Abstract
Myocarditis in response to COVID-19 vaccination has been reported since early 2021. In particular, young male individuals have been identified to exhibit an increased risk of myocardial inflammation following the administration of mRNA-based vaccines. Even though the first epidemiological analyses and numerous case reports investigated potential relationships, endomyocardial biopsy (EMB)-proven cases are limited. Here, we present a comprehensive histopathological analysis of EMBs from 15 patients with reduced ejection fraction (LVEF = 30 (14–39)%) and the clinical suspicion of myocarditis following vaccination with Comirnaty® (Pfizer-BioNTech) (n = 11), Vaxzevria® (AstraZenica) (n = 2) and Janssen® (Johnson & Johnson) (n = 2). Immunohistochemical EMB analyses reveal myocardial inflammation in 14 of 15 patients, with the histopathological diagnosis of active myocarditis according the Dallas criteria (n = 2), severe giant cell myocarditis (n = 2) and inflammatory cardiomyopathy (n = 10). Importantly, infectious causes have been excluded in all patients. The SARS-CoV-2 spike protein has been detected sparsely on cardiomyocytes of nine patients, and differential analysis of inflammatory markers such as CD4+ and CD8+ T cells suggests that the inflammatory response triggered by the vaccine may be of autoimmunological origin. Although a definitive causal relationship between COVID-19 vaccination and the occurrence of myocardial inflammation cannot be demonstrated in this study, data suggest a temporal connection. The expression of SARS-CoV-2 spike protein within the heart and the dominance of CD4+ lymphocytic infiltrates indicate an autoimmunological response to the vaccination.
1. Introduction
Myocarditis and pericarditis have been reported as a potential complication of the COVID-19 mRNA (messenger RNA) vaccines. Since the first observations of myocarditis in response to the Pfizer-BioNTech vaccine in April 2021 [1], numerous case reports, indicating a relationship between COVID-19 vaccination and the occurrence of myocarditis/pericarditis, have been published [2,3,4,5,6,7,8].
Meanwhile, large epidemiological studies reveal that the majority of COVID-19-vaccine-related myocarditis occur in young male individuals (median age of 21 years) following the second dose of mRNA based vaccines [9]. Most cases (76.8%) have been observed in response to the Pfizer-BioNTech vaccine (Comirnaty®), whereas one quarter (23.2%) received the Moderna vaccine (Spikevax®). Despite total case numbers, general incidence of myocarditis was shown to be higher in patients receiving Spikevax® (1.3 to 1.9 (Spikevax®), and 0.26 to 0.57 (Comirnaty®) extra cases of myocarditis per 10,000 subjects) [10]. Consequently, numerous national authorities recommended the use of Comirnaty® instead of Spikevax® for people aged 30 years and younger.
Signs of myocarditis after COVID-19 vaccination usually develop within 2 weeks [11,12] and clinical symptoms generally ease quickly without impairment of cardiac function [13]. Although COVID-19-vaccine-related myocarditis has an overall marginal incidence and affected patients show a fast recovery in absence of short-term complications, myocarditis is an alarming side effect, which needs to be monitored carefully.
While the majority of published cases are based on cardiac magnetic resonance imaging (cMRI) and laboratory evaluation [2,3,4,5,6,7], endomyocardial biopsy (EMB)-proven cases are limited [8,14,15]. Here, we present a comprehensive analysis of EMBs from 15 patients with the suspicion of myocarditis after vaccination against SARS-CoV-2 and reveal a temporal relation between the vaccination and the occurrence of myocardial inflammation, ranging from mild inflammatory cardiomyopathy to severe active myocarditis and giant cell myocarditis.
2. Results
Patients’ characteristics, clinical picture and laboratory findings at hospital admission are depicted in Table 1. Importantly, the suspicion of myocarditis in response to vaccination against SARS-CoV-2 was expressed in all cases. Median left ventricular ejection fraction (LVEF) was 30 (14–39)%. The analyzed cohort ranges from 18 to 68 years of age and is dominated by male sex (9/15, 60%). All patients were Caucasian. Vaccine type, onset of symptoms, clinical suspicion and clinical course differ a lot in the presented cohort (Table 1).

Table 1.
Patients’ characteristics, data on vaccination, clinical findings and EMB based diagnosis.
All EMBs were negatively tested for SARS-CoV-2 using E-gene-specific sequences [16]. Besides 10 cases with latent parvovirus B19 infection, no other viral pathogens including adenovirus, enterovirus, Epstein–Barr virus and human herpesvirus 6 were detected in the biopsies by nested- and qRT-PCR (Table 2). From 15 patients, two cases of active myocarditis (AMC, patients 1 and 2), two cases of giant cell myocarditis (GCMC, patients 3 and 14), nine cases of inflammatory cardiomyopathy (DCMi) and one case of dilated cardiomyopathy (DCM, patient 12) were diagnosed on the basis of EMB differential diagnostics. The majority of cases (11/15, 73%; AMC = 2; GCMC = 1; DCMi = 8) were observed in relation to the Comirnaty® mRNA vaccine, and four cases (27%) were related to Vaxzevria® (DCMi = 1; DCM = 1) and Janssen® (AMC = 1; DCMi = 1) vector vaccines. The onset of symptoms varied from 0 to 56 days (median 14 days) for Comirnaty®, from 1 to 14 days (median 7.5 days) for Vaxzevria®, and from 14 to 28 days (median 21 days) for Janssen® vaccines.

Table 2.
Biopsy findings. Virological analysis includes detection of genomes from SARS-CoV-2, Parvovirus B19 (B19V), Enterovirus, Adenovirus, human Herpesvirus 6 and Epstein–Barr virus. Immunohistochemical analysis includes detection of CD3+, CD4+ and CD8+ T cells, CD45R0+ T-memory cells, LFA-1+ lymphocytes, MAC-1+ macrophages, perforin+ cytotoxic cells, HLA-DR+-presenting cells, ICAM-1+ and VCAM-1+ endothelial cell-adhesion molecules, and SARS-CoV-2 spike protein. All inflammation markers were quantified by digital image analysis and are depicted as cells/mm2 or area %, respectively. Spike protein was evaluated as not expressed (− or sparsely expressed (+).
The majority of patients in the Comirnaty® group were male (8/11, 73%), while suspicion of myocarditis after vaccination with the vector vaccines was mainly made in female patients (1/2 for Janssen®; 2/2 for Vaxzevria®). The average age of patients receiving the Comirnaty® vaccine was 34 (28–46) years, of patients receiving Vaxzevria® 64 (61–66) years and of patients receiving Janssen® vaccines 38 (35–42) years (Table 1).
All patients revealed a sudden onset of severe left ventricular failure (LVEF ≤ 45%) and most had additional symptoms such as dyspnea, chest pain, reduced respiratory rate and diminished exercise capacity (Table 1). Data on cardiac MRI were available for six patients, showing no signs of myocarditis in four of them (patients 2, 4, 5 and 11) and typical signs of active myocarditis in two patients (patients 8 and 9) (Table 1). Five patients showed severe complications including cardiac decompensation (patients 5 and 8, both DCMi) and fulminant cardiogenic shock (patient 14, GCMC), and two patients (patients 2 and 3, AMC and GCMC, respectively) had to be resuscitated before admission to the intensive care unit (Table 1). Data on laboratory cardiac and inflammatory biomarkers including troponin, brain natriuretic peptide (BNP), creatinine kinase (CK) and C-reactive peptide (CRP) are heterogeneous and were elevated in 12 of 15 patients (Table 1).
Immunohistochemical EMB analysis revealed increased presence of inflammation markers in 14/15 patients (Figure 1, Table 2). Only 1 patient was found to have consistently low levels of inflammatory cells, compatible with the diagnosis of a DCM (patient 12, Figure 1, Table 2). All other patients with myocardial inflammation showed a diverse pattern of inflammatory markers, ranging from mild to severe degree of inflammation. From 14 patients with proof of inflammation, 9 (64%) showed elevated infiltration of CD3+ T cells, 12 (86%) showed increased levels of CD45R0+ T-memory cells and LFA-1+ lymphocytes, 11 (79%) showed increased numbers of MAC-1+ macrophages and HLA-DR+-presenting cells, and none showed augmented levels of perforin+ cytotoxic cells (Figure 1, Table 2). Cellular adhesion molecules ICAM-1 and VCAM-1 were normal in all patients, with the exception of two patient with AMC (patient 2) and GCMC (patient 14), respectively (Table 2). Further immunohistochemical analysis revealed positive detection of the SARS-CoV-2 spike protein in cardiac tissue in a couple of patients. In particular, the spike protein was found in sparse cells (cardiomyocytes) in 9 of 15 cases (Figure 2, Table 2). Moreover, except the cases with active myocarditis (patients 1 and 2), giant cell myocarditis (patients 3 and 14), and one case of DCMi (patient 9), CD4+-T-cell-to-CD8+-T-cell ratio was ≥1, suggesting a predominantly autoimmunological origin of the observed inflammation (Figure 3, Table 2).

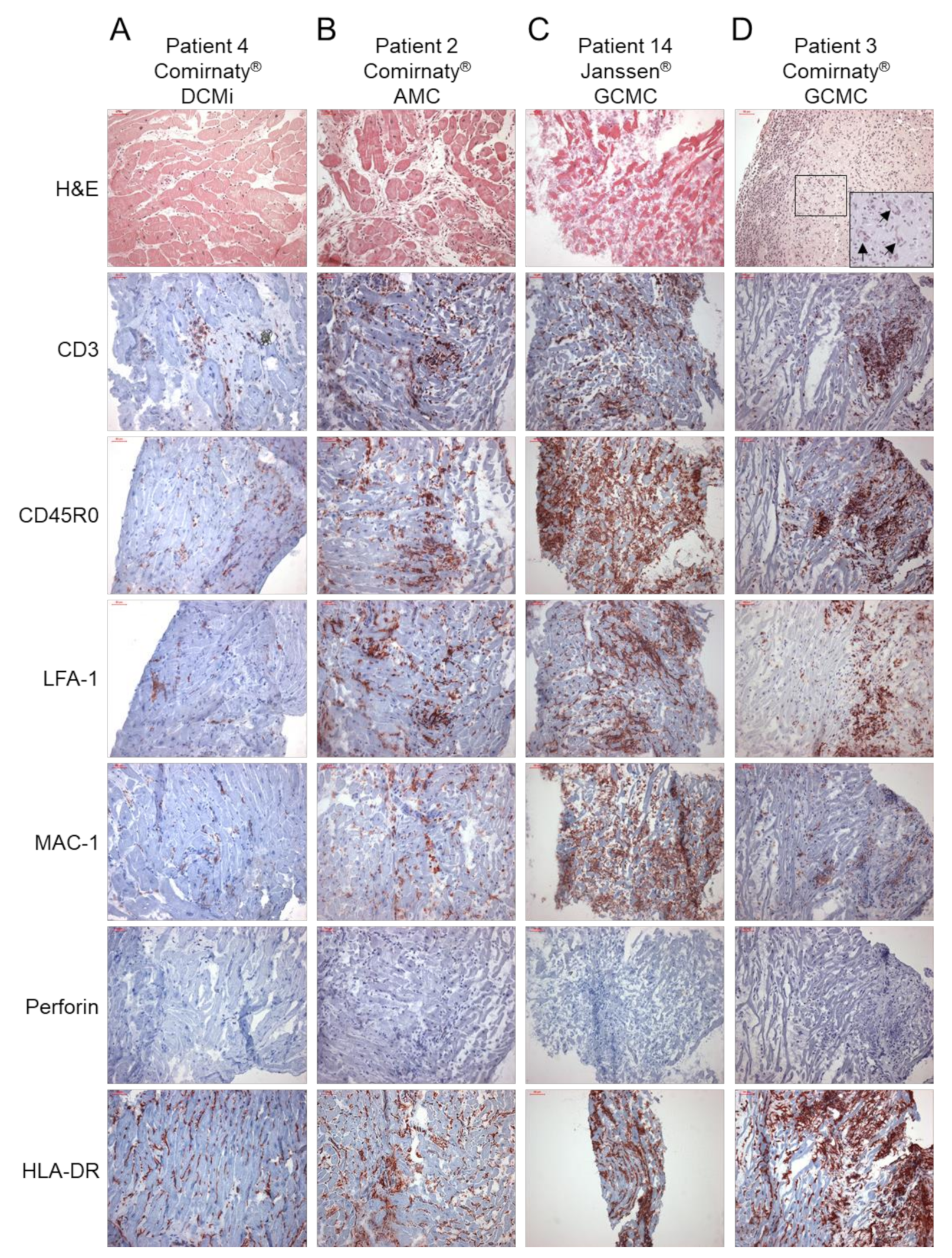
Figure 1.
Representative images of haematoxylin and eosin (H & E) staining, and immunohistochemical stainings for the assessment of inflammation in endomyocardial biopsies from patients with the suspicion of myocarditis after COVID-19 vaccination. Immunohistochemical detection of CD3+ T cells, CD45R0+ T-memory cells, LFA-1+ lymphocytes, MAC-1+ macrophages, perforin+ cytotoxic cells and HLA-DR+ activated T cells in patients diagnosed for (A) inflammatory cardiomyopathy (DCMi, patient 4, Comirnaty® vaccine), (B) acute myocarditis (AMC, patient 2, Comirnaty® vaccine) and (C,D) giant cell myocarditis (GCMC, patient 14, Janssen® vaccine; patient 3, Cormirnaty® vaccine; giant cells are marked by arrows in H & E staining). Immunohistochemical staining was quantified by digital image analysis and is depicted for each patient in Table 2. Magnification 200×. Scale bars 50 μm.
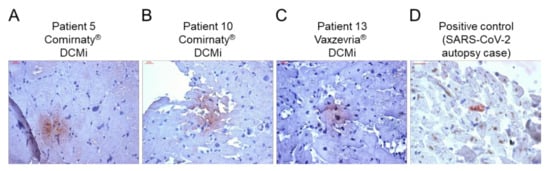
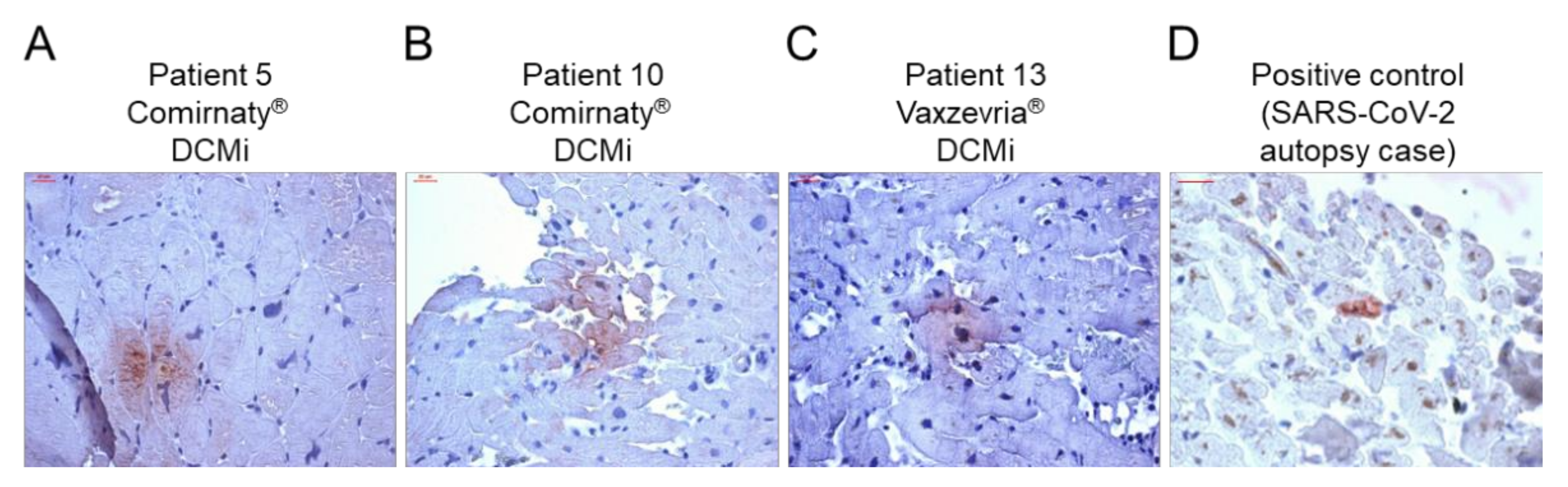
Figure 2.
Evidence of SARS-CoV-2 spike protein in cardiac tissue after COVID-19 vaccination. (A–C) Representative immunohistochemical stainings of SARS-CoV-2 spike protein in EMBs from patients diagnosed with DCMi after receiving Comirnaty® (panel A and B, patients 5 and 10) or Vaxzevria® (panel C, patient 13). (D) SARS-CoV-2-positive cardiac tissue served as positive control. Magnification 400×. Scale bars 20 μm.

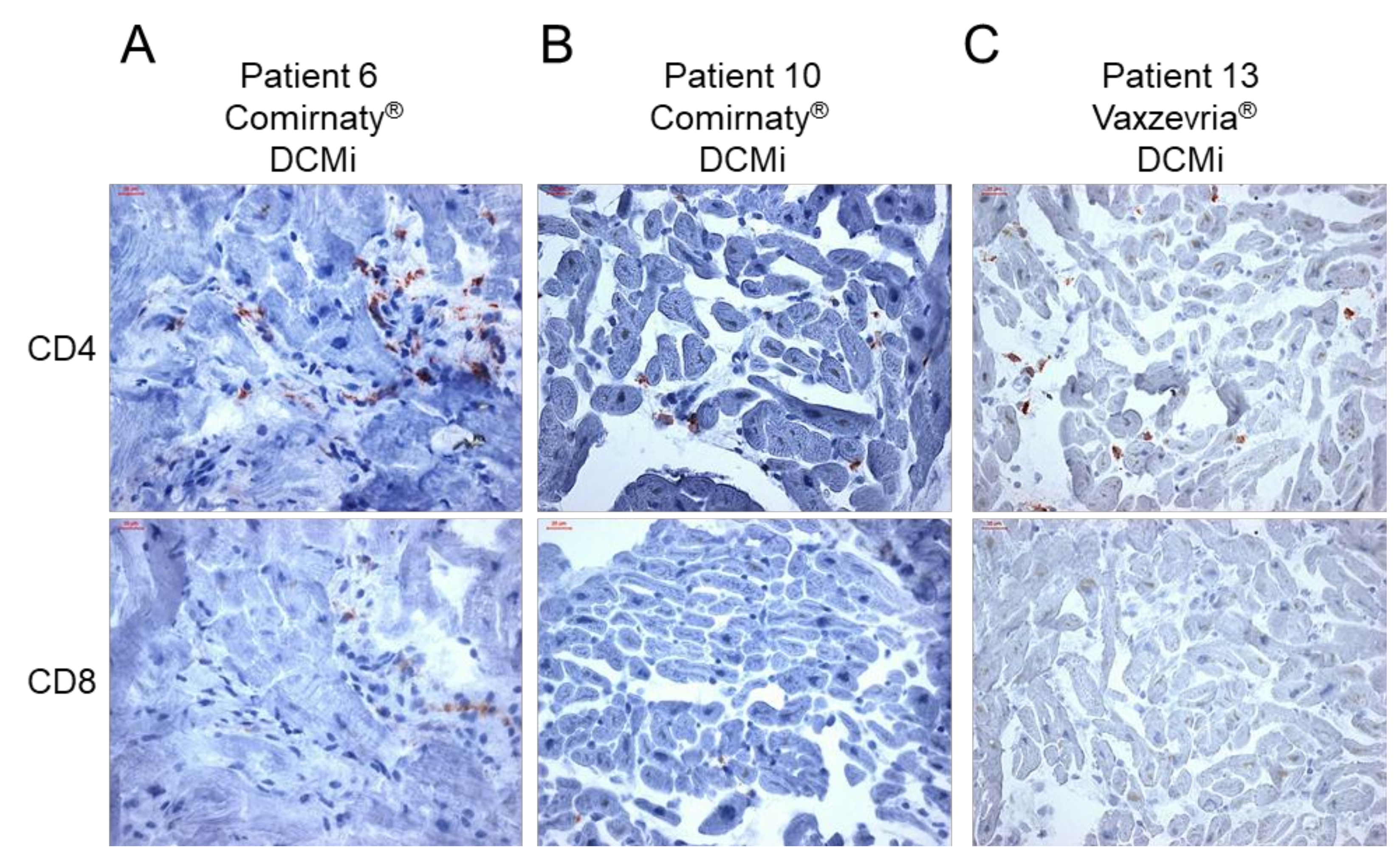
Figure 3.
Inflammatory cardiomyopathy in response to COVID-19 vaccination is dominated by CD4+ T cells. (A–C) Representative immunohistochemical stainings of CD4+ and CD8+ T cells in endomyocardial biopsies from patients diagnosed for inflammatory cardiomyopathy (DCMi) after receiving Comirnaty® (panel A and B, patients 6 and 10) or Vaxzevria® (panel C, patient 13) vaccines, respectively. Immunohistochemical staining was quantified by digital image analysis and CD4-to-CD8 ratio is depicted for each patient in Table 2. Magnification 400×. Scale bars 20 μm.
3. Discussion
In the present study, we identified 14/15 (93%) patients with suspected myocarditis after COVID-19 vaccination, with EMB-proven myocardial inflammation. Four of them had acute myocarditis (including active myocarditis and giant cell myocarditis), and 10 were diagnosed for inflammatory cardiomyopathy (DCMi). The absence of inflammation in EMBs of one patient might be explained by a sampling error. The onset of symptoms varied from 0 to 56 days, which is within the range of previous epidemiological findings [17]. Even though a causal relationship cannot be made, infectious reasons fro myocarditis had been excluded by molecular diagnostics of the most relevant viral pathogens (including SARS-CoV-2). Although latent viral genomes of B19V were found in 10/15 (67%) cases, active B19V transcription was not confirmed in these patients. B19V is the most frequently detected viral species in the human heart, and its contribution to myocardial inflammation and thus, to long-term patient outcome, is highly dependent on its transcriptional activity [18,19]. Therefore, identified latent B19V infections are unlikely causative of the myocardial inflammation in these patients.
Because viral infections have been ruled out as the cause for myocarditis/myocardial inflammation, autoimmunological mechanisms might be an explanation. Cross-reactivity of spike protein antibodies with myocardial contractile proteins, mRNA immune reactivity and hormonal involvement, have been discussed as potential mechanisms by which COVID-19 mRNA vaccines induce hyperimmunity [20]. In the present cohort, the SARS-CoV-2 spike protein was found to be expressed on cardiomyocytes in 9 of 15 patients. Thus, vaccine-encoded spike protein seems to reach the heart, where it might trigger an inflammatory response, resulting in the development of myocarditis or DCMi. Previous mouse data indicated an intramyocardial expression of spike protein after mRNA vaccination [21]; however, human data are scarce. Interestingly, except for five cases (AMC = 2, GCMC = 2 and DCMi = 1), the number of CD4+ T lymphocytes was either equal or higher than those of CD8+ T cells. In acute myocarditis, a shift of CD4-to-CD8 ratio towards CD8+ T cells is known [22], explaining the elevated CD8+ cells in AMC and GCMC patients. However, as CD4+ T cells are considered to be the major driver of autoimmune myocarditis [20], our data support the idea that vaccine-induced myocardial inflammation is a consequence of excessive CD4+ T-cell infiltration, and thus, a potential driver of autoimmunological myocardial damage. Moreover, the expression of HLA-DR was increased in 11 of 14 (79%) patients with evident inflammation. Due to the fact that HLA class II regions strongly associate with several autoimmune diseases [20], induction of HLA-DR in response to the vaccination supports an autoimmunological contribution to myocardial inflammation after vaccination. Expression of ICAM-1 and VCAM-1 adhesion molecules on the cardiac (micro)vascular endothelium is upregulated upon infection with myocarditis-associated viruses [23]. Besides one patient with AMC (patient 2) and one patient with GCMC (patient 14), none of the patients showed increased levels of ICAM-1 and VCAM-1, supporting a non-infectious origin of the observed inflammation. Perforin-mediated cardiac damage is known to be involved in viral [24,25] and acute idiopathic myocarditis [26]. Interestingly, none of the patients showed increased presence of perforin+ cells, indicating no contribution of cytotoxic events following the COVID-19 vaccination. Moreover, data on perforin+ cells in patients with reduced LVEF show that the lack of perforin+ cardiac infiltrates is associated with spontaneous LVEF improvement [23]. Thus, improvement of cardiac function could be assumed, as described for most of the vaccine-related myocarditis patients [13].
Among EMB diagnostics, non-invasive imaging techniques have been used to identify COVID-19-vaccine-related myocarditis [2,3,4,5,6,7]. Besides cMRI markers such as T2-weighted ratio, early gadolinium enhancement and late gadolinium enhancement within the Lake Louise criteria and somatostatin receptor positron emission tomography/computed tomography (PET/CT) have been applied for the diagnosis of myocarditis following COVID-19 vaccination [27]. However, as cardiac imaging techniques give helpful, but limited information about severe myocardial inflammation, they are unable to correlate with mild inflammation and viral persistence. The present case series reveal that cMRI was able to detect myocardial inflammation in only 33.3% (2/6) of the cases (Table 1). Thus, EMB remains the definite standard in the diagnosis of viral and inflammatory cardiomyopathy.
Study Limitations
There are several limitations of this study which have to be considered when interpreting the obtained results. First, no causality can be assumed or established due to the lack of a control group and the observational character of a case series. Although other causes of myocardial inflammation have been ruled out, there is no direct evidence of a vaccine-induced inflammatory response in the presented cases. Second, an incidence of vaccine-mediated myocarditis/inflammatory cardiomyopathy cannot be determined due to the selective approach of choosing cases with the suspicion of myocarditis after COVID-19 vaccination and the simultaneous availability of EMBs. Third, there is a lack extended clinical data for all of the patients covered in this multicenter study, and clinical data and evaluations were collected from different physicians. Finally, vaccine type and onset of symptoms after first or second dose differs between the cases and are therefore difficult to compare.
4. Materials and Methods
4.1. Patients and Clinical Investigations
In total, 15 patients (median age 38 (31–52) years; 9 men) from 11 different clinics were included in this this multicenter study. For all patients, the clinical suspicion of myocarditis after COVID-19 vaccination was expressed by the physician in charge due to a temporal relationship of the vaccination and the onset of myocarditis symptoms. All patients underwent key clinical investigations including laboratory testing, echocardiography and electrocardiogram, and some patients (n = 6) underwent additional cardiac magnetic resonance imaging (cMRI) [28]. Furthermore, EMBs were obtained from each patient and sent to the Institute for Cardiac Diagnostics and Therapy (IKDT, Berlin, Germany), for routine workup including histology, immunohistochemistry and molecular virology. The decision to perform EMB was made by the attending physician based on the setting of unexplained heart failure and sudden onset of severe left ventricular failure [29]. Coronary artery disease and other possible causes of myocardial dysfunction have been excluded by angiography prior to EMB in all patients. Left ventricular ejection fraction (LVEF) was determined by echocardiography with the Simpson method. All numerical data are presented with median and interquartile range.
4.2. Molecular Virology of EMBs
Immediately after withdrawal of EMBs, samples were transferred to RNAlater solution for nucleic acids stabilization (Thermo Fisher Scientific, Waltham, MA, USA). Total DNA was extracted by Gentra Puregene Kits (Qiagen, Hilden, Germany) according to manufacturer’s instructions [16]. Total RNA isolation was performed using TRIzol Reagent (Thermo Fisher Scientific, Waltham, MA, USA), followed by DNAse treatment (Promega, Walldorf, Germany) and cDNA synthesis using High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific, Waltham, MA, USA) and random hexamer primers (Thermo Fisher Scientific, Waltham, MA, USA) [19]. Then, the detection of viral genomes including Parvovirus B19 (B19V), Enteroviruses (including Coxsackievirus B3 and Echoviruses), Adenoviruses, Epstein–Barr virus, human Herpesvirus 6 and Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) was accomplished using nested polymerase chain reaction (nested-PCR) and quantitative reverse transcriptase (qRT)-PCR, as described before [19,30].
4.3. Histology, Immunohistochemistry and Digital Imaging Analysis
Histological examinations were carried out on formalin-fixed, paraffin-embedded specimens stained with hematoxylin and eosin (H & E) and trichrome stains according to standard procedures [31]. Active myocarditis was diagnosed according to the histomorphological Dallas criteria [32]. For qualification and quantification of inflammatory infiltrates, immunohistochemical staining was carried out on RNAlater-fixed, cryo-embedded EMBs, as described before [31]. Myocardial inflammation was diagnosed by the presence of ≥14 leucocytes with the presence of CD3+ T cells according to the European Society of Cardiology (ESC) position statement [33]. Furthermore, the number of CD11a+/LFA-1+ lymphocytes (threshold ≥ 14 cells/mm2), CD11b+/MAC-1+ macrophages (threshold ≥ 40 cells/mm2), CD45R0+ T-memory cells (threshold ≥ 50 cells/mm2), perforin+ cytotoxic cells (threshold ≥ 2.9 cells/mm2), and HLA-DR+-presenting cells (threshold ≥ 4.6 area%) was determined. In addition, ratio of CD4+-to-CD8+ T cells was analyzed. Tissue and endothelial activation were measured by the expression of the intercellular adhesion molecule 1 (ICAM-1, threshold ≥ 2.8 area%) and vascular cell adhesion molecule 1 (VCAM-1, threshold ≥ 0.08 area%). All immunohistochemical markers were quantified using digital imaging analysis, as described previously [23]. Detection of SARS-CoV-2 spike protein was performed on paraffin-embedded EMBs using an appropriate antibody (GeneTex, 1A9, GTX632604; 1:100) [34]. Autopsy cardiac tissue from a SARS-CoV-2 qRT-PCR-confirmed patient was used as positive control.
5. Conclusions
The present study summarizes EMB-based diagnostics of 15 patients with clinical suspicion of myocarditis following vaccination against SARS-CoV-2. It identifies 14 of 15 patients with myocardial inflammation, ranging from inflammatory cardiomyopathy to active myocarditis and severe giant cell myocarditis. Although a causal relationship between vaccination and the occurrence of myocardial inflammation cannot be established based on the findings, the cardiac detection of spike protein, the CD4+ T-cell-dominated inflammation and the close temporal relationship argue for a vaccine-triggered autoimmune reaction.
Author Contributions
Conceptualization, C.B., F.E. and H.-P.S.; methodology, C.B., G.A. and D.H.; formal analysis, C.B., G.A., D.H. and U.G.; investigation, C.B., G.A., D.H., U.G., C.H., B.A., R.W., M.K., S.R., P.W., T.M., A.E., M.G., C.P., A.B., M.F., T.K., J.B. and F.E.; writing—original draft preparation, C.B.; writing—review and editing, C.B.; funding acquisition, G.A., F.E. and H.-P.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by a ProFIT grant of the INVESTITIONSBANK BERLIN (IBB)/cofounded by European Regional Development Fund (EFRE), grant numbers 10169096, 10169098, 10169028.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board at Campus Benjamin Franklin, Charité, Berlin (EA4/236/20, 27 December 2020).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
All authors confirm that all related data supporting the findings of this study are given in the article.
Acknowledgments
The authors thank Susanne Ochmann, Kitty Winter, Jenny Klostermann, Katrin Errami (IKDT Berlin), and Hannes Jarmuth (Charite Berlin) for their skillful technical assistance. Furthermore, we thank Dirk Westermann and Diana Lindner from the University Heart and Vascular Center Hamburg for providing the SARS-CoV-2-positive control.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- CDC Centers for Disease Control and Prevention. Myocarditis and Pericarditis Following MRNA COVID-19 Vaccination. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/myocarditis.html (accessed on 6 April 2022).
- Minocha, P.K.; Better, D.; Singh, R.K.; Hoque, T. Recurrence of Acute Myocarditis Temporally Associated with Receipt of the MRNA Coronavirus Disease 2019 (COVID-19) Vaccine in a Male Adolescent. J. Pediatr. 2021, 238, 321–323. [Google Scholar] [CrossRef]
- Muthukumar, A.; Narasimhan, M.; Li, Q.-Z.; Mahimainathan, L.; Hitto, I.; Fuda, F.; Batra, K.; Jiang, X.; Zhu, C.; Schoggins, J.; et al. In-Depth Evaluation of a Case of Presumed Myocarditis After the Second Dose of COVID-19 MRNA Vaccine. Circulation 2021, 144, 487–498. [Google Scholar] [CrossRef] [PubMed]
- McLean, K.; Johnson, T.J. Myopericarditis in a Previously Healthy Adolescent Male Following COVID-19 Vaccination: A Case Report. Acad. Emerg. Med. 2021, 28, 918–921. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, T.; Cattafi, A.; Carerj, M.L.; Booz, C.; Ascenti, G.; Cicero, G.; Blandino, A.; Mazziotti, S. Myocarditis After SARS-CoV-2 Vaccination: A Vaccine-Induced Reaction? Can. J. Cardiol. 2021, 37, 1665–1667. [Google Scholar] [CrossRef]
- Albert, E.; Aurigemma, G.; Saucedo, J.; Gerson, D.S. Myocarditis Following COVID-19 Vaccination. Radiol. Case Rep. 2021, 16, 2142–2145. [Google Scholar] [CrossRef] [PubMed]
- Bautista García, J.; Peña Ortega, P.; Bonilla Fernández, J.A.; Cárdenes León, A.; Ramírez Burgos, L.; Caballero Dorta, E. Acute Myocarditis after Administration of the BNT162b2 Vaccine against COVID-19. Rev. Española Cardiol. 2021, 74, 812–814. [Google Scholar] [CrossRef]
- Nagasaka, T.; Koitabashi, N.; Ishibashi, Y.; Aihara, K.; Takama, N.; Ohyama, Y.; Yokoyama, T.; Kaneko, Y. Acute Myocarditis Associated with COVID-19 Vaccination: A Case Report. J. Cardiol. Cases 2021, 25, 285–288. [Google Scholar] [CrossRef]
- Matta, A.; Kunadharaju, R.; Osman, M.; Jesme, C.; McMiller, Z.; Johnson, E.M.; Matta, D.; Kallamadi, R.; Bande, D. Clinical Presentation and Outcomes of Myocarditis Post MRNA Vaccination: A Meta-Analysis and Systematic Review. Cureus 2021, 13, e19240. [Google Scholar] [CrossRef]
- European Medicine Agency Meeting Highlights from the Pharmacovigilance Risk Assessment Committee (PRAC) 29 November—2 December 2021. Available online: https://www.ema.europa.eu/en/news/meeting-highlights-pharmacovigilance-risk-assessment-committee-prac-29-november-2-december-2021 (accessed on 1 April 2022).
- Verma, A.K.; Lavine, K.J.; Lin, C.-Y. Myocarditis after COVID-19 MRNA Vaccination. N. Engl. J. Med. 2021, 385, 1332–1334. [Google Scholar] [CrossRef]
- Viskin, D.; Topilsky, Y.; Aviram, G.; Mann, T.; Sadon, S.; Hadad, Y.; Flint, N.; Shmilovich, H.; Banai, S.; Havakuk, O. Myocarditis Associated With COVID-19 Vaccination: Echocardiography, Cardiac Tomography, and Magnetic Resonance Imaging Findings. Circ. Cardiovasc. Imaging 2021, 14, e013236. [Google Scholar] [CrossRef]
- Salah, H.M.; Mehta, J.L. COVID-19 Vaccine and Myocarditis. Am. J. Cardiol. 2021, 157, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Kadwalwala, M.; Chadha, B.; Ortoleva, J.; Joyce, M. Multimodality Imaging and Histopathology in a Young Man Presenting with Fulminant Lymphocytic Myocarditis and Cardiogenic Shock after MRNA-1273 Vaccination. BMJ Case Rep. 2021, 14, e246059. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, P.; Klingel, K.; Ohlmann-Knafo, S.; Hüttinger, S.; Sood, N.; Pickuth, D.; Kindermann, M. Biopsy-Proven Lymphocytic Myocarditis Following First MRNA COVID-19 Vaccination in a 40-Year-Old Male: Case Report. Clin. Res. Cardiol. 2021, 110, 1855–1859. [Google Scholar] [CrossRef]
- Escher, F.; Pietsch, H.; Aleshcheva, G.; Bock, T.; Baumeier, C.; Elsaesser, A.; Wenzel, P.; Hamm, C.; Westenfeld, R.; Schultheiss, M.; et al. Detection of Viral SARS-CoV-2 Genomes and Histopathological Changes in Endomyocardial Biopsies. ESC Hear. Fail. 2020, 7, 2440–2447. [Google Scholar] [CrossRef] [PubMed]
- Witberg, G.; Barda, N.; Hoss, S.; Richter, I.; Wiessman, M.; Aviv, Y.; Grinberg, T.; Auster, O.; Dagan, N.; Balicer, R.D.; et al. Myocarditis after COVID-19 Vaccination in a Large Health Care Organization. N. Engl. J. Med. 2021, 385, 2132–2139. [Google Scholar] [CrossRef] [PubMed]
- Escher, F.; Aleshcheva, G.; Pietsch, H.; Baumeier, C.; Gross, U.M.; Schrage, B.N.; Westermann, D.; Bock, C.-T.; Schultheiss, H.-P. Transcriptional Active Parvovirus B19 Infection Predicts Adverse Long-Term Outcome in Patients with Non-Ischemic Cardiomyopathy. Biomedicines 2021, 9, 1898. [Google Scholar] [CrossRef] [PubMed]
- Pietsch, H.; Escher, F.; Aleshcheva, G.; Lassner, D.; Bock, C.-T.; Schultheiss, H.-P. Detection of Parvovirus MRNAs as Markers for Viral Activity in Endomyocardial Biopsy-Based Diagnosis of Patients with Unexplained Heart Failure. Sci. Rep. 2020, 10, 22354. [Google Scholar] [CrossRef] [PubMed]
- Vdovenko, D.; Eriksson, U. Regulatory Role of CD4+ T Cells in Myocarditis. J. Immunol. Res. 2018, 2018, 4396351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Chen, Y.; Zhao, Y.; Lung, D.C.; Ye, Z.; Song, W.; Liu, F.-F.; Cai, J.-P.; Wong, W.-M.; Yip, C.C.-Y.; et al. Intravenous Injection of Coronavirus Disease 2019 (COVID-19) MRNA Vaccine Can Induce Acute Myopericarditis in Mouse Model. Clin. Infect. Dis. 2021, 73, 2372–2373. [Google Scholar] [CrossRef]
- Jurcova, I.; Rocek, J.; Bracamonte-Baran, W.; Zelizko, M.; Netuka, I.; Maluskova, J.; Kautzner, J.; Cihakova, D.; Melenovsky, V.; Maly, J. Complete Recovery of Fulminant Cytotoxic CD8 T-cell-mediated Myocarditis after ECMELLA Unloading and Immunosuppression. ESC Heart Failure 2020, 7, 1976–1981. [Google Scholar] [CrossRef]
- Escher, F.; Kühl, U.; Lassner, D.; Stroux, A.; Gross, U.; Westermann, D.; Pieske, B.; Poller, W.; Schultheiss, H. High Perforin-Positive Cardiac Cell Infiltration and Male Sex Predict Adverse Long-Term Mortality in Patients with Inflammatory Cardiomyopathy. J. Am. Heart Assoc. 2017, 6, e005352. [Google Scholar] [CrossRef]
- Gebhard, J.R.; Perry, C.M.; Harkins, S.; Lane, T.; Mena, I.; Asensio, V.C.; Campbell, I.L.; Whitton, J.L. Coxsackievirus B3-Induced Myocarditis. Am. J. Pathol. 1998, 153, 417–428. [Google Scholar] [CrossRef]
- Seko, Y.; Shinkai, Y.; Kawasaki, A.; Yagita, H.; Okumura, K.; Takaku, F.; Yazaki, Y. Expression of Perforin in Infiltrating Cells in Murine Hearts with Acute Myocarditis Caused by Coxsackievirus B3. Circulation 1991, 84, 788–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, L.; Joag, S.; Zheng, L.; Young, J.; Lee, C.; Lee, Y. Perforin-Mediated Myocardial Damage in Acute Myocarditis. Lancet 1990, 336, 1019–1021. [Google Scholar] [CrossRef]
- Bollano, E.; Bergh, N.; Dudás, A.; Bobbio, E.; Polte, C.L. Somatostatin Receptor Positron Emission Tomography/Computed Tomography in Myocarditis Following MRNA COVID-19 Vaccination. Eur. Hear. J. Case Rep. 2022, 6, ytac117. [Google Scholar] [CrossRef]
- Lampejo, T.; Durkin, S.M.; Bhatt, N.; Guttmann, O. Acute Myocarditis: Aetiology, Diagnosis and Management. Clin. Med. 2021, 21, e505–e510. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.T.; Baughman, K.L.; Feldman, A.M.; Frustaci, A.; Jessup, M.; Kuhl, U.; Levine, G.N.; Narula, J.; Starling, R.C.; Towbin, J.; et al. The Role of Endomyocardial Biopsy in the Management of Cardiovascular Disease. Circulation 2007, 116, 2216–2233. [Google Scholar] [CrossRef] [Green Version]
- Pietsch, H.; Escher, F.; Aleshcheva, G.; Baumeier, C.; Morawietz, L.; Elsaesser, A.; Schultheiss, H.-P. Proof of SARS-CoV-2 Genomes in Endomyocardial Biopsy with Latency after Acute Infection. Int. J. Infect. Dis. 2021, 102, 70–72. [Google Scholar] [CrossRef]
- Baumeier, C.; Escher, F.; Aleshcheva, G.; Pietsch, H.; Schultheiss, H.-P. Plasminogen Activator Inhibitor-1 Reduces Cardiac Fibrosis and Promotes M2 Macrophage Polarization in Inflammatory Cardiomyopathy. Basic Res. Cardiol. 2021, 116, 1. [Google Scholar] [CrossRef]
- Aretz, H.T. Myocarditis: The Dallas Criteria. Hum. Pathol. 1987, 18, 619–624. [Google Scholar] [CrossRef]
- Caforio, A.L.P.; Pankuweit, S.; Arbustini, E.; Basso, C.; Gimeno-Blanes, J.; Felix, S.B.; Fu, M.; Heliö, T.; Heymans, S.; Jahns, R.; et al. Current State of Knowledge on Aetiology, Diagnosis, Management, and Therapy of Myocarditis: A Position Statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2013, 34, 2636–2648. [Google Scholar] [CrossRef] [PubMed]
- Dal Ferro, M.; Bussani, R.; Paldino, A.; Nuzzi, V.; Collesi, C.; Zentilin, L.; Schneider, E.; Correa, R.; Silvestri, F.; Zacchigna, S.; et al. SARS-CoV-2, Myocardial Injury and Inflammation: Insights from a Large Clinical and Autopsy Study. Clin. Res. Cardiol. 2021, 110, 1822–1831. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).