Microbial Contributions for Rice Production: From Conventional Crop Management to the Use of ‘Omics’ Technologies
Abstract
1. Introduction
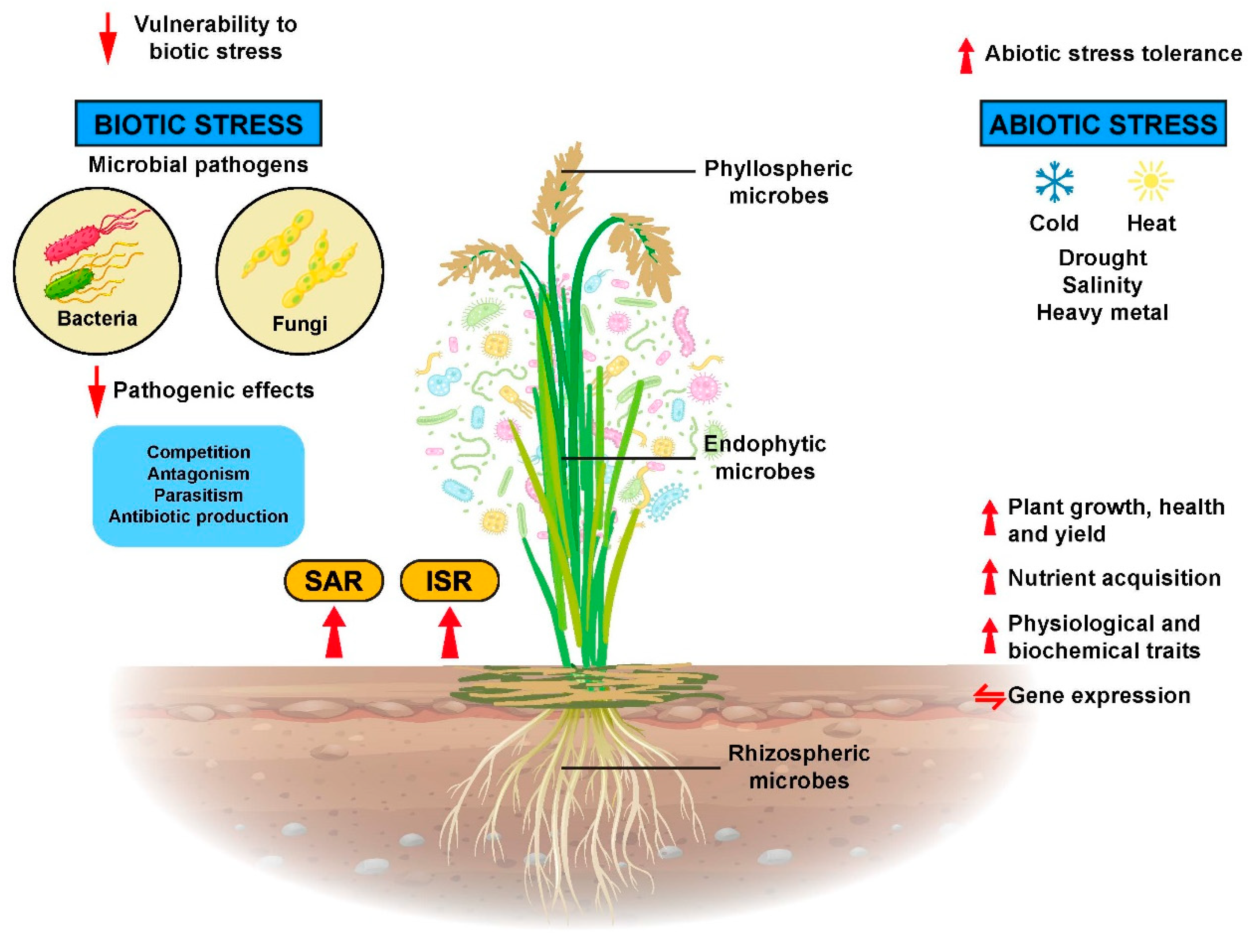
2. The Diverse and Dynamic Structures of Rice Microbiomes
2.1. The Rhizosphere
2.2. The Phyllosphere
2.3. The Endosphere
3. Microbial Contributions for Promoting Rice Growth and Yield: From Earlier Views to the Omics Era
4. Services of Microbes in Protecting Rice Plants against Plant Diseases
4.1. Fungal Diseases
4.2. Bacterial Diseases
5. Microbial Involvement in Conferring Abiotic Stress Tolerance in Rice
5.1. Drought Stress
5.2. Salinity Stress
5.3. Heavy Metal Stress
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- FAO. World Food and Agriculture—Statistical Yearbook 2021; FAO: Rome, Italy, 2021; Available online: https://www.fao.org/3/cb4477en/cb4477en.pdf (accessed on 23 September 2021).
- Yuan, S.; Nie, L.; Wang, F.; Huang, J.; Peng, S. Agronomic performance of inbred and hybrid rice cultivars under simplified and reduced-input practices. Field Crops Res. 2017, 210, 129–135. [Google Scholar] [CrossRef]
- Wu, K.; Wang, S.; Song, W.; Zhang, J.; Wang, Y.; Liu, Q.; Yu, J.; Ye, Y.; Li, S.; Chen, J.; et al. Enhanced sustainable green revolution yield via nitrogen-responsive chromatin modulation in rice. Science 2020, 367, eaaz2046. [Google Scholar] [CrossRef]
- Ray, D.K.; Ramankutty, N.; Mueller, N.D.; West, P.C.; Foley, J.A. Recent patterns of crop yield growth and stagnation. Nat. Commun. 2012, 3, 1293. [Google Scholar] [CrossRef] [PubMed]
- Muehe, E.M.; Wang, T.; Kerl, C.F.; Planer-Friedrich, B.; Fendorf, S. Rice production threatened by coupled stresses of climate and soil arsenic. Nat. Commun. 2019, 10, 4985. [Google Scholar] [CrossRef]
- Hussain, S.; Huang, J.; Huang, J.; Ahmad, S.; Nanda, S. Rice production under climate change: Adaptations and mitigating strategies. In Environment, Climate, Plant and Vegetation Growth; Fahad, S., Ed.; Springer: Cham, Switzerland, 2020; pp. 659–686. [Google Scholar]
- Heong, K.L.; Escalada, M.M.; Chien, H.V.; Delos, R.J.H. Are there productivity gains from insecticide applications in rice production? In Rice Planthoppers: Ecology, Management, Socio Economics and Policy; Heong, K.L., Cheng, J.A., Escalada, M.M., Eds.; Zhejiang University Press: Hangzhou, China; Springer Science+Business Media: Dordrecht, The Netherlands, 2015; pp. 181–192. [Google Scholar]
- Li, S.; Tian, Y.; Wu, K.; Ye, Y.; Yu, J.; Zhang, J.; Liu, Q.; Hu, M.; Li, H.; Tong, Y.; et al. Modulating plant growth-metabolism coordination for sustainable agriculture. Nature 2018, 560, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.I.D.S.; Chaibub, A.A.; Sousa, T.P.; Cortes, M.V.; de Souza, A.C.; da Conceição, E.C.; de Filippi, M.C. Formulations of Pseudomonas fluorescens and Burkholderia pyrrocinia control rice blast of upland rice cultivated under no-tillage system. Biol. Control. 2019, 27, 104153. [Google Scholar] [CrossRef]
- Harman, G.; Khadka, R.; Doni, F.; Uphoff, N. Benefits to plant health and productivity from enhancing plant microbial symbionts. Front. Plant Sci. 2021, 11, 610065. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; White, J.F., Jr.; Arnold, A.E.; Redman, R.S. Fungal endophytes: Diversity and functional roles. New Phytol. 2009, 182, 314–330. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.; Baird, A.; Cho, S.; Gray, Z.; Groover, E.; Harto, R.; Hsieh, M.; Malmberg, K.; Manglona, R.; Mercer, M.; et al. Programming plants for climate resilience through symbiogenics. In Seed Endophytes; Verma, S., White, J., Jr., Eds.; Springer: Cham, Switzerland, 2019; pp. 127–137. [Google Scholar]
- Nosheen, S.; Ajmal, I.; Song, Y. Microbes as biofertilizers, a potential approach for sustainable crop production. Sustainability 2021, 13, 1868. [Google Scholar] [CrossRef]
- Van der Heijden, M.G.; Hartmann, M. Networking in the plant microbiome. PLoS Biol. 2016, 14, e1002378. [Google Scholar] [CrossRef]
- Fitzpatrick, C.R.; Salas-González, I.; Conway, J.M.; Finkel, O.M.; Gilbert, S.; Russ, D. The plant microbiome: From ecology to reductionism and beyond. Ann. Rev. Microbiol. 2020, 74, 81–100. [Google Scholar] [CrossRef] [PubMed]
- Doni, F.; Suhaimi, N.S.M.; Irawan, B.; Mohamed, Z.; Mispan, M.S. Associations of Pantoea with rice plants: As friends or foes? Agriculture 2021, 11, 1278. [Google Scholar] [CrossRef]
- Harman, G.; Uphoff, N. Symbiotic root-endophytic soil microbes improve crop productivity and provide environmental benefits. Scientifica 2019, 9106395, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, N.S.; Doni, F.; Mispan, M.S.; Saiman, M.Z.; Yusuf, Y.M.; Oke, M.A.; Suhaimi, N.S.M. Harnessing Trichoderma in agriculture for productivity and sustainability. Agronomy 2021, 11, 2559. [Google Scholar] [CrossRef]
- Raio, A.; Puopolo, G. Pseudomonas chlororaphis metabolites as biocontrol promoters of plant health and improved crop yield. World J. Microbio. Biotechnol. 2021, 37, 1–8. [Google Scholar] [CrossRef]
- Finkel, O.M.; Castrillo, G.; Paredes, S.H.; González, I.S.; Dangl, J.L. Understanding and exploiting plant beneficial microbes. Curr. Opin. Plant. Biol. 2017, 38, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Stoop, W.A.; Sabarmatee, S.; Sivasubramanian, P.; Ravindra, A.; Sen, D.; Shambu, P.C.; Thakur, A.K. Opportunities for ecological intensification: Lessons and insights from the System of Rice/Crop Intensification—Their implications for agricultural research and development approaches. CAB Rev. 2017, 12, 1–19. [Google Scholar] [CrossRef]
- Ma, Y.; Freitas, H.; Vosatka, M. Beneficial microbes alleviate climatic stresses in plants. Front. Plant Sci. 2019, 10, 595. [Google Scholar] [CrossRef]
- Kim, H.; Lee, Y.H. The rice microbiome: A model platform for crop holobiome. Phytobiomes J. 2020, 4, 5–18. [Google Scholar] [CrossRef]
- Pang, Z.; Zhao, Y.; Xu, P.; Yu, D. Microbial diversity of upland rice roots and their influence on rice growth and drought tolerance. Microorganisms 2020, 8, 1329. [Google Scholar] [CrossRef]
- Ngalimat, M.S.; Mohd Hata, E.; Zulperi, D.; Ismail, S.I.; Ismail, M.R.; Mohd Zainudin, N.A.I. Plant growth-promoting bacteria as an emerging tool to manage bacterial rice pathogens. Microorganisms 2021, 9, 682. [Google Scholar] [CrossRef]
- Prasanna, R.; Sharma, E.; Sharma, P.; Kumar, A.; Kumar, R. Soil fertility and establishment potential of inoculated cyanobacteria in rice crop grown under non-flooded conditions. Paddy Water Environ. 2013, 11, 175–183. [Google Scholar] [CrossRef]
- Diem, G.; Rougier, M.; Hamad-Fares, I.; Balandreau, J.P.; Dommergues, Y.R. Colonization of rice roots by diazotroph bacteria. Ecol. Bull. 1978, 26, 305–311. [Google Scholar]
- Prasanna, R.; Nain, L.; Pandey, A.K.; Saxena, A.K. Microbial diversity and multidimensional interactions in the rice ecosystem. Arch. Agron. Soil Sci. 2012, 58, 723–744. [Google Scholar] [CrossRef]
- Kumar, U.; Shahid, M.; Tripathi, R.; Mohanty, S.; Kumar, A.; Bhattacharyya, P. Variation of functional diversity of soil microbial community in sub-humid tropical rice-rice cropping system under long-term organic and inorganic fertilization. Ecol. Indic. 2017, 73, 536–543. [Google Scholar] [CrossRef]
- Khan, M.I.; Gwon, H.S.; Alam, M.A.; Song, H.J.; Das, S.; Kim, P.J. Short term effects of different green manure amendments on the composition of main microbial groups and microbial activity of a submerged rice cropping system. Appl. Soil Ecol. 2020, 147, 103400. [Google Scholar] [CrossRef]
- Kimura, M.; Asakawa, S. Comparison of community structures of microbiota at main habitats in rice field ecosystems based on phospholipid fatty acid analysis. Biol. Fertil. Soils 2006, 43, 20–29. [Google Scholar] [CrossRef]
- Xu, Y.; Ge, Y.; Song, J.; Rensing, C. Assembly of root-associated microbial community of typical rice cultivars in different soil types. Biol. Fert. Soils 2020, 56, 249–260. [Google Scholar] [CrossRef]
- Liu, L.; Ding, M.; Zhou, L.; Chen, Y.; Li, H.; Zhang, F. Effects of different rice straw on soil microbial community structure. Agron. J. 2021, 113, 794–805. [Google Scholar] [CrossRef]
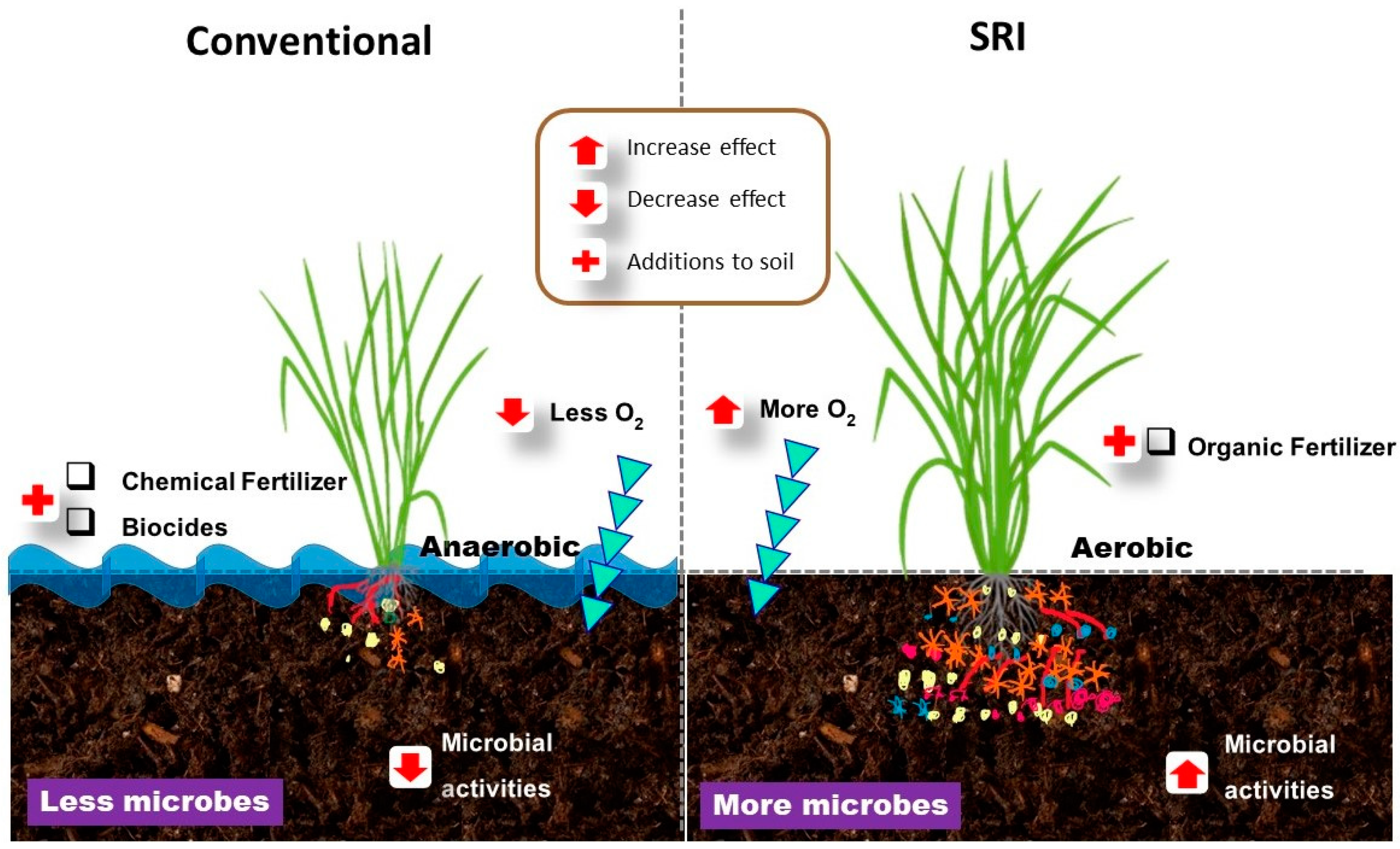
- Doni, F.; Mispan, M.S.; Suhaimi, N.S.M.; Ishak, N.; Uphoff, N. Roles of microbes in supporting sustainable rice production using the system of rice intensification. Appl. Microbiol. Biotechnol. 2019, 103, 5131–5142. [Google Scholar] [CrossRef]
- Chen, X.P.; Zhu, Y.G.; Xia, Y.; Shen, J.P.; He, J.Z. Ammonia-oxidizing archaea: Important players in paddy rhizosphere soil? Environ. Microbiol. 2008, 10, 1978–1987. [Google Scholar] [CrossRef]
- Wang, Y.; Ke, X.; Wu, L.; Lu, Y. Community composition of ammonia-oxidizing bacteria and archaea in rice field soil as affected by nitrogen fertilization. Syst. Appl. Microbiol. 2009, 32, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Salmaninezhad, F.; Mostowfizadeh-Ghalamfarsa, R. Three new Pythium species from rice paddy fields. Mycologia 2019, 111, 274–290. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Q.; Liu, Y.; Zhang, A.; Pan, G.; Li, L.; Zhang, X.; Song, C.L.; Jin, Z. Variation of bacterial and fungal community structures in the rhizosphere of hybrid and standard rice cultivars and linkage to CO2 flux. FEMS Microbiol. Ecol. 2011, 78, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.J.; Liang, Y.T.; Li, C.M.; Wang, F.; Sui, Y.Y.; Suvannang, N.; Zhou, J.Z.; Sun, B. Crop rotations alter bacterial and fungal diversity in paddy soils across East Asia. Soil Biol. Biochem. 2016, 95, 250–261. [Google Scholar] [CrossRef]
- Yuan, C.L.; Zhang, L.M.; Wang, J.T.; Hu, H.W.; Shen, J.P.; Cao, P.; He, J.Z. Distributions and environmental drivers of archaea and bacteria in paddy soils. J. Soils Sediments 2019, 19, 23–37. [Google Scholar] [CrossRef]
- Thapa, S.; Ranjan, K.; Ramakrishnan, B.; Velmourougane, K.; Prasanna, R. Influence of fertilizers and rice cultivation methods on the abundance and diversity of phyllosphere microbiome. J. Basic Microbiol. 2018, 58, 172–186. [Google Scholar] [CrossRef]
- Klinnawee, L.; Noirungsee, N.; Nopphakat, K.; Runsaeng, P.; Chantarachot, T. Flooding overshadows phosphorus availability in controlling the intensity of arbuscular mycorrhizal colonization in Sangyod Muang Phatthalung lowland indica rice. ScienceAsia 2021, 47, 202–210. [Google Scholar] [CrossRef]
- Somenahally, A.C.; Hollister, E.B.; Loeppert, R.H.; Yan, W.; Gentry, T.J. Microbial communities in rice rhizosphere altered by intermittent and continuous flooding in fields with long-term arsenic application. Soil Biol. Biochem. 2011, 43, 1220–1228. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, J.T.; Zhang, Z.F.; Li, W.; Chen, W.; Cai, L. Microbiota in the rhizosphere and seed of rice from China, with reference to their transmission and biogeography. Front. Microbiol. 2020, 11, 995. [Google Scholar] [CrossRef]
- Edwards, J.; Johnson, C.; Santos-Medellín, C.; Lurie, E.; Podishetty, N.K.; Bhatnagar, S.; Eisen, J.A.; Sundaresan, V. Structure, variation, and assembly of the root-associated microbiomes of rice. Proc. Natl. Acad. Sci. USA 2015, 112, E911–E920. [Google Scholar] [CrossRef] [PubMed]
- Rajkishore, S.K.; Doraisamy, P.; Subramanian, K.S.; Maheswari, M. Methane emissions patterns and their associated microflora with SRI and conventional systems of rice cultivation in Tamil Nadu, India. Taiwan Water Conserv. 2013, 61, 126–134. [Google Scholar]
- Venturi, V.; Keel, C. Signaling in the rhizosphere. Trends Plant Sci. 2016, 21, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.F.; Robert, C.A.M.; Cadot, S.; Zhang, X.; Ye, M.; Li, B.B. Root exudate metabolites drive plant-soil feedbacks on growth and defence by shaping the rhizosphere microbiota. Nat. Commun. 2018, 9, 2738. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Sun, Y.; Tian, L.; Ji, L.; Luo, S. The structure of rhizosphere fungal communities of wild and domesticated rice: Changes in diversity and co-occurrence patterns. Front. Microbiol. 2021, 12, 45. [Google Scholar] [CrossRef]
- Watanarojanaporn, N.; Boonkerd, N.; Tittabutr, P.; Longtonglang, A.; Young, J.P.W.; Teaumroong, N. Effect of rice cultivation systems on indigenous arbuscular mycorrhizal fungal community structure. Microbes Environ. 2013, 28, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, R.; Jaiswal, P.; Nayak, S.; Sood, A.; Kaushik, B.D. Cyanobacterial diversity in the rhizosphere of rice and its ecological significance. Indian J. Microbiol. 2009, 49, 89–97. [Google Scholar] [CrossRef]
- Anas, I.; Rupela, O.P.; Thiyagarajan, T.M.; Uphoff, N. A review of studies on SRI effects on beneficial organisms in rice soil rhizospheres. Paddy Water Environ. 2011, 9, 53–64. [Google Scholar] [CrossRef]
- Mwajita, M.R.; Murage, H.; Tani, A.; Kahangi, E.M. Evaluation of rhizosphere, rhizoplane and phyllosphere bacteria and fungi isolated from rice in Kenya for plant growth promoters. Springerplus 2013, 2, 606. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Kumar, R.; Humayun, P.; Srinivas, V.; Kumari, B. Assessment of different methods of rice (Oryza sativa. L) cultivation affecting growth parameters, soil chemical, biological, and microbiological properties, water saving, and grain yield in rice–rice system. Paddy Water Environ. 2014, 12, 79–87. [Google Scholar] [CrossRef]
- Leveau, J.H. A brief from the leaf: Latest research to inform our understanding of the phyllosphere microbiome. Curr. Opin. Microbiol. 2019, 49, 41–49. [Google Scholar] [CrossRef]
- Venkatachalam, S.; Ranjan, K.; Prasanna, R.; Ramakrishnan, B.; Thapa, S.; Kanchan, A. Diversity and functional traits of culturable microbiome members, including cyanobacteria in the rice phyllosphere. Plant Biol. 2016, 18, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Brettell, L.E.; Singh, B. Linking the phyllosphere microbiome to plant health. Trends Plant Sci. 2020, 25, 841–844. [Google Scholar] [CrossRef] [PubMed]
- Roman-Reyna, V.; Pinili, D.; Borja, F.N.; Quibod, I.L.; Groen, S.C.; Alexandrov, N. Characterization of the leaf microbiome from whole-genome sequencing data of the 3000 rice genomes project. Rice 2020, 13, 72. [Google Scholar] [CrossRef] [PubMed]
- Mano, H.; Morisaki, H. Endophytic bacteria in the rice plant. Microbes Environ. 2008, 23, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Adeleke, B.S.; Babalola, O.O. Roles of plant endosphere microbes in agriculture—A review. J. Plant Growth Regul. 2021. [Google Scholar] [CrossRef]
- Chebotar, V.K.; Malfanova, N.V.; Shcherbakov, A.V.; Ahtemova, G.A.; Borisov, A.Y.; Lugtenberg, B.; Tikhonovich, I.A. Endophytic bacteria in microbial preparations that improve plant development (review). Appl. Biochem. Microbiol. 2015, 51, 271–277. [Google Scholar] [CrossRef]
- Loaces, I.; Ferrando, L.; Scavino, A.F. Dynamics, diversity and function of endophytic siderophore-producing bacteria in rice. Microb. Ecol. 2010, 61, 606–618. [Google Scholar] [CrossRef]
- Zhong, Y.; Hu, J.; Xia, Q.; Zhang, S.; Li, X. Soil microbial mechanisms promoting ultrahigh rice yield. Soil Biol. Biochem. 2020, 143, 107741. [Google Scholar] [CrossRef]
- Hardoim, P.R.; van Overbeek, L.S.; van Elsas, J.D. Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol. 2008, 16, 463–471. [Google Scholar] [CrossRef]
- Torres-Cortés, G.; Bonneau, S.; Bouchez, O.; Genthon, C.; Briand, M.; Jacques, M.A.; Barret, M. Functional microbial features driving community assembly during seed germination and emergence. Front. Plant Sci. 2018, 9, 902. [Google Scholar] [CrossRef]
- Rodríguez, C.E.; Antonielli, L.; Mitter, B.; Trognitz, F.; Sessitsch, A. Heritability and functional importance of the Setaria viridis bacterial seed microbiome. Phytobiomes J. 2020, 4, 40–52. [Google Scholar] [CrossRef]
- Watanabe, I.; Barraquio, W.L.; de Guzman, M.; Cabrera, D.A. Nitrogen fixation (acetylene reduction) activity and population of aerobic heterotrophic nitrogen-fixing bacteria associated with wetland rice. Appl. Environ. Microbiol. 1979, 37, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Rao, V.R.; Jena, P.K.; Adhya, T.K. Inoculation of rice with nitrogen-fixing bacteria—problems and perspectives. Biol. Fertil. Soils 1987, 4, 21–26. [Google Scholar]
- Watanabe, I.; Lin, C. Response of wetland rice to inoculation with Azospirillum lipoferum and Pseudomonas sp. Soil Sci. Plant Nutr. 1984, 30, 117–124. [Google Scholar] [CrossRef]
- Roger, P.A. Biological N2-fixation and its management in wetland rice cultivation. Fertil. Res. 1995, 42, 261–276. [Google Scholar] [CrossRef]
- Yanni, Y.G.; Rizk, R.Y.; Corich, V.; Squartini, A.; Ninke, K.; Philip-Hollingsworth, S. Natural endophytic association between Rhizobium leguminosarum bv. trifolii and rice roots and assessment of its potential to promote rice growth. Plant Soil 1997, 194, 99–114. [Google Scholar] [CrossRef]
- Biswas, J.C.; Ladha, J.K.; Dazzo, F.B.; Yanni, Y.G.; Rolfe, B.G. Rhizobial inoculation influences seedling vigor and yield of rice. J. Agron. 2000, 92, 880–886. [Google Scholar] [CrossRef]
- Yanni, Y.; Rizk, R.Y.; Abd El-Fattah, F.K. The beneficial plant growth-promoting association of Rhizobium leguminosarum bv. trifolii with rice roots. Funct. Plant Biol. 2001, 28, 845–870. [Google Scholar] [CrossRef]
- Chi, F.; Shen, S.H.; Cheng, H.; Jing, Y.X.; Yanni, Y.G.; Dazzo, F.B. Ascending migration of endophytic rhizobia, from roots to leaves, inside rice plants and assessment of benefits to rice growth physiology. Appl. Environ. Microbiol. 2005, 71, 7271–7278. [Google Scholar] [CrossRef]
- Sasaki, K.; Ikeda, S.; Eda, S.; Mitsui, H.; Hanzawa, E.; Kisara, C. Impact of plant genotype and nitrogen level on rice growth response to inoculation with Azospirillum sp. strain B510 under paddy field conditions. Soil Sci. Plant Nutr. 2010, 56, 636–644. [Google Scholar] [CrossRef]
- Uphoff, N.; Chi, F.; Dazzo, F.B.; Rodriguez, R.J. Soil fertility as a contingent rather than inherent characteristic: Considering the contributions of crop–symbiotic soil biota. In Principles of Sustainable Soil Management in Agroecosystems; Lal, R., Stewart, B., Eds.; Taylor & Francis: Boca Raton, FL, USA, 2013; pp. 141–166. [Google Scholar]
- Mondal, S.; Halder, S.K.; Yadav, A.N.; Mondal, K.C. Microbial consortium with multifunctional plant growth-promoting attributes: Future perspective in agriculture. In Advances in Plant Microbiome and Sustainable Agriculture: Microorganisms for Sustainability; Yadav, A., Rastegari, A., Yadav, N., Kour, D., Eds.; Springer: Singapore, 2020; pp. 219–258. [Google Scholar]
- Jha, M.; Chourasia, S.; Sinha, S. Microbial consortium for sustainable rice production. Agroecol. Sustain. Food Syst. 2013, 37, 340–362. [Google Scholar] [CrossRef]
- Prasanna, R.; Joshi, M.; Rana, A.; Shivay, Y.S.; Nain, L. Influence of co-inoculation of bacteria-cyanobacteria on crop yield and C–N sequestration in soil under rice crop. World J. Microbiol. Biotechnol. 2012, 28, 1223–1235. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Gupta, R.; Kwon, S.J.; Wang, Y.; Agrawal, G.K.; Rakwal, R. Transcriptomic analysis of Oryza sativa leaves reveals key changes in response to Magnaporthe oryzae MSP1. Plant Pathol. J. 2018, 34, 257. [Google Scholar] [CrossRef] [PubMed]
- Chi, F.; Yang, P.; Han, F.; Jing, Y.; Shen, S. Proteomic analysis of rice seedlings infected by Sinorhizobium meliloti 1021. Proteomics 2010, 10, 1861–1874. [Google Scholar] [CrossRef]
- Wu, Q.; Peng, X.; Yang, M.; Zhang, W.; Dazzo, F.B.; Uphoff, N.; Jing, Y.; Shen, S. Rhizobia promote the growth of rice shoots by targeting cell signalling, division and expansion. Plant Molec. Biol. 2018, 97, 507–523. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.J.; Lu, F.; Yang, Y.; Sun, K.; Zhu, Q.; Xu, F.J.; Zang, W.; Dai, C.C. Benefits of endophytic fungus Phomopsis liquidambaris inoculation for improving mineral nutrition, quality, and yield of rice grains under low nitrogen and phosphorus condition. J. Plant Growth Regul. 2021, 1–15. [Google Scholar] [CrossRef]
- Harman, G.E.; Doni, F.; Khadka, R.B.; Uphoff, N. Endophytic strains of Trichoderma increase plants’ photosynthetic capability. J. Appl. Microbiol. 2021, 130, 529–546. [Google Scholar] [CrossRef]
- Anhar, A.; Putri, D.H.; Advinda, L.; Atika, V.; Amimi, S.; Aldo, W.; Ruchi, W. Molecular characterization of Trichoderma strains from West Sumatera, Indonesia and their beneficial effects on rice seedling growth. J. Crop Sci. Biotechnol. 2021, 24, 441–448. [Google Scholar] [CrossRef]
- Khadka, R.B.; Uphoff, N. Effects of Trichoderma seedling treatment with System of Rice Intensification management and with conventional management of transplanted rice. PeerJ 2019, 7, e5877. [Google Scholar] [CrossRef]
- Doni, F.; Che Radziah, C.M.Z.; Anizan, I.; Norela, S.; Fathurrahman, F.; Uphoff, N.; Wan Mohtar, W.Y. Relationships observed between Trichoderma inoculation and characteristics of rice grown under System of Rice Intensification (SRI) vs. conventional methods of cultivation. Symbiosis 2017, 72, 45–59. [Google Scholar] [CrossRef]
- Doni, F.; Fathurrahman, F.; Mispan, M.S.; Suhaimi, N.S.M.; Yusoff, W.M.W.; Uphoff, N. Transcriptomic profiling of rice seedlings inoculated with the symbiotic fungus Trichoderma asperellum SL2. J. Plant Growth Regul. 2019, 38, 1507–1515. [Google Scholar] [CrossRef]
- Doni, F.; Isahak, A.; Yusoff, W.M.W.; Uphoff, N. Physiological Effects and Transcriptomic Profiling of Rice Plant-Microbe Interactions in System of Rice Intensification (SRI) Management. In Proceedings of the 5th International Rice Congress, Singapore, 16 October 2018; Available online: https://www.slideshare.net/SRI.CORNELL/1809-physiological-effects-and-transcriptomic-profiling-of-rice-plant-microbe-interatctions-in-system-of-rice-intensification-sri-management (accessed on 23 September 2021).
- Pati, B.R. Effect of spraying nitrogen fixing phyllospheric bacterial isolates on rice plants. Zentralbl. Mikrobiol. 1992, 147, 441–446. [Google Scholar] [CrossRef]
- Biswas, J.C.; Ladha, J.K.; Dazzo, F.B. Rhizobia inoculation improves nutrient uptake and growth of lowland rice. Soil Sci. Soc. Am. J. 2000, 64, 1644–1650. [Google Scholar] [CrossRef]
- Alam, M.S.; Cui, Z.J.; Yamagishi, T.; Ishii, R. Grain yield and related physiological characteristics of rice plants (Oryza sativa L.) inoculated with free-living rhizobacteria. Plant Prod. Sci. 2001, 4, 126–130. [Google Scholar] [CrossRef]
- Cuevas, V.C. Soil inoculation with Trichoderma pseudokoningii Rifai enhances yield of rice. Philip. J. Sci. 2006, 135, 31–37. [Google Scholar]
- Dhar, D.W.; Prasanna, R.; Singh, B.V. Comparative performance of three carrier-based blue green algal biofertilizers for sustainable rice cultivation. J. Sust. Agric. 2007, 30, 41–50. [Google Scholar] [CrossRef]
- Razie, F.; Anas, I. Effect of Azotobacter and Azospirillum on growth and yield of rice grown on tidal swamp rice fields in south Kalimantan. J. Tanah Lingk. 2008, 10, 41–45. [Google Scholar] [CrossRef]
- Isawa, T.; Yasuda, M.; Awazaki, H.; Minamisawa, K.; Shinozaki, S.; Nakashita, H. Azospirillum sp. strain B510 enhances rice growth and yield. Microbes Environ. 2010, 25, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Seenivasan, N. Efficacy of Pseudomonas fluorescens and Paecilomyces lilacinus against Meloidogyne graminicola infesting rice under system of rice intensification. Arch. Phytopathol. Plant. Prot. 2011, 44, 1467–1482. [Google Scholar] [CrossRef]
- Lavakush, L.; Yadav, J.; Verma, J.P.; Jaiswal, D.K.; Kumar, A. Evaluation of PGPR and different concentration of phosphorus level on plant growth, yield and nutrient content of rice (Oryza sativa). Ecol. Eng. 2014, 62, 123–128. [Google Scholar] [CrossRef]
- Duy, M.; Hoi, N.; Ve, N.; Thuc, L.; Trang, N. Influence of cellulomonas Flavigena, Azospirillum sp. and Psudomonas sp. on rice growth and yield grown in submerged soil amended in rice straw. Recent Trends PGPR Res. Sust. Crop. Product. 2016, 8, 238–242. [Google Scholar]
- Doni, F.; Zain, C.R.; Isahak, A.; Fathurrahman, F.; Anhar, A.; Mohamad, W.N.; Yusoff, W.M.; Uphoff, N. A simple, efficient, and farmer-friendly Trichoderma-based biofertilizer evaluated with the SRI rice management system. Org. Agric. 2018, 8, 207–223. [Google Scholar] [CrossRef]
- Nascente, A.S.; Lanna, A.C.; de Sousa, T.P.; Chaibub, A.A.; de Souza, A.C.; de Filippi, M.C. N fertilizer dose-dependent efficiency of Serratia spp. for improving growth and yield of upland rice (Oryza sativa L.). Int. J. Plant Prod. 2019, 13, 217–226. [Google Scholar] [CrossRef]
- Ríos-Ruiz, W.F.; Torres-Chávez, E.E.; Torres-Delgado, J.; Rojas-García, J.C.; Bedmar, E.J.; Valdez-Nuñez, R.A. Inoculation of bacterial consortium increases rice yield (Oryza sativa L.) reducing applications of nitrogen fertilizer in San Martin region, Peru. Rhizosphere 2020, 14, 100200. [Google Scholar] [CrossRef]
- Fitriatin, B.N.; Sofyan, E.T.; Turmuktini, T. Increasing soil P and yield of upland rice through application phosphate solubilizing microbes. Haya Saudi J. Life. Sci. 2021, 6, 163–167. [Google Scholar]
- Mhlongo, M.I.; Piater, L.A.; Madala, N.E.; Labuschagne, N.; Dubery, I.A. The chemistry of plant–microbe interactions in the rhizosphere and the potential for metabolomics to reveal signaling related to defense priming and induced systemic resistance. Front. Plant Sci. 2018, 9, 112. [Google Scholar] [CrossRef]
- Rahman, S.F.S.A.; Singh, E.; Pieterse, C.M.J.; Schenk, P.M. Emerging microbial biocontrol strategies for plant pathogens. Plant Sci. 2018, 267, 102–111. [Google Scholar] [CrossRef]
- Mathivanan, N.; Prabavathy, V.R.; Vijayanandraj, V.R. Application of talc formulations of Pseudomonas fluorescens Migula and Trichoderma viride Pers. Ex S.F. gray decrease the sheath blight disease and enhance the plant growth and yield in rice. J. Phytopathol. 2005, 153, 697–701. [Google Scholar] [CrossRef]
- França, S.K.S.; Cardoso, A.F.; Lustosa, D.C.; Ramos, M.L.S.; Filippi, M.C.; Silva, G.B. Biocontrol of sheath blight by Trichoderma asperellum in tropical lowland rice. Agron. Sustain. Dev. 2015, 35, 317–324. [Google Scholar] [CrossRef]
- Singh, P.K.; Singh, A.K.; Singh, H.B.; Dhakal, B.K. Biological control of rice blast disease with Trichoderma harzianum in direct-seeded rice under medium low land rainfed conditions. Environ. Ecol. 2012, 30, 834–837. [Google Scholar]
- Abdel-Fattah, G.M.; Shabana, Y.M.; Ismail, A.E.; Rashad, Y.M. Trichoderma harzianum: A biocontrol agent against Bipolaris oryzae. Mycopathology 2007, 164, 81–89. [Google Scholar] [CrossRef]
- Harman, G.E. Overview of mechanisms and uses of Trichoderma spp. Phytopathology 2006, 96, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; Cortés-Penagos, C.; López-Bucio, J. Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol. 2009, 149, 1579–1592. [Google Scholar] [CrossRef]
- Stockwell, V.O.; Stack, J.P. Using Pseudomonas spp. for integrated biological control. Phytopathology 2007, 97, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Oni, F.E.; Olorunleke, O.F.; Höfte, M. Phenazines and cyclic lipopeptides produced by Pseudomonas sp. CMR12a are involved in the biological control of Pythium myriotylum on cocoyam (Xanthosoma sagittifolium). Biol. Control. 2019, 129, 109–114. [Google Scholar] [CrossRef]
- Nandakumar, R.; Babu, S.; Viswanathan, R.; Raguchander, T.; Samiyappan, R. Induction of systemic resistance in rice against sheath blight disease by Pseudomonas fluorescens. Soil Biol. Biochem. 2001, 33, 603–612. [Google Scholar] [CrossRef]
- Patel, A.; Kumar, A.; Sheoran, N.; Kumar, M.; Sahu, K.P. Antifungal and defense elicitor activities of pyrazines identified in endophytic Pseudomonas putida BP25 against fungal blast incited by Magnaporthe oryzae in rice. J. Plant Dis. Prot. 2021, 128, 261–272. [Google Scholar] [CrossRef]
- Radjacommare, R.; Nandakumar, R.; Kandan, A.; Suresh, S.; Bharathi, M.; Raguchander, T.; Samiyappan, R. Pseudomonas fluorescens based bioformulation for the management of sheath blight and leaffolder in rice. Crop Prot. 2002, 21, 671–677. [Google Scholar] [CrossRef]
- Leelasuphakul, W.; Sivanunsakul, P.; Phongpaichit, S. Purification, characterization and synergistic activity of β-1,3-glucanase and antibiotic extract from an antagonistic Bacillus subtilis NSRS 89-24 against rice blast and sheath blight. Enzyme Microb. Technol. 2006, 38, 990–997. [Google Scholar] [CrossRef]
- Durgadevi, D.; Harish, S.; Manikandan, R.; Prabhukarthikeyan, S.R.; Alice, D.; Raguchander, T. Proteomic profiling of defense/resistant genes induced during the tripartite interaction of Oryza sativa, Rhizoctonia solani AG1-1A, and Bacillus subtilis against rice sheath blight. Physiol. Mol. Plant Pathol. 2021, 115, 101669. [Google Scholar] [CrossRef]
- Vidhyasekaran, P.; Rabindran, R.; Muthamilan, R.; Nayar, M.; Rajappan, K.; Subramanian, K.; Vasumathi, K. Development of powder formulation of Pseudomonas fluorescens for control of rice blast. Plant Pathol. 1997, 46, 291–297. [Google Scholar] [CrossRef]
- Someya, N.; Nakajima, M.; Watanabe, K.; Hibi, T.; Akutsu, K. Potential of Serratia marcescens strain B2 for biological control of rice sheath blight. Biocontrol Sci. Technol. 2005, 15, 105–109. [Google Scholar] [CrossRef]
- Yang, D.; Wang, B.; Wang, J.; Chen, Y.; Zhou, M. Activity and efficacy of Bacillus subtilis strain NJ-18 against rice sheath blight and Sclerotinia stem rot of rape. Biol. Control. 2009, 51, 61–65. [Google Scholar] [CrossRef]
- Campos-Soriano, L.; Garcia-Martinez, J.; San, S.B. The arbuscular mycorrhizal symbiosis promotes the systemic induction of regulatory defence-related genes in rice leaves and confers resistance to pathogen infection. Mol. Plant Pathol. 2012, 13, 579–592. [Google Scholar] [CrossRef]
- Hossain, M.T.; Khan, A.; Chung, E.J.; Rashid, M.H.; Chung, Y.R. Biological control of rice bakanae by an endophytic Bacillus oryzicola YC7007. Plant Pathol. J. 2016, 32, 228–241. [Google Scholar] [CrossRef]
- Shao, Z.; Li, Z.; Fu, Y.; Wen, Y.; Wei, S. Induction of defense responses against Magnaporthe oryzae in rice seedling by a new potential biocontrol agent Streptomyces JD211. J. Basic Microbiol. 2018, 58, 686–697. [Google Scholar] [CrossRef]
- Chaibub, A.A.; Carvalho, J.C.B.; Silva, C.S.; Collevatti, R.G. Defence responses in rice plants in prior and simultaneous applications of Cladosporium sp. during leaf blast suppression. Environ. Sci. Pollut. Res. 2016, 23, 21554–21564. [Google Scholar] [CrossRef] [PubMed]
- Chaibub, A.A.; de Sousa, T.P.; de Araújo, L.G.; de Filippi, M.C. Cladosporium cladosporioides C24G modulates gene expression and enzymatic activity during leaf blast suppression in rice plants. J. Plant Growth Regul. 2020, 39, 1140–1152. [Google Scholar] [CrossRef]
- Chaibub, A.A.; de Sousa, T.P.; de Oliveira, M.I.; Arriel-Elias, M.T.; de Araújo, L.G.; de Filippi, M.C. Efficacy of Cladosporium cladosporioides C24G as a multifunctional agent in upland rice in agroecological systems. Int. J. Plant Prod. 2020, 14, 463–474. [Google Scholar] [CrossRef]
- Zhu, H.; Zhou, H.; Ren, Z.; Liu, E. Control of Magnaporthe oryzae and rice growth promotion by Bacillus subtilis JN005. J. Plant Growth Regul. 2021, 1–9. [Google Scholar] [CrossRef]
- Abbas, A.; Fu, Y.; Qu, Z.; Zhao, H.; Sun, Y. Isolation and evaluation of the biocontrol potential of Talaromyces spp. against rice sheath blight guided by soil microbiome. Environ. Microbiol. 2021, 23, 5946–5961. [Google Scholar] [CrossRef]
- Chukwu, S.C.; Rafii, M.Y.; Ramlee, S.; Ismail, S.I. Bacterial leaf blight resistance in rice: A review of conventional breeding to molecular approach. Mol. Biol. Rep. 2019, 46, 1519–1532. [Google Scholar] [CrossRef]
- Wu, L.M.; Wu, H.J.; Chen, L.; Yu, X.F.; Borriss, R.; Gao, X.W. Difficidin and bacilysin from Bacillus amyloliquefaciens FZB42 have antibacterial activity against Xanthomonas oryzae rice pathogens. Sci. Rep. 2015, 5, 12975. [Google Scholar] [CrossRef] [PubMed]
- Gangwar, G.P.; Sinha, A.P. Evaluation of Trichoderma spp. and fluorescent pseudomonads for the management of bacterial leaf blight of rice. Indian Phytopath. 2012, 65, 89–91. [Google Scholar]
- Jambhulkar, P.P.; Sharma, P.; Manokaran, R.; Lakshman, D.K.; Rokadia, P.; Jambhulkar, N. Assessing synergism of combined applications of Trichoderma harzianum and Pseudomonas fluorescens to control blast and bacterial leaf blight of rice. Eur. J. Plant Pathol. 2018, 152, 747–757. [Google Scholar] [CrossRef]
- Shrestha, B.K.; Karki, H.S.; Groth, D.E.; Jungkhun, N.; Ham, J.H. Biological control activities of rice-associated Bacillus sp. strains against sheath blight and bacterial panicle blight of rice. PLoS ONE 2016, 11, e0146764. [Google Scholar]
- Pedraza-Herrera, L.A.; Bautista, J.P.; Cruz-Ramírez, C.A.; Uribe-Vélez, D. IBUN2755 Bacillus strain controls seedling root and bacterial panicle blight caused by Burkholderia glumae. Biol. Control. 2021, 153, 104494. [Google Scholar] [CrossRef]
- Chung, E.J.; Hossain, M.T.; Khan, A.; Kim, K.H.; Jeon, C.O. Bacillus oryzicola sp. nov., an endophytic bacterium isolated from the roots of rice with antimicrobial, plant growth promoting, and systemic resistance inducing activities in rice. Plant Pathol. J. 2015, 31, 152–164. [Google Scholar] [CrossRef] [PubMed]
- Elshakh, A.S.; Anjum, S.I.; Qiu, W.; Almoneafy, A.A.; Li, W.; Yang, Z.; Cui, Z.Q.; Li, B.; Sun, G.C.; Xie, G.L. Controling and defence-related mechanisms of Bacillus strains against bacterial leaf blight of rice. J. Phytopathol. 2016, 164, 534–546. [Google Scholar] [CrossRef]
- Yasmin, S.; Hafeez, F.Y.; Mirza, M.S.; Rasul, M.; Arshad, H.M.; Zubair, M.; Iqbal, M. Biocontrol of bacterial leaf blight of rice and profiling of secondary metabolites produced by rhizospheric Pseudomonas aeruginosa BRp3. Front. Microbiol. 2017, 8, 1895. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Moreno, Z.R.; Vinchira-Villarraga, D.M.; Vergara-Morales, D.I.; Castellanos, L. Plant-growth promotion and biocontrol properties of three Streptomyces spp. isolates to control bacterial rice pathogens. Front. Microbiol. 2019, 10, 290. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Chatterjee, A.; Das, S. Synergistic consortium of beneficial microorganisms in rice rhizosphere promotes host defense to blight-causing Xanthomonas oryzae pv. oryzae. Planta 2020, 252, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Saikia, K.; Bora, L.C. Exploring actinomycetes and endophytes of rice ecosystem for induction of disease resistance against bacterial blight of rice. Eur. J. Plant. Pathol. 2021, 159, 67–79. [Google Scholar] [CrossRef]
- Compant, S.; Samad, A.; Faist, H. A review on the plant microbiome: Ecology, functions, and emerging trends in microbial application. J. Adv. Res. 2019, 19, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Shukla, N.; Awasthi, R.P.; Rawat, L.; Kumar, J. Biochemical and physiological responses of rice (Oryza sativa L.) as influenced by Trichoderma harzianum under drought stress. Plant Physiol. Biochem. 2012, 54, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Ansari, M.W.; Tula, S.; Yadav, S.; Sahoo, R.K.; Shukla, N. Dose-dependent response of Trichoderma harzianum in improving drought tolerance in rice genotypes. Planta 2016, 243, 1251–1264. [Google Scholar] [CrossRef]
- Singh, D.P.; Singh, V.; Gupta, V.K.; Shukla, R.; Prabha, R.; Sarma, B.K.; Patel, J.S. Microbial inoculation in rice regulates antioxidative reactions and defense-related genes to mitigate drought stress. Sci. Rep. 2020, 10, 4818. [Google Scholar] [CrossRef]
- Bashyal, B.M.; Parmar, P.; Zaidi, N.W.; Aggarwal, R. Molecular programming of drought-challenged Trichoderma harzianum-bioprimed rice (Oryza sativa L.). Front. Microbiol. 2021, 12, 655165. [Google Scholar] [CrossRef]
- Prathuangwong, S.; Chuaboon, W.; Chatnaparat, T.; Kladsuwan, L.; Choorin, M.; Kasem, S. Induction of disease and drought resistance in rice by Pseudomonas fluorescens SP007s. CMU J. Nat. Sci. Special Issue Agric. Nat. Res. 2012, 11, 45–55. [Google Scholar]
- Saakre, M.; Baburao, T.M.; Salim, A.P.; Ffancies, R.M.; Achuthan, V.P.; Thomas, G.; Sivarajan, S.R. Identification and characterization of genes responsible for drought tolerance in rice mediated by Pseudomonas Fluorescens. Rice Sci. 2017, 24, 291–298. [Google Scholar] [CrossRef]
- Tiwari, S.; Prasad, V.; Chauhan, P.S.; Lata, C. Bacillus amyloliquefaciens confers tolerance to various abiotic stresses and modulates plant response to phytohormones through osmoprotection and gene expression regulation in rice. Front. Plant Sci. 2017, 8, 1510. [Google Scholar] [CrossRef]
- Ruiz-Sanchez, M.; Armada, E.; Munoz, Y.; Garcia de Salamone, I.E.; Aroca, R.; Ruiz-Lozano, J.M.; Azcon, R. Azospirillum and arbuscular mycorrhizal colonization enhance rice growth and physiological traits under well-watered and drought conditions. J. Plant Physiol. 2011, 168, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.J.; Woodward, C.J.; Redman, R.S. Fungal influence on plant tolerance to stress. In Biocomplexity of Plant–Fungal Interactions; Southworth, D., Ed.; Wiley-Blackwell: Oxford, UK, 2012; pp. 155–163. [Google Scholar]
- Redman, R.S.; Rodriguez, R.J. The symbiogenic tango: Achieving climate-resilient crops via mutualistic plant-fungal relationships. In Functional Importance of the Plant Microbiome; Doty, S.L., Ed.; Springer: Cham, Switzerland, 2017; pp. 71–87. [Google Scholar]
- Rawat, L.; Singh, Y.; Shukla, N.; Kumar, J. Seed biopriming with salinity tolerant isolates of Trichoderma harzianum alleviates salt stress in rice: Growth, physiological and biochemical characteristics. J. Plant Pathol. 2012, 94, 353–365. [Google Scholar]
- Yasmeen, R.; Siddiqui, Z. Physiological responses of crop plants against Trichoderma harzianum in saline environment. Acta Bot. Croat. 2017, 76, 154–162. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, S.; Mukherjee, A.; Rastogi, R.P.; Verma, J.P. Salt-tolerant plant growth-promoting Bacillus pumilus strain JPVS11 to enhance plant growth attributes of rice and improve soil health under salinity stress. Microbiol. Res. 2021, 242, 126616. [Google Scholar] [CrossRef] [PubMed]
- Jogawat, A.; Vadassery, J.; Verma, N.; Oelmüller, R. PiHOG1, a stress regulator MAP kinase from the root endophyte fungus Piriformospora indica, confers salinity stress tolerance in rice plants. Sci. Rep. 2016, 6, 36765. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Lei, P.; Wang, Q.; Ma, J.; Zhan, Y.; Jiang, K.; Xu, Z.; Xu, H. The endophyte Pantoea alhagi NX-11 alleviates salt stress damage to rice seedlings by secreting exopolysaccharides. Front. Microbiol. 2020, 10, 3112. [Google Scholar] [CrossRef] [PubMed]
- Mishra, J.; Singh, R.; Arora, N.K. Alleviation of heavy metal stress in plants and remediation of soil by rhizosphere microorganisms. Front. Microbiol. 2017, 8, 1706. [Google Scholar] [CrossRef]
- Tiwari, S.; Lata, C. Heavy metal stress, signaling, and tolerance due to plant-associated microbes: An overview. J. Front. Plant. Sci. 2018, 9, 452. [Google Scholar] [CrossRef]
- Siripornadulsil, S.; Siripornadulsil, W. Cadmium-tolerant bacteria reduce the uptake of cadmium in rice: Potential for microbial bioremediation. Ecotoxicol. Environ. Saf. 2013, 94, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Nongmaithem, N.; Roy, A.; Bhattacharya, P.M. Potential of Trichoderma spp. on growth promotion and mitigating cadmium uptake in rice plant under the metal stress ecosystem. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 992–1010. [Google Scholar] [CrossRef][Green Version]
- Pandey, S.; Ghosh, P.K.; Ghosh, S. Role of heavy metal resistant Ochrobactrum sp. and Bacillus spp. strains in bioremediation of a rice cultivar and their PGPR like activities. J. Microbiol. 2013, 51, 11–17. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, X.; He, X.; Lü, Q.; Qian, X.; Xiao, Q.; Lin, R. Effects of Pseudomonas TCd-1 on rice (Oryza sativa L.) cadmium uptake, rhizosphere soils enzyme activities and cadmium bioavailability under cadmium contamination. Ecotoxicol. Environ. Saf. 2021, 218, 112249. [Google Scholar] [CrossRef] [PubMed]
- Pramanik, K.; Mandal, S.; Banerjee, S.; Ghosh, A.; Maiti, T.K.; Mandal, N.C. Unraveling the heavy metal resistance and biocontrol potential of Pseudomonas sp. K32 strain facilitating rice seedling growth under Cd stress. Chemosphere 2021, 274, 129819. [Google Scholar] [CrossRef]
- Zhang, X.H.; Zhu, Y.G.; Chen, B.D.; Lin, A.J.; Smith, S.E.; Smith, F.A. Arbuscular mycorrhizal fungi contribute to resistance of upland rice to combined metal contamination of soil. J. Plant Nutr. 2005, 28, 2065–2077. [Google Scholar] [CrossRef]
- Chan, W.F.; Li, H.; Wu, F.Y. Arsenic uptake in upland rice inoculated with a combination or single arbuscular mycorrhizal fungi. J. Hazard Mater. 2013, 262, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; Pramanik, K.; Bhattacharya, S.; Mondal, S.; Ghosh, S.K.; Ghosh, P.K.; Maiti, T.K. Abatement of arsenic-induced phytotoxic effects in rice seedlings by an arsenic-resistant Pantoea dispersa strain. Environ. Sci. Pollut. Res. 2021, 28, 21633–21649. [Google Scholar] [CrossRef]


| Microbes | Experimental Set-Up | Growth Enhancement Effects | Yield Increment (%) | References |
|---|---|---|---|---|
| Beijerinckia indica | Field | Enhanced number of tillers and plant height | 27–29 | [90] |
| R. leguminosarum E11; Rhizobium sp. IRBG74; and Bradyrhizobium sp. IRBG271 | Pot | Enhanced shoot biomass and NPK uptake | 8–22 | [91] |
| Consortium of Azotobacter, Bacillus, Enterobacter, and Xanthobacter | Pot | Enhanced shoot biomass and N uptake | 15–18 | [92] |
| Trichoderma spp. | Field | Enhanced number of tillers, plant height, and biomass | 18 | [93] |
| Consortium of Anabaena variabilis, Tolypothrix tenuis, Nostoc muscorum, and Aulosira fertilissima | Field | Enhanced grain yield | 37.97 | [94] |
| Azotobacter spp. | Field | Enhanced N uptake | 67–93 | [95] |
| Azospirillum sp. B510 | Field | Enhanced number of tillers and plant height | 17 | [96] |
| P. fluorescens | Field | Enhanced number of tillers, plant height, and biomass | 20.6–26.9 | [97] |
| Consortium of Providencia sp., Brevundimonas sp., and Ochrobacterium sp. | Pot | Enhanced plant biomass | 19.02 | [79] |
| T. viride | Field | Enhanced number of tillers, plant height, and biomass | 31 | [86] |
| Consortium of Pseudomonas spp., Azotobacter chroococcum, and Azospirillum brasilense | Pot | Enhanced shoot biomass and NPK uptake | 15.3 | [98] |
| Cellulomonas flavigena | Pot | Enhanced number of tillers and plant height | 18 | [99] |
| T. asperellum SL2 | Field | Enhanced plant height, biomass, number of tillers, photosynthetic rate, water-use efficiency, and gene expression | 30 | [100] |
| Serratia spp. | Field | Enhanced stomatal conductance, nutrient uptake, shoot dry matter, number of grains per plant, and grain yield | 7–22 | [101] |
| Consortium of B. ubonensis la3c3, B. vietnamiensis la1a4, and Citrobacter bitternis p9a3m | Field | Enhanced NPK uptake | 2.5–13.5 | [102] |
| Consortium of Pseudomonas sp. and Penicillium sp. | Field | Enhanced P uptake | 16 | [103] |
| Microbes | Phytopathogens | Observed Effects | References |
|---|---|---|---|
| P. fluorescens | P. oryzae | Triggered ISR in rice against P. oryzae | [119] |
| Serratia marcescens | R. solani | Reduced the incidence of sheath blight | [120] |
| Bacillus subtilis | R. solani | Reduced disease incidence and severity of sheath blight | [121] |
| Glomus intraradices | M. oryzae | Up-regulation of defense-response genes such as OsNPR1, OsAP2, OsEREBP and OsJAmyb | [122] |
| B. oryzicola | Gibberella fujikuroi | Reduced bakanae severity by 46–78% | [123] |
| Streptomyces spp. | M. oryzae | Acceleration of defense enzyme activities such as synthesis of catalase, phenylalanine ammonia-lyase, and β-1,3-glucanase | [124] |
| P. fluorescens | M. oryzae | Reduced the physiological damage caused by M. oryzae | [9] |
| Cladosporium cladosporioides | M. oryzae | Modulation of enzymatic activity, and enhanced expression of defense-related genes such as JIOsPR10, LOX-RLL, and PR1b | [125,126,127] |
| B. subtlis | M. oryzae | Reduction in blast disease by >50% | [128] |
| Talaromyces spp. | R. solani | Up-regulation of defense-related genes, and acceleration of defense enzyme activities | [129] |
| Microbes | Phytopathogens | Observed effects | References |
|---|---|---|---|
| B. oryzicola | B. glumae | Induction of systemic resistance and promotion of plant growth | [136] |
| B. subtilis; B. amyloliquefaciens; and B. methyltrophicus | X. oryzae pv. oryzae | Activation of ISR, resulting in enhanced activity of defense-related enzymes | [137] |
| P. aeruginosa | X. oryzae pv. oryzae | Induction of defense-related enzymes | [138] |
| Streptomyces spp. | B. glumae | Inhibited the growth of B. glumae and increased plant growth | [139] |
| B. amyloliquefaciens and Aspergillus pseudoporous | X. oryzae pv. oryzae | Up-regulation of defense-related enzymes, and acceleration of the activities of defense-related proteins and total phenols | [140] |
| Consortium of S. fimicarius, S. laurentii, P. putida, and Metarhizium anisopliae | X. oryzae pv. oryzae | Reduced the incidence of leaf blight | [141] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doni, F.; Suhaimi, N.S.M.; Mispan, M.S.; Fathurrahman, F.; Marzuki, B.M.; Kusmoro, J.; Uphoff, N. Microbial Contributions for Rice Production: From Conventional Crop Management to the Use of ‘Omics’ Technologies. Int. J. Mol. Sci. 2022, 23, 737. https://doi.org/10.3390/ijms23020737
Doni F, Suhaimi NSM, Mispan MS, Fathurrahman F, Marzuki BM, Kusmoro J, Uphoff N. Microbial Contributions for Rice Production: From Conventional Crop Management to the Use of ‘Omics’ Technologies. International Journal of Molecular Sciences. 2022; 23(2):737. https://doi.org/10.3390/ijms23020737
Chicago/Turabian StyleDoni, Febri, Nurul Shamsinah Mohd Suhaimi, Muhamad Shakirin Mispan, F Fathurrahman, Betty Mayawatie Marzuki, Joko Kusmoro, and Norman Uphoff. 2022. "Microbial Contributions for Rice Production: From Conventional Crop Management to the Use of ‘Omics’ Technologies" International Journal of Molecular Sciences 23, no. 2: 737. https://doi.org/10.3390/ijms23020737
APA StyleDoni, F., Suhaimi, N. S. M., Mispan, M. S., Fathurrahman, F., Marzuki, B. M., Kusmoro, J., & Uphoff, N. (2022). Microbial Contributions for Rice Production: From Conventional Crop Management to the Use of ‘Omics’ Technologies. International Journal of Molecular Sciences, 23(2), 737. https://doi.org/10.3390/ijms23020737







