PxTret1-like Affects the Temperature Adaptability of a Cosmopolitan Pest by Altering Trehalose Tissue Distribution
Abstract
1. Introduction
2. Results
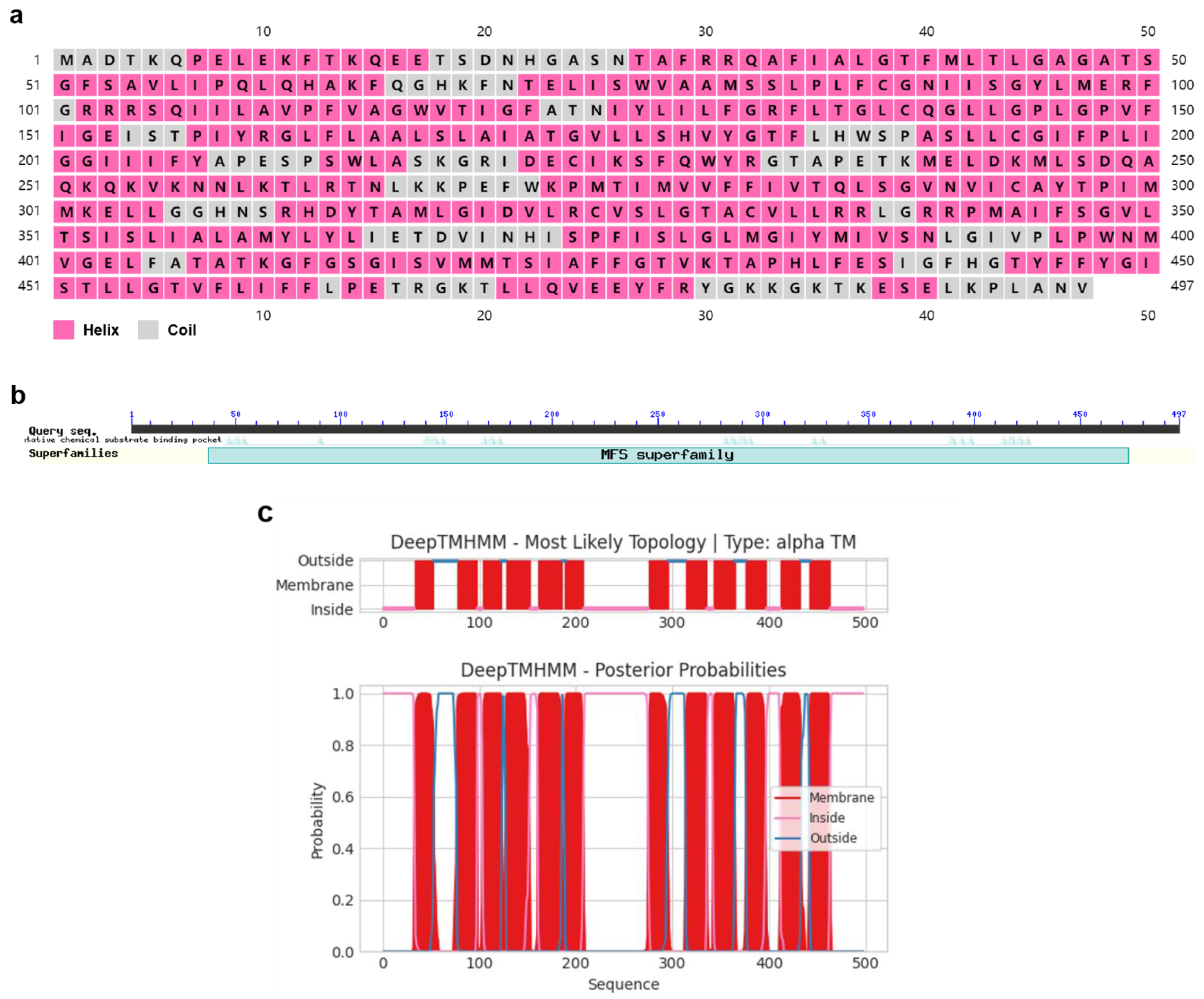
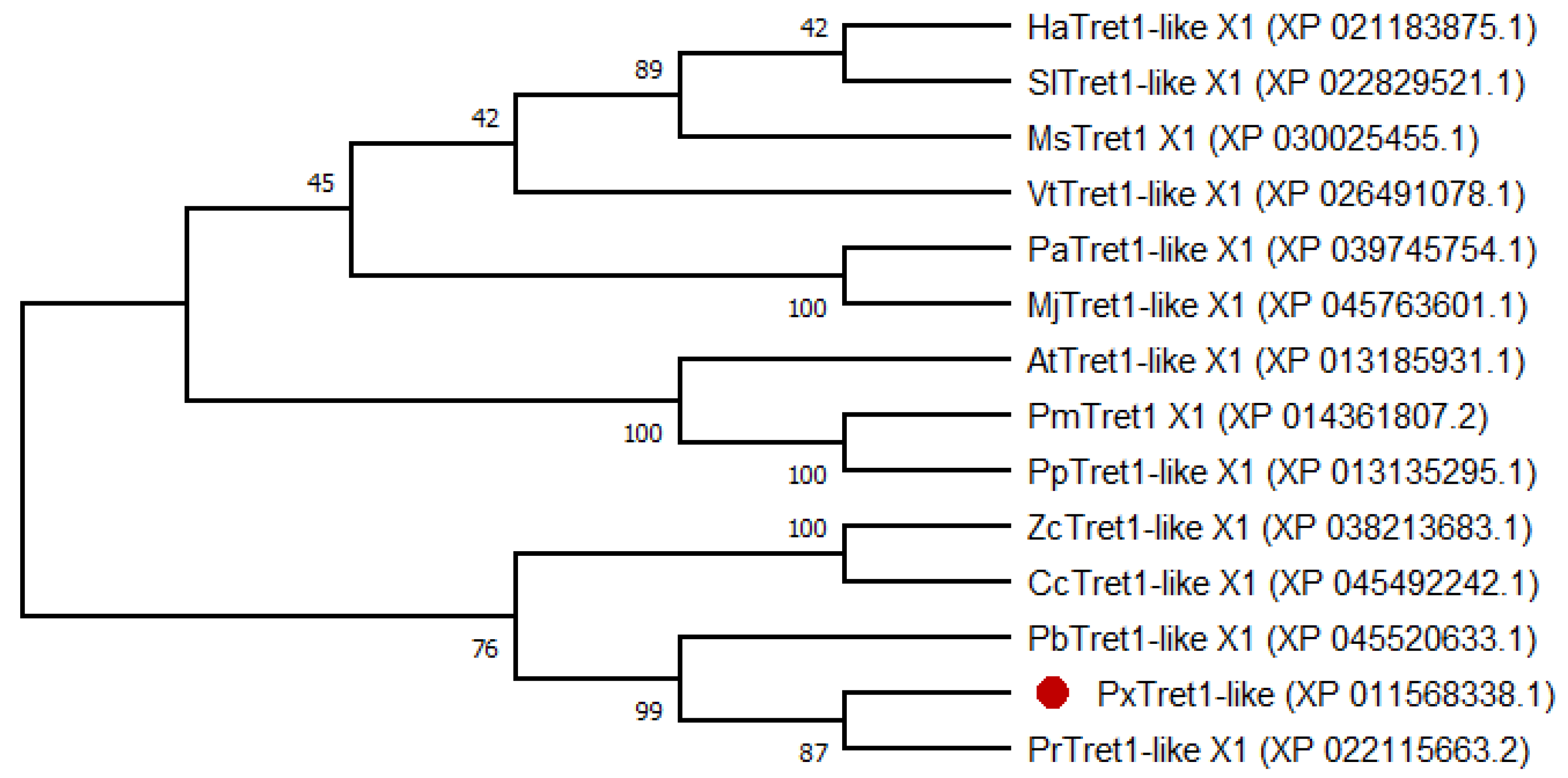
2.1. Identification and Characterization of PxTret1-like
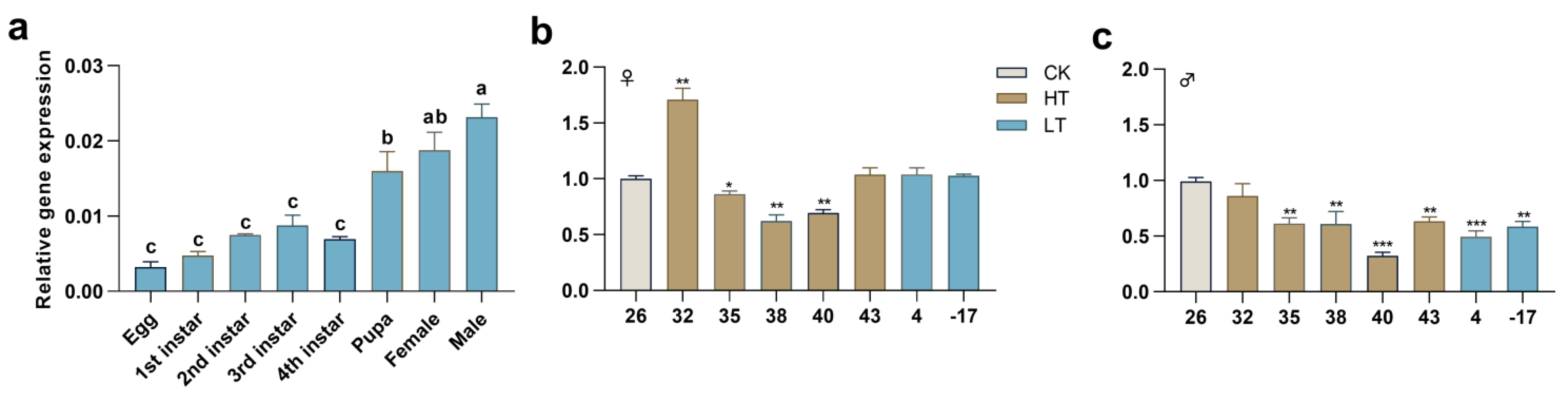
2.2. Expression Profiles of PxTret1-like in Different Stages and at Extreme Temperature
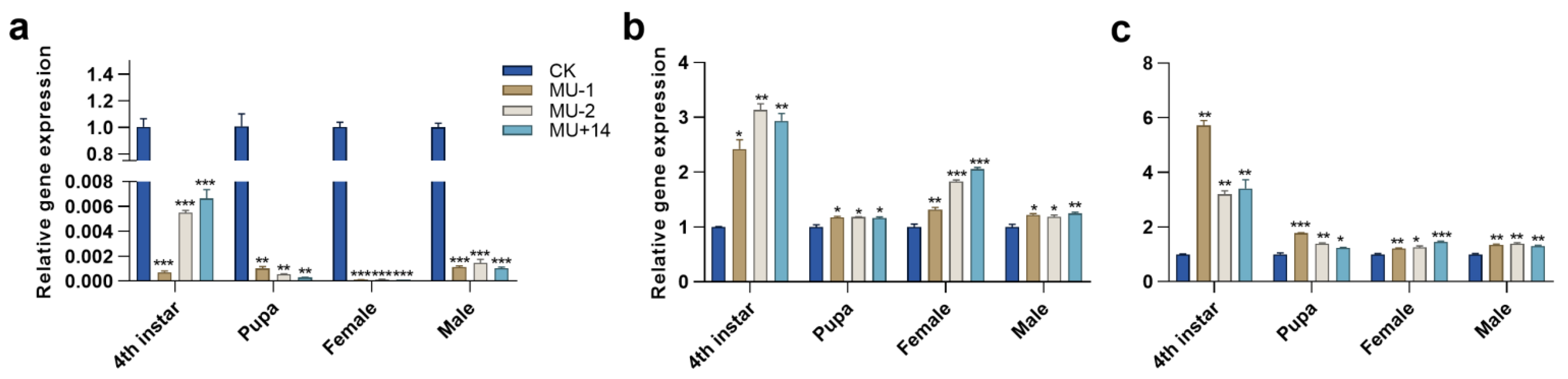
2.3. Establishment of Homozygous PxTret1-like Knockout Strains
2.4. Expression Profiles of TPS and TRE
2.5. Trehalose, Glucose and Trehalose Metabolic Enzymes
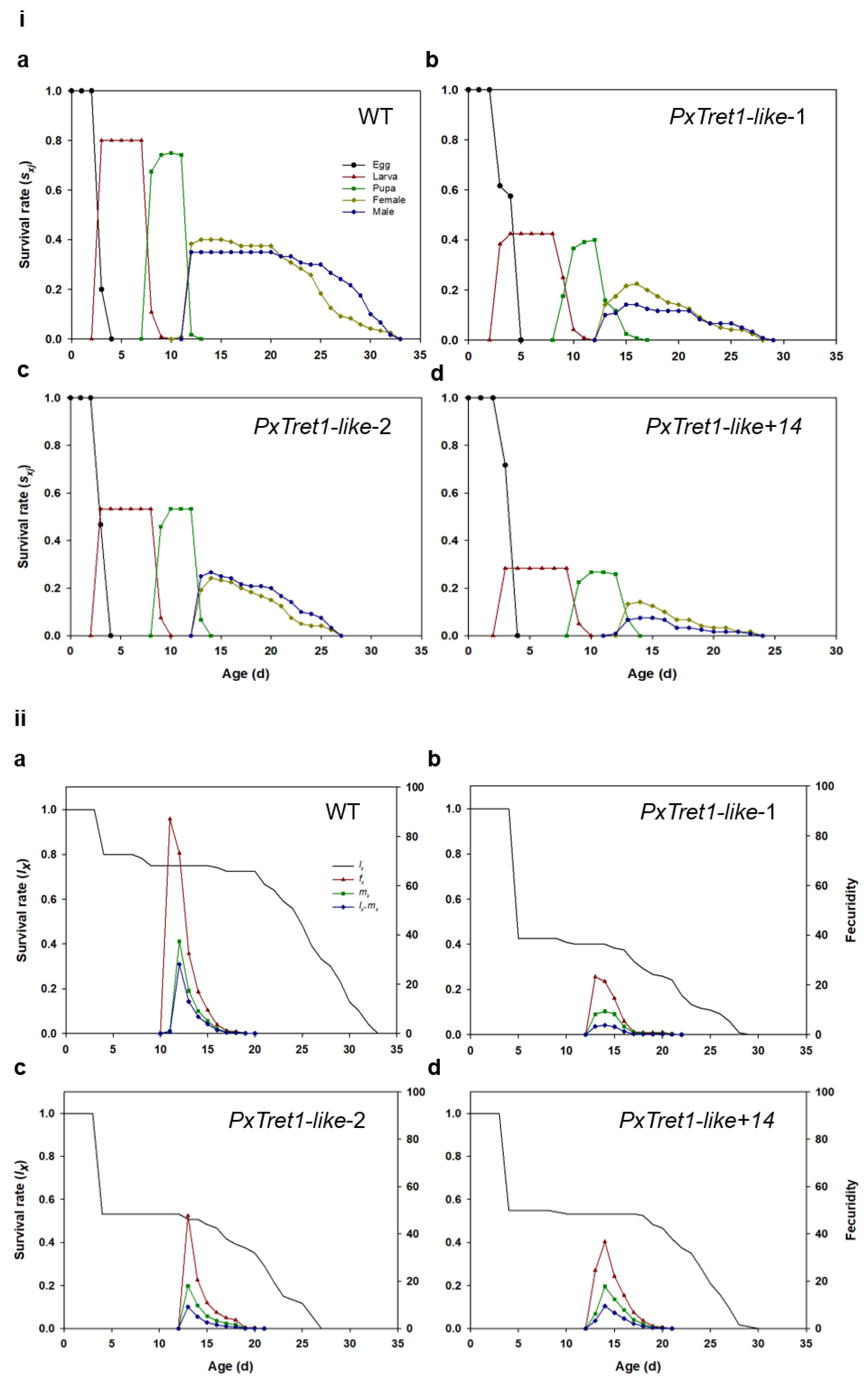
2.6. Age-Stage-Specific Sex Life Table
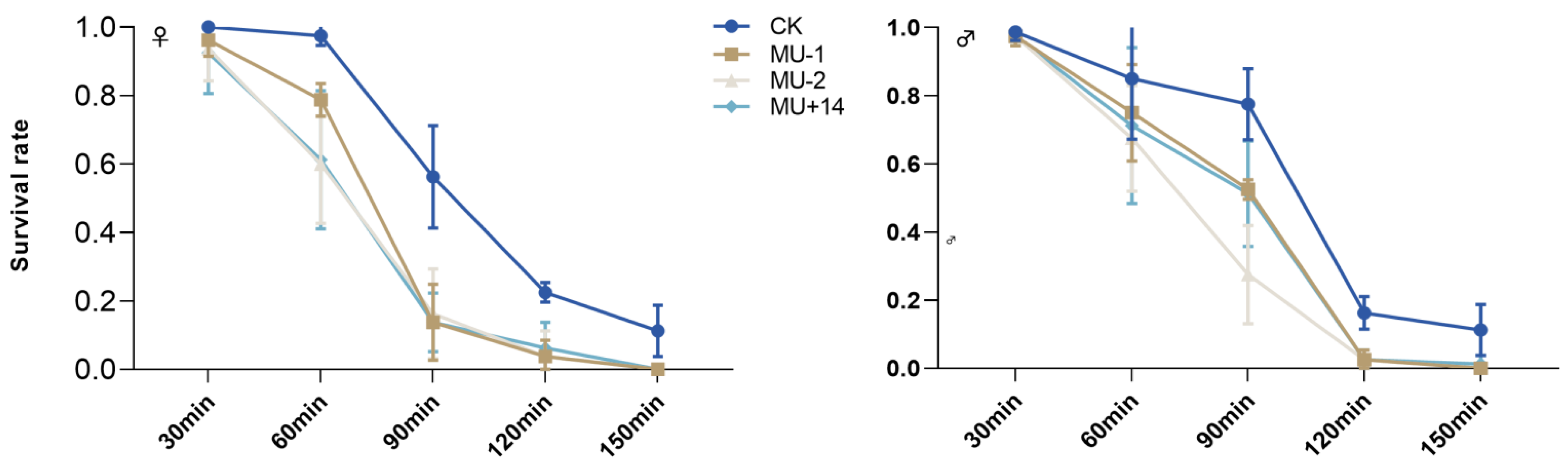
2.7. Response to Extreme Temperature
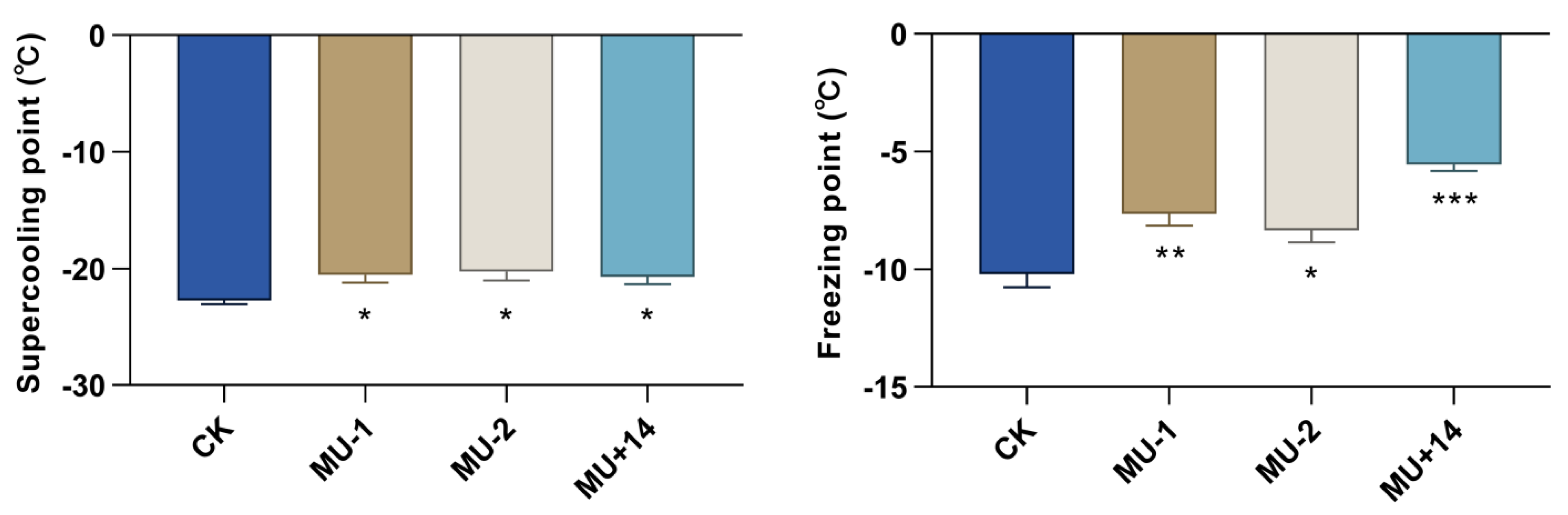
2.8. Supercooling Point and Freezing Point
3. Discussion
4. Materials and Methods
4.1. Insect Strains and Rearing
4.2. Gene Cloning
4.3. Sequence Analysis and Phylogenetic Tree Construction
4.4. Expression Profiling of PxTret1-like
4.4.1. Expression Patterns of Different Stages
4.4.2. Expression Patterns at Extreme Temperatures
4.5. sgRNA Design and Synthesis
4.5.1. sgRNA Design and Off-Target Analysis
4.5.2. sgRNA Synthesis and Purification
4.5.3. sgRNA/Cas9 Protein Microinjection
4.6. Mutation Screening
4.7. Expression Patterns of TPS and TRE
4.8. Determination of the of Trehalose, Glucose and Trehalose Metabolic Enzymes
4.9. Age-Stage-Specific Sex Life Table
4.10. Response to Extreme Temperature
4.11. Supercooling Point and Freezing Point
4.12. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | WT vs. PxTret1-like-1 | WT vs. PxTret1-like-2 | WT vs. PxTret1-like+14 |
|---|---|---|---|
| Egg (d) | p = 0.02347 | p = 1.00000 | p = 1.00000 |
| Larva (d) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| Pupa (d) | p = 0.09222 | p = 0.03240 | p = 0.00009 |
| Preadult All (d) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| Adult F (d) | p < 0.00001 | p < 0.00001 | p = 0.00044 |
| Adult M (d) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| Adult Al l(d) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| longevity All (d) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| APOP (d) | p = 0.00874 | p = 0.05235 | p = 0.0277 |
| TPOP (d) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| Oviposition (d) | p = 0.00062 | p = 0.05839 | p = 0.36337 |
| Fecundity (eggs) | p < 0.00001 | p < 0.00001 | p < 0.00001 |
| r (d−1) | p = 0.00008 | p < 0.00001 | p < 0.00001 |
| R0 (eggs/female) | p < 0.00001 | p = 0.00002 | p = 0.00100 |
| T (d) | p = 0.00003 | p < 0.00001 | p < 0.00001 |
| λ (d−1) | p = 0.00003 | p < 0.00001 | p < 0.00001 |
References
- Bauerfeind, S.S.; Fischer, K. Simulating climate change: Temperature extremes but not means diminish performance in a widespread butterfly. Popul. Ecol. 2014, 56, 239–250. [Google Scholar] [CrossRef]
- Sala, O.E.; Chapin, F.S.; Armesto, J.J.; Berlow, E.; Bloomfield, J.; Dirzo, R.; Huber–Sanwald, E.; Huenneke, L.F.; Jackson, R.B.; Kinzig, A.; et al. Global biodiversity scenarios for the year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef] [PubMed]
- Neven, L.G. Physiological responses of insects to heat. Postharvest Biol. Technol. 2000, 21, 103–111. [Google Scholar] [CrossRef]
- Horne, C.R.; Hirst, A.G.; Atkinson, D. Insect temperature–body size trends common to laboratory, latitudinal and seasonal gradients are not found across altitudes. Funct. Ecol. 2018, 32, 948–957. [Google Scholar] [CrossRef]
- Brakefield, P.M.; Willmer, P.G. The basis of thermal melanism in the ladybird Adalia bipunctata: Differences in reflectance and thermal properties between the morphs. Heredity 1985, 54, 9–14. [Google Scholar] [CrossRef]
- Ma, C.S.; Ma, G.; Pincebourde, S. Survive a warming climate: Insect responses to extreme high temperatures. Annu. Rev. Entomol. 2021, 66, 163–184. [Google Scholar] [CrossRef]
- Lale, N.E.S.; Vidal, S. Simulation studies on the effects of solar heat on egg-laying, development and survival of Callosobruchus maculatus (F.) and Callosobruchus subinnotatus (Pic) in stored bambara groundnut Vigna subterranea (L.) Verdcourt. J. Stored Prod. Res. 2003, 39, 447–458. [Google Scholar] [CrossRef]
- Bijlsma, R.; Loeschcke, V. Environmental stress, adaptation and evolution: An overview. J. Evol. Biol. 2005, 18, 744–749. [Google Scholar] [CrossRef]
- Mallard, F.; Nolte, V.; Tobler, R.; Kapun, M.; Schlötterer, C. A simple genetic basis of adaptation to a novel thermal environment results in complex metabolic rewiring in Drosophila. Genome Biol. 2018, 19, 119. [Google Scholar] [CrossRef]
- Polidori, C.; Gutiérrez–Cánovas, C.; Sánchez, E.; Tormos, J.; Castro, L.; Sánchez–Fernández, D. Climate change-driven body size shrinking in a social wasp. Ecol. Entomol. 2020, 45, 130–141. [Google Scholar] [CrossRef]
- Gibo, D.L.; Pallett, M.J. Soaring flight of monarch butterflies, Danaus plexippus (Lepidoptera: Danaidae), during the late summer migration in southern Ontario. Can. J. Zool. 1979, 57, 1393–1401. [Google Scholar] [CrossRef]
- Wolfe, G.R.; Hendrix, D.L.; Salvucci, M.E. A thermoprotective role for sorbitol in the silverleaf whitefly, Bemisia argentifolii. J. Insect Physiol. 1998, 44, 597–603. [Google Scholar] [CrossRef]
- Fiehn, O. Metabolomics—The link between genotypes and phenotypes. Funct. Genom. 2002, 48, 155–171. [Google Scholar]
- Viant, M.R. Recent developments in environmental metabolomics. Mol. Biosyst. 2008, 4, 980–986. [Google Scholar] [CrossRef] [PubMed]
- Goto, M.; Sekine, Y.; Outa, H.; Hujikura, M.; Suzuki, K. Relationships between cold hardiness and diapause, and between glycerol and free amino acid contents in overwintering larvae of the oriental corn borer, Ostrinia furnacalis. J. Insect Physiol. 2001, 47, 157–165. [Google Scholar] [CrossRef]
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J.; et al. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Hottiger, T.; Boller, T.; Wiemken, A. Rapid changes of heat and desiccation tolerance correlated with changes of trehalose content in Saccharomyces cerevisiae cells subjected to temperature shifts. FEBS Lett. 1987, 220, 113–115. [Google Scholar] [CrossRef]
- Tang, B.; Qin, Z.; Shi, Z.K.; Wang, S.; Guo, X.J.; Wang, S.G.; Zhang, F. Trehalase in Harmonia axyridis (Coleoptera: Coccinellidae): Effects on beetle locomotory activity and the correlation with trehalose metabolism under starvation conditions. Appl. Entomol. Zool. 2014, 49, 255–264. [Google Scholar] [CrossRef]
- Feofilova, E.P.; Usov, A.I.; Mysyakina, I.S.; Kochkina, G.A. Trehalose: Chemical structure, biological functions, and practical application. Microbiology 2014, 83, 184–194. [Google Scholar] [CrossRef]
- Salvucci, M.E.; Hendrix, D.L.; Wolfe, G.R. Effect of high temperature on the metabolic processes affecting sorbitol synthesis in the silverleaf whitefly, Bemisia argentifolii. J. Insect Physiol. 1999, 45, 21–27. [Google Scholar] [CrossRef]
- Khani, A.; Moharramipour, S.; Barzegar, M. Cold tolerance and trehalose accumulation in overwintering larvae of the codling moth, Cydia pomonella (Lepidoptera: Tortricidae). Eur. J. Entomol. 2007, 104, 385–392. [Google Scholar] [CrossRef]
- Wen, X.; Wang, S.; Duman, J.G.; Henling, L.M. Antifreeze proteins govern the precipitation of trehalose in a freezing–avoiding insect at low temperature. Proc. Natl. Acad. Sci. USA 2016, 113, 6683–6688. [Google Scholar] [CrossRef] [PubMed]
- Kikuta, S.; Nakamura, Y.; Hattori, M.; Sato, R.; Kikawada, T.; Noda, H. Herbivory-induced glucose transporter gene expression in the brown planthopper, Nilaparvata lugens. Insect Biochem. Mol. Biol. 2015, 64, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.B.; Wright, J.P.; Mittler, T.E. Osmoregulation by the aphid Myzus persicae: A physiological role for honeydew oligosaccharides. J. Insect Physiol. 1984, 30, 387–393. [Google Scholar] [CrossRef]
- Ashford, D.A.; Smith, W.A.; Douglas, A.E. Living on a high sugar diet: The fate of sucrose ingested by a phloem-feeding insect, the pea aphid Acyrthosiphon pisum. J. Insect Physiol. 2000, 46, 335–341. [Google Scholar] [CrossRef]
- Price, D.R.G.; Wilkinson, H.S.; Gatehouse, J.A. Functional expression and characterisation of a gut facilitative glucose transporter, NlHT1, from the phloem-feeding insect Nilaparvata lugens (rice brown planthopper). Insect Biochem. Mol. Biol. 2007, 37, 1138–1148. [Google Scholar] [CrossRef]
- Fraga, A.; Ribeiro, L.; Lobato, M.; Santos, V.; Silva, J.R.; Gomes, H.; da Cunha Moraes, J.L.; de Souza Menezes, J.; de Oliveira, C.J.L.; Campos, E.; et al. Glycogen and glucose metabolism are essential for early embryonic development of the red flour beetle Tribolium castaneum. PLoS ONE 2013, 8, e65125. [Google Scholar] [CrossRef]
- Ge, L.Q.; Jiang, Y.P.; Xia, T.; Song, Q.S.; Stanley, D.; Kuai, P.; Lu, X.L.; Yang, G.Q.; Wu, J.C. Silencing a sugar transporter gene reduces growth and fecundity in the brown planthopper, Nilaparvata lugens (Stål) (Hemiptera: Delphacidae). Sci. Rep. 2015, 5, 12194. [Google Scholar] [CrossRef]
- Tang, B.; Wang, S.; Wang, S.G.; Wang, H.J.; Zhang, J.Y.; Cui, S.Y. Invertebrate trehalose-6-phosphate synthase gene: Genetic architecture, biochemistry, physiological function, and potential applications. Front. Physiol. 2018, 9, 30. [Google Scholar] [CrossRef]
- De Castro, A.G.; Tunnacliffe, A. Intracellular trehalose improves osmotolerance but not desiccation tolerance in mammalian cells. FEBS Lett. 2000, 487, 199–202. [Google Scholar] [CrossRef]
- Stambuk, B.U.; Panek, A.D.; Crowe, J.H.; Crowe, L.M.; de Araujo, P.S. Expression of high-affinity trehalose–H+ symport in Saccharomyces cerevisiae. Biochim. Biophys. Acta Gen. Subj. 1998, 1379, 118–128. [Google Scholar] [CrossRef]
- Kikawada, T.; Saito, A.; Kanamori, Y.; Okuda, T. Trehalose transporter 1, a facilitated and high-capacity trehalose transporter, allows exogenous trehalose uptake into cells. Proc. Natl. Acad. Sci. USA 2007, 104, 11585–11590. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, M.; Niimi, T.; Su, Z.H.; Yaginuma, T. Trehalase from male accessory gland of an insect, Tenebrio molitor. cDNA sequencing and developmental profile of the gene expression. Biochem. J. 1992, 288, 19–22. [Google Scholar] [PubMed]
- Tang, B.; Yang, M.M.; Shen, Q.D.; Xu, Y.X.; Wang, H.J.; Wang, S.G. Suppressing the activity of trehalase with validamycin disrupts the trehalose and chitin biosynthesis pathways in the rice brown planthopper, Nilaparvata lugens. Pestic. Biochem. Phys. 2017, 137, 81–90. [Google Scholar]
- Talekar, N.S.; Shelton, A.M. Biology, ecology, and management of the diamondback moth. Annu. Rev. Entomol. 1993, 38, 275–301. [Google Scholar] [CrossRef]
- Furlong, M.J.; Wright, D.J.; Dosdall, L.M. Diamondback moth ecology and management: Problems, progress, and prospects. Annu. Rev. Entomol. 2013, 58, 517–541. [Google Scholar]
- You, M.S.; Yue, Z.; He, W.Y.; Yang, X.H.; Yang, G.; Xie, M.; Zhan, D.L.; Baxter, S.W.; Vasseur, L.; Gurr, G.M.; et al. A heterozygous moth genome provides insights into herbivory and detoxification. Nat. Genet. 2013, 45, 220–225. [Google Scholar] [CrossRef]
- Deutsch, C.A.; Tewksbury, J.J.; Huey, R.B.; Sheldon, K.S.; Ghalambor, C.K.; Haak, D.C.; Martin, P.R. Impacts of climate warming on terrestrial ectotherms across latitude. Proc. Natl. Acad. Sci. USA 2008, 105, 6668–6672. [Google Scholar] [CrossRef]
- Chen, Y.T.; Liu, Z.X.; Régnière, J.; Vasseur, L.; Lin, J.; Huang, S.G.; Ke, F.S.; Chen, S.P.; Li, J.Y.; Huang, J.L.; et al. Large-scale genome-wide study reveals climate adaptive variability in a cosmopolitan pest. Nat. Commun. 2021, 12, 7206. [Google Scholar]
- You, M.S.; Ke, F.S.; You, S.J.; Wu, Z.Y.; Liu, Q.F.; He, W.Y.; Baxter, S.W.; Yuchi, Z.G.; Vasseur, L.; Gurr, G.M.; et al. Variation among 532 genomes unveils the origin and evolutionary history of a global insect herbivore. Nat. Commun. 2020, 11, 2321. [Google Scholar]
- Kang, L.; Chen, B.; Wei, J.N.; Liu, T.X. Roles of thermal adaptation and chemical ecology in Liriomyza distribution and control. Annu. Rev. Entomol. 2009, 54, 127–145. [Google Scholar] [CrossRef] [PubMed]
- Misof, B.; Liu, S.; Meusemann, K.; Peters, R.; Donath, A.; Mayrt, C.; Frandsen, P.; Ware, J.; Flouri, T.; Beutel, R.; et al. Phylogenomics resolves the timing and pattern of insect evolution. Science 2014, 346, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, S.S.; Liu, Y.K.; Lu, Y.T.; Zhou, M.; Wang, S.; Wang, S.G. The effect of different dietary sugars on the development and fecundity of Harmonia axyridis. Front. Physiol. 2020, 11, 574851. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, Y.; Saito, A.; Hagiwara–Komoda, Y.; Tanaka, D.; Mitsumasu, K.; Kikuta, S.; Watanabe, M.; Cornette, R.; Kikawada, T.; Okuda, T. The trehalose transporter 1 gene sequence is conserved in insects and encodes proteins with different kinetic properties involved in trehalose import into peripheral tissues. Insect Biochem. Mol. Biol. 2010, 40, 30–37. [Google Scholar] [CrossRef]
- Yang, Z.Z.; Xia, J.X.; Pan, H.P.; Gong, C.; Xie, W.; Guo, Z.J.; Zheng, H.X.; Yang, X.; Yang, F.S.; Wu, Q.J.; et al. Genome-wide characterization and expression profiling of sugar transporter family in the whitefly, Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae). Front. Physiol. 2017, 8, 322. [Google Scholar] [CrossRef]
- Marten, A.D.; Stothard, A.I.; Kalera, K.; Swarts, B.M.; Conway, M.J. Validamycin A delays development and prevents flight in Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 2020, 57, 1096–1103. [Google Scholar] [CrossRef]
- Ge, L.Q.; Zhao, K.F.; Huang, L.J.; Wu, J.C. The effects of triazophos on the trehalose content, trehalase activity and their gene expression in the brown planthopper Nilaparvata lugens (Stål) (Hemiptera: Delphacidae). Pestic. Biochem. Phys. 2011, 100, 172–181. [Google Scholar] [CrossRef][Green Version]
- Zhang, B.C.; Jiang, C.J.; An, C.J.; Zhang, Q.W.; Zhao, Z.W. Variations in fuel use in the flight muscles of wing-dimorphic Gryllus firmus and implications for morph–specific dispersal. Environ. Entomol. 2011, 40, 1566–1571. [Google Scholar] [CrossRef]
- Shi, Z.K.; Liu, X.J.; Xu, Q.Y.; Qin, Z.; Wang, S.; Zhang, F.; Wang, S.G.; Tang, B. Two novel soluble trehalase genes cloned from Harmonia axyridis and regulation of the enzyme in a rapid changing temperature. Comp. Biochem. Phys. B. 2016, 198, 10–18. [Google Scholar] [CrossRef]
- Thorat, L.J.; Gaikwad, S.M.; Nath, B.B. Trehalose as an indicator of desiccation stress in Drosophila melanogaster larvae: A potential marker of anhydrobiosis. Biochem. Bioph. Res. Commun. 2012, 419, 638–642. [Google Scholar] [CrossRef]
- Becker, A.; Schlöder, P.; Steele, J.E.; Wegener, G. The regulation of trehalose metabolism in insects. Experientia 1996, 52, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Nardelli, A.; Vecchi, M.; Mandrioli, M.; Manicardi, G.C. The evolutionary history and functional divergence of trehalase (treh) genes in insects. Front. Physiol. 2019, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Shukla, E.; Thorat, L.J.; Nath, B.B.; Gaikwad, S.M. Insect trehalase: Physiological significance and potential applications. Glycobiology 2015, 25, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Matsuda, H.; Kubo, H.; Nishimura, T. Molecular characterization of Tps1 and Treh genes in Drosophila and their role in body water homeostasis. Sci. Rep. 2016, 6, 30582. [Google Scholar] [CrossRef] [PubMed]
- Santos–Ortega, Y.; Killiny, N. Silencing of sucrose hydrolase causes nymph mortality and disturbs adult osmotic homeostasis in Diaphorina citri (Hemiptera: Liviidae). Insect Biochem. Molec. 2018, 101, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Ozalp, P.; Emre, M. The effects of carbohydrates upon the survival and reproduction of adult female Pimpla turionellae L. (Hym., Ichneumonidae). J. Appl. Ent. 2001, 125, 177–180. [Google Scholar] [CrossRef]
- Li, Y.; Wang, S.S.; Wang, S.; Wang, S.G.; Tang, B.; Liu, F. Involvement of glucose transporter 4 in ovarian development and reproductive maturation of Harmonia axyridis (Coleoptera: Coccinellidae). Insect Sci. 2022, 29, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Kilby, B.A.; Candy, D. Insect Biochemistry and Function; Chapman and Hall: London, UK, 1975. [Google Scholar]
- Werren, J.H. Labile sex ratios in wasps and bees. Bioscience 1987, 37, 498–506. [Google Scholar] [CrossRef]
- Coskun, M.; Emre, I. Role of lipids, amino acids, and sucrose on the total adult and female emergence, and content of glycogen and protein in Pimpla turionellae (Hymenoptera: Ichneumonidae). Ann. Entomol. Soc. Am. 2015, 108, 820–826. [Google Scholar] [CrossRef]
- Huang, J.H.; Lee, H.J. RNA interference unveils functions of the hypertrehalosemic hormone on cyclic fluctuation of hemolymph trehalose and oviposition in the virgin female Blattella germanica. J. Insect physiol. 2011, 57, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Yamada, T.; Yoshida, M.; Nishimura, T. Flies without trehalose. J. Biol. Chem. 2015, 290, 1244–1255. [Google Scholar] [CrossRef] [PubMed]
- Li, J.X.; Cao, Z.; Guo, S.; Tian, Z.; Liu, W.; Zhu, F.; Wang, X.P. Molecular characterization and functional analysis of two trehalose transporter genes in the cabbage beetle, Colaphellus bowringi. J. Asia Pac. Entomol. 2020, 23, 627–633. [Google Scholar] [CrossRef]
- Lü, X.; Han, S.; Li, Z.; Li, L.Y.; Li, J. Gene characterization and enzymatic activities related to trehalose metabolism of in vitro reared Trichogramma dendrolimi Matsumura (Hymenoptera: Trichogrammatidae) under sustained cold stress. Insects 2020, 11, 767. [Google Scholar] [CrossRef]
- Liu, K.; Dong, Y.; Huang, Y.; Rasgon, J.L.; Agre, P. Impact of trehalose transporter knockdown on Anopheles gambiae stress adaptation and susceptibility to Plasmodium falciparum infection. Proc. Natl. Acad. Sci. USA 2013, 110, 17504–17509. [Google Scholar] [CrossRef] [PubMed]
- Hwang, W.Y.; Fu, Y.; Reyon, D.; Maeder, M.L.; Tsai, S.Q.; Sander, J.D.; Peterson, R.T.; Yeh, J.J.; Joung, J.K. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.J.; Xue, J.; Lu, B.O.; Zhang, X.C.; Zhuo, J.C.; He, S.F.; Ma, X.F.; Jiang, Y.Q.; Fan, H.W.; Xu, J.Y.; et al. Two insulin receptors determine alternative wing morphs in planthoppers. Nature 2015, 519, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.H.; Bellés, X.; Lee, H.J. Functional characterization of hypertrehalosemic hormone receptor in relation to hemolymph trehalose and to oxidative stress in the cockroach Blattella germanica. Front. Endocrinol. 2012, 2, 114. [Google Scholar] [CrossRef] [PubMed]
- Chi, H. Life-table analysis incorporating both sexes and variable development rates among individuals. Environ. Entomol. 1988, 17, 26–34. [Google Scholar] [CrossRef]
- Hao, S.G.; Kang, L. Supercooling capacity and cold hardiness of the eggs of the grasshopper Chorthippus fallax (Orthoptera: Acrididae). Eur. J. Entomol. 2004, 101, 231–236. [Google Scholar]


| Parameter | WT | PxTret1-like-1 | PxTret1-like-2 | PxTret-like+14 |
|---|---|---|---|---|
| Egg (d) | 3.00 ± 0.00 | 3.10 ± 0.04 * | 3.00 ± 0.00 | 3.00 ± 0.00 |
| Larva (d) | 5.11 ± 0.04 | 6.58 ± 0.08 * | 6.14 ± 0.04 * | 6.44 ± 0.08 * |
| Pupa (d) | 3.90 ± 0.04 | 4.08 ± 0.10 * | 3.98 ± 0.02 | 4.16 ± 0.05 * |
| Preadult All (d) | 12.01 ± 0.02 | 13.74 ± 0.16 * | 13.13 ± 0.04 * | 13.59 ± 0.08 * |
| Adult F (d) | 13.27 ± 0.57 | 8.00 ± 0.73 * | 8.07 ± 0.62 * | 10.50 ± 0.56 * |
| Adult M (d) | 16.60 ± 0.49 | 9.21 ± 1.06 * | 9.19 ± 0.65 * | 11.07 ± 0.53 * |
| Adult All (d) | 14.82 ± 0.42 | 8.49 ± 0.61 * | 8.66 ± 0.45 * | 10.75 ± 0.39 * |
| longevity All (d) | 21.36 ± 0.93 | 11.97 ± 0.80 * | 13.27 ± 0.84 * | 14.94 ± 0.95 * |
| APOP (d) | 0.00 ± 0.00 | 0.53 ± 0.19 * | 0.19 ± 0.09 | 0.17 ± 0.08 * |
| TPOP (d) | 11.98 ± 0.02 | 13.83 ± 0.23 * | 13.33 ± 0.13 * | 13.83 ± 0.13 * |
| Oviposition (d) | 4.93 ± 0.24 | 3.26 ± 0.41 * | 4.07 ± 0.38 | 4.60 ± 0.28 |
| Fecundity (eggs) | 151.9 ± 7.70 | 75.95 ± 13.88 * | 88.04 ± 9.93 * | 92.29 ± 7.45 * |
| Parameter | WT | PxTret1-like-1 | PxTret1-like-2 | PxTret-like+14 |
|---|---|---|---|---|
| r (d−1) | 0.29 ± 0.01 | 0.16 ± 0.02 * | 0.20 ± 0.01 * | 0.21 ± 0.01 * |
| R0 (eggs/female) | 54.43 ± 7.19 | 12.03 ± 3.31 * | 19.81 ± 4.03 * | 26.92 ± 4.38 * |
| T (d) | 13.68 ± 0.05 | 15.84 ± 0.23 * | 14.94 ± 0.01 * | 15.70 ± 0.11 * |
| λ (d−1) | 1.34 ± 0.01 | 1.18 ± 0.02 * | 1.22 ± 0.02 * | 1.23 ± 0.01 * |
| Primer Name | Primer Sequence 5′-3′ | Position | |
|---|---|---|---|
| PCR | CDS F | ATGGCGGACACGAAACAGC | 1–19 |
| CDS R | TCAAACATTAGCTAAAGGCTTCAATTC | 1468–1494 | |
| Quantitative PCR | Q-PxTret1-like F | GCTTCTCCGCCGTGCTTATCC | 2016–2036 |
| Q-PxTret1-like R | AGAACAGTGGAAGCGATGACA | 2307–2327 | |
| Q-PxTPS F | GTCCGACCCCAATGACAACACG | 827–848 | |
| Q-PxTPS R | CAGCGGCCAGAAGGTCCC | 2913–2930 | |
| Q-PxTRE1-1 F | CAGCAGCAGCTCTACTCCATCATC | 6369–6392 | |
| Q-PxTRE1-1 R | GTGTCGCGCATCTCCGACAG | 10,015–10,034 | |
| Q-PxTRE1-2 F | CTCCGAGGACTACGAGAATGCG | 885–906 | |
| Q-PxTRE1-2 R | GATGGCGTTCTGGTCGACGG | 1087–1106 | |
| sgRNA synthesis | SgRNA F a | TAATACGACTCACTATAGGGTCGCCGGCTGGGTCACCATG | 2405–2424 |
| TTTTAGAGCTAGAAATAGCAAGTTAAAATAAGGCTAGTCC | |||
| sgRNA-ComR b | AAAAGCACCGACTCGGTGCCACTTTTTCAAGTTGATAAC | - | |
| GGACTAGCCTTATTTTAACTTGCTATTTCTAGCTCTAAAA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, H.; Lei, G.; Chen, Y.; You, M.; You, S. PxTret1-like Affects the Temperature Adaptability of a Cosmopolitan Pest by Altering Trehalose Tissue Distribution. Int. J. Mol. Sci. 2022, 23, 9019. https://doi.org/10.3390/ijms23169019
Zhou H, Lei G, Chen Y, You M, You S. PxTret1-like Affects the Temperature Adaptability of a Cosmopolitan Pest by Altering Trehalose Tissue Distribution. International Journal of Molecular Sciences. 2022; 23(16):9019. https://doi.org/10.3390/ijms23169019
Chicago/Turabian StyleZhou, Huiling, Gaoke Lei, Yanting Chen, Minsheng You, and Shijun You. 2022. "PxTret1-like Affects the Temperature Adaptability of a Cosmopolitan Pest by Altering Trehalose Tissue Distribution" International Journal of Molecular Sciences 23, no. 16: 9019. https://doi.org/10.3390/ijms23169019
APA StyleZhou, H., Lei, G., Chen, Y., You, M., & You, S. (2022). PxTret1-like Affects the Temperature Adaptability of a Cosmopolitan Pest by Altering Trehalose Tissue Distribution. International Journal of Molecular Sciences, 23(16), 9019. https://doi.org/10.3390/ijms23169019






