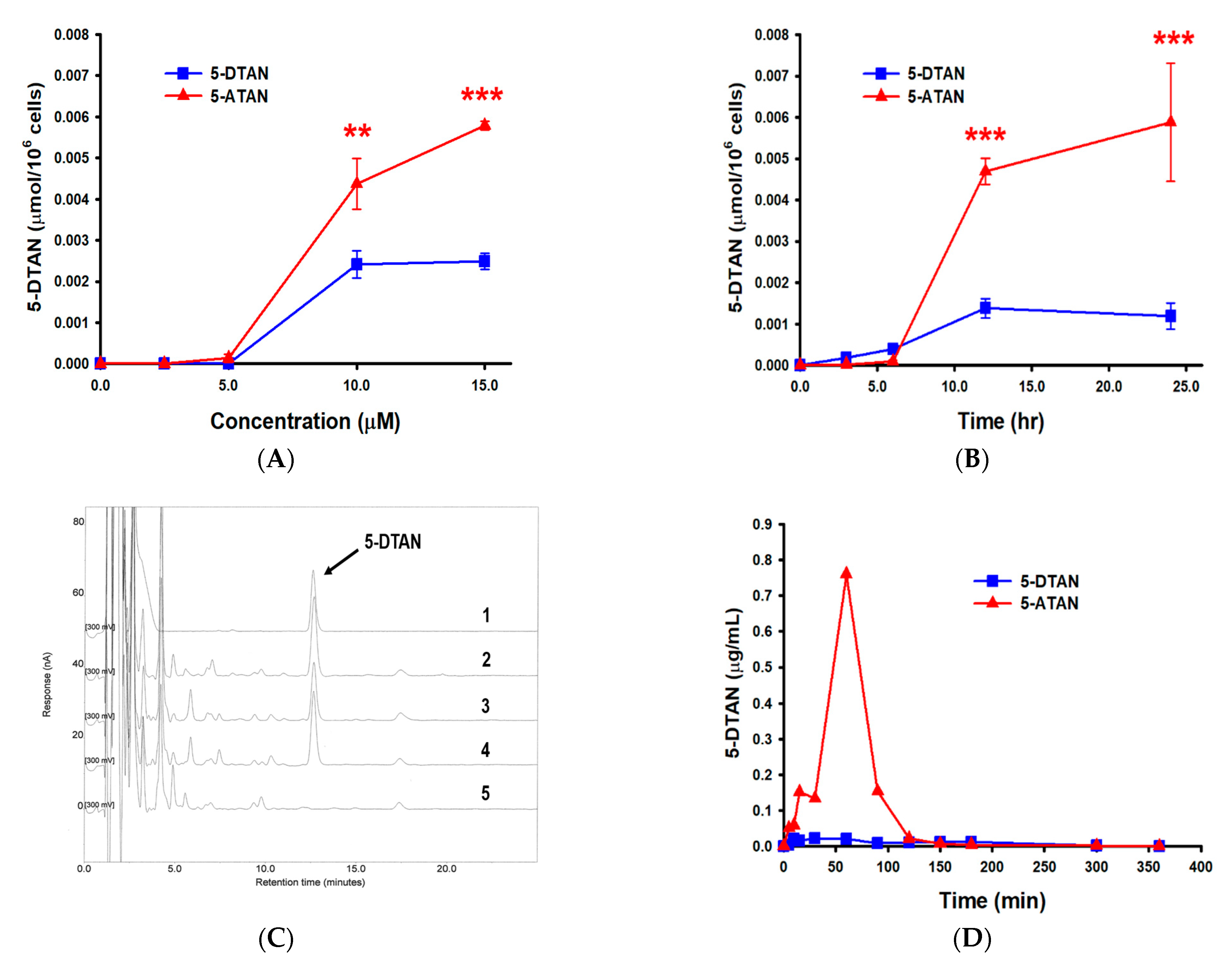
2.6. Effects of Acetylation on the Cell Uptake of 5-DTAN in PC-3 Cells and Its Oral Bioavailability in C57BL/6J Mice
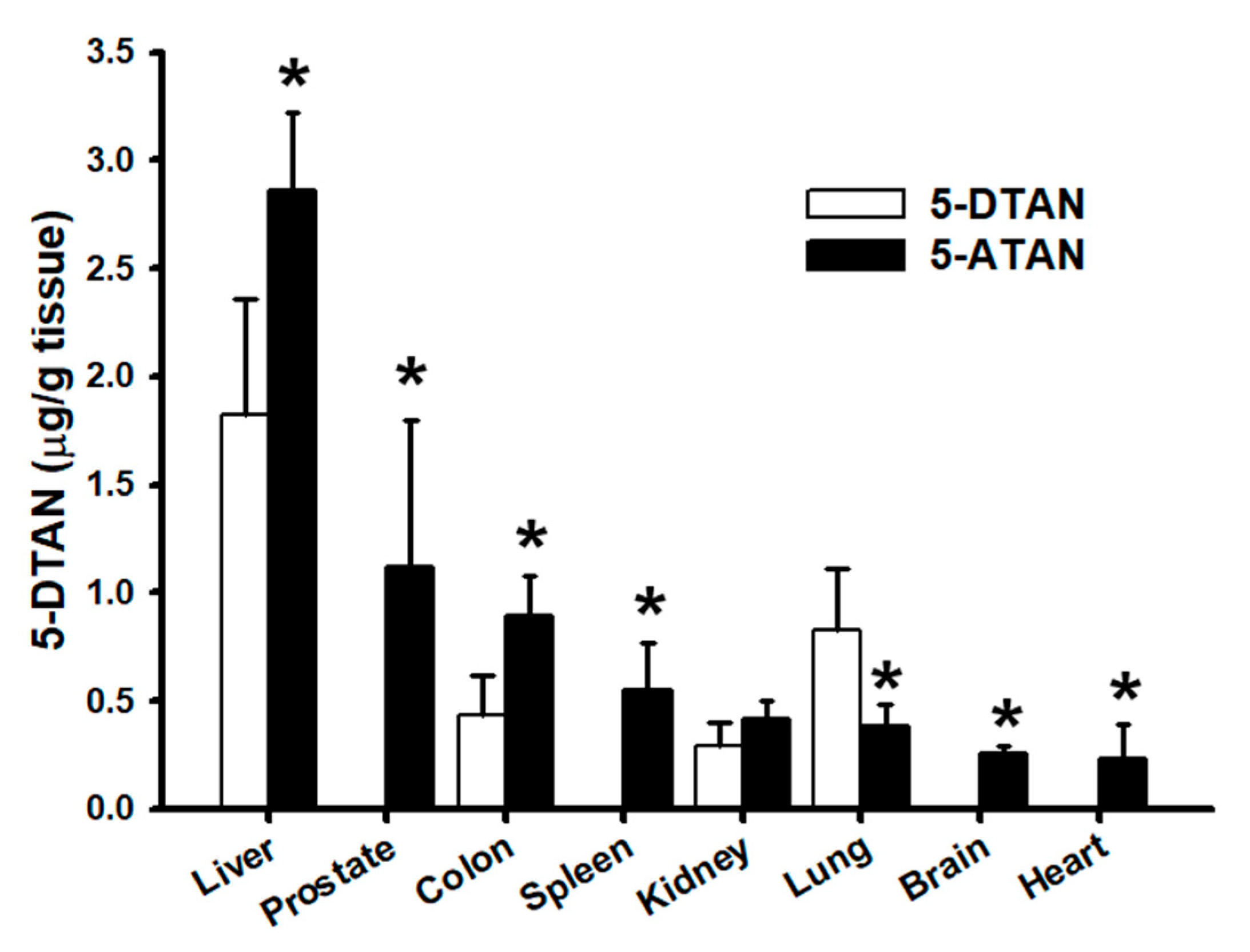
2.7. Effects of Acetylation on Tissue Distribution of 5-DTAN in C57BL/6J Mice
3. Materials and Methods
3.1. Chemicals
3.2. Cell Culture and Treatments
3.3. Cell Viability, Cell Cycle, and DNA Fragmentation Analysis
3.4. Caspase-3 Activity and Mitochondria Membrane Potential Analysis
3.5. Colony Formation and Cell Migration Analysis
3.6. Protein Expression Analysis
3.7. Animals
3.8. Inhibition of Tumor Growth in the Xenograft Model
3.9. Cellular Uptake, Oral Bioavailibiltiy, and Tissue Distribution Analysis
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jayaprakasha, G.K.; Negi, P.S.; Sikder, S.; Rao, L.J.; Sakariah, K.K. Antibacterial activity of Citrus reticulata peel extracts. Z. Naturforsch. C J. Biosci. 2000, 55, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Meng, D.; Zhang, P.; Wang, X.; Du, G.; Brennan, C.; Li, S.; Ho, C.T.; Zhao, H. Antioxidant protection of nobiletin, 5-demethylnobiletin, tangeretin, and 5-demethyltangeretin from citrus peel in Saccharomyces cerevisiae. J. Agric. Food Chem. 2018, 66, 3155–3160. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Luo, J.; Han, Y.; Du, H.; Liu, J.; He, W.; Zhu, J.; Xiao, J.; Wang, J.; Cao, Y.; et al. Dietary tangeretin alleviated dextran sulfate sodium-induced colitis in mice via inhibiting inflammatory response, restoring intestinal barrier function, and modulating gut microbiota. J. Agric. Food Chem. 2021, 69, 7663–7674. [Google Scholar] [CrossRef]
- Omar, H.A.; Mohamed, W.R.; Arab, H.H.; Arafa, E.-S. Tangeretin alleviates cisplatin-induced acute hepatic injury in rats: Targeting MAPKs and apoptosis. PLoS ONE 2016, 11, e0151649. [Google Scholar] [CrossRef]
- Bao, J.; Liang, Z.; Gong, X.; Zhao, Y.; Wu, M.; Liu, W.; Tu, C.; Wang, X.; Shu, X. Tangeretin inhibits BACE1 activity and attenuates cognitive impairments in AD model mice. J. Agric. Food Chem. 2022, 70, 1536–1546. [Google Scholar] [CrossRef]
- Feng, K.; Lan, Y.; Zhu, X.; Li, J.; Chen, T.; Huang, Q.; Ho, C.T.; Chen, Y.; Cao, Y. Hepatic lipidomics analysis reveals the antiobesity and cholesterol-lowering effects of tangeretin in high-fat diet-fed rats. J. Agric. Food Chem. 2020, 68, 6142–6153. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Chen, W.J.; Lin-Shiau, S.Y.; Ho, C.T.; Lin, J.K. Tangeretin induces cell-cycle G1 arrest through inhibiting cyclin-dependent kinases 2 and 4 activities as well as elevating Cdk inhibitors p21 and p27 in human colorectal carcinoma cells. Carcinogenesis 2002, 23, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Song, M.; Rakariyatham, K.; Zheng, J.; Wang, M.; Xu, F.; Gao, Z.; Xiao, H. Inhibitory Effects of 4′-Demethylnobiletin, a Metabolite of Nobiletin, on 12-O-Tetradecanoylphorbol-13-acetate (TPA)-Induced Inflammation in Mouse Ears. J. Agric. Food Chem. 2015, 63, 10921–10927. [Google Scholar] [CrossRef]
- Wu, X.; Song, M.; Rakariyatham, K.; Zheng, J.; Guo, S.; Tang, Z.; Zhou, S.; Xiao, H. Anti-inflammatory effects of 4′-demethylnobiletin, a major metabolite of nobiletin. J. Funct. Foods 2015, 19, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Charoensinphon, N.; Wu, X.; Zheng, J.; Gao, Z.; Xu, F.; Wang, M.; Xiao, H. Inhibitory effects of metabolites of 5-demethylnobiletin on human nonsmall cell lung cancer cells. J. Agric. Food Chem. 2016, 64, 4943–4949. [Google Scholar] [CrossRef] [PubMed]
- Qiu, P.; Dong, P.; Guan, H.; Li, S.; Ho, C.T.; Pan, M.H.; McClements, D.J.; Xiao, H. Inhibitory effects of 5-hydroxy polymethoxyflavones on colon cancer cells. Mol. Nutr. Food Res. 2010, 54 (Suppl. S2), S244–S252. [Google Scholar] [CrossRef] [PubMed]
- Qiu, P.; Guan, H.; Dong, P.; Li, S.; Ho, C.-T.; Pan, M.-H.; McClements, D.J.; Xiao, H. The p53-, Bax- and p21-dependent inhibition of colon cancer cell growth by 5-hydroxy polymethoxyflavones. Mol. Nutr. Food Res. 2011, 55, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Charoensinphon, N.; Qiu, P.; Dong, P.; Zheng, J.; Ngauv, P.; Cao, Y.; Li, S.; Ho, C.T.; Xiao, H. 5-demethyltangeretin inhibits human nonsmall cell lung cancer cell growth by inducing G2/M cell cycle arrest and apoptosis. Mol. Nutr. Food Res. 2013, 57, 2103–2111. [Google Scholar] [CrossRef]
- Ma, N.; Lai, C.-S.; Chung, C.-H.; Yang, J.-M.; Hsu, K.-C.; Chen, C.-Y.; Chung, T.-S.; Li, S.; Ho, C.-T.; Pan, M.-H. 5-Demethyltangeretin is more potent than tangeretin in inhibiting dimethylbenz(a)anthracene (DMBA)/12-O-tetradecanoylphorbol-13-acetate (TPA)-induced skin tumorigenesis. J. Funct. Foods 2014, 11, 528–537. [Google Scholar] [CrossRef]
- Li, S.; Pan, M.-H.; Lo, C.-Y.; Tan, D.; Wang, Y.; Shahidi, F.; Ho, C.-T. Chemistry and health effects of polymethoxyflavones and hydroxylated polymethoxyflavones. J. Funct. Foods 2009, 1, 2–12. [Google Scholar] [CrossRef]
- Rautio, J.; Kumpulainen, H.; Heimbach, T.; Oliyai, R.; Oh, D.; Järvinen, T.; Savolainen, J. Prodrugs: Design and clinical applications. Nat. Rev. Drug Discov. 2008, 7, 255–270. [Google Scholar] [CrossRef]
- Beaumont, K.; Webster, R.; Gardner, I.; Dack, K. Design of ester prodrugs to enhance oral Absorption of poorly permeable compounds: Challenges to the discovery scientist. Curr. Drug Metab. 2003, 4, 461–485. [Google Scholar] [CrossRef]
- Liederer, B.M.; Borchardt, R.T. Enzymes involved in the bioconversion of ester-based prodrugs. J. Pharm. Sci. 2006, 95, 1177–1195. [Google Scholar] [CrossRef]
- Nielsen, N.M.; Bundgaard, H. Evaluation of glycolamide esters and various other esters of aspirin as true aspirin prodrugs. J. Med. Chem. 1989, 32, 727–734. [Google Scholar] [CrossRef]
- Wright, C.A. XLIX.—On the action of organic acids and their anhydrides on the natural alkaloïds. Part I. J. Chem. Soc. 1874, 27, 1031–1043. [Google Scholar] [CrossRef]
- Lambert, J.D.; Sang, S.; Hong, J.; Kwon, S.J.; Lee, M.J.; Ho, C.T.; Yang, C.S. Peracetylation as a means of enhancing in vitro bioactivity and bioavailability of epigallocatechin-3-gallate. Drug Metab. Dispos. 2006, 34, 2111–2116. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Liu, X.; Wang, Q.; Cheng, S.; Zhang, S.; Zhang, M. Pharmacokinetics, tissue distribution and excretion study of resveratrol and its prodrug 3,5,4′-tri-O-acetylresveratrol in rats. Phytomedicine 2013, 20, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Duan, Y.; Zhi, D.; Li, G.; Wang, L.; Zhang, H.; Gu, L.; Ruan, H.; Zhang, K.; Liu, Q.; et al. Pro-apoptotic effects of the novel tangeretin derivate 5-acetyl-6,7,8,4’-tetramethylnortangeretin on MCF-7 breast cancer cells. Cell Biochem. Biophys. 2014, 70, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Zhi, D.; Liu, S.; Lin, L.; Wang, L.; Wang, J.; Ma, J.; Wang, S.; Zhao, H.; Ho, C.-T.; Wang, Y.; et al. 5-Acetyl-6,7,8,4′-tetramethylnortangeretin induces apoptosis in multiple myeloma U266 cells. Food Sci. Hum. Wellness 2014, 3, 197–203. [Google Scholar] [CrossRef][Green Version]
- Li, Y.R.; Li, S.; Ho, C.-T.; Chang, Y.-H.; Tan, K.-T.; Chung, T.-W.; Wang, B.-Y.; Chen, Y.-K.; Lin, C.-C. Tangeretin derivative, 5-acetyloxy-6,7,8,4′-tetramethoxyflavone induces G2/M arrest, apoptosis and autophagy in human non-small cell lung cancer cells in vitro and in vivo. Cancer Biol. Ther. 2016, 17, 48–64. [Google Scholar] [CrossRef]
- Cheng, Y.-P.; Li, S.; Chuang, W.-L.; Li, C.-H.; Chen, G.-J.; Chang, C.-C.; Or, C.-H.R.; Lin, P.-Y.; Chang, C.-C. Blockade of STAT3 signaling contributes to anticancer effect of 5-acetyloxy-6,7,8,4′-tetra-methoxyflavone, a tangeretin derivative, on human glioblastoma multiforme cells. Int. J. Mol. Sci. 2019, 20, 3366. [Google Scholar] [CrossRef]
- Robertson, J.D.; Orrenius, S.; Zhivotovsky, B. Review: Nuclear events in apoptosis. J. Struct. Biol. 2000, 129, 346–358. [Google Scholar] [CrossRef]
- Igney, F.H.; Krammer, P.H. Death and anti-death: Tumour resistance to apoptosis. Nat. Rev. Cancer 2002, 2, 277–288. [Google Scholar] [CrossRef]
- Kajstura, M.; Halicka, H.D.; Pryjma, J.; Darzynkiewicz, Z. Discontinuous fragmentation of nuclear DNA during apoptosis revealed by discrete “sub-G1” peaks on DNA content histograms. Cytom. Part A 2007, 71, 125–131. [Google Scholar] [CrossRef]
- Plesca, D.; Mazumder, S.; Almasan, A. DNA damage response and apoptosis. Methods Enzymol. 2008, 446, 107–122. [Google Scholar] [CrossRef]
- Xiao, H.; Yang, C.S.; Li, S.; Jin, H.; Ho, C.T.; Patel, T. Monodemethylated polymethoxyflavones from sweet orange (Citrus sinensis) peel inhibit growth of human lung cancer cells by apoptosis. Mol. Nutr. Food Res. 2009, 53, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Logue, S.E.; Martin, S.J. Caspase activation cascades in apoptosis. Biochem. Soc. Trans. 2008, 36, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.L.; Lin, J.Y.; Shieh, J.C.; Yeh, H.F.; Hsieh, Y.H.; Cheng, Y.C.; Lee, H.J.; Shen, C.Y.; Cheng, C.W. Induction of G2/M phase arrest by diosgenin via activation of Chk1 kinase and Cdc25C regulatory pathways to promote apoptosis in human breast cancer cells. Int. J. Mol. Sci. 2019, 21, 172. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Jiang, P.W.; Li, C.; Gao, M.X.; Sun, Y.S.; Zhang, D.Y.; Du, W.Q.; Zhao, J.; Shi, S.T.; Li, Y.; et al. Cdc25C/cdc2/cyclin B, raf/MEK/ERK and PERK/eIF2alpha/CHOP pathways are involved in forskolin-induced growth inhibition of MM.1S cells by G2/M arrest and mitochondrion-dependent apoptosis. Cell Cycle 2021, 20, 2402–2412. [Google Scholar] [CrossRef]
- Rafehi, H.; Orlowski, C.; Georgiadis, G.T.; Ververis, K.; El-Osta, A.; Karagiannis, T.C. Clonogenic assay: Adherent cells. J. Vis. Exp. 2011, 49, e2573. [Google Scholar] [CrossRef]
- Rajendran, V.; Jain, M.V. In Vitro Tumorigenic Assay: Colony forming assay for cancer stem cells. Methods Mol. Biol. 2018, 1692, 89–95. [Google Scholar] [CrossRef]
- Freitas, J.T.; Jozic, I.; Bedogni, B. Wound healing assay for melanoma cell migration. Methods Mol. Biol. 2021, 2265, 65–71. [Google Scholar] [CrossRef]
- Palmer, T.D.; Ashby, W.J.; Lewis, J.D.; Zijlstra, A. Targeting tumor cell motility to prevent metastasis. Adv. Drug. Deliv Rev. 2011, 63, 568–581. [Google Scholar] [CrossRef]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a005058. [Google Scholar] [CrossRef]
- Gialeli, C.; Theocharis, A.D.; Karamanos, N.K. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011, 278, 16–27. [Google Scholar] [CrossRef]
- Jablonska-Trypuc, A.; Matejczyk, M.; Rosochacki, S. Matrix metalloproteinases (MMPs), the main extracellular matrix (ECM) enzymes in collagen degradation, as a target for anticancer drugs. J. Enzyme. Inhib. Med. Chem. 2016, 31, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, Y.; Zhang, H.; Chen, J.; Cao, J.; Chen, Q.; Li, X.; Sun, C. Polymethoxyflavones from citrus inhibited gastric cancer cell proliferation through inducing apoptosis by upregulating RARbeta, both in vitro and in vivo. Food Chem. Toxicol. 2020, 146, 111811. [Google Scholar] [CrossRef] [PubMed]
- Lam, W.H.; Kazi, A.; Kuhn, D.J.; Chow, L.M.; Chan, A.S.; Dou, Q.P.; Chan, T.H. A potential prodrug for a green tea polyphenol proteasome inhibitor: Evaluation of the peracetate ester of (-)-epigallocatechin gallate [(-)-EGCG]. Bioorg. Med. Chem. 2004, 12, 5587–5593. [Google Scholar] [CrossRef]
- Saturnino, C.; Sinicropi, M.S.; Parisi, O.I.; Iacopetta, D.; Popolo, A.; Marzocco, S.; Autore, G.; Caruso, A.; Cappello, A.R.; Longo, P.; et al. Acetylated hyaluronic acid: Enhanced bioavailability and biological studies. BioMed Res. Int. 2014, 2014, 921549. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.K.; Kuo, Y.H.; Chiang, B.H.; Lo, J.M.; Sheen, L.Y. Cytotoxic activities of 9,11-dehydroergosterol peroxide and ergosterol peroxide from the fermentation mycelia of ganoderma lucidum cultivated in the medium containing leguminous plants on Hep 3B cells. J. Agric. Food Chem. 2009, 57, 5713–5719. [Google Scholar] [CrossRef]
- Tsai, H.Y.; Shih, Y.Y.; Yeh, Y.T.; Huang, C.H.; Liao, C.A.; Hu, C.Y.; Nagabhushanam, K.; Ho, C.T.; Chen, Y.K. Pterostilbene and its derivative 3’-hydroxypterostilbene ameliorated nonalcoholic fatty liver disease through synergistic modulation of the gut microbiota and SIRT1/AMPK signaling pathway. J. Agric. Food Chem. 2022, 70, 4966–4980. [Google Scholar] [CrossRef]
- Tsai, H.Y.; Yang, J.F.; Chen, H.H.; You, F.N.; Zhao, Y.J.; Lin, Y.H.; Hsu, J.L.; Chang, C.I.; Chen, Y.K. The effect of hot water extract of tilapia on exercise capacity in mice. Appl. Sci. 2022, 12, 2601. [Google Scholar] [CrossRef]
- Dong, P.; Qiu, P.; Zhu, Y.; Li, S.; Ho, C.T.; McClements, D.J.; Xiao, H. Simultaneous determination of four 5-hydroxy polymethoxyflavones by reversed-phase high performance liquid chromatography with electrochemical detection. J. Chromatogr. A 2010, 1217, 642–647. [Google Scholar] [CrossRef]
| Concentration (μM) | Cell Cycle Distribution (%) | |||
|---|---|---|---|---|
| G0/G1 | S | G2/M | ||
| 5-DTAN | 0 | 63.8 ± 1.7 a | 8.5 ± 0.6 | 27.7 ± 1.3 b |
| 2.5 | 60.2 ± 2.8 b | 8.2 ± 0.1 | 31.6 ± 2.7 a | |
| 5 | 59.9 ± 0.4 b | 8.7 ± 0.3 | 31.4 ± 0.5 a | |
| 10 | 58.2 ± 2.4 b | 8.5 ± 0.9 | 32.6 ± 1.1 a | |
| 5-ATAN | 0 | 55.3 ± 0.6 AB | 12.6 ± 0.4 A | 32.2 ± 0.9 D |
| 2.5 | 58.5 ± 1.1 A | 6.8 ± 0.3 B | 34.7 ± 0.8 C | |
| 5 | 49.1 ± 0.3 B | 5.2 ± 0.3 C | 45.7 ± 0.5 B | |
| 10 | 44.7 ± 0.3 C | 4.6 ± 0.5 D | 50.3 ± 1.3 A | |
| Parameters | 5-DTAN in Plasma | |
|---|---|---|
| 5-DTAN (50 mg/kg bw) | 5-ATAN (50 mg/kg bw) | |
| Cmax (μg/mL) | 0.030 ± 0.009 | 0.773 ± 0.196 * |
| Tmax (min) | 29.1 ± 17.1 | 59.7 ± 0.1 * |
| t1/2 (min) | 173.4 ± 120.3 | 33.4 ± 9.8 * |
| AUC (μg/mL · min) | 3.15 ± 0.24 | 34.73 ± 10.08 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, H.-Y.; Yang, J.-F.; Chen, Y.-B.; Guo, J.-L.; Li, S.; Wei, G.-J.; Ho, C.-T.; Hsu, J.-L.; Chang, C.-I.; Liang, Y.-S.; et al. Acetylation Enhances the Anticancer Activity and Oral Bioavailability of 5-Demethyltangeretin. Int. J. Mol. Sci. 2022, 23, 13284. https://doi.org/10.3390/ijms232113284
Tsai H-Y, Yang J-F, Chen Y-B, Guo J-L, Li S, Wei G-J, Ho C-T, Hsu J-L, Chang C-I, Liang Y-S, et al. Acetylation Enhances the Anticancer Activity and Oral Bioavailability of 5-Demethyltangeretin. International Journal of Molecular Sciences. 2022; 23(21):13284. https://doi.org/10.3390/ijms232113284
Chicago/Turabian StyleTsai, Hui-Yun, Jia-Fang Yang, Yin-Bo Chen, Jia-Lin Guo, Shiming Li, Guor-Jien Wei, Chi-Tang Ho, Jue-Liang Hsu, Chi-I Chang, Yu-Shen Liang, and et al. 2022. "Acetylation Enhances the Anticancer Activity and Oral Bioavailability of 5-Demethyltangeretin" International Journal of Molecular Sciences 23, no. 21: 13284. https://doi.org/10.3390/ijms232113284
APA StyleTsai, H.-Y., Yang, J.-F., Chen, Y.-B., Guo, J.-L., Li, S., Wei, G.-J., Ho, C.-T., Hsu, J.-L., Chang, C.-I., Liang, Y.-S., Yu, H.-S., & Chen, Y.-K. (2022). Acetylation Enhances the Anticancer Activity and Oral Bioavailability of 5-Demethyltangeretin. International Journal of Molecular Sciences, 23(21), 13284. https://doi.org/10.3390/ijms232113284









