New Pyrrole Derivatives as Promising Biological Agents: Design, Synthesis, Characterization, In Silico, and Cytotoxicity Evaluation
Abstract
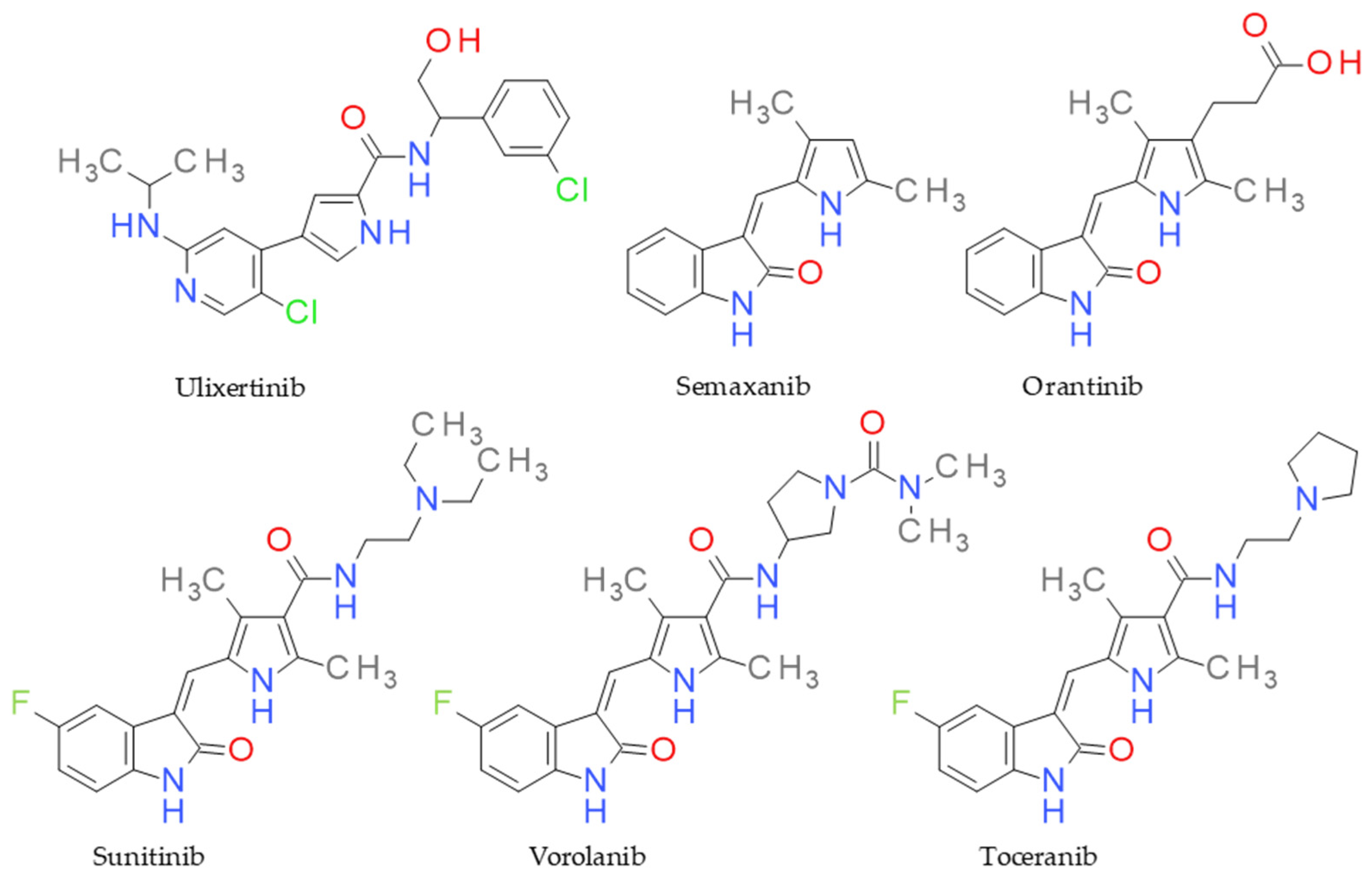
1. Introduction
2. Results and Discussion
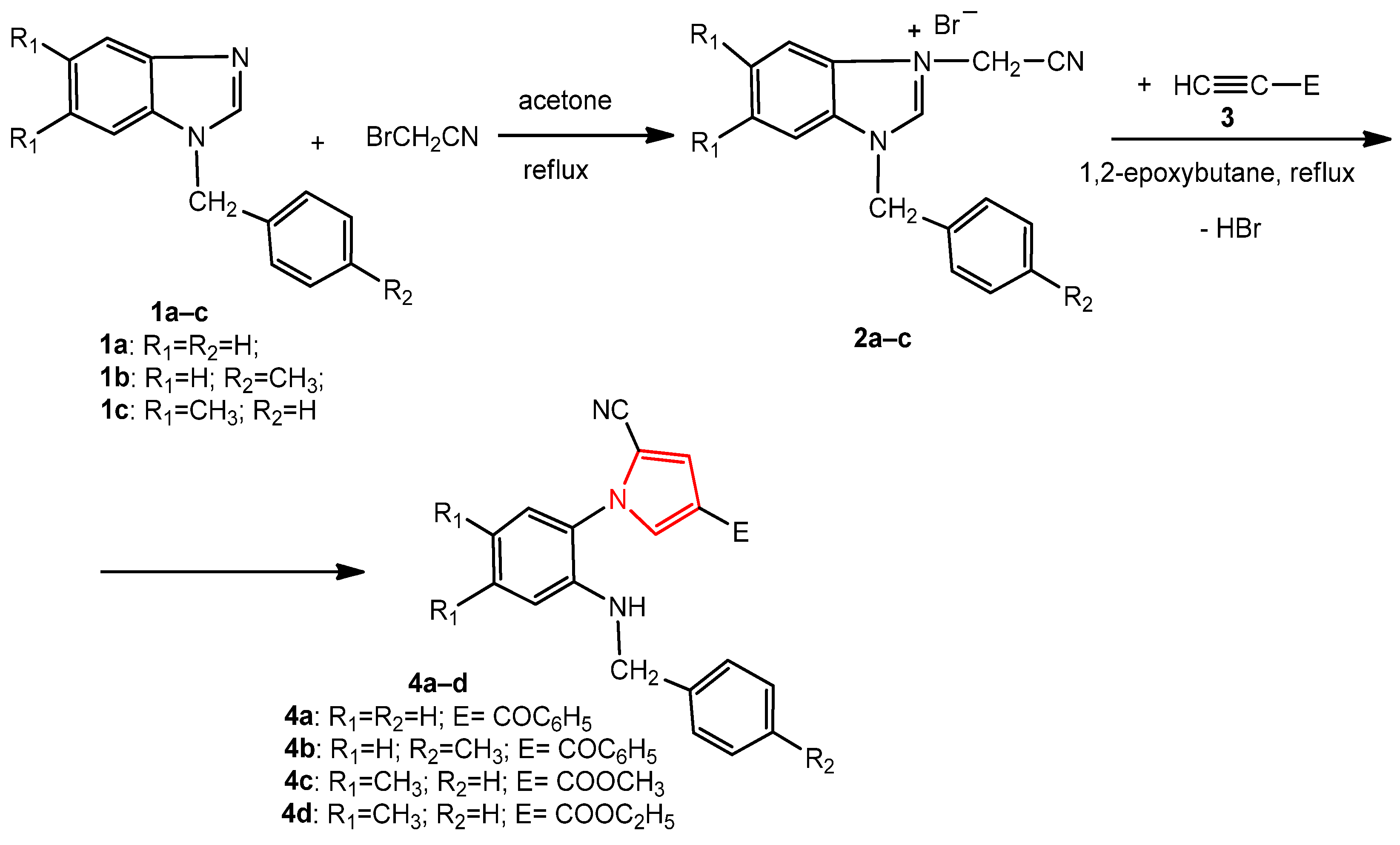
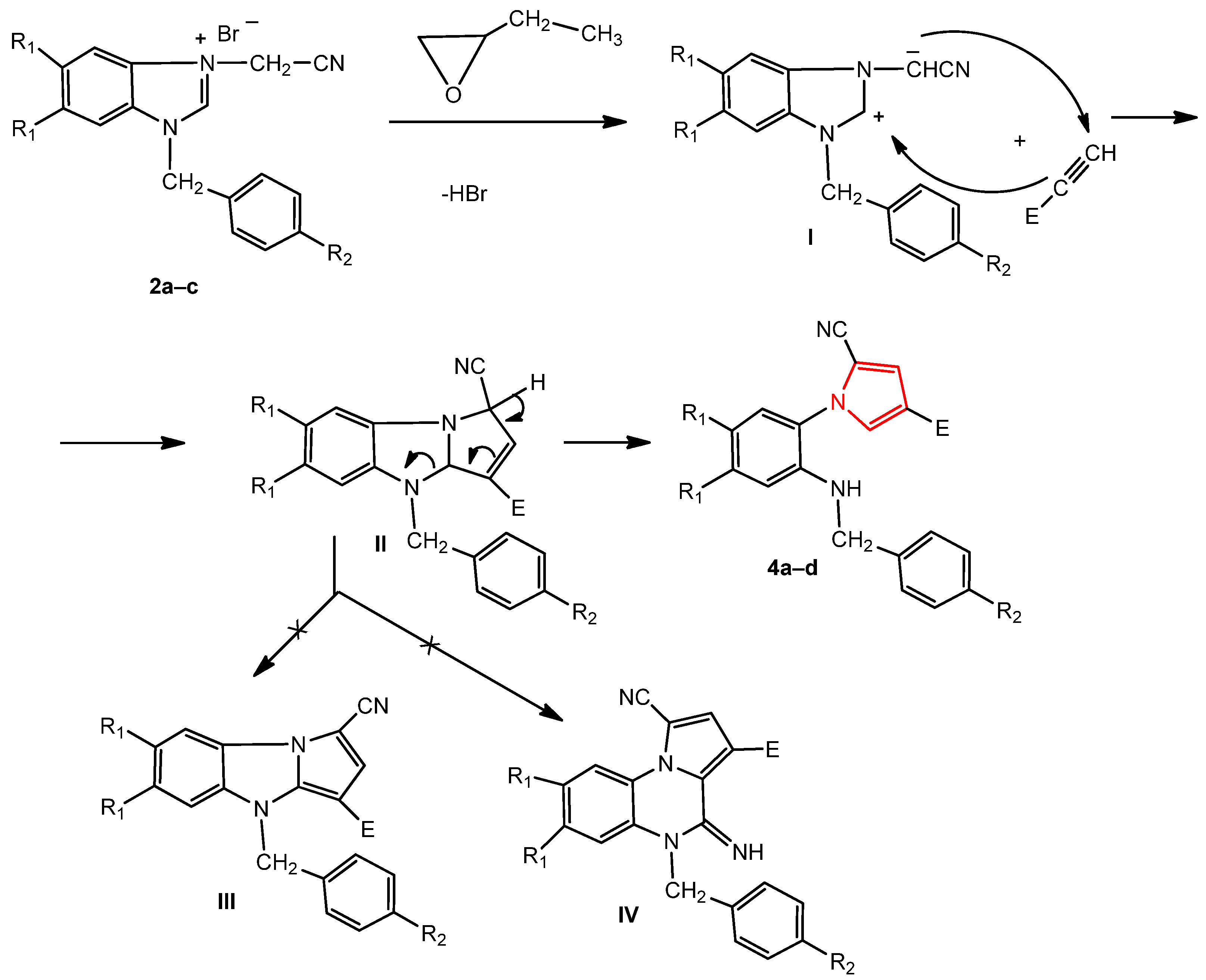
2.1. Chemistry
2.2. Toxicity Evaluation
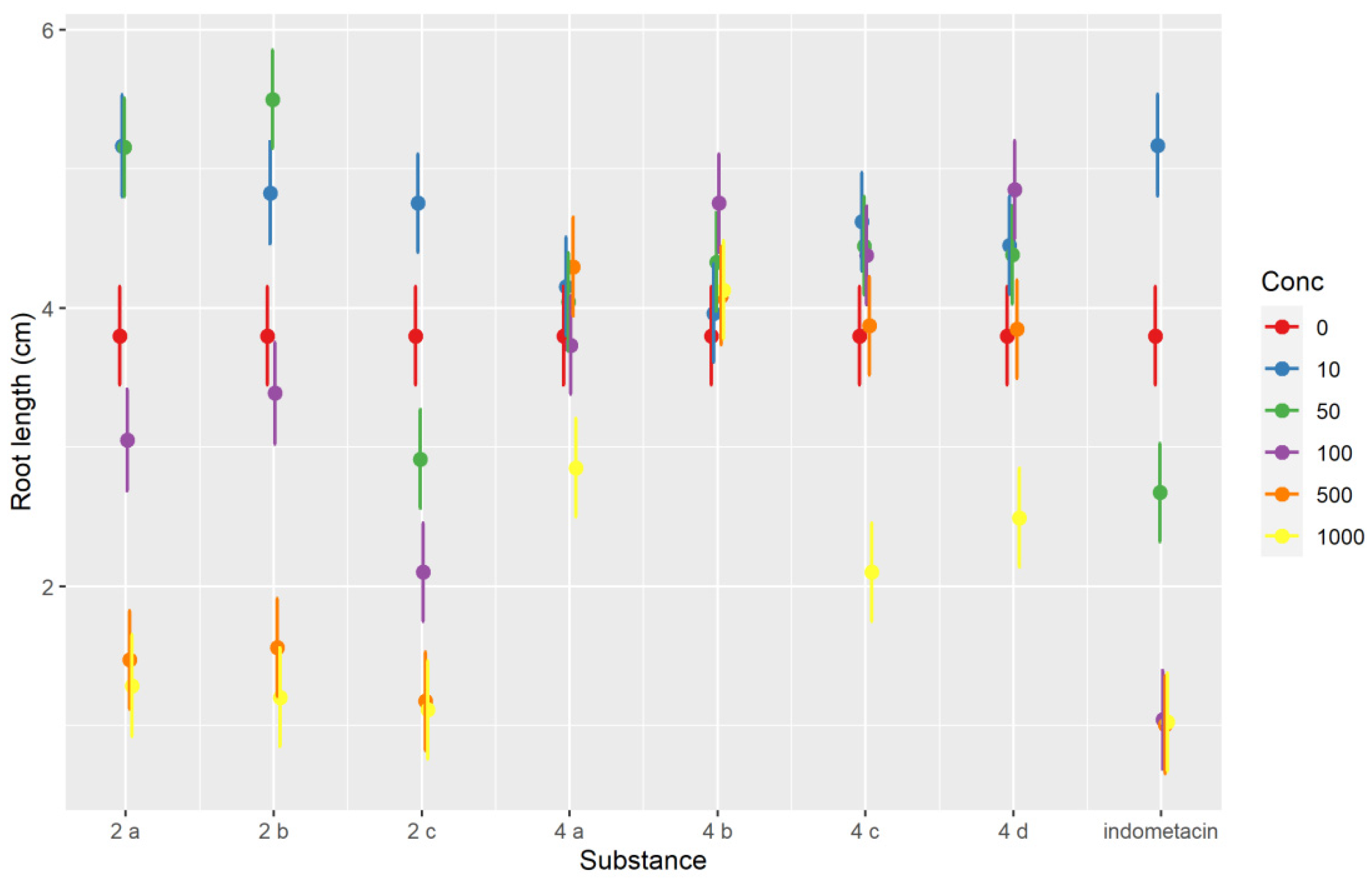
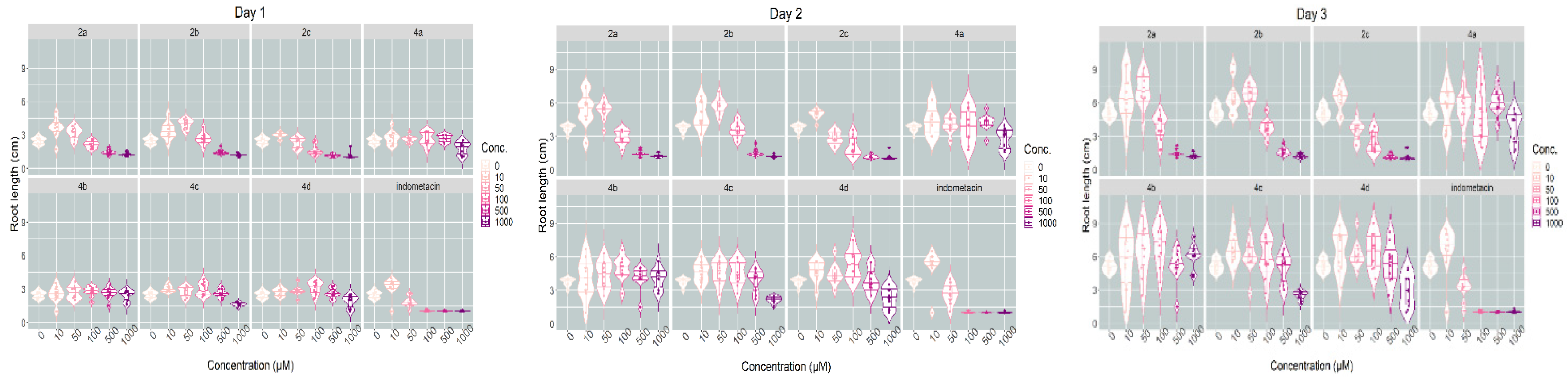
2.2.1. Plant Toxicity Assay
2.2.2. Animal Toxicity Assay
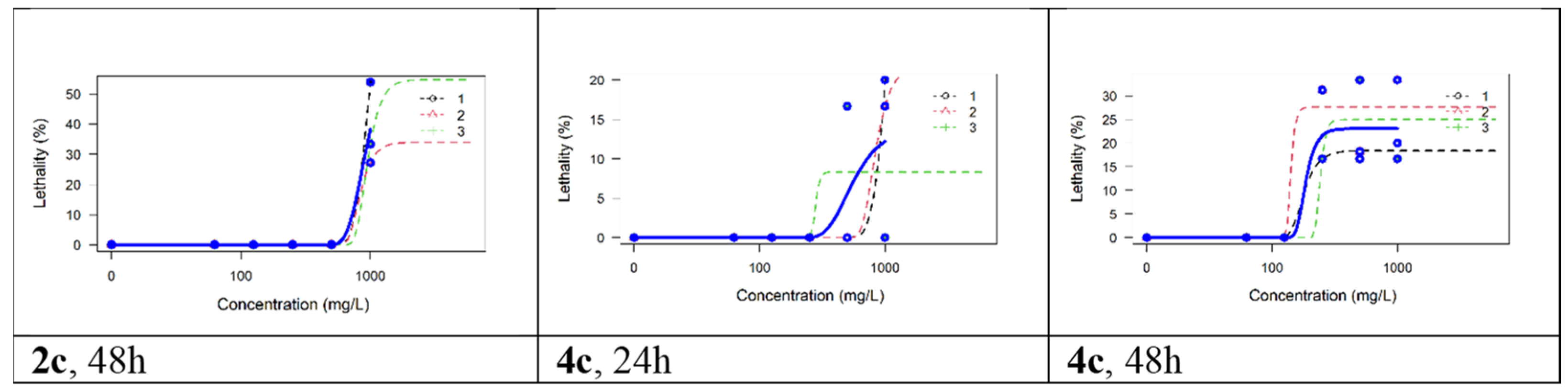
Artemia franciscana Toxicity Assay
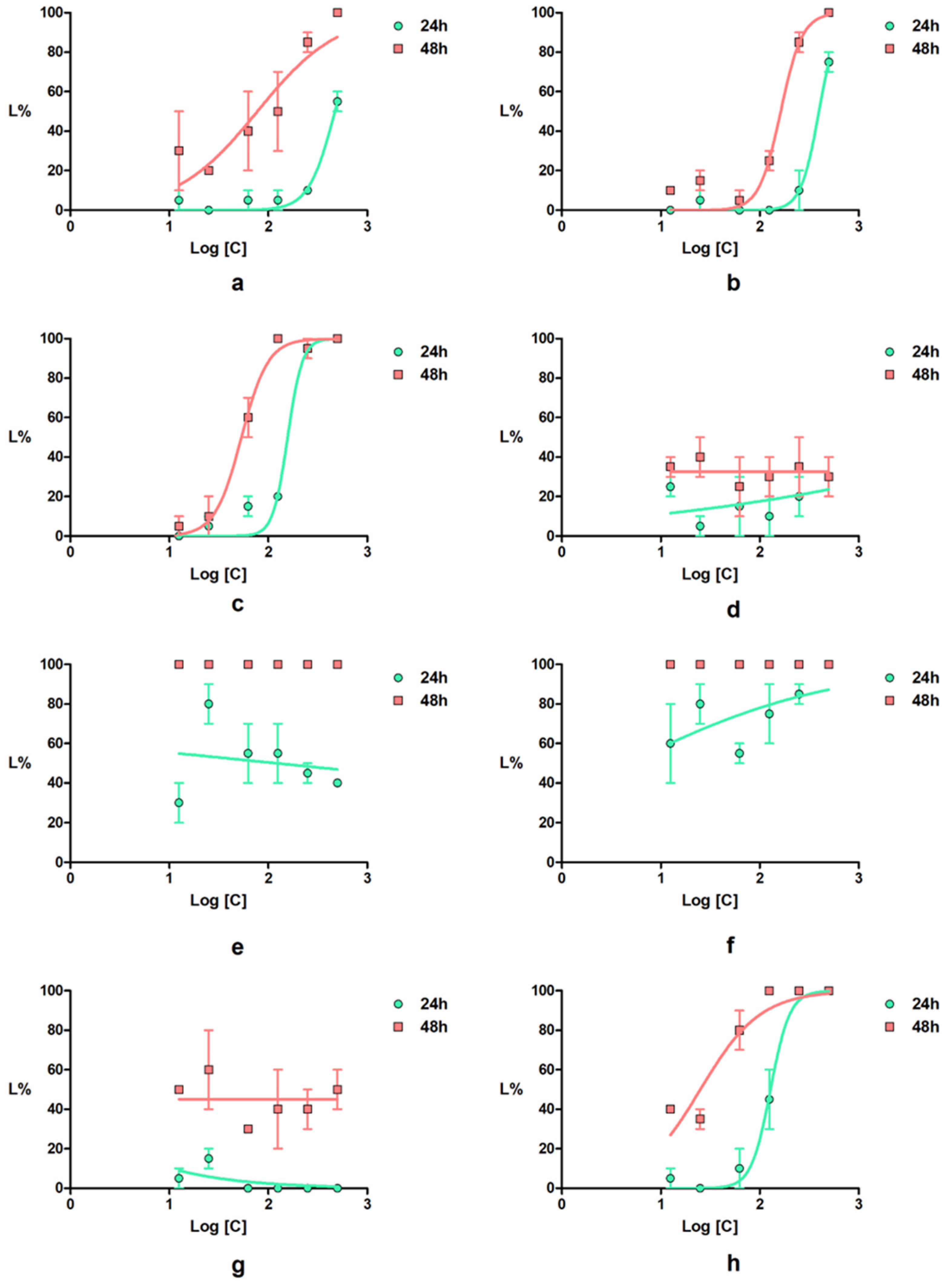
Daphnia magna Toxicity Assay
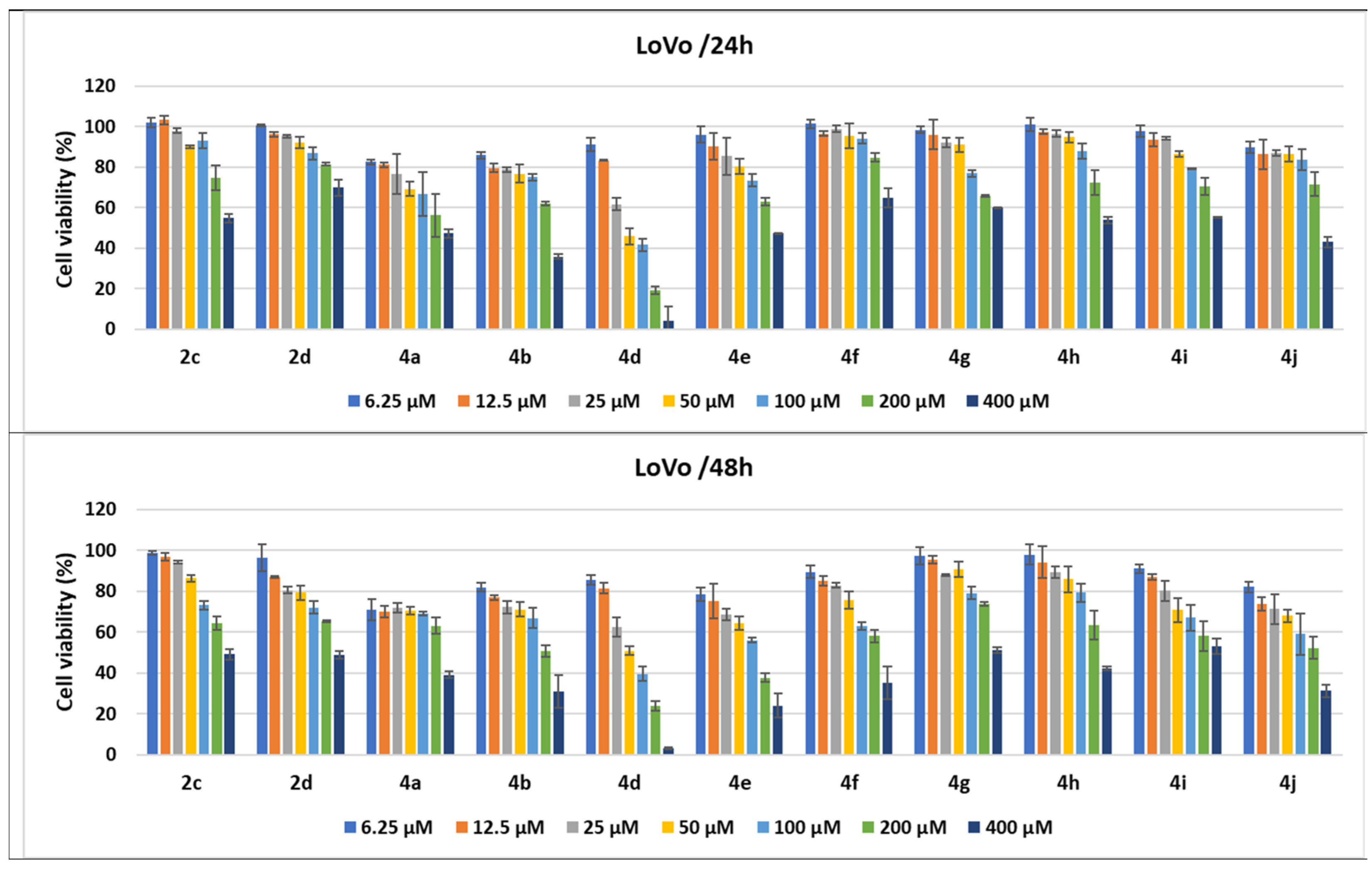
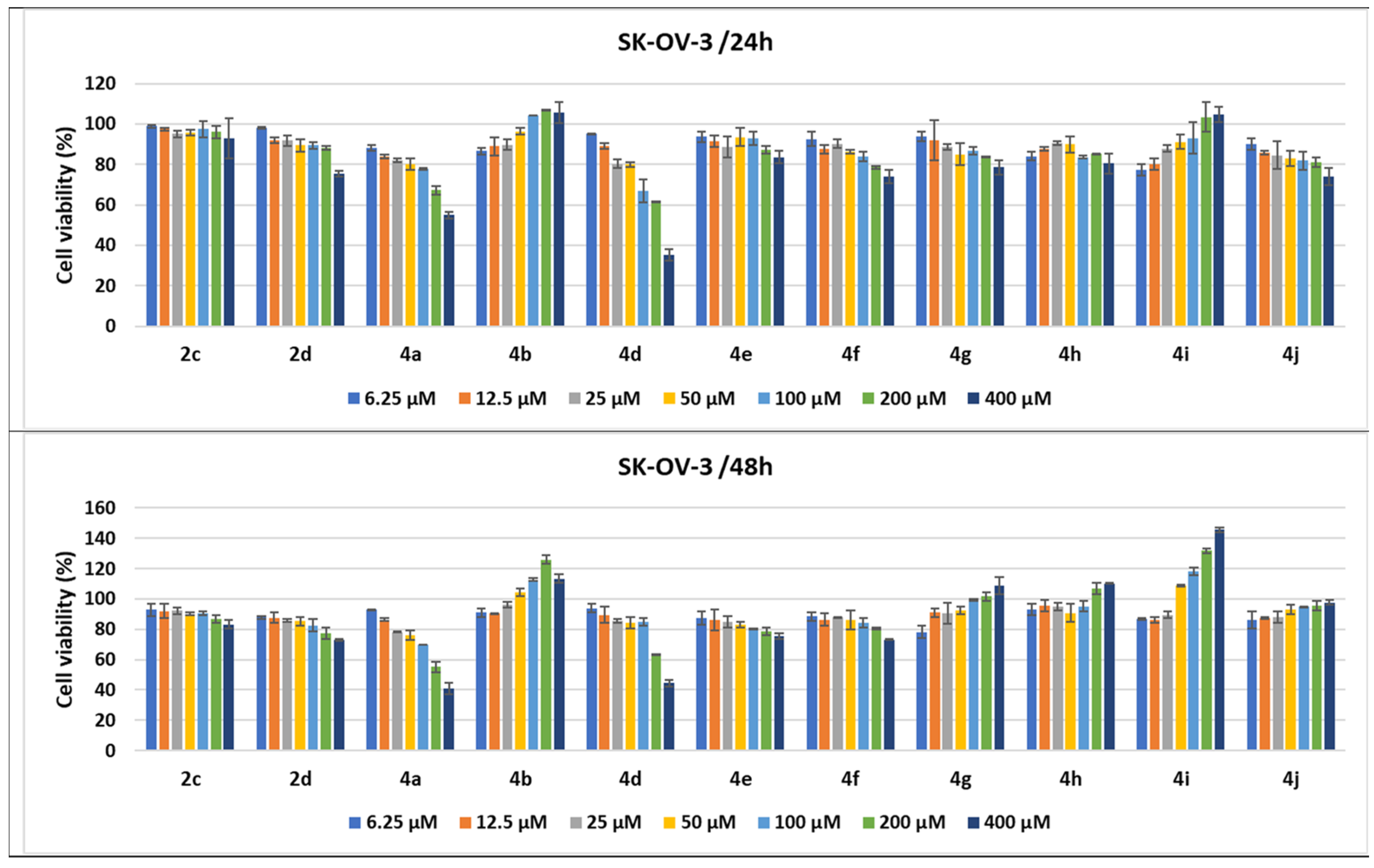
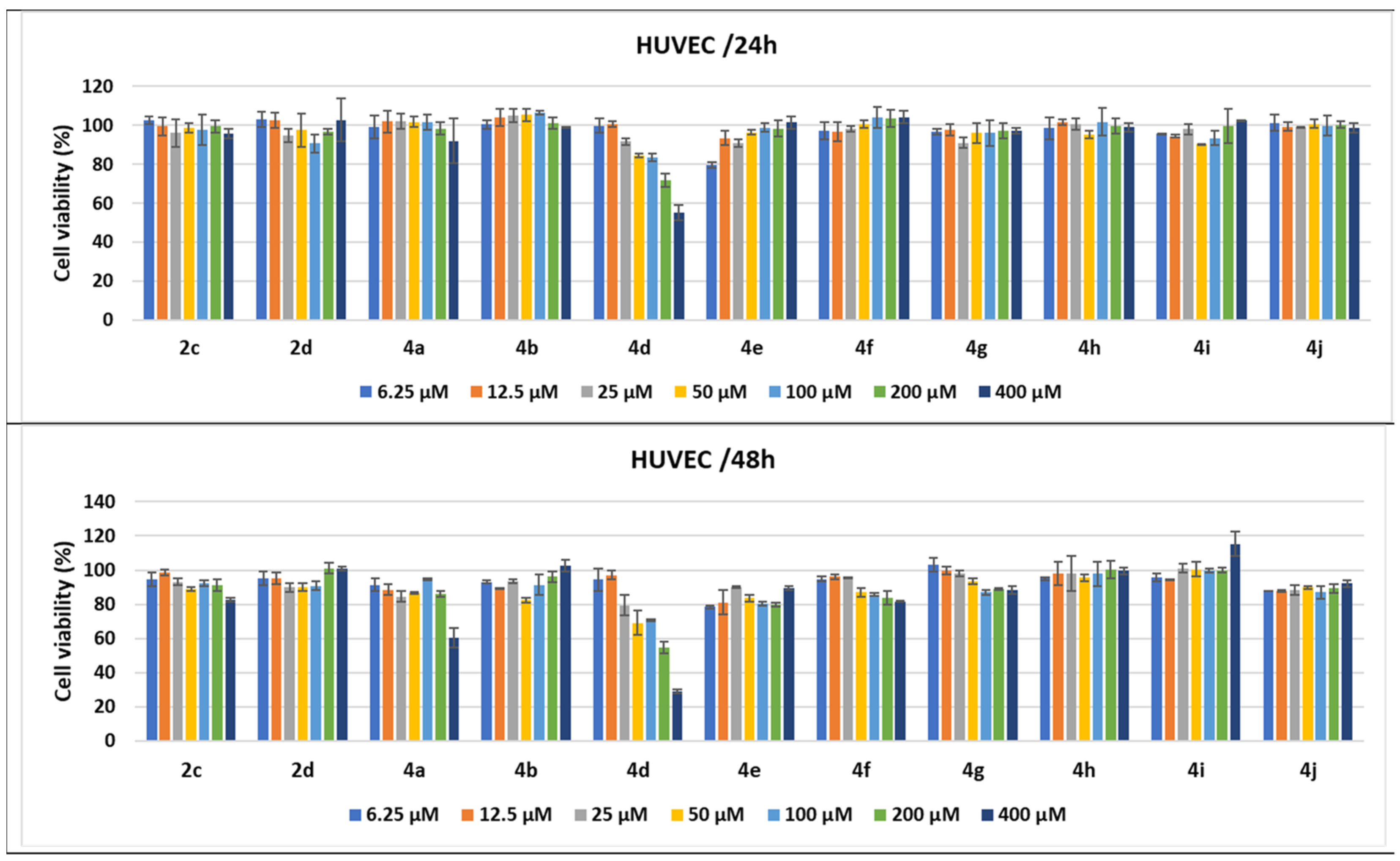
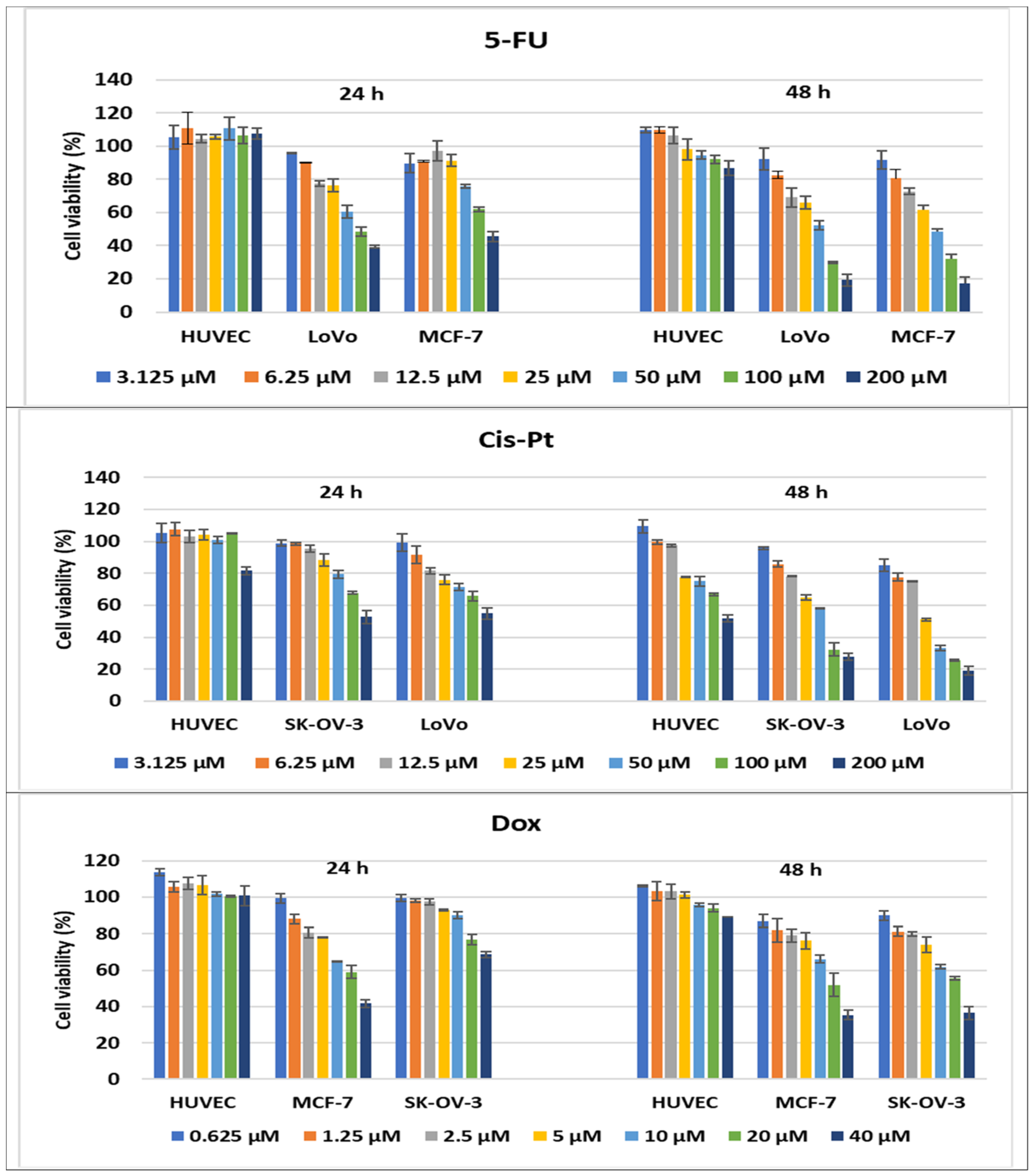
2.2.3. Compound-Mediated Cytotoxicity Assays
2.3. Prediction of the Molecular Mechanism of Action
3. Materials and Methods
3.1. Chemistry
3.1.1. Synthesis of 1-Benzyl-5,6-Dimethyl-3-Cyanomethylbenzimidazolium Bromide 2c
3.1.2. General Procedure for the Synthesis of Pyrroles 4a–d
3.2. Toxicity Evaluation
3.2.1. Phytotoxicity Evaluation
3.2.2. Animal Toxicity Assay
Artemia franciscana Toxicity Assay
Daphnia magna Toxicity Assay
3.3. Prediction of the Molecular Mechanism of Action
3.4. Statistical Analyses
3.5. Cell Cytotoxicity
3.5.1. Cell Cultures and Treatments
3.5.2. MTS Cytotoxicity Assay
3.5.3. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Mihaila, M.; Hotnog, C.M.; Bostan, M.; Munteanu, A.C.; Vacaroiu, I.A.; Brasoveanu, L.I.; Uivarosi, V. Anticancer activity of some ruthenium (III) complexes with quinolone antibiotics: In vitro cytotoxicity, cell cycle modulation, and apoptosis-inducing properties in LoVo colon cancer cell line. Appl. Sci. 2021, 11, 8594. [Google Scholar] [CrossRef]
- Tomi, I.H.R.; Tomma, J.H.; Al-Daraji, A.H.R.; Al-Dujaili, A.H. Synthesis, characterization and comparative study the microbial activity of some heterocyclic compounds containing oxazole and benzothiazole moieties. J. Saudi Chem. Soc. 2015, 19, 392–398. [Google Scholar] [CrossRef]
- Li Petri, G.; Spanò, V.; Spatola, R.; Holl, R.; Raimondi, M.V.; Barraja, P.; Montalbano, A. Bioactive pyrrole-based compounds with target selectivity. Eur. J. Med. Chem. 2020, 208, 112783. [Google Scholar] [CrossRef]
- Vitaku, E.; Smith, D.T.; Njardarson, J.T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 2014, 57, 10257–10274. [Google Scholar] [CrossRef]
- Sarg, M.T.; Koraa, M.M.; Bayoumi, A.H.; Gilil, S.M.A.E. Synthesis of Pyrroles and Condensed Pyrroles as Anti-Inflammatory Agents with Multiple Activities and Their Molecular Docking Study. Open J. Med. Chem. 2015, 05, 49–96. [Google Scholar] [CrossRef]
- Biava, M.; Porretta, G.C.; Poce, G.; Battilocchio, C.; Manetti, F.; Botta, M.; Forli, S.; Sautebin, L.; Rossi, A.; Pergola, C.; et al. Novel Ester and acid derivatives of the 1,5-diarylpyrrole Scaffold as anti-inflammatory and analgesic agents. Synthesis and in vitro and in vivo biological evaluation. J. Med. Chem. 2010, 53, 723–733. [Google Scholar] [CrossRef]
- Anzini, M.; Di Capua, A.; Valenti, S.; Brogi, S.; Rovini, M.; Giuliani, G.; Cappelli, A.; Vomero, S.; Chiasserini, L.; Sega, A.; et al. Novel analgesic/anti-inflammatory agents: 1,5-Diarylpyrrole nitrooxyalkyl ethers and related compounds as cyclooxygenase-2 inhibiting nitric oxide donors. J. Med. Chem. 2013, 56, 3191–3206. [Google Scholar] [CrossRef]
- Battilocchio, C.; Poce, G.; Alfonso, S.; Porretta, G.C.; Consalvi, S.; Sautebin, L.; Pace, S.; Rossi, A.; Ghelardini, C.; Di Cesare Mannelli, L.D.C.; et al. A class of pyrrole derivatives endowed with analgesic/anti-inflammatory activity. Bioorganic Med. Chem. 2013, 21, 3695–3701. [Google Scholar] [CrossRef]
- Sowmya, P.V.; Poojary, B.; Kumar, V.; Vishwanatha, U.; Shetty, P. Fluorinated pyrrole incorporated 2-thiazolyl hydrazone motifs: A new class of antimicrobial and anti tuberculosis agents. Arch. Pharmacal Res. 2017, 1–21. [Google Scholar] [CrossRef]
- Zhang, S.; Tan, X.; Liang, C.; Zhang, W. Design, synthesis, and antifungal evaluation of novel coumarin-pyrrole hybrids. J. Heterocycl. Chem. 2021, 58, 450–458. [Google Scholar] [CrossRef]
- Carvalho, R.C.C.; Martins, W.A.; Silva, T.P.; Kaiser, C.R.; Bastos, M.M.; Pinheiro, L.C.S.; Krettli, A.U.; Boechat, N. New pentasubstituted pyrrole hybrid atorvastatin-quinoline derivatives with antiplasmodial activity. Bioorg. Med. Chem. Lett. 2016, 26, 1881–1884. [Google Scholar] [CrossRef] [PubMed]
- Curreli, F.; Ahmed, S.; Benedict Victor, S.M.; Iusupov, I.R.; Belov, D.S.; Markov, P.O.; Kurkin, A.V.; Altieri, A.; Debnath, A.K. Preclinical Optimization of gp120 Entry Antagonists as anti-HIV-1 Agents with Improved Cytotoxicity and ADME Properties through Rational Design, Synthesis, and Antiviral Evaluation. J. Med. Chem. 2020, 63, 1724–1749. [Google Scholar] [CrossRef] [PubMed]
- Idhayadhulla, A.; Kumar, R.S.; Nasser, A.J.A.; Kavimani, S.; Indumathy, S. Synthesis and anticonvulsant activity of some new series of pyrrole derivatives. Med. Chem. Res. 2012, 21, 3699–3708. [Google Scholar] [CrossRef]
- Mateev, E.; Georgieva, M.; Zlatkov, A. Pyrrole as an Important Scaffold of Anticancer Drugs: Recent Advances. J. Pharm. Pharm. Sci. 2022, 25, 24–40. [Google Scholar] [CrossRef]
- Khalilpour, A.; Asghari, S. Synthesis, characterization and evaluation of cytotoxic and antioxidant activities of dihydropyrimidone substituted pyrrole derivatives. Med. Chem. Res. 2018, 27, 15–22. [Google Scholar] [CrossRef]
- Jung, E.K.; Leung, E.; Barker, D. Synthesis and biological activity of pyrrole analogues of combretastatin A-4. Bioorganic Med. Chem. Lett. 2016, 26, 3001–3005. [Google Scholar] [CrossRef]
- Zhan, X.; Lan, L.; Zhang, Y.; Chen, J.; Zhao, K.; Wang, S.; Xin, Y.; Mao, Z. Synthesis and cytotoxicity evaluation of new 3-substituted 4-(4-methyloxy phenyl)-1H-pyrrole derivatives. Bull. Korean Chem. Soc. 2016, 37, 200–206. [Google Scholar] [CrossRef]
- Imperatore, C.; Scuotto, M.; Valadan, M.; Rivieccio, E.; Saide, A.; Russo, A.; Altucci, C.; Menna, M.; Ramunno, A.; Mayol, L.; et al. Photo-control of cancer cell growth by benzodiazo N-substituted pyrrole derivatives. J. Photochem. Photobiol. A Chem. 2019, 377, 109–118. [Google Scholar] [CrossRef]
- Rashmi, K.C.; Harsha Raj, M.; Paul, M.; Girish, K.S.; Salimath, B.P.; Aparna, H.S. A new pyrrole based small molecule from Tinospora cordifolia induces apoptosis in MDA-MB-231 breast cancer cells via ROS mediated mitochondrial damage and restoration of p53 activity. Chem.-Biol. Interact. 2019, 299, 120–130. [Google Scholar] [CrossRef]
- Azad, I.; Jafri, A.; Khan, T.; Akhter, Y.; Arshad, M.; Hassan, F.; Ahmad, N.; Khan, A.R.; Nasibullah, M. Evaluation of pyrrole-2,3-dicarboxylate derivatives: Synthesis, DFT analysis, molecular docking, virtual screening and in vitro anti-hepatic cancer study. J. Mol. Struct. 2019, 1176, 314–334. [Google Scholar] [CrossRef]
- Jeelan Basha, N.; Basavarajaiah, S.M.; Shyamsunder, K. Therapeutic potential of pyrrole and pyrrolidine analogs: An update. Mol. Divers. 2022, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Nitulescu, G.M. Quantitative and Qualitative Analysis of the Anti-Proliferative Potential of the Pyrazole Scaffold in the Design of Anticancer Agents. Molecules 2022, 27, 3300. [Google Scholar] [CrossRef]
- Khajuria, R.; Dham, S.; Kapoor, K.K. Active methylenes in the synthesis of a pyrrole motif: An imperative structural unit of pharmaceuticals, natural products and optoelectronic materials. RSC Adv. 2016, 6, 37039–37066. [Google Scholar] [CrossRef]
- Papaetis, G.S.; Syrigos, K.N. Sunitinib: A multitargeted receptor tyrosine kinase inhibitor in the era of molecular cancer therapies. BioDrugs 2009, 23, 377–389. [Google Scholar] [CrossRef]
- Musser, M.L.; Johannes, C.M. Toceranib phosphate (Palladia) for the treatment of canine exocrine pancreatic adenocarcinoma. BMC Vet. Res. 2021, 17, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, K.S.; Grierson, P.M.; Picus, J.; Lockhart, A.C.; Roth, B.J.; Liu, J.; Morton, A.; Chan, E.; Huffman, J.; Liang, C.; et al. Vorolanib (X-82), an oral anti-VEGFR/PDGFR/CSF1R tyrosine kinase inhibitor, with everolimus in solid tumors: Results of a phase I study. Investig. New Drugs 2021, 39, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Germann, U.A.; Furey, B.F.; Markland, W.; Hoover, R.R.; Aronov, A.M.; Roix, J.J.; Hale, M.; Boucher, D.M.; Sorrell, D.A.; Martinez-Botella, G.; et al. Targeting the MAPK signaling pathway in cancer: Promising preclinical activity with the novel selective ERK1/2 inhibitor BVD-523 (ulixertinib). Mol. Cancer Ther. 2017, 16, 2351–2363. [Google Scholar] [CrossRef]
- Haddad, J.J. The immunopharmacologic potential of Semaxanib and new generation directed therapeutic drugs: Receptor tyrosine kinase regulation with anti-tumorigenensis/angiogenesis properties. Saudi Pharm. J. 2012, 20, 103–123. [Google Scholar] [CrossRef]
- Yurovskaya, M.A.; Alekseyev, R.S. New perspectives on classical heterocyclic reactions involving pyrrole derivatives (review). Chem. Heterocycl. Compd. 2014, 49, 1400–1425. [Google Scholar] [CrossRef]
- Behera, B.K.; Sahu, A.K.; Devi, N.R.; Saikia, A.K. K2S2O8-Mediated Synthesis of Highly Functionalized Pyrroles via Oxidative Self-Dimerization of N-Propargylamines. J. Org. Chem. 2021, 86, 12481–12493. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.C.; Huang, W.Y. Reaction of 1-alkylbenzimidazolium 3-ylides with ethyl 2,2- dihydropolyfluoroalkanoates. Tetrahedron 1998, 54, 12465–12474. [Google Scholar] [CrossRef]
- Moldoveanu, C.; Zbancioc, G.; Mantu, D.; Maftei, D.; Mangalagiu, I. The cycloaddition of the benzimidazolium ylides with alkynes: New mechanistic insights. PLoS ONE 2016, 11, e0156129. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, D.; Georgescu, E.; Caira, M.R.; Draghici, C.; Dumitrascu, F. A New Synthesis of Pyrroles from Benzimidazolium N-Cyanomethyl Ylides and Alkyne Dipolarophiles. Synlett 2017, 28, 2241–2246. [Google Scholar] [CrossRef]
- Gao, P.; Wang, J.; Bai, Z.J.; Shen, L.; Yan, Y.Y.; Yang, D.S.; Fan, M.J.; Guan, Z.H. Synthesis of Polycarbonyl Pyrroles via K2S2O8-Mediated Oxidative Cyclization of Enamines. Org. Lett. 2016, 18, 6074–6077. [Google Scholar] [CrossRef] [PubMed]
- Ivan, B.C.; Dumitrascu, F.; Anghel, A.I.; Ancuceanu, R.V.; Shova, S.; Dumitrescu, D.; Draghici, C.; Olaru, O.T.; Nitulescu, G.M.; Dinu, M.; et al. Synthesis and toxicity evaluation of new pyrroles obtained by the reaction of activated alkynes with 1-methyl-3-(Cyanomethyl)benzimidazolium bromide. Molecules 2021, 26, 6435. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, D.; Popa, M.M.; Georgescu, F.; Georgescu, E.; Barbu, L.; Dumitrascu, F. New Substituted Pyrroles Obtained In Searching For Pyrrolo [1,2-a]Quinazoline Framework. Rev. Roum. Chim. 2013, 58, 785–790. [Google Scholar]
- Caira, M.R.; Georgescu, E.; Barbu, L.; Georgescu, F.; Dumitrascu, F. New tri- and tetra-substituted pyrroles via quinazolinium N1-ylides. Arkivoc 2011, 10, 44–54. [Google Scholar] [CrossRef]
- Dumitrascu, F.; Georgescu, E.; Caira, M.R.; Georgescu, F.; Popa, M.; Draghici, B.; Dumitrescu, D.G. A Novel Approach for the Synthesis of N-Arylpyrroles. Synlett 2009, 20, 3336–3340. [Google Scholar] [CrossRef]
- Georgescu, E.; Nicolescu, A.; Georgescu, F.; Teodorescu, F.; Shova, S.; Marinoiu, A.T.; Dumitrascu, F.; Deleanu, C. Fine tuning the outcome of 1,3-dipolar cycloaddition reactions of benzimidazolium ylides to activated alkynes. Tetrahedron 2016, 72, 2507–2520. [Google Scholar] [CrossRef]
- Olaru, O.T.; Zanfirescu, A.; Nitulescu, G.; Dinu-Pirvu, C.E.; Anuta, V.; Tsatsakis, A.; Spandidos, D.A.; Margina, D.; Seremet, O.C.; Nitulescu, G.M. Predictive power of the Triticum root elongation test for the assessment of novel anti-proliferative therapies. Int. J. Mol. Med. 2019, 44, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hensman, J.; Kutkaite, G.; Toh, T.S.; Galhoz, A.; Dry, J.R.; Saez-Rodriguez, J.; Garnett, M.J.; Menden, M.P.; Dondelinger, F. A statistical framework for assessing pharmacological responses and biomarkers using uncertainty estimates. Elife 2020, 9, e60352. [Google Scholar] [CrossRef]
- Ekonomou, G.; Lolas, A.; Castritsi-Catharios, J.; Neofitou, C.; Zouganelis, G.D.; Tsiropoulos, N.; Exadactylos, A. Mortality and effect on growth of Artemia franciscana exposed to two common organic pollutants. Water 2019, 11, 1614. [Google Scholar] [CrossRef]
- Ali, A.J.; Mohamed, A.J.; Kumar, M.S.A.; John, B.A. Organophosphorus pesticides toxicity on brine shrimp artemia. J. CleanWAS 2018, 1, 23–26. [Google Scholar] [CrossRef]
- Qiu, J.; Huang, Y.; Wu, Y.; Shi, P.; Xu, B.; Chu, W.; Pan, Y. Detection, transformation, and toxicity of indole-derivative nonsteroidal anti-inflammatory drugs during chlorine disinfection. Chemosphere 2020, 260, 127579. [Google Scholar] [CrossRef] [PubMed]
- Davoren, M.; Fogarty, A.M. A test battery for the ecotoxicological evaluation of the agri-chemical Environ. Ecotoxicol. Environ. Saf. 2004, 59, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Cruzeiro, C.; Amaral, S.; Rocha, E.; Rocha, M.J. Determination of 54 pesticides in waters of the Iberian Douro River estuary and risk assessment of environmentally relevant mixtures using theoretical approaches and Artemia salina and Daphnia magna bioassays. Ecotoxicol. Environ. Saf. 2017, 145, 126–134. [Google Scholar] [CrossRef]
- Filimonov, D.A.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Druzhilovskii, D.S.; Pogodin, P.V.; Poroikov, V.V. Prediction of the biological activity spectra of organic compounds using the pass online web resource. Chem. Heterocycl. Compd. 2014, 50, 444–457. [Google Scholar] [CrossRef]
- Stecoza, C.E.; Nitulescu, G.M.; Draghici, C.; Caproiu, M.T.; Olaru, O.T.; Bostan, M.; Mihaila, M. Synthesis and anticancer evaluation of new 1,3,4-oxadiazole derivatives. Pharmaceuticals 2021, 14, 438. [Google Scholar] [CrossRef]
- Rigiracciolo, D.C.; Cirillo, F.; Talia, M.; Muglia, L.; Gutkind, J.S.; Maggiolini, M.; Lappano, R. Focal adhesion kinase fine tunes multifaced signals toward breast cancer progression. Cancers 2021, 13, 645. [Google Scholar] [CrossRef]
- Xiong, G.; Wu, Z.; Yi, J.; Fu, L.; Yang, Z.; Hsieh, C.; Yin, M.; Zeng, X.; Wu, C.; Lu, A.; et al. ADMETlab 2.0: An integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic Acids Res. 2021, 49, W5–W14. [Google Scholar] [CrossRef] [PubMed]
- CrysAlisPro Software System; Version 1.171.41.123a; Rigaku Corporation: Oxford, UK, 2015.
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT–Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Crystallogr. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, C71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Anghel, A.I.; Ancuceanu, R.; Popescu, M.L.; Gârd, C.E.; Dinu, M.; Hovaneț, M.V.; Nencu, I. Preliminary research regarding the cytotoxicity and antioxidant activity of arbutus unedo L. Leaves. Farmacia 2021, 69, 260–266. [Google Scholar] [CrossRef]
- Dinu, M.; Anghel, A.I.; Olaru, O.T.; Seremet, O.C.; Calalb, T.; Cojocaru-Toma, M.; Negres, S.; Hovanet, M.V.; Zbarcea, C.E.; Ancuceanu, R. Toxicity investigation of an extract of Amaranthus retroflexus L. (Amaranthaceae) leaves. Farmacia 2017, 65, 289–294. [Google Scholar]
- Wang, H.; Zhang, G. Experimental study of the inhibitory effect of indomethacin on the growth and angiogenesis of human colon cancer xenografts. Di Yi Jun Yi Da Xue Xue Bao 2004, 24, 184–187. [Google Scholar]
- Kashtwari, M.; Zargar, S.A.; Wani, A.A. Laboratory Techniques of Studying Plant Chromosomes. In Chromosome Structure and Aberrations; Bhat, T.A., Wani, A.A., Eds.; Springer: New Delhi, India, 2017; pp. 75–107. ISBN 978-81-322-3673-3. [Google Scholar]
- Nitulescu, G.; Nicorescu, I.M.; Olaru, O.T.; Ungurianu, A.; Mihai, D.P.; Zanfirescu, A.; Nitulescu, G.M.; Margina, D. Molecular Docking and Screening Studies of New Natural Sortase A Inhibitors. Int. J. Mol. Sci. 2017, 18, 2217. [Google Scholar] [CrossRef]
- Nitulescu, G.M.; Draghici, C.; Olaru, O.T. New potential antitumor pyrazole derivatives: Synthesis and cytotoxic evaluation. Int. J. Mol. Sci. 2013, 14, 21805–21818. [Google Scholar] [CrossRef]
- Nitulescu, G.; Mihai, D.P.; Nicorescu, I.M.; Olaru, O.T.; Ungurianu, A.; Zanfirescu, A.; Nitulescu, G.M.; Margina, D. Discovery of natural naphthoquinones as sortase A inhibitors and potential anti-infective solutions against Staphylococcus aureus. Drug Dev. Res. 2019, 80, 1136–1145. [Google Scholar] [CrossRef]
- Seremet, O.C.; Olaru, O.T.; Gutu, C.M.; Nitulescu, G.M.; Ilie, M.; Negres, S.; Zbarcea, C.E.; Purdel, C.N.; Spandidos, D.A.; Tsatsakis, A.M.; et al. Toxicity of plant extracts containing pyrrolizidine alkaloids using alternative invertebrate models. Mol. Med. Rep. 2018, 17, 7757–7763. [Google Scholar] [CrossRef] [PubMed]
- R Core Team R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 9 June 2022).
- RStudio Team. RStudio: Integrated Development Environment for R. RStudio, PBC, Boston. Available online: http://www.rstudio.com/ (accessed on 9 June 2022).
- Koller, M. Robustlmm: An R package for Robust estimation of linear Mixed-Effects models. J. Stat. Softw. 2016, 75, 1–24. [Google Scholar] [CrossRef]
- Lüdecke, D. sjPlot: Data Visualization for Statistics in Social Science. R Package Version 2.8.10. Available online: https://cran.r-project.org/package=sjPlot%3E (accessed on 9 June 2022).
- Ritz, C.; Baty, F.; Streibig, J.C.; Gerhard, D. Dose-response analysis using R. PLoS ONE 2015, 10, e0146021. [Google Scholar] [CrossRef] [PubMed]
- Hotnog, D.; Mihaila, M.; Botezatu, A.; Matei, G.G.; Hotnog, C.; Anton, G.; Bostan, M.; Brasoveanu, L.I. Genistein Potentiates The Apoptotic Effect Of 5-Fluorouracyl In Colon Cancer Cell Lines. Rom. Biotechnol. Lett. 2013, 18, 8751–8760. [Google Scholar]
- Munteanu, A.C.; Musat, M.G.; Mihaila, M.; Badea, M.; Olar, R.; Nitulescu, G.M.; Radulescu, F.S.; Brasoveanu, L.I.; Uivarosi, V. New heteroleptic lanthanide complexes as multimodal drugs: Cytotoxicity studies, apoptosis, cell cycle analysis, DNA interactions, and protein binding. Appl. Organomet. Chem. 2021, 35, e6062. [Google Scholar] [CrossRef]
- Munteanu, A.C.; Badea, M.; Olar, R.; Silvestro, L.; Mihaila, M.; Brasoveanu, L.I.; Musat, M.G.; Andries, A.; Uivarosi, V. Cytotoxicity studies, DNA interaction and protein binding of new Al (III), Ga (III) and In (III) complexes with 5-hydroxyflavone. Appl. Organomet. Chem. 2018, 32, e4579. [Google Scholar] [CrossRef]
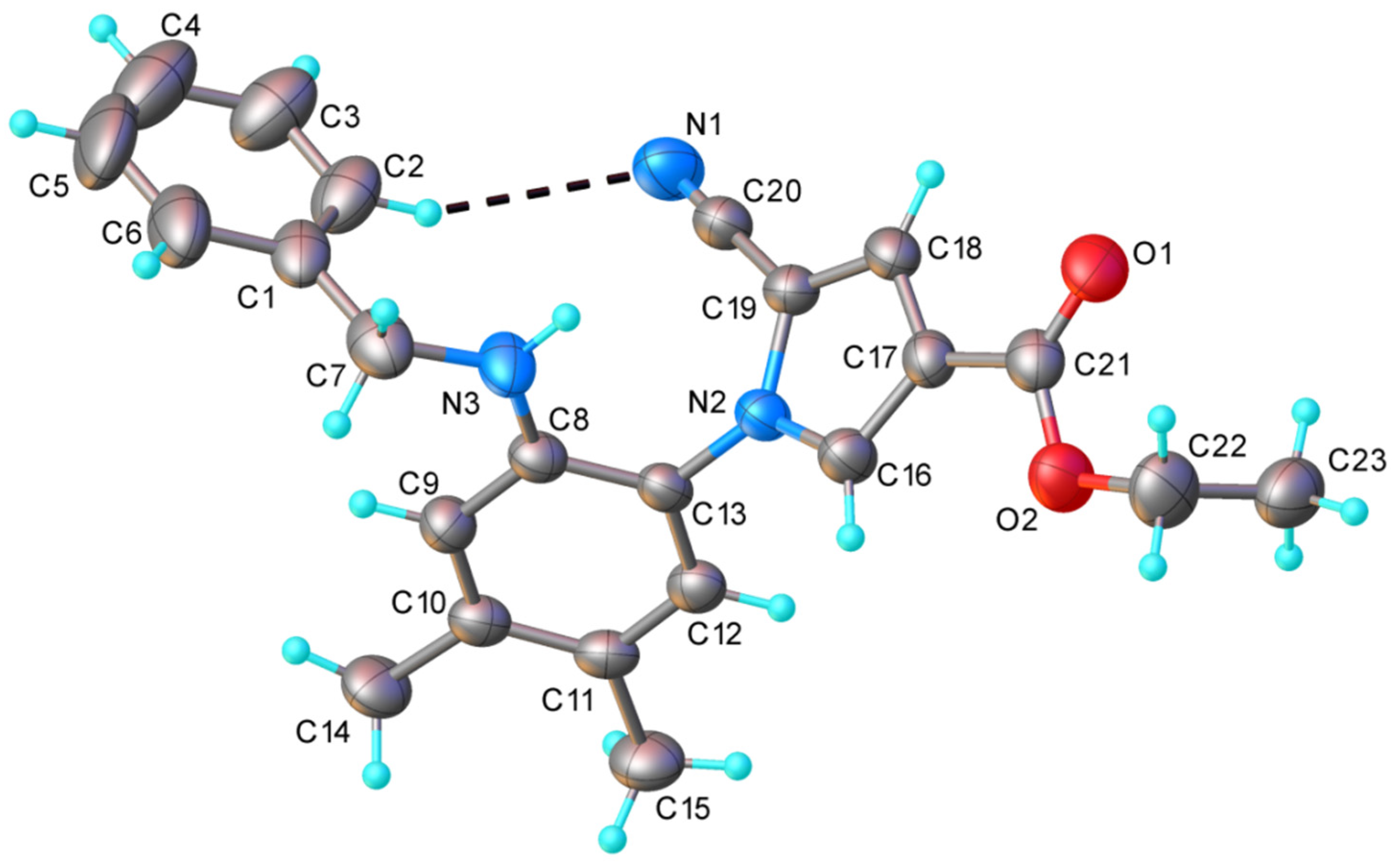
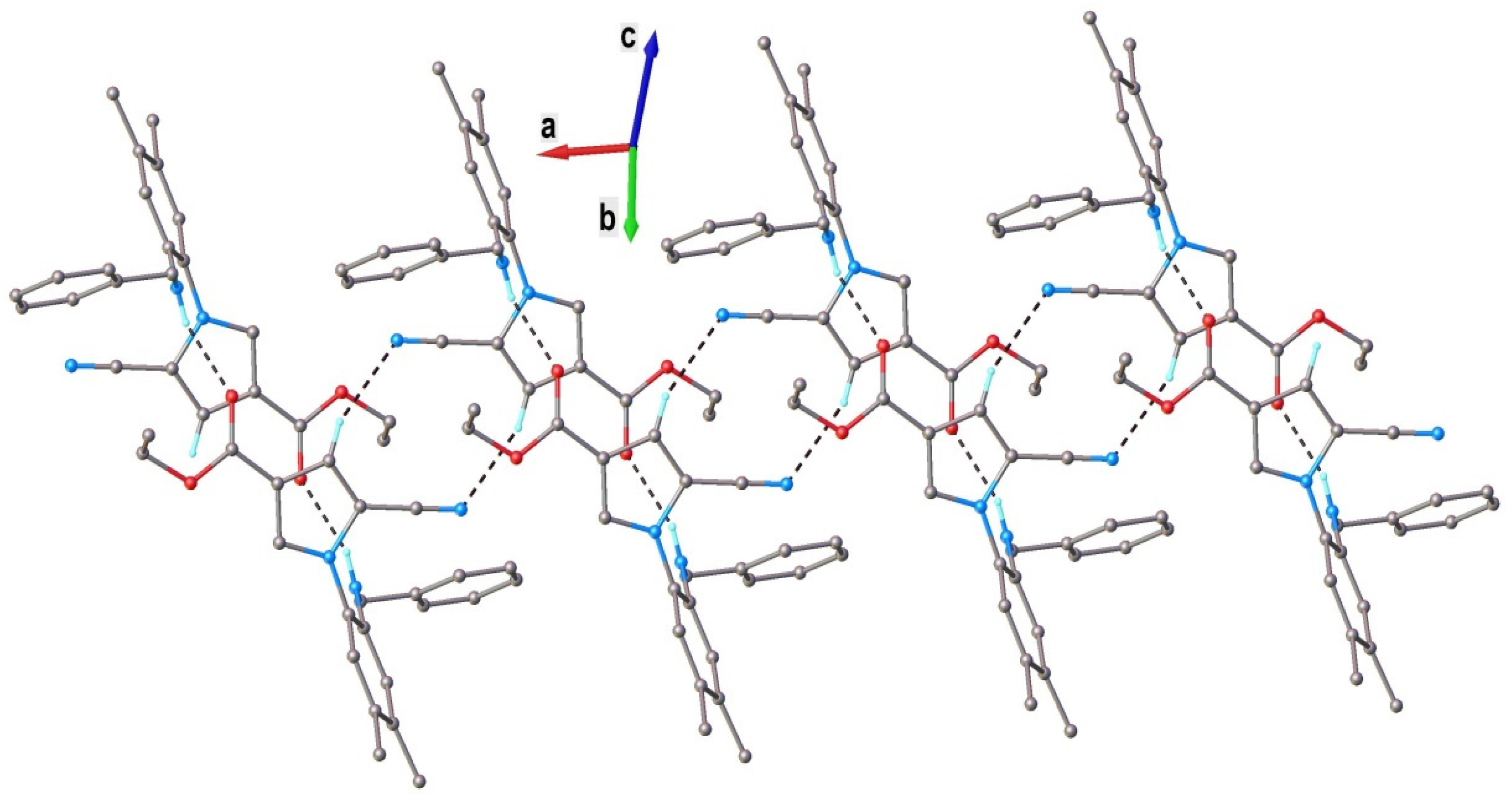



| Empirical Formula | C23H23N3O2 |
|---|---|
| Fw | 373.44 |
| Space group | P-1 |
| a (Å) | 8.0707(3) |
| b (Å) | 16.1583(6) |
| c (Å) | 17.0040(6) |
| α (°) | 73.404(3) |
| β (°) | 103.491(3) |
| γ (°) | 90.540(3) |
| V (Å3) | 2062.55(13) |
| Z | 4 |
| rcalcd (g cm−3) | 1.203 |
| Crystal size (mm) | 0.20 × 0.10 × 0.04 |
| T (K) | 293 |
| μ (mm−1) | 0.078 |
| 2Θ range (°) | 4.188 to 58.826 |
| Reflections collected | 30622 |
| Independent reflections | 9845 (Rint = 0.0443) |
| Data/restraints/parameters | 9845/73/494 |
| R1 (a) | 0.0738 |
| wR2 (b) | 0.2087 |
| GOF (c) | 1.000 |
| Largest difference in peak/hole (e·Å−3) | 0.23/−0.29 |
| CCDC No. | 2166919 |
| Compound | 24 h | 48 h | ||||
|---|---|---|---|---|---|---|
| Maximum L% (%) | LC5050 (µM) | 95% CI of LC50 (µM) | Maximum L% (µM) | LC50 (%) | 95% CI of LC50 (µM) | |
| 2a | 55 | 471.7 | 424.8–523.7 | 100 | 77.51 | 40.44–148.6 |
| 2b | 75 | 396.9 | 359.0–438.9 | 100 | 163.1 | 139.0–191.4 |
| 2c | 100 | 157 | 135.2–182.3 | 100 | 53.59 | 45.33–63.34 |
| 4a | 35 | ND * | ND * | 35 | ND * | ND * |
| 4b | 85 | ND ** | ND ** | 100 | ND ** | ND ** |
| 4c | 100 | 4.583 | 0.1717–122.3 | 100 | ND ** | ND ** |
| 4d | 15 | 0.4 | ND * | 60 | ND * | ND * |
| Indomethacin | 100 | 128.6 | 112.9–146.5 | 100 | 25.12 | 18.01–35.03 |
| Target/Effect | 2c | 2d | 4a | 4b | 4c | 4d | 4e | 4f | 4g | 4h | 4i | 4j | Su |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiogenesis inhibitor | - | - | 0.45 | 0.41 | 0.37 | 0.32 | 0.36 | 0.31 | 0.24 | 0.25 | 0.36 | 0.29 | 0.84 |
| Colony-stimulating factor antagonist | - | - | 0.51 | 0.48 | 0.40 | 0.37 | 0.53 | 0.46 | 0.41 | 0.40 | 0.45 | 0.41 | - |
| Focal adhesion kinase 2 inhibitor | - | 0.24 | 0.26 | 0.25 | 0.28 | 0.25 | 0.31 | 0.36 | 0.33 | 0.19 | 0.39 | 0.36 | - |
| Focal adhesion kinase inhibitor | - | - | 0.19 | 0.18 | 0.21 | 0.18 | 0.20 | 0.24 | 0.20 | - | 0.27 | 0.22 | - |
| Kinase inhibitor | - | - | - | 0.25 | 0.29 | - | 0.24 | 0.33 | - | - | - | - | 0.83 |
| Platelet-derived growth factor receptor kinase inhibitor | - | - | 0.34 | 0.37 | 0.32 | 0.35 | - | - | - | - | - | - | 0.86 |
| Proto-oncogene tyrosine-protein kinase Fgr inhibitor | - | 0.18 | 0.29 | 0.30 | 0.28 | 0.25 | 0.36 | 0.34 | 0.31 | 0.29 | 0.35 | 0.33 | - |
| Property | 4a | 4b | 4c | 4d |
|---|---|---|---|---|
| Lipinski rule | yes | yes | yes | yes |
| PAINS | no | no | no | no |
| Human intestinal absorption | <30% | <30% | <30% | <30% |
| Human hepatotoxicity | ++ | ++ | ++ | ++ |
| Drug-induced liver injury | +++ | +++ | + | + |
| AMES toxicity | ++ | ++ | −− | −−− |
| Carcinogenicity | + | + | −−− | −−− |
| Skin sensitization | −− | −−− | − | −− |
| Respiratory toxicity | −−− | −−− | −−− | −−− |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivan, B.-C.; Barbuceanu, S.-F.; Hotnog, C.M.; Anghel, A.I.; Ancuceanu, R.V.; Mihaila, M.A.; Brasoveanu, L.I.; Shova, S.; Draghici, C.; Olaru, O.T.; et al. New Pyrrole Derivatives as Promising Biological Agents: Design, Synthesis, Characterization, In Silico, and Cytotoxicity Evaluation. Int. J. Mol. Sci. 2022, 23, 8854. https://doi.org/10.3390/ijms23168854
Ivan B-C, Barbuceanu S-F, Hotnog CM, Anghel AI, Ancuceanu RV, Mihaila MA, Brasoveanu LI, Shova S, Draghici C, Olaru OT, et al. New Pyrrole Derivatives as Promising Biological Agents: Design, Synthesis, Characterization, In Silico, and Cytotoxicity Evaluation. International Journal of Molecular Sciences. 2022; 23(16):8854. https://doi.org/10.3390/ijms23168854
Chicago/Turabian StyleIvan, Beatrice-Cristina, Stefania-Felicia Barbuceanu, Camelia Mia Hotnog, Adriana Iuliana Anghel, Robert Viorel Ancuceanu, Mirela Antonela Mihaila, Lorelei Irina Brasoveanu, Sergiu Shova, Constantin Draghici, Octavian Tudorel Olaru, and et al. 2022. "New Pyrrole Derivatives as Promising Biological Agents: Design, Synthesis, Characterization, In Silico, and Cytotoxicity Evaluation" International Journal of Molecular Sciences 23, no. 16: 8854. https://doi.org/10.3390/ijms23168854
APA StyleIvan, B.-C., Barbuceanu, S.-F., Hotnog, C. M., Anghel, A. I., Ancuceanu, R. V., Mihaila, M. A., Brasoveanu, L. I., Shova, S., Draghici, C., Olaru, O. T., Nitulescu, G. M., Dinu, M., & Dumitrascu, F. (2022). New Pyrrole Derivatives as Promising Biological Agents: Design, Synthesis, Characterization, In Silico, and Cytotoxicity Evaluation. International Journal of Molecular Sciences, 23(16), 8854. https://doi.org/10.3390/ijms23168854








