Prebiotics as a Tool for the Prevention and Treatment of Obesity and Diabetes: Classification and Ability to Modulate the Gut Microbiota
Abstract
1. Introduction
2. Prebiotics
3. Classification of Prebiotics
3.1. Inulin (Fructan)
3.2. Fructo-Oligosaccharides (Fructan)
3.3. Galactooligosaccharides
3.4. Human Milk Oligosaccharides
3.5. Glucose-Derived Oligosaccharides
3.6. Resistant Starches
3.7. Pectic Oligosaccharides
| Abbreviation | Chemical Composition | DP | Chemical Formula | References |
|---|---|---|---|---|
| Inulin | linear chain of fructose with β(2→1) linkages | 3–60 | 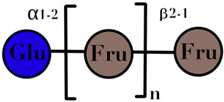 n = up to 100 | [59,60] |
| FOS | linear chain of fructose with β(2→1) linkages | <10 | 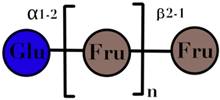 n = 1–5 | [59,60] |
| GOS | Chain of galactosyl residues and a terminal glucose linked by β-(1–2), β-(1–3), β-(1–4), or β-(1–6) glycosidic bonds | 2–8 | 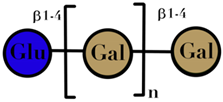 n = 1–4 | [60,103] |
| HMO | composed of five monosaccharides: glucose, galactose, N-acetylglucosamine, fucose, and N-acetylneuraminic acid or sialic acid | <7 | 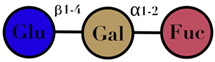 2′fucosyllactose 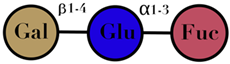 3′-fucosyllactose 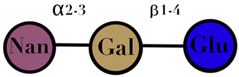 3′-sialyllactose 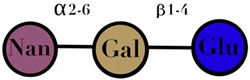 6′-sialyllactose 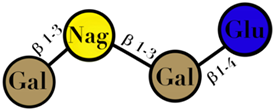 Lacto-N-tetraose 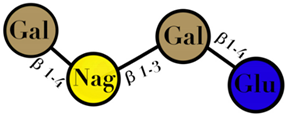 Lacto-N-neotetraose 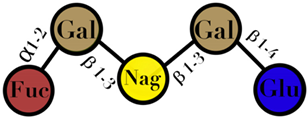 Lacto-N-fucopentaose I | [104,105] |
| Lactulose | consisting of galactose and fructose moieties | - | 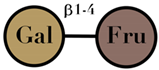 | [106,107] |
| Lactosucrose | composed of galactose, fructose, and glucose monomers | - | 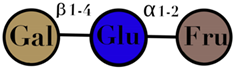 | [108] |
| AX | β-1,4-linked D-xylopyranoside units substituted with arabinose residues on the c(o)-2 or c(o)-3 position | 1–60 | 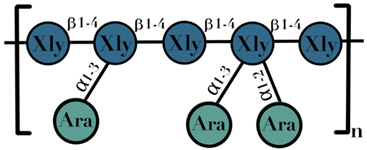 | [109] |
| XOS | xylose moeities linked by β-(1→4) glyosidic bonds | 2–4 | 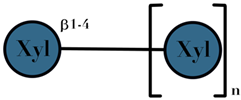 n = 2–4 | [110,111,112] |
| Symbols used in Table 1: their meaning and chemical structure. | 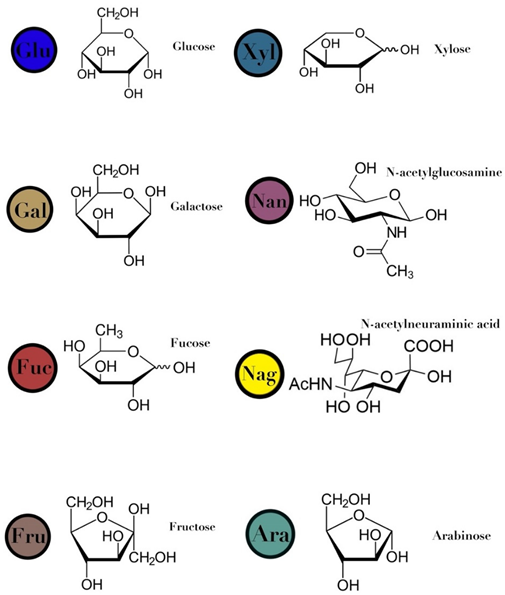 | |||
3.8. Lactulose
3.9. Lactosucrose
3.10. Arabinoxylans
3.11. Xylooligosaccharides
4. Efficacy of Prebiotics on Gut Microbiota Composition: In Vivo and In Vitro Studies
4.1. Inulin
4.2. FOS
4.3. GOS
4.4. HMO
4.5. PDX
4.6. RS
4.7. POS
4.8. Lactulose
4.9. Lactosucrose
4.10. AX
| Prebiotics | Model | Strategy/Duration of Feeding | Dose Supplemented | Form | No. of Applications | Re-Calculated Dose * | Fecal Microbial Changes Relative to Control | Reference |
|---|---|---|---|---|---|---|---|---|
| Inulin | 17 elderly women (mean age = 76.4 years body weight not reported) | 8 days, (3 days adaptation) Feeding was continued for 8 days | 20 g/day and increased to 40 g/day | Dissolved in drinking water | Once/day | 285.7 mg/kg/day and increased to 571.4 mg/kg/day | significant ↑ in Bifidobacteria ↓ in Enterococci and Enterobacteriaceae | [154] |
| 10 healthy volunteers (age = between 20 and 55 years Body weight not reported) | 14 days | 8 g/day | Dissolved in drinking water | Twice/day | 114.3 mg/kg/day | significant ↑ in Bifidobacteria ↑ in the number of Clostridia | [155] | |
| Germ-free adult male Fischer rats (age = 10 weeks and body weight = 280 g) | 8 weeks | 1.84 g/day of the diet | Mixed with chow | During the day | 6.57 g/kg/day | significant ↑ in producing SCFAs | [156] | |
| FOS | Male Wistar rats (age = 2 months and body weight 403.2 ± 48.1 g) | 7 days | 8% of the diet | Mixed with chow | During the day | 3.4 g/kg/day | ↑ the bioavailability of nutritionally important minerals | [158] |
| FOS + GOS | 10 Male C57BL/6J mice (age 8 weeks old mice; mean body weight = 28 g) | 10 weeks | 0.3 –0.4 g/mouse/day | Dissolved in drinking water | During the day | 1.1–1.43 g/kg/day | ↑ Akkermansia abundance | [164] |
| GOS | 18 healthy human (age and body weight not indicated) | 3 weeks | 2.5 g/day 5 g/day, 10 g/day | Administered in edible chews | Once/day | 35.7 mg/kg/day 71.4 mg/kg/day, 142 mg/kg/day | significant ↑ in abundance of Bifidobacteria and Faecalibacterium prausnitzii, ↓ in Bacteroides | [161] |
| Mud crab (age not reported and body weight 63.6 ± 8.8 g) | 24 h | 0.05 g/day | Dissolved in water | During the day | 786 mg/kg/day | ↑ Bacteroidetes | [163] | |
| PDX/FOS | 77 Children (age 5.8 ± 1.3; body weight not reported) | 2 weeks | 4.17 g PDX + 0.45 g FOS | Dissolved in drinking water | Once/day | PDX 200 mg/kg/day + FOS 22 mg/kg/day | ↑ in number of Bifidobacterium and Lactobacillus | [176] |
| PDX | 20 Healthy men (Age = 27.5 6 ± 4.33; body weight = 86.26 ± 13.48 kg) | 21 days | 21 g/day | Mixed in bar | Once/day | 243.4 mg/kg/day | ↑ in number of Faecalibacterium, Phascolarctobacterium, and Dialister | [175] |
| 15 Healthy volunteers (age = 18–50 body weight not reported) | 3 weeks | 8 g/day | Powder | Once/day | 243.5 mg/kg/day | ↑ Ruminococcus intestinalis, Clostridium clusters I, II and IV, significantly ↓ levels of Lactobacillus and Enterococcus group | [177] | |
| RS | 6 Male C57BL/6J mice (18–20 month old and body weight not reported) | 8 weeks | 0.54 g/day | Mixed with chow | During the day | 18 g/kg/day | ↑ in number of Bacteroidetes, Bifidobacterium and Akkermansia species | [178] |
| Sprague-Dawley rats (age 6 weeks and body weight not reported) | 12 weeks | 27% of the diet | Mixed with chow | During the day | 18 g/kg/day | ↑ in SCFAs | [180] | |
| POS | Pigs’ fecal inoculum (age 4 years and the mean body weight 233.0 ± 10.02 kg) | 48 h | 9 g/of POS to 1 mL of inoculum | Mixed with the chow | - | - | ↑ in SCFAs | [183] |
| Lactulose | 12 healthy volunteers (age = (24 to 31 years and body weight not reported) | 4 weeks | 20 g/day | Mixed with chow | Twice/day | 285.7 mg/kg/day | ↑ in number of Bifidobacterium and Lactobacillus. | [186] |
| Lactosucrose | Red seabream Pagrus major (age and body weight not reported) | 9 months | 20 mg/kg/day | Mixed with chow | Once/day | 20 mg/kg/day | ↑ production of SCFAs | [192] |
| 8 Shepherd dogs (body weight = 22 to 32 kg; mean age = 13.5 months) | 2 weeks | 1.5 g/day | Mixed with chow | Twice/day | 55.6 mg/kg/day | ↓ in the levels of f Clostridium pefringms ↑ Bifidobacterium | [122] | |
| 16 Broiler chickens (20–62 days and body weight not reported) | 62 days | 825 mg/day | Mixed with chow | During the day | 458 mg/kg/day | ↑ in the number of Bifidobacterium ↓ the number of Bacteriodaceae; Staphylococci; and total anaerobic bacteria, C. perfringens | [198] | |
| 8 Cats (Mean agae + 7; body weight 3.5 kg) | 2 weeks | 50 mg of lactosucrose/day | Mixed with the chow | During the day | 14 mg/kg/day | ↑ in Lactobacilli and Bifidobacterium ↓ in Clostridium perfringens, clostridia, Spirochaetaceae, and Enterobacteriaceae | [185] | |
| AX | 10 human children (mean age, 3 years, 7 months body weight not reported) (in vitro) | 48 h | 10 g/liter | Dissolved in drinking water | - | -- | ↑ in number of Lactobacillus | [196] |
| XOS | 12 healthy adult women (mean age for women = 33.6 years and body weight not reported) and 11 healthy men (mean age = 30.1 and body weight not reported) | 8 weeks | 1.4 g/day or 2.8 g/day | Capsule | Once/day | 20 or 40 mg/kg/day | ↑ Bacteroides fragilis, ↑ Bifidobacterium | [199] |
| 13 elderly human (body weight = 58.6 ± 10.1 kg body weight not reported) | 3 weeks | 4 g/day | Mixed with chow | Once/day | 68.3 mg/kg/day | ↑ in number of Bifidobacterium species | [200] |
4.11. XOS
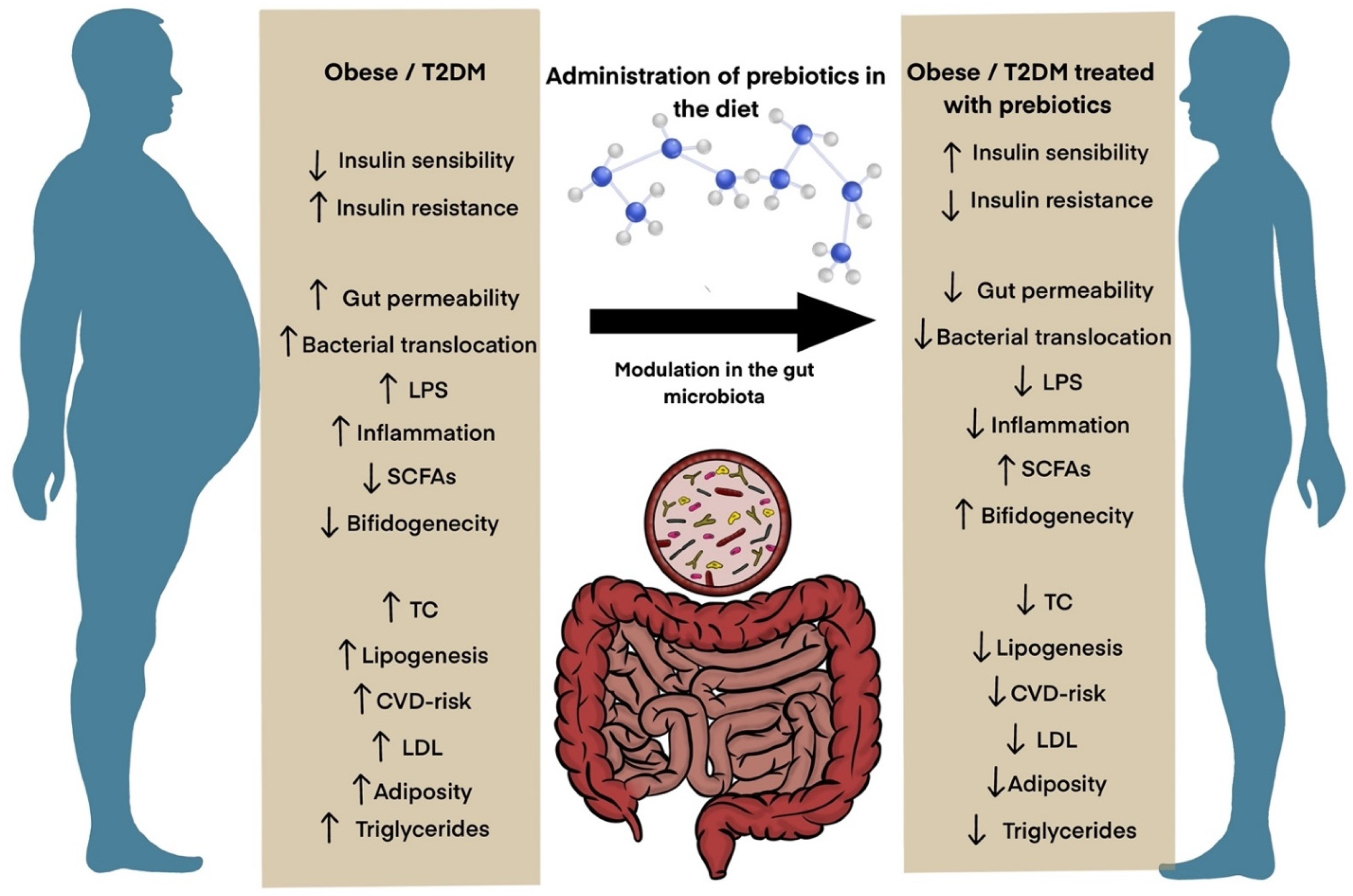
5. Prebiotics for the Treatment of Obesity and Diabetes
| Prebiotic Used | Tested Species | Dose | Re-Calculated Dose | Period | Outcomes | Reference |
|---|---|---|---|---|---|---|
| FOS | 27 women with Type-2 diabetes, age = 20–65 years; 76.0 (12.2) | 10 g/day | 131.6 mg/kg/day | 8 weeks |
| [220] |
| FOS | 10-week-old C57BL/6J mice, body weight not reported | 0.3 g/mouse/day | 8 weeks |
| [226] | |
| GOS | 6 rats alloxan-induced diabetic rats, 6 weeks old; Average weight = 90 g | 100 g/kg of diet | 1.11 g/kg of diet | 42 days |
| [227] |
| Human (women with overweight age 18–65 years and body weight not reported) | 5.5 g/day of GOS | 5.5 g/kg/day | 12 weeks |
| [228] | |
| PDX | Rats (Wistar rats age not reported and body weight 43.0 ± 4.5 g) | 5 g/100 g diet | 5 g/100 g diet | 60 days |
| [229] |
| RS | Human (over weight and obese adults—11 men and 22 women age 18–69 years and body weight not reported) | 15 g/kg/day of HAM-RS2 v. 30 g/kg/day HAM-RS21 | 15 g/kg/day of HAM-RS2 v. 30 g/kg/day HAM-RS21 | 4 weeks |
| [230] |
| Human (diabetic adults age = 55 ± 2.4 years body weight not reported) | 40 g/day of | 571.4 mg/kg/day | 12 weeks |
| [231] | |
| Lactulose | Human (patients with obesity age and body weight not reported) | 8.2 g/day | - | 2 days |
| [232] |
| AX | Rats (wild type rats with high cholesterol diet age 7 weeks body weight not reported) | 8% corn arabinoxylan | 5.8 g/kg/day | 20 days |
| [233] |
| Human (T2DM); mean age = 55 years and body weight not reported) | 49.2 g/day | 702.9 mg/kg/day | 35 days |
| [234] | |
| XOS | Rats (Male Wistar rats treated with streptozotocin to induce diabetes, age = 8 weeks; body weight = 180 ± 8 g) | 0.325 g/day | 1.81 mg/kg/day | 5 weeks |
| [235] |
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed]
- Morigny, P.; Houssier, M.; Mouisel, E.; Langin, D. Adipocyte lipolysis and insulin resistance. Biochimie 2016, 125, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Pascale, A.; Marchesi, N.; Govoni, S.; Coppola, A.; Gazzaruso, C. The role of gut microbiota in obesity, diabetes mellitus, and effect of metformin: New insights into old diseases. Curr. Opin. Pharmacol. 2019, 49, 1–5. [Google Scholar] [CrossRef]
- Arnoriaga-Rodríguez, M.; Mayneris-Perxachs, J.; Contreras-Rodríguez, O.; Burokas, A.; Ortega-Sanchez, J.-A.; Blasco, G.; Coll, C.; Biarnés, C.; Castells-Nobau, A.; Puig, J.; et al. Obesity-associated deficits in inhibitory control are phenocopied to mice through gut microbiota changes in one-carbon and aromatic amino acids metabolic pathways. Gut 2021, 70, 2283–2296. [Google Scholar] [CrossRef]
- Arnoriaga-Rodríguez, M.; Mayneris-Perxachs, J.; Burokas, A.; Contreras-Rodríguez, O.; Blasco, G.; Coll, C.; Biarnés, C.; Miranda-Olivos, R.; Latorre, J.; Moreno-Navarrete, J.-M.; et al. Obesity Impairs Short-Term and Working Memory through Gut Microbial Metabolism of Aromatic Amino Acids. Cell Metab. 2020, 32, 548–560.e7. [Google Scholar] [CrossRef] [PubMed]
- Arnoriaga-Rodríguez, M.; Mayneris-Perxachs, J.; Burokas, A.; Pérez-Brocal, V.; Moya, A.; Portero-Otin, M.; Ricart, W.; Maldonado, R.; Fernández-Real, J.-M. Gut bacterial ClpB-like gene function is associated with decreased body weight and a characteristic microbiota profile. Microbiome 2020, 8, 59. [Google Scholar] [CrossRef]
- Milani, C.; Duranti, S.; Bottacini, E.; Casey, E.; Turroni, F.; Mahony, J.; Belzer, C.; Montes, A.S.; Mancabelli, L.; Lugli, G.A.; et al. The First Microbial Colonizers of the Human Gut: Composition, Activities, and Health Implications of the Infant Gut Microbiota. Microbiol. Mol. Biol. Rev. 2017, 81, 1–67. [Google Scholar] [CrossRef]
- Adak, A.; Khan, M.R. An insight into gut microbiota and its functionalities. Cell. Mol. Life Sci. 2019, 76, 473–493. [Google Scholar] [CrossRef]
- Avershina, E.; Lundgård, K.; Sekelja, M.; Dotterud, C.; Storrø, O.; Øien, T.; Johnsen, R.; Rudi, K. Transition from infant- to adult-like gut microbiota. Environ. Microbiol. 2016, 18, 2226–2236. [Google Scholar] [CrossRef]
- Cerdó, T.; García-Santos, J.A.; Bermúdez, M.G.; Campoy, C. The role of probiotics and prebiotics in the prevention and treatment of obesity. Nutrients 2019, 11, 635. [Google Scholar] [CrossRef]
- Peterson, J.; Garges, S.; Giovanni, M.; McInnes, P.; Wang, L.; Schloss, J.A.; Bonazzi, V.; McEwen, J.E.; Wetterstrand, K.A.; Deal, C.; et al. The NIH Human Microbiome Project. Genome Res. 2009, 19, 2317–2323. [Google Scholar] [CrossRef] [PubMed]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Bello, M.G.; Godoy-Vitorino, F.; Knight, R.; Blaser, M.J. Role of the microbiome in human development. Gut 2019, 68, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Megur, A.; Baltriukienė, D.; Bukelskienė, V.; Burokas, A. The microbiota–gut–brain axis and Alzheimer’s disease: Neuroinflammation is to blame? Nutrients 2021, 13, 37. [Google Scholar] [CrossRef]
- Sommer, F.; Bäckhed, F. The gut microbiota-masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238. [Google Scholar] [CrossRef]
- Moschen, A.R.; Wieser, V.; Tilg, H. Dietary factors: Major regulators of the Gut’s microbiota. Gut Liver 2012, 6, 411–416. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The Human Microbiome Project. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- Mayneris-Perxachs, J.; Arnoriaga-Rodríguez, M.; Luque-Córdoba, D.; Priego-Capote, F.; Pérez-Brocal, V.; Moya, A.; Burokas, A.; Maldonado, R.; Fernández-Real, J.-M. Gut microbiota steroid sexual dimorphism and its impact on gonadal steroids: Influences of obesity and menopausal status. Microbiome 2020, 8, 136. [Google Scholar] [CrossRef]
- De Angelis, M.; Garruti, G.; Minervini, F.; Bonfrate, L.; Portincasa, P.; Gobbetti, M. The Food-gut Human Axis: The Effects of Diet on Gut Microbiota and Metabolome. Curr. Med. Chem. 2019, 26, 3567–3583. [Google Scholar] [CrossRef]
- Janssen, A.W.F.; Kersten, S. The role of the gut microbiota in metabolic health. FASEB J. 2015, 29, 3111–3123. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef] [PubMed]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, types, sources, mechanisms, and clinical applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.S.; Tsigalou, C.; Dalamaga, M. Probiotics, Prebiotics, Synbiotics, Postbiotics, and Obesity: Current Evidence, Controversies, and Perspectives. Curr. Obes. Rep. 2020, 9, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, T.F.; Casarotti, S.N.; de Oliveira, G.L.V.; Penna, A.L.B. The impact of probiotics, prebiotics, and synbiotics on the biochemical, clinical, and immunological markers, as well as on the gut microbiota of obese hosts. Crit. Rev. Food Sci. Nutr. 2021, 61, 337–355. [Google Scholar] [CrossRef]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Pluta, R.; Ułamek-Kozioł, M.; Januszewski, S.; Czuczwar, S.J. Gut microbiota and pro/prebiotics in Alzheimer’s disease. Aging 2020, 12, 5539–5550. [Google Scholar] [CrossRef]
- Shokryazdan, P.; Faseleh Jahromi, M.; Navidshad, B.; Liang, J.B. Effects of prebiotics on immune system and cytokine expression. Med. Microbiol. Immunol. 2017, 206, 1–9. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Macfarlane, S. Fermentation in the human large intestine: Its physiologic consequences and the potential contribution of prebiotics. J. Clin. Gastroenterol. 2011, 45, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Venegas, D.P.; De La Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short chain fatty acids (SCFAs)mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Li, Z.-R.; Green, R.S.; Holzman, I.R.; Lin, J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J. Nutr. 2009, 139, 1619–1625. [Google Scholar] [CrossRef] [PubMed]
- Gaudier, E.; Rival, M.; Buisine, M.-P.; Robineau, I.; Hoebler, C. Butyrate enemas upregulate Muc genes expression but decrease adherent mucus thickness in mice colon. Physiol. Res. 2009, 58, 111–119. [Google Scholar] [CrossRef]
- O’Keefe, S.J.D. Diet, microorganisms and their metabolites, and colon cancer. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 691–706. [Google Scholar] [CrossRef]
- Wan, M.L.Y.; Ling, K.H.; El-Nezami, H.; Wang, M.F. Influence of functional food components on gut health. Crit. Rev. Food Sci. Nutr. 2019, 59, 1927–1936. [Google Scholar] [CrossRef]
- Bengmark, S. Colonic food: Pre- and probiotics. Am. J. Gastroenterol. 2000, 95, 9–11. [Google Scholar] [CrossRef]
- Zdunczyk, Z. Physiological effect of low digestible oligosaccharides in diets for animals and humans. Pol. J. Food Nutr. Sci. 2004, 13, 115–130. [Google Scholar]
- Leeman, A.; Sjöö, M.; Eliasson, A.-C.; Björck, I. Resistant starch formation in temperature treated potato starches varying in amylose/amylopectin ratio. Carbohydr. Polym. 2006, 65, 306–313. [Google Scholar] [CrossRef]
- Dai, F.J.; Chau, C.F. Classification and regulatory perspectives of dietary fiber. J. Food Drug Anal. 2017, 25, 37–42. [Google Scholar] [CrossRef]
- Howlett, J.F.; Betteridge, V.A.; Champ, M.; Craig, S.A.S.; Meheust, A.; Jones, J.M. The definition of dietary fiber—Discussions at the Ninth Vahouny Fiber Symposium: Building scientific agreement. Food Nutr. Res. 2010, 54, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Slizewska, K.; Chlebicz-Wójcik, A. The in vitro analysis of prebiotics to be used as a component of a synbiotic preparation. Nutrients 2020, 12, 1272. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, D.P.; Mohapatra, S.; Misra, S.; Sahu, P.S. Milk derived bioactive peptides and their impact on human health—A review. Saudi J. Biol. Sci. 2016, 23, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Delzenne, N.M.; Cani, P.D.; Walter, J. Towards a more comprehensive concept for prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Simpson, H.L.; Campbell, B.J. Review article: Dietary fibre-microbiota interactions. Aliment. Pharmacol. Ther. 2015, 42, 158–179. [Google Scholar] [CrossRef]
- Hanau, S.; Almugadam, S.H.; Sapienza, E.; Cacciari, B.; Manfrinato, M.C.; Trentini, A.; Kennedy, J.F. Schematic overview of oligosaccharides, with survey on their major physiological effects and a focus on milk ones. Carbohydr. Polym. Technol. Appl. 2020, 1, 100013. [Google Scholar] [CrossRef]
- Swanson, K.S.; de Vos, W.M.; Martens, E.C.; Gilbert, J.A.; Menon, R.S.; Soto-Vaca, A.; Hautvast, J.; Meyer, P.D.; Borewicz, K.; Vaughan, E.E.; et al. Effect of fructans, prebiotics and fibres on the human gut microbiome assessed by 16S rRNA-based approaches: A review. Benef. Microbes 2020, 11, 101–129. [Google Scholar] [CrossRef]
- Singla, V.; Chakkaravarthi, S. Applications of prebiotics in food industry: A review. Food Sci. Technol. Int. 2017, 23, 649–667. [Google Scholar] [CrossRef]
- De Jesus Raposo, M.F.; de Morais, A.M.M.B.; de Morais, R.M.S.C. Emergent Sources of Prebiotics: Seaweeds and Microalgae. Mar. Drugs 2016, 14, 27. [Google Scholar] [CrossRef]
- Flesch, A.G.T.; Poziomyck, A.K.; Damin, D.C. The therapeutic use of symbiotics. Braz. Arch. Dig. Surg. 2014, 27, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for enriching next-generation health-promoting gut bacteria through prebiotics and other dietary components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Den Besten, G.; Van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef] [PubMed]
- Guarino, M.P.L.; Altomare, A.; Emerenziani, S.; Di Rosa, C.; Ribolsi, M.; Balestrieri, P.; Iovino, P.; Rocchi, G.; Cicala, M. Mechanisms of action of prebiotics and their effects on gastro-intestinal disorders in adults. Nutrients 2020, 12, 1037. [Google Scholar] [CrossRef]
- Uauy, R. Foreword. Br. J. Nutr. 2009, 101, S1. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Cardoso, B.B.; Amorim, C.; Silvério, S.C.; Rodrigues, L.R. Novel and emerging prebiotics: Advances and opportunities. Adv. Food Nutr. Res. 2021, 95, 41–95. [Google Scholar] [CrossRef]
- Kelly, G. Inulin-type prebiotics: A review (Part 2). Altern. Med. Rev. 2009, 14, 36–55. [Google Scholar]
- Yeung, C.K.; Glahn, R.P.; Welch, R.M.; Miller, D.D. Prebiotics and iron bioavailability—Is there a connection? J. Food Sci. 2005, 70, 584–590. [Google Scholar] [CrossRef]
- Ibrahim, O.O. Functional Oligosaccharide: Chemicals Structure, Manufacturing, Health Benefits, Applications and Regulations. J. Food Chem. Nanotechnol. 2018, 4, 65–76. [Google Scholar] [CrossRef]
- Roberfroid, M. Prebiotics: The concept revisited. J. Nutr. 2007, 137, 830S–837S. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.W.; Roberts, K.T. Dietary Fiber. Fulfilling the Promise of Added-Value Formulations; Academic Press: Cambridge, MA, USA, 2009; ISBN 9780123741950. [Google Scholar]
- Chatterjee, P.; Ojiambo, M. Food Ingredients; Elsevier Inc.: Amsterdam, The Netherlands, 2014; Volume 11, ISBN 9780128113721. [Google Scholar]
- Korczak, R.; Slavin, J.L. Fructooligosaccharides and appetite. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.W. Fructooligosaccharides—Occurrence, preparation, and application. Enzyme Microb. Technol. 1996, 19, 107–117. [Google Scholar] [CrossRef]
- Crittenden, R.; Playne, M. Production, properties and applications of food-grade oligosaccharides. Trends Food Sci. Technol. 1996, 7, 353–361. [Google Scholar] [CrossRef]
- Sako, T.; Tanaka, R. Prebiotics: Types. In Encyclopedia of Dairy Sciences, 2nd ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2011; pp. 354–364. [Google Scholar] [CrossRef]
- Commercially Available Oligosaccharides. Available online: https://agris.fao.org/agris-search/search.do?recordID=BE9601560 (accessed on 1 April 2022).
- Tanaka, R.; Takayama, H.; Morotomi, M.; Kuroshima, T.; Ueyama, S.; Matsumoto, K.; Kuroda, A.; Mutai, M. Effects of Administration of TOS and Bifidobacterium breve 4006 on the Human Fecal Flora. Bifidobact. Microflora 1983, 2, 17–24. [Google Scholar] [CrossRef]
- Mahoney, R.R. Galactosyl-oligosaccharide formation during lactose hydrolysis: A review. Food Chem. 1998, 63, 147–154. [Google Scholar] [CrossRef]
- Bao, Y.; Chen, C.; Newburg, D.S. Quantification of neutral human milk oligosaccharides by graphitic carbon high-performance liquid chromatography with tandem mass spectrometry. Anal. Biochem. 2013, 433, 28–35. [Google Scholar] [CrossRef]
- Thongaram, T.; Hoeflinger, J.L.; Chow, J.; Miller, M.J. Human milk oligosaccharide consumption by probiotic and human-associated bifidobacteria and lactobacilli. J. Dairy Sci. 2017, 100, 7825–7833. [Google Scholar] [CrossRef]
- Akkerman, R.; Faas, M.M.; de Vos, P. Non-digestible carbohydrates in infant formula as substitution for human milk oligosaccharide functions: Effects on microbiota and gut maturation. Crit. Rev. Food Sci. Nutr. 2019, 59, 1486–1497. [Google Scholar] [CrossRef]
- Xu, G.; Davis, J.C.; Goonatilleke, E.; Smilowitz, J.T.; German, J.B.; Lebrilla, C.B. Absolute Quantitation of Human Milk Oligosaccharides Reveals Phenotypic Variations during Lactation. J. Nutr. 2017, 147, 117–124. [Google Scholar] [CrossRef]
- Chaturvedi, P.; Warren, C.D.; Altaye, M.; Morrow, A.L.; Ruiz-Palacios, G.; Pickering, L.K.; Newburg, D.S. Fucosylated human milk oligosaccharides vary between individuals and over the course of lactation. Glycobiology 2001, 11, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Thurl, S.; Munzert, M.; Henker, J.; Boehm, G.; Müller-Werner, B.; Jelinek, J.; Stahl, B. Variation of human milk oligosaccharides in relation to milk groups and lactational periods. Br. J. Nutr. 2010, 104, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Thomson, P.; Medina, D.A.; Garrido, D. Human milk oligosaccharides and infant gut bifidobacteria: Molecular strategies for their utilization. Food Microbiol. 2018, 75, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef]
- Cheng, Y.-J.; Yeung, C.-Y. Recent advance in infant nutrition: Human milk oligosaccharides. Pediatr. Neonatol. 2021, 62, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Morozov, V.; Hansman, G.; Hanisch, F.G.; Schroten, H.; Kunz, C. Human Milk Oligosaccharides as Promising Antivirals. Mol. Nutr. Food Res. 2018, 62, 1–46. [Google Scholar] [CrossRef]
- Asakuma, S.; Hatakeyama, E.; Urashima, T.; Yoshida, E.; Katayama, T.; Yamamoto, K.; Kumagai, H.; Ashida, H.; Hirose, J.; Kitaoka, M. Physiology of Consumption of Human Milk Oligosaccharides by Infant Gut-associated Bifidobacteria. J. Biol. Chem. 2011, 286, 34583–34592. [Google Scholar] [CrossRef]
- Vandenplas, Y.; Berger, B.; Carnielli, V.P.; Ksiazyk, J.; Lagström, H.; Luna, M.S.; Migacheva, N.; Mosselmans, J.M.; Picaud, J.C.; Possner, M.; et al. Human milk oligosaccharides: 2’-fucosyllactose (2’-FL) and lacto-n-neotetraose (LNnT) in infant formula. Nutrients 2018, 10, 1161. [Google Scholar] [CrossRef]
- Corona, L.; Lussu, A.; Bosco, A.; Pintus, R.; Cesare Marincola, F.; Fanos, V.; Dessì, A. Human Milk Oligosaccharides: A Comprehensive Review towards Metabolomics. Children 2021, 8, 804. [Google Scholar] [CrossRef]
- Marcobal, A.; Barboza, M.; Froehlich, J.W.; Block, D.E.; German, J.B.; Lebrilla, C.B.; Mills, D.A. Consumption of human milk oligosaccharides by gut-related microbes. J. Agric. Food Chem. 2010, 58, 5334–5340. [Google Scholar] [CrossRef]
- Donovan, S.M.; Comstock, S.S. Human Milk Oligosaccharides Influence Neonatal Mucosal and Systemic Immunity. Ann. Nutr. Metab. 2016, 69 (Suppl. 2), 42–51. [Google Scholar] [CrossRef] [PubMed]
- Comstock, S.S.; Li, M.; Wang, M.; Monaco, M.H.; Kuhlenschmidt, T.B.; Kuhlenschmidt, M.S.; Donovan, S.M. Dietary Human Milk Oligosaccharides but Not Prebiotic Oligosaccharides Increase Circulating Natural Killer Cell and Mesenteric Lymph Node Memory T Cell Populations in Noninfected and Rotavirus-Infected Neonatal Piglets. J. Nutr. 2017, 147, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Bode, L. The functional biology of human milk oligosaccharides. Early Hum. Dev. 2015, 91, 619–622. [Google Scholar] [CrossRef]
- Jacobi, S.K.; Yatsunenko, T.; Li, D.; Dasgupta, S.; Yu, R.K.; Berg, B.M.; Chichlowski, M.; Odle, J. Dietary Isomers of Sialyllactose Increase Ganglioside Sialic Acid Concentrations in the Corpus Callosum and Cerebellum and Modulate the Colonic Microbiota of Formula-Fed Piglets. J. Nutr. 2016, 146, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Rousseaux, A.; Brosseau, C.; Le Gall, S.; Piloquet, H.; Barbarot, S.; Bodinier, M. Human Milk Oligosaccharides: Their Effects on the Host and Their Potential as Therapeutic Agents. Front. Immunol. 2021, 12, 680911. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Tu, Y.; Ren, F.; Zhang, H. Characterization and functional properties of Maillard reaction products of β-lactoglobulin and polydextrose. Food Chem. 2021, 377, 131749. [Google Scholar] [CrossRef]
- Lahtinen, S.J.; Knoblock, K.; Drakoularakou, A.; Jacob, M.; Stowell, J.; Gibson, G.R.; Ouwehand, A.C. Effect of molecule branching and glycosidic linkage on the degradation of polydextrose by gut microbiota. Biosci. Biotechnol. Biochem. 2010, 74, 2016–2021. [Google Scholar] [CrossRef]
- Caselato, V.; Ouwehand, A.; Flavors, I.; Andreollo, N.A. Polydextrose: Physiological Function, and Effects on Health. Nutrients 2016, 8, 553. [Google Scholar] [CrossRef]
- Zhong, J.; Luo, B.; Xiang, M.; Liu, H.; Zhai, Z.; Wang, T.; Craig, S.A.S. Studies on the effects of polydextrose intake on physiologic funtions in Chinese people. Am. J. Clin. Nutr. 2000, 72, 1503–1509. [Google Scholar] [CrossRef]
- Yao, N.; Paez, A.V.; White, P.J. Structure and function of starch and resistant starch from corn with different doses of mutant amylose-extender and floury-1 alleles. J. Agric. Food Chem. 2009, 57, 2040–2048. [Google Scholar] [CrossRef]
- Fuentes-Zaragoza, E.; Sánchez-Zapata, E.; Sendra, E.; Sayas, E.; Navarro, C.; Fernández-Lõpez, J.; Pérez-Alvarez, J.A. Resistant starch as prebiotic: A review. Starch/Staerke 2011, 63, 406–415. [Google Scholar] [CrossRef]
- Marlatt, K.L.; White, U.A.; Beyl, R.A.; Peterson, C.M.; Corby, K.; Marco, M.L.; Keenan, M.J.; Martin, R.J.; Aryana, K.J.; Ravussin, E.; et al. Role of resistant starch on diabetes risk factors in people with prediabetes: Design, conduct, and baseline results of the STARCH trial. Contemp. Clin. Trials 2018, 65, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Tingirikari, J.M.R. Microbiota-accessible pectic poly- and oligosaccharides in gut health. Food Funct. 2018, 9, 5059–5073. [Google Scholar] [CrossRef]
- Yoo, H.D.; Kim, D.; Paek, S.H.; Oh, S.E. Plant cell wall polysaccharides as potential resources for the development of novel prebiotics. Biomol. Ther. 2012, 20, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Maldonado-Gómez, M.X.; Hutkins, R.W.; Rose, D.J. Production and in vitro Fermentation of Soluble, Non-digestible, Feruloylated Oligo- and Polysaccharides from Maize and Wheat Brans. J. Agric. Food Chem. 2014, 62, 159–166. [Google Scholar] [CrossRef]
- Moon, J.S.; Shin, S.Y.; Choi, H.S.; Joo, W.; Cho, S.K.; Li, L.; Kang, J.-H.; Kim, T.-J.; Han, N.S. In vitro digestion and fermentation properties of linear sugar-beet arabinan and its oligosaccharides. Carbohydr. Polym. 2015, 131, 50–56. [Google Scholar] [CrossRef]
- Wilkowska, A.; Nowak, A.; Antczak-Chrobot, A.; Motyl, I.; Czyżowska, A.; Paliwoda, A. Structurally Different Pectic Oligosaccharides Produced from Apple Pomace and Their Biological Activity In Vitro. Foods 2019, 8, 365. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, P.; Zhang, H. Pectin in cancer therapy: A review. Trends Food Sci. Technol. 2015, 44, 258–271. [Google Scholar] [CrossRef]
- Vera, C.; Córdova, A.; Aburto, C.; Guerrero, C.; Suárez, S.; Illanes, A. Synthesis and purification of galacto-oligosaccharides: State of the art. World J. Microbiol. Biotechnol. 2016, 32, 197. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, T.; Chen, X.; Pang, X.; Zhang, S.; Obaroakpo, J.U.; Shilong, J.; Lu, J.; Lv, J. Absolute quantification of twelve oligosaccharides in human milk using a targeted mass spectrometry-based approach. Carbohydr. Polym. 2019, 219, 328–333. [Google Scholar] [CrossRef]
- German, J.; Freeman, S.; Lebrilla, C.; Mills, D. Human milk oligosaccharides: Evolution, structures and bioselectivity as substrates for intestinal bacteria. Nestle Nutr. Work. Ser. Pediatr. Progr. 2008, 62, 205–218. [Google Scholar] [CrossRef]
- Ackerman, D.L.; Craft, K.M.; Townsend, S.D. Infant food applications of complex carbohydrates: Structure, synthesis, and function. Carbohydr. Res. 2017, 437, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Tungland, B.C.; Meyer, D. Nondigestible oligo-and polysaccharides (dietary fiber): Their physiology and role in human health and food. Compr. Rev. Food Sci. Food Saf. 2002, 1, 90–109. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Liu, Q.; Yu, S.; Zhang, T.; Mu, W. Synthesis of Lactosucrose Using a Recombinant Levansucrase from Brenneria goodwinii. Appl. Biochem. Biotechnol. 2018, 186, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Langenaeken, N.A.; De Schutter, D.P.; Courtin, C.M. Arabinoxylan from non-malted cereals can act as mouthfeel contributor in beer. Carbohydr. Polym. 2020, 239, 116257. [Google Scholar] [CrossRef] [PubMed]
- Gérard, D.; Méline, T.; Muzard, M.; Deleu, M.; Plantier-Royon, R.; Rémond, C. Enzymatically-synthesized xylo-oligosaccharides laurate esters as surfactants of interest. Carbohydr. Res. 2020, 495, 108090. [Google Scholar] [CrossRef] [PubMed]
- Aachary, A.A.; Prapulla, S.G. Corncob-Induced endo-1,4-β-d-Xylanase of Aspergillus oryzae MTCC 5154: Production and Characterization of Xylobiose from Glucuronoxylan. J. Agric. Food Chem. 2008, 56, 3981–3988. [Google Scholar] [CrossRef]
- Carvalho, A.F.A.; de Oliva Neto, P.; da Silva, D.F.; Pastore, G.M. Xylo-oligosaccharides from lignocellulosic materials: Chemical structure, health benefits and production by chemical and enzymatic hydrolysis. Food Res. Int. 2013, 51, 75–85. [Google Scholar] [CrossRef]
- Shendurse, A.M.; Khedkar, C.D. Lactose. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 509–516. ISBN 978-0-12-384953-3. [Google Scholar]
- Panesar, P.S.; Bali, V. Prebiotics. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 464–471. ISBN 978-0-12-384953-3. [Google Scholar]
- Sivaprakasam, S.; Prasad, P.D.; Singh, N. Benefits of short-chain fatty acids and their receptors in inflammation and carcinogenesis. Pharmacol. Ther. 2016, 164, 144–151. [Google Scholar] [CrossRef]
- Prasad, S.; Dhiman, R.K.; Duseja, A.; Chawla, Y.K.; Sharma, A.; Agarwal, R. Lactulose improves cognitive functions and health-related quality of life in patients with cirrhosis who have minimal hepatic encephalopathy. Hepatology 2007, 45, 549–559. [Google Scholar] [CrossRef]
- Elkington, S.G. Lactulose. Gut 1970, 11, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Zeng, J.; Jia, T.; Gao, H.; Zhang, R.; Jiang, J.; Li, G.; Su, T. Effects of sialylated lactulose on the mouse intestinal microbiome using Illumina high-throughput sequencing. Appl. Microbiol. Biotechnol. 2019, 103, 9067–9076. [Google Scholar] [CrossRef] [PubMed]
- Mu, W.; Chen, Q.; Wang, X.; Zhang, T.; Jiang, B. Current studies on physiological functions and biological production of lactosucrose. Appl. Microbiol. Biotechnol. 2013, 97, 7073–7080. [Google Scholar] [CrossRef] [PubMed]
- Fernando, W.M.U.; Hill, J.E.; Zello, G.A.; Tyler, R.T.; Dahl, W.J.; Van Kessel, A.G. Diets supplemented with chickpea or its main oligosaccharide component raffinose modify faecal microbial composition in healthy adults. Benef. Microbes 2010, 1, 197–207. [Google Scholar] [CrossRef]
- Silvério, S.C.; Macedo, E.A.; Teixeira, J.A.; Rodrigues, L.R. Perspectives on the biotechnological production and potential applications of lactosucrose: A review. J. Funct. Foods 2015, 19, 74–90. [Google Scholar] [CrossRef]
- Terada, A.; Hara, H.; Oishi, T.; Matsui, S.; Mitsuoka, T.; Nakajyo, S.; Fujimori, I.; Hara, K. Effect of Dietary Lactosucrose on Faecal Flora and Faecal Metabolites of Dogs. Microb. Ecol. Health Dis. 1992, 5, 87–92. [Google Scholar] [CrossRef]
- Hino, K.; Kurose, M.; Sakurai, T.; Inoue, S.; Oku, K.; Chaen, H.; Fukuda, S. Effect of Dietary Lactosucrose (4G-β-D-Galactosylsucrose) on the Intestinal Immune Functions in Mice. J. Appl. Glycosci. 2007, 54, 169–172. [Google Scholar] [CrossRef][Green Version]
- Hoffman, W.F.; Gortner, R.A. The preparation and analysis of the various proteins of wheat flour with special reference to the globulin, albumin and proteose fractions. Cereal Chem. 2021, 4, 221–229. [Google Scholar]
- Bastos, R.; Coelho, E.; Coimbra, M.A. 8-Arabinoxylans from cereal by-products: Insights into structural features, recovery, and applications. In Woodhead Publishing Series in Food Science, Technology and Nutrition; Galanakis, C., Ed.; Woodhead Publishing: Sawston, UK, 2018; pp. 227–251. ISBN 978-0-08-102162-0. [Google Scholar]
- Izydorczyk, M.S. Arabinoxylans. In Handbook of Hydrocolloids, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 653–692. [Google Scholar] [CrossRef]
- Paz-Samaniego, R.; Méndez-Encinas, M.; Fierro-Islas, J.M.; Marquez-Escalante, J.; Rascón-Chu, A.; Martinez-Lopez, A.L.; Carvajal-Millan, E. Ferulated arabinoxylans recovered from low-value maize by-products: Gelation and antioxidant capacity. In Ferulic Acid: Antioxidant Properties, Uses and Potential Health Benefits; Nova Science Publishers: Hauppauge, NY, USA, 2014; pp. 151–164. [Google Scholar]
- Mendis, M.; Simsek, S. Arabinoxylans and human health. Food Hydrocoll. 2014, 42, 239–243. [Google Scholar] [CrossRef]
- Lu, Z.X.; Walker, K.Z.; Muir, J.G.; Mascara, T.; O’Dea, K. Arabinoxylan fiber, a byproduct of wheat flour processing, reduces the postprandial glucose response in normoglycemic subjects. Am. J. Clin. Nutr. 2000, 71, 1123–1128. [Google Scholar] [CrossRef]
- Mendis, M.; Leclerc, E.; Simsek, S. Arabinoxylans, gut microbiota and immunity. Carbohydr. Polym. 2016, 139, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Althubiani, A.S.; Al-Ghamdi, S.B.; Samreen; Qais, F.A.; Khan, M.S.; Ahmad, I.; Malak, H.A. Plant-Derived Prebiotics and Its Health Benefits; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128146200. [Google Scholar]
- Gibson, G.R.; Probert, H.M.; Van Loo, J.; Rastall, R.A.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef] [PubMed]
- Mitmesser, S.; Combs, M. Prebiotics: Inulin and Other Oligosaccharides; Elsevier Inc.: Amsterdam, The Netherlands, 2017; ISBN 9780128040621. [Google Scholar]
- Lin, S.-H.; Chou, L.-M.; Chien, Y.-W.; Chang, J.-S.; Lin, C.-I. Prebiotic Effects of Xylooligosaccharides on the Improvement of Microbiota Balance in Human Subjects. Gastroenterol. Res. Pract. 2016, 2016, 5789232. [Google Scholar] [CrossRef] [PubMed]
- Parajó, J.C.; Garrote, G.; Cruz, J.M.; Dominguez, H. Production of xylooligosaccharides by autohydrolysis of lignocellulosic materials. Trends Food Sci. Technol. 2004, 15, 115–120. [Google Scholar] [CrossRef]
- Scott, K.P.; Martin, J.C.; Duncan, S.H.; Flint, H.J. Prebiotic stimulation of human colonic butyrate-producing bacteria and bifidobacteria, in vitro. FEMS Microbiol. Ecol. 2014, 87, 30–40. [Google Scholar] [CrossRef]
- Tuohy, K.; Rouzaud, G.; Bruck, W.; Gibson, G. Modulation of the Human Gut Microflora Towards Improved Health Using Prebiotics—Assessment of Efficacy. Curr. Pharm. Des. 2005, 11, 75–90. [Google Scholar] [CrossRef]
- Modler, H.W. Bifidogenic factors—Sources, metabolism and applications. Int. Dairy J. 1994, 4, 383–407. [Google Scholar] [CrossRef]
- Sabater-Molina, M.; Larqué, E.; Torrella, F.; Zamora, S. Dietary fructooligosaccharides and potential benefits on health. J. Physiol. Biochem. 2009, 65, 315–328. [Google Scholar] [CrossRef]
- Carabin, I.G.; Flamm, W.G. Evaluation of safety of inulin and oligofructose as dietary fiber. Regul. Toxicol. Pharmacol. 1999, 30, 268–282. [Google Scholar] [CrossRef]
- Elison, E.; Vigsnaes, L.K.; Rindom Krogsgaard, L.; Rasmussen, J.; Sørensen, N.; McConnell, B.; Hennet, T.; Sommer, M.O.A.; Bytzer, P. Oral supplementation of healthy adults with 2’-O-fucosyllactose and lacto-N-neotetraose is well tolerated and shifts the intestinal microbiota. Br. J. Nutr. 2016, 116, 1356–1368. [Google Scholar] [CrossRef]
- Chen, L.; Liu, R.; Qin, C.; Meng, Y.; Zhang, J.; Wang, Y.; Xu, G. Sources and intake of resistant starch in the Chinese diet. Asia Pac. J. Clin. Nutr. 2010, 19, 274–282. [Google Scholar] [PubMed]
- Brouns, F.; Theuwissen, E.; Adam, A.; Bell, M.; Berger, A.; Mensink, R.P. Cholesterol-lowering properties of different pectin types in mildly hyper-cholesterolemic men and women. Eur. J. Clin. Nutr. 2012, 66, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Salminen, E. Lactulose, lactic acid bacteria, intestinal microecology and mucosal protection. Scand. J. Gastroenterol. Suppl. 1997, 222, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Barreteau, H.; Delattre, C.; Michaud, P. Production of oligosaccharides as promising new food additive generation. Food Technol. Biotechnol. 2006, 44, 323–333. [Google Scholar]
- Denji, K.A.; Mansour, M.R.; Akrami, R.; Ghobadi, S.; Jafarpour, S.A.; Mirbeygi, S.K. Effect of dietary prebiotic mannan oligosaccharide (mos) on growth performance, intestinal microflora, body composition, haematological and blood serum biochemical parameters of rainbow trout (oncorhynchus mykiss) juveniles. J. Fish. Aquat. Sci. 2015, 10, 255–265. [Google Scholar] [CrossRef]
- Klatt, N.R.; Canary, L.A.; Sun, X.; Vinton, C.L.; Funderburg, N.T.; Morcock, D.R.; Quiñones, M.; Deming, C.B.; Perkins, M.; Hazuda, D.J.; et al. Probiotic/prebiotic supplementation of antiretrovirals improves gastrointestinal immunity in SIV-infected macaques. J. Clin. Investig. 2013, 123, 903–907. [Google Scholar] [CrossRef]
- Champagne, C.P. Some Technological Challenges in the Addition of Probiotic Bacteria to Foods; Springer: New York, NY, USA, 2009; ISBN 9780387790572. [Google Scholar]
- Akutko, K.; Stawarski, A. Probiotics, prebiotics and synbiotics in inflammatory bowel diseases. J. Clin. Med. 2021, 10, 2466. [Google Scholar] [CrossRef]
- Cunningham, M.; Azcarate-Peril, M.A.; Barnard, A.; Benoit, V.; Grimaldi, R.; Guyonnet, D.; Holscher, H.D.; Hunter, K.; Manurung, S.; Obis, D.; et al. Shaping the Future of Probiotics and Prebiotics. Trends Microbiol. 2021, 29, 667–685. [Google Scholar] [CrossRef]
- Wan, X.; Guo, H.; Liang, Y.; Zhou, C.; Liu, Z.; Li, K.; Niu, F.; Zhai, X.; Wang, L. The physiological functions and pharmaceutical applications of inulin: A review. Carbohydr. Polym. 2020, 246, 116589. [Google Scholar] [CrossRef]
- Rezende, E.S.V.; Lima, G.C.; Naves, M.M.V. Dietary fibers as beneficial microbiota modulators: A proposal classification by prebiotic categories. Nutrition 2021, 89, 111217. [Google Scholar] [CrossRef]
- Yu, X.; Gurry, T.; Nguyen, L.T.T.; Richardson, H.S.; Alm, E.J. Prebiotics and Community Composition Influence Gas Production of the Human Gut Microbiota. MBio 2020, 11, e00217-20. [Google Scholar] [CrossRef] [PubMed]
- Kleessen, B.; Sykura, B.; Zunft, H.J.; Blaut, M. Effects of inulin and lactose on fecal microflora, microbial activity, and bowel habit in elderly constipated persons. Am. J. Clin. Nutr. 1997, 65, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Finlay, R.K.; Wynne, A.G.; Gibson, G.R. A Human Volunteer Study on the Prebiotic Effects of HP-Inulin—Faecal Bacteria Enumerated Using Fluorescent In Situ Hybridisation (FISH). Anaerobe 2001, 7, 113–118. [Google Scholar] [CrossRef]
- Roland, N.; Nugon-Baudon, L.; Andrieux, C.; Szylit, O. Comparative study of the fermentative characteristics of inulin and different types of fibre in rats inoculated with a human Whole faecal flora. Br. J. Nutr. 1995, 74, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Flores-Maltos, D.A.; Mussatto, S.I.; Contreras-Esquivel, J.C.; Rodríguez-Herrera, R.; Teixeira, J.A.; Aguilar, C.N. Biotechnological production and application of fructooligosaccharides. Crit. Rev. Biotechnol. 2016, 36, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Sakaguchi, E.; Bai, G. Short-term supplementation with dietary fructooligosaccharide and dietary mannitol elevated the absorption of calcium and magnesium in adult rats. Czech J. Anim. Sci. 2016, 61, 281–289. [Google Scholar] [CrossRef]
- Sangeetha, P.T.; Ramesh, M.N.; Prapulla, S.G. Recent trends in the microbial production, analysis and application of Fructooligosaccharides. Trends Food Sci. Technol. 2005, 16, 442–457. [Google Scholar] [CrossRef]
- Maathuis, A.J.H.; van den Heuvel, E.G.; Schoterman, M.H.C.; Venema, K. Galacto-Oligosaccharides Have Prebiotic Activity in a Dynamic In Vitro Colon Model Using a 13C-Labeling Technique. J. Nutr. 2012, 142, 1205–1212. [Google Scholar] [CrossRef]
- Davis, L.M.G.; Martínez, I.; Walter, J.; Goin, C.; Hutkins, R.W. Barcoded pyrosequencing reveals that consumption of galactooligosaccharides results in a highly specific bifidogenic response in humans. PLoS ONE 2011, 6, e25200. [Google Scholar] [CrossRef]
- Van Loo, J.; Cummings, J.; Delzenne, N.; Englyst, H.; Franck, A.; Hopkins, M.; Kok, N.; Macfarlane, G.; Newton, D.; Quigley, M.; et al. Functional food properties of non-digestible oligosaccharides: A consensus report from the ENDO project (DGXII AIRII-CT94-1095). Br. J. Nutr. 1999, 81, 121–132. [Google Scholar] [CrossRef]
- Tran, N.T.; Tang, Y.; Li, Z.; Zhang, M.; Wen, X.; Ma, H.; Li, S. Galactooligosaccharides and Resistant Starch Altered Microbiota and Short-Chain Fatty Acids in an in vitro Fermentation Study Using Gut Contents of Mud Crab (Scylla paramamosain). Front. Microbiol. 2020, 11, 1352. [Google Scholar] [CrossRef] [PubMed]
- Burokas, A.; Arboleya, S.; Moloney, R.D.; Peterson, V.L.; Murphy, K.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Targeting the Microbiota-Gut-Brain Axis: Prebiotics Have Anxiolytic and Antidepressant-like Effects and Reverse the Impact of Chronic Stress in Mice. Biol. Psychiatry 2017, 82, 472–487. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.; Cowen, P.J.; Harmer, C.J.; Tzortzis, G.; Errington, S.; Burnet, P.W.J. Prebiotic intake reduces the waking cortisol response and alters emotional bias in healthy volunteers. Psychopharmacology 2015, 232, 1793–1801. [Google Scholar] [CrossRef] [PubMed]
- Ray, C.; Kerketta, J.A.; Rao, S.; Patel, S.; Dutt, S.; Arora, K.; Pournami, F.; Bhushan, P. Human Milk Oligosaccharides: The Journey Ahead. Int. J. Pediatr. 2019, 2019, 2390240. [Google Scholar] [CrossRef]
- LoCascio, R.G.; Ninonuevo, M.R.; Freeman, S.L.; Sela, D.A.; Grimm, R.; Lebrilla, C.B.; Mills, D.A.; German, J.B. Glycoprofiling of bifidobacterial consumption of human milk oligosaccharides demonstrates strain specific, preferential consumption of small chain glycans secreted in early human lactation. J. Agric. Food Chem. 2007, 55, 8914–8919. [Google Scholar] [CrossRef]
- Bunesova, V.; Lacroix, C.; Schwab, C. Fucosyllactose and L-fucose utilization of infant Bifidobacterium longum and Bifidobacterium kashiwanohense. BMC Microbiol. 2016, 16, 248. [Google Scholar] [CrossRef]
- Gibson, G.R.; Wang, X. Regulatory effects of bifidobacteria on the growth of other colonic bacteria. J. Appl. Bacteriol. 1994, 77, 412–420. [Google Scholar] [CrossRef]
- Collado, M.C.; Cernada, M.; Baüerl, C.; Vento, M.; Pérez-Martínez, G. Microbial ecology and host-microbiota interactions during early life stages. Gut Microbes 2012, 3, 352–365. [Google Scholar] [CrossRef]
- Schwab, C.; Ruscheweyh, H.J.; Bunesova, V.; Pham, V.T.; Beerenwinkel, N.; Lacroix, C. Trophic interactions of infant bifidobacteria and eubacterium hallii during L-fucose and fucosyllactose degradation. Front. Microbiol. 2017, 8, 95. [Google Scholar] [CrossRef]
- Weichert, S.; Jennewein, S.; Hüfner, E.; Weiss, C.; Borkowski, J.; Putze, J.; Schroten, H. Bioengineered 2′-fucosyllactose and 3-fucosyllactose inhibit the adhesion of Pseudomonas aeruginosa and enteric pathogens to human intestinal and respiratory cell lines. Nutr. Res. 2013, 33, 831–838. [Google Scholar] [CrossRef]
- Reverri, E.J.; Devitt, A.A.; Kajzer, J.A.; Baggs, G.E.; Borschel, M.W. Review of the clinical experiences of feeding infants formula containing the human milk oligosaccharide 2′-fucosyllactose. Nutrients 2018, 10, 1346. [Google Scholar] [CrossRef] [PubMed]
- Holscher, H.D.; Caporaso, J.G.; Hooda, S.; Brulc, J.M.; Fahey, G.C.J.; Swanson, K.S. Fiber supplementation influences phylogenetic structure and functional capacity of the human intestinal microbiome: Follow-up of a randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Hooda, S.; Boler, B.M.V.; Serao, M.C.R.; Brulc, J.M.; Staeger, M.A.; Boileau, T.W.; Dowd, S.E.; Fahey, G.C.J.; Swanson, K.S. 454 pyrosequencing reveals a shift in fecal microbiota of healthy adult men consuming polydextrose or soluble corn fiber. J. Nutr. 2012, 142, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Toporovski, M.S.; de Morais, M.B.; Abuhab, A.; Crippa Júnior, M.A. Effect of polydextrose/fructooligosaccharide mixture on constipation symptoms in children aged 4 to 8 years. Nutrients 2021, 13, 1634. [Google Scholar] [CrossRef] [PubMed]
- Costabile, A.; Fava, F.; Röytiö, H.; Forssten, S.D.; Olli, K.; Klievink, J.; Rowland, I.R.; Ouwehand, A.C.; Rastall, R.A.; Gibson, G.R.; et al. Impact of polydextrose on the faecal microbiota: A double-blind, crossover, placebo-controlled feeding study in healthy human subjects. Br. J. Nutr. 2012, 108, 471–481. [Google Scholar] [CrossRef]
- Tachon, S.; Zhou, J.; Keenan, M.; Martin, R.; Marco, M.L. The intestinal microbiota in aged mice is modulated by dietary resistant starch and correlated with improvements in host responses. FEMS Microbiol. Ecol. 2013, 83, 299–309. [Google Scholar] [CrossRef]
- Brüssow, H. Microbiota and healthy ageing: Observational and nutritional intervention studies. Microb. Biotechnol. 2013, 6, 326–334. [Google Scholar] [CrossRef]
- Charrier, J.A.; Martin, R.J.; McCutcheon, K.L.; Raggio, A.M.; Goldsmith, F.; Goita, M.; Senevirathne, R.N.; Brown, I.L.; Pelkman, C.; Zhou, J.; et al. High fat diet partially attenuates fermentation responses in rats fed resistant starch from high-amylose maize. Obesity 2013, 21, 2350–2355. [Google Scholar] [CrossRef]
- Gullón, B.; Gómez, B.; Martínez-Sabajanes, M.; Yáñez, R.; Parajó, J.C.; Alonso, J.L. Pectic oligosaccharides: Manufacture and functional properties. Trends Food Sci. Technol. 2013, 30, 153–161. [Google Scholar] [CrossRef]
- Gómez, B.; Gullón, B.; Yáñez, R.; Schols, H.; Alonso, J.L. Prebiotic potential of pectins and pectic oligosaccharides derived from lemon peel wastes and sugar beet pulp: A comparative evaluation. J. Funct. Foods 2016, 20, 108–121. [Google Scholar] [CrossRef]
- Leijdekkers, A.G.M.; Aguirre, M.; Venema, K.; Bosch, G.; Gruppen, H.; Schols, H.A. In vitro fermentability of sugar beet pulp derived oligosaccharides using human and pig fecal inocula. J. Agric. Food Chem. 2014, 62, 1079–1087. [Google Scholar] [CrossRef]
- Gibson, G.; Angus, F. Leatherhead Ingredients Handbook: Prebiotics and Probiotics; Springer: New York, NY, USA; Wiley–Blackwell: Hoboken, NJ, USA, 2000. [Google Scholar]
- Terada, A.; Hara, H.; Kato, S.; Kimura, T.; Fujimori, I.; Hara, K.; Maruyama, T.; Mitsuoka, T. Effect of Lactosucrose (4G-β-D-Galactosylsucrose) on Fecal Flora and Fecal Putrefactive Products of Cats. J. Vet. Med. Sci. 1993, 55, 291–295. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ballongue, J.; Schumann, C.; Quignon, P. Effects of lactulose and lactitol on colonic microflora and enzymatic activity. Scand. J. Gastroenterol. Suppl. 1997, 222, 41–44. [Google Scholar] [CrossRef] [PubMed]
- García-Cayuela, T.; Díez-Municio, M.; Herrero, M.; Martínez-Cuesta, M.C.; Peláez, C.; Requena, T.; Moreno, F.J. Selective fermentation of potential prebiotic lactose-derived oligosaccharides by probiotic bacteria. Int. Dairy J. 2014, 38, 11–15. [Google Scholar] [CrossRef]
- Li, W.; Yu, S.; Zhang, T.; Jiang, B.; Stressler, T.; Fischer, L.; Mu, W. Efficient Biosynthesis of Lactosucrose from Sucrose and Lactose by the Purified Recombinant Levansucrase from Leuconostoc mesenteroides B-512 FMC. J. Agric. Food Chem. 2015, 63, 9755–9763. [Google Scholar] [CrossRef]
- Maruyama, T.; Terada, A.; Ogata, Y.; Fujita, K.; Ishigami, H.; Hara, K.; Pet, T.; Co, F.; Kaijo, T.; Shiba, B. Effective Dose of Lactosucrose on Fecal Flora and Fecal Metabolites of Humans vitro by bifidobacteria and lactobacilli, and to a limited degree by the The effects of dietary lactosucrose on the fecal flora and fecal metab- olites were studied in eight he. Bifidobact. Microflora 1994, 13, 51–63. [Google Scholar]
- Nath, A.; Haktanirlar, G.; Varga, Á.; Molnár, M.A.; Albert, K.; Galambos, I.; Koris, A.; Vatai, G. Biological Activities of Lactose-Derived Prebiotics and Symbiotic with Probiotics on Gastrointestinal System. Medicina 2018, 54, 18. [Google Scholar] [CrossRef]
- Díez-Municio, M.; Herrero, M.; Olano, A.; Moreno, F.J. Synthesis of novel bioactive lactose-derived oligosaccharides by microbial glycoside hydrolases. Microb. Biotechnol. 2014, 7, 315–331. [Google Scholar] [CrossRef]
- Kihara, M.; Ohba, K.; Sakata, T. Trophic effect of dietary lactosucrose on intestinal tunica muscularis and utilization of this sugar by gut microbes in red seabream Pagrus major, a marine carnivorous teleost, under artificial rearing. Comp. Biochem. Physiol. Part A Physiol. 1995, 112, 629–634. [Google Scholar] [CrossRef]
- Kihara, M.; Sakata, T. Production of short-chain fatty acids and gas from various oligosaccharides by gut microbes of carp (Cyprinus carpio L.) in micro-scale batch culture. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2002, 132, 333–340. [Google Scholar] [CrossRef]
- Kihara, M.; Sakata, T. Influences of incubation temperature and various saccharides on the production of organic acids and gases by gut microbes of rainbow trout Oncorhynchus mykiss in a micro-scale batch culture. J. Comp. Physiol. B Biochem. Syst. Environ. Physiol. 2001, 171, 441–447. [Google Scholar] [CrossRef]
- Kamel, R.; Afifi, S.M.; Kassem, I.A.A.; Elkasabgy, N.A.; Farag, M.A. Arabinoxylan and rhamnogalacturonan mucilage: Outgoing and potential trends of pharmaceutical, environmental, and medicinal merits. Int. J. Biol. Macromol. 2020, 165, 2550–2564. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, M.J.; Englyst, H.N.; Macfarlane, S.; Furrie, E.; Macfarlane, G.T.; McBain, A.J. Degradation of Cross-Linked and Non-Cross-Linked Arabinoxylans by the Intestinal Microbiota in Children. Appl. Environ. Microbiol. 2003, 69, 6354–6360. [Google Scholar] [CrossRef] [PubMed]
- Glitsø, L.V.; Gruppen, H.; Schols, H.A.; Højsgaard, S.; Sandström, B.; Bach Knudsen, K.E. Degradation of rye arabinoxylans in the large intestine of pigs. J. Sci. Food Agric. 1999, 79, 961–969. [Google Scholar] [CrossRef]
- Terada, A.; Hara, H.; Sakamoto, J.; Sato, N.; Takagi, S.; Mitsuoka, T.; Mino, R.; Hara, K.; Fujimori, I.; Yamada, T. Effects of Dietary Supplementation with Lactosucrose (4G-β-D-Galactosylsucrose) on Cecal Flora, Cecal Metabolites, and Performance in Broiler Chickens. Poult. Sci. 1994, 73, 1663–1672. [Google Scholar] [CrossRef]
- Finegold, S.M.; Li, Z.; Summanen, P.H.; Downes, J.; Thames, G.; Corbett, K.; Dowd, S.; Krak, M.; Heber, D. Xylooligosaccharide increases bifidobacteria but not lactobacilli in human gut microbiota. Food Funct. 2014, 5, 436–445. [Google Scholar] [CrossRef]
- Chung, Y.-C.; Hsu, C.-K.; Ko, C.-Y.; Chan, Y.-C. Dietary intake of xylooligosaccharides improves the intestinal microbiota, fecal moisture, and pH value in the elderly. Nutr. Res. 2007, 27, 756–761. [Google Scholar] [CrossRef]
- Berger, K.; Burleigh, S.; Lindahl, M.; Bhattacharya, A.; Patil, P.; Stålbrand, H.; Nordberg Karlsson, E.; Hållenius, F.; Nyman, M.; Adlercreutz, P. Xylooligosaccharides Increase Bifidobacteria and Lachnospiraceae in Mice on a High-Fat Diet, with a Concomitant Increase in Short-Chain Fatty Acids, Especially Butyric Acid. J. Agric. Food Chem. 2021, 69, 3617–3625. [Google Scholar] [CrossRef]
- Hsu, C.K.; Liao, J.W.; Chung, Y.C.; Hsieh, C.P.; Chan, Y.C. Xylooligosaccharides and fructooligosaccharides affect the intestinal microbiota and precancerous colonic lesion development in rats. J. Nutr. 2004, 134, 1523–1528. [Google Scholar] [CrossRef]
- Roberfroid, M.B.; Van Loo, J.A.E.; Gibson, G.R. The Bifidogenic Nature of Chicory Inulin and Its Hydrolysis Products. J. Nutr. 1998, 128, 11–19. [Google Scholar] [CrossRef]
- Walker, A.W.; Duncan, S.H.; Carol McWilliam Leitch, E.; Child, M.W.; Flint, H.J. pH and peptide supply can radically alter bacterial populations and short-chain fatty acid ratios within microbial communities from the human colon. Appl. Environ. Microbiol. 2005, 71, 3692–3700. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Louis, P.; Thomson, J.M.; Flint, H.J. The role of pH in determining the species composition of the human colonic microbiota. Environ. Microbiol. 2009, 11, 2112–2122. [Google Scholar] [CrossRef] [PubMed]
- Guariguata, L.; Whiting, D.R.; Hambleton, I.; Beagley, J.; Linnenkamp, U.; Shaw, J.E. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res. Clin. Pract. 2014, 103, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, R.S.; Anderson, R.M.; Buse, J.B.; Chin, M.H.; Eddy, D.; Fradkin, J.; Ganiats, T.G.; Ginsberg, H.; Kahn, R.; Nwankwo, R.; et al. The prevention or delay of type 2 diabetes. Diabetes Care 2003, 26 (Suppl. 1), S62–S69. [Google Scholar] [CrossRef] [PubMed]
- Nathan, D.M. Long-term complications of diabetes mellitus. N. Engl. J. Med. 1993, 328, 1676–1685. [Google Scholar] [CrossRef]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef]
- Moya-Pérez, A.; Luczynski, P.; Renes, I.B.; Wang, S.; Borre, Y.; Anthony Ryan, C.; Knol, J.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Intervention strategies for cesarean section-induced alterations in the microbiota-gut-brain axis. Nutr. Rev. 2017, 75, 225–240. [Google Scholar] [CrossRef]
- Rabot, S.; Membrez, M.; Bruneau, A.; Gérard, P.; Harach, T.; Moser, M.; Raymond, F.; Mansourian, R.; Chou, C.J. Germ-free C57BL/6J mice are resistant to high-fat-diet-induced insulin resistance and have altered cholesterol metabolism. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2010, 24, 4948–4959. [Google Scholar] [CrossRef]
- Yde, C.C.; Jensen, H.M.; Christensen, N.; Servant, F.; Lelouvier, B.; Lahtinen, S.; Stenman, L.K.; Airaksinen, K.; Kailanto, H.-M. Polydextrose with and without Bifidobacterium animalis ssp. lactis 420 drives the prevalence of Akkermansia and improves liver health in a multi-compartmental obesogenic mice study. PLoS ONE 2021, 16, e0260765. [Google Scholar] [CrossRef]
- Snelson, M.; de Pasquale, C.; Ekinci, E.I.; Coughlan, M.T. Gut microbiome, prebiotics, intestinal permeability and diabetes complications. Best Pract. Res. Clin. Endocrinol. Metab. 2021, 35, 101507. [Google Scholar] [CrossRef]
- Tajabadi-Ebrahimi, M.; Sharifi, N.; Farrokhian, A.; Raygan, F.; Karamali, F.; Razzaghi, R.; Taheri, S.; Asemi, Z. A Randomized Controlled Clinical Trial Investigating the Effect of Synbiotic Administration on Markers of Insulin Metabolism and Lipid Profiles in Overweight Type 2 Diabetic Patients with Coronary Heart Disease. Exp. Clin. Endocrinol. Diabetes Off. J. Ger. Soc. Endocrinol. Ger. Diabetes Assoc. 2017, 125, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Y.; Zhou, D.D.; Gan, R.Y.; Huang, S.Y.; Zhao, C.N.; Shang, A.; Xu, X.Y.; Li, H. Bin Effects and mechanisms of probiotics, prebiotics, synbiotics, and postbiotics on metabolic diseases targeting gut microbiota: A narrative review. Nutrients 2021, 13, 3211. [Google Scholar] [CrossRef] [PubMed]
- Vezza, T.; Abad-Jiménez, Z.; Marti-Cabrera, M.; Rocha, M.; Víctor, V.M. Microbiota-mitochondria inter-talk: A potential therapeutic strategy in obesity and type 2 diabetes. Antioxidants 2020, 9, 848. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.A.; Keogh, J.B.; Clifton, P.M. Probiotics, prebiotics, synbiotics and insulin sensitivity. Nutr. Res. Rev. 2018, 31, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.I.A.; Gibson, G.R. Effects of consumption of probiotics and prebiotics on serum lipid levels in humans. Crit. Rev. Biochem. Mol. Biol. 2002, 37, 259–281. [Google Scholar] [CrossRef]
- Costa, G.T.; Guimarães, S.B.; Sampaio, H.A.C. Fructo-oligosaccharide effects on blood glucose. An overview [Efeitos dos fruto-oligossacar{í}deos no controle glic{ê}mico. revis{ã}o]. Acta Cir. Bras. 2012, 27, 279–282. [Google Scholar] [CrossRef]
- Dehghan, P.; Pourghassem Gargari, B.; Asghari Jafar-abadi, M. Oligofructose-enriched inulin improves some inflammatory markers and metabolic endotoxemia in women with type 2 diabetes mellitus: A randomized controlled clinical trial. Nutrition 2014, 30, 418–423. [Google Scholar] [CrossRef]
- Letexier, D.; Diraison, F.; Beylot, M. Addition of inulin to a moderately high-carbohydrate diet reduces hepatic lipogenesis and plasma triacylglycerol concentrations in humans. Am. J. Clin. Nutr. 2003, 77, 559–564. [Google Scholar] [CrossRef]
- Aliasgharzadeh, A.; Khalili, M.; Mirtaheri, E.; Pourghassem Gargari, B.; Tavakoli, F.; Abbasalizad Farhangi, M.; Babaei, H.; Dehghan, P. A Combination of Prebiotic Inulin and Oligofructose Improve Some of Cardiovascular Disease Risk Factors in Women with Type 2 Diabetes: A Randomized Controlled Clinical Trial. Adv. Pharm. Bull. 2015, 5, 507–514. [Google Scholar] [CrossRef]
- Jackson, K.G.; Taylor, G.R.J.; Clohessy, A.M.; Williams, C.M. The effect of the daily intake of inulin on fasting lipid, insulin and glucose concentrations in middle-aged men and women. Br. J. Nutr. 1999, 82, 23–30. [Google Scholar] [CrossRef]
- Dewulf, E.M.; Cani, P.D.; Claus, S.P.; Fuentes, S.; Puylaert, P.G.B.; Neyrinck, A.M.; Bindels, L.B.; De Vos, W.M.; Gibson, G.R.; Thissen, J.P.; et al. Insight into the prebiotic concept: Lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut 2013, 62, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Schellekens, H.; Torres-Fuentes, C.; van de Wouw, M.; Long-Smith, C.M.; Mitchell, A.; Strain, C.; Berding, K.; Bastiaanssen, T.F.S.; Rea, K.; Golubeva, A.V.; et al. Bifidobacterium longum counters the effects of obesity: Partial successful translation from rodent to human. EBioMedicine 2021, 63, 103176. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Lazarevic, V.; Derrien, M.; Girard, M.; Muccioli, G.G.; Neyrinck, A.M.; Possemiers, S.; Van Holle, A.; François, P.; de Vos, W.M.; et al. Responses of gut microbiota and glucose and lipid metabolism to prebiotics in genetic obese and diet-induced leptin-resistant mice. Diabetes 2011, 60, 2775–2786. [Google Scholar] [CrossRef] [PubMed]
- Sangwan, V.; Tomar, S.K.; Ali, B.; Singh, R.R.B.; Singh, A.K. Hypoglycaemic effect of galactooligosaccharides in alloxan-induced diabetic rats. J. Dairy Res. 2015, 82, 70–77. [Google Scholar] [CrossRef]
- Vulevic, J.; Juric, A.; Tzortzis, G.; Gibson, G.R. A mixture of trans-galactooligosaccharides reduces markers of metabolic syndrome and modulates the fecal microbiota and immune function of overweight adults. J. Nutr. 2013, 143, 324–331. [Google Scholar] [CrossRef]
- Albarracín, M.; Weisstaub, A.R.; Zuleta, Á.; Mandalunis, P.; González, R.J.; Drago, S.R. Effects of extruded whole maize, polydextrose and cellulose as sources of fibre on calcium bioavailability and metabolic parameters of growing Wistar rats. Food Funct. 2014, 5, 804–810. [Google Scholar] [CrossRef]
- Maki, K.C.; Pelkman, C.L.; Finocchiaro, E.T.; Kelley, K.M.; Lawless, A.L.; Schild, A.L.; Rains, T.M. Resistant starch from high-amylose maize increases insulin sensitivity in overweight and obese men. J. Nutr. 2012, 142, 717–723. [Google Scholar] [CrossRef]
- Bodinham, C.L.; Smith, L.; Thomas, E.L.; Bell, J.D.; Swann, J.R.; Costabile, A.; Russell-Jones, D.; Umpleby, A.M.; Robertson, M.D. Efficacy of increased resistant starch consumption in human type 2 diabetes. Endocr. Connect. 2014, 3, 75–84. [Google Scholar] [CrossRef]
- Bianchi, G.; Ronchi, M.; Marchesini, G. Effect of lactulose on carbohydrate metabolism and diabetes mellitus. Scand. J. Gastroenterol. Suppl. 1997, 222, 62–64. [Google Scholar] [CrossRef]
- Lopez, H.W.; Levrat, M.A.; Guy, C.; Messager, A.; Demigné, C.; Rémésy, C. Effects of soluble corn bran arabinoxylans on cecal digestion, lipid metabolism, and mineral balance (Ca, Mg) in rats. J. Nutr. Biochem. 1999, 10, 500–509. [Google Scholar] [CrossRef]
- Lu, Z.X.; Walker, K.Z.; Muir, J.G.; O’Dea, K. Arabinoxylan fibre improves metabolic control in people with Type II diabetes. Eur. J. Clin. Nutr. 2004, 58, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Imaizumi, K.; Nakatsu, Y.; Sato, M.; Sedarnawati, Y.; Sugano, M. Effects of xylooligosaccharides on blood glucose, serum and liver lipids and-cecum short-chain fatty acids in diabetic rats. Agric. Biol. Chem. 1991, 55, 199–205. [Google Scholar] [CrossRef][Green Version]
- Kim, S.; Rhee, I.; Joo, G.; Ha, H. Effects of dietary xylooligosaccharide on lipid levels of serum in rats fed high cholesterol diet. J. Korean Soc. Food Sci. Nutr. 1998, 27, 945–951. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Megur, A.; Daliri, E.B.-M.; Baltriukienė, D.; Burokas, A. Prebiotics as a Tool for the Prevention and Treatment of Obesity and Diabetes: Classification and Ability to Modulate the Gut Microbiota. Int. J. Mol. Sci. 2022, 23, 6097. https://doi.org/10.3390/ijms23116097
Megur A, Daliri EB-M, Baltriukienė D, Burokas A. Prebiotics as a Tool for the Prevention and Treatment of Obesity and Diabetes: Classification and Ability to Modulate the Gut Microbiota. International Journal of Molecular Sciences. 2022; 23(11):6097. https://doi.org/10.3390/ijms23116097
Chicago/Turabian StyleMegur, Ashwinipriyadarshini, Eric Banan-Mwine Daliri, Daiva Baltriukienė, and Aurelijus Burokas. 2022. "Prebiotics as a Tool for the Prevention and Treatment of Obesity and Diabetes: Classification and Ability to Modulate the Gut Microbiota" International Journal of Molecular Sciences 23, no. 11: 6097. https://doi.org/10.3390/ijms23116097
APA StyleMegur, A., Daliri, E. B.-M., Baltriukienė, D., & Burokas, A. (2022). Prebiotics as a Tool for the Prevention and Treatment of Obesity and Diabetes: Classification and Ability to Modulate the Gut Microbiota. International Journal of Molecular Sciences, 23(11), 6097. https://doi.org/10.3390/ijms23116097






