BMP3 Affects Cortical and Trabecular Long Bone Development in Mice
Abstract
1. Introduction
2. Results
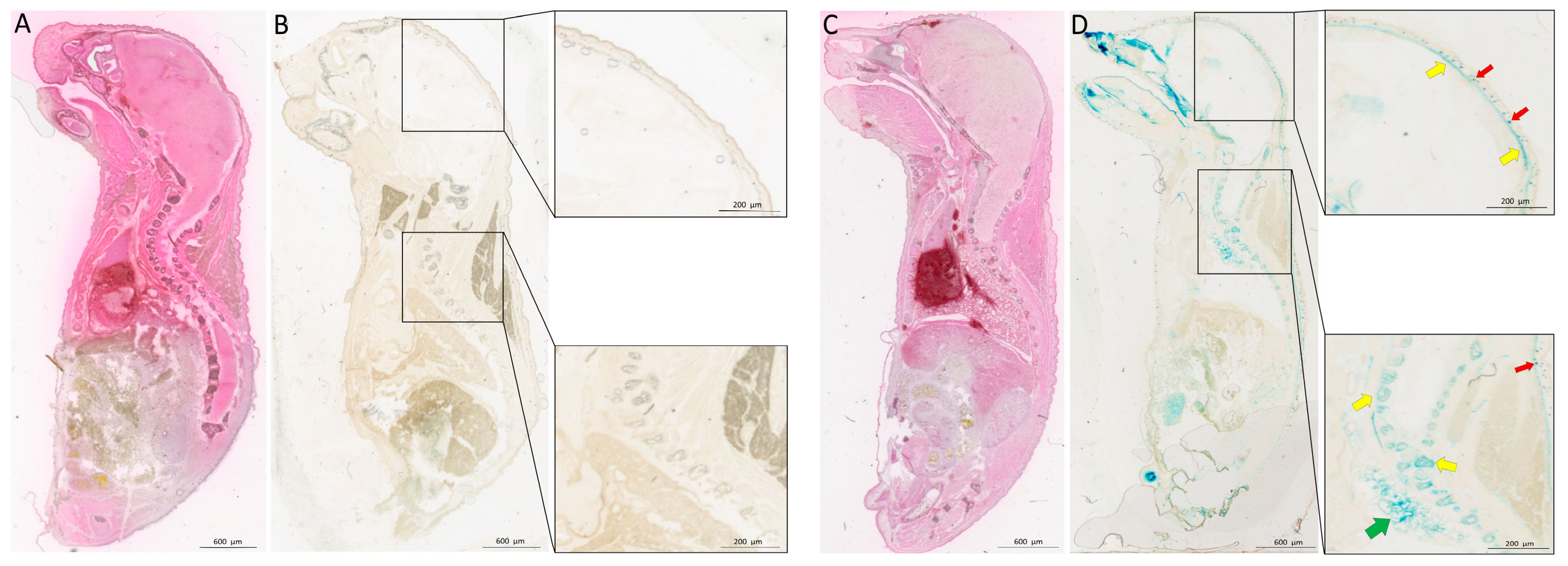
2.1. Bmp3 Tissue Localization
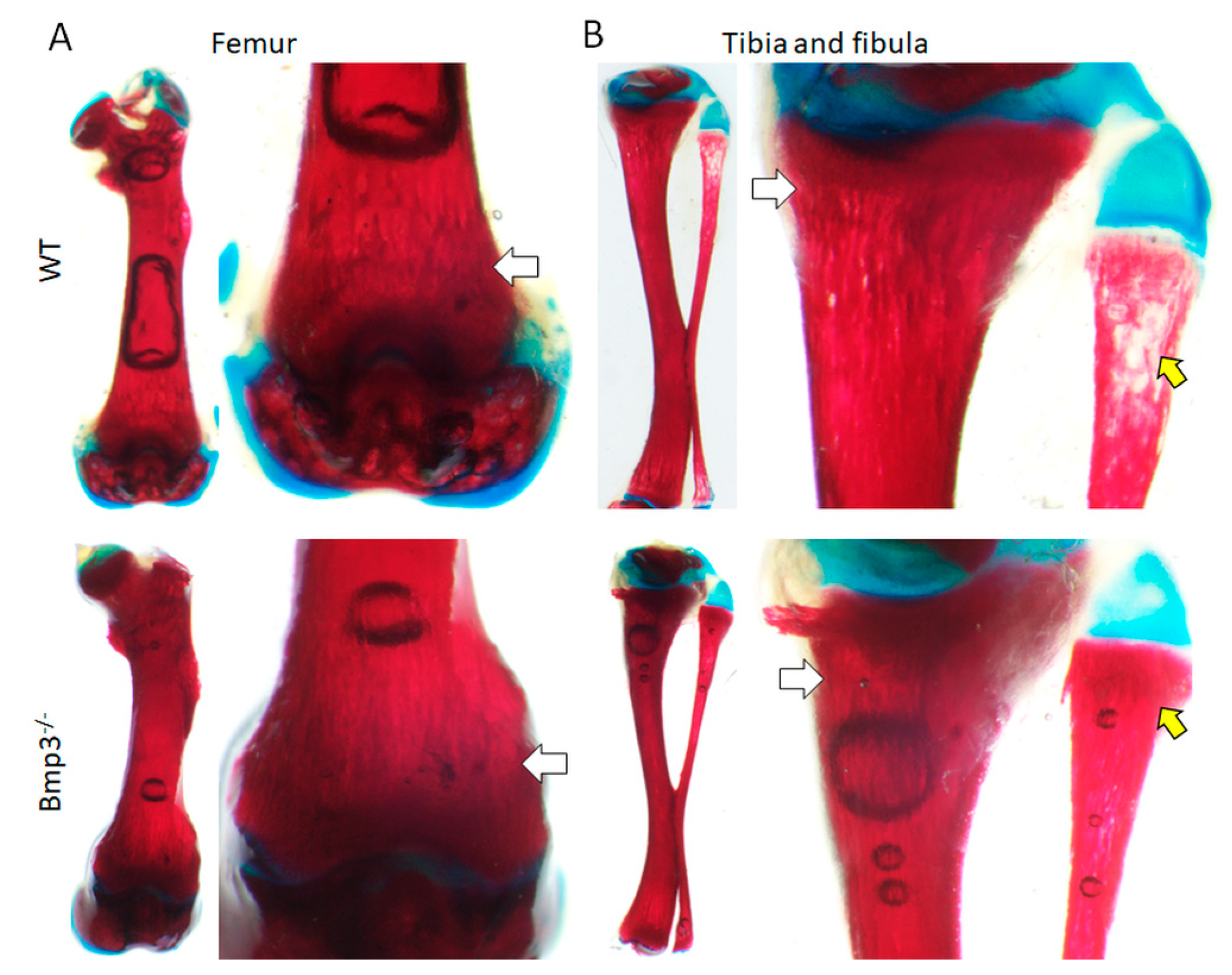
2.2. Effect of Bmp3 on Early Bone Development
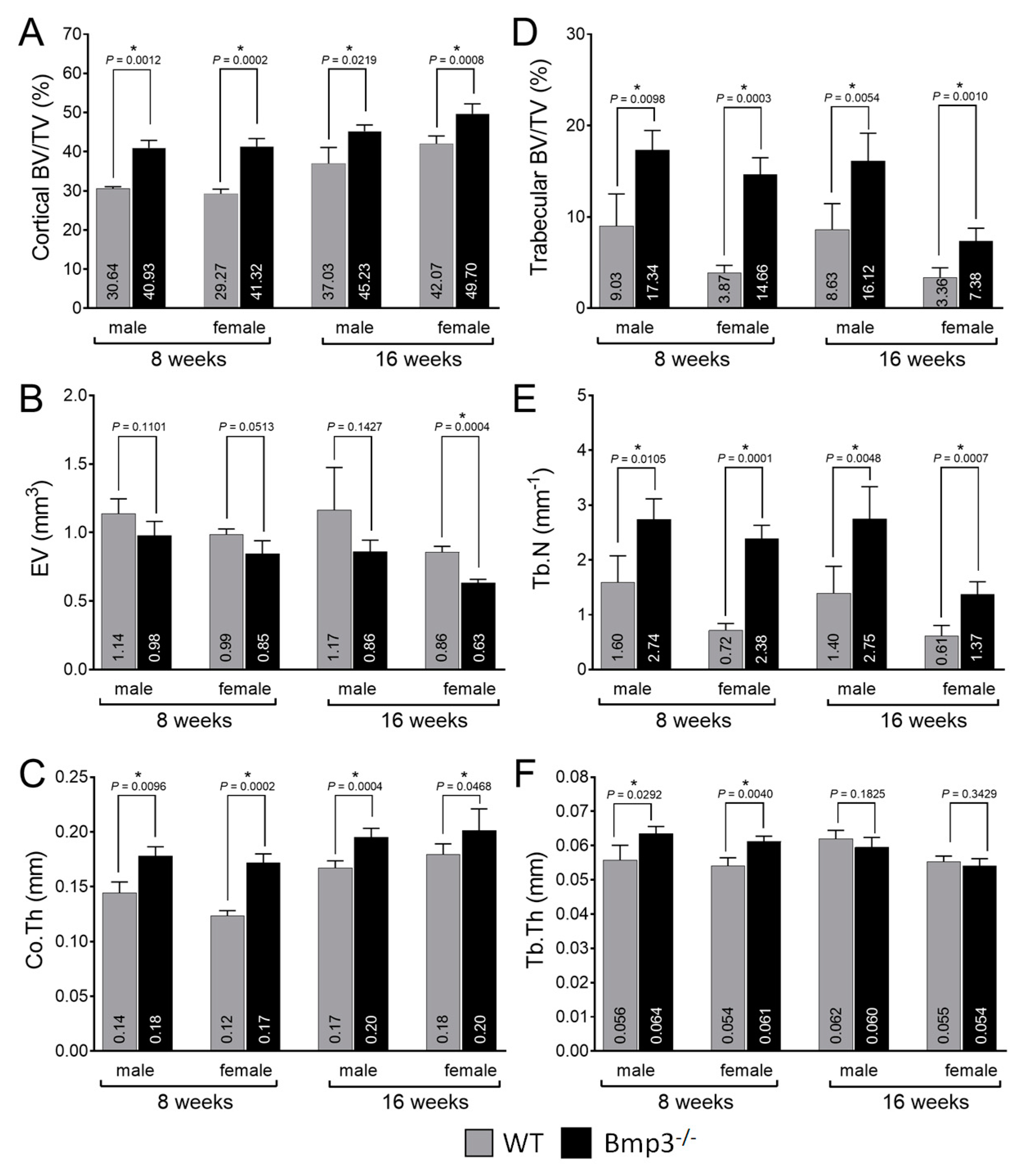
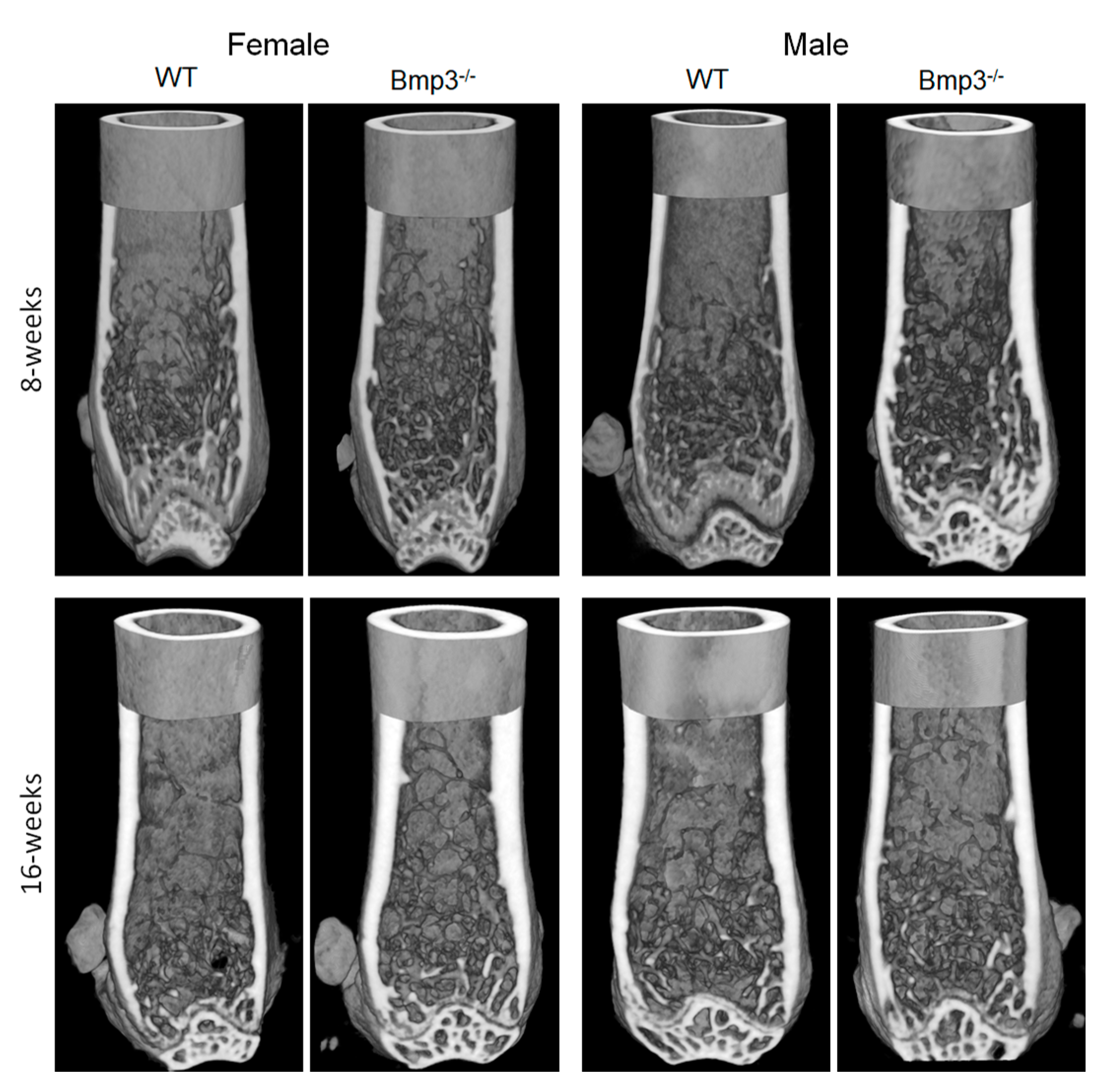
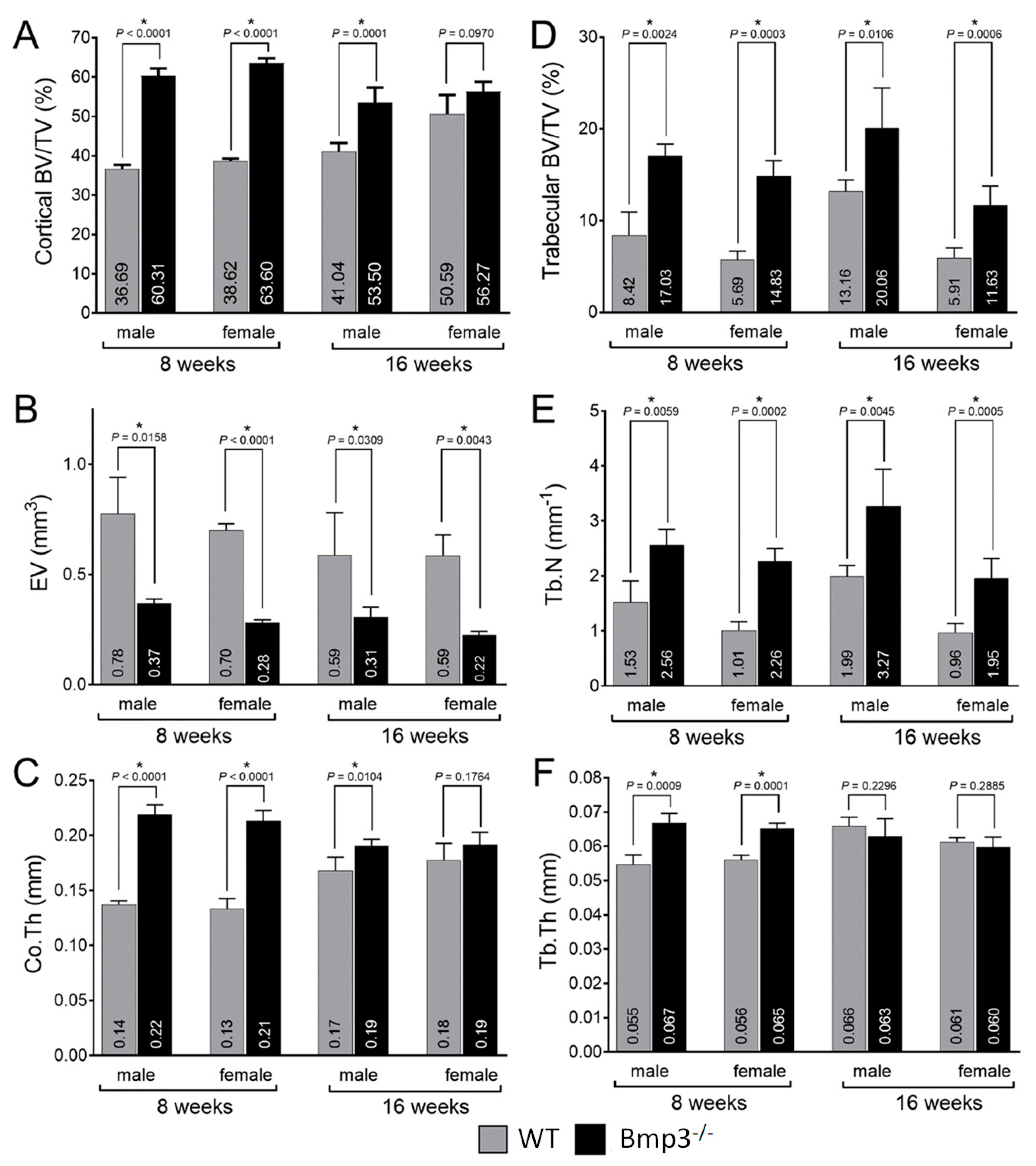
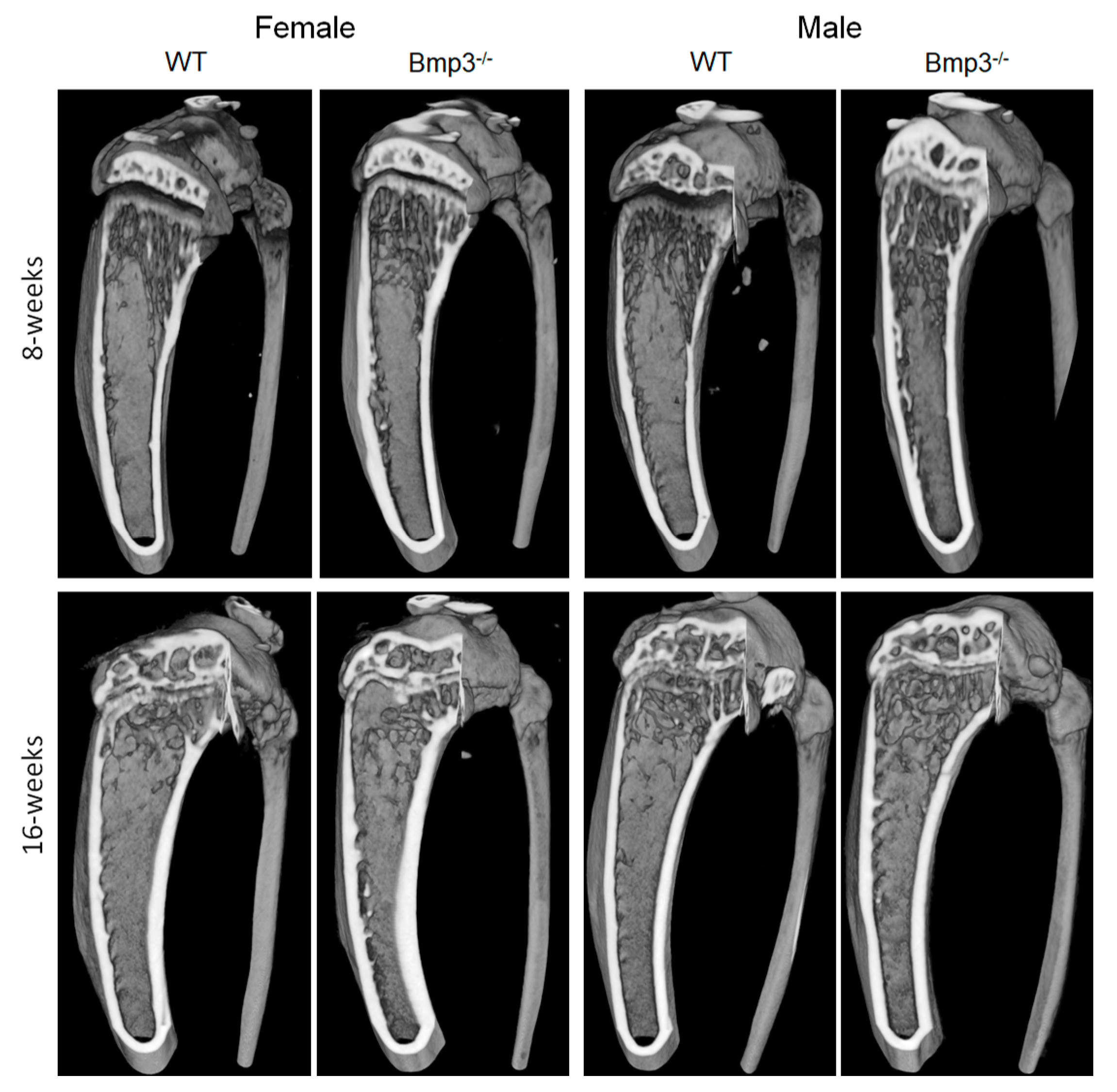
2.3. Changes in Cortical and Trabecular Long Bone Parameters of Bmp3−/− Mice Are Not Age- or Sex-Dependent
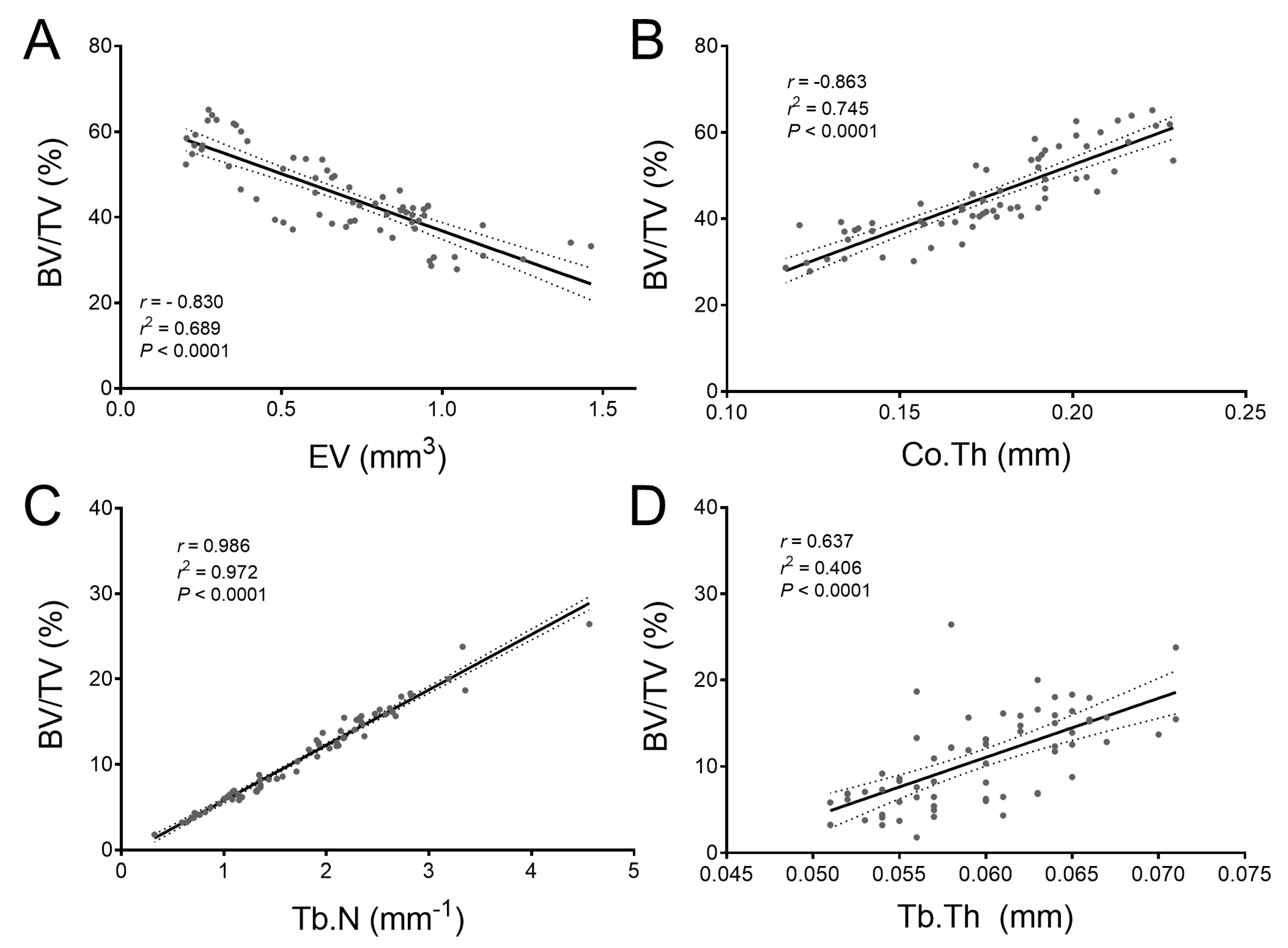
2.4. Bmp3 Effect Size and Association Analysis of Long Bone Parameters
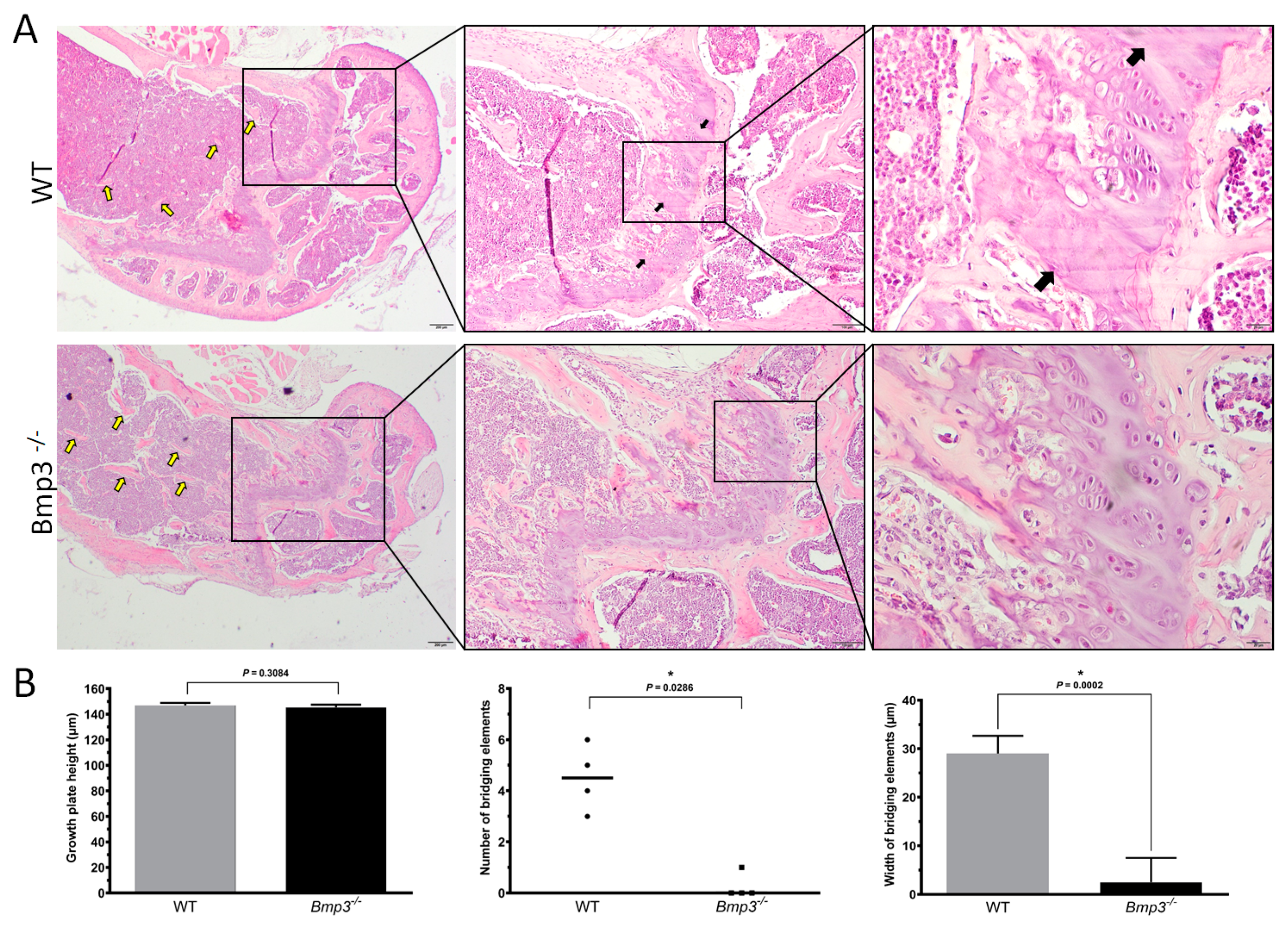
2.5. Bmp3 Deficiency Delays Epiphyseal Cartilage Mineralization and Growth Plate Bridging
3. Discussion
4. Materials and Methods
4.1. Animal Model
4.2. Genotype Determination
4.3. Bmp3 Tissue Expression
4.4. Differential Skeleton Staining
4.5. Bone Characterization
4.6. Micro-CT Analysis
4.7. Histological and Immunohistochemical Analysis
4.8. Data Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vukicevic, S.; Sampath, K.T. (Eds.) Bone Morphogenetic Proteins: From Laboratory to Clinical Practice; Birkhäuser Verlag: Basel, Switzerland, 2002; ISBN 3-7643-6509-9. [Google Scholar]
- Chen, D.; Zhao, M.; Mundy, G.R. Bone morphogenetic proteins. Growth Factors 2004, 22, 233–241. [Google Scholar] [CrossRef]
- Katagiri, T.; Watabe, T. Bone Morphogenetic Proteins. Cold Spring Harb. Perspect. Biol. 2016, 8, a021899. [Google Scholar] [CrossRef] [PubMed]
- Wozney, J.M. Bone morphogenetic proteins. Prog. Growth Factor Res. 1989, 1, 267–280. [Google Scholar] [CrossRef]
- Wozney, J.M.; Rosen, V. Bone morphogenetic protein and bone morphogenetic protein gene family in bone formation and repair. Clin. Orthop. Relat. Res. 1998, 346, 26–37. [Google Scholar] [CrossRef]
- Vukicevic, S.; Luyten, F.P.; Reddi, A.H. Stimulation of the expression of osteogenic and chondrogenic phenotypes in vitro by osteogenin. Proc. Natl. Acad. Sci. USA 1989, 86, 8793–8797. [Google Scholar] [CrossRef] [PubMed]
- Luyten, F.P.; Cunningham, N.S.; Vukicevic, S.; Paralkar, V.; Ripamonti, U.; Reddi, A.H. Advances in osteogenin and related bone morphogenetic proteins in bone induction and repair. Acta Orthop. Belg. 1992, 58 (Suppl. 1), 263–267. [Google Scholar] [PubMed]
- Luyten, F.P.; Yu, Y.M.; Yanagishita, M.; Vukicevic, S.; Hammonds, R.G.; Reddi, A.H. Natural bovine osteogenin and recombinant human bone morphogenetic protein-2B are equipotent in the maintenance of proteoglycans in bovine articular cartilage explant cultures. J. Biol. Chem. 1992, 267, 3691–3695. [Google Scholar] [CrossRef]
- Vukicevic, S.; Paralkar, V.M.; Cunningham, N.S.; Gutkind, J.S.; Reddi, A.H. Autoradiographic localization of osteogenin binding sites in cartilage and bone during rat embryonic development. Dev. Biol. 1990, 140, 209–214. [Google Scholar] [CrossRef]
- Bahamonde, M.E.; Lyons, K.M. BMP3: To be or not to be a BMP. J. Bone Jt. Surg. Am. 2001, 83 (Suppl. 1), S56–S62. [Google Scholar] [CrossRef]
- Bialek, P.; Parkington, J.; Li, X.; Gavin, D.; Wallace, C.; Zhang, J.; Root, A.; Yan, G.; Warner, L.; Seeherman, H.J.; et al. A myostatin and activin decoy receptor enhances bone formation in mice. Bone 2014, 60, 162–171. [Google Scholar] [CrossRef]
- Daluiski, A.; Engstrand, T.; Bahamonde, M.E.; Gamer, L.W.; Agius, E.; Stevenson, S.L.; Cox, K.; Rosen, V.; Lyons, K.M. Bone morphogenetic protein-3 is a negative regulator of bone density. Nat. Genet. 2001, 27, 84–88. [Google Scholar] [CrossRef]
- Zoricic, S.; Maric, I.; Bobinac, D.; Vukicevic, S. Expression of bone morphogenetic proteins and cartilage-derived morphogenetic proteins during osteophyte formation in humans. J. Anat. 2003, 202, 269–277. [Google Scholar] [CrossRef]
- Kokabu, S.; Gamer, L.; Cox, K.; Lowery, J.; Tsuji, K.; Raz, R.; Economides, A.; Katagiri, T.; Rosen, V. BMP3 suppresses osteoblast differentiation of bone marrow stromal cells via interaction with Acvr2b. Mol. Endocrinol. 2012, 26, 87–94. [Google Scholar] [CrossRef]
- Yamashita, K.; Mikawa, S.; Sato, K. BMP3 expression in the adult rat CNS. Brain Res. 2016, 1643, 35–50. [Google Scholar] [CrossRef] [PubMed]
- Ciller, I.M.; Palanisamy, S.K.; Ciller, U.A.; McFarlane, J.R. Postnatal expression of bone morphogenetic proteins and their receptors in the mouse testis. Physiol. Res. 2016, 65, 673–682. [Google Scholar] [CrossRef]
- Vukicevic, S.; Helder, M.N.; Luyten, F.P. Developing human lung and kidney are major sites for synthesis of bone morphogenetic protein-3 (osteogenin). J. Histochem. Cytochem. 1994, 42, 869–875. [Google Scholar] [CrossRef]
- Zhou, X.; Tao, Y.; Liang, C.; Zhang, Y.; Li, H.; Chen, Q. BMP3 Alone and Together with TGF-β Promote the Differentiation of Human Mesenchymal Stem Cells into a Nucleus Pulposus-Like Phenotype. Int. J. Mol. Sci. 2015, 16, 20344–20359. [Google Scholar] [CrossRef]
- Cernea, M.; Tang, W.; Guan, H.; Yang, K. Wisp1 mediates Bmp3-stimulated mesenchymal stem cell proliferation. J. Mol. Endocrinol. 2016, 56, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yang, W.; Cao, Y.; Shi, Y.; Lei, C.; Du, B.; Li, X.; Zhang, Q. The Functions of BMP3 in Rabbit Articular Cartilage Repair. Int. J. Mol. Sci. 2015, 16, 25934–25946. [Google Scholar] [CrossRef]
- Gamer, L.W.; Ho, V.; Cox, K.; Rosen, V. Expression and function of BMP3 during chick limb development. Dev. Dyn. 2008, 237, 1691–1698. [Google Scholar] [CrossRef] [PubMed]
- Gamer, L.W.; Cox, K.; Carlo, J.M.; Rosen, V. Overexpression of BMP3 in the developing skeleton alters endochondral bone formation resulting in spontaneous rib fractures. Dev. Dyn. 2009, 238, 2374–2381. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hu, Y.; Ma, Z. The experimental study on expression of BMP3 gene during fracture healing. Zhonghua Wai Ke Za Zhi 1996, 34, 585–588. [Google Scholar]
- Zheng, L.; Yamashiro, T.; Fukunaga, T.; Balam, T.A.; Takano-Yamamoto, T. Bone morphogenetic protein 3 expression pattern in rat condylar cartilage, femoral cartilage and mandibular fracture callus. Eur. J. Oral Sci. 2005, 113, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Kloen, P.; Lauzier, D.; Hamdy, R.C. Co-expression of BMPs and BMP-inhibitors in human fractures and non-unions. Bone 2012, 51, 59–68. [Google Scholar] [CrossRef]
- Matzelle, M.M.; Shaw, A.T.; Baum, R.; Maeda, Y.; Li, J.; Karmakar, S.; Manning, C.A.; Walsh, N.C.; Rosen, V.; Gravallese, E.M. Inflammation in arthritis induces expression of BMP3, an inhibitor of bone formation. Scand. J. Rheumatol. 2016, 45, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Fan, J.; Liu, Y.; Li, T.; Xu, H.; Yang, Y.; Deng, L.; Li, H.; Zhao, R.C. miR-450b Promotes Osteogenic Differentiation In Vitro and Enhances Bone Formation In Vivo by Targeting BMP3. Stem Cells Dev. 2018, 27, 600–611. [Google Scholar] [CrossRef]
- Aspenberg, P.; Basic, N.; Tägil, M.; Vukicevic, S. Reduced expression of BMP-3 due to mechanical loading: A link between mechanical stimuli and tissue differentiation. Acta Orthop. Scand. 2000, 71, 558–562. [Google Scholar] [CrossRef]
- Dai, Z.; Popkie, A.P.; Zhu, W.-G.; Timmers, C.D.; Raval, A.; Tannehill-Gregg, S.; Morrison, C.D.; Auer, H.; Kratzke, R.A.; Niehans, G.; et al. Bone morphogenetic protein 3B silencing in non-small-cell lung cancer. Oncogene 2004, 23, 3521–3529. [Google Scholar] [CrossRef]
- Loh, K.; Chia, J.A.; Greco, S.; Cozzi, S.-J.; Buttenshaw, R.L.; Bond, C.E.; Simms, L.A.; Pike, T.; Young, J.P.; Jass, J.R.; et al. Bone morphogenic protein 3 inactivation is an early and frequent event in colorectal cancer development. Genes Chromosomes Cancer 2008, 47, 449–460. [Google Scholar] [CrossRef]
- Chen, X.-R.; Wang, J.-W.; Li, X.; Zhang, H.; Ye, Z.-Y. Role of BMP3 in progression of gastric carcinoma in Chinese people. World J. Gastroenterol. 2010, 16, 1409–1413. [Google Scholar] [CrossRef]
- Kim, Y.O.; Hong, I.K.; Eun, Y.G.; Nah, S.-S.; Lee, S.; Heo, S.-H.; Kim, H.-K.; Song, H.-Y.; Kim, H.-J. Polymorphisms in bone morphogenetic protein 3 and the risk of papillary thyroid cancer. Oncol. Lett. 2013, 5, 336–340. [Google Scholar] [CrossRef]
- Kisiel, J.B.; Li, J.; Zou, H.; Oseini, A.M.; Strauss, B.B.; Gulaid, K.H.; Moser, C.D.; Aderca, I.; Ahlquist, D.A.; Roberts, L.R.; et al. Methylated Bone Morphogenetic Protein 3 (BMP3) Gene: Evaluation of Tumor Suppressor Function and Biomarker Potential in Biliary Cancer. J. Mol. Biomark. Diagn. 2013, 4, 1000145. [Google Scholar] [CrossRef]
- Valenzuela, D.M.; Murphy, A.J.; Frendewey, D.; Gale, N.W.; Economides, A.N.; Auerbach, W.; Poueymirou, W.T.; Adams, N.C.; Rojas, J.; Yasenchak, J.; et al. High-throughput engineering of the mouse genome coupled with high-resolution expression analysis. Nat. Biotechnol. 2003, 21, 652–659. [Google Scholar] [CrossRef]
- Takahashi, H.; Ikeda, T. Transcripts for two members of the transforming growth factor-beta superfamily BMP-3 and BMP-7 are expressed in developing rat embryos. Dev. Dyn. 1996, 207, 439–449. [Google Scholar] [CrossRef]
- Groppe, J.; Greenwald, J.; Wiater, E.; Rodriguez-Leon, J.; Economides, A.N.; Kwiatkowski, W.; Baban, K.; Affolter, M.; Vale, W.W.; Izpisua Belmonte, J.C.; et al. Structural basis of BMP signaling inhibition by Noggin, a novel twelve-membered cystine knot protein. J. Bone Jt. Surg. Am. 2003, 85 (Suppl. 3), 52–58. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Otsuka, F.; Hino, J.; Miyoshi, T.; Takano, M.; Miyazato, M.; Makino, H.; Kangawa, K. Bone morphogenetic protein-3b (BMP-3b) inhibits osteoblast differentiation via Smad2/3 pathway by counteracting Smad1/5/8 signaling. Mol. Cell. Endocrinol. 2012, 350, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Wozney, J.M.; Rosen, V.; Celeste, A.J.; Mitsock, L.M.; Whitters, M.J.; Kriz, R.W.; Hewick, R.M.; Wang, E.A. Novel regulators of bone formation: Molecular clones and activities. Science 1988, 242, 1528–1534. [Google Scholar] [CrossRef]
- Goodman, C.A.; Hornberger, T.A.; Robling, A.G. Bone and skeletal muscle: Key players in mechanotransduction and potential overlapping mechanisms. Bone 2015, 80, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Sanger, T.J.; Norgard, E.A.; Pletscher, L.S.; Bevilacqua, M.; Brooks, V.R.; Sandell, L.J.; Cheverud, J.M. Developmental and genetic origins of murine long bone length variation. J. Exp. Zool. B Mol. Dev. Evol. 2011, 316, 146–161. [Google Scholar] [CrossRef] [PubMed]
- Sims, N.; Dupont, S.; Krust, A.; Clement-Lacroix, P.; Minet, D.; Resche-Rigon, M.; Gaillard-Kelly, M.; Baron, R. Deletion of estrogen receptors reveals a regulatory role for estrogen receptors-β in bone remodeling in females but not in males. Bone 2002, 30, 18–25. [Google Scholar] [CrossRef]
- Cadet, E.R.; Gafni, R.I.; McCarthy, E.F.; McCray, D.R.; Bacher, J.D.; Barnes, K.M.; Baron, J. Mechanisms responsible for longitudinal growth of the cortex: Coalescence of trabecular bone into cortical bone. J. Bone Jt. Surg. Am. 2003, 85, 1739–1748. [Google Scholar] [CrossRef]
- Cho, D.-C.; Brennan, H.J.; Johnson, R.W.; Poulton, I.J.; Gooi, J.H.; Tonkin, B.A.; McGregor, N.E.; Walker, E.C.; Handelsman, D.J.; Martin, T.J.; et al. Bone corticalization requires local SOCS3 activity and is promoted by androgen action via interleukin-6. Nat. Commun. 2017, 8, 806. [Google Scholar] [CrossRef] [PubMed]
- Piemontese, M.; Almeida, M.; Robling, A.G.; Kim, H.-N.; Xiong, J.; Thostenson, J.D.; Weinstein, R.S.; Manolagas, S.C.; O’Brien, C.A.; Jilka, R.L. Old age causes de novo intracortical bone remodeling and porosity in mice. JCI Insight 2017, 2, e93771. [Google Scholar] [CrossRef] [PubMed]
- Birkhold, A.I.; Razi, H.; Duda, G.N.; Weinkamer, R.; Checa, S.; Willie, B.M. The Periosteal Bone Surface is Less Mechano-Responsive than the Endocortical. Sci. Rep. 2016, 6, 23480. [Google Scholar] [CrossRef]
- Jilka, R.L. The relevance of mouse models for investigating age-related bone loss in humans. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Hoshi, K.; Ogata, N.; Shimoaka, T.; Terauchi, Y.; Kadowaki, T.; Kenmotsu, S.-I.; Chung, U.-I.; Ozawa, H.; Nakamura, K.; Kawaguchi, H. Deficiency of insulin receptor substrate-1 impairs skeletal growth through early closure of epiphyseal cartilage. J. Bone Miner. Res. 2004, 19, 214–223. [Google Scholar] [CrossRef]
- Staines, K.A.; Madi, K.; Javaheri, B.; Lee, P.D.; Pitsillides, A.A. A Computed Microtomography Method for Understanding Epiphyseal Growth Plate Fusion. Front. Mater. 2018, 4, 48. [Google Scholar] [CrossRef]
- Wang, Z.; Storm, D.R. Extraction of DNA from mouse tails. BioTechniques 2006, 41, 412. [Google Scholar] [CrossRef]
- Takahashi, E.; Miyamoto, N.; Kajiwara, N.; Furuya, K.; Yanai-Taniguchi, K.; Sugiyama, F.; Yagami, K. Expression analysis of Escherichia coli lacZ reporter gene in transgenic mice. Brain Res. Protoc. 2000, 5, 159–166. [Google Scholar] [CrossRef]
- Webb, G.N.; Byrd, R.A. Simultaneous differential staining of cartilage and bone in rodent fetuses: An alcian blue and alizarin red S procedure without glacial acetic acid. Biotech. Histochem. 1994, 69, 181–185. [Google Scholar] [CrossRef]
- Campbell, G.M.; Sophocleous, A. Quantitative analysis of bone and soft tissue by micro-computed tomography: Applications to ex vivo and in vivo studies. Bonekey Rep. 2014, 3, 564. [Google Scholar] [CrossRef] [PubMed]
- Erjavec, I.; Bordukalo-Niksic, T.; Brkljacic, J.; Grcevic, D.; Mokrovic, G.; Kesic, M.; Rogic, D.; Zavadoski, W.; Paralkar, V.M.; Grgurevic, L.; et al. Constitutively Elevated Blood Serotonin Is Associated with Bone Loss and Type 2 Diabetes in Rats. PLoS ONE 2016, 11, e0150102. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.H.D.; Alves, J.M.; Silva, O.L.D.; Silva Junior, N.F.D. Two and three-dimensional morphometric analysis of trabecular bone using X-ray microtomography (µCT). J. Bone Jt. Surg. Am. 2014, 30, 93–101. [Google Scholar] [CrossRef]
- Cardiff, R.D.; Miller, C.H.; Munn, R.J. Manual hematoxylin and eosin staining of mouse tissue sections. Cold Spring Harb. Protoc. 2014, 2014, 655–658. [Google Scholar] [CrossRef] [PubMed]
- Hunziker, E.B.; Schenk, R.K.; Cruz-Orive, L.M. Quantitation of chondrocyte performance in growth-plate cartilage during longitudinal bone growth. J. Bone Jt. Surg Am. 1987, 69, 162–173. [Google Scholar] [CrossRef]
- Ruxton, G.D. The unequal variance t-test is an underused alternative to Student’s t-test and the Mann–Whitney U test. Behav. Ecol. 2006, 17, 688–690. [Google Scholar] [CrossRef]
- Mukaka, M.M. Statistics corner: A guide to appropriate use of correlation coefficient in medical research. Malawi Med. J. 2012, 24, 69–71. [Google Scholar]
| 8 Weeks | 16 Weeks | |||
|---|---|---|---|---|
| WT | Bmp3−/− | WT | Bmp3−/− | |
| M | 20.68 ± 1.49 | 19.30 ± 2.67 | 25.73 ± 0.49 | 23.98 ± 1.91 |
| F | 18.62 ± 1.22 | 18.31 ± 2.67 | 20.31 ± 1.14 | 21.65 ± 3.15 |
| Femur | Tibia | ||||
|---|---|---|---|---|---|
| Cortical Bone | Trabecular Bone | Cortical Bone | Trabecular Bone | ||
| 8 weeks | M | 6.6931 | 2.8748 | 15.7512 | 4.2907 |
| F | 7.1881 | 7.6275 | 26.5651 | 6.6429 | |
| 16 weeks | M | 2.8073 | 2.9811 | 4.065 | 2.0948 |
| F | 3.2661 | 3.1393 | 1.5804 | 3.1762 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banovac, I.; Grgurevic, L.; Rumenovic, V.; Vukicevic, S.; Erjavec, I. BMP3 Affects Cortical and Trabecular Long Bone Development in Mice. Int. J. Mol. Sci. 2022, 23, 785. https://doi.org/10.3390/ijms23020785
Banovac I, Grgurevic L, Rumenovic V, Vukicevic S, Erjavec I. BMP3 Affects Cortical and Trabecular Long Bone Development in Mice. International Journal of Molecular Sciences. 2022; 23(2):785. https://doi.org/10.3390/ijms23020785
Chicago/Turabian StyleBanovac, Ivan, Lovorka Grgurevic, Viktorija Rumenovic, Slobodan Vukicevic, and Igor Erjavec. 2022. "BMP3 Affects Cortical and Trabecular Long Bone Development in Mice" International Journal of Molecular Sciences 23, no. 2: 785. https://doi.org/10.3390/ijms23020785
APA StyleBanovac, I., Grgurevic, L., Rumenovic, V., Vukicevic, S., & Erjavec, I. (2022). BMP3 Affects Cortical and Trabecular Long Bone Development in Mice. International Journal of Molecular Sciences, 23(2), 785. https://doi.org/10.3390/ijms23020785






