Molecular Subtyping of Invasive Breast Cancer Using a PAM50-Based Multigene Expression Test-Comparison with Molecular-Like Subtyping by Tumor Grade/Immunohistochemistry and Influence on Oncologist’s Decision on Systemic Therapy in a Real-World Setting
Abstract
:1. Introduction
2. Results
2.1. Characteristics of Cohort
2.2. Comparison of Local versus Central Molecular-Like Subtyping
2.3. Distribution of Prosigna® Molecular Subtypes
2.4. Comparison between Surrogate Subtyping and Molecular Prosigna® Subtype
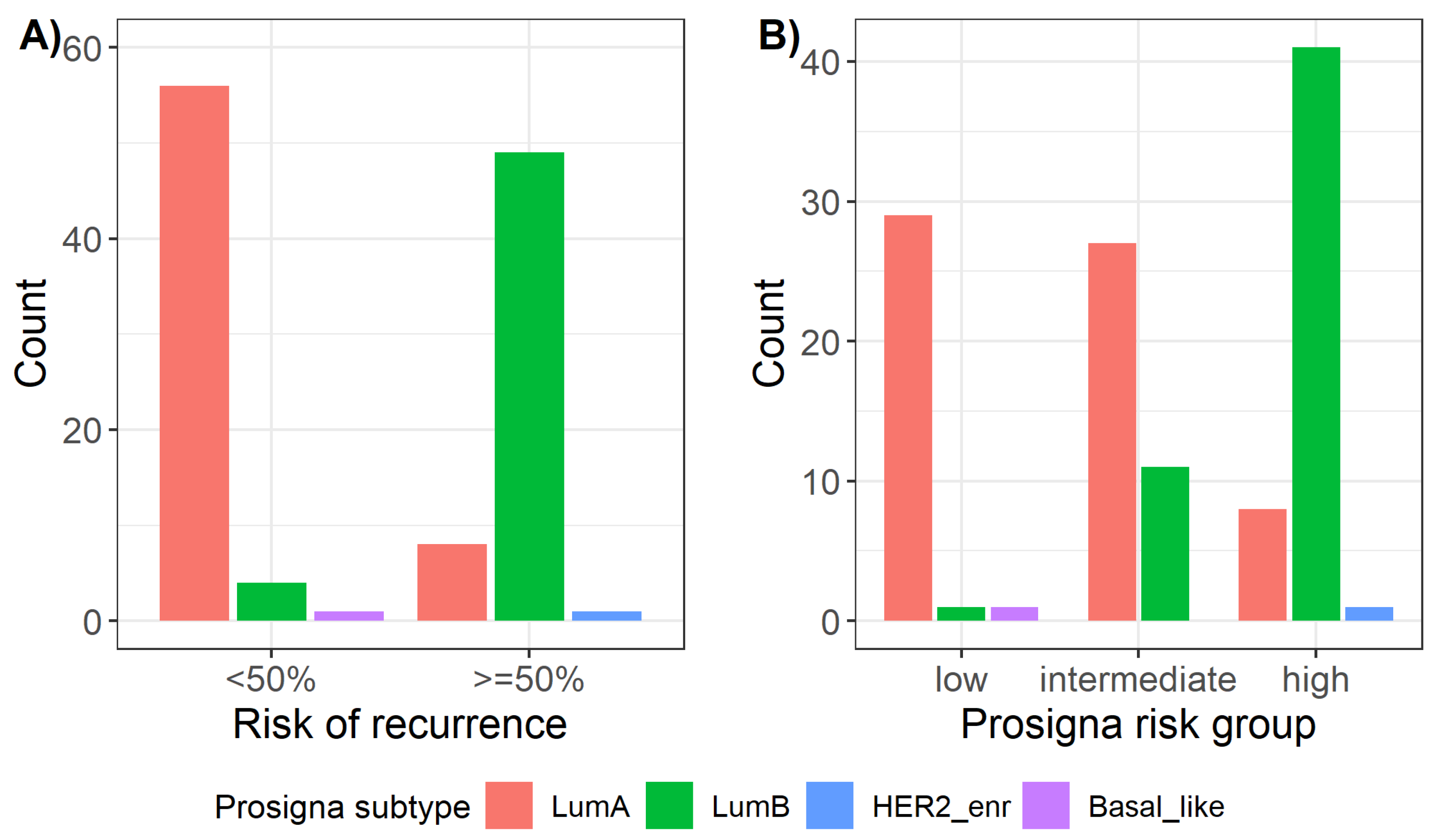
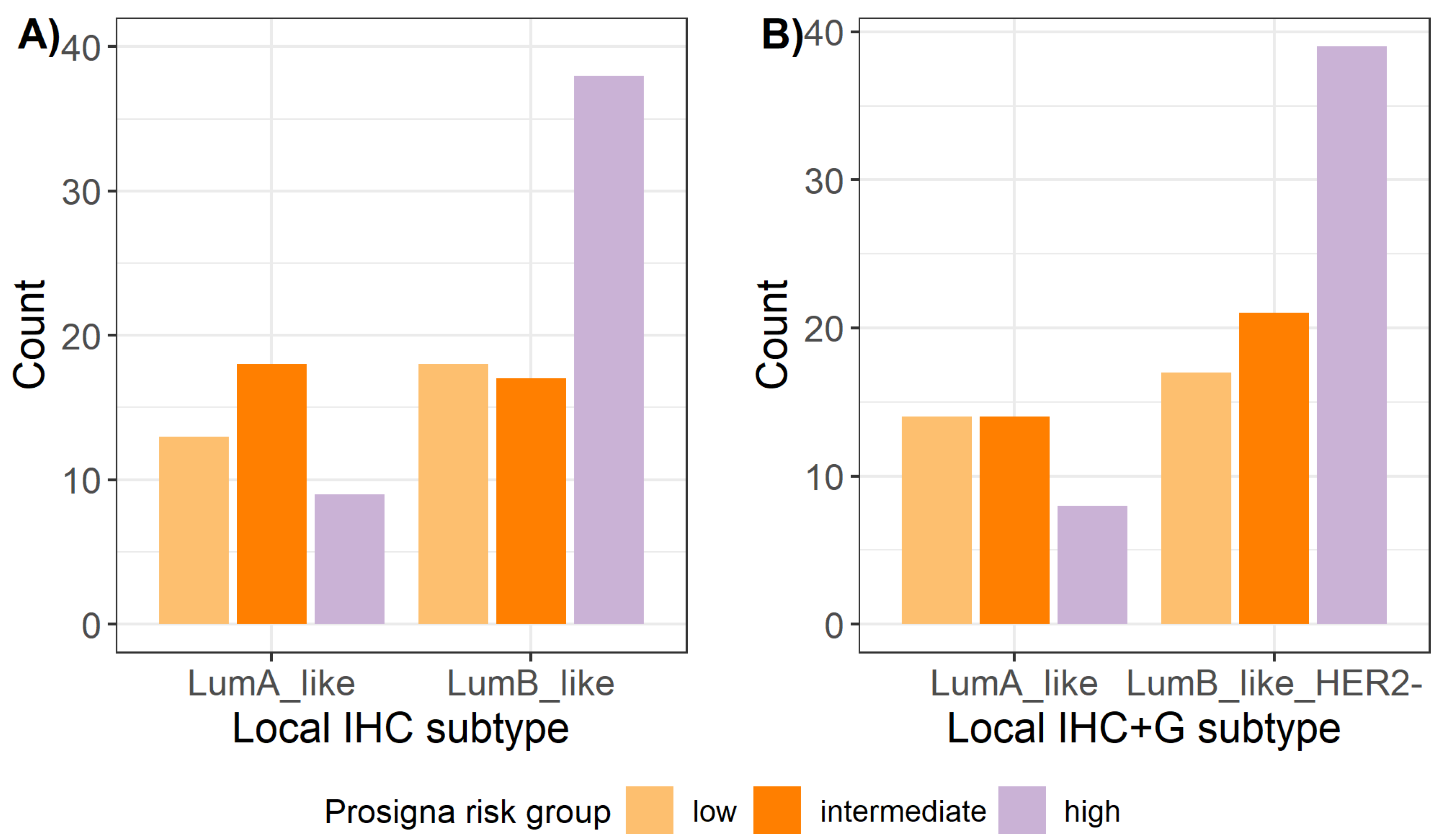
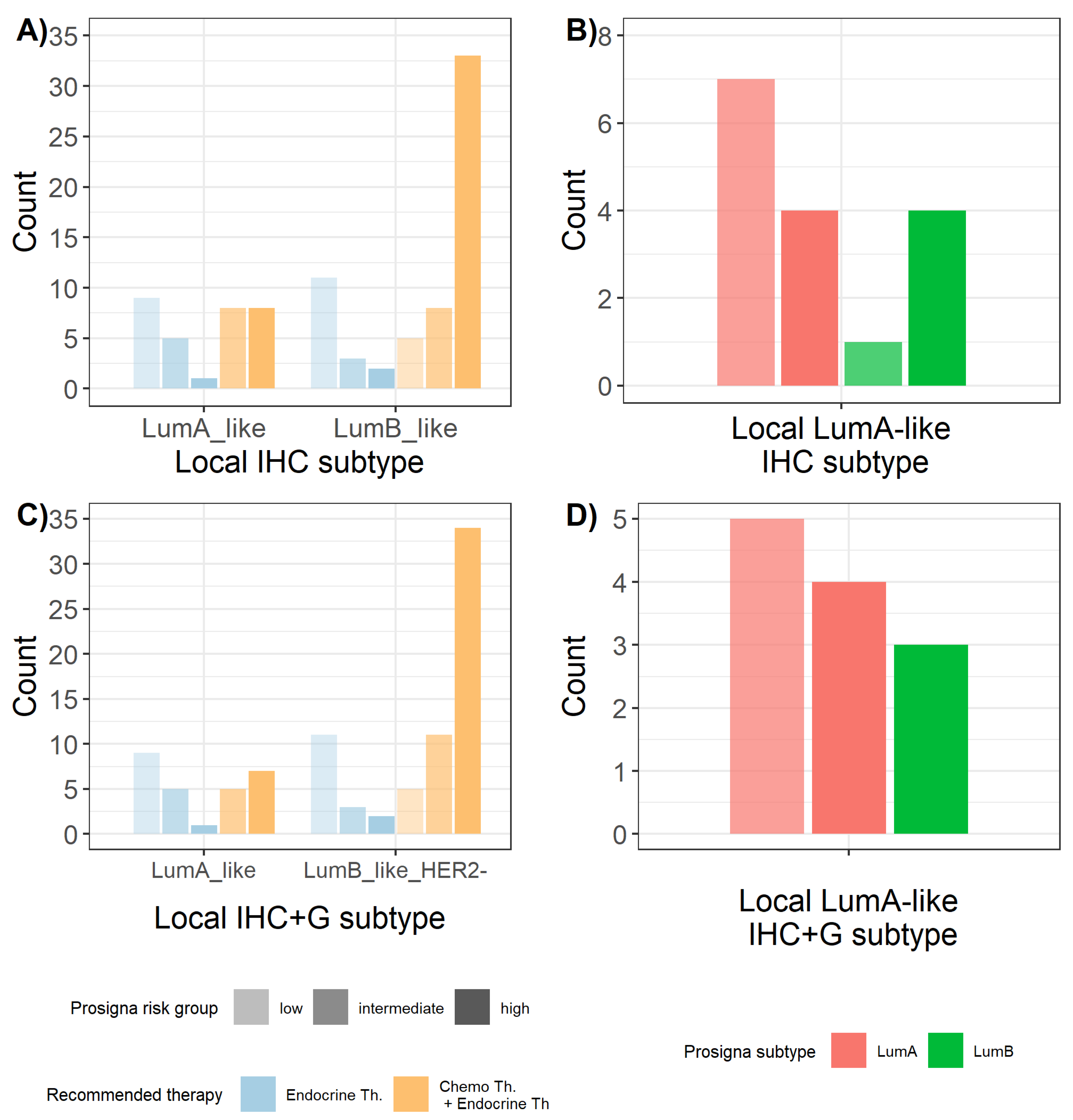
2.5. Prosigna® Risk Groups and Correlation with Local Surrogate Subtypes
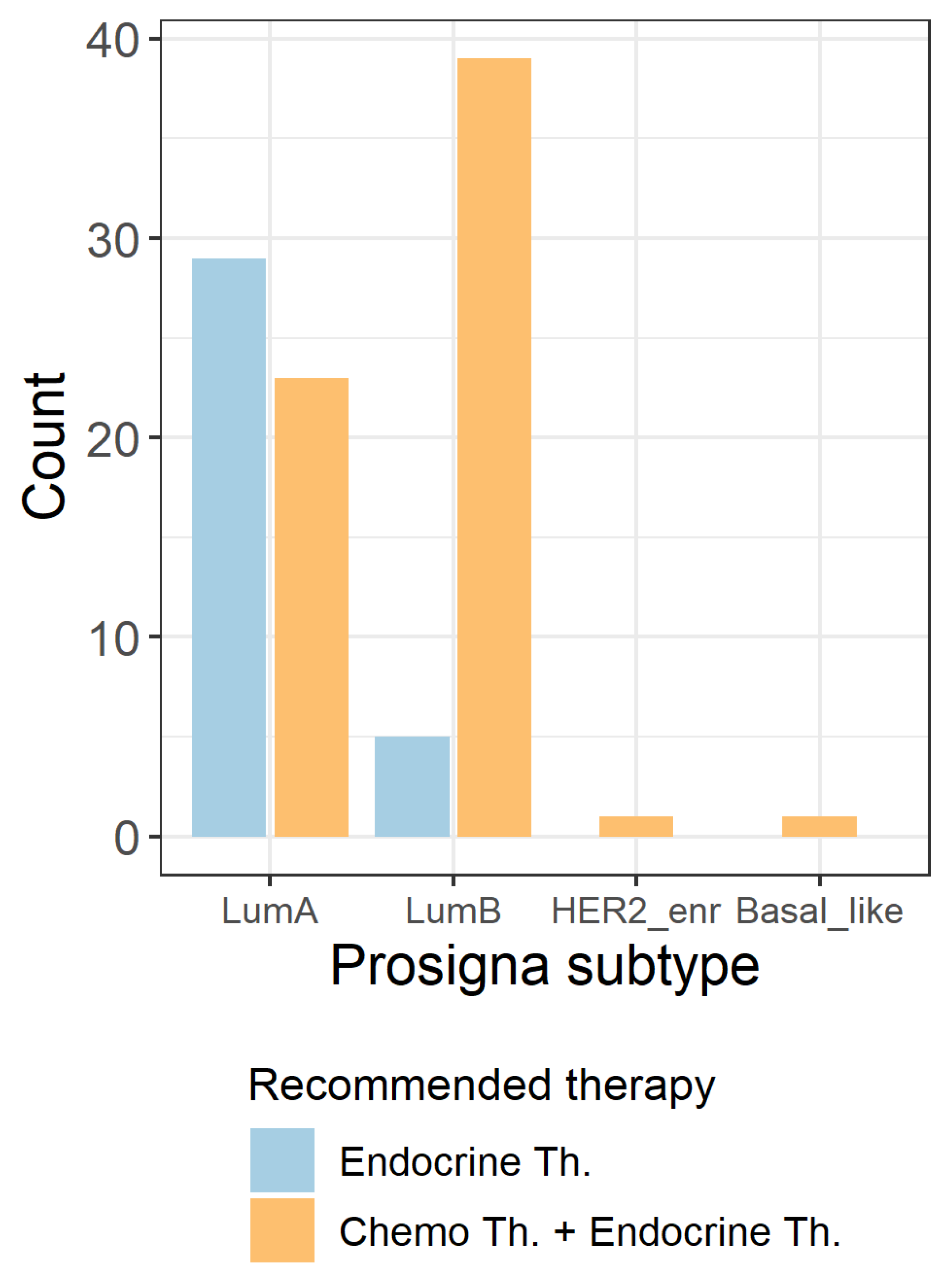
2.6. Influence of Prosigna® Assay Result on Treatment Recommendation
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Molecular-Like Surrogate Subtyping
4.3. Prosigna® Assay
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Available online: https://www.krebsdaten.de/Krebs/DE/Content/Krebsarten/Brustkrebs/brustkrebs_node.html (accessed on 27 April 2020).
- Interdisziplinäre S3-Leitlinie für die Früherkennung, Diagnostik, Therapie und Nachsorge des Mammakarzinoms. Langversion 4. 3 February 2020 AWMF-Registernummer: 032-045OL. Available online: https://www.leitlinienprogramm-onkologie.de/leitlinien/mammakarzinom/ (accessed on 2 August 2022).
- Arbeitsgemeinschaft Gynäkologische Onkologie e.V. Guidelines Breast Version 2020.1 (Download Gesamtdateien). Available online: www.ago-online.de (accessed on 26 April 2020).
- Allison, K.H.; Hammond, M.E.H.; Dowsett, M.; McKernin, S.E.; Carey, L.A.; Fitzgibbons, P.L.; Hayes, D.F.; Lakhani, S.R.; Chavez-MacGregor, M.; Perlmutter, J.; et al. Estrogen and Progesterone Receptor Testing in Breast Cancer: ASCO/CAP Guideline Update. J. Clin. Oncol. 2020, 38, 1346–1366. [Google Scholar] [CrossRef] [PubMed]
- Wolff, A.C.; Hammond, M.E.H.; Allison, K.H.; Harvey, B.E.; Mangu, P.B.; Bartlett, J.M.S.; Bilous, M.; Ellis, I.O.; Fitzgibbons, P.; Hanna, W.; et al. Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer. Am. Soc. Clin. Oncol./Coll. Am. Pathol. Clin. Pract. Guidel. Focused Update 2018, 142, 1364–1382. [Google Scholar] [CrossRef] [Green Version]
- Lokuhetty, D.; White, V.A.; Watanabe, R.; Cree, I.A. WHO Classification of Tumours Editorial Board. Breast Tumours. In WHO Classification of Tumours Series, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2019; Volume 2, pp. 88–97. [Google Scholar]
- Lux, M.P.; Önügören, O.; Wunderle, M.; Hein, A.; Rauh, C.; Schrauder, M.G.; Fasching, P.A.; Beckmann, M.W. (Neo-)Adjuvanten und First-Line-Therapie—Prognostische und prädiktive Faktoren. Gynäkologe 2015, 48, 876–884. [Google Scholar] [CrossRef]
- Cardoso, F.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rubio, I.T.; Zackrisson, S.; Senkus, E. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up†. Ann. Oncol. 2019, 30, 1194–1220. [Google Scholar] [CrossRef] [Green Version]
- Perou, C.M.; Sorlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Sorlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef] [Green Version]
- Schettini, F.; Brasó-Maristany, F.; Kuderer, N.M.; Prat, A. A perspective on the development and lack of interchangeability of the breast cancer intrinsic subtypes. npj Breast Cancer 2022, 8, 85. [Google Scholar] [CrossRef]
- Parker, J.S.; Mullins, M.; Cheang, M.C.; Leung, S.; Voduc, D.; Vickery, T.; Davies, S.; Fauron, C.; He, X.; Hu, Z.; et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2009, 27, 1160–1167. [Google Scholar] [CrossRef]
- Paik, S.; Shak, S.; Tang, G.; Kim, C.; Baker, J.; Cronin, M.; Baehner, F.L.; Walker, M.G.; Watson, D.; Park, T.; et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N. Engl. J. Med. 2004, 351, 2817–2826. [Google Scholar] [CrossRef] [Green Version]
- Schettini, F.; Prat, A. Dissecting the biological heterogeneity of HER2-positive breast cancer. Breast 2021, 59, 339–350. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group. Adjuvant chemotherapy in oestrogen-receptor-poor breast cancer: Patient-level meta-analysis of randomised trials. Lancet 2008, 371, 29–40. [Google Scholar] [CrossRef]
- Goldhirsch, A.; Winer, E.P.; Coates, A.S.; Gelber, R.D.; Piccart-Gebhart, M.; Thurlimann, B.; Senn, H.J.; Panel, M. Personalizing the treatment of women with early breast cancer: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2013, 24, 2206–2223. [Google Scholar] [CrossRef]
- Von Minckwitz, G.; Untch, M.; Blohmer, J.-U.; Costa, S.D.; Eidtmann, H.; Fasching, P.A.; Gerber, B.; Eiermann, W.; Hilfrich, J.; Huober, J.; et al. Definition and Impact of Pathologic Complete Response on Prognosis after Neoadjuvant Chemotherapy in Various Intrinsic Breast Cancer Subtypes. J. Clin. Oncol. 2012, 30, 1796–1804. [Google Scholar] [CrossRef] [Green Version]
- Sparano, J.A.; Gray, R.J.; Makower, D.F.; Pritchard, K.I.; Albain, K.S.; Hayes, D.F.; Geyer, C.E.J.; Dees, E.C.; Perez, E.A.; Olson, J.A.J.; et al. Prospective Validation of a 21-Gene Expression Assay in Breast Cancer. N. Engl. J. Med. 2015, 373, 2005–2014. [Google Scholar] [CrossRef]
- Gluz, O.; Nitz, U.A.; Christgen, M.; Kates, R.E.; Shak, S.; Clemens, M.; Kraemer, S.; Aktas, B.; Kuemmel, S.; Reimer, T.; et al. West German Study Group Phase III PlanB Trial: First Prospective Outcome Data for the 21-Gene Recurrence Score Assay and Concordance of Prognostic Markers by Central and Local Pathology Assessment. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 2341–2349. [Google Scholar] [CrossRef]
- Cardoso, F.; van’t Veer, L.J.; Bogaerts, J.; Slaets, L.; Viale, G.; Delaloge, S.; Pierga, J.-Y.; Brain, E.; Causeret, S.; DeLorenzi, M.; et al. 70-Gene Signature as an Aid to Treatment Decisions in Early-Stage Breast Cancer. N. Engl. J. Med. 2016, 375, 717–729. [Google Scholar] [CrossRef] [Green Version]
- Available online: https://www.ago-online.de/fileadmin/ago-online/downloads/_leitlinien/kommission_mamma/2021/Einzeldateien_Literatur/2021D_05_Prognostische_und_praediktive_Faktoren_MASTER_final_20210302_inklRef.pdf (accessed on 10 April 2021).
- Sestak, I.; Buus, R.; Cuzick, J.; Dubsky, P.; Kronenwett, R.; Ferree, S.; Sgroi, D.; Schnabel, C.; Baehner, R.; Mallon, E.; et al. Abstract S6-05: Comprehensive comparison of prognostic signatures for breast cancer in TransATAC. Cancer Res. 2017, 77 (Suppl. S4), S06-05. [Google Scholar] [CrossRef]
- Dubsky, P.; Brase, J.C.; Jakesz, R.; Rudas, M.; Singer, C.F.; Greil, R.; Dietze, O.; Luisser, I.; Klug, E.; Sedivy, R.; et al. The EndoPredict score provides prognostic information on late distant metastases in ER+/HER2- breast cancer patients. Br. J. Cancer 2013, 109, 2959–2964. [Google Scholar] [CrossRef] [Green Version]
- Dubsky, P.; Filipits, M.; Jakesz, R.; Rudas, M.; Singer, C.F.; Greil, R.; Dietze, O.; Luisser, I.; Klug, E.; Sedivy, R.; et al. EndoPredict improves the prognostic classification derived from common clinical guidelines in ER-positive, HER2-negative early breast cancer. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2013, 24, 640–647. [Google Scholar] [CrossRef]
- Filipits, M.; Rudas, M.; Jakesz, R.; Dubsky, P.; Fitzal, F.; Singer, C.F.; Dietze, O.; Greil, R.; Jelen, A.; Sevelda, P.; et al. A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2011, 17, 6012–6020. [Google Scholar] [CrossRef] [Green Version]
- Martin, M.; Brase, J.C.; Calvo, L.; Krappmann, K.; Ruiz-Borrego, M.; Fisch, K.; Ruiz, A.; Weber, K.E.; Munarriz, B.; Petry, C.; et al. Clinical validation of the EndoPredict test in node-positive, chemotherapy-treated ER+/HER2− breast cancer patients: Results from the GEICAM 9906 trial. Breast Cancer Res. 2014, 16, R38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paik, S.; Tang, G.; Shak, S.; Kim, C.; Baker, J.; Kim, W.; Cronin, M.; Baehner, F.L.; Watson, D.; Bryant, J.; et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2006, 24, 3726–3734. [Google Scholar] [CrossRef] [PubMed]
- Wolmark, N.; Mamounas, E.P.; Baehner, F.L.; Butler, S.M.; Tang, G.; Jamshidian, F.; Sing, A.P.; Shak, S.; Paik, S. Prognostic Impact of the Combination of Recurrence Score and Quantitative Estrogen Receptor Expression (ESR1) on Predicting Late Distant Recurrence Risk in Estrogen Receptor-Positive Breast Cancer after 5 Years of Tamoxifen: Results from NRG Oncology/National Surgical Adjuvant Breast and Bowel Project B-28 and B-14. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 2350–2358. [Google Scholar] [CrossRef] [Green Version]
- Albain, K.S.; Barlow, W.E.; Shak, S.; Hortobagyi, G.N.; Livingston, R.B.; Yeh, I.T.; Ravdin, P.; Bugarini, R.; Baehner, F.L.; Davidson, N.E.; et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: A retrospective analysis of a randomised trial. Lancet Oncol. 2010, 11, 55–65. [Google Scholar] [CrossRef] [Green Version]
- Sgroi, D.C.; Sestak, I.; Cuzick, J.; Zhang, Y.; Schnabel, C.A.; Schroeder, B.; Erlander, M.G.; Dunbier, A.; Sidhu, K.; Lopez-Knowles, E.; et al. Prediction of late distant recurrence in patients with oestrogen-receptor-positive breast cancer: A prospective comparison of the breast-cancer index (BCI) assay, 21-gene recurrence score, and IHC4 in the TransATAC study population. Lancet Oncol. 2013, 14, 1067–1076. [Google Scholar] [CrossRef] [Green Version]
- Filipits, M.; Nielsen, T.O.; Rudas, M.; Greil, R.; Stöger, H.; Jakesz, R.; Bago-Horvath, Z.; Dietze, O.; Regitnig, P.; Gruber-Rossipal, C.; et al. The PAM50 risk-of-recurrence score predicts risk for late distant recurrence after endocrine therapy in postmenopausal women with endocrine-responsive early breast cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 1298–1305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martín, M.; Prat, A.; Rodríguez-Lescure, A.; Caballero, R.; Ebbert, M.T.; Munárriz, B.; Ruiz-Borrego, M.; Bastien, R.R.; Crespo, C.; Davis, C.; et al. PAM50 proliferation score as a predictor of weekly paclitaxel benefit in breast cancer. Breast Cancer Res. Treat. 2013, 138, 457–466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gnant, M.; Filipits, M.; Greil, R.; Stoeger, H.; Rudas, M.; Bago-Horvath, Z.; Mlineritsch, B.; Kwasny, W.; Knauer, M.; Singer, C.; et al. Predicting distant recurrence in receptor-positive breast cancer patients with limited clinicopathological risk: Using the PAM50 Risk of Recurrence score in 1478 postmenopausal patients of the ABCSG-8 trial treated with adjuvant endocrine therapy alone. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2014, 25, 339–345. [Google Scholar] [CrossRef]
- Gnant, M.; Sestak, I.; Filipits, M.; Dowsett, M.; Balic, M.; Lopez-Knowles, E.; Greil, R.; Dubsky, P.; Stoeger, H.; Rudas, M.; et al. Identifying clinically relevant prognostic subgroups of postmenopausal women with node-positive hormone receptor-positive early-stage breast cancer treated with endocrine therapy: A combined analysis of ABCSG-8 and ATAC using the PAM50 risk of recurrence score and intrinsic subtype. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2015, 26, 1685–1691. [Google Scholar] [CrossRef]
- Andre, F.; Ismaila, N.; Henry, N.L.; Somerfield, M.R.; Bast, R.C.; Barlow, W.; Collyar, D.E.; Hammond, M.E.; Kuderer, N.M.; Liu, M.C.; et al. Use of Biomarkers to Guide Decisions on Adjuvant Systemic Therapy for Women With Early-Stage Invasive Breast Cancer: ASCO Clinical Practice Guideline Update—Integration of Results From TAILORx. Journal of Clinical Oncology 2019, 37, 1956–1964. [Google Scholar] [CrossRef] [Green Version]
- Krop, I.; Ismaila, N.; Andre, F.; Bast, R.C.; Barlow, W.; Collyar, D.E.; Hammond, M.E.; Kuderer, N.M.; Liu, M.C.; Mennel, R.G.; et al. Use of Biomarkers to Guide Decisions on Adjuvant Systemic Therapy for Women with Early-Stage Invasive Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline Focused Update. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2017, 35, 2838–2847. [Google Scholar] [CrossRef]
- Available online: https://www.accessdata.fda.gov/cdrh_docs/reviews/K130010.pdf (accessed on 9 April 2021).
- Available online: https://www.prosigna.com/de/overview/ (accessed on 9 April 2021).
- Dowsett, M.; Nielsen, T.O.; A’Hern, R.; Bartlett, J.; Coombes, R.C.; Cuzick, J.; Ellis, M.; Henry, N.L.; Hugh, J.C.; Lively, T.; et al. Assessment of Ki67 in breast cancer: Recommendations from the International Ki67 in Breast Cancer working group. J. Natl. Cancer Inst. 2011, 103, 1656–1664. [Google Scholar] [CrossRef] [Green Version]
- Harris, L.; Fritsche, H.; Mennel, R.; Norton, L.; Ravdin, P.; Taube, S.; Somerfield, M.R.; Hayes, D.F.; Bast, R.C., Jr. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2007, 25, 5287–5312. [Google Scholar] [CrossRef] [Green Version]
- Prat, A.; Cheang, M.C.; Martin, M.; Parker, J.S.; Carrasco, E.; Caballero, R.; Tyldesley, S.; Gelmon, K.; Bernard, P.S.; Nielsen, T.O.; et al. Prognostic significance of progesterone receptor-positive tumor cells within immunohistochemically defined luminal A breast cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2013, 31, 203–209. [Google Scholar] [CrossRef]
- Braun, L.; Mietzsch, F.; Seibold, P.; Schneeweiss, A.; Schirmacher, P.; Chang-Claude, J.; Peter Sinn, H.; Aulmann, S. Intrinsic breast cancer subtypes defined by estrogen receptor signalling—Prognostic relevance of progesterone receptor loss. Mod. Pathol. Off. J. United States Can. Acad. Pathol. Inc. 2013, 26, 1161–1171. [Google Scholar] [CrossRef] [Green Version]
- Rimm, D.L.; Leung, S.C.Y.; McShane, L.M.; Bai, Y.; Bane, A.L.; Bartlett, J.M.S.; Bayani, J.; Chang, M.C.; Dean, M.; Denkert, C.; et al. An international multicenter study to evaluate reproducibility of automated scoring for assessment of Ki67 in breast cancer. Mod. Pathol. Off. J. United States Can. Acad. Pathol. Inc. 2019, 32, 59–69. [Google Scholar] [CrossRef]
- Nielsen, T.O.; Leung, S.C.Y.; Rimm, D.L.; Dodson, A.; Acs, B.; Badve, S.; Denkert, C.; Ellis, M.J.; Fineberg, S.; Flowers, M.; et al. Assessment of Ki67 in Breast Cancer: Updated Recommendations from the International Ki67 in Breast Cancer Working Group. J. Natl. Cancer Inst. 2020. [Google Scholar] [CrossRef]
- Polley, M.Y.; Leung, S.C.; McShane, L.M.; Gao, D.; Hugh, J.C.; Mastropasqua, M.G.; Viale, G.; Zabaglo, L.A.; Penault-Llorca, F.; Bartlett, J.M.; et al. An international Ki67 reproducibility study. J. Natl. Cancer Inst. 2013, 105, 1897–1906. [Google Scholar] [CrossRef]
- Gnant, M.; Thomssen, C.; Harbeck, N. St. Gallen/Vienna 2015: A Brief Summary of the Consensus Discussion. Breast Care 2015, 10, 124–130. [Google Scholar] [CrossRef] [Green Version]
- Available online: https://documents.cap.org/protocols/cp-breast-biomarker-20-1400.pdf (accessed on 10 April 2021).
- Harris, L.N.; Ismaila, N.; McShane, L.M.; Andre, F.; Collyar, D.E.; Gonzalez-Angulo, A.M.; Hammond, E.H.; Kuderer, N.M.; Liu, M.C.; Mennel, R.G.; et al. Use of Biomarkers to Guide Decisions on Adjuvant Systemic Therapy for Women with Early-Stage Invasive Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 1134–1150. [Google Scholar] [CrossRef] [Green Version]
- Bastien, R.R.L.; Rodríguez-Lescure, Á.; Ebbert, M.T.W.; Prat, A.; Munárriz, B.; Rowe, L.; Miller, P.; Ruiz-Borrego, M.; Anderson, D.; Lyons, B.; et al. PAM50 Breast Cancer Subtyping by RT-qPCR and Concordance with Standard Clinical Molecular Markers. BMC Med. Genom. 2012, 5, 44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.K.; Park, K.H.; Kim, Y.; Park, S.E.; Lee, H.S.; Lim, S.W.; Cho, J.H.; Kim, J.Y.; Lee, J.E.; Ahn, J.S.; et al. Discordance of the PAM50 Intrinsic Subtypes Compared with Immunohistochemistry-Based Surrogate in Breast Cancer Patients: Potential Implication of Genomic Alterations of Discordance. Cancer Res. Treat. Off. J. Korean Cancer Assoc. 2019, 51, 737–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012, 490, 61–70. [Google Scholar] [CrossRef] [Green Version]
- Sweeney, C.; Bernard, P.S.; Factor, R.E.; Kwan, M.L.; Habel, L.A.; Quesenberry, C.P.; Shakespear, K.; Weltzien, E.K.; Stijleman, I.J.; Davis, C.A.; et al. Intrinsic subtypes from PAM50 gene expression assay in a population-based breast cancer cohort: Differences by age, race, and tumor characteristics. Cancer Epidemiol. Biomark. Prev. A Publ. Am. Assoc. Cancer Res. Cosponsored By Am. Soc. Prev. Oncol. 2014, 23, 714–724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picornell, A.C.; Echavarria, I.; Alvarez, E.; López-Tarruella, S.; Jerez, Y.; Hoadley, K.; Parker, J.S.; del Monte-Millán, M.; Ramos-Medina, R.; Gayarre, J.; et al. Breast cancer PAM50 signature: Correlation and concordance between RNA-Seq and digital multiplexed gene expression technologies in a triple negative breast cancer series. BMC Genom. 2019, 20, 452. [Google Scholar] [CrossRef] [Green Version]
- Sinn, P.; Aulmann, S.; Wirtz, R.; Schott, S.; Marmé, F.; Varga, Z.; Lebeau, A.; Kreipe, H.; Schneeweiss, A. Multigene Assays for Classification, Prognosis, and Prediction in Breast Cancer: A Critical Review on the Background and Clinical Utility. Geburtshilfe Und Frauenheilkd. 2013, 73, 932–940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prat, A.; Parker, J.S.; Fan, C.; Cheang, M.C.U.; Miller, L.D.; Bergh, J.; Chia, S.K.L.; Bernard, P.S.; Nielsen, T.O.; Ellis, M.J.; et al. Concordance among gene expression-based predictors for ER-positive breast cancer treated with adjuvant tamoxifen. Ann. Oncol. 2012, 23, 2866–2873. [Google Scholar] [CrossRef]
- Weigelt, B.; Reis-Filho, J.S. Histological and molecular types of breast cancer: Is there a unifying taxonomy? Nat. Rev. Clin. Oncol. 2009, 6, 718–730. [Google Scholar] [CrossRef]
- Prat, A.; Lluch, A.; Turnbull, A.K.; Dunbier, A.K.; Calvo, L.; Albanell, J.; de la Haba-Rodríguez, J.; Arcusa, A.; Chacón, J.I.; Sánchez-Rovira, P.; et al. A PAM50-Based Chemoendocrine Score for Hormone Receptor-Positive Breast Cancer with an Intermediate Risk of Relapse. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 3035–3044. [Google Scholar] [CrossRef] [Green Version]
- McVeigh, T.P.; Hughes, L.M.; Miller, N.; Sheehan, M.; Keane, M.; Sweeney, K.J.; Kerin, M.J. The impact of Oncotype DX testing on breast cancer management and chemotherapy prescribing patterns in a tertiary referral centre. Eur. J. Cancer 2014, 50, 2763–2770. [Google Scholar] [CrossRef] [Green Version]
- Ward, S.; Scope, A.; Rafia, R.; Pandor, A.; Harnan, S.; Evans, P.; Wyld, L. Gene expression profiling and expanded immunohistochemistry tests to guide the use of adjuvant chemotherapy in breast cancer management: A systematic review and cost-effectiveness analysis. Health Technol. Assess. 2013, 17, 1–302. [Google Scholar] [CrossRef] [Green Version]
- Seguí, M.Á.; Crespo, C.; Cortés, J.; Lluch, A.; Brosa, M.; Becerra, V.; Chiavenna, S.M.; Gracia, A. Genomic profile of breast cancer: Cost–effectiveness analysis from the Spanish National Healthcare System perspective. Expert Rev. Pharm. Outcomes Res. 2014, 14, 889–899. [Google Scholar] [CrossRef] [Green Version]
- Blohmer, J.U.; Rezai, M.; Kümmel, S.; Kühn, T.; Warm, M.; Friedrichs, K.; Benkow, A.; Valentine, W.J.; Eiermann, W. Using the 21-gene assay to guide adjuvant chemotherapy decision-making in early-stage breast cancer: A cost-effectiveness evaluation in the German setting. J. Med. Econ. 2013, 16, 30–40. [Google Scholar] [CrossRef]
- Hall, P.S.; Smith, A.; Hulme, C.; Vargas-Palacios, A.; Makris, A.; Hughes-Davies, L.; Dunn, J.A.; Bartlett, J.M.S.; Cameron, D.A.; Marshall, A.; et al. Value of Information Analysis of Multiparameter Tests for Chemotherapy in Early Breast Cancer: The OPTIMA Prelim Trial. Value Health 2017, 20, 1311–1318. [Google Scholar] [CrossRef] [Green Version]
- Arbeitsgemeinschaft Gynäkologische Onkologie e.V. Guidelines Breast Versions of 2015/2016 (Chapter Pathologie). Available online: www.ago-online.de (accessed on 7 April 2021).
- Von Minckwitz, G.; Blohmer, J.U.; Costa, S.D.; Denkert, C.; Eidtmann, H.; Eiermann, W.; Gerber, B.; Hanusch, C.; Hilfrich, J.; Huober, J.; et al. Response-guided neoadjuvant chemotherapy for breast cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2013, 31, 3623–3630. [Google Scholar] [CrossRef]
- Available online: https://www.prosigna.com/en-gb/downloads/ (accessed on 7 April 2021).
- R Core Team. R: A Language and Environment for Statistical Computing. 2020. Available online: https://www.R-project.org/ (accessed on 19 December 2021).
- Elston, C.W.; Ellis, I.O. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: Experience from a large study with long-term follow-up. Histopathology 1991, 19, 403–410. [Google Scholar]
- Hammond, M.E.; Hayes, D.F.; Dowsett, M.; Allred, D.C.; Hagerty, K.L.; Badve, S.; Fitzgibbons, P.L.; Francis, G.; Goldstein, N.S.; Hayes, M.; et al. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Clin. Oncol. 2010, 28, 2784–2795. [Google Scholar] [CrossRef] [Green Version]
- Wolff, A.C.; Hammond, D.E.; Hicks, D.G.; Dowsett, M.; McShane, L.M.; Allison, K.H.; Allred, D.C.; Bartlett, J.M.; Bilous, M.; Fitzgibbons, P.; et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J. Clin. Oncol. 2013, 31, 3997–4013. [Google Scholar] [CrossRef]
- Gamer, M.; Lemon, J.; Fellows, I.; Singh, P. irr: Various Coefficients of Interrater Reliability and Agreement. Available online: https://CRAN.R-project.org/package=irr (accessed on 26 January 2019).
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar]
- Bewick, V.; Cheek, L.; Ball, J. Statistics review 8: Qualitative data–tests of association. Crit. Care 2003, 8, 1–8. [Google Scholar]
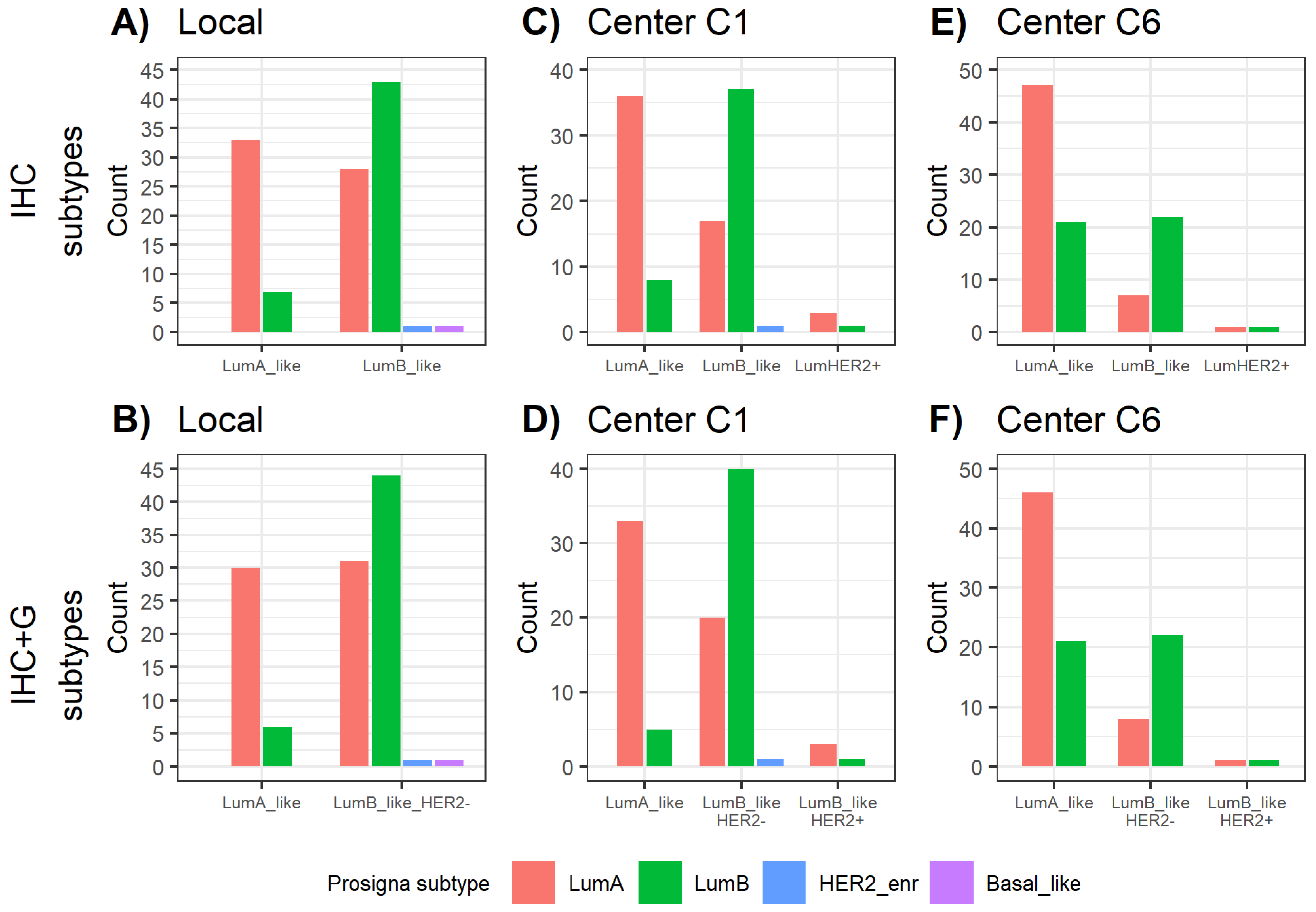
| Surrogate Subtypes | |||
|---|---|---|---|
| Luminal A-like | Luminal B-like HER2 negative | Luminal B-like HER2 positive | |
| Local IHC subtyping | 35.4% | 64.6% | // |
| Local IHC+G subtyping | 31.9% | 68.1% | // |
| C1 IHC subtyping | 42.7% | 53.4% | 3.9% |
| C1 IHC+G subtyping | 36.9% | 59.2% | 3.9% |
| C6 IHC subtyping | 68.7% | 29.3% | 2.0% |
| C6 IHC+G subtyping | 67.7% | 30.3% | 2.0% |
| Luminal A-Like | Luminal B-Like | |||
|---|---|---|---|---|
| Match with Prosigna® Luminal A | Upgrade to Prosigna® Luminal B | Match with Prosigna® Luminal B | Downgrade to Prosigna® Luminal A | |
| Local IHC subtyping | 82.5% (33/40) | 17.5% (7/40) | 58.9% (43/73) | 38.4% (28/73) |
| Local IHC+G subtyping | 83.3% (30/36) | 16.7% (6/36) | 57.1% (44/77) | 40.3% (31/77) |
| C1 IHC subtyping | 81.8% (36/44) | 18.2% (8/44) | 67.3% (37/55) | 30.9% (17/55) |
| C1 IHC+G subtyping | 86.8% (33/38) | 13.2% (5/38) | 65.6% (40/61) | 32.8% (20/61) |
| C6 IHC subtyping | 69.1% (47/68) | 30.9% (21/68) | 75.9% (22/29) | 24.1% (7/29) |
| C6 IHC+G subtyping | 68.7% (46/67) | 31.3% (21/67) | 73.3% (22/30) | 26.7% (8/30) |
| κ (IHC Subtype) | κ (IHC+G Subtype) | |
|---|---|---|
| Prosigna vs. local institutes | 0.374 | 0.344 |
| Prosigna vs. center C1 | 0.449 | 0.455 |
| Prosigna vs. center C6 | 0.379 | 0.36 |
| Surrogate Subtype | Subgroup | ER | PR | HER2 | Ki-67 (%) | |
|---|---|---|---|---|---|---|
| Luminal A-like | + | +/− | − | and | Low (<20%) | |
| Luminal B-like | HER2 negative | + | +/− | − | and | High (≥20%) |
| HER2 positive | + | +/− | + | Any value | ||
| HER2 positive (non-luminal) | − | − | + | Any value | ||
| Triple negative | − | − | − | Any value |
| Surrogate Subtype | Subgroup | ER | PR | HER2 | Tumor Grade | Ki-67 (%) | ||
|---|---|---|---|---|---|---|---|---|
| Luminal A-like | + | +/− | − | and | G1, G2 | or | Low (<20%) | |
| Luminal B-like | HER2 negative | + | +/− | − | and | G3 | or | High (≥20%) |
| HER2 positive | + | +/− | + | G1, G2, G3 | Any value | |||
| HER2 positive (non-luminal) | − | − | + | G1, G2, G3 | Any value | |||
| Triple negative | − | − | − | G1, G2, G3 | Any value |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erber, R.; Angeloni, M.; Stöhr, R.; Lux, M.P.; Ulbrich-Gebauer, D.; Pelz, E.; Bankfalvi, A.; Schmid, K.W.; Walter, R.F.H.; Vetter, M.; et al. Molecular Subtyping of Invasive Breast Cancer Using a PAM50-Based Multigene Expression Test-Comparison with Molecular-Like Subtyping by Tumor Grade/Immunohistochemistry and Influence on Oncologist’s Decision on Systemic Therapy in a Real-World Setting. Int. J. Mol. Sci. 2022, 23, 8716. https://doi.org/10.3390/ijms23158716
Erber R, Angeloni M, Stöhr R, Lux MP, Ulbrich-Gebauer D, Pelz E, Bankfalvi A, Schmid KW, Walter RFH, Vetter M, et al. Molecular Subtyping of Invasive Breast Cancer Using a PAM50-Based Multigene Expression Test-Comparison with Molecular-Like Subtyping by Tumor Grade/Immunohistochemistry and Influence on Oncologist’s Decision on Systemic Therapy in a Real-World Setting. International Journal of Molecular Sciences. 2022; 23(15):8716. https://doi.org/10.3390/ijms23158716
Chicago/Turabian StyleErber, Ramona, Miriam Angeloni, Robert Stöhr, Michael P. Lux, Daniel Ulbrich-Gebauer, Enrico Pelz, Agnes Bankfalvi, Kurt W. Schmid, Robert F. H. Walter, Martina Vetter, and et al. 2022. "Molecular Subtyping of Invasive Breast Cancer Using a PAM50-Based Multigene Expression Test-Comparison with Molecular-Like Subtyping by Tumor Grade/Immunohistochemistry and Influence on Oncologist’s Decision on Systemic Therapy in a Real-World Setting" International Journal of Molecular Sciences 23, no. 15: 8716. https://doi.org/10.3390/ijms23158716
APA StyleErber, R., Angeloni, M., Stöhr, R., Lux, M. P., Ulbrich-Gebauer, D., Pelz, E., Bankfalvi, A., Schmid, K. W., Walter, R. F. H., Vetter, M., Thomssen, C., Mayr, D., Klauschen, F., Sinn, P., Sotlar, K., Stering, K., Stenzinger, A., Wunderle, M., Fasching, P. A., ... Hartmann, A. (2022). Molecular Subtyping of Invasive Breast Cancer Using a PAM50-Based Multigene Expression Test-Comparison with Molecular-Like Subtyping by Tumor Grade/Immunohistochemistry and Influence on Oncologist’s Decision on Systemic Therapy in a Real-World Setting. International Journal of Molecular Sciences, 23(15), 8716. https://doi.org/10.3390/ijms23158716












