Red Upconverter Nanocrystals Functionalized with Verteporfin for Photodynamic Therapy Triggered by Upconversion
Abstract
:1. Introduction
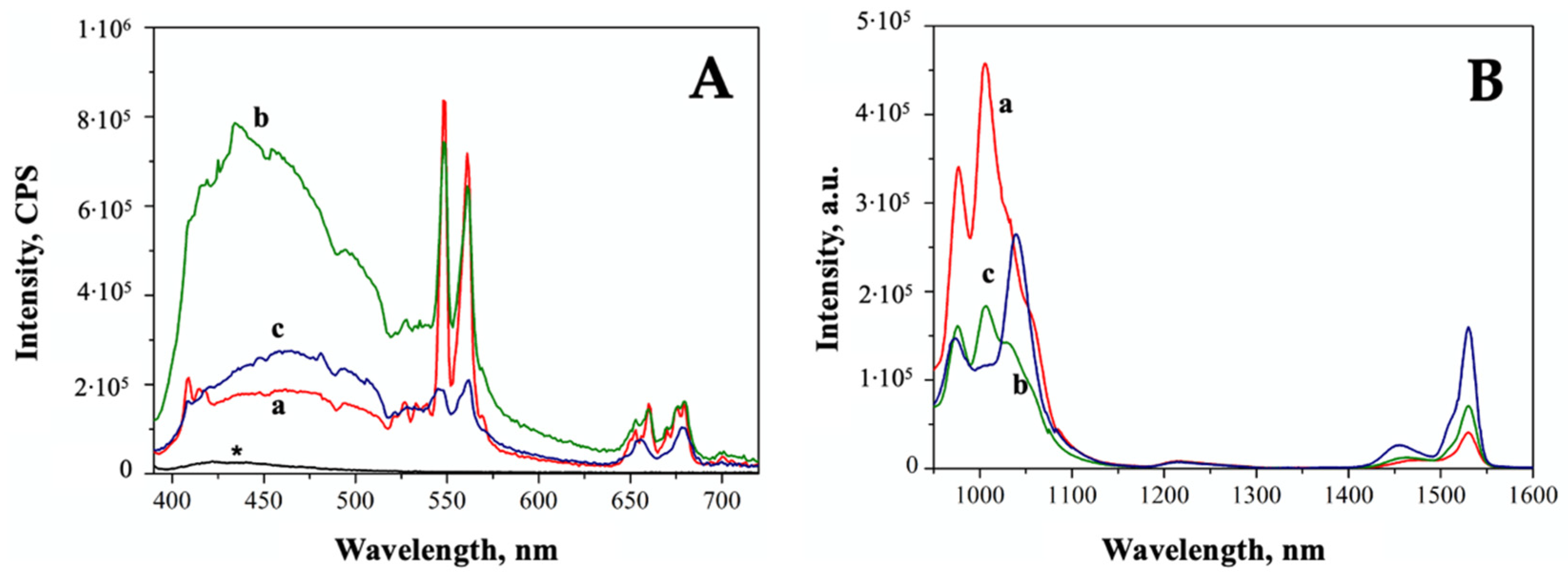
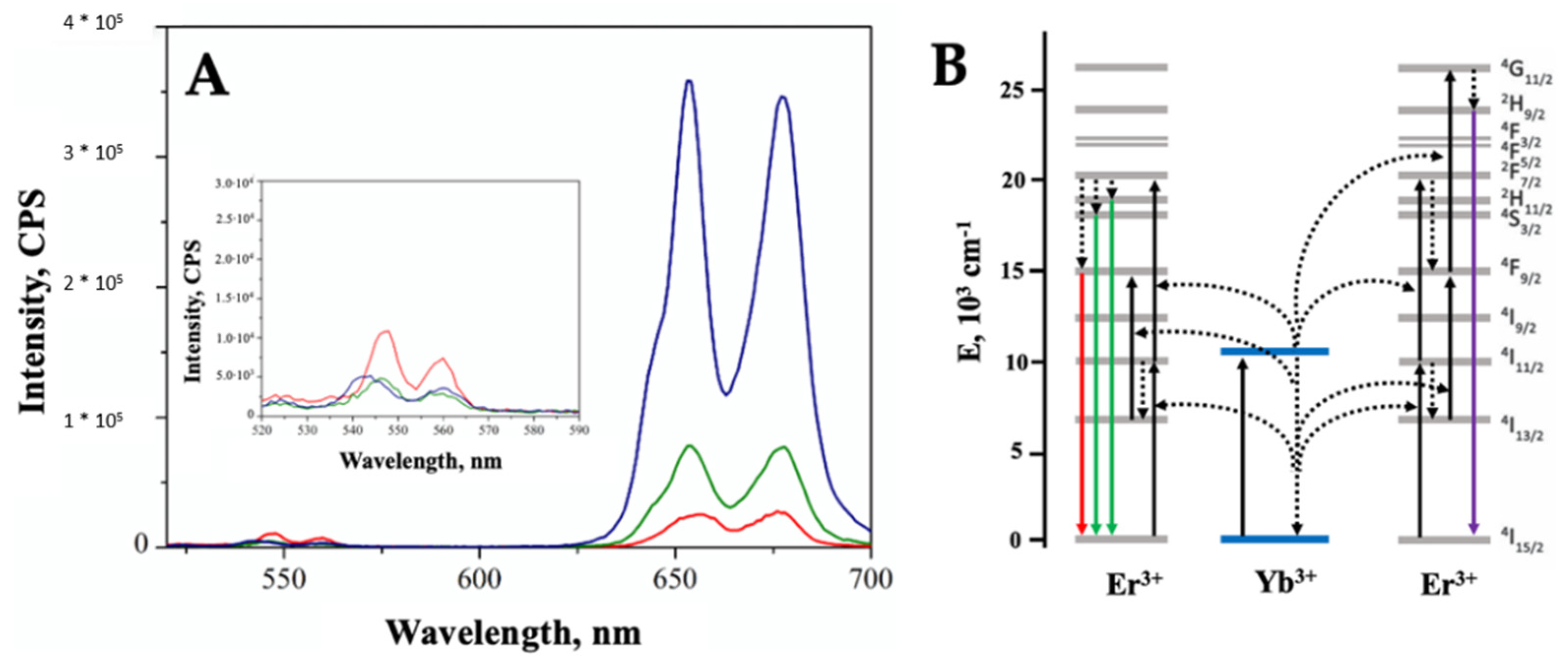
2. Results
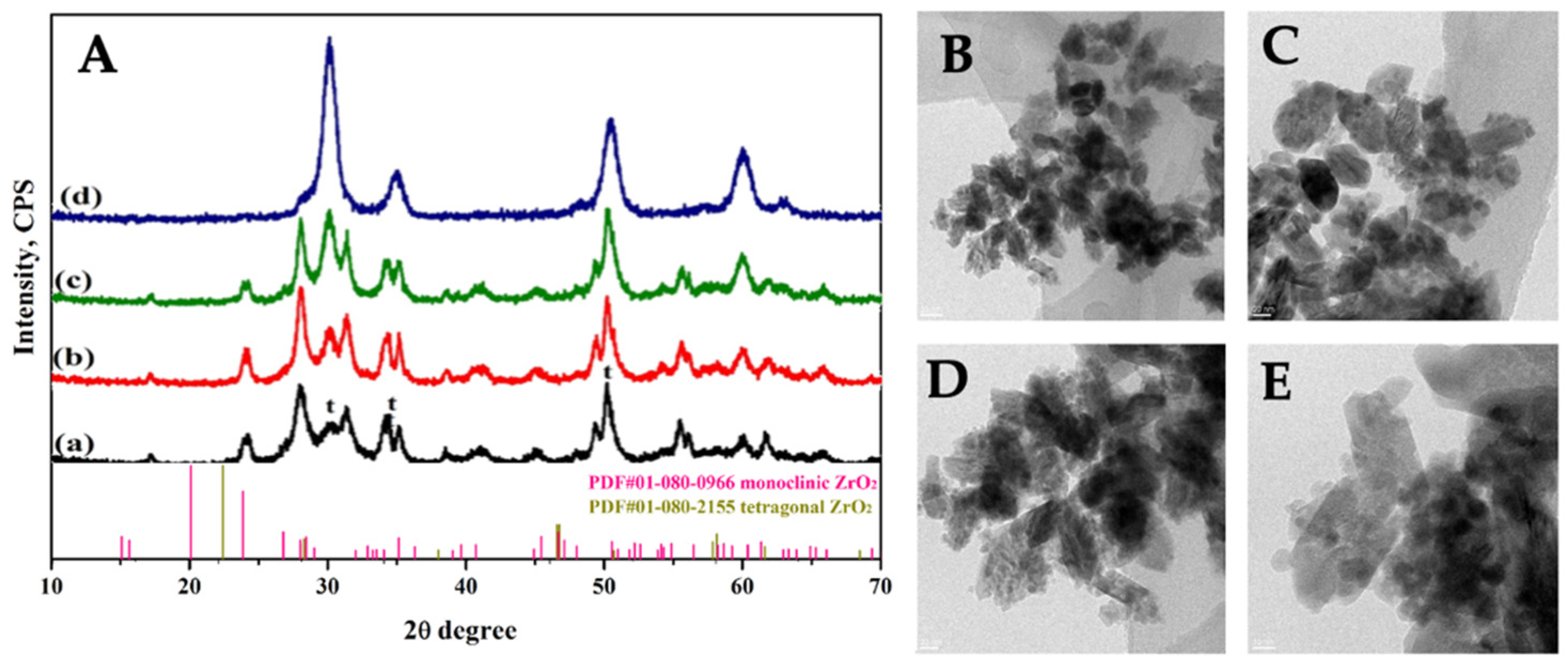
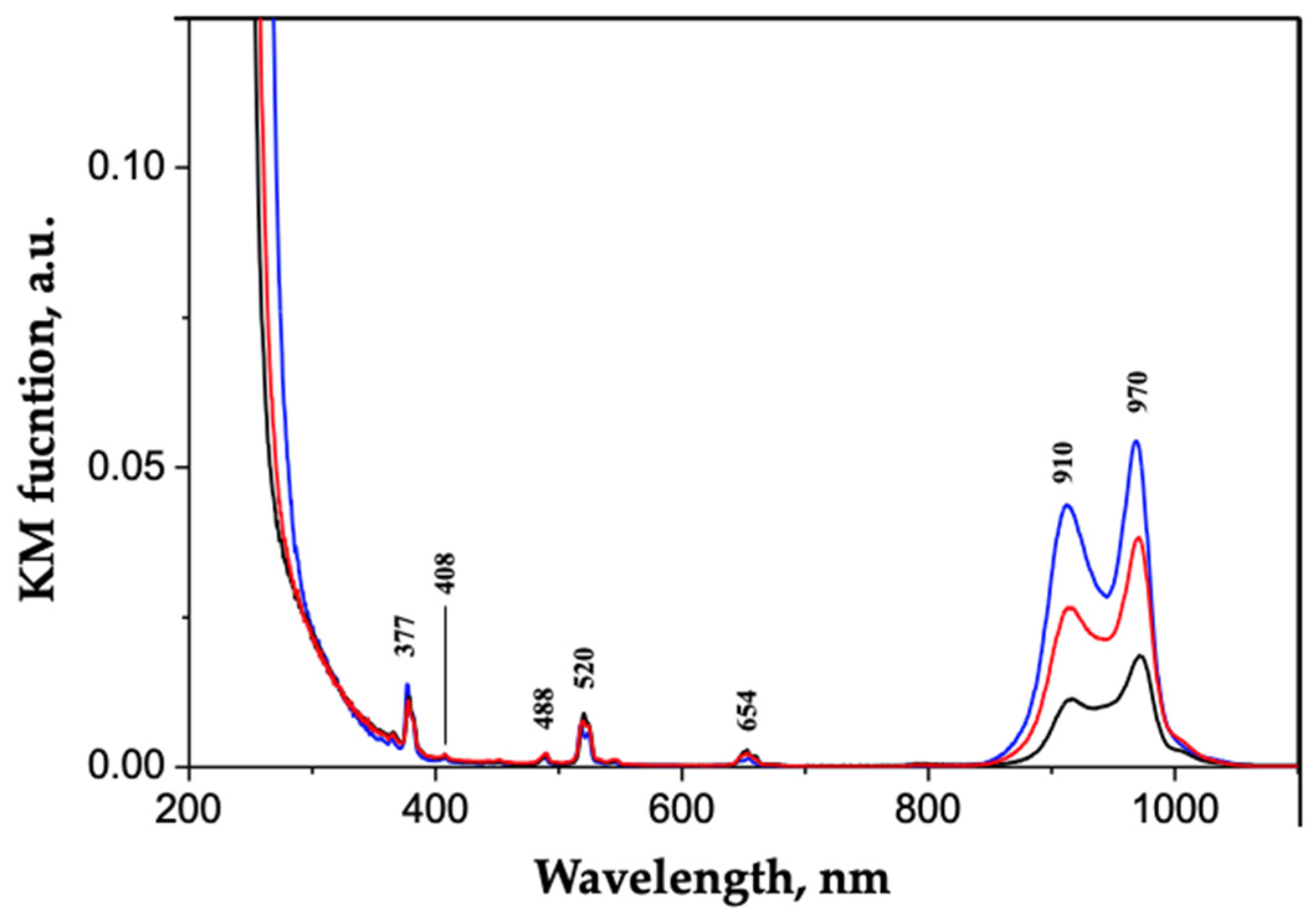
Synthesis and Characterization of Pristine ZrO2 Nanocrystals Co-Doped with Er3+ and Yb3+ Ions
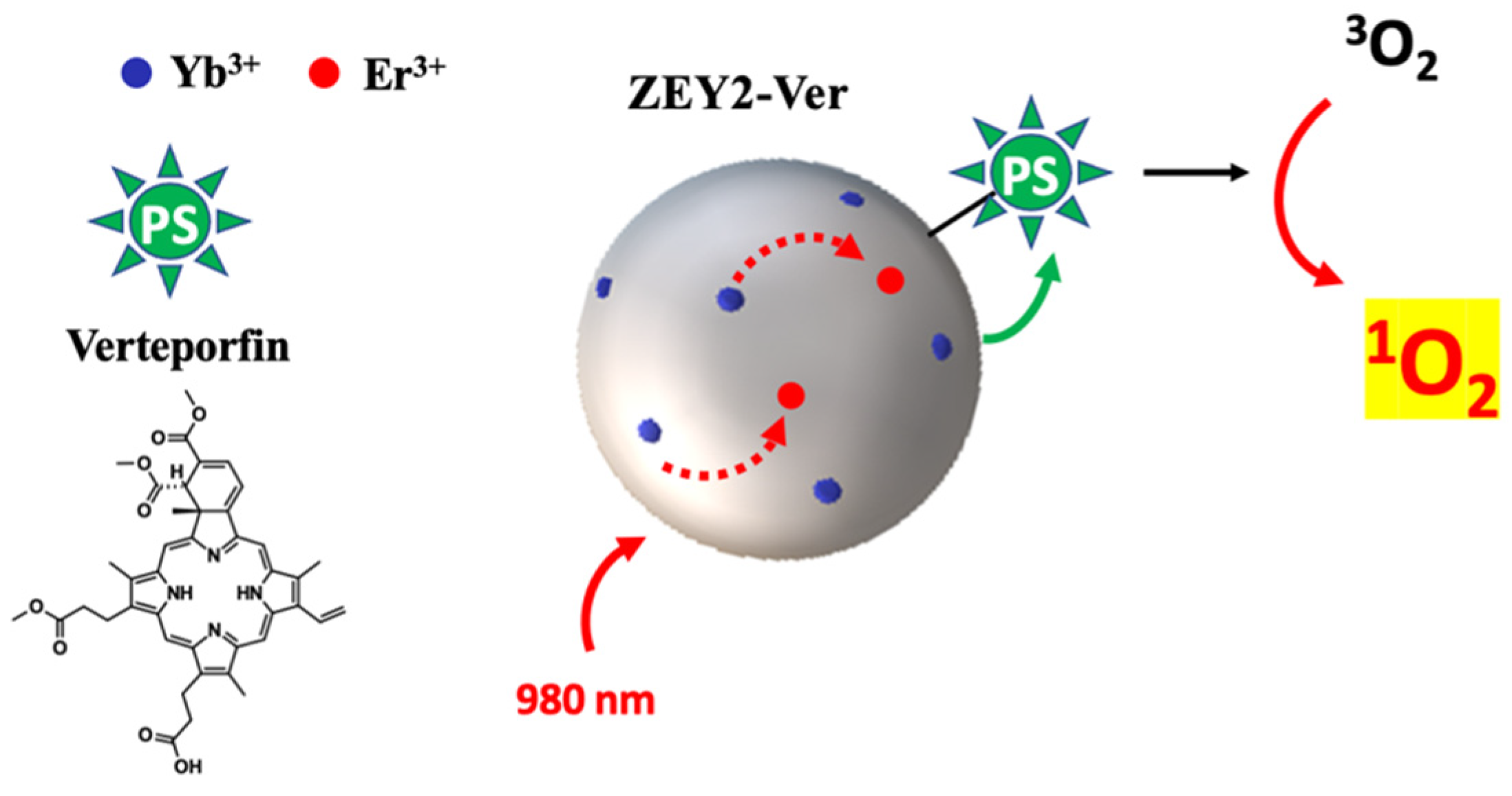
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Synthesis of Er3+/Yb3+ Doped ZrO2 Nanocrystals
5.2. Coating of Er0.005Yb0.02ZrO2 with Amino-Modified Amorphous Silica
5.3. Immobilization of Verteporfin on Silica Coated ZEY2 Nanoparticles
5.4. Characterization Methodologies
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, G.; Qiu, H.; Prasad, P.N.; Chen, X. Upconversion Nanoparticles: Design, Nanochemistry, and Applications in Theranostics. Chem. Rev. 2014, 114, 5161–5214. [Google Scholar] [CrossRef]
- Xie, J.; Lee, S.; Chen, X.Y. Nanoparticle-based theranostic agents. Adv. Drug Deliv. Rev. 2010, 62, 1064–1079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagoto, A.; Garello, F.; Marini, G.M.; Tripepi, M.; Arena, F.; Bardini, P.; Stefania, R.; Lanzardo, S.; Valbusa, G.; Porpiglia, F.; et al. Novel Gastrin-Releasing Peptide Receptor Targeted Near-Infrared Fluorescence Dye for Image-Guided Surgery of Prostate Cancer. Mol. Imaging Biol. 2020, 22, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Handula, M.; Verhoeven, M.; Chen, K.-T.; Haeck, J.; de Jong, M.; Dalm, S.U.; Seimbille, Y. Towards Complete Tumor Resection: Novel Dual-Modality Probes for Improved Image-Guided Surgery of GRPR-Expressing Prostate Cancer. Pharmaceutics 2022, 14, 195. [Google Scholar] [CrossRef] [PubMed]
- Sivagnanam, S.; Das, K.; Basak, M.; Mahata, T.; Stewart, A.; Maity, B.; Das, P. Self-assembled dipeptide based fluorescent nanoparticles as a platform for developing cellular imaging probes and targeted drug delivery chaperones. Nanoscale Adv. 2022, 4, 1694–1706. [Google Scholar] [CrossRef]
- Rubio-Camacho, M.; Alacid, Y.; Mallavia, R.; Martínez-Tomé, M.J.; Mateo, C.R. Polyfluorene-Based Multicolor Fluorescent Nanoparticles Activated by Temperature for Bioimaging and Drug Delivery. Nanomaterials 2019, 9, 1485. [Google Scholar] [CrossRef] [Green Version]
- Flak, D.; Przysiecka, Ł.; Nowaczyk, G.; Scheibe, B.; Kościński, M.; Jesionowski, T.; Jurga, S. GQDs-MSNs nanocomposite nanoparticles for simultaneous intracellular drug delivery and fluorescent imaging. J. Nanopart. Res. 2018, 20, 306. [Google Scholar] [CrossRef] [Green Version]
- Guglielmi, V.; Carton, F.; Vattemi, G.; Arpicco, S.; Stella, B.; Berlier, G.; Marengo, A.; Boschi, F.; Malatesta, M. Uptake and intracellular distribution of different types of nanoparticles in primary human myoblasts and myotubes. Int. J. Pharm. 2019, 560, 347–356. [Google Scholar] [CrossRef]
- Peng, Y.-K.; Tsang, S.C.E.; Chou, P.-T. Chemical design of nanoprobes for T1-weighted magnetic resonance imaging. Mater. Today 2016, 19, 336–348. [Google Scholar] [CrossRef]
- Ferrauto, G.; Carniato, F.; Tei, L.; Hu, H.; Aime, S.; Botta, M. MRI nanoprobes based on chemical exchange saturation transfer: LnIII chelates anchored on the surface of mesoporous silica nanoparticles. Nanoscale 2014, 6, 9604–9607. [Google Scholar] [CrossRef] [Green Version]
- Padovan, S.; Carrera, C.; Catanzaro, V.; Grange, C.; Koni, M.; Digilio, G. Glycol Chitosan Functionalized with a Gd(III) Chelate as a Redox-responsive Magnetic Resonance Imaging Probe to Label Cell Embedding Alginate Capsules. Chem. Eur. J. 2021, 27, 12289–12293. [Google Scholar] [CrossRef] [PubMed]
- Miletto, I.; Fraccarollo, A.; Barbero, N.; Barolo, C.; Cossi, M.; Marchese, L.; Gianotti, E. Mesoporous silica nanoparticles incorporating squaraine-based photosensitizers: A combined experimental and computational approach. Dalton Trans. 2018, 47, 3038–3046. [Google Scholar] [CrossRef] [PubMed]
- Garin, C.; Alejo, T.; Perez-Laguna, V.; Prieto, M.; Mendoza, G.; Arruebo, M.; Sebastian, V.; Rezusta, A. Chalcogenide nanoparticles and organic photosensitizers for synergetic antimicrobial photodynamic therapy. J. Mater. Chem. B 2021, 9, 6246–6259. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Thakur, N.S.; Chandna, S.; Reddy, Y.N.; Bhaumik, J. Development of a light activatable lignin nanosphere based spray coating for bioimaging and antimicrobial photodynamic therapy. J. Mater. Chem. B 2021, 9, 1592–1603. [Google Scholar] [CrossRef] [PubMed]
- Bilici, K.; Atac, N.; Muti, A.; Baylam, I.; Dogan, O.; Sennaroglu, A.; Can, F.; Acar, H.Y. Broad spectrum antibacterial photodynamic and photothermal therapy achieved with indocyanine green loaded SPIONs under near infrared irradiation. Biomater. Sci. 2020, 8, 4616–4625. [Google Scholar] [CrossRef]
- Pham, T.-C.; Nguyen, V.-N.; Choi, Y.; Lee, S.; Yoon, J. Recent Strategies to Develop Innovative Photosensitizers for Enhanced Photodynamic Therapy. Chem. Rev. 2021, 121, 13454–13619. [Google Scholar] [CrossRef]
- Yang, D.; Chen, F.; He, S.; Shen, H.; Hu, Y.; Feng, N.; Wang, S.; Weng, L.; Luo, Z.; Wang, L. One-pot growth of triangular SnS nanopyramids for photoacoustic imaging and photothermal ablation of tumors. New J. Chem. 2019, 43, 13256–13262. [Google Scholar] [CrossRef]
- Jaque, D.; Martinex Maestro, L.; del Rosal, B.; Haro-Gonzales, P.; Benayas, A.; Plaza, J.L.; Martin Rodriguez, E.; Garcia Sole, J. Nanoparticles for photothermal therapies. Nanoscale 2014, 6, 9494–9530. [Google Scholar] [CrossRef]
- Welsher, K.; Liu, Z.; Sherlock, S.P.; Robinson, J.T.; Chen, Z.; Daranciang, D.; Dai, H.J. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat. Nanotechnol. 2009, 4, 773–780. [Google Scholar] [CrossRef]
- Yaneva, Z.; Ivanova, D.; Nikolova, N.; Toneva, M. Organic dyes in contemporary medicinal chemistry and biomedicine. I. From the chromophore to the bioimaging/bioassay agent. Biotechnol. Biotechnol. Equip. 2022, 36, 1–14. [Google Scholar] [CrossRef]
- Mason, W.T. Fluorescent and Luminescent Probes for Biological Activity, 2nd ed.; Academic Press: London, UK, 1999. [Google Scholar]
- Shaner, N.C.; Steinbach, P.A.; Tsien, R.Y. A guide to choosing fluorescent proteins. Nat. Methods 2005, 2, 905–909. [Google Scholar] [CrossRef]
- Thorp-Greenwood, F.L. An Introduction to Organometallic Complexes in Fluorescence Cell Imaging: Current Applications and Future Prospects. Organometallics 2012, 31, 5686–5692. [Google Scholar] [CrossRef]
- Montalti, M.; Prodi, L.; Rampazzo, E.; Zaccheroni, N. Dye-doped silica nanoparticles as luminescent organized systems for nanomedicine. Chem. Soc. Rev. 2014, 43, 4243–4268. [Google Scholar] [CrossRef] [PubMed]
- Prilloff, S.; Fan, J.; Henrich-Noack, P.; Sabel, B.A. In vivo confocal neuroimaging (ICON): Non-invasive, functional imaging of the mammalian CNS with cellular resolution. Eur. J. Neurosci. 2010, 31, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hu, R.; Lin, G.; Roy, I.; Yong, K.T. Functionalized Quantum Dots for Biosensing and Bioimaging and Concerns on Toxicity. ACS Appl. Mater. Interfaces 2013, 5, 2786–2799. [Google Scholar] [CrossRef] [PubMed]
- Richards-Kortum, R.; Drezek, R.; Sokolov, K.; Pavlova, I.; Follen, M. Survey of endogenous biological fluorophores. In Handbook of Biomedical Fluorescence; Marcel Dekker Inc.: New York, NY, USA, 2003; pp. 237–264. [Google Scholar]
- Zhou, J.; Shirahata, N.; Sun, H.T.; Ghosh, B.; Ogawara, M.; Teng, Y.; Zhou, S.; Chu, R.G.S.; Fujii, M.; Qiu, J. Efficient Dual-Modal NIR-to-NIR Emission of Rare Earth Ions Co-doped Nanocrystals for Biological Fluorescence Imaging. J. Phys. Chem. Lett. 2013, 4, 402. [Google Scholar] [CrossRef]
- Hemmer, E.; Vetrone, F.; Soga, K. Lanthanide-based nanostructures for optical bioimaging: Small particles with large promise. MRS Bull. 2014, 39, 960–964. [Google Scholar] [CrossRef]
- Vetrone, F.; Capobianco, J.A. Lanthanide-doped fluoride nanoparticles: Luminescence, upconversion, and biological applications. Int. J. Nanotechnol. 2008, 5, 1306–1309. [Google Scholar] [CrossRef]
- Wu, Y.; Lin, S.; Liu, J.; Ji, Y.; Xu, J.; Xu, L.; Chen, K. Efficient up-conversion red emission from TiO2:Yb,Er nanocrystals. Opt. Express 2017, 25, 22648–22657. [Google Scholar] [CrossRef]
- Ivaturi, A.; MacDougall, S.K.W.; Martín-Rodríguez, R.; Quintanilla, M.; Marques-Hueso, J.; Krämer, K.W.; Meijerink, A.; Richards, B.S. Optimizing infrared to near infrared upconversion quantum yield of β-NaYF4:Er3+ in fluoropolymer matrix for photovoltaic devices. J. Appl. Phys. 2013, 114, 013505. [Google Scholar] [CrossRef] [Green Version]
- Mialon, G.; Turkcan, S.; Dantelle, G.; Collins, D.P.; Hadjipanayi, M.; Taylor, R.A.; Gacoin, T.; Alexandrou, A.; Boilott, J.P. High Up-Conversion Efficiency of YVO4:Yb,Er Nanoparticles in Water down to the Single-Particle Level. J. Phys. Chem. C 2010, 114, 22449–22454. [Google Scholar] [CrossRef]
- Kumar, G.A.; Pokhrel, M.; Martinez, A.; Dennis, R.C.; Villegas, I.L.; Sardar, D.K. Synthesis and spectroscopy of color tunable Y2O2S:Yb3+,Er3+ phosphors with intense emission. J. Alloys Compd. 2012, 513, 559–565. [Google Scholar] [CrossRef]
- Li, Y.; Wei, X.T.; Yin, M. Synthesis and upconversion luminescent properties of Er3+ doped and Er3+–Yb3+ codoped GdOCl powders. J. Alloys Compd. 2011, 509, 9865–9868. [Google Scholar] [CrossRef]
- Martinez, A.; Morales, J.; Diaz-Torres, L.A.; Salas, P.; de la Rosa, E.; Oliva, J.; Desirena, H. Green and red upconverted emission of hydrothermal synthesized Y2O3: Er3+–Yb3+ nanophosphors using different solvent ratio conditions. Mater. Sci. Eng. B 2010, 174, 164–168. [Google Scholar] [CrossRef]
- Patra, A.; Friend, C.S.; Kapoor, R.; Prasad, P.N. Effect of crystal nature on upconversion luminescence in Er3+:ZrO2 nanocrystals. Appl. Phys. Lett. 2003, 83, 284. [Google Scholar] [CrossRef]
- Wen, S.; Zhou, J.; Zheng, K.; Bednarkiewicz, A.; Liu, X.; Jin, D. Advances in highly doped upconversion nanoparticles. Nat. Commun. 2018, 9, 2415. [Google Scholar] [CrossRef]
- Hamblin, M.R. Upconversion in photodynamic therapy: Plumbing the depths. Dalton Trans. 2018, 47, 8571–8580. [Google Scholar] [CrossRef] [PubMed]
- Gionco, C.; Paganini, M.C.; Giamello, E.; Sacco, O.; Vaiano, V.; Sannino, D. Rare earth oxides in zirconium dioxide: How to turn a wide band gap metal oxide into a visible light active photocatalyst. J. Energy Chem. 2017, 26, 270–276. [Google Scholar] [CrossRef]
- Miletto, I.; Gioco, C.; Paganini, M.C.; Martinotti, S.; Ranzato, E.; Giamello, E.; Marchese, L.; Gianotti, E. Vis-NIR luminescent lanthanide-doped core-shell nanoparticles for imaging and photodynamic therapy. J. Photochem. Photobiol. A Chem. 2020, 403, 112840. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, W.; Seo, H.J. Analysis of Energy Transfer and Concentration Quenching in Sm3+-Activated Borate GdB3O6 Phosphors by Means of Fluorescence Dynamics. Spectrosc. Lett. 2015, 48, 27–31. [Google Scholar] [CrossRef]
- Wang, H.; Chen, Y.; Huang, J.; Gong, X.; Lin, Y.; Luo, Z.; Huang, Y. Spectroscopic properties and continuous-wave laser operation of Yb:Bi4Si3O12 crystals. Opt. Express 2014, 22, 21866–21871. [Google Scholar] [CrossRef]
- Zhao, J.; Sun, Y.; Kong, X.; Tian, L.; Wang, Y.; Tu, L.; Zhao, J.; Zhang, H. Controlled Synthesis, Formation Mechanism, and Great Enhancement of Red Upconversion Luminescence of NaYF4:Yb3+, Er3+ Nanocrystals/Submicroplates at Low Doping Level. J. Phys. Chem. B 2008, 112, 15666–15672. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Sun, L.-D.; Yan, C.-H. Energy transfer in lanthanide upconversion studies for extended optical applications. Chem. Soc. Rev. 2015, 44, 1608–1634. [Google Scholar] [CrossRef]
- Wang, F.; Liu, X. Upconversion Multicolor Fine-Tuning: Visible to Near-Infrared Emission from Lanthanide-Doped NaYF4 Nanoparticles. J. Am. Chem. Soc. 2008, 130, 5642–5643. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Liu, X.; Zeng, Q.; Zhang, Y.; Tu, L.; Liu, T.; Kong, X.; Wang, Y.; Cao, F.; Lambrechts, S.A.G.; et al. Covalently Assembled NIR Nanoplatform for Simultaneous Fluorescence Imaging and Photodynamic Therapy of Cancer Cells. ACS Nano 2012, 6, 4054–4062. [Google Scholar] [CrossRef] [PubMed]
- Ormond, A.B.; Freeman, H.S. Dye sensitizers for photodynamic therapy. Materials 2013, 6, 817–840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Estevao, B.M.; Miletto, I.; Hioka, N.; Marchese, L.; Gianotti, E. Mesoporous Silica Nanoparticles Functionalized with Amino Groups for Biomedical Applications. ChemistryOpen 2021, 10, 1251–1259. [Google Scholar] [CrossRef]
- Kobayashi, T. Vibronic coupling in J-aggregates. In J-Aggregates; World Scientific Publishing Co., Pte. Ltd.: Singapore, 2012; Volume 2, p. 49. [Google Scholar]
- Gianotti, E.; Estevão, B.M.; Miletto, I.; Tonello, S.; Renò, F.; Marchese, L. Verteporfin based silica nanoplatform for photodynamic therapy. ChemistrySelect 2016, 1, 127–131. [Google Scholar] [CrossRef]
- Wang, F.; Wang, J.; Liu, X. Direct Evidence of a Surface Quenching Effect on Size-Dependent Luminescence of Upconversion Nanoparticles. Angew. Chem. Int. Ed. 2010, 49, 7456–7460. [Google Scholar] [CrossRef]
- Johnson, N.J.J.; He, S.; Diao, S.; Chan, E.M.; Dai, H.; Almutairi, A. Direct Evidence for Coupled Surface and Concentration Quenching Dynamics in Lanthanide-Doped Nanocrystals. J. Am. Chem. Soc. 2017, 139, 3275–3282. [Google Scholar] [CrossRef]
- Zuo, J.; Li, Q.; Xue, B.; Li, C.; Chang, Y.; Zhang, Y.; Liu, X.; Tu, L.; Zhang, H.; Kong, X. Employing shells to eliminate concentration quenching in photonic upconversion nanostructure. Nanoscale 2017, 9, 7941–7946. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Zhao, J. Enhancing Upconversion Luminescence in Rare-Earth Doped Particles. U.S. Patent US20150252259A1, 10 September 2015. [Google Scholar]
- Gerola, A.P.; Semensato, J.; Pellosi, D.S.; Batistela, V.R.; Rabello, B.; Hioka, N.; Caetano, W. Chemical determination of singlet oxygen from photosensitizers illuminated with LED: New calculation methodology considering the influence of photobleaching. J. Photochem. Photobiol. A Chem. 2012, 232, 14–21. [Google Scholar] [CrossRef]
- Rabello, B.; Gerola, A.P.; Pellosi, D.; Tessaro, A.; Aparício, J.; Caetano, W.; Hioka, N. Singlet oxygen dosimetry using uric acid as a chemical probe: Systematic evaluation. J. Photochem. Photobiol. A Chem. 2012, 238, 53–62. [Google Scholar] [CrossRef]
- Locardi, F.; Gianotti, E.; Nelli, I.; Caratto, V.; Martinelli, A.; Ferretti, M.; Costa, G.A.; Canesi, L.; Balbi, T.; Fasoli, M.; et al. Facile synthesis of NIR and Visible luminescent Sm3+ doped lutetium oxide nanoparticles. Mater. Res. Bull. 2017, 86, 220–227. [Google Scholar] [CrossRef]
- Caratto, V.; Locardi, F.; Costa, G.A.; Masini, R.; Fasoli, M.; Panzeri, L.; Martini, M.; Bottinelli, E.; Gianotti, E.; Miletto, I. NIR Persistent Luminescence of Lanthanide Ion-Doped Rare-Earth Oxycarbonates: The Effect of Dopants. ACS Appl. Mater. Interfaces 2014, 6, 17346–17451. [Google Scholar] [CrossRef]
- Tu, L.; Liu, X.; Wu, F.; Zhang, H. Excitation energy migration dynamics in upconversion nanomaterials. Chem. Soc. Rev. 2015, 44, 1331–1345. [Google Scholar] [CrossRef] [Green Version]
- Stober, W.; Fink, A.; Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Estevao, B.M.; Miletto, I.; Marchese, L.; Gianotti, E. Optimized Rhodamine B labeled mesoporous silica nanoparticles as fluorescent scaffolds for the immobilization of photosensitizers: A theranostic platform for optical imaging and photodynamic therapy. Phys. Chem. Chem. Phys. 2016, 18, 9042–9052. [Google Scholar] [CrossRef]
- Rizzi, M.; Tonello, S.; Estevão, B.M.; Gianotti, E.; Marchese, L.; Renò, F. Verteporfin based silica nanoparticle for in vitro selective inhibition of human highly invasive melanoma cell proliferation. J. Photochem. Photobiol. B 2017, 167, 1–6. [Google Scholar] [CrossRef]
- Clemente, N.; Miletto, I.; Gianotti, E.; Invernizzi, M.; Marchese, L.; Dianzani, U.; Renò, F. Verteporfin-loaded mesoporous silica nanoparticles inhibit mouse melanoma proliferation in vitro and in vivo. J. Photochem. Photobiol. B 2019, 197, 111533. [Google Scholar] [CrossRef]
- Clemente, N.; Miletto, I.; Gianotti, E.; Sabbatini, M.; Invernizzi, M.; Marchese, L.; Dianzani, U.; Renò, F. Verteporfin-loaded mesoporous silica nanoparticles topical applications inhibit mouse melanoma lymphoangiogenesis and micrometastasis in vivo. Int. J. Mol. Sci. 2021, 22, 13443. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Qi, D.; Deng, C.; Zhang, X.; Zhao, D. Superparamagnetic High-Magnetization Microspheres with an Fe3O4@SiO2 Core and Perpendicularly Aligned Mesoporous SiO2 Shell for Removal of Microcystins. J. Am. Chem. Soc. 2008, 130, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Carpino, L.A. 1-Hydroxy-7-azabenzotriazole. An efficient peptide coupling additive. J. Am. Chem. Soc. 1993, 115, 4397–4398. [Google Scholar] [CrossRef]

| Acronyms | Sample Composition | Sample Description |
|---|---|---|
| ZEY2 | Er0.005Yb0.02Zr1-(0.025)O2 | ZrO2 doped with Er3+ (0.5%) and Yb3+ (2%) |
| ZEY5 | Er0.005Yb0.05Zr1-(0.055)O2 | ZrO2 doped with Er3+ (0.5%) and Yb3+ (5%) |
| ZEY10 | Er0.005Yb0.1Zr1-(0.105)O2 | ZrO2 doped with Er3+ (0.5%) and Yb3+ (10%) |
| ZEY2@SiO2 | Er0.005Yb0.02Zr1-(0.025)O2 | Silica-coated ZEY2 |
| ZEY2-Ver | Er0.005Yb0.02Zr1-(0.025)O2 | Ver-modified ZEY2@SiO2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miletto, I.; Gionco, C.; Paganini, M.C.; Cerrato, E.; Marchese, L.; Gianotti, E. Red Upconverter Nanocrystals Functionalized with Verteporfin for Photodynamic Therapy Triggered by Upconversion. Int. J. Mol. Sci. 2022, 23, 6951. https://doi.org/10.3390/ijms23136951
Miletto I, Gionco C, Paganini MC, Cerrato E, Marchese L, Gianotti E. Red Upconverter Nanocrystals Functionalized with Verteporfin for Photodynamic Therapy Triggered by Upconversion. International Journal of Molecular Sciences. 2022; 23(13):6951. https://doi.org/10.3390/ijms23136951
Chicago/Turabian StyleMiletto, Ivana, Chiara Gionco, Maria Cristina Paganini, Erik Cerrato, Leonardo Marchese, and Enrica Gianotti. 2022. "Red Upconverter Nanocrystals Functionalized with Verteporfin for Photodynamic Therapy Triggered by Upconversion" International Journal of Molecular Sciences 23, no. 13: 6951. https://doi.org/10.3390/ijms23136951
APA StyleMiletto, I., Gionco, C., Paganini, M. C., Cerrato, E., Marchese, L., & Gianotti, E. (2022). Red Upconverter Nanocrystals Functionalized with Verteporfin for Photodynamic Therapy Triggered by Upconversion. International Journal of Molecular Sciences, 23(13), 6951. https://doi.org/10.3390/ijms23136951









