Nanofiber Carriers of Therapeutic Load: Current Trends
Abstract
:1. Introduction
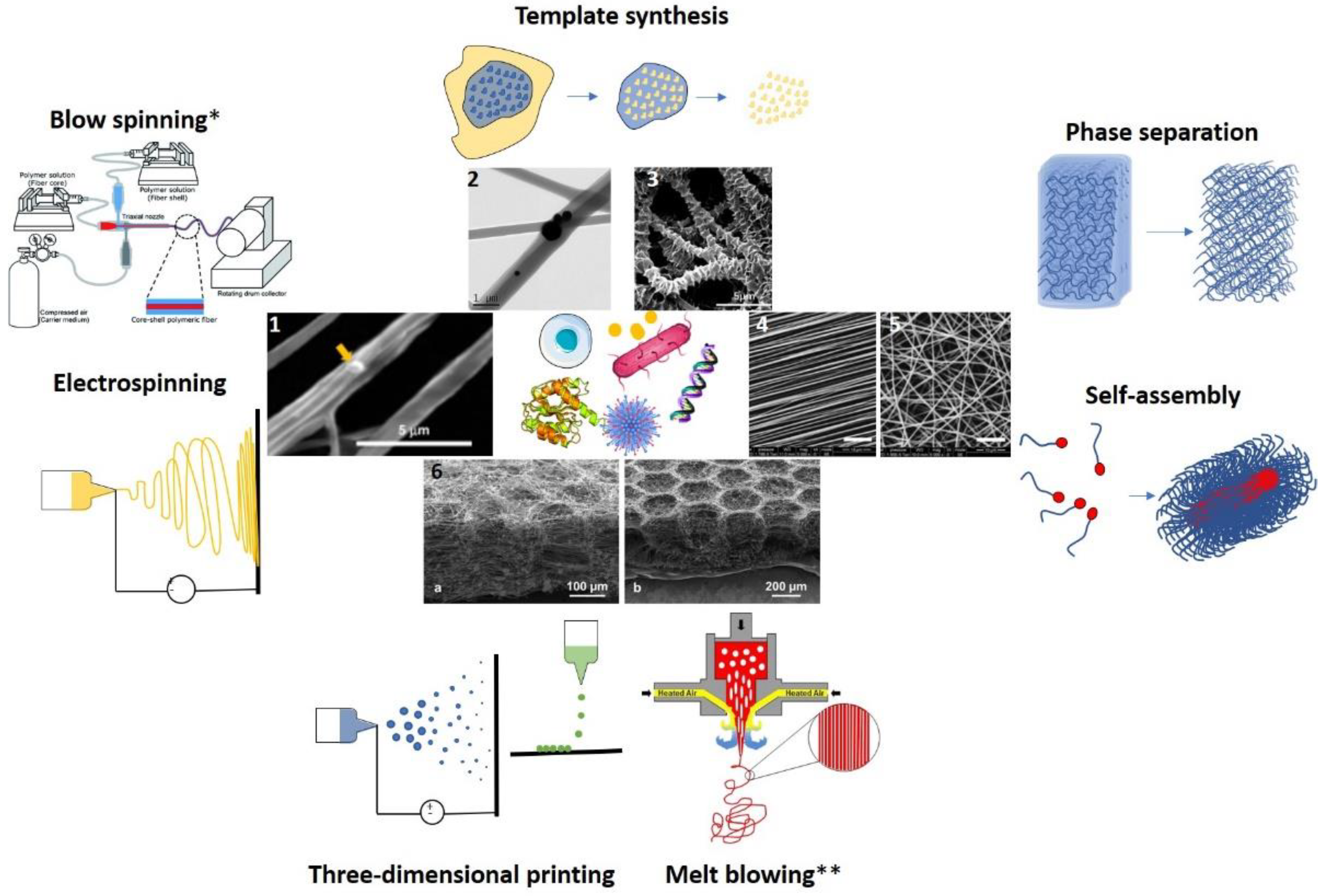
2. Methods of Preparation
2.1. Temple-Based Synthesis
2.2. Phase Separation
2.3. Self-Assembly
2.4. Three-Dimensional Printing
2.5. Electrospinning
2.5.1. Solution Parameters
2.5.2. Processing Parameters
2.5.3. Ambient Parameters
2.6. Blow-Spinning
2.7. Melt-Blowing
2.8. Other Methods
3. Delivery of Therapeutics
4. Therapeutic Applications
4.1. Delivery of Small Chemotherapeutic Molecules
4.2. Delivery of Peptides and Proteins
4.3. Delivery of Polynucleotides
4.4. Cell Delivery
5. Final Remarks and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ramakrishna, S.; Fujihara, K.; Teo, W.E.; Yong, T.; Ma, Z.; Ramaseshan, R. Electrospun nanofibers: Solving global issues. Mater. Today 2006, 9, 40–50. [Google Scholar] [CrossRef]
- Reddy, V.S.; Tian, Y.; Zhang, C.; Ye, Z.; Roy, K.; Chinnappan, A.; Ramakrishna, S.; Liu, W.; Ghosh, R. A Review on Electrospun Nanofibers Based Advanced Applications: From Health Care to Energy Devices. Polymers 2021, 13, 3746. [Google Scholar] [CrossRef] [PubMed]
- Intelligence, M. Nanofiber Market—Growth, Trends, COVID-19 Impact, and Forecasts (2022–2027); Global Information, Inc.: West Hartford, CT, USA, 2022. [Google Scholar]
- Stojanov, S.; Berlec, A. Electrospun Nanofibers as Carriers of Microorganisms, Stem Cells, Proteins, and Nucleic Acids in Therapeutic and Other Applications. Front. Bioeng. Biotechnol. 2020, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Omer, S.; Forgách, L.; Zelkó, R.; Sebe, I. Scale-up of Electrospinning: Market Overview of Products and Devices for Pharmaceutical and Biomedical Purposes. Pharmaceutics 2021, 13, 286. [Google Scholar] [CrossRef] [PubMed]
- Morelli, S.; Piscioneri, A.; Salerno, S.; De Bartolo, L. Hollow Fiber and Nanofiber Membranes in Bioartificial Liver and Neuronal Tissue Engineering. Cells Tissues Organs 2021, 211, 447–476. [Google Scholar] [CrossRef] [PubMed]
- US National Library of Medicine-NIH. Rotator Cuff Healing Using a Nanofiber Scaffold in Patients Greater Than 55 Years; NCT04325789; National Institutes of Health: Bethesda, MD, USA, 2020. [Google Scholar]
- Golestannejad, Z.; Khozeimeh, F.; Mehrasa, M.; Mirzaeei, S.; Sarfaraz, D. A novel drug delivery system using acyclovir nanofiber patch for topical treatment of recurrent herpes labialis: A randomized clinical trial. Clin. Exp. Dent. Res. 2022, 8, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Xi, K.; Gu, Y.; Tang, J.; Chen, H.; Xu, Y.; Wu, L.; Cai, F.; Deng, L.; Yang, H.; Shi, Q.; et al. Microenvironment-responsive immunoregulatory electrospun fibers for promoting nerve function recovery. Nat. Commun. 2020, 11, 4504. [Google Scholar] [CrossRef]
- Yang, J.; Wang, K.; Yu, D.-G.; Yang, Y.; Bligh, S.W.A.; Williams, G.R. Electrospun Janus nanofibers loaded with a drug and inorganic nanoparticles as an effective antibacterial wound dressing. Mater. Sci. Eng. C 2020, 111, 110805. [Google Scholar] [CrossRef]
- Huang, C.; Yang, G.; Zhou, S.; Luo, E.; Pan, J.; Bao, C.; Liu, X. Controlled Delivery of Growth Factor by Hierarchical Nanostructured Core–Shell Nanofibers for the Efficient Repair of Critical-Sized Rat Calvarial Defect. ACS Biomater. Sci. Eng. 2020, 6, 5758–5770. [Google Scholar] [CrossRef]
- Yu, C.-C.; Chen, Y.-W.; Yeh, P.-Y.; Hsiao, Y.-S.; Lin, W.-T.; Kuo, C.-W.; Chueh, D.-Y.; You, Y.-W.; Shyue, J.-J.; Chang, Y.-C.; et al. Random and aligned electrospun PLGA nanofibers embedded in microfluidic chips for cancer cell isolation and integration with air foam technology for cell release. J. Nanobiotechnol. 2019, 17, 31. [Google Scholar] [CrossRef] [Green Version]
- Wittmer, C.R.; Hébraud, A.; Nedjari, S.; Schlatter, G. Well-organized 3D nanofibrous composite constructs using cooperative effects between electrospinning and electrospraying. Polymer 2014, 55, 5781–5787. [Google Scholar] [CrossRef]
- Chan Park, S.; Kim, M.J.; Choi, K.; Kim, J.; Choi, S.-O. Influence of shell compositions of solution blown PVP/PCL core–shell fibers on drug release and cell growth. RSC Adv. 2018, 8, 32470–32480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuo, F.; Tan, D.H.; Wang, Z.; Jeung, S.; Macosko, C.W.; Bates, F.S. Nanofibers from Melt Blown Fiber-in-Fiber Polymer Blends. ACS Macro Lett. 2013, 2, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Lamm, M.E.; Li, K.; Qian, J.; Wang, L.; Lavoine, N.; Newman, R.; Gardner, D.J.; Li, T.; Hu, L.; Ragauskas, A.J.; et al. Recent Advances in Functional Materials through Cellulose Nanofiber Templating. Adv. Mater. 2021, 33, 2005538. [Google Scholar] [CrossRef] [PubMed]
- Hua, F.; Kim, G.; Lee, J.; Son, Y.; Lee, D.-S. Macroporous poly(L-lactide) scaffold 1. Preparation of a macroporous scaffold by liquid–liquid phase separation of a PLLA–dioxane–water system. J. Biomed. Mater. Res. 2002, 63, 161–167. [Google Scholar] [CrossRef]
- Kang, H.J.; Chen, N.; Dash, B.C.; Hsia, H.C.; Berthiaume, F. Self-Assembled Nanomaterials for Chronic Skin Wound Healing. Adv. Wound Care 2021, 10, 221–233. [Google Scholar] [CrossRef] [PubMed]
- La Manna, S.; Di Natale, C.; Onesto, V.; Marasco, D. Self-Assembling Peptides: From Design to Biomedical Applications. Int. J. Mol. Sci. 2021, 22, 12662. [Google Scholar] [CrossRef]
- Hartgerink, J.; Beniash, E.; Stupp, S. Self-Assembly and Mineralization of Peptide-Amphiphile Nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, P.; Cheetham, A.G.; Lin, Y.-a.; Cui, H. Self-Assembled Tat Nanofibers as Effective Drug Carrier and Transporter. ACS Nano 2013, 7, 5965–5977. [Google Scholar] [CrossRef] [Green Version]
- Mohamad, S.A.; Zahran, E.M.; Abdel Fadeel, M.R.; Albohy, A.; Safwat, M.A. New Acaciin-Loaded Self-Assembled Nanofibers as M(Pro) Inhibitors Against BCV as a Surrogate Model for SARS-CoV-2. Int. J. Nanomed. 2021, 16, 1789–1804. [Google Scholar] [CrossRef]
- Mittal, A.; Krishna; Aarti; Prasad, S.; Mishra, P.K.; Sharma, S.K.; Parshad, B. Self-assembly of carbohydrate-based small amphiphiles and their applications in pathogen inhibition and drug delivery: A review. Mater. Adv. 2021, 2, 3459–3473. [Google Scholar] [CrossRef]
- Mendes, A.C.; Strohmenger, T.; Goycoolea, F.; Chronakis, I.S. Electrostatic self-assembly of polysaccharides into nanofibers. Colloids Surf. A Physicochem. Eng. Asp. 2017, 531, 182–188. [Google Scholar] [CrossRef]
- Liu, N.; Zhu, L.; Li, Z.; Liu, W.; Sun, M.; Zhou, Z. In situ self-assembled peptide nanofibers for cancer theranostics. Biomater. Sci. 2021, 9, 5427–5436. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.-B.; Gao, Y.-J.; Wang, L.; Wang, H. Self-Assembled Peptide-Based Nanomaterials for Biomedical Imaging and Therapy. Adv. Mater. 2018, 30, 1703444. [Google Scholar] [CrossRef]
- Xu, L.; Wang, Y.; Zhu, C.; Ren, S.; Shao, Y.; Wu, L.; Li, W.; Jia, X.; Hu, R.; Chen, R.; et al. Morphological transformation enhances Tumor Retention by Regulating the Self-assembly of Doxorubicin-peptide Conjugates. Theranostics 2020, 10, 8162–8178. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, L.H.; Qiu, W.X.; Zhang, Y.H.; Song, W.; Zhang, L.; Wang, S.B.; Zhang, X.Z. A Transformable Chimeric Peptide for Cell Encapsulation to Overcome Multidrug Resistance. Small 2018, 14, e1703321. [Google Scholar] [CrossRef]
- Kalafatovic, D.; Nobis, M.; Son, J.; Anderson, K.I.; Ulijn, R.V. MMP-9 triggered self-assembly of doxorubicin nanofiber depots halts tumor growth. Biomaterials 2016, 98, 192–202. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Zhang, W.; Huang, Y.; Gao, F.; Fang, X. Dual-functional c(RGDyK)-decorated Pluronic micelles designed for antiangiogenesis and the treatment of drug-resistant tumor. Int. J. Nanomed. 2015, 10, 4863–4881. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.-X.; Li, L.-L.; Zhao, Y.; An, H.-W.; Cai, Q.; Lang, J.-Y.; Han, X.-X.; Peng, B.; Fei, Y.; Liu, H.; et al. In Situ Self-Assembled Nanofibers Precisely Target Cancer-Associated Fibroblasts for Improved Tumor Imaging. Angew. Chem. 2019, 58, 15287–15294. [Google Scholar] [CrossRef]
- Liu, L.-H.; Zhang, X.-Z. Carrier-free nanomedicines for cancer treatment. Prog. Mater. Sci. 2022, 125, 100919. [Google Scholar] [CrossRef]
- Mello, L.R.; Hamley, I.W.; Castelletto, V.; Garcia, B.B.M.; Lourenço, T.C.; Vassiliades, S.V.; Alves, W.A.; Han, S.W.; Silva, E.R. Self-assembly and intracellular delivery of DNA by a truncated fragment derived from the Trojan peptide Penetratin. Soft Matter 2020, 16, 4746–4755. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, Y.; Yan, J.; Zhang, K.; Lin, F.; Xiang, L.; Deng, L.; Guan, Z.; Cui, W.; Zhang, H. Pharmaceutical electrospinning and 3D printing scaffold design for bone regeneration. Adv. Drug Deliv. Rev. 2021, 174, 504–534. [Google Scholar] [CrossRef]
- Levato, R.; Jungst, T.; Scheuring, R.G.; Blunk, T.; Groll, J.; Malda, J. From Shape to Function: The Next Step in Bioprinting. Adv. Mater. 2020, 32, 1906423. [Google Scholar] [CrossRef] [PubMed]
- Olmos-Juste, R.; Alonso-Lerma, B.; Pérez-Jiménez, R.; Gabilondo, N.; Eceiza, A. 3D printed alginate-cellulose nanofibers based patches for local curcumin administration. Carbohydr. Polym. 2021, 264, 118026. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Mele, E. Electrospinning and Additive Manufacturing: Adding Three-Dimensionality to Electrospun Scaffolds for Tissue Engineering. Front. Bioeng. Biotechnol. 2021, 9, 1238. [Google Scholar] [CrossRef] [PubMed]
- Rajzer, I.; Kurowska, A.; Jabłoński, A.; Jatteau, S.; Śliwka, M.; Ziąbka, M.; Menaszek, E. Layered gelatin/PLLA scaffolds fabricated by electrospinning and 3D printing- for nasal cartilages and subchondral bone reconstruction. Mater. Des. 2018, 155, 297–306. [Google Scholar] [CrossRef]
- Wang, C.; Yang, Y.; Ji, J.; Fang, Y.; Ouyang, L.; Zhang, L.; Sun, W. 3D-printed Bioresorbable Stent Coated with Dipyridamole-Loaded Nanofiber for Restenosis Prevention and Endothelialization. Int. J. Bioprinting 2022, 8, 543. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Y.; Yu, H.-C.; Liu, Y.; Liang, G.; Zhang, T.; Hu, Q.-X. Dual drug spatiotemporal release from functional gradient scaffolds prepared using 3D bioprinting and electrospinning. Polym. Eng. Sci. 2016, 56, 170–177. [Google Scholar] [CrossRef]
- Duraj-Thatte, A.M.; Manjula-Basavanna, A.; Rutledge, J.; Xia, J.; Hassan, S.; Sourlis, A.; Rubio, A.G.; Lesha, A.; Zenkl, M.; Kan, A.; et al. Programmable microbial ink for 3D printing of living materials produced from genetically engineered protein nanofibers. Nat. Commun. 2021, 12, 6600. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L.; Fong, H.; Koombhongse, S. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J. Appl. Phys. 2000, 87, 4531–4547. [Google Scholar] [CrossRef] [Green Version]
- Rošic, R.; Pelipenko, J.; Kocbek, P.; Baumgartner, S.; Bešter-Rogač, M.; Kristl, J. The role of rheology of polymer solutions in predicting nanofiber formation by electrospinning. Eur. Polym. J. 2012, 48, 1374–1384. [Google Scholar] [CrossRef]
- Gupta, P.; Elkins, C.; Long, T.E.; Wilkes, G.L. Electrospinning of linear homopolymers of poly(methyl methacrylate): Exploring relationships between fiber formation, viscosity, molecular weight and concentration in a good solvent. Polymer 2005, 46, 4799–4810. [Google Scholar] [CrossRef]
- McKee, M.G.; Wilkes, G.L.; Colby, R.H.; Long, T.E. Correlations of Solution Rheology with Electrospun Fiber Formation of Linear and Branched Polyesters. Macromolecules 2004, 37, 1760–1767. [Google Scholar] [CrossRef]
- Palangetic, L.; Reddy, N.K.; Srinivasan, S.; Cohen, R.E.; McKinley, G.H.; Clasen, C. Dispersity and spinnability: Why highly polydisperse polymer solutions are desirable for electrospinning. Polymer 2014, 55, 4920–4931. [Google Scholar] [CrossRef] [Green Version]
- Mirtič, J.; Balažic, H.; Zupančič, Š.; Kristl, J. Effect of Solution Composition Variables on Electrospun Alginate Nanofibers: Response Surface Analysis. Polymers 2019, 11, 692. [Google Scholar] [CrossRef] [Green Version]
- Geng, X.; Kwon, O.H.; Jang, J. Electrospinning of chitosan dissolved in concentrated acetic acid solution. Biomaterials 2005, 26, 5427–5432. [Google Scholar] [CrossRef]
- Josef, E.; Guterman, R. Designing Solutions for Electrospinning of Poly(ionic liquid)s. Macromolecules 2019, 52, 5223–5230. [Google Scholar] [CrossRef] [Green Version]
- Song, Z.; Chiang, S.W.; Chu, X.; Du, H.; Li, J.; Gan, L.; Xu, C.; Yao, Y.; He, Y.; Li, B.; et al. Effects of solvent on structures and properties of electrospun poly(ethylene oxide) nanofibers. J. Appl. Polym. Sci. 2018, 135, 45787. [Google Scholar] [CrossRef]
- Eren Boncu, T.; Ozdemir, N.; Uskudar Guclu, A. Electrospinning of linezolid loaded PLGA nanofibers: Effect of solvents on its spinnability, drug delivery, mechanical properties, and antibacterial activities. Drug Dev. Ind. Pharm. 2020, 46, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Deitzel, J.M.; Knopf, J.; Chen, X.; Gillespie, J.W., Jr. The effect of solvent dielectric properties on the collection of oriented electrospun fibers. J. Appl. Polym. Sci. 2012, 125, 2585–2594. [Google Scholar] [CrossRef]
- Luo, C.J.; Stride, E.; Edirisinghe, M. Mapping the Influence of Solubility and Dielectric Constant on Electrospinning Polycaprolactone Solutions. Macromolecules 2012, 45, 4669–4680. [Google Scholar] [CrossRef]
- Angammana, C.; Jayaram, S. Analysis of the Effects of Solution Conductivity on Electrospinning Process and Fiber Morphology. IEEE Trans. Ind. Appl. 2011, 47, 1109–1117. [Google Scholar] [CrossRef]
- Yalcinkaya, F.; Yalcinkaya, B.; Jirsak, O. Influence of Salts on Electrospinning of Aqueous and Nonaqueous Polymer Solutions. J. Nanomater. 2015, 2015, 134251. [Google Scholar] [CrossRef]
- Wang, L.; Yang, H.; Hou, J.; Zhang, W.; Xiang, C.; Li, L. Effect of the electrical conductivity of core solutions on the morphology and structure of core–shell CA-PCL/CS nanofibers. New J. Chem. 2017, 41, 15072–15078. [Google Scholar] [CrossRef]
- Cramariuc, B.; Cramariuc, R.; Scarlet, R.; Manea, L.R.; Lupu, I.G.; Cramariuc, O. Fiber diameter in electrospinning process. J. Electrost. 2013, 71, 189–198. [Google Scholar] [CrossRef]
- Korycka, P.; Mirek, A.; Kramek-Romanowska, K.; Grzeczkowicz, M.; Lewińska, D. Effect of electrospinning process variables on the size of polymer fibers and bead-on-string structures established with a 2(3) factorial design. Beilstein J. Nanotechnol. 2018, 9, 2466–2478. [Google Scholar] [CrossRef] [Green Version]
- Haider, A.; Haider, S.; Kang, I.-K. A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 2018, 11, 1165–1188. [Google Scholar] [CrossRef]
- Liu, Z.; Ju, K.; Wang, Z.; Li, W.; Ke, H.; He, J. Electrospun Jets Number and Nanofiber Morphology Effected by Voltage Value: Numerical Simulation and Experimental Verification. Nanoscale Res. Lett. 2019, 14, 310. [Google Scholar] [CrossRef] [PubMed]
- Mirek, A.; Korycka, P.; Grzeczkowicz, M.; Lewińska, D. Polymer fibers electrospun using pulsed voltage. Mater. Des. 2019, 183, 108106. [Google Scholar] [CrossRef]
- Koenig, K.; Beukenberg, K.; Langensiepen, F.; Seide, G. A new prototype melt-electrospinning device for the production of biobased thermoplastic sub-microfibers and nanofibers. Biomater. Res. 2019, 23, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pathalamuthu, P.; Siddharthan, A.; Giridev, V.R. Spirograph based electrospinning system for producing fibre mat with near uniform mechanical property. J. Fibre Text. Res. 2019, 44, 279–285. [Google Scholar]
- Nitti, P.; Gallo, N.; Natta, L.; Scalera, F.; Palazzo, B.; Sannino, A.; Gervaso, F. Influence of Nanofiber Orientation on Morphological and Mechanical Properties of Electrospun Chitosan Mats. J. Healthc. Eng. 2018, 2018, 3651480. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Hussain, T.; Nazir, A.; Khenoussi, N.; Cheema, S.A. Oriented electrospun nanofibers on stand-alone multi-segmented cylindrical collectors. J. Text. Inst. 2021, 112, 955–964. [Google Scholar] [CrossRef]
- Eom, S.; Park, S.M.; Hong, H.; Kwon, J.; Oh, S.-R.; Kim, J.; Kim, D.S. Hydrogel-Assisted Electrospinning for Fabrication of a 3D Complex Tailored Nanofiber Macrostructure. ACS Appl. Mater. Interfaces 2020, 12, 51212–51224. [Google Scholar] [CrossRef]
- Kim, I.G.; Lee, J.-H.; Unnithan, A.R.; Park, C.-H.; Kim, C.S. A comprehensive electric field analysis of cylinder-type multi-nozzle electrospinning system for mass production of nanofibers. J. Ind. Eng. Chem. 2015, 31, 251–256. [Google Scholar] [CrossRef]
- Hirsch, E.; Vass, P.; Démuth, B.; Pethő, Z.; Bitay, E.; Andersen, S.; Vigh, T.; Verreck, G.; Molnar, K.; Nagy, Z.; et al. Electrospinning scale-up and formulation development of PVA nanofibers aiming oral delivery of biopharmaceuticals. Express Polym. Lett. 2019, 13, 590–603. [Google Scholar] [CrossRef]
- Moon, S.; Gil, M.; Lee, K.J. Syringeless Electrospinning toward Versatile Fabrication of Nanofiber Web. Sci. Rep. 2017, 7, 41424. [Google Scholar] [CrossRef] [Green Version]
- Hardick, O.; Stevens, B.; Bracewell, D.G. Nanofibre fabrication in a temperature and humidity controlled environment for improved fibre consistency. J. Mater. Sci. 2011, 46, 3890–3898. [Google Scholar] [CrossRef] [Green Version]
- Putti, M.; Simonet, M.; Solberg, R.; Peters, G.W.M. Electrospinning poly(ε-caprolactone) under controlled environmental conditions: Influence on fiber morphology and orientation. Polymer 2015, 63, 189–195. [Google Scholar] [CrossRef]
- Zhang, D.; Davoodi, P.; Li, X.; Liu, Y.; Wang, W.; Huang, Y.Y.S. An empirical model to evaluate the effects of environmental humidity on the formation of wrinkled, creased and porous fibre morphology from electrospinning. Sci. Rep. 2020, 10, 18783. [Google Scholar] [CrossRef]
- Yang, G.-Z.; Li, H.-P.; Yang, J.-H.; Wan, J.; Yu, D.-G. Influence of Working Temperature on The Formation of Electrospun Polymer Nanofibers. Nanoscale Res. Lett. 2017, 12, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van-Pham, D.-T.; Tran, Q.; Toan, P.; Nguyen, C.-N.; Ho, M.; Thien, H. Temperature effects on electrospun chitosan nanofibers. Green Processing Synth. 2020, 9, 488–495. [Google Scholar] [CrossRef]
- Li, Z.; Cui, Z.; Zhao, L.; Hussain, N.; Zhao, Y.; Yang, C.; Jiang, X.; Li, L.; Song, J.; Zhang, B.; et al. High-throughput production of kilogram-scale nanofibers by Kármán vortex solution blow spinning. Sci. Adv. 2022, 8, eabn3690. [Google Scholar] [CrossRef]
- Vasireddi, R.; Kruse, J.; Vakili, M.; Kulkarni, S.; Keller, T.F.; Monteiro, D.C.F.; Trebbin, M. Solution blow spinning of polymer/nanocomposite micro-/nanofibers with tunable diameters and morphologies using a gas dynamic virtual nozzle. Sci. Rep. 2019, 9, 14297. [Google Scholar] [CrossRef] [Green Version]
- Schuchard, K.; Joijode, A.; Willard, V.P.; Anderson, B.; Grondin, P.; Pourdeyhimi, B.; Shirwaiker, R. Fabrication of drug-loaded ultrafine polymer fibers via solution blowing and their drug release kinetics. Procedia Manuf. 2021, 53, 128–135. [Google Scholar] [CrossRef]
- Czarnecka, K.; Wojasiński, M.; Ciach, T.; Sajkiewicz, P. Solution Blow Spinning of Polycaprolactone-Rheological Determination of Spinnability and the Effect of Processing Conditions on Fiber Diameter and Alignment. Materials 2021, 14, 1463. [Google Scholar] [CrossRef]
- Singh, R.; Ahmed, F.; Polley, P.; Giri, J. Fabrication and Characterization of Core-Shell Nanofibers Using a Next-Generation Airbrush for Biomedical Applications. ACS Appl. Mater. Interfaces 2018, 10, 41924–41934. [Google Scholar] [CrossRef]
- Tutak, W.; Gelven, G.; Markle, C.; Palmer, X.-L. Rapid polymer fiber airbrushing: Impact of a device design on the fiber fabrication and matrix quality. J. Appl. Polym. Sci. 2015, 132, 42813. [Google Scholar] [CrossRef]
- Daristotle, J.L.; Behrens, A.M.; Sandler, A.D.; Kofinas, P. A Review of the Fundamental Principles and Applications of Solution Blow Spinning. ACS Appl. Mater. Interfaces 2016, 8, 34951–34963. [Google Scholar] [CrossRef] [Green Version]
- Wojasiński, M.; Pilarek, M.; Ciach, T. Comparative Studies of Electrospinning and Solution Blow Spinning Processes for the Production of Nanofibrous Poly(L-Lactic Acid) Materials for Biomedical Engineering. Pol. J. Chem. Technol. 2014, 16, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Souza, M.A.; Sakamoto, K.Y.; Mattoso, L.H.C. Release of the Diclofenac Sodium by Nanofibers of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) Obtained from Electrospinning and Solution Blow Spinning. J. Nanomater. 2014, 2014, 129035. [Google Scholar] [CrossRef] [Green Version]
- DeFrates, K.; Markiewicz, T.; Xue, Y.; Callaway, K.; Gough, C.; Moore, R.; Bessette, K.; Mou, X.; Hu, X. Air-jet spinning corn zein protein nanofibers for drug delivery: Effect of biomaterial structure and shape on release properties. Mater. Sci. Eng. C 2021, 118, 111419. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Matharu, R.; Shams, T.; Illangakoon, E.; Edirisinghe, M. A Comparison of Electric-Field-Driven and Pressure-Driven Fiber Generation Methods for Drug Delivery. Macromol. Mater. Eng. 2018, 303, 1700577. [Google Scholar] [CrossRef]
- Heseltine, P.; Ahmed, J.; Edirisinghe, M. Developments in Pressurized Gyration for the Mass Production of Polymeric Fibers. Macromol. Mater. Eng. 2018, 303, 1800218. [Google Scholar] [CrossRef] [Green Version]
- Brako, F.; Raimi-Abraham, B.T.; Mahalingam, S.; Craig, D.Q.M.; Edirisinghe, M. The development of progesterone-loaded nanofibers using pressurized gyration: A novel approach to vaginal delivery for the prevention of pre-term birth. Int. J. Pharm. 2018, 540, 31–39. [Google Scholar] [CrossRef]
- Mahalingam, S.; Huo, S.; Homer-Vanniasinkam, S.; Edirisinghe, M. Generation of Core-Sheath Polymer Nanofibers by Pressurised Gyration. Polymers 2020, 12, 1709. [Google Scholar] [CrossRef] [PubMed]
- Balogh, A.; Farkas, B.; Faragó, K.; Farkas, A.; Wagner, I.; Van Assche, I.; Verreck, G.; Nagy, Z.; Marosi, G. Melt-Blown and Electrospun Drug-Loaded Polymer Fiber Mats for Dissolution Enhancement: A Comparative Study. J. Pharm. Sci. 2015, 104, 1767–1776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, P.; Meena, A. Application of enzyme-mediated cellulose nanofibers from lemongrass waste for the controlled release of anticancer drugs. Environ. Sci. Pollut. Res. 2021, 28, 46343–46355. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, H.; Xu, G.; Hou, X.; He, H.; Wang, S. A biocompatible cellulose-nanofiber-based multifunctional material for Fe3+ detection and drug delivery. J. Mater. Chem. C 2020, 8, 11796–11804. [Google Scholar] [CrossRef]
- Zhong, C. Industrial-Scale Production and Applications of Bacterial Cellulose. Front. Bioeng. Biotechnol. 2020, 8, 605374. [Google Scholar] [CrossRef]
- Murase, S.K.; Lv, L.P.; Kaltbeitzel, A.; Landfester, K.; del Valle, L.J.; Katsarava, R.; Puiggali, J.; Crespy, D. Amino acid-based poly(ester amide) nanofibers for tailored enzymatic degradation prepared by miniemulsion-electrospinning. RSC Adv. 2015, 5, 55006–55014. [Google Scholar] [CrossRef] [Green Version]
- Zuidema, J.M.; Kumeria, T.; Kim, D.; Kang, J.; Wang, J.; Hollett, G.; Zhang, X.; Roberts, D.S.; Chan, N.; Dowling, C.; et al. Oriented Nanofibrous Polymer Scaffolds Containing Protein-Loaded Porous Silicon Generated by Spray Nebulization. Adv. Mater. 2018, 30, 1706785. [Google Scholar] [CrossRef] [PubMed]
- Alisani, R.; Rakhshani, N.; Abolhallaj, M.; Motevalli, F.; Abadi, P.G.-S.; Akrami, M.; Shahrousvand, M.; Jazi, F.S.; Irani, M. Adsorption, and controlled release of doxorubicin from cellulose acetate/polyurethane/multi-walled carbon nanotubes composite nanofibers. Nanotechnology 2022, 33, 155102. [Google Scholar] [CrossRef]
- Lee, S.J.; Nah, H.; Ko, W.-K.; Lee, D.; Moon, H.-J.; Lee, J.S.; Heo, M.; Hwang, Y.-S.; Bang, J.B.; An, S.-H.; et al. Facile Preparation of β-Cyclodextrin-grafted Chitosan Electrospun Nanofibrous Scaffolds as a Hydrophobic Drug Delivery Vehicle for Tissue Engineering Applications. ACS Omega 2021, 6, 28307–28315. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Nakayama, H.; Morita, Y.; Sakamoto, H.; Kitamura, T.; Hashimoto, M.; Suye, S.-I. Enhanced and Prolonged Activity of Enzymes Adsorbed on TEMPO-Oxidized Cellulose Nanofibers. ACS Omega 2020, 5, 18826–18830. [Google Scholar] [CrossRef] [PubMed]
- Udomluck, N.; Lee, H.; Hong, S.; Lee, S.-H.; Park, H. Surface functionalization of dual growth factor on hydroxyapatite-coated nanofibers for bone tissue engineering. Appl. Surf. Sci. 2020, 520, 146311. [Google Scholar] [CrossRef]
- Okada, T.; Niiyama, E.; Uto, K.; Aoyagi, T.; Ebara, M. Inactivated Sendai Virus (HVJ-E) Immobilized Electrospun Nanofiber for Cancer Therapy. Materials 2015, 9, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niemczyk-Soczynska, B.; Gradys, A.; Sajkiewicz, P. Hydrophilic Surface Functionalization of Electrospun Nanofibrous Scaffolds in Tissue Engineering. Polymers 2020, 12, 2636. [Google Scholar] [CrossRef]
- Schneider, R.; Facure, M.H.M.; Chagas, P.A.M.; Andre, R.S.; dos Santos, D.M.; Correa, D.S. Tailoring the Surface Properties of Micro/Nanofibers Using 0D, 1D, 2D, and 3D Nanostructures: A Review on Post-Modification Methods. Adv. Mater. Interfaces 2021, 8, 2100430. [Google Scholar] [CrossRef]
- Smith, S.; Goodge, K.; Delaney, M.; Struzyk, A.; Tansey, N.; Frey, M. A Comprehensive Review of the Covalent Immobilization of Biomolecules onto Electrospun Nanofibers. Nanomaterials 2020, 10, 2142. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, Z.; Gu, J.g.; Zhou, W.; Liang, X.; Zhou, G.; Han, C.C.; Xu, S.; Liu, Y. Mechanism of a long-term controlled drug release system based on simple blended electrospun fibers. J. Control. Release 2020, 320, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Amarjargal, A.; Altenried, S.; Liu, M.; Zuber, F.; Zeng, Z.; Rossi, R.M.; Maniura-Weber, K.; Ren, Q. Bioresponsive Hybrid Nanofibers Enable Controlled Drug Delivery through Glass Transition Switching at Physiological Temperature. ACS Appl. Bio Mater. 2021, 4, 4271–4279. [Google Scholar] [CrossRef] [PubMed]
- Amarjargal, A.; Brunelli, M.; Fortunato, G.; Spano, F.; Kim, C.S.; Rossi, R.M. On-demand drug release from tailored blended electrospun nanofibers. J. Drug Deliv. Sci. Technol. 2019, 52, 8–14. [Google Scholar] [CrossRef]
- Arroub, K.; Gessner, I.; Fischer, T.; Mathur, D.S. Thermo-Responsive Poly (N-isopropylacrylamide)/Polycaprolacton Nanofibrous Scaffolds for Controlled Release of Antibiotics. Adv. Eng. Mater. 2021, 23, 2100221. [Google Scholar] [CrossRef]
- Pawłowska, S.; Rinoldi, C.; Nakielski, P.; Ziai, Y.; Urbanek, O.; Li, X.; Kowalewski, T.A.; Ding, B.; Pierini, F. Ultraviolet Light-Assisted Electrospinning of Core–Shell Fully Cross-Linked P(NIPAAm-co-NIPMAAm) Hydrogel-Based Nanofibers for Thermally Induced Drug Delivery Self-Regulation. Adv. Mater. Interfaces 2020, 7, 2000247. [Google Scholar] [CrossRef]
- Chen, R.; Lin, L.; Wang, H.; Zhai, X.; Liang, Y.; Zhao, B.; Yu, Z.; Li, K.; Shen, W. Effects of Morphologies of Thermo-sensitive Electrospun Nanofibers on Controllable Drug Release. Tissue Eng. Part A 2020, 27, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Williams, G.R.; Wu, J.; Lv, Y.; Sun, X.; Wu, H.; Zhu, L.M. Thermosensitive nanofibers loaded with ciprofloxacin as antibacterial wound dressing materials. Int. J. Pharm. 2017, 517, 135–147. [Google Scholar] [CrossRef]
- Li, H.; Liu, K.; Williams, G.R.; Wu, J.; Wu, J.; Wang, H.; Niu, S.; Zhu, L.-M. Dual temperature and pH responsive nanofiber formulations prepared by electrospinning. Colloids Surf. B Biointerfaces 2018, 171, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Nakielski, P.; Pawłowska, S.; Rinoldi, C.; Ziai, Y.; De Sio, L.; Urbanek, O.; Zembrzycki, K.; Pruchniewski, M.; Lanzi, M.; Salatelli, E.; et al. Multifunctional Platform Based on Electrospun Nanofibers and Plasmonic Hydrogel: A Smart Nanostructured Pillow for Near-Infrared Light-Driven Biomedical Applications. ACS Appl. Mater. Interfaces 2020, 12, 54328–54342. [Google Scholar] [CrossRef]
- Jung, C.W.; Lee, J.S.; Jalani, G.; Hwang, E.Y.; Lim, D.W. Thermally-Induced Actuations of Stimuli-Responsive, Bicompartmental Nanofibers for Decoupled Drug Release. Front. Chem. 2019, 7, 73. [Google Scholar] [CrossRef]
- Tamayol, A.; Hassani Najafabadi, A.; Mostafalu, P.; Yetisen, A.K.; Commotto, M.; Aldhahri, M.; Abdel-wahab, M.S.; Najafabadi, Z.I.; Latifi, S.; Akbari, M.; et al. Biodegradable elastic nanofibrous platforms with integrated flexible heaters for on-demand drug delivery. Sci. Rep. 2017, 7, 9220. [Google Scholar] [CrossRef] [PubMed]
- Sayin, S.; Tufani, A.; Emanet, M.; Genchi, G.G.; Sen, O.; Shemshad, S.; Ozdemir, E.; Ciofani, G.; Ozaydin Ince, G. Electrospun Nanofibers With pH-Responsive Coatings for Control of Release Kinetics. Front. Bioeng. Biotechnol. 2019, 7, 309. [Google Scholar] [CrossRef] [PubMed]
- Gong, T.; Liu, T.; Zhang, L.; Ye, W.; Guo, X.; Wang, L.; Quan, L.; Pan, C. Design Redox-Sensitive Drug-Loaded Nanofibers for Bone Reconstruction. ACS Biomater. Sci. Eng. 2018, 4, 240–247. [Google Scholar] [CrossRef]
- Agrahari, V.; Meng, J.; Ezoulin, M.J.; Youm, I.; Dim, D.C.; Molteni, A.; Hung, W.T.; Christenson, L.K.; Youan, B.C. Stimuli-sensitive thiolated hyaluronic acid based nanofibers: Synthesis, preclinical safety and in vitro anti-HIV activity. Nanomedicine 2016, 11, 2935–2958. [Google Scholar] [CrossRef]
- Singh, B.; Shukla, N.; Kim, J.; Kim, K.; Park, M.H. Stimuli-Responsive Nanofibers Containing Gold Nanorods for On-Demand Drug Delivery Platforms. Pharmaceutics 2021, 13, 1319. [Google Scholar] [CrossRef]
- Sutka, A. Core-Shell PVA/PVP-FeOOH Nanofibers as a Potential Visible Light Responsive Drug Delivery System. Key Eng. Mater. 2020, 850, 259–263. [Google Scholar] [CrossRef]
- Perera, A.S.; Zhang, S.; Homer-Vanniasinkam, S.; Coppens, M.-O.; Edirisinghe, M. Polymer–Magnetic Composite Fibers for Remote-Controlled Drug Release. ACS Appl. Mater. Interfaces 2018, 10, 15524–15531. [Google Scholar] [CrossRef]
- Jariwala, T.; Ico, G.; Tai, Y.; Park, H.; Myung, N.V.; Nam, J. Mechano-Responsive Piezoelectric Nanofiber as an On-Demand Drug Delivery Vehicle. ACS Appl. Bio Mater. 2021, 4, 3706–3715. [Google Scholar] [CrossRef] [PubMed]
- Sasikala, A.R.K.; Unnithan, A.R.; Yun, Y.-H.; Park, C.H.; Kim, C.S. An implantable smart magnetic nanofiber device for endoscopic hyperthermia treatment and tumor-triggered controlled drug release. Acta Biomater. 2016, 31, 122–133. [Google Scholar] [CrossRef]
- Phromviyo, N.; Lert-Itthiporn, A.; Swatsitang, E.; Chompoosor, A. Biodegradable poly(vinyl alcohol)/polyoxalate electrospun nanofibers for hydrogen peroxide-triggered drug release. J. Biomater. Science. Polym. Ed. 2015, 26, 975–987. [Google Scholar] [CrossRef]
- Mansukhani, N.A.; Peters, E.B.; So, M.M.; Albaghdadi, M.S.; Wang, Z.; Karver, M.R.; Clemons, T.D.; Laux, J.P.; Tsihlis, N.D.; Stupp, S.I.; et al. Peptide Amphiphile Supramolecular Nanostructures as a Targeted Therapy for Atherosclerosis. Macromol. Biosci. 2019, 19, 1900066. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Yao, Y.; Wang, Z.; Tian, Q.; Wang, J.; Gu, L.; Li, B.; Zheng, Q.; Wu, Y. Local Delivery of Taxol From FGL-Functionalized Self-Assembling Peptide Nanofiber Scaffold Promotes Recovery After Spinal Cord Injury. Front. Cell Dev. Biol. 2020, 8, 820. [Google Scholar] [CrossRef] [PubMed]
- Chi, H.Y.; Chan, V.; Li, C.; Hsieh, J.H.; Lin, P.H.; Tsai, Y.-H.; Chen, Y. Fabrication of polylactic acid/paclitaxel nano fibers by electrospinning for cancer therapeutics. BMC Chem. 2020, 14, 63. [Google Scholar] [CrossRef] [PubMed]
- Do Pham, D.D.; Jenčová, V.; Kaňuchová, M.; Bayram, J.; Grossová, I.; Šuca, H.; Urban, L.; Havlíčková, K.; Novotný, V.; Mikeš, P.; et al. Novel lipophosphonoxin-loaded polycaprolactone electrospun nanofiber dressing reduces Staphylococcus aureus induced wound infection in mice. Sci. Rep. 2021, 11, 17688. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Wang, J.; Wang, Y.; Li, L.; Guo, X.; Zhou, S. An Implantable Active-Targeting Micelle-in-Nanofiber Device for Efficient and Safe Cancer Therapy. ACS Nano 2015, 9, 1161–1174. [Google Scholar] [CrossRef]
- Nasari, M.; Semnani, D.; Hadjianfar, M.; Amanpour, S. Poly (ε-caprolactone)/poly (N-vinyl-2-pyrrolidone) core–shell nanofibers loaded by multi-walled carbon nanotubes and 5-fluorouracil: An anticancer drug delivery system. J. Mater. Sci. 2020, 55, 10185–10201. [Google Scholar] [CrossRef]
- Padmakumar, S.; Joseph, J.; Neppalli, M.H.; Mathew, S.E.; Nair, S.V.; Shankarappa, S.A.; Menon, D. Electrospun Polymeric Core-sheath Yarns as Drug Eluting Surgical Sutures. ACS Appl. Mater. Interfaces 2016, 8, 6925–6934. [Google Scholar] [CrossRef] [PubMed]
- Zuidema, J.M.; Dumont, C.M.; Wang, J.; Batchelor, W.M.; Lu, Y.-S.; Kang, J.; Bertucci, A.; Ziebarth, N.M.; Shea, L.D.; Sailor, M.J. Porous Silicon Nanoparticles Embedded in Poly(lactic-co-glycolic acid) Nanofiber Scaffolds Deliver Neurotrophic Payloads to Enhance Neuronal Growth. Adv. Funct. Mater. 2020, 30, 2002560. [Google Scholar] [CrossRef] [PubMed]
- Jalvandi, J.; White, M.; Gao, Y.; Truong, Y.B.; Padhye, R.; Kyratzis, I.L. Slow release of levofloxacin conjugated on silica nanoparticles from poly(ɛ-caprolactone) nanofibers. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 507–513. [Google Scholar] [CrossRef]
- Nunes, R.; Bogas, S.; Faria, M.J.; Gonçalves, H.; Lúcio, M.; Viseu, T.; Sarmento, B.; das Neves, J. Electrospun fibers for vaginal administration of tenofovir disoproxil fumarate and emtricitabine in the context of topical pre-exposure prophylaxis. J. Control. Release 2021, 334, 453–462. [Google Scholar] [CrossRef]
- Ranjbar-Mohammadi, M.; Sa’di, V.; Moezzi, M.; Saghafi, R. Fabrication and Characterization of Antibacterial Suture Yarns Containing PLA/Tetracycline Hydrochloride-PVA/Chitosan Nanofibers. Fibers Polym. 2022, 23, 1538–1547. [Google Scholar] [CrossRef]
- Padmakumar, S.; Paul-Prasanth, B.; Pavithran, K.; Vijaykumar, D.K.; Rajanbabu, A.; Sivanarayanan, T.B.; Kadakia, E.; Amiji, M.M.; Nair, S.V.; Menon, D. Long-term drug delivery using implantable electrospun woven polymeric nanotextiles. Nanomed. Nanotechnol. Biol. Med. 2019, 15, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Yu, Y.; Wang, S.; Xu, H.; He, Q.; Wen, J.; Xu, J.; Li, K.; Huang, Z.; Xu, P. Preparation and characterization of conductive nerve guide conduit filled with dual drug-loaded nanofibers. J. Bioact. Compat. Polym. 2021, 36, 531–547. [Google Scholar] [CrossRef]
- Lee, C.-H.; Hsieh, M.-J.; Roth, J.G.; Fu, X.; Lu, C.-H.; Hung, K.-C.; Kuo, C.-C.; Liu, S.-J. Hybrid Core–Shell Nanofibrous Scaffolds/Stents Deliver Angiotensin II Receptor Blocker to Treat Diabetic Artery Disease. ACS Appl. Polym. Mater. 2022, 4, 4199–4207. [Google Scholar] [CrossRef]
- Kersani, D.; Mougin, J.; Lopez, M.; Degoutin, S.; Tabary, N.; Cazaux, F.; Janus, L.; Maton, M.; Chai, F.; Sobocinski, J.; et al. Stent coating by electrospinning with chitosan/poly-cyclodextrin based nanofibers loaded with simvastatin for restenosis prevention. Eur. J. Pharm. Biopharm. 2020, 150, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.-H.; Shen, S.-J.; Hsu, Y.-H.; Chou, Y.-C.; Yu, P.-C.; Liu, S.-J. Tri-Layered Doxycycline-, Collagen- and Bupivacaine-Loaded Poly(lactic-co-glycolic acid) Nanofibrous Scaffolds for Tendon Rupture Repair. Polymers 2022, 14, 2659. [Google Scholar] [CrossRef]
- Mitropoulou, A.; Markatos, D.N.; Antimisiaris, S.G.; Mavrilas, D. A Novel Design of a PVA Electrospun Nanofibrous Scaffold Incorporating Liposomes as Drug Delivery Carriers for Tissue Engineering. Ann. Biomed. Technol. Eng. 2018, 1, 1003. [Google Scholar]
- Pires, F.; Santos, J.F.; Bitoque, D.; Silva, G.A.; Marletta, A.; Nunes, V.A.; Ribeiro, P.A.; Silva, J.C.; Raposo, M. Polycaprolactone/Gelatin Nanofiber Membranes Containing EGCG-Loaded Liposomes and Their Potential Use for Skin Regeneration. ACS Appl. Bio Mater. 2019, 2, 4790–4800. [Google Scholar] [CrossRef]
- Sousa, M.G.C.; Rezende, T.M.B.; Franco, O.L. Nanofibers as drug-delivery systems for antimicrobial peptides. Drug Discov. Today 2021, 26, 2064–2074. [Google Scholar] [CrossRef]
- Si, Y.; Wen, Y.; Kelly, S.H.; Chong, A.S.; Collier, J.H. Intranasal delivery of adjuvant-free peptide nanofibers elicits resident CD8(+) T cell responses. J. Control. Release Off. J. Control. Release Soc. 2018, 282, 120–130. [Google Scholar] [CrossRef]
- Kelly, S.H.; Cossette, B.J.; Varadhan, A.K.; Wu, Y.; Collier, J.H. Titrating Polyarginine into Nanofibers Enhances Cyclic-Dinucleotide Adjuvanticity in Vitro and after Sublingual Immunization. ACS Biomater. Sci. Eng. 2021, 7, 1876–1888. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Yin, C.; Tu, H.; Jiang, S.; Wang, Q.; Zhou, X.; Xing, X.; Xie, C.; Shi, X.; Du, Y.; et al. Controlled Co-delivery of Growth Factors through Layer-by-Layer Assembly of Core–Shell Nanofibers for Improving Bone Regeneration. ACS Nano 2019, 13, 6372–6382. [Google Scholar] [CrossRef]
- Zheng, Z.; Liu, Y.; Huang, W.; Mo, Y.; Lan, Y.; Guo, R.; Cheng, B. Neurotensin-loaded PLGA/CNC composite nanofiber membranes accelerate diabetic wound healing. Artif. Cells Nanomed. Biotechnol. 2018, 46, 493–501. [Google Scholar] [CrossRef] [Green Version]
- Chaiarwut, S.; Ekabutr, P.; Chuysinuan, P.; Chanamuangkon, T.; Supaphol, P. Surface immobilization of PCL electrospun nanofibers with pexiganan for wound dressing. J. Polym. Res. 2021, 28, 344. [Google Scholar] [CrossRef]
- Voronova, A.; Prieto, C.; Pardo-Figuerez, M.; Lagaron, J.M.; Sanyal, A.; Demir, B.; Hubert, T.; Plaisance, V.; Pawlowski, V.; Vignoud-Despond, S.; et al. Photothermal Activatable Mucoadhesive Fiber Mats for On-Demand Delivery of Insulin via Buccal and Corneal Mucosa. ACS Appl. Bio Mater. 2022, 5, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, F.; Karumidze, N.; Kusradze, I.; Goderdzishvili, M.; Teixeira, P.; Gouveia, I.C. Immobilization of bacteriophage in wound-dressing nanostructure. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2475–2484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fries, C.N.; Wu, Y.; Kelly, S.H.; Wolf, M.; Votaw, N.L.; Zauscher, S.; Collier, J.H. Controlled Lengthwise Assembly of Helical Peptide Nanofibers to Modulate CD8+ T-Cell Responses. Adv. Mater. 2020, 32, 2003310. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Kelly, S.H.; Sanchez-Perez, L.; Sampson, J.H.; Collier, J.H. Comparative study of α-helical and β-sheet self-assembled peptide nanofiber vaccine platforms: Influence of integrated T-cell epitopes. Biomater. Sci. 2020, 8, 3522–3535. [Google Scholar] [CrossRef]
- Fries, C.N.; Chen, J.-L.; Dennis, M.L.; Votaw, N.L.; Eudailey, J.; Watts, B.E.; Hainline, K.M.; Cain, D.W.; Barfield, R.; Chan, C.; et al. HIV envelope antigen valency on peptide nanofibers modulates antibody magnitude and binding breadth. Sci. Rep. 2021, 11, 14494. [Google Scholar] [CrossRef]
- Mazza, M.; Ahmad, H.; Hadjidemetriou, M.; Agliardi, G.; Pathmanaban, O.N.; King, A.T.; Bigger, B.W.; Vranic, S.; Kostarelos, K. Hampering brain tumor proliferation and migration using peptide nanofiber:siPLK1/MMP2 complexes. Nanomedicine 2019, 14, 3127–3142. [Google Scholar] [CrossRef]
- Neuberg, P.; Hamaidi, I.; Danilin, S.; Ripoll, M.; Lindner, V.; Nothisen, M.; Wagner, A.; Kichler, A.; Massfelder, T.; Remy, J.S. Polydiacetylenic nanofibers as new siRNA vehicles for in vitro and in vivo delivery. Nanoscale 2018, 10, 1587–1590. [Google Scholar] [CrossRef]
- Zheng, Y.; Wu, Y.; Zhou, Y.; Wu, J.; Wang, X.; Qu, Y.; Wang, Y.; Zhang, Y.; Yu, Q. Photothermally Activated Electrospun Nanofiber Mats for High-Efficiency Surface-Mediated Gene Transfection. ACS Appl. Mater. Interfaces 2020, 12, 7905–7914. [Google Scholar] [CrossRef]
- Mulholland, E.J.; McErlean, E.M.; Dunne, N.; McCarthy, H.O. Design of a novel electrospun PVA platform for gene therapy applications using the CHAT peptide. Int. J. Pharm. 2021, 598, 120366. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, T.; Jin, S.; Xue, X.; Yang, X.; Gong, N.; Zhang, J.; Wang, P.C.; Tian, J.-H.; Xing, J.; et al. Virus-Inspired Self-Assembled Nanofibers with Aggregation-Induced Emission for Highly Efficient and Visible Gene Delivery. ACS Appl. Mater. Interfaces 2017, 9, 4425–4432. [Google Scholar] [CrossRef] [Green Version]
- Tahmasebi, A.; Enderami, S.E.; Saburi, E.; Islami, M.; Yaslianifard, S.; Mahabadi, J.A.; Ardeshirylajimi, A.; Soleimanifar, F.; Moghadam, A.S. Micro-RNA-incorporated electrospun nanofibers improve osteogenic differentiation of human-induced pluripotent stem cells. J. Biomed. Mater. Res. Part A 2020, 108, 377–386. [Google Scholar] [CrossRef]
- Ho, T.-C.; Kim, H.S.; Chen, Y.; Li, Y.; LaMere, M.W.; Chen, C.; Wang, H.; Gong, J.; Palumbo, C.D.; Ashton, J.M.; et al. Scaffold-mediated CRISPR-Cas9 delivery system for acute myeloid leukemia therapy. Sci. Adv. 2021, 7, eabg3217. [Google Scholar] [CrossRef]
- Kharaghani, D.; Kurniwan, E.B.; Khan, M.Q.; Yoshiko, Y. MiRNA-Nanofiber, the Next Generation of Bioactive Scaffolds for Bone Regeneration: A Review. Micromachines 2021, 12, 1472. [Google Scholar] [CrossRef]
- Achille, C.; Sundaresh, S.; Chu, B.; Hadjiargyrou, M. Cdk2 silencing via a DNA/PCL electrospun scaffold suppresses proliferation and increases death of breast cancer cells. PLoS ONE 2012, 7, e52356. [Google Scholar] [CrossRef] [Green Version]
- Zhou, F.; Jia, X.; Yang, Y.; Yang, Q.; Gao, C.; Hu, S.; Zhao, Y.; Fan, Y.; Yuan, X. Nanofiber-mediated microRNA-126 delivery to vascular endothelial cells for blood vessel regeneration. Acta Biomater. 2016, 43, 303–313. [Google Scholar] [CrossRef]
- Li, W.; Wu, D.; Zhu, S.; Liu, Z.; Luo, B.; Lu, L.; Zhou, C. Sustained release of plasmid DNA from PLLA/POSS nanofibers for angiogenic therapy. Chem. Eng. J. 2019, 365, 270–281. [Google Scholar] [CrossRef]
- Xiong, R.; Hua, D.; Van Hoeck, J.; Berdecka, D.; Léger, L.; De Munter, S.; Fraire, J.C.; Raes, L.; Harizaj, A.; Sauvage, F.; et al. Photothermal nanofibres enable safe engineering of therapeutic cells. Nat. Nanotechnol. 2021, 16, 1281–1291. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y.; Wu, M.; Zou, R.; Mao, S.; Cong, P.; Hou, M.; Jin, H.; Zhao, Y.; Bao, Y. Adipose-derived mesenchymal stem cell-loaded β-chitin nanofiber hydrogel promote wound healing in rats. J. Mater. Sci. Mater. Med. 2022, 33, 12. [Google Scholar] [CrossRef] [PubMed]
- Shou, K.; Huang, Y.; Qi, B.; Hu, X.; Ma, Z.; Lu, A.; Jian, C.; Zhang, L.; Yu, A. Induction of mesenchymal stem cell differentiation in the absence of soluble inducer for cutaneous wound regeneration by a chitin nanofiber-based hydrogel. J. Tissue Eng. Regen. Med. 2018, 12, e867–e880. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Qian, Y.; Li, L.; Yao, G.; Yang, L.; Sun, Y. Polycaprolactone nanofiber scaffold enhances the osteogenic differentiation potency of various human tissue-derived mesenchymal stem cells. Stem Cell Res. Ther. 2017, 8, 148. [Google Scholar] [CrossRef] [PubMed]
- Boda, S.K.; Chen, S.; Chu, K.; Kim, H.J.; Xie, J. Electrospraying Electrospun Nanofiber Segments into Injectable Microspheres for Potential Cell Delivery. ACS Appl. Mater. Interfaces 2018, 10, 25069–25079. [Google Scholar] [CrossRef] [PubMed]
- Alakhov, V.; Moskaleva, E.; Batrakova, E.V.; Kabanov, A.V. Hypersensitization of multidrug resistant human ovarian carcinoma cells by pluronic P85 block copolymer. Bioconjugate Chem. 1996, 7, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yun, S.; Park, K.I.; Jang, J.-H. Sliding Fibers: Slidable, Injectable, and Gel-like Electrospun Nanofibers as Versatile Cell Carriers. ACS Nano 2016, 10, 3282–3294. [Google Scholar] [CrossRef]
- Schüttler, K.F.; Bauhofer, M.W.; Ketter, V.; Giese, K.; Eschbach, D.A.; Yenigün, M.; Fuchs-Winkelmann, S.; Paletta, J.R.J. Direct incorporation of mesenchymal stem cells into a Nanofiber scaffold—In vitro and in vivo analysis. Sci. Rep. 2020, 10, 9557. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Shin, H.J.; Lee, J.; Shin, Y.M.; Perikamana, S.K.M.; Park, S.Y.; Jung, H.S.; Shin, H. Fabrication of in vitro 3D mineralized tissue by fusion of composite spheroids incorporating biomineral-coated nanofibers and human adipose-derived stem cells. Acta Biomater. 2018, 74, 464–477. [Google Scholar] [CrossRef]
- Vocetkova, K.; Sovkova, V.; Buzgo, M.; Lukasova, V.; Divin, R.; Rampichova, M.; Blazek, P.; Zikmund, T.; Kaiser, J.; Karpisek, Z.; et al. A Simple Drug Delivery System for Platelet-Derived Bioactive Molecules, to Improve Melanocyte Stimulation in Vitiligo Treatment. Nanomaterials 2020, 10, 1801. [Google Scholar] [CrossRef]
- Millán-Rivero, J.E.; Martínez, C.M.; Romecín, P.A.; Aznar-Cervantes, S.D.; Carpes-Ruiz, M.; Cenis, J.L.; Moraleda, J.M.; Atucha, N.M.; García-Bernal, D. Silk fibroin scaffolds seeded with Wharton’s jelly mesenchymal stem cells enhance re-epithelialization and reduce formation of scar tissue after cutaneous wound healing. Stem Cell Res. Ther. 2019, 10, 126. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Sun, Y.; Dong, X.; Yin, Q.; Zhu, H.; Li, S.; Zhou, J.; Wang, C. Cell-derived extracellular matrix-coated silk fibroin scaffold for cardiogenesis of brown adipose stem cells through modulation of TGF-β pathway. Regen. Biomater. 2020, 7, 403–412. [Google Scholar] [CrossRef] [Green Version]
- Nour-Eldeen, G.; Abdel-Rasheed, M.; El-Rafei, A.M.; Azmy, O.; El-Bassyouni, G.T. Adipose tissue-derived mesenchymal stem cells and chitosan/poly (vinyl alcohol) nanofibrous scaffolds for cartilage tissue engineering. Cell Regen. 2020, 9, 7. [Google Scholar] [CrossRef]
- Fathi, A.; Khanmohammadi, M.; Goodarzi, A.; Foroutani, L.; Mobarakeh, Z.T.; Saremi, J.; Arabpour, Z.; Ai, J. Fabrication of chitosan-polyvinyl alcohol and silk electrospun fiber seeded with differentiated keratinocyte for skin tissue regeneration in animal wound model. J. Biol. Eng. 2020, 14, 27. [Google Scholar] [CrossRef]
- Lupone, F.; Padovano, E.; Veca, A.; Franceschetti, L.; Badini, C. Innovative processing route combining fused deposition modelling and laser writing for the manufacturing of multifunctional polyamide/carbon fiber composites. Mater. Des. 2020, 193, 108869. [Google Scholar] [CrossRef]
- Chang, M.Y.; Chang, C.H.; Chen, C.H.; Cheng, B.; Lin, Y.D.; Luo, C.Y.; Wu, H.L.; Yang, Y.J.; Chen, J.H.; Hsieh, P.C. The time window for therapy with peptide nanofibers combined with autologous bone marrow cells in pigs after acute myocardial infarction. PLoS ONE 2015, 10, e0115430. [Google Scholar] [CrossRef]
- Amagat Molas, J.; Chen, M. Injectable PLCL/gelatin core-shell nanofibers support noninvasive 3D delivery of stem cells. Int. J. Pharm. 2019, 568, 118566. [Google Scholar] [CrossRef]
- Hauser, M.; Li, G.; Nowack, B. Environmental hazard assessment for polymeric and inorganic nanobiomaterials used in drug delivery. J. Nanobiotechnol. 2019, 17, 56. [Google Scholar] [CrossRef]
- Guimarães, A.T.B.; Malafaia, G. Multiple toxicity endpoints induced by carbon nanofibers in Amazon turtle juveniles: Outspreading warns about toxicological risks to reptiles. Sci. Total Environ. 2021, 779, 146514. [Google Scholar] [CrossRef]
- Chen, X.; Feng, B.; Zhu, D.-Q.; Chen, Y.-W.; Ji, W.; Ji, T.-J.; Li, F. Characteristics and toxicity assessment of electrospun gelatin/PCL nanofibrous scaffold loaded with graphene in vitro and in vivo. Int. J. Nanomed. 2019, 14, 3669–3678. [Google Scholar] [CrossRef] [Green Version]
- Soo Min, K.; Eun Ji, G.; Seung Hwan, J.; Sang Mock, L.; Woo Jong, S.; Jin Sik, K.J.J.o.T.; Assessment, R. Toxicity Evaluation of Cellulose Nanofibers (Cnfs) for Cosmetic Industry Application. J. Toxicol. Risk Assess 2019, 5, 29. [Google Scholar] [CrossRef]
- Yang, C.; Chu, L.; Zhang, Y.; Shi, Y.; Liu, J.; Liu, Q.; Fan, S.; Yang, Z.; Ding, D.; Kong, D.; et al. Dynamic biostability, biodistribution, and toxicity of L/D-peptide-based supramolecular nanofibers. ACS Appl. Mater. Interfaces 2015, 7, 2735–2744. [Google Scholar] [CrossRef] [PubMed]
- Balusamy, B.; Senthamizhan, A.; Uyar, T. 6—In vivo safety evaluations of electrospun nanofibers for biomedical applications. In Electrospun Materials for Tissue Engineering and Biomedical Applications; Uyar, T., Kny, E., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 101–113. [Google Scholar] [CrossRef]
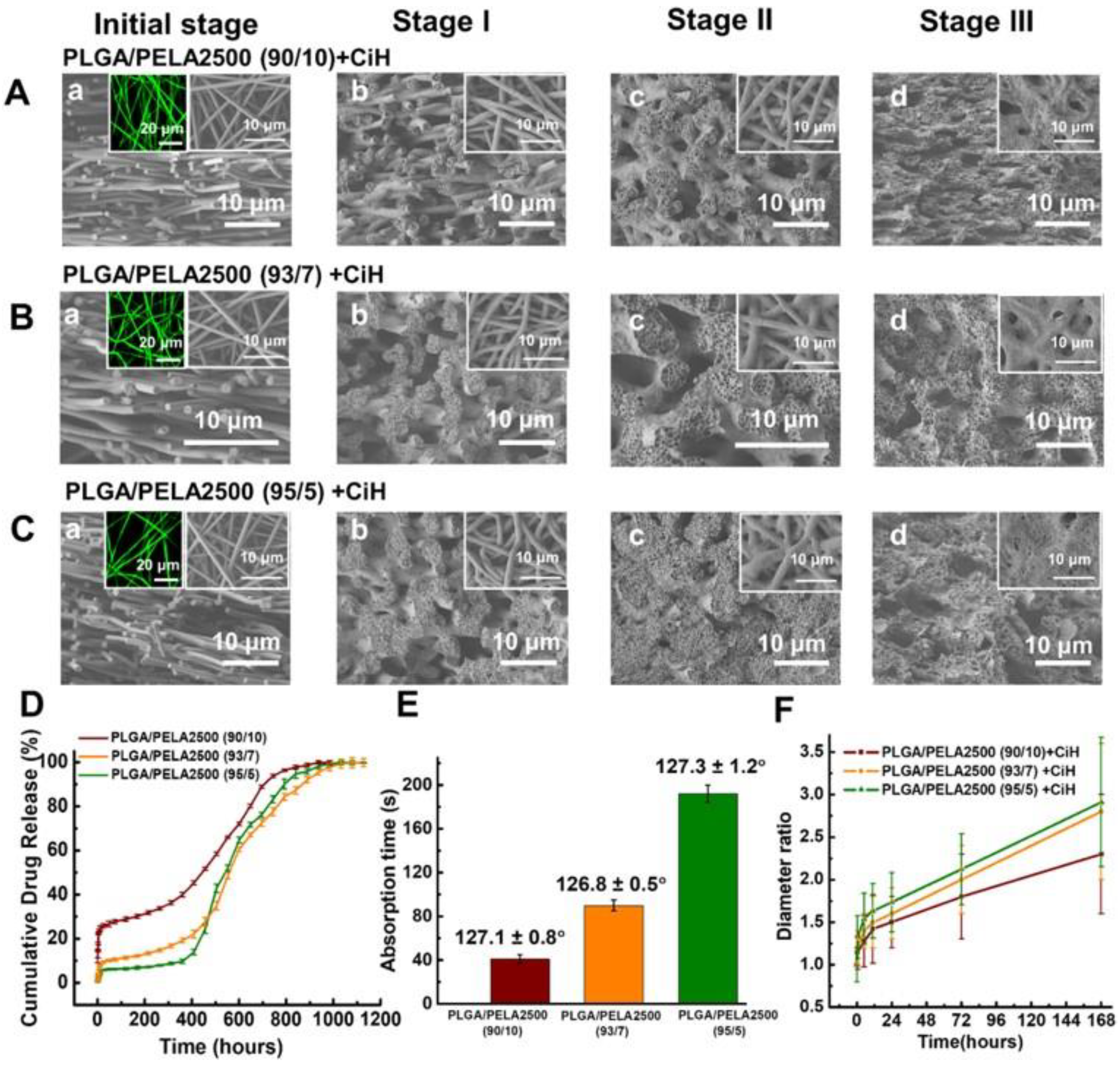
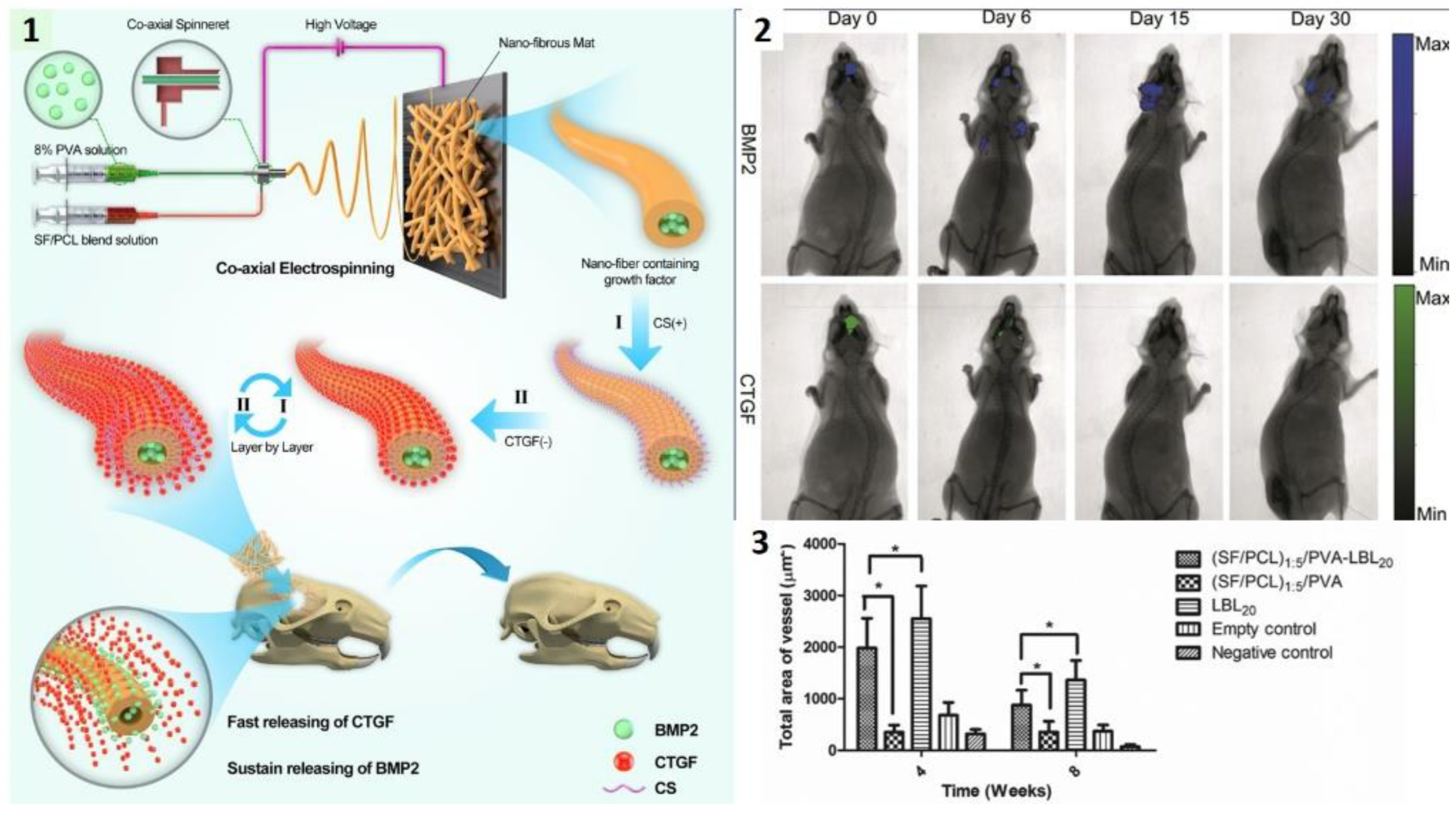
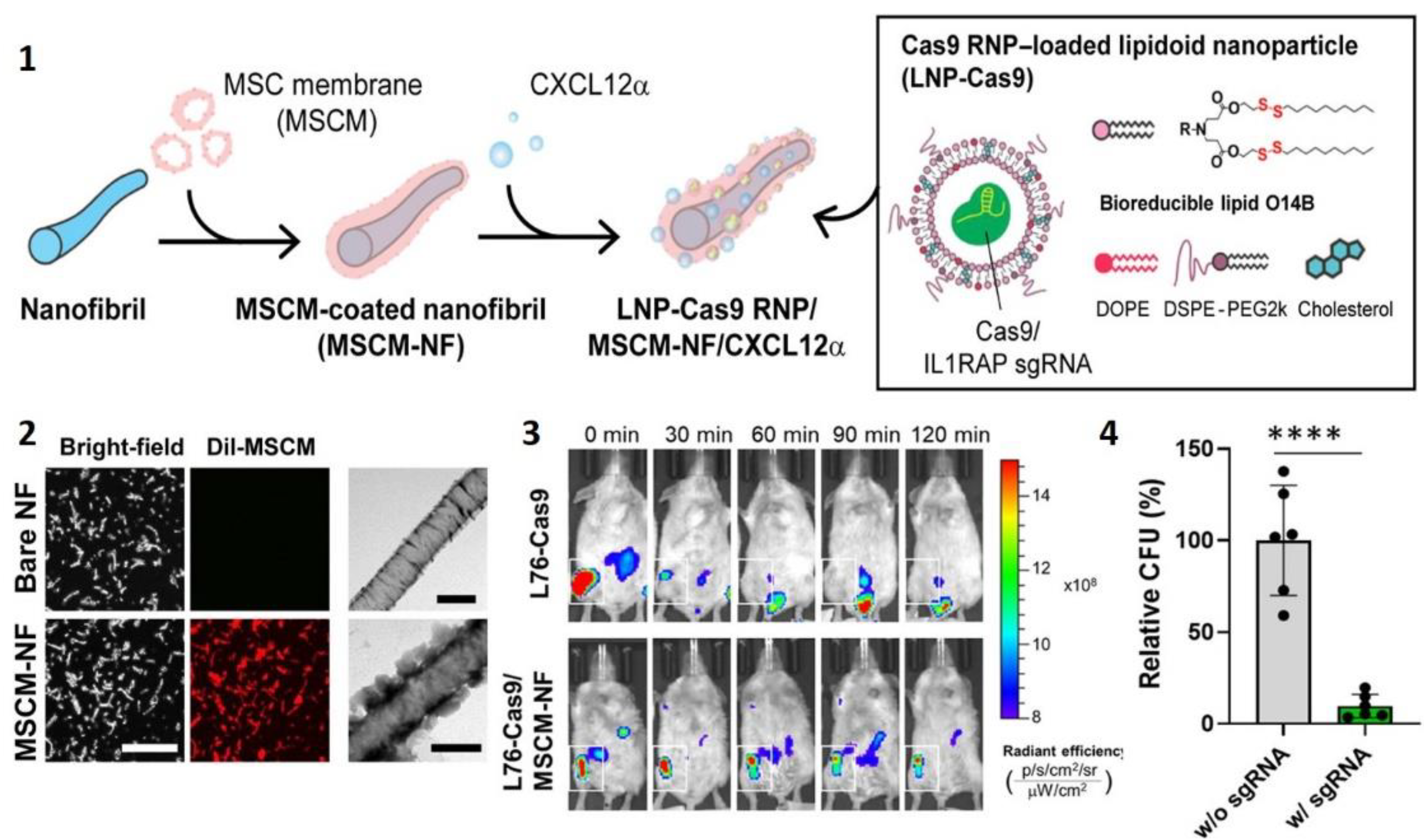
| Polymer | Solvent | Therapeutic Agent | Fabrication | Fiber Diameter | Architecture | Therapeutic Outcome | Ref |
|---|---|---|---|---|---|---|---|
| Thermo-responsive Nanofibers | |||||||
| Eudragit® RS 100 PMMA | DMF | Octenidine | Single nozzle electrospinning | 134–168 nm | Single mesh | Reduced in vitro bacterial colony formation | [104] |
| PCL/PNIPAAM | TFE | Doxycycline hyclate | Single nozzle electrospinning followed by mesh immersion and UV crosslinking | 275–490 nm | Core-crosslinked shell mesh | Reduced in vitro bacterial colony formation | [106] |
| P(NIPAAm-co-NIPMAAm)/PLCL | H2O CH2Cl2/DMF | Light-assisted co-axial electrospinning | 1400 nm | Crosslinked core-shell | Enhanced drug penetration in soft tissues by NIR-light assistance | [111] | |
| poly(NIPAAM-co-AAh)/PEGDMA | Ethanol TFE | Bovine serum albumin Dexamethasone phosphate | EHD side-by-side co-jetting followed by UV crosslinking | ~1000 nm | Bicompartmental nanofibers | - | [112] |
| PGS/PCL | CH2Cl2/ Ethanol | NP-encapsulated cefazolin and ceftriaxone | Single nozzle electrospinning followed by printing of electrically conductive pattern | 350–1100 nm | Elastic sheets | Reduced in vitro bacterial colony formation | [113] |
| pH-responsive Nanofibers | |||||||
| PVA/p(4VP-co-EGDMA) | H2O | Rose Bengal | Single nozzle electrospinning followed by chemical vapor deposition | 580 nm | Core-coated mats | In vitro antiproliferative activity against cancer cells (U87MG) | [114] |
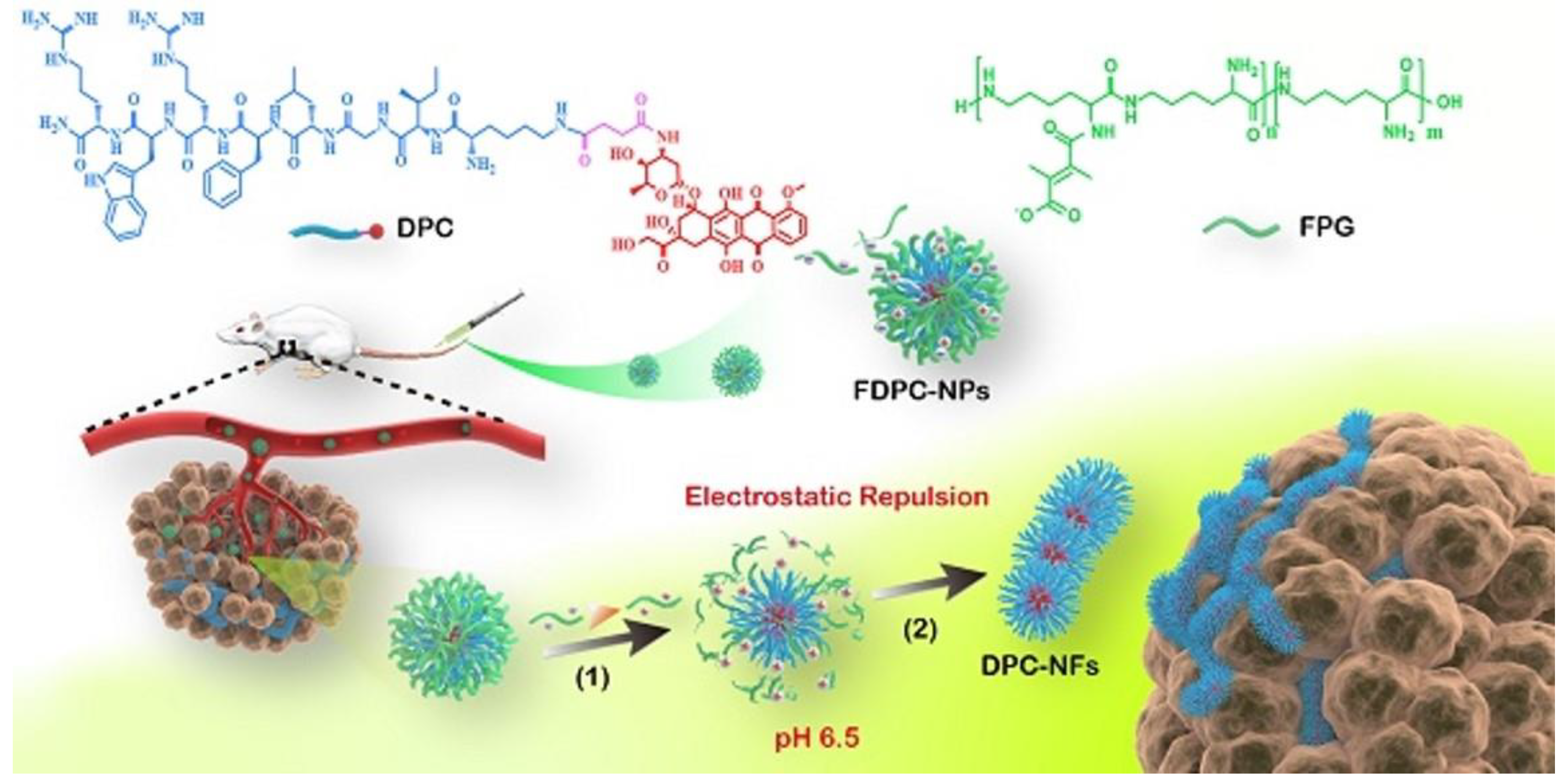
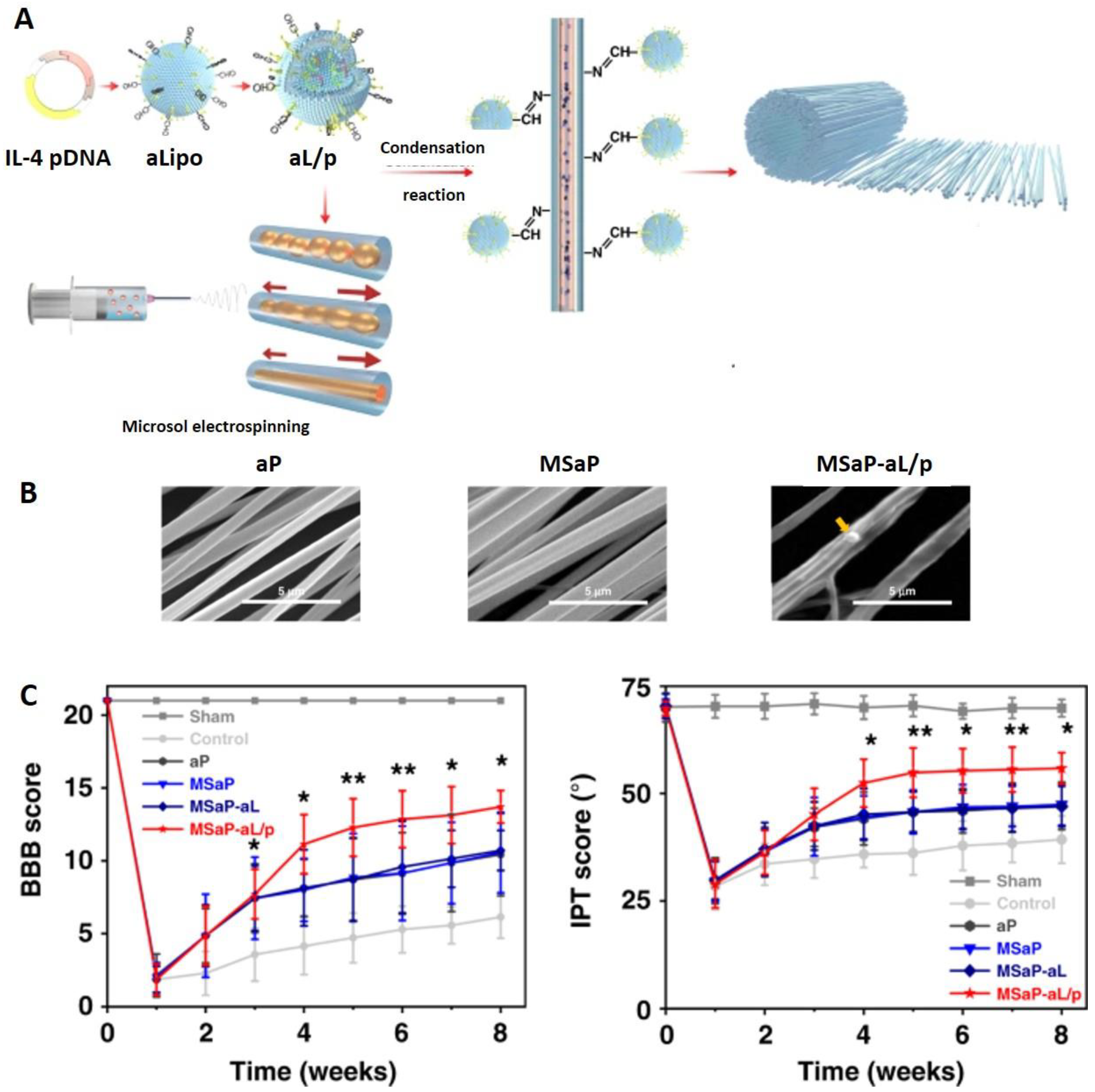
| aPLA/HA | CH2Cl2/DMF H2O | Interleukin-4 pDNA-loaded liposome Nerve growth factor | Microsol electrospinning followed by chemical grafting of liposomes | 550–570 nm | Oriented core-shell fiber scaffolds | Reduced in vivo inflammatory response, increased nerve repair and recovery of motor function | [9] |
| Redox-responsive Nanofibers | |||||||
| PEO/PCL and redox responsive c-6A PEG-PCL/6A PEG-PCLSH NG | CH2Cl2 H2O | BMP-2 | Co-axial electrospinning | 220–340 nm | Core-shell | In vitro stem cell osteogenesis differentiation. In vivo mandible bone reconstruction | [115] |
| Enzyme-responsive Nanofibers | |||||||
| coPEA 8-[L-Phe-6] 0.95–[L-Leu-6]0.05/ PCL | H2O CH2Cl2/C6H12 | a-chymotrypsin Non-specific lipase Fluorophore | Colloid electrospinning | 375–1380 nm | Single scaffolds | - | [93] |
| HA-SH PEO | H2O | Tenofovir | Co-axial electrospinning | 75 nm | Core-shell scaffolds | In vitro anti-HIV effect | [116] |
| P18-PLGVRGRGD | H2O | P18 | Gelatinase-triggered in situ formation | 30 nm | Fibrous superstructures | Improved photoacoustic tumor imaging and therapeutic efficacy | [30] |
| Light-responsive Nanofibers | |||||||
| PNIPAM/ Au nanorods | DMF/THF | Camptothecin | Single nozzle electrospinning followed by high temperature cross-linking | 600–700 nm | Single scaffolds | In vitro decreased viability of malignant glioma cells (U-87 MG) | [117] |
| PVA/PVP-FeOOH | H2O | Methylene blue | Co-axial electrospinning | 580 nm | Core-shell mat | - | [118] |
| PLLA/P(NIPAAm-co-PNIPMAAm) AuNR hydrogel | CHCl3/DMF | Rhodamine B | Single nozzle electrospinning followed by pillow assembly and interface UV-crosslinking | Nanostructured pillow composed of NF membrane and hydrogel core | [111] | ||
| Magneto-responsive Nanofibers | |||||||
| PVA/IONP | H2O | Acetaminophen | Infusion gyration followed by drug adsorption | 100−300 nm | Single mats of beaded fibers | - | [119] |
| Mechano-responsive Nanofibers | |||||||
| P(VDF-TrFE) | DMF/THF | Crystal violet poly(L-lysine)-Vivotag-645 | Single nozzle electrospinning | 70–500 nm | Nanofibrous membranes | - | [120] |
| Multi-responsive Nanofibers | |||||||
| PNVCL/EC Eudragit L100 | Ethanol | Ketoprofen | Twin-jet electrospinning | 350–670 nm | Thermo- and pH-sensitive hybrid mats | - | [110] |
| PLGA/ IONP | HFIP | Bortezomib | Single nozzle electrospinning followed by functionalization | 600 nm | Not-like nanofibers | In vitro apoptosis in breast cancer cells (4T1) | [121] |
| Polymer | Therapeutic Agent | Functionalization | Fabrication | Architecture | Administration Route | Therapeutic Outcome | Ref |
|---|---|---|---|---|---|---|---|
| Amphiphilic peptides | Liver X receptor agonist GW3965 | ApoA1-targeting peptide | Self-assembly | Individual NFs with secondary β-sheet structure | Intravenous | Reduced plaque burden | [123] |
| Amphiphilic peptides | Taxol | FGL | Self-assembly | NFs hydrogel | Intraspinal microinjection post-SCI | In vitro and in vivo neurite elongation. Decreased inflammatory response and neurobehavioral recovery in vivo | [124] |
| PLA | Paclitaxel | - | Single nozzle electrospinning | NF membrane | - | Inhibition of cancer cells growth (HCT-116) | [125] |
| PCL | Lipophosphonoxin | - | Single nozzle electrospinning | NF scaffold | NF wound dressing | Reduced Staphylococcus aureus count in infected wound. No systemic absorption | [126] |
| PVA/ crosslinked gelatin | Dox-loaded PCL-PEG micelle | Folate-decorated micelles | Coaxial electrospinning | Core-shell NFs | Localized implant | In vitro and in vivo antitumor effect against mammary tumor | [127] |
| PCL/PVP | Multi-walled carbon nanotubes 5-fluorouracil | - | Coaxial electrospinning | Core-shell NF | - | In vitro cytotoxicity against HeLa cervical tumor cells | [128] |
| PLA/PLGA | Aceclofenac or insulin | - | Core electrospinning followed by sheet electrospinning | Twisted core yarn coated with drug-loaded sheet | Surgical sutures | Sustained drug release attenuated skin inflammation in in vivo animal model | [129] |
| PCL | pSiNP/drug, enzyme or RNA aptamer pSiNP/levofloxacin | - - | Airbrush spray nebulization. Single nozzle electrospinning | Aligned NFs NF mat | - - | Directional growth of neuronal cells | [94,130,131] |
| PVA | Liposomes loaded with tenofovir disoproxil fumarate and emtricitabine | - | Single nozzle electrospinning | NF mat | - | [132] |
| Polymer | Therapeutic Agent | Fabrication | Architecture | Administration Route | Therapeutic Outcome | Ref |
|---|---|---|---|---|---|---|
| Q11 self-assembly domain | Influenza acid polymerase epitope | Self-assembly | Individual NFs | Intranasal Subcutaneous | Reduced viral load in the lungs 6 weeks after 2nd vaccination | [142] |
| PEG-Q11R9 | CDN mucosal adjuvants | Self-assembly | Individual NFs | Sublingual | Activation of dendritic cells in the draining lymph nodes | [143] |
| SF/PCL/PVA | BMP2 CTGF | Coaxial electrospinning followed by layer-by-layer deposition | Multilayer core-shell NF | Implant | In vitro and in vivo temporal control of GF release Promoted bona and vessel formation | [144] |
| PLGL/CNC | Neurotensin | Single nozzle electrospinning | Composite NF membrane | Wound patch | Accelerated skin regeneration | [145] |
| PCL | Pexiganan | Single nozzle electrospinning followed by hydrolysis | NF mats | - | In vitro antimicrobial activity on gram-positive and negative bacteria | [146] |
| PAA/rGO β-cyclodextrin | Insulin | Single nozzle electrospinning | Hydrogel-like fiber mats | Buccal Eye cornea | Ex vivo delivery via eye cornea and the buccal mouth lining | [147] |
| PCL | Bacteriophage capsid | Single nozzle electrospinning followed by bacteriophage conjugation | Elastic NF mat | - | In vitro bactericidal activity (P. aeruginosa) | [148] |
| Polymer | Therapeutic Agent | Functionalization | Fabrication | Architecture | Administration Route | Therapeutic Outcome | Ref |
|---|---|---|---|---|---|---|---|
| palmitoyl-GGGAAAKRK peptide amphiphile | siRNA | - | Self-assembly | Individual NFs | Intra-tumoral administration | Extended survival of glioblastoma bearing mice | [152] |
| PDA | siRNA | - | Photopolymerization | Individual NFs | Intraperitoneal | In vitro and in vivo transfection of cancer cells and oncogene silencing | [153] |
| pDNA-PEI PLA-gelatin | pDNA polyplex | Au nanorods | Coaxial electrospinning | Nanofiber mats | - | Improved in vitro transfection | [154] |
| PVA | pDNA-CHAT polyplex | Cell penetrating CHAT peptide | Single nozzle electrospinning followed by DNAsoak-loading | Crosslinked nanofibers | - | Improved in vitro transfection | [155] |
| Virus-inspired polypeptide gene vector capped with lipophilic tail | pDNA | tetraphenylethene | Self-assembly | Individual NFs | - | In vitro transfection of wide cell range, including stem cells | [156] |
| PCL/gelatin | miRNA polyplex | - | Dual power electrospinning | Bilayer electrospun membranes | - | Improved in vitro osteogenic differentiation of human-induced pluripotent stem cells | [157] |
| PCL | LNPs loaded with CRISPR-Cas9 RNP complex | Biomimetic MSCM coating CXCL12 chimokine | Single nozzle electrospinning | Fragmented nanofibrils with biomimetic coating | Intra bone-marrow injection | Reduced human leukemic burden in mice | [158] |
| Combination therapy | |||||||
| HA/aPLA | pDNA-loaded aLiposome nerve GF | - | Microsol Electrospinning followed by chemical grafting of pDNA-aLiposome | Oriented core-shell NF | Implantation | Reduced in vivo inflammatory response, increased nerve repair and recovery of motor function | [9] |
| Polymer | Therapeutic Cell | Fabrication | Architecture | Therapeutic Outcome | Ref |
|---|---|---|---|---|---|
| β-chitin | ADSC | Chemically from natural sources | NF hydrogel | Improved in vivo wound healing | [164] |
| α-chitin | BMSC | Chemically from natural sources | NF hydrogel | Improved in vivo wound healing | [165] |
| PCL | BMSC, ADSC, UCSC | Fused deposition modeling | NF directed scaffolds | Enhanced in vitro proliferation and osteogenic potency | [166] |
| PCA/gelatin PLGA/gelatin | MSC, ESC | Electrospinning followed by electrospraying | Injectable NF microspheres | In vitro neural differentiation of ESC | [167] |
| Amphiphilic peptides | MuSC, VEGF, bFGF | Self-assembly followed by annealing and gelation | Injectable liquid crystalline NF gel | Enhanced engraftment of transplanted MuSCs | [168] |
| PCL-PS/MeHA | hNSC | Axial electrospinning followed by gel coating and crosslinking | SLIDING NFs | Prolonged survival of cells | [169] |
| PLA/collagen | MSC | Simultaneous co-axial cell electrospraying and polymer electrospinning | Cell-loaded NFs | Enhanced bone regeneration | [170] |
| Chitosan/PVA/SF | BMSC-derived keratinocytes | Single nozzle electrospinning followed by crosslinking | NF mat | Improved wound healing and skin regeneration | |
| PLA | ADSC | Single nozzle electrospinning followed by fragmentation and mineralizarion | Composite spheroids based on mineralized NF | Sustained ADSC viability and enhanced expression of osteogenic genes | [171] |
| PCL | Melanocyte | Single nozzle electrospinning followed by platelet soaking | Functionalized NF mats | Sustained melanocyte growth under the influence of GFs secreted by platelets Stimulated melanin synthesis for vitiligo treatment | [172] |
| SF | Wharton’s jelly MSC | Single nozzle electrospinning | Crosslinked NF 3D mat | Improved in vivo wound healing | [173] |
| SF | BASC | Salt sinking followed by CF growth and decullarization | ECM-coated NF mat | In vitro differentiation of BASC into CF | [174] |
| Collagen/PVA | ADSC | Single nozzle electrospinning | NF 3D mat | In vitro differentiation of ADSC into chondrogenic cells | [175] |
| NCT Number | Phase | Status | Condition(s) | Intervention(s) |
|---|---|---|---|---|
| NCT 04325789 | Not Applicable | Recruiting | Rotator Cuff Tears | Nanofiber scaffold |
| NCT 03690960 | Not Applicable | Unknown | Necrosis, Pulp | Electrospun TAP nanofibers |
| NCT 03242291 | Not Applicable | Unknown | Marginal Integrity of Hydroxyapatite Nanofiber Reinforced Flowable Composite | Conventional resin-based flowable composite. Hydroxyapatite Nanofiber reinforced flowable composite |
| NCT 03264105 | Not Applicable | Unknown | Retention Rate of Flowable Composite in Demineralized Pits and Fissure | Conventional resin-based flowable composite. Hydroxyapatite Nanofiber reinforced flowable composite |
| NCT 04867265 | Not Applicable | Completed | Ventilation During Resuscitation | Mouth-to-mouth ventilation |
| NCT 04870736 | Withdrawn | Basic Life Support Ventilation | Mouth-to-mouth ventilation | |
| NCT 02237287 | Early Phase 1 | Terminated | Wounds|Pressure Ulcer | Wound dressing with VAC and sNAG without anti-aggregation |
| NCT 02680106 | Not Applicable | Completed | Wound of Skin | Experimental: SPINNER. Active Comparator: JELONET/IBU Biatain |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jarak, I.; Silva, I.; Domingues, C.; Santos, A.I.; Veiga, F.; Figueiras, A. Nanofiber Carriers of Therapeutic Load: Current Trends. Int. J. Mol. Sci. 2022, 23, 8581. https://doi.org/10.3390/ijms23158581
Jarak I, Silva I, Domingues C, Santos AI, Veiga F, Figueiras A. Nanofiber Carriers of Therapeutic Load: Current Trends. International Journal of Molecular Sciences. 2022; 23(15):8581. https://doi.org/10.3390/ijms23158581
Chicago/Turabian StyleJarak, Ivana, Inês Silva, Cátia Domingues, Ana Isabel Santos, Francisco Veiga, and Ana Figueiras. 2022. "Nanofiber Carriers of Therapeutic Load: Current Trends" International Journal of Molecular Sciences 23, no. 15: 8581. https://doi.org/10.3390/ijms23158581
APA StyleJarak, I., Silva, I., Domingues, C., Santos, A. I., Veiga, F., & Figueiras, A. (2022). Nanofiber Carriers of Therapeutic Load: Current Trends. International Journal of Molecular Sciences, 23(15), 8581. https://doi.org/10.3390/ijms23158581











