Low Efficacy of Genetic Tests for the Diagnosis of Primary Lymphedema Prompts Novel Insights into the Underlying Molecular Pathways
Abstract
1. Introduction
2. Results
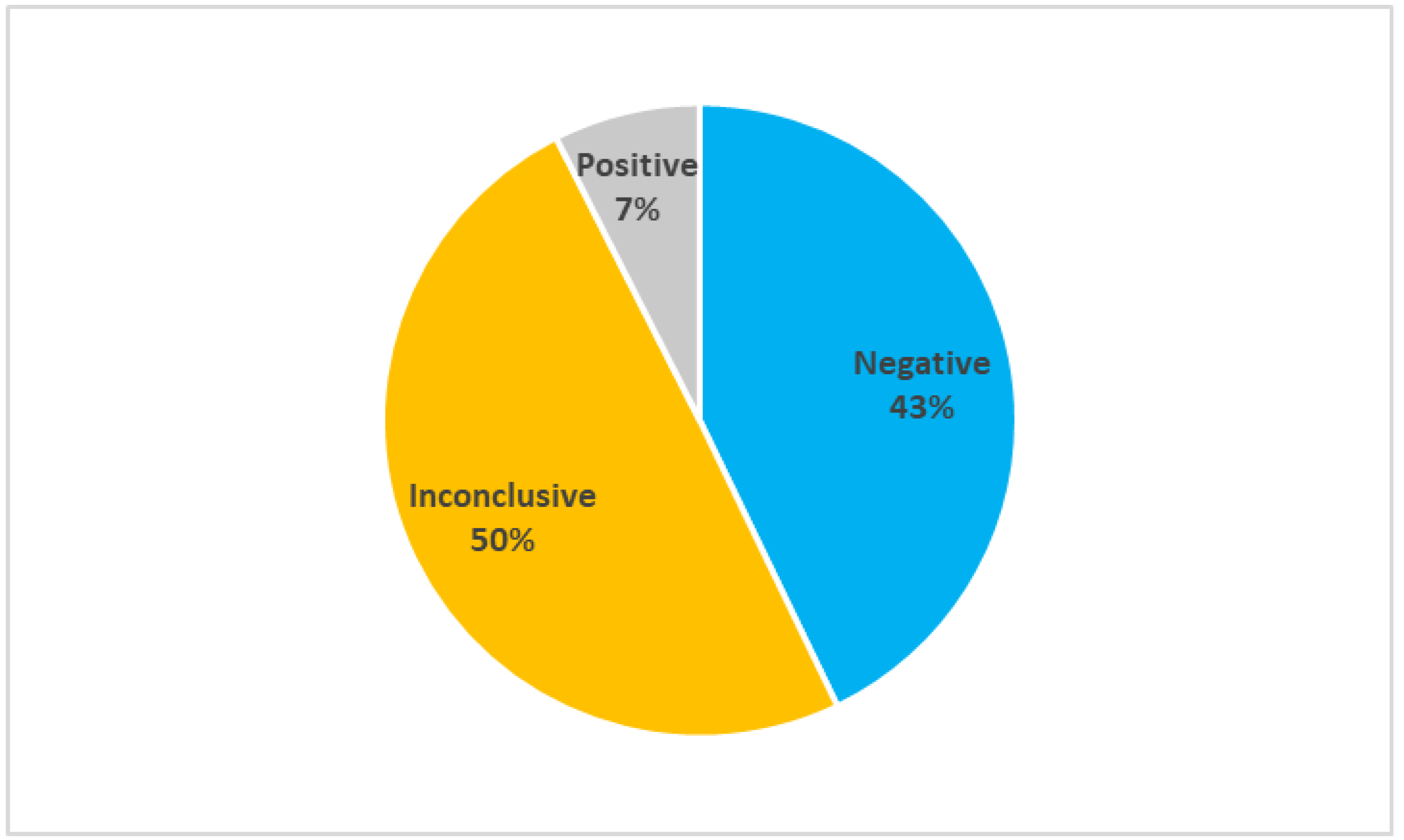
2.1. Efficacy of the Current Genetic Tests
2.2. Study Cohort Characteristics
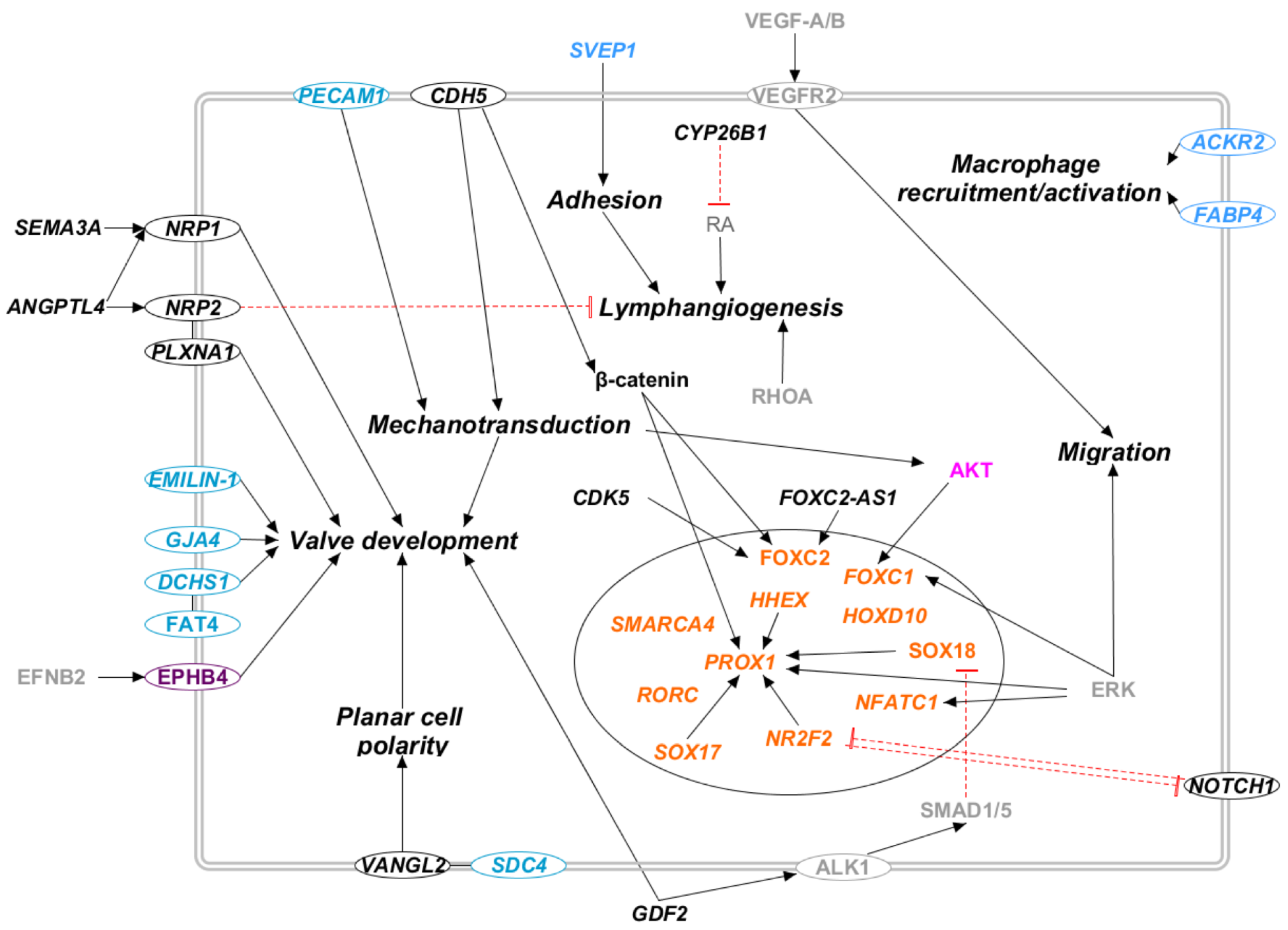
2.3. Molecular Pathways Involved in Primary Lymphedema
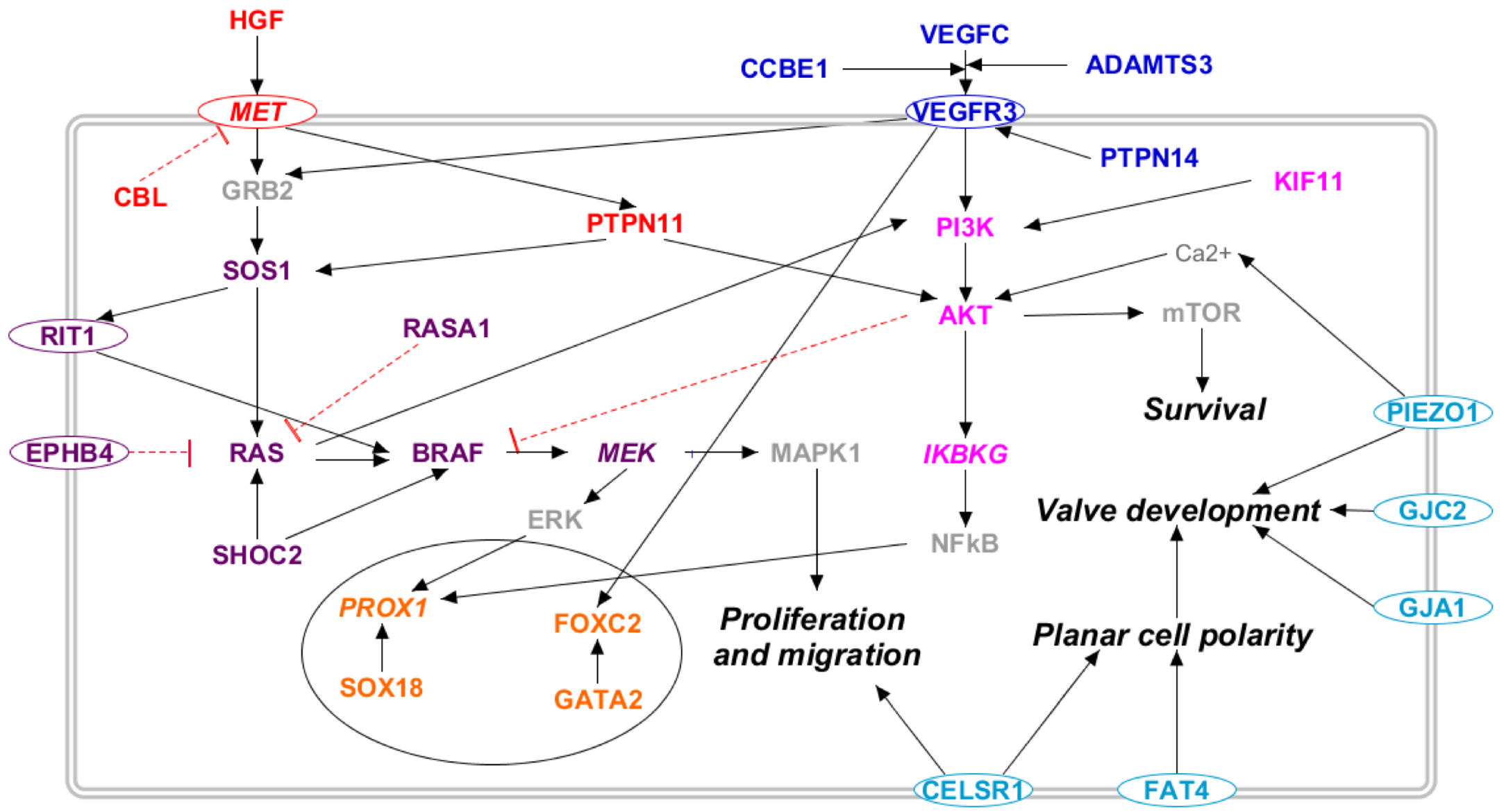
2.3.1. Diagnostic Gene Pathways
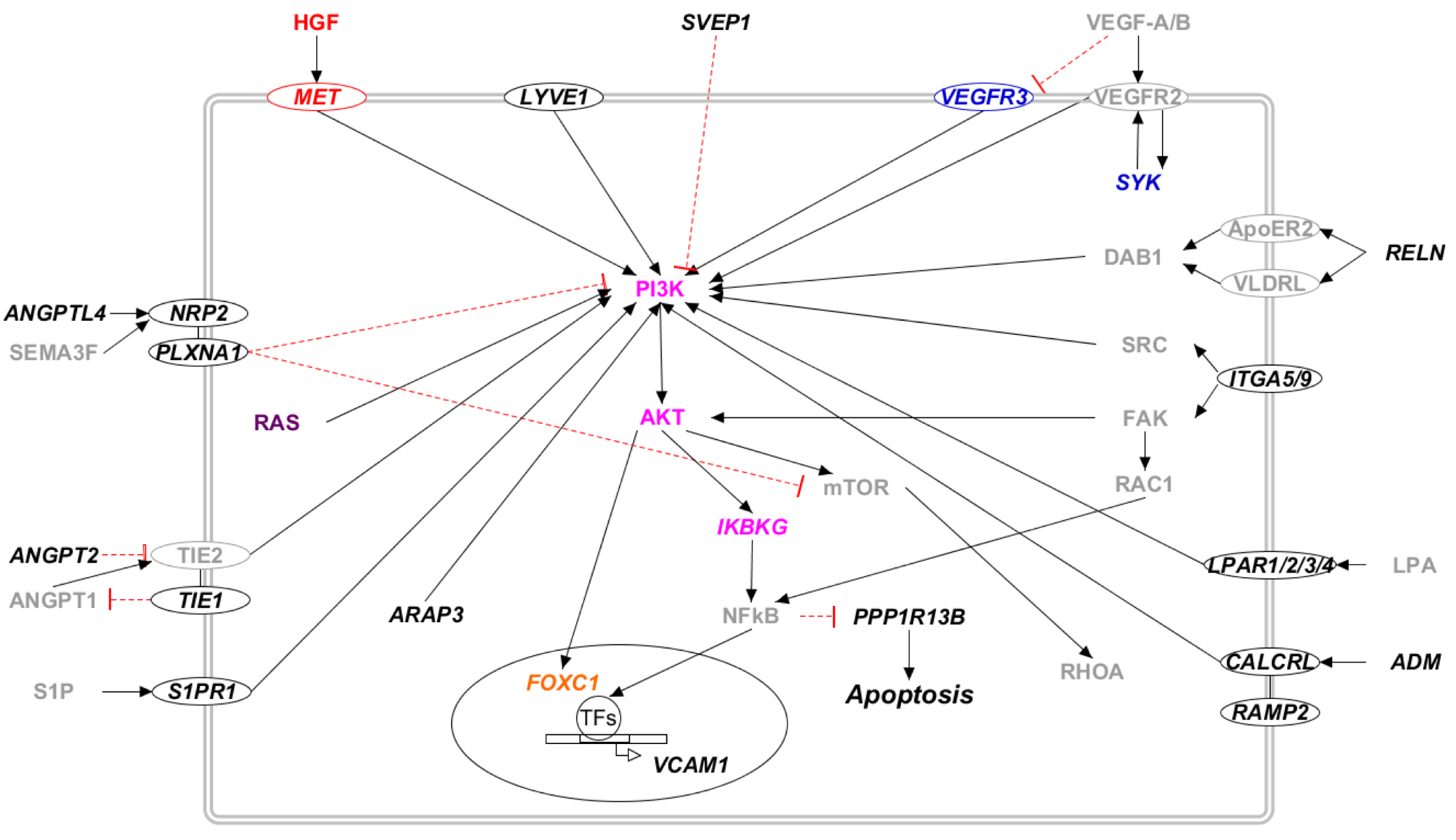
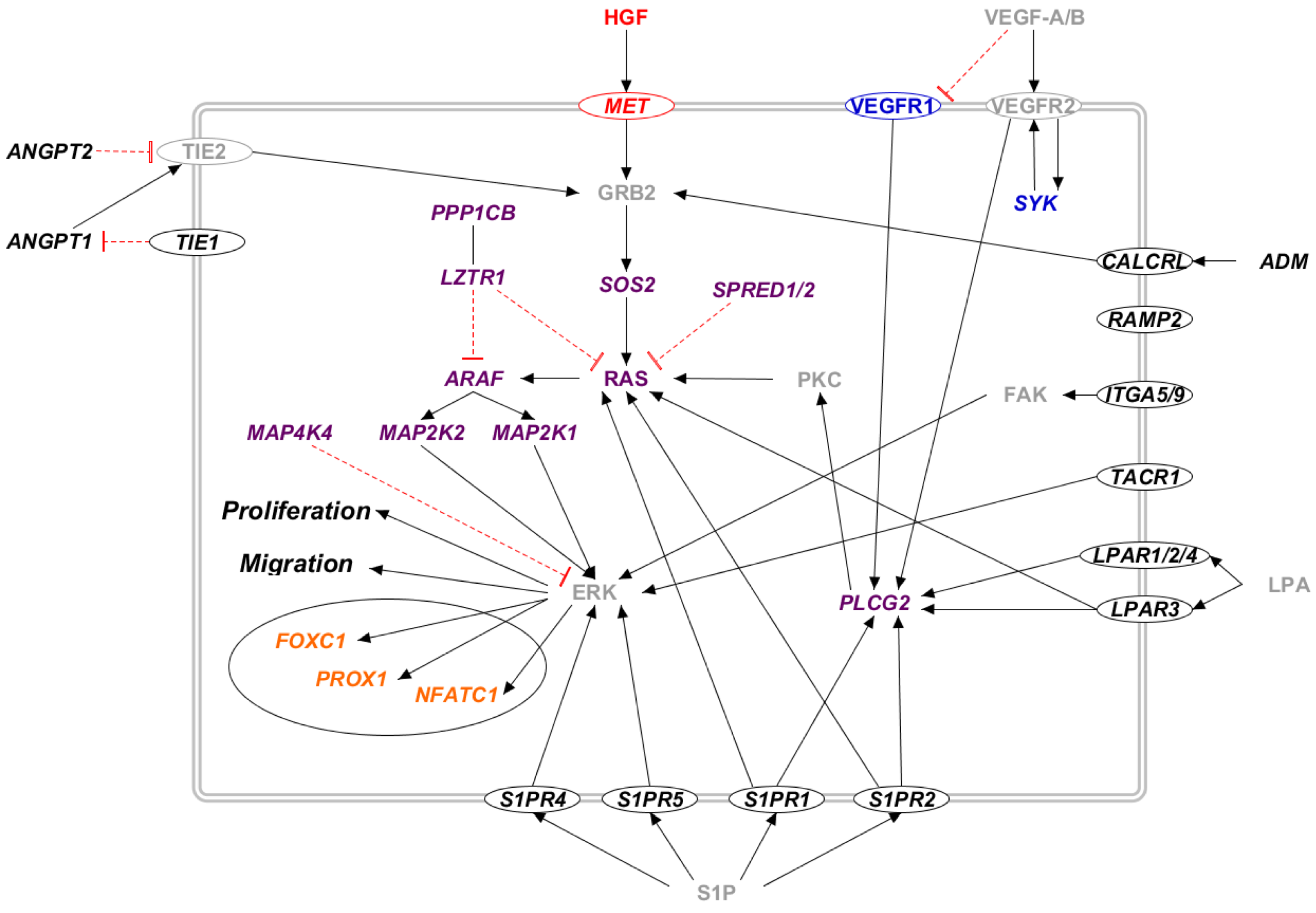
2.3.2. Candidate Gene Pathways
3. Discussion
3.1. Molecular Pathways Involved in Primary Lymphedema
3.1.1. VEGF-C/VEGFR-3 Pathway
3.1.2. HGF/MET Pathway
3.1.3. PI3K/AKT Pathway
3.1.4. RAS/MAPK Pathway
3.1.5. Rho/ROCK Pathway
3.1.6. Transcription Factors
3.2. Physiological Outcomes
3.2.1. Planar Cell Polarity and Lymphatic Valve Development
3.2.2. Lymph Pumping
3.2.3. Macrophage Activation and Lymphangiogenesis
3.3. Limitations and Future Perspectives
4. Materials and Methods
4.1. Genetic and Data Analysis
- Negative when no variants were found in the analyzed genes;
- Inconclusive when the detected variants could be causative of the disease, but their effect is uncertain;
- Positive when the detected variants were correlated with the disease.
4.2. Creation of Molecular Pathway Diagrams
- The gene is involved in the development of the lymphatic system in vivo;
- Gene mutations cause in vivo lymphatic defects;
- The gene participates in a molecular pathway that is important in lymphedema pathogenesis.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fossum, T.W.; Miller, M.W. Lymphedema: Etiopathogenesis. J. Vet. Intern. Med. 1992, 6, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.M. Lymphedema-Distichiasis Syndrome: Report of a Case and Review. Arch. Dermatol. 1999, 135, 347–348. [Google Scholar] [CrossRef] [PubMed]
- Okeke, A. Lymphoedema in Urological Cancer. Eur. Urol. 2004, 45, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Bhusan Tripathi, Y.; Pandey, N.; Mishra, P.; Tripathi, P.; Coatto, M.; Anpilogov, K.; Dhuli, K.; Donato, K.; Michelini, S.; Cecchin, S.; et al. Effect of a Dietary Supplement on the Reduction of Lymphedema-Progression in Mouse Tail-Cut Model. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Bertelli, M.; Kiani, A.K.; Paolacci, S.; Manara, E.; Kurti, D.; Dhuli, K.; Bushati, V.; Miertus, J.; Pangallo, D.; Baglivo, M.; et al. Hydroxytyrosol: A natural compound with promising pharmacological activities. J. Biotechnol. 2019, 309, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Michelini, S.; Paolacci, S.; Manara, E.; Eretta, C.; Mattassi, R.; Lee, B.-B.; Bertelli, M. Genetic tests in lymphatic vascular malformations and lymphedema. J. Med. Genet. 2018, 55, 222–232. [Google Scholar] [CrossRef]
- Michelini, S.; Degiorgio, D.; Cestari, M.; Corda, D.; Ricci, M.; Cardone, M.; Mander, A.; Famoso, L.; Contini, E.; Serrani, R.; et al. Clinical and genetic study of 46 Italian patients with primary lymphedema. Lymphology 2012, 45, 3–12. [Google Scholar]
- Michelini, S.; Vettori, A.; Maltese, P.E.; Cardone, M.; Bruson, A.; Fiorentino, A.; Cappellino, F.; Sainato, V.; Guerri, G.; Marceddu, G.; et al. Genetic Screening in a Large Cohort of Italian Patients Affected by Primary Lymphedema Using a Next Generation Sequencing (NGS) Approach. Lymphology 2016, 49, 57–72. [Google Scholar]
- Dayan, J.H.; Ly, C.L.; Kataru, R.P.; Mehrara, B.J. Lymphedema: Pathogenesis and Novel Therapies. Annu. Rev. Med. 2018, 69, 263–276. [Google Scholar] [CrossRef]
- Baglivo, M.; Martelli, F.; Paolacci, S.; Manara, E.; Michelini, S.; Bertelli, M. Electrical Stimulation in the Treatment of Lymphedema and Associated Skin Ulcers. Lymphat. Res. Biol. 2020, 18, 270–276. [Google Scholar] [CrossRef]
- Bertelli, M.; Kiani, A.K.; Paolacci, S.; Manara, E.; Dautaj, A.; Beccari, T.; Michelini, S. Molecular pathways involved in lymphedema: Hydroxytyrosol as a candidate natural compound for treating the effects of lymph accumulation. J. Biotechnol. 2019, 308, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Brouillard, P.; Boon, L.; Vikkula, M. Genetics of Lymphatic Anomalies. J. Clin. Investig. 2014, 124, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Balboa-Beltran, E.; Fernández-Seara, M.J.; Pérez-Muñuzuri, A.; Lago, R.; García-Magán, C.; Couce, M.L.; Sobrino, B.; Amigo, J.; Carracedo, A.; Barros, F. A novel stop mutation in the vascular endothelial growth factor-C gene (VEGFC) results in Milroy-like disease. J. Med. Genet. 2014, 51, 475–478. [Google Scholar] [CrossRef] [PubMed]
- Karkkainen, M.J.; Ferrell, R.E.; Lawrence, E.C.; Kimak, M.A.; Levinson, K.L.; McTigue, M.A.; Alitalo, K.; Finegold, D. Missense mutations interfere with VEGFR-3 signalling in primary lymphoedema. Nat. Genet. 2000, 25, 153–159. [Google Scholar] [CrossRef]
- Ghalamkarpour, A.; Morlot, S.; Raas-Rothschild, A.; Utkus, A.; Mulliken, J.; Boon, L.; Vikkula, M. Hereditary lymphedema type I associated with VEGFR3 mutation: The first de novo case and atypical presentations. Clin. Genet. 2006, 70, 330–335. [Google Scholar] [CrossRef]
- Lohela, M.; Bry, M.; Tammela, T.; Alitalo, K. VEGFs and receptors involved in angiogenesis versus lymphangiogenesis. Curr. Opin. Cell Biol. 2009, 21, 154–165. [Google Scholar] [CrossRef]
- Karkkainen, M.J.; Saaristo, A.; Jussila, L.; Karila, K.A.; Lawrence, E.C.; Pajusola, K.; Bueler, H.; Eichmann, A.; Kauppinen, R.; Kettunen, M.I.; et al. A model for gene therapy of human hereditary lymphedema. Proc. Natl. Acad. Sci. USA 2001, 98, 12677–12682. [Google Scholar] [CrossRef]
- Karkkainen, M.J.; Haiko, P.; Sainio, K.; Partanen, J.; Taipale, J.; Petrova, T.V.; Jeltsch, M.; Jackson, D.G.; Talikka, M.; Rauvala, H.; et al. Vascular endothelial growth factor C is required for sprouting of the first lymphatic vessels from embryonic veins. Nat. Immunol. 2003, 5, 74–80. [Google Scholar] [CrossRef]
- Dumont, D.J.; Jussila, L.; Taipale, J.; Lymboussaki, A.; Mustonen, T.; Pajusola, K.; Breitman, M.; Alitalo, K. Cardiovascular Failure in Mouse Embryos Deficient in VEGF Receptor-3. Science 1998, 282, 946–949. [Google Scholar] [CrossRef]
- Mäkinen, T.; Veikkola, T.; Mustjoki, S.; Karpanen, T.; Catimel, B.; Nice, E.C.; Wise, L.; Mercer, A.; Kowalski, H.; Kerjaschki, D.; et al. Isolated lymphatic endothelial cells transduce growth, survival and migratory signals via the VEGF-C/D receptor VEGFR-3. EMBO J. 2001, 20, 4762–4773. [Google Scholar] [CrossRef]
- Le Guen, L.; Karpanen, T.; Schulte, D.; Harris, N.C.; Koltowska, K.; Roukens, G.; Bower, N.; van Impel, A.; Stacker, S.; Achen, M.; et al. Ccbe1 regulates Vegfc-mediated induction of Vegfr3 signaling during embryonic lymphangiogenesis. Development 2014, 141, 1239–1249. [Google Scholar] [CrossRef] [PubMed]
- Connell, F.; Kalidas, K.; Ostergaard, P.; Brice, G.; Homfray, T.; Roberts, L.; Bunyan, D.J.; Mitton, S.; Mansour, S.; Mortimer, P.; et al. Linkage and sequence analysis indicate that CCBE1 is mutated in recessively inherited generalised lymphatic dysplasia. Qual. Life Res. 2009, 127, 231–241. [Google Scholar] [CrossRef]
- Alders, M.; Hogan, B.; Gjini, E.; Salehi, F.; Al-Gazali, L.; Hennekam, E.A.; Holmberg, E.E.; Mannens, M.M.A.M.; Mulder, M.F.; Offerhaus, G.J.A.; et al. Mutations in CCBE1 cause generalized lymph vessel dysplasia in humans. Nat. Genet. 2009, 41, 1272–1274. [Google Scholar] [CrossRef]
- Brouillard, P.; Dupont, L.; Helaers, R.; Coulie, R.; E Tiller, G.; Peeden, J.; Colige, A.; Vikkula, M. Loss of ADAMTS3 activity causes Hennekam lymphangiectasia–lymphedema syndrome 3. Hum. Mol. Genet. 2017, 26, 4095–4104. [Google Scholar] [CrossRef] [PubMed]
- Au, A.C.; Hernandez, P.A.; Lieber, E.; Nadroo, A.M.; Shen, Y.-M.; Kelley, K.A.; Gelb, B.D.; Diaz, G.A. Protein Tyrosine Phosphatase PTPN14 Is a Regulator of Lymphatic Function and Choanal Development in Humans. Am. J. Hum. Genet. 2010, 87, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Bordbar, A.; Maroofian, R.; Ostergaard, P.; Kashaki, M.; Nikpour, S.; Gordon, K.; Crosby, A.; Khosravi, P.; Shojaei, A. A homozygous loss-of-function mutation in PTPN14 causes a syndrome of bilateral choanal atresia and early infantile-onset lymphedema. Meta Gene 2017, 14, 53–58. [Google Scholar] [CrossRef]
- Finegold, D.N.; Schacht, V.; Kimak, M.A.; Lawrence, E.C.; Foeldi, E.; Karlsson, J.M.; Baty, C.J.; Ferrell, R.E. HGF and MET Mutations in Primary and Secondary Lymphedema. Lymphat. Res. Biol. 2008, 6, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Kajiya, K.; Hirakawa, S.; Ma, B.; Drinnenberg, I.; Detmar, M. Hepatocyte growth factor promotes lymphatic vessel formation and function. EMBO J. 2005, 24, 2885–2895. [Google Scholar] [CrossRef]
- Martinelli, S.; De Luca, A.; Stellacci, E.; Rossi, C.; Checquolo, S.; Lepri, F.; Caputo, V.; Silvano, M.; Buscherini, F.; Consoli, F.; et al. Heterozygous Germline Mutations in the CBL Tumor-Suppressor Gene Cause a Noonan Syndrome-like Phenotype. Am. J. Hum. Genet. 2010, 87, 250–257. [Google Scholar] [CrossRef]
- Tartaglia, M.; Kalidas, K.; Shaw, A.; Song, X.; Musat, D.L.; van der Burgt, I.; Brunner, H.G.; Bertola, D.R.; Crosby, A.; Ion, A.; et al. PTPN11 Mutations in Noonan Syndrome: Molecular Spectrum, Genotype-Phenotype Correlation, and Phenotypic Heterogeneity. Am. J. Hum. Genet. 2002, 70, 1555–1563. [Google Scholar] [CrossRef]
- Kosaki, K.; Suzuki, T.; Muroya, K.; Hasegawa, T.; Sato, S.; Matsuo, N.; Kosaki, R.; Nagai, T.; Hasegawa, Y.; Ogata, T. PTPN11(Protein-Tyrosine Phosphatase, Nonreceptor-Type 11) Mutations in Seven Japanese Patients with Noonan Syndrome. J. Clin. Endocrinol. Metab. 2002, 87, 3529–3533. [Google Scholar] [CrossRef] [PubMed]
- Shchelochkov, O.A.; Patel, A.; Weissenberger, G.M.; Chinault, A.C.; Wiszniewska, J.; Fernandes, P.H.; Eng, C.; Kukolich, M.K.; Sutton, V.R. Duplication of chromosome band 12q24.11q24.23 results in apparent Noonan syndrome. Am. J. Med. Genet. Part A 2008, 146A, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.M.; Kramer, N.; Bejjani, B.A.; Thiel, C.; Carta, C.; Neri, G.; Tartaglia, M.; Zenker, M. Genomic duplication ofPTPN11is an uncommon cause of Noonan syndrome. Am. J. Med. Genet. Part A 2009, 149A, 2122–2128. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.M. Proteus syndrome review: Molecular, clinical, and pathologic features. Clin. Genet. 2013, 85, 111–119. [Google Scholar] [CrossRef]
- Luks, V.L.; Kamitaki, N.; Vivero, M.P.; Uller, W.; Rab, R.; Bovée, J.V.; Rialon, K.L.; Guevara, C.J.; Alomari, A.I.; Greene, A.K.; et al. Lymphatic and Other Vascular Malformative/Overgrowth Disorders Are Caused by Somatic Mutations in PIK3CA. J. Pediatr. 2015, 166, 1048–1054. [Google Scholar] [CrossRef]
- Kurek, K.C.; Luks, V.L.; Ayturk, U.M.; Alomari, A.I.; Fishman, S.J.; Spencer, S.A.; Mulliken, J.B.; Bowen, M.E.; Yamamoto, G.L.; Kozakewich, H.P.; et al. Somatic Mosaic Activating Mutations in PIK3CA Cause CLOVES Syndrome. Am. J. Hum. Genet. 2012, 90, 1108–1115. [Google Scholar] [CrossRef]
- Rodriguez-Laguna, L.; Agra, N.; Ibáñez, K.; Oliva-Molina, G.; Gordo, G.; Khurana, N.; Hominick, D.; Beato, M.; Colmenero, I.; Herranz, G.; et al. Somatic activating mutations in PIK3CA cause generalized lymphatic anomaly. J. Exp. Med. 2018, 216, 407–418. [Google Scholar] [CrossRef]
- Bui, K.; Hong, Y.-K. Ras Pathways on Prox1 and Lymphangiogenesis: Insights for Therapeutics. Front. Cardiovasc. Med. 2020, 7, 597374. [Google Scholar] [CrossRef]
- Baluk, P.; Yao, L.-C.; Flores, J.C.; Choi, D.; Hong, Y.-K.; McDonald, D.M. Rapamycin reversal of VEGF-C–driven lymphatic anomalies in the respiratory tract. JCI Insight 2017, 2, e90103. [Google Scholar] [CrossRef]
- Flister, M.J.; Wilber, A.; Hall, K.L.; Iwata, C.; Miyazono, K.; Nisato, R.E.; Pepper, M.; Zawieja, D.; Ran, S. Inflammation induces lymphangiogenesis through up-regulation of VEGFR-3 mediated by NF-κB and Prox1. Blood 2010, 115, 418–429. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, L.; Rogers, D.; Su, W.; Odaka, Y.; Zhou, H.; Chen, W.; Shen, T.; Alexander, J.S.; Huang, S. Rapamycin Inhibits Lymphatic Endothelial Cell Tube Formation by Downregulating Vascular Endothelial Growth Factor Receptor 3 Protein Expression. Neoplasia 2012, 14, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, P.; Simpson, M.A.; Mendola, A.; Vasudevan, P.; Connell, F.C.; van Impel, A.; Moore, A.T.; Loeys, B.L.; Ghalamkarpour, A.; Onoufriadis, A.; et al. Mutations in KIF11 Cause Autosomal-Dominant Microcephaly Variably Associated with Congenital Lymphedema and Chorioretinopathy. Am. J. Hum. Genet. 2012, 90, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Fotiou, E.; Martin-Almedina, S.; Simpson, M.; Lin, S.; Gordon, K.; Brice, G.; Atton, G.; A Jeffery, I.T.; Rees, D.C.; Mignot, C.; et al. Novel mutations in PIEZO1 cause an autosomal recessive generalized lymphatic dysplasia with non-immune hydrops fetalis. Nat. Commun. 2015, 6, 8085. [Google Scholar] [CrossRef] [PubMed]
- Crompton, M.; Purnell, T.; Tyrer, H.E.; Parker, A.; Ball, G.; Hardisty-Hughes, R.E.; Gale, R.; Williams, D.; Dean, C.H.; Simon, M.M.; et al. A mutation in Nischarin causes otitis media via LIMK1 and NF-κB pathways. PLoS Genet. 2017, 13, e1006969. [Google Scholar] [CrossRef]
- Sakurai, A.; Doci, C.; Gutkind, J.S. Semaphorin signaling in angiogenesis, lymphangiogenesis and cancer. Cell Res. 2011, 22, 23–32. [Google Scholar] [CrossRef]
- Van Trier, D.C.; Rinne, T.; Noordam, K.; Draaisma, J.M.; Van Der Burgt, I. Variable phenotypic expression in a large Noonan syndrome family segregating a novel SOS1 mutation. Am. J. Med. Genet. Part A 2017, 173, 2968–2972. [Google Scholar] [CrossRef]
- Tartaglia, M.; Pennacchio, L.; Zhao, C.; Yadav, K.K.; Fodale, V.; Sarkozy, A.; Pandit, B.; Oishi, K.; Martinelli, S.; Schackwitz, W.; et al. Gain-of-function SOS1 mutations cause a distinctive form of Noonan syndrome. Nat. Genet. 2006, 39, 75–79. [Google Scholar] [CrossRef]
- Roberts, A.E.; Araki, T.; Swanson, K.D.; Montgomery, K.T.; A Schiripo, T.; A Joshi, V.; Li, L.; Yassin, Y.; Tamburino, A.M.; Neel, B.G.; et al. Germline gain-of-function mutations in SOS1 cause Noonan syndrome. Nat. Genet. 2006, 39, 70–74. [Google Scholar] [CrossRef]
- Gos, M.; Fahiminiya, S.; Poznański, J.; Klapecki, J.; Obersztyn, E.; Piotrowicz, M.; Wierzba, J.; Posmyk, R.; Bal, J.; Majewski, J. Contribution ofRIT1mutations to the pathogenesis of Noonan syndrome: Four new cases and further evidence of heterogeneity. Am. J. Med. Genet. Part A 2014, 164, 2310–2316. [Google Scholar] [CrossRef]
- Aoki, Y.; Niihori, T.; Banjo, T.; Okamoto, N.; Mizuno, S.; Kurosawa, K.; Ogata, T.; Takada, F.; Yano, M.; Ando, T.; et al. Gain-of-Function Mutations in RIT1 Cause Noonan Syndrome, a RAS/MAPK Pathway Syndrome. Am. J. Hum. Genet. 2013, 93, 173–180. [Google Scholar] [CrossRef]
- Castellano, E.; Santos, E. Functional Specificity of Ras Isoforms: So Similar but So Different. Genes Cancer 2011, 2, 216–231. [Google Scholar] [CrossRef] [PubMed]
- Niihori, T.; Aoki, Y.; Narumi, Y.; Neri, G.; Cavé, H.; Verloes, A.; Okamoto, N.; Hennekam, R.C.M.; Gillessen-Kaesbach, G.; Wieczorek, D.; et al. Germline KRAS and BRAF mutations in cardio-facio-cutaneous syndrome. Nat. Genet. 2006, 38, 294–296. [Google Scholar] [CrossRef] [PubMed]
- Carta, C.; Pantaleoni, F.; Bocchinfuso, G.; Stella, L.; Vasta, I.; Sarkozy, A.; Digilio, C.; Palleschi, A.; Pizzuti, A.; Grammatico, P.; et al. Germline Missense Mutations Affecting KRAS Isoform B Are Associated with a Severe Noonan Syndrome Phenotype. Am. J. Hum. Genet. 2006, 79, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Cirstea, I.C.; Kutsche, K.; Dvorsky, R.; Gremer, L.; Carta, C.; Horn, D.; E Roberts, A.; Lepri, F.R.; Merbitz-Zahradnik, T.; König, R.; et al. A restricted spectrum of NRAS mutations causes Noonan syndrome. Nat. Genet. 2009, 42, 27–29. [Google Scholar] [CrossRef]
- Gremer, L.; De Luca, A.; Merbitz-Zahradnik, T.; Dallapiccola, B.; Morlot, S.; Tartaglia, M.; Kutsche, K.; Ahmadian, M.R.; Rosenberger, G. Duplication of Glu37 in the switch I region of HRAS impairs effector/GAP binding and underlies Costello syndrome by promoting enhanced growth factor-dependent MAPK and AKT activation. Hum. Mol. Genet. 2009, 19, 790–802. [Google Scholar] [CrossRef]
- Aoki, Y.; Niihori, T.; Kawame, H.; Kurosawa, K.; Ohashi, H.; Tanaka, Y.; Filocamo, M.; Kato, K.; Suzuki, Y.; Kure, S.; et al. Germline mutations in HRAS proto-oncogene cause Costello syndrome. Nat. Genet. 2005, 37, 1038–1040. [Google Scholar] [CrossRef]
- Sarkozy, A.; Carta, C.; Moretti, S.; Zampino, G.; Digilio, M.C.; Pantaleoni, F.; Scioletti, A.P.; Esposito, G.; Cordeddu, V.; Lepri, F.; et al. GermlineBRAFmutations in Noonan, LEOPARD, and cardiofaciocutaneous syndromes: Molecular diversity and associated phenotypic spectrum. Hum. Mutat. 2009, 30, 695–702. [Google Scholar] [CrossRef]
- Ma, Y.; Xia, Z.; Ye, C.; Lu, C.; Zhou, S.; Pan, J.; Liu, C.; Zhang, J.; Liu, T.; Hu, T.; et al. AGTR1 promotes lymph node metastasis in breast cancer by upregulating CXCR4/SDF-1α and inducing cell migration and invasion. Aging 2019, 11, 3969–3992. [Google Scholar] [CrossRef]
- Basile, J.R.; Gavard, J.; Gutkind, J.S. Plexin-B1 Utilizes RhoA and Rho Kinase to Promote the Integrin-dependent Activation of Akt and ERK and Endothelial Cell Motility. J. Biol. Chem. 2007, 282, 34888–34895. [Google Scholar] [CrossRef]
- Krishnan, H.; Miller, W.T.; Blanco, F.J.; Goldberg, G.S. Src and podoplanin forge a path to destruction. Drug Discov. Today 2018, 24, 241–249. [Google Scholar] [CrossRef]
- Geng, X.; Yanagida, K.; Akwii, R.G.; Choi, D.; Chen, L.; Ho, Y.; Cha, B.; Mahamud, R.; de Ruiz, K.B.; Ichise, H.; et al. S1PR1 regulates the quiescence of lymphatic vessels by inhibiting laminar shear stress–dependent VEGF-C signaling. JCI Insight 2020, 5, e137652. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.-C.; Yang, J.; Liu, D.; Wu, M.-F.; Qiao, L.; Wang, J.-N.; Ma, Q.-F.; Zeng, Z.; Ye, S.-M.; Guo, E.-S.; et al. Tumor-associated Lymphatic Endothelial Cells Promote Lymphatic Metastasis by Highly Expressing and Secreting SEMA4C. Clin. Cancer Res. 2017, 23, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Valtcheva, N.; Primorac, A.; Jurisic, G.; Hollmén, M.; Detmar, M. The Orphan Adhesion G Protein-coupled Receptor GPR97 Regulates Migration of Lymphatic Endothelial Cells via the Small GTPases RhoA and Cdc42. J. Biol. Chem. 2013, 288, 35736–35748. [Google Scholar] [CrossRef] [PubMed]
- Basile, J.R.; Barac, A.; Zhu, T.; Guan, K.-L.; Gutkind, J.S. Class IV Semaphorins Promote Angiogenesis by Stimulating Rho-Initiated Pathways through Plexin-B. Cancer Res. 2004, 64, 5212–5224. [Google Scholar] [CrossRef]
- Cheong, S.-S.; Akram, K.M.; Matellan, C.; Kim, S.Y.; Gaboriau, D.C.A.; Hind, M.; Hernández, A.E.D.R.; Griffiths, M.; Dean, C.H. The Planar Polarity Component VANGL2 Is a Key Regulator of Mechanosignaling. Front. Cell Dev. Biol. 2020, 8, 577201. [Google Scholar] [CrossRef]
- Hosaka, K.; Mizuno, R.; Ohhashi, T. Rho-Rho kinase pathway is involved in the regulation of myogenic tone and pump activity in isolated lymph vessels. Am. J. Physiol. Circ. Physiol. 2003, 284, H2015–H2025. [Google Scholar] [CrossRef][Green Version]
- Maltese, P.E.; Michelini, S.; Ricci, M.; Maitz, S.; Fiorentino, A.; Serrani, R.; Lazzerotti, A.; Bruson, A.; Paolacci, S.; Benedetti, S.; et al. Increasing evidence of hereditary lymphedema caused byCELSR1loss-of-function variants. Am. J. Med. Genet. Part A 2019, 179, 1718–1724. [Google Scholar] [CrossRef]
- Wünnemann, F.; Kokta, V.; Leclerc, S.; Thibeault, M.; McCuaig, C.; Hatami, A.; Stheneur, C.; Grenier, J.-C.; Awadalla, P.; Mitchell, G.A.; et al. Aortic Dilatation Associated with a de novo Mutation in the SOX18 Gene: Expanding the Clinical Spectrum of Hypotrichosis-Lymphedema-Telangiectasia Syndrome. Can. J. Cardiol. 2016, 32, 135.e1–135.e7. [Google Scholar] [CrossRef]
- Moalem, S.; Brouillard, P.; Kuypers, D.; Legius, E.; Harvey, E.; Taylor, G.; Francois, M.; Vikkula, M.; Chitayat, D. Hypotrichosis-lymphedema-telangiectasia-renal defect associated with a truncating mutation in the SOX18 gene. Clin. Genet. 2014, 87, 378–382. [Google Scholar] [CrossRef]
- Irrthum, A.; Devriendt, K.; Chitayat, D.; Matthijs, G.; Glade, C.; Steijlen, P.M.; Fryns, J.-P.; Van Steensel, M.A.M.; Vikkula, M. Mutations in the Transcription Factor Gene SOX18 Underlie Recessive and Dominant Forms of Hypotrichosis-Lymphedema-Telangiectasia. Am. J. Hum. Genet. 2003, 72, 1470–1478. [Google Scholar] [CrossRef]
- Ostergaard, P.; Simpson, M.; Connell, F.C.; Steward, C.; Brice, G.; Woollard, W.J.; Dafou, D.; Kilo, T.; Smithson, S.; Lunt, P.; et al. Mutations in GATA2 cause primary lymphedema associated with a predisposition to acute myeloid leukemia (Emberger syndrome). Nat. Genet. 2011, 43, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Tavian, D.; Missaglia, S.; Maltese, P.E.; Michelini, S.; Fiorentino, A.; Ricci, M.; Serrani, R.; Walter, M.A.; Bertelli, M. FOXC2 disease-mutations identified in lymphedema-distichiasis patients cause both loss and gain of protein function. Oncotarget 2016, 7, 54228–54239. [Google Scholar] [CrossRef] [PubMed]
- Van Steensel, M.; Damstra, R.; Heitink, M.; Bladergroen, R.; Veraart, J.; Steijlen, P.M.; van Geel, M. Novel missense mutations in theFOXC2gene alter transcriptional activity. Hum. Mutat. 2009, 30, E1002–E1009. [Google Scholar] [CrossRef]
- Ricci, M.; Amato, B.; Barati, S.; Compagna, R.; Veselenyiova, D.; Kenanoglu, S.; Stuppia, L.; Beccari, T.; Baglivo, M.; Kurti, D.; et al. Two rare PROX1 variants in patients with lymphedema. Mol. Genet. Genom. Med. 2020, 8, e1424. [Google Scholar] [CrossRef]
- Missaglia, S.; Tavian, D.; Michelini, S.; Maltese, P.; Bonanomi, A.; Bertelli, M. Imbalance between Expression of FOXC2 and Its lncRNA in Lymphedema-Distichiasis Caused by Frameshift Mutations. Genes 2021, 12, 650. [Google Scholar] [CrossRef]
- Tavian, D.; Missaglia, S.; Michelini, S.; Maltese, P.E.; Manara, E.; Mordente, A.; Bertelli, M. FOXC2 Disease Mutations Identified in Lymphedema Distichiasis Patients Impair Transcriptional Activity and Cell Proliferation. Int. J. Mol. Sci. 2020, 21, 5112. [Google Scholar] [CrossRef]
- Lee, Y.J. Cell Fate Determination of Lymphatic Endothelial Cells. Int. J. Mol. Sci. 2020, 21, 4790. [Google Scholar] [CrossRef]
- Liebl, J.; Zhang, S.; Moser, M.; Agalarov, Y.; Demir, C.S.; Hager, B.; Bibb, J.A.; Adams, R.H.; Kiefer, F.; Miura, N.; et al. Cdk5 controls lymphatic vessel development and function by phosphorylation of Foxc2. Nat. Commun. 2015, 6, 7274. [Google Scholar] [CrossRef]
- Yang, Y.; Cha, B.; Motawe, Z.Y.; Srinivasan, R.S.; Scallan, J.P. VE-Cadherin Is Required for Lymphatic Valve Formation and Maintenance. Cell Rep. 2019, 28, 2397–2412.e4. [Google Scholar] [CrossRef]
- Cha, B.; Srinivasan, R.S. Mechanosensitive β-catenin signaling regulates lymphatic vascular development. BMB Rep. 2016, 49, 403–404. [Google Scholar] [CrossRef]
- Hosking, B.M.; Wang, S.-C.M.; Downes, M.; Koopman, P.; Muscat, G.E.O. The VCAM-1 Gene That Encodes the Vascular Cell Adhesion Molecule Is a Target of the Sry-related High Mobility Group Box Gene, Sox18. J. Biol. Chem. 2004, 279, 5314–5322. [Google Scholar] [CrossRef] [PubMed]
- Derynck, R.; Akhurst, R.J. BMP-9 balances endothelial cell fate. Proc. Natl. Acad. Sci. USA 2013, 110, 18746–18747. [Google Scholar] [CrossRef] [PubMed]
- Michelini, S.; Ricci, M.; Serrani, R.; Stuppia, L.; Beccari, T.; Vešelényiová, D.; Kenanoglu, S.; Barati, S.; Kurti, D.; Baglivo, M.; et al. Possible Role of the RORC Gene in Primary and Secondary Lymphedema: Review of the Literature and Genetic Study of Two Rare Causative Variants. Lymphat. Res. Biol. 2021, 19, 129–133. [Google Scholar] [CrossRef]
- Knirsh, R.; Ben Dror, I.; Modai, S.; Shomron, N.; Vardimon, L. MicroRNA 10b promotes abnormal expression of the proto-oncogene c-Jun in metastatic breast cancer cells. Oncotarget 2016, 7, 59932–59944. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shou, J.; Jing, J.; Xie, J.; You, L.; Jing, Z.; Yao, J.; Han, W.; Pan, H. Nuclear factor of activated T cells in cancer development and treatment. Cancer Lett. 2015, 361, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Castorena-Gonzalez, J.A.; Zawieja, S.D.; Li, M.; Srinivasan, R.S.; Simon, A.M.; De Wit, C.; De La Torre, R.; Martinez-Lemus, L.A.; Hennig, G.W.; Davis, M.J. Mechanisms of Connexin-Related Lymphedema. Circ. Res. 2018, 123, 964–985. [Google Scholar] [CrossRef] [PubMed]
- Erickson, R.P.; Lai, L.; Mustacich, D.J.; Bernas, M.J.; Kuo, P.H.; Witte, M.H. Sex-limited penetrance of lymphedema to females with CELSR1 haploinsufficiency: A second family. Clin. Genet. 2019, 96, 478–482. [Google Scholar] [CrossRef]
- Gonzalez-Garay, M.; Aldrich, M.B.; Rasmussen, J.; Guilliod, R.; Lapinski, P.E.; King, P.D.; Sevick-Muraca, E.M. A novel mutation in CELSR1 is associated with hereditary lymphedema. Vasc. Cell 2016, 8, 1. [Google Scholar] [CrossRef]
- Alders, M.; Al-Gazali, L.; Cordeiro, I.; Dallapiccola, B.; Garavelli, L.; Tüysüz, B.; Salehi, F.; Haagmans, M.A.; Mook, O.R.; Majoie, C.B.; et al. Hennekam syndrome can be caused by FAT4 mutations and be allelic to Van Maldergem syndrome. Qual. Life Res. 2014, 133, 1161–1167. [Google Scholar] [CrossRef]
- Pujol, F.; Hodgson, T.; Martinez-Corral, I.; Prats, A.-C.; Devenport, D.; Takeichi, M.; Genot, E.; Mäkinen, T.; Francis-West, P.; Garmy-Susini, B.; et al. Dachsous1–Fat4 Signaling Controls Endothelial Cell Polarization During Lymphatic Valve Morphogenesis—Brief Report. Arter. Thromb. Vasc. Biol. 2017, 37, 1732–1735. [Google Scholar] [CrossRef]
- Brice, G.; Ostergaard, P.; Jeffery, S.; Gordon, K.; Mortimer, P.; Mansour, S. A novel mutation inGJA1causing oculodentodigital syndrome and primary lymphoedema in a three generation family. Clin. Genet. 2013, 84, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Ferrell, R.E.; Baty, C.J.; Kimak, M.A.; Karlsson, J.M.; Lawrence, E.C.; Franke-Snyder, M.; Meriney, S.D.; Feingold, E.; Finegold, D.N. GJC2 Missense Mutations Cause Human Lymphedema. Am. J. Hum. Genet. 2010, 86, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Paznekas, W.A.; Boyadjiev, S.A.; Shapiro, R.E.; Daniels, O.; Wollnik, B.; Keegan, C.E.; Innis, J.W.; Dinulos, M.B.; Christian, C.; Hannibal, M.C.; et al. Connexin 43 (GJA1) Mutations Cause the Pleiotropic Phenotype of Oculodentodigital Dysplasia. Am. J. Hum. Genet. 2003, 72, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Nonomura, K.; Lukacs, V.; Sweet, D.T.; Goddard, L.M.; Kanie, A.; Whitwam, T.; Ranade, S.S.; Fujimori, T.; Kahn, M.L.; Patapoutian, A. Mechanically activated ion channel PIEZO1 is required for lymphatic valve formation. Proc. Natl. Acad. Sci. USA 2018, 115, 12817–12822. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, L.A.; Gordon, K.; Kholová, I.; Meijer-Jorna, L.B.; Telinius, N.; Gallagher, P.J.; Van Der Wal, A.C.; Baandrup, U. Lymph vessels: The forgotten second circulation in health and disease. Virchows Arch. 2016, 469, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Tatin, F.; Taddei, A.; Weston, A.; Fuchs, E.; Devenport, D.; Tissir, F.; Makinen, T. Planar Cell Polarity Protein Celsr1 Regulates Endothelial Adherens Junctions and Directed Cell Rearrangements during Valve Morphogenesis. Dev. Cell 2013, 26, 31–44. [Google Scholar] [CrossRef]
- Sabine, A.; Bovay, E.; Demir, C.S.; Kimura, W.; Jaquet, M.; Agalarov, Y.; Zangger, N.; Scallan, J.P.; Graber, W.; Gulpinar, E.; et al. FOXC2 and fluid shear stress stabilize postnatal lymphatic vasculature. J. Clin. Investig. 2015, 125, 3861–3877. [Google Scholar] [CrossRef]
- Betterman, K.L.; Sutton, D.L.; Secker, G.A.; Kazenwadel, J.; Oszmiana, A.; Lim, L.; Miura, N.; Sorokin, L.; Hogan, B.M.; Kahn, M.L.; et al. Atypical cadherin FAT4 orchestrates lymphatic endothelial cell polarity in response to flow. J. Clin. Investig. 2020, 130, 3315–3328. [Google Scholar] [CrossRef]
- Conway, D.E.; Breckenridge, M.T.; Hinde, E.; Gratton, E.; Chen, C.S.; Schwartz, M.A. Fluid Shear Stress on Endothelial Cells Modulates Mechanical Tension across VE-Cadherin and PECAM-1. Curr. Biol. 2013, 23, 1024–1030. [Google Scholar] [CrossRef]
- Bálint, L.; Jakus, Z. Mechanosensation and Mechanotransduction by Lymphatic Endothelial Cells Act as Important Regulators of Lymphatic Development and Function. Int. J. Mol. Sci. 2021, 22, 3955. [Google Scholar] [CrossRef]
- Bowles, J.; Secker, G.; Nguyen, C.; Kazenwadel, J.; Truong, V.; Frampton, E.; Curtis, C.; Skoczylas, R.; Davidson, T.-L.; Miura, N.; et al. Control of retinoid levels by CYP26B1 is important for lymphatic vascular development in the mouse embryo. Dev. Biol. 2014, 386, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Ricci, M.; Serrani, R.; Amato, B.; Compagna, R.; Veselenyiova, D.; Kenanoglu, S.; Kurti, D.; Baglivo, M.; Krajcovic, J.; Miggiano, G.; et al. CYP26B1 and its implications in lymphangiogenesis: Literature review and study of rare variants in two families. Lymphology 2020, 53, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Von Der Weid, P.-Y. Review article: Lymphatic vessel pumping and inflammation-the role of spontaneous constrictions and underlying electrical pacemaker potentials. Aliment. Pharmacol. Ther. 2001, 15, 1115–1129. [Google Scholar] [CrossRef] [PubMed]
- Michelini, S.; Amato, B.; Ricci, M.; Serrani, R.; Veselenyiova, D.; Kenanoglu, S.; Kurti, D.; Dautaj, A.; Baglivo, M.; Compagna, R.; et al. SVEP1 Is Important for Morphogenesis of Lymphatic System: Possible Implications in Lymphedema. Lymphology 2021, 54, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Danuser, R.; Stein, J.V.; Graham, D.; Nibbs, R.J.; Graham, G.J. The chemokine receptors ACKR 2 and CCR 2 reciprocally regulate lymphatic vessel density. EMBO J. 2014, 33, 2564–2580. [Google Scholar] [CrossRef] [PubMed]
- Wagner, M.; Steinskog, E.S.; Wiig, H. Blockade of Lymphangiogenesis Shapes Tumor-Promoting Adipose Tissue Inflammation. Am. J. Pathol. 2019, 189, 2102–2114. [Google Scholar] [CrossRef]
- Lvovs, D.; Favorova, O.O.; Favorov, A.V. A Polygenic Approach to the Study of Polygenic Diseases. Acta Nat. 2012, 4, 59–71. [Google Scholar] [CrossRef]
- Meghnani, V.; Mohammed, N.; Giauque, C.; Nahire, R.; David, T. Performance Characterization and Validation of Saliva as an Alternative Specimen Source for Detecting Hereditary Breast Cancer Mutations by Next Generation Sequencing. J. Genom. 2016, 2016, 2059041. [Google Scholar] [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–423. [Google Scholar] [CrossRef]
- OMIM® Online Mendelian Inheritance in Man®. Available online: https://www.omim.org/ (accessed on 1 December 2021).
- KEGG: Kyoto Encyclopedia of Genes and Genomes. Available online: https://www.genome.jp/kegg/ (accessed on 1 December 2021).
- Zhu, M.; Zhao, S. Candidate Gene Identification Approach: Progress and Challenges. Int. J. Biol. Sci. 2007, 3, 420–427. [Google Scholar] [CrossRef]
- PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 1 December 2021).
- Scopus. Available online: https://www.scopus.com/home.uri (accessed on 1 December 2021).
- PathVisio Biological Pathway Editor. Available online: https://pathvisio.org/ (accessed on 1 December 2021).
- Petrelli, A.; Gilestro, G.F.; Lanzardo, S.; Comoglio, P.; Migone, N.; Giordano, S. The endophilin–CIN85–Cbl complex mediates ligand-dependent downregulation of c-Met. Nature 2002, 416, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Robinson, A.; Escuin, S.; Doudney, K.; Vekemans, M.; Stevenson, R.E.; Greene, N.D.; Copp, A.J.; Stanier, P. Mutations in the planar cell polarity genesCELSR1andSCRIBare associated with the severe neural tube defect craniorachischisis. Hum. Mutat. 2011, 33, 440–447. [Google Scholar] [CrossRef]
- Amyere, M.; Revencu, N.; Helaers, R.; Pairet, E.; Baselga, E.; Cordisco, M.; Chung, W.; Dubois, J.; Lacour, J.-P.; Martorell, L.; et al. Germline Loss-of-Function Mutations in EPHB4 Cause a Second Form of Capillary Malformation-Arteriovenous Malformation (CM-AVM2) Deregulating RAS-MAPK Signaling. Circulation 2017, 136, 1037–1048. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Rui, B.; Qingquan, F.; Chen, C.; Ping, H.Y.; Xiaoling, S.; Hao, W.; Jun, G. KIF11 promotes cell proliferation via ERBB2/PI3K/AKT signaling pathway in gallbladder cancer. Int. J. Biol. Sci. 2021, 17, 514–526. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Zhang, G.; Jiang, S.; Ning, Y.; Deng, B.; Pan, X.; Liu, S.; He, Y.; Zhang, L.; Wan, R.; et al. Mechanosensitive Piezo1 in endothelial cells promotes angiogenesis to support bone fracture repair. Cell Calcium 2021, 97, 102431. [Google Scholar] [CrossRef]
- Idrees, M.; Xu, L.; Song, S.-H.; Joo, M.-D.; Lee, K.-L.; Muhammad, T.; El Sheikh, M.; Sidrat, T.; Kong, I.-K. PTPN11 (SHP2) Is Indispensable for Growth Factors and Cytokine Signal Transduction during Bovine Oocyte Maturation and Blastocyst Development. Cells 2019, 8, 1272. [Google Scholar] [CrossRef]
- Paolacci, S.; Rakhmanov, Y.; Maltese, P.E.; Zulian, A.; Michelini, S.; Bertelli, M. Genetic testing for lymphatic malformations with or without primary lymphedema. EuroBiotech J. 2018, 2, 5–9. [Google Scholar] [CrossRef]
- Fritz-Six, K.L.; Dunworth, W.P.; Li, M.; Caron, K.M. Adrenomedullin signaling is necessary for murine lymphatic vascular development. J. Clin. Investig. 2008, 118, 40–50. [Google Scholar] [CrossRef]
- Wong, H.K.; Cheung, T.T.; Cheung, B.M.Y. Adrenomedullin and cardiovascular diseases. JRSM Cardiovasc. Dis. 2012, 1, 1–7. [Google Scholar] [CrossRef]
- Nikitenko, L.L.; Shimosawa, T.; Henderson, S.; Mäkinen, T.; Shimosawa, H.; Qureshi, U.; Pedley, R.B.; Rees, M.C.; Fujita, T.; Boshoff, C. Adrenomedullin Haploinsufficiency Predisposes to Secondary Lymphedema. J. Investig. Dermatol. 2013, 133, 1768–1776. [Google Scholar] [CrossRef]
- Bäckhed, F.; Crawford, P.A.; O’Donnell, D.; Gordon, J.I. Postnatal lymphatic partitioning from the blood vasculature in the small intestine requires fasting-induced adipose factor. Proc. Natl. Acad. Sci. USA 2007, 104, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Sodhi, A.; Ma, T.; Menon, D.; Deshpande, M.; Jee, K.; Dinabandhu, A.; Vancel, J.; Lu, D.; Montaner, S. Angiopoietin-like 4 binds neuropilins and cooperates with VEGF to induce diabetic macular edema. J. Clin. Investig. 2019, 129, 4593–4608. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Son, M.K.; Yan, H.H.; Fang, Z.; Kim, J.; Kim, S.J.; Park, J.H.; Lee, J.E.; Yoon, Y.; Seo, M.S.; et al. ANGPTL 4 exacerbates pancreatitis by augmenting acinar cell injury through upregulation of C5a. EMBO Mol. Med. 2020, 12, e11222. [Google Scholar] [CrossRef] [PubMed]
- Kartopawiro, J.; Bower, N.I.; Karnezis, T.; Kazenwadel, J.; Betterman, K.L.; Lesieur, E.; Koltowska, K.; Astin, J.; Crosier, P.; Vermeren, S.; et al. Arap3 is dysregulated in a mouse model of hypotrichosis–lymphedema–telangiectasia and regulates lymphatic vascular development. Hum. Mol. Genet. 2013, 23, 1286–1297. [Google Scholar] [CrossRef]
- Krugmann, S.; Anderson, K.; Ridley, S.; Risso, N.; McGregor, A.; Coadwell, J.; Davidson, K.; Eguinoa, A.; Ellson, C.; Lipp, P.; et al. Identification of ARAP3, a Novel PI3K Effector Regulating Both Arf and Rho GTPases, by Selective Capture on Phosphoinositide Affinity Matrices. Mol. Cell 2002, 9, 95–108. [Google Scholar] [CrossRef]
- Hirashima, M.; Sano, K.; Morisada, T.; Murakami, K.; Rossant, J.; Suda, T. Lymphatic vessel assembly is impaired in Aspp1-deficient mouse embryos. Dev. Biol. 2008, 316, 149–159. [Google Scholar] [CrossRef]
- Trigiante, G.; Lu, X. ASPPs and cancer. Nat. Cancer 2006, 6, 217–226. [Google Scholar] [CrossRef]
- Michelini, S.; Ricci, M.; Amato, B.; Gentileschi, S.; Veselenyiova, D.; Kenanoglu, S.; Fiorentino, A.; Kurti, D.; Baglivo, M.; Manara, E.; et al. CDH5, a Possible New Candidate Gene for Genetic Testing of Lymphedema. In Lymphatic Research and Biology; Mary Ann Liebert, Inc.: Larchmont, NY, USA, 2021. [Google Scholar] [CrossRef]
- Hägerling, R.; Hoppe, E.; Dierkes, C.; Stehling, M.; Makinen, T.; Butz, S.; Vestweber, D.; Kiefer, F. Distinct roles of VE -cadherin for development and maintenance of specific lymph vessel beds. EMBO J. 2018, 37, e98271. [Google Scholar] [CrossRef]
- Ozeki, M.; Fukao, T. Generalized Lymphatic Anomaly and Gorham–Stout Disease: Overview and Recent Insights. Adv. Wound Care 2019, 8, 230–245. [Google Scholar] [CrossRef]
- Danussi, C.; Belluz, L.D.B.; Pivetta, E.; Modica, T.M.E.; Muro, A.; Wassermann, B.; Doliana, R.; Sabatelli, P.; Colombatti, A.; Spessotto, P. EMILIN1/α9β1 Integrin Interaction Is Crucial in Lymphatic Valve Formation and Maintenance. Mol. Cell. Biol. 2013, 33, 4381–4394. [Google Scholar] [CrossRef]
- Ferrell, R.E.; Kimak, M.A.; Lawrence, E.C.; Finegold, D.N. Candidate Gene Analysis in Primary Lymphedema. Lymphat. Res. Biol. 2008, 6, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y. VEGFR1 for Lymphangiogenesis. Arter. Thromb. Vasc. Biol. 2008, 28, 604–605. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fatima, A.; Wang, Y.; Uchida, Y.; Norden, P.; Liu, T.; Culver, A.; Dietz, W.H.; Culver, F.; Millay, M.; Mukouyama, Y.-S.; et al. Foxc1 and Foxc2 deletion causes abnormal lymphangiogenesis and correlates with ERK hyperactivation. J. Clin. Investig. 2016, 126, 2437–2451. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Bhowmick, N.; Qu, Y.; Chung, S.; E Giuliano, A.; Cui, X. FOXC1: An emerging marker and therapeutic target for cancer. Oncogene 2017, 36, 3957–3963. [Google Scholar] [CrossRef] [PubMed]
- Levet, S.; Ciais, D.; Merdzhanova, G.; Mallet, C.; Zimmers, T.A.; Lee, S.-J.; Navarro, F.; Texier, I.; Feige, J.-J.; Bailly, S.; et al. Bone morphogenetic protein 9 (BMP9) controls lymphatic vessel maturation and valve formation. Blood 2013, 122, 598–607. [Google Scholar] [CrossRef]
- Kanady, J.D.; Dellinger, M.T.; Munger, S.J.; Witte, M.H.; Simon, A.M. Connexin37 and Connexin43 deficiencies in mice disrupt lymphatic valve development and result in lymphatic disorders including lymphedema and chylothorax. Dev. Biol. 2011, 354, 253–266. [Google Scholar] [CrossRef]
- Hadizadeh, M.; Ardebili, S.M.M.; Salehi, M.; Young, C.; Mokarian, F.; McClellan, J.; Xu, Q.; Kazemi, M.; Moazam, E.; Mahaki, B.; et al. GJA4/Connexin 37 Mutations Correlate with Secondary Lymphedema Following Surgery in Breast Cancer Patients. Biomedicines 2018, 6, 23. [Google Scholar] [CrossRef]
- Gauvrit, S.; Villasenor, A.; Strilic, B.; Kitchen, P.; Collins, M.M.; Marín-Juez, R.; Guenther, S.; Maischein, H.-M.; Fukuda, N.; Canham, M.A.; et al. HHEX is a transcriptional regulator of the VEGFC/FLT4/PROX1 signaling axis during vascular development. Nat. Commun. 2018, 9, 2704. [Google Scholar] [CrossRef]
- Klein, S.; Dieterich, L.; Mathelier, A.; Chong, C.; Sliwa-Primorac, A.; Hong, Y.-K.; Shin, J.W.; Lizio, M.; Itoh, M.; Kawaji, H.; et al. DeepCAGE transcriptomics identify HOXD10 as transcription factor regulating lymphatic endothelial responses to VEGF-C. J. Cell Sci. 2016, 129, 2573–2585. [Google Scholar] [CrossRef]
- Tilstra, J.S.; Clauson, C.L.; Niedernhofer, L.J.; Robbins, P.D. NF-ΚB in Aging and Disease. Aging Dis. 2011, 2, 449–465. [Google Scholar]
- Geng, X.; Cha, B.; Mahamud, R.; Lim, K.-C.; Silasi-Mansat, R.; Uddin, M.K.; Miura, N.; Xia, L.; Simon, A.M.; Engel, J.D.; et al. Multiple mouse models of primary lymphedema exhibit distinct defects in lymphovenous valve development. Dev. Biol. 2015, 409, 218–233. [Google Scholar] [CrossRef] [PubMed]
- Bazigou, E.; Xie, S.; Chen, C.; Weston, A.; Miura, N.; Sorokin, L.; Adams, R.; Muro, A.F.; Sheppard, D.; Makinen, T. Integrin-α9 Is Required for Fibronectin Matrix Assembly during Lymphatic Valve Morphogenesis. Dev. Cell 2009, 17, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Visser, J.; Van Geel, M.; Cornelissen, A.J.; Van Der Hulst, R.R.; Qiu, S.S. Breast Cancer-Related Lymphedema and Genetic Predisposition: A Systematic Review of the Literature. Lymphat. Res. Biol. 2019, 17, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Lev, A.; Lee, Y.N.; Sun, G.; Hallumi, E.; Simon, A.J.; Zrihen, K.S.; Levy, S.; Halevi, T.B.; Papazian, M.; Shwartz, N.; et al. Inherited SLP76 deficiency in humans causes severe combined immunodeficiency, neutrophil and platelet defects. J. Exp. Med. 2020, 218, e20201062. [Google Scholar] [CrossRef]
- Meng, G.; Wuest, M.; Tang, X.; Dufour, J.; McMullen, T.P.; Wuest, F.; Murray, D.; Brindley, D.N. Dexamethasone Attenuates X-Ray-Induced Activation of the Autotaxin-Lysophosphatidate-Inflammatory Cycle in Breast Tissue and Subsequent Breast Fibrosis. Cancers 2020, 12, 999. [Google Scholar] [CrossRef]
- Dunworth, W.P.; Caron, K.M. G Protein–Coupled Receptors as Potential Drug Targets for Lymphangiogenesis and Lymphatic Vascular Diseases. Arter. Thromb. Vasc. Biol. 2009, 29, 650–656. [Google Scholar] [CrossRef]
- Xiang, H.; Lu, Y.; Shao, M.; Wu, T. Lysophosphatidic Acid Receptors: Biochemical and Clinical Implications in Different Diseases. J. Cancer 2020, 11, 3519–3535. [Google Scholar] [CrossRef]
- Liu, N.F.; Yu, Z.; Luo, Y.; Sun, D. A LYVE-1/CRSBP-1 Mutation in Inherited Primary Lymphedema. Lymphology 2017, 50, 9–15. [Google Scholar]
- Abe, T.; Umeki, I.; Kanno, S.-I.; Inoue, S.-I.; Niihori, T.; Aoki, Y. LZTR1 facilitates polyubiquitination and degradation of RAS-GTPases. Cell Death Differ. 2019, 27, 1023–1035. [Google Scholar] [CrossRef]
- Flach, R.J.R.; Guo, C.-A.; Danai, L.V.; Yawe, J.C.; Gujja, S.; Edwards, Y.J.K.; Czech, M.P. Endothelial Mitogen-Activated Protein Kinase Kinase Kinase Kinase 4 Is Critical for Lymphatic Vascular Development and Function. Mol. Cell. Biol. 2016, 36, 1740–1749. [Google Scholar] [CrossRef]
- Gao, X.; Chen, G.; Gao, C.; Zhang, D.H.; Kuan, S.-F.; Stabile, L.P.; Liu, G.; Hu, J. MAP4K4 is a novel MAPK/ERK pathway regulator required for lung adenocarcinoma maintenance. Mol. Oncol. 2017, 11, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Munger, S.J.; Geng, X.; Srinivasan, R.S.; Witte, M.H.; Paul, D.L.; Simon, A.M. Segregated Foxc2, NFATc1 and Connexin expression at normal developing venous valves, and Connexin-specific differences in the valve phenotypes of Cx37, Cx43, and Cx47 knockout mice. Dev. Biol. 2016, 412, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Scallan, J.P.; Davis, M.J.; Huxley, V.H. Permeability and contractile responses of collecting lymphatic vessels elicited by atrial and brain natriuretic peptides. J. Physiol. 2013, 591, 5071–5081. [Google Scholar] [CrossRef] [PubMed]
- Michelini, S.; Amato, B.; Ricci, M.; Kenanoglu, S.; Veselenyiova, D.; Kurti, D.; Baglivo, M.; Manara, E.; Dundar, M.; Krajcovic, J.; et al. Segregation Analysis of Rare NRP1 and NRP2 Variants in Families with Lymphedema. Genes 2020, 11, 1361. [Google Scholar] [CrossRef] [PubMed]
- Schacht, V.; Ramirez, M.I.; Hong, Y.-K.; Hirakawa, S.; Feng, D.; Harvey, N.; Williams, M.; Dvorak, A.M.; Dvorak, H.F.; Oliver, G.; et al. T1α/podoplanin deficiency disrupts normal lymphatic vasculature formation and causes lymphedema. EMBO J. 2003, 22, 3546–3556. [Google Scholar] [CrossRef]
- Ichise, H.; Ichise, T.; Ohtani, O.; Yoshida, N. Phospholipase Cγ2 is necessary for separation of blood and lymphatic vasculature in mice. Development 2009, 136, 191–195. [Google Scholar] [CrossRef]
- Bouvrée, K.; Brunet, I.; Del Toro, R.; Gordon, E.; Prahst, C.; Cristofaro, B.; Mathivet, T.; Xu, Y.; Soueid, J.; Fortuna, V.; et al. Semaphorin3A, Neuropilin-1, and PlexinA1 Are Required for Lymphatic Valve Formation. Circ. Res. 2012, 111, 437–445. [Google Scholar] [CrossRef]
- Nakayama, H.; Kusumoto, C.; Nakahara, M.; Fujiwara, A.; Higashiyama, S. Semaphorin 3F and Netrin-1: The Novel Function as a Regulator of Tumor Microenvironment. Front. Physiol. 2018, 9, 1662. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, X.; Wang, J.; Li, Q.; Zhao, Y.; Jin, X. LZTR1: A promising adaptor of the CUL3 family. Oncol. Lett. 2021, 22, 564. [Google Scholar] [CrossRef]
- Wang, P.; Yuan, Y.; Lin, W.; Zhong, H.; Xu, K.; Qi, X. Roles of sphingosine-1-phosphate signaling in cancer. Cancer Cell Int. 2019, 19, 1–12. [Google Scholar] [CrossRef]
- Kimizuka, K.; Kawai, Y.; Maejima, D.; Ajima, K.; Kaidoh, M.; Ohhashi, T. Sphingosine 1-Phosphate (S1P) Induces S1P2 Receptor-Dependent Tonic Contraction in Murine Iliac Lymph Vessels. Microcirculation 2012, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Zhang, H.; Liu, Y.; He, Y.; Yang, C.; Du, Y.; Wu, M.; Zhang, G.; Gao, F. The cooperative role of S1P3 with LYVE-1 in LMW-HA-induced lymphangiogenesis. Exp. Cell Res. 2015, 336, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Secker, G.; Harvey, N. Regulation of VEGFR Signalling in Lymphatic Vascular Development and Disease: An Update. Int. J. Mol. Sci. 2021, 22, 7760. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Baeyens, N.; Corti, F.; Tanaka, K.; Fang, J.; Zhang, J.; Jin, Y.; Coon, B.; Hirschi, K.K.; Schwartz, M.A.; et al. Syndecan-4 controls lymphatic vasculature remodeling during embryonic development. Development 2016, 143, 4441–4451. [Google Scholar] [CrossRef] [PubMed]
- Ricci, M.; Daolio, C.; Amato, B.; Kenanoglu, S.; Veselenyiova, D.; Kurti, D.; Dautaj, A.; Baglivo, M.; Basha, S.; Priya, S.; et al. Review of the Function of Sema3a in Lymphatic Vessel Maturation and Its Potential as a Candidate Gene for Lymphedema: Analysis of Three Families with Rare Causative Variants. Lymphology 2020, 53, 63–75. [Google Scholar] [CrossRef]
- Li, Z.; Hao, J.; Duan, X.; Wu, N.; Zhou, Z.; Yang, F.; Li, J.; Zhao, Z.; Huang, S. The Role of Semaphorin 3A in Bone Remodeling. Front. Cell. Neurosci. 2017, 11, 40. [Google Scholar] [CrossRef]
- Singh, A.P.; Foley, J.; Tandon, A.; Phadke, D.; Kinyamu, H.; Archer, T.K. A role for BRG1 in the regulation of genes required for development of the lymphatic system. Oncotarget 2017, 8, 54925–54938. [Google Scholar] [CrossRef]
- Kriaucionis, S. LKB1 cooperates with Sox17 to drive metastasis. Nat. Cell Biol. 2021, 23, 816–817. [Google Scholar] [CrossRef]
- Hosking, B.; François, M.; Wilhelm, D.; Orsenigo, F.; Caprini, A.; Svingen, T.; Tutt, D.; Davidson, T.; Browne, C.; Dejana, E.; et al. Sox7 and Sox17 are strain-specific modifiers of the lymphangiogenic defects caused by Sox18 dysfunction in mice. Development 2009, 136, 2385–2391. [Google Scholar] [CrossRef]
- Motta, M.; Fasano, G.; Gredy, S.; Brinkmann, J.; Bonnard, A.A.; Simsek-Kiper, P.O.; Gulec, E.Y.; Essaddam, L.; Utine, G.E.; Prandi, I.G.; et al. SPRED2 loss-of-function causes a recessive Noonan syndrome-like phenotype. Am. J. Hum. Genet. 2021, 108, 2112–2129. [Google Scholar] [CrossRef]
- Chen, L.; Liu, D.; Yi, X.; Qi, L.; Tian, X.; Sun, B.; Dong, Q.; Han, Z.; Li, Q.; Song, T.; et al. The novel miR-1269b-regulated protein SVEP1 induces hepatocellular carcinoma proliferation and metastasis likely through the PI3K/Akt pathway. Cell Death Dis. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hasselhof, V.; Sperling, A.; Buttler, K.; Ströbel, P.; Becker, J.; Aung, T.; Felmerer, G.; Wilting, J. Morphological and Molecular Characterization of Human Dermal Lymphatic Collectors. PLoS ONE 2016, 11, e0164964. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Misawa, K.; Misawa, Y.; Uehara, T.; Fukushima, H.; Kusaka, G.; Maruta, M.; Carey, T.E. G-Protein-Coupled Receptors: Next Generation Therapeutic Targets in Head and Neck Cancer? Toxins 2015, 7, 2959–2984. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Bhat, A.; Woodnutt, G.; Lappe, R. Targeting the ANGPT–TIE2 pathway in malignancy. Nat. Cancer 2010, 10, 575–585. [Google Scholar] [CrossRef]

| Characteristic | Case Subjects (n = 147) | |
|---|---|---|
| Age | Mean | 46 ± 18 |
| Median | 47 ± 18 | |
| Females/Males | 116/31 (79%/21%) | |
| Period of onset | Congenital | 7 (5%) |
| Childhood (1–10 years) | 26 (18%) | |
| Youth (11–17 years) | 43 (29%) | |
| Adult (>18 years) | 71 (48%) | |
| Age of onset | Mean | 27 ± 18 |
| Median | 25 ± 18 | |
| Unknown | n = 39 | |
| Sporadic | 79 (54%) | |
| Familiarity | Familiar | 40 (27%) |
| Unknown | 28 (19%) | |
| Location | Lower limb | 49 (33%) |
| Lower limbs | 77 (53%) | |
| Upper limb | 6 (4%) | |
| Lower and upper limbs | 12 (8%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonetti, G.; Paolacci, S.; Samaja, M.; Maltese, P.E.; Michelini, S.; Michelini, S.; Michelini, S.; Ricci, M.; Cestari, M.; Dautaj, A.; et al. Low Efficacy of Genetic Tests for the Diagnosis of Primary Lymphedema Prompts Novel Insights into the Underlying Molecular Pathways. Int. J. Mol. Sci. 2022, 23, 7414. https://doi.org/10.3390/ijms23137414
Bonetti G, Paolacci S, Samaja M, Maltese PE, Michelini S, Michelini S, Michelini S, Ricci M, Cestari M, Dautaj A, et al. Low Efficacy of Genetic Tests for the Diagnosis of Primary Lymphedema Prompts Novel Insights into the Underlying Molecular Pathways. International Journal of Molecular Sciences. 2022; 23(13):7414. https://doi.org/10.3390/ijms23137414
Chicago/Turabian StyleBonetti, Gabriele, Stefano Paolacci, Michele Samaja, Paolo Enrico Maltese, Sandro Michelini, Serena Michelini, Silvia Michelini, Maurizio Ricci, Marina Cestari, Astrit Dautaj, and et al. 2022. "Low Efficacy of Genetic Tests for the Diagnosis of Primary Lymphedema Prompts Novel Insights into the Underlying Molecular Pathways" International Journal of Molecular Sciences 23, no. 13: 7414. https://doi.org/10.3390/ijms23137414
APA StyleBonetti, G., Paolacci, S., Samaja, M., Maltese, P. E., Michelini, S., Michelini, S., Michelini, S., Ricci, M., Cestari, M., Dautaj, A., Medori, M. C., & Bertelli, M. (2022). Low Efficacy of Genetic Tests for the Diagnosis of Primary Lymphedema Prompts Novel Insights into the Underlying Molecular Pathways. International Journal of Molecular Sciences, 23(13), 7414. https://doi.org/10.3390/ijms23137414







