Hazard Assessment of the Effects of Acute and Chronic Exposure to Permethrin, Copper Hydroxide, Acephate, and Validamycin Nanopesticides on the Physiology of Drosophila: Novel Insights into the Cellular Internalization and Biological Effects
Abstract
:1. Introduction
2. Results
2.1. Characterization of Permethrin Nanopesticides, Cu(OH)2 Nanopesticides, Acephate Nanopesticides, and Validamycin Nanopesticides
2.2. The Endotoxin Levels of Permethrin Nanopesticides, Cu(OH)2 Nanopesticides, Acephate Nanopesticides, and Validamycin Nanopesticides
2.3. Determination of the LC50 and Mortality Values of Nanopesticides
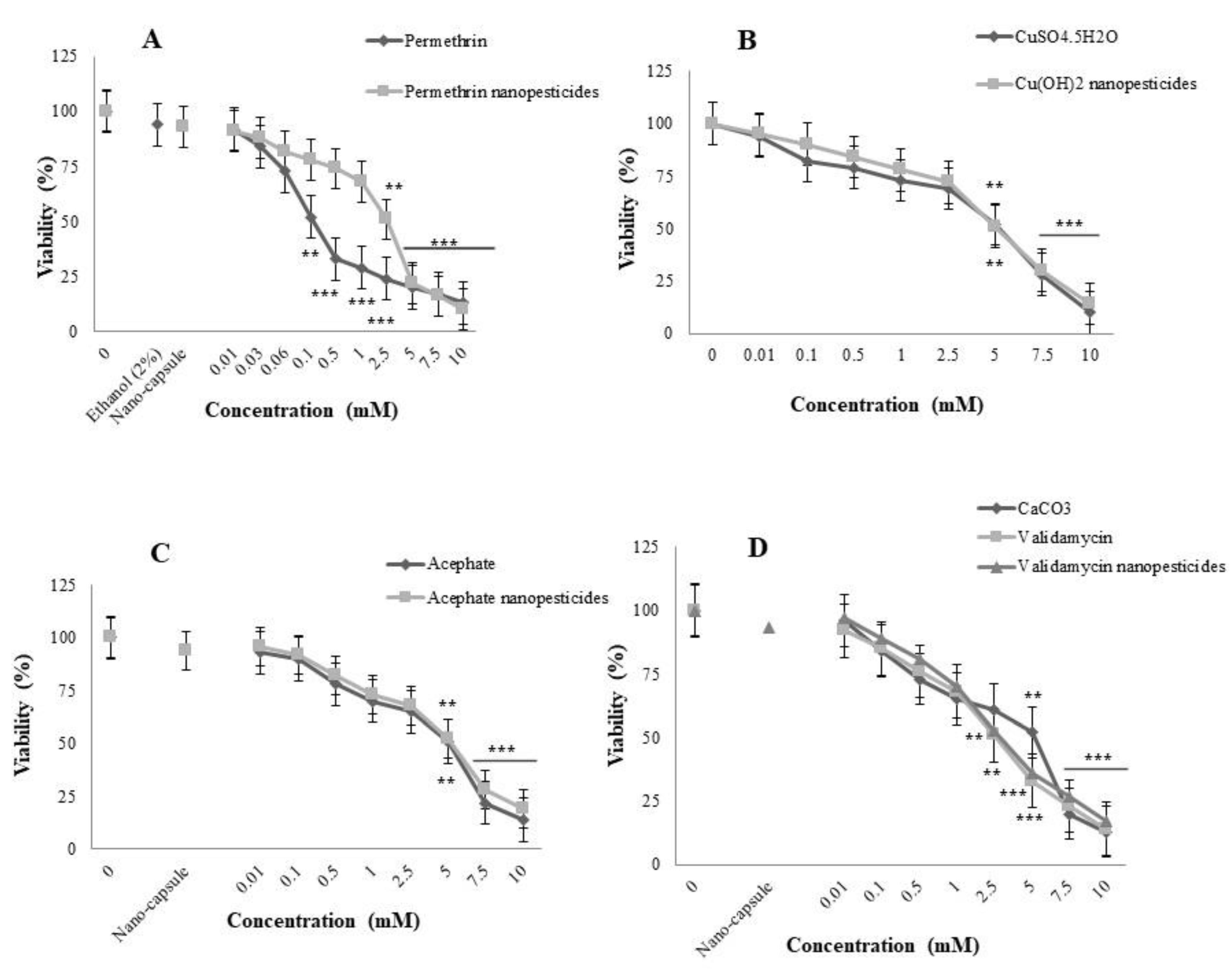
2.4. Toxicity
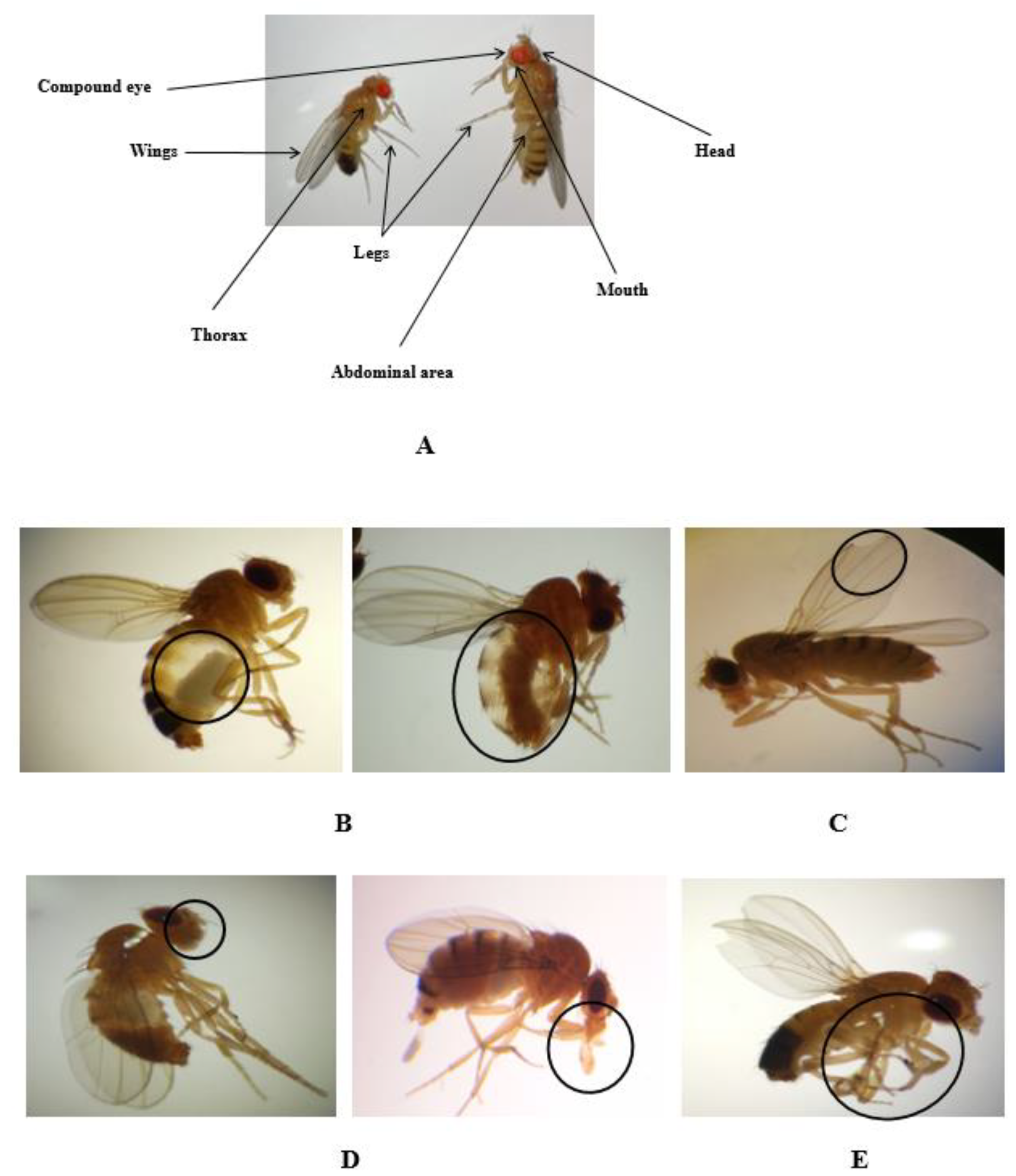
2.5. Morphological Alterations
2.6. Phenotypic Variations
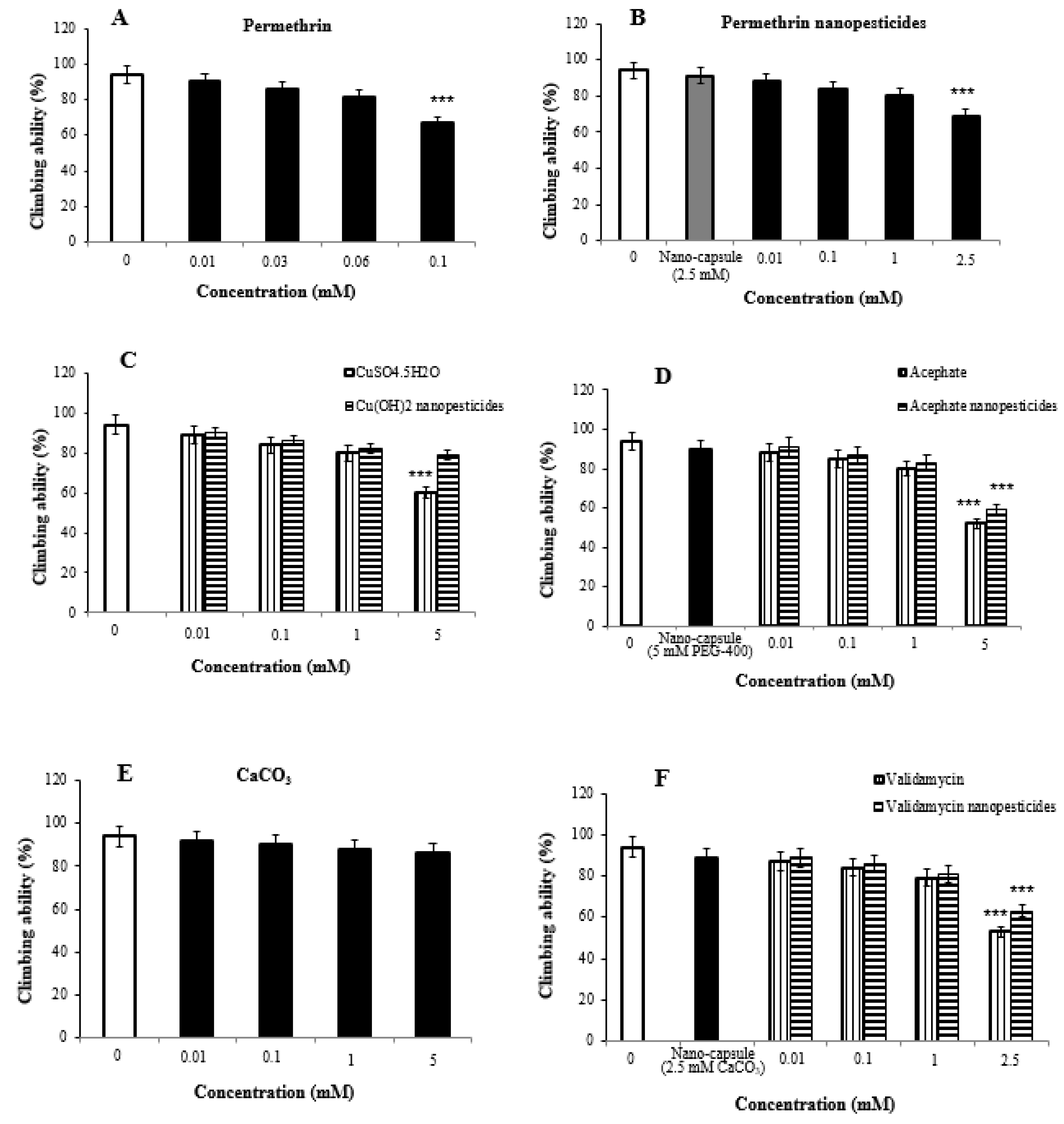
2.7. Impact of Nanopesticides on Climbing Behavior in Flies
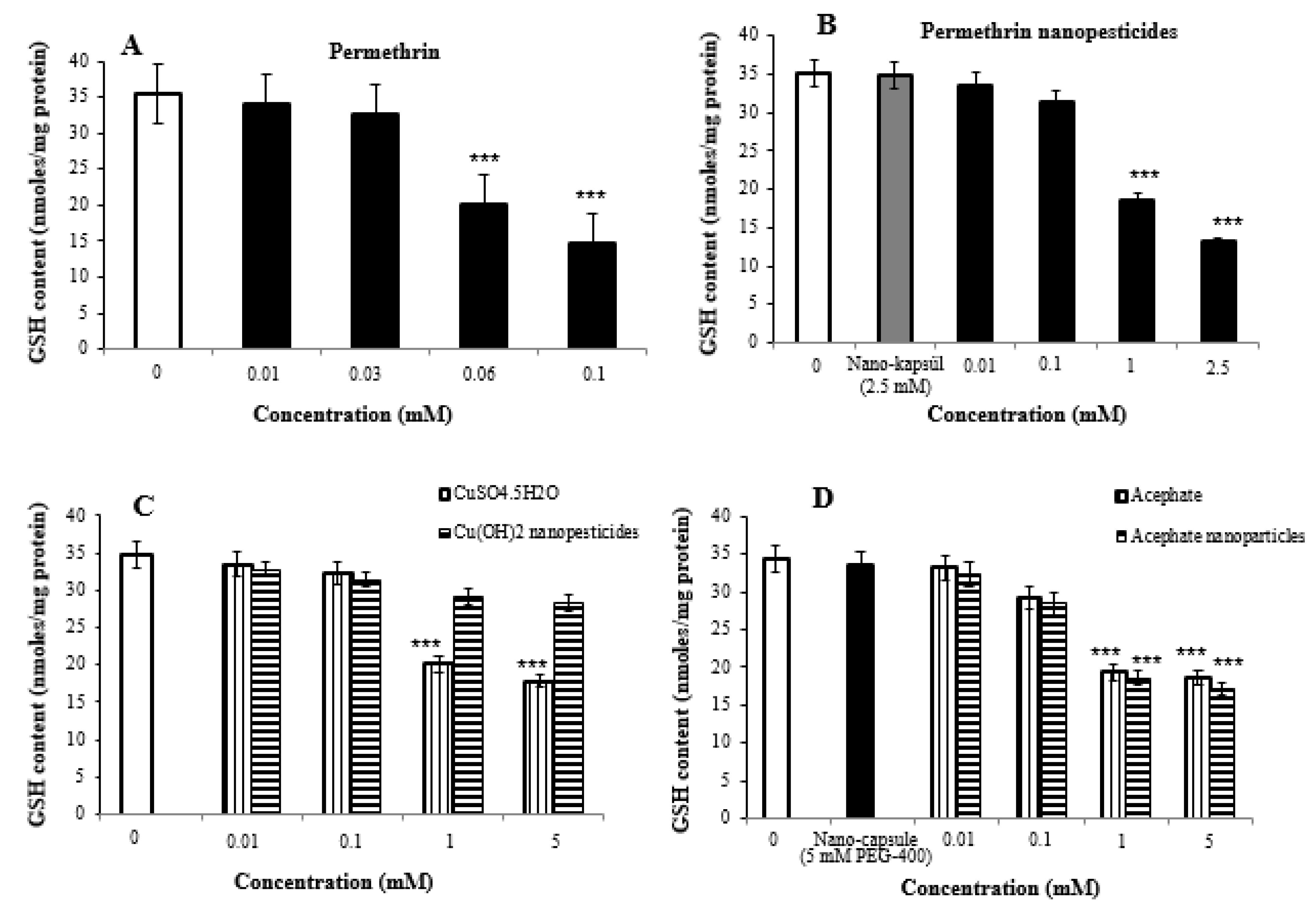
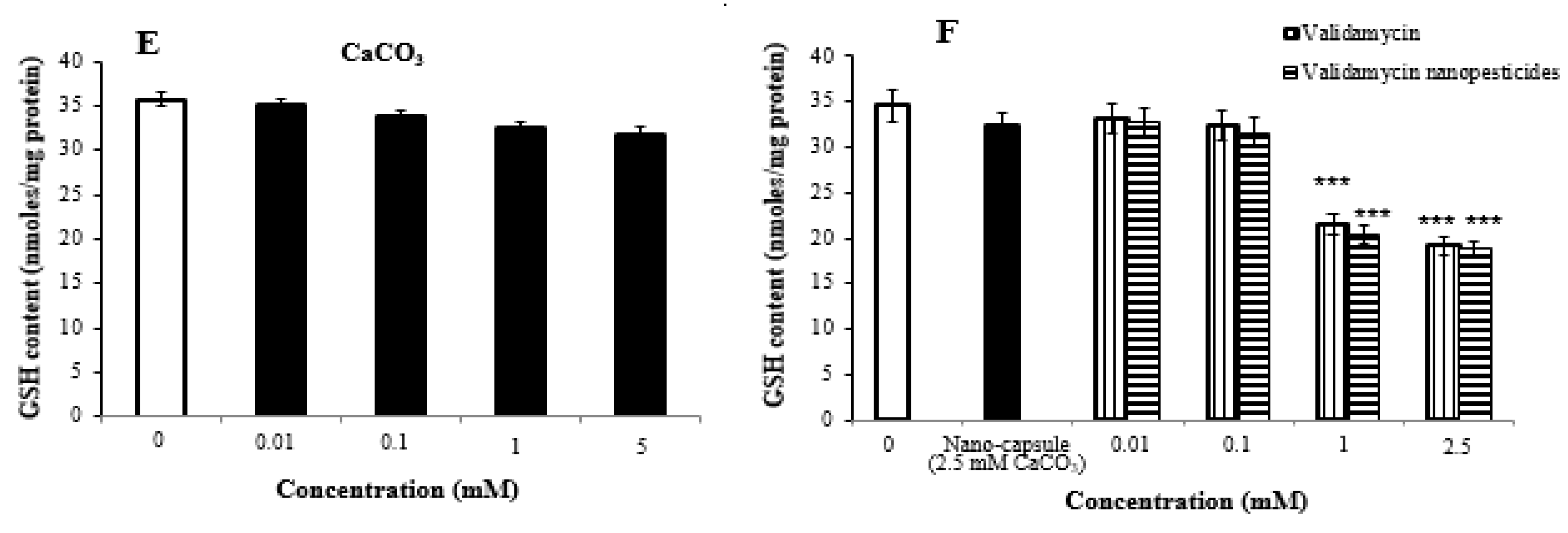
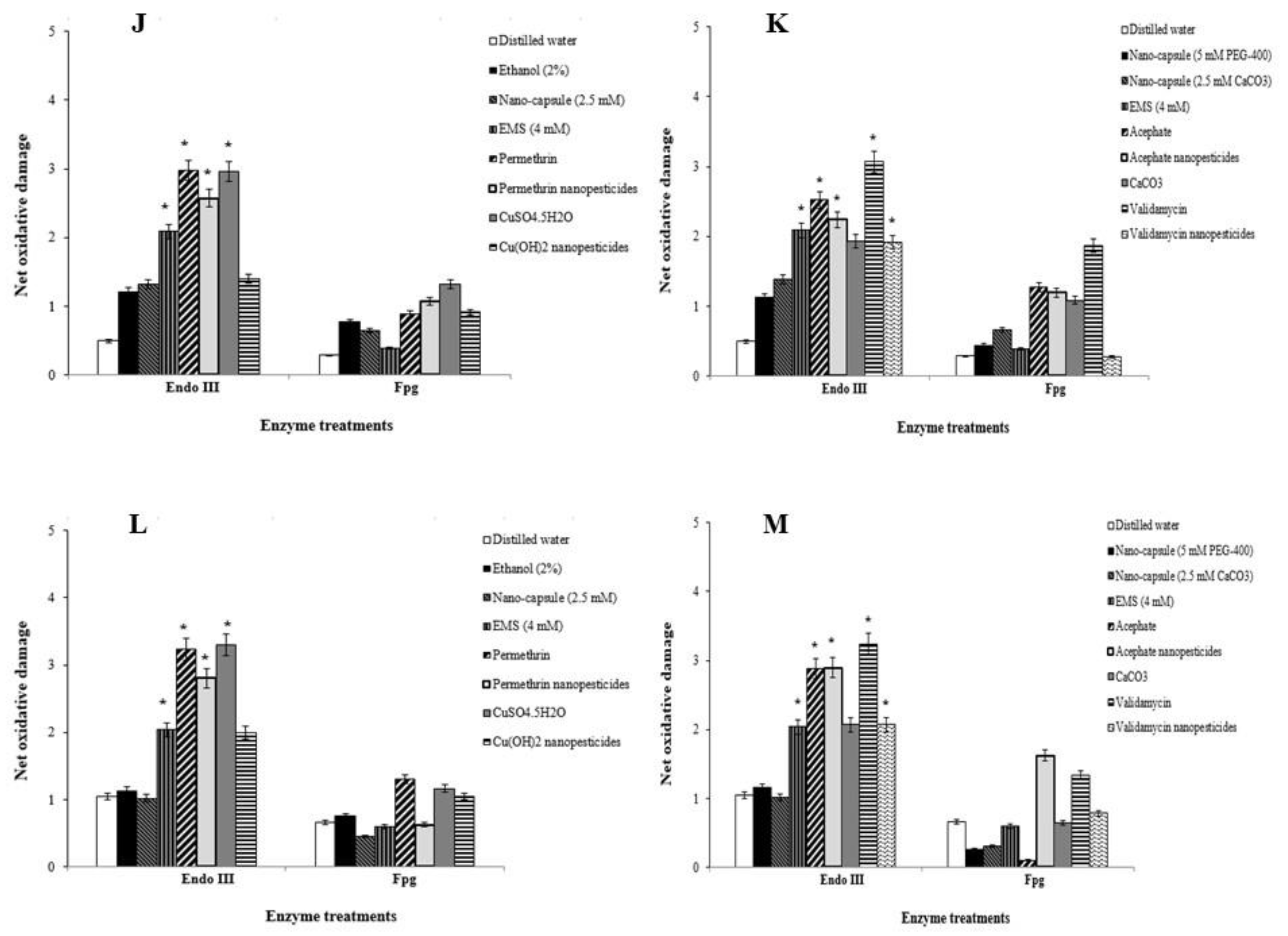
2.8. Oxidative Stress Assay
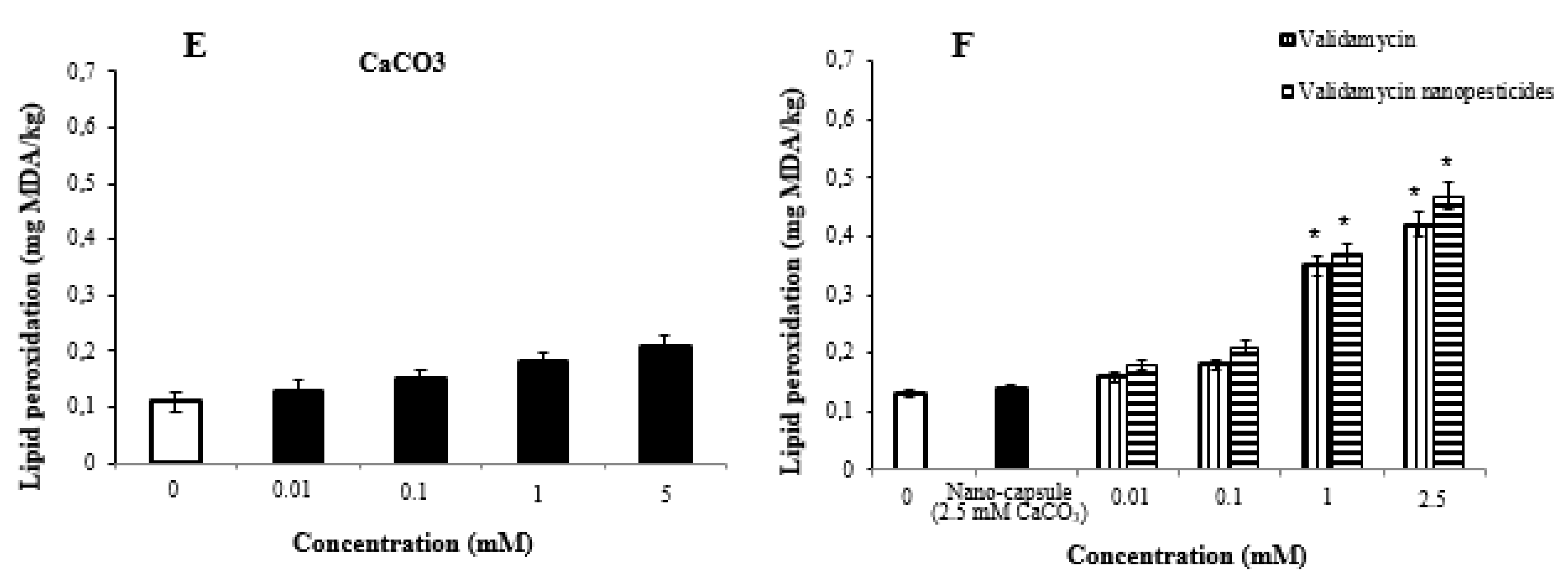
2.9. Lipid Peroxidation Assay
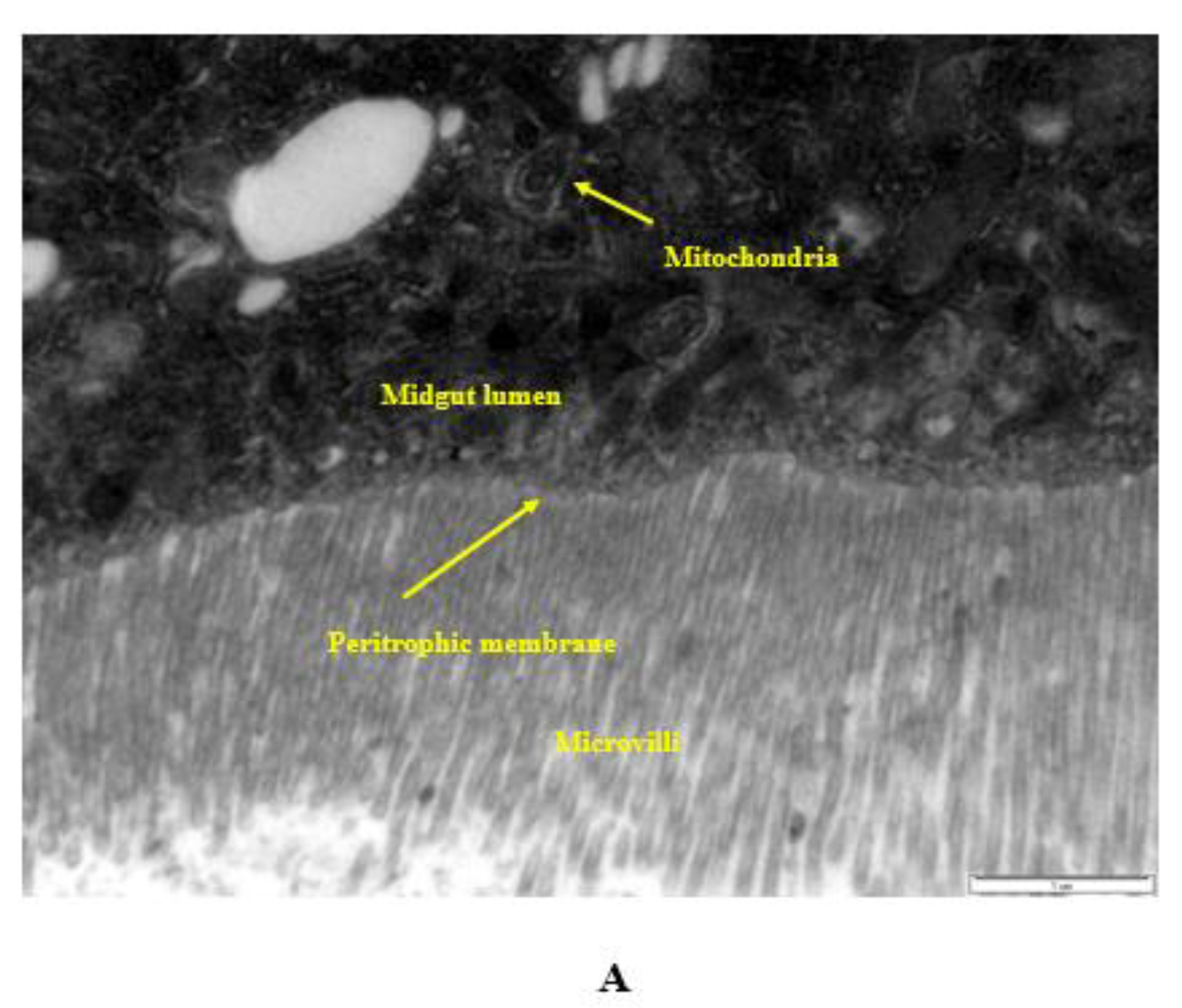
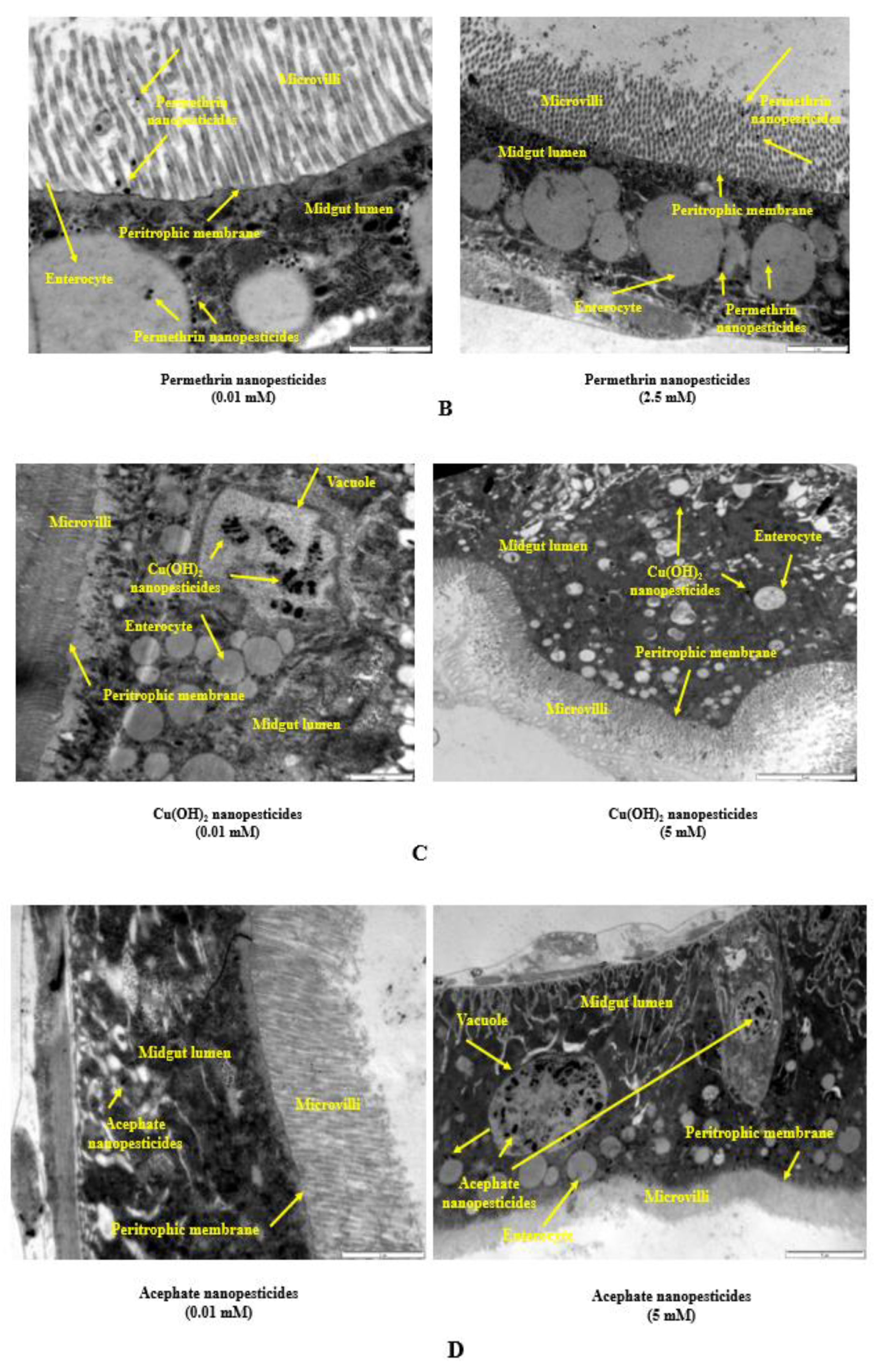
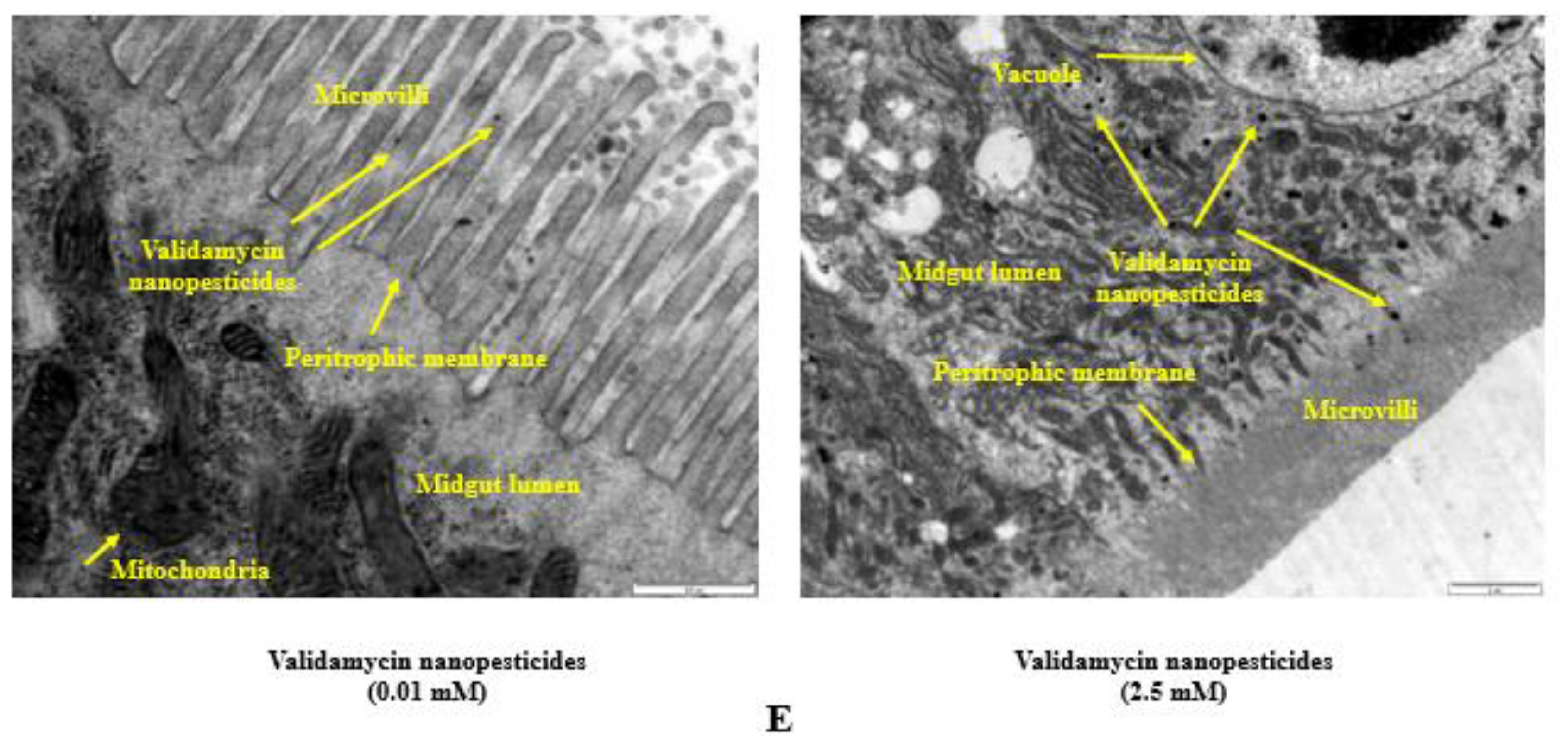
2.10. Internalization via the Intestinal Barrier
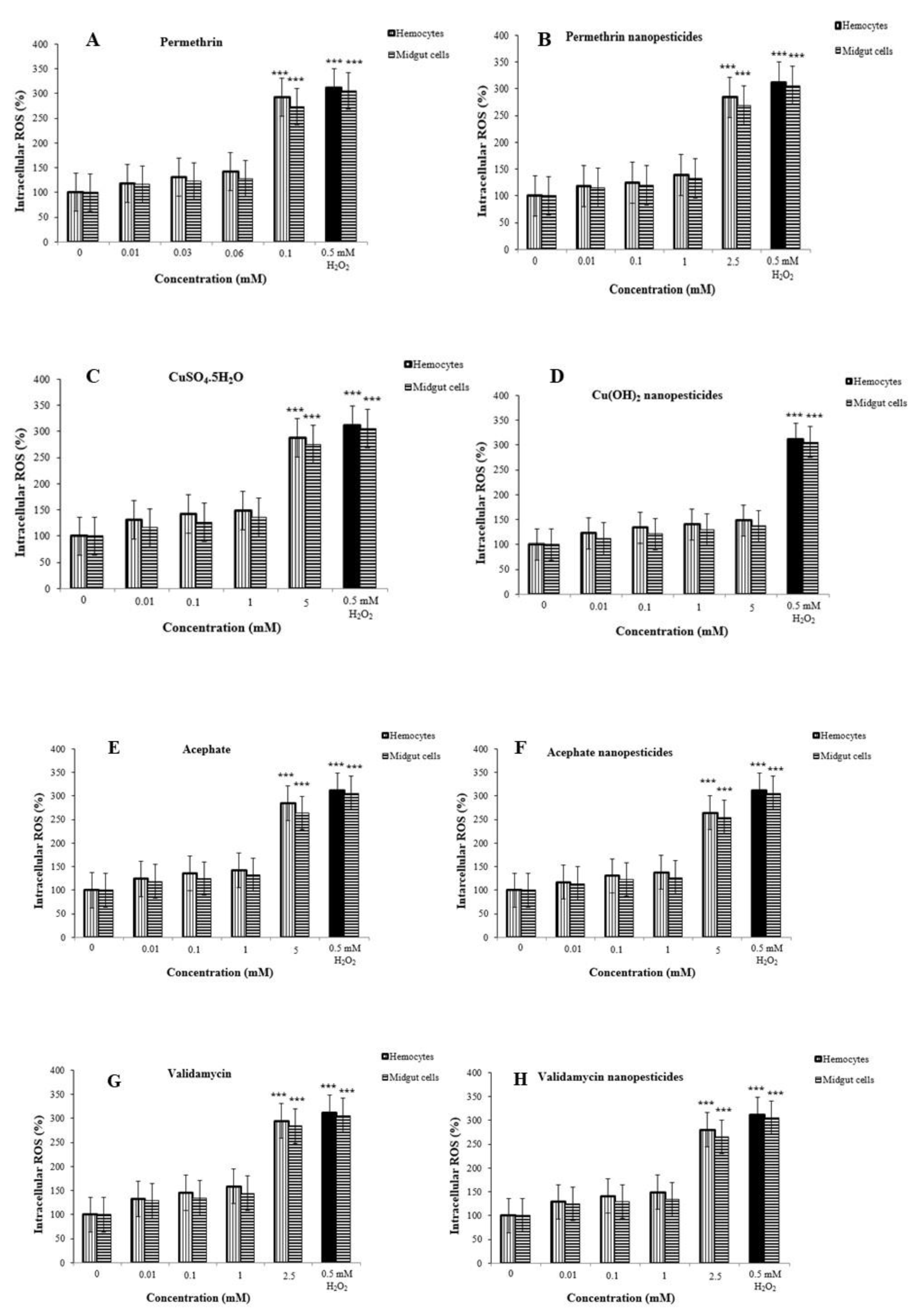
2.11. Reactive Oxygen Species (ROS) in Hemocytes and Midgut Cells
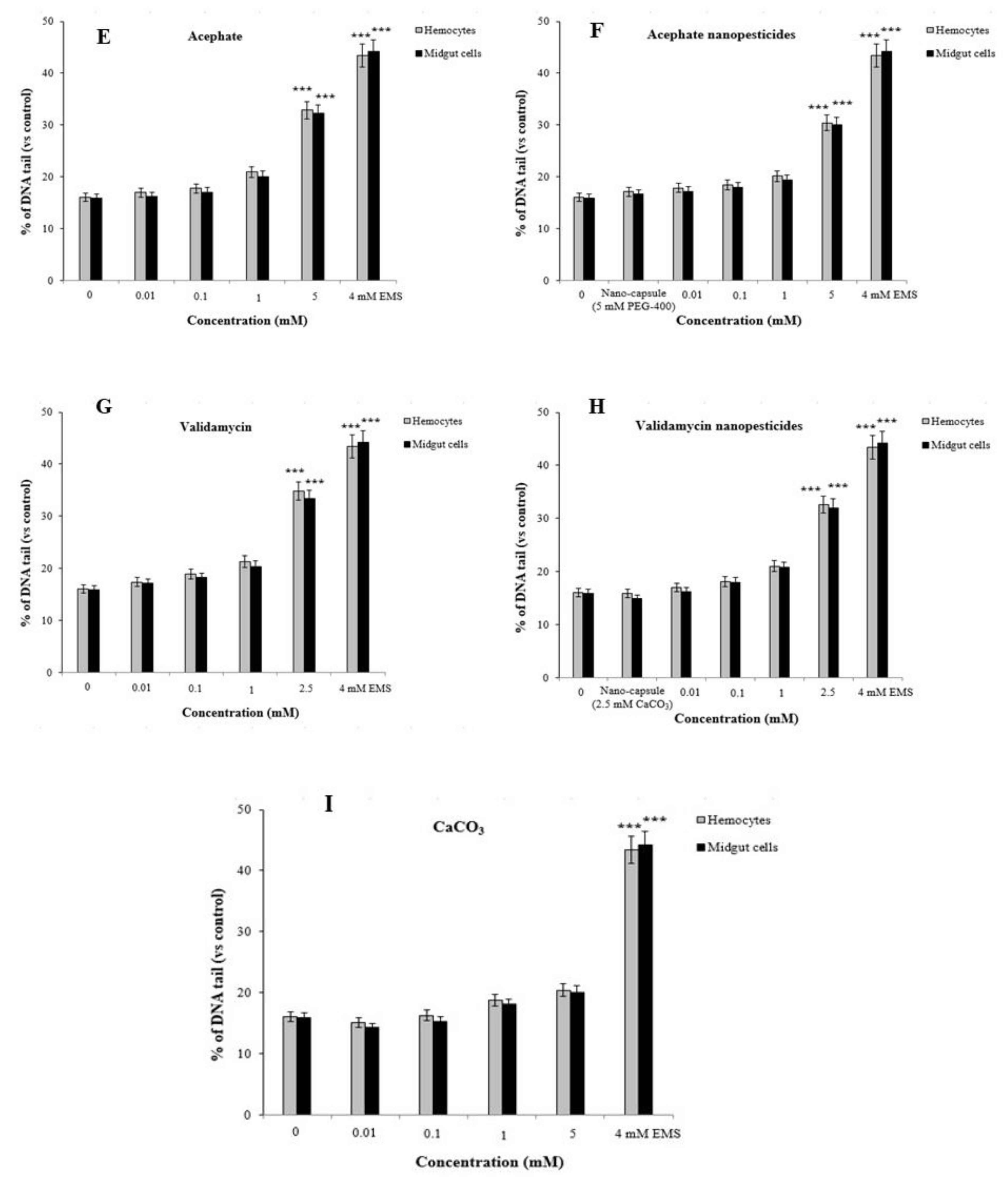
2.12. Genotoxicity Studies
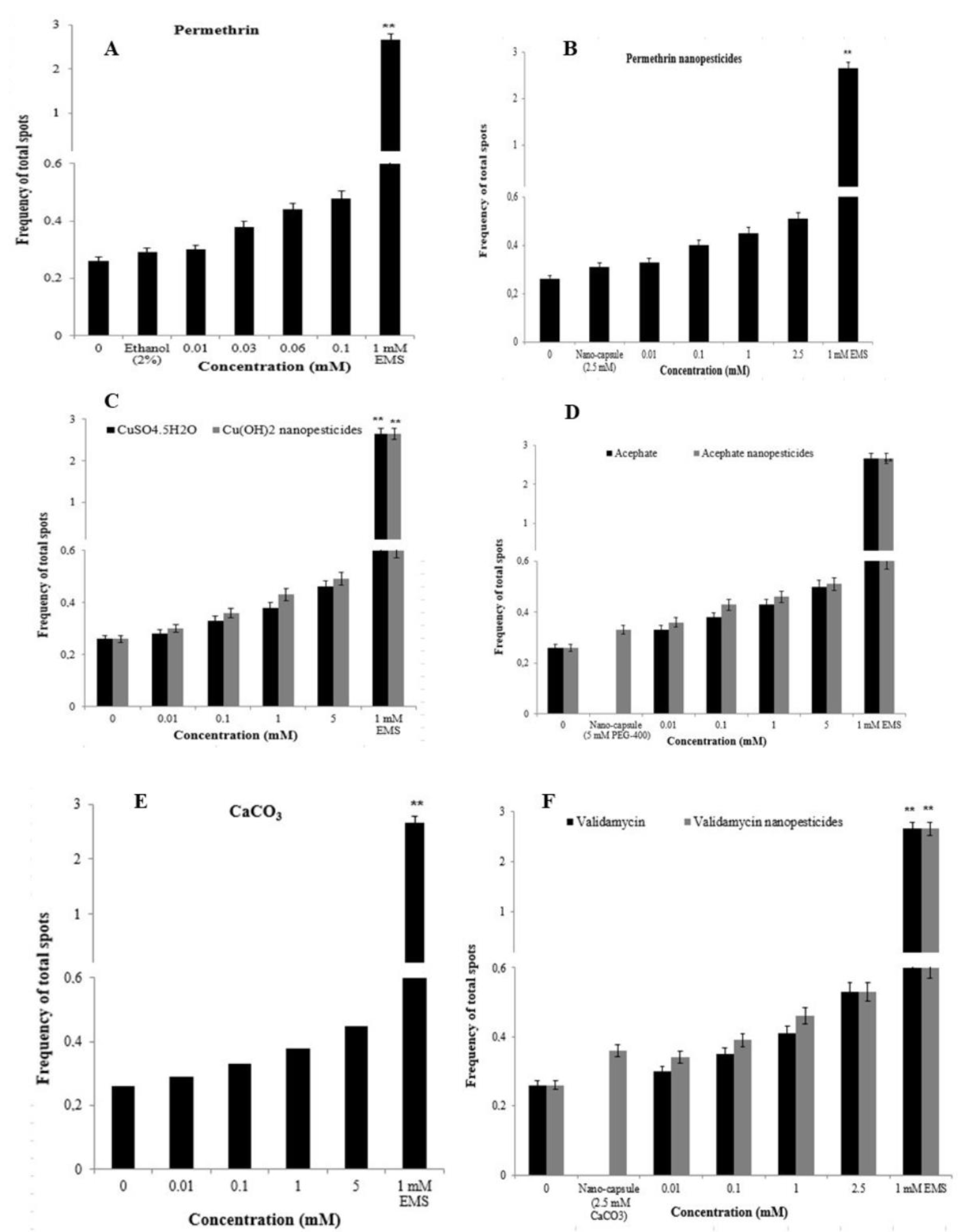
2.12.1. The Wing-Spot Assay
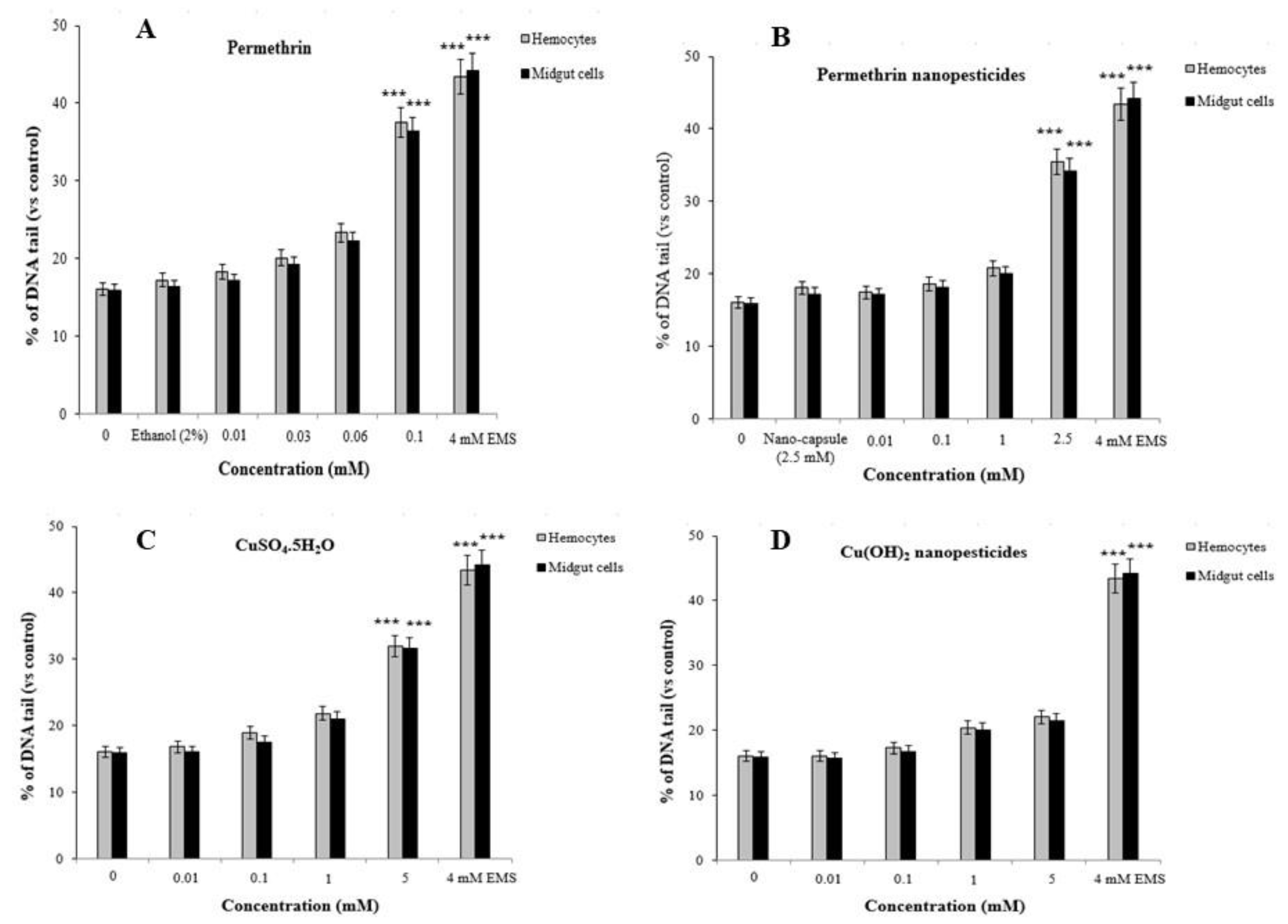
2.12.2. Comet Assay
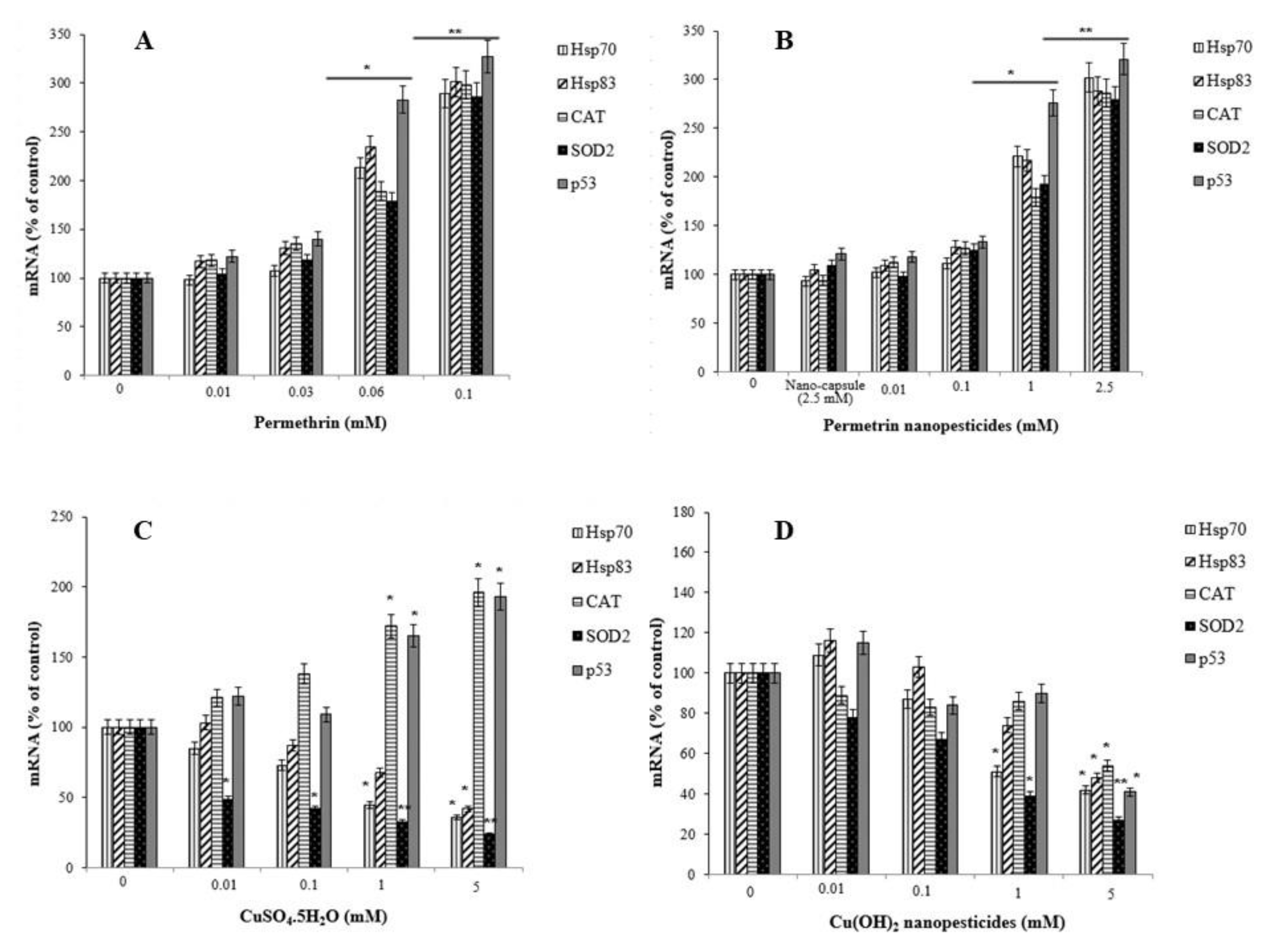
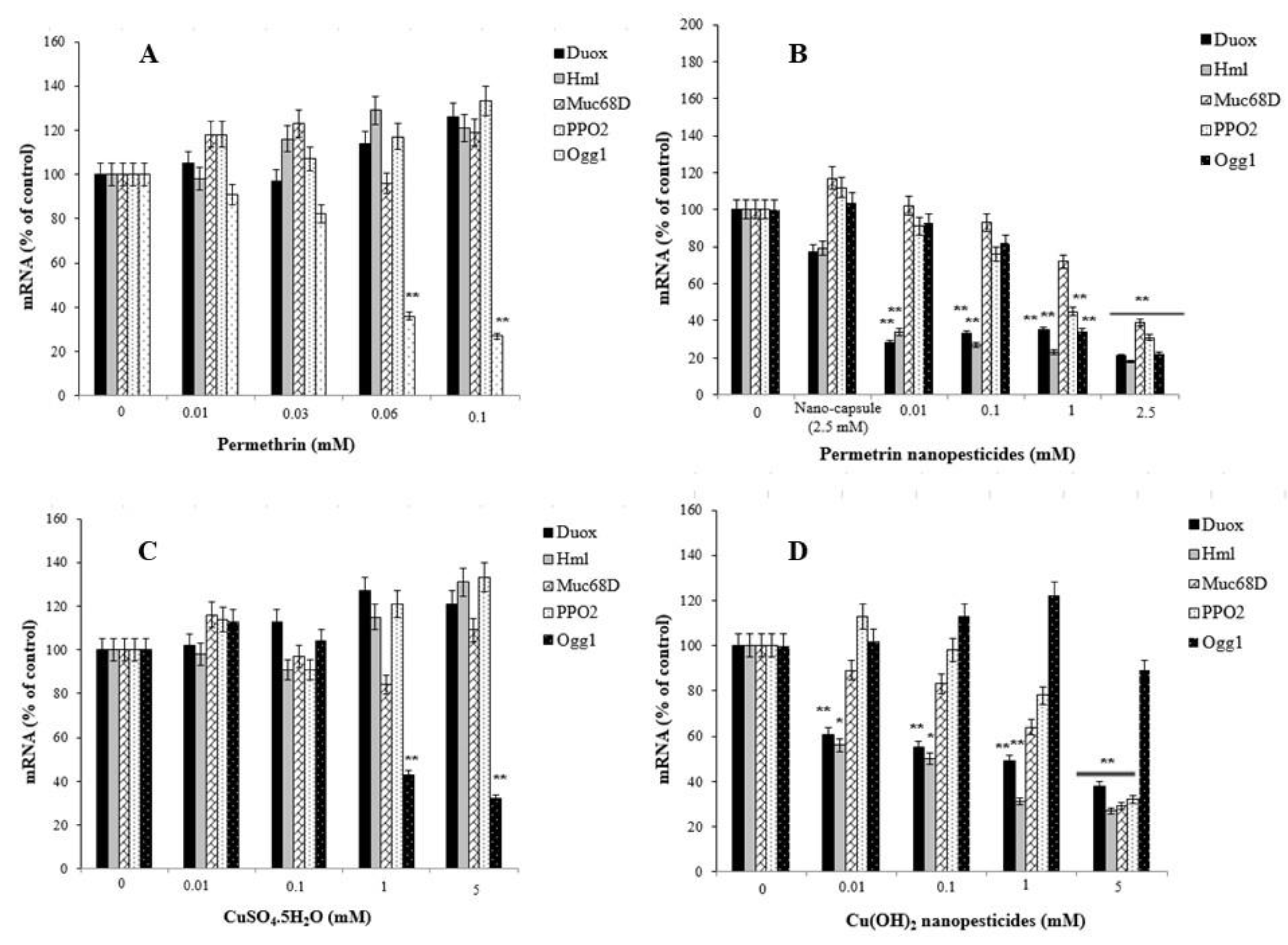
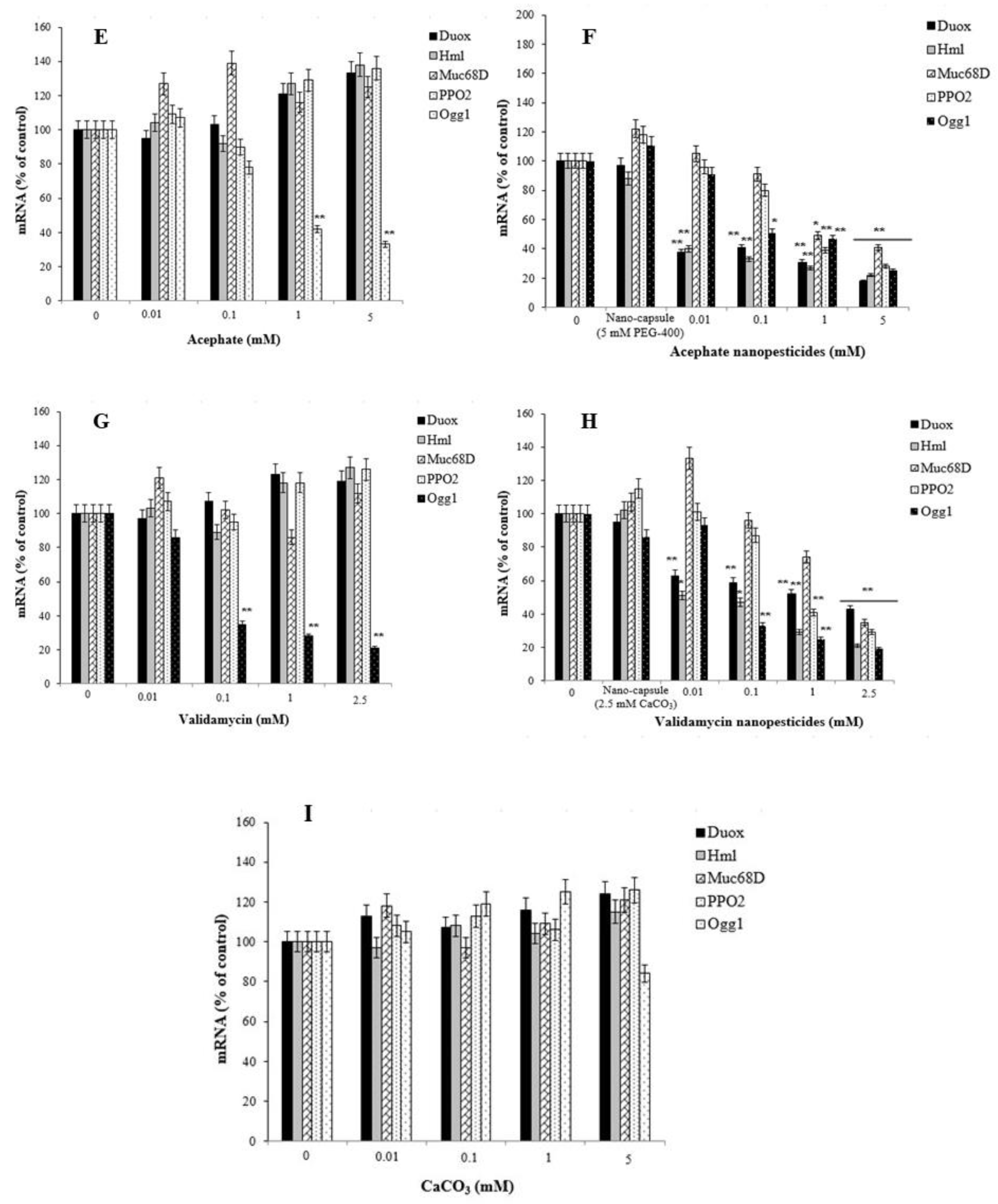
2.13. Gene Expression Changes
3. Discussion
4. Methods
4.1. Chemicals
4.2. Synthesis of Permethrin Nanopesticides, Cu(OH)2 Nanopesticides, Acephate Nanopesticides, and Validamycin Nanopesticides
4.3. Characterization and Dispersion of Permethrin Nanopesticides, Cu(OH)2 Nanopesticides, Acephate Nanopesticides, and Validamycin Nanopesticides
4.4. Endotoxin Assay
4.5. Determination of 50% Lethal Concentration (LC50) and Mortality Values
4.6. D. melanogaster Strains, Exposure, Toxicity, and Morphological Alterations
4.7. Phenotypic Variations
4.8. Climbing Assay
4.9. Oxidative Stress Assay
4.10. Lipid Peroxidation Assay
4.11. Internalization via the Intestinal Barrier
4.12. Intracellular Oxidative Stress (ROS) Detection
4.13. Comet Assay
4.14. Gene Expression Changes
4.15. Statistical Analysis
5. Conclusions
- (1)
- Characterization of nanoparticles for size and diameter should be specified in detail by TEM and SEM imaging techniques, along with XRD and HPLC.
- (2)
- Endotoxin levels at all different doses of test chemicals were below the detectable limit level (0.116667 EU/mL), and these nanopesticides were not contaminated with endotoxins.
- (3)
- Lower doses of the tested nanopesticides showed no hazardous effects on the ability of fruit flies to reach the adult stage.
- (4)
- All nanopesticides other than copper-based pesticides caused morphological changes in the abdomen, wing, mouth, and leg regions of adult flies.
- (5)
- All nanopesticides at the highest doses—except for copper-based ones—had significant pro-oxidant effects in Drosophila.
- (6)
- High doses of pesticides caused significant changes in glutathione and lipid peroxidation formation in Drosophila larvae.
- (7)
- Single-stranded and oxidative DNA damage was mediated by oxidative damage to the pyrimidine bases.
- (8)
- Nanopesticides and microparticle forms caused no mutagenic and/or recombinogenic effects.
- (9)
- All pesticides at the highest doses—except for copper-based ones—caused phenotypic variations across three generations of fruit flies.
- (10)
- Nanopesticides and microparticle forms significantly impaired the climbing and walking ability of adult flies.
- (11)
- Ingested nanopesticides found their way through the intestinal barrier into the intestinal lumen.
- (12)
- Pesticides/nanopesticides caused a significant increase in the expression of stress genes (Hsp70 and Hsp83), antioxidant defense genes (CAT and SOD2), and a genomic integrity gene (p53).
- (13)
- Nanopesticides and microparticle pesticides induced significant impairment in the expression of the DUOX, Hml, Muc68D, and/or PPO2 genes, as well as in the mRNA expression of Ogg1—a gene associated with DNA repair.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations. World Population Prospects: The 2017 Revision. Department of Economic and Social Affairs; Population Division: New York, NY, USA, 2017. [Google Scholar]
- Wiseguyreports. Agrochemicals Market 2018 Global Trend, Segmentation and Opportunities Forecast To 2021, 2018. Available online: http://www.wiseguyreports.com (accessed on 17 November 2011).
- Nuruzzaman, M.D.; Rahman, M.M.; Liu, Y.; Naidu, R. Nanoencapsulation, nano-guard for pesticides: A new window for safe application. J. Agric. Food Chem. 2016, 64, 1447–1483. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.P. Pesticides, environment, and food safety. Food Energy Secur. 2017, 6, 48–60. [Google Scholar] [CrossRef]
- Anandhi, S.; Saminathan, V.R.; Yasotha, P.; Saravanan, P.T.; Rajanbabu, V. Nano-pesticides in pest management. J. Entomol. Zool. Stud. 2020, 8, 685–690. [Google Scholar]
- Kah, M.; Hofmann, T. Nanopesticide research: Current trends and future priorities. Environ. Int. 2014, 63, 224–235. [Google Scholar] [CrossRef]
- Singh, A.; Dhiman, N.; Kar, A.K.; Singh, D.; Purohit, M.P.; Ghosh, D.; Patnaik, S. Advances in controlled release pesticide formulations: Prospects to safer integrated pest management and sustainable agriculture. J. Hazard. Mater. 2020, 385, 121525. [Google Scholar] [CrossRef]
- Elmer, W.; De La Torre-Roche, R.; Pagano, L.; Majumdar, S.; Zuverza-Mena, N.; Dimkpa, C.; Gardea-Torresdey, J.; White, J.C. Effect of metalloid and metal oxide nanoparticles on Fusarium wilt of watermelon. Plant Dis. 2018, 102, 1394–1401. [Google Scholar] [CrossRef]
- Zhao, X.; Cui, H.; Wang, Y.; Sun, C.; Cui, B.; Zeng, Z. Development strategies and prospects of nano-based smart pesticide formulation. J. Agric. Food Chem. 2018, 66, 6504–6512. [Google Scholar] [CrossRef]
- Demir, E. Drosophila as a model for assessing nanopesticide toxicity. Nanotoxicology 2020, 14, 1271–1279. [Google Scholar] [CrossRef] [PubMed]
- Gogos, A.; Knauer, K.; Bucheli, T. Nanomaterials in plant protection and fertilization: Current state, foreseen applications, and research priorities. J. Agric. Food Chem. 2012, 60, 9781–9792. [Google Scholar] [CrossRef]
- Kookana, R.S.; Boxall, A.B.A.; Reeves, P.T.; Ashauer, R.; Beulke, S.; Chaudhry, Q.; Cornelis, G.; Fernandes, T.F.; Gan, J.; Kah, M.; et al. Nanopesticides: Guiding Principles for Regulatory Evaluation of Environmental Risks. J. Agric. Food Chem. 2014, 62, 4227–4240. [Google Scholar] [CrossRef]
- Sekhon, B.S. Nanotechnology in agri-food production: An overview. Nanotechnol. Sci. Appl. 2014, 7, 31–53. [Google Scholar] [CrossRef] [PubMed]
- Kah, M. Nanopesticides and Nanofertilizers: Emerging Contaminants or Oppurtunities for Risk Mitigation? Front. Chem. 2015, 3, 64. [Google Scholar] [CrossRef]
- Kah, M.; Weniger, A.K.; Hofmann, T. Impacts of (Nano)formulations on the Fate of an Insecticide in Soil and Consequences for Environmental Exposure Assessment. Environ. Sci. Technol. 2016, 50, 10960–10967. [Google Scholar] [CrossRef] [PubMed]
- Unsworth, J.B.; Corsi, C.; Van Emon, J.M.; Farenhorst, A.; Hamilton, D.J.; Howard, C.J.; Hunter, R.; Jenkins, J.J.; Kleter, K.A.; Kookana, R.S. Developing Global Leaders for Research, Regulation, and Stewardship of Crop Protection Chemistry in the 21st Century. J. Agric. Food Chem. 2016, 4, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Aksakal, F.I.; Arslan, H. Detoxification and reproductive system-related gene expression following exposure to Cu(OH)2 nanopesticide in water flea (Daphnia magna Straus 1820). Environ. Sci. Pollut. Res. 2020, 27, 6103–6111. [Google Scholar] [CrossRef]
- Grillo, R.; Fraceto, L.F.; Amorim, M.; Scott-Fordsmand, J.J.; Schoonjans, R.; Chaudhry, Q. Ecotoxicological and regulatory aspects of environmental sustainability of nanopesticides. J. Hazard. Mater. 2021, 404, 124148. [Google Scholar] [CrossRef]
- Yusoff, S.N.M.; Kamari, A.; Aljafree, N.F.A. A review of materials used as carrier agents in pesticide formulations. Int. J. Environ. Sci. Technol. 2016, 13, 2977–2994. [Google Scholar] [CrossRef]
- Morgado, R.G.; Pavlaki, M.D.; Soares, A.M.; Loureiro, S. Terrestrial organisms react differently to nano and non-nano Cu (OH) 2 forms. Sci. Total Environ. 2022, 807, 150679. [Google Scholar] [CrossRef]
- Anjali, C.H.; Sharma, Y.; Mukherjee, A.; Chandrasekaran, N. Neem oil (Azadirachta indica) nanoemulsion as potent larvicidal agent against Culex quinquefasciatus. Pest Manag. Sci. 2012, 68, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.S.S.; Shiny, P.J.; Anjali, C.H.; Jerobin, J.; Goshen, K.M.; Magdassi, S.; Mukherjee, A.; Chandrasekaran, N. Distinctive effects of nano-sized permethrin in the environment. Environ. Sci. Pollut. Res. 2013, 20, 2593–2602. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.C.; Basri, M.; Omar, D.; Rahman, M.B.A.; Salleh, A.B.; Rahman, R.N.Z.R.A.; Selamat, A. Green nano-emulsion intervention for water-soluble glyphosate isopropylamine (IPA) formulations in controlling Eleusine indica (E-indica). Pestic. Biochem. Phys. 2012, 102, 19–29. [Google Scholar] [CrossRef]
- Lim, C.J.; Basri, M.; Omar, D.; Rahman, M.B.A.; Salleh, A.B.; Rahman, R.N.Z.R.A. Physicochemical characterization and formation of glyphosate-laden nano-emulsion for herbicide formulation. Ind. Crops Prod. 2012, 36, 607–613. [Google Scholar] [CrossRef]
- Lim, C.J.; Basri, M.; Omar, D.; Rahman, M.B.A.; Salleh, A.B.; Rahman, R.N.Z.R.A. Green nanoemulsion-laden glyphosate isopropylamine formulation in suppressing creeping foxglove (A. gangetica), slender button weed (D. ocimifolia) and buffalo grass (P. conjugatum). Pest Manag. Sci. 2013, 69, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, F.; Shafiq, S.; Haq, N.; Alanazi, F.K.; Alsarra, I.A. Nanoemulsions as potential vehicles for transdermal and dermal delivery of hydrophobic compounds: An overview. Expert Opin. Drug Deliv. 2012, 9, 953–974. [Google Scholar] [CrossRef] [PubMed]
- Parrino, V.; Costa, G.; Cannavà, C.; Fazio, E.; Bonsignore, M.; Concetta, S.; Piccione, G.; Fazio, F. Flow cytometry and micro-Raman spectroscopy: Identification of hemocyte populations in the mussel Mytilus galloprovincialis (Bivalvia: Mytilidae) from Faro Lake and Tyrrhenian Sea (Sicily, Italy). Fish Shellfish Immunol. 2019, 87, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Parrino, V.; Costa, G.; Giannetto, A.; De Marco, G.; Cammilleri, G.; Acar, Ü.; Piccione, G.; Fazio, F. Trace elements (Al, Cd, Cr, Cu, Fe, Mn, Ni, Pb and Zn) in Mytilus galloprovincialis and Tapes decussatus from Faro and Ganzirri Lakes (Sicily, Italy): Flow cytometry applied for hemocytes analysis. J. Trace Elem. Med. Biol. 2021, 68, 126870. [Google Scholar] [CrossRef] [PubMed]
- WHO, World Health Organization. Permethrin Environmental Health Criteria; World Health Organization: Geneva, Switzerland, 1990; Volume 94. [Google Scholar]
- Cetin, H.; Demir, E.; Kocaoğlu, S.; Kaya, B. Insecticidal Activity of Some Synthetic Pyrethroids with Different Rates of Piperonyl Butoxide (PBO) Combinations on Drosophila melanogaster (Diptera: Drosophilidae). Ekoloji 2010, 19, 27–32. [Google Scholar] [CrossRef]
- Demir, E.; Kaya, B.; Cenkci Kocaoğlu, S.; Çetin, H.; Marcos, R. In vivo genotoxicity of four synthetic pyrethroids with combinations piperonyl butoxide (PBO) in the Drosophila SMART assay. Ekoloji 2014, 23, 9–18. [Google Scholar] [CrossRef]
- Sundaramoorthy, R.; Velusamy, Y.; Balaji, A.P.B.; Mukherjee, A.; Chandrasekaran, N. Comparative cytotoxic and genotoxic effects of permethrin and its nanometric form on human erythrocytes and lymphocytes in vitro. Chem. Biol. Interact. 2016, 257, 119–124. [Google Scholar] [CrossRef]
- Mishra, P.; Balaji, A.P.B.; Swathy, J.S.; Paari, A.L.; Kezhiah, M.; Tyagi, B.K.; Mukherjee, A.; Chandrasekaran, N. Stability assessment of hydro dispersive nanometric permethrin and its biosafety study towards the beneficial bacterial isolate from paddy rhizome. Environ. Sci. Pollut. Res. 2016, 23, 24970–24982. [Google Scholar] [CrossRef]
- Debnath, N.; Mitra, S.; Das, S.; Goswami, A. Synthesis of surface functionalized silica nanoparticles and their use as entomotoxic nanocides. Powder Technol. 2012, 221, 252–256. [Google Scholar] [CrossRef]
- Shukla, G.; Gaurav, S.S.; Singh, A. Synthesis of mycogenic zinc oxide nanoparticles and preliminary determination of its efficacy as a larvicide against white grubs (Holotrichia sp.). Int. Nano Lett. 2020, 10, 131–139. [Google Scholar] [CrossRef]
- Paret, M.L.; Vallad, G.E.; Averett, D.R.; Jones, J.B.; Olson, S.M. Photocatalysis: Effect of light-activated nanoscale formulations of TiO2 on Xanthomonas perforans and control of bacterial spot of tomato. Phytopathology 2013, 103, 228–236. [Google Scholar] [CrossRef]
- Guilger-Casagrande, M.; Germano-Costa, T.; Pasquoto-Stigliani, T.; Fraceto, L.F.; Lima, R. Biosynthesis of silver nanoparticles employing Trichoderma harzianum with enzymatic stimulation for the control of Sclerotinia sclerotiorum. Sci. Rep. 2019, 9, 14351. [Google Scholar] [CrossRef]
- Mondal, K.K.; Mani, C. Investigation of the antibacterial properties of nanocopper against Xanthomonas axonopodis pv. punicae, the incitant of pomegranate bacterial blight. Ann. Microbiol. 2012, 62, 889–893. [Google Scholar] [CrossRef]
- Stadler, T.; Buteler, M.; Weaver, D.K.; Sofie, S. Comparative toxicity of nanostructured alumina and a commercial inert dust for Sitophilus oryzae (L.) and Rhyzopertha dominica (F.) at varying ambient humidity levels. J. Stored Prod. Res. 2012, 48, 81–90. [Google Scholar] [CrossRef]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanoparticle Res. 2013, 15, 1692. [Google Scholar] [CrossRef]
- Ren, G.G.; Hu, D.W.; Cheng, E.W.C.; Vargas-Reus, M.A.; Reip, P.; Allaker, R.P. Characterisation of copper oxide nanoparticles for antimicrobial applications. Int. J. Antimicrob. Agents 2019, 33, 587–590. [Google Scholar] [CrossRef]
- Rubilar, O.; Rai, M.; Tortella, G.; Diez, M.C.; Seabra, A.B.; Durán, N. Biogenic nanoparticles: Copper, copper oxides, copper sulphides, complex copper nanostructures and their applications. Biotechnol. Lett. 2013, 35, 1365–1375. [Google Scholar] [CrossRef]
- Carmona, E.R.; Inostroza-Blancheteau, C.; Obando, V.; Rubio, L.; Marcos, R. Genotoxicity of copper oxide nanoparticles in Drosophila melanogaster. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2015, 791, 1–11. [Google Scholar] [CrossRef]
- Alaraby, M.; Hernandez, A.; Marcos, R. New insights in the toxic/genotoxic effects of CuO nanoparticles in the in vivo Drosophila model. Nanotoxicology 2016, 10, 749–760. [Google Scholar] [CrossRef]
- Alaraby, M.; Hernández, A.; Marcos, R. Copper oxide nanoparticles and copper sulphate act as anti-genotoxic agents in Drosophila melanogaster. Environ. Mol. Mutagen. 2017, 58, 46–55. [Google Scholar] [CrossRef]
- Baeg, E.; Sooklert, K.; Sereemaspun, A. Copper oxide nanoparticles cause a dose-dependent toxicity via inducing reactive oxygen species in Drosophila. Nanomaterials 2018, 8, 824. [Google Scholar] [CrossRef] [PubMed]
- Carmona, E.R.; García-Rodríguez, A.; Marcos, R. Genotoxicity of copper and nickel nanoparticles in somatic cells of Drosophila melanogaster. J. Toxicol. 2018, 2018, 7278036. [Google Scholar] [CrossRef]
- Gaetke, L.M.; Chow-Johnson, H.S.; Chow, C.K. Copper: Toxicological relevance and mechanisms. Arch. Toxicol. 2014, 88, 1929–1938. [Google Scholar] [CrossRef] [PubMed]
- Navrotsky, A. Nanomaterials in the environment, agriculture, and technology (NEAT). J. Nanoparticle Res. 2000, 2, 321–323. [Google Scholar] [CrossRef]
- Choudhury, S.R.; Pradhan, S.; Goswami, A. Preparation and characterisation of acephate nano-encapsulated complex. Nanosci. Methods 2012, 1, 9–15. [Google Scholar] [CrossRef]
- Rajak, P.; Dutta, M.; Khatun, S.; Mandi, M.; Roy, S. Exploring hazards of acute exposure of Acephate in Drosophila melanogaster and search for l-ascorbic acid mediated defense in it. J. Hazard. Mater. 2017, 321, 690–702. [Google Scholar] [CrossRef] [PubMed]
- Rajak, P.; Khatun, S.; Dutta, M.; Mandi, M.; Roy, S. Chronic exposure to acephate triggers ROS-mediated injuries at organismal and sub-organismal levels of Drosophila melanogaster. Toxicol. Res. 2018, 7, 874–887. [Google Scholar] [CrossRef]
- Matassini, C.; Parmeggiani, C.; Cardona, F. New Frontiers on Human Safe Insecticides and Fungicides: An Opinion on Trehalase Inhibitors. Molecules 2020, 25, 3013. [Google Scholar] [CrossRef]
- Kah, M.; Beulke, S.; Tiede, K.; Hofmann, T. Nano-pesticides: State of knowledge, environmental fate and exposure modelling. Crit. Rev. Environ. Sci. Technol. 2013, 43, 1823–1867. [Google Scholar] [CrossRef]
- Song, M.R.; Cui, S.M.; Gao, F.; Liu, Y.R.; Fan, C.L.; Lei, T.Q.; Liu, D.C. Dispersible silica nanoparticles as carrier for enhanced bioactivity of chlorfenapyr. J. Pest. Sci. 2012, 37, 258–260. [Google Scholar] [CrossRef]
- Mingming, A.; Yuncong, Z.; Shun, H.; Deguang, L.; Pingliang, L.; Jianqiang, L.; Yongsong, C. Preparation and characterization of 1-naphthylacetic acid-silica conjugated nanospheres for enhancement of controlled-release performance. Nanotechnology 2013, 24, 035601–035608. [Google Scholar]
- Qian, K.; Shi, T.; Tang, T.; Zhang, S.; Liu, X.; Cao, Y. Preparation and characterization of nano-sized calcium carbonate as controlled release pesticide carrier for validamycin against Rhizoctonia solani. Microchim. Acta 2011, 173, 51–57. [Google Scholar] [CrossRef]
- Addadi, L.; Raz, S.; Weiner, S. Taking advantage of disorder: Amorphous calcium carbonate and its roles in biomineralization. Adv. Mater. 2003, 15, 959–970. [Google Scholar] [CrossRef]
- Huang, Y.X.; Adeleye, A.S.; Zhao, L.J.; Minakova, A.S.; Anumol, T.; Keller, A.A. Antioxidant response of cucumber (Cucumis sativus) exposed to nano copper pesticide: Quantitative determination via LC-MS/MS. Food Chem. 2019, 270, 47–52. [Google Scholar] [CrossRef]
- Peng, C.; Zhao, Q.; Gao, C. Sustained delivery of doxorubicin by porous CaCO3 and chitosan/alginate multilayers-coated CaCO3 microparticles. Colloids Surf. A 2010, 353, 132–139. [Google Scholar] [CrossRef]
- Trinci, A.P.J. Effect of validamycin A and l-sorbose on the growth and morphology of Rhizoctonia cerealis and Rhizoctonia solani. Exp. Mycol. 1985, 1, 20–27. [Google Scholar] [CrossRef]
- Marten, A.D.; Stothard, A.I.; Kalera, K.; Swarts, B.M.; Conway, M.J. Validamycin A delays development and prevents flight in Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 2020, 57, 1096–1103. [Google Scholar] [CrossRef]
- Bernards, A.; Hariharan, I.K. Of flies and men- studying human disease in Drosophila. Curr. Opin. Genet. Dev. 2001, 11, 274–278. [Google Scholar] [CrossRef]
- Lloyd, T.E.; Taylor, J.P. Flightless flies: Drosophila models of neuromuscular disease. Ann. N. Y. Acad. Sci. 2010, 1184, E1–E20. [Google Scholar] [CrossRef] [PubMed]
- Demir, E.; Turna Demir, F. Drosophila melanogaster as a dynamic in vivo model organism reveal the hidden effects of interactions between microplastic/nanoplastic and heavy metals. J. Appl. Toxicol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Lee, W.J. Role of DUOX in gut inflammation: Lessons from Drosophila model of gut-microbiota interactions. Front. Cell Infect. Microbiol. 2014, 3, 116. [Google Scholar] [CrossRef] [PubMed]
- Grillo, R.; Pereira, A.E.; Nishisaka, C.S.; De Lima, R.; Oehlke, K.; Greiner, R.; Fraceto, L.F. Chitosan/tripolyphosphate nanoparticles loaded with paraquat herbicide: An environmentally safer alternative for weed control. J. Hazard. Mater. 2014, 278, 163–171. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, J.L.; Campos, E.V.; Pereira, A.E.; Pasquoto, T.; Lima, R.; Grillo, R.; de Andrade, D.J.; dos Santos, F.A.; Fraceto, L.F. Zein nanoparticles as eco-friendly carrier systems for botanical repellents aiming sustainable agriculture. J. Agric. Food Chem. 2018, 66, 1330–1340. [Google Scholar] [CrossRef]
- Pascoli, M.; de Albuquerque, F.P.; Calzavara, A.K.; Tinoco-Nunes, B.; Oliveira, W.H.C.; Goncalves, K.C.; Polanczyk, R.A.; Della Vechia, J.F.; de Matoss, S.T.S.; de Andrade, D.; et al. The potential of nanobiopesticide based on zein nanoparticles and neem oil for enhanced control of agricultural pests. J. Pest Sci. 2020, 93, 793–806. [Google Scholar] [CrossRef]
- Adeleye, A.S.; Conway, J.R.; Perez, T.; Rutten, P.; Keller, A.A. Influence of extracellular polymeric substances on the long-term fate, dissolution, and speciation of copper-based nanoparticles. Environ. Sci. Technol. 2014, 48, 12561–12568. [Google Scholar] [CrossRef] [PubMed]
- Awwad, A.M.; Albiss, B. Biosynthesis of colloidal copper hydroxide nanowires using Pistachio leaf extract. Adv. Mater. Lett. 2015, 6, 51–54. [Google Scholar] [CrossRef]
- Kontoyannis, C.G.; Vagenas, N.V. Calcium carbonate phase analysis using XRD and FT-Raman spectroscopy. Analyst 2000, 125, 251–255. [Google Scholar] [CrossRef]
- Guo, H.; Sun, P.; Qin, Z.; Shan, L.; Zhang, G.; Cui, S.; Liang, Y. Sodium Lignosulfonate Induced Vaterite Calcium Carbonate with Multilayered Structure. Eur. J. Inorg. Chem. 2014, 6, 1001–1009. [Google Scholar] [CrossRef]
- Demir, E.; Marcos, R.; Kaya, B. Genotoxicity studies in the ST cross of the Drosophila wing spot test of sunflower and soybean oils before and after frying and boiling procedures. Food Chem. Toxicol. 2012, 50, 3619–3624. [Google Scholar] [CrossRef]
- Domenech, J.; Hernández, A.; Demir, E.; Marcos, R.; Cortés, C. Interactions of graphene oxide and graphene nanoplatelets with the in vitro Caco-2/HT29 model of intestinal barrier. Sci. Rep. 2020, 10, 2793. [Google Scholar] [CrossRef]
- Collins, A.R. Assays for oxidative stres and antioxidant status: Applications to research into the biological effectiveness of polyphenols. Am. J. Clin. Nutr. 2005, 81, 261–267. [Google Scholar] [CrossRef]
- Kastenbaum, M.A.; Bowman, K.O. Tables for determining the statistical significance of mutation frequencies. Mutat. Res. 1970, 9, 527–549. [Google Scholar] [CrossRef]
- Garcia-Quispes, W.A.; Carmona, E.R.; Creus, A.; Marcos, R. Genotoxic evaluation of two halonitromethane disinfection by-products in the Drosophila wing-spot test. Chemosphere 2009, 75, 906–909. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.E.; do Amaral, V.S.; Dihl, R.R.; Lehmann, M.; Reguly, M.L.; de Andrade, H.H.R. Assessment of genotoxicity of Lidocaine, Prilonest and Septanest in the Drosophila wing-spot test. Food Chem. Toxicol. 2009, 47, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Henderson, L.; Wolfreys, A.; Fedyk, J.; Bourner, C.; Windebank, S. The ability of the Comet assay to discriminate between genotoxins and cytotoxins. Mutagenesis 1998, 13, 89–94. [Google Scholar] [CrossRef]
- Mukhopadhyay, I.; Nazir, A.; Saxena, D.K.; Chowdhuri, D.K. Heat shock response: hsp70 in environmental monitoring. J. Biochem. Mol. Toxicol. 2003, 17, 249–254. [Google Scholar] [CrossRef]
- Demir, E.; Akça, H.; Kaya, B.; Burgucu, D.; Tokgün, O.; Turna, F.; Aksakal, S.; Vales, G.; Creus, A.; Marcos, R. Zinc oxide nanoparticles: Genotoxicity, interactions with UV-light and cell-transforming potential. J. Hazard. Mater. 2014, 264, 420–429. [Google Scholar] [CrossRef]
- Demir, E.; Akça, H.; Turna, F.; Aksakal, S.; Burgucu, D.; Kaya, B.; Tokgün, O.; Vales, G.; Creus, A.; Marcos, R. Genotoxic and cell-transforming effects of titanium dioxide nanoparticles. Environ. Res. 2015, 136, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Demir, E.; Aksakal, S.; Turna, F.; Kaya, B.; Marcos, R. In vivo genotoxic effects of four different nano-sizes forms of silica nanoparticles in Drosophila melanogaster. J. Hazard. Mater. 2015, 283, 260–266. [Google Scholar] [CrossRef]
- Siddique, H.R.; Mitra, K.; Bajpai, V.K.; Ravi Ram, K.; Saxena, D.K.; Chowdhuri, D.K. Hazardous effect of tannery solid waste leachates on development and reproduction in Drosophila melanogaster: 70kDa heat shock protein as a marker of cellular damage. Ecotoxicol. Environ. Saf. 2009, 72, 1652–1662. [Google Scholar] [CrossRef]
- Inoue, T.; Wu, L.; Stuart, J.; Maki, C.G. Control of p53 nuclear accumulation in stressed cells. FEBS Letters 2005, 579, 4978–4984. [Google Scholar] [CrossRef]
- Hayakawa, T.; Shitomi, Y.; Miyamoto, K.; Hori, H. GalNAc pretreatment inhibits trapping of Bacillus thuringiensis Cry1Ac on the peritrophic membrane of Bombyx mori. FEBS Lett. 2004, 576, 331–335. [Google Scholar] [CrossRef]
- Alaraby, M.; Demir, E.; Domenech, J.; Velázquez, A.; Hernández, A.; Marcos, R. In vivo evaluation of the toxic and genotoxic effects of exposure to cobalt nanoparticles in Drosophila melanogaster. Environ. Sci. Nano 2020, 7, 610–622. [Google Scholar] [CrossRef]
- Shibata, T.; Sekihara, S.; Fujikawa, T.; Miyaji, R.; Maki, K.; Ishihara, T. Transglutaminase-catalyzed protein-protein cross-linking suppresses the activity of the NF-kB-like transcription factor relish. Sci. Signal. 2013, 6, 61. [Google Scholar] [CrossRef]
- Syed, Z.A.; Hard, T.; Uv, A.; van Dijk-Hard, I.F. A potential role for Drosophila mucins in development and physiology. PLoS ONE 2008, 3, e3041. [Google Scholar] [CrossRef]
- Kuraishi, T.; Hori, A.; Kurata, S. Host-microbe interactions in the gut of Drosophila melanogaster. Front. Physiol. 2013, 4, 375. [Google Scholar] [CrossRef]
- Bae, Y.S.; Choi, M.K.; Lee, W.J. Dual oxidase in mucosal immunity and host-microbe homeostasis. Trends Immunol. 2010, 31, 278–287. [Google Scholar] [CrossRef]
- Kumar, S.; Molina-Cruz, A.; Gupta, L.; Rodrigues, J.; Barillas-Mury, C. A peroxidase/dual oxidase system modulates midgut epithelial immunity in Anopheles gambiae. Science 2010, 327, 1644–1648. [Google Scholar] [CrossRef]
- Scherfer, C.; Karlsson, C.; Loseva, O.; Bidla, G.; Goto, A.; Havemann, J. Isolation and characterization of hemolymph clotting factors in Drosophila melanogaster by a pullout method. Curr. Biol. 2004, 14, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Goto, A.; Kumagai, T.; Kumagai, C.; Hirose, J.; Narita, H.; Mori, H.; Kadowaki, T.; Beck, K.; Kitagawa, Y. A Drosophila haemocyte-specific protein, hemolectin, similar to human von Willebrand factor. Biochem. J. 2001, 359, 99–108. [Google Scholar] [CrossRef]
- Binggeli, O.; Neyen, C.; Poidevin, M.; Lemaitre, B. Prophenoloxidase activation is required for survival to microbial infections in Drosophila. PLoS Pathog. 2014, 10, e1004067. [Google Scholar] [CrossRef] [PubMed]
- Pascoli, M.; Jacques, M.T.; Agarrayua, D.A.; Avila, D.S.; Lima, R.; Fraceto, L.F. Neem oil based nanopesticide as an environmentally-friendly formulation for applications in sustainable agriculture: An ecotoxicological perspective. Sci. Total Environ. 2019, 677, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Kah, M.; Kookana, R.S.; Gogos, A.; Bucheli, T.D. A critical evaluation of nanopesticides and nanofertilizers against their conventional analogues. Nat. Nanotechnol. 2018, 13, 677–684. [Google Scholar] [CrossRef]
- Oliveira, H.C.; Stolf-Moreira, R.; Martinez, C.B.R.; Sousa, G.F.M.; Grillo, R.; de Jesus, M.B.; Fraceto, L.F. Evaluation of the side effects of poly(epsilon-caprolactone) nanocapsules containing atrazine toward maize plants. Front. Chem. 2015, 3, 61. [Google Scholar] [CrossRef] [PubMed]
- Lykogianni, M.; Papadopoulou, E.A.; Sapalidis, A.; Tsiourvas, D.; Sideratou, Z.; Aliferis, K.A. Metabolomics reveals differential mechanisms of toxicity of hyperbranched poly(ethyleneimine)-derived nanoparticles to the soil-borne fungus Verticillium dahliae Kleb. Pestic. Biochem. Phys. 2020, 165, 104535. [Google Scholar] [CrossRef]
- OECD (Organisation for Economic Co-operation and Development). No. 36: Guidance on Sample Preparation and Dosimetry for the Safety Testing of Manufactured Nanomaterials; OECD: Paris, France, 2012. [Google Scholar]
- OECD (Organisation for Economic Co-operation and Development). No. 63: Physical-Chemical Parameters: Measurements and Methods Relevant for the Regulation of Nanomaterials; OECD: Paris, France, 2016. [Google Scholar]
- Hund-Rinke, K.; Baun, A.; Cupi, D.; Fernandes, T.F.; Handy, R.; Kinross, J.H.; Navas, J.M.; Peijnenburg, W.; Schlich, K.; Shaw, B.J.; et al. Regulatory ecotoxicity testing of nanomaterials-proposed modifications of OECD test guidelines based on laboratory experience with silver and titanium dioxide nanoparticles. Nanotoxicology 2016, 10, 1442–1447. [Google Scholar] [CrossRef] [PubMed]
- Amorim, M.J.B.; Fernandez-Cruz, M.L.; Hund-Rinke, K.; Scott-Fordsmand, J.J. Environmental hazard testing of nanobiomaterials. Environ. Sci. Eur. 2020, 32, 101. [Google Scholar] [CrossRef]
- Gomes, S.I.L.; Scott-Fordsmand, J.J.; Campos, E.V.R.; Grillo, R.; Fraceto, L.F.; Amorim, M.J.B. On the safety of nanoformulations to non-target soil invertebrates—An atrazine case study. Environ. Sci. Nano 2019, 6, 1950–1958. [Google Scholar] [CrossRef]
- Fojtova, D.; Vasickova, J.; Grillo, R.; Bilkova, Z.; Simek, Z.; Neuwirthova, N.; Kah, M.; Hofman, J. Nanoformulations can significantly affect pesticide degradation and uptake by earthworms and plants. Environ. Chem. 2019, 16, 470–481. [Google Scholar] [CrossRef]
- Neves, J.; Cardoso, D.N.; Malheiro, C.; Kah, M.; Soares, A.M.V.M.; Wrona, F.J.; Loureiro, S. 2019. Copper toxicity to Folsomia candida in different soils: A comparison between nano and conventional formulations. Environ. Chem. 2019, 16, 419–429. [Google Scholar] [CrossRef]
- Jacques, M.T.; Oliveira, J.L.; Campos, E.V.R.; Fraceto, L.F.; Avila, D.S. Safety assessment of nanopesticides using the roundworm Caenorhabditis elegans. Ecotoxicol. Environ. Saf. 2017, 139, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Firdaus, M.A.M.; Agatz, A.; Hodson, M.E.; Al-Khazrajy, O.S.A.; Boxall, A.B.A. 2018. Fate, uptake, and distribution of nanoencapsulated pesticides in soil-earthworm systems and implications for environmental risk assessment. Environ. Toxicol. Chem. 2018, 37, 1420–1429. [Google Scholar]
- Phanse, Y.; Dunphy, B.M.; Perry, J.L.; Airs, P.M.; Paquette, C.C.H.; Carlson, J.O.; Xu, J.; Luft, J.C.; DeSimone, J.M.; Beaty, B.J.; et al. Biodistribution and toxicity studies of PRINT hydrogel nanoparticles in mosquito larvae and cells. PLoS Negl. Trop. Dis. 2015, 9, e0003735. [Google Scholar] [CrossRef]
- He, X.J.; Aker, W.G.; Fu, P.P.; Hwang, H.M. Toxicity of engineered metal oxide nanomaterials mediated by nano-bio-eco-interactions: A review and perspective. Environ. Sci. Nano 2015, 2, 564–582. [Google Scholar] [CrossRef]
- Hjorth, R.; Skjolding, L.M.; Sorensen, S.N.; Baun, A. Regulatory adequacy of aquatic ecotoxicity testing of nanomaterials. Nanoimpact 2017, 8, 28–37. [Google Scholar] [CrossRef]
- Jenning, V.; Goodhead, R.; Tyler, C.R. Ecotoxicology of nanomaterials in aquatic systems. Front. Nanosci. 2015, 8, 3–45. [Google Scholar]
- Blewett, T.A.; Qi, A.A.; Zhang, Y.Y.; Weinrauch, A.M.; Blair, S.D.; Folkerts, E.J.; Sheedy, C.; Nilsson, D.; Goss, G.G. Toxicity of nanoencapsulated bifenthrin to rainbow trout (Oncorhynchus mykiss). Environ. Sci. Nano 2019, 6, 2777–2785. [Google Scholar] [CrossRef]
- Amjad, S.; Sharma, A.K.; Serajuddin, M. Toxicity assessment of cypermethrin nanoparticles in Channa punctatus: Behavioural response, micronuclei induction and enzyme alteration. Regul. Toxicol. Pharmacol. 2018, 100, 127–133. [Google Scholar] [CrossRef]
- de Andrade, L.L.; Pereira, A.D.S.; Fraceto, L.F.; Martinez, C.B.D. Can atrazine loaded nanocapsules reduce the toxic effects of this herbicide on the fish Prochilodus lineatus? A multibiomarker approach. Sci. Total Environ. 2019, 663, 548–559. [Google Scholar] [CrossRef]
- Mishra, P.; Dutta, S.; Haldar, M.; Dey, P.; Kumar, D.; Mukherjee, A.; Chandrasekaran, N. Enhanced mosquitocidal efficacy of colloidal dispersion of pyrethroid nanometric emulsion with benignity towards non-target species. Ecotoxicol. Environ. Saf. 2019, 176, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Sheedy, C.; Nilsson, D.; Goss, G.G. Evaluation of interactive effects of UV light and nano encapsulation on the toxicity of azoxystrobin on zebrafish. Nanotoxicology 2020, 14, 232–249. [Google Scholar] [CrossRef] [PubMed]
- Clemente, Z.; Grillo, R.; Jonsson, M.; Santos, N.Z.P.; Feitosa, L.O.; Lima, R.; Fraceto, L.F. Ecotoxicological evaluation of poly(epsilon-caprolactone) nanocapsules containing triazine herbicides. J. Nanosci. Nanotechnol. 2014, 14, 4911–4917. [Google Scholar] [CrossRef]
- Vignardi, C.P.; Muller, E.B.; Tran, K.; Couture, J.L.; Means, J.C.; Murray, J.L.S.; Ortiz, C.; Keller, A.A.; Smith Sanchez, N.; Lenihan, H.S. Conventional and nano-copper pesticides are equally toxic to the estuarine amphipod Leptocheirus plumulosus. Aquat. Toxicol. 2020, 224, 105481. [Google Scholar] [CrossRef]
- Slattery, M.; Harper, B.; Harper, S. Pesticide encapsulation at the nanoscale drives changes to the hydrophobic partitioning and toxicity of an active ingredient. Nanomaterials 2019, 9, 1. [Google Scholar] [CrossRef]
- Wang, L.; Huang, X.L.; Sun, W.L.; Too, H.Z.; Laserna, A.K.C.; Li, S.F.Y. A global metabolomic insight into the oxidative stress and membrane damage of copper oxide nanoparticles and microparticles on microalga Chlorella vulgaris. Env. Pollut. 2020, 258, 113647. [Google Scholar] [CrossRef]
- Pontes, M.S.; Graciano, D.E.; Antunes, D.R.; Santos, J.S.; Arruda, G.J.; Botero, E.R.; Grillo, R.; Lima, S.M.; Andrade, L.H.C.; Caires, A.R.L.; et al. In vitro and in vivo impact assessment of eco-designed CuO nanoparticles on non-target aquatic photoautotrophic organisms. J. Hazard. Mater. 2020, 396, 122484. [Google Scholar] [CrossRef]
- De Oliveira, C.R.; Fraceto, L.F.; Rizzi, G.M.; Salla, R.F.; Abdalla, F.C.; Costa, M.J.; SilvaZacarin, E.C.M. Hepatic effects of the clomazone herbicide in both its free form and associated with chitosan-alginate nanoparticles in bullfrog tadpoles. Chemosphere 2016, 149, 304–313. [Google Scholar] [CrossRef]
- Oliveira, C.R.; Garcia, T.D.; Franco-Belussi, L.; Salla, R.F.; Souza, B.F.S.; de Melo, N.F.S.; Irazusta, S.P.; Jones-Costa, M.; Silva-Zacarin, E.C.M.; Fraceto, L.F. Pyrethrum extract encapsulated in nanoparticles: Toxicity studies based on genotoxic and hematological effects in bullfrog tadpoles. Environ. Pollut. 2019, 253, 1009–1020. [Google Scholar] [CrossRef]
- Ahmed, M.A.I.; Othman, A.A.E. Piperonyl butoxide enhances the insecticidal toxicity of nanoformulation of imidacloprid on Culex pipiens (Diptera: Culicidae) mosquito. Vector-Borne Zoonotic Dis. 2020, 20, 134–142. [Google Scholar] [CrossRef]
- Balaji, A.P.B.; Ashu, A.; Manigandan, S.; Sastry, T.P.; Mukherjee, A.; Chandrasekaran, N. Polymeric nanoencapsulation of insect repellent: Evaluation of its bioefficacy on Culex quinquefasciatus mosquito population and effective impregnation onto cotton fabrics for insect repellent clothing. J. King Saud Univ. Sci. 2017, 29, 517–527. [Google Scholar] [CrossRef]
- Ayoub, H.A.; Khairy, M.; Elsaid, S.; Rashwan, F.A.; Abdel-Hafez, H.F. Pesticidal activity of nanostructured metal oxides for generation of alternative pesticide formulations. J. Agric. Food Chem. 2018, 66, 5491–5498. [Google Scholar] [CrossRef]
- Gonzalez, J.W.; Yeguerman, C.; Marcovecchio, D.; Delrieux, C.; Ferrero, A.; Fernandez, B. Evaluation of sublethal effects of polymer-based essential oils nanoformulation on the german cockroach. Ecotoxicol. Environ. Saf. 2016, 130, 11–18. [Google Scholar] [CrossRef] [PubMed]
- FAO (Food and Agriculture Organization). The Importance of Bees and Other Pollinators for Food and Agriculture; Food and Agriculture Organization of the United Nations: Slovenia, Balkans, 2018. [Google Scholar]
- Fishel, F.M. Pesticides Effects of Nontarget Organisms; PI-85; Pesticide Information Office; Florida Cooperative Extension Service, IFAS, University of Florida: Gainesville, FL, USA, 2011. [Google Scholar]
- Conway, J.R.; Adeleye, A.S.; Gardea-Torresdey, J.; Keller, A.A. Aggregation, dissolution, and transformation of copper nanoparticles in natural waters. Environ. Sci. Technol. 2015, 49, 2749–2756. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Ortiz, C.; Adeleye, A.S.; Hu, Q.; Zhou, H.; Huang, Y.; Keller, A.A. Metabolomics to Detect Response of Lettuce (Lactuca sativa) to Cu(OH)2 Nanopesticides: Oxidative Stress Response and Detoxification Mechanisms. Environ. Sci. Technol. 2016, 50, 9697–9707. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, N.R.; Pojano, G.; Wallin, H.; Jensen, K.A. Nanomaterial dispersion protocol for toxicological studies in ENPRA. Intern. ENPRA Rep. 2010, 6, 8. [Google Scholar]
- Nanogenotox. Available online: http://www.nanogenotox.eu/files/PDF/Deliverables/nanogenotox%20deliverable%203_wp4_%20dispersion%20protocol.pdf (accessed on 17 November 2011).
- Lankoff, A.; Arabski, M.; Wegierek-Ciuk, A.; Kruszewski, M.; Lisowska, H.; Banasik-Nowak, A.; Rozga-Wijas, K.; Wojewodzka, M.; Slomkowski, S. Effect of surface modification of silica nanoparticles on toxicity and cellular uptake by human peripheral blood lymphocytes in vitro. Nanotoxicology 2013, 7, 235–250. [Google Scholar] [CrossRef]
- Demir, E.; Marcos, R. Antigenotoxic potential of boron nitride nanotubes. Nanotoxicology 2018, 12, 868–884. [Google Scholar] [CrossRef] [PubMed]
- Demir, E.; Marcos, R. Toxic and genotoxic effects of graphene and multi-walled carbon nanotubes. J. Toxicol. Environ. Health A Curr. Issues 2018, 81, 645–660. [Google Scholar] [CrossRef]
- Demir, E. Mechanisms and biological impacts of graphene and multi-walled carbon nanotubes on Drosophila melanogaster: Oxidative stress, genotoxic damage, phenotypic variations, locomotor behavior, parasitoid resistance, and cellular immune response. J. Appl. Toxicol. 2022, 42, 450–474. [Google Scholar] [CrossRef] [PubMed]
- Chromogenic LAL Assay. Available online: http://www.lonza.com/qcl1000 (accessed on 8 August 2022).
- Busvine, J.R. Techniques for Testing Insecticides, 2nd ed.; Commonwealth Agricultural Bureau: London, UK, 1971. [Google Scholar]
- WHO (World Health Organization). Criteria and Meaning of Tests for Determining the Susceptibility or Resistance of Insects to Insecticides-Diagnostic Test; World Health Organization: Geneva, Switzerland, 1981. [Google Scholar]
- WHO (World Health Organization). Instructions for Determining the Susceptibility or Resistance of Adult Mosquitos to Organochlorine, Organophosphate and Carbamate Insecticides-Diagnostic Test; World Health Organization: Geneva, Switzerland, 1981. [Google Scholar]
- Lindsley, D.L.; Zimm, G.G. The Genome of Drosophila Melanogaster; Academic Press: San Diego, CA, USA, 1992. [Google Scholar]
- Khot, L.R.; Sankaran, S.; Maja, J.M.; Ehsani, R.; Schuster, E. Application of nanomaterials in agricultural production and crop protection: A review. J. Crop Prot. 2012, 35, 64–70. [Google Scholar] [CrossRef]
- Mishra, M.; Sabat, D.; Ekka, B.; Sahu, S.; Unnikannan, P.; Dash, P. Oral intake of zirconia nanoparticle alters neuronal development and behaviour of Drosophila melanogaster. J. Nanoparticle Res. 2017, 19, 282. [Google Scholar] [CrossRef]
- Turna, F.; Aksakal, S.; Demir, E.; Kaya, B. Antigenotoxic effects of Resveratrol in somatic cells of Drosophila melanogaster. Fresenius Environ. Bull. 2014, 23, 2116–2125. [Google Scholar]
- Anand, A.S.; Gahlot, U.; Prasad, D.N.; Kohli, E. Aluminum oxide nanoparticles mediated toxicity, loss of appendages in progeny of Drosophila melanogaster on chronic exposure. Nanotoxicology 2019, 13, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.S.; Prasad, D.N.; Singh, S.B.; Kohli, E. Chronic exposure of zinc oxide nanoparticles causes deviant phenotype in Drosophila melanogaster. J. Hazard Mater. 2017, 327, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Priyadarsini, S.; Sahoo, S.K.; Sahu, S.; Mukherjee, S.; Hota, G.; Mishra, M. Oral administration of graphene oxide nano-sheets induces oxidative stress, genotoxicity, and behavioral teratogenicity in Drosophila melanogaster. Environ. Sci. Pollut. Res. 2019, 26, 19560–19574. [Google Scholar] [CrossRef] [PubMed]
- Pendleton, R.G.; Parvez, F.; Sayed, M.; Hillman, R. Effects of pharmacological agents upon a transgenic model of Parkinson’s disease in Drosophila melanogaster. J. Pharmacol. Exp. Ther. 2002, 300, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Sood, K.; Kaur, J.; Singh, H.; Arya, S.K.; Khatri, M. Comparative toxicity evaluation of graphene oxide (GO) and zinc oxide (ZnO) nanoparticles on Drosophila melanogaster. Toxicol. Rep. 2019, 6, 768–781. [Google Scholar] [CrossRef] [PubMed]
- Elmann, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Singh, M.P.; Krishna Reddy, M.M.; Mathur, N.; Saxena, D.K.; Kar Chowdhuri, D. Induction of hsp70, hsp60, hsp83 and hsp26 and oxidative stress markers in benzene, toluene and xylene exposed Drosophila melanogaster: Role of ROS generation. Toxicol. Appl. Pharmacol. 2009, 235, 226–243. [Google Scholar] [CrossRef]
- Tironi, V.A.; Tomás, M.C.; Añón, M.C. Lipid and protein deterioration during the chilled storage of minced sea salmon (Pseudopercis semifasciata). J. Sci. Food Agric. 2007, 87, 2239–2246. [Google Scholar] [CrossRef]
- Alaraby, M.; Demir, E.; Hernandez, A.; Marcos, R. Assessing potential harmful effects of CdSe quantum dots by using Drosophila melanogaster as in vivo model. Sci. Total Environ. 2015, 530, 66–75. [Google Scholar] [CrossRef]
- Alaraby, M.; Hernandez, A.; Annangi, B.; Demir, E.; Bach, J.; Rubio, L.; Creus, A.; Marcos, R. Antioxidant and antigenotoxic properties of CeO2 NPs and cerium sulphate: Studies with Drosophila melanogaster as a promising in vivo model. Nanotoxicology 2015, 9, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Demir, E. An in vivo study of nanorod, nanosphere, and nanowire forms of titanium dioxide using Drosophila melanogaster: Toxicity, cellular uptake, oxidative stress, and DNA damage. J. Toxicol. Environ. Health A Curr. Issues 2020, 83, 456–469. [Google Scholar] [CrossRef]
- Chen, J.; Li, J.; Huang, H.; Xi, R. Gene expression analysis of sorted cells by RNA-seq in Drosophila intestine. Bio-Protoc. 2016, 6, e2079. [Google Scholar] [CrossRef]
- Demir, E.; Marcos, R. Assessing the genotoxic effects of two lipid peroxidation products (4-oxo-2-nonenal and 4-hydroxy-hexenal) in haemocytes and midgut cells of Drosophila melanogaster larvae. Food Chem. Toxicol. 2017, 105, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef]
- Demir, E.; Turna, F.; Aksakal, S.; Emre, Y.; Emre, N.; Yağcı, A.; Kaya, B. The Comet assay using Rainbow trout (Oncorhynchus mykiss) for the detection of nutrient pollution generated from overfed fish farms in the Esen Stream. Fresenius Environ. Bull. 2015, 24, 3665–3671. [Google Scholar]
- Demir, E.; Turna, F.; Vales, G.; Kaya, B.; Creus, A.; Marcos, R. In vivo genotoxicity assessment of titanium, zirconium and aluminium nanoparticles, and their microparticulated forms, in Drosophila. Chemosphere 2013, 93, 2304–2310. [Google Scholar] [CrossRef]
- Frei, H.; Würgler, F.E. Statistical methods to decide whether mutagenicity test data from Drosophila assays indicate a positive, negative, or inconclusive results. Mutat. Res. 1988, 203, 297–308. [Google Scholar] [CrossRef]
- Frei, H.; Würgler, F.E. Optimal Experimental Design and Sample Size for the Statistical Evaluation of Data from Somatic Mutation and Recombination Tests (SMART) in Drosophila. Mutat. Res. 1995, 334, 247–258. [Google Scholar] [CrossRef]
- Finney, D.J. Probit Analysis; Cambridge University Press: New York, NY, USA, 1971. [Google Scholar]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demir, E.; Kansız, S.; Doğan, M.; Topel, Ö.; Akkoyunlu, G.; Kandur, M.Y.; Turna Demir, F. Hazard Assessment of the Effects of Acute and Chronic Exposure to Permethrin, Copper Hydroxide, Acephate, and Validamycin Nanopesticides on the Physiology of Drosophila: Novel Insights into the Cellular Internalization and Biological Effects. Int. J. Mol. Sci. 2022, 23, 9121. https://doi.org/10.3390/ijms23169121
Demir E, Kansız S, Doğan M, Topel Ö, Akkoyunlu G, Kandur MY, Turna Demir F. Hazard Assessment of the Effects of Acute and Chronic Exposure to Permethrin, Copper Hydroxide, Acephate, and Validamycin Nanopesticides on the Physiology of Drosophila: Novel Insights into the Cellular Internalization and Biological Effects. International Journal of Molecular Sciences. 2022; 23(16):9121. https://doi.org/10.3390/ijms23169121
Chicago/Turabian StyleDemir, Eşref, Seyithan Kansız, Mehmet Doğan, Önder Topel, Gökhan Akkoyunlu, Muhammed Yusuf Kandur, and Fatma Turna Demir. 2022. "Hazard Assessment of the Effects of Acute and Chronic Exposure to Permethrin, Copper Hydroxide, Acephate, and Validamycin Nanopesticides on the Physiology of Drosophila: Novel Insights into the Cellular Internalization and Biological Effects" International Journal of Molecular Sciences 23, no. 16: 9121. https://doi.org/10.3390/ijms23169121
APA StyleDemir, E., Kansız, S., Doğan, M., Topel, Ö., Akkoyunlu, G., Kandur, M. Y., & Turna Demir, F. (2022). Hazard Assessment of the Effects of Acute and Chronic Exposure to Permethrin, Copper Hydroxide, Acephate, and Validamycin Nanopesticides on the Physiology of Drosophila: Novel Insights into the Cellular Internalization and Biological Effects. International Journal of Molecular Sciences, 23(16), 9121. https://doi.org/10.3390/ijms23169121






