Galectins—Potential Therapeutic Targets for Neurodegenerative Disorders
Abstract
1. Introduction
2. Physiological Role of Gals
2.1. Gal-1
2.2. Gal-3
3. Galectins as Extracellular Binding Proteins
4. Galectins Auto-Antibodies
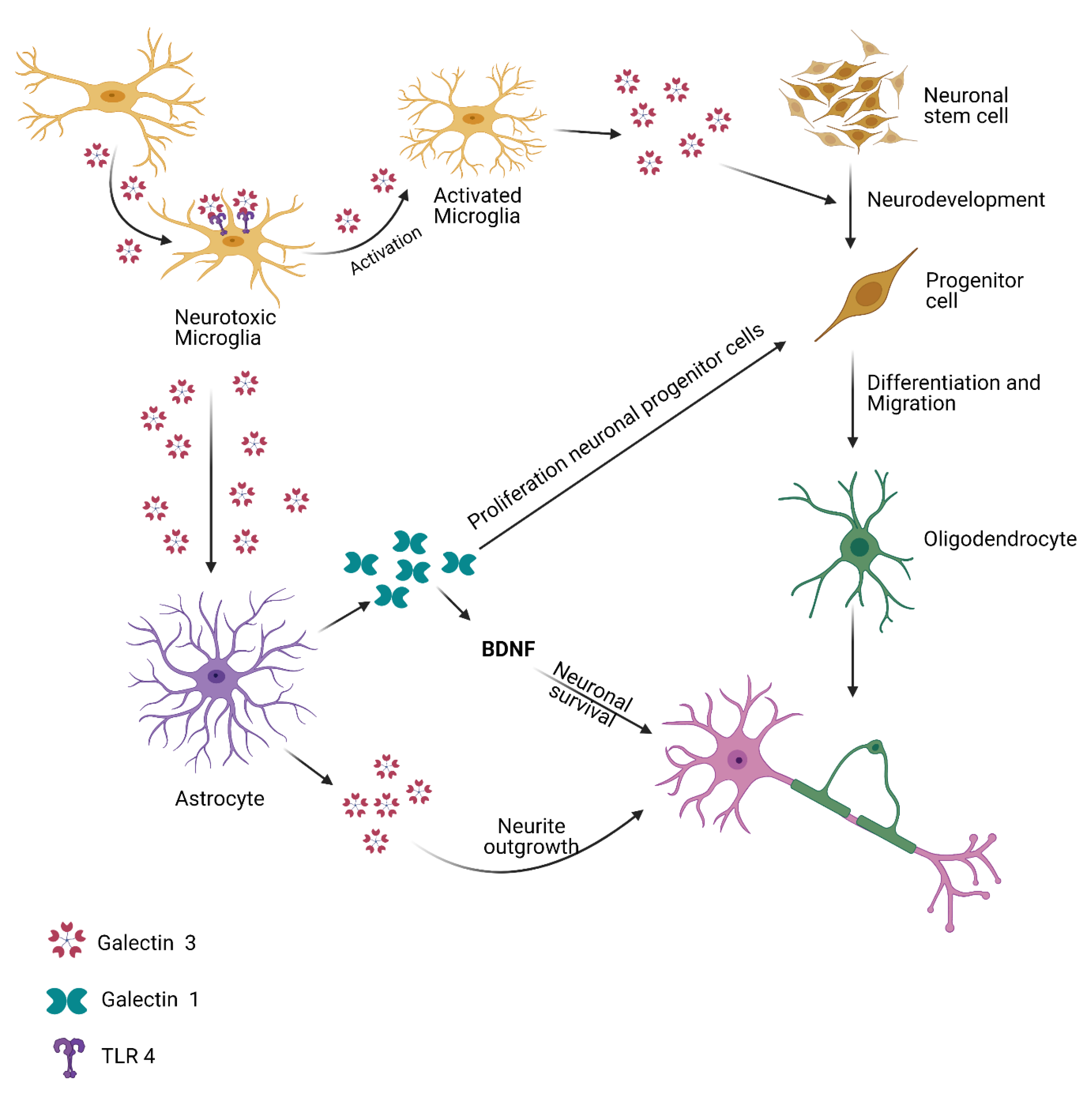
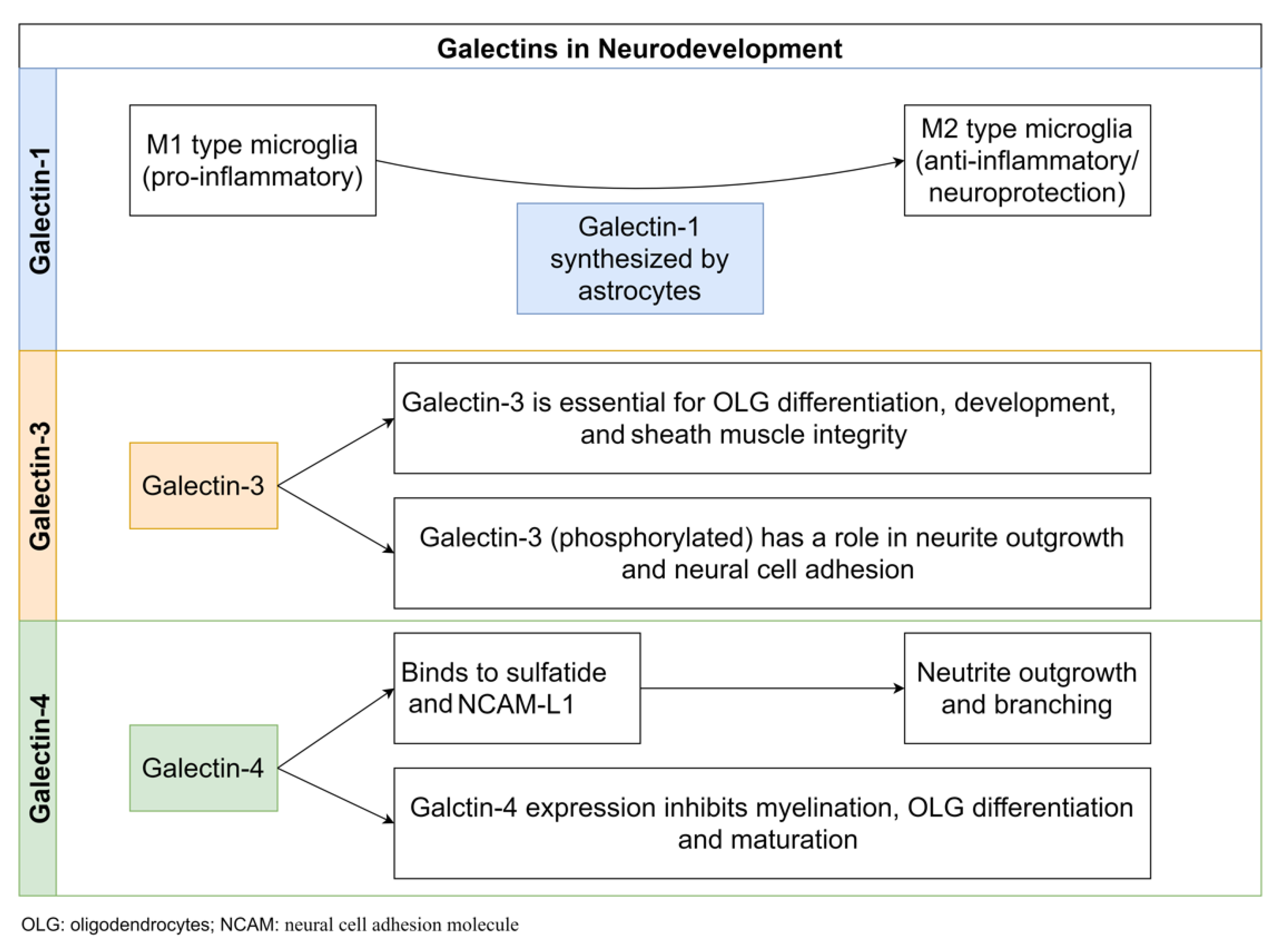
5. Gals and Neurodevelopment
5.1. Gal-1
5.2. Gal-3
5.3. Gal-4
6. Gals in Neuroinflammation
6.1. Gal-1
6.2. Gal-3
6.3. Gal-4
6.4. Gal-9
7. Role of Gals in Neuronal Re- and Degeneration
7.1. Gal-1
7.2. Gal-3
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amor, S.; Puentes, F.; Baker, D.; van der Valk, P. Inflammation in neurodegenerative diseases. Immunology 2010, 129, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Aldaz, C.M.; Hussain, T. WWOX Loss of Function in Neurodevelopmental and Neurodegenerative Disorders. Int. J. Mol. Sci. 2020, 21, 8922. [Google Scholar] [CrossRef] [PubMed]
- Li, C.H.; Chang, Y.C.; Chan, M.H.; Yang, Y.F.; Liang, S.M.; Hsiao, M. Galectins in Cancer and the Microenvironment: Functional Roles, Therapeutic Developments, and Perspectives. Biomedicines 2021, 9, 1159. [Google Scholar] [CrossRef] [PubMed]
- Md Ashraf, G.; Perveen, A.; Tabrez, S.; Jabir, N.R.; Damanhouri, G.A.; Zaidi, S.K.; Banu, N. Altered galectin glycosylation: Potential factor for the diagnostics and therapeutics of various cardiovascular and neurological disorders. Adv. Exp. Med. Biol. 2015, 822, 67–84. [Google Scholar] [CrossRef]
- Ashraf, G.M.; Perveen, A.; Tabrez, S.; Zaidi, S.K.; Kamal, M.A.; Banu, N. Studies on the role of goat heart galectin-1 as a tool for detecting post-malignant changes in glycosylation pattern. Saudi J. Biol. Sci. 2015, 22, 85–89. [Google Scholar] [CrossRef][Green Version]
- Ashraf, G.M.; Bilal, N.; Suhail, N.; Hasan, S.; Banu, N. Glycosylation of purified buffalo heart galectin-1 plays crucial role in maintaining its structural and functional integrity. Biochemistry 2010, 75, 1450–1457. [Google Scholar] [CrossRef]
- Rabinovich, G.A.; Toscano, M.A.; Jackson, S.S.; Vasta, G.R. Functions of cell surface galectin-glycoprotein lattices. Curr. Opin. Struct. Biol. 2007, 17, 513–520. [Google Scholar] [CrossRef]
- Nielsen, M.I.; Stegmayr, J.; Grant, O.C.; Yang, Z.; Nilsson, U.J.; Boos, I.; Carlsson, M.C.; Woods, R.J.; Unverzagt, C.; Leffler, H.; et al. Galectin binding to cells and glycoproteins with genetically modified glycosylation reveals galectin–glycan specificities in a natural context. J. Biol. Chem. 2018, 293, 20249–20262. [Google Scholar] [CrossRef]
- Cummings, R.D.; Liu, F.-T. Galectins. In Essentials of Glycobiology, 2nd ed.; National Library of Medicine: Rockville Pike, MD, USA, 2009; pp. 1–12. [Google Scholar]
- He, J.; Baum, L.G. Galectin Interactions with Extracellular Matrix and Effects on Cellular Function. Methods Enzymol. 2006, 417, 247–256. [Google Scholar] [CrossRef]
- Ashraf, G.M.; Baeesa, S.S. Investigation of Gal-3 Expression Pattern in Serum and Cerebrospinal Fluid of Patients Suffering from Neurodegenerative Disorders. Front. Neurosci. 2018, 12, 430. [Google Scholar] [CrossRef]
- Aalinkeel, R.; Mangum, C.S.; Abou-Jaoude, E.; Reynolds, J.L.; Liu, M.; Sundquist, K.; Parikh, N.U.; Chaves, L.D.; Mammen, M.J.; Schwartz, S.A.; et al. Galectin-1 Reduces Neuroinflammation via Modulation of Nitric Oxide-Arginase Signaling in HIV-1 Transfected Microglia: A Gold Nanoparticle-Galectin-1 “Nanoplex” a Possible Neurotherapeutic? J. Neuroimmune Pharmacol. 2016, 12, 133–151. [Google Scholar] [CrossRef]
- Aalinkeel, R.; Mahajan, S.D. Neuroprotective role of galectin-1 in central nervous system pathophysiology. Neural Regen. Res. 2016, 11, 896. [Google Scholar] [CrossRef]
- Ashraf, G.; Perveen, A.; Zaidi, S.K.; Ahmad, A.; Shakil, S.; Firoz, C.K.; Jabir, N.R.; Hassan, I.; Khan, T.A.; Yarla, N.S.; et al. Galectins-A Potential Target for Cardiovascular Therapy. Curr. Vasc. Pharmacol. 2017, 15, 296–312. [Google Scholar] [CrossRef]
- Hasan, S.S.; Ashraf, G.M.; Banu, N. Galectins—Potential targets for cancer therapy. Cancer Lett. 2007, 253, 25–33. [Google Scholar] [CrossRef]
- Radosavljevic, G.; Volarevic, V.; Jovanovic, I.; Milovanovic, M.; Pejnovic, N.; Arsenijevic, N.; Hsu, D.K.; Lukic, M.L. The roles of Galectin-3 in autoimmunity and tumor progression. Immunol. Res. 2012, 52, 100–110. [Google Scholar] [CrossRef]
- Budd Haeberlein, S.L.; Harris, T.J.R. Promising Targets for the Treatment of Neurodegenerative Diseases. Clin. Pharmacol. Ther. 2015, 98, 492–501. [Google Scholar] [CrossRef]
- Dugger, B.N.; Dickson, D.W. Pathology of Neurodegenerative Diseases. Cold Spring Harb. Perspect. Biol. 2017, 9, a028035. [Google Scholar] [CrossRef]
- Zhou, J.Y.; Afjehi-Sadat, L.; Asress, S.; Duong, D.M.; Cudkowicz, M.; Glass, J.D.; Peng, J. Galectin-3 is a candidate biomarker for amyotrophic lateral sclerosis: Discovery by a proteomics approach. J. Proteome Res. 2010, 9, 5133–5141. [Google Scholar] [CrossRef]
- Yazar, T.; Olgun Yazar, H.; Cihan, M. Evaluation of serum galectin-3 levels at Alzheimer patients by stages: A preliminary report. Acta Neurol. Belg. 2021, 121, 949–954. [Google Scholar] [CrossRef]
- Boza-Serrano, A.; Ruiz, R.; Sanchez-Varo, R.; García-Revilla, J.; Yang, Y.; Jimenez-Ferrer, I.; Paulus, A.; Wennström, M.; Vilalta, A.; Allendorf, D.; et al. Galectin-3, a novel endogenous TREM2 ligand, detrimentally regulates inflammatory response in Alzheimer’s disease. Acta Neuropathol. 2019, 138, 251–273. [Google Scholar] [CrossRef]
- .Wang, X.; Niu, Y.; Yue, C.X.; Fu, S.; Wang, R.T. Increased ileal bile acid binding protein and galectin-9 are associated with mild cognitive impairment and Alzheimer’s disease. J. Psychiatr. Res. 2019, 119, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Sciacchitano, S.; Lavra, L.; Morgante, A.; Ulivieri, A.; Magi, F.; de Francesco, G.P.; Bellotti, C.; Salehi, L.B.; Ricci, A. Galectin-3: One molecule for an alphabet of diseases, from A to Z. Int. J. Mol. Sci. 2018, 19, 379. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, G.; Rancourt, A.; Sato, S. Metazoan soluble β-Galactoside-Binding Lectins, Galectins: Methods for purification, characterization of their carbohydrate-binding specificity, and probing their ligands. Methods Mol. Biol. 2020, 2132, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, H.; Bum-Erdene, K.; Bohari, M.H.; Yu, X. Galectin-1 inhibitors and their potential therapeutic applications: A patent review. Expert Opin. Ther. Pat. 2016, 26, 537–554. [Google Scholar] [CrossRef]
- Barondes, S.H.; Cooper, D.N.W.; Gitt, M.A.; Leffler, H. Galectins. Structure and function of a large family of animal lectins. J. Biol. Chem. 1994, 269, 20807–20810. [Google Scholar] [CrossRef]
- Leonidas, D.D.; Elbert, B.L.; Zhou, Z.; Leffler, H.; Ackerman, S.J.; Acharya, K.R. Crystal structure of human Charcot-Leyden crystal protein, an eosinophil lysophospholipase, identifies it as a new member of the carbohydrate-binding family of galectins. Structure 1995, 3, 1379–1393. [Google Scholar] [CrossRef]
- Lobsanov, Y.D.; Gitt, M.A.; Leffler, H.; Barondes, S.H.; Rini, J.M. X-ray crystal structure of the human dimeric S-Lac lectin, L-14-II, in complex with lactose at 2.9-A resolution. J. Biol. Chem. 1993, 268, 27034–27038. [Google Scholar] [CrossRef]
- Yang, R.Y.; Rabinovich, G.A.; Liu, F.T. Galectins: Structure, function and therapeutic potential. Expert Rev. Mol. Med. 2008, 10, e17. [Google Scholar] [CrossRef]
- Barondes, S.; Castronovo, V.; Cooper, D.N.W.; Cummings, R.D.; Drickamer, K.; Feizi, T.; Gltt, M.A.; Hirabayashi, J.; Hughes, C.; Kasai, K.-k.; et al. Galectins: A family of animal beta-galactoside-binding lectins. Cell 1994, 76, 597–598. [Google Scholar] [CrossRef]
- Li, Y.; Chen, N.; Wu, C.; Lu, Y.; Gao, G.; Duan, C.; Yang, H.; Lu, L. Galectin-1 attenuates neurodegeneration in Parkinson’s disease model by modulating microglial MAPK/IκB/NFκB axis through its carbohydrate-recognition domain. Brain Behav. Immun. 2020, 83, 214–225. [Google Scholar] [CrossRef]
- Bonsack, F.; Sukumari-Ramesh, S. Differential cellular expression of galectin-1 and galectin-3 after intracerebral hemorrhage. Front. Cell. Neurosci. 2019, 13, 157. [Google Scholar] [CrossRef]
- He, X.W.; Li, W.L.; Li, C.; Liu, P.; Shen, Y.G.; Zhu, M.; Jin, X.P. Serum levels of galectin-1, galectin-3, and galectin-9 are associated with large artery atherosclerotic stroke. Sci. Rep. 2017, 7, 40994. [Google Scholar] [CrossRef]
- Huflejt, M.E.; Turck, C.W.; Lindstedt, R.; Barondes, S.H.; Leffler, H. L-29, a soluble lactose-binding lectin, is phosphorylated on serine 6 and serine 12 in vivo and by casein kinase I. J. Biol. Chem. 1993, 268, 26712–26718. [Google Scholar] [CrossRef]
- Sundblad, V.; Quintar, A.A.; Morosi, L.G.; Niveloni, S.I.; Cabanne, A.; Smecuol, E.; Mauriño, E.; Mariño, K.V.; Bai, J.C.; Maldonado, C.A.; et al. Galectins in Intestinal Inflammation: Galectin-1 Expression Delineates Response to Treatment in Celiac Disease Patients. Front. Immunol. 2018, 9, 379. [Google Scholar] [CrossRef]
- Tenne-Brown, J.; Puche, A.C.; Key, B. Expression of galectin-1 in the mouse olfactory system. Int. J. Dev. Biol. 1998, 42, 791–799. [Google Scholar]
- Carlos, C.P.; Silva, A.A.; Gil, C.D.; Oliani, S.M. Pharmacological treatment with galectin-1 protects against renal ischaemia-reperfusion injury. Sci. Rep. 2018, 8, 9568. [Google Scholar] [CrossRef]
- Starossom, S.C.; Mascanfroni, I.D.; Imitola, J.; Cao, L.; Raddassi, K.; Hernandez, S.F.; Bassil, R.; Croci, D.O.; Cerliani, J.P.; Delacour, D.; et al. Galectin-1 Deactivates Classically Activated Microglia and Protects from Inflammation-Induced Neurodegeneration. Immunity 2012, 37, 249–263. [Google Scholar] [CrossRef]
- Yan, J.; Xu, Y.; Zhang, L.; Zhao, H.; Jin, L.; Liu, W.G.; Weng, L.H.; Li, Z.H.; Chen, L. Increased Expressions of Plasma Galectin-3 in Patients with Amyotrophic Lateral Sclerosis. Chin. Med. J. 2016, 129, 2797. [Google Scholar] [CrossRef]
- Nangia-Makker, P.; Nakahara, S.; Hogan, V.; Raz, A. Galectin-3 in apoptosis, a novel therapeutic target. J. Bioenerg. Biomembr. 2007, 39, 79–84. [Google Scholar] [CrossRef]
- Gao, P.; Simpson, J.L.; Zhang, J.; Gibson, P.G. Galectin-3: Its role in asthma and potential as an anti-inflammatory target. Respir. Res. 2013, 14, 136. [Google Scholar] [CrossRef]
- Cunha, E.G.C.; de Lima, C.A.D.; Vilar, K.M.; Nóbrega, M.F.; Almeida, A.R.; Pereira, M.C.; Dantas, A.T.; Gonçalves, R.S.G.; Rêgo, M.J.B.M.; Duarte, A.L.B.P.; et al. Genetic variants in LGALS3 are related to lower galectin-3 serum levels and clinical outcomes in systemic sclerosis patients: A case-control study. Autoimmunity 2021, 54, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Trompet, S.; Jukema, W.; Mooijaart, S.P.; Ford, I.; Stott, D.J.; Westendorp, R.G.; de Craen, A.J. Genetic variation in galectin-3 gene associates with cognitive function at old age. Neurobiol. Aging 2012, 33, 2232.e1–2232.e9. [Google Scholar] [CrossRef] [PubMed]
- Nickel, W. The mystery of nonclassical protein secretion. Eur. J. Biochem. 2003, 270, 2109–2119. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.C. Secretion of the galectin family of mammalian carbohydrate-binding proteins. Biochim. Biophys. Acta (BBA) Gen. Subj. 1999, 1473, 172–185. [Google Scholar] [CrossRef]
- le Mercier, M.; Fortin, S.; Mathieu, V.; Kiss, R.; Lefranc, F. Galectins and gliomas. Brain Pathol. 2010, 20, 17–27. [Google Scholar] [CrossRef]
- Rabinovich, G.A.; Baum, L.G.; Tinari, N.; Paganelli, R.; Natoli, C.; Liu, F.T.; Iacobelli, S. Galectins and their ligands: Amplifiers, silencers or tuners of the inflammatory response? Trends Immunol. 2002, 23, 313–320. [Google Scholar] [CrossRef]
- Liu, F.T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef]
- Hernandez, J.D.; Baum, L.G. Ah, sweet mystery of death! Galectins and control of cell fate. Glycobiology 2002, 12, 127R–136R. [Google Scholar] [CrossRef]
- Elola, M.T.; Wolfenstein-Todel, C.; Troncoso, M.F.; Vasta, G.R.; Rabinovich, G.A. Galectins: Matricellular glycan-binding proteins linking cell adhesion, migration, and survival. Cell. Mol. Life Sci. 2007, 64, 1679–1700. [Google Scholar] [CrossRef]
- Garner, O.B.; Baum, L.G. Galectin-glycan lattices regulate cell-surface glycoprotein organization and signalling. Biochem. Soc. Trans. 2008, 36, 1472–1477. [Google Scholar] [CrossRef]
- Nishihara, H.; Shimizu, F.; Kitagawa, T.; Yamanaka, N.; Akada, J.; Kuramitsu, Y.; Sano, Y.; Takeshita, Y.; Maeda, T.; Abe, M.; et al. Identification of galectin-3 as a possible antibody target for secondary progressive multiple sclerosis. Mult. Scler. 2017, 23, 382–394. [Google Scholar] [CrossRef]
- Pardo, E.; Cárcamo, C.; Martín, R.U.S.; Ciampi, E.; Segovia-Miranda, F.; Curkovic-Peña, C.; Montecino, F.; Holmes, C.; Tichauer, J.E.; Acuña, E.; et al. Galectin-8 as an immunosuppressor in experimental autoimmune encephalomyelitis and a target of human early prognostic antibodies in multiple sclerosis. PLoS ONE 2017, 12, e0177472. [Google Scholar] [CrossRef]
- Massardo, L.; Metz, C.; Pardo, E.; Mezzano, V.; Babul, M.; Jarpa, E.; Guzmán, A.M.; André, S.; Kaltner, H.; Gabius, H.J.; et al. Autoantibodies against galectin-8: Their specificity, association with lymphopenia in systemic lupus erythematosus and detection in rheumatoid arthritis and acute inflammation. Lupus 2009, 18, 539–546. [Google Scholar] [CrossRef]
- Vicuña, L.; Pardo, E.; Curkovic, C.; Döger, R.; Oyanadel, C.; Metz, C.; Massardo, L.; González, A.; Soza, A. Galectin-8 binds to LFA-1, blocks its interaction with ICAM-1 and is counteracted by anti-Gal-8 autoantibodies isolated from lupus patients. Biol. Res. 2013, 46, 275–280. [Google Scholar] [CrossRef]
- Norambuena, A.; Metz, C.; Vicuña, L.; Silva, A.; Pardo, E.; Oyanadel, C.; Massardo, L.; González, A.; Soza, A. Galectin-8 induces apoptosis in Jurkat T cells by phosphatidic acid-mediated ERK1/2 activation supported byprotein kinase A down-regulation. J. Biol. Chem. 2009, 284, 12670–12679. [Google Scholar] [CrossRef]
- Rabinovich, G.A.; Croci, D.O. Regulatory Circuits Mediated by Lectin-Glycan Interactions in Autoimmunity and Cancer. Immunity 2012, 36, 322–335. [Google Scholar] [CrossRef]
- Partridge, E.A.; le Roy, C.; di Guglielmo, G.M.; Pawling, J.; Cheung, P.; Granovsky, M.; Nabi, I.R.; Wrana, J.L.; Dennis, J.W. Regulation of cytokine receptors by golgi N-glycan processing and endocytosis. Science 2004, 306, 120–124. [Google Scholar] [CrossRef]
- Penninger, J.M.; Irie-Sasaki, J.; Sasaki, T.; Oliveira-dos-Santos, A.J. CD45: New jobs for an old acquaintance. Nat. Immunol. 2001, 2, 389–396. [Google Scholar] [CrossRef]
- Nonaka, M.; Fukuda, M. Galectin-1 for Neuroprotection? Immunity 2012, 37, 187–189. [Google Scholar] [CrossRef]
- Trapp, B.D.; Nave, K.A. Multiple sclerosis: An immune or neurodegenerative disorder? Annu. Rev. Neurosci. 2008, 31, 247–269. [Google Scholar] [CrossRef]
- Bø, L.; Vedeler, C.A.; Nyland, H.; Trapp, B.D.; Mørk, S.J. Intracortical multiple sclerosis lesions are not associated with increased lymphocyte infiltration. Mult. Scler. 2003, 9, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Stowell, S.R.; Qian, Y.; Karmakar, S.; Koyama, N.S.; Dias-Baruffi, M.; Leffler, H.; McEver, R.P.; Cummings, R.D. Differential roles of galectin-1 and galectin-3 in regulating leukocyte viability and cytokine secretion. J. Immunol. 2008, 180, 3091–3102. [Google Scholar] [CrossRef] [PubMed]
- Walther, M.; Kuklinski, S.; Pesheva, P.; Guntinas-Lichius, O.; Angelov, D.N.; Neiss, W.F.; Asou, H.; Probstmeier, R. Galectin-3 is upregulated in microglial cells in response to ischemic brain lesions, but not to facial nerve axotomy. J. Neurosci. Res. 2000, 61, 430–435. [Google Scholar] [CrossRef]
- Yoo, H.; Kim, E.G.; Lee, E.J.; Hong, S.Y.; Yoon, C.S.; Hong, M.J.; Park, S.J.; Woo, R.S.; Baik, T.K.; Song, D.Y. Neuroanatomical distribution of galectin-3 in the adult rat brain. J. Mol. Histol. 2017, 48, 133–146. [Google Scholar] [CrossRef]
- Comte, I.; Kim, Y.; Young, C.C.; van der Harg, J.M.; Hockberger, P.; Bolam, P.J.; Poirier, F.; Szele, F.G. Galectin-3 maintains cell motility from the subventricular zone to the olfactory bulb. J. Cell Sci. 2011, 124, 2438–2447. [Google Scholar] [CrossRef]
- Liu, W.; Hsu, D.K.; Chen, H.Y.; Yang, R.Y.; Carraway, K.L.; Isseroff, R.R.; Liu, F.T. Galectin-3 regulates intracellular trafficking of EGFR through alix and promotes keratinocyte migration. J. Investig. Dermatol. 2012, 132, 2828–2837. [Google Scholar] [CrossRef]
- de Jong, C.G.H.M.; Gabius, H.J.; Baron, W. The emerging role of galectins in (re)myelination and its potential for developing new approaches to treat multiple sclerosis. Cell. Mol. Life Sci. 2020, 77, 1289–1317. [Google Scholar] [CrossRef]
- Thomas, L.; Pasquini, L.A. Galectin-3-mediated glial crosstalk drives oligodendrocyte differentiation and (Re)myelination. Front. Cell. Neurosci. 2018, 12, 297. [Google Scholar] [CrossRef]
- Fatemi, A.; Wilson, M.A.; Johnston, M.V. Hypoxic-Ischemic Encephalopathy in the Term Infant. Clin. Perinatol. 2009, 36, 835–858. [Google Scholar] [CrossRef]
- Lee, A.C.C.; Kozuki, N.; Blencowe, H.; Vos, T.; Bahalim, A.; Darmstadt, G.L.; Niermeyer, S.; Ellis, M.; Robertson, N.J.; Cousens, S.; et al. Intrapartum-related neonatal encephalopathy incidence and impairment at regional and global levels for 2010 with trends from 1990. Pediatr. Res. 2013, 74 (Suppl. S1), 50–72. [Google Scholar] [CrossRef]
- Torres-Cuevas, I.; Corral-Debrinski, M.; Gressens, P. Brain oxidative damage in murine models of neonatal hypoxia/ischemia and reoxygenation. Free Radic. Biol. Med. 2019, 142, 3–15. [Google Scholar] [CrossRef]
- Schiering, I.A.M.; de Haan, T.R.; Niermeijer, J.M.F.; Koelman, J.H.; Majoie, C.B.L.M.; Reneman, L.; Aronica, E. Correlation Between Clinical and Histologic Findings in the Human Neonatal Hippocampus After Perinatal Asphyxia. J. Neuropathol. Exp. Neurol. 2014, 73, 324–334. [Google Scholar] [CrossRef]
- Vlassaks, E.; Brudek, T.; Pakkenberg, B.; Gavilanes, A.W.D. Cerebellar cytokine expression in a rat model for fetal asphyctic preconditioning and perinatal asphyxia. Cerebellum 2014, 13, 471–478. [Google Scholar] [CrossRef]
- Lalancette-Hébert, M.; Swarup, V.; Beaulieu, J.M.; Bohacek, I.; Abdelhamid, E.; Weng, Y.C.; Sato, S.; Kriz, J. Galectin-3 Is Required for Resident Microglia Activation and Proliferation in Response to Ischemic Injury. J. Neurosci. 2012, 32, 10383–10395. [Google Scholar] [CrossRef]
- Rahimian, R.; Béland, L.C.; Kriz, J. Galectin-3: Mediator of microglia responses in injured brain. Drug Discov. Today 2018, 23, 375–381. [Google Scholar] [CrossRef]
- Doverhag, C.; Hedtjärn, M.; Poirier, F.; Mallard, C.; Hagberg, H.; Karlsson, A.; Sävman, K. Galectin-3 contributes to neonatal hypoxic-ischemic brain injury. Neurobiol. Dis. 2010, 38, 36–46. [Google Scholar] [CrossRef]
- Doverhag, C.; Keller, M.; Karlsson, A.; Hedtjarn, M.; Nilsson, U.; Kapeller, E.; Sarkozy, G.; Klimaschewski, L.; Humpel, C.; Hagberg, H.; et al. Pharmacological and genetic inhibition of NADPH oxidase does not reduce brain damage in different models of perinatal brain injury in newborn mice. Neurobiol. Dis. 2008, 31, 133–144. [Google Scholar] [CrossRef]
- Pesheva, P.; Kuklinski, S.; Biersack, H.J.; Probstmeier, R. Nerve growth factor-mediated expression of galectin-3 in mouse dorsal root ganglion neurons. Neurosci. Lett. 2000, 293, 37–40. [Google Scholar] [CrossRef]
- Pesheva, P.; Kuklinski, S.; Schmitz, B.; Probstmeier, R. Galectin-3 promotes neural cell adhesion and neurite growth. J. Neurosci. Res. 1998, 54, 639–654. [Google Scholar] [CrossRef]
- Kuklinski, S.; Vladimirova, V.; Waha, A.; Kamata, H.; Pesheva, P.; Probstmeier, R. Expression of galectin-3 in neuronally differentiating PC12 cells is regulated both via Ras/MAPK-dependent and -independent signalling pathways. J. Neurochem. 2003, 87, 1112–1124. [Google Scholar] [CrossRef]
- Al-Dalahmah, O.; Campos Soares, L.; Nicholson, J.; Draijer, S.; Mundim, M.; Lu, V.M.; Sun, B.; Tyler, T.; Adorján, I.; O’Neill, E.; et al. Galectin-3 modulates postnatal subventricular zone gliogenesis. GLIA 2020, 68, 435–450. [Google Scholar] [CrossRef]
- Velasco, S.; Díez-Revuelta, N.; Hernández-Iglesias, T.; Kaltner, H.; André, S.; Gabius, H.J.; Abad-Rodríguez, J. Neuronal Galectin-4 is required for axon growth and for the organization of axonal membrane L1 delivery and clustering. J. Neurochem. 2013, 125, 49–62. [Google Scholar] [CrossRef]
- Cheng, L.; Lemmon, V. Pathological missense mutations of neural cell adhesion molecule L1 affect neurite outgrowth and branching on an L1 substrate. Mol. Cell. Neurosci. 2004, 27, 522–530. [Google Scholar] [CrossRef]
- Schmid, R.S.; Maness, P.F. L1 and NCAM adhesion molecules as signaling coreceptors in neuronal migration and process outgrowth. Curr. Opin. Neurobiol. 2008, 18, 245–250. [Google Scholar] [CrossRef]
- Stajic, D.; Selakovic, D.; Jovicic, N.; Joksimovic, J.; Arsenijevic, N.; Lukic, M.L.; Rosic, G. The role of galectin-3 in modulation of anxiety state level in mice. Brain Behav. Immun. 2019, 78, 177–187. [Google Scholar] [CrossRef]
- Winzeler, A.M.; Mandemakers, W.J.; Sun, M.Z.; Stafford, M.; Phillips, C.T.; Barres, B.A. The lipid sulfatide is a novel myelin-associated inhibitor of CNS axon outgrowth. J. Neurosci. 2011, 31, 6481–6492. [Google Scholar] [CrossRef]
- Kölliker-Frers, R.; Udovin, L.; Otero-Losada, M.; Kobiec, T.; Herrera, M.I.; Palacios, J.; Razzitte, G.; Capani, F. Neuroinflammation: An Integrating Overview of Reactive-Neuroimmune Cell Interactions in Health and Disease. Mediat. Inflamm. 2021, 2021, 9999146. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, S.; Kuroiwa, T.; Sakaguchi, M.; Sun, L.; Kadoya, T.; Okano, H.; Mizusawa, H. Galectin-1 regulates neurogenesis in the subventricular zone and promotes functional recovery after stroke. Exp. Neurol. 2007, 207, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.; Chalasani, N.; Lawitz, E.; Marri, S.; Noureddin, M.; Sanyal, A.; Schiano, T.; Siddiqui, M.; Neuschwander-Tetri, B.; Traber, P. Early phase 1 clinical trial results of GR-MD-02, a galectin-3 inhibitor, in patients having non-alcoholic steatohepatitis (NASH) with advanced fibrosis. Hepatology 2014, 60, 225A–226A. [Google Scholar]
- Shin, T. The pleiotropic effects of galectin-3 in neuroinflammation: A review. Acta Histochem. 2013, 115, 407–411. [Google Scholar] [CrossRef]
- Steinman, L.; Zamvil, S.S. How to successfully apply animal studies in experimental allergic encephalomyelitis to research on multiple sclerosis. Ann. Neurol. 2006, 60, 12–21. [Google Scholar] [CrossRef]
- Jiang, H.-R.; Rasebi, Z.; Mensah-Brown, E.; Shahin, A.; Xu, D.; Goodyear, C.S.; Fukada, S.Y.; Liu, F.-T.; Liew, F.Y.; Lukic, M.L. Galectin-3 Deficiency Reduces the Severity of Experimental Autoimmune Encephalomyelitis. J. Immunol. 2009, 182, 1167–1173. [Google Scholar] [CrossRef]
- Wolf, S.A.; Boddeke, H.W.G.M.; Kettenmann, H. Microglia in Physiology and Disease. Annu. Rev. Physiol. 2017, 79, 619–643. [Google Scholar] [CrossRef]
- Reichert, F.; Rotshenker, S. Galectin-3/MAC-2 in experimental allergic encephalomyelitis. Exp. Neurol. 1999, 160, 508–514. [Google Scholar] [CrossRef]
- Rotshenker, S. Wallerian degeneration: The innate-immune response to traumatic nerve injury. J. Neuroinflammation 2011, 8, 109. [Google Scholar] [CrossRef]
- Rotshenker, S.; Reichert, F.; Gitik, M.; Haklai, R.; Elad-Sfadia, G.; Kloog, Y. Galectin-3/MAC-2, ras and PI3K activate complement receptor-3 and scavenger receptor-AI/II mediated myelin phagocytosis in microglia. GLIA 2008, 56, 1607–1613. [Google Scholar] [CrossRef]
- Rotshenker, S. The Role of Galectin-3/MAC-2 in the Activation of the Innate-Immune Function of Phagocytosis in Microglia in Injury and Disease. J. Mol. Neurosci. 2009, 39, 99–103. [Google Scholar] [CrossRef]
- Mietto, B.S.; Jurgensen, S.; Alves, L.; Pecli, C.; Narciso, M.S.; Assunção-Miranda, I.; Villa-Verde, D.M.S.; de Souza Lima, F.R.; de Menezes, J.R.L.; Benjamim, C.F.; et al. Lack of galectin-3 speeds Wallerian degeneration by altering TLR and pro-inflammatory cytokine expressions in injured sciatic nerve. Eur. J. Neurosci. 2013, 37, 1682–1690. [Google Scholar] [CrossRef]
- Chaudhary, P.; Marracci, G.; Galipeau, D.; Pocius, E.; Morris, B.; Bourdette, D. Lipoic Acid Reduces Inflammation in a Mouse Focal Cortical Experimental Autoimmune Encephalomyelitis Model. J. Neuroimmunol. 2015, 289, 68. [Google Scholar] [CrossRef][Green Version]
- Mensah-Brown, E.P.K.; al Rabesi, Z.; Shahin, A.; al Shamsi, M.; Arsenijevic, N.; Hsu, D.K.; Liu, F.T.; Lukic, M.L. Targeted disruption of the galectin-3 gene results in decreased susceptibility to multiple low dose streptozotocin-induced diabetes in mice. Clin. Immunol. 2009, 130, 83–88. [Google Scholar] [CrossRef]
- Petrovic, I.; Pejnovic, N.; Ljujic, B.; Pavlovic, S.; Miletic Kovacevic, M.; Jeftic, I.; Djukic, A.; Draginic, N.; Andjic, M.; Arsenijevic, N.; et al. Overexpression of Galectin 3 in Pancreatic β Cells Amplifies β-Cell Apoptosis and Islet Inflammation in Type-2 Diabetes in Mice. Front. Endocrinol. 2020, 11, 30. [Google Scholar] [CrossRef]
- de Giusti, C.J.; Alberdi, L.; Frik, J.; Ferrer, M.F.; Scharrig, E.; Schattner, M.; Gomez, R.M. Galectin-3 is upregulated in activated glia during Junin virus-induced murine encephalitis. Neurosci. Lett. 2011, 501, 163–166. [Google Scholar] [CrossRef]
- James, R.E.; Hillis, J.; Adorján, I.; Gration, B.; Mundim, M.V.; Iqbal, A.J.; Majumdar, M.M.; Yates, R.L.; Richards, M.M.H.; Goings, G.E.; et al. Loss of galectin-3 decreases the number of immune cells in the subventricular zone and restores proliferation in a viral model of multiple sclerosis. GLIA 2016, 64, 105–121. [Google Scholar] [CrossRef]
- Stechly, L.; Morelle, W.; Dessein, A.F.; André, S.; Grard, G.; Trinel, D.; Dejonghe, M.J.; Leteurtre, E.; Drobecq, H.; Trugnan, G.; et al. Galectin-4-regulated delivery of glycoproteins to the brush border membrane of enterocyte-like cells. Traffic 2009, 10, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Delacour, D.; Gouyer, V.; Zanetta, J.P.; Drobecq, H.; Leteurtre, E.; Grard, G.; Moreau-Hannedouche, O.; Maes, E.; Pons, A.; André, S.; et al. Galectin-4 and sulfatides in apical membrane trafficking in enterocyte-like cells. J. Cell Biol. 2005, 169, 491–501. [Google Scholar] [CrossRef]
- Hokama, A.; Mizoguchi, E.; Sugimoto, K.; Shimomura, Y.; Tanaka, Y.; Yoshida, M.; Rietdijk, S.T.; de Jong, Y.P.; Snapper, S.B.; Terhorst, C.; et al. Induced reactivity of intestinal CD4+ T cells with an epithelial cell lectin, galectin-4, contributes to exacerbation of intestinal inflammation. Immunity 2004, 20, 681–693. [Google Scholar] [CrossRef]
- Steelman, A.J.; Smith, R.; Welsh, C.J.; Li, J. Galectin-9 protein is up-regulated in astrocytes by tumor necrosis factor and promotes encephalitogenic T-cell apoptosis. J. Biol. Chem. 2013, 288, 23776–23787. [Google Scholar] [CrossRef]
- Imaizumi, T.; Kumagai, M.; Sasaki, N.; Kurotaki, H.; Mori, F.; Seki, M.; Nishi, N.; Fujimoto, K.; Tanji, K.; Shibata, T.; et al. Interferon-gamma stimulates the expression of galectin-9 in cultured human endothelial cells. J. Leukoc. Biol. 2002, 72, 486–491. [Google Scholar]
- Heusschen, R.; Griffioen, A.W.; Thijssen, V.L. Galectin-9 in tumor biology: A jack of multiple trades. Biochim. Biophys. Acta (BBA) Rev. Cancer 2013, 1836, 177–185. [Google Scholar] [CrossRef]
- Chou, F.C.; Chen, H.Y.; Kuo, C.C.; Sytwu, H.K. Role of galectins in tumors and in clinical immunotherapy. In Int. J. Mol. Sci. 2018, 19, 430. [Google Scholar] [CrossRef] [PubMed]
- Klyosov, A.A.; Traber, P.G. Galectins in Disease and Potential Therapeutic Approaches; ACS Publications: Washington, DC, USA, 2012. [Google Scholar]
- Rinaldi, M.; Thomas, L.; Mathieu, P.; Carabias, P.; Troncoso, M.F.; Pasquini, J.M.; Rabinovich, G.A.; Pasquini, L.A. Galectin-1 circumvents lysolecithin-induced demyelination through the modulation of microglial polarization/phagocytosis and oligodendroglial differentiation. Neurobiol. Dis. 2016, 96, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Camby, I.; le Mercier, M.; Lefranc, F.; Kiss, R. Galectin-1: A small protein with major functions. Glycobiology 2006, 16, 137R–157R. [Google Scholar] [CrossRef] [PubMed]
- Phyo, P.; Zhao, X.; Templeton, A.C.; Xu, W.; Cheung, J.K.; Su, Y. Understanding molecular mechanisms of biologics drug delivery and stability from NMR spectroscopy. Adv. Drug Deliv. Rev. 2021, 174, 1–29. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnology 2018, 16, 71. [Google Scholar] [CrossRef]
- Stowell, S.R.; Cho, M.; Feasley, C.L.; Arthur, C.M.; Song, X.; Colucci, J.K.; Karmakar, S.; Mehta, P.; Dias-Baruffi, M.; McEver, R.P.; et al. Ligand reduces galectin-1 sensitivity to oxidative inactivation by enhancing dimer formation. J. Biol. Chem. 2009, 284, 4989–4999. [Google Scholar] [CrossRef]
- McGraw, J.; Gaudet, A.D.; Oschipok, L.W.; Kadoya, T.; Horie, H.; Steeves, J.D.; Tetzlaff, W.; Ramer, M.S. Regulation of neuronal and glial galectin-1 expression by peripheral and central axotomy of rat primary afferent neurons. Exp. Neurol. 2005, 195, 103–114. [Google Scholar] [CrossRef]

| Galectin | Individual Functions |
|---|---|
| Galectin-1 |
|
| Galectin-2 |
|
| Galectin-3 |
|
| Galectin-4 |
|
| Galectin-7 |
|
| Galectin-8 |
|
| Galectin-9 |
|
| Galectin-10 |
|
| Galectin-12 |
|
| Pathogenesis/ Pathophysiology | Galectin Expressed | Therapeutic Potential | |
|---|---|---|---|
| Cancer |
| Gal-1 Gal-3 |
|
| Cardio-vascular diseases (CVD) |
| Gal-1 Gal-3 Gal-9 |
|
| Fatty Liver Disease and Fibrosis |
| Gal-3 |
|
| Multiple Sclerosis (NDD) |
| Gal-1 Gal-3 Gal-4 Gal-9 |
|
| Amyotrophic Lateral Sclerosis (NDD) |
| Gal-1 Gal-3 |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaudhary, S.; Chaudhary, S.; Rawat, S.; Kulkarni, A.; Bilgrami, A.L.; Perveen, A.; Alghamdi, B.S.; Zughaibi, T.A.; Ashraf, G.M.; Alam, M.Z.; et al. Galectins—Potential Therapeutic Targets for Neurodegenerative Disorders. Int. J. Mol. Sci. 2022, 23, 11012. https://doi.org/10.3390/ijms231911012
Chaudhary S, Chaudhary S, Rawat S, Kulkarni A, Bilgrami AL, Perveen A, Alghamdi BS, Zughaibi TA, Ashraf GM, Alam MZ, et al. Galectins—Potential Therapeutic Targets for Neurodegenerative Disorders. International Journal of Molecular Sciences. 2022; 23(19):11012. https://doi.org/10.3390/ijms231911012
Chicago/Turabian StyleChaudhary, Sapana, Sameer Chaudhary, Sakshi Rawat, Archana Kulkarni, Anwar L. Bilgrami, Asma Perveen, Badrah S. Alghamdi, Torki Al Zughaibi, Ghulam Md Ashraf, Mohammad Zubair Alam, and et al. 2022. "Galectins—Potential Therapeutic Targets for Neurodegenerative Disorders" International Journal of Molecular Sciences 23, no. 19: 11012. https://doi.org/10.3390/ijms231911012
APA StyleChaudhary, S., Chaudhary, S., Rawat, S., Kulkarni, A., Bilgrami, A. L., Perveen, A., Alghamdi, B. S., Zughaibi, T. A., Ashraf, G. M., Alam, M. Z., & Hussain, T. (2022). Galectins—Potential Therapeutic Targets for Neurodegenerative Disorders. International Journal of Molecular Sciences, 23(19), 11012. https://doi.org/10.3390/ijms231911012









