Synergistic Activity of Ketoconazole and Miconazole with Prochloraz in Inducing Oxidative Stress, GSH Depletion, Mitochondrial Dysfunction, and Apoptosis in Mouse Sertoli TM4 Cells
Abstract
:1. Introduction
2. Results
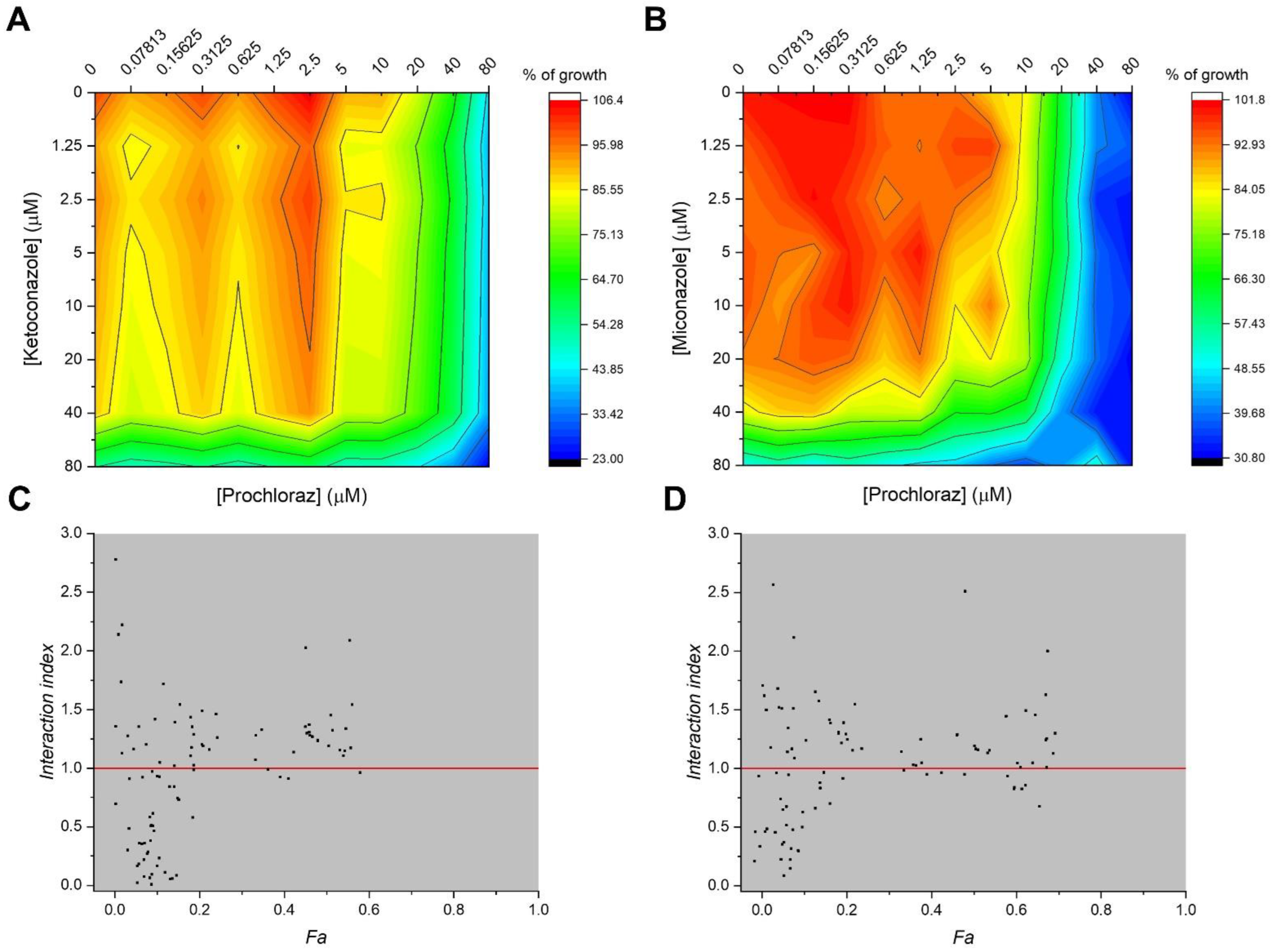
2.1. Synergistic Antiproliferative Effects of the Combined Treatment with PCZ, KCZ, and MCZ in TM4 Cells
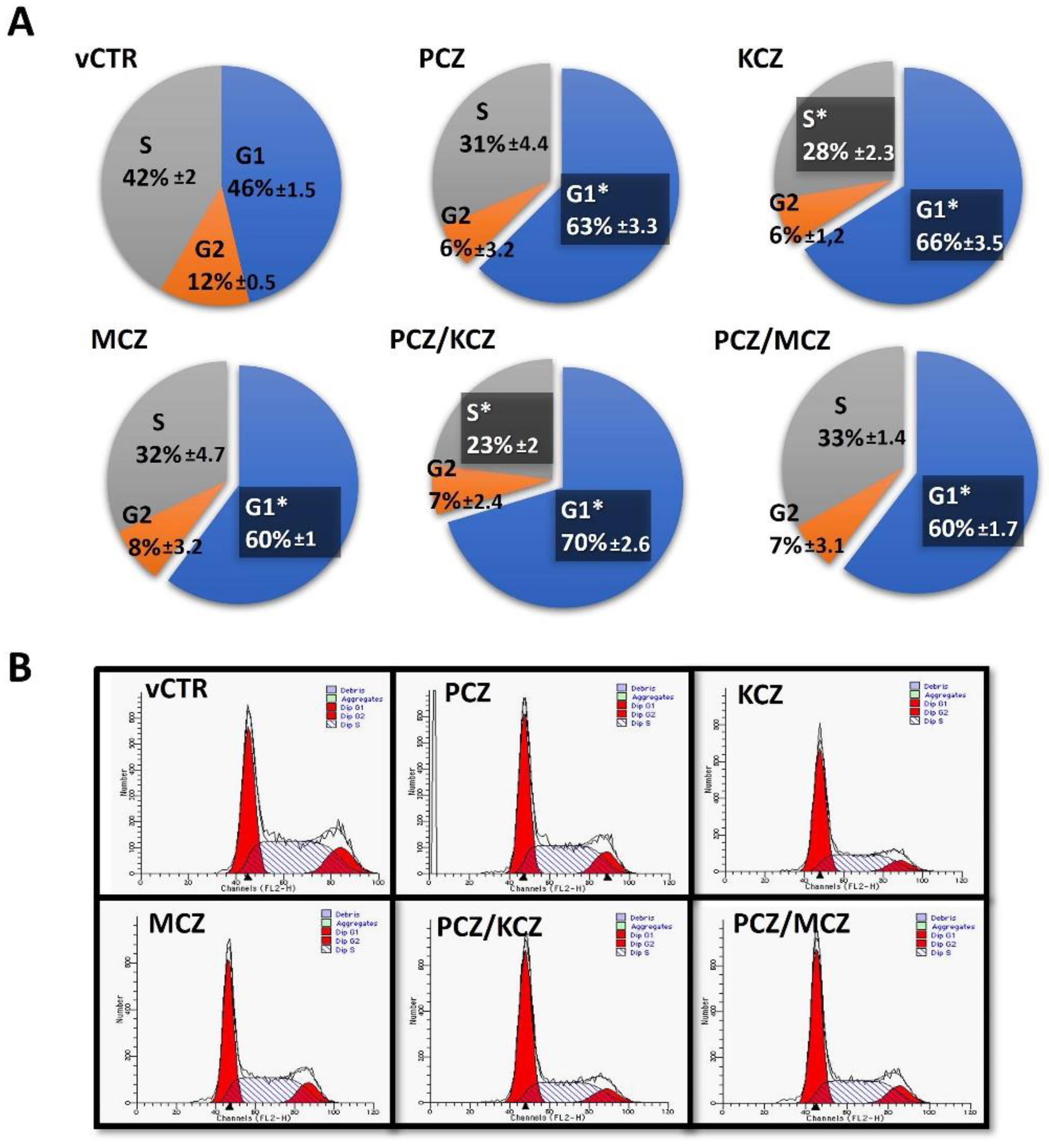
2.2. PCZ/KCZ Combination Enhances the G0/G1 Phase Arrest in TM4 Cells
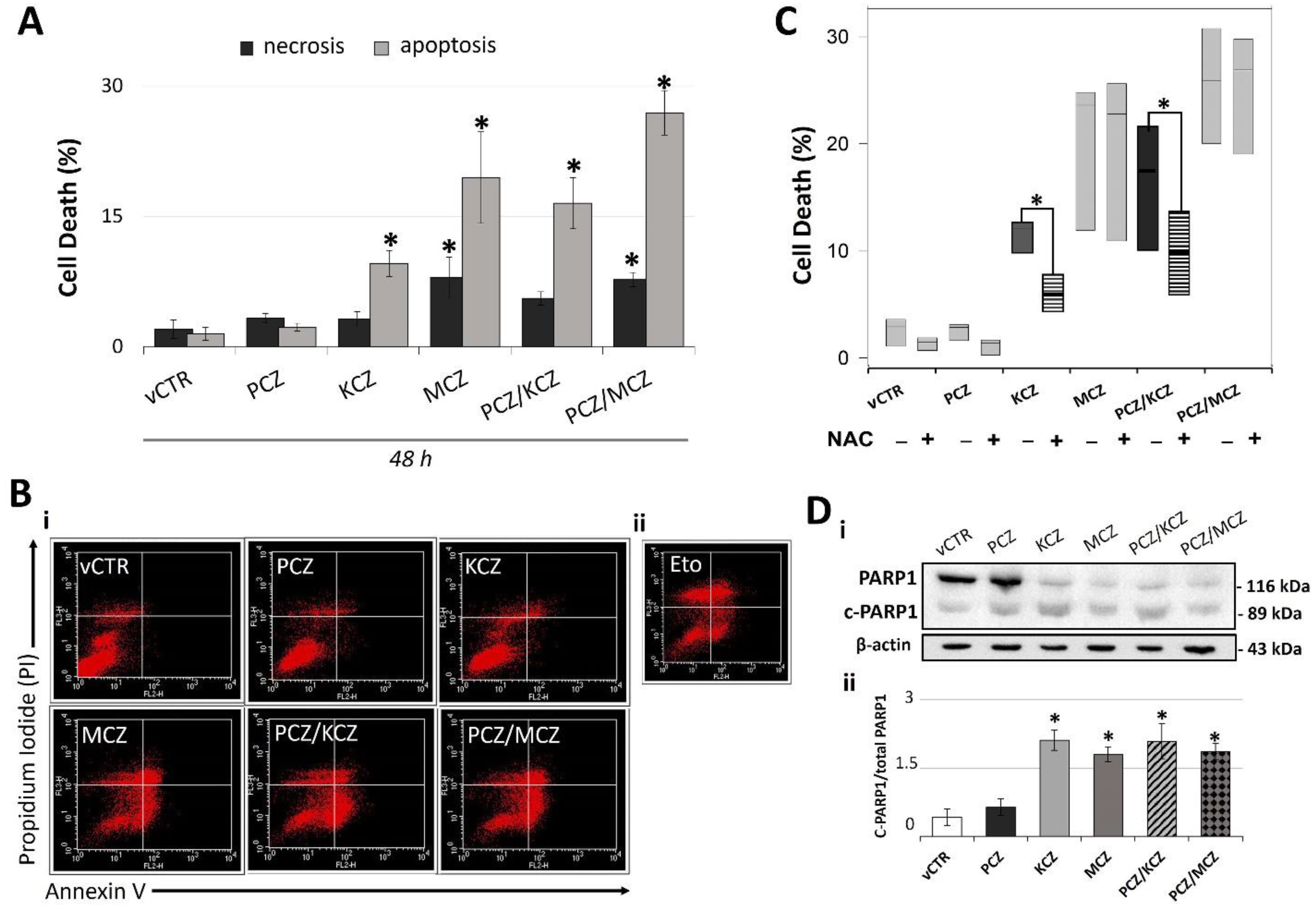
2.3. PCZ Enhances KCZ- and MCZ-Induced Apoptosis in TM4 Cells
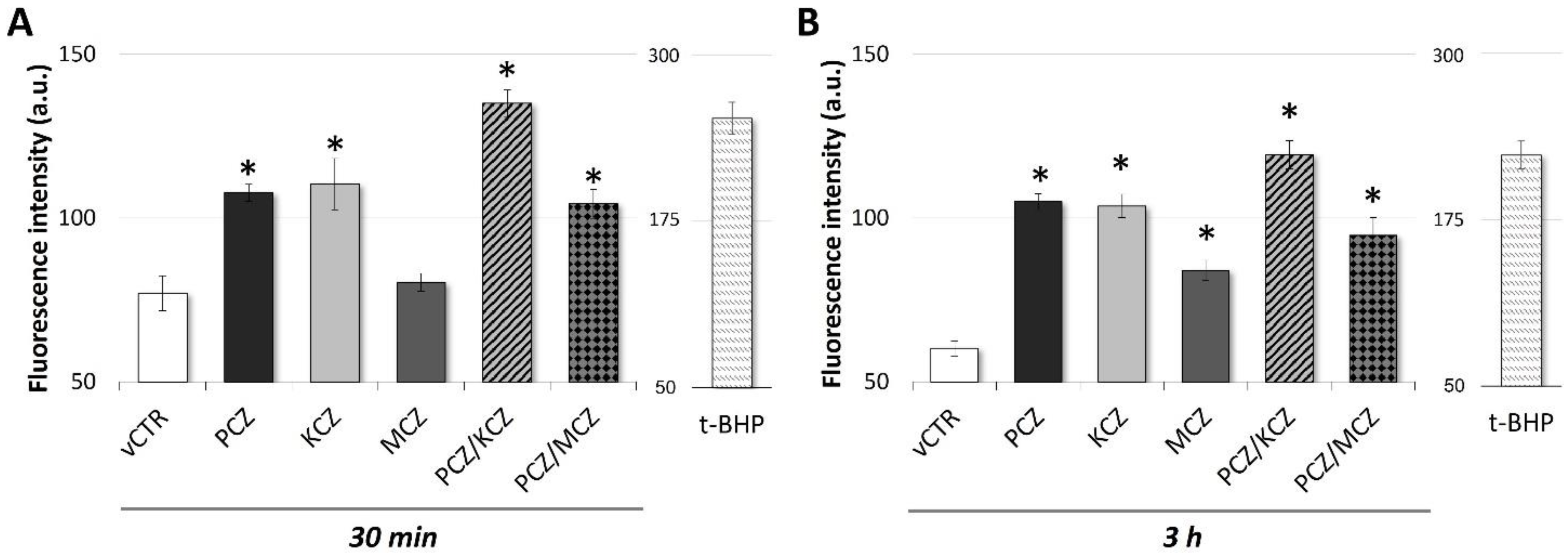
2.4. Early Increased ROS Production in Azoles-Induced Apoptosis
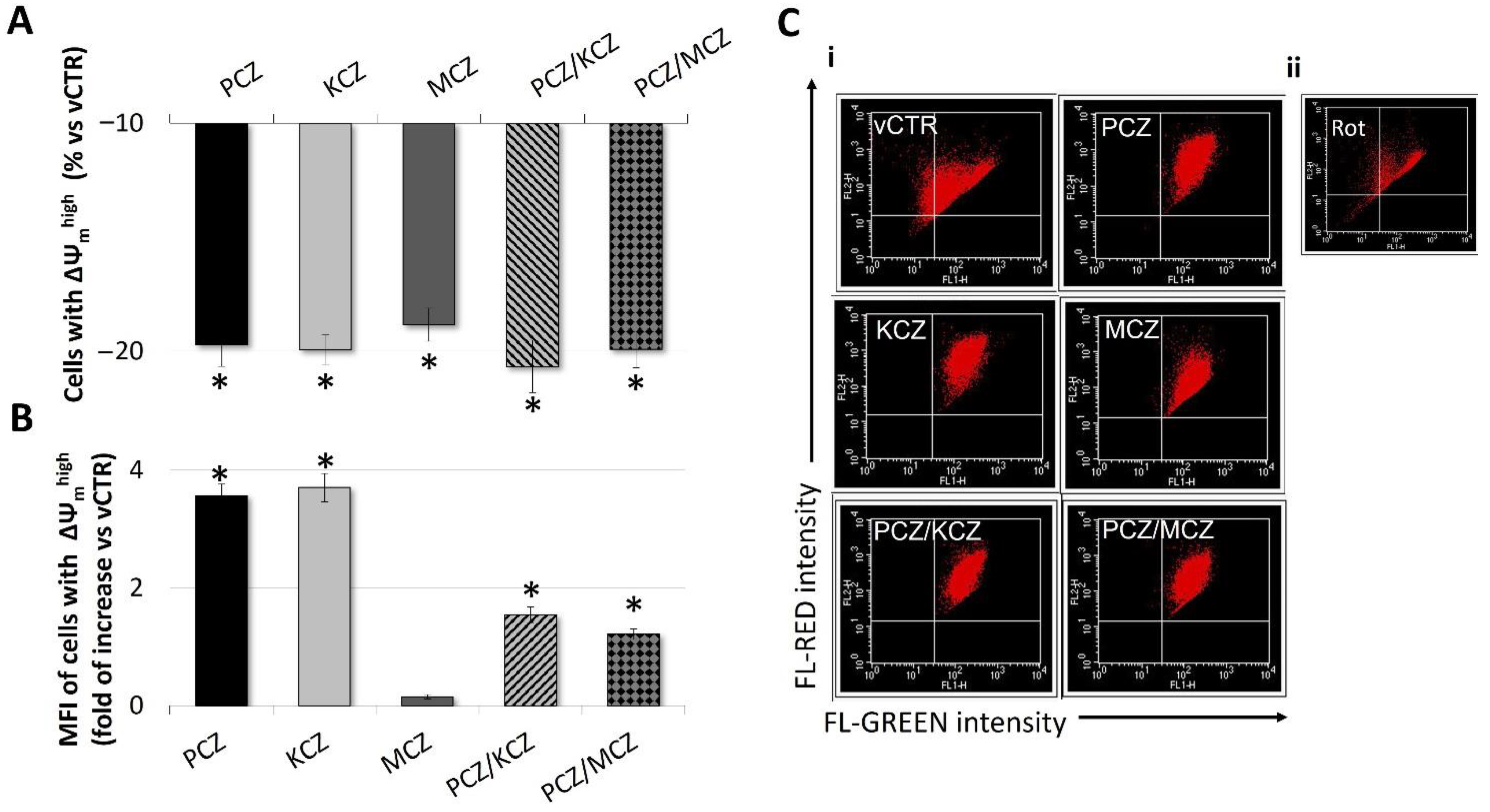
2.5. Mitochondrial Membrane Potential (ΔΨm) Loss in Azoles-Treated Cells
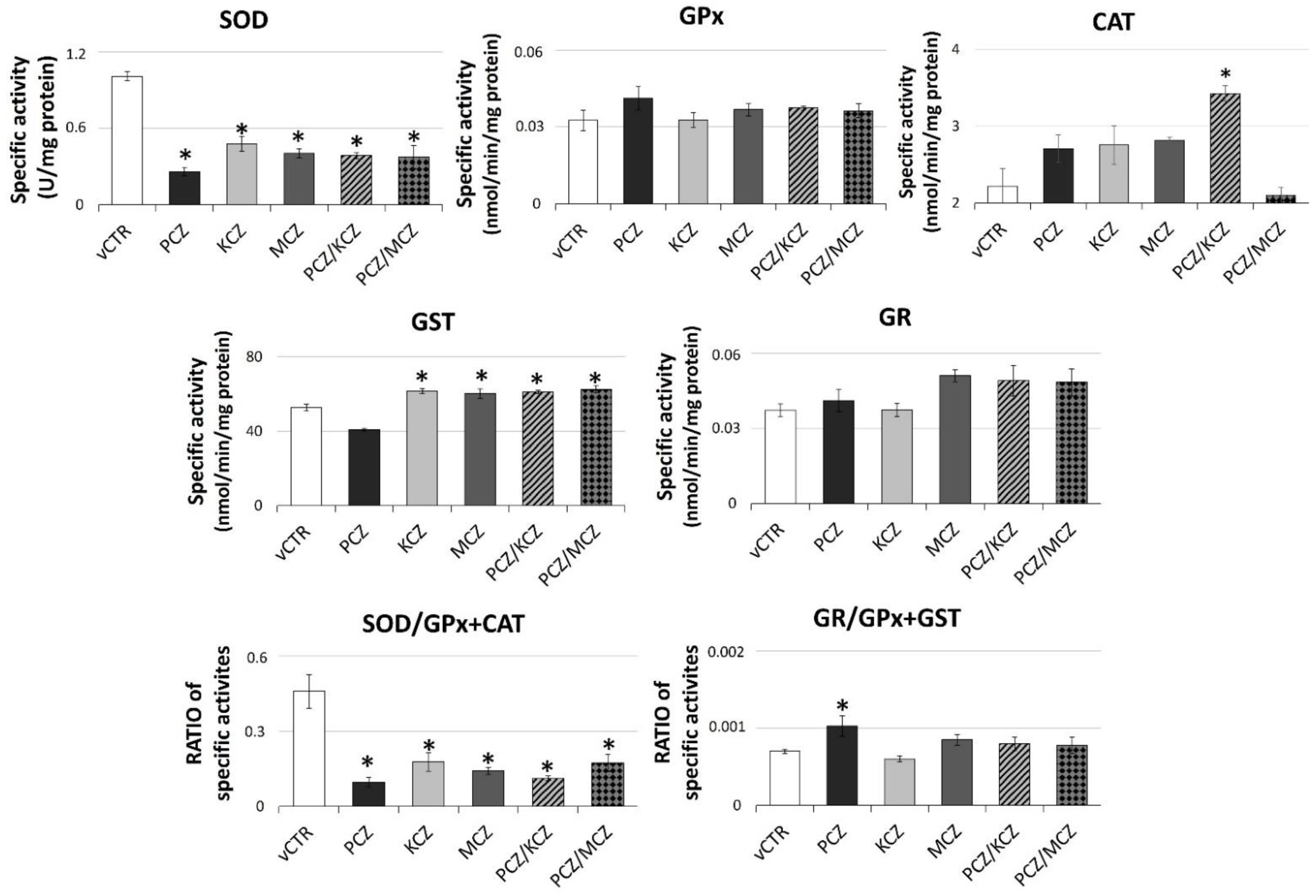
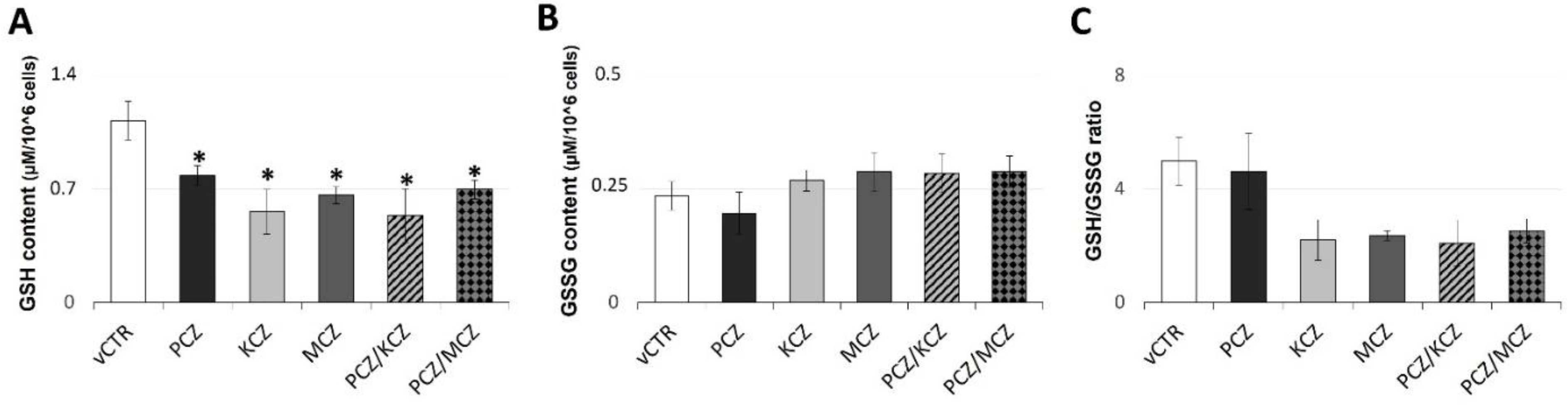
2.6. Azoles Exposure Drastically Reduces SOD Activity Levels and Down-Regulates the Intracellular GSH Pool
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatments
4.2. Cell Growth and Viability
4.3. Calculation of the Interaction Index
4.4. Flow Cytometry Analyses of Cell Cycle
4.5. Detection of Intracellular Reactive Oxygen Species (ROS)
4.6. Enzymatic Activity Assays
4.7. Detection of GSH and GSSG Intracellular Content
4.8. Assessment of ΔΨm by Flow Cytometry
4.9. Annexin V-FITC and Propidium Iodide Assay
4.10. Western Blot Analysis
4.11. Statistical Analyses
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lotti, F.; Maggi, M. Sexual dysfunction and male infertility. Nat. Rev. Urol. 2018, 15, 287–307. [Google Scholar] [CrossRef] [PubMed]
- Vander Borght, M.; Wyns, C. Fertility and infertility: Definition and epidemiology. Clin. Biochem. 2018, 62, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Gong, T.-T.; Jiang, Y.-T.; Zhang, S.; Zhao, Y.-H.; Wu, Q.-J. Global, regional, and national prevalence and disability-adjusted life-years for infertility in 195 countries and territories, 1990–2017: Results from a global burden of disease study, 2017. Aging 2019, 11, 10952–10991. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Baskaran, S.; Parekh, N.; Cho, C.-L.; Henkel, R.; Vij, S.; Arafa, M.; Selvam, M.K.P.; Shah, R. Male infertility. Lancet 2021, 397, 319–333. [Google Scholar] [CrossRef]
- Ma, Y.; He, X.; Qi, K.; Wang, T.; Qi, Y.; Cui, L.; Wang, F.; Song, M. Effects of environmental contaminants on fertility and reproductive health. J. Environ. Sci. 2019, 77, 210–217. [Google Scholar] [CrossRef]
- Agarwal, A.; Parekh, N.; Selvam, M.K.P.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S.; et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J. Men’s Health 2019, 37, 296–312. [Google Scholar] [CrossRef]
- Sifakis, S.; Androutsopoulos, V.P.; Tsatsakis, A.M.; Spandidos, D.A. Human exposure to endocrine disrupting chemicals: Effects on the male and female reproductive systems. Environ. Toxicol. Pharmacol. 2017, 51, 56–70. [Google Scholar] [CrossRef]
- Moreira, S.; Pereira, S.C.; Seco-Rovira, V.; Oliveira, P.F.; Alves, M.G.; Pereira, M.D.L. Pesticides and Male Fertility: A Dangerous Crosstalk. Metabolites 2021, 11, 799. [Google Scholar] [CrossRef]
- Brehm, E.; Flaws, J.A. Transgenerational Effects of Endocrine-Disrupting Chemicals on Male and Female Reproduction. Endocrinology 2019, 160, 1421–1435. [Google Scholar] [CrossRef] [Green Version]
- Jenardhanan, P.; Panneerselvam, M.; Mathur, P.P. Effect of environmental contaminants on spermatogenesis. Semin. Cell Dev. Biol. 2016, 59, 126–140. [Google Scholar] [CrossRef]
- Barati, E.; Nikzad, H.; Karimian, M. Oxidative stress and male infertility: Current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell. Mol. Life Sci. 2020, 77, 93–113. [Google Scholar] [CrossRef] [PubMed]
- Asadi, N. The Impact of Oxidative Stress on Testicular Function and the Role of Antioxidants in Improving it: A Review. J. Clin. Diagn. Res. 2017, 11, IE01–IE05. [Google Scholar] [CrossRef] [PubMed]
- Mannucci, A.; Argento, F.R.; Fini, E.; Coccia, M.E.; Taddei, N.; Becatti, M.; Fiorillo, C. The Impact of Oxidative Stress in Male Infertility. Front. Mol. Biosci. 2022, 8, 799294. [Google Scholar] [CrossRef] [PubMed]
- Bui, A.D.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, e13012. [Google Scholar] [CrossRef]
- Gao, Y.; Mruk, D.D.; Cheng, C.Y. Sertoli cells are the target of environmental toxicants in the testis—A mechanistic and therapeutic insight. Expert Opin. Ther. Targets 2015, 19, 1073–1090. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.; Dufrusine, B.; Lizzi, A.R.; Luzi, C.; Piccoli, A.; Fezza, F.; Iorio, R.; D’Andrea, G.; Dainese, E.; Cecconi, S.; et al. Bisphenol A Deranges the Endocannabinoid System of Primary Sertoli Cells with an Impact on Inhibin B Production. Int. J. Mol. Sci. 2020, 21, 8986. [Google Scholar] [CrossRef]
- Oduwole, O.O.; Peltoketo, H.; Huhtaniemi, I.T. Role of Follicle-Stimulating Hormone in Spermatogenesis. Front. Endocrinol. 2018, 9, 763. [Google Scholar] [CrossRef] [Green Version]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef]
- Chen, Z.-F.; Ying, G.-G. Occurrence, fate and ecological risk of five typical azole fungicides as therapeutic and personal care products in the environment: A review. Environ. Int. 2015, 84, 142–153. [Google Scholar] [CrossRef]
- A systematic procedure for the identification of emerging chemical risks in the food and feed chain. EFSA Support. Publ. 2014, 11, 547E. [CrossRef]
- Zarn, J.A.; Brüschweiler, B.J.; Schlatter, J.R. Azole fungicides affect mammalian steroidogenesis by inhibiting sterol 14 alpha-demethylase and aromatase. Environ. Health Perspect. 2003, 111, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, A.; Cedergreen, N.; Oskarsson, A.; Ullerås, E. Mixture effects of imidazole fungicides on cortisol and aldosterone secretion in human adrenocortical H295R cells. Toxicology 2010, 275, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Kjaerstad, M.B.; Taxvig, C.; Andersen, H.R.; Nellemann, C. Mixture effects of endocrine disrupting compounds in vitro. Int. J. Androl. 2010, 33, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pas, R.; Hofland, L.J.; Hofland, J.; Taylor, A.E.; Arlt, W.; Steenbergen, J.; Van Koetsveld, P.M.; De Herder, W.W.; De Jong, F.H.; Feelders, R.A. Fluconazole inhibits human adrenocortical steroidogenesis in vitro. J. Endocrinol. 2012, 215, 403–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christen, V.; Crettaz, P.; Fent, K. Additive and synergistic antiandrogenic activities of mixtures of azol fungicides and vinclozolin. Toxicol. Appl. Pharmacol. 2014, 279, 455–466. [Google Scholar] [CrossRef]
- Glynn, M.; Jo, W.; Minowa, K.; Sanada, H.; Nejishima, H.; Matsuuchi, H.; Okamura, H.; Pillai, R.; Mutter, L. Efinaconazole: Developmental and reproductive toxicity potential of a novel antifungal azole. Reprod. Toxicol. 2015, 52, 18–25. [Google Scholar] [CrossRef] [Green Version]
- Rieke, S.; Heise, T.; Schmidt, F.; Haider, W.; Bednarz, H.; Niehaus, K.; Mentz, A.; Kalinowski, J.; Hirsch-Ernst, K.I.; Steinberg, P.; et al. Mixture effects of azole fungicides on the adrenal gland in a broad dose range. Toxicology 2017, 385, 28–37. [Google Scholar] [CrossRef]
- Hamdi, H.; Ben Salem, I.; Ben Othmène, Y.; Annabi, E.; Abid-Essefi, S. The involvement of ROS generation on Epoxiconazole-induced toxicity in HCT116 cells. Pestic. Biochem. Physiol. 2018, 148, 62–67. [Google Scholar] [CrossRef]
- Ben Othmène, Y.; Hamdi, H.; Annabi, E.; Amara, I.; Ben Salem, I.; Neffati, F.; Najjar, M.F.; Abid-Essefi, S. Tebuconazole induced cardiotoxicity in male adult rat. Food Chem. Toxicol. 2020, 137, 111134. [Google Scholar] [CrossRef]
- Ben Othmène, Y.; Monceaux, K.; Karoui, A.; Ben Salem, I.; Belhadef, A.; Abid-Essefi, S.; Lemaire, C. Tebuconazole induces ROS-dependent cardiac cell toxicity by activating DNA damage and mitochondrial apoptotic pathway. Ecotoxicol. Environ. Saf. 2020, 204, 111040. [Google Scholar] [CrossRef]
- Hamdi, H.; Ben Othmene, Y.; Khlifi, A.; Hallara, E.; Houas, Z.; Najjar, M.F.; Abid-Essefi, S. Subchronic exposure to Epoxiconazole induced-heart damage in male Wistar rats. Pestic. Biochem. Physiol. 2022, 182, 105034. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, D.; Kondo, K.; Uehara, N.; Otokozawa, S.; Tsuji, N.; Yagihashi, A.; Watanabe, N. Endogenous Reactive Oxygen Species Is an Important Mediator of Miconazole Antifungal Effect. Antimicrob. Agents Chemother. 2002, 46, 3113–3117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.P.; Kim, J.-E.; Park, W.-H. Cytoprotective effect of rhamnetin on miconazole-induced H9c2 cell damage. Nutr. Res. Pract. 2015, 9, 586–591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, H.-J.; Seo, I.; Jha, B.K.; Suh, S.-I.; Baek, W.-K. Miconazole induces autophagic death in glioblastoma cells via reactive oxygen species-mediated endoplasmic reticulum stress. Oncol. Lett. 2021, 21, 335. [Google Scholar] [CrossRef]
- Chengzhu, W.U.; Gao, M.; Shen, L.; Bohan, L.I.; Bai, X.; Gui, J.; Hongmei, L.I.; Huo, Q.; Tao, M.A. Miconazole triggers various forms of cell death in human breast cancer MDA-MB-231 cells. Die Pharm.-Int. J. Pharm. Sci. 2019, 74, 290–294. [Google Scholar] [CrossRef]
- Tits, J.; Berman, J.; Cammue, B.P.A.; Thevissen, K. Combining Miconazole and Domiphen Bromide Results in Excess of Reactive Oxygen Species and Killing of Biofilm Cells. Front. Cell Dev. Biol. 2021, 8, 617214. [Google Scholar] [CrossRef]
- Lundqvist, J.; Hellman, B.; Oskarsson, A. Fungicide prochloraz induces oxidative stress and DNA damage in vitro. Food Chem. Toxicol. 2016, 91, 36–41. [Google Scholar] [CrossRef]
- Alpertunga, B.; Kara, M.; Abudayyak, M.; Oztas, E.; Ozden, S.; Özhan, G. Effects of prochloraz on DNA damage, lipid peroxidation and antioxidant systemin vitro. Toxicol. Mech. Methods 2014, 24, 268–275. [Google Scholar] [CrossRef]
- Haegler, P.; Joerin, L.; Krähenbühl, S.; Bouitbir, J. Hepatocellular Toxicity of Imidazole and Triazole Antimycotic Agents. Toxicol. Sci. 2017, 157, 183–195. [Google Scholar] [CrossRef] [Green Version]
- Wewering, F.; Jouy, F.; Wissenbach, D.K.; Gebauer, S.; Blüher, M.; Gebhardt, R.; Pirow, R.; Von Bergen, M.; Kalkhof, S.; Luch, A.; et al. Characterization of chemical-induced sterile inflammation in vitro: Application of the model compound ketoconazole in a human hepatic co-culture system. Arch. Toxicol. 2017, 91, 799–810. [Google Scholar] [CrossRef]
- da Silva, C.R.; Neto, J.B.d.A.; Sidrim, J.J.C.; Ângelo, M.R.F.; Magalhães, H.I.F.; Cavalcanti, B.C.; Brilhante, R.S.N.; Macedo, D.S.; de Moraes, M.O.; Lobo, M.D.P.; et al. Synergistic Effects of Amiodarone and Fluconazole on Candida tropicalis Resistant to Fluconazole. Antimicrob. Agents Chemother. 2013, 57, 1691–1700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahl, C.D.; Behling, C.S.; Hackenhaar, F.S.; Silva, M.N.D.C.E.; Putti, J.; Salomon, T.B.; Alves, S.H.; Fuentefria, A.; Benfato, M.S. Induction of ROS generation by fluconazole in Candida glabrata: Activation of antioxidant enzymes and oxidative DNA damage. Diagn. Microbiol. Infect. Dis. 2015, 82, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.A.; Gaertner, A.A.E.; Henriquez, S.A.; Fang, D.; Colon-Reyes, R.J.; Brumaghim, J.L.; Kozubowski, L. Fluconazole induces ROS in Cryptococcus neoformans and contributes to DNA damage in vitro. PLoS ONE 2018, 13, e0208471. [Google Scholar] [CrossRef] [PubMed]
- Petricca, S.; Flati, V.; Celenza, G.; Di Gregorio, J.; Lizzi, A.R.; Luzi, C.; Cristiano, L.; Cinque, B.; Rossi, G.; Festuccia, C.; et al. Tebuconazole and Econazole Act Synergistically in Mediating Mitochondrial Stress, Energy Imbalance, and Sequential Activation of Autophagy and Apoptosis in Mouse Sertoli TM4 Cells: Possible Role of AMPK/ULK1 Axis. Toxicol. Sci. 2019, 169, 209–223. [Google Scholar] [CrossRef]
- Ben Saad, H.; Ben Abdallah, F.; Bkhairia, I.; Boudawara, O.; Nasri, M.; Hakim, A.; Ben Amara, I. Efficacy of Essential Trace Elements Supplementation on Mineral Composition, Sperm Characteristics, Antioxidant Status, and Genotoxicity in Testis of Tebuconazole-treated Rats. Biomed. Environ. Sci. 2020, 33, 760–770. [Google Scholar] [CrossRef]
- Feijó, M.; Martins, R.V.L.; Socorro, S.; Pereira, L.; Correia, S. Effects of the endocrine disruptor vinclozolin in male reproduction: A systematic review and meta-analysis. Biol. Reprod. 2021, 104, 962–975. [Google Scholar] [CrossRef]
- Caporale, N.; Leemans, M.; Birgersson, L.; Germain, P.-L.; Cheroni, C.; Borbély, G.; Engdahl, E.; Lindh, C.; Bressan, R.B.; Cavallo, F.; et al. From cohorts to molecules: Adverse impacts of endocrine disrupting mixtures. Science 2022, 375, eabe8244. [Google Scholar] [CrossRef]
- Redza-Dutordoir, M.; Averill-Bates, D.A. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2016, 1863, 2977–2992. [Google Scholar] [CrossRef]
- Reis, M.M.S.; Moreira, A.C.; Sousa, M.; Mathur, P.P.; Oliveira, P.F.; Alves, M.G. Sertoli cell as a model in male reproductive toxicology: Advantages and disadvantages. J. Appl. Toxicol. 2015, 35, 870–883. [Google Scholar] [CrossRef]
- Mostafalou, S.; Abdollahi, M. Pesticides: An update of human exposure and toxicity. Arch. Toxicol. 2017, 91, 549–599. [Google Scholar] [CrossRef]
- Sengupta, P.; Banerjee, R. Environmental toxins: Alarming impacts of pesticides on male fertility. Hum. Exp. Toxicol. 2014, 33, 1017–1039. [Google Scholar] [CrossRef] [PubMed]
- Meli, R.; Monnolo, A.; Annunziata, C.; Pirozzi, C.; Ferrante, M.C. Oxidative Stress and BPA Toxicity: An Antioxidant Approach for Male and Female Reproductive Dysfunction. Antioxidants 2020, 9, 405. [Google Scholar] [CrossRef] [PubMed]
- Wong, E.W.P.; Cheng, C.Y. Impacts of environmental toxicants on male reproductive dysfunction. Trends Pharmacol. Sci. 2011, 32, 290–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Y.; Li, D.; Lu, Y.; Han, X. Monobutyl phthalate induces the expression change of G-Protein-Coupled Receptor 30 in rat testicular Sertoli cells. Folia Histochem. Cytobiol. 2013, 51, 18–24. [Google Scholar] [CrossRef] [Green Version]
- Rasool, M.; Naseer, M.I.; Zaigham, K.; Malik, A.; e Habiba, U.; Manan, A.; Qazi, M.H.; Asif, M. Potential reproductive health effects and oxidative stress associated with exposure to potassium dichromate (K2CR2O7) and magnesium sulphate (MgSO4) in male mice. Pak. J. Med. Sci. 2014, 30, 819–823. [Google Scholar] [CrossRef]
- Choi, M.-S.; Park, H.-J.; Oh, J.-H.; Lee, E.-H.; Park, S.-M.; Yoon, S. Nonylphenol-induced apoptotic cell death in mouse TM4 Sertoli cells via the generation of reactive oxygen species and activation of the ERK signaling pathway. J. Appl. Toxicol. 2014, 34, 628–636. [Google Scholar] [CrossRef]
- Liu, X.; Nie, S.; Chen, Y.; Huang, D.; Xie, M. Effects of 4-nonylphenol isomers on cell receptors and mitogen-activated protein kinase pathway in mouse Sertoli TM4 cells. Toxicology 2014, 326, 1–8. [Google Scholar] [CrossRef]
- Duan, P.; Hu, C.; Quan, C.; Yu, T.; Zhou, W.; Yuan, M.; Shi, Y.; Yang, K. 4-Nonylphenol induces apoptosis, autophagy and necrosis in Sertoli cells: Involvement of ROS-mediated AMPK/AKT-mTOR and JNK pathways. Toxicology 2016, 341–343, 28–40. [Google Scholar] [CrossRef]
- De Freitas, A.T.A.G.; Pinho, C.F.; de Aquino, A.M.; Fernandes, A.A.H.; Domeniconi, R.F.; Justulin, L.A.; Scarano, W.R. Panax ginseng methabolit (GIM-1) prevents oxidative stress and apoptosis in human Sertoli cells exposed to Monobutyl-phthalate (MBP). Reprod. Toxicol. 2019, 86, 68–75. [Google Scholar] [CrossRef]
- Zhang, L.; Ji, X.; Ding, F.; Wu, X.; Tang, N.; Wu, Q. Apoptosis and blood-testis barrier disruption during male reproductive dysfunction induced by PAHs of different molecular weights. Environ. Pollut. 2022, 300, 118959. [Google Scholar] [CrossRef]
- Castinetti, F.; Guignat, L.; Giraud, P.; Muller, M.; Kamenicky, P.; Drui, D.; Caron, P.; Luca, F.; Donadille, B.; Vantyghem, M.C.; et al. Ketoconazole in Cushing’s Disease: Is It Worth a Try? J. Clin. Endocrinol. Metab. 2014, 99, 1623–1630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, K.; Wu, Y.; Chi, Z.; Shu, C.; Li, L.; Wei, J.; Tao, L.; Ma, P.; Ding, L. A highly sensitive LC–MS/MS method for determination of ketoconazole in human plasma: Application to a clinical study of the exposure to ketoconazole in patients after topical administration. J. Pharm. Biomed. Anal. 2016, 128, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.-L.; Xu, M.; Ye, Q. Quantitative Determination of Ketoconazole by UPLC-MS/MS in Human Plasma and its Application to Pharmacokinetic Study. Drug Res. 2014, 64, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Lewi, P.J.; Boelaert, J.; Daneels, R.; De Meyere, R.; Van Landuyt, H.; Heykants, J.J.P.; Symoens, J.; Wynants, J. Pharmacokinetic profile of intravenous miconazole in man. Eur. J. Clin. Pharmacol. 1976, 10, 49–54. [Google Scholar] [CrossRef]
- European Food Safety Authority. Conclusion on the peer review of the pesticide risk assessment of the active substance prochloraz. EFSA J. 2011, 9, 2323. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; Acosta, D. Comparison of ketoconazole- and fluconazole-induced hepatotoxicity in a primary culture system of rat hepatocytes. Toxicology 1995, 96, 83–92. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; Acosta, D. Inhibition of mitochondrial function in isolated rate liver mitochondria by azole antifungals. J. Biochem. Toxicol. 1996, 11, 127–131. [Google Scholar] [CrossRef]
- Martin, L.A.; Kennedy, B.E.; Karten, B. Mitochondrial cholesterol: Mechanisms of import and effects on mitochondrial function. J. Bioenerg. Biomembr. 2016, 48, 137–151. [Google Scholar] [CrossRef]
- Wages, P.A.; Joshi, P.; Tallman, K.A.; Kim, H.-Y.H.; Bowman, A.B.; Porter, N.A. Screening ToxCast™ for Chemicals That Affect Cholesterol Biosynthesis: Studies in Cell Culture and Human Induced Pluripotent Stem Cell–Derived Neuroprogenitors. Environ. Health Perspect. 2020, 128, 017014. [Google Scholar] [CrossRef] [Green Version]
- Iorio, R.; Celenza, G.; Petricca, S. Mitophagy: Molecular Mechanisms, New Concepts on Parkin Activation and the Emerging Role of AMPK/ULK1 Axis. Cells 2021, 11, 30. [Google Scholar] [CrossRef]
- Keyer, K.; Imlay, J.A. Superoxide accelerates DNA damage by elevating free-iron levels. Proc. Natl. Acad. Sci. USA 1996, 93, 13635–13640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benov, L. How superoxide radical damages the cell. Protoplasma 2001, 217, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Albertolle, M.E.; Guengerich, F.P. The relationships between cytochromes P450 and H2O2: Production, reaction, and inhibition. J. Inorg. Biochem. 2018, 186, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Circu, M.L.; Aw, T.Y. Glutathione and apoptosis. Free Radic. Res. 2008, 42, 689–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalinina, E.V.; Chernov, N.N.; Novichkova, M.D. Role of glutathione, glutathione transferase, and glutaredoxin in regulation of redox-dependent processes. Biochemistry 2014, 79, 1562–1583. [Google Scholar] [CrossRef]
- Fujii, J.; Iuchi, Y.; Matsuki, S.; Ishii, T. Cooperative function of antioxidant and redox systems against oxidative stress in male reproductive tissues. Asian J. Androl. 2003, 5, 231–242. [Google Scholar] [PubMed]
- Kumar, T.R.; Wiseman, A.L.; Kala, G.; Kala, S.V.; Matzuk, M.M.; Lieberman, M.W. Reproductive Defects in γ-Glutamyl Transpeptidase-Deficient Mice. Endocrinology 2000, 141, 4270–4277. [Google Scholar] [CrossRef]
- Chou, T.C.; Talalay, P. Quantitative analysis of dose-effect relationships: The combined effects of multiple drugs or enzyme inhibitors. Adv. Enzym. Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef]
- Greco, W.R.; Park, H.S.; Rustum, Y.M. Application of a new approach for the quantitation of drug synergism to the combination of cis-diamminedichloroplatinum and 1-beta-D-arabinofuranosylcytosine. Cancer Res. 1990, 50, 5318–5327. [Google Scholar]
- Iorio, R.; Castellucci, A.; Rossi, G.; Cinque, B.; Cifone, M.G.; Macchiarelli, G.; Cecconi, S. Mancozeb affects mitochondrial activity, redox status and ATP production in mouse granulosa cells. Toxicol. In Vitro 2015, 30, 438–445. [Google Scholar] [CrossRef]
- Iorio, R.; Petricca, S.; Luzi, C.; Bellio, P.; Cristiano, L.; Festuccia, C.; Amicosante, G.; Celenza, G. Lactobacillus sakei Pro-Bio65 Reduces TNF-α Expression and Upregulates GSH Content and Antioxidant Enzymatic Activities in Human Conjunctival Cells. Transl. Vis. Sci. Technol. 2021, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Fagnani, L.; Nazzicone, L.; Brisdelli, F.; Giansanti, L.; Battista, S.; Iorio, R.; Petricca, S.; Amicosante, G.; Perilli, M.; Celenza, G.; et al. Cyclic and Acyclic Amine Oxide Alkyl Derivatives as Potential Adjuvants in Antimicrobial Chemotherapy against Methicillin-Resistant Staphylococcus aureus with an MDR Profile. Antibiotics 2021, 10, 952. [Google Scholar] [CrossRef] [PubMed]
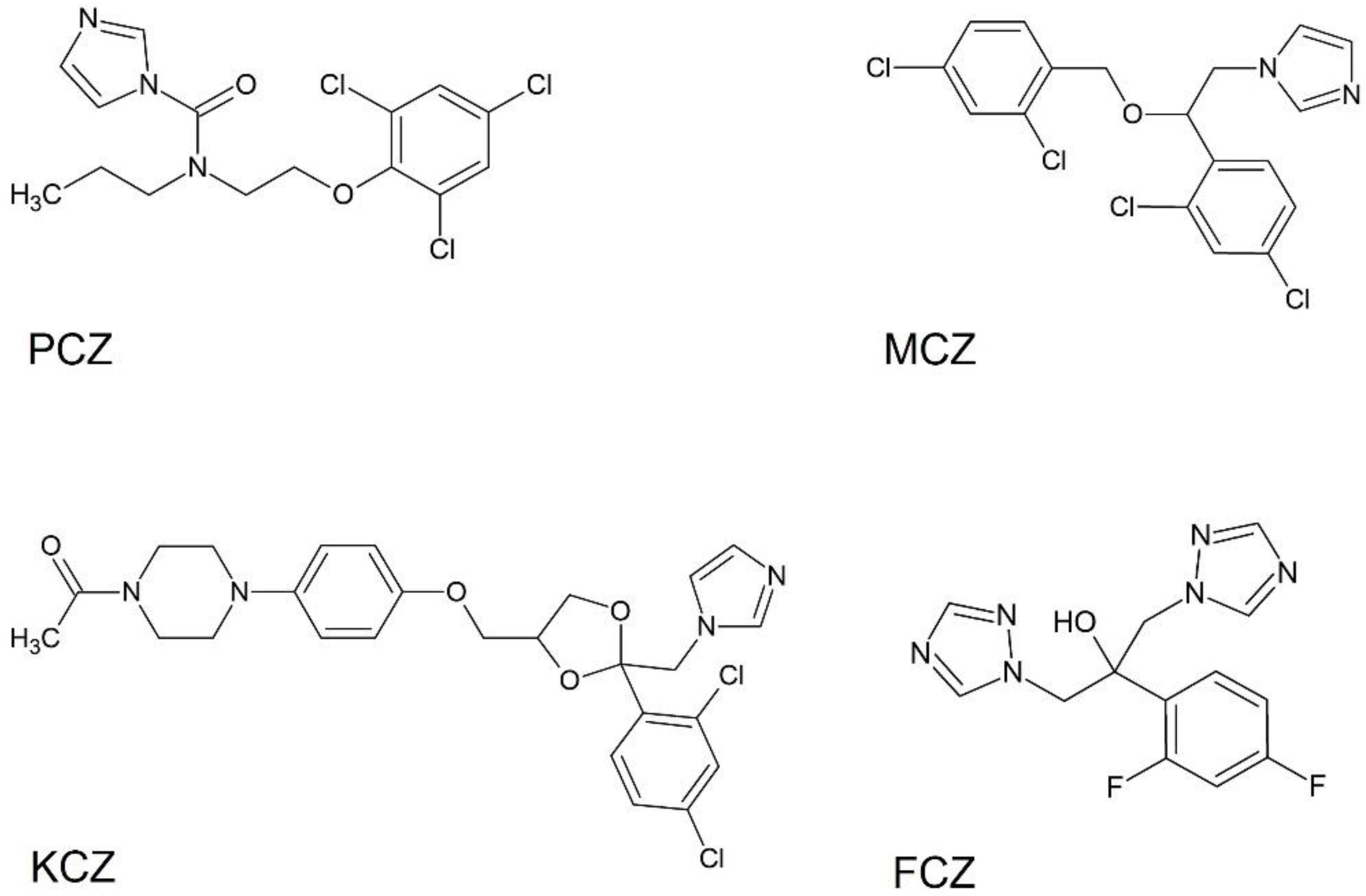
| Compound | IC20 ± SE (µM) | IC50 ± SE(µM) | IC80 ± SE(µM) |
|---|---|---|---|
| PCZ | 18.5 ± 3.9 | 73.8 ± 15.6 | 295.2 ± 62.3 |
| MCZ | 8.6 ± 0.6 | 34.4 ± 2.5 | 137.6 ± 9.8 |
| KCZ | 18.3 ± 2.4 | 73.2 ± 9.5 | 293.0 ± 38.1 |
| FCZ | Not detectable | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petricca, S.; Celenza, G.; Luzi, C.; Cinque, B.; Lizzi, A.R.; Franceschini, N.; Festuccia, C.; Iorio, R. Synergistic Activity of Ketoconazole and Miconazole with Prochloraz in Inducing Oxidative Stress, GSH Depletion, Mitochondrial Dysfunction, and Apoptosis in Mouse Sertoli TM4 Cells. Int. J. Mol. Sci. 2022, 23, 5429. https://doi.org/10.3390/ijms23105429
Petricca S, Celenza G, Luzi C, Cinque B, Lizzi AR, Franceschini N, Festuccia C, Iorio R. Synergistic Activity of Ketoconazole and Miconazole with Prochloraz in Inducing Oxidative Stress, GSH Depletion, Mitochondrial Dysfunction, and Apoptosis in Mouse Sertoli TM4 Cells. International Journal of Molecular Sciences. 2022; 23(10):5429. https://doi.org/10.3390/ijms23105429
Chicago/Turabian StylePetricca, Sabrina, Giuseppe Celenza, Carla Luzi, Benedetta Cinque, Anna Rita Lizzi, Nicola Franceschini, Claudio Festuccia, and Roberto Iorio. 2022. "Synergistic Activity of Ketoconazole and Miconazole with Prochloraz in Inducing Oxidative Stress, GSH Depletion, Mitochondrial Dysfunction, and Apoptosis in Mouse Sertoli TM4 Cells" International Journal of Molecular Sciences 23, no. 10: 5429. https://doi.org/10.3390/ijms23105429
APA StylePetricca, S., Celenza, G., Luzi, C., Cinque, B., Lizzi, A. R., Franceschini, N., Festuccia, C., & Iorio, R. (2022). Synergistic Activity of Ketoconazole and Miconazole with Prochloraz in Inducing Oxidative Stress, GSH Depletion, Mitochondrial Dysfunction, and Apoptosis in Mouse Sertoli TM4 Cells. International Journal of Molecular Sciences, 23(10), 5429. https://doi.org/10.3390/ijms23105429






