Probing the Potential Mechanism of Quercetin and Kaempferol against Heat Stress-Induced Sertoli Cell Injury: Through Integrating Network Pharmacology and Experimental Validation
Abstract
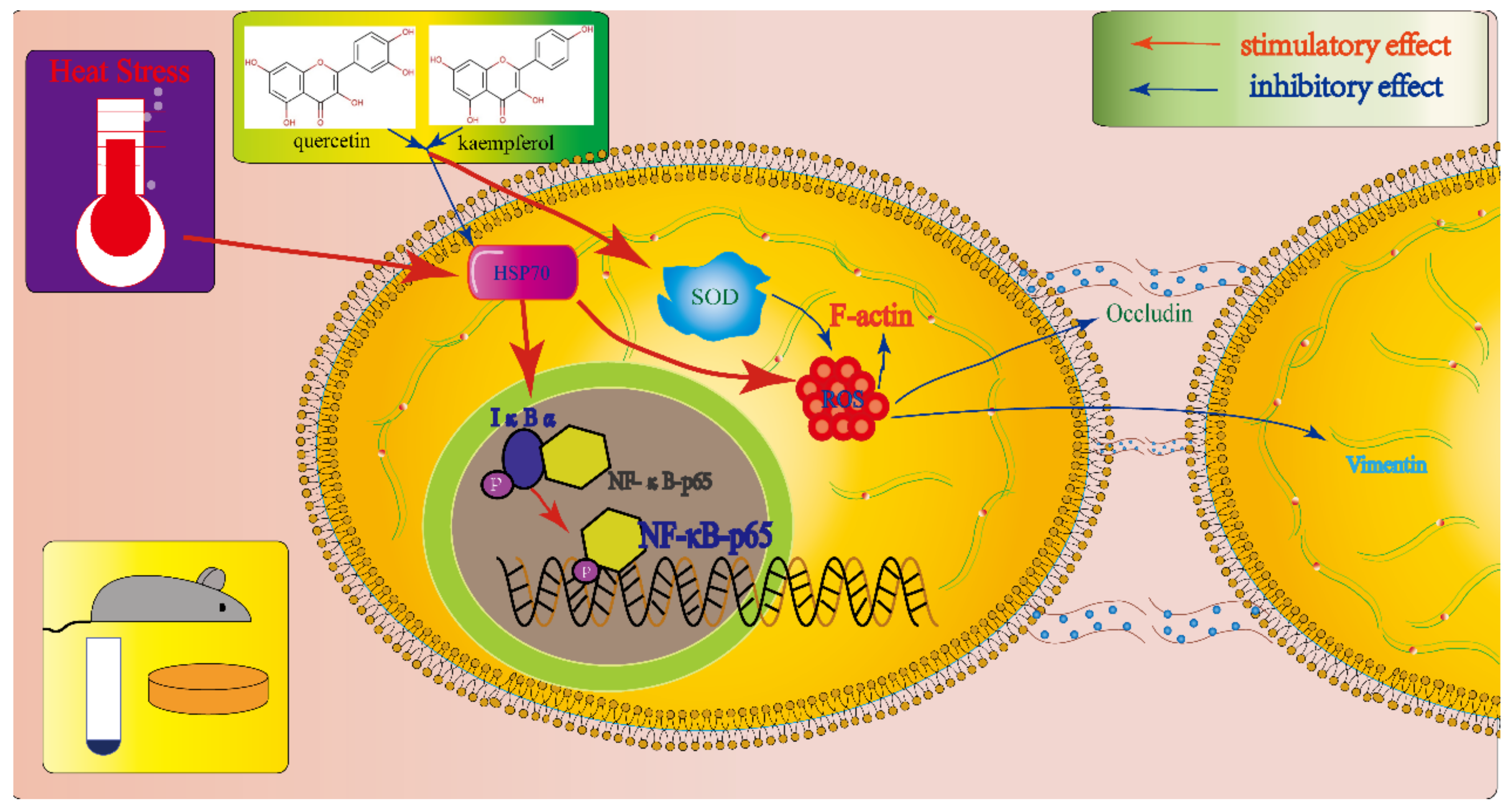
1. Introduction
2. Results
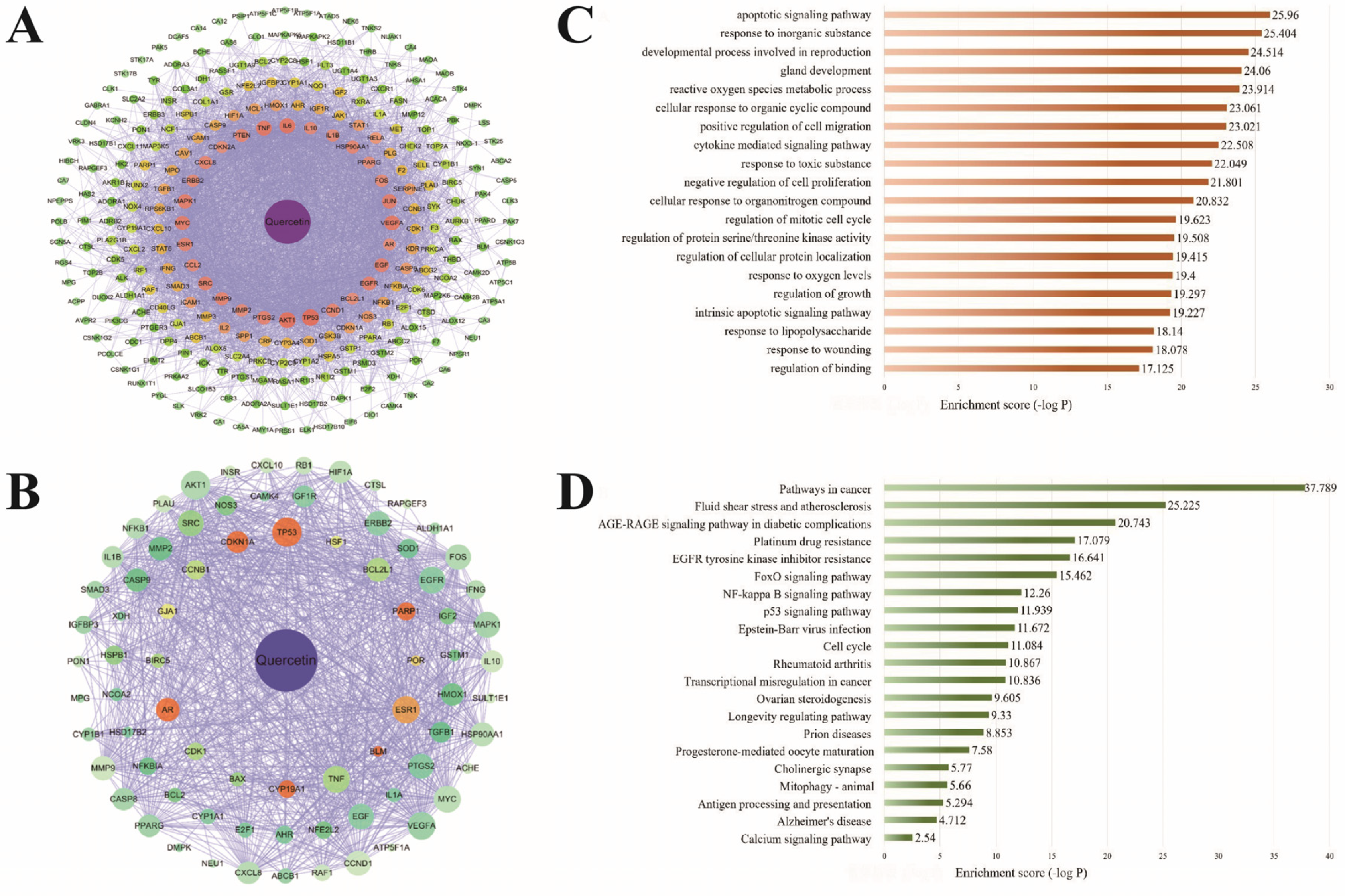
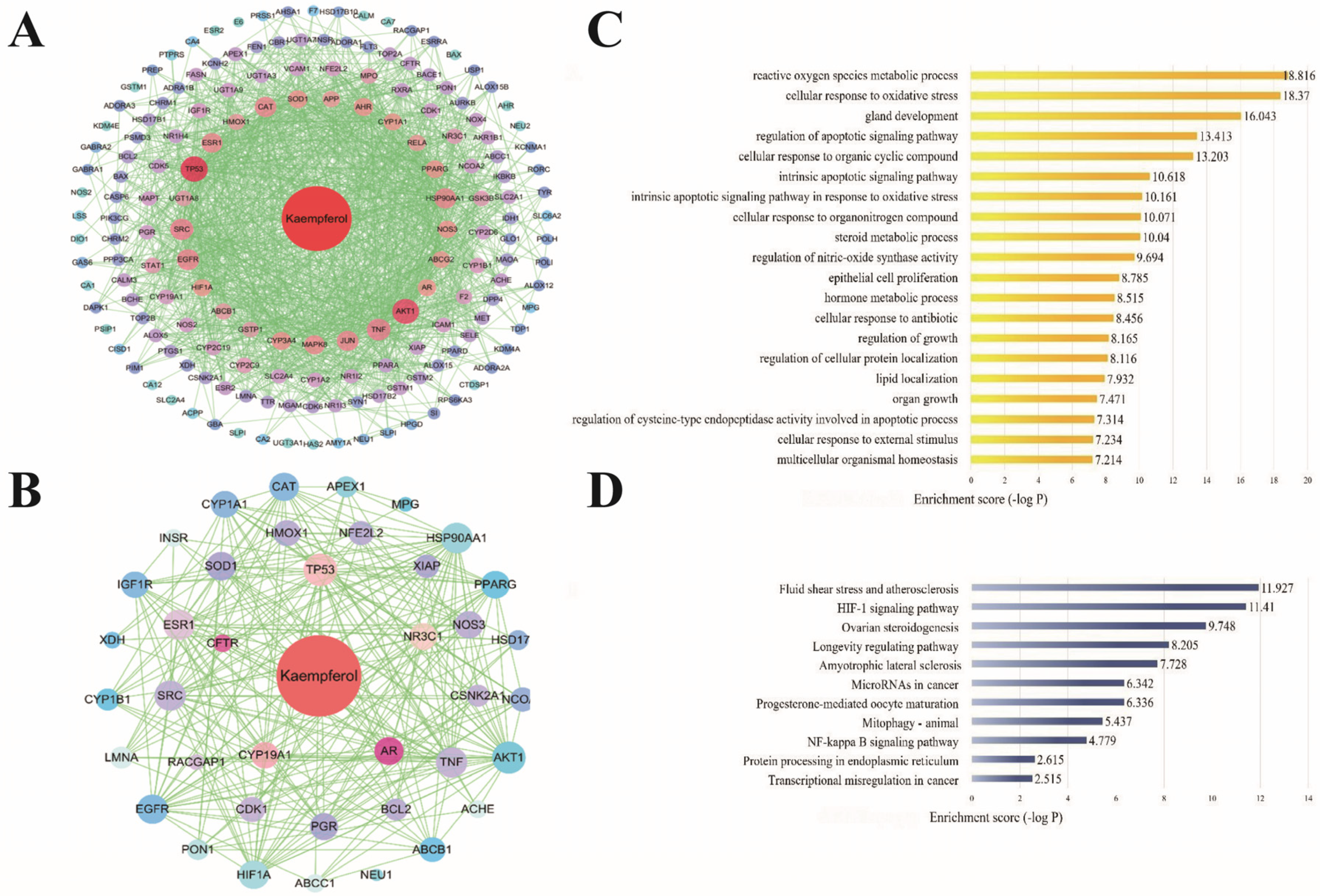
2.1. Target Identification
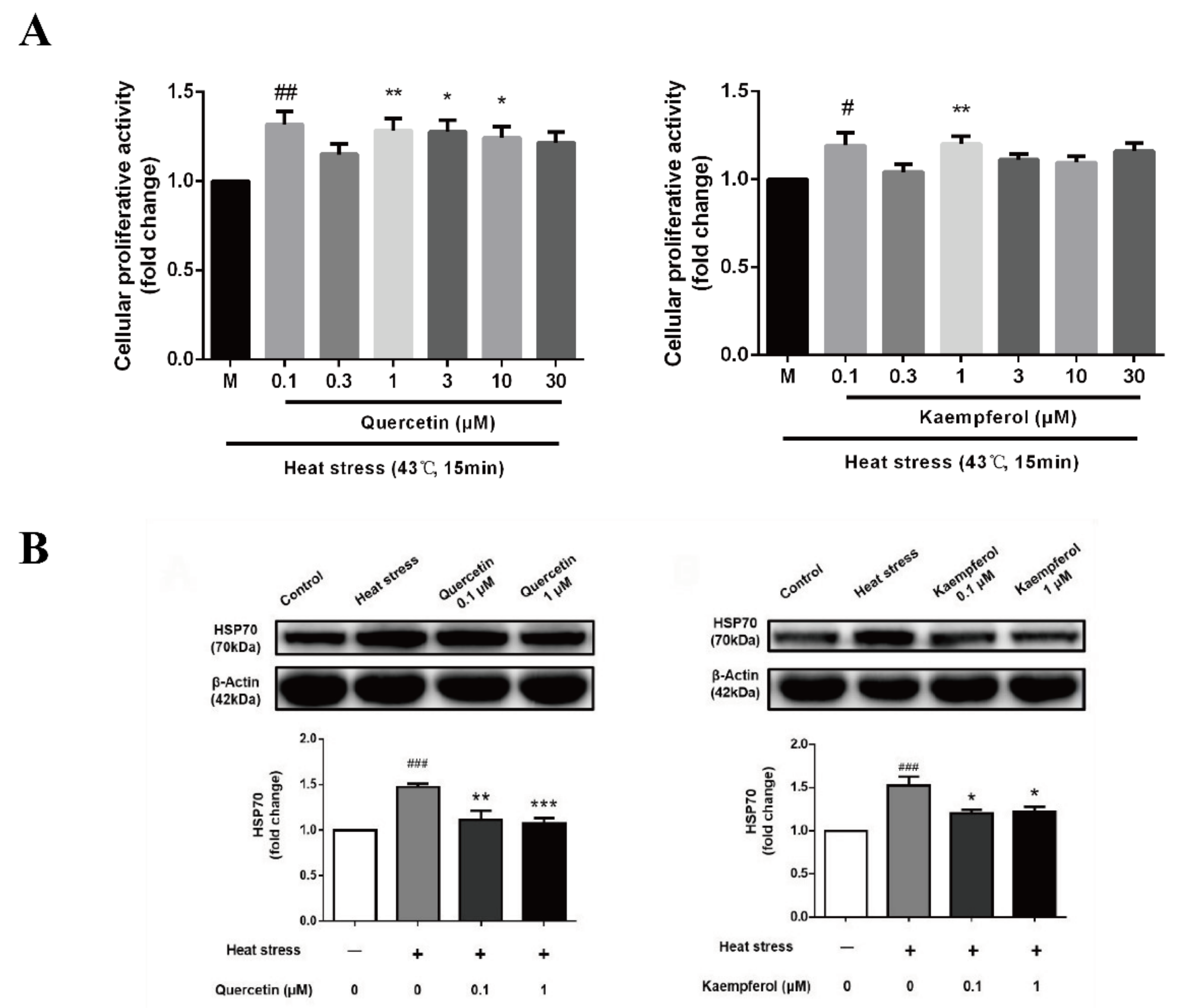
2.2. Effects of Quercetin and Kaempferol on SC Viability and Heat Shock Protein Expression after Heat Stress
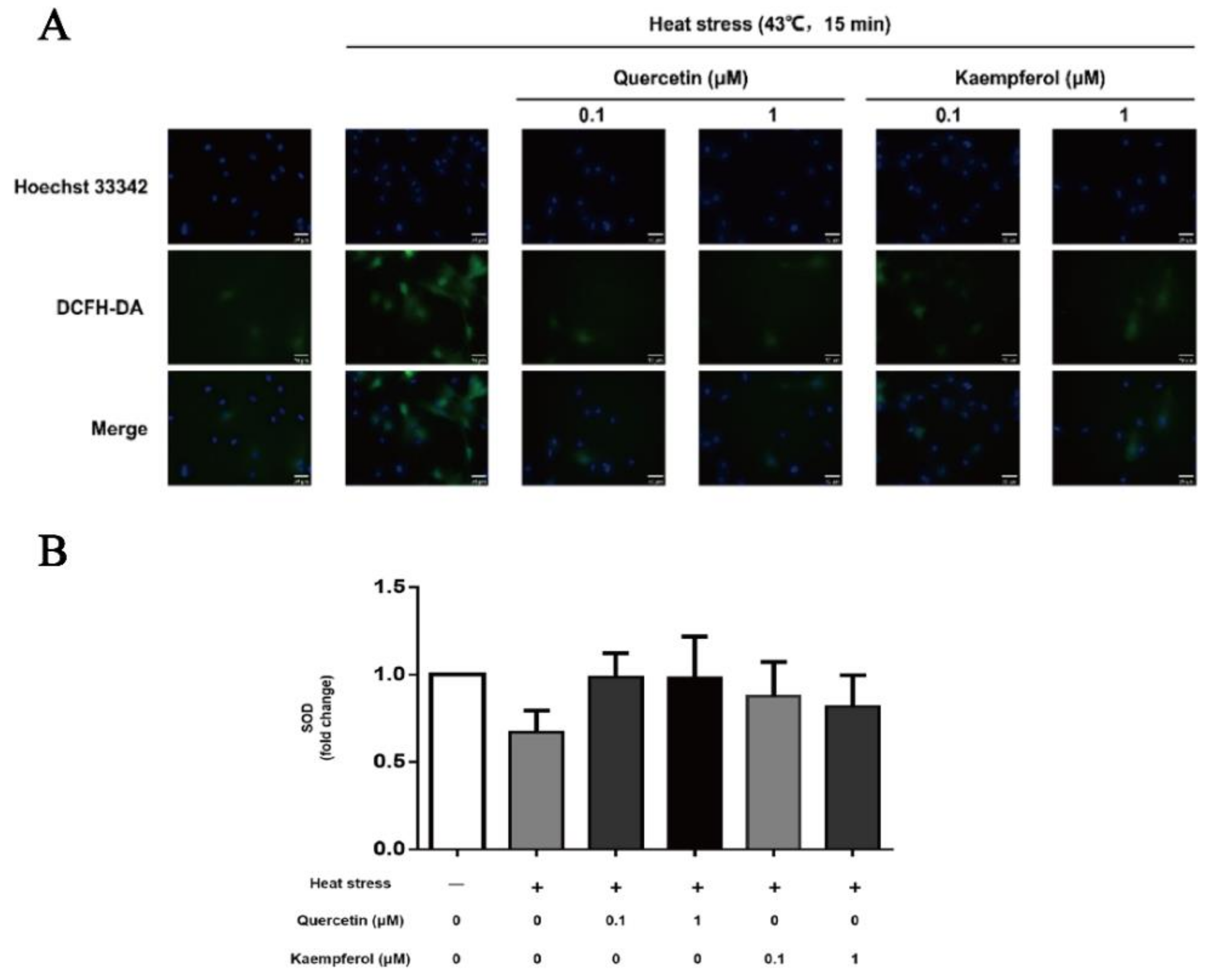
2.3. Effects of Quercetin and Kaempferol on the Expression of ROS and SOD in SCs after Heat Stress
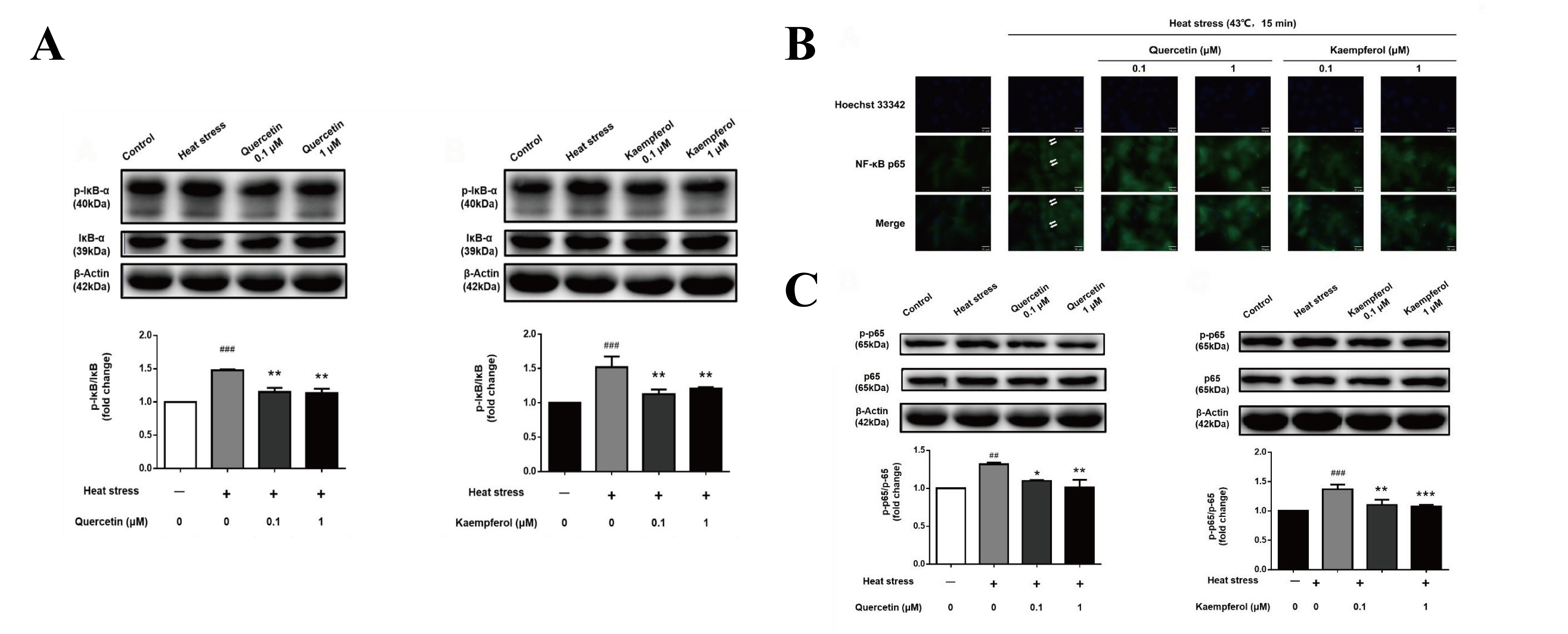
2.4. Effects of Quercetin and Kaempferol on the Expression of p-IκB and p-NFκB-p65 in SCs after Heat Stress
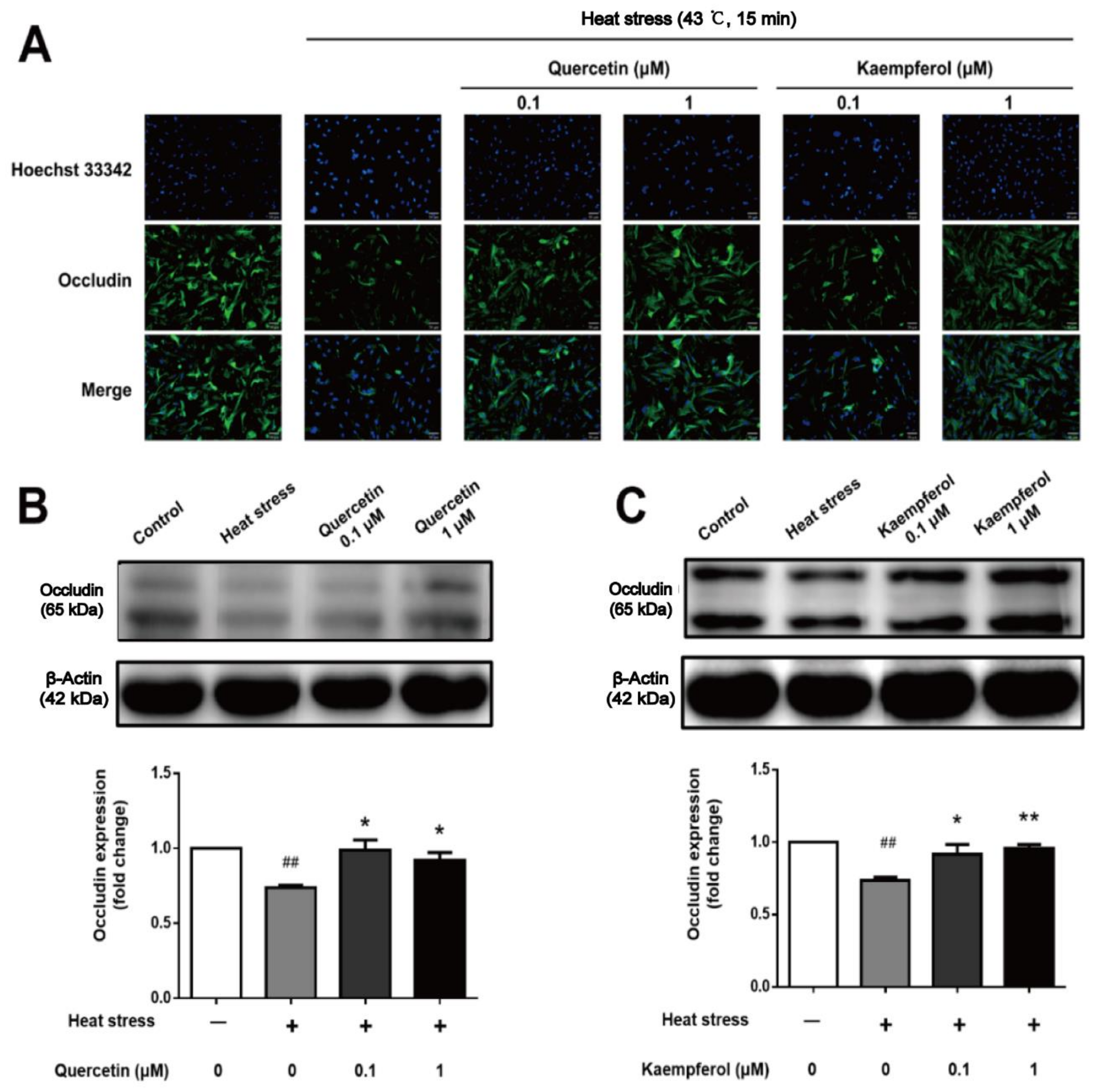
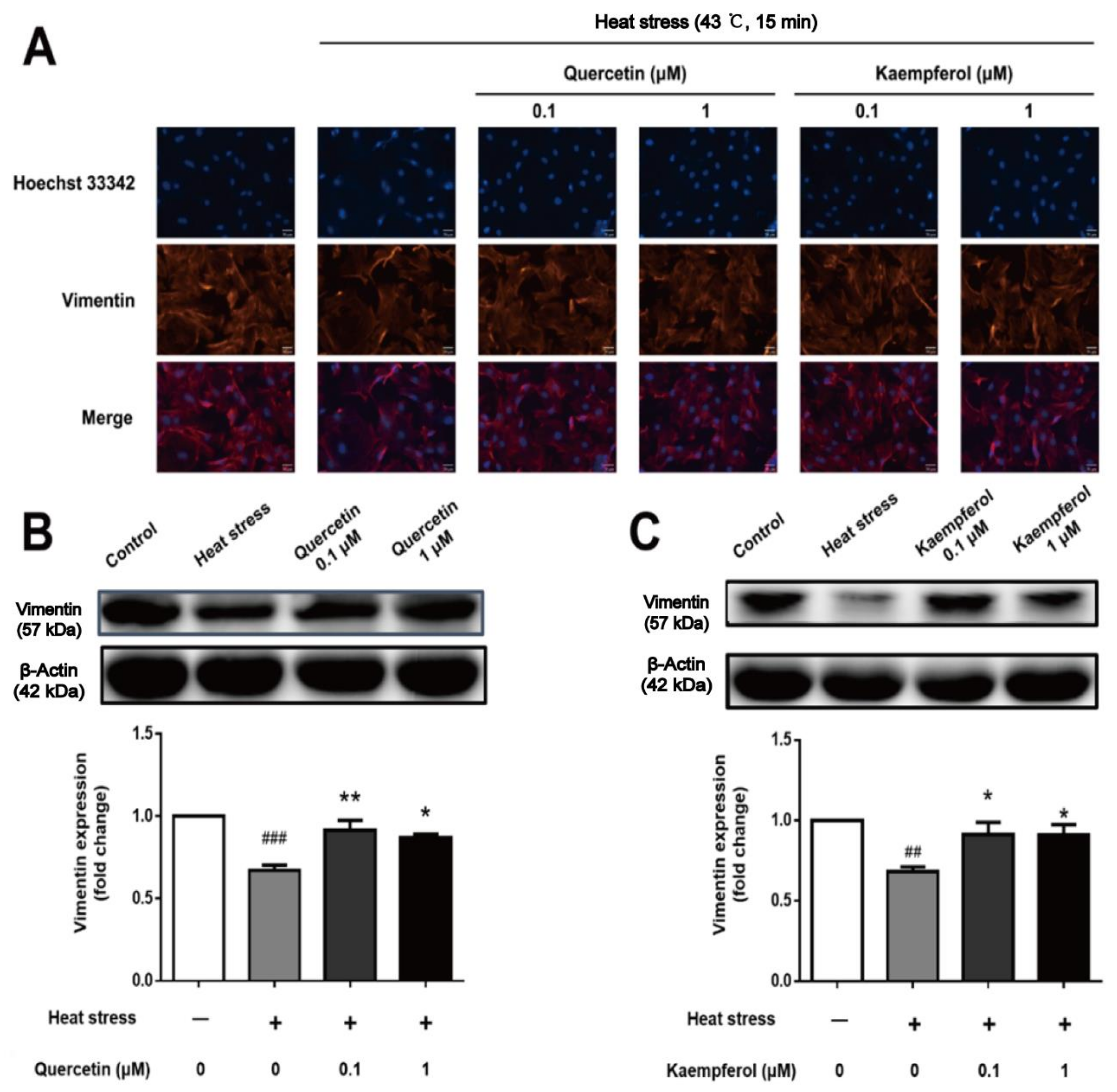
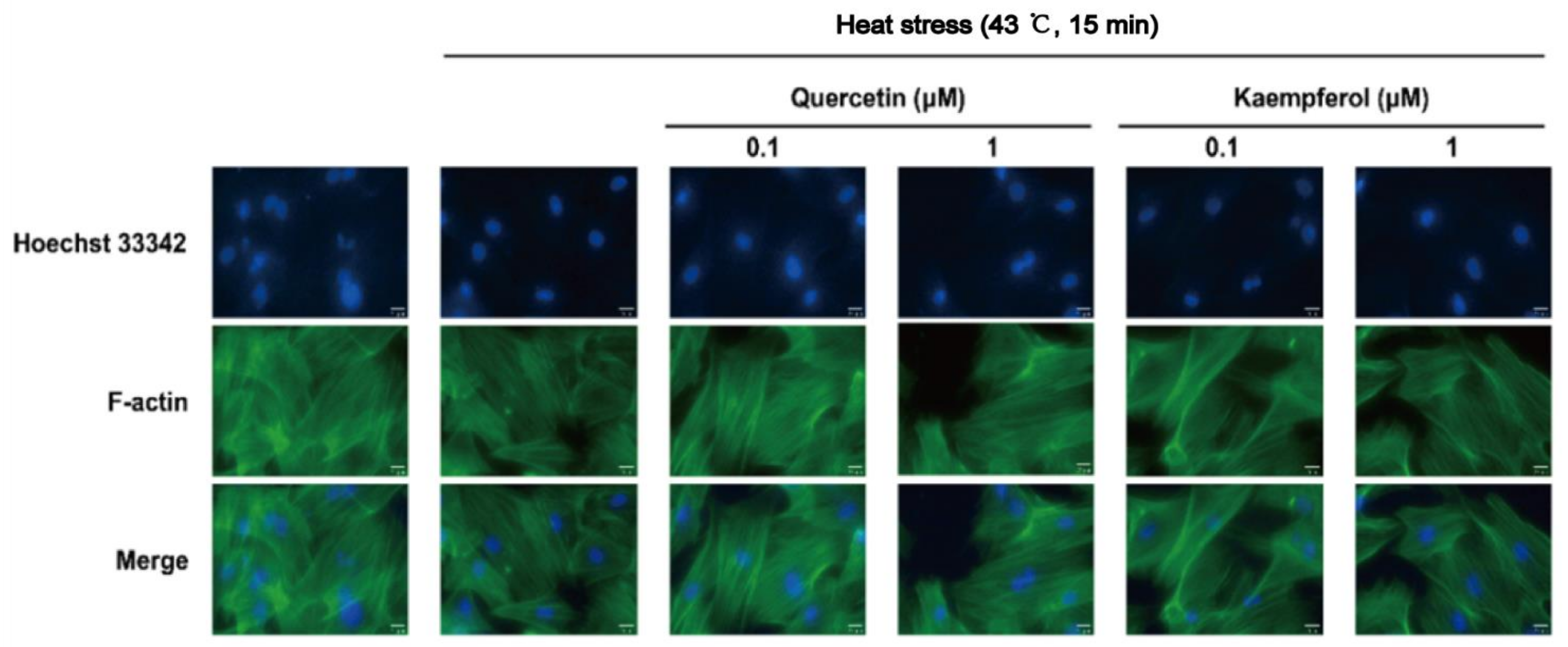
2.5. Effects of Quercetin and Kaempferol on Occludin, Vimentin, and F-actin in SCs after Heat Stress
3. Discussion
4. Materials and Methods
4.1. Online Databases and Software
4.2. Materials and Reagents
4.3. Cell Isolation, Culture, and Heat Stress Procedure
4.4. Cell Proliferation Assay
4.5. Determination of ROS by DCFH-DA
4.6. SOD Activity Assay
4.7. Western Blots
4.8. Immunofluorescence Staining
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Agarwal, A.; Finelli, R.; Selvam, M.K.P.; Leisegang, K.; Majzoub, A.; Tadros, N.; Ko, E.; Parekh, N.; Henkel, R.; Durairajanayagam, D.; et al. A Global Survey of Reproductive Specialists to Determine the Clinical Utility of Oxidative Stress Testing and Antioxidant Use in Male Infertility. World J. Mens. Health 2021, 39, 470–488. [Google Scholar] [CrossRef] [PubMed]
- Shahat, A.M.; Rizzoto, G.; Kastelic, J.P. Amelioration of heat stress-induced damage to testes and sperm quality. Theriogenology 2020, 158, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Kastelic, J.P.; Rizzoto, G.; Thundathil, J. Review: Testicular vascular cone development and its association with scrotal thermoregulation, semen quality and sperm production in bulls. Animal 2018, 12, s133–s141. [Google Scholar] [CrossRef] [PubMed]
- Afshar, A.; Aliaghaei, A.; Nazarian, H.; Abbaszadeh, H.A.; Naserzadeh, P.; Fathabadi, F.F.; Abdi, S.; Raee, P.; Aghajanpour, F.; Norouzian, M.; et al. Curcumin-Loaded Iron Particle Improvement of Spermatogenesis in Azoospermic Mouse Induced by Long-Term Scrotal Hyperthermia. Reprod. Sci. 2021, 28, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Ilkhani, S.; Moradi, A.; Aliaghaei, A.; Norouzian, M.; Abdi, S.; Rojhani, E.; Ebadinejad, A.; Sajadi, E.; Abdollahifar, M.A. Spatial arrangement of testicular cells disrupted by transient scrotal hyperthermia and subsequent impairment of spermatogenesis. Andrologia 2020, 52, e13664. [Google Scholar] [CrossRef]
- Shadmehr, S.; Fatemi Tabatabaei, S.R.; Hosseinifar, S.; Tabandeh, M.R.; Amiri, A. Attenuation of heat stress-induced spermatogenesis complications by betaine in mice. Theriogenology 2018, 106, 117–126. [Google Scholar] [CrossRef]
- Takeshima, T.; Usui, K.; Mori, K.; Asai, T.; Yasuda, K.; Kuroda, S.; Yumura, Y. Oxidative stress and male infertility. Reprod. Med. Biol. 2021, 20, 41–52. [Google Scholar] [CrossRef]
- Hedger, M.P. Macrophages and the immune responsiveness of the testis. J. Reprod. Immunol. 2002, 57, 19–34. [Google Scholar] [CrossRef]
- Samali, A.; Cotter, T.G. Heat shock proteins increase resistance to apoptosis. Exp. Cell Res. 1996, 223, 163–170. [Google Scholar] [CrossRef]
- Hespel, C.; Moser, M. Role of inflammatory dendritic cells in innate and adaptive immunity. Eur. J. Immunol. 2012, 42, 2535–2543. [Google Scholar] [CrossRef]
- Nicolas, N.; Michel, V.; Bhushan, S.; Wahle, E.; Hayward, S.; Ludlow, H.; de Kretser, D.M.; Loveland, K.L.; Schuppe, H.C.; Meinhardt, A.; et al. Testicular activin and follistatin levels are elevated during the course of experimental autoimmune epididymo-orchitis in mice. Sci. Rep. 2017, 7, 42391. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Wang, J.M.; Cai, Y.; Lu, S.F.; Zhu, B.M. Correlation between ischemic myocardial injury and inflammatory reaction, and anti-inflammatory effect of acupuncture. Zhen Ci Yan Jiu 2019, 44, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Sharma, R.K.; Desai, N.R.; Prabakaran, S.; Tavares, A.; Sabanegh, E. Role of oxidative stress in pathogenesis of varicocele and infertility. Urology 2009, 73, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Capela, L.; Leites, I.; Romao, R.; Lopes-da-Costa, L.; Pereira, R. Impact of Heat Stress on Bovine Sperm Quality and Competence. Animals 2022, 12, 975. [Google Scholar] [CrossRef]
- Pirani, M.; Novin, M.G.; Abdollahifar, M.A.; Piryaei, A.; Kuroshli, Z.; Mofarahe, Z.S. Protective Effects of Fisetin in the Mice Induced by Long-Term Scrotal Hyperthermia. Reprod. Sci. 2021, 28, 3123–3136. [Google Scholar] [CrossRef]
- Ziaeipour, S.; Ahrabi, B.; Naserzadeh, P.; Aliaghaei, A.; Sajadi, E.; Abbaszadeh, H.A.; Amini, A.; Abdi, S.; Darabi, S.; Abdollahifar, M.A. Effects of Sertoli Cell Transplantation on Spermatogenesis in Azoospermic Mice. Cell Physiol. Biochem. 2019, 52, 421–434. [Google Scholar] [CrossRef]
- Panahi, S.; Karamian, A.; Sajadi, E.; Aliaghaei, A.; Nazarian, H.; Abdi, S.; Danyali, S.; Paktinat, S.; Abdollahifar, M.A.; Farahani, R.M. Sertoli cell-conditioned medium restores spermatogenesis in azoospermic mouse testis. Cell Tissue Res. 2020, 379, 577–587. [Google Scholar] [CrossRef]
- Nash, S.; Rahman, M.S. Short-term heat stress impairs testicular functions in the American oyster, Crassostrea virginica: Molecular mechanisms and induction of oxidative stress and apoptosis in spermatogenic cells. Mol. Reprod. Dev. 2019, 86, 1444–1458. [Google Scholar] [CrossRef]
- Matschurat, C.; Rode, K.; Hollenbach, J.; Wolf, K.; Urhausen, C.; Beineke, A.; Gunzel-Apel, A.R.; Brehm, R. Impaired spermatogenesis, tubular wall disruption, altered blood-testis barrier composition and intratubular lymphocytes in an infertile Beagle dog—A putative case of autoimmune orchitis. Histol. Histopathol. 2019, 34, 525–535. [Google Scholar] [CrossRef]
- Wang, H.; Yuan, Q.; Sun, M.; Niu, M.; Wen, L.; Fu, H.; Zhou, F.; Chen, Z.; Yao, C.; Hou, J.; et al. BMP6 Regulates Proliferation and Apoptosis of Human Sertoli Cells Via Smad2/3 and Cyclin D1 Pathway and DACH1 and TFAP2A Activation. Sci. Rep. 2017, 7, 45298. [Google Scholar] [CrossRef]
- Cai, H.; Ren, Y.; Li, X.X.; Yang, J.L.; Zhang, C.P.; Chen, M.; Fan, C.H.; Hu, X.Q.; Hu, Z.Y.; Gao, F.; et al. Scrotal heat stress causes a transient alteration in tight junctions and induction of TGF-beta expression. Int. J. Androl. 2011, 34, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.S.; Zhao, Y.Y.; Zhao, L.N.; Wang, P.; Liu, Y.H.; Ma, J.; Xue, Y.X. MiR-18a increased the permeability of BTB via RUNX1 mediated down-regulation of ZO-1, occludin and claudin-5. Cell. Signal. 2015, 27, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Al-Obaidy, K.I.; Idrees, M.T.; Grignon, D.J.; Ulbright, T.M. Adenocarcinoma of the Rete Testis: Clinicopathologic and Immunohistochemical Characterization of 6 Cases and Review of the Literature. Am. J. Surg. Pathol. 2019, 43, 670–681. [Google Scholar] [CrossRef] [PubMed]
- Duan, M.; Xing, Y.; Guo, J.; Chen, H.; Zhang, R. Borneol increases blood-tumour barrier permeability by regulating the expression levels of tight junction-associated proteins. Pharm. Biol. 2016, 54, 3009–3018. [Google Scholar] [CrossRef][Green Version]
- Filardo, S.; Skilton, R.J.; O’Neill, C.E.; Di Pietro, M.; Sessa, R.; Clarke, I.N. Growth kinetics of Chlamydia trachomatis in primary human Sertoli cells. Sci. Rep. 2019, 9, 5847. [Google Scholar] [CrossRef]
- Khosravi, A.; Hasani, A.; Rahimi, K.; Aliaghaei, A.; Pirani, M.; Azad, N.; Ramezani, F.; Tamimi, A.; Behnam, P.; Raoofi, A.; et al. Ameliorating effects of curcumin-loaded superparamagnetic iron oxide nanoparticles (SPIONs) on the mouse testis exposed to the transient hyperthermia: A molecular and stereological study. Acta Histochem. 2020, 122, 151632. [Google Scholar] [CrossRef]
- Shen, L.; Tang, X.; Wei, Y.; Long, C.; Tan, B.; Wu, S.; Sun, M.; Zhou, Y.; Cao, X.; Wei, G. Vitamin E and vitamin C attenuate Di-(2-ethylhexyl) phthalate-induced blood-testis barrier disruption by p38 MAPK in immature SD rats. Reprod. Toxicol. 2018, 81, 17–27. [Google Scholar] [CrossRef]
- Zhang, P.; Zheng, Y.; Lv, Y.; Li, F.; Su, L.; Qin, Y.; Zeng, W. Melatonin protects the mouse testis against heat-induced damage. Mol. Hum. Reprod. 2020, 26, 65–79. [Google Scholar] [CrossRef]
- Zou, H.; Ye, H.; Kamaraj, R.; Zhang, T.; Zhang, J.; Pavek, P. A review on pharmacological activities and synergistic effect of quercetin with small molecule agents. Phytomed. Int. J. Phytother. Phytopharm. 2021, 92, 153736. [Google Scholar] [CrossRef]
- Tie, F.; Ding, J.; Hu, N.; Dong, Q.; Chen, Z.; Wang, H. Kaempferol and Kaempferide Attenuate Oleic Acid-Induced Lipid Accumulation and Oxidative Stress in HepG2 Cells. Int. J. Mol. Sci. 2021, 22, 8847. [Google Scholar] [CrossRef]
- Osawe, S.O.; Farombi, E.O. Quercetin and rutin ameliorates sulphasalazine-induced spermiotoxicity, alterations in reproductive hormones and steroidogenic enzyme imbalance in rats. Andrologia 2018, 50, e12981. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Li, P.L.; Hua, Y.L.; Ji, P.; Yao, W.L.; Zhang, X.S.; Zhong, L.J.; Wei, Y.M. Effects of Tao-Hong-Si-Wu decoction on acute blood stasis in rats based on a LC-Q/TOF-MS metabolomics and network approach. Biomed. Chromatogr. 2018, 32, e4144. [Google Scholar] [CrossRef] [PubMed]
- Archana, P.R.; Sejian, V.; Ruban, W.; Bagath, M.; Krishnan, G.; Aleena, J.; Manjunathareddy, G.B.; Beena, V.; Bhatta, R. Comparative assessment of heat stress induced changes in carcass traits, plasma leptin profile and skeletal muscle myostatin and HSP70 gene expression patterns between indigenous Osmanabadi and Salem Black goat breeds. Meat Sci. 2018, 141, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Bukau, B.; Weissman, J.; Horwich, A. Molecular chaperones and protein quality control. Cell 2006, 125, 443–451. [Google Scholar] [CrossRef]
- Asea, A.; Kraeft, S.K.; Kurt-Jones, E.A.; Stevenson, M.A.; Chen, L.B.; Finberg, R.W.; Koo, G.C.; Calderwood, S.K. HSP70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat. Med. 2000, 6, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Oghbaei, H.; Hosseini, L.; Farajdokht, F.; Rahigh Aghsan, S.; Majdi, A.; Sadigh-Eteghad, S.; Sandoghchian Shotorbani, S.; Mahmoudi, J. Heat stress aggravates oxidative stress, apoptosis, and endoplasmic reticulum stress in the cerebellum of male C57 mice. Mol. Biol. Rep. 2021, 48, 5881–5887. [Google Scholar] [CrossRef]
- Kim, S.J.; Park, J.H.; Lee, S.A.; Lee, J.G.; Shin, J.M.; Lee, H.M. All-trans retinoic acid regulates TGF-β1-induced extracellular matrix production via p38, JNK, and NF-κB-signaling pathways in nasal polyp-derived fibroblasts. Int. Forum Allergy Rhinol. 2020, 10, 636–645. [Google Scholar] [CrossRef]
- Cheng, C.Y. Toxicants target cell junctions in the testis: Insights from the indazole-carboxylic acid model. Spermatogenesis 2014, 4, e981485. [Google Scholar] [CrossRef]
- Ozden, C.; Ozdal, O.L.; Bulut, S.; Guzel, O.; Koyuncu, H.H.; Memis, A. Effect of varicocelectomy on serum inhibin B levels in infertile patients with varicocele. Scand. J. Urol. Nephrol. 2008, 42, 441–443. [Google Scholar] [CrossRef]
- Boyle, C.J.; Plotczyk, M.; Villalta, S.F.; Patel, S.; Hettiaratchy, S.; Masouros, S.D.; Masen, M.A.; Higgins, C.A. Morphology and composition play distinct and complementary roles in the tolerance of plantar skin to mechanical load. Sci. Adv. 2019, 5, eaay0244. [Google Scholar] [CrossRef]
- Liu, Z.; Zou, D.; Yang, X.; Xue, X.; Zuo, L.; Zhou, Q.; Hu, R.; Wang, Y. Melatonin inhibits colon cancer RKO cell migration by downregulating Rhoassociated protein kinase expression via the p38/MAPK signaling pathway. Mol. Med. Rep. 2017, 16, 9383–9392. [Google Scholar] [CrossRef] [PubMed]
- Kothakota, S.; Azuma, T.; Reinhard, C.; Klippel, A.; Tang, J.; Chu, K.; McGarry, T.J.; Kirschner, M.W.; Koths, K.; Kwiatkowski, D.J.; et al. Caspase-3-generated fragment of gelsolin: Effector of morphological change in apoptosis. Science 1997, 278, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Wen, K.; Fang, X.; Yang, J.; Yao, Y.; Nandakumar, K.S.; Salem, M.L.; Cheng, K. Recent Research on Flavonoids and their Biomedical Applications. Curr. Med. Chem. 2021, 28, 1042–1066. [Google Scholar] [CrossRef] [PubMed]
- Zeng, P.; Wang, X.M.; Ye, C.Y.; Su, H.F.; Tian, Q. The Main Alkaloids in Uncaria rhynchophylla and Their Anti-Alzheimer’s Disease Mechanism Determined by a Network Pharmacology Approach. Int. J. Mol. Sci. 2021, 22, 3612. [Google Scholar] [CrossRef]
- Habtemariam, S.; Belai, A. Natural Therapies of the Inflammatory Bowel Disease: The Case of Rutin and its Aglycone, Quercetin. Mini Rev. Med. Chem. 2018, 18, 234–243. [Google Scholar] [CrossRef]
- Khodabandeh, Z.; Dolati, P.; Zamiri, M.J.; Mehrabani, D.; Bordbar, H.; Alaee, S.; Jamhiri, I.; Azarpira, N. Protective Effect of Quercetin on Testis Structure and Apoptosis Against Lead Acetate Toxicity: An Stereological Study. Biol. Trace Elem. Res. 2021, 199, 3371–3381. [Google Scholar] [CrossRef]
- Amasheh, M.; Schlichter, S.; Amasheh, S.; Mankertz, J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Quercetin enhances epithelial barrier function and increases claudin-4 expression in Caco-2 cells. J. Nutr. 2008, 138, 1067–1073. [Google Scholar] [CrossRef]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Analysis of Cell Viability by the MTT Assay. Cold Spring Harbor Protoc. 2018, 2018. [Google Scholar] [CrossRef]
- Qin, D.Z.; Cai, H.; He, C.; Yang, D.H.; Sun, J.; He, W.L.; Li, B.L.; Hua, J.L.; Peng, S. Melatonin relieves heat-induced spermatocyte apoptosis in mouse testes by inhibition of ATF6 and PERK signaling pathways. Zool. Res. 2021, 42, 514–524. [Google Scholar] [CrossRef]
- Alahmar, A.T.; Calogero, A.E.; Singh, R.; Cannarella, R.; Sengupta, P.; Dutta, S. Coenzyme Q10, oxidative stress, and male infertility: A review. Clin. Exp. Reprod. Med. 2021, 48, 97–104. [Google Scholar] [CrossRef]
- Yan, W.; Qiao, Y.; He, J.; Qu, J.; Liu, Y.; Zhang, Q.; Wang, X. Molecular Mechanism Based on Histopathology, Antioxidant System and Transcriptomic Profiles in Heat Stress Response in the Gills of Japanese Flounder. Int. J. Mol. Sci. 2022, 23, 3286. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Hashmi, S.A.; Rehman, R. Oxidative stress, vitamin D deficiency and male infertility: An under-looked aspect. J. Pak. Med. Assoc. 2021, 71, 786–787. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, C.; Ko, E.Y. Oxidative stress in the pathophysiology of male infertility. Andrologia 2021, 53, e13581. [Google Scholar] [CrossRef] [PubMed]
- Hallak, J.; Teixeira, T.A. Oxidative Stress & Male Infertility—A necessary and conflicted indissociable marriage: How and when to call for evaluation? Int. Braz. J. Urol. 2021, 47, 686–689. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Khan, S.; Ashraf, M.; Akram Mudassir, H.; Rehman, R. Male infertility: Role of vitamin D and oxidative stress markers. Andrologia 2021, 53, e14147. [Google Scholar] [CrossRef]
- Kapuscinski, J. DAPI: A DNA-specific fluorescent probe. Biotech. Histochem. 1995, 70, 220–233. [Google Scholar] [CrossRef]
- Zhao, H.; Li, J.; Traganos, F.; Halicka, H.D.; Zarebski, M.; Dobrucki, J.; Darzynkiewicz, Z. Cell fixation in zinc salt solution is compatible with DNA damage response detection by phospho-specific antibodies. Cytom. Part A J. Int. Soc. Anal. Cytol. 2011, 79, 470–476. [Google Scholar] [CrossRef]
- Gregoire, M.; Hernandez-Verdun, D.; Bouteille, M. Visualization of chromatin distribution in living PTO cells by Hoechst 33342 fluorescent staining. Exp. Cell Res. 1984, 152, 38–46. [Google Scholar] [CrossRef]
- Crowley, L.C.; Marfell, B.J.; Waterhouse, N.J. Analyzing Cell Death by Nuclear Staining with Hoechst 33342. Cold Spring Harbor Protoc. 2016, 2016, pdb-prot087205. [Google Scholar] [CrossRef]
- Hasani, A.; Khosravi, A.; Rahimi, K.; Afshar, A.; Fadaei-Fathabadi, F.; Raoofi, A.; Raee, P.; Aghajanpour, F.; Aliaghaei, A.; Abdi, S.; et al. Photobiomodulation restores spermatogenesis in the transient scrotal hyperthermia-induced mice. Life Sci. 2020, 254, 117767. [Google Scholar] [CrossRef]
- Hung, J.H.; Chen, C.Y.; Omar, H.A.; Huang, K.Y.; Tsao, C.C.; Chiu, C.C.; Chen, Y.L.; Chen, P.H.; Teng, Y.N. Reactive oxygen species mediate Terbufos-induced apoptosis in mouse testicular cell lines via the modulation of cell cycle and pro-apoptotic proteins. Environ. Toxicol. 2016, 31, 1888–1898. [Google Scholar] [CrossRef] [PubMed]
- Yin, B.; Tang, S.; Sun, J.; Zhang, X.; Xu, J.; Di, L.; Li, Z.; Hu, Y.; Bao, E. Vitamin C and sodium bicarbonate enhance the antioxidant ability of H9C2 cells and induce HSPs to relieve heat stress. Cell Stress Chaperones 2018, 23, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, T.; Tan, P.; Shi, H.; Cheng, Y.; Cai, T.; Bai, J.; Du, Y.; Fu, W. Kaempferol from Penthorum chinense Pursh Attenuates Hepatic Ischemia/Reperfusion Injury by Suppressing Oxidative Stress and Inflammation Through Activation of the Nrf2/HO-1 Signaling Pathway. Front. Pharmacol. 2022, 13, 857015. [Google Scholar] [CrossRef] [PubMed]
- Bakhshii, S.; Khezri, S.; Ahangari, R.; Jahedsani, A.; Salimi, A. Protection of clozapine-induced oxidative stress and mitochondrial dysfunction by kaempferol in rat cardiomyocytes. Drug Dev. Res. 2021, 82, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Aslani, F.; Schuppe, H.C.; Guazzone, V.A.; Bhushan, S.; Wahle, E.; Lochnit, G.; Lustig, L.; Meinhardt, A.; Fijak, M. Targeting high mobility group box protein 1 ameliorates testicular inflammation in experimental autoimmune orchitis. Hum. Reprod. 2015, 30, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Wahlqvist, M.L. Antioxidant relevance to human health. Asia Pac. J. Clin. Nutr. 2013, 22, 171–176. [Google Scholar] [CrossRef]
- Li, Q.; Mi, Y.; Tan, W.; Guo, Z. Highly efficient free radical-scavenging property of phenolic-functionalized chitosan derivatives: Chemical modification and activity assessment. Int. J. Biol. Macromol. 2020, 164, 4279–4288. [Google Scholar] [CrossRef]
- El-Beltagy, A.E.; Alharthi, S. Free Radical Scavenging Activity of Some Legumes Hulls Extract and Its Efficacy on Oil Oxidative Stability. J. AOAC Int. 2021, 104, 472–478. [Google Scholar] [CrossRef]
- Boulebd, H.; Amine Khodja, I. A detailed DFT-based study of the free radical scavenging activity and mechanism of daphnetin in physiological environments. Phytochemistry 2021, 189, 112831. [Google Scholar] [CrossRef]
- Akefe, I.O.; Ayo, J.O.; Sinkalu, V.O. Kaempferol and zinc gluconate mitigate neurobehavioral deficits and oxidative stress induced by noise exposure in Wistar rats. PLoS ONE 2020, 15, e0236251. [Google Scholar] [CrossRef]
- Lin, M.-K.; Yu, Y.-L.; Chen, K.-C.; Chang, W.-T.; Lee, M.-S.; Yang, M.-J.; Cheng, H.-C.; Liu, C.-H.; Chen, C.-D.; Chu, C.-L. Kaempferol from Semen cuscutae attenuates the immune function of dendritic cells. Immunobiology 2011, 216, 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Kucheryavenko, O.; Wordsworth, J.; von Zglinicki, T. The senescent bystander effect is caused by ROS-activated NF-kappaB signalling. Mech. Ageing Dev. 2018, 170, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.R.; Li, B.B.; Hu, Y.; Zhang, L.; Wang, X.Z. Oxidative stress mediates heat-induced changes of tight junction proteins in porcine sertoli cells via inhibiting CaMKKbeta-AMPK pathway. Theriogenology 2020, 142, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, T.; Feng, J.; Li, L.; Wang, R.; Cheng, H.; Yuan, Y. Kaempferol protects against gamma radiation-induced mortality and damage via inhibiting oxidative stress and modulating apoptotic molecules in vivo and vitro. Environ. Toxicol. Pharmacol. 2018, 60, 128–137. [Google Scholar] [CrossRef]
- Pentikainen, V.; Erkkila, K.; Suomalainen, L.; Parvinen, M.; Dunkel, L. Estradiol acts as a germ cell survival factor in the human testis in vitro. J. Clin. Endocrinol. Metab. 2000, 85, 2057–2067. [Google Scholar] [CrossRef]
- Hong, C.Y.; Park, J.H.; Seo, K.H.; Kim, J.M.; Im, S.Y.; Lee, J.W.; Choi, H.S.; Lee, K. Expression of MIS in the testis is downregulated by tumor necrosis factor alpha through the negative regulation of SF-1 transactivation by NF-kappa B. Mol. Cell. Biol. 2003, 23, 6000–6012. [Google Scholar] [CrossRef]
- Silva, E.J.R.; Ribeiro, C.M.; Mirim, A.F.M.; Silva, A.A.S.; Romano, R.M.; Hallak, J.; Avellar, M.C.W. Lipopolysaccharide and lipotheicoic acid differentially modulate epididymal cytokine and chemokine profiles and sperm parameters in experimental acute epididymitis. Sci. Rep. 2018, 8, 103. [Google Scholar] [CrossRef]
- Ray, M.; Yu, S.; Sharda, D.R.; Wilson, C.B.; Liu, Q.; Kaushal, N.; Prabhu, K.S.; Hankey, P.A. Inhibition of TLR4-induced IkappaB kinase activity by the RON receptor tyrosine kinase and its ligand, macrophage-stimulating protein. J. Immunol. 2010, 185, 7309–7316. [Google Scholar] [CrossRef]
- Endale, M.; Park, S.C.; Kim, S.; Kim, S.H.; Yang, Y.; Cho, J.Y.; Rhee, M.H. Quercetin disrupts tyrosine-phosphorylated phosphatidylinositol 3-kinase and myeloid differentiation factor-88 association, and inhibits MAPK/AP-1 and IKK/NF-kappaB-induced inflammatory mediators production in RAW 264.7 cells. Immunobiology 2013, 218, 1452–1467. [Google Scholar] [CrossRef]
- Ramgir, S.S.; Renu, K.; Vellingiri, B.; George, A.; Tirupapuliyur, D.; Thiagarajan, P.; Valsala Gopalakrishnan, A. Phytomedicinal therapeutics for male infertility: Critical insights and scientific updates. J. Nat. Med. 2022, 76, 546–573. [Google Scholar] [CrossRef]
- Kim, M.; Lim, S.J.; Kang, S.W.; Um, B.H.; Nho, C.W. Aceriphyllum rossii extract and its active compounds, quercetin and kaempferol inhibit IgE-mediated mast cell activation and passive cutaneous anaphylaxis. J. Agric. Food Chem. 2014, 62, 3750–3758. [Google Scholar] [CrossRef] [PubMed]
- Ni, F.D.; Hao, S.L.; Yang, W.X. Molecular insights into hormone regulation via signaling pathways in Sertoli cells: With discussion on infertility and testicular tumor. Gene 2020, 753, 144812. [Google Scholar] [CrossRef] [PubMed]
- Hayrabedyan, S.; Todorova, K.; Pashova, S.; Mollova, M.; Fernandez, N. Sertoli Cell Quiescence—New Insights. Am. J. Reprod. Immunol. 2012, 68, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Hayrabedyan, S.B.; Zasheva, D.Y.; Todorova, K.O. NLRs Challenge Impacts Tight Junction Claudins in Sertoli Cells. Folia Med. 2015, 57, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Chung, N.P.Y.; Cheng, C.Y. Is cadmium chloride-induced inter-sertoli tight junction permeability barrier disruption a suitable in vitro model to study the events of junction disassembly during spermatogenesis in the rat testis? Endocrinology 2001, 142, 1878–1888. [Google Scholar] [CrossRef]
- Zheng, Y.; Gao, Q.; Li, T.; Liu, R.; Cheng, Z.; Guo, M.; Xiao, J.; Wu, D.; Zeng, W. Sertoli cell and spermatogonial development in pigs. J. Anim. Sci. Biotechnol. 2022, 13, 45. [Google Scholar] [CrossRef]
- Gul, U.; Turunc, T.; Haydardedeoglu, B.; Yaycioglu, O.; Kuzgunbay, B.; Ozkardes, H. Sperm retrieval and live birth rates in presumed Sertoli-cell-only syndrome in testis biopsy: A single centre experience. Andrology 2013, 1, 47–51. [Google Scholar] [CrossRef]
- Taitson, P.F.; Mourthe, A.F.; Radaelli, M.R.M. Testicular sperm extraction in men with sertoli cell-only testicular histology—1680 cases. JBRA Assist. Reprod. 2019, 23, 246–249. [Google Scholar] [CrossRef]
- Abofoul-Azab, M.; Lunenfeld, E.; Levitas, E.; Zeadna, A.; Younis, J.S.; Bar-Ami, S.; Huleihel, M. Identification of Premeiotic, Meiotic, and Postmeiotic Cells in Testicular Biopsies Without Sperm from Sertoli Cell-Only Syndrome Patients. Int. J. Mol. Sci. 2019, 20, 470. [Google Scholar] [CrossRef]
- Modarresi, T.; Hosseinifar, H.; Daliri Hampa, A.; Chehrazi, M.; Hosseini, J.; Farrahi, F.; Dadkhah, F.; Sabbaghian, M.; Sadighi Gilani, M.A. Predictive factors of successful microdissection testicular sperm extraction in patients with presumed sertoli cell-only syndrome. Int. J. Fertil. Steril. 2015, 9, 107–112. [Google Scholar] [CrossRef]
- Xie, X.; Zhou, W.; Hu, Y.; Chen, Y.; Zhang, H.; Li, Y. A dual-function epidermal growth factor receptor pathway substrate 8 (Eps8)-derived peptide exhibits a potent cytotoxic T lymphocyte-activating effect and a specific inhibitory activity. Cell Death Dis. 2018, 9, 379. [Google Scholar] [CrossRef] [PubMed]
- Lie, P.P.; Mruk, D.D.; Lee, W.M.; Cheng, C.Y. Epidermal growth factor receptor pathway substrate 8 (Eps8) is a novel regulator of cell adhesion and the blood-testis barrier integrity in the seminiferous epithelium. FASEB J. 2009, 23, 2555–2567. [Google Scholar] [CrossRef]
- He, X.; Sun, Z.; Manthari, R.K.; Wu, P.; Wang, J. Fluoride altered rat’s blood testis barrier by affecting the F-actin via IL-1alpha. Chemosphere 2018, 211, 826–833. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Mruk, D.D.; Tang, E.I.; Wong, C.K.; Lee, W.M.; John, C.M.; Turek, P.J.; Silvestrini, B.; Cheng, C.Y. Environmental toxicants perturb human Sertoli cell adhesive function via changes in F-actin organization mediated by actin regulatory proteins. Hum. Reprod. 2014, 29, 1279–1291. [Google Scholar] [CrossRef] [PubMed]
- Goley, E.D.; Welch, M.D. The ARP2/3 complex: An actin nucleator comes of age. Nat. Rev. Mol. Cell Biol. 2006, 7, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Liu, Y.; Wang, L.; Xue, F.; Hu, Y.; Hu, R.; Xu, C. MKP-1 attenuates LPS-induced blood-testis barrier dysfunction and inflammatory response through p38 and IkappaBalpha pathways. Oncotarget 2016, 7, 84907–84923. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Kar, A.; Mukherjee, P.K.; Haldar, P.K.; Sharma, N.; Katiyar, C.K. Immunoprotective potential of Ayurvedic herb Kalmegh (Andrographis paniculata) against respiratory viral infections—LC-MS/MS and network pharmacology analysis. Phytochem. Anal. 2021, 32, 629–639. [Google Scholar] [CrossRef]
- Ru, J.; Li, P.; Wang, J.; Zhou, W.; Li, B.; Huang, C.; Li, P.; Guo, Z.; Tao, W.; Yang, Y.; et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J. Cheminform. 2014, 6, 13. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef]
- UniProt Consortium. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D480–D489. [Google Scholar] [CrossRef]
- Amberger, J.S.; Bocchini, C.A.; Schiettecatte, F.; Scott, A.F.; Hamosh, A. OMIM.org: Online Mendelian Inheritance in Man (OMIM(R)), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 2015, 43, D789–D798. [Google Scholar] [CrossRef] [PubMed]
- Stelzer, G.; Rosen, N.; Plaschkes, I.; Zimmerman, S.; Twik, M.; Fishilevich, S.; Stein, T.I.; Nudel, R.; Lieder, I.; Mazor, Y.; et al. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Curr. Protoc. Bioinform. 2016, 54, 1–30. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Stelzer, G.; Plaschkes, I.; Oz-Levi, D.; Alkelai, A.; Olender, T.; Zimmerman, S.; Twik, M.; Belinky, F.; Fishilevich, S.; Nudel, R.; et al. VarElect: The phenotype-based variation prioritizer of the GeneCards Suite. BMC Genom. 2016, 17 (Suppl. S2). [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, D.-L.; Liu, S.-J.; Hu, S.-Q.; Chen, Y.-C.; Guo, J. Probing the Potential Mechanism of Quercetin and Kaempferol against Heat Stress-Induced Sertoli Cell Injury: Through Integrating Network Pharmacology and Experimental Validation. Int. J. Mol. Sci. 2022, 23, 11163. https://doi.org/10.3390/ijms231911163
Liu D-L, Liu S-J, Hu S-Q, Chen Y-C, Guo J. Probing the Potential Mechanism of Quercetin and Kaempferol against Heat Stress-Induced Sertoli Cell Injury: Through Integrating Network Pharmacology and Experimental Validation. International Journal of Molecular Sciences. 2022; 23(19):11163. https://doi.org/10.3390/ijms231911163
Chicago/Turabian StyleLiu, Dian-Long, Si-Jia Liu, Su-Qin Hu, Yu-Cai Chen, and Jian Guo. 2022. "Probing the Potential Mechanism of Quercetin and Kaempferol against Heat Stress-Induced Sertoli Cell Injury: Through Integrating Network Pharmacology and Experimental Validation" International Journal of Molecular Sciences 23, no. 19: 11163. https://doi.org/10.3390/ijms231911163
APA StyleLiu, D.-L., Liu, S.-J., Hu, S.-Q., Chen, Y.-C., & Guo, J. (2022). Probing the Potential Mechanism of Quercetin and Kaempferol against Heat Stress-Induced Sertoli Cell Injury: Through Integrating Network Pharmacology and Experimental Validation. International Journal of Molecular Sciences, 23(19), 11163. https://doi.org/10.3390/ijms231911163





