Light-Control over Casein Kinase 1δ Activity with Photopharmacology: A Clear Case for Arylazopyrazole-Based Inhibitors
Abstract
:1. Introduction
2. Results
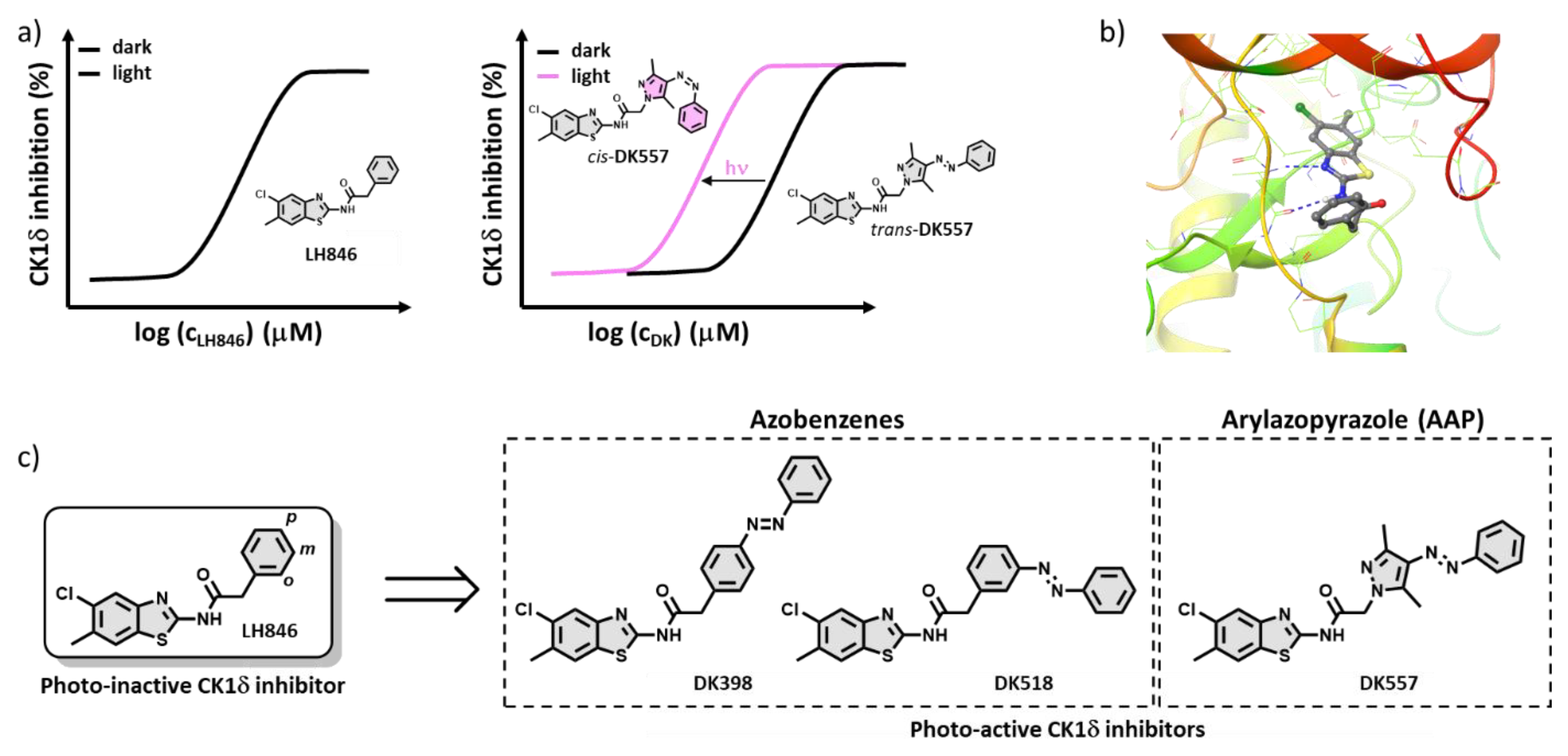
2.1. Design of the Photoswitchable Inhibitors
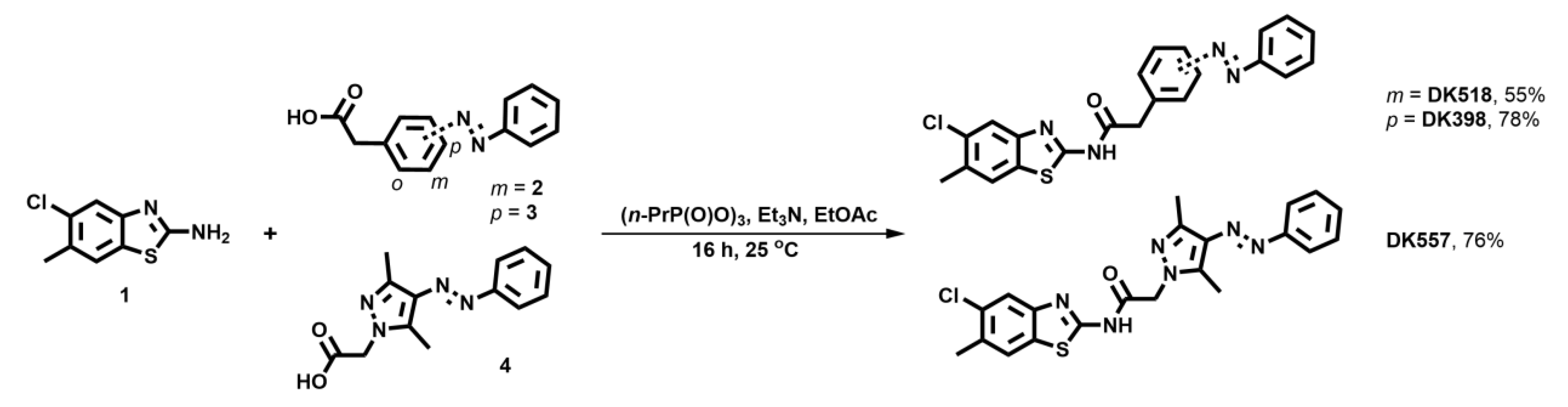
2.2. Synthesis
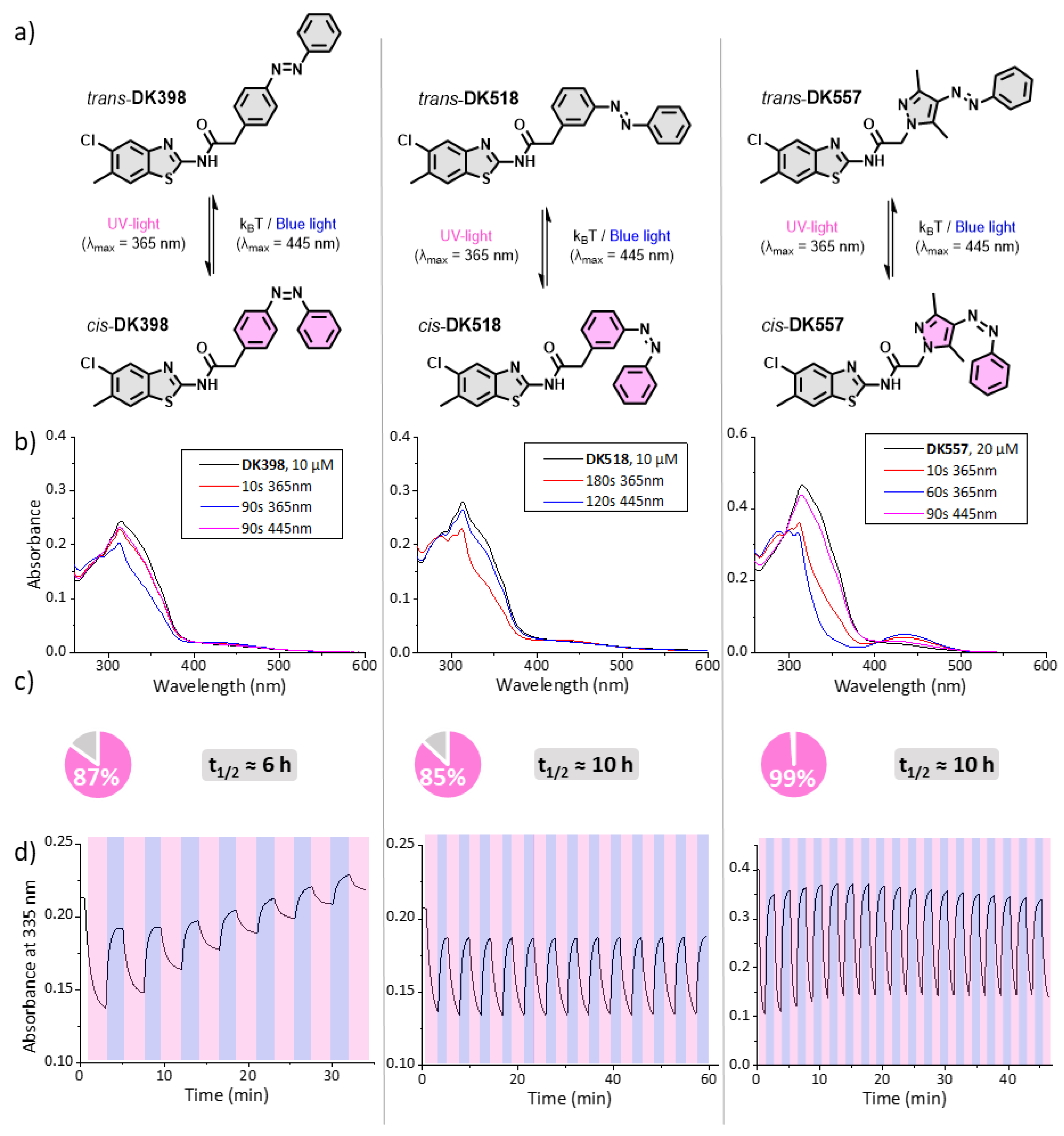
2.3. Photochemical Evaluation
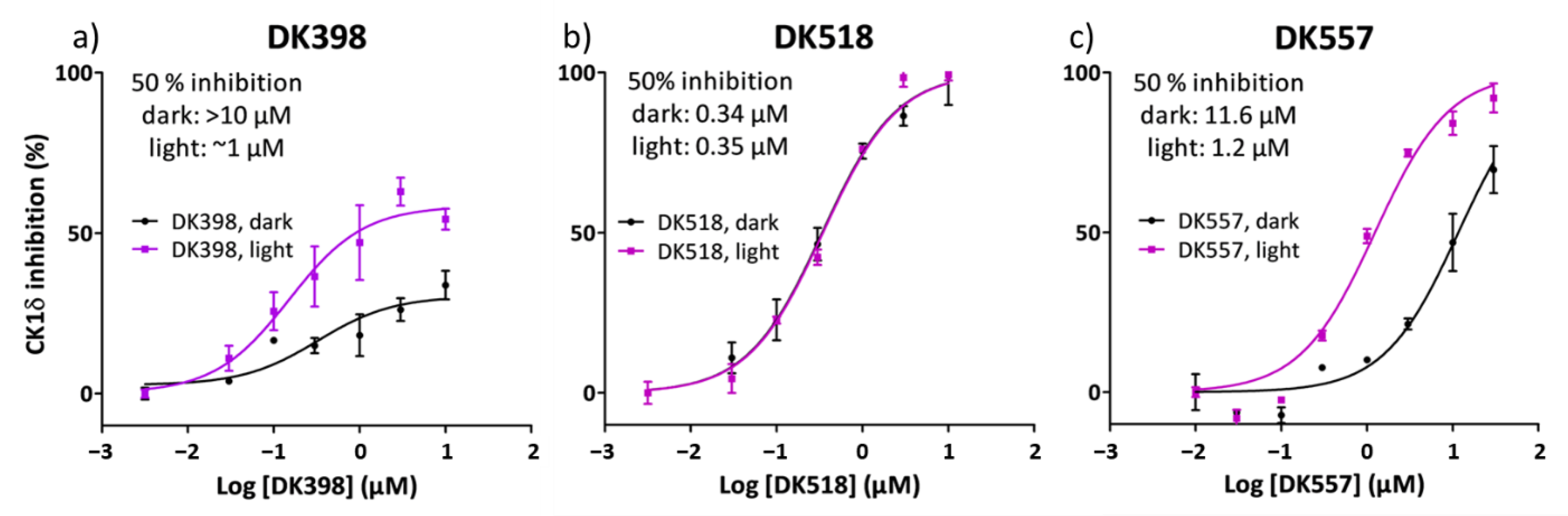
2.4. Light Modulation of CK1δ Activity
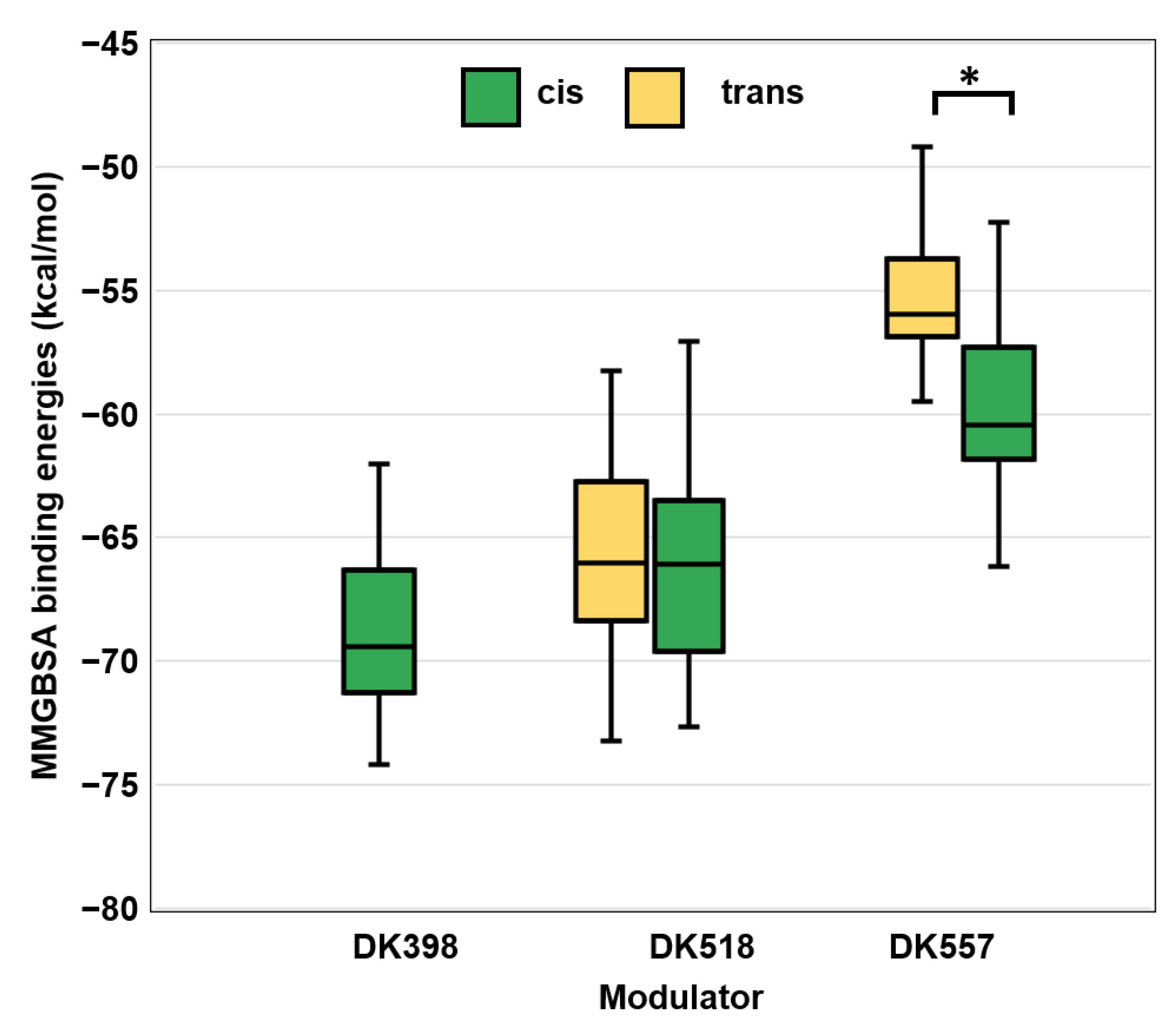
2.5. Molecular Docking and Dynamic Simulations
3. Material and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knippschild, U.; Gocht, A.; Wolff, S.; Huber, N.; Löhler, J.; Stöter, M. The Casein Kinase 1 Family: Participation in Multiple Cellular Processes in Eukaryotes. Cell. Signal. 2005, 17, 675–689. [Google Scholar] [CrossRef] [PubMed]
- Cheong, J.K.; Virshup, D.M. Casein Kinase 1: Complexity in the Family. Int. J. Biochem. Cell Biol. 2011, 43, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Knippschild, U.; Krüger, M.; Richter, J.; Xu, P.; Balbina, G.-R.; Peifer, C.; Halekotte, J.; Bakulev, V.; Bischof, J. The CK1 Family: Contribution to Cellular Stress Response and Its Role in Carcinogenesis. Front. Oncol. 2014, 4, 96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eide, E.J.; Virshup, D.M. Casein kinase I: Another cog in the circadian clockworks. Chronobiol. Int. 2001, 18, 389–398. [Google Scholar] [CrossRef]
- Xu, Y.; Padiath, Q.S.; Shapiro, R.E.; Jones, C.R.; Wu, S.C.; Saigoh, N.; Saigoh, K.; Ptáček, L.J.; Fu, Y.-H. Functional Consequences of a CKIδ Mutation Causing Familial Advanced Sleep Phase Syndrome. Nature 2005, 434, 640–644. [Google Scholar] [CrossRef]
- Schwab, C.; Demaggio, A.J.; Ghoshal, N.; Binder, L.I.; Kuret, J.; McGeer, P.L. Casein Kinase 1 Delta Is Associated with Pathological Accumulation of Tau in Several Neurodegenerative Diseases. Neurobiol. Aging 2000, 21, 503–510. [Google Scholar] [CrossRef]
- Kurihara, T.; Sakurai, E.; Toyomoto, M.; Kii, I.; Kawamoto, D.; Asada, T.; Tanabe, T.; Yoshimura, M.; Hagiwara, M.; Miyata, A. Alleviation of Behavioral Hypersensitivity in Mouse Models of Inflammatory Pain with Two Structurally Different Casein Kinase 1 (CK1) Inhibitors. Mol. Pain 2014, 10, 17. [Google Scholar] [CrossRef] [Green Version]
- McAlpine, C.S.; Swirski, F.K. Circadian Influence on Metabolism and Inflammation in Atherosclerosis. Circ. Res. 2016, 119, 131–141. [Google Scholar] [CrossRef] [Green Version]
- García-Reyes, B.; Witt, L.; Jansen, B.; Karasu, E.; Gehring, T.; Leban, J.; Henne-Bruns, D.; Pichlo, C.; Brunstein, E.; Baumann, U.; et al. Discovery of Inhibitor of Wnt Production 2 (IWP-2) and Related Compounds as Selective ATP-Competitive Inhibitors of Casein Kinase 1 (CK1) δ/ϵ. J. Med. Chem. 2018, 61, 4087–4102. [Google Scholar] [CrossRef]
- Velema, W.A.; Szymanski, W.; Feringa, B.L. Photopharmacology: Beyond Proof of Principle. J. Am. Chem. Soc. 2014, 136, 2178–2191. [Google Scholar] [CrossRef] [Green Version]
- Hoorens, M.W.H.; Szymanski, W. Reversible, Spatial and Temporal Control over Protein Activity Using Light. Trends Biochem. Sci. 2018, 43, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Hüll, K.; Morstein, J.; Trauner, D. In Vivo Photopharmacology. Chem. Rev. 2018, 118, 10710–10747. [Google Scholar] [CrossRef]
- Lerch, M.M.; Hansen, M.J.; van Dam, G.M.; Szymanski, W.; Feringa, B.L. Emerging Targets in Photopharmacology. Angew. Chem. Int. Ed. 2016, 55, 10978–10999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Welleman, I.M.; Hoorens, M.W.H.; Feringa, B.L.; Boersma, H.H.; Szymański, W. Photoresponsive Molecular Tools for Emerging Applications of Light in Medicine. Chem. Sci. 2020, 11, 11672–11691. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, K.; Irie, M. Diarylethene as a Photoswitching Unit. J. Photochem. Photobiol. C Photochem. Rev. 2004, 5, 169–182. [Google Scholar] [CrossRef]
- Wilson, D.; Li, J.W.; Branda, N.R. Visible-Light-Triggered Activation of a Protein Kinase Inhibitor. ChemMedChem 2017, 12, 284–287. [Google Scholar] [CrossRef]
- Grathwol, C.W.; Wössner, N.; Swyter, S.; Smith, A.C.; Tapavicza, E.; Hofstetter, R.K.; Bodtke, A.; Jung, M.; Link, A. Azologization and Repurposing of a Hetero-Stilbene-Based Kinase Inhibitor: Towards the Design of Photoswitchable Sirtuin Inhibitors. Beilstein J. Org. Chem. 2019, 15, 2170–2183. [Google Scholar] [CrossRef] [Green Version]
- Hammerich, M.; Schütt, C.; Stähler, C.; Lentes, P.; Röhricht, F.; Höppner, R.; Herges, R. Heterodiazocines: Synthesis and Photochromic Properties, Trans to Cis Switching within the Bio-Optical Window. J. Am. Chem. Soc. 2016, 138, 13111–13114. [Google Scholar] [CrossRef]
- Heintze, L.; Schmidt, D.; Rodat, T.; Witt, L.; Ewert, J.; Kriegs, M.; Herges, R.; Peifer, C. Photoswitchable Azo- and Diazocine-Functionalized Derivatives of the VEGFR-2 Inhibitor Axitinib. Int. J. Mol. Sci. 2020, 21, 8961. [Google Scholar] [CrossRef]
- Lachmann, D.; Lahmy, R.; König, B. Fulgimides as Light-Activated Tools in Biological Investigations. Eur. J. Org. Chem. 2019, 2019, 5018–5024. [Google Scholar] [CrossRef]
- Beharry, A.A.; Woolley, G.A.; Nass, M.M.; Wassermann, N.H.; Erlanger, B.F.; Takagi, M.; Komiyama, M.; Kokkinidis, M.; Rompp, A.; Spengler, B.; et al. Azobenzene Photoswitches for Biomolecules. Chem. Soc. Rev. 2011, 40, 4422. [Google Scholar] [CrossRef] [PubMed]
- Weston, C.E.; Richardson, R.D.; Haycock, P.R.; White, A.J.P.; Fuchter, M.J. Arylazopyrazoles: Azoheteroarene Photoswitches Offering Quantitative Isomerization and Long Thermal Half-Lives. J. Am. Chem. Soc. 2014, 136, 11878–11881. [Google Scholar] [CrossRef] [PubMed]
- Broichhagen, J.; Frank, J.A.; Trauner, D. A Roadmap to Success in Photopharmacology. Acc. Chem. Res. 2015, 48, 1947–1960. [Google Scholar] [CrossRef]
- Szymański, W.; Beierle, J.M.; Kistemaker, H.A.V.; Velema, W.A.; Feringa, B.L. Reversible Photocontrol of Biological Systems by the Incorporation of Molecular Photoswitches. Chem. Rev. 2013, 113, 6114–6178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, R.; Nilsson, J.R.; Solano, C.; Andréasson, J.; Grøtli, M. Design, Synthesis and Inhibitory Activity of Photoswitchable RET Kinase Inhibitors. Sci. Rep. 2015, 5, 9769. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleming, C.L.; Grøtli, M.; Andréasson, J. On-Command Regulation of Kinase Activity Using Photonic Stimuli. ChemPhotoChem 2019, 3, 318–326. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, D.; Rodat, T.; Heintze, L.; Weber, J.; Horbert, R.; Girreser, U.; Raeker, T.; Bußmann, L.; Kriegs, M.; Hartke, B.; et al. Axitinib: A Photoswitchable Approved Tyrosine Kinase Inhibitor. ChemMedChem 2018, 13, 2415–2426. [Google Scholar] [CrossRef] [PubMed]
- Hoorens, M.W.H.; Ourailidou, M.E.; Rodat, T.; van der Wouden, P.E.; Kobauri, P.; Kriegs, M.; Peifer, C.; Feringa, B.L.; Dekker, F.J.; Szymanski, W. Light-Controlled Inhibition of BRAFV600E Kinase. Eur. J. Med. Chem. 2019, 179, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Thayyil, S.; Kawaguchi, M.; Nakagawa, H.; Tamaoki, N. A Visible Light-Controllable Rho Kinase Inhibitor Based on a Photochromic Phenylazothiazole. Chem. Commun. 2021, 57, 12500–12503. [Google Scholar] [CrossRef]
- Xu, Y.; Gao, C.; Håversen, L.; Lundbäck, T.; Andréasson, J.; Grøtli, M. Design and Development of a Photoswitchable DFG-out Kinase Inhibitor. Chem. Commun. 2021, 57, 10043–10046. [Google Scholar] [CrossRef]
- Kolarski, D.; Vinyals, C.M.; Sugiyama, A.; Srivastava, A.; Ono, D.; Nagai, Y.; Iida, M.; Kenichiro Kenichiro, I.; Tama, F.; Szymanski, W.; et al. Reversible Modulation of Circadian Time with Chronophotopharmacology. Nat. Commun. 2021, 12, 3164. [Google Scholar] [CrossRef] [PubMed]
- Kolarski, D.; Sugiyama, A.; Rodat, T.; Schulte, A.; Peifer, C.; Itami, K.; Hirota, T.; Feringa, B.L.; Szymanski, W. Reductive Stability Evaluation of 6-Azopurine Photoswitches for the Regulation of CKIα Activity and Circadian Rhythms. Org. Biomol. Chem. 2021, 19, 2312–2321. [Google Scholar] [CrossRef] [PubMed]
- Schehr, M.; Ianes, C.; Weisner, J.; Heintze, L.; Müller, M.P.; Pichlo, C.; Charl, J.; Brunstein, E.; Ewert, J.; Lehr, M.; et al. 2-Azo-, 2-Diazocine-Thiazols and 2-Azo-Imidazoles as Photoswitchable Kinase Inhibitors: Limitations and Pitfalls of the Photoswitchable Inhibitor Approach. Photochem. Photobiol. Sci. 2019, 18, 1398–1407. [Google Scholar] [CrossRef] [PubMed]
- Reynders, M.; Chaikuad, A.; Berger, B.T.; Bauer, K.; Koch, P.; Laufer, S.; Knapp, S.; Trauner, D. Controlling the Covalent Reactivity of a Kinase Inhibitor with Light. Angew. Chem. Int. Ed. 2021, 60, 20178–20183. [Google Scholar] [CrossRef]
- Volarić, J.; Szymanski, W.; Simeth, N.A.; Feringa, B.L. Molecular Photoswitches in Aqueous Environments. Chem. Soc. Rev. 2021, 50, 12377–12449. [Google Scholar] [CrossRef]
- Kannaiyan, R.; Mahadevan, D. A Comprehensive Review of Protein Kinase Inhibitors for Cancer Therapy. Expert Rev. Anticancer. Ther. 2018, 18, 1249–1270. [Google Scholar] [CrossRef]
- Crespi, S.; Simeth, N.A.; König, B. Heteroaryl Azo Dyes as Molecular Photoswitches. Nat. Rev. Chem. 2019, 3, 133–146. [Google Scholar] [CrossRef]
- Calbo, J.; Thawani, A.R.; Gibson, R.S.L.; White, A.J.P.; Fuchter, M.J. A Combinatorial Approach to Improving the Performance of Azoarene Photoswitches. Beilstein J. Org. Chem. 2019, 15, 2753–2764. [Google Scholar] [CrossRef]
- Bhunia, S.; Dolai, A.; Samanta, S. Robust Bi-Directional Photoswitching of Thiomethyl Substituted Arylazopyrazoles under Visible Light. Chem. Commun. 2020, 56, 10247–10250. [Google Scholar] [CrossRef]
- Wang, Y.T.; Liu, X.Y.; Cui, G.; Fang, W.H.; Thiel, W. Photoisomerization of Arylazopyrazole Photoswitches: Stereospecific Excited-State Relaxation. Angew. Chem. Int. Ed. 2016, 55, 14009–14013. [Google Scholar] [CrossRef]
- Stricker, L.; Böckmann, M.; Kirse, T.M.; Doltsinis, N.L.; Ravoo, B.J. Arylazopyrazole Photoswitches in Aqueous Solution: Substituent Effects, Photophysical Properties, and Host–Guest Chemistry. Chem. Eur. J. 2018, 24, 8639–8647. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Hirota, T.; Peters, E.C.; Garcia, M.; Gonzalez, R.; Cho, C.Y.; Wu, X.; Schultz, P.G.; Kay, S.A. A Small Molecule Modulates Circadian Rhythms through Phosphorylation of the Period Protein. Angew. Chem. Int. Ed. 2011, 50, 10608–10611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stafforst, T.; Hilvert, D.; Stafforst, T.; Hilvert, D. Modulating PNA/DNA Hybridization by Light. Angew. Chem. Int. Ed. 2010, 49, 9998–10001. [Google Scholar] [CrossRef] [PubMed]
- Stricker, L.; Fritz, E.C.; Peterlechner, M.; Doltsinis, N.L.; Ravoo, B.J. Arylazopyrazoles as Light-Responsive Molecular Switches in Cyclodextrin-Based Supramolecular Systems. J. Am. Chem. Soc. 2016, 138, 4547–4554. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schulte, A.M.; Kolarski, D.; Sundaram, V.; Srivastava, A.; Tama, F.; Feringa, B.L.; Szymanski, W. Light-Control over Casein Kinase 1δ Activity with Photopharmacology: A Clear Case for Arylazopyrazole-Based Inhibitors. Int. J. Mol. Sci. 2022, 23, 5326. https://doi.org/10.3390/ijms23105326
Schulte AM, Kolarski D, Sundaram V, Srivastava A, Tama F, Feringa BL, Szymanski W. Light-Control over Casein Kinase 1δ Activity with Photopharmacology: A Clear Case for Arylazopyrazole-Based Inhibitors. International Journal of Molecular Sciences. 2022; 23(10):5326. https://doi.org/10.3390/ijms23105326
Chicago/Turabian StyleSchulte, Albert M., Dušan Kolarski, Vidya Sundaram, Ashutosh Srivastava, Florence Tama, Ben L. Feringa, and Wiktor Szymanski. 2022. "Light-Control over Casein Kinase 1δ Activity with Photopharmacology: A Clear Case for Arylazopyrazole-Based Inhibitors" International Journal of Molecular Sciences 23, no. 10: 5326. https://doi.org/10.3390/ijms23105326
APA StyleSchulte, A. M., Kolarski, D., Sundaram, V., Srivastava, A., Tama, F., Feringa, B. L., & Szymanski, W. (2022). Light-Control over Casein Kinase 1δ Activity with Photopharmacology: A Clear Case for Arylazopyrazole-Based Inhibitors. International Journal of Molecular Sciences, 23(10), 5326. https://doi.org/10.3390/ijms23105326





