Nephritis-Associated Plasmin Receptor (NAPlr): An Essential Inducer of C3-Dominant Glomerular Injury and a Potential Key Diagnostic Biomarker of Infection-Related Glomerulonephritis (IRGN)
Abstract
1. Introduction
2. Isolation and Characterization of NAPlr
3. Clinical and Histological Characteristics of PSAGN in Relation to Complements, ICs, and NAPlr
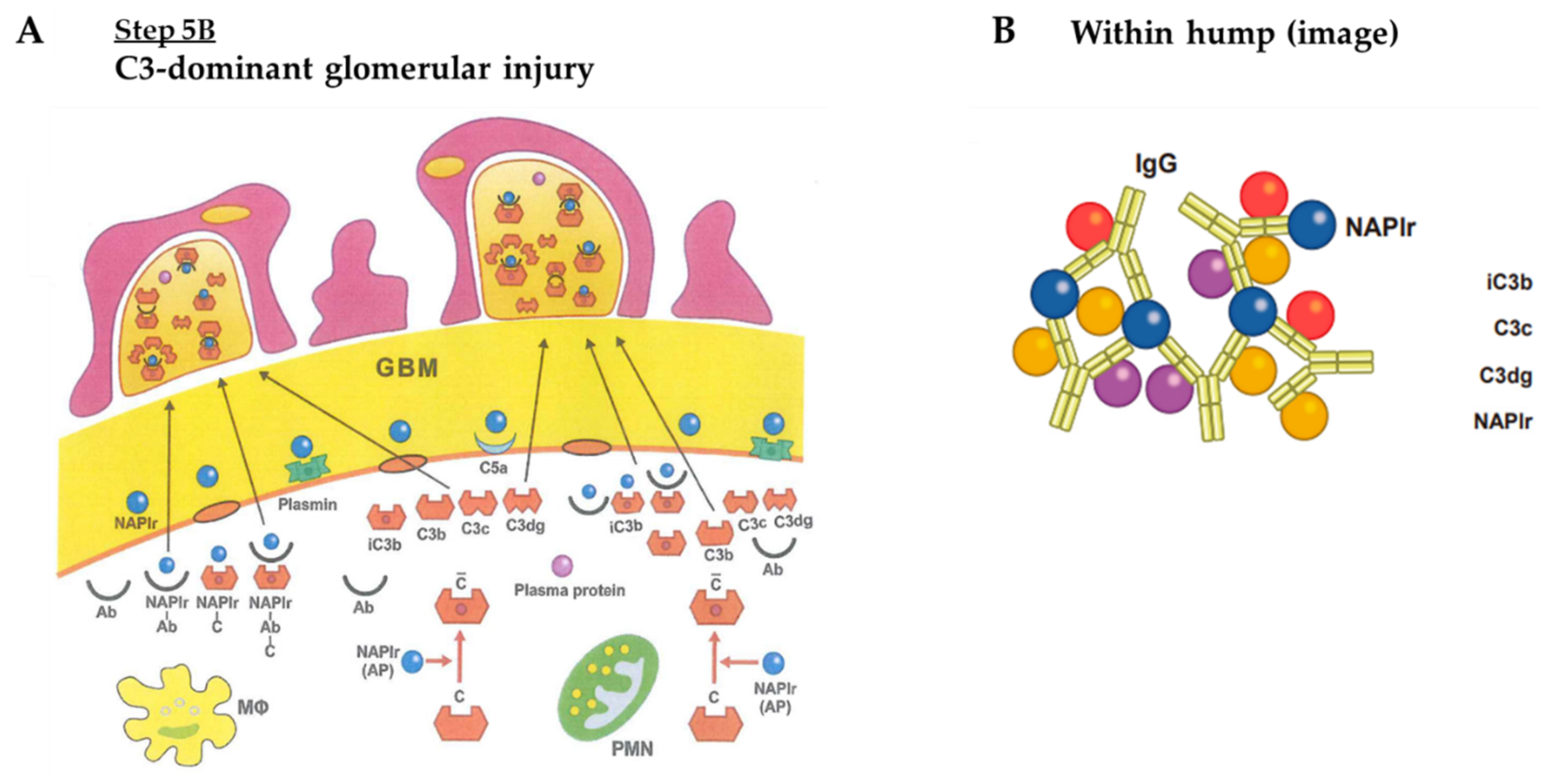
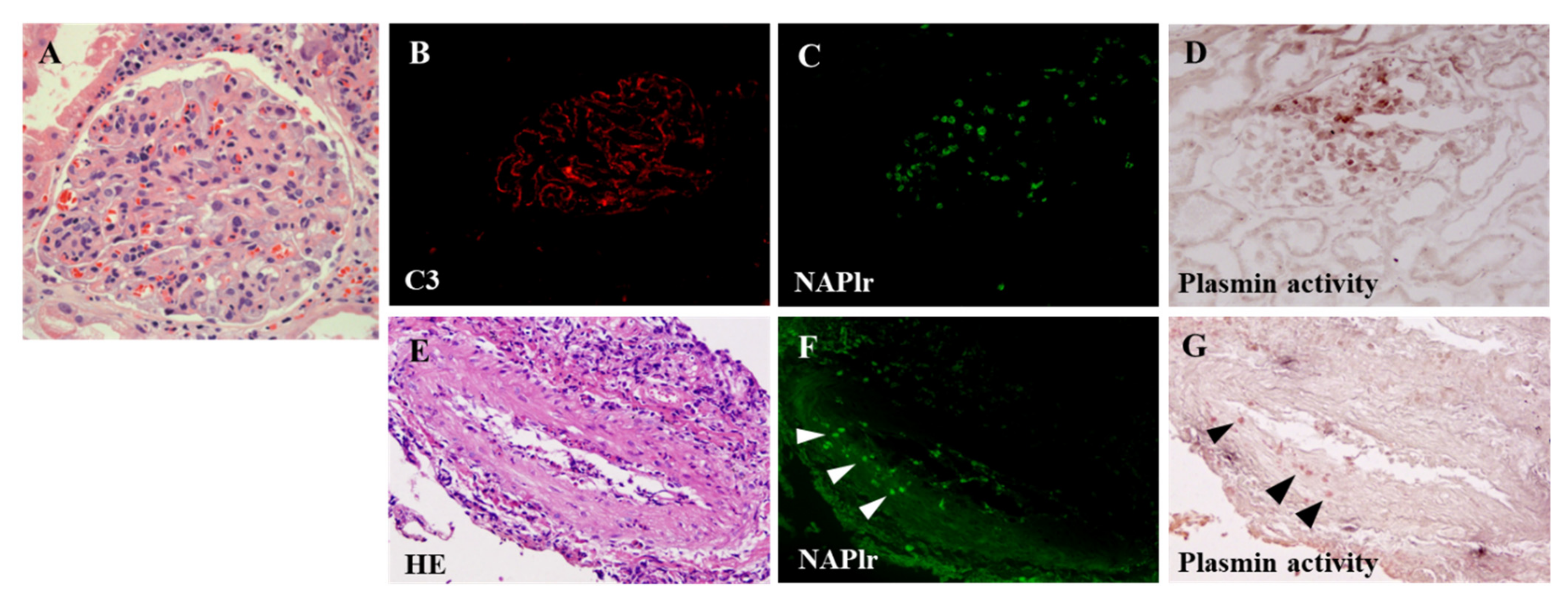
4. Proposed Mechanism for PSAGN
- (1)
- Complement profile in PSAGN
- (2)
- Complement activation by NAPlr
- (3)
- C3-dominant glomerular injury
| IC-Dominant GI | C3-Dominant GI | ||
|---|---|---|---|
| Major difference | C3 with IgG | C3 without IgG | |
| NAPlr deposition | +~++ | +~++ | |
| Plasmin activation | +~++ | +~++ | |
| Complement activation | |||
| Alternative pathway | + | ++ | |
| Classical pathway | + | + | |
| Serum complement | |||
| CH50 | ↓ | ↓ | |
| C3 | ↓ | ↓ | |
| C4 | → | → | |
| Underlying complement dysregulation | unknown | unknown | |
| C3 Nephritic factor | + ? | + ? | [68] |
| Anti-factor B antibody | ++ ? | ++ ? | [67] |
| Pathology | |||
| LM | |||
| Endocapillary GN | ++ | ++ | |
| Exudative change | ++ | ++ | |
| IF | |||
| C3 | ++~+++ | ++~+++ | |
| IgG | +~++ | − | |
| C4 | +~− | − | |
| EM | |||
| Hump | ++ | ++ | |
| Circulating IC | ++ | ++ | [63] |
| Circulating NAPlr antibody | ++ | ++ | [2] |
5. NAPlr as a Key Diagnostic Biomarker of IRGN
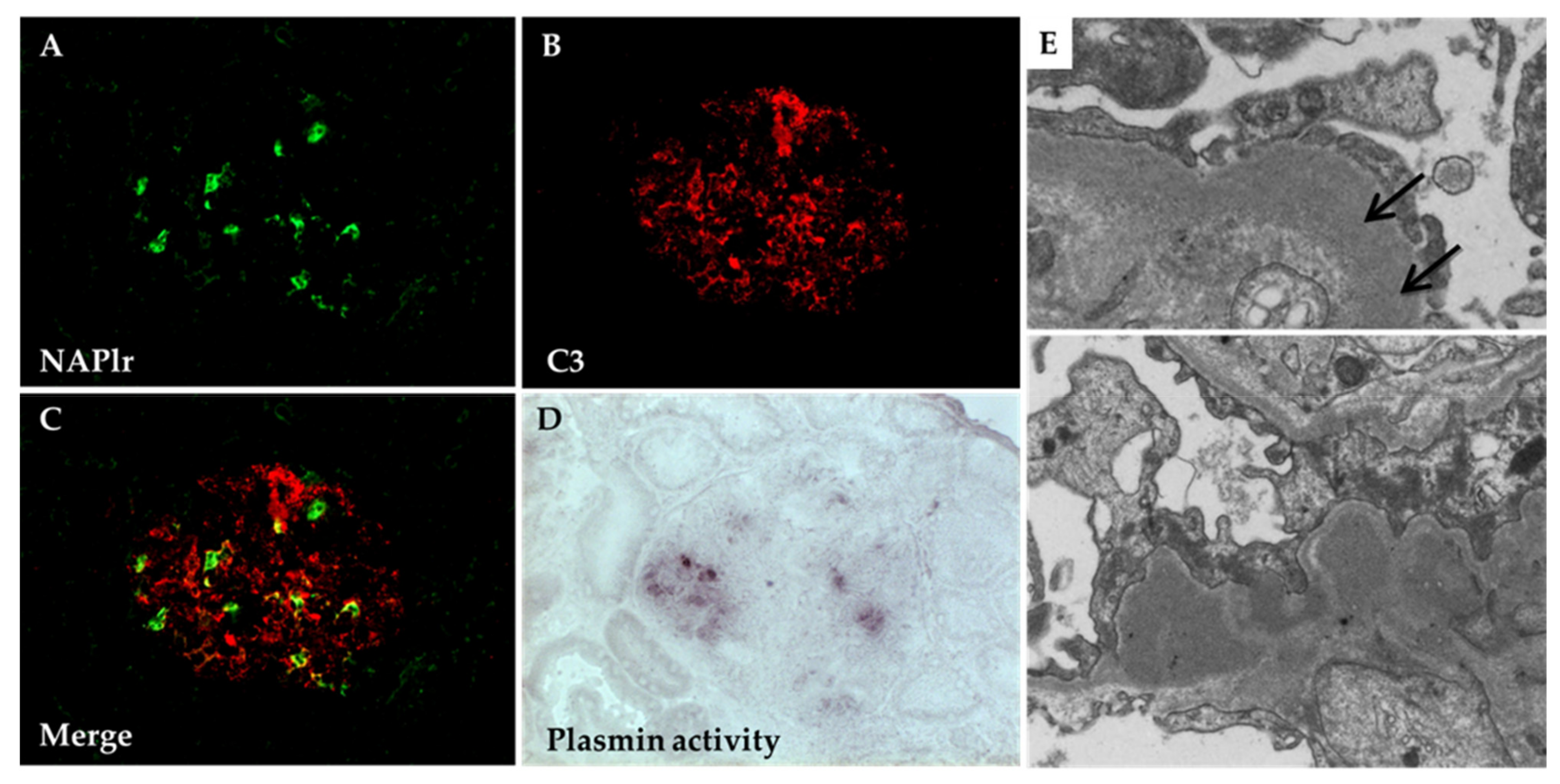
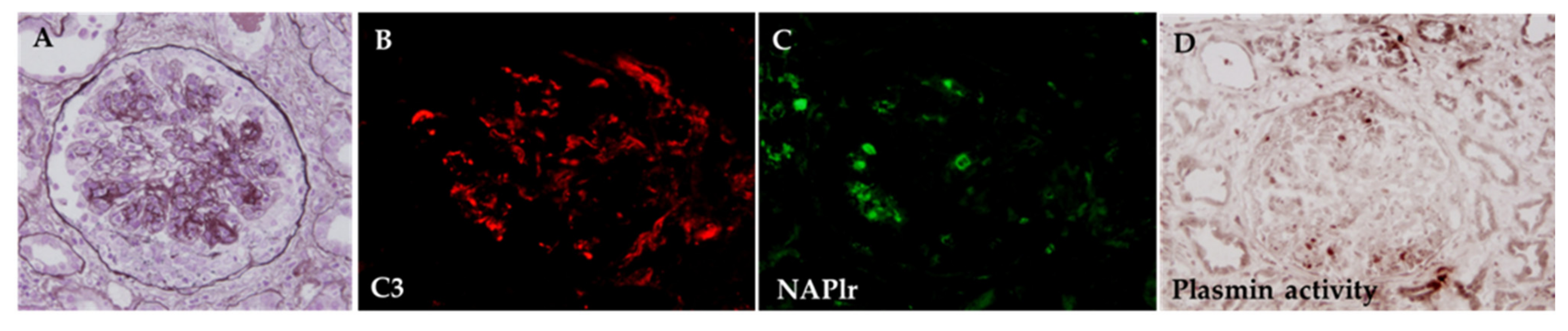
5.1. Glomerular NAPlr Deposition and Related Plasmin Activity in Streptococcal Infection-Related Nephritis
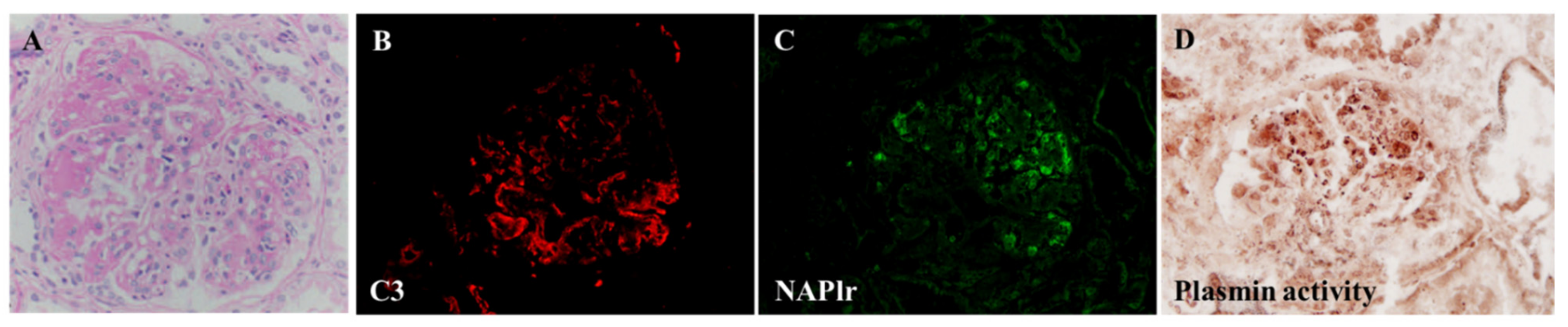
5.2. Positive Staining for NAPlr/Plasmin Activity in Non-Streptococcal IRGN
5.3. Plasmin-Binding Capacity of GAPDH in Non-Streptococcal Bacteria
5.4. NAPlr as a General Diagnostic Biomarker of IRGN
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Yamakami, K.; Yoshizawa, N.; Wakabayashi, K.; Takeuchi, A.; Tadakuma, T.; Boyle, M.D. The potential role for nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. Methods 2000, 21, 185–197. [Google Scholar] [CrossRef]
- Yoshizawa, N.; Yamakami, K.; Fujino, M.; Oda, T.; Tamura, K.; Matsumoto, K.; Sugisaki, T.; Boyle, M.D. Nephritis-associated plasmin receptor and acute poststreptococcal glomerulonephritis: Characterization of the antigen and associated immune response. J. Am. Soc. Nephrol. 2004, 15, 1785–1793. [Google Scholar] [CrossRef] [PubMed]
- Couser, W.G.; Johnson, R.J. Post-infecitous glomerulonephritis. In Immunologic Renal Diseases; Neilson, E.G., Couser, W.G., Eds.; Lippincott-Raven: Philadelphia, PA, USA, 1997; pp. 915–943. [Google Scholar]
- Nadasdy, T.; Silva, F.G. Acute postinfectious glomerulonephritis and glomerulonephritis complicating persistent bacterial infection. In Heptinstall’s Pathology of the Kidney, 6th ed.; Jennette, J.C., Olson, J.L., Schwartz, M.M., Silva, F.G., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 321–396. [Google Scholar]
- Nasr, S.H.; Fidler, M.E.; Valeri, A.M.; Cornell, L.D.; Sethi, S.; Zoller, A.; Stokes, M.B.; Markowitz, G.S.; D’Agati, V.D. Postinfectious glomerulonephritis in the elderly. J. Am. Soc. Nephrol. 2011, 22, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Oda, T.; Yoshizawa, N.; Yamakami, K.; Tamura, K.; Kuroki, A.; Sugisaki, T.; Sawanobori, E.; Higashida, K.; Ohtomo, Y.; Hotta, O.; et al. Localization of nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. Hum. Pathol. 2010, 41, 1276–1285. [Google Scholar] [CrossRef] [PubMed]
- Treser, G.; Semar, M.; McVicar, M.; Franklin, M.; Ty, A.; Sagel, I.; Lange, K. Antigenic streptococcal components in acute glomerulonephritis. Science 1969, 163, 676–677. [Google Scholar] [CrossRef]
- Winram, S.B.; Lottenberg, R. The plasmin-binding protein Plr of group A streptococci is identified as glyceraldehyde-3-phosphate dehydrogenase. Microbiology 1996, 142, 2311–2320. [Google Scholar] [CrossRef] [PubMed]
- Lottenberg, R.; Broder, C.C.; Boyle, M.D. Identification of a specific receptor for plasmin on a group A streptococcus. Infect. Immun. 1987, 55, 1914–1918. [Google Scholar] [CrossRef] [PubMed]
- Lottenberg, R.; DesJardin, L.E.; Wang, H.; Boyle, M.D. Streptokinase-producing streptococci grown in human plasma acquire unregulated cell-associated plasmin activity. J. Infect. Dis. 1992, 166, 436–440. [Google Scholar] [CrossRef]
- Oda, T.; Yamakami, K.; Omasu, F.; Suzuki, S.; Miura, S.; Sugisaki, T.; Yoshizawa, N. Glomerular plasmin-like activity in relation to nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. J. Am. Soc. Nephrol. 2005, 16, 247–254. [Google Scholar] [CrossRef]
- Oda, T.; Yoshizawa, N.; Yamakami, K.; Sakurai, Y.; Takechi, H.; Yamamoto, K.; Oshima, N.; Kumagai, H. The role of nephritis-associated plasmin receptor (NAPlr) in glomerulonephritis associated with streptococcal infection. J. Biomed. Biotechnol. 2012, 2012, 417675. [Google Scholar] [CrossRef]
- Vogt, A.; Batsford, S.; Rodriguez-Iturbe, B.; Garcia, R. Cationic antigens in poststreptococcal glomerulonephritis. Clin. Nephrol. 1983, 20, 271–279. [Google Scholar]
- Rodriguez-Iturbe, B. Autoimmunity in Acute Poststreptococcal GN: A Neglected Aspect of the Disease. J. Am. Soc. Nephrol. 2021, 32, 534–542. [Google Scholar] [CrossRef]
- Fish, A.J.; Herdman, R.C.; Michael, A.F.; Pickering, R.J.; Good, R.A. Epidemic acute glomerulonephritis associated with type 49 streptococcal pyoderma. II. Correlative study of light, immunofluorescent and electron microscopic findings. Am. J. Med. 1970, 48, 28–39. [Google Scholar] [CrossRef]
- Westberg, N.G.; Naff, G.B.; Boyer, J.T.; Michael, A.F. Glomerular deposition of properdin in acute and chronic glomerulonephritis with hypocomplementemia. J. Clin. Investig. 1971, 50, 642–649. [Google Scholar] [CrossRef]
- Verroust, P.J.; Wilson, C.B.; Cooper, N.R.; Edgington, T.S.; Dixon, F.J. Glomerular complement components in human glomerulonephritis. J. Clin. Investig. 1974, 53, 77–84. [Google Scholar] [CrossRef]
- Kluthe, R.; Vogt, A.; Batsford, S.R. Glomerulonephritis: International Conference on Pathogenesis, Pathology and Treatment, Freiburg, Germany, 18–19th June 1976; Thieme: New York, NY, USA, 1976. [Google Scholar]
- Wyatt, R.J.; McAdams, A.J.; Forristal, J.; Snyder, J.; West, C.D. Glomerular deposition of complement-control proteins in acute and chronic glomerulonephritis. Kidney Int. 1979, 16, 505–512. [Google Scholar] [CrossRef]
- Sorger, K.; Gessler, U.; Hubner, F.K.; Kohler, H.; Schulz, W.; Stuhlinger, W.; Thoenes, G.H.; Thoenes, W. Subtypes of acute postinfectious glomerulonephritis. Synopsis of clinical and pathological features. Clin. Nephrol. 1982, 17, 114–128. [Google Scholar]
- Madaio, M.P.; Harrington, J.T. Current concepts. The diagnosis of acute glomerulonephritis. N. Engl. J. Med. 1983, 309, 1299–1302. [Google Scholar] [CrossRef]
- Al-Ghaithi, B.; Chanchlani, R.; Riedl, M.; Thorner, P.; Licht, C. C3 Glomerulopathy and post-infectious glomerulonephritis define a disease spectrum. Pediatr. Nephrol. 2016, 31, 2079–2086. [Google Scholar] [CrossRef]
- Oda, T. Acute glomerulonephritis syndrome (AGS). In Shin Rinsho Naikagaku (PRACTICE OF INTERNAL MEDICINE), 10th ed; Yazaki, Y., Ed.; Igaku-Shoin Ltd.: Tokyo, Japan, 2020; pp. 1181–1183. [Google Scholar]
- Yoshizawa, N. Acute glomerulonephritis. Intern. Med. 2000, 39, 687–694. [Google Scholar] [CrossRef]
- Plesner, T.; Ploug, M.; Ellis, V.; Ronne, E.; Hoyer-Hansen, G.; Wittrup, M.; Pedersen, T.L.; Tscherning, T.; Dano, K.; Hansen, N.E. The receptor for urokinase-type plasminogen activator and urokinase is translocated from two distinct intracellular compartments to the plasma membrane on stimulation of human neutrophils. Blood 1994, 83, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Song, Y.P.; Boel, G.; Kochar, J.; Pancholi, V. Group A streptococcal surface GAPDH, SDH, recognizes uPAR/CD87 as its receptor on the human pharyngeal cell and mediates bacterial adherence to host cells. J. Mol. Biol. 2005, 350, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Boyle, M.D.; Lottenberg, R. Plasminogen activation by invasive human pathogens. Thromb. Haemost. 1997, 77, 001–010. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Ploplis, V.A.; Castellino, F.J. Bacterial plasminogen receptors utilize host plasminogen system for effective invasion and dissemination. J. Biomed. Biotechnol. 2012, 2012, 482096. [Google Scholar] [CrossRef]
- Pancholi, V.; Fischetti, V.A. A major surface protein on group A streptococci is a glyceraldehyde-3-phosphate-dehydrogenase with multiple binding activity. J. Exp. Med. 1992, 176, 415–426. [Google Scholar] [CrossRef]
- Uchida, T.; Oda, T. Glomerular Deposition of Nephritis-Associated Plasmin Receptor (NAPlr) and Related Plasmin Activity: Key Diagnostic Biomarkers of Bacterial Infection-related Glomerulonephritis. Int. J. Mol. Sci. 2020, 21, 2595. [Google Scholar] [CrossRef]
- Vassalli, J.D.; Sappino, A.P.; Belin, D. The plasminogen activator/plasmin system. J. Clin. Investig. 1991, 88, 1067–1072. [Google Scholar] [CrossRef]
- Mignatti, P.; Rifkin, D.B. Biology and biochemistry of proteinases in tumor invasion. Physiol. Rev. 1993, 73, 161–195. [Google Scholar] [CrossRef]
- Montrucchio, G.; Lupia, E.; De Martino, A.; Silvestro, L.; Savu, S.R.; Cacace, G.; De Filippi, P.G.; Emanuelli, G.; Camussi, G. Plasmin promotes an endothelium-dependent adhesion of neutrophils. Involvement of platelet activating factor and P-selectin. Circulation 1996, 93, 2152–2160. [Google Scholar] [CrossRef]
- Burysek, L.; Syrovets, T.; Simmet, T. The serine protease plasmin triggers expression of MCP-1 and CD40 in human primary monocytes via activation of p38 MAPK and janus kinase (JAK)/STAT signaling pathways. J. Biol. Chem. 2002, 277, 33509–33517. [Google Scholar] [CrossRef]
- Syrovets, T.; Simmet, T. Novel aspects and new roles for the serine protease plasmin. Cell Mol. Life Sci. 2004, 61, 873–885. [Google Scholar] [CrossRef]
- Yoshizawa, N.; Oda, T.; Oshikawa, Y.; Akashi, Y.; Suzuki, Y.; Shimizu, J.; Shimizu, E.; Niwa, H.; Treser, G. Cell-mediated immune response in acute poststreptococcal glomerulonephritis. Nihon Jinzo Gakkai Shi 1994, 36, 322–330. [Google Scholar]
- Oda, T.; Yoshizawa, N.; Takeuchi, A.; Nakabayashi, I.; Nishiyama, J.; Ishida, A.; Tazawa, K.; Murayama, M.; Hotta, O.; Taguma, Y. Glomerular proliferating cell kinetics in acute post-streptococcal glomerulonephritis (APSGN). J. Pathol. 1997, 183, 359–368. [Google Scholar] [CrossRef]
- Platt, J.L.; Grant, B.W.; Eddy, A.A.; Michael, A.F. Immune cell populations in cutaneous delayed-type hypersensitivity. J. Exp. Med. 1983, 158, 1227–1242. [Google Scholar] [CrossRef]
- Arai, M.; Mii, A.; Kashiwagi, T.; Shimizu, A.; Sakai, Y. The severity of glomerular endothelial cell injury is associated with infiltrating macrophage heterogeneity in endocapillary proliferative glomerulonephritis. Sci. Rep. 2021, 11, 13339. [Google Scholar] [CrossRef]
- Yoshizawa, N.; Treser, G.; Sagel, I.; Ty, A.; Ahmed, U.; Lange, K. Demonstration of antigenic sites in glomeruli of patients with acute poststreptococcal glomerulonephritis by immunofluorescein and immunoferritin technics. Am. J. Pathol. 1973, 70, 131–150. [Google Scholar]
- Seegal, B.C.; Andres, G.A.; Hsu, K.C.; Zabriskie, J.B. Studies on the Pathogenesis of Acute and Progressive Glomerulonephritis in Man by Immunofluorescein and Immunoferritin Techniques. Fed. Proc. 1965, 24, 100–108. [Google Scholar]
- Andres, G.A.; Accinni, L.; Hsu, K.C.; Zabriskie, J.B.; Seegal, B.C. Electron microscopic studies of human glomerulonephritis with ferritin-conjugated antibody. Localization of antigen-antibody complexes in glomerular structures of patients with acute glomerulonephritis. J. Exp. Med. 1966, 123, 399–412. [Google Scholar] [CrossRef]
- Batsford, S.R.; Mezzano, S.; Mihatsch, M.; Schiltz, E.; Rodriguez-Iturbe, B. Is the nephritogenic antigen in post-streptococcal glomerulonephritis pyrogenic exotoxin B (SPE B) or GAPDH? Kidney Int. 2005, 68, 1120–1129. [Google Scholar] [CrossRef]
- Oshima, S.; Yoshizawa, N. Role of the membrane attack complex (MAC) in acute poststreptococcal glomerulonephritis. Nihon Jinzo Gakkai Shi 1988, 30, 811–819. [Google Scholar]
- Yoshizawa, N.; Oshima, S.; Takeuchi, A.; Kondo, S.; Oda, T.; Shimizu, J.; Nishiyama, J.; Ishida, A.; Nakabayashi, I.; Tazawa, K.; et al. Experimental acute glomerulonephritis induced in the rabbit with a specific streptococcal antigen. Clin. Exp. Immunol. 1997, 107, 61–67. [Google Scholar] [CrossRef]
- Yoshizawa, N.; Oshima, S.; Sagel, I.; Shimizu, J.; Treser, G. Role of a streptococcal antigen in the pathogenesis of acute poststreptococcal glomerulonephritis. Characterization of the antigen and a proposed mechanism for the disease. J. Immunol. 1992, 148, 3110–3116. [Google Scholar]
- Satoskar, A.A.; Silva, F.G. Acute postinfectious glomerulonephritis and glomerulonephritis caused by persistent bacterial infection. In Heptinstall’s Pathology of the Kidney, 7th ed.; Jennette, J.C., Olson, J.L., Silva, F.G., D’Agati, V.D., Eds.; Wolters Kluwer: Philadelphia, PA, USA, 2015; pp. 367–436. [Google Scholar]
- Pickering, M.C.; D’Agati, V.D.; Nester, C.M.; Smith, R.J.; Haas, M.; Appel, G.B.; Alpers, C.E.; Bajema, I.M.; Bedrosian, C.; Braun, M.; et al. C3 glomerulopathy: Consensus report. Kidney Int. 2013, 84, 1079–1089. [Google Scholar] [CrossRef]
- Sethi, S.; Gamez, J.D.; Vrana, J.A.; Theis, J.D.; Bergen, H.R., 3rd; Zipfel, P.F.; Dogan, A.; Smith, R.J. Glomeruli of Dense Deposit Disease contain components of the alternative and terminal complement pathway. Kidney Int. 2009, 75, 952–960. [Google Scholar] [CrossRef]
- Ito, N.; Ohashi, R.; Nagata, M. C3 glomerulopathy and current dilemmas. Clin. Exp. Nephrol. 2017, 21, 541–551. [Google Scholar] [CrossRef]
- Ravindran, A.; Fervenza, F.C.; Smith, R.J.H.; Sethi, S. C3 glomerulopathy associated with monoclonal Ig is a distinct subtype. Kidney Int. 2018, 94, 178–186. [Google Scholar] [CrossRef]
- Fakhouri, F.; Le Quintrec, M.; Fremeaux-Bacchi, V. Practical management of C3 glomerulopathy and Ig-mediated MPGN: Facts and uncertainties. Kidney Int. 2020, 98, 1135–1148. [Google Scholar] [CrossRef]
- Levine, A.P.; Chan, M.M.Y.; Sadeghi-Alavijeh, O.; Wong, E.K.S.; Cook, H.T.; Ashford, S.; Carss, K.; Christian, M.T.; Hall, M.; Harris, C.L.; et al. Large-Scale Whole-Genome Sequencing Reveals the Genetic Architecture of Primary Membranoproliferative GN and C3 Glomerulopathy. J. Am. Soc. Nephrol. 2020, 31, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Chauvet, S.; Roumenina, L.T.; Aucouturier, P.; Marinozzi, M.C.; Dragon-Durey, M.A.; Karras, A.; Delmas, Y.; Le Quintrec, M.; Guerrot, D.; Jourde-Chiche, N.; et al. Both Monoclonal and Polyclonal Immunoglobulin Contingents Mediate Complement Activation in Monoclonal Gammopathy Associated-C3 Glomerulopathy. Front. Immunol. 2018, 9, 2260. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Vrana, J.A.; Fervenza, F.C.; Theis, J.D.; Sethi, A.; Kurtin, P.J.; Zhang, Y.; Smith, R.J.H. Characterization of C3 in C3 glomerulopathy. Nephrol. Dial. Transplant. 2017, 32, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Bomback, A.S.; Santoriello, D.; Avasare, R.S.; Regunathan-Shenk, R.; Canetta, P.A.; Ahn, W.; Radhakrishnan, J.; Marasa, M.; Rosenstiel, P.E.; Herlitz, L.C.; et al. C3 glomerulonephritis and dense deposit disease share a similar disease course in a large United States cohort of patients with C3 glomerulopathy. Kidney Int. 2018, 93, 977–985. [Google Scholar] [CrossRef]
- Kaartinen, K.; Safa, A.; Kotha, S.; Ratti, G.; Meri, S. Complement dysregulation in glomerulonephritis. Semin. Immunol. 2019, 45, 101331. [Google Scholar] [CrossRef]
- Lemaire, M.; Noone, D.; Lapeyraque, A.L.; Licht, C.; Fremeaux-Bacchi, V. Inherited Kidney Complement Diseases. Clin. J. Am. Soc. Nephrol. 2021, 16, 942–956. [Google Scholar] [CrossRef]
- Cook, H.T.; Pickering, M.C. C3 glomerulopathies, including dense deposit disease. In Heptinstall’s Pathology of the Kidney, 7th ed.; Jennette, J.C., Olson, J.L., Silva, F.G., D’Agati, V.D., Eds.; Wolters Kluwer: Philadelphia, PA, USA, 2015; pp. 341–366. [Google Scholar]
- Takahashi, M.; Takahashi, S.; Hirose, S. Solubilization of antigen-antibody complexes: A new function of complement as a regulator of immune reactions. Prog. Allergy 1980, 27, 134–166. [Google Scholar]
- Gadd, K.J.; Reid, K.B. The binding of complement component C3 to antibody-antigen aggregates after activation of the alternative pathway in human serum. Biochem. J. 1981, 195, 471–480. [Google Scholar] [CrossRef]
- Takata, Y.; Tamura, N.; Fujita, T. Interaction of C3 with antigen-antibody complexes in the process of solubilization of immune precipitates. J. Immunol. 1984, 132, 2531–2537. [Google Scholar]
- Yoshizawa, N.; Treser, G.; McClung, J.A.; Sagel, I.; Takahashi, K. Circulating immune complexes in patients with uncomplicated group A streptococcal pharyngitis and patients with acute poststreptococcal glomerulonephritis. Am. J. Nephrol. 1983, 3, 23–29. [Google Scholar] [CrossRef]
- Messias, N.C.; Walker, P.D.; Larsen, C.P. Paraffin immunofluorescence in the renal pathology laboratory: More than a salvage technique. Mod. Pathol. 2015, 28, 854–860. [Google Scholar] [CrossRef]
- Tauber, J.W.; Polley, M.J.; Zabriskie, J.B. Nonspecific complement activation by streptococcal structures. II. Properdin-independent initiation of the alternate pathway. J. Exp. Med. 1976, 143, 1352–1366. [Google Scholar] [CrossRef]
- Vernon, K.A.; Goicoechea de Jorge, E.; Hall, A.E.; Fremeaux-Bacchi, V.; Aitman, T.J.; Cook, H.T.; Hangartner, R.; Koziell, A.; Pickering, M.C. Acute presentation and persistent glomerulonephritis following streptococcal infection in a patient with heterozygous complement factor H-related protein 5 deficiency. Am. J. Kidney Dis. 2012, 60, 121–125. [Google Scholar] [CrossRef]
- Chauvet, S.; Berthaud, R.; Devriese, M.; Mignotet, M.; Vieira Martins, P.; Robe-Rybkine, T.; Miteva, M.A.; Gyulkhandanyan, A.; Ryckewaert, A.; Louillet, F.; et al. Anti-Factor B Antibodies and Acute Postinfectious GN in Children. J. Am. Soc. Nephrol. 2020, 31, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Fervenza, F.C.; Zhang, Y.; Zand, L.; Meyer, N.C.; Borsa, N.; Nasr, S.H.; Smith, R.J. Atypical postinfectious glomerulonephritis is associated with abnormalities in the alternative pathway of complement. Kidney Int. 2013, 83, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Radhakrishnan, J.; D’Agati, V.D. Bacterial infection-related glomerulonephritis in adults. Kidney Int. 2013, 83, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Sawanobori, E.; Umino, A.; Kanai, H.; Matsushita, K.; Iwasa, S.; Kitamura, H.; Oda, T.; Yoshizawa, N.; Sugita, K.; Higashida, K. A prolonged course of Group A streptococcus-associated nephritis: A mild case of dense deposit disease (DDD)? Clin. Nephrol. 2009, 71, 703–707. [Google Scholar] [CrossRef]
- Suga, K.; Kondo, S.; Matsuura, S.; Kinoshita, Y.; Kitano, E.; Hatanaka, M.; Kitamura, H.; Hidaka, Y.; Oda, T.; Kagami, S. A case of dense deposit disease associated with a group A streptococcal infection without the involvement of C3NeF or complement factor H deficiency. Pediatr. Nephrol. 2010, 25, 1547–1550. [Google Scholar] [CrossRef]
- Okabe, M.; Tsuboi, N.; Yokoo, T.; Miyazaki, Y.; Utsunomiya, Y.; Hosoya, T. A case of idiopathic membranoproliferative glomerulonephritis with a transient glomerular deposition of nephritis-associated plasmin receptor antigen. Clin. Exp. Nephrol. 2012, 16, 337–341. [Google Scholar] [CrossRef]
- Iseri, K.; Iyoda, M.; Yamamoto, Y.; Kobayashi, N.; Oda, T.; Yamaguchi, Y.; Shibata, T. Streptococcal Infection-related Nephritis (SIRN) Manifesting Membranoproliferative Glomerulonephritis Type I. Intern. Med. 2016, 55, 647–650. [Google Scholar] [CrossRef][Green Version]
- Kohatsu, K.; Suzuki, T.; Yazawa, M.; Yahagi, K.; Ichikawa, D.; Koike, J.; Oda, T.; Shibagaki, Y. Granulomatosis with Polyangiitis Induced by Infection. Kidney Int. Rep. 2019, 4, 341–345. [Google Scholar] [CrossRef]
- Yano, K.; Suzuki, H.; Oda, T.; Ueda, Y.; Tsukamoto, T.; Muso, E. Crescentic poststreptococcal acute glomerulonephritis accompanied by small vessel vasculitis: Case report of an elderly male. BMC Nephrol. 2019, 20, 471. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Yoshizawa, N.; Oda, T.; Imakiire, T.; Suzuki, S.; Miura, S. Streptococcal origin of a case of Henoch-Schoenlein purpura nephritis. Clin. Nephrol. 2006, 65, 124–128. [Google Scholar] [CrossRef]
- Odaka, J.; Kanai, T.; Ito, T.; Saito, T.; Aoyagi, J.; Betsui, H.; Oda, T.; Ueda, Y.; Yamagata, T. A case of post-pneumococcal acute glomerulonephritis with glomerular depositions of nephritis-associated plasmin receptor. CEN Case Rep. 2015, 4, 112–116. [Google Scholar] [CrossRef]
- Komaru, Y.; Ishioka, K.; Oda, T.; Ohtake, T.; Kobayashi, S. Nephritis-associated plasmin receptor (NAPlr) positive glomerulonephritis caused by Aggregatibacter actinomycetemcomitans bacteremia: A case report. Clin. Nephrol. 2018, 90, 155–160. [Google Scholar] [CrossRef]
- Hirano, D.; Oda, T.; Ito, A.; Yamada, A.; Kakegawa, D.; Miwa, S.; Umeda, C.; Takemasa, Y.; Tokunaga, A.; Wajima, T.; et al. Glyceraldehyde-3-phosphate dehydrogenase of Mycoplasma pneumoniae induces infection-related glomerulonephritis. Clin. Nephrol. 2019, 92, 263–272. [Google Scholar] [CrossRef]
- Grundel, A.; Pfeiffer, M.; Jacobs, E.; Dumke, R. Network of Surface-Displayed Glycolytic Enzymes in Mycoplasma pneumoniae and Their Interactions with Human Plasminogen. Infect. Immun. 2015, 84, 666–676. [Google Scholar] [CrossRef]
- Lottenberg, R.; Broder, C.C.; Boyle, M.D.; Kain, S.J.; Schroeder, B.L.; Curtiss, R., 3rd. Cloning, sequence analysis, and expression in Escherichia coli of a streptococcal plasmin receptor. J. Bacteriol. 1992, 174, 5204–5210. [Google Scholar] [CrossRef][Green Version]
- Winram, S.B.; Lottenberg, R. Site-directed mutagenesis of streptococcal plasmin receptor protein (Plr) identifies the C-terminal Lys334 as essential for plasmin binding, but mutation of the plr gene does not reduce plasmin binding to group A streptococci. Microbiology 1998, 144, 2025–2035. [Google Scholar] [CrossRef]
- Takehara, E.; Mandai, S.; Shikuma, S.; Akita, W.; Chiga, M.; Mori, T.; Oda, T.; Kuwahara, M.; Uchida, S. Post-infectious Proliferative Glomerulonephritis with Monoclonal Immunoglobulin G Deposits Associated with Complement Factor H Mutation. Intern. Med. 2017, 56, 811–817. [Google Scholar] [CrossRef]
- Okabe, M.; Takamura, T.; Tajiri, A.; Tsuboi, N.; Ishikawa, M.; Ogura, M.; Ohashi, R.; Oda, T.; Yokoo, T. A case of infection-related glomerulonephritis with massive eosinophilic infiltration. Clin. Nephrol. 2018, 90, 142–147. [Google Scholar] [CrossRef]
- Asano, M.; Oda, T.; Mizuno, M. A case of C3 glomerulopathy with nephritis-associated plasmin receptor positivity without a history of streptococcal infection. CEN Case Rep. 2021, 11, 259–264. [Google Scholar] [CrossRef]
- Komaki, K.; Shiotsu, Y.; Adachi, H.; Urata, N.; Hara, M.; Nakayama, M.; Kusaba, T.; Masuzawa, N.; Konishi, E.; Oda, T.; et al. Nephritis-associated plasmin receptor (NAPlr)-positive glomerulonephritis in a case of ANCA-negative small vessel vasculitis. CEN Case Rep. 2022, 11, 90–96. [Google Scholar] [CrossRef]
- Onishi, A.; Mizumoto, A.; Mitsumoto, K.; Katsunuma, R.; Shingu, T.; Oda, T.; Uzu, T. A man with immunoglobulin A nephropathy complicated by infection-related glomerulonephritis with glomerular depositions of nephritis-associated plasmin receptor. CEN Case Rep. 2021, 10, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Suzuki, T.; Watanabe, S.; Nakata, M.; Ichikawa, D.; Koike, J.; Oda, T.; Suzuki, H.; Suzuki, Y.; Shibagaki, Y. Galactose-deficient IgA1 and nephritis-associated plasmin receptors as markers for IgA-dominant infection-related glomerulonephritis: A case report. Medicine 2021, 100, e24460. [Google Scholar] [CrossRef] [PubMed]
- Noda, S.; Mandai, S.; Oda, T.; Shinoto, T.; Sato, H.; Sato, K.; Hirokawa, K.; Noda, Y.; Uchida, S. Asymptomatic sinusitis as an origin of infection-related glomerulonephritis manifesting steroid-resistant nephrotic syndrome: A case report. Medicine 2020, 99, e20572. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Galgano, S.J.; Markowitz, G.S.; Stokes, M.B.; D’Agati, V.D. Immunofluorescence on pronase-digested paraffin sections: A valuable salvage technique for renal biopsies. Kidney Int. 2006, 70, 2148–2151. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.H.; Fidler, M.E.; Said, S.M. Paraffin Immunofluorescence: A Valuable Ancillary Technique in Renal Pathology. Kidney Int. Rep. 2018, 3, 1260–1266. [Google Scholar] [CrossRef]
- Nasr, S.H.; Fidler, M.E.; Said, S.M.; Koepplin, J.W.; Altamirano-Alonso, J.M.; Leung, N. Immunofluorescence staining for immunoglobulin heavy chain/light chain on kidney biopsies is a valuable ancillary technique for the diagnosis of monoclonal gammopathy-associated kidney diseases. Kidney Int. 2021, 100, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Larsen, C.P.; Messias, N.C.; Walker, P.D.; Fidler, M.E.; Cornell, L.D.; Hernandez, L.H.; Alexander, M.P.; Sethi, S.; Nasr, S.H. Membranoproliferative glomerulonephritis with masked monotypic immunoglobulin deposits. Kidney Int. 2015, 88, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Larsen, C.P.; Ambuzs, J.M.; Bonsib, S.M.; Boils, C.L.; Cossey, L.N.; Messias, N.C.; Silva, F.G.; Wang, Y.H.; Gokden, N.; Walker, P.D. Membranous-like glomerulopathy with masked IgG kappa deposits. Kidney Int. 2014, 86, 154–161. [Google Scholar] [CrossRef]
- Howlader, A.; Thajudeen, B.; Sussman, A.N.; Bracamonte, E.; Krahl, L.; Nasr, S.H. Proliferative Glomerulonephritis with Masked Monoclonal Deposits Responsive to Myeloma Therapy. Kidney Int. Rep. 2017, 2, 1233–1237. [Google Scholar] [CrossRef][Green Version]
- DiFranza, L.T.; Markowitz, G.S.; D’Agati, V.D.; Santoriello, D. Atypical Infection-Related Glomerulonephritis With “Masked” IgG-Kappa Crystalline Hump-Like Deposits. Kidney Int. Rep. 2021, 6, 228–233. [Google Scholar] [CrossRef]
- Yu, X.J.; Hu, N.; Wang, S.X.; Zhou, F.D.; Zhao, M.H. Membranoproliferative glomerulonephritis with deposition of monoclonal IgG evolved from polyclonal IgG: A case report with two consecutive renal biopsies. BMC Nephrol. 2019, 20, 275. [Google Scholar] [CrossRef]
- Johnson, C.K.; Zuniga, S.C.; Dhawale, T.; Zhang, Y.; Smith, R.J.H.; Blosser, C.D. Monoclonal Gammopathy of Renal Significance Causes C3 Glomerulonephritis Via Monoclonal IgG Kappa Inhibition of Complement Factor H. Kidney Int. Rep. 2021, 6, 2505–2509. [Google Scholar] [CrossRef]
- Sethi, S.; Fervenza, F.C.; Zhang, Y.; Zand, L.; Vrana, J.A.; Nasr, S.H.; Theis, J.D.; Dogan, A.; Smith, R.J. C3 glomerulonephritis: Clinicopathological findings, complement abnormalities, glomerular proteomic profile, treatment, and follow-up. Kidney Int. 2012, 82, 465–473. [Google Scholar] [CrossRef]

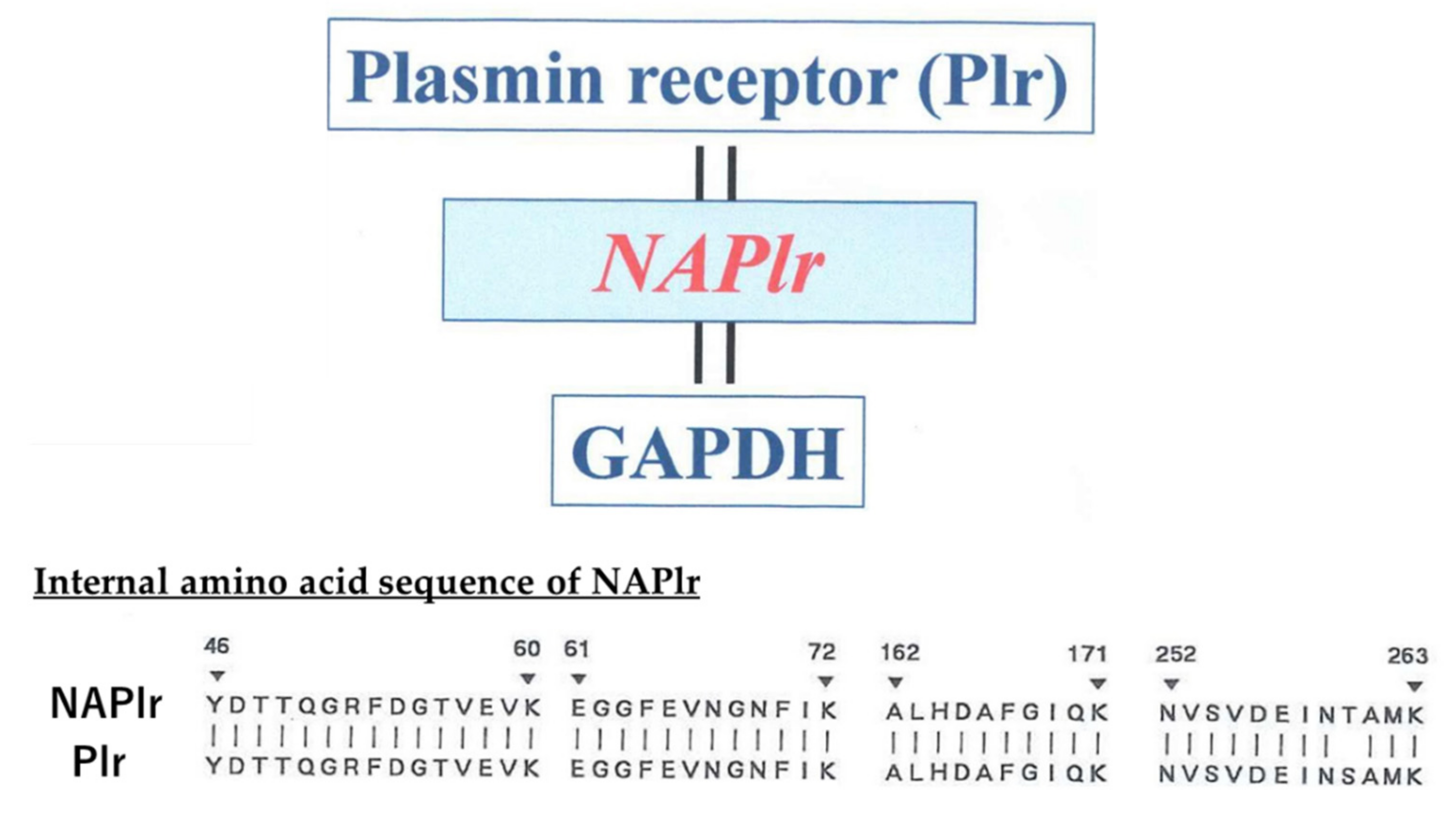

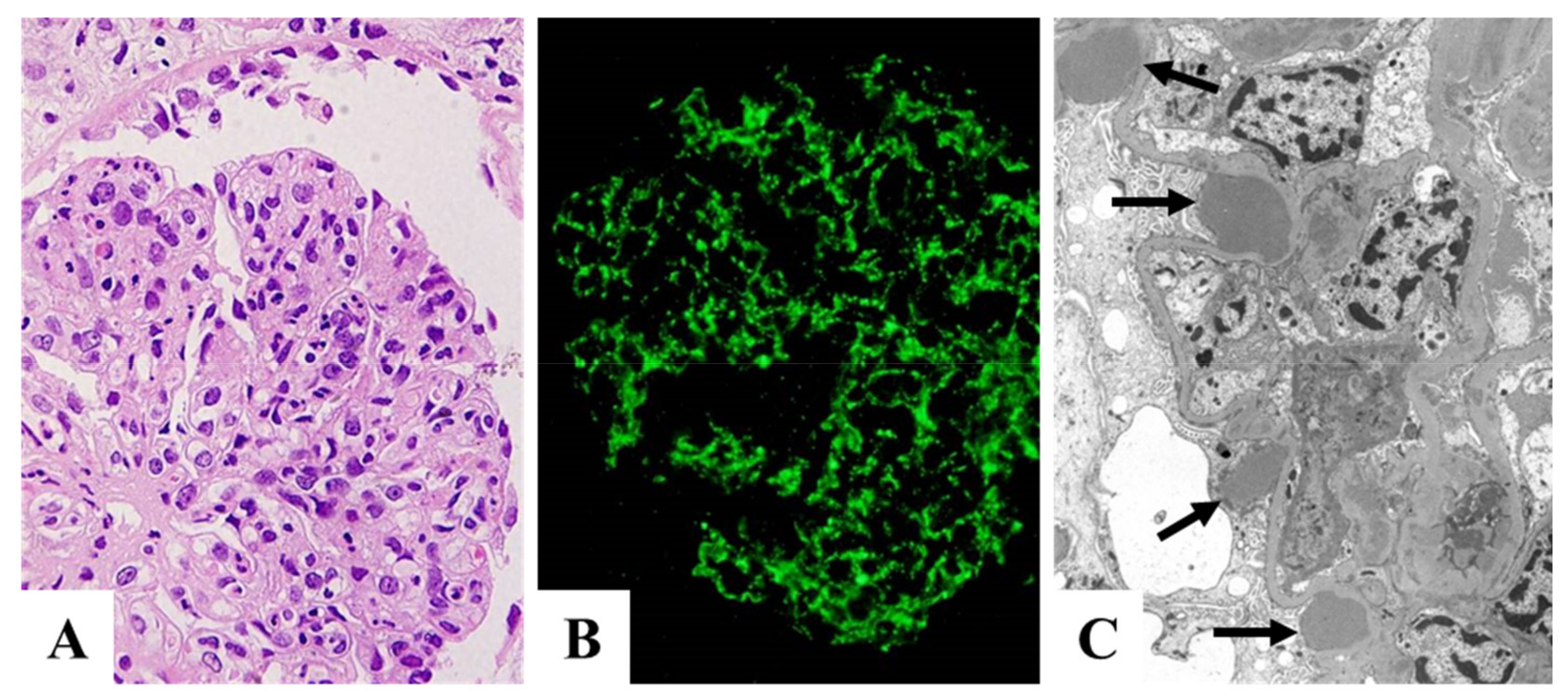
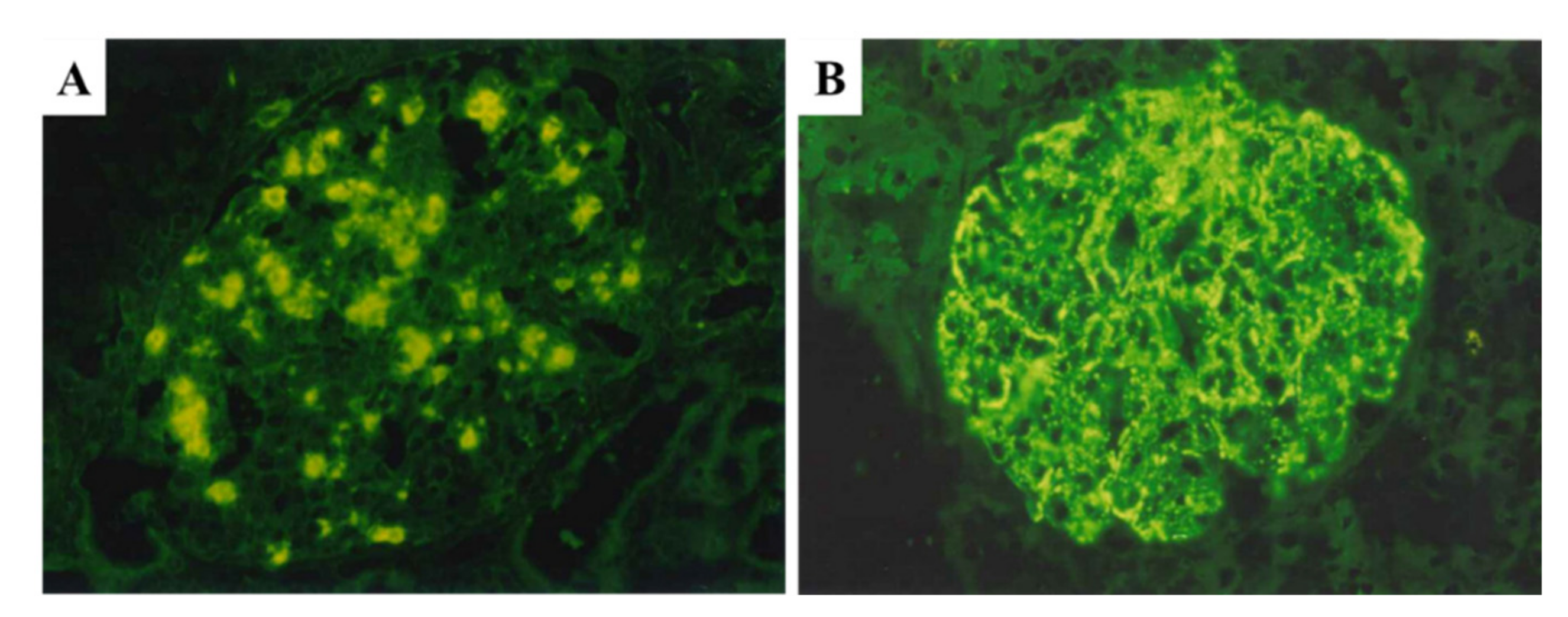
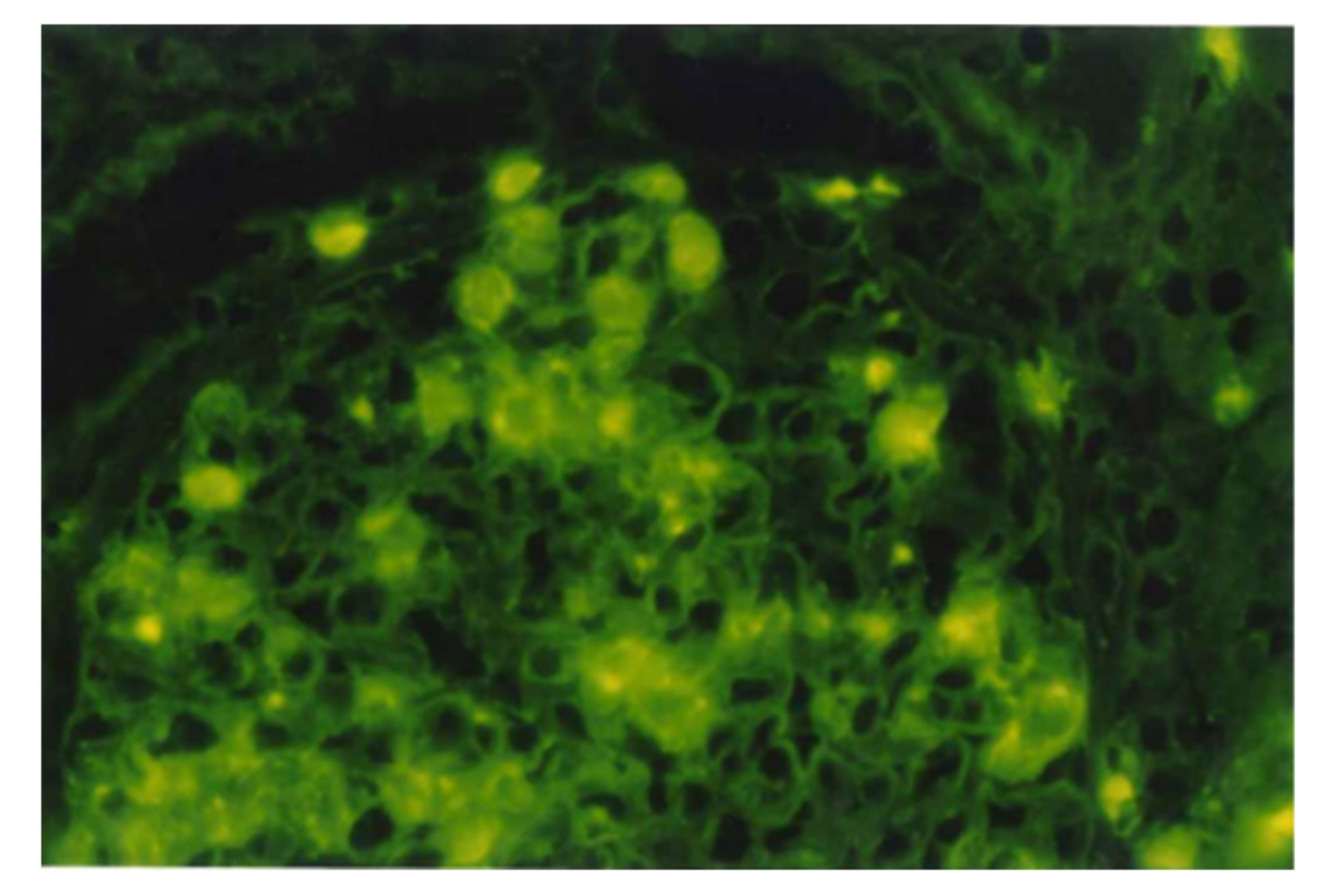
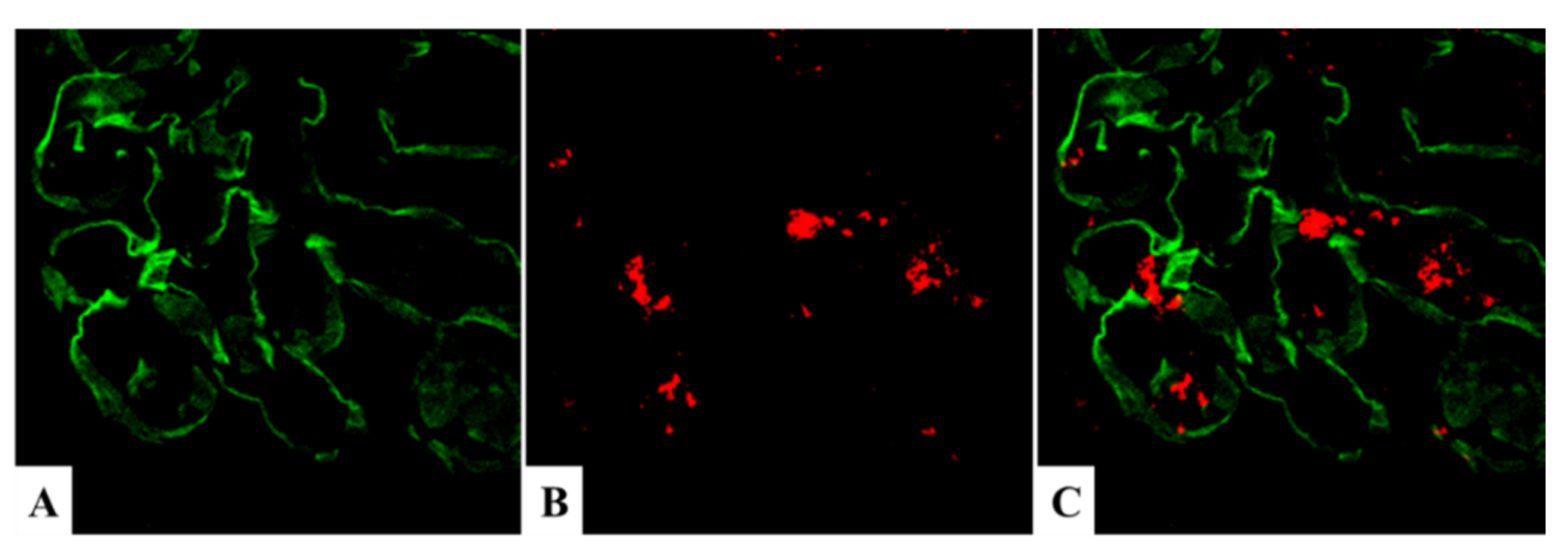
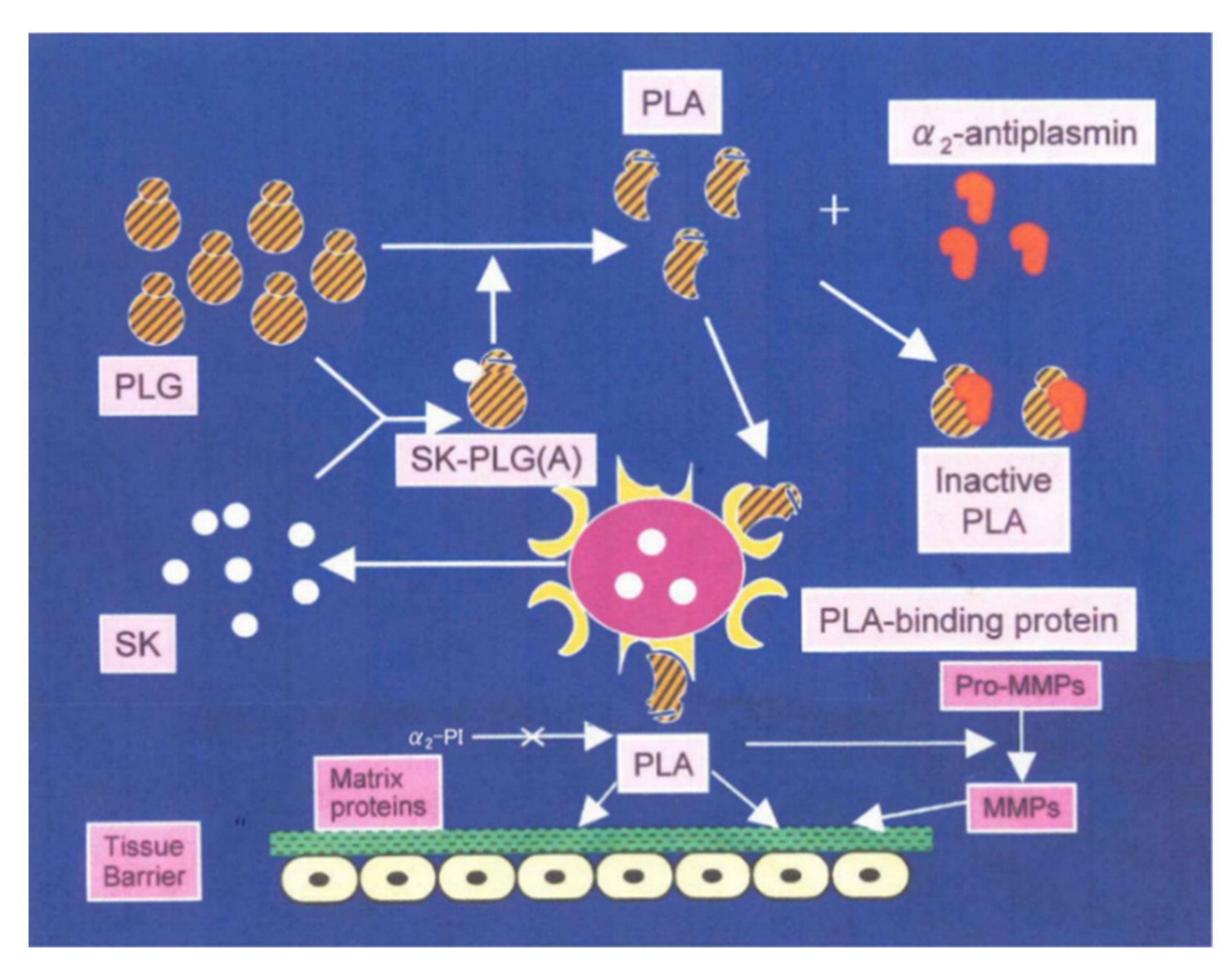

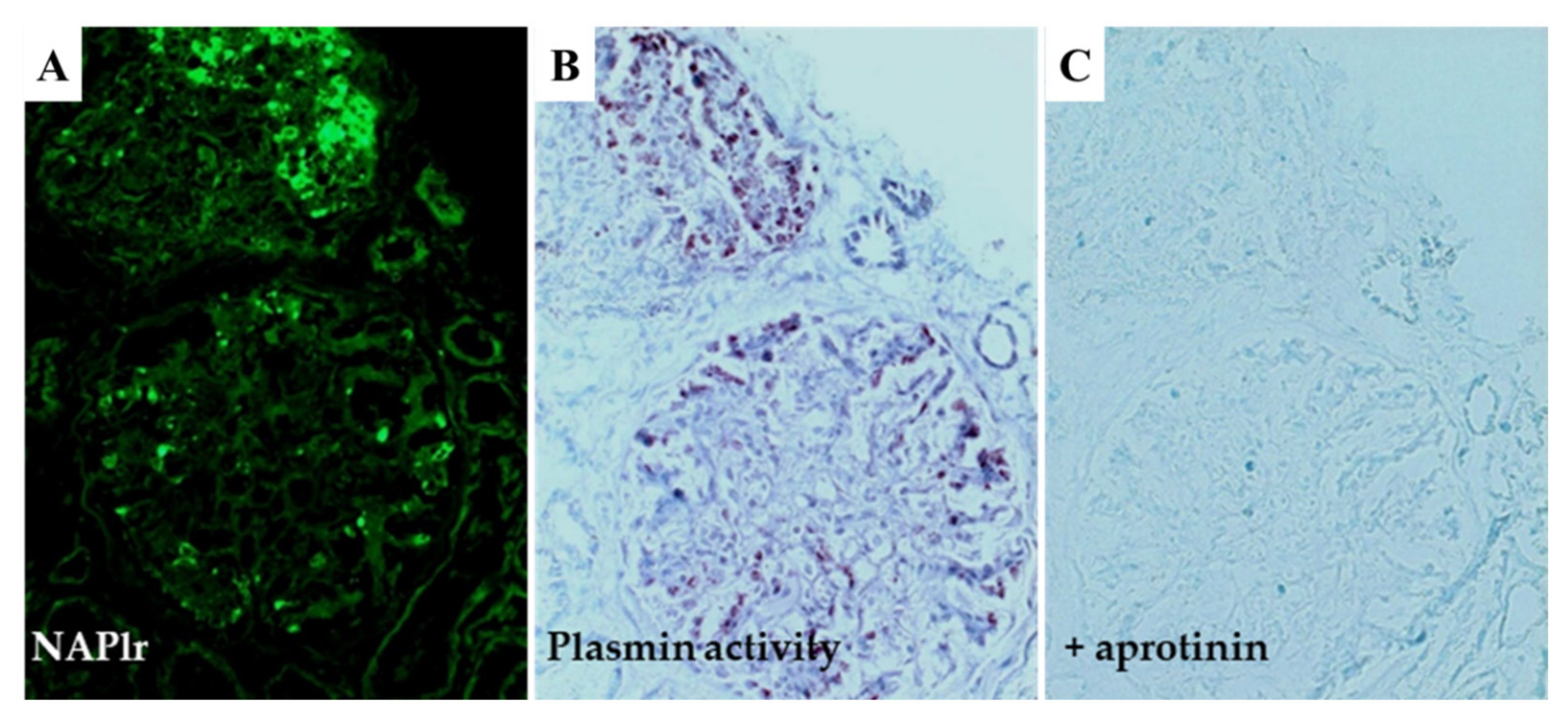
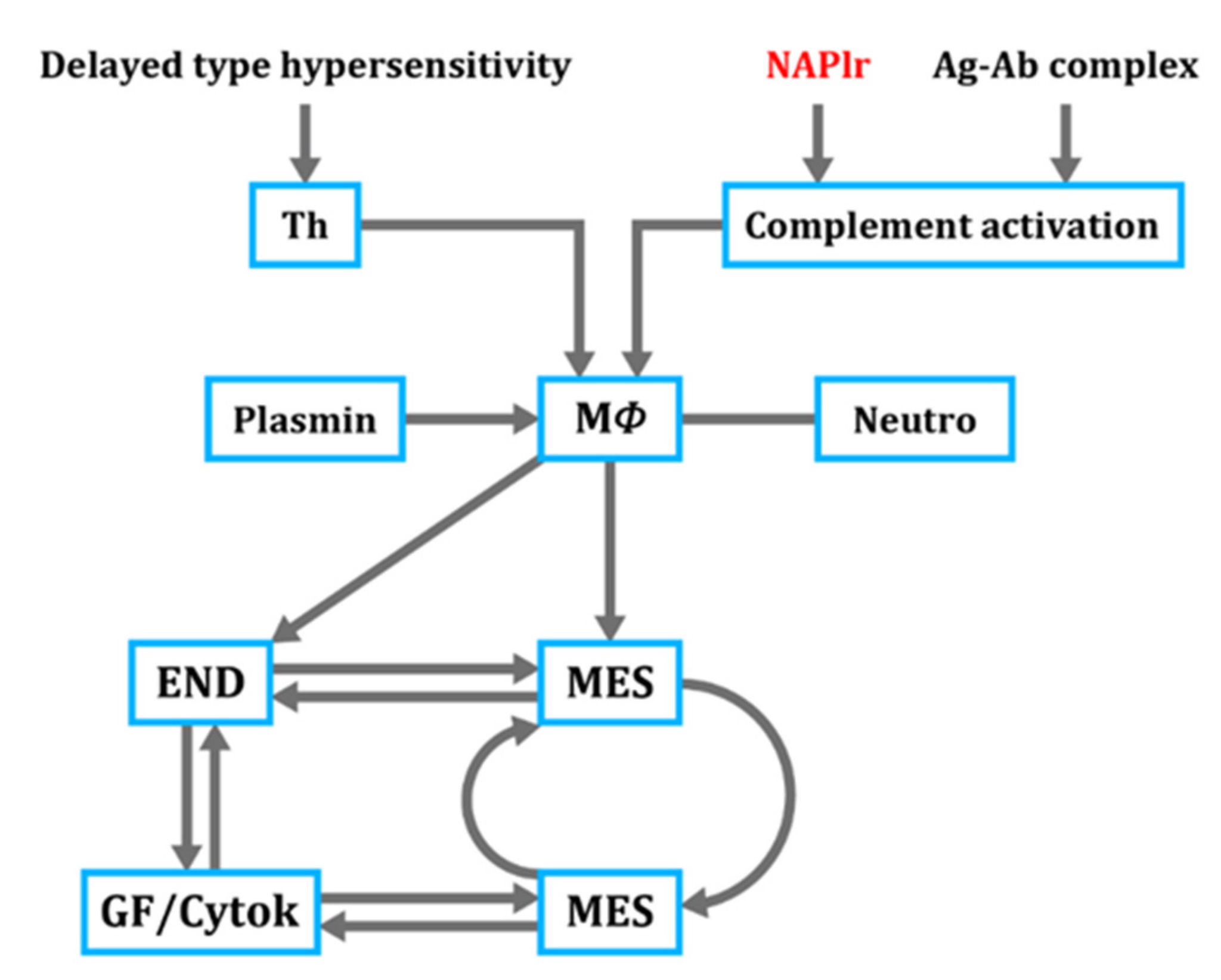

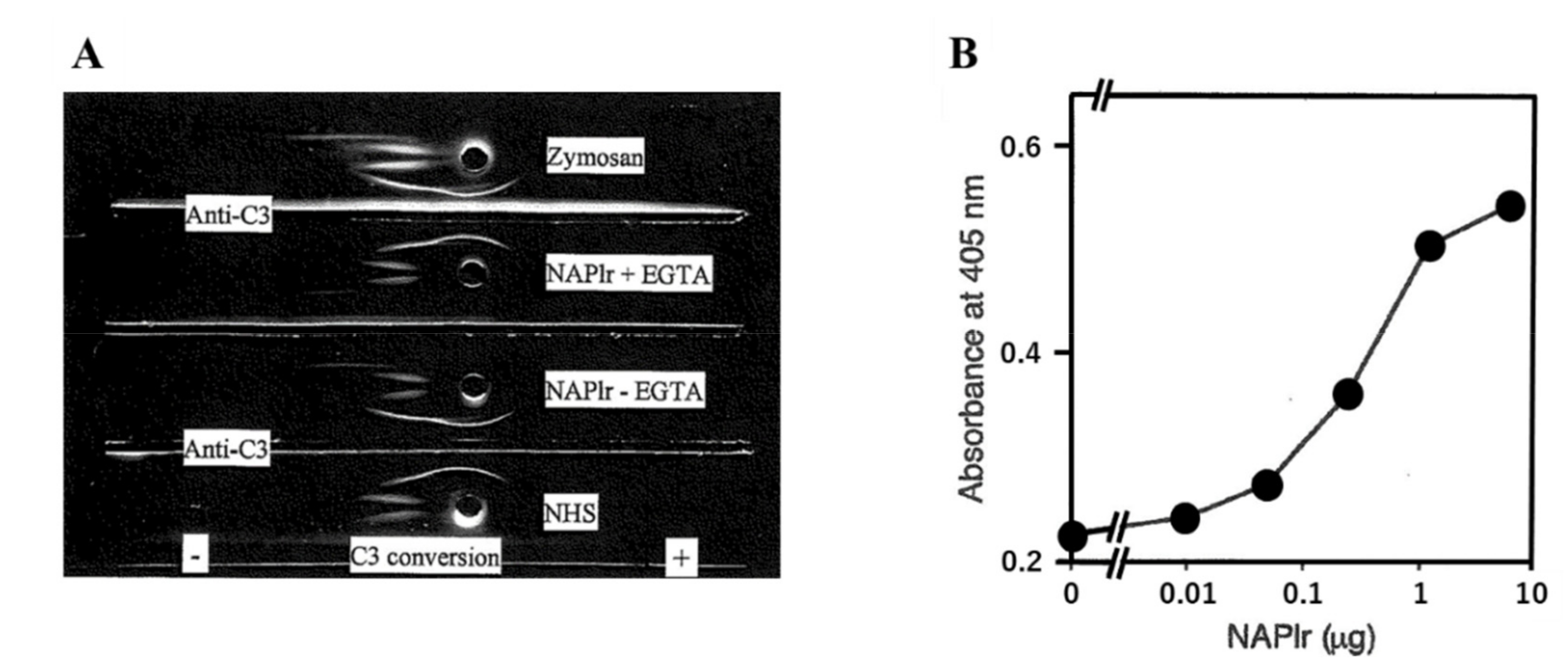
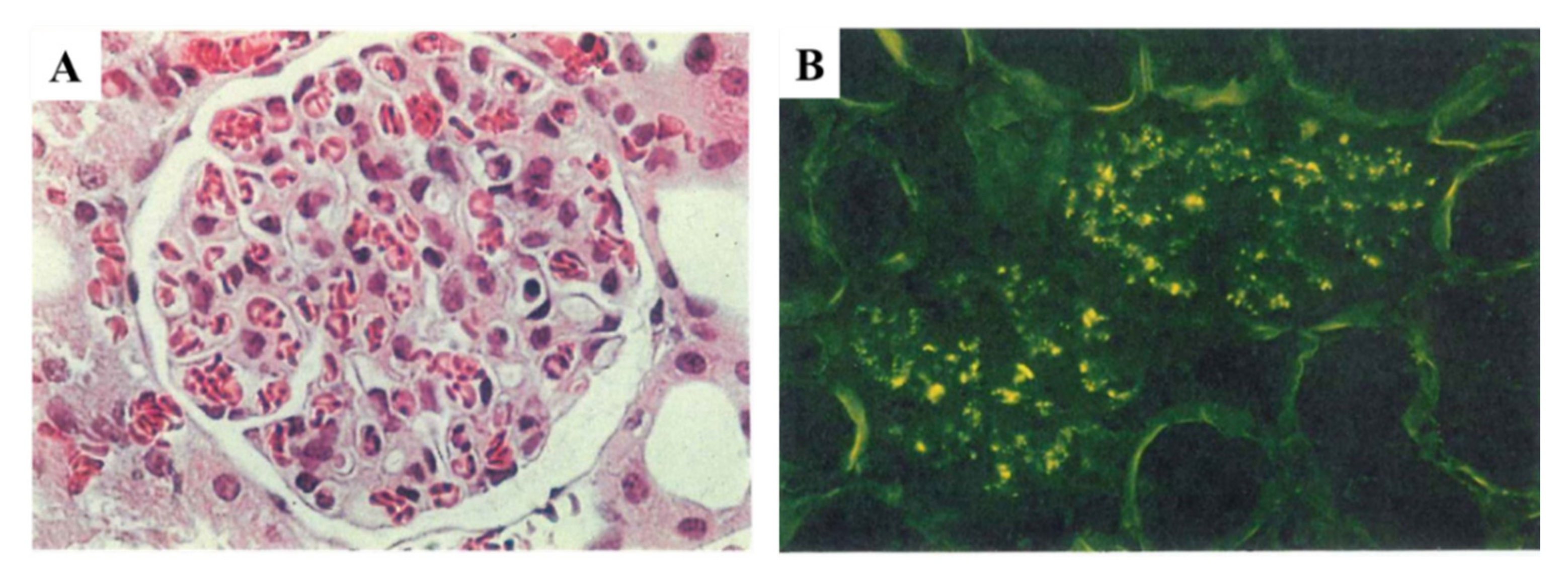
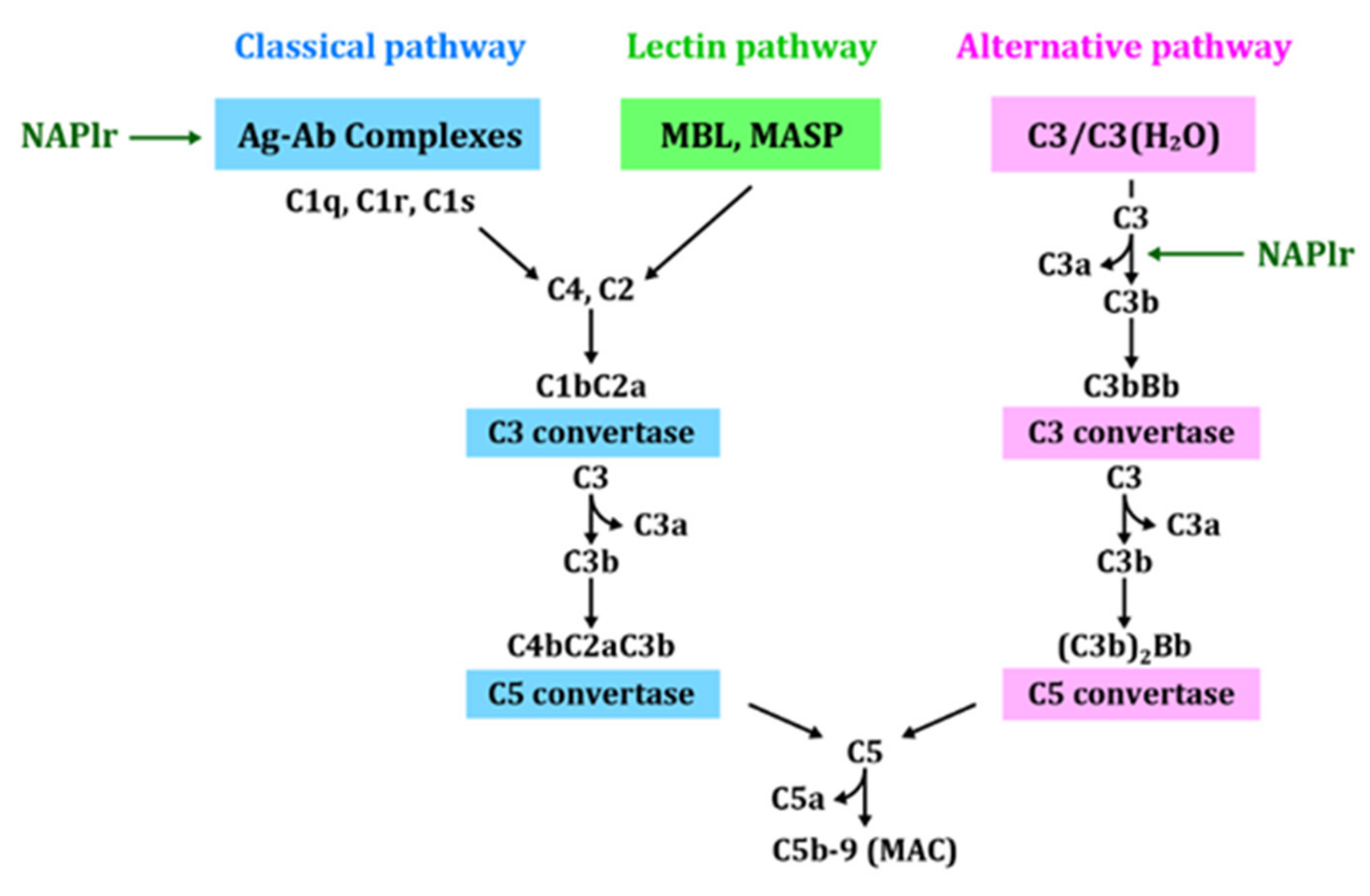
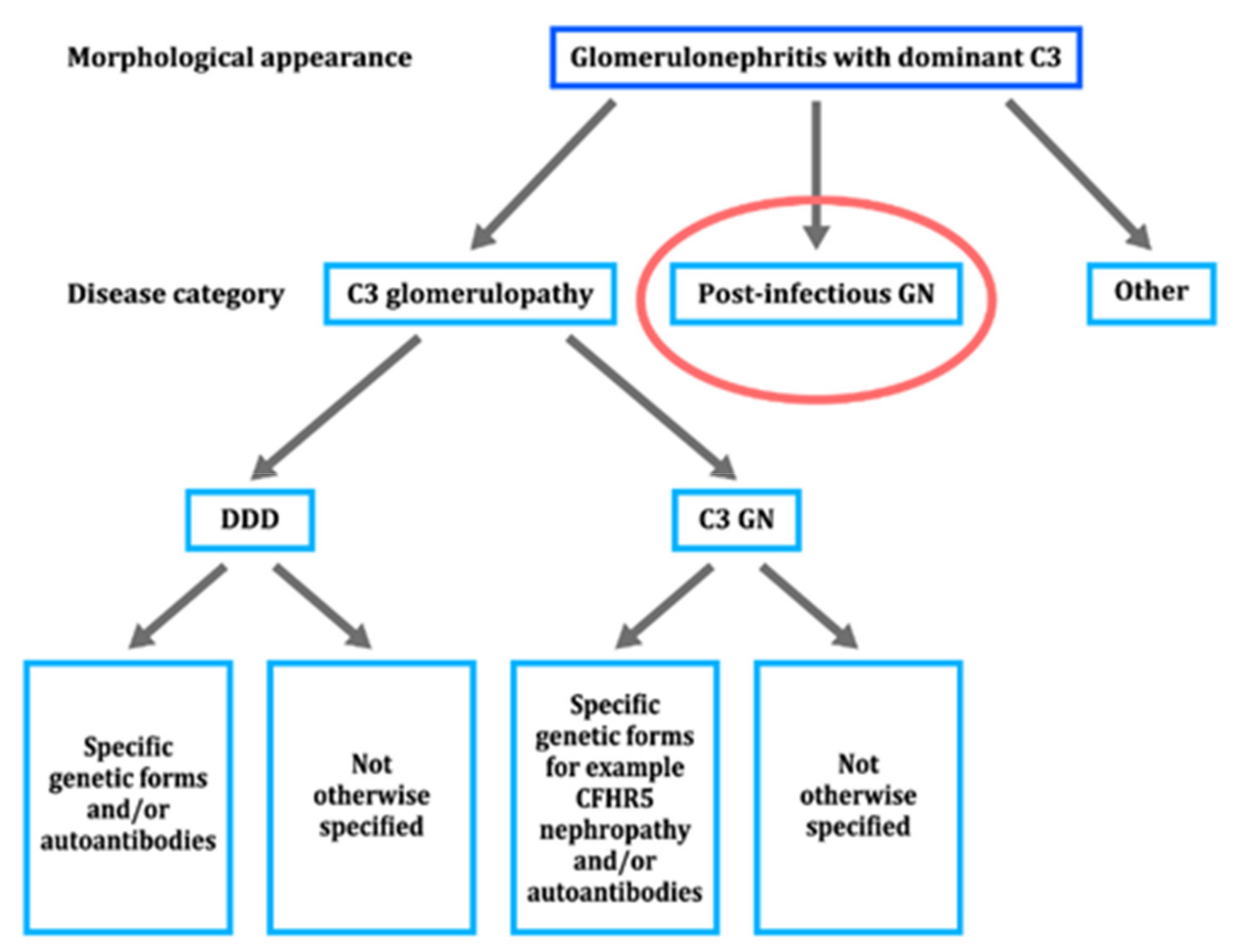

| Time from Onset to Biopsy | n | NAPlr | Plasminogen | Fibrinogen | C3 | IgG | IgA | IgM |
|---|---|---|---|---|---|---|---|---|
| 1–14 d | 25 | 25/25 (100%) | 10/25 (40) | 15/25 (60) | 25/25 (100) | 16/25 (64) | 11/25 (44) | 10/25 (40) |
| 15–30 d | 18 | 11/18 (61) | 5/18 (28) | 11/18 (61) | 18/18 (100) | 11/18 (61) | 8/18 (44) | 9/18 (50) |
| 31–90 d | 7 | 0/7 (0) | 0/7 (0) | 4/7 (57) | 6/7 (86) | 3/7 (43) | 3/7 (43) | 3/7 (43) |
| Total | 50 | 36/50 (72) | 15/50 (30) | 30/50 (60) | 49/50 (98) | 30/50 (60) | 22/50 (44) | 22/50 (44) |
| Time from Onset to Biopsy | n | P | C1q | C4 | C5 | C9 | S | MAC |
| 1–14 d | 25 | 23/25 (92%) | 7/25 (28) | 8/25 (32) | 25/25 (100) | 24/25 (96) | 24/25 (96) | 25/25 (100) |
| 15–30 d | 18 | 16/18 (89) | 6/18 (33) | 3/18 (17) | 18/18 (100) | 17/18 (94) | 17/18 (94) | 18/18 (100) |
| 31–90 d | 7 | 5/7 (71) | 2/7 (29) | 1/7 (14) | 6/7 (86) | 5/7 (71) | 5/7 (71) | 6/7 (86) |
| Total | 50 | 44/50 (88) | 15/50 (30) | 12/50 (24) | 49/50 (98) | 46/50 (92) | 46/50 (92) | 49/50 (98) |
| Biopsy Specimens | Onset to Biopsy | Glomerular NAPlr (+) | |
|---|---|---|---|
| PSAGN | 1–14 days | 25/25 | (100%) |
| 15–30 days | 11/18 | (61%) | |
| 31–90 days | 0/7 | (0%) | |
| Total | 36/50 | (72%) | |
| Non-PSAGN | 4/100 | (4%) | |
| Normal kidneys | 0/10 | (0%) | |
| Age in Years Range | Anti-NAPlr Antibody (+) | Anti-NAPlr Antibody Titers | |
|---|---|---|---|
| PSAGN | (5–72 y, mean 29.3) | 46/50 (92%) | 566 ± 106.1 |
| Streptococcal infection | (8–64 y, mean 29.0) | 30/50 (60%) | 227.1 ± 51.2 |
| Pediatric I | (0.2–10 y, mean 7.2) | 13/50 (26%) | 138.9 ± 23.4 |
| Pediatric II | (11–20 y, mean 14.1) | 18/50 (36%) | 166.0 ± 25.7 |
| Normal adults I | (25–35 y, mean 30.0) | 24/50 (48%) | 100.1 ± 18.0 |
| Normal adults II | (52–59 y, mean 53.0) | 36/50 (72%) | 186.0 ± 17.3 |
| Case | Age/ Gender | Diagnosis | Onset | UP | U-RBC (/hpf) | SCr (mg/dL) | CH50 (U/mL) | C3 (mg/dL) | C4 (mg/dL) | CH50 (U/mL) | EP | IF | NAPlr | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 14/F | DDD | AGN | +++ | +++ | 0.48 | <10 | 8 | - | 1090 | +++ | C3 cap | +++ | [71] |
| 2 | 12/M | DDD | AKI on CGN | +++ | 30–49 | 0.44 | 31.1 | 48 | 27 | 333 | +++ | C3 cap | +++ | [70] |
| 3 | 6/F | DDD | Asymptomatic | ± | 20–29 | 0.36 | 23.5 | 36 | 19 | 290 | ++ | C3 cap | + | our case |
| 4 | 62/F | MPGN I | AKI on CGN | +++ | 20–29 | 3.6 | 21.7 | - | - | 170 | ++ | IgG, IgM, C3 cap | ++ | [73] |
| 5 | 6/F | HSPN | Abd pain, purpura | +++ | many | 0.35 | 67.1 | 89 | 32 | 154 | ++ | IgA, C3 mes-cap | ++ | our case |
| 6 | 25/M | HSPN | Abd pain, purpura | + | 30–49 | 1.3 | 28 | 50 | 22 | 743 | +++ | IgA, C3 mes-cap | +++ | [76] |
| 7 | 71/M | SVV | Pain and swelling of the rt. ankle | +++ | 50–99 | 3.28 | - | 42 | 27 | 512 | +++ | C3 mes-cap | +++ | [75] |
| 8 | 17/M | GPA | AGN | ++ | 100< | 7.53 | - | 10 | 26 | 301 | − | C3 mes | + | [74] |
| Case | Age/ Gender | Infection Focus | Pathogen | Complement C3/C4/CH50 | NAPlr /PA | Light Microscopy | IF | EM (Dense Deposits) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 12/F | respiratory infection | Streptococcus pneumoniae | 10/33/- | +/+ | cellular crescent formation, endocapillary proliferative GN | C3 | GBM hump (−) | [77] |
| 2 | 64/M | endocarditis | Aggregatibacter actinomycetemcomitans | 86/16/- | +/− | proliferative GN with inflammatory cell infiltration | IgG IgM C3 C1q IgA | Subend Mes hump (−) | [78] |
| 3 | 7/F | respiratory infection | Mycoplasma pneumoniae | WNL | +/+ | endocapillary proliferation and cellular crescents | IgA IgM C3 | Mes Parames Subendo hump (−) | [79] |
| 4 | 70/M | cellulitis | Staphylococcus aureus (MSSA) | 118/43.9/75.3 | +/+ | endocapillary proliferative GN | IgG IgA IgM C3 C1q | GBM Parames hump (−) | Our case |
| Pathogen | Amino Acid Sequence of GAPDH | |||
|---|---|---|---|---|
| Total Amino Acid | Identity Similarity (%) | C Terminal Amino Acid | ||
| Streptococcal pyogenes | 336 | - | - | T L E Y F A K I A K |
| Streptococcus pneumoniae | 359 | 92 | 99 | T L E Y F A K I A K |
| Aggregatibacter actinomycetemcomitans | 334 | 50 | 85 | L V A H V Y N Y K D |
| Mycoplasma pneumoniae | 337 | 54 | 87 | V R V V N Y C A K L |
| Staphylococcus aureus (MSSA) | 336 | 67 | 92 | T L A Y L A E L S K |
| Case | Age/ Gender | Infection Focus | Underlying Disease | Pathogen | Complement C3/C4/CH50 | NAPlr/ PA | Light Microscopy | IF | EM (Dense Deposits) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 68/M | sinusitis | - | unknown | 25.1/20.8/<12 | +/+ | proliferative and exudative GN, MPGN type I | IgG C3 | Parames hump (−) | [89] |
| 6 | 17/F | unknown | C3G | unknown | 6/15/<12 | +/+ | lobulation of the glomerular capillary tufts, proliferation of glomerular mesangial cells, increased mesangial matrix, MPGN type III | C3 | Subendo Subepi | [85] |
| 7 | 55/M | unknown | CFH mutation | unknown | 108/43/61.6 | +/+ | cellular crescents and diffuse endocapillary and mesangial proliferation with double contour formation along the capillary walls | C3 IgG | Subepi Subendo hump (−) | [83] |
| 8 | 70/M | respiratory infection | - | unknown | 80/11.8/15.8 | +/+ | endocapillary proliferative GN | IgG C3 | Subepi hump (+) | [84] |
| 9 | 75/M | unknown | ANCA negative SVV | unknown | 105/26/52.7 | +/+ | cellular crescents/segmental endocapillary proliferation with neutrophils and tuft necrosis | C3 | Mes, GBM, Subepi, hump (+) | [86] |
| 10 | 82/M | unknown | - | unknown | 64/25/- | +/+ | subendothelial deposits including wire loop lesions, endocapillary proliferation, cellular crescents | IgA C3 IgG/IgM C1q/C4 | Subendo, Mes hump (−) | [88] |
| 11 | 27/M | unknown | IgA nephropathy | unknown | 18/23/- | +/+ | mesangial cell proliferation, increased mesangial matrix increasing, endocapillary proliferation | IgA C3 | Mes hump (−) | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshizawa, N.; Yamada, M.; Fujino, M.; Oda, T. Nephritis-Associated Plasmin Receptor (NAPlr): An Essential Inducer of C3-Dominant Glomerular Injury and a Potential Key Diagnostic Biomarker of Infection-Related Glomerulonephritis (IRGN). Int. J. Mol. Sci. 2022, 23, 9974. https://doi.org/10.3390/ijms23179974
Yoshizawa N, Yamada M, Fujino M, Oda T. Nephritis-Associated Plasmin Receptor (NAPlr): An Essential Inducer of C3-Dominant Glomerular Injury and a Potential Key Diagnostic Biomarker of Infection-Related Glomerulonephritis (IRGN). International Journal of Molecular Sciences. 2022; 23(17):9974. https://doi.org/10.3390/ijms23179974
Chicago/Turabian StyleYoshizawa, Nobuyuki, Muneharu Yamada, Masayuki Fujino, and Takashi Oda. 2022. "Nephritis-Associated Plasmin Receptor (NAPlr): An Essential Inducer of C3-Dominant Glomerular Injury and a Potential Key Diagnostic Biomarker of Infection-Related Glomerulonephritis (IRGN)" International Journal of Molecular Sciences 23, no. 17: 9974. https://doi.org/10.3390/ijms23179974
APA StyleYoshizawa, N., Yamada, M., Fujino, M., & Oda, T. (2022). Nephritis-Associated Plasmin Receptor (NAPlr): An Essential Inducer of C3-Dominant Glomerular Injury and a Potential Key Diagnostic Biomarker of Infection-Related Glomerulonephritis (IRGN). International Journal of Molecular Sciences, 23(17), 9974. https://doi.org/10.3390/ijms23179974






