The Effect of Tau and Taxol on Polymerization of MCF7 Microtubules In Vitro
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Protein Preparation
4.2. Visualization, Measurements, and Data Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Howard, J. Mechanics of Motor Proteins and the Cytoskeleton; Sinauer Associates: Sunderland, MA, USA, 2001. [Google Scholar]
- Nogales, E.; Whittaker, M.; Milligan, R.A.; Downing, K.H. High-resolution model of the microtube. Cell 1999, 96, 79–88. [Google Scholar] [CrossRef] [Green Version]
- Rao, S.; He, L.; Chakravarty, S.; Ojima, I.; Orr, G.A.; Horwitz, S.B. Characterization of the Taxol binding site on the microtubule. Identification of Arg(282) in beta-tubulin as the site of photoincorporation of a 7-benzophenone analogue of Taxol. J. Biol. Chem. 1999, 274, 37990–37994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kar, S.; Fan, J.; Smith, M.J.; Goedert, M.; Amos, L.A. Repeat motifs of tau bind to the insides of microtubules in the absence of taxol. EMBO J. 2003, 22, 70–77. [Google Scholar] [CrossRef]
- Smoter, M.; Bodnar, L.; Duchnowska, R.; Stec, R.; Grala, B.; Szczylik, C. The role of Tau protein in resistance to paclitaxel. Cancer Chemother. Pharmacol. 2011, 68, 553–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makrides, V.; Massie, M.R.; Feinstein, S.C.; Lew, J. Evidence for two distinct binding sites for tau on microtubules. Proc. Natl. Acad. Sci. USA 2004, 101, 6746–6751. [Google Scholar] [CrossRef] [Green Version]
- Parness, J.; Horwitz, S.B. Taxol binds to polymerized tubulin in vitro. J. Cell Biol. 1981, 92, 479–487. [Google Scholar] [CrossRef]
- Arnal, I.; Wade, R.H. How does taxol stabilize microtubules? Curr. Biol. 1995, 5, 900–908. [Google Scholar] [CrossRef] [Green Version]
- Weingarten, M.D.; Lockwood, A.H.; Hwo, S.Y.; Kirschner, M.W. A protein factor essential for microtubule assembly. Proc. Natl. Acad. Sci. USA 1975, 72, 1858–1862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, D.B.; Johnson, K.A.; Borisy, G.G. Role of tubulin-associated proteins in microtubule nucleation and elongation. J. Mol. Biol. 1977, 117, 33–52. [Google Scholar] [CrossRef]
- Drechsel, D.N.; Hyman, A.A.; Cobb, M.H.; Kirschner, M.W. Modulation of the dynamic instability of tubulin assembly by the microtubule-associated protein Tau. Mol. Biol. Cell 1992, 3, 1141–1154. [Google Scholar]
- Panda, D.; Samuel, J.; Massia, M.; Feinstein, S.; Wilson, L. Differential regulation of microtubule dynamics by three- and four-repeat tau: Implications for the onset of neurodegenerative disease. Proc. Natl. Acad. Sci. USA 2003, 100, 9548–9553. [Google Scholar] [CrossRef] [Green Version]
- Gallo, J.M.; Hanger, D.P.; Twist, E.C.; Kosik, K.S.; Anderton, B.H. Expression and phosphorylation of a three-repeat isoform of tau in transfected non-neuronal cells. Biochem. J. 1992, 286, 399–404. [Google Scholar] [CrossRef] [Green Version]
- Lobert, S.; Hiser, L.; Correia, J. Expression profiling of tubulin isotypes and microtubule-interacting proteins using real-time polymerase chain reaction. Methods Cell Biol. 2010, 95, 47–58. [Google Scholar] [PubMed]
- Sirajuddin, M.; Rice, L.M.; Vale, R.D. Regulation of microtubule motors by tubulin isotypes and post-translational modifications. Nat. Cell Biol. 2014, 16, 335–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feizabadi, M.S.; Jun, Y. Kinesin-1 translocation: Surprising differences between bovine brain and MCF7-drived microtubules. Biochem. Biophys. Res. Commun. 2014, 4, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Feizabadi, M.S.; Janakaloti Narayanareddy, B.R.; Vadpey, O.; Jun, Y.; Chapman, D.; Rosenfeld, S.; Gross, S.P. Microtubule c-terminal tails can change characteristics of motor force production. Traffic 2015, 16, 1075–1087. [Google Scholar] [CrossRef]
- Feizabadi, M.S. The contribution of the c-terminal tails of microtubules in altering the force production specification of multiple kinesin-1. Cell Biochem. Biophys. 2016, 74, 373–380. [Google Scholar] [CrossRef]
- Feizabadi, M.S.; Jun, Y.; Reddy, J.N. Distinction between dynamic characteristic of the single Eg5 motor protein along neural vs. cancerous microtubules. Biochem. Biophys. Res. Commun. 2016, 478, 1630–1633. [Google Scholar] [CrossRef]
- Nami, B.; Wang, Z. Genetics and Expression Profile of the Tubulin Gene Superfamily in Breast Cancer Subtypes and Its Relation to Taxane Resistance. Cancers 2018, 10, 274. [Google Scholar] [CrossRef] [Green Version]
- Schroeder, C.; Grell, J.; Hube-Magg, C.; Kluth, M.; Lang, D.; Simon, R.; Höflmayer, D.; Minner, S.; Burandt, E.; Clauditz, T.S.; et al. Aberrant expression of the microtubule-associated protein tau is an independent prognostic feature in prostate cancer. BMC Cancer 2019, 19, 193. [Google Scholar] [CrossRef] [Green Version]
- Rouzier, R.; Rajan, R.; Wagner, P.; Hess, K.R.; Gold, D.L.; Stec, J.; Ayers, M.; Ross, J.S.; Zhang, P.; Buchholz, T.A.; et al. Microtubule-associated protein tau: A marker of paclitaxel sensitivity in breast cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 8315–8320. [Google Scholar] [CrossRef] [Green Version]
- Bonneau, C.; Gurard-Levin, Z.A.; Andre, F.; Pusztai, L.; Rouzier, R. Predictive and prognostic value of the Tau protein in breast cancer. Anticancer Res. 2015, 35, 5179–5184. [Google Scholar] [PubMed]
- Feizabadi, M.S.; Hernandez, M.A.V.; Breslin, J.B.; Akintola, I.I. The regulatory effect of Tau protein on polymerization of MCF7 microtubules in vitro. Biochem. Biophys. Rep. 2019, 17, 151–156. [Google Scholar] [CrossRef]
- Bhandare, V.V.; Kumbhar, B.V.; Kunwar, A. Differential binding affinity of tau repeat region R2 with neuronal-specific β-tubulin isotypes. Sci. Rep. 2019, 9, 10795. [Google Scholar] [CrossRef]
- Schiff, P.B.; Fant, J.; Horwitz, S.B. Promotion of microtubule assembly in vitro by Taxol. Nature 1979, 277, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Newton, C.N.; DeLuca, J.G.; Himes, R.H.; Miller, H.P.; Jordan, M.A.; Wilson, L. Intrinsically slow dynamic instability of Hela cell microtubules in vitro. J. Biol. Chem. 2002, 227, 42456–42462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feizabadi, M.S.; Rosario, B. MCF7 microtubules: Cancer microtubules with relatively slow and stable dynamic in vitro. Biochem. Biophys. Res. Commun. 2017, 484, 354–357. [Google Scholar] [CrossRef]
- Walker, R.A.; O’Brien, E.T.; Pryer, N.K.; Soboeiro, M.F.; Voter, W.A.; Erickson, H.P.; Salmon, E.D. Dynamic instability of individual, MAP-free microtubules analyzed by video light microscopy: Rate constants and transition frequencies. J. Cell Biol. 1988, 107, 1437–1448. [Google Scholar] [CrossRef]
- Steiner, B.; Mandelkow, E.M.; Biernat, J.; Gustke, N.; Meyer, H.E.; Schmidt, B.; Mieskes, G.; Soling, H.D.; Drechsel, D.; Kirschner, M.W. Phosphorylation of microtubule-associated protein tau: Identification of the site for Ca2(þ)-calmodulin dependent kinase and relationship with tau phosphorylation in Alzheimer tangles. EMBO J. 1990, 9, 3539–3544. [Google Scholar] [CrossRef]
- Mietelska-Porowska, A.; Wasik, U.; Goras, M.; Filipek, A.; Niewiadomska, G. Tau protein modifications and interactions: Their role in function and dysfunction. Int. J. Mol. Sci. 2014, 15, 4671–4713. [Google Scholar] [CrossRef] [Green Version]
- Roll-Mecak, A. Intrinsically disordered tubulin tails; Complex tuners of microtubule functions? Semin. Cell Dev. Biol. 2015, 37, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Jho, Y.S.; Zhulina, E.B.; Kim, M.W.; Pincus, P.A. Monte Carlo simulations of tau proteins: Effect of phosphorylation. Biophys. J. 2010, 99, 2387–2397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, D.; Mukrasch, M.D.; Biernat, J.; Bibow, S.; Blackledge, M.; Griesinger, C.; Mandelkow, E.; Zweckstetter, M. Conformational changes specific for pseudophosphorylation at serine 262 selectively impair binding of tau to microtubules. Biochemistry 2009, 48, 10047–10055. [Google Scholar] [CrossRef] [PubMed]
- Barbier, P.; Zejneli, O.; Martinho, M.; Lasorsa, A.; Belle, V.; Smet-Nocca, C.; Tsvetkov, P.O.; Devred, F.; Landrieu, I. Role of Tau as a microtubule-associated protein: Structural and functional aspects. Front. Aging Neurosci. 2019, 11, 204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alquezar, C.; Arya, S.; Kao, A.W. Tau Post-translational Modifications: Dynamic Transformers of Tau Function, Degradation, and Aggregation. Front. Neurol. 2021, 7, 595532. [Google Scholar] [CrossRef] [PubMed]
- Kavallaris, M.; Kuo, D.Y.; Burkhart, C.A.; Regl, D.L.; Norris, M.D.; Haber, M.; Horwitz, S.B. Taxol-resistant epithelial ovarian tumors are associated with altered expression of specific beta-tubulin isotypes. J. Clin. Investig. 1997, 100, 1282–1293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albrethsen, J.; Angeletti, R.H.; Horwitz, S.B.; Yang, C.P. Proteomics of cancer cell lines resistant to microtubule-stabilizing agents. Mol. Cancer Ther. 2014, 13, 260–269. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.P.H.; Yap, E.H.; Xiao, H.; Fiser, A.; Horwitz, S.B. 2-(m-Azidobenzoyl) taxol binds differentially to distinct beta-tubulin isotypes. Proc. Natl. Acad. Sci. USA 2016, 113, 11294–11299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huzil, J.T.; Chen, K.; Kurgan, L.; Tuszynski, J.A. The roles of beta-tubulin mutations and isotype expression in acquired drug resistance. Cancer Inform. 2007, 3, 159–181. [Google Scholar] [CrossRef]

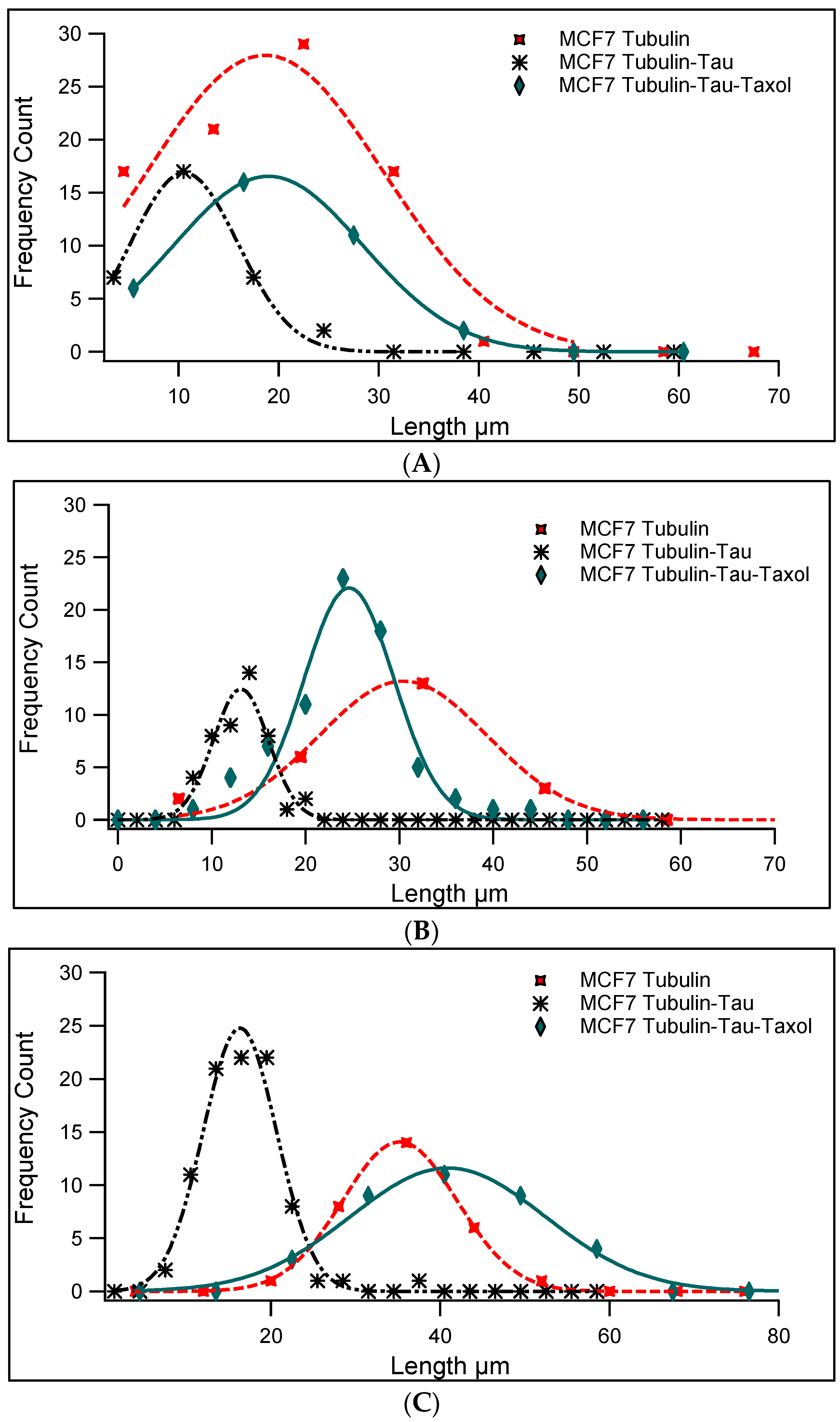
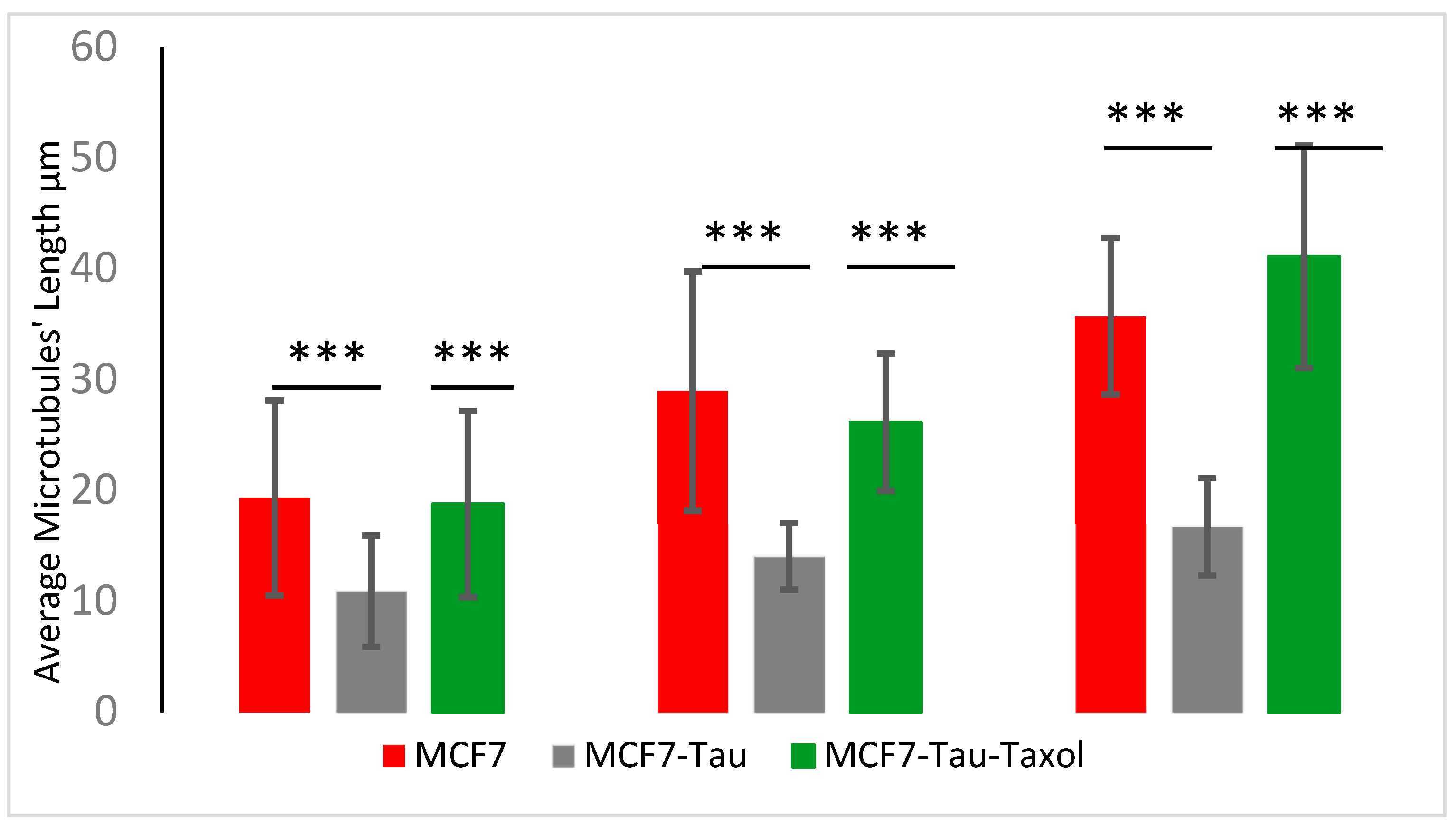
| Samples (Horizontal) Concentrations (Vertical) | Pure Tubulin MTs’ Average Length | Tau-Induced MTs’ Average Length | Tau and Taxol-Induced MTs’ Average Length |
|---|---|---|---|
| Tubulin and Tau (1.25, 0.1) mg/mL | 19.3 ± 8.8 µm, | 10.9 ± 5.0 µm, | 18.8 ± 8.4 µm, |
| n = 85 | n = 33 | n = 35 | |
| Tubulin and Tau (2, 0.2) mg/mL | 29.0 ± 10.8 µm, | 14.0 ± 3.0 µm, | 26.2 ± 6.3 µm, |
| n = 24 | n = 46 | n = 73 | |
| Tubulin and Tau (2.5, 0.25) mg/mL | 35.7 ± 7.1 µm, | 16.7 ± 4.4 µm, | 41.1 ± 10.0 µm, |
| n = 30 | n = 89 | n = 36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feizabadi, M.S.; Castillon, V.J. The Effect of Tau and Taxol on Polymerization of MCF7 Microtubules In Vitro. Int. J. Mol. Sci. 2022, 23, 677. https://doi.org/10.3390/ijms23020677
Feizabadi MS, Castillon VJ. The Effect of Tau and Taxol on Polymerization of MCF7 Microtubules In Vitro. International Journal of Molecular Sciences. 2022; 23(2):677. https://doi.org/10.3390/ijms23020677
Chicago/Turabian StyleFeizabadi, Mitra Shojania, and Venise Jan Castillon. 2022. "The Effect of Tau and Taxol on Polymerization of MCF7 Microtubules In Vitro" International Journal of Molecular Sciences 23, no. 2: 677. https://doi.org/10.3390/ijms23020677
APA StyleFeizabadi, M. S., & Castillon, V. J. (2022). The Effect of Tau and Taxol on Polymerization of MCF7 Microtubules In Vitro. International Journal of Molecular Sciences, 23(2), 677. https://doi.org/10.3390/ijms23020677





