Incretins as a Potential Treatment Option for Gestational Diabetes Mellitus
Abstract
:1. Introduction
2. Management of GDM
3. GLP-1—State of the Art
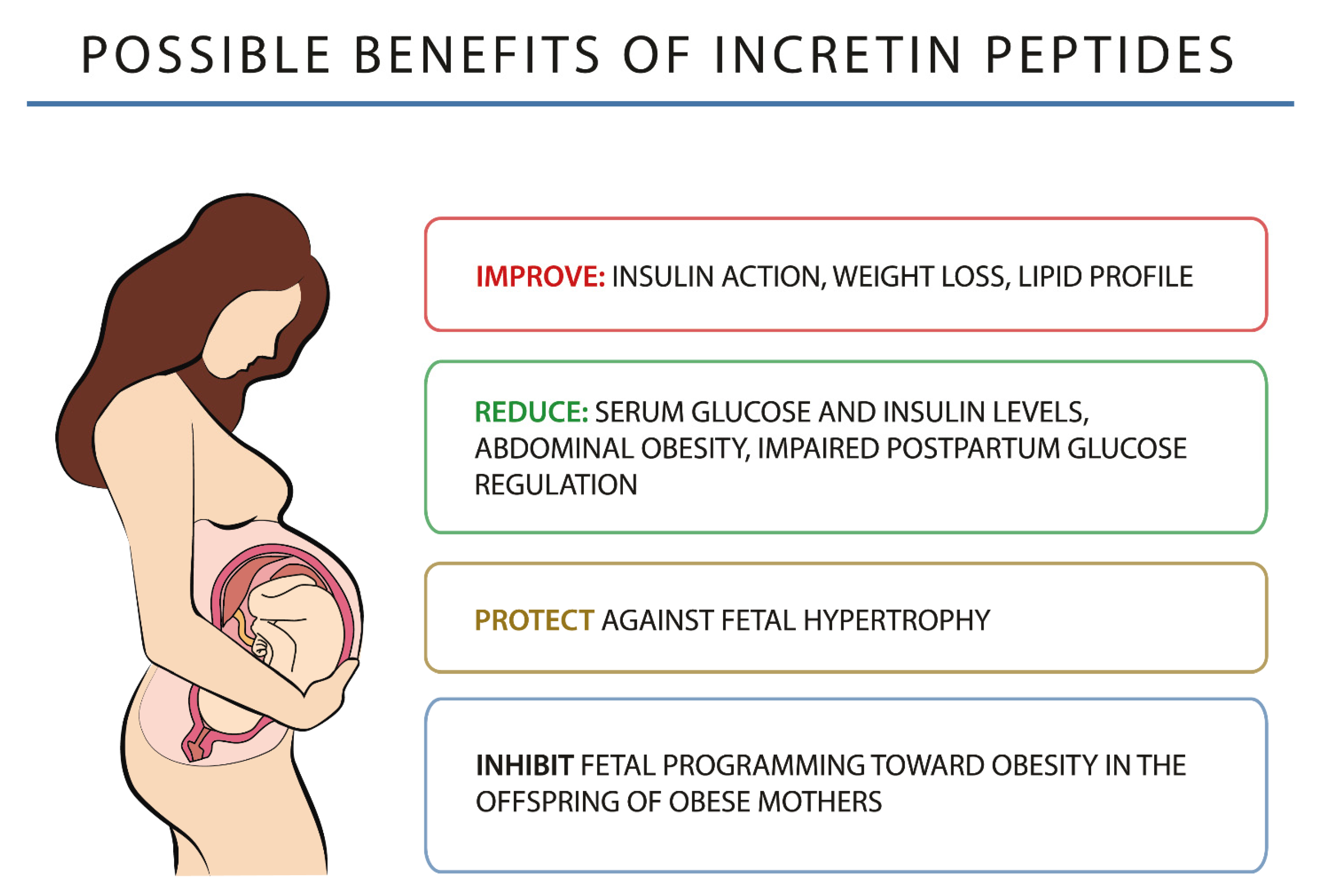
4. GLP-1 and Pregnancy
5. GLP-1R Agonists in GDM
6. DPP-4—State of the Art
7. DPP-4 in GDM
8. Potential Side Effects of the Use of Incretins in Pregnant Women and Their Offspring
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Plows, J.F.; Stanley, J.L.; Baker, P.N.; Reynolds, C.M.; Vickers, M.H. The Pathophysiology of Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2018, 19, 3342. [Google Scholar] [CrossRef]
- Fritsche, L.; Heni, M.; Eckstein, S.S.; Hummel, J.; Schürmann, A.; Häring, H.U.; Preißl, H.; Birkenfeld, A.L.; Peter, A.; Fritsche, A.; et al. Incretin hypersecretion in gestational diabetes mellitus. J. Clin. Endocrinol. Metab. 2022, 107, e2425–e2430. [Google Scholar] [CrossRef]
- Patro-Małysza, J.; Trojnar, M.; Kimber-Trojnar, Ż.; Mierzyński, R.; Bartosiewicz, J.; Oleszczuk, J.; Leszczyńska-Gorzelak, B. FABP4 in Gestational Diabetes-Association between Mothers and Offspring. J. Clin. Med. 2019, 8, 285. [Google Scholar] [CrossRef]
- Zhang, C.; Tobias, D.K.; Chavarro, J.E.; Bao, W.; Wang, D.; Ley, S.H.; Hu, F.B. Adherence to healthy lifestyle and risk of gestational diabetes mellitus: Prospective cohort study. BMJ 2014, 349, g5450. [Google Scholar] [CrossRef]
- You, H.; Hu, J.; Liu, Y.; Luo, B.; Lei, A. Risk of type 2 diabetes mellitus after gestational diabetes mellitus: A systematic review & meta-analysis. Indian J. Med. Res. 2021, 154, 62–77. [Google Scholar]
- Gascho, C.L.L.; Leandro, D.M.K.; Ribeiro, E.; Silva, T.; Silva, J.C. Predictors of cesarean delivery in pregnant women with gestational diabetes mellitus. Rev. Bras. Ginecol. Obstet. 2017, 39, 60–65. [Google Scholar] [CrossRef]
- Shostrom, D.C.V.; Sun, Y.; Oleson, J.J.; Snetselaar, L.G.; Bao, W. History of Gestational Diabetes Mellitus in Relation to Cardiovascular Disease and Cardiovascular Risk Factors in US Women. Front. Endocrinol. 2017, 26, 8–144. [Google Scholar] [CrossRef]
- Marciniak, A.; Patro-Małysza, J.; Kimber-Trojnar, Ż.; Marciniak, B.; Oleszczuk, J.; Leszczyńska-Gorzelak, B. Fetal programming of the metabolic syndrome. Taiwan J. Obstet. Gynecol. 2017, 56, 133–138. [Google Scholar] [CrossRef]
- Kimber-Trojnar, Ż.; Patro-Małysza, J.; Trojnar, M.; Skórzyńska-Dziduszko, K.E.; Bartosiewicz, J.; Oleszczuk, J.; Leszczyńska-Gorzelak, B. Fatty Acid-Binding Protein 4-An “Inauspicious” Adipokine-In Serum and Urine of Post-Partum Women with Excessive Gestational Weight Gain and Gestational Diabetes Mellitus. J. Clin. Med. 2018, 7, 505. [Google Scholar] [CrossRef] [PubMed]
- Dearden, L.; Bouret, S.G.; Ozanne, S.E. Nutritional and developmental programming effects of insulin. J. Neuroendocrinol. 2021, 33, e12933. [Google Scholar] [CrossRef]
- Nielsen, J.H. Beta cell adaptation in pregnancy: A tribute to Claes Hellerström. Ups. J. Med. Sci. 2016, 121, 151–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruszała, M.; Pilszyk, A.; Niebrzydowska, M.; Kimber-Trojnar, Ż.; Trojnar, M.; Leszczyńska-Gorzelak, B. Novel Biomolecules in the Pathogenesis of Gestational Diabetes Mellitus 2.0. Int. J. Mol. Sci. 2022, 23, 4364. [Google Scholar] [CrossRef]
- Ruszała, M.; Niebrzydowska, M.; Pilszyk, A.; Kimber-Trojnar, Ż.; Trojnar, M.; Leszczyńska-Gorzelak, B. Novel Biomolecules in the Pathogenesis of Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2021, 22, 11578. [Google Scholar] [CrossRef] [PubMed]
- Barbour, L.A.; McCurdy, C.E.; Hernandez, T.L.; Kirwan, J.P.; Catalano, P.M.; Friedman, J.E. Cellular mechanisms for insulin resistance in normal pregnancy and gestational diabetes. Diabetes Care 2007, 30, S112–S119. [Google Scholar] [CrossRef]
- Alfadhli, E.M. Gestational diabetes mellitus. Saudi Med. J. 2015, 36, 399–406. [Google Scholar] [CrossRef]
- Hill, D.J. Placental control of metabolic adaptations in the mother for an optimal pregnancy outcome. What goes wrong in gestational diabetes? Placenta 2018, 69, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Skórzyńska-Dziduszko, K.E.; Kimber-Trojnar, Ż.; Patro-Małysza, J.; Olszewska, A.; Zaborowski, T.; Małecka-Massalska, T. An Interplay between Obesity and Inflammation in Gestational Diabetes Mellitus. Curr. Pharm. Biotechnol. 2016, 17, 603–613. [Google Scholar] [CrossRef]
- Friedrichsen, B.N.; Neubauer, N.; Lee, Y.C.; Gram, V.K.; Blume, N.; Petersen, J.S.; Nielsen, J.H.; Møldrup, A. Stimulation of pancreatic beta-cell replication by incretins involves transcriptional induction of cyclin D1 via multiple signalling pathways. J. Endocrinol. 2006, 188, 481–492. [Google Scholar] [CrossRef]
- Mosavat, M.; Omar, S.Z.; Jamalpour, S.; Tan, P.C. Serum Glucose-Dependent Insulinotropic Polypeptide (GIP) and Glucagon-Like Peptide-1 (GLP-1) in association with the Risk of Gestational Diabetes: A Prospective Case-Control Study. J. Diabetes Res. 2020, 29, 9072492. [Google Scholar] [CrossRef]
- Johns, E.C.; Denison, F.C.; Norman, J.E.; Reynolds, R.M. Gestational Diabetes Mellitus: Mechanisms, Treatment, and Complications. Trends Endocrinol. Metab. 2018, 29, 743–754. [Google Scholar] [CrossRef]
- Lende, M.; Rijhsinghani, A. Gestational Diabetes: Overview with Emphasis on Medical Management. Int. J. Environ. Res. Public Health 2020, 17, 9573. [Google Scholar] [CrossRef] [PubMed]
- Schoonejans, J.M.; Blackmore, H.L.; Ashmore, T.J.; Aiken, C.E.; Fernandez-Twinn, D.S.; Ozanne, S.E. Maternal Metformin Intervention during Obese Glucose-Intolerant Pregnancy Affects Adiposity in Young Adult Mouse Offspring in a Sex-Specific Manner. Int. J. Mol. Sci. 2021, 22, 8104. [Google Scholar] [CrossRef]
- Kimber-Trojnar, Ż.; Dłuski, D.F.; Wierzchowska-Opoka, M.; Ruszała, M.; Leszczyńska-Gorzelak, B. Metformin as a Potential Treatment Option for Endometriosis. Cancers 2022, 14, 577. [Google Scholar] [CrossRef] [PubMed]
- Kimber-Trojnar, Z.; Marciniak, B.; Leszczyńska-Gorzelak, B.; Trojnar, M.; Oleszczuk, J. Glyburide for the treatment of gestational diabetes mellitus. Pharmacol. Rep. 2008, 60, 308–318. [Google Scholar] [PubMed]
- Kimber-Trojnar, Ż.; Marciniak, B.; Patro-Malysza, J.; Skorzynska-Dziduszko, K.; Poniedzialek-Czajkowska, E.; Mierzynski, R.; Galczynski, K.; Trojnar, M.; Leszczynska-Gorzelak, B.; Oleszczuk, J. Is glyburide safe in pregnancy? Curr. Pharm. Biotechnol. 2014, 15, 100–112. [Google Scholar] [CrossRef]
- Drucker, D.J.; Habener, J.F.; Holst, J.J. Discovery, characterization, and clinical development of the glucagon-like peptides. J. Clin. Investig. 2017, 127, 4217–4227. [Google Scholar] [CrossRef]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 receptor agonists in the treatment of type 2 diabetes—State-of-the-art. Mol. Metab. 2021, 46, 101102. [Google Scholar] [CrossRef]
- Drucker, D.J. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metab. 2018, 27, 740–756. [Google Scholar] [CrossRef]
- Maselli, D.B.; Camilleri, M. Effects of GLP-1 and Its Analogs on Gastric Physiology in Diabetes Mellitus and Obesity. Adv. Exp. Med. Biol. 2021, 1307, 171–192. [Google Scholar]
- D’Alessio, D. Is GLP-1 a hormone: Whether and When? J. Diabetes Investig. 2016, 7 (Suppl. S1), 50–55. [Google Scholar] [CrossRef]
- Meier, J.J. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2012, 8, 728–742. [Google Scholar] [CrossRef] [PubMed]
- Smith, N.K.; Hackett, T.A.; Galli, A.; Flynn, C.R. GLP-1: Molecular mechanisms and outcomes of a complex signaling system. Neurochem. Int. 2019, 128, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Drucker, D.J.; Nauck, M.A. The incretin system: Glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006, 368, 1696–1705. [Google Scholar] [CrossRef]
- Jansen, T.J.P.; van Lith, S.A.M.; Boss, M.; Brom, M.; Joosten, L.; Béhé, M.; Buitinga, M.; Gotthardt, M. Exendin-4 analogs in insulinoma theranostics. J. Labelled Comp. Radiopharm. 2019, 62, 656–672. [Google Scholar] [CrossRef]
- Patro-Malysza, J.; Kimber-Trojnar, Z.; Skorzynska-Dziduszko, K.; Marciniak, B.; Darmochwal-Kolarz, D.; Bartosiewicz, J.; Leszczynska-Gorzelak, B.; Oleszczuk, J. The impact of substance P on the pathogenesis of insulin resistance leading to gestational diabetes. Curr. Pharm. Biotechnol. 2014, 15, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Mayer, F.; Gunawan, A.L.; Tso, P.; Aponte, G.W. Glucagon-like peptide 1 and glucose-dependent insulinotropic polypeptide stimulate release of substance P from TRPV1- and TRPA1-expressing sensory nerves. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 319, G23–G35. [Google Scholar] [CrossRef] [PubMed]
- Baggio, L.L.; Drucker, D.J. Glucagon-like peptide-1 receptor co-agonists for treating metabolic disease. Mol. Metab. 2021, 46, 101090. [Google Scholar] [CrossRef]
- Valsamakis, G.; Margeli, A.; Vitoratos, N.; Boutsiadis, A.; Sakkas, E.G.; Papadimitriou, G.; Al-Daghri, N.M.; Botsis, D.; Kumar, S.; Papassotiriou, I.; et al. The role of maternal gut hormones in normal pregnancy: Fasting plasma active glucagon-like peptide 1 level is a negative predictor of fetal abdomen circumference and maternal weight change. Eur. J. Endocrinol. 2010, 162, 897–903. [Google Scholar] [CrossRef]
- Kimber-Trojnar, Ż.; Patro-Małysza, J.; Skórzyńska-Dziduszko, K.E.; Oleszczuk, J.; Trojnar, M.; Mierzyński, R.; Leszczyńska-Gorzelak, B. Ghrelin in Serum and Urine of Post-Partum Women with Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2018, 19, 3001. [Google Scholar] [CrossRef]
- Bonde, L.; Vilsbøll, T.; Nielsen, T.; Bagger, J.I.; Svare, J.A.; Holst, J.J.; Larsen, S.; Knop, F.K. Reduced postprandial GLP-1 responses in women with gestational diabetes mellitus. Diabetes Obes. Metab. 2013, 15, 713–720. [Google Scholar] [CrossRef]
- Kosinski, M.; Knop, F.K.; Vedtofte, L.; Grycewiczv, J.; Swierzewska, P.; Cypryk, K.; Vilsbøll, T. Postpartum reversibility of impaired incretin effect in gestational diabetes mellitus. Regul. Pept. 2013, 186, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Elkind-Hirsch, K.E.; Shaler, D.; Harris, R. Postpartum treatment with liraglutide in combination with metformin versus metformin monotherapy to improve metabolic status and reduce body weight in overweight/obese women with recent gestational diabetes: A double-blind, randomized, placebo-controlled study. J. Diabetes Complicat. 2020, 34, 107548. [Google Scholar]
- Foghsgaard, S.; Andreasen, C.; Vedtofte, L.; Andersen, E.S.; Bahne, E.; Strandberg, C.; Buhl, T.; Holst, J.J.; Svare, J.A.; Clausen, T.D.; et al. Nonalcoholic fatty liver disease is prevalent in women with prior gestational diabetes mellitus and independently associated with insulin resistance and waist circumference. Diabetes Care 2017, 40, 109–116. [Google Scholar] [CrossRef] [Green Version]
- Vedtofte, L.; Bahne, E.; Foghsgaard, S.; Bagger, J.I.; Andreasen, C.; Strandberg, C.; Gørtz, P.M.; Holst, J.J.; Grønbæk, H.; Svare, J.A.; et al. One Year’s Treatment with the Glucagon-Like Peptide 1 Receptor Agonist Liraglutide Decreases Hepatic Fat Content in Women with Nonalcoholic Fatty Liver Disease and Prior Gestational Diabetes Mellitus in a Randomized, Placebo-Controlled Trial. J. Clin. Med. 2020, 9, 3213. [Google Scholar] [CrossRef] [PubMed]
- Foghsgaard, S.; Vedtofte, L.; Bahne, E.; Andreasen, C.; Mathiesen, E.R.; Svare, J.A.; Christiansen, L.K.; Holst, J.J.; Clausen, T.D.; Damm, P.; et al. Intervention with a glucagon-like peptide-1 receptor agonist improves glycaemic control in women with prior gestational diabetes: A randomised, placebo-controlled trial. Diabetologia 2016, 59, S386. [Google Scholar]
- Shao, S.; Xu, Q.; Yu, X.; Pan, R.; Chen, Y. Dipeptidyl peptidase 4 inhibitors and their potential immune modulatory functions. Pharmacol. Ther. 2020, 209, 107503. [Google Scholar] [CrossRef]
- Deacon, C.F. Physiology and Pharmacology of DPP-4 in Glucose Homeostasis and the Treatment of Type 2 Diabetes. Front. Endocrinol. 2019, 10, 80. [Google Scholar] [CrossRef]
- Montaniel, K.R.C.; Bucher, M.; Phillips, E.A.; Li, C.; Sullivan, E.L.; Kievit, P.; Rugonyi, S.; Nathanielsz, P.W.; Maloyan, A. Dipeptidyl peptidase IV inhibition delays developmental programming of obesity and metabolic disease in male offspring of obese mothers. J. Dev. Orig. Health Dis. 2022, 24, 1–14. [Google Scholar] [CrossRef]
- Kumar, S.; Mittal, A.; Mittal, A. A review upon medicinal perspective and designing rationale of DPP-4 inhibitors. Bioorg. Med. Chem. 2021, 46, 116354. [Google Scholar] [CrossRef]
- Singh, A.K.; Yadav, D.; Sharma, N.; Jin, J.O. Dipeptidyl Peptidase (DPP)-IV Inhibitors with Antioxidant Potential Isolated from Natural Sources: A Novel Approach for the Management of Diabetes. Pharmaceuticals 2021, 14, 586. [Google Scholar] [CrossRef]
- Sesti, G.; Avogaro, A.; Belcastro, S.; Bonora, B.M.; Croci, M.; Daniele, G.; Dauriz, M.; Dotta, F.; Formichi, C.; Frontoni, S.; et al. Ten years of experience with DPP-4 inhibitors for the treatment of type 2 diabetes mellitus. Acta Diabetol. 2019, 56, 605–617. [Google Scholar] [CrossRef]
- Cao, F.; Wu, K.; Zhu, Y.Z.; Bao, Z.W. Roles and Mechanisms of Dipeptidyl Peptidase 4 Inhibitors in Vascular Aging. Front. Endocrinol. 2021, 12, 731273. [Google Scholar] [CrossRef] [PubMed]
- Florentin, M.; Kostapanos, M.S.; Papazafiropoulou, A.K. Role of dipeptidyl peptidase 4 inhibitors in the new era of antidiabetic treatment. World J. Diabetes 2022, 13, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Yin, R.; Xu, Y.; Wang, X.; Yang, L.; Zhao, D. Role of Dipeptidyl Peptidase 4 Inhibitors in Antidiabetic Treatment. Molecules 2022, 27, 3055. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.P.; Pratley, R.E. GLP-1 Analogs and DPP-4 Inhibitors in Type 2 Diabetes Therapy: Review of Head-to-Head Clinical Trials. Front. Endocrinol. 2020, 11, 178. [Google Scholar] [CrossRef]
- Lee, S.; Lee, H.; Kim, Y.; Kim, E. Effect of DPP-IV Inhibitors on Glycemic Variability in Patients with T2DM: A Systematic Review and Meta-Analysis. Sci. Rep. 2019, 19, 13296. [Google Scholar] [CrossRef]
- Scheen, A.J. DPP-4 inhibitors in the management of type 2 diabetes: A critical review of head-to-head trials. Diabetes Metab. 2012, 38, 89–101. [Google Scholar] [CrossRef]
- Hoffmann-Petersen, I.T.; Holt, C.B.; Jensen, L.; Hage, C.; Mellbin, L.G.; Thiel, S.; Hansen, T.K.; Østergaard, J.A. Effect of dipeptidyl peptidase-4 inhibitors on complement activation. Diabetes Metab. Res. Rev. 2021, 37, e3385. [Google Scholar] [CrossRef]
- Deng, X.; Tavallaie, M.S.; Sun, R.; Wang, J.; Cai, Q.; Shen, J.; Lei, S.; Fu, L.; Jiang, F. Drug discovery approaches targeting the incretin pathway. Bioorg. Chem. 2020, 99, 103810. [Google Scholar] [CrossRef]
- Al-Aissa, Z.; Rosta, K.; Hadarits, O.; Harreiter, J.; Zóka, A.; Bancher-Todesca, D.; Patócs, A.; Kiss, K.; Sármán, B.; Pusztai, P.; et al. Cord serum dipeptidyl-peptidase 4 activity in gestational diabetes. Eur. J. Clin. Investig. 2015, 45, 196–203. [Google Scholar] [CrossRef]
- Liu, B.; Deng, S.; Xu, Y.; Yang, J.; Geng, H.; Wang, Z. Association between maternal and umbilical cord serum dipeptidyl peptidase IV in pregnant women with and without gestational diabetes mellitus. J. Obstet. Gynaecol. Res. 2016, 42, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Kandzija, N.; Zhang, W.; Motta-Mejia, C.; Mhlomi, V.; McGowan-Downey, J.; James, T.; Cerdeira, A.S.; Tannetta, D.; Sargent, I.; Redman, C.W.; et al. Placental extracellular vesicles express active dipeptidyl peptidase IV; levels are increased in gestational diabetes mellitus. J. Extracell. Vesicles 2019, 8, 1617000. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, Z.; Ning, H.; Sun, H.; Ji, X. Sitagliptin down-regulates retinol-binding protein 4 and reduces insulin resistance in gestational diabetes mellitus: A randomized and double-blind trial. Metab. Brain Dis. 2017, 32, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Hummel, S.; Beyerlein, A.; Pfirrmann, M.; Hofelich, A.; Much, D.; Hivner, S.; Bunk, M.; Herbst, M.; Peplow, C.; Walter, M.; et al. Efficacy of vildagliptin for prevention of postpartum diabetes in women with a recent history of insulin-requiring gestational diabetes: A phase II, randomized, double-blind, placebo-controlled study. Mol. Metab. 2018, 9, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Elkind-Hirsch, K.E.; Paterson, M.S.; Shaler, D.; Gutowski, H.C. Short-Term Sitagliptin-Metformin Therapy Is More Effective Than Metformin or Placebo in Prior Gestational Diabetic Women with Impaired Glucose Regulation. Endocr. Pract. 2018, 24, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Chia, C.W.; Egan, J.M. Incretins in obesity and diabetes. Ann. N. Y. Acad. Sci. 2019, 1461, 104–126. [Google Scholar] [CrossRef] [PubMed]
- Borner, T.; Shaulson, E.D.; Tinsley, I.C.; Stein, L.M.; Horn, C.C.; Hayes, M.R.; Doyle, R.P.; De Jonghe, B.C. A second-generation glucagon-like peptide-1 receptor agonist mitigates vomiting and anorexia while retaining glucoregulatory potency in lean diabetic and emetic mammalian models. Diabetes Obes. Metab. 2020, 22, 1729–1741. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wang, M.; Wen, Z.; Lu, Z.; Cui, L.; Fu, C.; Xue, H.; Liu, Y.; Zhang, Y. GLP-1 Receptor Agonists: Beyond Their Pancreatic Effects. Front. Endocrinol. 2021, 12, 721135. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilszyk, A.; Niebrzydowska, M.; Pilszyk, Z.; Wierzchowska-Opoka, M.; Kimber-Trojnar, Ż. Incretins as a Potential Treatment Option for Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2022, 23, 10101. https://doi.org/10.3390/ijms231710101
Pilszyk A, Niebrzydowska M, Pilszyk Z, Wierzchowska-Opoka M, Kimber-Trojnar Ż. Incretins as a Potential Treatment Option for Gestational Diabetes Mellitus. International Journal of Molecular Sciences. 2022; 23(17):10101. https://doi.org/10.3390/ijms231710101
Chicago/Turabian StylePilszyk, Aleksandra, Magdalena Niebrzydowska, Zuzanna Pilszyk, Magdalena Wierzchowska-Opoka, and Żaneta Kimber-Trojnar. 2022. "Incretins as a Potential Treatment Option for Gestational Diabetes Mellitus" International Journal of Molecular Sciences 23, no. 17: 10101. https://doi.org/10.3390/ijms231710101
APA StylePilszyk, A., Niebrzydowska, M., Pilszyk, Z., Wierzchowska-Opoka, M., & Kimber-Trojnar, Ż. (2022). Incretins as a Potential Treatment Option for Gestational Diabetes Mellitus. International Journal of Molecular Sciences, 23(17), 10101. https://doi.org/10.3390/ijms231710101







