Insights into Four NAC Transcription Factors Involved in Grain Development and in Response to Moderate Heat in the Triticeae Tribe
Abstract
1. Introduction
2. Results
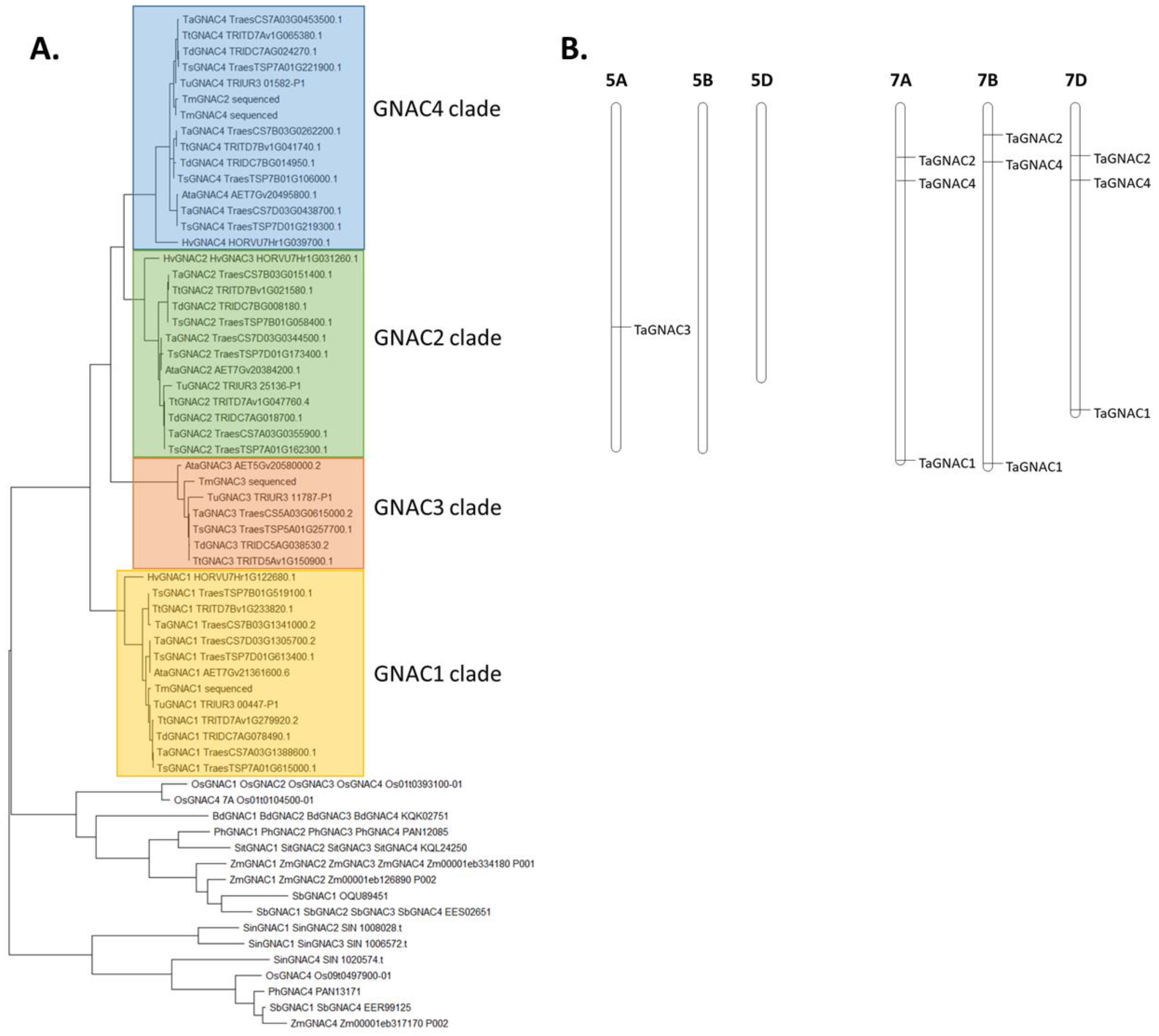
2.1. Identification of the Four NAC Genes Grain-Expressed (GNAC) in 15 Cereal Species
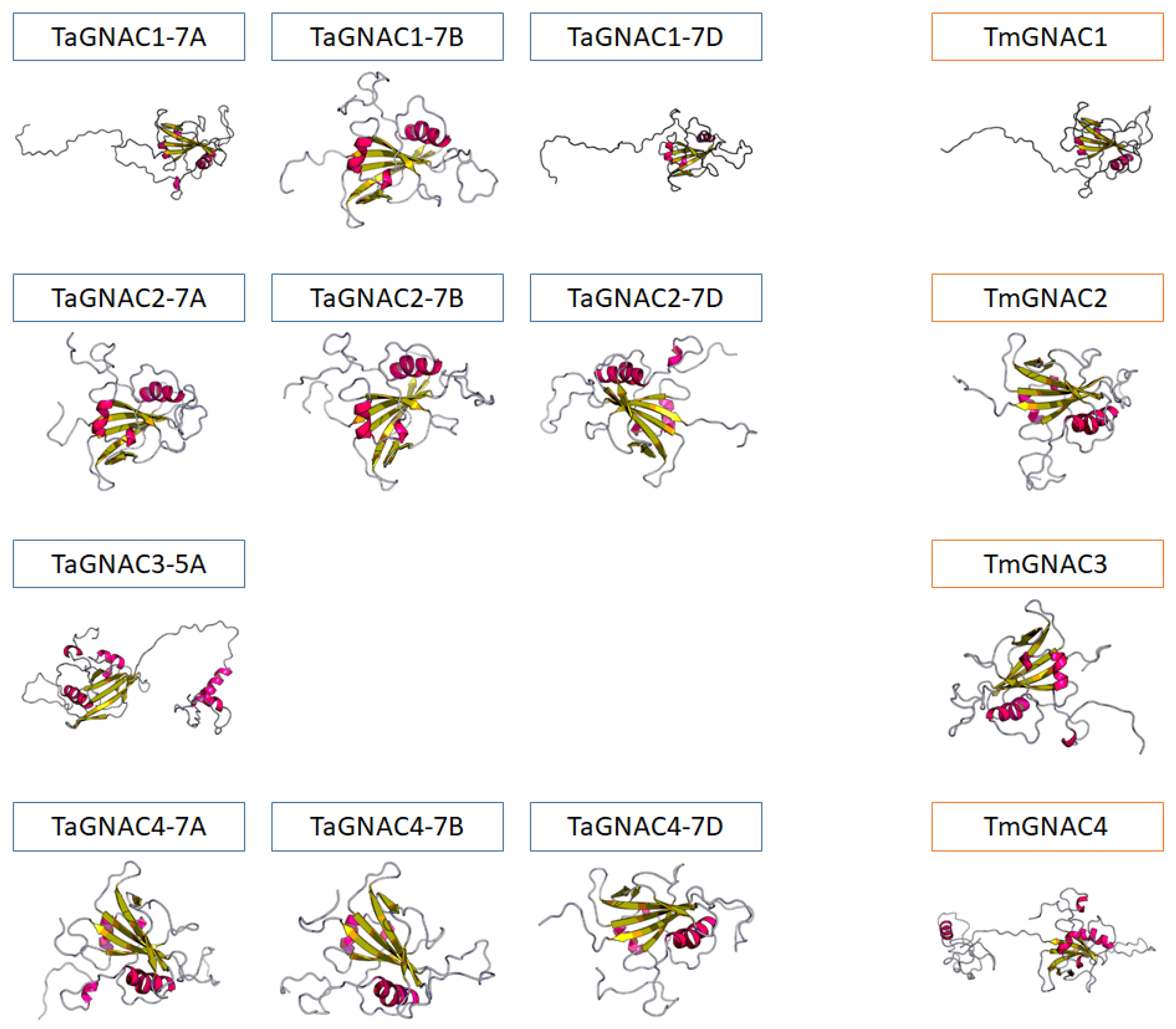
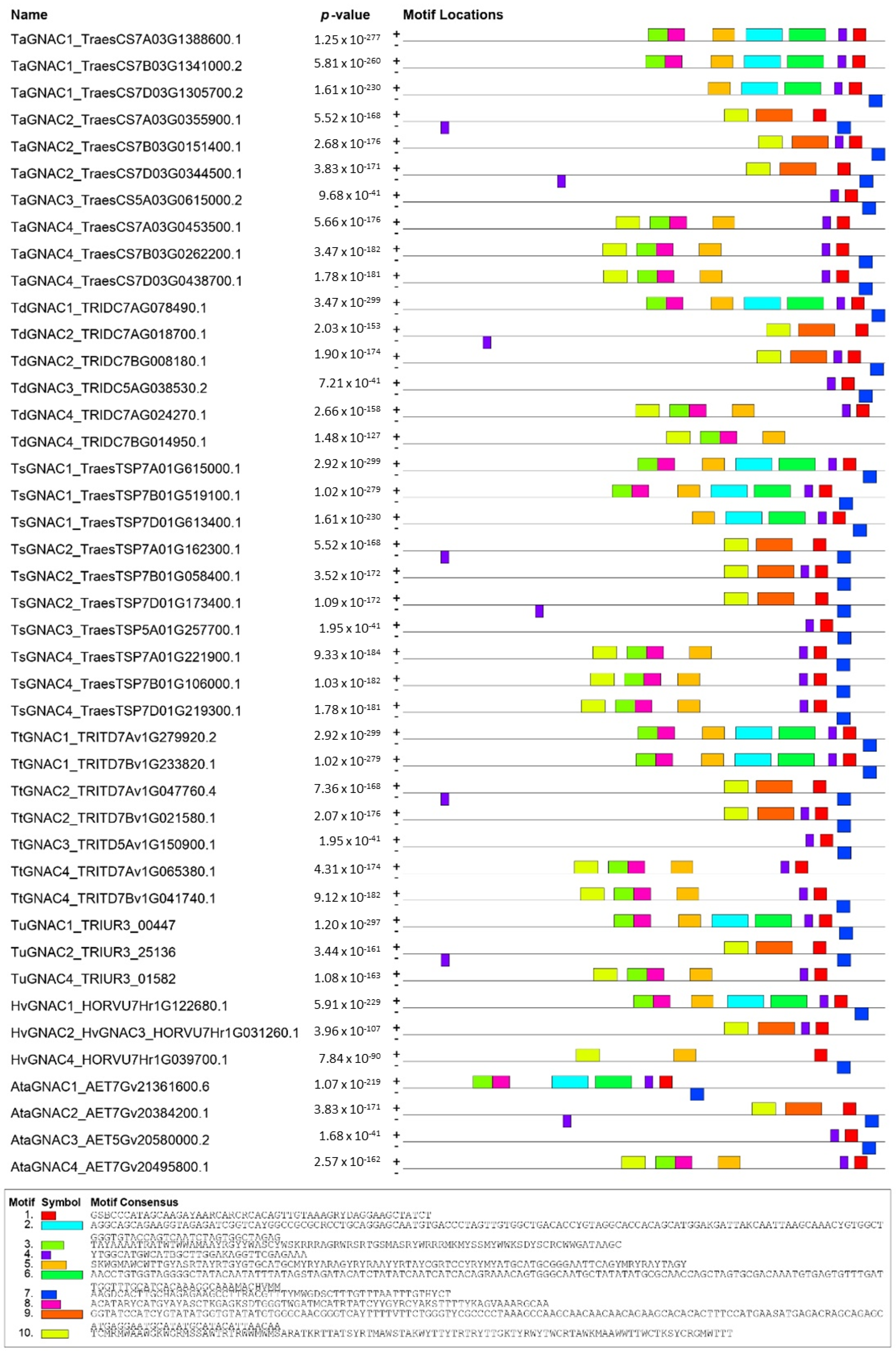
2.2. Structure and Functional Domains of GNAC Proteins
2.3. Expression of Orthologs of the Four TaGNAC Genes in Einkorn Grain (TmGNAC) and TaGNAC Homeologs during Development
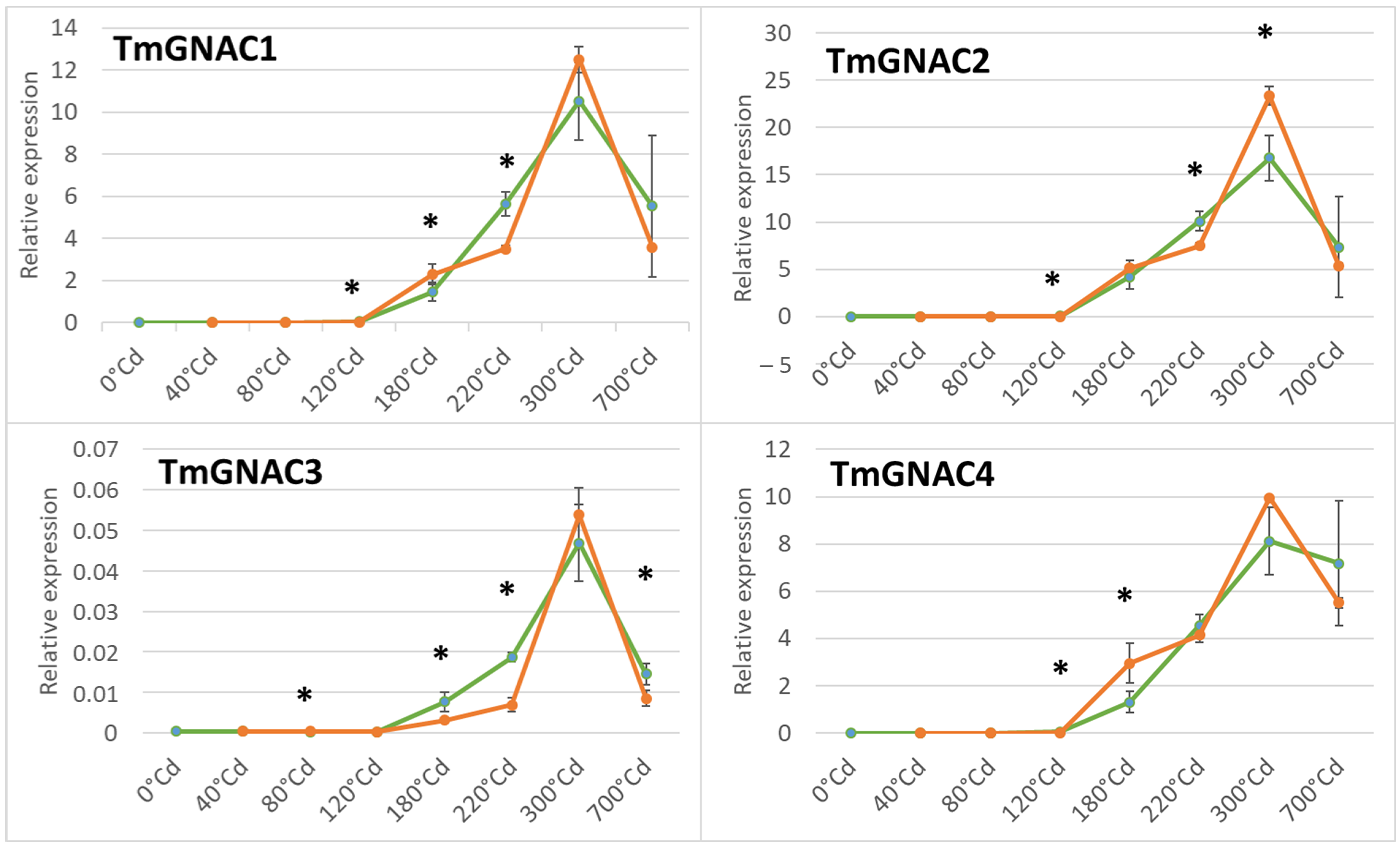
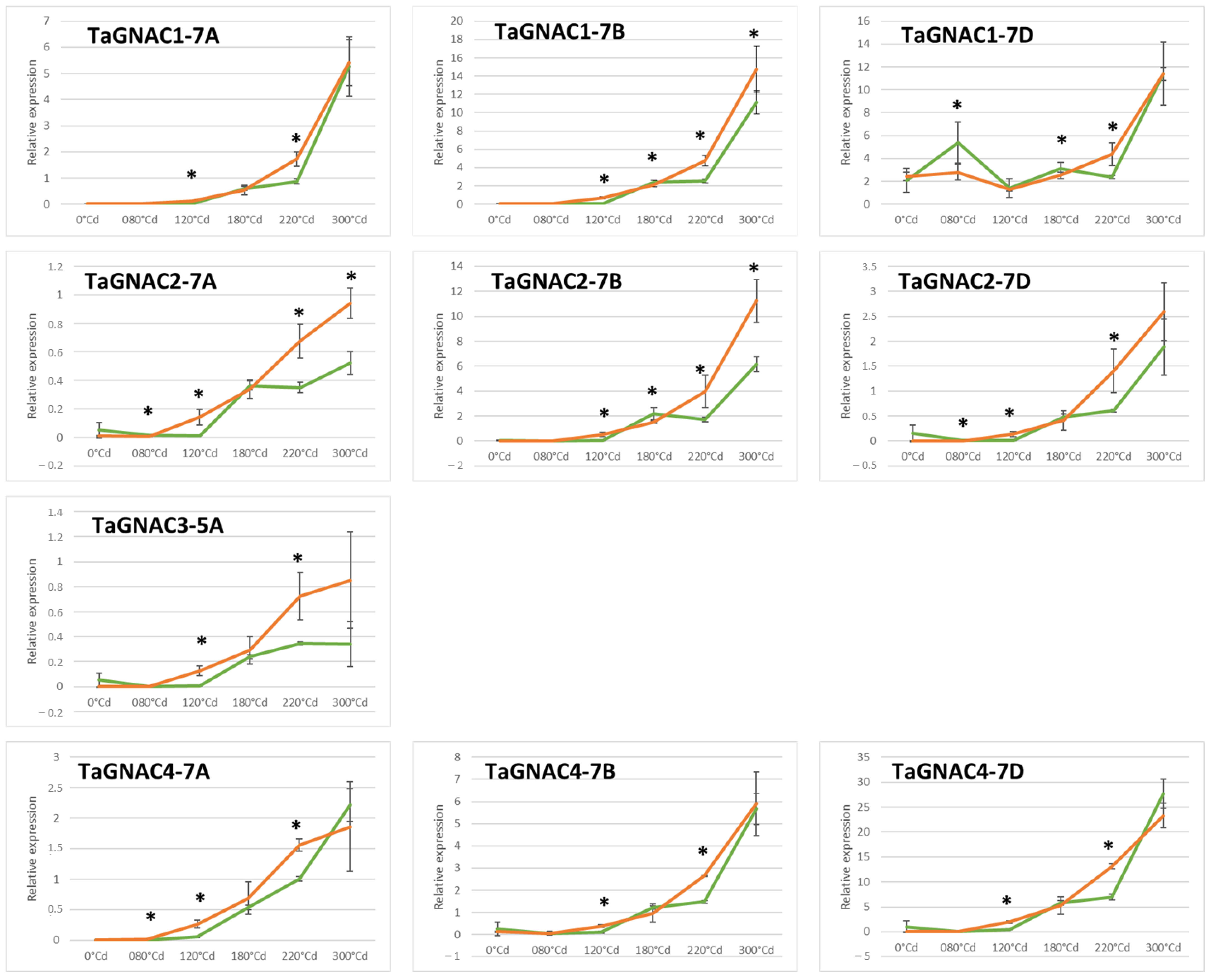
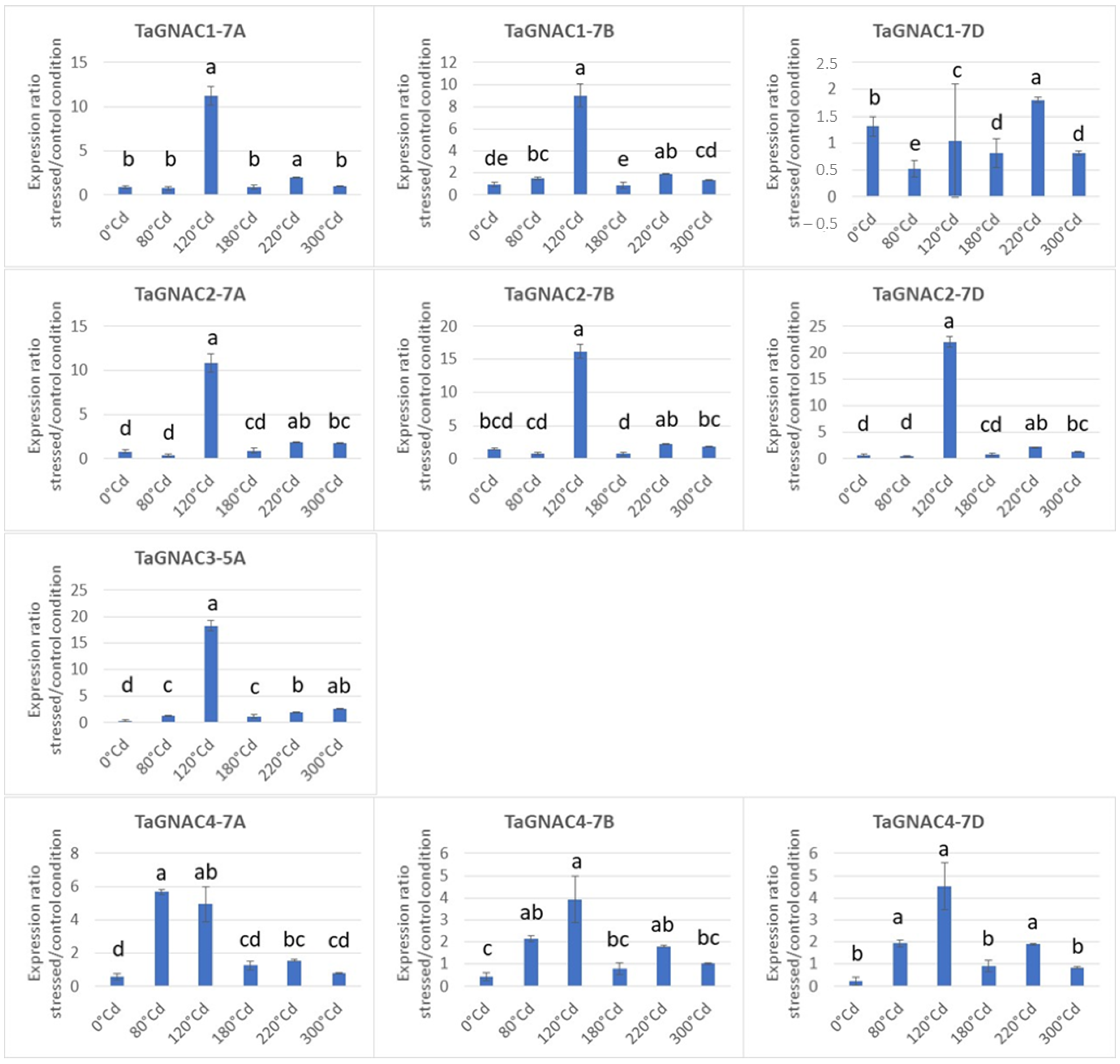
2.4. Gene Expression Profile of the TmGNAC in Einkorn and TaGNAC A Homeologs in Bread Wheat in Response to Heat Stress
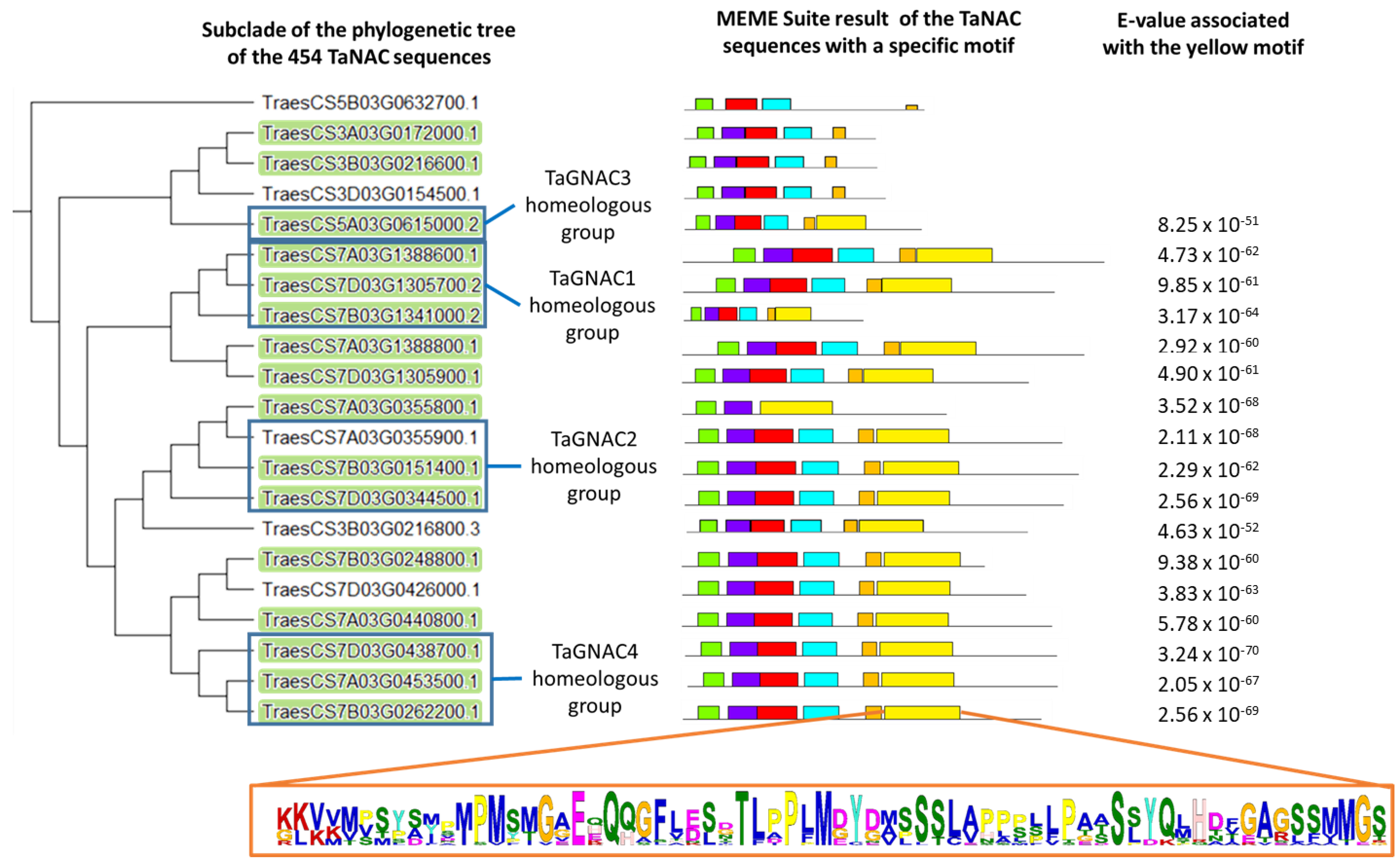
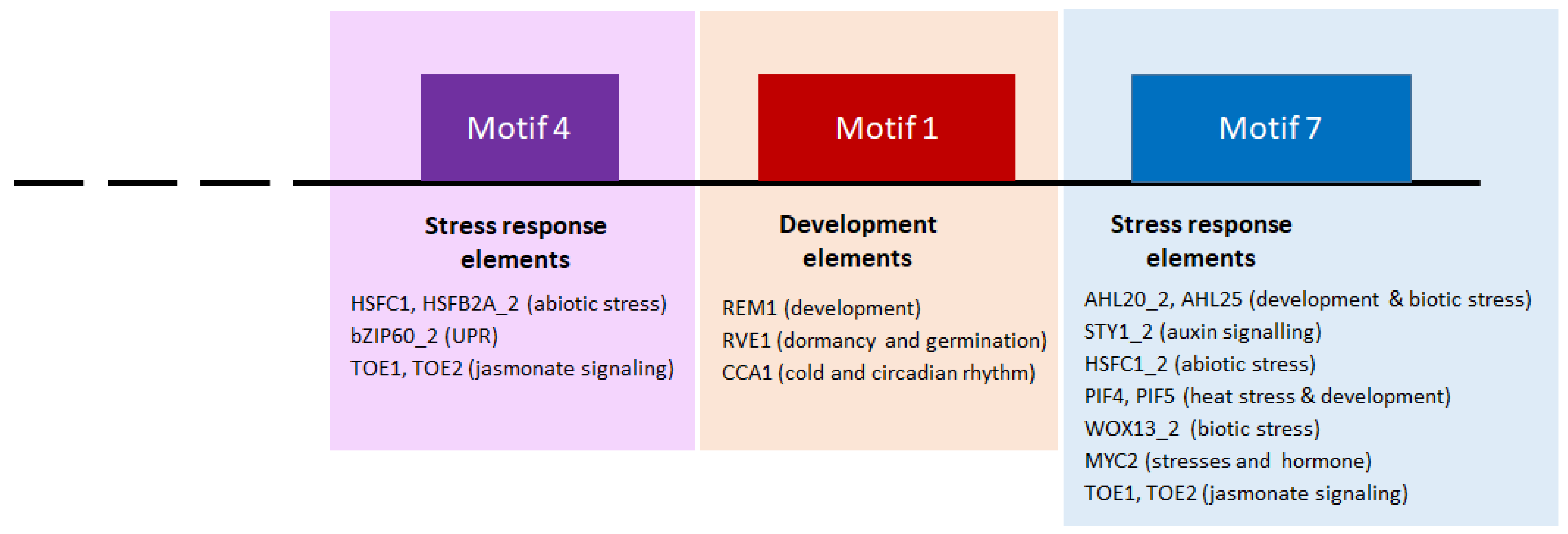
2.5. Prediction of the Transcriptional Regulation of the GNAC Expression
3. Discussion
3.1. TaGNAC Genes Carry a Recognizable Pattern and Are Highly Conserved through the Triticeae Tribe
3.2. The Four GNACs Harbour a Similar Expression Pattern during Seed Development in Wheat and Einkorn
3.3. The Four GNAC Genes Are Expressed in the Seed Earlier in Response to Thermal Stress
4. Materials and Methods
4.1. Sequence Identification and Analysis in Bread Wheat
4.2. Sequence Identification in Einkorn from Bread Wheat
4.3. Identification of the Four TaGNAC Orthologs in the Genome of 13 Other Cereals
4.4. Plant Material and Thermal Stress Treatment
4.5. RNA Extraction and RT-qPCR of Candidate Genes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, S.; Koyama, H.; Bhati, K.K.; Alok, A. The Biotechnological Importance of the Plant-Specific NAC Transcription Factor Family in Crop Improvement. J. Plant Res. 2021, 134, 475–495. [Google Scholar] [CrossRef] [PubMed]
- Nuruzzaman, M.; Manimekalai, R.; Sharoni, A.M.; Satoh, K.; Kondoh, H.; Ooka, H.; Kikuchi, S. Genome-Wide Analysis of NAC Transcription Factor Family in Rice. Gene 2010, 465, 30–44. [Google Scholar] [CrossRef]
- Li, W.; Li, X.; Chao, J.; Zhang, Z.; Wang, W.; Guo, Y. NAC Family Transcription Factors in Tobacco and Their Potential Role in Regulating Leaf Senescence. Front. Plant Sci. 2018, 9, 1900. [Google Scholar] [CrossRef] [PubMed]
- Melo, B.P.; Fraga, O.T.; Silva, J.C.F.; Ferreira, D.O.; Brustolini, O.J.B.; Carpinetti, P.A.; Machado, J.P.B.; Reis, P.A.B.; Fontes, E.P.B. Revisiting the Soybean GmNAC Superfamily. Front. Plant Sci 2018, 9, 1864. [Google Scholar] [CrossRef] [PubMed]
- Moyano, E.; Martínez-Rivas, F.J.; Blanco-Portales, R.; Molina-Hidalgo, F.J.; Ric-Varas, P.; Matas-Arroyo, A.J.; Caballero, J.L.; Muñoz-Blanco, J.; Rodríguez-Franco, A. Genome-Wide Analysis of the NAC Transcription Factor Family and Their Expression during the Development and Ripening of the Fragaria × Ananassa Fruits. PLoS ONE 2018, 13, e0196953. [Google Scholar] [CrossRef]
- Jin, J.F.; Wang, Z.Q.; He, Q.Y.; Wang, J.Y.; Li, P.F.; Xu, J.M.; Zheng, S.J.; Fan, W.; Yang, J.L. Genome-Wide Identification and Expression Analysis of the NAC Transcription Factor Family in Tomato (Solanum Lycopersicum) during Aluminum Stress. BMC Genom. 2020, 21, 288. [Google Scholar] [CrossRef]
- Zong, X.; Yan, Q.; Wu, F.; Ma, Q.; Zhang, J. Genome-Wide Analysis of the Role of NAC Family in Flower Development and Abiotic Stress Responses in Cleistogenes Songorica. Genes 2020, 11, 927. [Google Scholar] [CrossRef]
- Lu, M.; Sun, Q.-P.; Zhang, D.-F.; Wang, T.-Y.; Pan, J.-B. Identification of 7 Stress-Related NAC Transcription Factor Members in Maize (Zea Mays L.) and Characterization of the Expression Pattern of These Genes. Biochem. Biophys. Res. Commun. 2015, 462, 144–150. [Google Scholar] [CrossRef]
- Zhu, G.; Chen, G.; Zhu, J.; Zhu, Y.; Lu, X.; Li, X.; Hu, Y.; Yan, Y. Molecular Characterization and Expression Profiling of NAC Transcription Factors in Brachypodium Distachyon L. PLoS ONE 2015, 10, e0139794. [Google Scholar] [CrossRef]
- Saidi, M.N.; Mergby, D.; Brini, F. Identification and Expression Analysis of the NAC Transcription Factor Family in Durum Wheat (Triticum Turgidum L. Ssp. Durum). Plant Physiol. Biochem. 2017, 112, 117–128. [Google Scholar] [CrossRef]
- Murozuka, E.; Massange-Sánchez, J.A.; Nielsen, K.; Gregersen, P.L.; Braumann, I. Genome Wide Characterization of Barley NAC Transcription Factors Enables the Identification of Grain-Specific Transcription Factors Exclusive for the Poaceae Family of Monocotyledonous Plants. PLoS ONE 2018, 13, e0209769. [Google Scholar] [CrossRef]
- Guérin, C.; Roche, J.; Allard, V.; Ravel, C.; Mouzeyar, S.; Bouzidi, M.F. Genome-Wide Analysis, Expansion and Expression of the NAC Family under Drought and Heat Stresses in Bread Wheat (T. Aestivum L.). PLoS ONE 2019, 14, e0213390. [Google Scholar] [CrossRef]
- Ooka, H.; Satoh, K.; Doi, K.; Nagata, T.; Otomo, Y.; Murakami, K.; Matsubara, K.; Osato, N.; Kawai, J.; Carninci, P.; et al. Comprehensive Analysis of NAC Family Genes in Oryza Sativa and Arabidopsis Thaliana. DNA Res. 2003, 10, 239–247. [Google Scholar] [CrossRef]
- Ernst, H.A.; Olsen, A.N.; Skriver, K.; Larsen, S.; Leggio, L.L. Structure of the Conserved Domain of ANAC, a Member of the NAC Family of Transcription Factors. EMBO Rep. 2004, 5, 297–303. [Google Scholar] [CrossRef]
- Hao, Y.-J.; Wei, W.; Song, Q.-X.; Chen, H.-W.; Zhang, Y.-Q.; Wang, F.; Zou, H.-F.; Lei, G.; Tian, A.-G.; Zhang, W.-K.; et al. Soybean NAC Transcription Factors Promote Abiotic Stress Tolerance and Lateral Root Formation in Transgenic Plants. Plant J. 2011, 68, 302–313. [Google Scholar] [CrossRef]
- Xie, Q.; Frugis, G.; Colgan, D.; Chua, N.-H. Arabidopsis NAC1 Transduces Auxin Signal Downstream of TIR1 to Promote Lateral Root Development. Genes Dev. 2000, 14, 3024–3036. [Google Scholar] [CrossRef]
- Olsen, A.N.; Ernst, H.A.; Leggio, L.L.; Skriver, K. NAC Transcription Factors: Structurally Distinct, Functionally Diverse. Trends Plant Sci. 2005, 10, 79–87. [Google Scholar] [CrossRef]
- Sablowski, R.W.; Meyerowitz, E.M. A Homolog of NO APICAL MERISTEM Is an Immediate Target of the Floral Homeotic Genes APETALA3/PISTILLATA. Cell 1998, 92, 93–103. [Google Scholar] [CrossRef]
- Mitsuda, N.; Seki, M.; Shinozaki, K.; Ohme-Takagi, M. The NAC Transcription Factors NST1 and NST2 of Arabidopsis Regulate Secondary Wall Thickenings and Are Required for Anther Dehiscence. Plant Cell 2005, 17, 2993–3006. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Kim, S.-G.; Park, J.-E.; Park, H.-Y.; Lim, M.-H.; Chua, N.-H.; Park, C.-M. A Membrane-Bound NAC Transcription Factor Regulates Cell Division in Arabidopsis. Plant Cell 2006, 18, 3132–3144. [Google Scholar] [CrossRef]
- Souer, E.; van Houwelingen, A.; Kloos, D.; Mol, J.; Koes, R. The No Apical Meristem Gene of Petunia Is Required for Pattern Formation in Embryos and Flowers and Is Expressed at Meristem and Primordia Boundaries. Cell 1996, 85, 159–170. [Google Scholar] [CrossRef]
- Kim, S.-G.; Lee, A.-K.; Yoon, H.-K.; Park, C.-M. A Membrane-Bound NAC Transcription Factor NTL8 Regulates Gibberellic Acid-Mediated Salt Signaling in Arabidopsis Seed Germination. Plant J. 2008, 55, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Uauy, C.; Distelfeld, A.; Fahima, T.; Blechl, A.; Dubcovsky, J. A NAC Gene Regulating Senescence Improves Grain Protein, Zinc, and Iron Content in Wheat. Science 2006, 314, 1298–1301. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Takasaki, H.; Mizoi, J.; Shinozaki, K.; Yamaguchi-Shinozaki, K. NAC Transcription Factors in Plant Abiotic Stress Responses. Biochim. Biophys. Acta (BBA) Gene Regul. Mech. 2012, 1819, 97–103. [Google Scholar] [CrossRef]
- Shahnejat-Bushehri, S.; Mueller-Roeber, B.; Balazadeh, S. Arabidopsis NAC Transcription Factor JUNGBRUNNEN1 Affects Thermomemory-Associated Genes and Enhances Heat Stress Tolerance in Primed and Unprimed Conditions. Plant Signal. Behav. 2012, 7, 1518–1521. [Google Scholar] [CrossRef]
- Wu, A.; Allu, A.D.; Garapati, P.; Siddiqui, H.; Dortay, H.; Zanor, M.-I.; Asensi-Fabado, M.A.; Munne-Bosch, S.; Antonio, C.; Tohge, T.; et al. JUNGBRUNNEN1, a Reactive Oxygen Species-Responsive NAC Transcription Factor, Regulates Longevity in Arabidopsis. Plant Cell 2012, 24, 482–506. [Google Scholar] [CrossRef]
- Thirumalaikumar, V.P.; Devkar, V.; Mehterov, N.; Ali, S.; Ozgur, R.; Turkan, I.; Mueller-Roeber, B.; Balazadeh, S. NAC Transcription Factor JUNGBRUNNEN1 Enhances Drought Tolerance in Tomato. Plant Biotechnol. J. 2018, 16, 354–366. [Google Scholar] [CrossRef]
- Guan, Q.; Yue, X.; Zeng, H.; Zhu, J. The Protein Phosphatase RCF2 and Its Interacting Partner NAC019 Are Critical for Heat Stress-Responsive Gene Regulation and Thermotolerance in Arabidopsis. Plant Cell 2014, 26, 438–453. [Google Scholar] [CrossRef]
- Guo, W.; Zhang, J.; Zhang, N.; Xin, M.; Peng, H.; Hu, Z.; Ni, Z.; Du, J. The Wheat NAC Transcription Factor TaNAC2L Is Regulated at the Transcriptional and Post-Translational Levels and Promotes Heat Stress Tolerance in Transgenic Arabidopsis. PLoS ONE 2015, 10, e0135667. [Google Scholar] [CrossRef]
- Kim, S.-G.; Lee, S.; Seo, P.J.; Kim, S.-K.; Kim, J.-K.; Park, C.-M. Genome-Scale Screening and Molecular Characterization of Membrane-Bound Transcription Factors in Arabidopsis and Rice. Genomics 2010, 95, 56–65. [Google Scholar] [CrossRef]
- Lee, S.; Seo, P.J.; Lee, H.-J.; Park, C.-M. A NAC Transcription Factor NTL4 Promotes Reactive Oxygen Species Production during Drought-Induced Leaf Senescence in Arabidopsis: NTL4 in Drought-Induced Leaf Senescence. Plant J. 2012, 70, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, H.-J.; Huh, S.U.; Paek, K.-H.; Ha, J.-H.; Park, C.-M. The Arabidopsis NAC Transcription Factor NTL4 Participates in a Positive Feedback Loop That Induces Programmed Cell Death under Heat Stress Conditions. Plant Sci. 2014, 227, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Liao, K.; Du, H.; Xu, Y.; Song, H.; Li, X.; Xiong, L. A Stress-Responsive NAC Transcription Factor SNAC3 Confers Heat and Drought Tolerance through Modulation of Reactive Oxygen Species in Rice. J. Exp. Bot. 2015, 66, 6803–6817. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yu, Y.; Liu, Z.; Li, S.; Wang, Z.; Xiang, F. Membrane-Bound NAC Transcription Factors in Maize and Their Contribution to the Oxidative Stress Response. Plant Sci. 2016, 250, 30–39. [Google Scholar] [CrossRef]
- Morishita, T.; Kojima, Y.; Maruta, T.; Nishizawa-Yokoi, A.; Yabuta, Y.; Shigeoka, S. Arabidopsis NAC Transcription Factor, ANAC078, Regulates Flavonoid Biosynthesis under High-Light. Plant Cell Physiol. 2009, 50, 2210–2222. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, M.; Gao, S.; Zhang, Z.; Zhao, X.; Zhao, C.; Zhang, F.; Chen, X. Molecular Characterization of Novel TaNAC Genes in Wheat and Overexpression of TaNAC2a Confers Drought Tolerance in Tobacco. Physiol. Plant 2012, 144, 210–224. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, Y. Overexpression of TaNAC2D Displays Opposite Responses to Abiotic Stresses between Seedling and Mature Stage of Transgenic Arabidopsis. Front. Plant Sci. 2016, 7, 1754. [Google Scholar] [CrossRef]
- Zhao, D.; Derkx, A.P.; Liu, D.-C.; Buchner, P.; Hawkesford, M.J. Overexpression of a NAC Transcription Factor Delays Leaf Senescence and Increases Grain Nitrogen Concentration in Wheat. Plant Biol. 2015, 17, 904–913. [Google Scholar] [CrossRef]
- Mathew, I.E.; Das, S.; Mahto, A.; Agarwal, P. Three Rice NAC Transcription Factors Heteromerize and Are Associated with Seed Size. Front. Plant Sci. 2016, 7, 1638. [Google Scholar] [CrossRef]
- Shen, H.; Yin, Y.; Chen, F.; Xu, Y.; Dixon, R.A. A Bioinformatic Analysis of NAC Genes for Plant Cell Wall Development in Relation to Lignocellulosic Bioenergy Production. BioEnergy Res. 2009, 2, 217–232. [Google Scholar] [CrossRef]
- Albà, M.M.; Pagès, M. Plant Proteins Containing the RNA-Recognition Motif. Trends Plant Sci. 1998, 3, 15–21. [Google Scholar] [CrossRef]
- Fierro-Monti, I.; Mathews, M.B. Proteins Binding to Duplexed RNA: One Motif, Multiple Functions. Trends Biochem. Sci. 2000, 25, 241–246. [Google Scholar] [CrossRef]
- Chang, C.-W.; Couñago, R.M.; Williams, S.J.; Boden, M.; Kobe, B. The Distribution of Different Classes of Nuclear Localization Signals (NLSs) in Diverse Organisms and the Utilization of the Minor NLS-Binding Site Inplantnuclear Import Factor Importin-α. Plant Signal. Behav. 2013, 8, e25976. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Wang, Z.; Liu, S.; Kong, X.; Wang, F.; Sun, G.; Geng, S.; Mao, L.; Zhou, P.; Li, A. Transcriptome Analysis of Developing Wheat Grains at Rapid Expanding Phase Reveals Dynamic Gene Expression Patterns. Biology 2022, 11, 281. [Google Scholar] [CrossRef]
- Plessis, A.; Ravel, C.; Bordes, J.; Balfourier, F.; Martre, P. Association Study of Wheat Grain Protein Composition Reveals That Gliadin and Glutenin Composition Are Trans-Regulated by Different Chromosome Regions. J. Exp. Bot. 2013, 64, 3627–3644. [Google Scholar] [CrossRef]
- Vincent, J.; Martre, P.; Ravel, C.; Baillif, A.; Agier, M. A Weboriented Platform for Gene Regulatory Network Inference. In Application to Seed Storage Proteins in Wheat; Barillot, E., Froidevaux, C., Rocha, E., Eds.; Institut Pasteur: Paris, France, 2011; pp. 293–294. [Google Scholar]
- Zhang, Z.; Dong, J.; Ji, C.; Wu, Y.; Messing, J. NAC-Type Transcription Factors Regulate Accumulation of Starch and Protein in Maize Seeds. Proc. Natl. Acad. Sci. USA 2019, 116, 11223–11228. [Google Scholar] [CrossRef]
- Thomas, M.S.; Flavell, R.B. Identification of an Enhancer Element for the Endosperm-Specific Expression of High Molecular Weight Glutenin. Plant Cell 1990, 2, 1171–1180. [Google Scholar]
- Gao, Y.; An, K.; Guo, W.; Chen, Y.; Zhang, R.; Zhang, X.; Chang, S.; Rossi, V.; Jin, F.; Cao, X.; et al. The Endosperm-Specific Transcription Factor TaNAC019 Regulates Glutenin and Starch Accumulation and Its Elite Allele Improves Wheat Grain Quality. Plant Cell 2021, 33, 603–622. [Google Scholar] [CrossRef]
- Girousse, C.; Roche, J.; Guerin, C.; Le Gouis, J.; Balzegue, S.; Mouzeyar, S.; Bouzidi, M.F. Coexpression Network and Phenotypic Analysis Identify Metabolic Pathways Associated with the Effect of Warming on Grain Yield Components in Wheat. PLoS ONE 2018, 13, e0199434. [Google Scholar] [CrossRef]
- Ma, J.; Yuan, M.; Sun, B.; Zhang, D.; Zhang, J.; Li, C.; Shao, Y.; Liu, W.; Jiang, L. Evolutionary Divergence and Biased Expression of NAC Transcription Factors in Hexaploid Bread Wheat (Triticum Aestivum L.). Plants 2021, 10, 382. [Google Scholar] [CrossRef]
- Swindell, W.R.; Huebner, M.; Weber, A.P. Transcriptional Profiling of Arabidopsis Heat Shock Proteins and Transcription Factors Reveals Extensive Overlap between Heat and Non-Heat Stress Response Pathways. BMC Genom. 2007, 8, 125. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, L.; Cao, W.; Wang, J.; Yu, J.; Yang, Z.; Huang, B. Characterization and Functional Analysis of FaHsfC1b from Festuca Arundinacea Conferring Heat Tolerance in Arabidopsis. Int. J. Mol. Sci. 2018, 19, E2702. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, M.; Gross-Hardt, R.; Schöffl, F. Heat Shock Factor HSFB2a Involved in Gametophyte Development of Arabidopsis Thaliana and Its Expression Is Controlled by a Heat-Inducible Long Non-Coding Antisense RNA. Plant Mol. Biol. 2014, 85, 541–550. [Google Scholar] [CrossRef]
- Mao, H.; Wang, H.; Liu, S.; Li, Z.; Yang, X.; Yan, J.; Li, J.; Tran, L.-S.P.; Qin, F. A Transposable Element in a NAC Gene Is Associated with Drought Tolerance in Maize Seedlings. Nat. Commun. 2015, 6, 8326. [Google Scholar] [CrossRef] [PubMed]
- International Wheat Genome Sequencing Consortium (IWGSC); IWGSC RefSeq Principal Investigators; Appels, R.; Eversole, K.; Feuillet, C.; Keller, B.; Rogers, J.; Stein, N.; IWGSC Whole-Genome Assembly Principal Investigators; Pozniak, C.J.; et al. Shifting the Limits in Wheat Research and Breeding Using a Fully Annotated Reference Genome. Science 2018, 361, 7191. [Google Scholar] [CrossRef]
- Voorrips, R.E. MapChart: Software for the Graphical Presentation of Linkage Maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Fox, S.E.; Geniza, M.; Hanumappa, M.; Naithani, S.; Sullivan, C.; Preece, J.; Tiwari, V.K.; Elser, J.; Leonard, J.M.; Sage, A.; et al. De Novo Transcriptome Assembly and Analyses of Gene Expression during Photomorphogenesis in Diploid Wheat Triticum Monococcum. PLoS ONE 2014, 9, e96855. [Google Scholar] [CrossRef]
- Gupta, S.; Stamatoyannopoulos, J.A.; Bailey, T.L.; Noble, W.S. Quantifying Similarity between Motifs. Genome Biol. 2007, 8, R24. [Google Scholar] [CrossRef]
- Franco-Zorrilla, J.M.; López-Vidriero, I.; Carrasco, J.L.; Godoy, M.; Vera, P.; Solano, R. DNA-Binding Specificities of Plant Transcription Factors and Their Potential to Define Target Genes. Proc. Natl. Acad. Sci. USA 2014, 111, 2367–2372. [Google Scholar] [CrossRef]
- Capron, D.; Mouzeyar, S.; Boulaflous, A.; Girousse, C.; Rustenholz, C.; Laugier, C.; Paux, E.; Bouzidi, M.F. Transcriptional Profile Analysis of E3 Ligase and Hormone-Related Genes Expressed during Wheat Grain Development. BMC Plant Biol. 2012, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Paolacci, A.R.; Tanzarella, O.A.; Porceddu, E.; Ciaffi, M. Identification and Validation of Reference Genes for Quantitative RT-PCR Normalization in Wheat. BMC Mol. Biol. 2009, 10, 11. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guérin, C.; Dupuits, C.; Mouzeyar, S.; Roche, J. Insights into Four NAC Transcription Factors Involved in Grain Development and in Response to Moderate Heat in the Triticeae Tribe. Int. J. Mol. Sci. 2022, 23, 11672. https://doi.org/10.3390/ijms231911672
Guérin C, Dupuits C, Mouzeyar S, Roche J. Insights into Four NAC Transcription Factors Involved in Grain Development and in Response to Moderate Heat in the Triticeae Tribe. International Journal of Molecular Sciences. 2022; 23(19):11672. https://doi.org/10.3390/ijms231911672
Chicago/Turabian StyleGuérin, Claire, Céline Dupuits, Said Mouzeyar, and Jane Roche. 2022. "Insights into Four NAC Transcription Factors Involved in Grain Development and in Response to Moderate Heat in the Triticeae Tribe" International Journal of Molecular Sciences 23, no. 19: 11672. https://doi.org/10.3390/ijms231911672
APA StyleGuérin, C., Dupuits, C., Mouzeyar, S., & Roche, J. (2022). Insights into Four NAC Transcription Factors Involved in Grain Development and in Response to Moderate Heat in the Triticeae Tribe. International Journal of Molecular Sciences, 23(19), 11672. https://doi.org/10.3390/ijms231911672







