Intrauterine and Postnatal Exposure to High Levels of Fluoride Is Associated with Motor Impairments, Oxidative Stress, and Morphological Damage in the Cerebellum of Offspring Rats
Abstract
:1. Introduction
2. Results
2.1. The Influence of F on the Body Mass of Progenitor Rats Exposed to F during Gestational and Lactation Periods
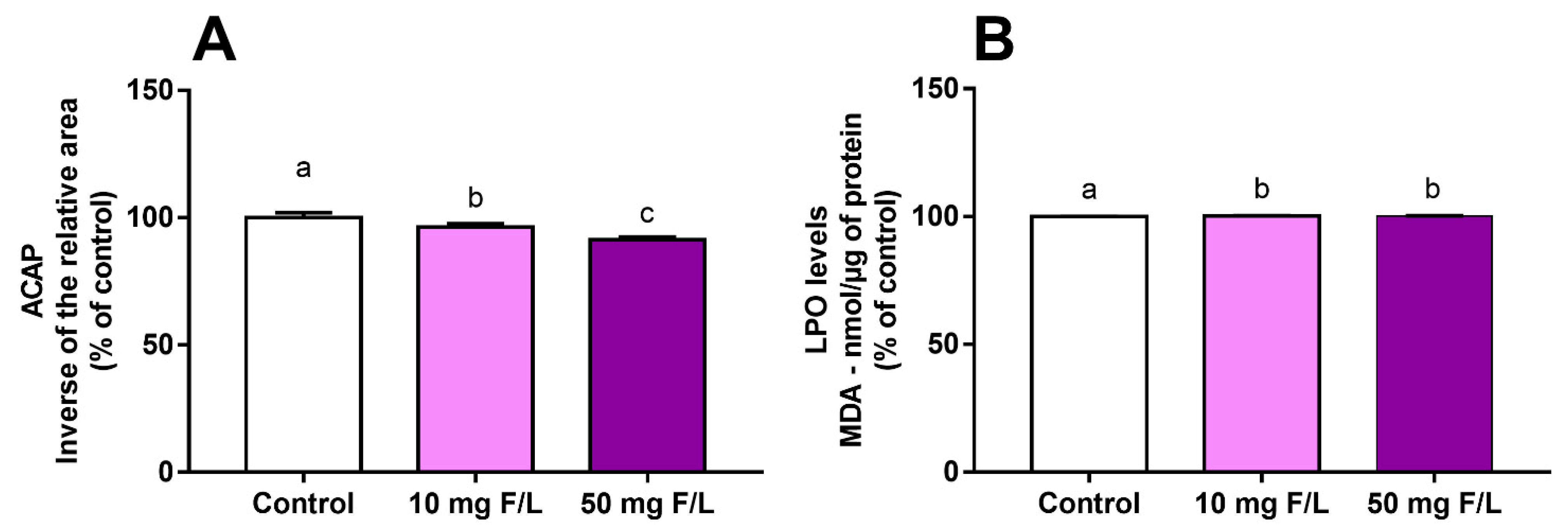
2.2. Fluoride Exposure Triggered a Disbalance in the Oxidative State of the Cerebellum of Rats
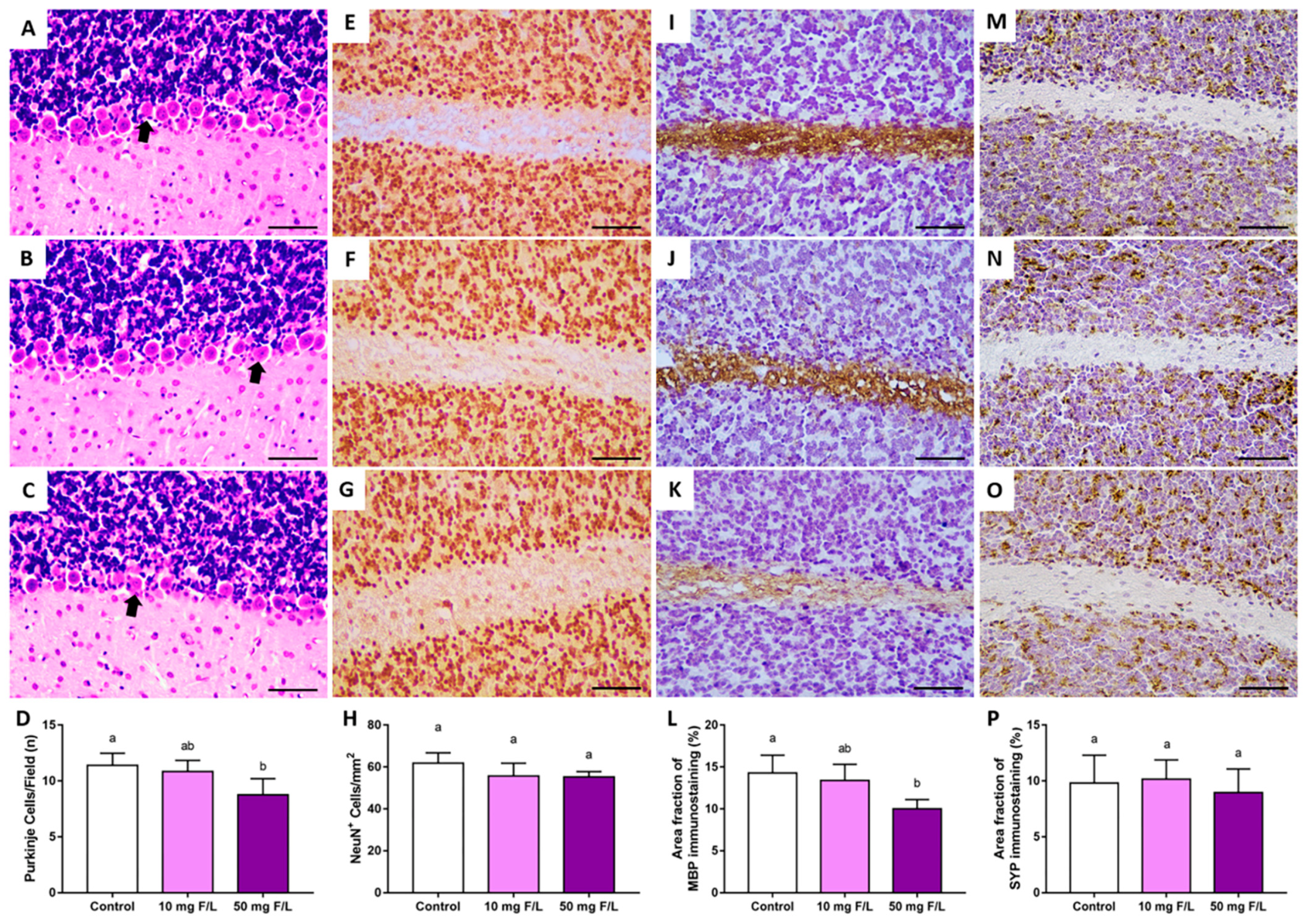
2.3. Prenatal and Postnatal Exposure to F at 50 Mg/L Was Associated with Cerebellar Morphology Damages in Rats
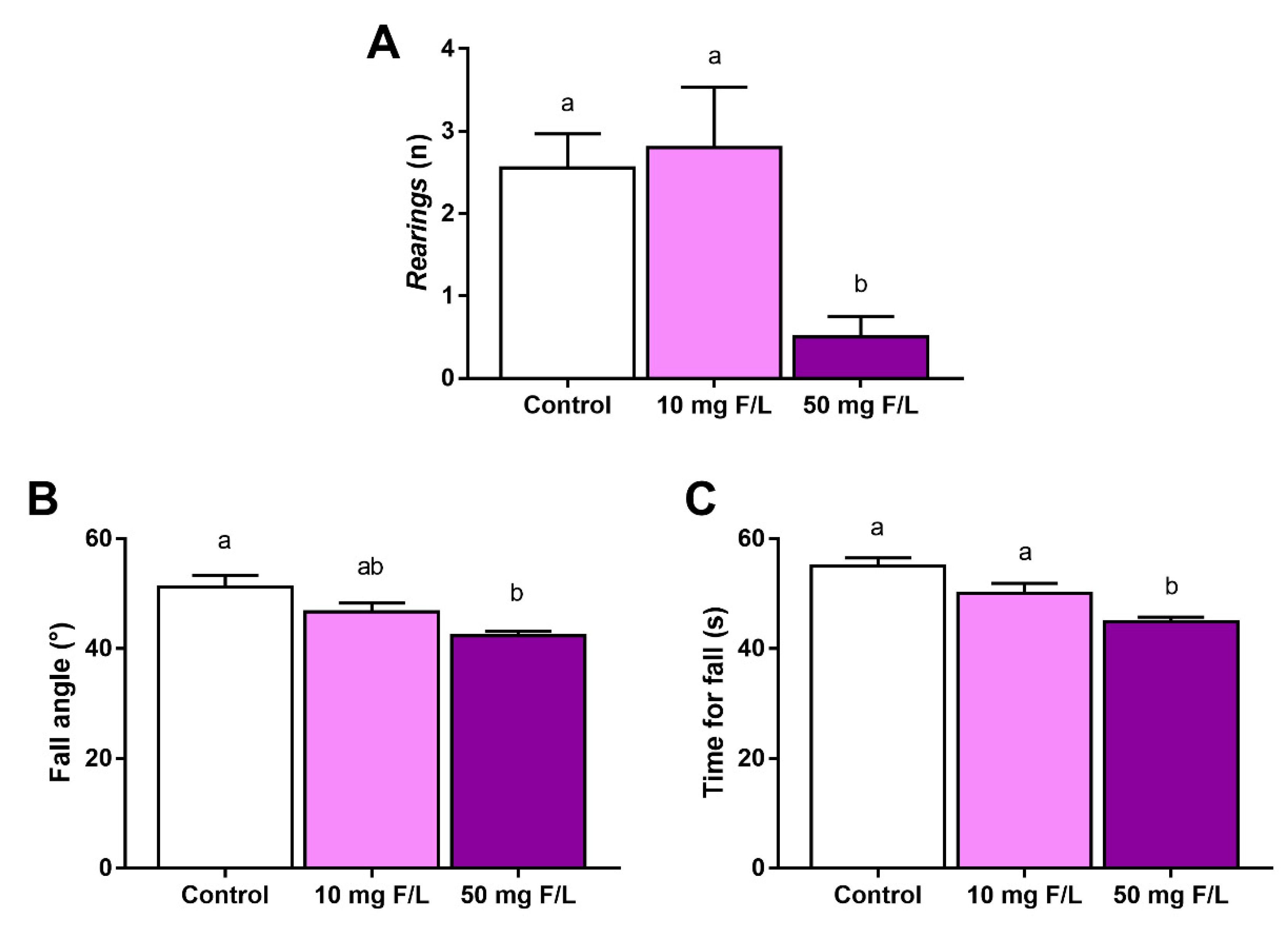
2.4. F Exposure during Prenatal and Postnatal Periods Was Able to Promote Alterations in Rats’ Exploratory Behavior and Motor Coordination
3. Discussion
4. Materials and Methods
4.1. Ethics Statement and Experimental Groups
4.2. Behavioral Tests
4.3. Biological Sample Resection
4.4. Perfusion and Histological Analysis
4.4.1. Purkinje Cell Counting
4.4.2. Immunohistochemical Assays
4.5. Oxidative Biochemistry Assays
4.5.1. Antioxidant Capacity against Peroxyl Radicals
4.5.2. Lipid Peroxidation
4.6. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dhar, V.; Bhatnagar, M. Physiology and toxicity of fluoride. Indian J. Dent. Res. 2009, 20, 350. [Google Scholar] [CrossRef] [PubMed]
- Ozsvath, D.L. Fluoride and environmental health: A review. Rev. Environ. Sci. Bio/Technol. 2009, 8, 59–79. [Google Scholar] [CrossRef]
- Buzalaf, M.A.R.; Pessan, J.P.; Honório, H.M.; Ten Cate, J.M. Mechanisms of action of fluoride for caries control. Monogr. Oral. Sci. 2011, 22, 97–114. [Google Scholar] [PubMed]
- Buzalaf, M.A.R.; Whitford, G.M. Fluoride metabolism. Monogr. Oral. Sci. 2011, 22, 20–36. [Google Scholar]
- Basha, P.M.; Rai, P.; Begum, S. Evaluation of fluoride-induced oxidative stress in rat brain: A multigeneration study. Biol. Trace Elem. Res. 2010, 142, 623–637. [Google Scholar] [CrossRef]
- Ding, Y.; Gao, Y.; Sun, H.; Han, H.; Wang, W.; Ji, X.; Liu, X.; Sun, D. The relationships between low levels of urine fluoride on children’s intelligence, dental fluorosis in endemic fluorosis areas in Hulunbuir, Inner Mongolia, China. J. Hazard. Mater. 2011, 186, 1942–1946. [Google Scholar] [CrossRef]
- Dec, K.; Łukomska, A.; Maciejewska, D.; Jakubczyk, K.; Baranowska-Bosiacka, I.; Chlubek, D.; Wąsik, A.; Gutowska, I. The Influence of Fluorine on the Disturbances of Homeostasis in the Central Nervous System. Biol. Trace Elem. Res. 2017, 177, 224–234. [Google Scholar] [CrossRef] [Green Version]
- Trivedi, M.H.; Sangai, N.P.; Patel, R.S.; Payak, M.; Vyas, S.J. Assessment of groundwater quality with special reference to fluoride and its impact on IQ of schoolchildren in six villages of the Mundra Region, Kachchh, Gujarat, India. Fluoride 2012, 45, 377–383. [Google Scholar]
- Karimzade, S.; Aghaei, M.; Mahvi, A.H. Investigation of intelligence quotient in 9–12-year-old children exposed to high- and low-drinking water fluoride in west azerbaijan province, Iran. Fluoride 2014, 47, 9–14. [Google Scholar]
- Rocha-Amador, D.; Navarro, M.E.; Carrizales, L.; Morales, R.; Calderón, J. Decreased intelligence in children and exposure to fluoride and arsenic in drinking water. Cad. Saúde Pública 2007, 23, S579–S587. [Google Scholar] [CrossRef]
- Choi, A.L.; Sun, G.; Zhang, Y.; Grandjean, P. Developmental fluoride neurotoxicity: A systematic review and meta-analysis. Environ. Health Perspect. 2012, 120, 1362–1368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miranda, G.H.N.; Alvarenga, M.O.P.; Ferreira, M.K.M.; Puty, B.; Bittencourt, L.O.; Fagundes, N.C.F.; Pessan, J.P.; Buzalaf, M.A.R.; Lima, R.R. A systematic review and meta-analysis of the association between fluoride exposure and neurological disorders. Sci. Rep. 2021, 11, 22659. [Google Scholar] [CrossRef]
- Grandjean, P. Developmental fluoride neurotoxicity: An updated review. Environ. Health 2019, 18, 110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanduti, D.; Sterbenk, P.; Artnik, A. Fluoride: A Review of Use and Effects on Health. Socio Med. 2016, 28, 133–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Till, C.; Green, R. Controversy: The evolving science of fluoride: When new evidence doesn’t conform with existing beliefs. Pediatr. Res. 2021, 90, 1093–1095. [Google Scholar] [CrossRef]
- Ferreira, M.K.M.; Aragão, W.A.B.; Bittencourt, L.O.; Puty, B.; Dionizio, A.; Souza, M.P.C.; Buzalaf, M.A.R.; Oliveira, E.H.; Crespo-Lopez, M.E.; Lima, R.R. Fluoride exposure during pregnancy and lactation triggers oxidative stress and molecular changes in hippocampus of offspring rats. Ecotoxicol. Environ. Saf. 2021, 208, 111437. [Google Scholar] [CrossRef] [PubMed]
- Lopes, G.O.; Ferreira, M.K.M.; Davis, L.; Bittencourt, L.O.; Aragão, W.A.B.; Dionizio, A.; Buzalaf, M.A.R.; Crespo-Lopez, M.E.; Maia, C.S.F.; Lima, R.R. Effects of Fluoride Long-Term Exposure over the Cerebellum: Global Proteomic Profile, Oxidative Biochemistry, Cell Density, and Motor Behavior Evaluation. Int. J. Mol. Sci. 2020, 21, 7297. [Google Scholar] [CrossRef]
- Wang, S.X.; Wang, Z.H.; Cheng, X.T.; Li, J.; Sang, Z.P.; Zhang, X.D.; Han, L.L.; Qiao, X.Y.; Wu, Z.M.; Wang, Z.Q. Arsenic and fluoride exposure in drinking water: Children’s IQ and growth in Shanyin county, Shanxi province, China. Environ. Health Perspect. 2007, 115, 643–647. [Google Scholar] [CrossRef] [Green Version]
- Guth, S.; Hüser, S.; Roth, A.; Degen, G.; Diel, P.; Edlund, K.; Eisenbrand, G.; Engel, K.; Epe, B.; Grune, T.; et al. Toxicity of fluoride: Critical evaluation of evidence for human developmental neurotoxicity in epidemiological studies, animal experiments and in vitro analyses. Arch. Toxicol. 2020, 94, 1375–1415. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Questions and Answers on Fluoride. Available online: https://www.epa.gov/sites/default/files/2015–10/documents/2011_fluoride_questionsanswers.pdf (accessed on 30 May 2022).
- Dionizio, A.S.; Melo, C.G.S.; Sabino-Arias, I.T.; Ventura, T.M.S.; Leite, A.L.; Souza, S.R.G.; Santos, E.X.; Heubel, A.D.; Souza, J.G.; Perles, J.V.C.M.; et al. Chronic treatment with fluoride affects the jejunum: Insights from proteomics and enteric innerva-tion analysis. Sci. Rep. 2018, 8, 3180. [Google Scholar] [CrossRef]
- Dunipace, A.J.; Brizendine, E.J.; Zhang, W.; Wilson, M.E.; Miller, L.L.; Katz, B.P.; Warrick, J.M.; Stookey, G.K. Effect of aging on animal response to chronic fluoride exposure. J. Dent. Res. 1995, 74, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Castiblanco-Rubio, G.A.; Muñoz-Rocha, T.V.; Téllez-Rojo, M.M.; Ettinger, A.S.; Mercado-García, A.; Peterson, K.E.; Hu, H.; Cantoral, A.; Martínez-Mier, E.A. Dietary Influences on Urinary Fluoride over the Course of Pregnancy and at One-Year Postpartum. Biol. Trace Elem. Res. 2022, 200, 1568–1579. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Verma, P.K.; Sood, S.; Singh, R.; Gupta, A.; Rastogi, A. Distribution of Fluoride in Plasma, Brain, and Bones and Associated Oxidative Damage After Induced Chronic Fluorosis in Wistar Rats. Biol. Trace Elem. Res. 2021, 200, 1710–1721. [Google Scholar] [CrossRef] [PubMed]
- Arber, S.; Costa, R.M. Connecting neuronal circuits for movement. Science 2018, 29, 1403–1404. [Google Scholar] [CrossRef] [Green Version]
- Grillner, S.; El Manira, A. Current Principles of Motor Control, with Special Reference to Vertebrate Locomotion. Physiol. Rev. 2020, 1, 271–320. [Google Scholar] [CrossRef] [Green Version]
- Ito, M. Error detection and representation in the olivo-cerebellar system. Front. Neural Circuits 2013, 7, 1. [Google Scholar] [CrossRef] [Green Version]
- Ran, S.; Sun, N.; Liu, Y.; Zhang, W.; Li, Y.; Wei, L.; Wang, J.; Liu, B. Fluoride resistance capacity in mammalian cells involves complex global gene expression changes. FEBS Open Bio 2017, 7, 968–980. [Google Scholar] [CrossRef] [Green Version]
- Chen, R.; Zhao, L.-D.; Liu, H.; Li, H.-H.; Ren, C.; Zhang, P.; Guo, K.-T.; Zhang, H.-X.; Geng, D.-Q.; Zhang, C.-Y. Fluoride Induces Neuroinflammation and Alters Wnt Signaling Pathway in BV2 Microglial Cells. Inflammation 2017, 40, 1123–1130. [Google Scholar] [CrossRef]
- Fina, B.L.; Lombarte, M.; Rigalli, J.P.; Rigalli, A. Fluoride increases superoxide production and impairs the respiratory chain in ROS 17/2.8 osteoblastic cells. PLoS ONE 2014, 9, e100768. [Google Scholar] [CrossRef]
- Johnston, N.R.; Strobel, S.A. Principles of fluoride toxicity and the cellular response: A review. Arch. Toxicol. 2020, 94, 1051–1069. [Google Scholar] [CrossRef]
- Perera, T.; Ranasinghe, S.; Alles, N.; Waduge, R. Effect of fluoride on major organs with the different time of exposure in rats. Environ. Health Prev. Med. 2018, 23, 17. [Google Scholar] [CrossRef]
- Preiser, J.C. Oxidative stress. J. Parenter. Enteral. Nutr. 2012, 36, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Tsikas, D. Assessment of lipid peroxidation by measuring malondialdehyde (MDA) and relatives in biological samples: Ana-lytical and biological challenges. Anal. Biochem. 2017, 524, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Reichmann, D.; Voth, W.; Jakob, U. Maintaining a Healthy Proteome during Oxidative Stress. Mol. Cell 2018, 18, 203–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, N.; Yao, Y.; Ma, Y.; Meng, X.; Sowanou, A.; Pei, J. Effects of Fluoride on Oxidative Stress Markers of Lipid, Gene, and Protein in Rats. Biol. Trace Elem. Res. 2020, 199, 2238–2246. [Google Scholar] [CrossRef] [PubMed]
- Owumi, S.E.; Aliyu-Banjo, N.O.; Danso, O.F. Fluoride and diethylnitrosamine coexposure enhances oxido-inflammatory re-sponses and caspase-3 activation in liver and kidney of adult rats. J. Biochem. Mol. Toxicol. 2019, 33, 22327. [Google Scholar] [CrossRef]
- Pereira, H.A.B.D.S.; Dionizio, A.S.; Araujo, T.T.; Fernandes, M.D.S.; Iano, F.G.; Buzalaf, M.A.R. Proposed mechanism for understanding the dose- and time-dependency of the effects of fluoride in the liver. Toxicol. Appl. Pharmacol. 2018, 358, 68–75. [Google Scholar] [CrossRef]
- Paul, M.S.; Limaiem, F. Histology, Purkinje Cells. In StatPearls; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Xia, G.; Santostefano, K.; Hamazaki, T.; Liu, J.; Subramony, S.H.; Terada, N.; Ashizawa, T. Generation of human-induced pluripotent stem cells to model spinocerebellar ataxia type 2 In Vitro. J. Mol. Neurosci. 2012, 51, 237–248. [Google Scholar] [CrossRef] [Green Version]
- Gusel’nikova, V.V.; Korzhevskiy, D.E. NeuN As a Neuronal Nuclear Antigen and Neuron Differentiation Marker. Acta Nat. 2015, 7, 42–47. [Google Scholar] [CrossRef]
- Leão, L.K.R.; Bittencourt, L.O.; Oliveira, A.C.; Nascimento, P.C.; Miranda, G.H.N.; Ferreira, R.O.; Nabiça, M.; Dantas, K.; Dionizio, A.; Cartágenes, S.; et al. Long-Term Lead Exposure Since Adolescence Causes Proteomic and Morphological Altera-tions in the Cerebellum Associated with Motor Deficits in Adult Rats. Int. J. Mol. Sci. 2020, 21, 3571. [Google Scholar] [CrossRef]
- Boggs, J.M. Myelin basic protein: A multifunctional protein. Cell Mol. Life Sci. 2006, 63, 1945–1961. [Google Scholar] [CrossRef]
- Chang, M.-W.; Young, M.-S.; Lin, M.-T. An inclined plane system with microcontroller to determine limb motor function of laboratory animals. J. Neurosci. Methods 2008, 168, 186–194. [Google Scholar] [CrossRef]
- Galante, M.; Jani, H.; Vanes, L.; Daniel, H.; Fisher, E.M.; Tybulewicz, V.L.; Bliss, T.V.; Morice, E. Impairments in motor coor-dination without major changes in cerebellar plasticity in the Tc1 mouse model of Down syndrome. Hum. Mol. Genet. 2009, 15, 1449–1463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Pandolfo, P.; Vendruscolo, L.F.; Sordi, R.; Takahashi, R.N. Cannabinoid induced conditioned place preference in the sponta-neously hypertensive rat–an animal model of attention deficit hyperactivity disorder. Psychopharmacology 2009, 205, 319–326. [Google Scholar] [CrossRef]
- Teixeira, F.B.; Santana, L.N.D.S.; Bezerra, F.R.; De Carvalho, S.; Fontes-Júnior, E.A.; Prediger, R.D.; Crespo-López, M.E.; Maia, C.S.F.; Lima, R.R. Chronic Ethanol Exposure during Adolescence in Rats Induces Motor Impairments and Cerebral Cortex Damage Associated with Oxidative Stress. PLoS ONE 2014, 9, e101074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bittencourt, L.O.; Chemelo, V.S.; Aragão, W.A.B.; Puty, B.; Dionizio, A.; Teixeira, F.B.; Fernandes, M.S.; Silva, M.C.F.; Fernandes, L.M.P.; de Oliveira, E.H.C.; et al. From Molecules to Behavior in Long-Term Inorganic Mercury Intoxication: Unraveling Proteomic Features in Cerebellar Neurodegeneration of Rats. Int. J. Mol. Sci. 2021, 23, 111. [Google Scholar] [CrossRef] [PubMed]
- Freire, M.A.M.; Faber, J.; Picanço-Diniz, C.W.; Franca, J.G.; Pereira, A. Morphometric variability of nicotinamide adenine dinucleotide phosphate diaphorase neurons in the primary sensory areas of the rat. Neuroscience 2012, 205, 140–153. [Google Scholar] [CrossRef]
- Amado, L.L.; Garcia, M.L.; Ramos, P.B.; Freitas, R.F.; Zafalon, B.; Ferreira, J.L.R.; Yunes, J.S.; Monserrat, J.M. A method to measure total antioxidant capacity against peroxyl radicals in aquatic organisms: Application to evaluate microcystins toxicity. Sci. Total Environ. 2009, 407, 2115–2123. [Google Scholar] [CrossRef]
- Esterbauer, H.; Cheeseman, K.H. Determination of aldehydic lipid peroxidation products: Malonaldehyde and 4-hydroxynonenal. Methods Enzymol. 1990, 186, 407–421. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souza-Monteiro, D.; Ferreira, M.K.M.; Bittencourt, L.O.; Aragão, W.A.B.; Oliveira, I.G.d.; Maia, C.S.F.; Freire, M.A.M.; Zohoori, F.V.; Buzalaf, M.A.R.; Lima, R.R. Intrauterine and Postnatal Exposure to High Levels of Fluoride Is Associated with Motor Impairments, Oxidative Stress, and Morphological Damage in the Cerebellum of Offspring Rats. Int. J. Mol. Sci. 2022, 23, 8556. https://doi.org/10.3390/ijms23158556
Souza-Monteiro D, Ferreira MKM, Bittencourt LO, Aragão WAB, Oliveira IGd, Maia CSF, Freire MAM, Zohoori FV, Buzalaf MAR, Lima RR. Intrauterine and Postnatal Exposure to High Levels of Fluoride Is Associated with Motor Impairments, Oxidative Stress, and Morphological Damage in the Cerebellum of Offspring Rats. International Journal of Molecular Sciences. 2022; 23(15):8556. https://doi.org/10.3390/ijms23158556
Chicago/Turabian StyleSouza-Monteiro, Deiweson, Maria Karolina Martins Ferreira, Leonardo Oliveira Bittencourt, Walessa Alana Bragança Aragão, Igor Gonçalves de Oliveira, Cristiane Socorro Ferraz Maia, Marco Aurelio M. Freire, Fatemeh Vida Zohoori, Marília Afonso Rabelo Buzalaf, and Rafael Rodrigues Lima. 2022. "Intrauterine and Postnatal Exposure to High Levels of Fluoride Is Associated with Motor Impairments, Oxidative Stress, and Morphological Damage in the Cerebellum of Offspring Rats" International Journal of Molecular Sciences 23, no. 15: 8556. https://doi.org/10.3390/ijms23158556
APA StyleSouza-Monteiro, D., Ferreira, M. K. M., Bittencourt, L. O., Aragão, W. A. B., Oliveira, I. G. d., Maia, C. S. F., Freire, M. A. M., Zohoori, F. V., Buzalaf, M. A. R., & Lima, R. R. (2022). Intrauterine and Postnatal Exposure to High Levels of Fluoride Is Associated with Motor Impairments, Oxidative Stress, and Morphological Damage in the Cerebellum of Offspring Rats. International Journal of Molecular Sciences, 23(15), 8556. https://doi.org/10.3390/ijms23158556










